Introduction: Vertebroplasty (VP) is a minimally invasive procedure where bone cement is injected into vertebral fractures for palliative relief via fracture stabilization. Basic requirements of VP cements include 5-20 min of injection time and lasting compression strength (σc) >30 MPa [1],[2]. Germanium containing glass ionomer cements (Ge-GICs) demonstrate appropriate handling characteristics for VP [3], but weaken significantly over time and little is known regarding their in vivo performance. The objectives of this study were to: (i) establish composition-property relationships to mitigate the known mechanical decline of Ge based GICs; (ii) quantify their potential as injectable cements; and (iii) conduct a pilot animal trial to gain preliminary biocompatibility data.
Materials and Methods: Design of mixtures (DOM) was used to identify 12 glass compositions. Glasses were synthesized via a melt-quench technique, ground to ≤45 µm, and annealed (Tg – 30˚C). GICs were prepared by mixing glass powders with polyacrylic acid (Mw=12,700 g/mol, 50 wt% aq. sol.) at a 4:3 ratio. GIC σc was evaluated up to 180 days and DOM response models were produced for relevant time points. An additional composition (DG302) was interpolated from within the design space and synthesized to validate the DOM models. Injectability of DG302 GIC was assessed by profiling the force required to extrude cement from a 1 cc syringe with a 12 G cannula over time. AccelLAB (Boisbriand, CAN) was contracted for the in vivo pilot, where DG302 filled 4 subcritical femoral defects in 2 male NZ white rabbits with a sham control over an 8-week period (e.g. Fig. 1). Post-sacrifice, defects were scanned using MicroCT to reconstruct 3-D images of the implants, then sectioned, stained with Goldner’s Trichrome, and graded according to cell type and local host response. Systemic effects of the DG302 implants were also assessed via blood work.
Results and Discussions: Ge-GIC mechanical performances were successfully improved, e.g. σc of DG302 increased from 51 MPa at 1 day to 71 MPa at 180 days. DOM models identified ZrO2/Na2O content as the most influential factor on σc, and SiO2:GeO2 ratio key to ensure long-term mechanical stability. DG302 injection profiles exhibited an initial injection force of 16 N that increased to 62 N after 15 min. All animals maintained healthy weights and appetites, and concerns regarding the long-term stability of Ge-GICs were further mitigated as DG302 implants remained intact over the 8 weeks studyA.
Conclusion: Precise manipulation of glass chemistry yields Ge-GICs with clinically viable handling and mechanical properties that are stable in vivo. These characteristics exemplify a practical foundation to further the investigation of Ge-GICs for minimally invasive procedures such as VP.
A. Full histopathological and hematological results to be obtained in October 2015 and not available at the time of abstract submission, but will be included in presentation of this work at WBC.
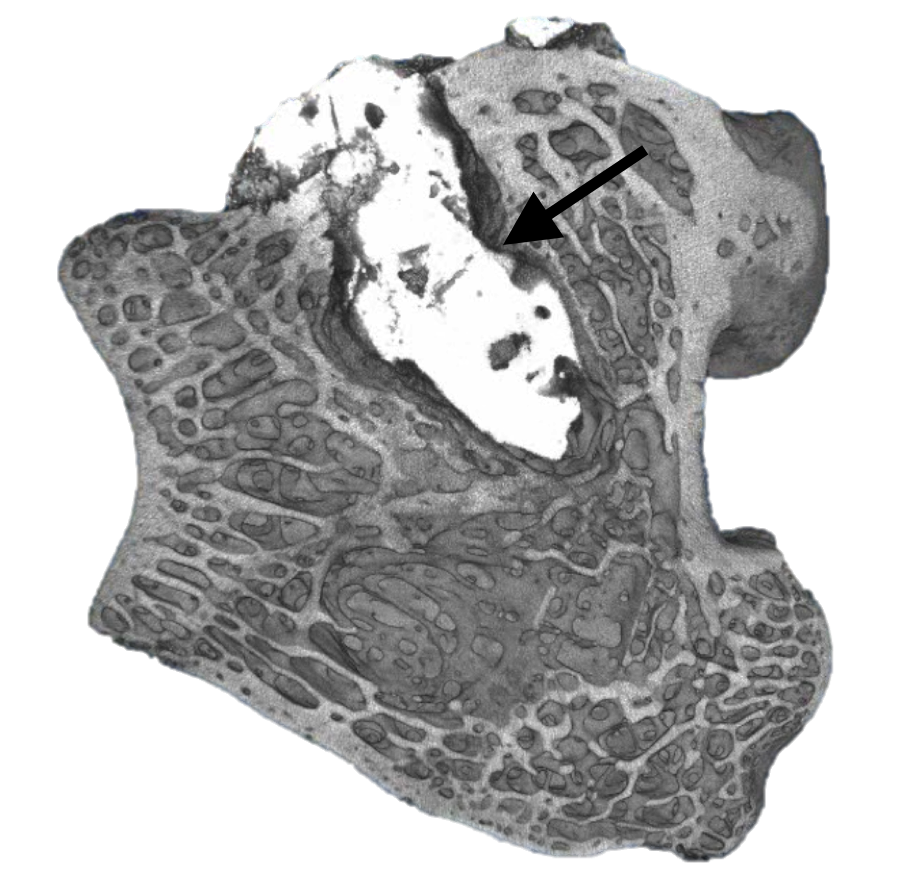
Figure 1: MicroCT section of DG302 implant (identified by black arrow) in rabbit femoral defect model after 8 weeks.
References:
[1] Heini and Berlemann, 2001 Eur Spine J 10, s205-s213
[2] Jansen et al. 2005, Orthop Clin N Am 36
[3] Dickey et al. 2013 JMBBM 23