- 1UR OPAALE, IRSTEA, Rennes, France
- 2Université Bretagne Loire, Rennes, France
- 3Agroécologie, AgroSup Dijon, Institut National de la Recherche Agronomique, Université Bourgogne Franche-Comté, Dijon, France
Understanding how Listeria monocytogenes, the causative agent of listeriosis, adapts to the environment is crucial. Adaptation to new matrices requires regulation of gene expression. To determine how the pathogen adapts to lagoon effluent and soil, two matrices where L. monocytogenes has been isolated, we compared the transcriptomes of L. monocytogenes CIP 110868 20 min and 24 h after its transfer to effluent and soil extract. Results showed major variations in the transcriptome of L. monocytogenes in the lagoon effluent but only minor modifications in the soil. In both the lagoon effluent and in the soil, genes involved in mobility and chemotaxis and in the transport of carbohydrates were the most frequently represented in the set of genes with higher transcript levels, and genes with phage-related functions were the most represented in the set of genes with lower transcript levels. A modification of the cell envelop was only found in the lagoon environment. Finally, the differential analysis included a large proportion of regulators, regulons, and ncRNAs.
Introduction
Listeria monocytogenes, the causative agent of listeriosis, can be found in farm environments (Nightingale et al., 2004; Vilar et al., 2007; Esteban et al., 2008; Boscher et al., 2012; Pourcher et al., 2015). Depending on the type of livestock waste (solid or liquid manure, litter) and management (static pile, composting, storage in a tank, aerobic or anaerobic digestion), L. monocytogenes may survive waste treatment. In previous studies on piggery effluents, we provided evidence for higher prevalence of L. monocytogenes in the liquid phase of manures treated aerobically (lagoon effluents) than in raw manures (Pourcher et al., 2015) and demonstrated that viable cells of L. monocytogenes entered the viable but non-culturable state within the first hours of contact with piggery lagoon effluents (Desneux et al., 2016). We also observed the presence of L. monocytogenes in soils located in the vicinity of the lagoons (Pourcher et al., 2015). L. monocytogenes can survive for more than a month in soil amended or not with livestock effluents (Jiang et al., 2004; Hutchison et al., 2005; Nicholson et al., 2005; Moynihan et al., 2015). Its persistence depends on intrinsic factors (physiological state, concentration), and on abiotic (physical and chemical properties, sunshine, precipitation, temperature) and biotic environmental factors (production of inhibitory molecules, competition for substrate, protozoan grazing). The manure application method also affects the survival of L. monocytogenes in the soil. Indeed, Hutchison et al. (2004) reported that incorporating livestock effluent increases the survival of manure-borne pathogens after spreading of the effluent. The presence of L. monocytogenes in livestock feces can therefore contaminate the soil when livestock effluents are spread and, following rainfall events, can contaminate surface waters through runoff and groundwater through infiltration. While rainfall appears to be a major factor in the dissemination of L. monocytogenes in water systems, temperature and the presence of livestock are also important (Lyautey et al., 2007; Wilkes et al., 2009; Cooley et al., 2014). For example, in Canada, Lyautey et al. (2007) analyzed 314 samples of surface water and observed that the number of samples positive for L. monocytogenes was higher in areas close to cattle farms. Similarly, among the 53 soil samples from 23 Burgundy sites analyzed by Locatelli et al. (2013a) (three cow pastures, eight cultivated fields, 11 meadows, and one forest), L. monocytogenes was isolated only in soils from pastures.
The ability to survive in these environments can be explained by the extensive regulatory repertoire identified in the genome of L. monocytogenes (Glaser et al., 2001), which represents 7.3% of the genome of L. monocytogenes EGD-e. In response to environmental cues including starvation (Herbert and Foster, 2001), changes in temperature (Liu et al., 2002; Cacace et al., 2010; Mattila et al., 2011), pH (Abram et al., 2008; Giotis et al., 2008; Chen et al., 2011), and osmotic variations (Duche et al., 2002; Bergholz et al., 2012), L. monocytogenes can adapt its physiology by regulating gene expression. This includes genes encoding cell surface proteins, secreted proteins, transporters, and proteins involved in motility and chemotaxis. In addition to the coding part of the genome, evidence suggests that non-coding RNAs (ncRNAs) are key regulators of the environmental adaptation of L. monocytogenes (Christiansen et al., 2006; Mandin et al., 2007; Nielsen et al., 2008, 2011; Mellin and Cossart, 2012).
Only a few authors have performed differential analysis at gene-level resolution in complex matrices. These include food-related conditions, food matrices (cabbage, milk, turkey; Palumbo et al., 2005; Liu and Ream, 2008; Bae et al., 2011), and disinfectants used in the food industry such as the benzalkonium chloride (BZT; Fox et al., 2011; Casey et al., 2014). To date, only one study (Piveteau et al., 2011), has described transcriptional modifications induced by the transfer of L. monocytogenes to an environmental matrix. Whatever the strain analyzed (EGD-e, 6179, F6854, F2365, or 10403) and the technology used (DNA microarray, RT-PCR, RNA-seq), a similar scenario was described with higher transcript levels of genes involved in the transport of carbohydrates (Bae et al., 2011; Piveteau et al., 2011; Casey et al., 2014), in the metabolism of amino acids (Liu and Ream, 2008; Piveteau et al., 2011) or in motility and in chemotaxis (Palumbo et al., 2005; Casey et al., 2014). Interestingly, while genes involved in the virulence were not differentially transcribed in food matrices or in presence of BZT (Liu and Ream, 2008; Casey et al., 2014), they had lower transcript levels in soil extracts (Piveteau et al., 2011), suggesting an effect of the matrix on the regulation of gene expression of L. monocytogenes.
Given the significant role of manure application in the dissemination of L. monocytogenes in farmland and the lack of data on its physiology in the agricultural environment, it is important to characterize the transcriptome of this pathogenic bacterium in both livestock effluents and soil. The main objective of this study was thus to identify the response of a strain isolated from manure (L. monocytogenes CIP 110868) 20 min and 24 h after being transferred to a lagoon effluent or a soil extract by differential analysis using RNAseq.
Materials and Methods
Bacterial Strain and Culture Media
L. monocytogenes CIP 110868 (serogroup IVb) was isolated from pig manure. The strain was grown at 30°C in a nutrient broth supplemented with 0.3% glucose (NBG). Three independent inocula were prepared by inoculating 100 ml of NBG to an O.D600 nm of 0.04 and incubating overnight at 30°C to an O.D600 nm of 0.4. The cultures were then centrifuged at 5,000 g for 5 min and the pellets were suspended in 0.8% NaCl solution. After a second washing step, the cells were suspended in saline to reach a final concentration of 5 × 109 CFU/ml.
Sampling and Preparation of Soil Extract and Lagoon Effluent Microcosms
Soil and lagoon effluent were collected in sterile flasks from a pig manure treatment processing unit located in Brittany (France). The treatment consists of pre-storage and centrifugation of the manure to remove phosphorus. The liquid phase, which is biologically treated to reduce the level of nitrogen, is then left to settle. The liquid fraction in the settling tank is sent to an open-air lagoon where it is stored for 9–12 months. The sample of silty topsoil (0–20 cm) was collected near the lagoon. The two matrices were stored at 4°C until analysis.
Soil extract was prepared according to Piveteau et al. (2011). Briefly, 500 g of soil were mixed in 750 mL of water and autoclaved at 130°C for 1 h. The soil suspension was centrifuged at 10,000 g for 20 min and the supernatant was filtered on Whatman® 3 MM paper. The soil extract obtained after filtration was used after autoclaving at 120°C for 20 min.
The lagoon effluent extract was prepared by centrifugation of the effluent at 10,000 g for 10 min. The supernatant was filtered through Whatman® 3 MM paper and Whatman® qualitative filter paper Grade 2 (8 μm). The extract was then filter-sterilized on cellulose ester membrane pore size 3, 1.2, 0.8, 0.45, and 0.22 μm and transferred to sterile flasks. Sterility was confirmed by measuring culturable bacteria on plate count agar (PCA) incubated at 30 and 37°C for 48 h. Culturable bacteria were not found in either the soil extract or in the effluent.
Microcosms (30 ml) of both the soil extract and the lagoon effluent were prepared in triplicate.
Inoculation of the Microcosms
Soil extract and lagoon effluent microcosms were inoculated with L. monocytogenes CIP 110868 to reach a final concentration of 5 × 108 CFU/ml. Immediately after inoculation, the microcosms were incubated under static conditions at 21°C for 20 min (T1) and 24 h (T2). The temperature of 21°C was selected to mimic the average summer temperature in piggery lagoons in Brittany. In each treatment, three independent microcosms were inoculated with three independent inocula.
RNA Extraction and Purification
Total RNA was extracted from the culture broth (T0) and from the inoculum after 20 min (T1) and 24 h (T2) of incubation. RNAs were immediately stabilized by treating 2 ml of the bacterial cultures with RNA protect bacterial reagent (Qiagen, France) according to the manufacturer's instructions. Three independent replicates were prepared and treated separately. Four RNA extractions were performed of each replicate.
RNA was isolated according to the protocol described by Piveteau et al. (2011). Briefly, bacteria were harvested by centrifugation at 5,500 g for 5 min. Pellets were suspended in 700 ml of RLT buffer supplemented with 1% β-mercaptoethanol and 0.2 g of RNase-free glass beads (100 μm). Cells were mechanically disrupted in a Fast Prep®-24 instrument (MP Bio, France) with four cycles (6 m/s, 30 s). Total RNA was isolated using the Qiagen RNeasy kit (Qiagen, France) according to the manufacturer's instructions. After treatment with DNase I (DNAse on column, 10 U, Qiagen), RNA was eluted in 40 μl of RNase-free water. The four eluates of the same replicate were then pooled and treated with RQ1 RNase free DNase (Promega, France) in RNasin (40 U, Promega). RNAs were purified and concentrated using the RNeasy MinElute cleanup kit (Qiagen, France) according to the manufacturer's instructions. The quality and purity of the RNAs were determined spectrophotometrically and by capillary electrophoresis using the Bioanalyser 2100 (Agilent, France).
All RNAs were stored at −80°C until analysis.
