Cytocompatible magnetic polymer nanoparticle covered with phospholipid polymer brush layer for in situ analysis of time-dependent cell functions
-
1
University of Tokyo, Department of Bioengineering, Japan
-
2
University of Tokyo, Department of Materials Engineering, Japan
Introduction: mRNA is one of the essential biomolecules that control the cell activities. In this regard, the analysis of the expression dynamics of mRNA in living cells is important. The polymerase chain reaction (PCR) technique is one of the powerful tools for the analysis of mRNA expression; however, the cytoclasis process is indispensable to extract mRNA. The purpose of this study is to fabricate the cytocompatible and cell-penetrating polymer nanoparticles that enable to control the movement by external forces. The multi-functional magnetic polymer nanoparticles could be fabricated by the encapsulation of the super paramagnetic nanoparticles, surface modification by zwitterionic and cationic polymers, and introduction of functional groups at the outermost surface to capture mRNA. The polymer nanoparticles could extract mRNA from living cells without cytoclasis. The time-dependent cell analysis could be achieved by the combination of PCR and magnetic polymer nanoparticles fabricated in this study.
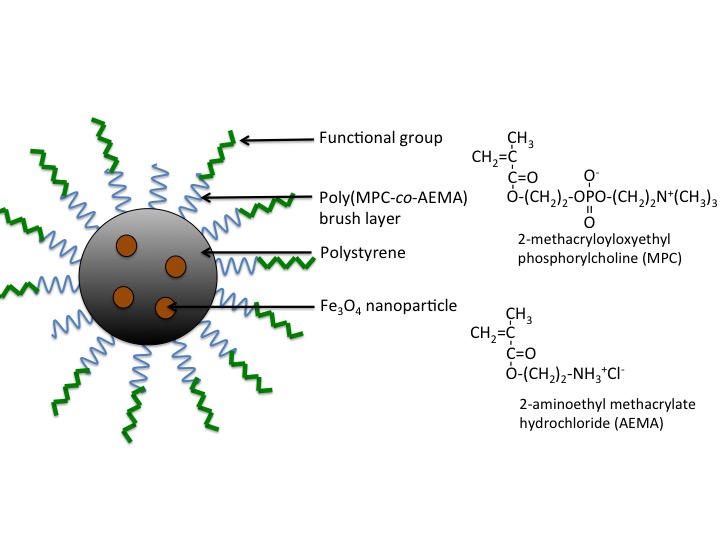
Methods: The magnetic core nanoparticles were synthesized from the miniemulsion polymerization of a styrene containing different amounts of Fe3O4 nanoparticles in feed. The diameter of nanoparticles and content of Fe3O4 in the nanoparticles were determined by dynamic light scattering (DLS) and thermo gravimetric (TG) measurements, respectively. The surface of the magnetic core nanoparticles was modified with a poly(MPC-co-AEMA) (PMA) or poly(MPC) brush layers. The magnetic response of the magnetic nanoparticles in an aqueous dispersion to a neodymium magnet was analyzed from the turbidity at 600 nm with the UV measurement. The rhodamine-labeled magnetic nanoparticles with the polymer brush layers were contacted with adherent HeLa cells in culture medium for 2h, and then, a confocal laser scanning microscope observation was performed.
Results: The diameter of magnetic core nanoparticle was about 100 nm, which would be small enough for entering into cells. The content of the Fe3O4 was almost the same as that in feed, indicating the fine control of the encapsulated amount of Fe3O4. More than 90% of the magnetic polymer nanoparticles could be collected by a neodymium magnet within 20 min, and easily re-dispersed by stirring. These results indicated that the magnetic polymer nanoparticles would have an excellent remote operation. In the case of the magnetic nanoparticles with the PMA brush layer, the fluorescence could be observed in cells, while no fluorescence could be observed in the case of those with the poly(MPC) brush layer. Furthermore, the phenotype of the adherent cells containing the magnetic polymer nanoparticles was almost the same as that of the cells normally cultured. These results indicated that the magnetic polymer nanoparticles with the PMA brush layer could enter into cytoplasm and display a good cytocompatibility. We could fabricate the platform nanoparticles for a sequential analysis of mRNA expression.
This research was supported from "Nanomedicine Molecular Science" (2306), NEXT, Japan
Keywords:
biomaterial,
nanoparticle,
Biocompatibility,
polymer brush
Conference:
10th World Biomaterials Congress, Montréal, Canada, 17 May - 22 May, 2016.
Presentation Type:
Poster
Topic:
Regeneration inducing biomaterials
Citation:
Bansyo
Y,
Inoue
Y and
Ishihara
K
(2016). Cytocompatible magnetic polymer nanoparticle covered with phospholipid polymer brush layer for in situ analysis of time-dependent cell functions.
Front. Bioeng. Biotechnol.
Conference Abstract:
10th World Biomaterials Congress.
doi: 10.3389/conf.FBIOE.2016.01.02611
Copyright:
The abstracts in this collection have not been subject to any Frontiers peer review or checks, and are not endorsed by Frontiers.
They are made available through the Frontiers publishing platform as a service to conference organizers and presenters.
The copyright in the individual abstracts is owned by the author of each abstract or his/her employer unless otherwise stated.
Each abstract, as well as the collection of abstracts, are published under a Creative Commons CC-BY 4.0 (attribution) licence (https://creativecommons.org/licenses/by/4.0/) and may thus be reproduced, translated, adapted and be the subject of derivative works provided the authors and Frontiers are attributed.
For Frontiers’ terms and conditions please see https://www.frontiersin.org/legal/terms-and-conditions.
Received:
27 Mar 2016;
Published Online:
30 Mar 2016.