The Role of Dysbiosis in Critically Ill Patients With COVID-19 and Acute Respiratory Distress Syndrome
- 1Anesthesia and Intensive Care, Ospedale Policlinico San Martino, Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS) per l'Oncologia e le Neuroscienze, Genova, Italy
- 2Department of Medicine, University of Barcelona, Barcelona, Spain
- 3Department of Surgical Sciences and Integrated Diagnostics (DISC), Università degli Studi di Genova, Genova, Italy
- 4Department of Anesthesia and Intensive Care II, Clinical Emergency County Hospital of Cluj, Iuliu Hatieganu, University of Medicine and Pharmacy, Cluj-Napoca, Romania
- 5Anaesthesia and Intensive Care 1, Clinical Emergency County Hospital Cluj-Napoca, Cluj-Napoca, Romania
- 6Dietetics and Clinical Nutrition Unit, Ospedale Policlinico San Martino, Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS) per l'Oncologia e le Neuroscienze, Genova, Italy
- 7Clinica Malattie Infettive, Istituto di Ricovero e Cura a Carattere Scientifico (IRCCS) per l'Oncologia e le Neuroscienze, Genova, Italy
- 8Dipartimento di Scienze della Salute (DISSAL), Università degli Studi di Genova, Genova, Italy
- 9Division of Animal Experimentation, Department of Pulmonology, Hospital Clinic, Barcelona, Spain
- 10Centro de Investigacion en Red de Enfermedades Respiratorias (CIBERES), Madrid, Spain
- 11Institut d'investigacions Biomediques August Pi i Sunyer (IDIBAPS), Barcelona, Spain
- 12Laboratory of Pulmonary Investigation, Carlos Chagas Filho Institute of Biophysics, Federal University of Rio de Janeiro, Rio de Janeiro, Brazil
- 13COVID-19-Network, Ministry of Science, Technology, Innovation and Communication, Brasilia, Brazil
In late December 2019, severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) quickly spread worldwide, and the syndrome it causes, coronavirus disease 2019 (COVID-19), has reached pandemic proportions. Around 30% of patients with COVID-19 experience severe respiratory distress and are admitted to the intensive care unit for comprehensive critical care. Patients with COVID-19 often present an enhanced immune response with a hyperinflammatory state characterized by a “cytokine storm,” which may reflect changes in the microbiota composition. Moreover, the evolution to acute respiratory distress syndrome (ARDS) may increase the severity of COVID-19 and related dysbiosis. During critical illness, the multitude of therapies administered, including antibiotics, sedatives, analgesics, body position, invasive mechanical ventilation, and nutritional support, may enhance the inflammatory response and alter the balance of patients' microbiota. This status of dysbiosis may lead to hyper vulnerability in patients and an inappropriate response to critical circumstances. In this context, the aim of our narrative review is to provide an overview of possible interaction between patients' microbiota dysbiosis and clinical status of severe COVID-19 with ARDS, taking into consideration the characteristic hyperinflammatory state of this condition, respiratory distress, and provide an overview on possible nutritional strategies for critically ill patients with COVID-19-ARDS.
Introduction
In late December 2019, a novel coronavirus able to induce an acute respiratory syndrome was identified in Wuhan, China (1). This virus, since named severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), quickly spread worldwide, and the syndrome it causes, coronavirus disease 2019 (COVID-19), was declared a pandemic by the World Health Organization on March 11, 2020. The standard presentation of COVID-19 includes fever, dry cough, respiratory distress, fatigue, myalgia, dyspnea, headache, and diarrhea (1, 2). Fecal samples collected from patients with COVID-19 remained positive for about 11 days, raising concerns about the possible fecal-oral transmission of the virus and gastrointestinal symptoms (3, 4).
Several patients with COVID-19 are admitted to the intensive care unit (ICU) due to severe respiratory distress, with a clear status of typical or atypical acute respiratory distress syndrome (ARDS), requiring critical care support (2, 5). The rate of patients admitted to ICU depends on the diversity of cares within countries and the pandemic global situation. Indeed, compared with the first wave of the pandemic, during the second wave 50% less of all hospitalized patients with COVID-19 were admitted to the ICU (6). Traditional critical care includes respiratory and cardiovascular support, management of renal, hepatic, infectious, and neurologic status, and nutritional management (2). Patients with severe COVID-19 often experience an enhanced immune response with a hyperinflammatory state characterized by a “cytokine storm” (7), with fever and respiratory distress considered to represent increased dysbiosis. During critical illness, the multitude of therapies administered, including antibiotics, sedatives, analgesics, invasive mechanical ventilation, and nutritional support, may enhance the inflammatory response and impact on the patients' microbiota, leading to dysbiosis. In turn, this status may lead to hyper vulnerability in patients and an inappropriate response to critical circumstances (8).
In this context, the aim of our narrative review is to provide an overview of possible interaction between patients' microbiota dysbiosis and the clinical status of severe COVID-19 with ARDS, taking into consideration the characteristic hyperinflammatory state of this condition, respiratory distress, and provide an overview of possible nutritional strategies for critically ill patients with COVID-19-ARDS.
Pathophysiology of the Microbiota Gut-Lung Axes
The gut-lung axis is bidirectional, which means that metabolites derived from the gut or lung bacteria can affect each other. Gut microbiota is often altered as early as the first days of ICU admission (9), altering both susceptibility to and severity of infections (10). Mechanisms implied in microbiota-lung-gut-axis alteration in COVID-19 include: (1.1) Direct lung damage (1.2) ACE2 expression; (1.3) gut microbiota as lungs' defense against SARS-CoV-2; and (1.4) immune response.
Direct Lung Damage in COVID-19
ARDS is a common complication of COVID-19. After binding to angiotensin-converting enzyme-2 receptors (ACE2) and transmembrane protease serine 2 (TMPRSS2), SARS-CoV-2 enters the host cells and causes pneumonia with possible ARDS in the most severe cases. The histologic presentation of severe COVID-19 pneumonia includes a progressive thickening of the alveolar wall with infiltrates of mononuclear cells and macrophages, associated with endotheliitis (11). Edema, fibroblasts, and myofibroblasts thicken the alveolar septa, with interstitial inflammatory cell infiltrates. In the late and organizing stage, the lungs become consolidated, and fibroblasts and myofibroblasts proliferate and migrate, forming granulation tissue and fibrosis by accretion, with possible thickening of interlobular septum and bronchial walls, thus leading to diffuse alveolar damage (DAD) (11). In this state, patients with severe COVID-19 may need to be admitted to the ICU for endotracheal intubation and mechanical ventilation. The evolution to COVID-19-ARDS is characterized by pulmonary edema, hypoxemia and inflammation, which are associated with changes in the lung microbiome (12). The microbiota is defined as the overall community of microbes included in a population (13), and the genetic content of the microbiota is known as the microbiome. In healthy conditions, the composition of the microbiota is generally characterized by high abundance and diversity of microorganisms with preponderance of potentially beneficial species, a condition known as eubiosis (13).
The microbiota is primarily involved in the immune response and host defense against pathogens, as well as in gut maturation, nutrient uptake and metabolism, mucosal barrier function, intestinal motility, and modulation of the enteric nervous system (14). Moreover, mechanical ventilation, decreased bowel transit time, reduced oxygenation, multiple antibiotic usage, sedatives, analgesics, muscle relaxants, gastric protectors, and abnormal nutritional intake may affect the composition of microbiota, which may increase the risk of dysbiosis and inflammation (15–17). Mice treated with broad spectrum or targeted antibiotics impaired their response to systemic and respiratory infections (18). Most prominent among these are gram-negative bacteria (e.g., Proteobacteria), which can lead to severe gut-lung dysbiosis (9, 19).
ACE2 Expression
Once affected the lungs' tissue, COVID-19 may extend to other organs by binding to ACE2 and TMPRSS2 (20, 21), which are broadly expressed in various tissues (22, 23). ACE2 are involved in the regulation of inflammation and microbial community, while regulating the host intestinal metabolism of tryptophan, which plays a key role in the composition of gut microbiota (24–26). Thus, ACE2 expression may alter both the lung and gut microbiomes in certain disease conditions (24–26). In fact, a down-regulation of ACE2 reduces the absorption of tryptophan in the gut, while reducing the secretion of antimicrobial peptides and triggering dysbiosis (27). Bacterioides dorei and other bacterial species down-regulate the expression of colonic ACE2, thus supporting the appearance of intestinal symptoms in some COVID-19 patients (28). SARS-CoV-2 infection of the intestinal tract impairs the absorption of nutrients altering the intestinal function and activation of the enteric nervous system, causing gastrointestinal manifestations (29). Recent findings confirmed the role of gut dysbiosis in the induction of ARDS and its importance in possibly determining tissue damage in SARS-CoV-2 infection (16, 30).
Gut Microbiota as Lungs' Defense Against SARS-CoV-2
The gut microbiota regulates the function of the immune system and cellular homeostasis of both gut and lung tissues due to antimicrobial peptides and metabolites derived from intestinal commensals (18, 31). The enteric nervous system is composed of the myenteric plexuses, which control fluid movement and intestinal motility; and is influenced by the activation pattern recognition receptors (PRRs), especially toll-like receptors (TLRs) which recognize pathogens (32).
SARS-CoV-2 infection may promote intestinal inflammation, epithelial barrier disruption, and decreased production of antimicrobial peptides (AMPs), until developing secondary enteric infections. An increased inflammatory status of the gut induced by SARS-CoV-2 may alter gut permeability causing epithelial leakage, which may enhance bacterial translocation and trigger systemic inflammation and inflammatory response to other organs (22). Additionally, over-expression of fecal calprotectin is implied in gut inflammation in COVID-19 (33). The passage of gut bacteria from the intestinal to the lung tissues is regulated by the ability of gut tight junctions in maintaining epithelial permeability, and intestinal bacteria in preserving the intestinal barrier. Among the proposed mechanisms of alteration, products of commensal bacteria fermentation like butyrate and other short-chain-fatty-acids (SCFAs) are responsible of intestinal barrier function and regulation of tight junctions' permeability (34). Additionally, the alteration in the secretion of mucin by goblet cells can lead to impaired mucus layer, increasing susceptibility to increased gut permeability (35). Dysbiosis results in diminished production of microbial-associated molecular patterns including TLRs and nucleotide oligomerization domain (NOD)-like agonists and microbial metabolites such as SCFAs, thus reducing antibacterial pulmonary immunity (18). Hence, by altering the gut immune homeostasis, respiratory viral superinfection may occur. Gut bacteria were found in lung microbiome, suggesting possible translocation from the intestinal tract to the lungs via the hematic circulation (36). The abundance of gut-associated pathogens in the lungs is increased in non-COVID-19 ARDS patients, but little is known regarding ARDS associated with COVID-19. Patients with ARDS revealed a higher prevalence of Lechnospiraceae as a strong predictor of reduced survival (37). Some authors investigated the lung bacterial burden and diversity of patients with non-COVID-19 ARDS, concluding that the lung microbiota is reduced in term of diversity and is increased in terms of bacterial burden (38).
Immune Response
The multifunctional SARS-CoV-2 Envelope (E) protein, which interact with host tight junction protein ZO1, showed great contribution to virus assembly, while contributing to epithelial barrier damage, pathogenesis (binding to ACE2 receptor), and disease severity (39). Human intestinal epithelial cells (in the esophageal upper epithelia, gland cells, enterocytes of the ileum and colon) are potential target of viral replication, also promoting spreading of SARS-CoV-2 and immune response mediated by type III interferon (IFN) (40, 41). At lungs level, studies have highlighted the impact of gut microbiota on the lungs' production of type I IFN, which is implied in the control of viral infections (42, 43). Microbial metabolites such as desaminotyrosine and SCFAs are critical for microbiota homeostasis. For example, significant changes in the composition of gut microbiota have been identified in an experimental model of pulmonary influenza (44). Desaminotyrosine is produced by an anaerobe clostridium called Clostridium orbiscindens to protect the lungs and activate the innate immune response passing through the blood system against influenza infection. Desaminotyrosine implements type I IFN signaling of lungs' phagocytes by promoting genes transcription (45). Similarly, SCFAs result important in priming the pulmonary immune innate system (18, 31). The subsequent inflammatory response can promote and encourage local inflammation followed by endothelial and epithelial injury (Figure 1).
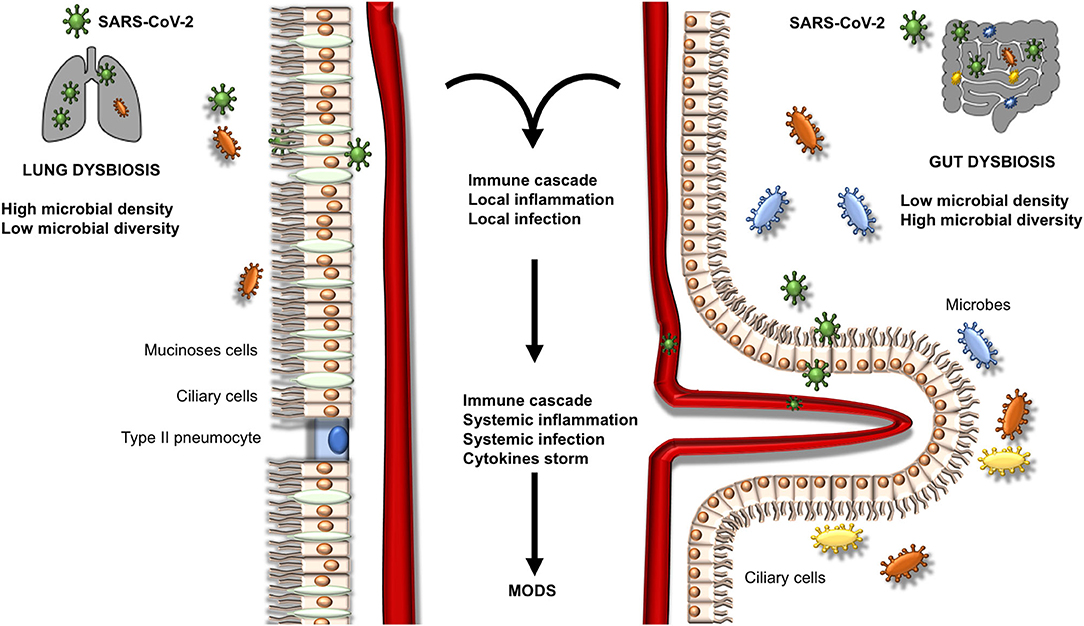
Figure 1. Mechanisms of microbiota gut-lung axis dysbiosis. This figure represents the possible evolution of dysbiosis in the lungs and intestine. A local inflammatory process is activated, thus converting in a systemic inflammatory process with possible infection and multiorgan disease syndrome (MODS).
The inflammatory response of SARS-CoV-2 infection is very complex. In fact, SARS-CoV-2 may interfere with the innate antiviral immune response that is made up two different antiviral pathways.
