Introduction: Prostate cancer (PCa) is the second most frequent malignancy in men, with 1.1 million cases diagnosed annually[1]. Fortunately, most men are diagnosed when the malignancy is confined to the gland (localised disease). However, approximately 10-33% of patients will go on to progress to advanced, metastatic disease, which is ultimately lethal[2]. It is widely acknowledged that the tumour microenvironment (TME; stroma, extracellular matrix (ECM), vasculature, immune cells and hormones) plays a fundamental role in this disease progression. Despite this, most in vitro PCa models are simple, 2D monocultures of epithelial tumour cell lines. There is an immediate need to develop multi-cellular in vitro constructs to investigate the effect of the TME on tumourigenicity. To this end, we have developed a novel 3D bioengineered in vitro scaffold-based model, which aims to recapitulate specific stages of early prostate tumourigenesis, namely fibroblast-epithelial and immune-epithelial interactions.
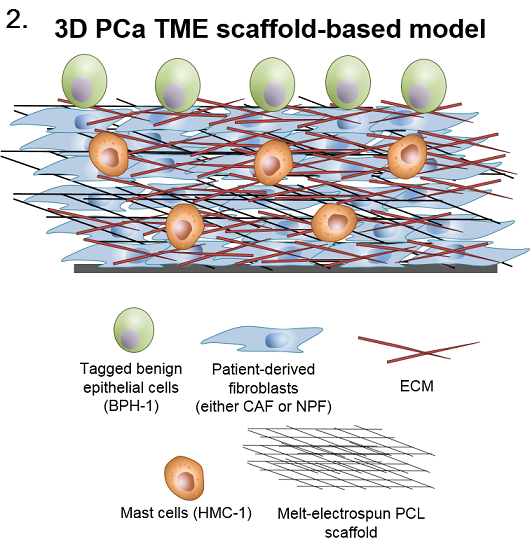
Materials and Methods: Melt-electrospun scaffolds were formatted from poly(ε-caprolactone) (PCL) polymer spun in a 0/90⁰ pattern, forming flexible 15x15mm2 meshes with a pore size of ~100µm. Scaffolds were plasma treated to enhance cell attachment. Validated patient-derived cancer-associated fibroblasts (CAF) or non-malignant prostatic fibroblasts (NPF)[3] were incorporated into the treated scaffolds. Once confluent, benign (initiated) epithelial cells (BPH-1) were co-cultured on the fibroblast constructs +/- mast cell (MC) conditioned media. At day 1, scaffolds were fixed and tumourigenicity was assessed by analysing the morphological transformation of BPH-1 cells (sphericity, volume, orientation, migration) using confocal microscopy and Imaris software.
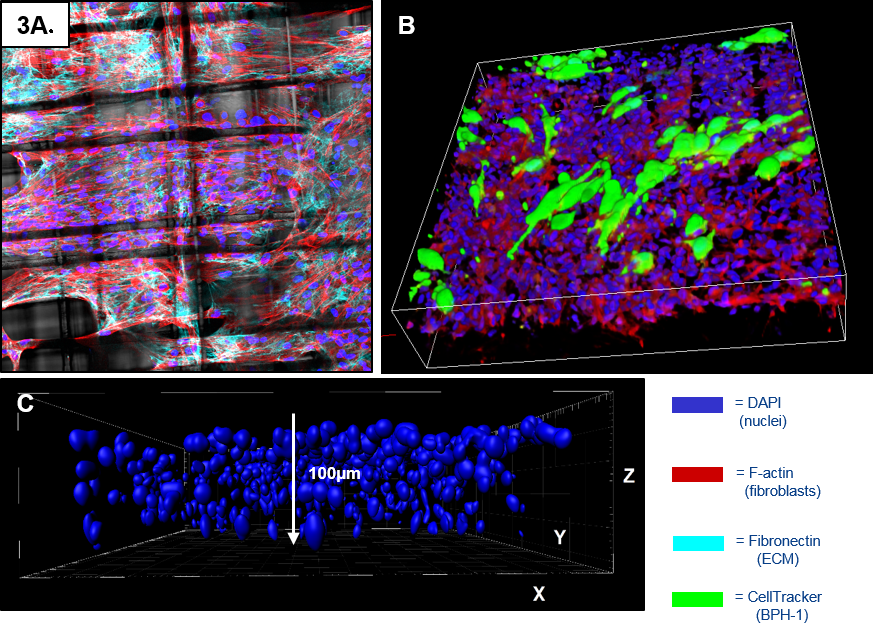
Results: CAFs and NPFs proliferate and deposit ECM to form a 3D stromal network within the scaffolds. Cell penetrance is ~90µm (6-8 cell layers). CAF, but not NPF, induce an invasive morphology in the benign epithelium, recapitulating the in vivo biology[4]. Significantly, when an immune component was incorporated – MC conditioned media –the CAF-induced transformation of BPH-1 cells was further potentiated. Ongoing work includes investigating the transcriptome of the BPH-1 cells when co-cultured with CAF/NPF +/- mast cells and proteomic analysis of CAF and NPF grown in both 2D and scaffold (3D) environments.
Conclusion: This 3D scaffold-based model provide a physiologically relevant in vitro platform to study the prostate TME, with data highlighting the fundamental contribution of fibroblast-epithelial and immune-epithelial interactions on early PCa progression.

Monash University; IHBI-QUT; IRTG Monash University-JLU Giessen; Prostate Cancer Foundation of Australia
References:
[1] Ferlay, J., et al. Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11. GLOBOCAN 2012 v1.0 2012 29/04/2014]; Available from: http://globocan.iarc.fr.
[2] Leach, D.A., et al., Stromal androgen receptor regulates the composition of the microenvironment to influence prostate cancer outcome. Oncotarget, 2015. 5.
[3] Lawrence, M.G., et al., A preclinical xenograft model of prostate cancer using human tumors. Nature protocols, 2013. 8(5): p. 836-848.
[4] Olumi, A.F., et al., Carcinoma-associated fibroblasts direct tumor progression of initiated human prostatic epithelium. Cancer research, 1999. 59(19): p. 5002-5011.