Introduction: The gonadotoxic effects of chemotherapy and radiation may result in premature ovarian failure in premenopausal oncology patients[1],[2]. Although autotransplantation of ovarian tissue has led to successful live births, reintroduction of latent malignant cells inducing relapse is a significant concern[3]-[5]. In this work, we investigated the design of biomaterial grafts for transplantation of isolated ovarian follicles as a means to preserve fertility.
Materials and Methods: Primordial and primary ovarian follicles from young female mice were extracted, encapsulated and cultured in vitro within a range of biomaterials for initial evaluation of suitability for subsequent transplantation into adult mice. Three biomaterial combinations (fibrin, fibrin/alginate, and fibrin/collagen) were then investigated for their effect on follicle survival and development in vivo. Biomaterial formulations that maximized in vitro growth and in vivo survival were identified, and employed for transplantation of primordial follicles in breeding studies. Follicles were encapsulated within fibrin and fibrin-VEGF hydrogels, and transplant recipients were mated and their ability to resume cycling and to achieve a pregnancy assessed. Finally, mice transplanted with human metastatic breast cancer cells were used as ovary donors for creation of a would-be transplantation graft to determine the ability of the current follicle isolation method to reduce the number of associated cancer cells relative to transplanted ovarian tissue.
Results and Discussion: Among the formulations tested, aggregated follicles encapsulated within fibrin had enhanced survival and integration with the host tissue following transplantation relative to the fibrin-alginate and fibrin-collagen composites.
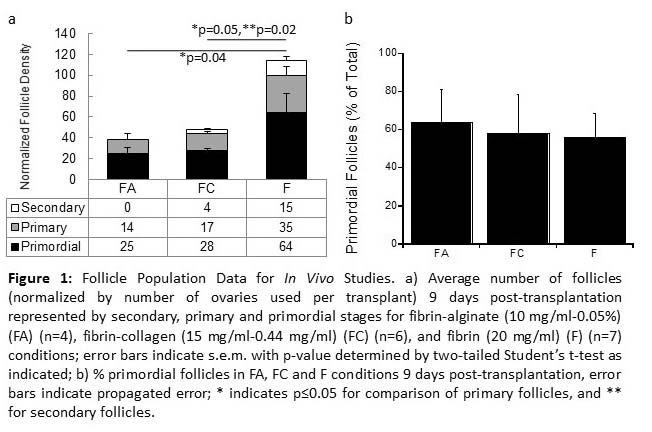
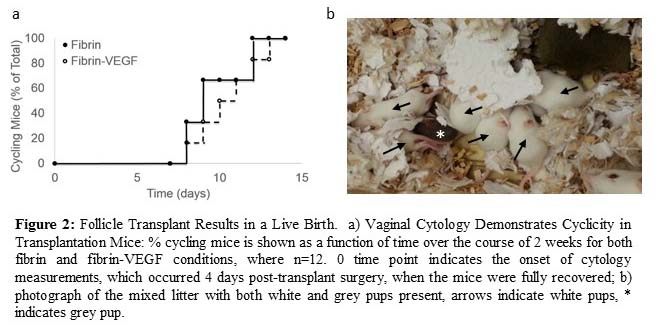
All mice transplanted with fibrin-encapsulated follicles resumed cycling in under three weeks, demonstrating the restoration of endocrine function of the graft. Importantly, live births were achieved, yet were observed only for follicles transplanted within VEGF-loaded fibrin beads.
The extent to which these procedures were capable of decreasing the presence of metastatic breast cancer cells among the isolated follicles was evaluated, demonstrating that the density of cancer cells in the graft was reduced more than 7-fold relative to the density of cancer cells in the intact ovary.
Conclusion: The ability to obtain live births by transplanting isolated primordial and primary follicles, while also reducing the risk of re-seeding disease relative to ovarian tissue transplantation, may ultimately provide a means to preserve fertility in premenopausal oncology patients. While the current approach makes significant strides towards this ultimate goal, additional developments are needed to completely eliminate this risk and fulfill the potential for restoring fertility and endocrine function through follicle autotransplantation for patients surviving cancer.
Kenan Moss; Sayali Kale; Eiji Saito; Samira Azarin; Shreyas Rao; Brian Aguado; Ariella Shikanov; Min Xu; Rachel Mullin; Woodruff lab members
References:
[1] Jeruss, J. S. & Woodruff, T. K. Preservation of fertility in patients with cancer. N Engl J Med 360, 902-911 (2009).
[2] Shapira, M., Raanani, H., Cohen, Y. & Meirow, D. Fertility preservation in young females with hematological malignancies. Acta Haematol 132, 400-413 (2014).
[3] Dolmans, M. M., Luyckx, V., Donnez, J., Andersen, C. Y. & Greve, T. Risk of transferring malignant cells with transplanted frozen-thawed ovarian tissue. Fertil Steril 99, 1514-1522 (2013).
[4] Bockstaele, L., Tsepelidis, S., Dechene, J., Englert, Y. & Demeestere, I. Safety of ovarian tissue autotransplantation for cancer patients. Obstet Gynecol Int 2012, 495142 (2012).
[5] Dolmans, M. M. et al. Reimplantation of cryopreserved ovarian tissue from patients with acute lymphoblastic leukemia is potentially unsafe. Blood 116, 2908-2914 (2010).