Phagocytes are recruited to fight against local infections and to repair and regenerate the epithelium. As aforementioned, the manipulation of cytokines and IFNs may play an important role in the prevention of infections and mucosal protection. Particularly, IL-22 and IFN-λ act as mucosal defenders and upregulate antimicrobial peptides (46). The IFN regulatory factors increase transcription of type I and III IFNs, which stimulate natural killer cells and cluster differentiation (CD)8+ T lymphocytes, whereas the nuclear factor-kB (NF-kB) promotes the activation of monocytes and their differentiation into macrophages (type M1). Cytokines are therefore released, and T-cells activated (inflammatory T-cells Th1 and Th17). Notably, a “cytokine storm” appears to occur in cases of severe COVID-19, as demonstrated by increased levels of interleukin (IL)-2, IL-17, granulocyte-colony stimulation factor, IFN-γ, inducible protein 10, monocyte chemoattractant protein-1, macrophage inflammatory protein 1-α, and tumor necrosis factor-α (7). Previous studies found that IL-22 is substantially expressed during viral infections, and animals with deficiency of IL-22 were unable to repair and regenerate epithelial tissues (47). Moreover, IL-22 usually enhance the recruitment of other inflammatory cells, thus amplifying the systemic inflammatory response (46, 48), which along with the local damage may predispose COVID-19 patients to secondary bacterial infections, capillary leakage syndrome, and systemic bacterial translocation (49), thus enhancing the risk of multiple organ damage (20–22). Nevertheless, in the COVID-19 era the role of cytokines and interferons on epithelial integrity and systemic reaction is still not clear, and IL-22 and IFN-λ might be considered as further promising targets to maintain the COVID-19 lungs' integrity, but more evidences are urgently needed (50). This exaggerates cytokines and interleukins release may increase the expression of markers like programmed death-1, T-cell immunoglobulin, mucin domain-containing protein-3 while favoring lymphocyte apoptosis and necrosis. Lymphopenia is frequent and is associated with disease severity and inflammation (51). Lymphopenia in COVID-19 may be induced by several mechanisms, including direct infection of lymphocytes, viral aggression of lymphatic organs, or continuous inflammation with cytokines release that could induce lymphocyte deficiency (52–54). Additionally, lymphopenia may be associated with glucocorticoids treatment (51). Since gut microbiota is one of the key components of the host immune system, and primary responses to infections and other immune insults, lymphopenia due to SARS-CoV-2 infection may interfere and predispose to changes in the normal flora by opportunistic germs (52–55).
Composition of the Gut-Lung Microbiota in Cases of Severe Covid-19 Pneumonia
The gut represents the largest microbial environment in humans. The healthy (eubiotic) intestinal microbiota represents a highly heterogeneous ecosystem including eukaryotic organisms (including Yeast), Virus, Archaea and Bacteria. The latters are the most represented members and include nine different phyla, of which Bacteroidetes and Firmicutes represent the most common populations (13, 32). Differently from the gut, the microbiota is scarcely represented in the lung, being mainly associated with mucosal surfaces. The gut microbiota of patients with COVID-19 demonstrated a high prevalence of opportunistic pathogens over commensals that persisted after negative swabs and resolution of respiratory symptoms. The abundance of Coprobacillus, Clostridium ramosum, and Clostridium hathewayi correlated with the severity of COVID-19 (33). Another study confirmed a high prevalence of opportunistic pathogens in patients with COVID-19, including Streptococcus, Rothia, Veillonella, and Actinomyces with a reduced relative abundance of symbionts (56).
The high prevalence of gastrointestinal disorders associated with acute infection by SARS-CoV-2 (anorexia, dysgeusia, ageusia, diarrhea, nausea, and hematemesis) (57) might be associated with the damage to the intestinal ecosystem that may be modified (58). In fact, SARS-CoV-2 infection can impact on some tight junction proteins (like PALS1), that compose the intestinal and lung epithelium. However, definitive confirmation on the impact of SARS-CoV-2 on tight junctions and intestinal permeability while potentially damaging to enterocytes are still limited and warrants further molecular researches (59). Gastrointestinal symptoms have been also associated with reduced number of circulating lymphocytes, and the circulating lymphocytes were inversely associated with virus discharge in stool (58). Moreover, a recent meta-analysis on gastrointestinal symptoms of SARS-CoV-2 infection concluded that gastrointestinal symptoms on admission were associated more with complications, including ARDS, acute cardiac injury, and acute kidney injury (57).
The main difference between gut and lung microenvironments is the higher turnover of lung bacteria with regard to the gut counterpart. This characteristic of the lung microbiota is due to the high rate of immigrated and eliminated pathogens (aspiration and mucosal dispersion vs. cough and muco-ciliary clearance). The gut microbiota, which is usually enriched in nutrients, makes tough competition with dense resident communities. In contrast, the lung microbiota is enriched in pharyngeal microbes (60, 61), as demonstrated in numerous studies (62, 63).
The oral cavity is the second largest microbiota in humans, and Neisseria, Corynebacterium, Leptotrichia, Streptococcus, Veillonella, Prevotella, Fusobacterium, and Capnocytophaga are among the most common bacterial taxa (64). Similarly, the healthy lung microbiota is predominantly composed of Streptococcus, Fusobacterium, Pseudomonas, Veillonella, Prevotella, and Capnocytophaga (65). In a recent study of eight patients with SARS-CoV-2 infection, samples of bronchoalveolar lavage fluid (BALF) were sequenced for meta-transcriptome. The host variants varied from 0 to 51 due to a high rate of evolution of the virus. Differences in microbiota composition between healthy controls and those with SARS-CoV-2 were observed, although the variation was not specific (65). Opportunistic bacteria have been found in BALF of patients with COVID-19, particularly Veillonella and Capnocytophaga, which are commensal of the oral cavity (66). Another study on BALF of patients with COVID-19 revealed predominance of Leptotrichia buccalis, Veillonella parvula, Capnocytophaga gingivalis, and Prevotella, whereas Acinetobacter baumannii, Klebsiella pneumoniae, Aspergillus flavus, Candida glabrata, and Candida albicans were detected in nasal swabs (67, 68). Several studies demonstrated a higher incidence of ventilator-associated pneumonia (VAP) in patients with ARDS due to SARS-CoV-2 infection. In a multicentric study in 586 patients admitted to ICU, VAP incidence resulted as high as 29%, being Pseudomonas aeruginosa and Staphylococcus aureus the most involved microorganisms. Septic shock at VAP onset and ARDS were associated with fatality (69). Another study which compared 81 COVID-19 and 144 non-COVID-19 patients, concluded that those with SARS-CoV-2 infection were significantly more likely to develop VAP (Cox proportional hazard ratio 2.01, 95%CI 1.14–3.54, p = 0.0015). The organisms responsible of VAP and microbiome were similar between groups, but COVID-19 patients were more susceptible to Aspergillus and Herpes infections than general ICU patients (70). Again, data extracted by Rouzé et al. (71) from an European multicentric cohort of 1,576 patients concluded that lower respiratory tract infections associated to ventilation were significantly higher in patients with COVID-19 as compared to influenza patients, and those without viral infections. The most common causative pathogens included Pseudomonas aeruginosa, Enterobacter spp, and Klebsiella spp. Further studies are warranted to confirm the real incidence of lung dysbiosis and VAP in cases of severe COVID-19 pneumonia. Figure 2 depicts differences in lung and gut microbiota composition in patients with severe COVID-19 pneumonia and patients with typical ARDS.
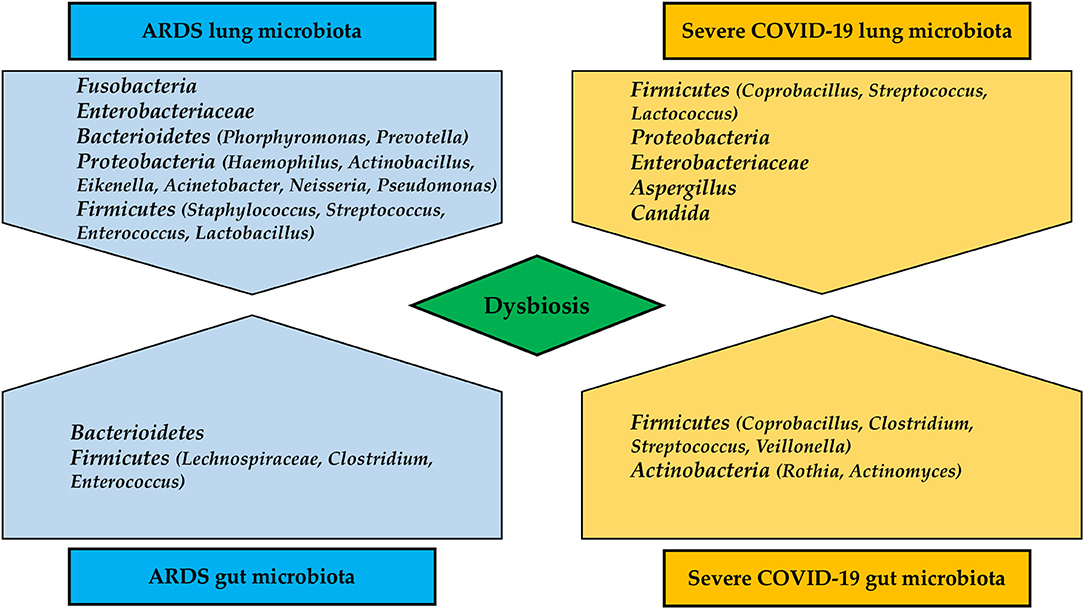
Figure 2. Differences in lung and gut microbiota composition in patients with severe COVID-19 pneumonia and typical ARDS.
Risk Factors and Prevention of Dysbiosis in Severe COVID-19-ARDS
Recognizing the causes of dysbiosis in critical illness is challenging (16). Literature confirm that hospital admission may only partially alter patients' ecosystem, while increasing severity is commonly seen when implementing the level of cares. Indeed, several and impacting iatrogenic forces are applied during ICU care, thus affecting the physiology of the host, which in turn alters the community structures of resident microbes. In healthy or minimally ill individuals the elimination of pathogens is normally rapid and mediated by the passage through the intestinal tract via defecation. During critically illness, glucose and electrolyte disturbances, endogenous and exogenous opioids, sedatives and catecholamines, myorelaxants, poor oral hygiene, endotracheal or nasotracheal intubation, cuff pressure balance, body position, patients' transport and mobilization represent only few of the possible risk factors that may influence dysbiosis (72, 73). The consequent systemic response includes a lowering of the stomach and intestinal transit-time, drops in bile salt production, impairment of immunoglobulin type A production, and loss of the mucosal barrier. Moreover, the intestinal wall is often hypoperfused, leading to mucosal inflammation, altered oxygen gradient and increased nitrate concentration, while reducing the commensal bacteria in favor of the pathogens, and lowering the transit-time and pathogens' elimination. Additionally, when mechanical ventilation is applied, the ecological system of the lungs is highly stressed with possible impaired muco-ciliary clearance, depressed cough reflex and pathogens' overgrowth (16). If to these important grounds typical COVID-19 patients' comorbidities are added, the risk of dysbiosis is dramatically increased.
Comorbidities of Patients With Severe COVID-19-ARDS and Their Role in Dysbiosis
The age of patients with severe COVID-19 is commonly high, and populations of gut bacteria normally change with age. In the gut of the elderly, less Bifidobacteria have been identified, maybe because of reduced gut epithelial barrier function, reduced immune function, and increased inflammation (74). In addition, obesity seems to be a typical characteristic of COVID-19, and it is associated with higher levels of pro-inflammatory cytokines and a poorer gut barrier. These mechanisms may favor the passage of gram-negative bacteria with possible endotoxemia (75). Intestinal bacteria along with their products (like SCFAs) play a key role in the protection of the mucosal intestinal barrier, and in the maintenance of adequate permeability through tight junctions, that may be down-regulated by pro-inflammatory cytokines and chemokines (76). Low-grade systemic inflammation is also present in those with chronic cardiovascular disease, type II diabetes mellitus, arthritis, and cancer. This may increase the risk of infection and altered microbiota (77). A great proportion of COVID-19 patients has hypertension (78). SCFAs has a crucial role in regulating blood pressure, while trimethyl amine-n-oxide (TMAO) is involved in atherosclerosis, hypertension, and coronary artery diseases' pathogenesis (79).
COVID-19 patients who present type II diabetes mellitus are around 30%. Lactobacilli are higher in diabetic patients, while the abundance of Firmicutes is correlated with inflammation (80). This basal diversity should be kept in mind when approaching dysbiosis in a COVID-19 patient with both SARS-CoV-2 infection and type II diabetes mellitus. Finally, according to the CDC's weekly report, around 35% of critically ill COVID-19 patients have an underlying chronic lung disease, such as asthma (81). As previously explained, the direct lung damage may be responsible for microbiota dysbiosis and over-infections. The airway mucosal barrier may lose the critical defense against SARS-CoV-2 and other infections (46).
Indirect Risk Factors of Dysbiosis Associated With Mechanical Ventilation in COVID-19
Oral Hygiene and Aseptic Mouthwashes
Poor oral hygiene has been associated with increased incidence of pneumonia and dysbiosis in critically ills (82), and no scientific evidence exists yet to recommend mouthwashes to control the SARS-CoV-2 load in the oral cavity. Some antiseptic mouth rinses have antiviral ingredients able to decrease the viral load, but conclusive evidences are still limited. Besides, changes in the normal oropharyngeal flora as a consequence of poor oral hygiene could be related, not only to a greater ease of infection by SARS-CoV-2 with consequent higher viral load and greater severity (83), but also to secondary superinfections (84).
Common periodontal pathogens have been identified in the lungs of ICU patients, including Treponema denticola, P. gingivalis, Fusobacterium nucleatum, Actinobacillus actinomycetemcomitans, and Veillonella parvula (85, 86). In a cohort of 122,251 patients, the risk of pneumonia increased in those who did not engage in good oral care, including the presence of dental caries and missing teeth (87). The use of mouthwashes to prevent pneumonia is still debated. A randomized controlled trial (RCT) on 80 ICU patients who were randomized to receive Nanosil mouthwashes and chlorhexidine 0.12% for 5 days demonstrated that the pneumonia rate was reduced in the Nanosil group (2.7 vs. 23.7%, p = 0.008), but mortality was similar in both groups (88). Another trial investigating the role of peroxide hydrogen over normal saline in the prevention of pneumonia concluded that patients treated with peroxide hydrogen had a lower risk of contracting VAP (relative risk [RR], 2.6; 95% confidence interval [CI], 1.04–6.49, p = 0.0279) (89). Although no data on patients infected with SARS-CoV-2 are available, mouthwashes containing cetylpyridinium chloride reduced in vitro SARS-CoV-2 infectivity. The reduction of SARS-CoV-2 infectivity may reduce lung dysbiosis, but the novelty of this study is limited by the fact that, being in vitro, it cannot reproduce the real condition of an in-vivo oral flora (90). Moreover, the efficacy of mouthwashes with hydrogen peroxide has not been in doubt, especially their capacity to inactivate corona and influenza viruses (91, 92). In conclusion, irrespective of the mouthwash agent, maintaining good oral hygiene is an effective strategy to reduce the rate of over-infections in all ICU patients, especially in those with COVID-19 and ARDS which may present higher risk of superinfections (70, 71).
Endotracheal Intubation, Cuff Pressure Control, and Chest Physiotherapy
Unfortunately, no specific study in severe COVID-19 pneumonia is actually available and current suggestions come from ICU patients. The choice of nasal intubation over the endotracheal route should be weighed against several factors, including a higher level of comfort, less use of sedatives and analgesics, but also the higher incidence of sinusitis and possible translocation of nasal bacteria to the lungs (93). A study comparing patients intubated endotracheally with polyurethane tubes with continuous assessment of cuff pressure and subglottic drainage with patients intubated with PVC and intermittent cuff pressure measurements and intermittent subglottic drainage, demonstrated that prevention of VAP could be performed by using polyurethane tubes, performing continuous subglottic drainage, and continuous cuff pressure measurement (94). The use of chest physiotherapy maneuvers such as subglottic secretion drainage has been identified as a valuable adjuvant for the prevention of VAP in ICU patients. This technique is currently in use in several ICUs during the COVID-19 pandemic, although with limited resources and higher risks. A modified technique has recently been proposed by our group to limit airborne exposure (95). A recent meta-analysis investigated the real benefits of this maneuver, concluding that subglottic secretion drainage is effective but not significant in reducing VAP (RR, 0.56; 95%CI, 0.48–0.63, p = 0.841) (96). A recent RCT compared chest physiotherapy with controls for the prevention of VAP, and found that VAP occurred in 39% of the intervention group vs. 8% of the control group (odds ratio, 14; 95% CI, 0.03–0.56; p = 0.002); no differences were found in terms of mortality and length of ICU stay (97). However, a meta-analysis concluded that chest physiotherapy does not reduce the incidence of VAP, although these results should be viewed cautiously due to the heterogeneity of the studies and poor evidence (98).
