- 1Faculty of Sports Science, Mersin University, Mersin, Türkiye
- 2Faculty of Sport Sciences, Aydin Adnan Menderes Universitesi, Aydın, Türkiye
- 3Akdeniz Universitesi, Antalya, Türkiye
- 4Faculty of Sport Sciences Department of Coaching Education, Mersin University, Mersin, Türkiye
- 5Faculty of Sport Sciences, Mus Alparslan University, Muş, Türkiye
- 6Hasan Dogan Faculty of Sport Sciences, Karabuk University, Karabük, Türkiye
This review investigates the transformative potential of wearable sensors for body fluid monitoring in sports and healthcare. These devices offer a non-invasive, real-time, in-situ glimpse into our health by continuously tracking vital biomarkers found in sweat, saliva, urine, and tears. We discuss various sensor technologies, including electrochemical, optical, and microfluidic, and the innovative materials like hydrogels and nanocomposites that enable their functionality. The integration of RFID and flexible electronics is also examined, highlighting how these advancements improve the connectivity, portability, and user-friendliness of the sensors. Moreover, we delve into the advanced manufacturing techniques, such as 3D printing, that are crucial for crafting these sophisticated devices with high precision and adaptability. In prospect, this article evaluates the transformative potential of integrating these in-situ sensors with artificial intelligence and machine learning, envisioning a paradigm shift in health monitoring and athletic performance optimization.
1 Introduction
The integration of wearable sensor technologies into sports and health monitoring marks a significant evolution in personalized healthcare and athletic performance. These devices transcend the limitations of traditional diagnostic methodologies, which are inherently constrained by their reliance on invasive, episodic, and laboratory-based analyses. By leveraging advancements in flexible materials, sophisticated electronics, and wireless communication protocols, wearable sensors facilitate the continuous and unobtrusive monitoring of physiological states. This capability not only democratizes access to health data but also empowers individuals with real-time insights, fostering proactive health management. Such advancements are particularly salient in sports science, where nuanced physiological data underpin performance optimization and recovery strategies, and in healthcare, where early intervention and the longitudinal management of chronic conditions are paramount.
The market dynamics surrounding wearable health sensors and sports devices reflect a robust growth trajectory. Projections indicate a 10.5% Compound Annual Growth Rate (CAGR) for the Wearable Health Sensors Market, driven primarily by remote patient monitoring applications that capitalize on continuous health data acquisition and analysis. Similarly, the wearable sports device market is poised for expansion, from an estimated USD 94.17 billion in 2025 to USD 115.57 billion by 2030, representing a 4.18% CAGR. This surge is propelled by the sports industry’s increasing reliance on real-time athletic performance and health metrics (Mordor Intelligence, 2025a; Mordor Intelligence, 2025b). The confluence of technological innovation, enhanced device comfort and portability, and heightened health and fitness awareness underpins this growth. These trends not only accelerate the adoption of wearable technologies but also facilitate their seamless integration into daily life, thereby redefining preventive healthcare and sports training paradigms. The increasing embedment of these devices within consumer and medical ecosystems heralds a shift towards data-driven personal and professional health decision-making.
Within the realm of sports and health monitoring, a diverse array of sensors—including Electrocardiography (ECG), Electromyography (EMG), Functional Near-Infrared Spectroscopy (fNIRS), and Inertial Measurement Units (IMU)—contributes to the optimization of athletic performance and the safeguarding of wellbeing (Seçkin et al., 2023). Notably, sensors designed for direct interaction with body fluids occupy a pivotal role. For instance, Pulse Oximeter sensors measure blood oxygen saturation (Seçkin et al., 2023), providing critical insights into respiratory efficiency and cardiovascular function. Blood Pressure Sensors (BPS) enable non-invasive monitoring of blood pressure, offering essential data on cardiovascular health, particularly during strenuous physical activity (Iqbal et al., 2024). Galvanic Skin Response (GSR) sensors (Mao et al., 2023) detect variations in skin conductivity due to perspiration, revealing stress levels and physiological responses to training.
Wearable body fluid sensors, specifically, distinguish themselves through their capacity to provide real-time, direct biomarkers of an athlete’s physiological state, enabling tailored training regimens and health monitoring. Building upon this foundation, wearable sensor technology has revolutionized health monitoring and athletic performance assessment through non-invasive, real-time tracking of critical physiological parameters. The capacity to analyze extracellular body fluids—including blood, sweat, saliva, urine, and tears—has yielded unprecedented insights into biomarkers such as pH, glucose, electrolytes, osmolality, and cortisol. These advancements have redefined approaches to metabolic health management, hydration strategies, stress response monitoring, and athletic optimization, paving the way for personalized healthcare and performance-driven strategies (Kristoffersson and Lindén, 2022; Kelly et al., 2024; Magdalena, 2024; Obaidur Rahman Khan et al., 2024; Jafleh et al., 2024; Vishnupriya, 2024; Wenfei, 2024).
Each body fluid presents unique characteristics and analytical challenges (Ates et al., 2021), dictating its suitability for wearable sensor applications. While blood offers a comprehensive biomarker profile and remains the gold standard for many clinical diagnostics, its invasive sampling requirements limit its practicality for continuous monitoring (Islam and Washington, 2024). Non-invasive alternatives—sweat, saliva, urine, and tears—offer significant advantages in accessibility and ease of use (Kim et al., 2019). Sweat provides biomarkers such as sodium, lactate, and cortisol, offering crucial insights into hydration and metabolic states (Min et al., 2023; Childs et al., 2024; Baker, 2017). Saliva serves as a convenient matrix for monitoring stress, immune function, and glucose levels (Chojnowska et al., 2021; Pfaffe et al., 2011; Kumari et al., 2024). Urine provides a cumulative reflection of hydration, renal health, and electrolyte balance (Xue et al., 2023; Wardenaar, 2022). Tears, with biomarkers such as osmolality and cortisol, offer a unique avenue for assessing ocular and systemic health (Potvin et al., 2015; Alkozi et al., 2024).
This transition signifies a transformative era in personalized healthcare and sports science, wherein in-situ wearable sensors facilitate precise real-time health monitoring, enhanced athletic performance insights, and early intervention capabilities. By enabling continuous data collection directly from the body, these sensors obviate the need for laboratory-based testing, offering unparalleled convenience and accuracy. The ensuing discussion will delve into the specific properties and applications of body fluids in wearable sensor technologies, addressing the challenges and innovations that are shaping the future of this dynamic field. Although many reviews have addressed individual biofluid sensors, few have systematically compared cross-fluid integration (blood–sweat–tear) within AI/IoT frameworks for athletic optimization and personalized healthcare.
2 Body fluids key sensor parameters
Wearable sensors for body fluid monitoring represent the intersection of technological innovation, scientific discovery, and practical utility. Their ability to provide continuous, non-invasive access to critical health metrics empowers individuals and professionals to make informed decisions, transforming the landscape of healthcare and performance optimization. The integration of these sensors into body fluid analysis has enabled precise and dynamic monitoring of key physiological parameters vital to health and performance. Among these, pH (Heikenfeld et al., 2018; Wolf et al., 2011), conductivity (Shirreffs and Maughan, 1998), dielectric constant (Wolf et al., 2011), and biomarker-specific metrics (Gao et al., 2021; Koh et al., 2016; Jaiswal et al., 2024) stand out as particularly informative. These parameters not only reflect the body’s immediate physiological state but also provide insights into long-term health trends. Advances in sensor technologies have enabled the development of highly sensitive and non-invasive devices capable of detecting subtle changes in these metrics, offering actionable insights for both healthcare practitioners and athletes. The ability to monitor these parameters in real time allows users to make timely adjustments to hydration, nutrition, and recovery strategies, minimizing risks and maximizing performance. Exploring the significance and applications of these parameters underscores how wearable sensors are reshaping the understanding and utilization of body fluid analytics in healthcare and sports science.
Among the various parameters, pH stands out as a critical measure of the acid-base balance in body fluids, essential for maintaining physiological stability and overall health. For athletes, pH monitoring in sweat, blood, urine, or tears reveals valuable data about metabolic processes, hydration levels, and recovery efficiency. Wearable sensors for pH measurement, employing technologies like electrochemical systems or optical methods, provide real-time tracking of acid-base shifts. Such insights allow timely interventions to mitigate fatigue, dehydration, or metabolic stress, enhancing performance and recovery. Non-invasive pH sensors, designed to be flexible and biocompatible, monitor various physiological parameters by detecting ionic changes in sweat (Hossain et al., 2023), which indicate the body’s metabolic state. When integrated with IoT platforms such as Blynk App (Sathya and Rajalakshmi, 2023) and AI analytics, these sensors promise to enhance personalized training and hydration strategies while addressing potential health risks related to pH imbalances.
Similarly, monitoring the conductivity of body fluids, linked to ion concentration, offers significant insights into hydration and electrolyte balance. Conductivity sensors integrated into wearable devices detect ionic changes, providing real-time data crucial for managing hydration and preventing imbalances. These compact, non-invasive sensors have gained traction in sports and health applications, and their future integration with IoT and AI is poised to enhance personalized electrolyte management, ultimately improving athletic performance and health outcomes.
The dielectric constant, indicative of a fluid’s ability to store electrical energy, further enhances the understanding of body fluid composition. By monitoring the dielectric constant of sweat, blood, saliva, or tears, wearable sensors offer critical insights into hydration, electrolyte balance, and overall physiological health. Advances in sensor technology, such as RF and capacitive systems, enable highly sensitive, real-time, and non-invasive monitoring. These capabilities provide athletes with actionable data to optimize performance and recovery while reducing risks associated with dehydration and electrolyte imbalances.
Electrolyte monitoring is another essential application of wearable sensors. Electrolytes such as sodium, potassium, calcium, and chloride are critical for hydration, nerve function, and muscle performance. Wearable sensors, employing advanced electrochemical and ion-selective technologies, allow real-time tracking of electrolyte levels, offering athletes precise data to adjust hydration and nutrition strategies. The integration of these sensors with IoT and AI systems ensures a personalized approach to optimizing health and performance, reducing risks associated with electrolyte imbalances.
Osmolality, a measure of solute concentration, is crucial for assessing hydration and metabolic health (Shirreffs and Maughan, 1998). Innovative sensor technologies, including microfluidic, conductivity-based, and optical systems, provide precise osmolality measurements, often integrated into wearable devices for real-time tracking. These advancements facilitate early detection of dehydration or overhydration and help maintain fluid and electrolyte balance, ensuring optimal performance. As sensor technologies evolve, non-invasive approaches using sweat, saliva, and tears will continue to improve, supported by IoT integration and AI-driven analysis for comprehensive health management.
Recent advancements in materials science and manufacturing have played a pivotal role in enabling the integration of wearable sensors with body fluids (Fakhr et al., 2024; Promphet et al., 2021; Yuan et al., 2022; Wu et al., 2022; Shahzad et al., 2024). The use of flexible substrates like polydimethylsiloxane (PDMS) and advanced conductive materials such as graphene and silver nanowires has led to the development of lightweight, biocompatible sensors. Hydrogels, enhancing biofluid interaction, ensure reliable biomarker detection even under dynamic conditions (Zazoum et al., 2022). Furthermore, wireless communication technologies, including RFID and Bluetooth, have expanded the scalability and usability of these devices, allowing seamless data collection and analysis. Table 1 provides examples of body fluids, including tears, and their parameters, highlighting their impact on an athlete’s body and the potential sensor types that can be used for detecting health issues, as documented in the literature. Figure 1 also shows some non-invasive chemical sensors used on the human body (Promphet et al., 2021).
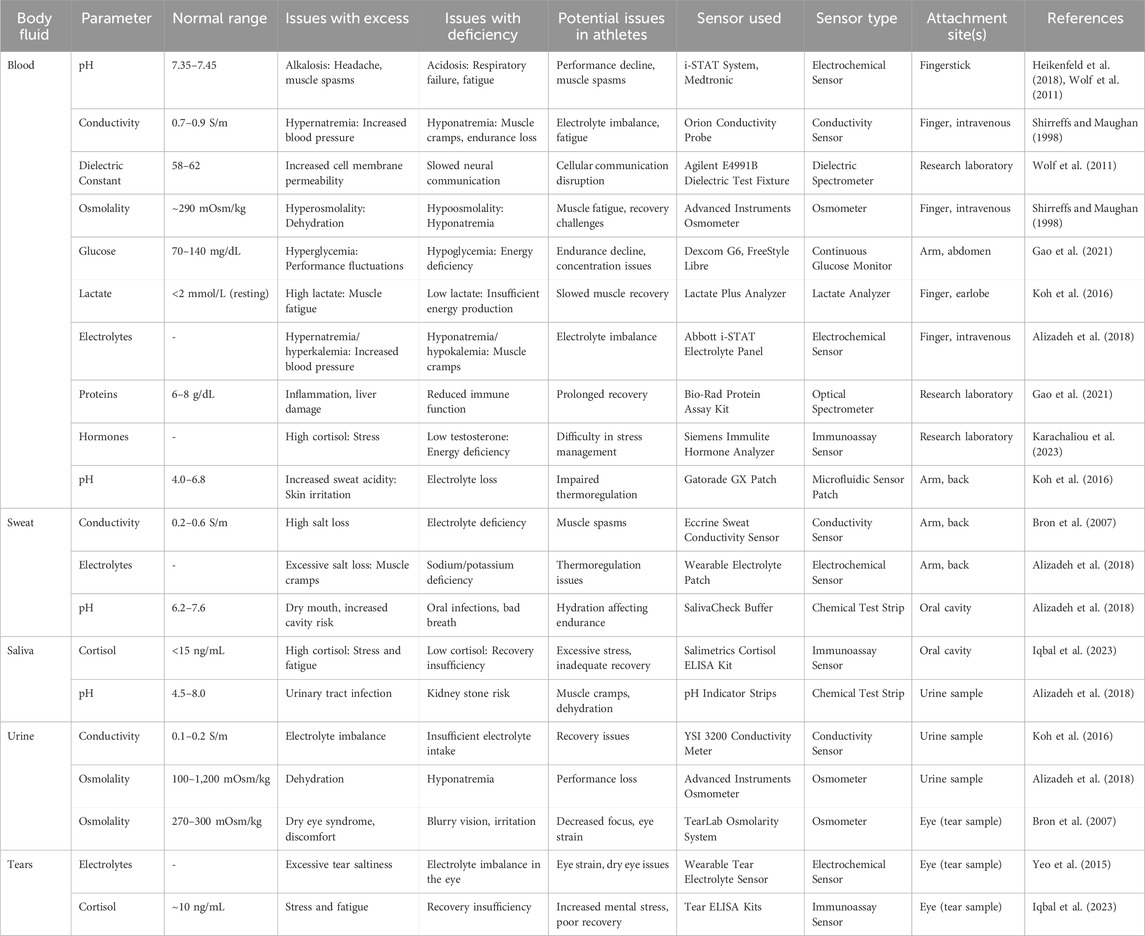
Table 1. Body Fluids, Measurement Parameters, In-situ Sensors, and Their Impact on Athletes’ Health: Wearable/nonwearable samples.