Sequencing and Data Analysis
RNA stabilization, 16S, 23S, and 5S RNA depletion, library preparation (“strand specific” method), clustering and sequencing (illumina HiSeq2000) were performed by Sistemas Genómicos (Valence, Spain) according to the manufacturer's instructions. Sequenced reads were quality-checked using FastQC. Reads were mapped against the most recent version of the L. monocytogenes EGD-e genome provided by NCBI database, and quantified. The statistical analysis performed by Sistemas Genómicos is described below. The differential expression study between groups of samples was achieved using Python and R statistic packages. The gene differential expression was studied with the algorithm proposed by DESeq2 (Anders and Huber, 2010; Anders et al., 2013) using a negative binomial distribution as dispersion model. Using the functional annotation from Uniprot database (http://www.uniprot.org/), Sistemas Genómicos realized a hyper-geometric test using different statistical packages from statistical platform R to performed the functional enrichment study. T-statistics and p-values (FDR adjusted p-value of 0.05) were calculated to identify differentially expressed genes. The fold changes at T1 and T2 were calculated by comparing results with expression levels at time 0.
Genes with at least a three-fold change were considered significantly different in the interpretation applied in this study. Finally, functional studies and differential analyses were performed between samples. Genes differentially transcribed were grouped according to their functional category (Glaser et al., 2001).
Results and Discussion
This study investigated variations in the transcriptome of a strain of L. monocytogenes isolated from pig manure after transfer to lagoon effluent or to soil extract to better understand the mechanisms behind its physiological adaptation to these environments. The two matrices selected were (i) a lagoon filled with biologically treated manure and (ii) soil located in the vicinity of the lagoon, which may receive manure containing L. monocytogenes. Raw reads ranged between 3.1 × 107 and 3.8 × 107 in all samples. After removing low quality reads, the average number of properly paired mapped reads ranged between 1.9 × 107 and 2.9 × 107 for the datasets from cells transferred in lagoon effluent (62.8–76.7% of properly paired reads), and from 3.0 × 107 to 3.6 × 107 for the datasets from cells transferred in soil extract (92.2–95.8% properly paired reads). Among the differentially expressed entries (with a fold change ≥2), the total number of genes which had mapped reads was 2,267 for the 4 conditions (soil and lagoon at 20 min and 24 h). As shown in Figure 1, each condition tested led to differences in transcriptomes. PCA also indicated homogeneity within each condition except after 20 min in the soil.
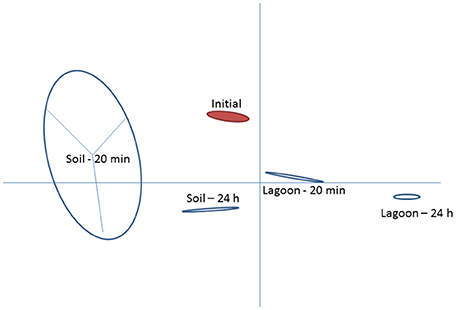
Figure 1. Principal component analysis of Listeria monocytogenes transcriptomes after 20 min and 24 h of incubation in lagoon effluent and soil extract.
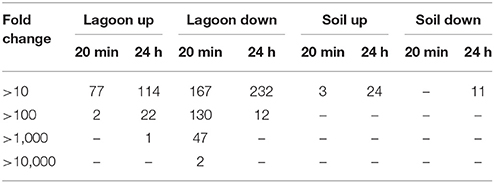
The variations of transcriptome were higher in lagoon than in soil. A total of 1,513 genes were differentially transcribed in the lagoon effluent, 585 with higher and 928 genes with lower transcript levels. In the soil, 458 genes were differentially expressed of which 212 with higher and 246 genes with lower transcript levels (Figure 2). While the percentage of properly paired reads supports a comprehensive overview of the transcriptome variations during incubation in soil, the fraction of unexplained changes was higher in the lagoon effluent as suggested by the lower percentage of paired reads. A total of 1,513 genes were differentially transcribed in the lagoon effluent, 585 with higher and 928 genes with lower transcript levels. In the soil, 458 genes were differentially expressed of which 212 with higher and 246 genes with lower transcript levels (Figure 2). Interestingly, the values of fold changes observed in differentially expressed genes were lower in the soil than in the lagoon effluent (Table 1). Indeed, in the soil, fold changes went from −14.7 to +48.3, whereas in the lagoon effluent, they went from −19,267 to +2,146.
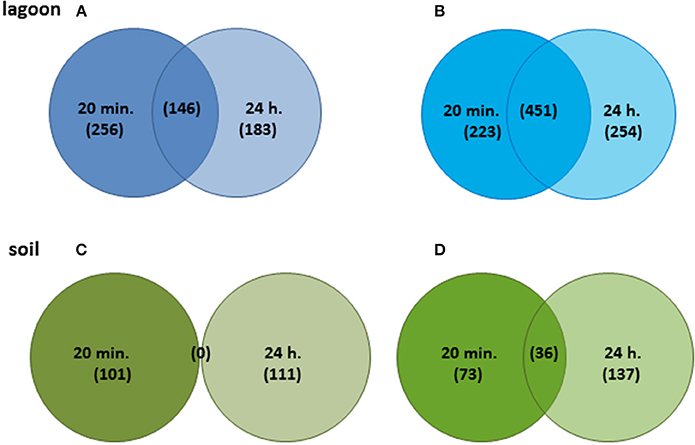
Figure 2. Venn diagrams of genes showing significant differences after 20 min and 24 h. (A) Genes with higher transcript levels in the lagoon effluent, (B) genes with lower transcript levels in the lagoon effluent, (C) genes with higher transcript levels in the soil extract, (D) genes with lower transcript levels in the soil extract.
Marked Variations in the Transcriptome Detected in Lagoon Effluent
Overview
The transfer of L. monocytogenes CIP 110868 into the lagoon effluent caused substantial changes of the transcriptome. Within 20 min, 1,076 genes were differentially transcribed. After 24 h, the number of up or down regulated genes remained stable, with 1,034 genes identified with a different transcript level, indicating that transcriptome reshaping took place within minutes following contact with the effluent (Table S1). A high proportion of operons were differentially transcribed (Table 2). Seventy-four complete operons including 218 genes were differentially expressed after 20 min of incubation, which represents 20% of the set of genes with different transcript levels. After 24 h, the proportion increased to 26% with 76 operons including 268 genes. A large set of transcriptional regulators was differentially transcribed at T1 and T2 (functional category 3.5.2, Table 3, Table S1). Among the 209 regulators described in the genome of L. monocytogenes EGD-e (Glaser et al., 2001), 76 (36%) were differentially transcribed after 20 min and 83 (40%) after 24 h. Interestingly, most had a lower transcript level. These results confirm the pivotal role of the extensive regulatory repertoire in the ability of L. monocytogenes to survive in various environments and to adapt its physiology (Gandhi and Chikindas, 2007). Therefore, it is not surprising that a high proportion of transcriptional regulators and response regulators was detected in the set of differentially transcribed genes. Table 4 lists the differentially transcribed regulators for which biological functions have been described. Similarly, high proportions of the regulons (from 31.4 to 59.4%) of sigma factors and other transcriptional regulators were differentially transcribed after 20 min and 24 h (Table 5). This suggests these regulons may be important for adaptation to the lagoon environment.
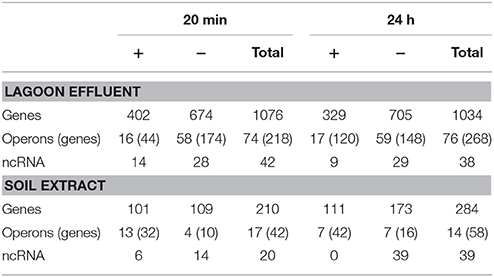
Table 2. Number of genes, operons and ncRNA differentially transcribed in the lagoon effluent and in the soil extract.

Table 3. Distribution of the genes differentially transcribed in the lagoon effluent in the different functional categories.
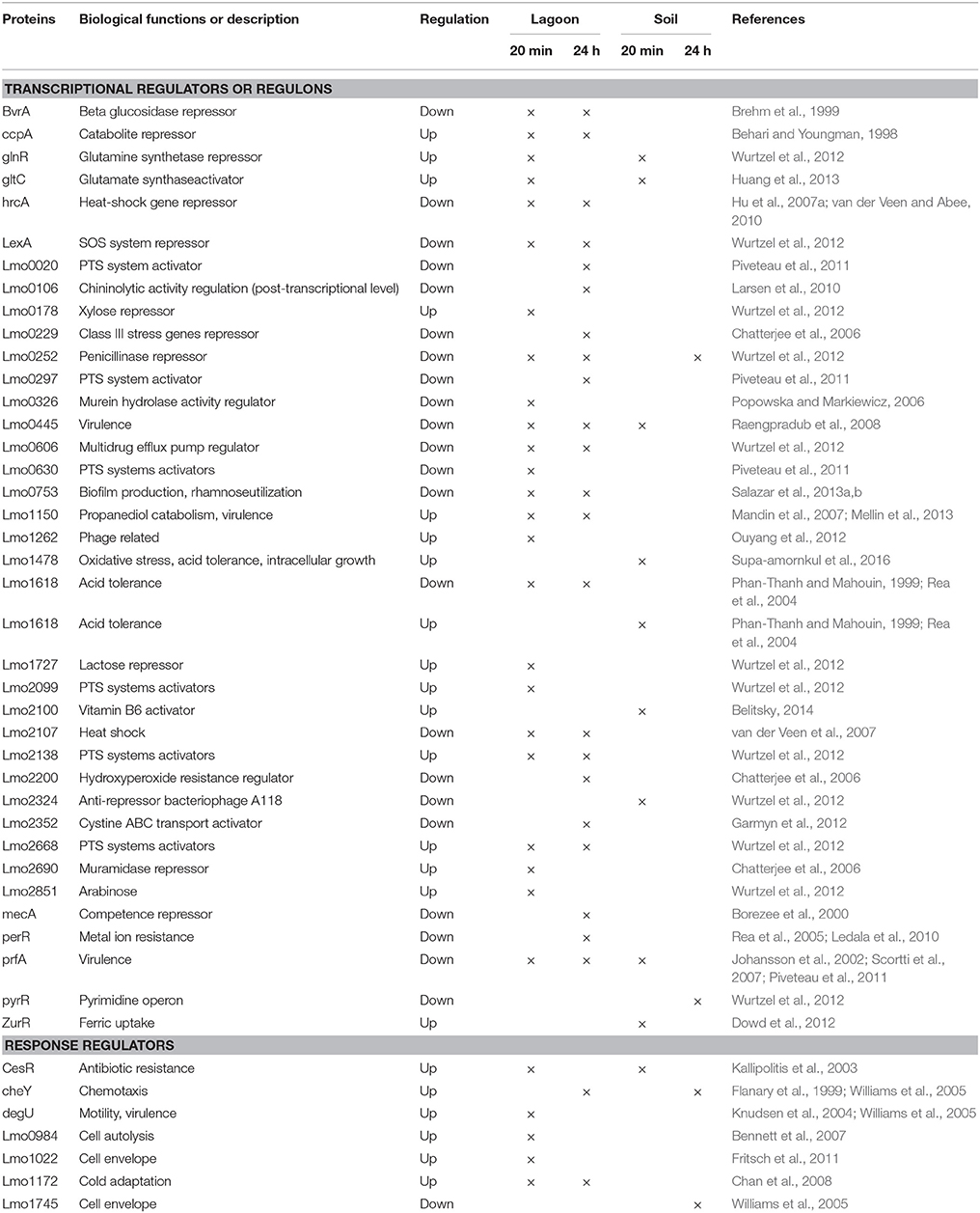
Table 4. Regulators differentially transcribed for which biological functions have already been described.
