- 1Department of Psychiatry, Faculty of Medicine, University of Geneva (UNIGE), Geneva, Switzerland
- 2Department of Psychiatry, ASO Santi Antonio e Biagio e Cesare Arrigo Hospital, Alessandria, Italy
- 3Faculty of Psychology, University of Geneva (UNIGE), Geneva, Switzerland
- 4Department of Psychiatry, Service of Liaison Psychiatry and Crisis Intervention (SPLIC), Geneva University Hospitals (HUG), Geneva, Switzerland
- 5Department of Neurosurgery, University and City of Health and Science Hospital, Turin, Italy
- 6Section of Psychiatry, Department of Neuroscience, Rehabilitation, Ophthalmology, Genetics, Maternal and Child Health (DINOGMI), University of Genova, Genova, Italy
- 7Department of Psychiatry, IRCCS Ospedale Policlinico San Martino, Genoa, Italy
- 8Mood Disorders Program, Tufts Medical Center, Boston, MA, United States
- 9Department of Neurosciences, Mental Health and Sensory Organs, Suicide Prevention Center, Sant’Andrea Hospital, Sapienza University of Rome, Rome, Italy
- 10Department of Microbiology and Immunology, Stanford University, Palo Alto, CA, United States
- 11Tranquis Therapeutics, Palo Alto, CA, United States
- 12Hong Kong University of Science and Technology, Hong Kong, China
Deep brain stimulation (DBS) is a very well-established and effective treatment for patients with extrapyramidal diseases. Despite its generally favorable clinical efficacy, some undesirable outcomes associated with DBS have been reported. Among such complications are incidences of suicidal ideation (SI) and behavior (SB) in patients undergoing this neurosurgical procedure. However, causal associations between DBS and increased suicide risk are not demonstrated and they constitute a debated issue. In light of these observations, the main objective of this work is to provide a comprehensive and unbiased overview of the literature on suicide risk in patients who received subthalamic nucleus (STN) and internal part of globus pallidum (GPi) DBS treatment. Additionally, putative mechanisms that might be involved in the development of SI and SB in these patients as well as caveats associated with these hypotheses are introduced. Finally, we briefly propose some clinical implications, including therapeutic strategies addressing these potential disease mechanisms. While a mechanistic connection between DBS and suicidality remains a controversial topic that requires further investigation, it is of critical importance to consider suicide risk as an integral component of candidate selection and post-operative care in DBS.
Introduction
Deep brain stimulation (DBS) has emerged as an effective therapy for patients with extrapyramidal disorders, particularly those with drug-refractory advanced Parkinson’s disease (PD). This neurosurgical procedure requires the implantation of electrodes to deliver electrical pulses from a neurostimulator into specific brain regions. Depending on various stimulatory parameters such as intensity (mA), desired frequency (Hz), and pulse duration (μs), DBS can differentially modulate neuronal activities to achieve desired therapeutic outcomes. Introduced more than two decades ago, this approach has been frequently employed to target the subthalamic nucleus (STN) and the internal globus pallidum (GPi) in PD (Pollak et al., 1993; Limousin et al., 1998). While STN has been the preferred DBS target with a greater impact on post-operative medication withdrawal (Moro et al., 2010; St George et al., 2015), GPi appears to be more programmable (easier to precisely target specific areas while avoiding off-targeting effects) due to its larger volume (Au et al., 2020). GPi DBS seems to have a more powerful anti-dyskinesia effect in comparison to STN DBS (Munhoz et al., 2014). Differences in efficacy between STN and GPi DBS have also been observed in studies of advanced PD with a particular emphasis on tremor, where the former provided enhanced motor improvement during the off-drug phase (Odekerken et al., 2016) and the latter resulted in less dyskinesia during the post-operative on-medication period (Tsuboi et al., 2020; Zhang et al., 2020). To date, DBS is the neurosurgical treatment of choice for drug-refractory PD with proven efficacy (Thobois et al., 2002; Herzog et al., 2003; Benabid et al., 2009; Schuepbach et al., 2013; Lezcano et al., 2016). The most profound effects of DBS are in the areas of rigidity (75% improvement) and akinesia (50% improvement), allowing a marked reduction in dopaminergic therapies, and consequentially, avoiding motor and non-motor complications (Benabid et al., 2009). While the generally high precision and efficacy of DBS represents a clinical success, its mechanism of action remains elusive (Denys et al., 2012; Chiken and Nambu, 2016; Herrington et al., 2016) with possible involvement in the modulation of primarily glutamatergic (STN-DBS) and GABAergic (GPi-DBS) neuronal circuits (Dostrovsky et al., 2000; Jakobs et al., 2019).