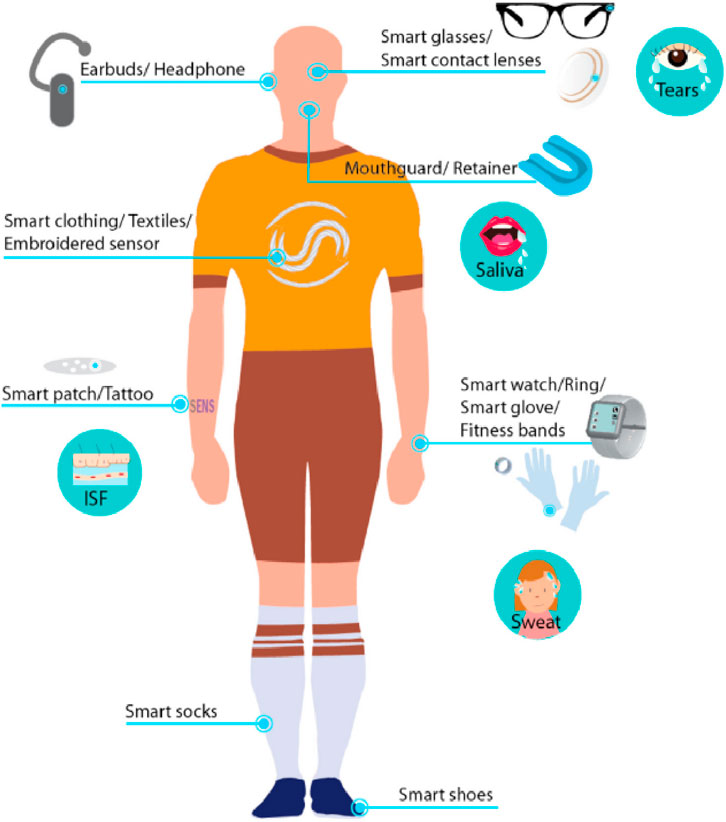
Figure 1. Depiction of various non-invasive chemical sensors applied to the human body, illustrating the diverse applications and sensor technologies used for monitoring different physiological parameters. (ISF: Interstitial fluid) (Promphet et al., 2021).
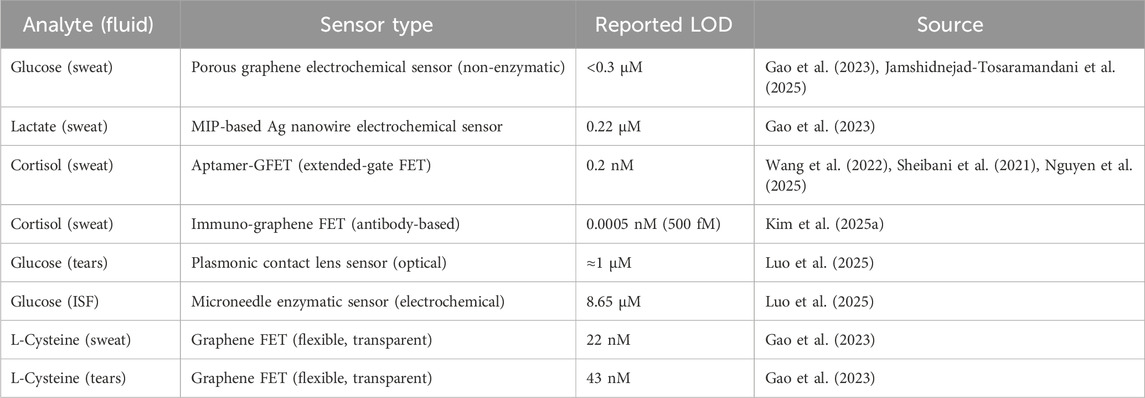
Wearable biosensors designed for body fluid analysis must not only identify key physiological parameters but also exhibit high analytical performance. Sensitivity, selectivity, and the limit of detection (LOD) are critical indicators of precision, stability, and clinical applicability in such systems. Among these, LOD plays a decisive role in determining whether a sensor can reliably detect the typically low concentrations of biomarkers found in sweat, tears, or interstitial fluid. A lower LOD enables the detection of subtle physiological changes and early deviations from normal, which is essential for timely health monitoring and intervention. Accordingly, recent research has focused on improving LOD through advanced materials and optimized transduction mechanisms. Nanostructured electrodes, high-surface-area nanomaterials, and signal amplification strategies have been widely adopted to enhance sensitivity, achieving nanomolar or even sub-nanomolar detection limits for various biomarkers. Table 2 summarizes representative wearable sensor technologies and their reported LODs, highlighting how different sensing platforms—electrochemical, optical, and field-effect transistor (FET)-based—compare in terms of sensitivity.
3 Sensors used for blood
3.1 Characteristics and parameters
Wearable sensors have transformed the monitoring of blood-related parameters in sports and healthcare through non-invasive technologies. Photoplethysmography (PPG) sensors are widely used to measure heart rate, heart rate variability (HRV), blood oxygen saturation (SpO2), and blood volume changes by analyzing light absorption in blood vessels (Seçkin et al., 2023; Yun et al., 2021). Optical sensors enhance these capabilities, enabling the measurement of blood oxygen levels and showing potential for non-invasive glucose monitoring (Quan et al., 2021). Electrical bioimpedance sensors track blood flow, volume, and hydration levels by assessing the body’s electrical resistance, while near-infrared spectroscopy (NIRS) sensors provide real-time insights into muscle oxygenation and tissue health (Ismail et al., 2023). Skin-interfaced microfluidic devices analyze sweat to estimate blood-related metabolites such as lactate and glucose, particularly valuable during physical activity (Iqbal et al., 2024). Among these, electrochemical sensors stand out for their versatility and precision. By measuring lactate, glucose, pH, and electrolytes in sweat, they offer a powerful tool for real-time metabolic and physiological monitoring, making them an essential component of wearable sensor technology for personalized health and performance optimization (Mao et al., 2023).
Blood serves as a primary diagnostic medium due to its complex biochemical composition. It maintains homeostasis by delivering oxygen and nutrients to tissues and removing metabolic waste. Key parameters include pH (7.35–7.45), osmolality (∼290 mOsm/kg), glucose levels (70–140 mg/dL), lactate concentration, and electrolyte balance. Deviations in these values can indicate metabolic disorders, dehydration, or physiological stress (Heikenfeld et al., 2018). Glucose monitoring is particularly critical for diabetic athletes, as it affects energy availability and performance. Similarly, blood lactate serves as an indicator of anaerobic threshold and recovery status (Jonathan et al., 2023).
3.2 Sensor technologies
The complexity of blood analysis has historically required laboratory-based methods; however, recent advances have brought about portable and wearable devices. Flexible nanomaterial sensors, such as those using GeSe, are integrated into wearable devices to measure blood glucose levels non-invasively (Figure 2). These sensors employ light polarization techniques and machine learning algorithms to enhance measurement accuracy, offering a seamless and reliable monitoring experience (Marco, 2024).
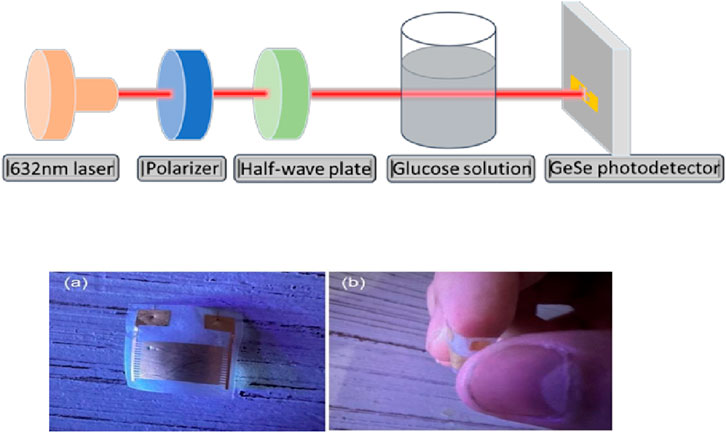
Figure 2. Representation of a non-invasive optical glucose-sensing system that measures blood glucose levels through light-polarization techniques enhanced with machine-learning algorithms for improved accuracy and user experience (Marco, 2024). (a) Illustration of the GeSe-based nanometric sensor architecture. (b) Demonstration of the sensor’s mechanical flexibility, showing that it can be freely bent without performance degradation.
Electrochemical sensors are the gold standard for detecting analytes in blood due to their sensitivity, specificity, and adaptability to miniaturized designs. The i-STAT system, widely used in clinical settings, measures pH, electrolytes, and glucose but lacks the portability needed for wearable applications. Emerging wearable technologies aim to bridge this gap. Microneedle-based sensors (Figure 3) provide a minimally invasive solution for sampling interstitial fluid (ISF), which is compositionally similar to blood (Aroche et al., 2024). These sensors use polymeric microneedles combined with electrochemical detectors to measure lactate, glucose, and electrolytes in real time (Chua et al., 2013).
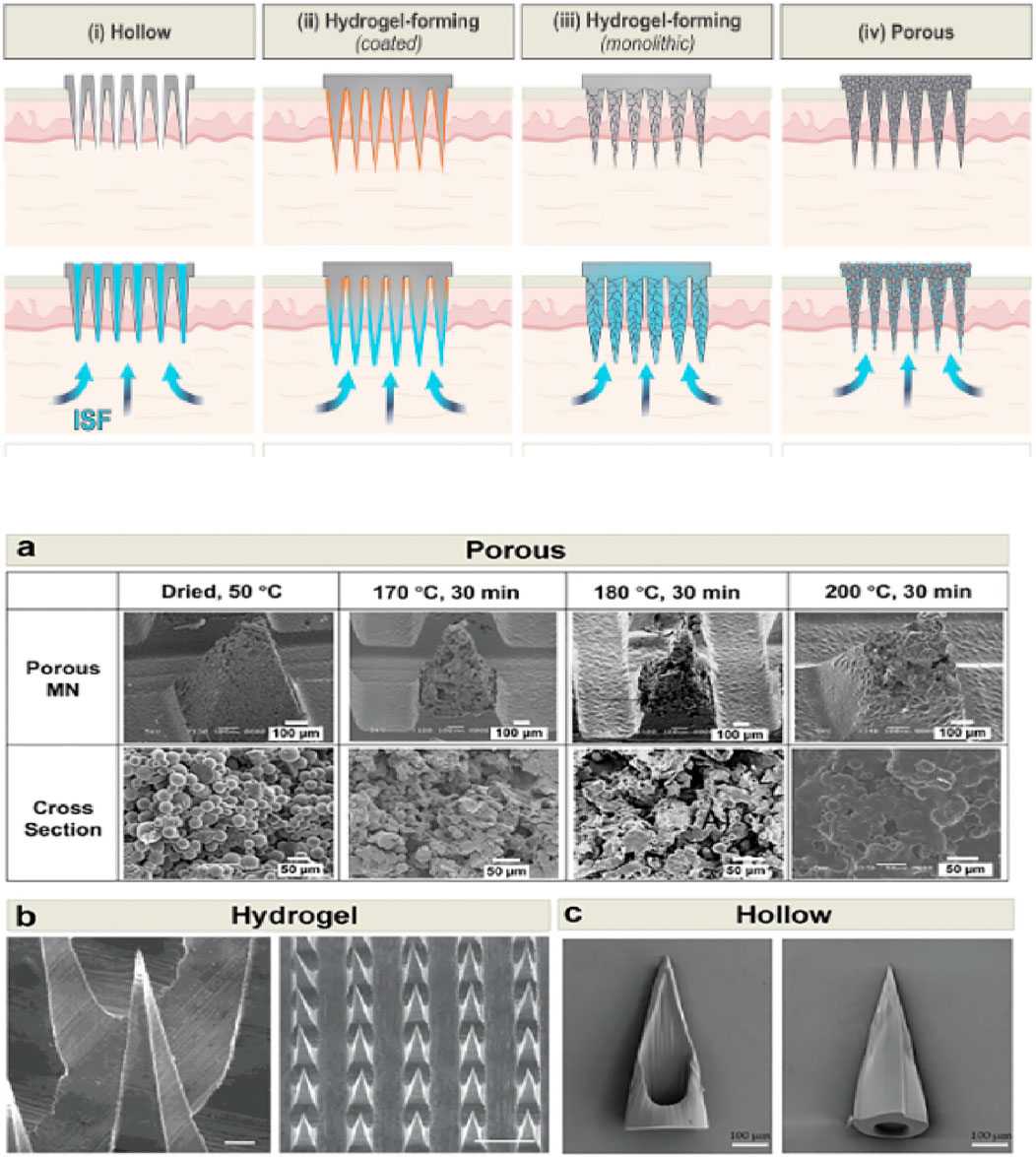
Figure 3. Detailed imagery of hydrogel-forming microneedle arrays combined with an electrochemical detector (the image above), highlighted by Scanning Electron Microscope (SEM) pictures of microneedles (the image below). This figure emphasizes the innovative approach to minimally invasive, real-time biochemical monitoring (Aroche et al., 2024). The SEM images include: (a) heat-treated porous PLA microneedles, (b) methacrylated hyaluronic acid (MeHA) hydrogel microneedles shown in top and side views, and (c) photolithographically fabricated hollow polymeric microneedles.
While ISF sensors with polymeric or hydrogel form microneedles and electrochemical detectors provide valuable insights into vital biomarkers, they still require skin penetration, which can be perceived as invasive and may cause discomfort or reluctance among users. This perception could impact user compliance. Furthermore, repeated application of these devices can lead to skin irritation or allergic reactions, and the variability in individual skin properties may affect the accuracy and reliability of the data collected. Additionally, the cost of these advanced sensors can be prohibitive, limiting accessibility, particularly in low-resource environments.
To enhance the appeal and utility of these technologies, there is a significant push towards improving and expanding non-invasive monitoring methods. Efforts are focused on refining sensor designs to eliminate skin penetration entirely, employing advanced algorithms and calibration techniques to adjust for individual differences, reducing manufacturing costs, and increasing educational outreach to inform potential users about the non-invasive options available. These strategies aim to make non-invasive health monitoring more acceptable, accurate, and accessible, thereby maximizing its potential in healthcare and sports science.
In addition, the incorporation of RFID technology enhances the functionality of wearable blood sensors. RFID modules allow real-time data transfer to mobile devices, enabling remote health monitoring. For instance, an RFID-integrated glucose sensor can continuously monitor and transmit blood glucose levels to smartphones, offering significant advantages for diabetic athletes (Sheng et al., 2022). While RFID offers passive operation, emerging BLE and Zigbee systems enable multi-node body networks for blood parameter telemetry, expanding connectivity and data throughput for real-time physiological monitoring.
3.3 Manufacturing approaches
Advancements in additive manufacturing have revolutionized the production of blood sensors. Techniques such as two-photon polymerization and stereolithography enable the fabrication of microstructures with sub-micrometer precision. These methods are particularly useful for creating microneedle arrays and sensor electrodes (Figure 3). Materials like polydimethylsiloxane (PDMS) and hydrogels are commonly used as substrates due to their biocompatibility and flexibility. Conductive materials, including graphene and silver nanowires, are integrated to enhance sensor performance. For example, graphene-coated microneedles exhibit superior electrical conductivity, making them ideal for electrochemical sensing (Yang et al., 2020). Hybrid manufacturing approaches are also gaining traction. Roll-to-roll printing is used to produce large-scale RFID antenna systems, which are integrated with 3D-printed sensor components to create compact, multifunctional devices. These methods reduce production costs while maintaining high performance and reliability.
In addition, the real-time monitoring of blood parameters has applications beyond athletic performance. For instance, in critical care, wearable blood sensors can track acid-base balance and electrolyte levels, reducing the need for frequent venipuncture. Additionally, in diabetes management, continuous glucose monitors (CGMs) improve glycemic control, reducing the risk of long-term complications. Lactate sensors are also used in clinical settings to assess sepsis severity and in sports science to optimize training regimens (Deulkar et al., 2024).
3.4 Hybrid and on-hand sensing platforms (lab-on-a-glove systems)
Recent “lab-on-a-glove” systems combine tactile sampling with on-site chemical sensing (UC San Diego Jacobs School of Engineering, 2017). Enzyme-coated electrodes on the fingertips can detect hazardous agents such as organophosphates or opioids upon surface contact (Mishra et al., 2017). Sweat-based glove sensors further enable in-motion monitoring of lactate and electrolytes during exercise (Luo et al., 2018). These glove-integrated biosensors bridge physical interaction and biochemical analytics—transforming the hand into an intuitive diagnostic interface.
