Introduction: The metastatic spread of cancer cells to distant sites of the body, in particular bone, is the major cause of death among prostate cancer patients[1]. Despite the current disease models, it has not yet been well understood how cancer cells attain the ability to survive and establish metastasis within the surrounding microenvironment, remaining one of the most challenging aspects in the treatment of metastatic cancers. Hence, to closely mimic the tumour microenvironment, this study aimed at developing a 3D bioengineered in vitro bone model for the study of bone metastasis.
Martials and Methods: Medical-grade Polycaprolactone (PCL) from Purac was used to produce tubular scaffolds by melt electrospinning writing on a custom-built machine[2]. Scaffolds (1 cm length, 5 mm diameter, 100 µm thickness, 250 pore size, 0-90° pattern) were further coated by calcium phosphate (CaP) for osteogenic properties[3] and seeded with 4×105 human osteoprogenitor bone cells (hOBs), isolated from human bone samples[4]. After reaching 3D confluency, the tubular constructs were transferred from static cultures to the vessel of a bi-axial rotating bioreactor[5] and mineralisation was induced for 6 weeks with osteogenic media (50 μg/ml ascorbate-2-phosphate, 10 mM β-glycerophosphate, 100 nM dexamethasone)[6]. After 6 weeks, human mesenchymal stem cells (MSCs) from the same patient and human umbilical vein endothelial cells (HUVECs) were mixed (1:1 ratio; 6×105 cells per gel) with a gelatin methacrylamide-based gel (gelMA) and injected in the centre of the osteoblast-derived tubular scaffold, followed by cross-linking with 365 nm wavelength UV for 10 minutes[7] to form a soft gel, 3-4 kPa in stiffness, inside the tubular scaffold. After one week of culture in the rotating bioreactor, allowing for capillaries to form, 2×104 human prostate cancer cells (LNCaP, C4-2B, PC3) were injected into the central part of the construct and co-cultured for an additional two weeks, in order to mimic and study human prostate cancer metastasis to the bone (Figure 1).
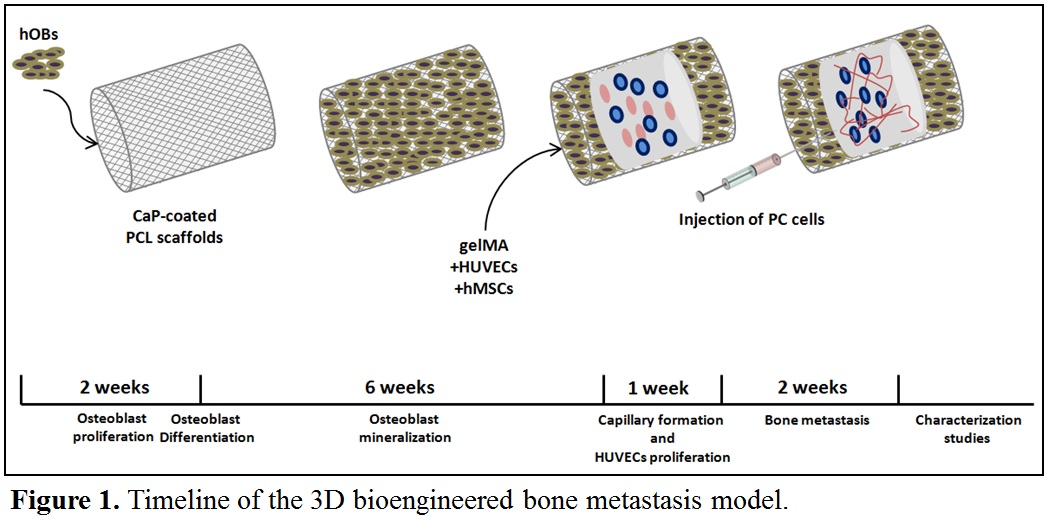
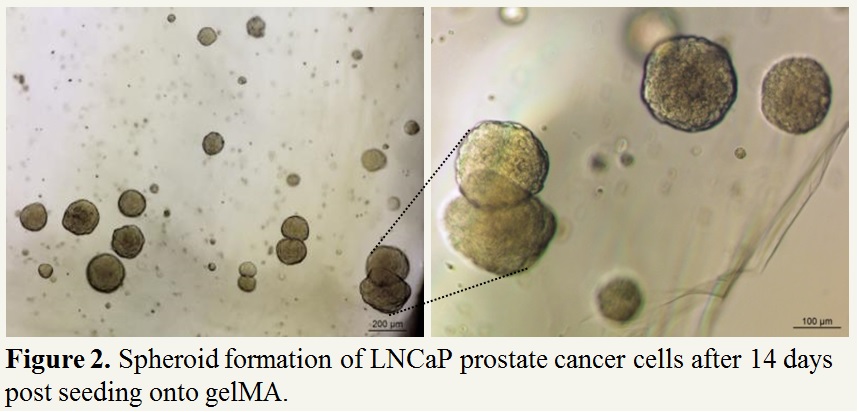
Results and Discussion: We have successfully grown human bone-derived osteoblast cells onto tubular PCL scaffolds with cells fully covering the scaffolds after six week post seeding. Furthermore, we have successfully grown cancer cells in gelMA where after 14 days cells are forming large spheroid-like colonies ranging from 100 to 200 µm, mimicking the tumour-like characteristics (Figure 2). We are currently optimizing the mechanical properties of gelMA for optimal growth of HUVECs and MSCs and capillary formation. Further studies involve micro-computed tomography, scanning electron microscopy (SEM) and immunohistochemistry for assessing the extent of bone metastatic lesions in the novel bioengineered model.
Conclusion: This novel bioengineered 3D in vitro model better reflects the natural 3D environment of prostate cancer metastasis to the bone and hence will provide an ideal platform to study current and novel treatments against metastasis, ultimately improving patient outcomes.
References:
[1] Bubendorf, L. et al. Metastatic patterns of prostate cancer: an autopsy study of 1,589 patients. Hum. Pathol. 31, 578–583 (2000).
[2] Brown, Toby D., et al. "Design and fabrication of tubular scaffolds via direct writing in a melt electrospinning mode." Biointerphases 7.1 (2012): 13.
[3] Vaquette, Cédryck, et al. "Effect of culture conditions and calcium phosphate coating on ectopic bone formation." Biomaterials 34.22 (2013): 5538-5551.
[4] Reichert, Johannes C., et al. "Mineralized human primary osteoblast matrices as a model system to analyse interactions of prostate cancer cells with the bone microenvironment." Biomaterials 31.31 (2010): 7928-7936.
[5] Singh, Harmeet, et al. "Flow modelling within a scaffold under the influence of uni-axial and bi-axial bioreactor rotation." Journal of biotechnology 119.2 (2005): 181-196.
[6] Thibaudeau, Laure, et al. "A tissue-engineered humanized xenograft model of human breast cancer metastasis to bone." Disease models & mechanisms 7.2 (2014): 299-309.
[7] Schuurman, Wouter, et al. "Gelatin‐Methacrylamide Hydrogels as Potential Biomaterials for Fabrication of Tissue‐Engineered Cartilage Constructs." Macromolecular bioscience 13.5 (2013): 551-561.