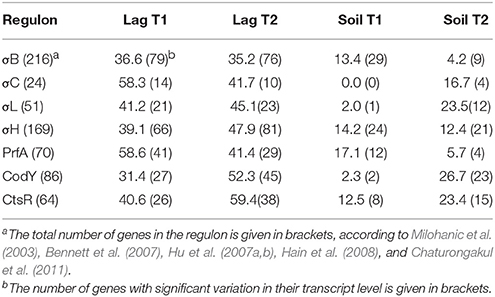
Table 5. Percentage of genes from sigma factors and other transcriptional regulators (regulons) with significant variations in transcription after 20 min (T1) and 24 h (T2) of incubation in lagoon effluent (Lag) and soil extract (Soil).
Upregulation of Transporters of Carbon and Energy Sources
A large repertoire of differentially transcribed genes illustrated the metabolic adaptation of L. monocytogenes CIP 110868 to its environment within the first minutes of incubation (Table 3). Transporters required for the use of available resources were highly represented, suggesting that a readjustment of the transcriptome occurred rapidly in order to access resources available in the lagoon environment. After 20 min of incubation in the lagoon, a total of 125 genes associated with transport were differentially transcribed. This represents 38% of the genes identified in the genome of L. monocytogenes EGD-e as being devoted to carbohydrate uptake (Glaser et al., 2001; Fichant et al., 2006). These genes included 42 phosphoenolpyruvate-dependent phosphotransferase systems (PTS systems) and 54 ATP-binding cassette transporters (ABC transporters). Among them, respectively 29 and 30 genes had higher transcript levels. These genes encoded proteins involved in glycerol (lmo1539), galactitol (lmo2667), fructose (lmo0039, lmo0400 and lmo0358), and maltose-maltodextrin (lmo2123-24-25) uptake. Moreover, arabinose and lactose transcriptional regulators (lmo2851 and lmo1727, respectively) and a xylose repressor (lmo0178) were identified in the set of genes with higher transcript levels. In addition, three known activators of PTS systems (lmo2138, lmo2099, and lmo2668) were also found to be upregulated. Genes encoding transporters found in the set of genes with lower transcript levels were mainly related to the transport of metal cations but none to the transport of carbohydrates.
The same pattern was observed after 24 h of incubation, except that the percentage of genes encoding transporters decreased from 38 to 31%.
Modification of the Cell Envelope
Throughout the experiment, genes involved in cell surface rearrangement were differentially transcribed. The functional categories “cell wall” and “cell surface proteins” were highly represented. Most of the genes were downregulated. For example, seven genes encoding teichoic acid synthesis had lower transcript levels. Similarly, the transcript level of genes encoding cell surface proteins also decreased. This included peptidoglycan bound proteins and internalins. As reviewed by Bierne and Cossart (2007), the adjustment of the cell envelope is of major importance for coping with new environmental conditions. Cell surface proteins fulfill many functions as diverse as bacterial growth, peptidoglycan metabolism, sensing of and protection from environmental stresses, adhesion and invasion of host cells and signaling. For example, teichoic acids, which are directly linked to the structure of the outer cell wall, enable reinforcement of the cell wall of L. monocytogenes when environmental conditions are unfavorable. The products of both genes of the tagGH operon are involved in translocation in teichoic acids. The operon, controlled by a σA-dependent promoter, was first described in Bacillus subtilis for which it is essential for cell growth (Lazarevic and Karamata, 1995). In L. monocytogenes, the operon plays a role in protecting cells against the biocide benzethonium chloride (Casey et al., 2014). Additional genes involved in the biosynthesis and the glycosylation of teichoic acids, such as ggaB, lmo1077, lmo1085, gtcA, tagB, and tagO, were also found in the set of genes with lower transcript levels. It is important to note that these genes were differentially transcribed with a fold change in the region of −1,000.
The variations in transcript levels observed in these functional categories point to the need for a readjustment of the cell surface to adjust to the new environment.
Protection of L. monocytogenes against Antibiotics and Metal Ions
lmo0441, pbpA, pbpB, lmo2229, and lmo1438 genes were positively transcribed. These penicillin-binding protein encoding genes have been shown to be involved in cell wall functioning (Rismondo et al., 2015), cell division during growth (Krawczyk-Balska et al., 2012) and resistance to β-lactam antibiotics (Van de Velde et al., 2009; Collins et al., 2012). Moreover, the penicillinase repressor lmo0252 had a lower transcript level, suggesting deactivation of repression and the production of penicillinase. Similarly, the transcriptional repressor (lmo2690) from the TetR family, which targets positive regulators of multi-drug efflux pumps, had a lower transcript level, indicating that the repression of the pumps may be lifted in the effluent. We also found the tetracycline resistance protein coding gene lmo0839 in the set of genes with higher transcript levels. The response regulator lmo1022, which resembles the LiaSR two-component system of B. subtilis known to be induced when antibiotics interfere with the cytoplasmic membrane (Fritsch et al., 2011), was found to be activated. Finally, lmo1967 was also activated. lmo1967, first described for toxic ion resistance, is required for optimum resistance to nisin and other antimicrobial molecules (oxacillin, cefuroxime, cefotaxime, methicillin, and bacitracin; Collins et al., 2010). These data suggest that mechanisms of protection against antibiotics had been triggered. Conversely, other genes (lmo0872, lmo1409, and lmo1617), also implicated in antibiotic resistance and in the functioning of multi-drug efflux transporter, were downregulated.
Antibiotics (mainly oxytetracycline and chlortetracycline) are frequently administered to pigs and animals may excrete from 25 to 90% of the administered antibiotics as parent compounds. As a consequence, high concentrations of antibiotics are detected in manures (Qian et al., 2016). For example, oxytetracycline and chlortetracycline have been detected at concentrations in the order of mg per kg of dry weight in manure samples (Widyasari-Mehta et al., 2016). As a result, it is possible that lagoon effluents (liquid fraction of biologically treated manure) are also contaminated by antibiotics and that L. monocytogenes had to face these molecules in the lagoon effluent, as our results suggest.
Metal ions are commonly detected in piggery effluents (Xiong et al., 2010; Hölzel et al., 2012). For example in the lagoon effluent used in this study, copper and zinc were detected at concentrations of 16 and 55 mg/L respectively (data not shown). The upregulation of lmo1478, a transcriptional regulator of the MerR family, within the first minutes of incubation in the effluent, suggests that L. monocytogenes responded to exposure to metal ions. In contrast, genes encoding efflux systems for cadmium/arsenic and for zinc/arsenic transport were downregulated.
Activation of Other Stress Responses
The introduction of L. monocytogenes in harsh environments triggers the regulation of genes involved in stress responses (Piveteau et al., 2011; Casey et al., 2014).
A battery of genes involved in stress response was detected among genes with higher transcript levels throughout the course of the experiment. Briefly, the expression of the cold shock genes cspD and cspL increased. Upregulation of the opuC operon (opuCA, opuCB, opuCD) suggests a response to osmotic stress. This system is involved in the uptake of the osmoprotectants glycine-betaine, carnitine, and choline. These molecules have previously been shown to play a role in the osmoregulation and cold adaptation of L. monocytogenes (Fraser et al., 2000). The GltC activator also had a higher transcript level, suggesting that oxidative stress occurred in the lagoon environment. The LexA SOS system repressor and the repressor of class III stress genes Lmo0229 were in the set of genes with lower transcript levels. Finally, it is important to note that σB, which plays a central role in the stress response of L. monocytogenes and the RsbU phosphatase involved in the regulation of σB, had higher transcript levels. Among the 216 genes of the σB regulon (Hain et al., 2008), 79 were differentially transcribed after 20 min of incubation and 76 after 24 h.
All these modifications suggest that incubation in the lagoon triggered responses to environmental stresses. Conversely, regulators involved in adaptation to heat shock (Lmo2107; van der Veen et al., 2007) and acid shock (Lmo1618; Phan-Thanh and Mahouin, 1999; Rea et al., 2004) were in the set of genes with lower transcript levels. Some of these variations may be explained by the conditions under which inocula were prepared. Indeed, the pH of the broth and temperature used as reference differed from the conditions of the lagoon.
Activation of Amino Acid Metabolic Pathways
The number of genes dedicated to the metabolism of amino acids and related molecules increased with time, from 55 genes differentially transcribed after 20 min to 63 genes after 24 h. These genes are particularly involved in the synthesis of leucine and isoleucine (ilvDBC, leuABCD, ilvA), histidine (hisEIA, hisJ, hisC), threonine (thrC), glutamate and glutamine (serC, glnA), alanine (daI), glycine (lmo1349-50, glyA), arginine (argFDBJC, argGH), lysine (dapF), and tryptophan (trpABCD; Figure S1). In addition, several riboswitches (Tbox, lysine, glycine, TPP, purine, pyrimidine, glmS riboswitch), which regulate amino acid biosynthesis, were also found in the set of upregulated genes (Table S2).