4 Sweat sensors
4.1 Characteristics and parameters
Sweat is one of the most extensively studied biofluids in wearable sensor research due to its easy accessibility and non-invasive collection, serving as a valuable proxy for assessing both metabolic and hydration status (Meng et al., 2024; Zhang et al., 2024). This makes it particularly useful for monitoring athletic performance and general health. Sweat primarily consists of water, electrolytes (Na+, K+, Cl−), metabolites such as lactate and glucose, and trace biomarkers like cortisol (Koh et al., 2016; Sonner et al., 2015). Its pH typically ranges from 4.0 to 6.8, and deviations from this range may reflect skin-barrier dysfunction or metabolic stress (Choi and Fluhr, 2024); elevated pH levels often result from dehydration or excessive sweating, whereas acidic conditions are associated with irritation and inflammation. Electrolyte concentrations, especially sodium and potassium, provide insights into hydration and thermoregulation, while sweat conductivity reflects total ionic strength and thus overall electrolyte loss. Lactate concentration correlates with the anaerobic threshold and metabolic efficiency, marking exercise intensity (Hiroki et al., 2023). Owing to this dynamic composition, sweat is an excellent medium for real-time monitoring through wearable technologies, particularly in sports and occupational health applications. Notably, sweat composition varies by anatomical region—axillary sweat contains more lipids and volatile organic compounds, forehead sweat primarily reflects thermoregulation, and scalp sweat is linked to sebaceous activity—necessitating region-specific sensor calibration and substrate design (Gallagher et al., 2008; Baker, 2019).
4.2 Sweat sensor innovations
The working mechanism of these sensors typically involves the integration of microfluidic systems and biorecognition elements on flexible substrates. Sweat is directed through microfluidic channels to designated sensing regions, where selective detection occurs using electrochemical, optical, or colorimetric methods (Figure 4) (Bandodkar et al., 2019). For instance, enzymatic electrodes in the sensor oxidize glucose or lactate in sweat, generating an electrical signal proportional to the analyte concentration. These signals are then processed by embedded electronics and transmitted wirelessly for real-time monitoring. The integration of nanomaterials, such as graphene and metal-organic frameworks, enhances sensitivity and specificity. Sweat sensors offer a non-intrusive, continuous, and real-time monitoring approach, making them highly applicable for personalized healthcare, sports performance tracking, and disease management.
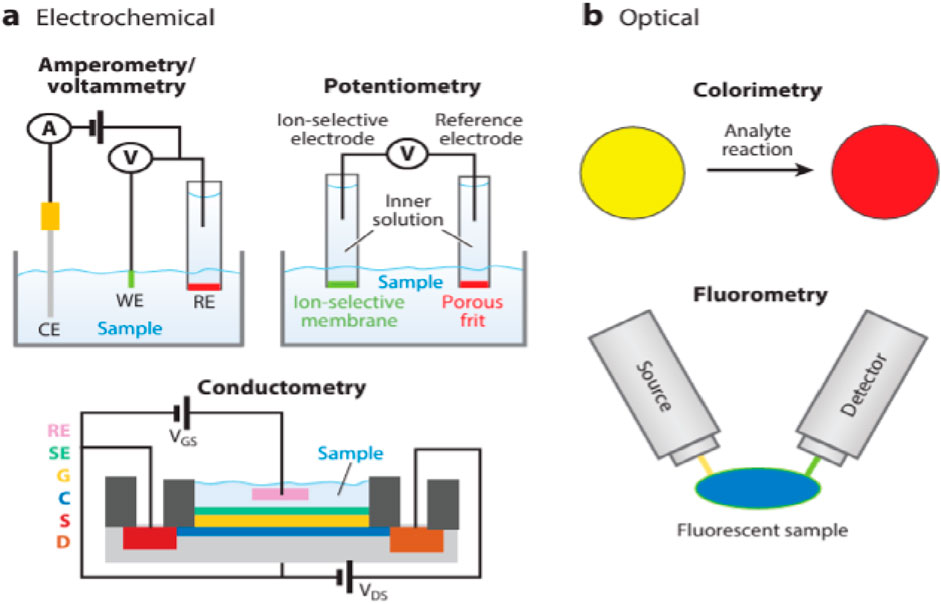
Figure 4. Illustration of the primary working mechanisms of sweat sensors, focusing on the electrochemical and optical detection methods. This figure highlights the sensors’ capabilities to analyze sweat for health monitoring, reflecting advancements in wearable technology ((a) Electrochemical, (b) Optical) (Bandodkar et al., 2019).
Recent advancements in sensor technology have driven the development of innovative devices for sweat monitoring, utilizing a variety of materials and techniques to enhance reliability, accuracy, and usability in real-time applications.
Metal oxide-based sensors, such as those combining RuO2 and TiO2, are renowned for their stability and ease of fabrication. Constructed using thick-film technology and optimized with an 80:20 ratio of RuO2 to TiO2, these sensors deliver excellent performance for sweat pH monitoring, fine-tuned through impedance spectroscopy (Pagar et al., 2024). Potentiometric sensors employ ion-selective electrodes (ISEs) crafted with advanced printing methods like aerosol-jet printing. These compact and cost-effective sensors feature pH-sensitive membranes on flexible substrates like TPU and PET, achieving reliable readings with a potentiometric response of −53.48 mV/pH on PET substrates (Dominiczak et al., 2024). Optical sensors provide a non-invasive solution by leveraging single-walled carbon nanotubes (SWCNTs) that emit near-infrared light for rapid, precise pH detection, particularly useful in healthcare and environmental monitoring (Sultana et al., 2023). Similarly, fiber-optic sensors, featuring tapered interferometers coated with pH-sensitive membranes, detect changes through wavelength shifts, offering broad-range sensitivity for physiological applications (Lei et al., 2023).
Flexible sensors are transforming sweat monitoring by conforming to the skin’s surface. Thin films made from Sb2O3/Sb offer lightweight, sensitive, and accurate readings across a wide pH range, while MoS2-polyaniline-based sensors provide stable, non-invasive monitoring for both natural and artificial sweat samples (Ghadge et al., 2024). Textile-based and colorimetric sensors further enhance practicality by integrating sweat monitoring directly into clothing. Curcumin-based fibers visually indicate pH changes through color variation, while biodegradable wool-based sensors like “Woolitmus” offer an eco-friendly, zero-waste alternative (Ghadge et al., 2024). Advanced wearable devices now combine microfluidic systems with electrochemical sensors to provide continuous analysis of sweat biomarkers. Systems like the Gatorade GX Patch direct sweat into sensing chambers using capillary action, measuring pH, electrolytes, and glucose levels. Flexible electronics using materials such as graphene and silver nanowires further enhance these devices by enabling wireless data transmission via Bluetooth or RFID. RFID-enabled sensors, particularly those integrated with microfluidic systems, are gaining popularity for their cost-effective real-time tracking of sweat pH, especially during physical activities, making them invaluable for athletes and individuals engaging in exercise (Ghadge et al., 2024).
Sweat cortisol sensors are another groundbreaking innovation, offering a non-invasive method for assessing stress and recovery. By using graphene oxide functionalized with antibodies or aptamers, these sensors achieve high specificity and sensitivity, enabling real-time stress monitoring (Riente et al., 2023). These advancements in sweat sensor technology are revolutionizing health and performance monitoring, providing precise and real-time insights with wide-ranging applications in sports, healthcare, and beyond.
4.3 Material and manufacturing insights for sweat sensors
Sweat sensors require biocompatible, flexible, and durable materials to withstand prolonged wear and mechanical stress during physical activities. Substrates such as polydimethylsiloxane (PDMS), thermoplastic polyurethane (TPU), and polyethylene terephthalate (PET) are commonly used for their desirable properties (Ates et al., 2022). Conductive materials like graphene, carbon nanotubes, and silver nanowires are integrated into electrodes to enhance electrical conductivity and sensitivity. For example, graphene-based materials are particularly suited for electrochemical sensing due to their high surface area and exceptional conductivity (Li et al., 2018). Additive manufacturing techniques, including fused deposition modeling (FDM) and stereolithography (SLA), have revolutionized the rapid prototyping of complex microfluidic systems. FDM is utilized for fabricating sweat channels and reservoirs, while SLA enables the creation of high-resolution sensing structures. Additionally, hybrid printing approaches, such as combining direct ink writing (DIW) of conductive inks with 3D-printed substrates, are increasingly employed to integrate sensing and communication modules (Gao et al., 2021). In literature, additive manufacturing, particularly the “one-step” fabrication process, has emerged as a key technique for developing flexible sensors. This method integrates layers of electrospun PVDF, printed silver electrodes, and electrospun TPU encapsulation to produce breathable, sweatproof, and highly responsive sensors ideal for wearable applications (Zhiqiang et al., 2024). Similarly, hydrothermal and one-pot preparation methods are utilized to create non-enzymatic electrochemical glucose sensors by functionalizing gold nanoparticles on aminated multi-walled carbon nanotubes, which are then crosslinked with carboxylated styrene-butadiene rubber and PEDOT: PSS. These flexible sensors, integrated onto screen-printed electrodes, enable high-sensitivity continuous glucose monitoring in sweat (Yuhua et al., 2024). The stamping-vacuum filtration dry transfer (SVFDT) technique offers a cost-effective approach for constructing wearable sweat glucose sensors. By combining a PVC stamp and vacuum-filtration, multi-walled carbon nanotube/PDMS film electrodes are fabricated, improving conductivity and stability for real-time sweat glucose detection (Youyuan et al., 2023). Another innovative approach, electro-assisted impregnation core-spinning technology (EAICST), is used to develop textile-based sweat sensors. This method combines impregnation coating and electrospinning to produce sheath-core electrochemical sensing yarns that are integrated into fabrics, enabling scalable production of durable, high-sensitivity, and washable sensors (Xiangda et al., 2024). Microfluidic and colorimetric analysis methods further advance wearable sensors, with hydrophilic yarn-based patches designed for sweat collection and real-time analysis through colorimetric indicators. Additionally, Janus nanofiber membranes improve directional sweat transfer, enhancing comfort and accuracy by preventing sweat accumulation (Wenze et al., 2023; Xuecui et al., 2024). Advanced materials and techniques such as 3D graphene foam and PDMS-based sensors have also demonstrated high sensitivity and stability in detecting uric acid in sweat, while waveguide and all-printed flexible sensors present innovative solutions for glucose and sweat monitoring (Mingrui et al., 2023; Balasubramanian et al., 2024; Chandradas et al., 2024). Together, these methods highlight the potential of wearable sweat sensors in healthcare, sports science, and beyond. To facilitate wireless data transfer, RFID technology is often incorporated into wearable sweat sensors, enabling remote monitoring by transmitting real-time data to smartphones or cloud-based platforms. Moreover, Bluetooth Low Energy (BLE) modules are frequently used for high-resolution data transmission over short distances.
In addition, wearable sweat sensors have a broad range of applications, particularly in sports science, occupational health, and medical diagnostics. For athletes, sweat monitoring provides real-time feedback on hydration and electrolyte levels, helping prevent dehydration and heat-related illnesses (Belabbaci et al., 2025). In occupational health, these devices are used to monitor workers in high-temperature environments, ensuring safe hydration and electrolyte balance. Medical applications include monitoring metabolic conditions, such as cystic fibrosis, through chloride concentration analysis. Recent studies have also explored the use of sweat sensors for early detection of infectious diseases, leveraging sweat biomarkers like cytokines and immune response markers (Ray et al., 2021).
4.4 Smart wound dressings and infection-responsive sweat sensors
Smart wound dressings now extend sweat sensing toward infection detection (Wang X. et al., 2024; Youssef et al., 2023). Changes in wound pH or bacterial metabolites such as pyocyanin signal infection onset, providing early indicators of bacterial colonization and inflammation (Wang X. et al., 2024; Gu et al., 2025). Aptamer-based hydrogels can even recognize pathogens such as Staphylococcus aureus or bacterial endotoxins, enabling selective and rapid detection in complex wound environments (Dsouza et al., 2022; Cristea et al., 2025). Some emerging “theranostic” systems not only detect infection but also release antibiotics or antimicrobial agents upon bacterial trigger, achieving on-demand therapy (Su et al., 2024; Cristea et al., 2025; Cao et al., 2023). This convergence of sweat and wound analytics redefines biosensing as both diagnostic and therapeutic, integrating real-time monitoring with intelligent drug delivery for advanced wound management (Yang et al., 2023; Vo and Trinh, 2025; Moradifar et al., 2024).
5 Saliva sensors
5.1 Characteristics and parameters
Saliva is increasingly recognized as a valuable biofluid for health monitoring due to its non-invasive collection, ease of handling, and rich biochemical composition. Secreted by salivary glands, it is primarily composed of water (approximately 99%) along with electrolytes, enzymes, hormones, glucose, and immune markers. Saliva acts as a mirror of systemic health, with many of its constituents closely correlating with blood levels (Lakshminrusimhan et al., 2024). Among the key parameters, pH typically ranges from 6.2 to 7.6 and serves as an indicator of oral health and systemic acid-based balance. A decrease in pH may signal oral infections or heightened acidity due to poor hydration, whereas an increase could point to reduced saliva flow or conditions like Sjögren’s syndrome. Cortisol, a stress biomarker, provides insights into hypothalamic-pituitary-adrenal (HPA) axis activity; elevated levels suggest acute stress, while chronic elevations may indicate long-term physiological strain or insufficient recovery (Choi et al., 2021). Salivary glucose emerges as a non-invasive metric for monitoring blood glucose levels in diabetic patients, offering a practical alternative to invasive blood sampling, especially for continuous monitoring. Additionally, immunoglobulin A (IgA), a critical antibody in mucosal immunity, reflects immune status. Low levels of salivary IgA are linked to a higher risk of infections, making it a significant parameter for athletes undergoing intense training (Ryland et al., 2022).
5.2 Wearable saliva sensors
Saliva sensors operate by detecting and analyzing biomarkers present in saliva, providing valuable insights into an individual’s health status (Anchidin-Norocel et al., 2024). These sensors typically utilize biochemical recognition elements, such as enzymes or antibodies, to specifically bind target analytes like glucose, lactate, or cortisol. Upon binding, a transducer converts this biochemical interaction into a measurable signal, often electrical or optical, which correlates with the concentration of the analyte. This non-invasive monitoring approach facilitates real-time health assessments, offering a convenient alternative to traditional methods.
Wearable saliva sensors represent a transformative advancement in healthcare and performance monitoring, leveraging flexible electronics and advanced materials to provide real-time, non-invasive analysis with high sensitivity. Graphene-based sensors are particularly prominent in this field due to their superior conductivity, mechanical flexibility, and biocompatibility. For instance, salivary cortisol detection is achieved using graphene oxide electrodes functionalized with aptamers or antibodies, ensuring specificity and selectivity (Zaryab et al., 2024). Disposable biosensors, such as inkjet-printed devices utilizing conductive inks on biocompatible substrates like polyethylene terephthalate (PET), offer a cost-effective solution for saliva analysis. These single-use sensors deliver rapid and accurate measurements of biomarkers, including glucose and immunoglobulin A (IgA) (Ray et al., 2021). Additionally, RFID technology has been integrated into wearable saliva sensors to enable wireless data transmission. RFID based sensor, which senses glucose level changing by RF responses, is shown in Figure 5. Thus, RFID chips can transmit data to a smartphone, providing real-time stress assessments. Such innovations are particularly advantageous in sports science, where timely feedback on stress and recovery is essential (Choi et al., 2021).
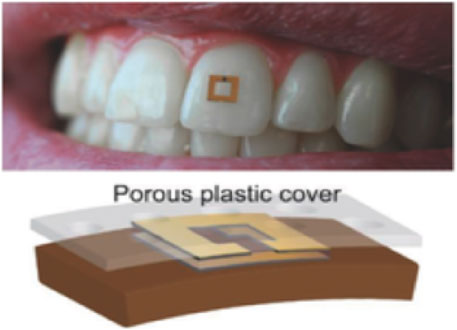
Figure 5. An RFID-based saliva sensor mounted on a tooth, illustrating the sensor’s functionality to detect changes in glucose levels by RF responses. This innovative approach enables real-time, non-invasive monitoring of stress and recovery in sports science (Tseng et al., 2018).
