Introduction: Scaffolds are among the key components of a tissue-engineered construct. Although many different proteins have been used in scaffold preparation, gelatin, which is obtained by degradation of collagen, is a completely biodegradable, highly compatible, and low-cost raw material. These characteristics justify its success in numerous applications in tissue engineering and other fields such as drug delivery, wound dressing and others[1],[2]. This work focuses on the study of mechanical characterization of porous gelatin gels, in particular, the viscoelastic and poro-elastic response to the application of strain or stress. Simulations have also been used to prove the necessity of introducing a nonlinear stress-strain relationship in order to reproduce experimental results.
Materials and Methods: Gelatin type A samples were made from porcine skin (Sigma-Aldrich, St-Louis, MO, USA). Samples were prepared by dissolving gelatin in deionized water stirred at 50°C for 1h. The solution was poured into Petri dishes and let jellify for 12 hrs at 25°C.Two cooling steps were used to freeze the samples: 6h at 4°C and then 8h at -20°C. Samples were then freeze-dried to create the porous matrix[3].
Viscoelastic and poro-elastic behaviors manifest themselves at different time-scales during relaxation and were studied using a method proposed by Wang[4]. This method relies on unconfined compression relaxation tests on disk-shaped samples of various sizes. For relaxation tests, a 25 mm diameter compression plate was attached to a 150 g load cell mounted on a Biomomentum Mach 1 (Laval, Qc, Canada) testing apparatus. Compressions were followed by a relaxation time as long as 12 hours (43200s). All tests were performed at room temperature in Phosphate Buffered Saline. Simulations were done with Simulink (Matlab) to assess strain-stress relationships using Neo-Hookean and Mooney-Rivlin models.
Results and Discussion: Figure 1 shows a typical scanning electronic microscope (SEM) image of the porous structure of the hydrogel. Poro-elasticity analysis was used to study the migration of fluids within a porous matrix. By mechanical compression, fluid diffusion was induced through the gels from the center to the outmost layer. Figure 2 shows mechanical responses of porous gelatin samples of different sizes. For comparison purposes, collagen hydrogel relaxation (viscoelastic only) was added to show the behavior difference between a poro-elastic sample and a viscoelastic sample. The mechanical properties of porous gelatin specimens at different concentrations and pore sizes were investigated. The presence of poro-elastic behavior was detected with the use of a simple method based on curve fitting techniques.

Figure 1: Scanning electron microscope image of the porous structure of gelatin. Average pore size is 50µm.
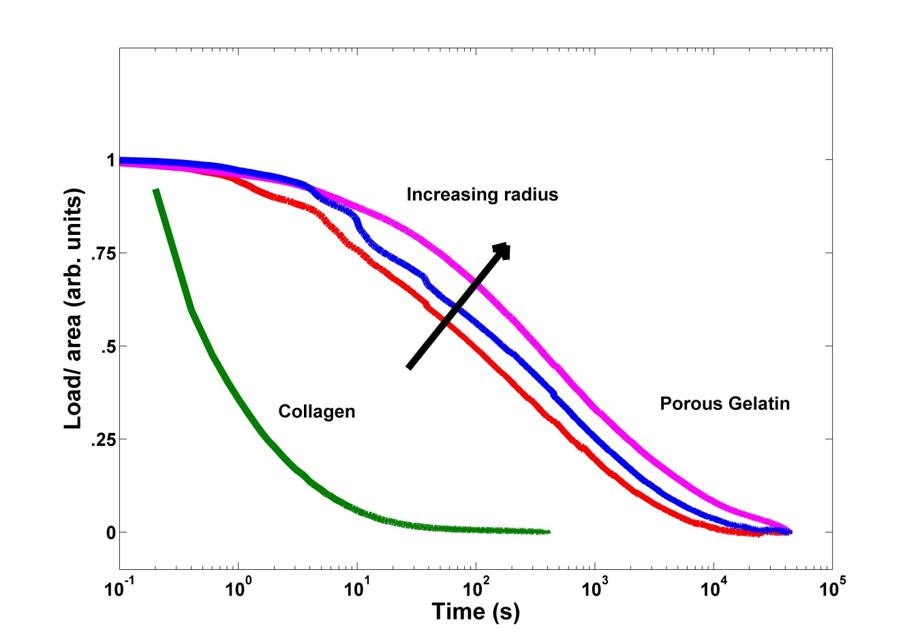
Figure 2: Stress relaxation under a 50% strain for disk shaped samples of different radius for porous gelatin gels and collagen hydrogel.
Conclusion: The method described in this work presents new techniques to assess the viscoelasticity and poro-elasticity of hydrogels. While the contribution of poro-elasticty relative to viscoelasticity might be negligible for some hydrogels, it constitutes a crucial part of mechanical behavior for other hydrogels like porous gelatin.
AL was awarded of a MSc Scholarship from NSERC CREATE Program in Regenerative Medicine (www.ncprm.ulaval.ca). This work was partially supported by NSERC-Canada, CIHR-Canada, CFI-Canada, FRQ-NT-Quebec, MRI-Quebec and, Heart and Stroke Foundation.
References:
[1] Islam, M.M.; Khanb, M.A.; Rahmana, M.M. Preparation of gelatin based porous biocomposite for bone tissue engineering and evaluation of gamma irradiation effect on its properties, Materials Science and Engineering C, 2015, 49, 648–655.
[2] Grover, C.N.; Cameron, R.E.; Best, S.M, Investigating the morphological, mechanical and degradation properties of scaffolds comprising collagen, gelatin and elastin for use in soft tissue engineering, Journal of the mechanical behavior of biomedical materials,2012, 10, 62-74
[3] Spinelli, A.; Vinci, B.; Tirella, A.; Matteucci,M.; Gargani, L.; Ahluwalia, A.; Domenici, C.; Picano E.; Chiarelli,P. Realization of a poro-elastic ultrasound replica of pulmonary tissue, Biomatter, 2012, 2:1, 37-42.
[4] Wang, Q.M.; Mohan, A.C.; Oyen, M.L.; Zhao, X.H. Separating viscoelasticity and poroelasticity of gels with different length and time scales, Acta Mechanica Sinica,2014,30,20-27.