Titanium plates and screws have been used for years for maxillofacial fractures fixation. However, titanium implants must be excised in revision surgery cases, mainly in the pediatric population. To prevent a second surgery, resorbable polylactide (PLA) based co-polymer implants have been developed. However, their limited strength restricts their use in higher load-bearing indications. Furthermore, the consistency of PLA devices complicates handling during surgery and usually requires the use of a water bath for the shaping of plates. Hence, the development of alternative resorbable biomaterials is desired. Magnesium alloys have attracted the interest of scientists as biocompatible metallic bone implants for orthopedic surgery because of their excellent mechanical properties and properties similar to those of cortical bone. However, the challenge with magnesium implants is to guarantee a suitable mechanical stability during the healing process despite their biodegradation. It is well-documented that hydrogen gas is formed during the degradation of magnesium, which could affect wound and fracture healing if it is not adequately evacuated. Based on these considerations, we developed a new fracture model in a large animal model, which permits the use of human-sized implants.
Zygoma and orbital rim fractures were performed by osteotomy on a total of five Yucatan miniature pigs. For a first group of three animals, the fractures were fixed with coated magnesium (mg) plates/screws as test materials, while a second group of two animals were implanted with polylactide-co-glycolide (PLGA) plates/screws as control materials. Two animals (one per group) were implanted for one month post-surgery, while three others (one control and two from the test group) were implanted for nine months. A continuous monitoring using in vivo computed tomography (CT; Siemens Somatom Sensation 16) was conducted to follow implant degradation and fracture healing over implantation time. CTs were performed immediately post-op and also before euthanasia for the 1-month cohort and at 0, 2, 4, 6 and 9 months post-surgery for the 9-month cohort. At necropsy, bone blocks containing the implants and surrounding tissue were harvested, processed for undecalcified histology, infiltrated with PMMA, microground (Exakt 400 CS), and stained with Goldner’s Trichome. A complete histopathological analysis was then performed.
The gas formation produced during magnesium degradation was detectable on radiological examination, but did not inhibit the fracture healing process. The magnesium implants as well as the PLGA implants remained visible 9 months after surgery. The initial osteotomy locations showed a physiological bone healing and good fracture alignement at every time points in both groups (see figure 1: CT of mg implants direct after surgery (left) and 9 months after surgery (right)).
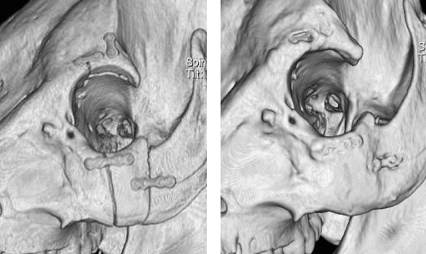
No major differences on histological evaluation were observed across the two groups.
This new maxillofacial fracture model allowed an accurate comparison of two different resorbable plate/screw implant systems. The use of a magnesium plate/screw system for maxillofacial fracture fixation was successful. Compared to PLGA implants, magnesium produced intraosseus cavities. However, no side effects due to gas formation and corrosion processes were observed. This study showed promising results for further development of magnesium plate/screws for maxillofacial fracture fixation.
The authors would like to sincerely thank Dr. Cathy Tkaczyk and Mr. Yannick Trudel for their respective scientific and surgical support