Body Position
As understood by decades of research, body position plays a pivotal role in the development of pneumonia and lungs' dysbiosis (99). The lateral position is known to be effective for improving oxygenation in monolateral pneumonia (100), but severe COVID-19 pneumonia seems to interest both lungs (101). Besides, lateral position in COVID-19 is applied (102, 103). Despite the confirmed application of lateral position in COVID-19, its effects on possible superinfections and subsequent dysbiosis has not been investigated yet. The majority of literature concerning the effects of body position on superinfections and possible dysbiosis come from non-COVID-19 setting. A meta-analysis from 10 RCTs compared a semi-recumbent position (30°-60°) and a supine position (0°-10). The semi-recumbent position, with the higher elevation of the head of the bed, reduced the risk of VAP (14 vs. 40%; RR, 0.36; 95% CI, 0.25–0.5) (104). The lateral Trendelenburg position and a semi-recumbent position were compared in a recent RCT, which concluded that the semi-recumbent position was associated with a higher incidence of VAP than the lateral Trendelenburg (4 vs. 0.5%; RR, 0.13; 95% CI, 0.02–1.03; p = 0.04), with no differences in mortality and other secondary outcomes (105). Finally, the prone position, which assumes a pivotal role for severe COVID-19 with ARDS (101), did not seem to increase the incidence of VAP (incidence rate per 100 days of mechanical ventilation of 1.18 vs. 1.54 for supine and prone positions, respectively, p = 0.1) (106), as confirmed by a previous similar RCT (107). Similar results were obtained from a multicentric study on 586 COVID-19 patients (69). In conclusion, as stressed above, few studies investigating the role of body position on lung dysbiosis in severe COVID-19 pneumonia are currently available. We suggest that body position may play a role in the development of dysbiosis.
Medications as Possible Risk Factors of Dysbiosis
Antibiotics
Numerous medications are administered in the ICU. Antibiotic consumption in ICUs is almost doubled that in non-ICU wards. During the COVID-19 pandemic, severely ill ICU patients received more antibiotics (66, 108). Antibiotic use is associated with important changes in gut microbial communities with a subsequent loss of the colonization resistance, a hallmark feature of the healthy gut microbiota, thus increasing the susceptibility to gastrointestinal infections by nosocomial pathogen (109, 110). Antibiotic exposure seems to increase the phyla of Actinobacteria, Bacteroidetes, Firmicutes, and Proteobacteria (111). The COVID-19 pandemic is associated with a higher and often unnecessary use of antibiotics in the early phases of the disease, in older people, and in mechanically ventilated patients (112, 113). Azithromycin is one of the largely used antibiotics in COVID-19 due to its antiviral and immunomodulatory effects in vitro, which include the interference with receptor mediated binding, viral lysosomal escape, intracellular pathways and enhancement of type I and III interferon expression (20). Besides, recent trials on the use of azithromycin combined or not with hydroxychloroquine in critically ill COVID-19 patients tended toward non-routine use (114). Similar results were obtained from a large observational study (115, 116). Another RCT on the use of azithromycin in hospitalized patients with COVID-19 is currently ongoing (117).
Sedatives, Analgesics, and Myorelaxants
Sedation and analgesia in mechanically ventilated COVID-19 patients are important pieces of this complex multisystemic puzzle. Patients with severe COVID-19 pneumonia and multiorgan disease are often kept sedated and curarized for longer periods than non-COVID-19 patients (median 10 days vs. 1 day) (118). Moreover, stopping sedatives, analgesics, and myorelaxants a greater proportion of COVID-19 patients experienced delirium (119). Growing evidence confirm the role of sedatives, analgesics opioids and myorelaxants on gut microbiota composition. Opioids receptors are located both in the digestive tract and central nervous system, and its effects on dysbiosis have been largely reported by literature. Moreover, some bacterial components can modify the expression of opioids receptors, changing the tolerance to pain (120). Larger studies are needed to confirm the effect of these medications on gut microbiota composition and outcome.
Inotropes and Vasopressors
Critically ill mechanically ventilated COVID-19 with ARDS frequently report the need of vasopressors and inotropes for treating septic shock or other multisystemic diseases (69, 121). Insights from animal models concluded that catecholamines may increase the growth of bacteria, virulence-associated factors, adhesions, and biofilm formation, while influencing the outcome of infections in many hosts (122). Inotropes have been associated with the growth of pathogens, and vasopressors inhibit growth (123, 124). To date, no evidence concerning the effects of inotropes and vasopressors on gut dysbiosis have been described.
Proton Pump Inhibitors (PPI) and H-2 Receptor Antagonists (H2RA)
PPI and H2RA are largely used in ICU for stress ulcer prophylaxis, and likely increases mortality but with low certainly evidence (125–127). The effect of PPI on gut microbiome has been largely investigated in animal studies. PPI showed increased intestinal permeability when compared to non-treated animals, thus changing the microbial composition, impairing colonization resistance, and inducing dysbiosis (128) and pneumonia in humans (129). This was also confirmed by other evidences in humans (130), but few specific investigations on COVID-19 are available yet. In a small monocentric study in 152 COVID-19 patients the impact of PPI was tested (131). Sixty-two patients were treated with PPI, of whom 48.4% without clear reason. Forty-eight percentage of patients treated with PPI, and only 20% of those non-treated presented with secondary infection. Forty-eight percentage of PPI treated patients and 12% of non-treated developed ARDS. The development of secondary infections remained significant after adjusting for other potential confounding (131). Although the sample size of this study is small, we believe that an association between the use of PPI and H2RA and superinfections in COVID-19 who are -per se- at higher risk of superinfections should be considered. Moreover, another study concluded that the pre-hospital use of PPI was associated with worse clinical outcome in hospitalized COVID-19 (132, 133).
Steroids
The Surviving Sepsis Campaign Coronavirus Disease 2019 recently published the first update of their known guidelines (134). High-quality evidence showing reduction in death and minimal adverse effects with short course of corticosteroids. Thus, the guidelines strongly recommended the use of a short course of systemic corticosteroids over not using corticosteroids. There are no trials comparing different corticosteroids with each other, but dexamethasone was associated with good treatment effect compared to no corticosteroids (135, 136). On one hand, corticosteroids reduce death and severity of COVID-19; on the other hand, corticosteroids remain mediators of the stress response that may enhance the hypothalamus-pituitary-adrenal axis which is implied in the control of immune response to stressor agents and intestinal microenvironment. Glucocorticoids may be therefore be involved in the alteration of gastrointestinal microbiota by enhancing the translocation of aerobe and gram negative enteric bacteria to extraintestinal tissues (137). A recent RCT reported no substantial differences in infections among critically ill COVID-19 patients treated with dexamethasone (21.9%) and those not treated (29.1%). However, few conclusive studies are warranted to confirm the real effects of corticosteroids on superinfections in severe COVID-19 pneumonia (138).
Treatment Proposals
Prebiotics and Probiotics
Probiotics are living micro-organisms which confer benefits to the host when administrated in adequate dose, and most used organisms include bifidobacteria, lactic acid bacteria, enterococci, and yeast (139). Probiotics usually have distinctive characteristics such as the ability of surviving under intestinal conditions, stimulating the immune system and acting against pathogens, also preventing health-care associated pneumonia (140). Furthermore, probiotics exert interesting properties by modulating cytokines production, interacting with TLRs, antagonizing pathogens in cell adhesion and mucin homeostasis, and by stimulating SCFAs production (141).
Probiotics act by enhancing epithelial barrier function and are anti-inflammatory, improving gut diversity and competing against opportunistic pathobionts for the same ecological niches in the gut (including competition for nutrients or cellular receptors on the mucosal surface). Specifically, they act by blocking or activating multiple signaling pathways (such as NF-kB and STAT1) and producing protective metabolites such as SCFAs. Gastrointestinal symptoms (including diarrhea) appear to be common in COVID-19, possibly reflecting alterations in the composition of gut microbiota (dysbiosis), inflammation and disruption of the epithelial barrier. In this context, administration of probiotics and/or prebiotics might be considered. As an example, Lactobacilli are well-known modulators of intestinal inflammation and immune response, so that their administration is recommended to counteract high level of inflammation, in prevention of diarrhea, and during infections sustained by enteric pathogens (139). Additionally, Bifidobacteria, are able to produce vitamins, enzymes, acetic and lactic acids, lowering the pH in the colon microenvironment and inhibiting (potential) pathogens (142). Evidence of beneficial effects, such as decreased infections frequency, shortening of the duration of episodes by 1–1.5 days, reduced shedding of rotaviruses or an increase in the production of rotavirus-specific antibodies, have been demonstrated for Lactobacillus rhamnosus GG (LGG), L. casei Shirota, L. reuteri, Bifidobacterium animalis ssp. lactis Bb-12, and a number of other probiotic strains (143).
Probiotics, prebiotics (formulation of nutrients exploited by probiotic bacteria), and symbiotics (a synergistic combinations of pro- and prebiotics) are currently used to improve gut dysbiosis, by favoring the proliferation of healthy protective bacteria in the intestine, ameliorating or preventing inflammation (balancing proinflammatory and immunoregulatory cytokines) and other intestinal diseases (144). The use of probiotics has also been associated with a reduction in the incidence and severity of VAP. Probiotics reduced the duration of mechanical ventilation in critically ill patients (145, 146). Specifically, use of the probiotic Streptococcus salivarius K12 has been proposed for patients with COVID-19 (146). Also, the presence of ACE2 was identified in certain probiotics strains. Oral delivery of human ACE2 through the probiotic species Lactobacillus paracasei increased ACE activity in the serum and tissues of mice. Similar results can be obtained with the bacteria-derived B38-CAP enzyme (147, 148). Recent research highlighted the role of mucin biopolymers as pivotal in regulating mucin production, which is implied in viral replication in the gut. Lactobacilli are known implementors of the mucus layer and glycocalyx, and inhibitors of pathogenic adherence, thus preventing intestinal inflammation (149). A recent network meta-analysis provided a rationale for the implementation of probiotics in preventing and treating COVID-19. They identified 90 genes potentially implicated in COVID-19 probiotics treatment. Moreover, the clearly shown that the application of probiotics could play a pivotal role on ACE2-mediated virus entry, activation of the systemic immune response, immunomodulatory pathways, lung tissue damage, cardiovascular complications, and altered metabolic pathways in the disease outcome (150). There are currently multiple lines of research with probiotics and numerous potential therapeutic indications, however studies with strong scientific evidence of therapeutic benefits are required.
Fecal Microbiota Transplantation
Fecal microbiota transplantation (FMT) is gaining ground as a treatment option for certain changes in the gut microbiota. The mechanism of action of FMT requires a fecal suspension from a healthy donor deposited into the gastrointestinal tract of a patient by using an endoscope, nasal tube, or capsule. However, FMT is still considered “off label” except for recurrent or refractory Clostridium difficile infections, where reconstitution of the intestinal microbiota by FMT has proved extremely successful and has definitively confirmed the role of dysbiosis in the pathogenesis such infection. Only one study reported FMT in COVID-19 population (151). Because COVID-19 frequently presents with gastrointestinal symptoms (such as diarrhea), fecal transplantation could potentially contribute to spreading the virus. Therefore, the authors suggested careful identification of donors, considering typical symptoms and history of possible contacts, as well as donor testing for SARS-CoV-2 by real-time PCR (152). Eleven COVID-19 patients who received fecal microbiota transplantation resulted in altered peripheral lymphocyte subset, restored gut microbiota and alleviated gastrointestinal disorders (151). FMT efficacy may be affected by some microbial metabolites as primary bile acids (such as cholic acid and chenodeoxycholic acid), that are conjugated by the gut microbiota and bile salt hydrolase to form secondary bile acids (such as deoxycholic acid, lithocholic acid, and ursodeoxycholic acid) (153). The post-antibiotic expansion of C. difficile population was shown to be strongly associated with the depletion of secondary bile salts, consequently to an antibiotic-mediated depletion of microbial taxa mediating the conversion of primary into secondary bile acids (154). Primary and secondary bile acids may also exert anti-inflammatory properties and inhibit several viruses by modulating the cytokine-storm via NF-kB (influenza A, and other viruses) (155).
Most intriguingly, while the treatment's success of FMT mostly revolves around intestinal viable healthy bacteria that are transferred through fecal suspensions, it should be considered that the viable bacteria fraction may not be the only factor affecting the recipient's biology. Viruses, archaea, fungi, donor's colonocytes, immunoglobulin, protists, and a number of metabolites, made by the donor's commensal bacteria (as SCFAs) or intestinal cells, can be implanted, potentially triggering a plethora of functionally different effects (156).
Dietary Composition
In the acute phase of ICU admission, inflammation, energy expenditure, and catabolic metabolism are enhanced (157). During their stay in the ICU, patients often develop post-ventilation-acquired dysphagia and ICU-acquired weakness, which mean nutritional support has a pivotal role in maintaining the necessary muscular strength to help wean patients from the ventilator (158–160). Moreover, critical illness is considered to be a major environmental factor in influencing gut homeostasis and dysbiosis, and nutritional therapy could play an essential role in these processes. Among the various environmental factors, indeed, diet is a source of dominant variation of the whole gut microbial community (161). As an example, nutritional models based on plant-based foods were shown to promote a more favorable gut microbiota profile based on the high amount of dietary fiber and SCFA (162). Therefore, nutrition may exert different indirect effect on intestinal function by modulating the gut microbial composition. A recent study on fecal samples of patients with COVID-19 revealed a high abundance of bacterial species Collinsella aerofaciens, Collinsella tanakaei, Streptococcus infantis, Morganella morganii, and higher nucleotide de novo biosynthesis, amino acid biosynthesis, and glycolysis. These were distinct from fecal samples of patients without COVID-19 who had higher abundance of SCFAs-producing bacteria, including Parabacteroides merdae, Bacteroides stercoris, Alistipes onderdonkii, and Lachnospiraceae bacterium 1_1_57FAA (163). It is now well-recognized that SCFAs exert several beneficial effects, influencing a number metabolic (as the lipids, cholesterol and glucose metabolism) and inflammatory (as the butyrate-mediated inhibition of macrophagic NF-κB or inhibition of the LPS-induced cytokines IL-6 and IL-12p40) responses (164).
Colon bacteria respond to fermentable substrates provided by the diet to produce SCFAs and gases through anaerobic metabolism (165). Within this context, dietary intake is an essential factor for resilience of patients' gut microbiota and its impact on upper respiratory tract infections (145).
The use of early enteral nutrition has been associated with improved immunologic function, decreased bacterial translocation, and better mucosal integrity (111). Moreover, the composition of enteral nutrition has a great impact on intestinal homeostasis. The gut microbiota is normally preserved be feeding with various dietary components in different concentrations. With that in mind, an inadequate dietary composition may alter the composition of the intestinal microbiota, thus increasing the growth of opportunistic pathogens over commensals (111).