Motility Triggered after 24 h Incubation
After 24 h of incubation in the lagoon environment, the genes involved in motility and chemotaxis were highly activated. In this functional category, 28 genes had higher transcript levels after 24 h but not after 20 min. Among the 28 genes, we identified the well-described flaA encoding flagellin (Grundling et al., 2004), motAB encoding flagellar motor rotation proteins (Michel et al., 1998) and fli, flg, and flh genes. Interestingly, these genes belong to two operons, all of whose genes were also activated (42 genes). Among them cheYA encodes the two component system responsible for the regulation of chemotaxis in L. monocytogenes (Flanary et al., 1999; Williams et al., 2005). Motility may be a strategy used by cells to cope with their environment. Several of these genes (for example flaA and motAB) are thermoregulated, with higher expression at 25°C (saprophyte life in the environment) and lower expression at 37°C (intracellular life; Michel et al., 1998; Grundling et al., 2004).
Virulence Genes Downregulated
As for some genes involved in motility, temperature is an environmental cue that affects PrfA activity and expression of its regulon (Freitag et al., 2009; de las Heras et al., 2011). During the saprophytic lifestyle of the bacteria (at temperatures below 30°C), the secondary structure of the mRNA of prfA forms a hairpin structure which masks the ribosome binding region. In this conformation, the thermosensor represses the translation of prfA mRNAs. prfA was downregulated throughout the course of our experiment. Among the 70 genes referenced in the PrfA regulon (Milohanic et al., 2003; Chaturongakul et al., 2011), 41 had different transcript levels after 20 min and 29 after 24 h. As a result of introducing L. monocytogenes in an environmental matrix, genes specifically involved in the adaptation of the pathogen to the intracellular environment were downregulated. Internalin-coding genes inlB, inlC, inlE, inlG, and inlH were in the set of genes with lower transcript levels. Similarly, lmo0754, lmo2067, and lmo0446, which encode bile acid hydrolases in response to acid stress, lmo0445, a candidate regulator of virulence genes (Raengpradub et al., 2008) and lmo2200, an ortholog regulator of ohrR in B. subtilis, which is essential for intracellular growth (Chatterjee et al., 2006) were downregulated.
Genes in the “Transformation/Competence” Category Upregulated
Genes of the ComG (comGA and comGB) and ComE (comEB and comEC) operons were found to have a higher transcript level in the lagoon environment both at 20 min and 24 h. They are similar to B. subtilis comG and comE operons, which encode proteins enabling he development of competence and the acquisition of extracellular DNA (Dubnau, 1997). The MecA regulatory protein, which represses competence genes (Borezee et al., 2000), was in the set of genes with lower transcript levels. While the Com master activator is disrupted by a Listeria-specific prophage in some strains including EGD-e, other strains harbor the full version of comK. Although competence has never been demonstrated in L. monocytogenes, Rabinovich et al. (2012) showed that excision of the prophage can allow functional transcription of comK and that the role of Com systems in Listeria might have diverged and could be useful for phagosome escape and virulence. Alternatively, competence may be possible under some environmental conditions yet to be identified such as lagoons. Some isolates might be competent even though signals inducing competence have not been identified (Buchrieser et al., 2003).
Downregulation of ncRNAs after Incubation in the Lagoon Effluent
Whatever the incubation time, a wide range of ncRNAs was detected in the sets of differentially transcribed targets (Table S2) most of which presented a lower transcript level. Like in other bacterial models, ncRNAs play a central role in the adaptation of L. monocytogenes to its environment. So far, a role for the non-coding part of the genome has been reported in the regulation of virulence (Mandin et al., 2007; Loh et al., 2009; Toledo-Arana et al., 2009; Mraheil et al., 2011; Nielsen et al., 2011; Mellin and Cossart, 2012), in the transport and metabolism of sugars (Mandin et al., 2007; Nielsen et al., 2011), in the transport of metal ions (Wurtzel et al., 2012), in the synthesis of vitamins (Mellin and Cossart, 2012), in the formation of biofilm (Peng et al., 2016) and in the regulation of motility (Mellin and Cossart, 2012). In our study, we found that Rli28, Rli31, Rli38, Rli49, and Rli50, which are involved in invasion of the host's blood system, in the regulation of virulence factors (Toledo-Arana et al., 2009), in intracellular multiplication in mouse macrophage P338D1 and complete virulence in Galleria mellonella models (Mraheil et al., 2011), had lower transcript levels after incubation in the lagoon environment. Similarly, the virulence regulators LhrC1, LhrC2, LhrC3, and LhrC4 riboswitches were deactivated. Conversely, Rli60 was found in the set of ncRNAs with higher transcript levels. This ncRNA has recently been reported to play a role in the response (growth and biofilm formation) of L. monocytogenes EGD-e to environmental stresses (temperature, pH, osmotic pressure, alcohol; Peng et al., 2016).
Highly Differentially Transcribed Genes
To extract more information on the response of L. monocytogenes to the lagoon environment, we next focused on genes for which at least 10-fold increase was observed. A total of 568 genes met this criterion (fold change >10 = 183 genes and fold change < −10 = 385 genes). Among the genes with a 10-fold higher transcript level, functional categories 1.2 (transport/binding proteins and lipoproteins), 2.1.1 (specific pathways), 3.5.2 (regulation), and 5.2 (similar to unknown proteins from other organisms) were the most widely represented. Interestingly, the genes lmo0049 (agrD) and lmo0050 (agrC), encoding respectively the propeptide AgrD (precursor of the auto-inducing peptide) and the histidine kinase AgrC, were in this set of genes. AgrD and AgrC are a part of the Agr communication system (Rieu et al., 2007). This indicates that communication between bacteria might have been induced in the lagoon environment. As the Agr communication system is involved in the overall adaptation of the cell (Rieu et al., 2007; Riedel et al., 2009; Vivant et al., 2014) by integrating environmental cues, it was not surprising to find a marked increase in the transcript levels of the agrD and agrC genes. The functional categories highly represented in the set of genes with fold changes lower than −10 differed. We identified categories 3.5.2 (regulation), 4.3 (phage-related functions), 5.2 (similar to unknown proteins in other organisms), and 6 (no similarity). Genes with phage-related functions are often described in studies involving differential analysis (Piveteau et al., 2011; Casey et al., 2014). As described in the study by Piveteau et al. (2011), transcript levels of phage-related genes decreased within the first minutes of incubation (42 of the 48 phage-related genes at 20 min) and throughout the course of the experiment (36 genes at 24 h). However, the function of these modifications is still not known. In our study, the majority of the genes in the “phage-related functions” category were related to the Listeria phage A118.
The Transfer of L. monocytogenes in Soil Led to a Slight Modification of Its Transcriptome
Smaller variations in the transcriptome were observed after incubation of L. monocytogenes CIP 110868 in the soil than in the lagoon (Table S3). We detected 210 genes with different transcripts level after 20 min of incubation and 284 genes after 24 h of incubation in the soil extract (Table 2, Figures 2C,D). Another difference between the sets of differentially transcribed genes from lagoon and soil is that, after incubation of L. monocytogenes CIP 110868 in soil extract, fold changes were low (Table 1). Indeed, of the 210 differentially transcribed genes at 20 min, only three had fold changes >10 (one transporter and two unknown proteins). On average, fold changes ranged from −3.5 to 4.4. A similar pattern was observed after 24 h of incubation, when 35 of the 284 genes were highly differentially transcribed (fold change >10 or < −10). These genes were mainly dedicated to transport, mobility and unknown functions.
As described in the lagoon environment, complete operons were differentially transcribed during incubation in soil extract (Table 2). Transcription and response regulators were differentially transcribed but their proportion was smaller than in the lagoon environment. The percentage of regulators differentially transcribed was 5.7 and 4.6% of the genes differentially transcribed at 20 min and 24 h in the soil, whereas these proportions were 7.1 and 8% in the lagoon. The proportion of genes under the control of regulators and sigma factors was also smaller. Interestingly, the proportion of genes of the σC, σL, and CodY regulons in the set of differentially transcribed genes increased after 24 h (Table 5).
Surprisingly, genes with higher transcript levels differed completely at T1 and T2 (Figure 2C). In contrast, 37 genes with lower transcript levels were common at the two incubation times. They encoded unknown proteins and the internalin InlC.
Motility
Like in the lagoon environment, activation of a large panel of genes involved in motility was observed after 24 h of incubation (Table 6), including the regulator CheY. This confirms that motility is a strategy used by L. monocytogenes during its saprophytic lifestyle. For example, motility offers the opportunity to access a wide range of nutrients.

Table 6. Distribution of the genes differentially transcribed in the soil extract in the functional categories.
Transport/Binding Proteins
Incubation of L. monocytogenes for 20 min in the soil extract led to the deactivation of transporters dedicated to fructose, sorbitol, and ribulose and the activation of ABC transporters of metal ions. Moreover, transporters of spermidine and putrescine were also present in the set of genes with higher transcript levels. These two polyamines are involved in numerous biological processes and are essential for biofilm formation (Burrell et al., 2010; Wortham et al., 2010; Sakamoto et al., 2012). As confirmation that L. monocytogenes was introduced in a natural environment, a PTS system dedicated to the transport of lichenan, a polysaccharide isolated from lichen, was also activated.
The proportion of genes from category transport and binding protein was higher at 24 h (Table 6). Our results illustrate the requirement to acquire substrates available in the soil extract. This included plant residues such as cellobiose (lmo2708, lmo2762-63 and lmo2765), mannitol (lmo2651), maltose and maltodextrin (lmo2122-26), rhamnose, and rhamnulose (lmo2847-49), residues of arthropods and fungi such as chitin (lmo0105 and lmo1883) and residues of algae and plant exudates such as galacticol (lmo2665-67). The upregulation of transporters to acquire substrates from various sources in soil has already been described (Piveteau et al., 2011).
Stresses Encountered
Lesser variations of transcripts of genes related to stress response were detected from soil than from the lagoon environment, as only a few genes dedicated to stress response were differentially transcribed. They are mainly involved in the adaptation of L. monocytogenes to changes in temperature. Broth and incubation temperatures differed by about 10°C in our experiment, which explains the response to thermal stimuli. We identified as downregulated the genes encoding the cold shock proteins CspB and CspL and the heat shock protein GroES. The transcriptional regulator NmlR involved in oxidative stress and in tolerance to acid (Supa-amornkul et al., 2016) was found to be activated while the general stress protein encoded by lmo1601 was downregulated. L. monocytogenes is ubiquitous in the environment. The pathogen has been detected in soils (Locatelli et al., 2013a) and its ability to survive in this environment for long periods has been repeatedly demonstrated (Dowe et al., 1997; Moshtaghi et al., 2009; McLaughlin et al., 2011; Locatelli et al., 2013b).
