Bone is a dynamic and vascularized tissue that has the ability of naturally healing upon damage. Nevertheless, in the case of critical size defects this potential is impaired. Present approaches mainly consider autografts and allografts, which presents several limitations. Bone Tissue Engineering (BTE) is based on the use of 3D matrices to guide both cellular growth and differentiation. We previously showed that a porous 3D hydrogel, Glycosyl-Nucleoside-Fluorinated (GNF) is: 1) non-cytotoxic to clustered human Adipose Mesenchymal Stem Cells (hASCs), 2) bioinjectable. Although hASCs could not adhere to the gel, preformed cell aggregates were shown to grow and differentiate towards the osteoblastic lineage, without the use of osteogenic factors, when entrapped within the GNF-based gel.
Therefore, this novel class of hydrogels show promise for the development of therapeutic solutions for BTE[1]. The hypothesis of this research was that improving the capacity to promote the adhesion of cells by adding collagen to the GNF could impact the regenerative potential of this GNF-based gel. Collagen is a protein matrix well known for its cytocompatibility[2]. Thereby, the overall aim of this work was to design, develop and validate a new composite hydrogel.
GNF was prepared as previously described in detail[1], at a concentration of 3% (w/v). Type I-collagen gel was prepared from rat-tail tendons at a concentration of 4 g/L[3].
hASCs were isolated from human adipose tissue and cultured in tissue culture flasks using DMEM. TdTomato-labelled hASCs were observed with a confocal microscope.
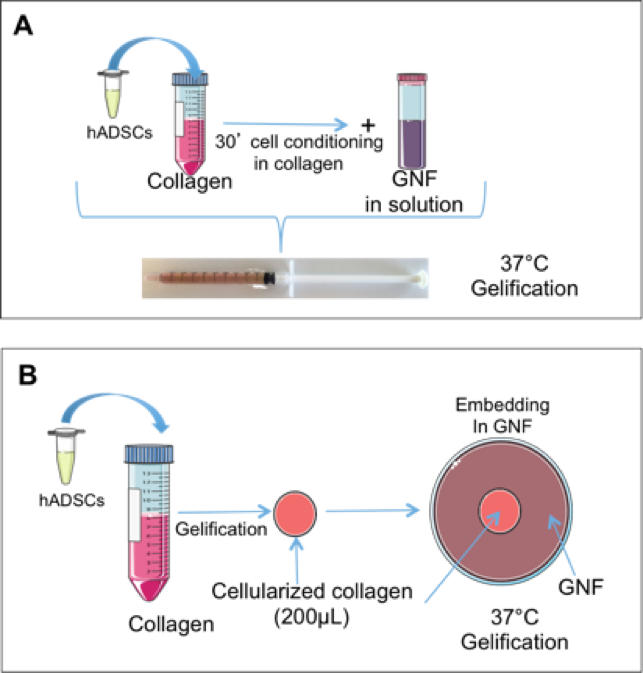
In order to establish a suitable microenvironment for cell proliferation and differentiation we designed two types of gel: 1) Cells were seeded in collagen for 30 minutes and then GNF was added and the resulting mixture was blended (Fig. 1A) ; 2) cells were pre-seeded on a collagen gel and brought to jellification. Then, 200 μL cellularized collagen hydrogel were included in GNF solution that was then cooled down until GNF forms a gel. This procedure leads to two separate gel phases (Fig.1B).
Results show that pre-seeding cells in collagen improve cell survival, spreading and proliferation rather than simple blending of GNF-collagen-hASCs (Fig.2A1). Cells seeded in GNF-encapsulated collagen gel allowed homogenous adhesion and growth up to 20 days of culture(Fig.2B1). ALP positive cells were observed at 7 days in both gel formulations, in absence of osteogenic factor(Fig.2A2&B2).
This novel gel composite has the ability to sustain hASCs adhesion, proliferation and differentiation towards the osteoblastic lineage.

Adding collagen to GNF allowed to obtain gels showing satisfactory cell-behaviour.
These results allow us to proceed towards the study of the in vivo fate of hASC embedded
in these composite hydrogel systems. Two implantation sites have been selected: the heterotopic subcutaneous site, where we can assess osteoinductive potential; orthotopic calvarium bone critical defect in order to challenge the bone repair capacity of
the cell-hydrogel complex.
In parallel, the suitable behaviour of ASCs in GNF-embedded collagen prompts us to design different 3D organizations of gel combinations, and to analyse the influence of these structures on cell fate in vitro. One of the potential applications of such systems is
the trapping of osteoinductive proteins in GNF that would be released towards the cell-based collagen gel.
This work was partially supported by NSERC-Canada, FRQ-NT-Quebec, FRQ-S-Quebec, and CFI-Canada. Mathieu Maisani was awarded of a NSERC CREATE Program in Regenerative Medicine, www.ncprm.ulaval.ca. This work was also supported by CHU de Québec Division of Regenerative Medicine
References:
[1] S. Ziane, S. Schlaubitz, S. Miraux, A. it Patwa, C. Lalande, I. Bilem, S. Lepreux, B. Rousseau, J.-F. Le Meins, L. Latxague, P. Barthelimy, and O. Chassande, “A Thermosensitive Low Molecular Weight Hydrogel As Scaffold,” Eur. cells Mater., vol. 23, pp. 147–160, 2012.
[2] N. Rajan, J. Habermehl, M.-F. Coté, C. J. Doillon, and D. Mantovani, “Preparation of ready-to-use, storable and reconstituted type I collagen from rat tail tendon for tissue engineering applications.,” Nat. Protoc., vol. 1, no. 6, pp. 2753–8, Jan. 2006.
[3] F. Boccafoschi, J. Habermehl, S. Vesentini, and D. Mantovani, “Biological performances of collagen-based scaffolds for vascular tissue engineering,” Biomaterials, vol. 26, no. 35, pp. 7410–7417, 2005.