General nutritional consideration for ICU patients should be applied to COVID-19 patients (166). As COVID-19 is frequently associated with gastrointestinal symptoms, patients can be at high risk of refeeding syndrome. If this risk is present, SCCM/ASPEN guidelines recommend starting at ~25% of the target energy intake (whether enteral or parenteral) (166). The ESPEN guideline estimates around 27 kcal/kg body weight/day, based on total energy expenditure, for patients aged >65 years with multiple comorbidities, or 30 kcal/kg body weight/day, based on total energy expenditure, for severely underweight patients with multiple comorbidities and older adults (individually adjusted on the basis of nutritional status, physical activity, and disease status) (167). An energy goal of 15–20 kcal/kg actual body weight should be reached within the first week of nutritional support even in COVID-19 (166). Recently, in a prospective observational study Cereda et al. in mechanically ventilated COVID-19 patients who have been fed with a low caloric intake in the early phase of ICU admission, found a higher risk of death. Additionally, patients with mild obesity were associated with higher mortality, while those with moderate-severe obesity were more difficult to wean from the ventilator (168). Early overfeeding should be avoided, because aggressive caloric intake can cause hyperglycemia or the need for insulin therapy (169). In case of contraindications to oral and enteral nutrition, parenteral nutrition should be implemented and increased within 3–7 days (170, 171). In critically ill patients intolerant to enteral feeding, intravenous erythromycin should be considered as the first-line prokinetic therapy, followed by intravenous metoclopramide or a combination of both (170). Prone positioning is being used with increasing frequency to treat both typical ARDS and respiratory distress in severe COVID-19 pneumonia. Traditionally, this leads to forced periods of rest from enteral nutrition (172), although enteral nutrition has recently been demonstrated to be feasible and safe in the prone position as well (173). In patients at high risk of aspiration, post-pyloric enteral nutrition can be provided instead (170) to reduce the possible risks related to prone positioning and development of pneumonia.
The specific recommendations for nutritional management in COVID-19 (167) suggest that a high-energy, low-to-normal carbohydrate (based on diabetic status and glycemic control), normal-to-high protein diet should be considered. Contrasting findings are available concerning the optimal protein intake for critically ill patients (174). Protein intake can influence the catabolic response. During the catabolic phase, within the first 10 days of ICU admission, a reduction in muscle mass of up to 1 kg/day in patients with multiorgan dysfunction can occur (175). A recent RCT compared enteral feeding with high-intact-protein formula (VHPF) with a standard high-protein formula (SHPF). The VHPF facilitated feeding without increasing energy intake, which is consistent with previous ESPEN guidelines (176). However, early high protein intake is associated with a lower mortality rate only in patients with a low skeletal muscle area at hospital admission, not in those with a normal skeletal muscle area (177). Another study found that improvement in daily protein intake could reduce 3-month mortality after hospital discharge (178). A standard high-protein (>20%) isosmotic enteral formula may be used in the early phase of critical illness, with possible addition of fibers (if tolerated) for maintenance of gut microbiota function (166). Consider 1 g protein/kg body weight/day in older persons (individually adjusted on the basis of nutritional status, physical activity, disease status, and tolerance), and 1.2–2.0 g protein/kg body weight/day (166) in patients with multiple comorbidities (167). An isocaloric, high-protein diet is recommended for obese patients, especially guided by urinary nitrogen losses (170); if this measurement is not available, a protein intake of 1.3 g/kg should be considered. The latest ESPEN guidelines recommend a daily protein intake of 1.3 g/kg, delivered progressively (170). However, a great number of COVID-19 patients require continuous renal replacement therapy as part of the systemic multiorgan dysfunction that they manifest. Thus, specific consideration of protein intake during the use of such filters for renal depurations should have been counted by novel guidelines (179).
The amount of glucose, whether parenteral or via carbohydrates by enteral feeding, should not exceed 5 mg/kg/min (170). However, current guidelines in COVID-19 did not account that hypertension, obesity, and diabetes mellitus are the most prevalent comorbidities that may alter patients' metabolic profile (180). Similar consideration may be done for lipids administration in a population composed by a large number of obese patients, as COVID-19 population is (180). Indeed, guidelines recommend that intravenous lipids for parenteral nutrition should not exceed 1.5 g/kg/day (170). The intake of carbohydrates and fat should be adapted according to energy ratio of 50:50 from fat and carbohydrates for ventilated patients (167). Additionally, a ketogenic diet for obese or diabetic patients should be considered (181). Since COVID-19 often leads to liver and renal failure, parenteral Gln dipeptide should not be administered (170). Blood glucose should be measured at ICU admission and at least every 4 h for the first 2 days. Insulin therapy should begin when glucose levels exceed 180 mg/dL (170). Triglyceride levels should be considered in cases of prolonged sedation with propofol or prolonged administration of intravenous lipid emulsion for parenteral nutrition (166). Adequate intake of vitamins and minerals is paramount for the prevention of viral infections. Particularly, vitamins A, E, B6, B12, C, and D; zinc; selenium; iron; and omega-3 polyunsaturated fatty acids should be administered with a view to ameliorating clinical outcomes, as advised for other viral illnesses (167, 182). A recent RCT demonstrated that no mortality advantages were found in critically ill patients who received early implementation of vitamin D (183). In COVID-19, a serum 25-hydroxyvitamin-D level of around 30 mg/mL reduced the risk for adverse clinical outcomes (184), but further studies are needed to confirm these findings. General dietetic recommendations for critically ill patients with COVID-19-ARDS are depicted in Figure 3.
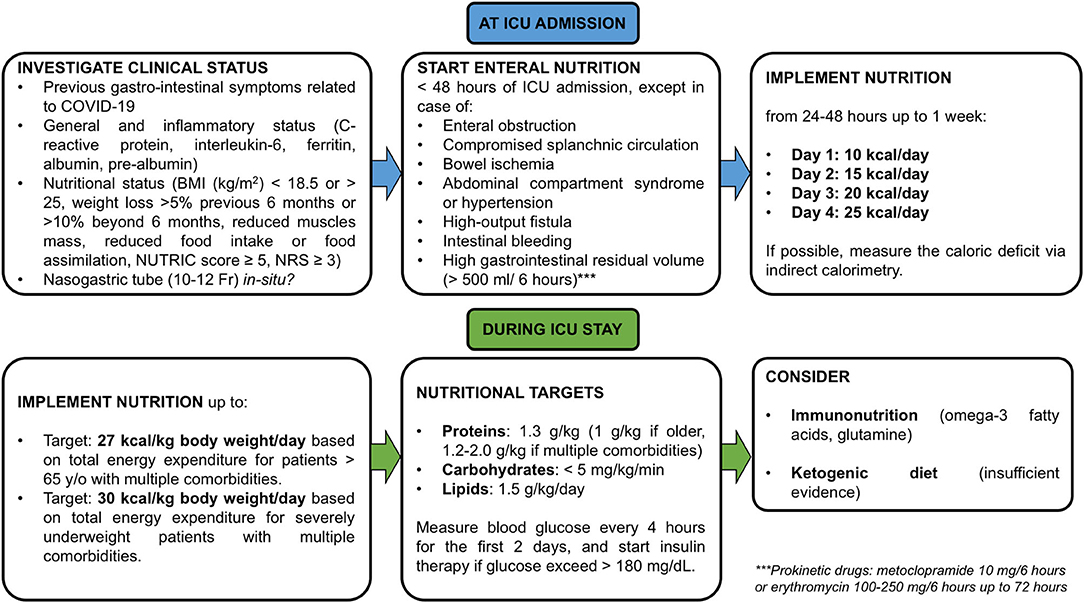
Figure 3. Dietetic recommendations in cases of COVID-19 with ARDS. Nutritional recommendations for critically ill patients with COVID-19 and ARDS from ICU admission to ICU stay. NRS, nutritional risk screening. Each nutritional support is suggested to be calculated on Ideal body weight.
Other Nutritional Interventions to Modulate Dysbiosis in COVID-19
Other nutritional interventions have been proposed to modulate the cytokine storm in ARDS and COVID-19, but studies are lacking. Therefore, the following sentences represent an overview of nutritional treatments for immune and inflammatory dysfunctions in COVID-19 patients. ARDS is considered an overwhelming systemic inflammatory process. Patients with COVID-19 frequently present with hypoalbuminemia and lymphopenia, which may reflect malnutrition and hyperinflammation and have been associated with a negative prognosis (1). Although the albumin level should not be considered a nutritional marker in patients with active inflammation, prealbumin levels are associated with progression to ARDS (185). Patients who survived severe COVID-19 pneumonia often present significant functional limitations, and experience higher morbidity and mortality (186).
Immunonutrition
Immunonutrition has been proposed for patients with severe COVID-19 pneumonia (187, 188) because supplementary immunonutrients and antioxidants have been shown to promote favorable outcomes in the general critically ill population (186, 189, 190). The severity of disease influences the efficacy of immunonutrition (190). In one meta-analysis, immunonutrition reduced mortality and improved oxygenation in patients with ARDS (191); however, more recent studies failed to replicate these findings (186, 192–194). Several products are available to provide immunonutrition. Broadly, these consist of antioxidant vitamins (e.g., vitamin E, vitamin C, carotene), trace elements (e.g., selenium, zinc), essential amino acids (e.g., glutamine, arginine), and essential fatty acids (e.g., omega-3 fatty acids, eicosapentaenoic acid, docosahexaenoic acid, linolenic acid) (186).
Monounsaturated and polyunsaturated fatty acids are involved in cytokine production (190). When immunonutritional enrichment of fatty acids is administered, many components of the immune response are modulated and suppressed (190) by modification of the lipid bilayer of multiple cell types. Omega-3 fatty acids are essential lipids that are able to suppress pro-inflammatory eicosanoid biosynthesis, reduce lung permeability, inhibit inflammation by enhancing T cell function, and decrease pulmonary edema (189, 195). On the other hand, the administration of omega-6 fatty acids may have opposite effects. Thus, the intravenous administration of lipid-enriched solutions may be detrimental, increasing mortality and complications in critically ill patients, because of the infusion of high amounts of omega-6 fatty acids (196).
Glutamine and arginine are sulfur-containing amino acids that have been proposed as components of immunonutrition for their immunomodulatory properties. Particularly, the properties of glutamine include improvement of gut barrier function and immunomodulation of lymphocyte, neutrophil, and macrophage function (186). Glutamine also enhances glutathione synthesis and cell proliferation, thus enhancing antioxidant mechanisms. Likewise, arginine enhances nitric oxide synthesis, lymphocyte function, growth hormone production, and anabolism (189). Arginine is synthesized from proline and participates in the synthesis of ornithine, which is essential for immune function. Arginine deficiency has been found to correlate with suppression of T cell proliferation and cluster of differentiation (CD)3 (190). An RCT of a specific anti-inflammatory and antioxidant nutritional therapy regimen for patients with COVID-19 is ongoing (197). Precursors of cysteine may be administered exogenously in the form of N-acetylcysteine or procysteine, although cysteine and methionine are not easily captured into cells (190). The putative mechanisms of immunonutrition are summarized in Figure 4. Although glutamine and antioxidants could be considered in patients with oxidative stress, benefits to outcomes have not been reported. On the contrary, an increase in mortality was found in critically ill patients with multiorgan failure (198). Therefore, caution is needed since conclusive evidences are not available yet.
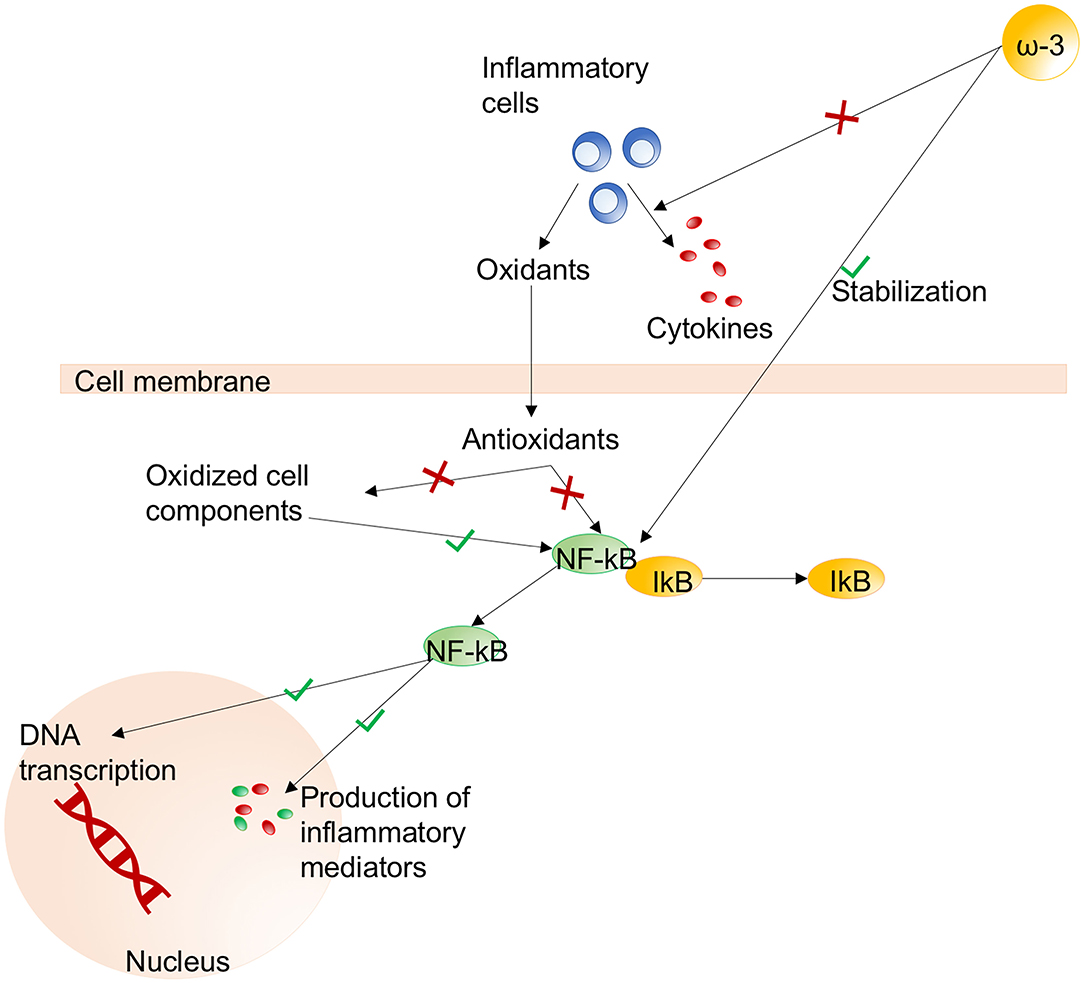
Figure 4. Immunonutrition. The main pathways activated (green) or inhibited (red) during immunonutritional therapy. Effects of omega-3 fatty acids on stabilization of the NF-κB/IκB pathway and reduced production of cytokines from inflammatory cells. Effects of NF-κB on the nucleus include DNA transcription and production of inflammatory mediators. NF-κB, nuclear factor kappa-B; ω-3, omega-3; DNA, deoxyribonucleic acid. Modified from Grimble (100).
Ketogenic Diet
Ketogenic diet is a nutritional alternative to mitigate inflammation in COVID-19 patients. The ketogenic diet is a low-carbohydrate, high-fat nutritional support strategy that promotes metabolic ketosis. This has proved to be efficient in controlling glucose levels and body weight, and in promoting anti-inflammatory effects in obesity and type 2 diabetes (199, 200). Ketogenic diets were initially proposed to control refractory status epilepticus and protect the central nervous system (201, 202). Over time, evidence emerged that very low-carbohydrate diets decreased energy intake while improving lipid and glucose homeostasis (203), as well as decreasing levels of inflammatory markers (181). Preliminary results in a murine model of beta coronavirus infection demonstrated that ketones protect against systemic inflammatory response (204). The rationale for using ketogenic diet in COVID-19 is summarized in the following paragraphs. Furthermore, a trial investigating the use of ketogenic diet for patients with COVID-19 is ongoing (205).