5.3 Insights into materials and manufacturing processes for saliva sensors
The development of saliva sensors necessitates materials capable of withstanding the wet and enzymatically active environment of the oral cavity. Hydrogels are often utilized as substrates due to their biocompatibility and ability to retain moisture. To enhance sensor performance, conductive materials such as carbon nanotubes (CNTs) and silver nanowires are integrated into these substrates. Additive manufacturing techniques, including stereolithography (SLA) and direct ink writing (DIW), have proven essential in the fabrication of saliva sensors, particularly for creating intricate microfluidic channels that direct saliva samples to specific sensing regions, thereby improving measurement accuracy. Inkjet printing is especially effective for depositing conductive materials, facilitating the production of lightweight, flexible, and disposable saliva sensors. In the literature, 3D printing is being utilized to fabricate flexible ion-selective field effect transistors (ISFETs) for saliva analysis (Chao et al., 2019). This technique involves the hybridization of printed organic ion-selective electrodes with inorganic transistors, providing high sensitivity and selectivity in detecting ions such as ammonium, potassium, and calcium, as reported by Bao et al. (2019). Another innovative approach is the Spin Coating and Plasma Treatment, part of the Spin Coating-Plasma treatment-Coprecipitation (SPC) strategy (Tingjun et al., 2024). This method is used to develop super-hydrophilic gel sensors, significantly enhancing the sensor’s surface properties to improve its effectiveness in saliva glucose monitoring, according to Chen et al. (2024). Additionally, Dip-Coating is a straightforward and efficient manufacturing technique for creating liquid flexible sensors. It involves coating a cellulose fabric with a conductive composite material, making it a cost-effective option suitable for large-scale production, as described by Siyi and Yinxiang (2019) (Bi and Lyu, 2019). Each of these techniques contributes uniquely to advancing sensor technology, offering tailored solutions for specific biochemical analyses. Additionally, the integration of communication modules, such as RFID and near-field communication (NFC) systems, allows for wireless data transfer. These modules are often fabricated using roll-to-roll printing techniques, enabling large-scale production while ensuring the sensors remain reliable and high-performing.
Furthermore, wearable saliva sensors have diverse applications, ranging from stress and hydration monitoring in athletes to chronic disease management. In sports science, salivary cortisol and IgA levels are used to assess recovery and immune function, guiding training regimens to minimize overtraining risks (Ryland et al., 2022). In medical diagnostics, saliva sensors are being developed for early detection of conditions such as diabetes and oral cancers. Glucose monitoring devices offer a non-invasive alternative for diabetic patients, while salivary biomarkers for oral cancers are being explored for rapid, point-of-care diagnostics.
5.4 Smart pacifier biosensors for neonatal monitoring
In neonatal care, saliva-based “smart pacifiers” replace invasive blood draws (Lim et al., 2022; García-Carmona et al., 2019). These pacifiers integrate microfluidic channels and ion-selective electrodes to continuously monitor sodium and potassium levels (Lim et al., 2022). Data transmitted via Bluetooth enables early detection of dehydration or kidney stress. Other prototypes target glucose monitoring in neonatal hypoglycemia (García-Carmona et al., 2019; Zhou et al., 2024). Such soft, baby-safe devices highlight how biosensing can be empathetic, non-invasive, and child-centered.
6 Urine sensors
6.1 Characteristics and parameters
Urine, a biofluid excreted by the kidneys, serves as an accessible and rich diagnostic medium, providing valuable insights into the body’s hydration status, electrolyte balance, and metabolic health (Gao et al., 2024; Lee et al., 2024). Unlike blood, urine analysis captures cumulative physiological changes over time, making it particularly useful for long-term monitoring. Key diagnostic parameters include pH, osmolality, electrolytes, proteins, glucose, and ketones. The pH of urine, typically ranging from 4.5 to 8.0, reflects the body’s acid-based balance, with alkaline urine potentially indicating urinary tract infections (UTIs) or dietary influences, while acidic urine is often linked to high protein intake or conditions like ketoacidosis. Urine osmolality, generally between 100 and 1,200 mOsm/kg, directly measures solute concentration and hydration status, with high values often signaling dehydration or kidney stress, and low values suggesting overhydration or renal dysfunction. Electrolyte levels, including sodium, potassium, and chloride, are critical for muscle function and physiological stability. Proteinuria, the presence of excess proteins in urine, is a marker of kidney dysfunction or systemic inflammation. Additionally, glucose and ketones are significant markers in diabetes management, indicating hyperglycemia or ketosis (Shirreffs and Maughan, 1998; Li et al., 2018). Its composition and variability, urine is an excellent candidate for wearable monitoring systems, particularly for athletes and patients with chronic conditions.
6.2 Urine sensor development
Wearable urine sensors are rapidly emerging as innovative and practical tools for non-invasive health monitoring, offering significant advancements in real-time health tracking (Fakhr et al., 2024). These sensors typically utilize electrochemical, optical, or piezoelectric principles to transduce chemical interactions into measurable electrical signals. These devices aim to deliver continuous data on hydration status, electrolyte balance, and metabolic health, eliminating the need for frequent laboratory visits and traditional invasive procedures. They are particularly beneficial in medical diagnostics and for individuals such as athletes or workers in physically demanding environments, who require consistent monitoring of their physiological status.
One of the key technologies driving this innovation is flexible electrochemical sensors, which are engineered to detect parameters like pH, osmolality, and electrolyte concentrations. These sensors often utilize screen-printed electrodes coated with conductive polymers such as poly (3,4-ethylenedioxythiophene) (PEDOT), a material renowned for its exceptional sensitivity and durability (Gao et al., 2021). In this context, flexible and wearable humidity sensors, which are used as urine sensors and manufactured with PEDOT: PSS, provide an effective solution for detecting urinary incontinence. These sensors can be integrated between the cover stock and the acquisition/distribution layer (ADL) of the adult diaper (Tekcin et al., 2022). This placement allows the sensor to effectively detect urinary incontinence while preventing direct contact with the skin. The use of such advanced materials ensures accurate and reliable performance, even in challenging conditions.
Another essential component of wearable urine sensors is the integration of microfluidic platforms, which streamline the sample collection process. These platforms leverage capillary forces to direct urine samples to the sensing regions passively, requiring minimal user intervention. This design is particularly advantageous for continuous monitoring, as it reduces the burden on users while maintaining consistent data collection. Such systems have proven valuable for monitoring individuals exposed to high-stress environments, including athletes and emergency responders (Yang et al., 2020).
In recent advancements, optical and multi-parameter sensing technologies have gained prominence. These hybrid sensors combine optical and electrochemical methods to measure multiple biomarkers simultaneously, such as pH, glucose, and ketones. This multifunctional capability provides a holistic view of an individual’s health, enhancing the potential for early detection of conditions like dehydration, metabolic disorders, or stress-related imbalances.
Collectively, these advancements in wearable urine sensor technology represent a paradigm shift in health monitoring, combining comfort, accessibility, and precision to empower individuals and healthcare professionals alike. As research continues to refine these systems, their potential applications are poised to expand, driving transformative impacts in personalized medicine and public health.
6.3 Advances in material and manufacturing for urine sensors
The materials used in wearable urine sensors must withstand the corrosive nature of urine while ensuring accurate biomarker detection (Anshin et al., 2024). Hydrophobic polymers like polyethylene terephthalate (PET) are often utilized for device housings due to their durability, while conductive materials such as silver nanowires and graphene are incorporated into sensing elements to enhance performance. Various manufacturing techniques have been developed to enhance the design and functionality of urine sensors, each employing unique methods and materials. The Pad Printing Technique, as detailed by Tekçin and Bahadır (2024), uses silver and silver/nickel inks to fabricate flexible urine sensors (Meltem and Kursun, 2024). This process involves multiple sintering passes to improve conductivity and surface morphology, essential for accurately detecting changes in resistance when exposed to urine. Another innovative approach is the use of Electrospinning and Carbon Nanofibers, where phosphated lignin is incorporated to enhance the flexibility and thermal stability of the fibers. This green strategy, described by Zheng et al. (2022), increases the sensor’s ability to detect uric acid in urine through improved molecular interactions and electron transmission (Hao et al., 2022). Lastly, Contactless Jet Printing, as implemented by Zhang et al. (2023), offers a rapid manufacturing solution for creating ultrathin, highly sensitive moisture sensors (Lan et al., 2023). These sensors are directly printed onto diapers or undergarments, facilitating continuous urine monitoring while maintaining user comfort. Each technique represents a significant advancement in sensor technology, addressing specific challenges in moisture detection and wearable comfort.
Furthermore, wearable urine sensors are transforming the way hydration, electrolyte balance, and kidney function are monitored, offering significant benefits for athletes, patients with chronic illnesses, and individuals in high-risk professions. In sports science, these sensors provide real-time feedback on hydration and electrolyte levels, helping athletes prevent dehydration and heat-related illnesses. By monitoring osmolality and sodium excretion during high-intensity activities, these devices enable tailored hydration strategies to optimize performance (Shirreffs and Maughan, 1998). In medical diagnostics, wearable urine sensors are invaluable for patients with chronic kidney disease, facilitating continuous monitoring of renal function. Diabetic patients also benefit from these devices, as they enable glucose and ketone monitoring, reducing the need for invasive blood tests. In occupational health, wearable urine sensors are critical for ensuring proper hydration and electrolyte balance under extreme conditions, such as in mining or firefighting environments (Li et al., 2018). Additionally, researchers are exploring the potential of urine biomarkers for the early detection of urinary tract infections (UTIs) and systemic inflammatory conditions, using wearable sensors to provide rapid, and point-of-care diagnostics (Yang et al., 2020).
6.4 Diaper-integrated smart urine sensors
Smart diapers transform routine care into real-time health tracking (Tanweer et al., 2024). Printed impedance or capacitive sensors detect urination events and transmit data via Bluetooth (Xiong et al., 2024). Some systems measure uric acid or ionic composition to assess hydration and renal health (Shitanda et al., 2021). Commercial devices like Monit™ and Opro9™ already demonstrate this concept in infant and elderly care, helping to prevent rashes and monitor urinary infections (Opro9, 2025). Disposable yet intelligent, these systems redefine the interface between comfort and diagnostics.
7 Tears sensors
Tears have become a focal point for wearable sensor applications, recognized for their easy accessibility and the wealth of biomarkers they carry. Parameters like osmolality, electrolytes, and stress-related hormones such as cortisol make tears especially valuable for monitoring. These biomarkers yield crucial insights into not only ocular health but also broader systemic conditions, making tear analysis an impactful tool in sports, healthcare, and ophthalmology (Bron et al., 2007). For athletes, in particular, the ability to monitor changes in tear osmolality and cortisol levels offers direct benefits for managing hydration and stress, critical factors in achieving optimal performance. The use of advanced microfluidic and electrochemical technologies in wearable devices facilitates the real-time, non-invasive tracking of these biomarkers, significantly advancing personalized healthcare and sports science initiatives.
7.1 Characteristics and parameters
Tear fluid contains various biomarkers that provide critical insights into ocular and systemic health. One such marker is osmolality, a vital parameter for evaluating dry eye syndrome and systemic hydration. Typically ranging between 270 and 300 mOsm/kg, tear osmolality serves as an important indicator of ocular surface health, as highlighted in studies by (Bron et al., 2007). This measurement is widely utilized in both clinical and research settings to monitor changes in the tear film associated with environmental stressors or pathological conditions.
Another significant biomarker is cortisol, a hormone presents in tears at concentrations of approximately 10 ng/mL. Elevated levels of tear cortisol have been linked to increased stress and mental fatigue, making it a valuable, non-invasive tool for assessing physiological strain. Research by (Iqbal et al., 2023) emphasizes the potential of tear cortisol measurements to provide real-time insights into an individual’s stress response, offering applications in both healthcare and performance monitoring.
Lastly, the levels of electrolytes such as sodium and potassium play a crucial role in maintaining ocular homeostasis. These electrolytes not only support the integrity of the tear film but also serve as indicators of electrolyte imbalances that could impact both eye and systemic health. Yeo et al. (2015) underline their importance in diagnosing and managing conditions related to hydration and ionic disturbances. Together, these biomarkers—osmolality, cortisol, and electrolytes—represent a comprehensive approach to understanding and monitoring the intricate connections between ocular health and systemic wellbeing (Wang Z. et al., 2024).
7.2 Tear sensor applications
Tear sensors are becoming increasingly important tools for monitoring our health, particularly in areas like ocular health, hydration, and stress management (Rajan et al., 2024). For people with conditions such as dry eye syndrome, devices like the TearLab Osmolarity System use microfluidic technology to measure tear osmolality, helping with diagnosis and treatment (Zazoum et al., 2022). Similarly, wearable glasses with built-in optical sensors provide real-time updates on hydration levels by tracking changes in tear osmolality, making it easier to maintain eye health (Lan et al., 2023).
When it comes to managing stress, tear sensors offer unique insights by detecting cortisol levels, a key stress hormone. Electrochemical sensors equipped with specialized molecules like aptamers or antibodies are highly sensitive, making them ideal for tracking stress, especially for athletes (Ghadge et al., 2024). Some advanced versions even use RFID technology to send real-time data to smartphones, making stress management more accessible (Ungaro et al., 2015).
For those concerned about hydration and electrolyte balance, wearable patches with ion-selective electrodes (ISEs) can measure sodium and potassium levels in tears, helping to prevent imbalances that could affect overall health (Yeo et al., 2015). On the inflammation front, tear sensors break new ground by detecting inflammatory markers, offering a way to diagnose conditions like conjunctivitis or autoimmune diseases. Wearable sensors that analyze proteins in tears are also emerging, using techniques like mass spectrometry to provide a deeper understanding of both eye and systemic inflammation (Iqbal et al., 2023).
In addition, tear sensors are expanding into broader health applications, such as glucose monitoring for diabetes. Devices using advanced nanomaterials now offer a non-invasive way to track glucose levels continuously, improving the lives of those managing diabetes (Sandra, 2023; Zhou, 2024). Tear sensors built into contact lenses, using fluorescence technology, are another breakthrough. These can detect key biomarkers for inflammation and disease, enabling personalized and immediate care (Shi et al., 2024; Shi et al., 2023). With these advancements, tear sensors are proving to be more than just medical devices—they are tools that empower people to take charge of their health in a non-invasive, real-time way. Whether managing stress, hydration, or chronic diseases, these sensors are paving the way for a future of more personalized and accessible healthcare.
7.3 Materials and manufacturing for tear sensors
Tear sensors detect biochemical markers in tear fluid using advanced technologies such as electrochemical, optical, and piezoelectric mechanisms. These sensors frequently incorporate nanomaterials like graphene, carbon nanotubes, and metallic nanoparticles to achieve higher sensitivity and specificity (Rajan et al., 2024). For example, electrochemical tear sensors measure glucose levels for diabetes management by detecting the oxidation of glucose molecules through enzymatic reactions on the sensor’s surface. Optical sensors utilize fluorescence or changes in refractive index to detect analytes like lactate or electrolytes, which are crucial for assessing hydration and metabolic health. Piezoelectric mechanisms excel in measuring tear flow and viscosity, offering valuable insights into ocular surface disorders such as dry eye disease. When integrated into wearable devices like smart contact lenses, tear sensors provide continuous and non-invasive health monitoring, presenting a transformative approach to personalized healthcare.
The functionality of tear sensors relies on specialized materials and precise manufacturing techniques to accommodate the sensitive ocular environment. Materials such as polydimethylsiloxane (PDMS) deliver the flexibility and biocompatibility needed for wearable tear sensors, while graphene and silver nanowires enhance electrical sensitivity, making them ideal for electrochemical applications targeting tear analysis (J et al., 2023). Advanced manufacturing methods, such as 3D printing, enable the precise fabrication of microfluidic channels to optimize tear collection and analysis (Kim et al., 2018). Additionally, inkjet printing facilitates the production of disposable electrodes, reducing contamination risks in single-use tear sensors (Riente et al., 2023).