Virulence and Intracellular Determinants
The virulence master regulator PrfA, the internalin InlC, and a bile acid hydrolase (lmo2067) were downregulated 20 min after L. monocytogenes was introduced in the soil extract. This is consistent with the saprophytic lifestyle of the bacteria. These observations are consistent with the observations made by Piveteau et al. (2011). In their study, transcript levels of virulence determinants including PrfA and internalins decreased 18 h after L. monocytogenes was transferred in soil extract microcosms.
ncRNAs
As confirmation that virulence factors were downregulated within the first minutes of incubation, ncRNAs involved in the regulation of virulence (Rli31, Rli50, and Rli47) were in the set of ncRNAs with lower transcript levels after 20 min of incubation (Table S4). Rli31 and Rli50 are required for full virulence in murine, larva, and macrophage infection models (Mraheil et al., 2011), and Rli47 (sreB) is a trans-acting ncRNA which controls the expression of the regulator PrfA by binding the untranslated region of its mRNA. Seven ncRNAs were found at both incubation times (Table S4) and all had lower transcript levels. Their functions are not yet described in the literature, except for RliI, which is involved in the regulation of the metabolism and the transport of carbohydrates (Mandin et al., 2007). Interestingly, after 24 h, no ncRNAs with higher transcript levels were detected, while a larger proportion had lower transcript levels. Among them, we mainly identified the four LhrC (1, 2, 3, and 4) riboswitches and Tbox and SAM riboswitches.
Phage-Related Functions
As observed in the lagoon effluent, several genes from functional category phage-related functions were in the set of genes with lower transcript levels within the first minutes of incubation in the soil extract (Table 6). These targets matched with genes coding proteins from bacteriophage A118.
Similar Readjustments of the Transcriptome of L. monocytogenes in the Lagoon Effluent and in the Soil
Regardless of the incubation time (20 min and 24 h), a large fraction of the genes differentially expressed in the soil extract (42–78%) were shared with the lagoon effluent (Figure S2). The adaptation of L. monocytogenes CIP 110868 to the natural environment led to similar readjustments of the transcriptome (Figure 3). Whether L. monocytogenes was inoculated in the lagoon effluent or in the soil extract, genes encoding proteins involved in motility and chemotaxis and in the transport of carbohydrates were the most widely represented in the set of genes with higher transcript levels. This is commonly reported in the literature, except that nutrients acquired by bacteria differ depending on the environment. Within genes with lower transcript levels, in both conditions, we mainly found genes in the “phage-related functions” category. It is worth mentioning that a large proportion of the genes differentially transcribed after the introduction of L. monocytogenes CIP 110868 in the natural matrices, encoded proteins with unknown functions. This represents from 25% of the set of genes with higher transcript levels to 60% of the set of genes with lower transcript levels. Finally, the proportion of genes from the PrfA, CodY, and CtsR regulons increased between 20 min and 24 h both in the lagoon and in the soil.
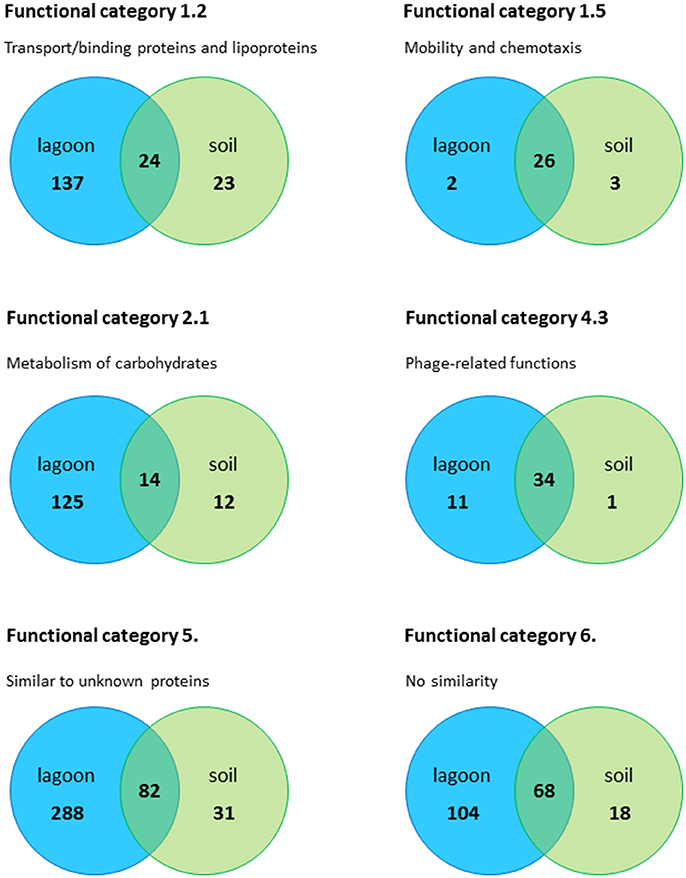
Figure 3. Number of genes in the lagoon effluent and in the soil extract, distributed in the most important functional categories.
Genes from the Category Cell Envelope Were Specifically Represented in the Lagoon Effluent
Comparison of differentially transcribed genes in soil and lagoon led to the identification of specific variations. Three functional categories (cell wall, competence, and metabolism of amino acids) were highly represented in the lagoon environment but not in the soil. Differences in the composition of the two matrices could explain such results. For example, the higher concentrations of detrimental molecules such as antibiotics and metal ions in lagoon effluents might trigger upregulation of genes leading to the adjustment of the cell wall for protection against toxic compounds.
Conclusion
In this study, we showed that the response of L. monocytogenes CIP 110868 differed according to the matrix and changed over time. Although, expression profiles fluctuated in the lagoon effluent and in the soil extract, similar mechanisms of adaptation were involved. The adaptation of strain CIP 110868 resulted in the activation of (i) genes encoding transport systems, (ii) genes encoding enzymes dedicated to the assimilation of carbohydrates and the production of energy and (iii) genes involved in motility and chemotaxis of the cells. Finally, we observed a higher proportion of differentially transcribed genes in the lagoon effluent than in the soil, suggesting that lagoon-specific environmental cues trigger multiple regulation routes required to coordinate the appropriate response to this environment.
Author Contributions
AP, PP, and JD designed laboratory work. JD performed all laboratory work. AP, PP, JD, and AV analyzed and interpreted of transcriptomic data. All authors co-wrote the manuscript. AV drafted the manuscript and AP finalized the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This work was supported by the French Agency for Food, Environmental and Occupational Health and Safety (ANSES) and by the French Environment and Energy Management Agency (ADEME). JD is recipient of an Irstea-Région Bretagne fellowship.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fmicb.2017.01811/full#supplementary-material
Table S1. Genes differentially expressed in the lagoon effluent.
Table S3. Genes differentially expressed in the soil extract.
References
Abram, F., Starr, E., Karatzas, K. A. G., Matlawska-Wasowska, K., Boyd, A., Wiedmann, M., et al. (2008). Identification of components of the sigma B regulon in Listeria monocytogenes that contribute to acid and salt tolerance. Appl. Environ. Microbiol. 74, 6848–6858. doi: 10.1128/AEM.00442-08
Anders, S., and Huber, W. (2010). Differential expression analysis for sequence count data. Genome Biol. 11:R106. doi: 10.1186/gb-2010-11-10-r106
Anders, S., McCarthy, D. J., Chen, Y. S., Okoniewski, M., Smyth, G. K., Huber, W., et al. (2013). Count-based differential expression analysis of RNA sequencing data using R and Bioconductor. Nat. Protoc. 8, 1765–1786. doi: 10.1038/nprot.2013.099
Bae, D., Crowley, M. R., and Wang, C. (2011). Transcriptome analysis of Listeria monocytogenes grown on a ready-to-eat meat matrix. J. Food Protoc. 74, 1104–1111. doi: 10.4315/0362-028X.JFP-10-508
Behari, J., and Youngman, P. (1998). A homolog of CcpA mediates catabolite control in Listeria monocytogenes but not carbon source regulation of virulence genes. J. Bacteriol. 180, 6316–6324.
Belitsky, B. R. (2014). Role of PdxR in the activation of vitamin B6 biosynthesis in Listeria monocytogenes. Mol. Microbiol. 92, 1113–1128. doi: 10.1111/mmi.12618
Bennett, H. J., Pearce, D. M., Glenn, S., Taylor, C. M., Kuhn, M., Sonenshein, A. L., et al. (2007). Characterization of relA and codY mutants of Listeria monocytogenes: identification of the CodY regulon and its role in virulence. Mol. Microbiol. 63, 1453–1467. doi: 10.1111/j.1365-2958.2007.05597.x
Bergholz, T. M., Bowen, B., Wiedmann, M., and Boor, K. J. (2012). Listeria monocytogenes shows temperature-dependent and -independent responses to salt stress, including responses that induce cross-protection against other stresses. Appl. Environ. Microbiol. 78, 2602–2612. doi: 10.1128/AEM.07658-11
Bierne, H., and Cossart, P. (2007). Listeria monocytogenes surface proteins: from genome predictions to function. Microbiol. Mol. Biol. Rev. 71, 377–397. doi: 10.1128/MMBR.00039-06
Borezee, E., Msadek, T., Durant, L., and Berche, P. (2000). Identification in Listeria monocytogenes of MecA, a homologue of the Bacillus subtilis competence regulatory protein. J. Bacteriol. 182, 5931–5934. doi: 10.1128/JB.182.20.5931-5934.2000
Boscher, E., Houard, E., and Denis, M. (2012). Prevalence and distribution of Listeria monocytogenes serotypes and pulsotypes in sows and fattening pigs in farrow-to-finish farms (France, 2008). J. Food Protoc. 75, 889–895. doi: 10.4315/0362-028X.JFP-11-340
Brehm, K., Ripio, M. T., Kreft, J., and Vazquez-Boland, J. A. (1999). The bvr locus of Listeria monocytogenes mediates virulence gene repression by beta-glucosides. J. Bacteriol. 181, 5024–5032.