The release of inflammatory cytokines and caspase-1, as occurs in SARS-CoV-2 infection following the activation of innate immunity in response to damage-associated molecular pattern (DAMPs) (7), can be modulated by the nod-like receptor protein-3 (NLRP3) inflammasome (206). Ablation of NLRP3 is able to attenuate type 2 diabetes and atherosclerosis (7), which have been identified in most patients with severe COVID-19. During a ketogenic diet, alternative sources of energy are produced by the liver, including the ketone bodies β-hydroxybutyrate (BHB) and acetoacetate (ACA), to maintain the metabolic functions of the brain, heart, and skeletal muscles. The increased consumption of liver glycogen stores that is characteristic of all ketogenic diets is also associated with altered immune cell function. Specifically, the use of lactate as a source of mitochondrial oxidative energy plays a key role in the production of innate immune type I cells and interferon type I, which are effective in the host defense against viral infections (207). In an experimental mouse model, caloric restriction implemented through a ketogenic diet was found to exert anti-inflammatory effects; ketone bodies attenuated caspase-1 activation and IL-1β secretion by modulating the NLRP3 inflammasome (208). Recent research has proposed that the inhibitor of glycolysis, deoxy-D-glucose, could be a reasonable therapeutic strategy for SARS-CoV-2 infection (209), because it has been found to reduce the duration of ventilator support and partial pressure of carbon dioxide in patients with acute respiratory failure (210). The mechanisms of action of a ketogenic diet are summarized in Figure 5.
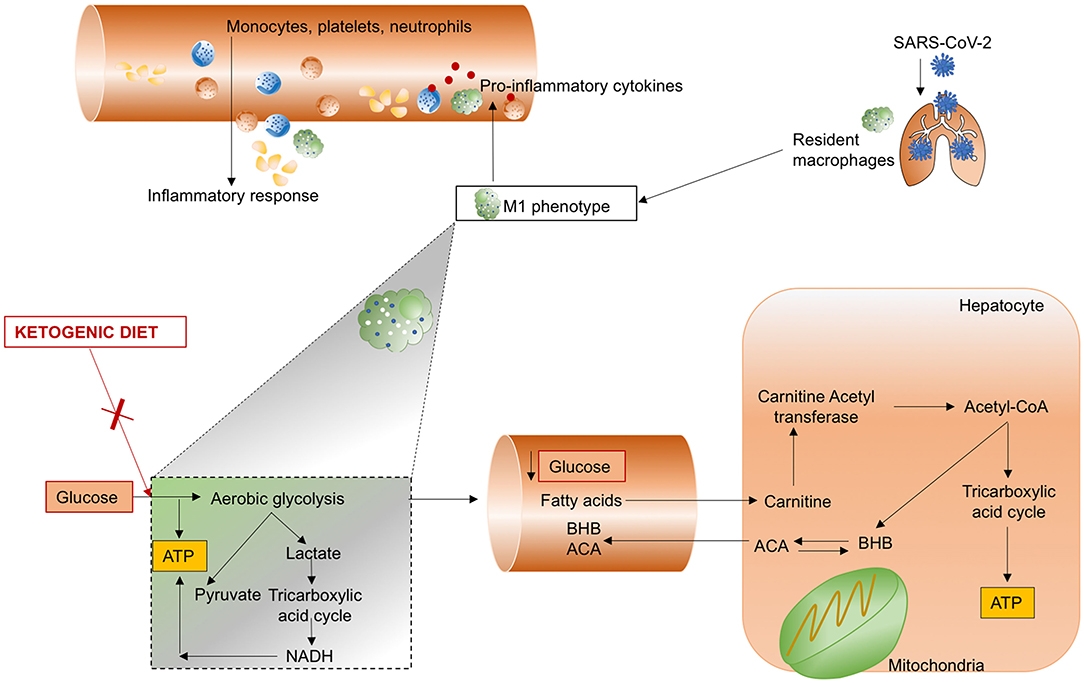
Figure 5. Ketogenic diet. SARS-CoV-2 infects the lung and induces hyperinflammation with recall of monocytes, platelets, and neutrophils by macrophages polarized to the M1 phenotype. A ketogenic diet is able to reduce the synthesis of adenosine triphosphate (ATP) from glucose by limiting aerobic glycolysis, usually implicated in the production of lactate and pyruvate, and activation of the tricarboxylic acid cycle, culminating in increased nicotinamide adenine dinucleotide (NADH). Glucose concentration in the blood is reduced, thereby increasing the production of β-hydroxybutyrate (BHB) and acetoacetate (ACA) from hepatocyte mitochondria.
Conclusions
Dysbiotic states of the microbiota may impact on the pathogenesis, as well as on the complexity, of immune and inflammatory diseases. Several mechanisms have been identified as potential targets to reduce inflammation and secondary infections. Particularly, novel nutritional interventions have been proposed to regulate the mechanisms underlying dysbiosis of the lung and intestinal microbiota. However, further studies on patients with severe COVID-19 are needed to confirm the effective benefit of such interventions.
Author Contributions
DB: conceptualization, writing original draft, review, and editing. CR, AF, ST, SS, VD, DG, AV, MB, IB, NP, LB, and AT: review and editing. PR and PP: writing, review, and editing. All authors contributed to the article and approved the submitted version.
Funding
This research was partly funded by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (401700/2020-8) and Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (E-26/210.181/ 2020).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. (2020) 395:1054–62. doi: 10.1016/S0140-6736(20)30566-3
2. Cao Y, Liu X, Xiong L, Cai K. Imaging and clinical features of patients with 2019 novel coronavirus SARS-CoV-2: a systematic review and meta-analysis. J Med Virol. (2020) 92:1449–59. doi: 10.1002/jmv.25822
3. Chen Y, Chen L, Deng Q, Zhang G, Wu K, Ni L, et al. The presence of SARS-CoV-2 RNA in the feces of COVID-19 patients. J Med Virol. (2020) 92:833–40. doi: 10.1002/jmv.25825
4. Ding S, Liang TJ. Is SARS-CoV-2 also an enteric pathogen with potential fecal–oral transmission? A COVID-19 virological and clinical review. Gastroenterology. (2020) 159:53–61. doi: 10.1053/j.gastro.2020.04.052
5. Heo J, Han D, Kim HJ, Kim D, Lee YK, Lim D, et al. Prediction of patients requiring intensive care for COVID-19: development and validation of an integer-based score using data from Centers for disease control and prevention of South Korea. J Intensive Care. (2021) 9:16. doi: 10.1186/s40560-021-00527-x
6. Karagiannidis C, Windisch W, McAuley DF, Welte T, Busse R. Major differences in ICU admissions during the first and second COVID-19 wave in Germany. Lancet Respir Med. (2021) 9:e47–8. doi: 10.1016/S2213-2600(21)00101-6
7. Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson JJ, et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. (2020). doi: 10.1016/S0140-6736(20)30628-0. [Epub ahead of print].
8. Martin-Loeches I, Dickson R, Torres A, Hanberger H, Lipman J, Antonelli M, et al. The importance of airway and lung microbiome in the critically ill. Crit Care. (2020) 24:537. doi: 10.1186/s13054-020-03219-4
9. Ojima M, Motooka D, Shimizu K, Gotoh K, Shintani A, Yoshiya K, et al. Metagenomic analysis reveals dynamic changes of whole gut microbiota in the acute phase of intensive care unit patients. Dig Dis Sci. (2016) 61:1628–34. doi: 10.1007/s10620-015-4011-3
10. Schuijt TJ, Lankelma JM, Scicluna BP, De Sousa E Melo F, Roelofs JJTH, De Boer JD, et al. The gut microbiota plays a protective role in the host defence against pneumococcal pneumonia. Gut. (2016) 65:575–83. doi: 10.1136/gutjnl-2015-309728
11. Barisione E, Grillo F, Ball L, Bianchi R, Grosso M, Morbini P, et al. Fibrotic progression and radiologic correlation in matched lung samples from COVID-19 post-mortems. Virchows Arch. (2020) 478:471–85. doi: 10.1007/s00428-020-02934-1
12. Chattopadhyay I, Shankar EM. SARS-CoV-2-indigenous microbiota nexus: does gut microbiota contribute to inflammation and disease severity in COVID-19? Front Cell Infect Microbiol. (2021) 11:590874. doi: 10.3389/fcimb.2021.590874
13. Rajilić-Stojanović M, de Vos WM. The first 1000 cultured species of the human gastrointestinal microbiota. FEMS Microbiol Rev. (2014) 38:996–1047. doi: 10.1111/1574-6976.12075
14. Wolff NS, Hugenholtz F, Wiersinga WJ. The emerging role of the microbiota in the ICU. Crit Care. (2018) 22:78. doi: 10.1186/s13054-018-1999-8
15. Boulangé CL, Neves AL, Chilloux J, Nicholson JK, Dumas ME. Impact of the gut microbiota on inflammation, obesity, and metabolic disease. Genome. (2016) 8:42. doi: 10.1186/s13073-016-0303-2
16. Dickson RP. The microbiome and critical illness. Lancet Respir Med. (2016) 4:59–72. doi: 10.1016/S2213-2600(15)00427-0
17. Lamarche D, Johnstone J, Zytaruk N, Clarke F, Hand L, Loukov D, et al. Microbial dysbiosis and mortality during mechanical ventilation: a prospective observational study. Respir Res. (2018) 19:245. doi: 10.1186/s12931-018-0950-5
18. Sencio V, Machado MG, Trottein F. The lung–gut axis during viral respiratory infections: the impact of gut dysbiosis on secondary disease outcomes. Mucosal Immunol. (2021) 14:296–304. doi: 10.1038/s41385-020-00361-8
19. Lankelma JM, van Vught LA, Belzer C, Schultz MJ, van der Poll T, de Vos WM, et al. Critically ill patients demonstrate large interpersonal variation in intestinal microbiota dysregulation: a pilot study. Intensive Care Medi. (2017) 43:59–68. doi: 10.1007/s00134-016-4613-z
20. Gyselinck I, Janssens W, Verhamme P, Vos R. Rationale for azithromycin in COVID-19: an overview of existing evidence. BMJ Open Respir Res. (2021) 8:e000806. doi: 10.1136/bmjresp-2020-000806
21. Wang F, Nie J, Wang H, Zhao Q, Xiong Y, Deng L, et al. Characteristics of peripheral lymphocyte subset alteration in COVID-19 pneumonia. J Infect Dis. (2020) 221:1762–9. doi: 10.1093/infdis/jiaa150
22. Robba C, Battaglini D, Pelosi P, Rocco RMP. Multiple organ dysfunction in SARS-CoV-2: MODS-CoV-2. Expert Rev Respir Med. (2020) 14:865–8. doi: 10.1080/17476348.2020.1778470
23. Cheung KS, Hung IFN, Chan PPY, Lung KC, Tso E, Liu R, et al. Gastrointestinal manifestations of SARS-CoV-2 infection and virus load in fecal samples from a hong kong cohort: systematic review and meta-analysis. Gastroenterology. (2020) 159:81–95. doi: 10.1053/j.gastro.2020.03.065
24. Cole-Jeffrey CT, Liu M, Katovich MJ, Raizada MK, Shenoy V. ACE2 and microbiota. J Cardiovasc Pharmacol. (2015) 66:540–50. doi: 10.1097/FJC.0000000000000307
25. Hashimoto T, Perlot T, Rehman A, Trichereau J, Ishiguro H, Paolino M, et al. ACE2 links amino acid malnutrition to microbial ecology and intestinal inflammation. Nature. (2012) 487:477–81. doi: 10.1038/nature11228
26. Li N, Ma WT, Pang M, Fan QL, Hua JL. The commensal microbiota and viral infection: a comprehensive review. Front Immunol. (2019) 10:1551. doi: 10.3389/fimmu.2019.01551
27. He Y, Wang J, Li F, Shi Y. Main clinical features of COVID-19 and potential prognostic and therapeutic value of the microbiota in SARS-CoV-2 infections. Front Microbiol. (2020) 11:1302. doi: 10.3389/fmicb.2020.01302
28. Du M, Cai G, Chen F, Christiani DC, Zhang Z, Wang M. Multiomics evaluation of gastrointestinal and other clinical characteristics of COVID-19. Gastroenterology. (2020) 158:2298–301.e7. doi: 10.1053/j.gastro.2020.03.045
29. Lamers MM, Beumer J, van der Vaart J, Knoops K, Puschhof J, Breugem TI, et al. SARS-CoV-2 productively infects human gut enterocytes. Science. (2020) 369:50–4. doi: 10.1126/science.abc1669
30. Girija ASS, Shankar EM, Larsson M. Could SARS-CoV-2-induced hyperinflammation magnify the severity of coronavirus disease (CoViD-19) leading to acute respiratory distress syndrome? Front Immunol. (2020) 11:1206. doi: 10.3389/fimmu.2020.01206
31. Negi S, Das DK, Pahari S, Nadeem S, Agrewala JN. Potential role of gut microbiota in induction and regulation of innate immune memory. Front Immunol. (2019) 10:2441. doi: 10.3389/fimmu.2019.02441
32. Battaglini D, Pimentel-Coelho PM, Robba C, dos Santos CC, Cruz FF, Pelosi P, et al. Gut microbiota in acute ischemic stroke: from pathophysiology to therapeutic implications. Front Neurol. (2020) 11:598. doi: 10.3389/fneur.2020.00598
33. Zuo T, Zhang F, Lui GCY, Yeoh YK, Li AYL, Zhan H, et al. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology. (2020) 159:944–55.e8. doi: 10.1053/j.gastro.2020.05.048
34. Hiippala K, Jouhten H, Ronkainen A, Hartikainen A, Kainulainen V, Jalanka J, et al. The potential of gut commensals in reinforcing intestinal barrier function and alleviating inflammation. Nutrients. (2018) 10:988. doi: 10.3390/nu10080988
35. Uzzan M, Corcos O, Martin JC, Treton X, Bouhnik Y. Why is SARS-CoV-2 infection more severe in obese men? The gut lymphatics – Lung axis hypothesis. Med Hypotheses. (2020) 144:110023. doi: 10.1016/j.mehy.2020.110023
36. Weinstock GM. Genomic approaches to studying the human microbiota. Nature. (2012) 489:250–6. doi: 10.1038/nature11553
37. Dickson RP, Schultz MJ, van der Poll T, Schouten LR, Falkowski NR, Luth JE, et al. Lung microbiota predict clinical outcomes in critically ill patients. Am J Respir Crit Care Med. (2020) 201:555–63. doi: 10.1164/rccm.201907-1487OC
38. Kyo M, Nishioka K, Nakaya T, Kida Y, Tanabe Y, Ohshimo S, et al. Unique patterns of lower respiratory tract microbiota are associated with inflammation and hospital mortality in acute respiratory distress syndrome. Respir Res. (2019) 20:246. doi: 10.1186/s12931-019-1203-y
39. Shepley-McTaggart A, Sagum CA, Oliva I, Rybakovsky E, DiGuilio K, Liang J, et al. SARS-CoV-2 envelope (E) protein interacts with PDZ-domain-2 of host tight junction protein ZO1. bioRxiv. (2020) 12:422708. doi: 10.1101/2020.12.22.422708
40. Hamming I, Timens W, Bulthuis M, Lely A, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. (2004) 203:631–7. doi: 10.1002/path.1570
41. Stanifer ML, Kee C, Cortese M, Zumaran CM, Triana S, Mukenhirn M, et al. Critical role of type III interferon in controlling SARS-CoV-2 infection in human intestinal epithelial cells. Cell Rep. (2020) 32:107863. doi: 10.1016/j.celrep.2020.107863
42. Bastard P, Rosen LB, Zhang Q, Michailidis E, Hoffmann H-H, Zhang Y, et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science. (2020) 370:eabd4585. doi: 10.1126/science.abd4585
43. Busnadiego I, Fernbach S, Pohl MO, Karakus U, Huber M, Trkola A, et al. Antiviral activity of type I, II, and III interferons counterbalances ACE2 inducibility and restricts SARS-CoV-2. MBio. (2020) 11:e01928–20. doi: 10.1128/mBio.01928-20