Some adverse outcomes in patients receiving this therapy have been reported. Notably, suicidal ideation (SI) and behavior (SB), including suicide attempts (SA) and completed suicides, have been observed but causal associations between DBS and increased suicide risk have not been established. Therefore, the primary goal of this work is to provide a concise and unbiased overview of studies on suicidality in patients receiving STN and GPi DBS. Additionally, putative mechanisms that might be involved in the development of suicidality in these patients as well as caveats associated with these hypotheses are introduced. Finally, we propose some clinical implications, including therapeutic strategies addressing these potential disease mechanisms to avoid the risk of suicidality in patients receiving DBS, with a particular emphasis on patient selection and neuropsychiatric post-operative care.
Literature Overview
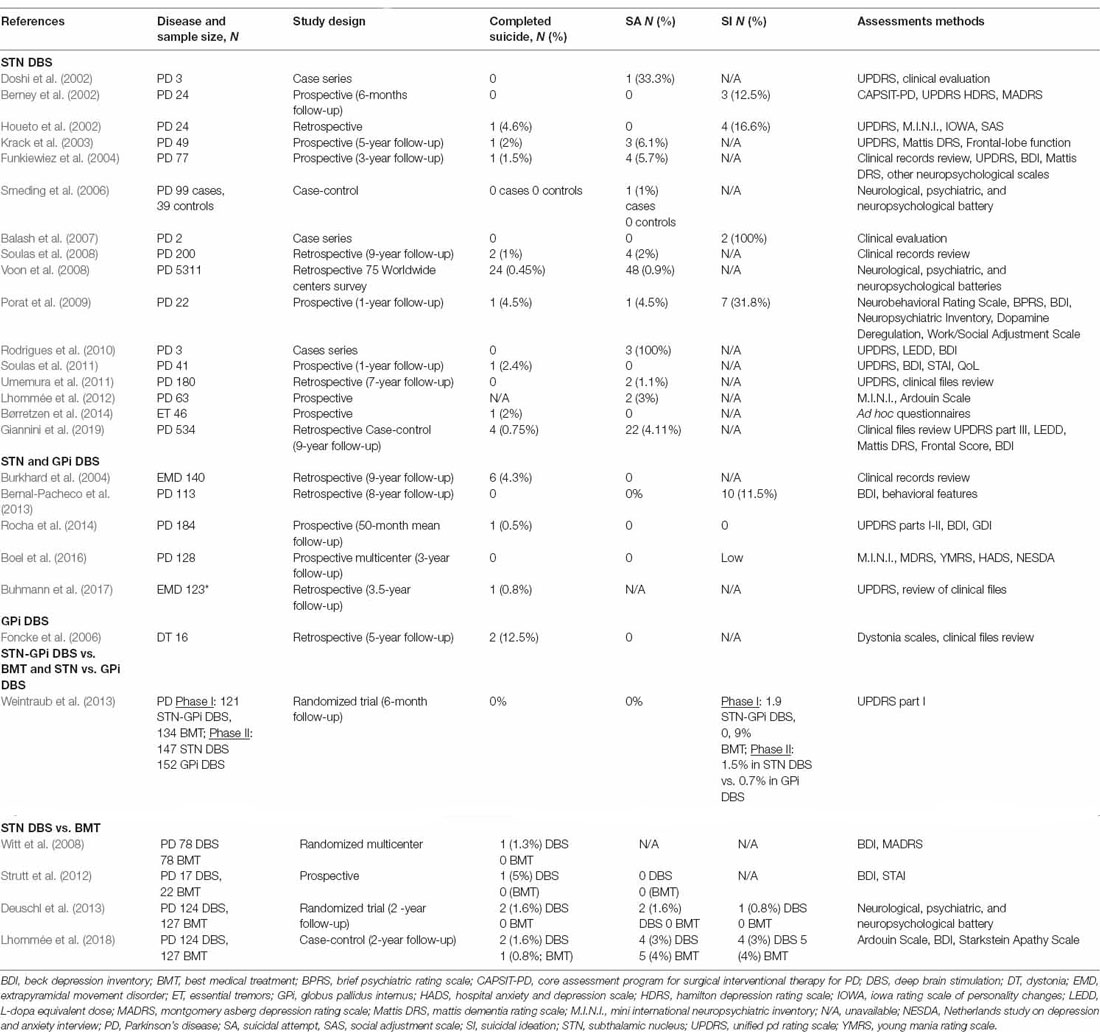
While comprehensive data on SI and SB rates of subjects with a co-morbid neurological condition or physical illness are often limited (Ostertag et al., 2019; Costanza et al., 2020d, i), clinical evidence of suicidality possibly associated with DBS treatment for PD subjects was first documented in a series of case reports (Doshi et al., 2002; Balash et al., 2007; Rodrigues et al., 2010). Additionally, analyses of initial cohorts of PD patients undergoing STN and GPi DBS in the early 2000s observed a notable rate of suicidality (1.5–4.6% for complete suicides, 0–6.1% for SA, and 12.5–16.6% for SI) despite the interventional success concerning motor symptom improvement (Berney et al., 2002; Houeto et al., 2002; Krack et al., 2003; Burkhard et al., 2004; Funkiewiez et al., 2004; Table 1). This phenomenon was particularly interesting because unlike other neurological conditions with well-established increases in suicide risk, such as multiple sclerosis and epilepsy (Costanza et al., 2015, 2020a), subjects with PD have a markedly lower risk in comparison to that of the general population (Li et al., 2018). Suicidality in patients with STN and GPi DBS was also reported in larger studies from 2006 to 2019 (Table 1), but with more varying frequencies of SI and SB compared to previous cohorts and it remained unclear whether this phenomenon was mechanistically related to DBS. This assertion was in particular challenged by two meta-analyses in a combined total of more than 10,000 subjects with extrapyramidal diseases, which yielded a substantially lower rate of suicide associated with DBS (~0.1–0.32%; Kleiner-Fisman et al., 2006; Appleby et al., 2007; Table 2). Conversely, a vast international multicenter study reported a 0.45% rate of suicide in more than 5,000 PD patients receiving DBS; interestingly, it was demonstrated that after adjustment for other demographic factors, the standardized mortality rate of DBS subjects appeared to be higher than that of the general population, especially in the first year after a DBS surgical procedure (Voon et al., 2008). While the incidences of SI and SB in patients who underwent DBS seemed to be significantly decreasing, probably due to improvement in candidate selection and postoperative follow-up (see paragraph 4), the debate was revived in 2019, when in a large retrospective case-controlled study by Giannini et al. (2019) in 534 PD patients who underwent bilateral STN DBS between 1993 and 2016, completed suicide and SA percentages were 0.75% and 4.11%, respectively. The observed suicide rate in the first postoperative year was higher than the expected National Observatory on Suicide Risks rate adjusted for age and sex and