These technological advances have significant implications for sports and healthcare. In sports, real-time cortisol tracking through tear sensors supports stress management and recovery optimization for athletes. In healthcare, tear osmolality sensors are vital for diagnosing and monitoring dry eye syndrome, a condition particularly common among athletes frequently exposed to outdoor environments. Furthermore, analyzing tear electrolytes and proteins enables the early detection of systemic health issues, including autoimmune diseases and electrolyte imbalances, offering valuable insights into overall health management.
7.4 Ring-based multimodal platforms for tear and skin biomarkers
Beyond ocular applications, the integration of tear sensing with multimodal wearable platforms such as smart rings bridges biochemical and physiological analytics, enabling holistic athlete monitoring. Smart rings provide continuous, non-invasive insight into physiological trends such as heart rate, sleep, and temperature (Fiore et al., 2024). Advanced prototypes integrate optical and electrochemical sensors to estimate glucose and blood pressure non-invasively (Sempionatto and Wang, 2017; Tang et al., 2025). Compact, waterproof, and discreet, they extend biosensing to daily life—merging comfort, analytics, and personalized health intelligence.
8 Other body fluids
Wearable sensor technology is breaking new ground by incorporating less conventional biofluids, such as semen and vaginal fluids, to provide advanced insights into health monitoring, including applications tailored for athletes. These biofluids contain critical biomarkers that offer valuable information on hydration, recovery, and reproductive health, presenting new opportunities for optimizing athletic performance and wellbeing.
Semen serves as a biomarker-rich fluid that can reflect systemic health and fertility parameters. Advanced sensor systems are being developed to analyze semen’s pH (7.2–8.0), sperm motility, and electrolyte concentrations in real time. Though wearable solutions are in the research phase, portable microfluidic and electrochemical sensors are being optimized for non-invasive sperm motility assessments and monitoring of oxidative stress markers like prostate-specific antigen (PSA). These technologies, when adapted to athletes, could monitor hormonal or oxidative imbalances, which directly impact recovery and physical performance (Heikenfeld et al., 2018).
Similarly, reproductive health features unique markers such as a specific pH range (3.8–4.5) within the female reproductive tract. Advanced pH sensors integrated into smart tampons or patches enable non-invasive monitoring of conditions like bacterial vaginosis and yeast infections, as well as the assessment of inflammatory markers such as cytokines. These technologies are particularly useful for athletes, providing real-time insights into hydration levels, immune function, and overall reproductive health. This information is vital for managing the demands of intense physical activities and optimizing recovery periods. Additionally, the use of biosensors to monitor cervical mucus can help in tracking electrolyte shifts and hormonal changes, which can be critical for tailoring training regimens to individual physiological cycles (Choi et al., 2021).
Emerging RFID-enabled wearable systems further enhance these applications. Devices such as sanitary pads equipped with biosensors measure blood parameters like hemoglobin A1c and electrolyte levels, providing a new layer of non-invasive, continuous monitoring. These can be particularly valuable for female athletes in managing menstrual health and its impact on performance. Furthermore, the integration of IoT platforms and AI analytics allows for real-time decision-making, offering athletes actionable insights for tailored hydration, nutrition, and training adjustments.
These innovative sensors, by monitoring biofluids like semen and vaginal fluids, are paving the way for highly specialized health assessments in athletes. By leveraging advanced materials such as graphene and flexible hydrogel interfaces, these devices ensure biocompatibility and precision in dynamic conditions. Future iterations of these sensors, combined with miniaturized electronics and cloud-based analytics, are poised to revolutionize athlete health monitoring, enabling peak performance and reducing injury risks through personalized, data-driven interventions.
9 Advanced materials and manufacturing techniques
9.1 Materials for wearable sensors
Materials science plays a critical role in the design and functionality of wearable sensors for body fluid monitoring. The materials used in these devices must meet strict criteria, including biocompatibility, flexibility, durability, and responsiveness to physiological environments. Given that wearable sensors often operate under dynamic conditions, they require materials capable of maintaining performance despite mechanical stress and exposure to biofluids.
Flexible polymers and substrates, such as polydimethylsiloxane (PDMS), thermoplastic polyurethane (TPU), and polyethylene terephthalate (PET), are widely used in wearable sensors. PDMS stands out for its biocompatibility, optical transparency, and elasticity, making it ideal for skin-conforming applications. TPU, known for its excellent mechanical strength, is frequently employed in flexible microfluidic systems designed for analyzing sweat and urine (Atsani Susanto et al., 2023).
Advanced conductive materials, including graphene, carbon nanotubes (CNTs), and silver nanowires, significantly enhance the electrical performance of wearable sensors. Graphene, with its high surface area and exceptional conductivity, is commonly integrated into electrochemical sensors for detecting biomarkers like glucose and lactate. Silver nanowires, prized for their stretchability and conductivity, are especially suitable for flexible electrodes.
In terms of enhancing sensitivity and expanding detection limits, the limits of detection (LoDs) and sensitivity are crucial parameters that determine the efficacy of wearable sensors in health monitoring, particularly for detecting low-concentration biomarkers essential for early diagnosis and management in personalized medicine and chronic disease scenarios. Notably, graphene-based sensors, such as graphene-based field-effect transistors (GFETs), have achieved remarkably low LoDs—0.022 × 10^−6 M for L-cysteine in sweat and 0.043 × 10^−6 M in artificial tears—demonstrating their capability for sensitive biomarker detection across various body fluids (Huang et al., 2022). Additionally, electrochemical sensors have been refined to provide low LoDs by incorporating advanced materials and microfluidic integration, making them essential for tracking a broad spectrum of biomarkers in sports medicine and chronic illness management (Raymond et al., 2024). The development of dual biofluid sampling platforms has enabled simultaneous monitoring of sweat and interstitial fluid, offering comprehensive biomarker analysis with correlations closely aligned to blood levels, which is vital for extending the sensors’ clinical applicability (Kim et al., 2018).
Hydrogels are another critical component, valued for their ability to retain moisture and mimic the mechanical properties of biological tissues. They are particularly effective in saliva and urine sensors, where direct contact with biofluids is essential. Additionally, hydrogels embedded with ionic conductors or nanomaterials can function as active sensing elements, enhancing signal transduction (Sun Y. et al., 2024).
Smart materials, such as shape-memory alloys and piezoelectric polymers, are being increasingly integrated into wearable sensors to enable advanced functionalities. For example, piezoelectric materials can generate electrical signals in response to mechanical deformation, creating self-powered sensor systems that eliminate the need for external batteries (Gao et al., 2021).
9.2 Additive manufacturing in wearable sensor fabrication
Additive manufacturing, commonly known as 3D printing, has revolutionized the production of wearable sensors by enabling rapid prototyping, customization, and the integration of complex structures. Several advanced 3D printing techniques are utilized in wearable sensor development to meet the specific demands of different applications. Stereolithography (SLA), which employs photopolymerization, is ideal for creating high-resolution structures and is widely used to fabricate microfluidic components and sensor housings. This technique excels in producing saliva and urine sensors with intricate channel designs that efficiently direct biofluids to sensing regions (Li et al., 2018). Fused Deposition Modeling (FDM), a versatile and cost-effective method, is commonly used to create flexible substrates and durable housing for wearable sensors. Materials such as TPU and PETG are frequently employed in FDM to fabricate sweat sensors with complex microfluidic paths, ensuring efficient sweat collection and analysis. Direct Ink Writing (DIW) allows for the precise deposition of conductive inks and polymers onto flexible substrates, facilitating the creation of integrated sensor-electrode systems. This approach is particularly effective for hybrid sensors that combine electrochemical and optical sensing modalities (Ray et al., 2021). Two-Photon Polymerization (TPP) is another innovative technique that enables the fabrication of nanoscale structures with exceptional precision. It is widely used for creating microneedle arrays in blood sensors, allowing for minimally invasive sampling and enhanced sensitivity (Yang et al., 2020). Together, these techniques have significantly advanced the capabilities and versatility of wearable sensor technologies.
9.3 Integration of electronics
The integration of electronic components such as RFID and Bluetooth modules significantly enhances the functionality of wearable sensors by enabling wireless data transmission and real-time monitoring. RFID technology serves as a cornerstone for wearable sensors, particularly in healthcare and sports applications. RFID-enabled sensors facilitate remote monitoring by transmitting data wirelessly to smartphones or cloud platforms, with these modules often fabricated using roll-to-roll printing to ensure scalability and cost-efficiency (Gao et al., 2021). Similarly, Bluetooth Low Energy (BLE) modules are widely employed in wearable sensors for short-range, high-resolution data transmission. BLE is particularly suited for real-time monitoring during physical activity, offering continuous feedback on physiological parameters (Liu et al., 2021). Additionally, advances in flexible electronics have enabled the creation of wearable sensors that conform seamlessly to the skin. Flexible printed circuit boards (FPCBs) integrate sensors, processors, and communication modules into compact, durable devices. These circuits, fabricated with conductive inks on polymer substrates, combine flexibility with reliability, making them ideal for wearable applications (Li et al., 2018).
9.4 Hybrid manufacturing approaches
Hybrid manufacturing combines multiple fabrication techniques to optimize sensor performance and reduce production costs. For instance, 3D printing is often combined with roll-to-roll printing to fabricate wearable sensors with integrated communication modules. These approaches allow for the simultaneous production of structural and electronic components, streamlining the manufacturing process.
9.5 Applications and future directions
The use of advanced materials and manufacturing techniques has significantly expanded the applications of wearable sensors across healthcare, sports science, and occupational health. Future research should prioritize the development of sustainable materials, such as biodegradable polymers, to minimize environmental impact. Efforts should also focus on enhancing sensor sensitivity and selectivity through the integration of nanomaterials, enabling more precise and reliable monitoring. Additionally, scaling up manufacturing processes will be essential to ensure these technologies remain both affordable and accessible. The synergy of additive manufacturing and flexible electronics is poised to drive the next-generation of wearable sensors, enabling seamless integration into daily life and advancing personalized healthcare solutions. Future research should converge soft bioelectronics, AI-driven interpretability, and sustainable 3D manufacturing toward clinically standardized, ethically compliant, and globally deployable wearable platforms. In parallel with these technological advances, data privacy, ethical governance, and sustainability must form the backbone of global-scale biosensing deployment.
9.5.1 Ethical, scalable, and secure data ecosystems
As wearable biosensors increasingly integrate with AI- and IoT-based health infrastructures, data privacy and ethical governance become pivotal. Beyond technical performance, sensor networks must comply with strict data protection frameworks such as the General Data Protection Regulation (GDPR) to ensure secure, anonymized data flow across interconnected systems. Recent frameworks emphasize GDPR-compliant data handling and federated learning models that preserve user privacy while enabling large-scale analytics (Zaguir et al., 2024).
Moreover, the long-term sustainability and scalability of IoT-enabled healthcare demand circular design principles—reducing electronic waste, optimizing energy use, and enabling device recyclability through modular manufacturing (Brauneck et al., 2023; Saha et al., 2025). Integrating these ethical, environmental, and economic considerations into next-generation biosensing platforms will be critical for achieving global deployment, clinical standardization, and equitable healthcare access.
9.6 Wireless data transmission technologies in wearable sensors
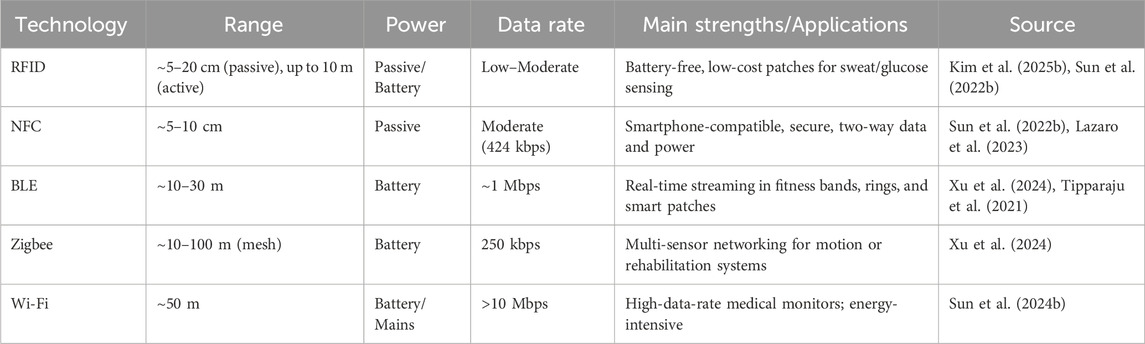
Modern wearable biosensors depend on seamless, low-power wireless communication. Among available options, Radio-Frequency Identification (RFID) remains the foundation for passive, battery-free data exchange (Kim C. et al., 2025). Yet, new standards—Near-Field Communication (NFC), Bluetooth Low Energy (BLE), and Zigbee—are extending range, bandwidth, and network flexibility (Sun et al., 2022a; Kong et al., 2024). Table 3 shows wireless technology used for body fluids sensors.
RFID and NFC: RFID enables wireless data transfer through electromagnetic coupling between a reader and tag. Passive RFID tags, powered directly by the reader field, are ideal for thin, battery-free patches that monitor biomarkers such as sweat pH or glucose. NFC, operating at 13.56 MHz, is essentially a short-range, two-way evolution of RFID. It allows a smartphone to both energize and read a wearable patch—for example, an NFC-based cortisol sensor scanned directly on skin (Sun et al., 2022a). These passive methods combine high security, simplicity, and comfort but are limited to a few centimeters.
BLE and Zigbee: BLE dominates consumer-grade wearables such as smart rings and watches. Operating at 2.4 GHz, it streams data continuously over 10–30 m with minimal power use, ideal for real-time heart rate or motion monitoring (Kong et al., 2024). Zigbee, by contrast, forms low-power mesh networks, linking multiple sensors into coordinated “body-area networks.” Though slower in data rate, its multi-node scalability suits rehabilitation suits and motion-tracking systems.
Rather than competing, these protocols now complement each other. RFID/NFC excel at short-range, battery-free sensing; BLE extends continuous real-time streaming; Zigbee enables distributed, multi-sensor intelligence. Hybrid systems—such as an NFC-powered patch that uploads data via BLE—illustrate the shift toward context-adaptive, connected biosensing (Kong et al., 2024). Future designs will merge these technologies to balance energy efficiency, user comfort, and data richness.
9.7 Integration of artificial intelligence (AI) and IoT: real-world applications
The convergence of Artificial Intelligence (AI) and the Internet of Things (IoT) with wearable biosensors for body-fluid analysis (sweat, interstitial fluid, tears, saliva) is reshaping precision diagnostics from passive monitoring to adaptive, predictive intelligence. Modern AI algorithms—particularly machine and deep learning—enhance signal reliability by filtering motion artifacts, baseline drift, and environmental noise inherent in body-fluid sensing. They enable multimodal data fusion across electrochemical, optical, and mechanical domains, extracting hidden biomarkers and physiological trends that traditional threshold-based methods miss (Kargarandehkordi et al., 2025; Zhang et al., 2023). For instance, AI-driven wearable networks have been shown to integrate sweat electrolyte data and motion cues to detect fatigue or dehydration with higher accuracy than linear statistical models (Huang et al., 2025).
IoT connectivity complements this intelligence by establishing continuous feedback loops between sensors, cloud servers, and clinicians, transforming single-use wearables into components of distributed health networks. Modular architectures combining local edge analytics with cloud computing minimize latency and energy consumption while maintaining scalability (Hosain et al., 2024). Real-world trials have demonstrated that IoT-enabled biosensors can remotely track hydration state, glucose variability, and renal stress in near real time, with automated alerts improving clinical responsiveness (Luo et al., 2024).