Buchrieser, C., Rusniok, C., Kunst, F., Cossart, P., Glaser, P., and Listeria, C. (2003). Comparison of the genome sequences of Listeria monocytogenes and Listeria innocua: clues for evolution and pathogenicity. FEMS Immunol. Med. Microbiol. 35, 207–213. doi: 10.1016/S0928-8244(02)00448-0
Burrell, M., Hanfrey, C. C., Murray, E. J., Stanley-Wall, N. R., and Michael, A. J. (2010). Evolution and multiplicity of arginine decarboxylases in polyamine biosynthesis and essential role in Bacillus subtilis biofilm formation. J. Biol. Chem. 285, 39224–39238. doi: 10.1074/jbc.M110.163154
Cacace, G., Mazzeo, M. F., Sorrentino, A., Spada, V., Malorni, A., and Siciliano, R. A. (2010). Proteomics for the elucidation of cold adaptation mechanisms in Listeria monocytogenes. J. Proteomics 73, 2021–2030. doi: 10.1016/j.jprot.2010.06.011
Casey, A., Fox, E. M., Schmitz-Esser, S., Coffey, A., McAuliffe, O., and Jordan, K. (2014). Transcriptome analysis of Listeria monocytogenes exposed to biocide stress reveals a multi-system response involving cell wall synthesis, sugar uptake, and motility. Front. Microbiol. 5:68. doi: 10.3389/fmicb.2014.00068
Chan, Y. C., Hu, Y., Chaturongakul, S., Files, K. D., Bowen, B. M., Boor, K. J., et al. (2008). Contributions of two-component regulatory systems, alternative sigma factors, and negative regulators to Listeria monocytogenes cold adaptation and cold growth. J. Food Protoc. 71, 420–425. doi: 10.4315/0362-028X-71.2.420
Chatterjee, S. S., Hossain, H., Otten, S., Kuenne, C., Kuchmina, K., Machata, S., et al. (2006). Intracellular gene expression profile of Listeria monocytogenes. Infect. Immun. 74, 1323–1338. doi: 10.1128/IAI.74.2.1323-1338.2006
Chaturongakul, S., Raengpradub, S., Palmer, M. E., Bergholz, T. M., Orsi, R. H., Hu, Y. W., et al. (2011). Transcriptomic and phenotypic analyses identify coregulated, overlapping regulons among PrfA, CtsR, HrcA, and the alternative sigma factors sigma(B), sigma(C), sigma(H), and sigma(L) in Listeria monocytogenes. Appl. Environ. Microbiol. 77, 187–200. doi: 10.1128/AEM.00952-10
Chen, J., Cheng, C., Xia, Y., Zhao, H., Fang, C., Shan, Y., et al. (2011). Lmo0036, an ornithine and putrescine carbamoyltransferase in Listeria monocytogenes, participates in arginine deiminase and agmatine deiminase pathways and mediates acid tolerance. Microbiology 157, 3150–3161. doi: 10.1099/mic.0.049619-0
Christiansen, J. K., Nielsen, J. S., Ebersbach, T., Valentin-Hansen, P., Sogaard-Andersen, L., and Kallipolitis, B. H. (2006). Identification of small Hfq-binding RNAs in Listeria monocytogenes. RNA 12, 1383–1396. doi: 10.1261/rna.49706
Collins, B., Guinane, C. M., Cotter, P. D., Hill, C., and Ross, R. P. (2012). Assessing the contributions of the LiaS histidine kinase to the innate resistance of Listeria monocytogenes to nisin, cephalosporins, and disinfectants. Appl. Environ. Microbiol. 78, 2923–2929. doi: 10.1128/AEM.07402-11
Collins, B., Joyce, S., Hill, C., Cotter, P. D., and Ross, R. P. (2010). TelA contributes to the innate resistance of Listeria monocytogenes to nisin and other cell wall-acting antibiotics. Antimicrob. Agents Chemother. 54, 4658–4663. doi: 10.1128/AAC.00290-10
Cooley, M. B., Quinones, B., Oryang, D., Mandrell, R. E., and Gorski, L. (2014). Prevalence of shiga toxin producing Escherichia coli Salmonella enterica, and Listeria monocytogenes at public access watershed sites in a California Central Coast agricultural region. Front. Cell. Infect. Microbiol. 4:30. doi: 10.3389/fcimb.2014.00030
de las Heras, A., Cain, R. J., Bielecka, M. K., and Vazquez-Boland, J. A. (2011). Regulation of Listeria virulence: PrfA master and commander. Curr. Opin. Microbiol. 14, 118–127. doi: 10.1016/j.mib.2011.01.005
Desneux, J., Biscuit, A., Picard, S., and Pourcher, A.-M. (2016). Fate of viable but non-culturable Listeria monocytogenes in pig manure microcosms. Front. Microbiol. 7:245. doi: 10.3389/fmicb.2016.00245
Dowd, G. C., Casey, P. G., Begley, M., Hill, C., and Gahan, C. G. M. (2012). Investigation of the role of ZurR in the physiology and pathogenesis of Listeria monocytogenes. FEMS Microbiol. Lett. 327, 118–125. doi: 10.1111/j.1574-6968.2011.02472.x
Dowe, M. J., Jackson, E. D., Mori, J. G., and Bell, C. R. (1997). Listeria monocytogenes survival in soil and incidence in agricultural soils. J. Food Protoc. 60, 1201–1207. doi: 10.4315/0362-028X-60.10.1201
Dubnau, D. (1997). Binding and transport of transforming DNA by Bacillus subtilis: the role of type-IV pilin-like proteins - A review. Gene 192, 191–198. doi: 10.1016/S0378-1119(96)00804-9
Duche, O., Tremoulet, F., Namane, A., Labadie, J., and European Listeria Genome Consortium (2002). A proteomic analysis of the salt stress response of Listeria monocytogenes. FEMS Microbiol. Lett. 215, 183–188. doi: 10.1111/j.1574-6968.2002.tb11389.x
Esteban, J. I., Oporto, B., Aduriz, G., Juste, R. A., and Hurtado, A. (2008). A survey of food-borne pathogens in free-range poultry farms. Int. J. Food Microbiol. 123, 177–182. doi: 10.1016/j.ijfoodmicro.2007.12.012
Fichant, G., Basse, M. J., and Quentin, Y. (2006). ABCdb: an online resource for ABC transporter repertories from sequenced archaeal and bacterial genomes. FEMS Microbiol. Lett. 256, 333–339. doi: 10.1111/j.1574-6968.2006.00139.x
Flanary, P. L., Allen, R. D., Dons, L., and Kathariou, S. (1999). Insertional inactivation of the Listeria monocytogenes cheYA operon abolishes response to oxygen gradients and reduces the number of flagella. Can. J. Microbiol. 45, 646–652. doi: 10.1139/w99-052
Fox, E. M., Leonard, N., and Jordan, K. (2011). Physiological and transcriptional characterization of persistent and nonpersistent Listeria monocytogenes isolates. Appl. Environ. Microbiol. 77, 6559–6569. doi: 10.1128/AEM.05529-11
Fraser, K. R., Harvie, D., Coote, P. J., and O'Byrne, C. P. (2000). Identification and characterization of an ATP binding cassette L-carnitine transporter in Listeria monocytogenes. Appl. Environ. Microbiol. 66, 4696–4704. doi: 10.1128/AEM.66.11.4696-4704.2000
Freitag, N. E., Port, G. C., and Miner, M. D. (2009). Listeria monocytogenes-from saprophyte to intracellular pathogen. Nat. Rev. Microbiol. 7, 623–628. doi: 10.1038/nrmicro2171
Fritsch, F., Mauder, N., Williams, T., Weiser, J., Oberle, M., and Beier, D. (2011). The cell envelope stress response mediated by the LiaFSR(Lm) three-component system of Listeria monocytogenes is controlled via the phosphatase activity of the bifunctional histidine kinase LiaS(Lm). Microbiology 157, 373–386. doi: 10.1099/mic.0.044776-0
Gandhi, M., and Chikindas, M. L. (2007). Listeria: a foodborne pathogen that knows how to survive. Int. J. Food Microbiol. 113, 1–15. doi: 10.1016/j.ijfoodmicro.2006.07.008
Garmyn, D., Augagneur, Y., Gal, L., Vivant, A.-L., and Piveteau, P. (2012). Listeria monocytogenes differential transcriptome analysis reveals temperature-dependent Agr regulation and suggests overlaps with other regulons. PLoS ONE 7:e43154. doi: 10.1371/journal.pone.0043154
Giotis, E. S., Muthaiyan, A., Blair, I. S., Wilkinson, B. J., and McDowell, D. A. (2008). Genomic and proteomic analysis of the alkali-tolerance response (AlTR) in Listeria monocytogenes 10403S. BMC Microbiol. 8:102. doi: 10.1186/1471-2180-8-102
Glaser, P., Frangeul, L., Buchrieser, C., Rusniok, C., Amend, A., Baquero, F., et al. (2001). Comparative genomics of Listeria species. Science 294, 849–852. doi: 10.1126/science.1063447
Grundling, A., Burrack, L. S., Bouwer, H. G. A., and Higgins, D. E. (2004). Listeria monocytogenes regulates flagellar motility gene expression through MogR, a transcriptional repressor required for virulence. Proc. Natl. Acad. Sci. U.S.A. 101, 12318–12323. doi: 10.1073/pnas.0404924101
Hain, T., Hossain, H., Chatterjee, S. S., Machata, S., Volk, U., Wagner, S., et al. (2008). Temporal transcriptomic analysis of the Listeria monocytogenes EGD-e sigma(B) regulon. BMC Microbiol. 8:20. doi: 10.1186/1471-2180-8-20
Herbert, K. C., and Foster, S. J. (2001). Starvation survival in Listeria monocytogenes: characterization of the response and the role of known and novel components. Microbiology 147, 2275–2284. doi: 10.1099/00221287-147-8-2275
Hölzel, C. S., Müller, C., Harms, K. S., Mikolajewski, S., Schäfer, S., Schwaiger, K., et al. (2012). Heavy metals in liquid pig manure in light of bacterial antimicrobial resistance. Environ. Res. 113, 21–27. doi: 10.1016/j.envres.2012.01.002