44. Deriu E, Boxx GM, He X, Pan C, Benavidez SD, Cen L, et al. Influenza virus affects intestinal microbiota and secondary salmonella infection in the gut through type I Interferons. PLoS Pathog. (2016) 12:e1005572. doi: 10.1371/journal.ppat.1005572
45. Steed AL, Christophi GP, Kaiko GE, Sun L, Goodwin VM, Jain U, et al. The microbial metabolite desaminotyrosine protects from influenza through type I interferon. Science. (2017) 357:498–502. doi: 10.1126/science.aam5336
46. Ahn D, Prince A. Participation of the IL-10RB related cytokines, IL-22 and IFN-λ in defense of the airway mucosal barrier. Front Cell Infect Microbiol. (2020) 10:300. doi: 10.3389/fcimb.2020.00300
47. Xue M, Zhao J, Ying L, Fu F, Li L, Ma Y, et al. IL-22 suppresses the infection of porcine enteric coronaviruses and rotavirus by activating STAT3 signal pathway. Antiviral Res. (2017) 142:68–75. doi: 10.1016/j.antiviral.2017.03.006
48. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. (2020) 395:497–506. doi: 10.1016/S0140-6736(20)30183-5
49. Sirivongrangson P, Kulvichit W, Payungporn S, Pisitkun T, Chindamporn A, Peerapornratana S, et al. Endotoxemia and circulating bacteriome in severe COVID-19 patients. Intensive Care Med Exp. (2020) 8:72. doi: 10.1186/s40635-020-00362-8
50. Prokunina-Olsson L, Alphonse N, Dickenson RE, Durbin JE, Glenn JS, Hartmann R, et al. COVID-19 and emerging viral infections: the case for interferon lambda. J Exp Med. (2020) 217:e20200653. doi: 10.1084/jem.20200653
51. Liu J, Li H, Luo M, Liu J, Wu L, Lin X, et al. Lymphopenia predicted illness severity and recovery in patients with COVID-19: a single-center, retrospective study. PLoS ONE. (2020) 15:e0241659. doi: 10.1371/journal.pone.0241659
52. Fathi N, Rezaei N. Lymphopenia in COVID-19: Therapeutic opportunities. Cell Biol Int. (2020) 44:1792–7. doi: 10.1002/cbin.11403
53. Huang I, Pranata R. Lymphopenia in severe coronavirus disease-2019 (COVID-19): systematic review and meta-analysis. J Intensive Care. (2020) 8:36. doi: 10.1186/s40560-020-00453-4
54. Diao B, Wang C, Tan Y, Chen X, Liu Y, Ning L, et al. Reduction and functional exhaustion of T cells in patients with coronavirus disease 2019 (COVID-19). Front Immunol. (2020) 11:827. doi: 10.3389/fimmu.2020.00827
55. Ferreira C, Viana SD, Reis F. Is gut microbiota dysbiosis a predictor of increased susceptibility to poor outcome of COVID-19 patients? An update. Microorganisms. (2020) 9:53. doi: 10.3390/microorganisms9010053
56. Gu S, Chen Y, Wu Z, Chen Y, Gao H, Lv L, et al. Alterations of the gut microbiota in patients with coronavirus disease 2019 or H1N1 influenza. Clin Infect Dis. (2020) 71:2669–78. doi: 10.1093/cid/ciaa709
57. Elshazli RM, Kline A, Elgaml A, Aboutaleb MH, Salim MM, Omar M, et al. Gastroenterology manifestations and COVID-19 outcomes: a meta-analysis of 25,252 cohorts among the first and second waves. J Med Virol. (2021) 93:2740–68. doi: 10.1002/jmv.26836
58. Chen W, Zheng KI, Liu S, Xu C, Xing C, Qiao Z. Virus discharge and initial gastrointestinal involvement are inversely associated with circulating lymphocyte count in COVID-19. Int J Med Sci. (2021) 18:1137–42. doi: 10.7150/ijms.51672
59. Teoh KT, Siu YL, Chan WL, Schlüter MA, Liu CJ, Peiris JSM, et al. The SARS coronavirus e protein interacts with PALS1 and alters tight junction formation and epithelial morphogenesis. Mol Biol Cell. (2010) 21:3838–52. doi: 10.1091/mbc.e10-04-0338
60. Dickson RP, Erb-Downward JR, Falkowski NR, Hunter EM, Ashley SL, Huffnagle GB. The lung microbiota of healthy mice are highly variable, cluster by environment, and reflect variation in baseline lung innate immunity. Am J Respir Crit Care Med. (2018) 198:497–508. doi: 10.1164/rccm.201711-2180OC
61. Dickson RP, Martinez FJ, Huffnagle GB. The role of the microbiome in exacerbations of chronic lung diseases. Lancet. (2014) 384:691–702. doi: 10.1016/S0140-6736(14)61136-3
62. Segal LN, Alekseyenko AV, Clemente JC, Kulkarni R, Wu B, Chen H, et al. Enrichment of lung microbiome with supraglottic taxa is associated with increased pulmonary inflammation. Microbiome. (2013) 1:19. doi: 10.1186/2049-2618-1-19
63. Venkataraman A, Bassis CM, Beck JM, Young VB, Curtis JL, Huffnagle GB, et al. Application of a neutral community model to assess structuring of the human lung microbiome. MBio. (2015) 6:e02284–14. doi: 10.1128/mBio.02284-14
64. Li Y, He J, He Z, Zhou Y, Yuan M, Xu X, et al. Phylogenetic and functional gene structure shifts of the oral microbiomes in periodontitis patients. ISME J. (2014) 8:1879–91. doi: 10.1038/ismej.2014.28
65. Beck JM, Schloss PD, Venkataraman A, Twigg H, Jablonski KA, Bushman FD, et al. Multicenter comparison of lung and oral microbiomes of HIV-infected and HIV-uninfected individuals. Am J Respir Crit Care Med. (2015) 192:1335–44. doi: 10.1164/rccm.201501-0128OC
66. Rawson TM, Moore LSP, Zhu N, Ranganathan N, Skolimowska K, Gilchrist M, et al. Bacterial and fungal coinfection in individuals with coronavirus: a rapid review to support COVID-19 antimicrobial prescribing. Clin Infect Dis. (2020) 71:2459–68. doi: 10.1093/cid/ciaa530
67. Ai JW, Zhang HC, Xu T, Wu J, Zhu M, Yu YQ, et al. Optimizing diagnostic strategy for novel coronavirus pneumonia, a multi-center study in Eastern China. medRxiv [Preprint]. (2020). doi: 10.1101/2020.02.13.20022673
68. Wu C, Guo S, Niu Y, Yang L, Liu B, Jiang N, et al. Heat-shock protein 60 of Porphyromonas gingivalis may induce dysfunction of human umbilical endothelial cells via regulation of endothelial-nitric oxide synthase and vascular endothelial-cadherin. Biomed Reports. (2016) 5:243–7. doi: 10.3892/br.2016.693
69. Giacobbe DR, Battaglini D, Enrile EM, Dentone C, Vena A, Robba C, et al. Incidence and prognosis of ventilator-associated pneumonia in critically ill patients with COVID-19: a multicenter study. J Clin Med. (2021) 10:555. doi: 10.3390/jcm10040555
70. Maes M, Higginson E, Pereira-Dias J, Curran MD, Parmar S, Khokhar F, et al. Ventilator-associated pneumonia in critically ill patients with COVID-19. Crit Care. (2021) 25:130. doi: 10.1186/s13054-021-03560-2
71. Rouzé A, Martin-Loeches I, Povoa P, Makris D, Artigas A, Bouchereau M, et al. Relationship between SARS-CoV-2 infection and the incidence of ventilator-associated lower respiratory tract infections: a European multicenter cohort study. Intensive Care Med. (2021) 47:188–98. doi: 10.1007/s00134-020-06323-9
72. Fernández-Barat L, López-Aladid R, Torres A. Reconsidering ventilator-associated pneumonia from a new dimension of the lung microbiome. EBioMedicine. (2020) 60:102995. doi: 10.1016/j.ebiom.2020.102995
73. Bao L, Zhang C, Dong J, Zhao L, Li Y, Sun J. Oral microbiome and SARS-CoV-2: beware of lung co-infection. Front Microbiol. (2020) 11:1840. doi: 10.3389/fmicb.2020.01840
74. Biagi E, Franceschi C, Rampelli S, Severgnini M, Ostan R, Turroni S, et al. Gut microbiota and extreme longevity. Curr Biol. (2016) 26:1480–5. doi: 10.1016/j.cub.2016.04.016
75. Muscogiuri G, Pugliese G, Barrea L, Savastano S, Colao A. Commentary: obesity: the “Achilles heel” for COVID-19? Metabolism. (2020) 108:154251. doi: 10.1016/j.metabol.2020.154251
76. De Maio F, Lo Cascio E, Babini G, Sali M, Della Longa S, Tilocca B, et al. Improved binding of SARS-CoV-2 Envelope protein to tight junction-associated PALS1 could play a key role in COVID-19 pathogenesis. Microbes Infect. (2020) 22:592–7. doi: 10.1016/j.micinf.2020.08.006
77. Libby P. Inflammatory mechanisms: the molecular basis of inflammation and disease. Nutr Rev. (2008) 65:S140–6. doi: 10.1111/j.1753-4887.2007.tb00352.x
78. Sanyaolu A, Okorie C, Marinkovic A, Patidar R, Younis K, Desai P, et al. Comorbidity and its impact on patients with COVID-19. SN Compr Clin Med. (2020) 2:1069–76. doi: 10.1007/s42399-020-00363-4
79. Naqvi S, Asar TO, Kumar V, Al-Abbasi FA, Alhayyani S, Kamal MA, et al. A cross-talk between gut microbiome, salt and hypertension. Biomed Pharmacother. (2021) 134:111156. doi: 10.1016/j.biopha.2020.111156
80. Umirah F, Neoh CF, Ramasamy K, Lim SM. Differential gut microbiota composition between type 2 diabetes mellitus patients and healthy controls: a systematic review. Diabetes Res Clin Pract. (2021) 173:108689. doi: 10.1016/j.diabres.2021.108689
81. Garg S, Kim L, Whitaker M, O'Halloran A, Cummings C, Holstein R, et al. Hospitalization rates and characteristics of patients hospitalized with laboratory-confirmed coronavirus disease 2019 — COVID-NET, 14 States, March 1–30, 2020. MMWR Morb Mortal Wkly Rep. (2020) 69:458–64. doi: 10.15585/mmwr.mm6915e3
82. Chiang TC, Huang MS, Lu PL, Huang ST, Lin YC. The effect of oral care intervention on pneumonia hospitalization, Staphylococcus aureus distribution, and salivary bacterial concentration in Taiwan nursing home residents: a pilot study. BMC Infect Dis. (2020) 20:374. doi: 10.1186/s12879-020-05061-z
83. Vena A, Taramasso L, Di Biagio A, Mikulska M, Dentone C, De Maria A, et al. Prevalence and clinical significance of persistent viral shedding in hospitalized adult patients with SARS-CoV-2 infection: a prospective observational study. Infect Dis Ther. (2021) 10:387–98. doi: 10.1007/s40121-020-00381-8
84. Carrouel F, Gonçalves LS, Conte MP, Campus G, Fisher J, Fraticelli L, et al. Antiviral activity of reagents in mouth rinses against SARS-CoV-2. J Dent Res. (2021) 100:124–32. doi: 10.1177/0022034520967933
85. Kumar PS. From focal sepsis to periodontal medicine: a century of exploring the role of the oral microbiome in systemic disease. J Physiol. (2017) 595:465–76. doi: 10.1113/JP272427
86. de Carvalho Baptista IM, Martinho FC, Nascimento GG, da Rocha Santos CE, Prado RF do, Valera MC. Colonization of oropharynx and lower respiratory tract in critical patients: risk of ventilator-associated pneumonia. Arch Oral Biol. (2018) 85:64–9. doi: 10.1016/j.archoralbio.2017.09.029
87. Son M, Jo S, Lee JS, Lee DH. Association between oral health and incidence of pneumonia: a population-based cohort study from Korea. Sci Rep. (2020) 10:9576. doi: 10.1038/s41598-020-66312-2
88. Khaky B, Yazdannik A, Mahjoubipour H. Evaluating the efficacy of nanosil mouthwash on the preventing pulmonary infection in intensive care unit: a randomized clinical trial. Med Arch. (2018) 72:206–9. doi: 10.5455/medarh.2018.72.206-209
89. Nobahar M, Razavi MR, Malek F, Ghorbani R. Effects of hydrogen peroxide mouthwash on preventing ventilator-associated pneumonia in patients admitted to the intensive care unit. Brazilian J Infect Dis. (2016) 20:444–50. doi: 10.1016/j.bjid.2016.06.005
90. Muñoz-Basagoiti J, Perez-Zsolt D, León R, Blanc V, Gispert J, Clotet B, et al. Cetylpyridinium chloride-containing mouthwashes reduce in vitro SARS-CoV-2 infectivity. bioRxiv [Preprint]. (2020). doi: 10.1101/2020.12.21.423779
91. Kampf G, Todt D, Pfaender S, Steinmann E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J Hosp Infect. (2020) 104:246–51. doi: 10.1016/j.jhin.2020.01.022
92. Caruso AA, Del Prete A, Lazzarino AI, Capaldi R, Grumetto L. Might hydrogen peroxide reduce the hospitalization rate and complications of SARS-CoV-2 infection? Infect Control Hosp Epidemiol. (2020) 41:1360–1. doi: 10.1017/ice.2020.170
94. Muzlovic I, Perme J, Stubljar D. Orotracheal tube as a risk factor for lower respiratory tract infection: preliminary data from a randomised trial. Wien Klin Wochenschr. (2018) 130:328–34. doi: 10.1007/s00508-017-1304-x
95. Battaglini D, Robba C, Caiffa S, Ball L, Brunetti I, Loconte M, et al. Chest physiotherapy: an important adjuvant in critically ill mechanically ventilated patients with COVID-19. Resp Physiol Neurobiol. (2020) 282:103529. doi: 10.1016/j.resp.2020.103529
96. Pozuelo-Carrascosa DP, Herráiz-Adillo Á, Alvarez-Bueno C, Añón JM, Martínez-Vizcaíno V, Cavero-Redondo I. Subglottic secretion drainage for preventing ventilator-associated pneumonia: an overview of systematic reviews and an updated meta-analysis. Eur Respir Rev. (2020) 29:190107. doi: 10.1183/16000617.0107-2019
97. Ntoumenopoulos G, Presneill J, McElholum M, Cade J. Chest physiotherapy for the prevention of ventilator-associated pneumonia. Intensive Care Med. (2002) 28:850–6. doi: 10.1007/s00134-002-1342-2
98. Wang MY, Pan L, Hu XJ. Chest physiotherapy for the prevention of ventilator-associated pneumonia: a meta-analysis. Am J Infect Control. (2019) 47:755–60. doi: 10.1016/j.ajic.2018.12.015