Despite this progress, the union of AI and IoT in chemical biosensing faces persistent challenges: limited generalizability of data-driven models across users and environments, energy constraints of on-device inference, and vulnerability of networked systems to synchronization failures and security breaches. Moreover, “black-box” algorithms undermine interpretability—an essential factor for clinical trust. Addressing these issues requires hybrid strategies coupling physics-based priors with data-driven learning, continual recalibration to correct for sensor drift, and on-device edge intelligence that ensures privacy and real-time decision-making. When properly integrated, AI supplies cognition and IoT provides communication—together enabling context-aware, connected biosensing. Yet, the impact of this synergy ultimately depends on sensor fidelity, calibration integrity, and algorithmic transparency. As research shifts toward scalable printed electronics, self-calibrating models, and secure, low-power networking, body-fluid biosensors are poised to evolve from passive monitors into intelligent companions for personalized healthcare.
10 Conclusion and future directions
Wearable sensors for body fluid monitoring have significantly transformed healthcare, sports science, and occupational health by providing real-time, non-invasive access to critical physiological data. These devices bridge the gap between traditional laboratory diagnostics and continuous, on-the-go health monitoring. Body fluids such as blood, sweat, saliva, and urine serve as rich sources of biomarkers, offering insights into metabolic health, hydration, and overall wellbeing.
The integration of advanced materials like PDMS, TPU, and graphene, along with manufacturing techniques such as 3D printing and stereolithography, has been instrumental in advancing wearable sensor technology. These innovations have enabled the development of flexible, durable, and sensitive devices that can adapt to various applications. Wearable sensors have demonstrated significant utility in domains ranging from sports science—where they optimize performance and recovery—to healthcare, where they monitor chronic conditions and support early disease detection. Furthermore, they play a crucial role in ensuring safety and efficiency in high-risk professions by tracking hydration and stress levels.
Despite these advancements, wearable sensor technology faces challenges that limit widespread adoption. Ensuring accuracy and reliability across diverse environments and skin types remains a technical hurdle. Power supply limitations and concerns over data security in wireless communication systems like RFID and Bluetooth also need to be addressed. Additionally, while advanced manufacturing methods have improved affordability, scaling production to meet global demand efficiently requires further innovation.
An increasing emphasis on environmental sustainability is reshaping the development of wearable sensors. The adoption of biodegradable and recyclable materials, such as cellulose-based substrates and graphene, addresses environmental concerns while ensuring long-term viability. Energy harvesting technologies, like triboelectric nanogenerators (TENGs), promise self-powered sensors that minimize reliance on disposable batteries, reducing electronic waste. These advancements align with global efforts to promote a circular economy and environmentally conscious technology (Zhang et al., 2025).
Artificial intelligence (AI) has emerged as a transformative force in wearable sensor technology, enabling smarter and more efficient health monitoring. AI algorithms can process vast amounts of sensor data in real time, providing actionable insights and predictive analytics. For instance, machine learning models can analyze patterns in body fluid biomarkers to predict hydration status, detect early signs of metabolic disorders, or assess recovery rates in athletes. AI-powered wearable systems can personalize health recommendations by integrating contextual information such as user activity levels, environmental conditions, and historical health data. Moreover, advanced AI techniques like deep learning enhance sensor calibration and fault detection, improving reliability and reducing errors caused by environmental or usage variability.
Moving forward, interdisciplinary collaboration among materials scientists, engineers, and healthcare professionals will play a pivotal role in driving sustainability. Sustainable innovations not only enhance device functionality but also contribute to user experience by increasing durability and facilitating end-of-life recycling. However, challenges remain in balancing production costs, supply chain logistics, and consumer awareness to achieve widespread adoption of eco-friendly materials and energy-efficient designs. Moreover, sustainable manufacturing practices can help scale production efficiently to meet growing global demand, ensuring wearable sensors contribute positively to both health outcomes and environmental preservation.
In the long term, wearable sensors are expected to evolve into fully integrated systems capable of monitoring multiple biomarkers, communicating with healthcare providers, and delivering therapeutic interventions. These systems will play a pivotal role in the transition toward personalized and preventive medicine, empowering individuals to take proactive control of their health. By addressing existing challenges, embracing sustainable materials, and integrating cutting-edge innovations, wearable sensors have the potential to revolutionize health monitoring, creating a healthier, more connected, and environmentally responsible future.
Author contributions
EG: Methodology, Supervision, Conceptualization, Formal analysis, Project administration, Investigation, Software, Funding acquisition, Resources, Writing – review and editing. SA: Methodology, Supervision, Conceptualization, Formal analysis, Project administration, Validation, Investigation, Software, Funding acquisition, Resources, Visualization, Writing – original draft, Writing – review and editing. AT: Data curation, Methodology, Supervision, Conceptualization, Formal analysis, Project administration, Validation, Investigation, Software, Funding acquisition, Resources, Visualization, Writing – original draft, Writing – review and editing. TT: Conceptualization, Formal analysis, Project administration, Validation, Investigation, Software, Funding acquisition, Resources, Visualization, Data curation, Methodology, Supervision, Writing – original draft, Writing – review and editing. ES: Data curation, Methodology, Supervision, Conceptualization, Formal analysis, Project administration, Validation, Investigation, Software, Funding acquisition, Resources, Writing – original draft, Writing – review and editing. MS: Conceptualization, Formal analysis, Project administration, Validation, Investigation, Software, Funding acquisition, Resources, Visualization, Data curation, Methodology, Supervision, Writing – original draft, Writing – review and editing. NBK: Conceptualization, Formal analysis, Project administration, Validation, Investigation, Software, Funding acquisition, Resources, Visualization, Data curation, Methodology, Writing – original draft, Writing – review and editing.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Alizadeh, A., Burns, A., Lenigk, R., Gettings, R., Ashe, J., Porter, A., et al. (2018). A wearable patch for continuous monitoring of sweat electrolytes during exertion. Lab a Chip 18 (17), 2632–2641. doi:10.1039/c8lc00510a
Alkozi, H. A., Alhudhayf, H. A., Alawad, N. M. A., et al. (2024). Cortisol levels in tears as a stress biomarker. J. Multidiscip. Healthc. 17, 217–226. doi:10.2147/JMDH.S488956
Anchidin-Norocel, L., Savage, W. K., Nemțoi, A., Dimian, M., and Cobuz, C. (2024). Recent progress in saliva-based sensors for continuous monitoring of heavy metal levels linked with diabetes and obesity. Chemosensors 12 (12), 269. doi:10.3390/chemosensors12120269
Anshin, S. M., Biryukov, V. P., Gradov, O., and Prince, A. N. (2024). Thermoplastic polyurethane (TPU) and nitrile Butadiene Rubbers (NBR) as promising materials for elastomeric microfluidics and cryomicrofluidics. Adv. Transdiscipl. Eng. doi:10.3233/atde240639
Aroche, A. F., Nissan, H. E., and Daniele, M. A. (2024). Hydrogel-forming microneedles and applications in interstitial fluid diagnostic devices. Adv. Healthc. Mater. 14, 2401782. doi:10.1002/adhm.202401782
Ates, H. C., Brunauer, A., von Stetten, F., Urban, G. A., Güder, F., Merkoçi, A., et al. (2021). Integrated devices for non-invasive diagnostics. Adv. Funct. Mater. 31 (15), 2010388. doi:10.1002/adfm.202010388
Ates, H. C., Nguyen, P. Q., Gonzalez-Macia, L., Morales-Narváez, E., Güder, F., Collins, J. J., et al. (2022). End-to-end design of wearable sensors. Nat. Rev. Mater 7, 887–907. doi:10.1038/s41578-022-00460-x
Atsani Susanto, R. T., Patel, B., Hsiao, Y. S., Tseng, H. Y., and Lin, P. T. (2023). Design optimization of a 3D microfluidic channel system for biomedical applications. 3D Print. Addit. Manuf. 11, e2075–e2088. doi:10.1089/3dp.2023.0169
Baker, L. B. (2017). Sweating rate and sweat sodium concentration in athletes: a review of methodology and intra/interindividual variability. Sports Med. 47 (Suppl. 1), 111–128. doi:10.1007/s40279-017-0691-5
Baker, L. B. (2019). Physiology of sweat gland function: the roles of sweating and sweat composition in human health. Temp. (Austin). 6 (3), 211–259. doi:10.1080/23328940.2019.1632145
Balasubramanian, V., Niknahad, F., and Zarifi, M. H. (2024). “Wireless real time sweat secretion monitoring using waveguide-based wearable sensor,” in 2024 IEEE MTT-S International Microwave Biomedical Conference (IMBioC), Montreal, QC, Canada, 11-13 June 2024, 81–83. doi:10.1109/imbioc60287.2024.10590247
Bandodkar, A. J., Jeang, W. J., Ghaffari, R., and Rogers, J. A. (2019). Wearable sensors for biochemical sweat analysis. Annu. Rev. Anal. Chem. 12 (1), 1–22. doi:10.1146/annurev-anchem-061318-114910
Belabbaci, N. A., Anaadumba, R., and Alam, M. A. U. (2025). Recent advancements in wearable hydration-monitoring technologies: scoping review of sensors, trends, and future directions. JMIR Mhealth Uhealth 13, e60569. doi:10.2196/60569
Brauneck, A., Schmalhorst, L., Kazemi Majdabadi, M., Bakhtiari, M., Völker, U., Baumbach, J., et al. (2023). Federated machine learning, privacy-enhancing technologies, and data protection laws in medical research: scoping review. J. Med. Internet Res. 25, e41588. doi:10.2196/41588
Bron, A. J., Abelson, M. B., Ousler, G., Pearce, E., Tomlinson, A., Yokoi, N., et al. (2007). Methodologies to diagnose and monitor dry eye disease: report of the diagnostic methodology subcommittee of the international dry eye WorkShop (2007). Ocul. Surf. 5 (2), 108–152. doi:10.1016/s1542-0124(12)70083-6
Cao, Z., Huo, X., Ma, Q., Song, J., Pan, Q., Chen, L., et al. (2023). TFT-CN/P3HT blending active layer based two-component organic field-effect transistor for improved H2S gas detection. Sensors Actuators B Chem. 385, 133685. doi:10.1016/j.snb.2023.133685
Chandradas, S., Adarsh, S., Pillai, S., Achu, C., Manoj, R., Varma, S., et al. (2024). All-printed wearable biosensor based on MWCNT-Iron oxide nanocomposite ink for physiological level detection of glucose in human sweat. Biosens. Bioelectron. 258, 116358. doi:10.1016/j.bios.2024.116358
Chao, B., Manpreet, K., and Woo, S. (2019). Toward a highly selective artificial saliva sensor using printed hybrid field effect transistors. Sensors Actuators B-chemical 285, 186–192. doi:10.1016/J.SNB.2019.01.062
Childs, A., Mayol, B., Lasalde-Ramírez, J. A., Childs, A., Childs, A., Childs, A., et al. (2024). Diving into sweat: advances, challenges, and future of sweat-based wearable sensors. ACS Nano 18 (1), 125–150. doi:10.1021/acsnano.4c10344
Choi, E. H., and Fluhr, J. W. (2024). Importance of stratum corneum acidification to restore healthy skin barrier function. Ann. Dermatology. doi:10.5021/ad.23.078
Choi, J., Ghaffari, R., Baker, L. B., and Rogers, J. A. (2021). Skin-interfaced systems for sweat collection and analytics. Sci. Adv. 7 (1), eabe3929. doi:10.1126/sciadv.abe3929
Chojnowska, S., Ptaszyńska-Sarosiek, I., Kępka, A., Knaś, M., and Waszkiewicz, N. (2021). Salivary biomarkers of stress, anxiety and depression. J. Clin. Med. 10 (3), 517. doi:10.3390/jcm10030517
Chua, B., Desai, S. P., Tierney, M. J., Tamada, J. A., and Jina, A. N. (2013). Effect of microneedles shape on skin penetration and minimally invasive continuous glucose monitoring in vivo. Sensors Actuators A Phys. 203, 373–381. doi:10.1016/j.sna.2013.09.026
Cristea, A.-G., Lisă, E. L., Ciobotaru, S. I., Dragostin, I., Ștefan, C. S., Fulga, I., et al. (2025). Antimicrobial smart dressings for combating antibiotic resistance in wound care. Pharmaceuticals 18 (6), 825. doi:10.3390/ph18060825
Deulkar, P., Singam, A., Mudiganti, V. S., and Jain, A. (2024). Lactate monitoring in intensive care: a comprehensive review of its utility and interpretation. Cureus 16 (8), e66356. doi:10.7759/cureus.66356
Dominiczak, J., Krzemiński, J., Wojcieszek, J., Baraniecki, D., Budny, F., Wojciechowska, I., et al. (2024). Aerosol-jet-printed potentiometric pH sensor for sweat measurements in smart patches. Sens. Bio-Sensing Res. 43, 100636. doi:10.1016/j.sbsr.2024.100636
Dsouza, A., Constantinidou, C., Arvanitis, T. N., Haddleton, D. M., Charmet, J., and Hand, R. A. (2022). Multifunctional composite hydrogels for bacterial capture, growth/elimination, and sensing applications. ACS Appl. Mater Interfaces 14, 47323–47344. doi:10.1021/acsami.2c08582
Fakhr, M. H., Carrasco, I. L., Belyaev, D., Kang, J., Shin, Y., Yeo, J. S., et al. (2024). Recent advances in wearable electrochemical biosensors towards technological and material aspects. Biosens. Bioelectron. X 19, 100503. doi:10.1016/j.biosx.2024.100503
Fiore, M., Bianconi, A., Sicari, G., Conni, A., Lenzi, J., Tomaiuolo, G., et al. (2024). The use of smart rings in health monitoring—A meta-analysis. Appl. Sci. 14 (23), 10778. doi:10.3390/app142310778
Gallagher, M., Wysocki, C. J., Leyden, J. J., Spielman, A. I., Sun, X., and Preti, G. (2008). Analyses of volatile organic compounds from human skin. Br. J. Dermatol 159 (4), 780–791. doi:10.1111/j.1365-2133.2008.08748.x
Gao, W., Brooks, G., and Wang, J. (2021). Advances in additive manufacturing for wearable sensors. Nano Today 40, 101215. doi:10.1016/j.nantod.2021.101215
Gao, F., Liu, C., Zhang, L., Liu, T., Wang, Z., Song, Z., et al. (2023). Wearable and flexible electrochemical sensors for sweat analysis: a review. Microsyst. Nanoeng. 9, 1. doi:10.1038/s41378-022-00443-6
Gao, Q., Fu, J., Xiong, F., Wang, J., Qin, Z., and Li, S. (2024). A multi-channel urine sensing detection system based on creatinine, uric acid, and pH. Biosensors 14 (10), 473. doi:10.3390/bios14100473
García-Carmona, L., Martín, A., Sempionatto, J. R., Moreto, J. R., González, M. C., Wang, J., et al. (2019). Pacifier biosensor: toward noninvasive saliva biomarker monitoring. Anal. Chem. 91 (21), 13883–13891. doi:10.1021/acs.analchem.9b03379
Ghadge, S., Marathe, A., Adivarekar, R., and More, S. (2024). Woolitmus: an approach to minimize E-waste by using wool-based wearable sensor for sweat pH detection. Fibers Polym. 25 (9), 3379–3386. doi:10.1007/s12221-024-00669-3