Hu, Y., Oliver, H. F., Raengpradub, S., Palmer, M. E., Orsi, R. H., Wiedmann, M., et al. (2007a). Transcriptomic and phenotypic analyses suggest a network between the transcriptional regulators HrcA and sigma(B) in Listeria monocytogenes. Appl. Environ. Microbiol. 73, 7981–7991. doi: 10.1128/AEM.01281-07
Hu, Y., Raengpradub, S., Schwab, U., Loss, C., Orsi, R. H., Wiedmann, M., et al. (2007b). Phenotypic and transcriptomic analyses demonstrate interactions between the transcriptional regulators CtsR and sigma B in Listeria monocytogenes. Appl. Environ. Microbiol. 73, 7967–7980. doi: 10.1128/AEM.01085-07
Huang, Y., Suo, Y., Shi, C., Szlavik, J., Shi, X.-M., and Knochel, S. (2013). Mutations in gltB and gltC reduce oxidative stress tolerance and biofilm formation in Listeria monocytogenes 4b G. Int. J. Food Microbiol. 163, 223–230. doi: 10.1016/j.ijfoodmicro.2013.02.023
Hutchison, M. L., Walters, L. D., Moore, A., Crookes, K. M., and Avery, S. M. (2004). Effect of length of time before incorporation on survival of pathogenic bacteria present in livestock wastes applied to agricultural soil. Appl. Environ. Microbiol. 70, 5111–5118. doi: 10.1128/AEM.70.9.5111-5118.2004
Hutchison, M. L., Walters, L. D., Moore, T., Thomas, D. J. I., and Avery, S. M. (2005). Fate of pathogens present in livestock wastes spread onto fescue plots. Appl. Environ. Microbiol. 71, 691–696. doi: 10.1128/AEM.71.2.691-696.2005
Jiang, X. P., Islam, M., Morgan, J., and Doyle, M. P. (2004). Fate of Listeria monocytogenes in bovine manure-amended soil. J. Food Protoc. 67, 1676–1681. doi: 10.4315/0362-028X-67.8.1676
Johansson, J., Mandin, P., Renzoni, A., Chiaruttini, C., Springer, M., and Cossart, P. (2002). An RNA thermosensor controls expression of virulence genes in Listeria monocytogenes. Cell 110, 551–561. doi: 10.1016/S0092-8674(02)00905-4
Kallipolitis, B. H., Ingmer, H., Gahan, C. G., Hill, C., and Sogaard-Andersen, L. (2003). CesRK, a two-component signal transduction system in Listeria monocytogenes, responds to the presence of cell wall-acting antibiotics and affects beta-lactam resistance. Antimicrob. Agents Chemother. 47, 3421–3429. doi: 10.1128/AAC.47.11.3421-3429.2003
Knudsen, G. M., Olsen, J. E., and Dons, L. (2004). Characterization of DegU, a response regulator in Listeria monocytogenes, involved in regulation of motility and contributes to virulence. FEMS Microbiol. Lett. 240, 171–179. doi: 10.1016/j.femsle.2004.09.039
Krawczyk-Balska, A., Popowska, M., and Markiewicz, Z. (2012). Re-evaluation of the significance of penicillin binding protein 3 in the susceptibility of Listeria monocytogenes to beta-lactam antibiotics. BMC Microbiol. 12:57. doi: 10.1186/1471-2180-12-57
Larsen, M. H., Leisner, J. J., and Ingmer, H. (2010). The chitinolytic activity of Listeria monocytogenes EGD is regulated by carbohydrates but also by the virulence regulator PrfA. Appl. Environ. Microbiol. 76, 6470–6476. doi: 10.1128/AEM.00297-10
Lazarevic, V., and Karamata, D. (1995). The tagGH operon of Bacillus-subtilis-168 encodes a 2-component ABC transporter involved in the metabolism of 2 wall teichoic-acids. Mol. Microbiol. 16, 345–355. doi: 10.1111/j.1365-2958.1995.tb02306.x
Ledala, N., Sengupta, M., Muthaiyan, A., Wilkinson, B. J., and Jayaswal, R. K. (2010). Transcriptomic response of Listeria monocytogenes to iron limitation and fur mutation. Appl. Environ. Microbiol. 76, 406–416. doi: 10.1128/AEM.01389-09
Liu, S. Q., Graham, J. E., Bigelow, L., Morse, P. D., and Wilkinson, B. J. (2002). Identification of Listeria monocytogenes genes expressed in response to growth at low temperature. Appl. Environ. Microbiol. 68, 1697–1705. doi: 10.1128/AEM.68.4.1697-1705.2002
Liu, Y., and Ream, A. (2008). Gene expression profiling of Listeria monocytogenes strain F2365 during growth in ultrahigh-temperature-processed skim milk. Appl. Environ. Microbiol. 74, 6859–6866. doi: 10.1128/AEM.00356-08
Locatelli, A., Depret, G., Jolivet, C., Henry, S., Dequiedt, S., Piveteau, P., et al. (2013a). Nation-wide study of the occurrence of Listeria monocytogenes in French soils using culture-based and molecular detection methods. J. Microbiol. Methods 93, 242–250. doi: 10.1016/j.mimet.2013.03.017
Locatelli, A., Spor, A., Jolivet, C., Piveteau, P., and Hartmann, A. (2013b). Biotic and abiotic soil properties influence survival of Listeria monocytogenes in soil. PLoS ONE 8:e75969. doi: 10.1371/journal.pone.0075969
Loh, E., Dussurget, O., Gripenland, J., Vaitkevicius, K., Tiensuu, T., Mandin, P., et al. (2009). A trans-acting riboswitch controls expression of the virulence regulator PrfA in Listeria monocytogenes. Cell 139, 770–779. doi: 10.1016/j.cell.2009.08.046
Lyautey, E., Lapen, D. R., Wilkes, G., McCleary, K., Pagotto, F., Tyler, K., et al. (2007). Distribution and characteristics of Listeria monocytogenes isolates from surface waters of the South Nation River watershed, Ontario, Canada. Appl. Environ. Microbiol. 73, 5401–5410. doi: 10.1128/AEM.00354-07
Mandin, P., Repoila, F., Vergassola, M., Geissmann, T., and Cossart, P. (2007). Identification of new noncoding RNAs in Listeria monocytogenes and prediction of mRNA targets. Nucleic Acids Res. 35, 962–974. doi: 10.1093/nar/gkl1096
Mattila, M., Lindstrom, M., Somervuo, P., Markkula, A., and Korkeala, H. (2011). Role of flhA and motA in growth of Listeria monocytogenes at low temperatures. Int. J. Food Microbiol. 148, 177–183. doi: 10.1016/j.ijfoodmicro.2011.05.022
McLaughlin, H. P., Casey, P. G., Cotter, J., Gahan, C. G. M., and Hill, C. (2011). Factors affecting survival of Listeria monocytogenes and Listeria innocua in soil samples. Arch. Microbiol. 193, 775–785. doi: 10.1007/s00203-011-0716-7
Mellin, J. R., and Cossart, P. (2012). The non-coding RNA world of the bacterial pathogen Listeria monocytogenes. RNA Biol. 9, 372–378. doi: 10.4161/rna.19235
Mellin, J. R., Tiensuu, T., Becavin, C., Gouin, E., Johansson, J., and Cossart, P. (2013). A riboswitch-regulated antisense RNA in Listeria monocytogenes. Proc. Natl. Acad. Sci. U.S.A. 110, 13132–13137. doi: 10.1073/pnas.1304795110
Michel, E., Mengaud, J., Galsworthy, S., and Cossart, P. (1998). Characterization of a large motility gene cluster containing the cheR, motAB genes of Listeria monocytogenes and evidence that PrfA downregulates motility genes. FEMS Microbiol. Lett. 169, 341–347. doi: 10.1111/j.1574-6968.1998.tb13338.x
Milohanic, E., Glaser, P., Coppee, J. Y., Frangeul, L., Vega, Y., Vazquez-Boland, J. A., et al. (2003). Transcriptome analysis of Listeria monocytogenes identifies three groups of genes differently regulated by PrfA. Mol. Microbiol. 47, 1613–1625. doi: 10.1046/j.1365-2958.2003.03413.x
Moshtaghi, H., Garg, S., and Mandokhot, U. V. (2009). Survivability of Listeria monocytogenes in agricultural field soil. Indian J. Vet. Res. 18, 1–7. Available online at: http://www.iaavr.org
Moynihan, E. L., Richards, K. G., Brennan, F. P., Tyrrel, S. F., and Ritz, K. (2015). Enteropathogen survival in soil from different land-uses is predominantly regulated by microbial community composition. Appl. Soil Ecol. 89, 76–84. doi: 10.1016/j.apsoil.2015.01.011
Mraheil, M. A., Billion, A., Mohamed, W., Mukherjee, K., Kuenne, C., Pischimarov, J., et al. (2011). The intracellular sRNA transcriptome of Listeria monocytogenes during growth in macrophages. Nucleic Acids Res. 39, 4235–4248. doi: 10.1093/nar/gkr033
Nicholson, F. A., Groves, S. J., and Chambers, B. J. (2005). Pathogen survival during livestock manure storage and following land application. Bioresour. Technol. 96, 135–143. doi: 10.1016/j.biortech.2004.02.030
Nielsen, J. S., Larsen, M. H., Lillebaek, E. M. S., Bergholz, T. M., Christiansen, M. H. G., Boor, K. J., et al. (2011). A small RNA controls expression of the chitinase ChiA in Listeria monocytogenes. PLoS ONE 6:e19019. doi: 10.1371/journal.pone.0019019
Nielsen, J. S., Olsen, A. S., Bonde, M., Valentin-Hansen, P., and Kallipolitis, B. H. (2008). Identification of a sigma(B)-dependent small noncoding RNA in Listeria monocytogenes. J. Bacteriol. 190, 6264–6270. doi: 10.1128/JB.00740-08
Nightingale, K. K., Schukken, Y. H., Nightingale, C. R., Fortes, E. D., Ho, A. J., Her, Z., et al. (2004). Ecology and transmission of Listeria monocytogenes infecting ruminants and in the farm environment. Appl. Environ. Microbiol. 70, 4458–4467. doi: 10.1128/AEM.70.8.4458-4467.2004
Ouyang, Y., Li, J., Dong, Y., Blakely, L. V., and Cao, M. (2012). Genome-wide screening of genes required for Listeria monocytogenes biofilm formation. J. Biotechnol. Res. 4, 13–25.