99. Li Bassi G, Prats RG, Artigas A, Xiol EA, Marti JD, Ranzani OT, et al. Appraisal of systemic inflammation and diagnostic markers in a porcine model of VAP: secondary analysis from a study on novel preventive strategies. Intensive Care Med Exp. (2018) 6:42. doi: 10.1186/s40635-018-0206-1
100. Meli A, Barbeta Viñas E, Battaglini D, Li Bassi G, Yang H, Yang M, et al. Lateral position during severe mono-lateral pneumonia: an experimental study. Sci Rep. (2020) 10:19372. doi: 10.1038/s41598-020-76216-w
101. Robba C, Battaglini D, Ball L, Patroniti N, Loconte M, Brunetti I, et al. Distinct phenotypes require distinct respiratory management strategies in severe COVID-19. Respir Physiol Neurobiol. (2020) 279:103455. doi: 10.1016/j.resp.2020.103455
102. Schifino G, de Grauw AJ, Daniele F, Comellini V, Fasano L, Pisani L. Effects of prone and lateral position in non-intubated patients with 2019 Novel Coronavirus (COVID-19) pneumonia. Pulmonology. (2020) 27:167–71. doi: 10.1016/j.pulmoe.2020.10.015
103. Retucci M, Aliberti S, Ceruti C, Santambrogio M, Tammaro S, Cuccarini F, et al. Prone and lateral positioning in spontaneously breathing patients with COVID-19 pneumonia undergoing noninvasive helmet CPAP treatment. Chest. (2020) 158:2431–5. doi: 10.1016/j.chest.2020.07.006
104. Wang L, Li X, Yang Z, Tang X, Yuan Q, Deng L, et al. Semi-recumbent position versus supine position for the prevention of ventilator-associated pneumonia in adults requiring mechanical ventilation. Cochrane Database Syst Rev. (2016) 1:CD009946. doi: 10.1002/14651858.CD009946.pub2
105. Li Bassi G, Panigada M, Ranzani OT, Zanella A, Berra L, Cressoni M, et al. Randomized, multicenter trial of lateral Trendelenburg versus semirecumbent body position for the prevention of ventilator-associated pneumonia. Intensive Care Med. (2017) 43:1572–84. doi: 10.1007/s00134-017-4858-1
106. Ayzac L, Girard R, Baboi L, Beuret P, Rabilloud M, Richard JC, et al. Ventilator-associated pneumonia in ARDS patients: the impact of prone positioning. A secondary analysis of the PROSEVA trial. Intensive Care Med. (2016) 42:871–8. doi: 10.1007/s00134-015-4167-5
107. Mounier R, Adrie C, Francais A, Garrouste-Orgeas M, Cheval C, Allaouchiche B, et al. Study of prone positioning to reduce ventilator-associated pneumonia in hypoxaemic patients. Eur Respir J. (2010) 35:795–804. doi: 10.1183/09031936.00057509
108. Cox MJ, Loman N, Bogaert D, O'Grady J. Co-infections: potentially lethal and unexplored in COVID-19. Lancet Microbe. (2020) 1:e11. doi: 10.1016/S2666-5247(20)30009-4
109. Preidis GA, Versalovic J. Targeting the human microbiome with antibiotics, probiotics, and prebiotics: gastroenterology enters the metagenomics era. Gastroenterology. (2009) 136:2015–31. doi: 10.1053/j.gastro.2009.01.072
110. Weber DJ, Raasch R, Rutala WA. Nosocomial infections in the ICU. Chest. (1999) 115:34S−41S. doi: 10.1378/chest.115.suppl_1.34S
111. Zaher S. Nutrition and the gut microbiome during critical illness: a new insight of nutritional therapy. Saudi J Gastroenterol. (2020) 26:290–8. doi: 10.4103/sjg.SJG_352_20
112. Langford BJ, So M, Raybardhan S, Leung V, Soucy JPR, Westwood D, et al. Antibiotic prescribing in patients with COVID-19: rapid review and meta-analysis. Clin Microbiol Infect. (2021) 27:520–31. doi: 10.1016/j.cmi.2020.12.018
113. Søgaard KK, Baettig V, Osthoff M, Marsch S, Leuzinger K, Schweitzer M, et al. Community-acquired and hospital-acquired respiratory tract infection and bloodstream infection in patients hospitalized with COVID-19 pneumonia. J Intensive Care. (2021) 9:10. doi: 10.1186/s40560-021-00526-y
114. Furtado RHM, Berwanger O, Fonseca HA, Corrêa TD, Ferraz LR, Lapa MG, et al. Azithromycin in addition to standard of care versus standard of care alone in the treatment of patients admitted to the hospital with severe COVID-19 in Brazil (COALITION II): a randomised clinical trial. Lancet. (2020) 396:959–67. doi: 10.1016/S0140-6736(20)31862-6
115. Cavalcanti AB, Zampieri FG, Rosa RG, Azevedo LCP, Veiga VC, Avezum A, et al. Hydroxychloroquine with or without azithromycin in mild-to-moderate covid-19. N Engl J Med. (2020) 383:2041–52. doi: 10.1056/NEJMoa2019014
116. Rosenberg ES, Dufort EM, Udo T, Wilberschied LA, Kumar J, Tesoriero J, et al. Association of treatment with hydroxychloroquine or azithromycin with in-hospital mortality in patients with COVID-19 in New York State. JAMA. (2020) 323:2493–502. doi: 10.1001/jama.2020.8630
117. Gyselinck I, Liesenborgs L, Landeloos E, Belmans A, Verbeke G, Verhamme P, et al. Direct antivirals working against the novel coronavirus: azithromycin (DAWn-AZITHRO), a randomized, multicenter, open-label, adaptive, proof-of-concept clinical trial of new antivirals working against SARS-CoV-2—azithromycin trial. Trials. (2021) 22:126. doi: 10.1186/s13063-021-05033-x
118. Page V. Sedation in mechanically ventilated patients with COVID-19. Lancet Respir Med. (2021) 9:218–9. doi: 10.1016/S2213-2600(20)30570-1
119. Pun BT, Badenes R, Heras La Calle G, Orun OM, Chen W, Raman R, et al. Prevalence and risk factors for delirium in critically ill patients with COVID-19 (COVID-D): a multicentre cohort study. Lancet Respir Med. (2021) 9:239–50. doi: 10.1016/S2213-2600(20)30552-X
120. Rueda-Ruzafa L, Cruz F, Cardona D, Hone AJ, Molina-Torres G, Sánchez-Labraca N, et al. Opioid system influences gut-brain axis: dysbiosis and related alterations. Pharmacol Res. (2020) 159:104928. doi: 10.1016/j.phrs.2020.104928
121. Bardi T, Pintado V, Gomez-Rojo M, Escudero-Sanchez R, Azzam Lopez A, Diez-Remesal Y, et al. Nosocomial infections associated to COVID-19 in the intensive care unit: clinical characteristics and outcome. Eur J Clin Microbiol Infect Dis. (2021) 40:495–502. doi: 10.1007/s10096-020-04142-w
122. Sarkodie EK, Zhou S, Baidoo SA, Chu W. Influences of stress hormones on microbial infections. Microb Pathog. (2019) 131:270–6. doi: 10.1016/j.micpath.2019.04.013
123. Freestone PPE, Haigh RD, Lyte M. Catecholamine inotrope resuscitation of antibiotic-damaged staphylococci and its blockade by specific receptor antagonists. J Infect Dis. (2008) 197:1044–52. doi: 10.1086/529202
124. Sandrini S, Aldriwesh M, Alruways M, Freestone P. Microbial endocrinology: host–bacteria communication within the gut microbiome. J Endocrinol. (2015) 225:R21–34. doi: 10.1530/JOE-14-0615
125. Lee TC, Goodwin Wilson M, Lawandi A, McDonald EG. Proton pump inhibitors versus histamine-2 receptor antagonists likely increase mortality in critical care: an updated meta-analysis. Am J Med. (2020) 134:e184–8. doi: 10.1016/j.amjmed.2020.08.004
126. Wang Y, Ge L, Ye Z, Siemieniuk RA, Reintam Blaser A, Wang X, et al. Efficacy and safety of gastrointestinal bleeding prophylaxis in critically ill patients: an updated systematic review and network meta-analysis of randomized trials. Intensive Care Med. (2020) 46:1987–2000. doi: 10.1007/s00134-020-06209-w
127. Young PJ, Bagshaw SM, Forbes AB, Nichol AD, Wright SE, Bailey M, et al. Effect of stress ulcer prophylaxis with proton pump inhibitors vs histamine-2 receptor blockers on in-hospital mortality among ICU patients receiving invasive mechanical ventilation. JAMA. (2020) 323:611–3. doi: 10.1001/jama.2019.22436
128. Takashima S, Tanaka F, Kawaguchi Y, Usui Y, Fujimoto K, Nadatani Y, et al. Proton pump inhibitors enhance intestinal permeability via dysbiosis of gut microbiota under stressed conditions in mice. Neurogastroenterol Motil. (2020) 32:e13841. doi: 10.1111/nmo.13841
129. Wang Y, Ye Z, Ge L, Siemieniuk RAC, Wang X, Wang Y, et al. Efficacy and safety of gastrointestinal bleeding prophylaxis in critically ill patients: systematic review and network meta-analysis. BMJ. (2020) 368:l6744. doi: 10.1136/bmj.l6744
130. Horvath A, Leber B, Feldbacher N, Steinwender M, Komarova I, Rainer F, et al. The effects of a multispecies synbiotic on microbiome-related side effects of long-term proton pump inhibitor use: a pilot study. Sci Rep. (2020) 10:2723. doi: 10.1038/s41598-020-59550-x
131. Luxenburger H, Sturm L, Biever P, Rieg S, Duerschmied D, Schultheiss M, et al. Treatment with proton pump inhibitors increases the risk of secondary infections and ARDS in hospitalized patients with COVID-19: coincidence or underestimated risk factor? J Intern Med. (2021) 289:121–4. doi: 10.1111/joim.13121
132. Ramachandran P, Perisetti A, Gajendran M, Jean-Louis F, Bansal P, Dwivedi AK, et al. Pre-hospitalization proton pump inhibitor use and clinical outcomes in COVID-19. Eur J Gastroenterol Hepatol. (2020). doi: 10.1097/MEG.0000000000002013. [Epub ahead of print].
133. Kow CS, Hasan SS. Use of proton pump inhibitors and risk of adverse clinical outcomes from COVID-19: a meta-analysis. J Intern Med. (2021) 289:125–8. doi: 10.1111/joim.13183
134. Alhazzani W, Evans L, Alshamsi F, Møller MH, Ostermann M, Prescott HC, et al. Surviving sepsis campaign guidelines on the management of adults with coronavirus disease 2019 (COVID-19) in the ICU. Crit Care Med. (2021) 49:e219–34. doi: 10.1097/CCM.0000000000004899
135. Sterne JAC, Murthy S, Diaz JV, Slutsky AS, Villar J, Angus DC, et al. Association between administration of systemic corticosteroids and mortality among critically ill patients with COVID-19. JAMA. (2020) 324:1330–41. doi: 10.1001/jama.2020.17023
136. Horby P, Shen Lim W, Emberson JR, Mafham M, Bell JL, Linsell L, et al. Dexamethasone in hospitalized patients with covid-19 — preliminary report. N Engl J Med. (2020) 324:1330–41. doi: 10.1101/2020.06.22.20137273
137. Ünsal H, Balkaya M, Ünsal C, Biyik H, Başbülbül G, Poyrazoglu E. The short-term effects of different doses of dexamethasone on the numbers of some bacteria in the ileum. Dig Dis Sci. (2008) 53:1842–5. doi: 10.1007/s10620-007-0089-6
138. Tomazini BM, Maia IS, Cavalcanti AB, Berwanger O, Rosa RG, Veiga VC. Effect of dexamethasone on days alive and ventilator-free in patients with moderate or severe acute respiratory distress syndrome and COVID-19. JAMA. (2020) 324:1307. doi: 10.1001/jama.2020.17021
139. Gagliardi A, Totino V, Cacciotti F, Iebba V, Neroni B, Bonfiglio G, et al. Rebuilding the gut microbiota ecosystem. Int J Environ Res Public Health. (2018) 15:1679. doi: 10.3390/ijerph15081679
140. Zhao J, Li L, Chen C, Zhang G, Cui W, Tian B. Do probiotics help prevent ventilator-associated pneumonia in critically ill patients? A systematic review with meta-analysis. ERJ Open Res. (2021) 7:00302–02020. doi: 10.1183/23120541.00302-2020
141. Plaza-Diaz J, Ruiz-Ojeda FJ, Gil-Campos M, Gil A. Mechanisms of action of probiotics. Adv Nutr. (2019) 10:S49–66. doi: 10.1093/advances/nmy063
142. López P, González-Rodríguez I, Gueimonde M, Margolles A, Suárez A. Immune response to bifidobacterium bifidum strains support Treg/Th17 plasticity. PLoS ONE. (2011) 6:e24776. doi: 10.1371/journal.pone.0024776
143. de Vrese M, Schrezenmeir J. Probiotics, Prebiotics, and Synbiotics. Adv Biochem Eng Biotechnol. (2008) 111:1–66. doi: 10.1007/10_2008_097
144. Hemarajata P, Versalovic J. Effects of probiotics on gut microbiota: mechanisms of intestinal immunomodulation and neuromodulation. Therap Adv Gastroenterol. (2013) 6:39–51. doi: 10.1177/1756283X12459294
145. Walton GE, Gibson GR, Hunter KA. Mechanisms linking the human gut microbiome to prophylactic and treatment strategies for COVID-19. Br J Nutr. (2020). doi: 10.1017/S0007114520003980. [Epub ahead of print].
146. Di Pierro F. A possible probiotic (S. salivarius K12) approach to improve oral and lung microbiotas and raise defenses against SARS-CoV-2. Minerva Med. (2020) 111:281–3. doi: 10.23736/S0026-4806.20.06570-2
147. Verma A, Xu K, Du T, Zhu P, Liang Z, Liao S, et al. Expression of human ACE2 in lactobacillus and beneficial effects in diabetic retinopathy in mice. Mol Ther Methods Clin Dev. (2019) 14:161–70. doi: 10.1016/j.omtm.2019.06.007
148. Minato S, Hirai K, Morino J, Kaneko S, Yanai K, Mutsuyoshi Y, et al. Factors associated with uremic pruritus in patients undergoing peritoneal dialysis. Int J Nephrol Renovasc Dis. (2020) 13:1–9. doi: 10.2147/IJNRD.S224871
149. Din AU, Mazhar M, Waseem M, Ahmad W, Bibi A, Hassan A, et al. SARS-CoV-2 microbiome dysbiosis linked disorders and possible probiotics role. Biomed Pharmacother. (2021) 133:110947. doi: 10.1016/j.biopha.2020.110947
150. Patra S, Saxena S, Sahu N, Pradhan B, Roychowdhury A. Systematic network and meta-analysis on the antiviral mechanisms of probiotics: a preventive and treatment strategy to mitigate SARS-CoV-2 infection. Probiotics Antimicrob Proteins. (2021). doi: 10.1007/s12602-021-09748-w. [Epub ahead of print].