Gu, H., Sun, X., Bao, H., Feng, X., and Chen, Y. (2025). Optically pH-Sensing in smart wound dressings towards real-time monitoring of wound states: a review. Anal. Chim. Acta 1350, 343808. doi:10.1016/j.aca.2025.343808
Hao, Z., Xiao, H., Qiulin, W., Xinliang, L., Youming, Li., and Jinghui, Z. (2022). A green flexible and wearable biosensor based on carbon nanofibers for sensitive detection of uric acid in artificial urine. J. Mater. Chem. B 10, 8450–8461. doi:10.1039/d2tb01547a
Heikenfeld, J., Jajack, A., Rogers, J., Gutruf, P., Tian, L., Pan, T., et al. (2018). Wearable sensors: modalities, challenges, and prospects. Lab a Chip 18 (2), 217–248. doi:10.1039/c7lc00914c
Hiroki, O., Yuji, I., Tomonori, S., Kazuhisa, S., Kyohei, D., Yuta, S., et al. (2023). Anaerobic threshold using sweat lactate sensor under hypoxia. Dent. Sci. Rep. 13, 22865. doi:10.1038/s41598-023-49369-7
Hosain, M. N., Kwak, Y. S., Lee, J., Choi, H., Park, J., and Kim, J. (2024). IoT-enabled biosensors for real-time monitoring and early detection of chronic diseases. Phys. Activity Nutr. 28 (4), 060–069. doi:10.20463/pan.2024.0033
Hossain, N. I., Sharma, A., and Sonkusale, S. (2023). A wearable and multiplexed electrochemical sensor suite for real-time sweat ionic content and pH monitoring with IoT integration. IEEE sensors Lett. 7, 1–4. doi:10.1109/lsens.2023.3300827
Huang, C., Hao, Z., Wang, Z., Wang, H., Zhao, X., and Pan, Y. L. (2022). An ultraflexible and transparent graphene-based wearable sensor for biofluid biomarkers detection. Adv. Mater. Technol. 7 (6), 2101131. doi:10.1002/admt.202101131
Huang, G., Chen, X., and Liao, C. (2025). AI-Driven wearable bioelectronics in digital healthcare. Biosens. (Basel). 15 (7), 410. doi:10.3390/bios15070410
Iqbal, T., Elahi, A., Wijns, W., and Shahzad, A. (2023). Cortisol detection methods for stress monitoring in connected health. Health Sci. Rev. 6, 100079. doi:10.1016/j.hsr.2023.100079
Iqbal, S. M. A., Leavitt, M. A., Mahgoub, I., and Asghar, W. (2024). A wearable internet of things device for noninvasive remote monitoring of vital signs related to heart failure. IoT 5 (1), 155–167. doi:10.3390/iot5010008
Islam, T., and Washington, P. (2024). Non-invasive biosensing for healthcare using artificial intelligence: a semi-systematic review. Biosensors 14 (4), 183. doi:10.3390/bios14040183
Ismail, S. N. A., Nayan, N. A., Mohammad Haniff, M. A. S., Jaafar, R., and May, Z. (2023). Wearable two-dimensional nanomaterial-based flexible sensors for blood pressure monitoring: a review. Nanomaterials 13 (5), 852. doi:10.3390/nano13050852
Jarnda, K. V., Wang, D., Qurrat-Ul-Ain, , Anaman, R., Johnson, V. E., Johnson, V. E., et al. (2023). Recent advances in electrochemical non-enzymatic glucose sensor for the detection of glucose in tears and saliva: a review. Sensors Actuators A Phys. 363, 114778. doi:10.1016/j.sna.2023.114778
Jafleh, E. A., Alnaqbi, F. A., Almaeeni, H. A., Faqeeh, S., Alzaabi, M. A., and Al Zaman, K. (2024). The role of wearable devices in chronic disease monitoring and patient care: a comprehensive review. Cureus 16, e68921. doi:10.7759/cureus.68921
Jaiswal, J., Mizushima, S., Kanno, S., Sasaki, K., Tsuchiya, K., Kaneda-Nakashima, K., et al. (2024). Highly sensitive on-skin flexible Sb2O3/Sb thin film pH sensor for real-time sweat sensing. Mater. Res. Bull. 175, 112795. doi:10.1016/j.materresbull.2024.112795
Jamshidnejad-Tosaramandani, T., Kashanian, S., Omidfar, K., and Schiöth, H. B. (2025). The role of nanomaterials in the wearable electrochemical glucose biosensors for diabetes management. Biosensors 15 (7), 451. doi:10.3390/bios15070451
Jonathan, F., Giorgia, S., and Ferdinando, F. (2023). “Lactate: anaerobic threshold and new discoveries,” in Technology in sports - recent advances, new perspectives and application. doi:10.5772/intechopen.1003067
Karachaliou, C. E., Koukouvinos, G., Goustouridis, D., Raptis, I., Kakabakos, S., Petrou, P., et al. (2023). Cortisol immunosensors: a literature review. Biosensors 13, 285. doi:10.3390/bios13020285
Kargarandehkordi, A., Li, S., Lin, K., Phillips, K. T., Benzo, R. M., and Washington, P. (2025). Fusing wearable biosensors with artificial intelligence for mental health monitoring: a systematic review. Biosensors 15 (4), 202. doi:10.3390/bios15040202
Kelly, F., Vivian, Li., Michelle, S., Julia, V.-B., and Grace, H. (2024). The role of wearable technology in real-time skin health monitoring. JEADV Clin. Pract. 4, 21–29. doi:10.1002/jvc2.587
Kim, J., Sempionatto, J. R., Imani, S., Hartel, M. C., Barfidokht, A., Tang, G., et al. (2018). Simultaneous monitoring of sweat and interstitial fluid using a single wearable biosensor platform. Adv. Sci. 5 (10), 1800880. doi:10.1002/ADVS.201800880
Kim, J., Campbell, A. S., Esteban-Fernández de Ávila, B., and Wang, J. (2019). Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 37, 389–406. doi:10.1038/s41587-019-0045-y
Kim, K. H., Kim, J. T., Seo, S. E., Lee, H. G., and Kwon, O. S. (2025a). Immuno-graphene field-effect transistor-based cortisol bioelectronics using a novel cortisol antibody. BioChip J. 19, 194–203. doi:10.1007/s13206-024-00176-w
Kim, C., Lee, J., Jeong, E. Y., Jang, Y., Choi, B., Han, D., et al. (2025b). Wireless technologies for wearable electronics: a review. Adv. Electron. Mater. 11 (10), 2400884. doi:10.1002/aelm.202400884
Koh, A., Kang, D., Xue, Y., Lee, S., Pielak, R. M., Kim, J., et al. (2016). A soft, wearable microfluidic device for the capture, storage, and colorimetric sensing of sweat. Sci. Transl. Med. 8, 366ra165. doi:10.1126/scitranslmed.aaf2593
Kong, L., Li, W., Zhang, T., Ma, H., Cao, Y., Wang, K., et al. (2024). Wireless technologies in flexible and wearable sensing: from materials design, system integration to applications. Adv. Mater. 36 (27), 2400333. doi:10.1002/adma.202400333
Kristoffersson, A., and Lindén, M. (2022). A systematic review of wearable sensors for monitoring physical activity. Sensors 22 (2), 573. doi:10.3390/s22020573
Kumari, S., Samara, M., Ampadi Ramachandran, R., Gosh, S., George, H., Wang, R., et al. (2024). A review on saliva-based health diagnostics: biomarker selection and future directions. Biomed. Mater. and Devices 2, 121–138. doi:10.1007/s44174-023-00090-z
Lakshminrusimhan, C. A., Preethi, D. K. S., Sowmya, R., and Sivagurunathan, N. (2024). Salivary diagnostics in oral and systemic diseases - a review. Int. J. innovative Sci. Res. Technol., 2083–2087. doi:10.38124/ijisrt/ijisrt24jun1344
Lan, Z., Jian, Lu., En, T., and Sohei, M. (2023). Rapid manufacturing approach of an ultrathin moisture sensor for health monitoring. Sensors 23, 4262. doi:10.3390/s23094262
Lazaro, A., Villarino, R., Lazaro, M., Canellas, N., Prieto-Simon, B., and Girbau, D. (2023). Recent advances in batteryless NFC sensors for chemical sensing and biosensing. Biosensors 13 (8), 775. doi:10.3390/bios13080775
Lee, S. Y., Ciou, D. S., Lee, H.-Y., Chen, J.-Y., Wei, Y.-C., and Shieh, M.-D. (2024). Portable electrochemical system and platform with point-of-care determination of urine albumin-to-creatinine ratio to evaluate chronic kidney disease and cardiorenal syndrome. Biosensors 14 (10), 463. doi:10.3390/bios14100463
Lei, Y., Gong, Z., Li, Y., Zhang, J., Liu, Z., Sun, Z., et al. (2023). Highly sensitive physiological sensor based on tapered fiber-optic interferometer for sweat pH detection. IEEE Sensors J. 23 (11), 11627–11634. doi:10.1109/JSEN.2023.3266346
Li, X., Liu, Y., and Zhao, T. (2018). Wearable biosensors for urine analysis in clinical and athletic settings. Adv. Mater. 30 (18), 1706981. doi:10.1002/adma.201706981
Lim, H.-R., Lee, S. M., Park, S., Choi, C., Kim, H., Kim, J., et al. (2022). Smart bioelectronic pacifier for real-time continuous monitoring of salivary electrolytes. Biosens. and Bioelectron. 210, 114329. doi:10.1016/j.bios.2022.114329
Liu, Q., Zhang, Y., Wu, X., Zhao, Z., Dobrzyn, A., and Brzozka, Z. (2021). Islet-on-a-chip: biomimetic micropillar-based microfluidic system for three-dimensional pancreatic islet cell culture. Biosens. Bioelectron. 183, 113215. doi:10.1016/j.bios.2021.113215
Luo, X., Shi, W., Yu, H., Xie, Z., Li, K., and Cui, Y. (2018). Wearable carbon nanotube-based biosensors on gloves for lactate. Sensors (Basel) 18 (10), 3398. doi:10.3390/s18103398
Luo, X., Tan, H., and Wen, W. (2024). Recent advances in wearable healthcare devices: from material to application. Bioeng. (Basel) 11 (4), 358. doi:10.3390/bioengineering11040358
Luo, R., Yang, Y., Huang, S., and Huang, Z. (2025). Wearable devices for monitoring sweat glucose: an integrated strategy for efficient electrochemical sensors. Sensors Actuators Rep. 9, 100339. doi:10.1016/j.snr.2025.100339
Magdalena, K. (2024). Wearable devices for training and patient monitoring: a comprehensive review. Qual. Sport 29, 55667. doi:10.12775/qs.2024.29.55667
Mao, P., Li, H., and Yu, Z. (2023). A review of skin-wearable sensors for non-invasive health monitoring applications. Sensors 23 (7), 3673. doi:10.3390/s23073673
Marco, K. (2024). Flexible nanomaterial sensors for non-invasive health monitoring. Appl. Comput. Eng. 72, 45–58. doi:10.54254/2755-2721/72/20240981
Meltem, T., and Kursun, S. (2024). Development of wearable textile-based moisture sensor for smart diaper applications and examination of its comfort properties. J. Text. Inst. 116, 1666–1677. doi:10.1080/00405000.2024.2396161
Meng, J., Zahran, M., and Li, X. (2024). Metal–organic framework-based nanostructures for electrochemical sensing of sweat biomarkers. Biosensors 14 (10), 495. doi:10.3390/bios14100495
Min, J., Tu, J., Xu, C., Lukas, H., Shin, S., Yang, Y., et al. (2023). Skin-interfaced wearable sweat sensors for precision medicine. Chem. Rev. 123 (8), 5049–5138. doi:10.1021/acs.chemrev.2c00823
Mingrui, Lv., Xiujuan, Q., Yanxin, Li., Xianghua, Z., and Xiliang, L. (2023). A stretchable wearable sensor with dual working electrodes for reliable detection of uric acid in sweat. Anal. Chim. Acta. doi:10.1016/j.aca.2023.342154
Mishra, R. K., Hubble, L. J., Martín, A., Kumar, R., Barfidokht, A., Kim, J., et al. (2017). Wearable flexible and stretchable glove biosensor for On-Site detection of organophosphorus chemical threats. ACS Sens. 2 (4), 553–561. doi:10.1021/acssensors.7b00051
Moradifar, F., Sepahdoost, N., Tavakoli, P., and Mirzapoor, A. (2024). Multi-functional dressings for recovery and screenable treatment of wounds: a review. Heliyon 11 (1), e41465. doi:10.1016/j.heliyon.2024.e41465
Mordor Intelligence (2025a). Wearable devices in sports market size. Available online at: https://www.mordorintelligence.com/industry-reports/wearable-devices-in-sports-market.
Mordor Intelligence (2025b). Wearable health sensors market size. Available online at: https://www.mordorintelligence.com/industry-reports/global-wearable-health-sensors-market-industry.
Nguyen, T. T. H., Nguyen, C. M., Huynh, M. A., Trinh, Q. T., Tanner, P., Ingebrandt, S., et al. (2025). Ultra-sensitive aptameric field-effect transistor for cortisol sensing. Sensors Actuators Rep. 9, 100324. doi:10.1016/j.snr.2025.100324
Obaidur Rahman Khan, A. J. M., Mohaiminul Islam, S. A., Sarkar, A., Islam, T., Rakesh, P., and Bari, S. (2024). Real-time predictive health monitoring using Ai-driven wearable sensors: enhancing early detection and personalized interventions in chronic disease management. Int. J. Multidiscip. Res. doi:10.36948/ijfmr.2024.v06i05.28497
Opro9 (2025). Smart diaper sensor (product page). Available online at: https://www.opro9.com/smart-diaper/.
Pagar, V., Jagtap, S., Shaligram, A., and Bhadane, P. (2024). “Fabrication of metal oxide based thick film pH sensor and its application for sweat pH measurement,” in Intelligent systems. ICMIB 2023. Lecture notes in networks and systems. Editors S. K. Udgata, S. Sethi, and X. Z. Gao (Singapore: Springer), 728.
Pfaffe, T., Cooper-White, J., Beyerlein, P., Kostner, K., and Punyadeera, C. (2011). Diagnostic potential of saliva: current state and future applications. Clin. Chem. 57 (5), 675–687. doi:10.1373/clinchem.2010.153767
Potvin, R., Makari, S., and Rapuano, C. J. (2015). Tear film osmolarity and dry eye disease: a review of the literature. Clin. Ophthalmol. 9, 2039–2047. doi:10.2147/OPTH.S95242
Promphet, N., Ummartyotin, S., Ngeontae, W., Puthongkham, P., and Rodthongkum, N. (2021). Non-invasive wearable chemical sensors in real-life applications. Anal. Chim. Acta 1179, 338643. doi:10.1016/j.aca.2021.338643
Quan, X., Liu, J., Roxlo, T., Siddharth, S., Leong, W., Muir, A., et al. (2021). Advances in non-invasive blood pressure monitoring. Sensors 21 (13), 4273. doi:10.3390/s21134273
Rajan, A., Vishnu, J., and Shankar, B. (2024). Tear-based ocular wearable biosensors for human health monitoring. Biosensors 14 (10), 483. doi:10.3390/bios14100483
Ray, T. R., Choi, J., Bandodkar, A. J., and Rogers, J. A. (2021). Printing technologies for wearable sensors. Biosens. Bioelectron. 180, 113105. doi:10.1016/j.bios.2021.113105
Raymond, D. A., Praveen, K., and Palash, G. (2024). “Wearable electrochemical sensors for real-time biomarkers detection,” in 2024 Second International Conference on Intelligent Cyber Physical Systems and Internet of Things (ICoICI), Coimbatore, India, 28-30 August 2024. doi:10.1109/icoici62503.2024.10696798
Riente, A., Bianco, G. M., Arduini, F., Marrocco, G., and Occhiuzzi, C. (2023). An RFID sensor with microfluidics for monitoring the pH of sweat during sport activity. J. Sports Med. 12 (7), 123–135. doi:10.23919/EuCAP57121.2023.10133044
Ryland, M., Patrick, O., Eduard, B., Rocco, D. , M., and Alexandre, M. (2022). The immunological and hormonal responses to competitive match-play in elite soccer players. Int. J. Environ. Res. Public Health 19, 11784. doi:10.3390/ijerph191811784
Saha, K., Farhanj, Z., and Kumar, V. (2025). A systematic review of circular economy literature in healthcare: transitioning from a ‘Post-Waste’Approach to sustainability. J. Clean. Prod. 512, 145427. doi:10.1016/j.jclepro.2025.145427
Sandra, S. (2023). Towards accessible personal stress Mobile information displays informed by physiological stress data fusion. doi:10.17760/d20467216
Sathya, P., and Rajalakshmi, S. (2023). Smart non-invasive physiological parameter monitoring system using IoT and blynk app. J. Commun., 689–696. doi:10.12720/jcm.18.11.689-696
Seçkin, A. Ç., Ateş, B., and Seçkin, M. (2023). Review on wearable technology in sports: concepts, challenges and opportunities. Appl. Sci. 13 (18), 10399. doi:10.3390/app131810399
Sempionatto, J. R., and Wang, J. (2017). Wearable ring-based sensing platform for detecting analytes via electrochemistry. ACS Sensors. doi:10.1021/acssensors.7b00603
Shahzad, S., Iftikhar, F. J., Shah, A., Rehman, H. A., and Iwuoha, E. (2024). Novel interfaces for internet of wearable electrochemical sensors. RSC Adv. 14 (49), 36713–36732. doi:10.1039/d4ra07165d
Sheibani, S., Capua, L., Kamaei, S., Akbari, S. S. A., Zhang, J., Guerin, H., et al. (2021). Extended gate field-effect-transistor for sensing cortisol stress hormone. Commun. Mater 2, 10. doi:10.1038/s43246-020-00114-x
Sheng, X., Chunlei, M., Renhai, F., Xingwei, X., and Peng, J. (2022). Wireless glucose sensing system based on dual-tag RFID technology. IEEE Sensors J. 22, 13632–13639. doi:10.1109/JSEN.2022.3179498
Shi, X., Li, J., and Wang, H. (2023). Fluorescence-based sensors in contact lenses for ocular health monitoring. Adv. Funct. Mater. 33 (6), 230058.