Palumbo, J. D., Kaneko, A., Nguyen, K. D., and Gorski, L. (2005). Identification of genes induced in Listeria monocytogenes during growth and attachment to cut cabbage, using differential display. Appl. Environ. Microbiol. 71, 5236–5243. doi: 10.1128/AEM.71.9.5236-5243.2005
Peng, Y. L., Meng, Q. L., Qiao, J., Xie, K., Chen, C., Liu, T. L., et al. (2016). The regulatory roles of ncRNA Rli60 in adaptability of Listeria monocytogenes to environmental stress and biofilm formation. Curr. Microbiol. 73, 77–83. doi: 10.1007/s00284-016-1028-6
Phan-Thanh, L., and Mahouin, F. (1999). A proteomic approach to study the acid response in Listeria monocytogenes. Electrophoresis 20, 2214–2224. doi: 10.1002/(SICI)1522-2683(19990801)20:11<2214::AID-ELPS2214>3.0.CO;2-G
Piveteau, P., Depret, G., Pivato, B., Garmyn, D., and Hartmann, A. (2011). Changes in gene expression during adaptation of Listeria monocytogenes to the soil environment. PLoS ONE 6:e24881. doi: 10.1371/journal.pone.0024881
Popowska, M., and Markiewicz, Z. (2006). Characterization of Listeria monocytogenes protein Lmo0327 with murein hydrolase activity. Arch. Microbiol. 186, 69–86. doi: 10.1007/s00203-006-0122-8
Pourcher, A. M., Ziebal, C., Boscher, E., Picard, S., Perrot, M., Desneux, J., et al. (2015). “Survey of Listeria monocytogenes in raw and treated manures from two pig farms,” in XVII International Congress on Animal Hygiene (Košice).
Qian, M., Wu, H., Wang, J., Zhang, H., Zhang, Z., Zhang, Y., et al. (2016). Occurrence of trace elements and antibiotics in manure- based fertilizers from the Zhejiang Province of China. Sci. Tot. Environ. 559, 174–181. doi: 10.1016/j.scitotenv.2016.03.123
Rabinovich, L., Sigal, N., Borovok, I., Nir-Paz, R., and Herskovits, A. A. (2012). Prophage excision activates listeria competence genes that promote phagosomal escape and virulence. Cell. 150, 792–802. doi: 10.1016/j.cell.2012.06.036
Raengpradub, S., Wiedmann, M., and Boor, K. J. (2008). Comparative analysis of the sigma(B)-dependent stress responses in Listeria monocytogenes and Listeria innocua strains exposed to selected stress conditions. Appl. Environ. Microbiol. 74, 158–171. doi: 10.1128/AEM.00951-07
Rea, R. B., Gahan, C. G. M., and Hill, C. (2004). Disruption of putative regulatory loci in Listeria monocytogenes demonstrates a significant role for fur and PerR in virulence. Infect. Immun. 72, 717–727. doi: 10.1128/IAI.72.2.717-727.2004
Rea, R., Hill, C., and Gahan, C. G. M. (2005). Listeria monocytogenes PerR mutants display a small-colony phenotype, increased sensitivity to hydrogen peroxide, and significantly reduced murine virulence. Appl. Environ. Microbiol. 71, 8314–8322. doi: 10.1128/AEM.71.12.8314-8322.2005
Riedel, C. U., Monk, I. R., Casey, P. G., Waidmann, M. S., Gahan, C. G. M., and Hill, C. (2009). AgrD-dependent quorum sensing affects biofilm formation, invasion, virulence and global gene expression profiles in Listeria monocytogenes. Mol. Microbiol. 71, 1177–1189. doi: 10.1111/j.1365-2958.2008.06589.x
Rieu, A., Weidmann, S., Garmyn, D., Piveteau, P., and Guzzo, J. (2007). agr system of Listeria monocytogenes EGD-e: role in adherence and differential expression pattern. Appl. Environ. Microbiol. 73, 6125–6133. doi: 10.1128/AEM.00608-07
Rismondo, J., Moeller, L., Aldridge, C., Gray, J., Vollmer, W., and Halbedel, S. (2015). Discrete and overlapping functions of peptidoglycan synthases in growth, cell division and virulence of Listeria monocytogenes. Mol. Microbiol. 95, 332–351. doi: 10.1111/mmi.12873
Sakamoto, A., Terui, Y., Yamamoto, T., Kasahara, T., Nakamura, M., Tomitori, H., et al. (2012). Enhanced biofilm formation and/or cell viability by polyamines through stimulation of response regulators UvrY and CpxR in the two-component signal transducing systems, and ribosome recycling factor. Int. J. Biochem. Cell Biol. 44, 1877–1886. doi: 10.1016/j.biocel.2012.07.010
Salazar, J. K., Wu, Z., McMullen, P. D., Luo, Q., Freitag, N. E., Tortorello, M. L., et al. (2013a). prfA-like transcription factor gene lmo0753 contributes to L-rhamnose utilization in Listeria monocytogenes strains associated with human food-borne infections. Appl. Environ. Microbiol. 79, 5584–5592. doi: 10.1128/AEM.01812-13
Salazar, J. K., Wu, Z., Yang, W., Freitag, N. E., Tortorello, M. L., Wang, H., et al. (2013b). Roles of a Novel Crp/Fnr family transcription factor Lmo0753 in soil survival, biofilm production and surface attachment to fresh produce of Listeria monocytogenes. PLoS ONE 8:e75736. doi: 10.1371/journal.pone.0075736
Scortti, M., Monzo, H. J., Lacharme-Lora, L., Lewis, D. A., and Vazquez-Boland, J. A. (2007). The PrfA virulence regulon. Microb. Infect. 9, 1196–1207. doi: 10.1016/j.micinf.2007.05.007
Supa-amornkul, S., Chantratita, W., Srichunrusami, C., Janchompoo, P., and Chaturongakul, S. (2016). Listeria monocytogenes MerR-like regulator NmlR(lm): its transcriptome and role in stress response. Foodborne Pathog. Dis. 13, 369–378. doi: 10.1089/fpd.2015.2101
Toledo-Arana, A., Dussurget, O., Nikitas, G., Sesto, N., Guet-Revillet, H., Balestrino, D., et al. (2009). The Listeria transcriptional landscape from saprophytism to virulence. Nature 459, 950–956. doi: 10.1038/nature08080
Van de Velde, S., Carryn, S., Van Bambeke, F., Hill, C., Tulkens, P. M., and Sleator, R. D. (2009). Penicillin-binding Proteins (PBP) and Lmo0441 (a PBP-like protein) play a role in Beta-lactam sensitivity of Listeria monocytogenes. Gut Pathogens 1:23. doi: 10.1186/1757-4749-1-23
van der Veen, S., and Abee, T. (2010). HrcA and DnaK are important for static and continuous-flow biofilm formation and disinfectant resistance in Listeria monocytogenes. Microbiology 156, 3782–3790. doi: 10.1099/mic.0.043000-0
van der Veen, S., Hain, T., Wouters, J. A., Hossain, H., de Vos, W. M., Abee, T., et al. (2007). The heat-shock response of Listeria monocytogenes comprises genes involved in heat shock, cell division, cell wall synthesis, and the SOS response. Microbiology 153, 3593–3607. doi: 10.1099/mic.0.2007/006361-0
Vilar, M. J., Yus, E., Sanjuan, M. L., Dieguez, F. J., and Rodriguez-Otero, J. L. (2007). Prevalence of and risk factors for listeria species on dairy farms. J. Dairy Sci. 90, 5083–5088. doi: 10.3168/jds.2007-0213
Vivant, A.-L., Garmyn, D., Gal, L., and Piveteau, P. (2014). The Agr communication system provides a benefit to the populations of Listeria monocytogenes in soil. Front. Cell. Infect. Microbiol. 4:160. doi: 10.3389/fcimb.2014.00160
Widyasari-Mehta, A., Hartung, S., and Kreuzig, R. (2016). From the application of antibiotics to antibiotic residues in liquid manures and digestates: a screening study in one European center of conventional pig husbandry. J. Environ. Manage. 177, 129–137. doi: 10.1016/j.jenvman.2016.04.012
Wilkes, G., Edge, T., Gannon, V., Jokinen, C., Lyautey, E., Medeiros, D., et al. (2009). Seasonal relationships among indicator bacteria, pathogenic bacteria, Cryptosporidium oocysts, Giardia cysts, and hydrological indices for surface waters within an agricultural landscape. Water Res. 43, 2209–2223. doi: 10.1016/j.watres.2009.01.033
Williams, T., Bauer, S., Beier, D., and Kuhn, M. (2005). Construction and characterization of Listeria monocytogenes mutants with in-frame deletions in the response regulator genes identified in the genome sequence. Infect. Immun. 73, 3152–3159. doi: 10.1128/IAI.73.5.3152-3159.2005
Wortham, B. W., Oliveira, M. A., Fetherston, J. D., and Perry, R. D. (2010). Polyamines are required for the expression of key Hms proteins important for Yersinia pestis biofilm formation. Environ. Microbiol. 12, 2034–2047. doi: 10.1111/j.1462-2920.2010.02219.x
Wurtzel, O., Sesto, N., Mellin, J. R., Karunker, I., Edelheit, S., Becavin, C., et al. (2012). Comparative transcriptomics of pathogenic and non-pathogenic Listeria species. Mol. Syst. Biol. 8:583. doi: 10.1038/msb.2012.11
Keywords: Listeria monocytogenes, transcriptome, pig manure, RNAseq, lagoon effluent, soil, microcosms
Citation: Vivant A-L, Desneux J, Pourcher A-M and Piveteau P (2017) Transcriptomic Analysis of the Adaptation of Listeria monocytogenes to Lagoon and Soil Matrices Associated with a Piggery Environment: Comparison of Expression Profiles. Front. Microbiol. 8:1811. doi: 10.3389/fmicb.2017.01811
Received: 28 April 2017; Accepted: 05 September 2017;
Published: 26 September 2017.
Edited by:
Daniela De Biase, Sapienza Università di Roma, ItalyReviewed by:
Edward M. Fox, CSIRO, AustraliaCatherine Maeve Burgess, Teagasc, The Irish Agriculture and Food Development Authority, Ireland
Copyright © 2017 Vivant, Desneux, Pourcher and Piveteau. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Pascal Piveteau, piveteau@u-bourgogne.fr
 Anne-Laure Vivant1,2
Anne-Laure Vivant1,2 Anne-Marie Pourcher
Anne-Marie Pourcher Pascal Piveteau
Pascal Piveteau