151. Liu F, Ye S, Zhu X, He X, Wang S, Li Y, et al. Gastrointestinal disturbance and effect of fecal microbiota transplantation in discharged COVID-19 patients. J Med Case Rep. (2021) 15:60. doi: 10.1186/s13256-020-02583-7
152. Ianiro G, Mullish BH, Kelly CR, Sokol H, Kassam Z, Ng S, et al. Screening of faecal microbiota transplant donors during the COVID-19 outbreak: suggestions for urgent updates from an international expert panel. Lancet Gastroenterol Hepatol. (2020) 5:430–2. doi: 10.1016/S2468-1253(20)30082-0
153. Mullish BH, McDonald JAK, Pechlivanis A, Allegretti JR, Kao D, Barker GF, et al. Microbial bile salt hydrolases mediate the efficacy of faecal microbiota transplant in the treatment of recurrent Clostridioides difficile infection. Gut. (2019) 68:1791–800. doi: 10.1136/gutjnl-2018-317842
154. Bäumler AJ, Sperandio V. Interactions between the microbiota and pathogenic bacteria in the gut. Nature. (2016) 535:85–93. doi: 10.1038/nature18849
155. Luo L, Han W, Du J, Yang X, Duan M, Xu C, et al. Chenodeoxycholic acid from bile inhibits influenza a virus replication via blocking nuclear export of viral ribonucleoprotein complexes. Molecules. (2018) 23:3315. doi: 10.3390/molecules23123315
156. Bojanova DP, Bordenstein SR. Fecal transplants: what is being transferred? PLOS Biol. (2016) 14:e1002503. doi: 10.1371/journal.pbio.1002503
157. Wernerman J, Christopher KB, Annane D, Casaer MP, Coopersmith CM, Deane AM, et al. Metabolic support in the critically ill: a consensus of 19. Crit Care. (2019) 23:318. doi: 10.1186/s13054-019-2597-0
158. Zusman O, Theilla M, Cohen J, Kagan I, Bendavid I, Singer P. Resting energy expenditure, calorie and protein consumption in critically ill patients: a retrospective cohort study. Crit Care. (2016) 20:367. doi: 10.1186/s13054-016-1538-4
159. Needham DM, Davidson J, Cohen H, Hopkins RO, Weinert C, Wunsch H, et al. Improving long-term outcomes after discharge from intensive care unit: Report from a stakeholders' conference. Crit Care Med. (2012) 40:502–9. doi: 10.1097/CCM.0b013e318232da75
160. Van Zanten ARH, De Waele E, Wischmeyer PE. Nutrition therapy and critical illness: practical guidance for the icu, post-icu, and long-term convalescence phases. Crit Care. (2019) 23:368. doi: 10.1186/s13054-019-2657-5
161. De Filippo C, Cavalieri D, Di Paola M, Ramazzotti M, Poullet JB, Massart S, et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci USA. (2010) 107:14691–6. doi: 10.1073/pnas.1005963107
162. De Filippis F, Pellegrini N, Vannini L, Jeffery IB, La Storia A, Laghi L, et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut. (2016) 65:1812–21. doi: 10.1136/gutjnl-2015-309957
163. Zuo T, Liu Q, Zhang F, Lui GCY, Tso EY, Yeoh YK, et al. Depicting SARS-CoV-2 faecal viral activity in association with gut microbiota composition in patients with COVID-19. Gut. (2020) 70:276–84. doi: 10.1136/gutjnl-2020-322294
164. Morrison DJ, Preston T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes. (2016) 7:189–200. doi: 10.1080/19490976.2015.1134082
165. Rowland I, Gibson G, Heinken A, Scott K, Swann J, Thiele I, et al. Gut microbiota functions: metabolism of nutrients and other food components. Eur J Nutr. (2018) 57:1–24. doi: 10.1007/s00394-017-1445-8
166. Martindale R, Patel JJ, Taylor B, Warren M, McClave SA. Nutrition therapy in the patient with COVID-19 disease requiring ICU Care (2020). Available online at: https://www.nutritioncare.org/uploadedFiles/Documents/Guidelines_and_Clinical_Resources/Nutrition%20Therapy%20COVID-19_SCCM-ASPEN.pdf (accessed May 23, 2021).
167. Barazzoni R, Bischoff SC, Krznaric Z, Pirlich M, Singer P. ESPEN expert statements and practical guidance for nutritional management of individuals with SARS-CoV-2 infection. Clin Nutr. (2020) 39:1631–8. doi: 10.1016/j.clnu.2020.03.022
168. Cereda E, Guzzardella A, Klersy C, Belliato M, Pellegrini A, Sciutti F, et al. Early caloric deficit is associated with a higher risk of death in invasive ventilated COVID-19 patients. Clin Nutr. (2021). doi: 10.1016/j.clnu.2021.02.020. [Epub ahead of print].
169. Chapman M, Peake SL, Bellomo R, Davies A, Deane A, Horowitz M, et al. Energy-dense versus routine enteral nutrition in the critically ill. N Engl J Med. (2018) 379:1823–34. doi: 10.1056/NEJMoa1811687
170. Singer P, Blaser AR, Berger MM, Alhazzani W, Calder PC, Casaer MP, et al. ESPEN guideline on clinical nutrition in the intensive care unit. Clin Nutr. (2019) 38:48–79. doi: 10.1016/j.clnu.2018.08.037
171. Alsharif DJ, Alsharif FJ, Aljuraiban GS, Abulmeaty MMA. Effect of supplemental parenteral nutrition versus enteral nutrition alone on clinical outcomes in critically ill adult patients: a systematic review and meta-analysis of randomized controlled trials. Nutrients. (2020) 12:2968. doi: 10.3390/nu12102968
172. Cintoni M, Rinninella E, Annetta MG, Mele MC. Nutritional management in hospital setting during SARS-CoV-2 pandemic: a real-life experience. Eur J Clin Nutr. (2020) 74:846–7. doi: 10.1038/s41430-020-0625-4
173. Pan L, Mu M, Yang P, Sun Y, Wang R, Yan J, et al. Clinical characteristics of COVID-19 patients with digestive symptoms in Hubei, China. Am J Gastroenterol. (2020) 115:766–73. doi: 10.14309/ajg.0000000000000620
174. Preiser JC, van Zanten ARH, Berger MM, Biolo G, Casaer MP, Doig GS, et al. Metabolic and nutritional support of critically ill patients: consensus and controversies. Crit Care. (2015) 19:35. doi: 10.1186/s13054-015-0737-8
175. Puthucheary ZA, Rawal J, McPhail M, Connolly B, Ratnayake G, Chan P, et al. Acute skeletal muscle wasting in critical illness. JAMA. (2013) 310:1591–600. doi: 10.1001/jama.2013.278481
176. van Zanten ARH, Petit L, De Waele J, Kieft H, de Wilde J, van Horssen P, et al. Very high intact-protein formula successfully provides protein intake according to nutritional recommendations in overweight critically ill patients: a double-blind randomized trial. Crit Care. (2018) 22:156. doi: 10.1186/s13054-018-2070-5
177. Looijaard WGPM, Dekker IM, Beishuizen A, Girbes ARJ, Oudemans-van Straaten HM, Weijs PJM. Early high protein intake and mortality in critically ill ICU patients with low skeletal muscle area and -density. Clin Nutr. (2019) 39:2192–201. doi: 10.1016/j.clnu.2019.09.007
178. Weijs PJM, Mogensen KM, Rawn JD, Christopher KB. Protein intake, nutritional status and outcomes in icu survivors: a single center cohort study. J Clin Med. (2019) 8:43. doi: 10.3390/jcm8010043
179. Xu Z, Tang Y, Huang Q, Fu S, Li X, Lin B, et al. Systematic review and subgroup analysis of the incidence of acute kidney injury (AKI) in patients with COVID-19. BMC Nephrol. (2021) 22:52. doi: 10.1186/s12882-021-02244-x
180. Ng WH, Tipih T, Makoah NA, Vermeulen JG, Goedhals D, Sempa JB, et al. Comorbidities in SARS-CoV-2 Patients: a Systematic Review and Meta-Analysis. MBio. (2021) 12:e03647–20. doi: 10.1128/mBio.03647-20
181. O'Brien KD, Brehm BJ, Seeley RJ, Bean J, Wener MH, Daniels S, et al. Diet-induced weight loss is associated with decreases in plasma serum amyloid A and C-reactive protein independent of dietary macronutrient composition in obese subjects. J Clin Endocrinol Metab. (2005) 90:2244–9. doi: 10.1210/jc.2004-1011
182. Semba RD, Tang AM. Micronutrients and the pathogenesis of human immunodeficiency virus infection. Br J Nutr. (1999) 81:181–9. doi: 10.1017/S0007114599000379
183. National Heart Lung Blood Institute PETAL Clinical Trials Network Ginde A, Brower R, Caterino J, Finck L, Banner-Goodspeed V. Early high-dose vitamin D 3 for critically ill, vitamin D–deficient patients. N Engl J Med. (2019) 381:2529–40. doi: 10.1056/NEJMoa1911124
184. Maghbooli Z, Sahraian MA, Ebrahimi M, Pazoki M, Kafan S, Tabriz HM, et al. Vitamin D sufficiency, a serum 25-hydroxyvitamin D at least 30 ng/mL reduced risk for adverse clinical outcomes in patients with COVID-19 infection. PLoS ONE. (2020) 15:e0239799. doi: 10.1371/journal.pone.0239799
185. Wu C, Chen X, Cai Y, Xia J, Zhou X, Xu S, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. (2020) 180:1–11. doi: 10.1001/jamainternmed.2020.0994
186. Dushianthan A, Cusack R, Burgess VA, Grocott MPW, Calder P. Immunonutrition for adults with ARDS: Results from a cochrane systematic review and meta-analysis. Respir Care. (2020) 65:99–110. doi: 10.4187/respcare.06965
187. Holford P, Carr AC, Jovic TH, Ali SR, Whitaker IS, Marik PE. Vitamin C—An adjunctive therapy for respiratory infection, sepsis and COVID-19. Nutrients. (2020) 12:3760. doi: 10.3390/nu12123760
188. Derbyshire E, Delange J. COVID-19: is there a role for immunonutrition, particularly in the over 65s? BMJ Nutr Prev Heal. (2020) 3:100–5. doi: 10.1136/bmjnph-2020-000071
189. Grimble RF. Educational Paper Basics in clinical nutrition: immunonutrition-Nutrients which influence immunity: Effect and mechanism of action. e-SPEN. (2009) 4:e10–3. doi: 10.1016/j.eclnm.2008.07.015
190. Grimble RF. Immunonutrition. Curr Opin Gastroenterol. (2005) 21:216–22. doi: 10.1097/01.mog.0000153360.90653.82
191. Pontes-Arruda A, DeMichele S, Seth A, Singer P. The use of an inflammation-modulating diet in patients with acute lung injury or acute respiratory distress syndrome: a meta-analysis of outcome data. JPEN J Parenter Enter Nutr. (2008) 32:596–605. doi: 10.1177/0148607108324203
192. Rice TW, Wheeler AP, Thompson BT, DeBoisblanc BP, Steingrub J, Rock P. Enteral omega-3 fatty acid, γ-linolenic acid, and antioxidant supplementation in acute lung injury. JAMA. (2011) 306:1574–81. doi: 10.1001/jama.2011.1435
193. Stapleton RD, Martin TR, Weiss NS, Crowley JJ, Gundel SJ, Nathens AB, et al. A phase II randomized placebo-controlled trial of omega-3 fatty acids for the treatment of acute lung injury. Crit Care Med. (2011) 39:1655–62. doi: 10.1097/CCM.0b013e318218669d
194. Zhu D, Zhang Y, Li S, Gan L, Feng H, Nie W. Enteral omega-3 fatty acid supplementation in adult patients with acute respiratory distress syndrome: a systematic review of randomized controlled trials with meta-analysis and trial sequential analysis. Intensive Care Med. (2014) 40:504–12. doi: 10.1007/s00134-014-3244-5
195. Anez-Bustillos L, Dao DT, Finkelstein A, Pan A, Cho BS, Mitchell PD, et al. Metabolic and inflammatory effects of an ω-3 fatty acid–based eucaloric ketogenic diet in mice with endotoxemia. JPEN J Parenter Enter Nutr. (2019) 43:986–97. doi: 10.1002/jpen.1688
196. Heyland DK, MacDonald S, Keefe L, Drover JW. Total parenteral nutrition in the critically III patient: a meta- analysis. JAMA. (1998) 280:2013–9. doi: 10.1001/jama.280.23.2013
197. Anti-inflammatory/Antioxidant Oral Nutrition Supplementation in COVID-19. Clin Identifier NCT04323228. (2020). Available online at: https://clinicaltrials.gov/ct2/show/NCT04323228 (accessed April 28, 2020).
198. Heyland D, Muscedere J, Wischmeyer PE, Cook D, Jones G, Albert M, et al. A randomized trial of glutamine and antioxidants in critically ill patients. N Engl J Med. (2013) 368:1489–97. doi: 10.1056/NEJMoa1212722
199. Rosenbaum M, Hall KD, Guo J, Ravussin E, Mayer LS, Reitman ML, et al. Glucose and lipid homeostasis and inflammation in humans following an isocaloric ketogenic diet. Obesity. (2019) 27:971–81. doi: 10.1002/oby.22468
200. Martin-Mcgill KJ, Jackson CF, Bresnahan R, Levy RG, Cooper PN. Ketogenic diets for drug-resistant epilepsy. Cochrane Database Syst Rev. (2018) 2018:CD001903. doi: 10.1002/14651858.CD001903.pub4
201. Thakur KT, Probasco JC, Hocker SE, Roehl K, Henry B, Kossoff EH, et al. Ketogenic diet for adults in super-refractory status epilepticus. Neurology. (2014) 82:665–70. doi: 10.1212/WNL.0000000000000151
202. Park EG, Lee J, Lee J. The ketogenic diet for super-refractory status epilepticus patients in intensive care units. Brain Dev. (2019) 41:420–7. doi: 10.1016/j.braindev.2018.12.007
203. Boden G, Sargrad K, Homko C, Mozzoli M, Stein TP. Effect of a low-carbohydrate diet on appetite, blood glucose levels, and insulin resistance in obese patients with type 2 diabetes. Ann Intern Med. (2005) 142:403–11. doi: 10.7326/0003-4819-142-6-200503150-00006
204. Ryu S, Shchukina I, Youm YH, Qing H, Hilliard BK, Dlugos T, et al. Ketogenesis restrains aging-induced exacerbation of COVID in a mouse model. bioRxiv [Preprint]. (2020). doi: 10.1101/2020.09.11.294363
205. Keto-diet for Intubated Critical Care COVID-19. Clin Identifier NCT04358835. (2020). Available online at: https://www.clinicaltrials.gov/ct2/show/NCT04358835 (accessed April 28, 2020).
206. Wen H, Miao EA, Ting JPY. Mechanisms of NOD-like receptor-associated inflammasome activation. Immunity. (2013) 39:432–41. doi: 10.1016/j.immuni.2013.08.037
207. Scagnolari C, Trombetti S, Cicetti S, Antonelli S, Selvaggi C, Perrone L, et al. Severe acute respiratory syndrome coronavirus elicits a weak interferon response compared to traditional interferon-inducing viruses. Intervirology. (2008) 51:217–23. doi: 10.1159/000154258
208. Youm YH, Nguyen KY, Grant RW, Goldberg EL, Bodogai M, Kim D, et al. The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease. Nat Med. (2015) 21:263–9. doi: 10.1038/nm.3804
209. Sukkar SG, Bassetti M. Induction of ketosis as a potential therapeutic option to limit hyperglycemia and prevent cytokine storm in COVID-19. Nutrition. (2020) 79–80:110967. doi: 10.1016/j.nut.2020.110967
Keywords: microbiota, dysbiosis, ARDS, critical care, infection, COVID-19
Citation: Battaglini D, Robba C, Fedele A, Trancǎ S, Sukkar SG, Di Pilato V, Bassetti M, Giacobbe DR, Vena A, Patroniti N, Ball L, Brunetti I, Torres Martí A, Rocco PRM and Pelosi P (2021) The Role of Dysbiosis in Critically Ill Patients With COVID-19 and Acute Respiratory Distress Syndrome. Front. Med. 8:671714. doi: 10.3389/fmed.2021.671714
Received: 24 February 2021; Accepted: 12 May 2021;
Published: 04 June 2021.
Edited by:
Ru-Ping Dai, Central South University, ChinaReviewed by:
Suzana Erico Tanni, Saõ Paulo State University, BrazilBob Eikemans, Maasstad Ziekenhuis, Netherlands
Copyright © 2021 Battaglini, Robba, Fedele, Trancǎ, Sukkar, Di Pilato, Bassetti, Giacobbe, Vena, Patroniti, Ball, Brunetti, Torres Martí, Rocco and Pelosi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Denise Battaglini, battaglini.denise@gmail.com
†These authors share senior authorship
 Denise Battaglini
Denise Battaglini Chiara Robba
Chiara Robba Andrea Fedele1
Andrea Fedele1  Sebastian Trancǎ
Sebastian Trancǎ Matteo Bassetti
Matteo Bassetti Daniele Roberto Giacobbe
Daniele Roberto Giacobbe Antonio Vena
Antonio Vena Antoni Torres Martí
Antoni Torres Martí Patricia Rieken Macedo Rocco
Patricia Rieken Macedo Rocco Paolo Pelosi
Paolo Pelosi