Shi, X., Zhang, Y., and Lin, C. (2024). Real-time ocular biomarker detection using fluorescence technology. Biosens. Bioelectron. 177, 113768. doi:10.1016/j.bios.2024.113768
Shirreffs, S. M., and Maughan, R. J. (1998). Urine osmolality and conductivity as indices of hydration status in athletes in the heat. Med. and Sci. Sports and Exerc. 30 (11), 1598–1602. doi:10.1097/00005768-199811000-00007
Shitanda, I., Fujimura, Y., Takarada, T., Suzuki, R., Aikawa, T., Itagaki, M., et al. (2021). Self-powered diaper sensor with wireless transmitter. ACS Sensors 6 (11), 4212–4220. doi:10.1021/acssensors.1c01266
Sonner, Z., Wilder, E., Heikenfeld, J., Kasting, G., Beyette, F., Swaile, D., et al. (2015). The microfluidics of the eccrine sweat gland, including biomarker partitioning, transport, and biosensing implications. Biomicrofluidics 9 (3), 031301. doi:10.1063/1.4921039
Su, R., Wang, L., Han, F., Bian, S., Meng, F., Qi, W., et al. (2024). A highly stretchable smart dressing for wound infection monitoring and treatment. Mater. Today Bio 26, 101107. doi:10.1016/j.mtbio.2024.101107
Sultana, N., Dewey, H., and Budhathoki-Uprety, J. (2023). (second place poster award) development of optical nanosensors for pH measurements in model biofluids. Meet. Abstr., 1166. doi:10.1149/ma2023-0191166mtgabs
Sun, X., Zhao, C., Li, H., Yu, H., Zhang, J., Qiu, H., et al. (2022a). Wearable near-field communication sensors for healthcare. Sensors (Basel). Available online at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9146494/.
Sun, X., Zhao, C., Li, H., Yu, H., Zhang, J., Qiu, H., et al. (2022b). Wearable near-field communication sensors for healthcare: materials, fabrication and application. Micromachines 13 (5), 784. doi:10.3390/mi13050784
Sun, Y., Thakur, V. K., and Luo, R. (2024a). High mechanical strength, strong adhesion, self-healing and conductive ionic hydrogels for motion detection. Polymer 299, 126946. doi:10.1016/j.polymer.2024.126946
Sun, J., Bian, X., and Li, M. (2024b). Non-contact heart rate monitoring method based on Wi-Fi CSI signal. Sensors 24 (7), 2111. doi:10.3390/s24072111
Tang, J., He, Z., Zhang, M., Geng, W., Zhou, C., Shi, W., et al. (2025). τ-Ring: a smart ring platform for multimodal physiological and behavioral sensing. Available online at: https://arxiv.org/abs/2508.00778.
Tanweer, M., Monga, D. C., Gillan, L., Sepponen, R., Tanzer, I. O., and Halonen, K. A. (2024). Smart diaper with printed capacitive sensors and integrated front-end to monitor voided fluid volume. IEEE Sensors J. 24 (9), 14443–14453. doi:10.1109/JSEN.2024.3380890
Tekcin, M., Sayar, E., Yalcin, M. K., and Bahadir, S. K. (2022). Wearable and flexible humidity sensor integrated to disposable diapers for wetness monitoring and urinary incontinence. Electronics 11 (7), 1025. doi:10.3390/electronics11071025
Tingjun, C., Jing, P., Xinchuan, L., Na, C., Chenchen, Wu., Yu, D., et al. (2024). Anti-biofilm super-hydrophilic gel sensor for saliva glucose monitoring. Nano Today 55, 102141. doi:10.1016/j.nantod.2023.102141
Tipparaju, V. V., Mallires, K. R., Wang, D., Tsow, F., and Xian, X. (2021). Mitigation of data packet loss in bluetooth low energy-based wearable healthcare ecosystem. Biosensors 11 (10), 350. doi:10.3390/bios11100350
Tseng, P., Napier, B., Garbarini, L., Kaplan, D. L., and Omenetto, F. G. (2018). Functional, RF-trilayer sensors for tooth-mounted, wireless monitoring of the oral cavity and food consumption. Adv. Mater. 30 (18), 1703257. doi:10.1002/adma.201703257
UC San Diego Jacobs School of Engineering (2017). ‘Lab-on-a-glove’ could bring nerve-agent detection to a wearer’s fingertips. Available online at: https://jacobsschool.ucsd.edu/news/release/2159?id=2159.
Ungaro, C. T., Reimel, A. J., Nuccio, R. P., Barnes, K. A., Pahnke, M. D., and Baker, L. B. (2015). Non-invasive estimation of hydration status changes through tear fluid osmolarity during exercise and post-exercise rehydration. Eur. J. Appl. Physiology 115, 1165–1175. doi:10.1007/S00421-015-3099-7
Vishnupriya, S. D. (2024). Wearable health technology: integrating devices and health care systems for better monitoring. Indian Sci. J. Res. Eng. Manag. 08, 1–8. doi:10.55041/ijsrem21895
Vo, D. K., and Trinh, K. T. L. (2025). Advances in wearable biosensors for wound healing and infection monitoring. Biosensors 15 (3), 139. doi:10.3390/bios15030139
Wang, B., Zhao, C., Wang, Z., Yang, K. A., Cheng, X., Liu, W., et al. (2022). Wearable aptamer-field-effect transistor sensing system for noninvasive cortisol monitoring. Sci. Adv. 8, eabk0967. doi:10.1126/sciadv.abk0967
Wang, X., Cheng, J., and Wang, H. (2024a). Chronic wound management: a liquid diode-based smart bandage with ultrasensitive pH sensing ability. Microsyst. Nanoeng. 10, 193. doi:10.1038/s41378-024-00801-6
Wang, Z., Dong, Y., Sui, X., Shao, X., Li, K., Zhang, H., et al. (2024b). An artificial intelligence-assisted microfluidic colorimetric wearable sensor system for monitoring of key tear biomarkers. Npj Flex. Electron 8, 35. doi:10.1038/s41528-024-00321-3
Wardenaar, F. C. (2022). Human hydration indices: spot urine sample reference values for urine concentration markers in athletic populations. Dietetics 1 (1), 39–51. doi:10.3390/dietetics1010005
Wenfei, Yu. (2024). Application of flexible wearable sensors for revolution of personalized medicine in the age of digital health. Trans. Mater. Biotechnol. Life Sci. 4, 20–24. doi:10.62051/nzhrvy28
Wenze, Yu., Qiu, J. , Li., Zhi-Li, Z., Jixian, G., Zheng, Li., and Jianfei, Z. (2023). A yarn/fabric-based microfluidic patch for sweat sensing and monitoring. J. Text. Inst. 115, 2331–2342. doi:10.1080/00405000.2023.2288357
Wolf, M., Gulich, R., Lunkenheimer, P., and Loidl, A. (2011). Broadband dielectric spectroscopy on human blood. Biochimica Biophysica Acta (BBA)-General Subj. 1810 (8), 727–740. doi:10.1016/j.bbagen.2011.05.012
Wu, J. Y., Ching, C. T. S., Wang, H. M. D., and Liao, L. D. (2022). Emerging wearable biosensor technologies for stress monitoring and their real-world applications. Biosensors 12 (12), 1097. doi:10.3390/bios12121097
Xiangda, M., Xueqi, W., Wencan, L., Zijin, L., Fei, W., and Hui, Y. (2024). Large-scale wearable textile-based sweat sensor with high sensitivity, rapid response, and stable electrochemical performance. ACS Appl. Mater. and Interfaces 16, 18202–18212. doi:10.1021/acsami.4c01521
Xiong, F., Li, Y., Xie, C., Wang, Z., Zhou, J., Yang, H., et al. (2024). Research on dynamic urine volume detection system based on smart flexible textile sensors. Sci. Rep. 14, 22459. doi:10.1038/s41598-024-73179-0
Xu, Z., Zhao, H., and Zhang, J. (2024). Technologies and applications in wireless biosensors for health monitoring. J. Wirel. Netw. Appl., 1–20. doi:10.1007/s44258-024-00041-3
Xue, C., Yang, B., Fu, L., Hou, H., Qiang, J., Zhou, C., et al. (2023). Urine biomarkers can outperform serum biomarkers in certain diseases. Urine 3, 57–64. doi:10.1016/j.urine.2023.10.001
Xuecui, M., Liang, Z., Jiao, Y., Yingchun, L., and Li, Y. (2024). Wearable janus nanofiber membrane-based colorimetric sensors for directional transfer and analysis of sweat. Biomed. Anal. 1, 64–72. doi:10.1016/j.bioana.2024.02.002
Yang, Z., Zhao, Y., Zhang, H., Dong, X., Guo, X., Ye, L., et al. (2020). Asymmetrically noncovalently fused-ring acceptor for high-efficiency organic solar cells with reduced voltage loss and excellent thermal stability. Nano Energy 74, 104861. doi:10.1016/j.nanoen.2020.104861
Yang, C., Chen, Y., Liu, J., Liu, Z., and Chen, H. J. (2023). The trends in wound management: sensing, therapeutic treatment, and “theranostics”. J. Sci. Adv. Mat. Devices 8 (1), 100619. doi:10.1016/j.jsamd.2023.100619
Yeo, J. C., Tan, E. P., and Lim, C. T. (2015). Ion-selective electrodes for wearable tear analysis. Trends Anal. Chem. 68, 48–57.
Youssef, K., Ullah, A., Rezai, P., Hasan, A., and Amirfazli, A. (2023). Recent advances in biosensors for real time monitoring of pH, temperature, and oxygen in chronic wounds. Mater Today Bio 22, 100764. doi:10.1016/j.mtbio.2023.100764
Youyuan, X., Tong, Su., Ziyi, Mi., Ziyou, F., Yawen, H., Xiaoya, Hu., et al. (2023). Wearable electrochemical sensor based on bimetallic MOF coated CNT/PDMS film electrode via a dual-stamping method for real-time sweat glucose analysis. Anal. Chim. Acta 1278, 341754. doi:10.1016/j.aca.2023.341754
Yuan, Y., Liu, B., Li, H., Li, M., Song, Y., Wang, R., et al. (2022). Flexible wearable sensors in medical monitoring. Biosensors 12 (12), 1069. doi:10.3390/bios12121069
Yuhua, C., Yanghan, S., Yi, Li., Zhuo, W., Xue, P., Yuanke, He., et al. (2024). A wearable non-enzymatic sensor for continuous monitoring of glucose in human sweat. Talanta 278, 126499. doi:10.1016/j.talanta.2024.126499
Yun, S. M., Kim, M., Kwon, Y. W., Kim, H., Kim, M. J., Park, Y.-G., et al. (2021). Recent advances in wearable devices for non-invasive sensing. Appl. Sci. 11 (3), 1235. doi:10.3390/app11031235
Zaguir, N. A., de Magalhães, G. H., and de Mesquita Spinola, M. (2024). Challenges and enablers for GDPR compliance: systematic literature review and future research directions. IEEE Access 12, 81608–81630. doi:10.1109/ACCESS.2024.3406724
Zaryab, H., Sarah, A., Alsalhi, N., Drissi, S. A. M., Jawariya, S., Parwasha, T., et al. (2024). Graphene nanoplatelets-polyaniline composite for the detection of cortisol. J. Phys. Chem. Solids 191, 112031. doi:10.1016/j.jpcs.2024.112031
Zazoum, B., Batoo, K. M., and Khan, M. A. A. (2022). Recent advances in flexible sensors and their applications. Sensors 22 (12), 4653. doi:10.3390/s22124653
Zhang, Y., Hu, Y., Jiang, N., and Yetisen, A. K. (2023). Wearable artificial intelligence biosensor networks. Biosens. and Bioelectron. 219, 114825. doi:10.1016/j.bios.2022.114825
Zhang, S., He, Z., Zhao, W., Liu, C., Zhou, S., Ibrahim, O. O., et al. (2024). Innovative material-based wearable non-invasive electrochemical sweat sensors towards biomedical applications. Nanomaterials 14 (10), 857. doi:10.3390/nano14100857
Zhang, Q., Soham, D., Liang, Z., and Wan, J. (2025). Advances in wearable energy storage and harvesting systems. Med-X 3, 3. doi:10.1007/s44258-024-00048-w
Zhiqiang, Y., Zilong, G., Weixia, L., and Yuanyuan, L. (2024). “Flexible, breathable and sweatproof piezoelectric sensors based on additive manufacturing,” in 2024 6th International Conference on Electronic Engineering and Informatics (EEI), Chongqing, China, 28-30 June 2024. doi:10.1109/eei63073.2024.10696538
Zhou, Y. (2024). Advanced nanomaterials for electrochemical sensors: application to wearable tear glucose sensing technology. J. Mater. Chem. B 12 (4), 785–793. doi:10.1039/d4tb00790e
Keywords: wearable sensors, body fluid monitoring, non-invasive technologies, advanced materials, sports and healthcare applications
Citation: Gulgosteren E, Agrali Ermis S, Algin Toros A, Toros T, Serin E, Sekeroglu MO and Bahadır Kayisoglu N (2025) Sweat, tears, and beyond: advanced wearable sensors for personalized health and athletic performance. Front. Bioeng. Biotechnol. 13:1684674. doi: 10.3389/fbioe.2025.1684674
Received: 25 August 2025; Accepted: 27 October 2025;
Published: 28 November 2025.
Edited by:
Hamed Golmohammadi, Chemistry and Chemical Engineering Research Center, IranReviewed by:
Tao Chen, Qingdao University, ChinaKhibar Dashtian, Iran University of Science and Technology, Iran
Alessia Riente, Università cattolica del Sacro Cuore di Roma, Italy
Copyright © 2025 Gulgosteren, Agrali Ermis, Algin Toros, Toros, Serin, Sekeroglu and Bahadır Kayisoglu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Aydolu Algin Toros, YXlkb2x1YWxnaW5AaG90bWFpbC5jb20=
†ORCID: Erkan Gulgosteren, orcid.org/0000-0003-4642-4880; Sermin Agrali Ermis, orcid.org/0000-0002-9653-233X; Aydolu Algin Toros, orcid.org/0000 0003 0376 114X; Turhan Toros, orcid.org/0000 0002 8328 2925; Emre Serin, orcid.org/0000-0001-9596-2912; Mustafa Onder Sekeroglu, orcid.org/0000-0002-8868-507X; Numan Bahadir Kayisoglu, orcid.org/0000-0002-2003-7973
 Erkan Gulgosteren1†
Erkan Gulgosteren1† Sermin Agrali Ermis
Sermin Agrali Ermis Turhan Toros
Turhan Toros Emre Serin
Emre Serin