this rate remained similar over the second and third postoperative years (Giannini et al., 2019; Table 1). Two recent meta-analyses revealed increased rates of SI and SB (Xu et al., 2021), particularly in patients who underwent DBS compared to the general population (Du et al., 2020; Table 2). Two recent systematic reviews reaffirm the relevance of the problem of suicidality in DBS while insisting on the heterogeneity of the results and the extreme complexity in interpreting them (Berardelli et al., 2019; Zarzycki and Domitrz, 2020), while a third is more incisive in excluding any causal associations between DBS and suicidality (Shepard et al., 2019).
Besides studies that focus on DBS at the STN or the STN and GPi, an examination of suicidality in patients receiving only GPi DBS treatment has been scarcely reported. In this regard, complete suicides (12.5%) were documented in a small cohort of patients with dystonia (Foncke et al., 2006). A comparative analysis of SI incidence in PD patients receiving DBS at two different neurological targets (GPi vs. STN) showed a markedly lower rate in the GPi group (Weintraub et al., 2013; Table 1). It was postulated that GPi DBS might be protective against the development of depression or SI and SB because it is comparable to STN DBS concerning motor outcomes but does not require postoperative reduction of dopaminergic medications as is usually the case for STN DBS (Weintraub et al., 2013). Moreover, GPi DBS might provide better protection against the development of depression or SI and SB as this treatment seemed to result in greater relief from psychiatric symptoms (Liu et al., 2014; Negida et al., 2018). A meta-analysis found that stimulating STN and GPi were equally effective at improving motor symptoms and dyskinesias; however, there would be has been discrepancy as to whether the cognitive, behavioral, and mood symptoms were affected differently between the two targets (Combs et al., 2015). This issue needs further research (Combs et al., 2015).
Greater SI and SB incidences in case-control studies among PD patients treated with DBS compared to PD patients treated with best medical treatment (BMT) were reported (0–5% for complete suicides, 0–3% for SA, and 0–3% for SI; Witt et al., 2008; Strutt et al., 2012; Deuschl et al., 2013; Weintraub et al., 2013; Lhommée et al., 2018). However, these studies do not yield uniform results. In the cohort of Lhommée et al. (2018), it was shown that there was a two-fold increase in complete suicides (1.6% vs. 0.8%) in patients with DBS compared to patients who received BMT, but SA and SI were less frequent in former patients than in latter (3% vs. 4%; Table 1).
To date, the topic of suicidality and DBS remains controversial due to differences in clinical assessment methods, study types, sample sizes, and patient characteristics. Nevertheless, these reports highlight possibly DBS-associated suicidality as an important phenomenon that deserves further examination and clinical attention.
Hypotheses on Mechanisms for Suicidality Associated with DBS
Neuroanatomical Circuit Dysfunctions
While many neurobiological factors have been associated with suicide (Hawton and van Heeringen, 2009; Turecki et al., 2012; Costanza et al., 2014; van Heeringen and Mann, 2014; Costanza et al., 2020j), little is known about the neuro-etiology of this phenomenon in the context of DBS. The primary hypothesis on putative neurobiological mechanisms of suicide after DBS revolves around the anatomical regions that might be undesirably targeted by this treatment, notably those structurally adjacent or functionally related to STN or GPi (14; Figure 1).
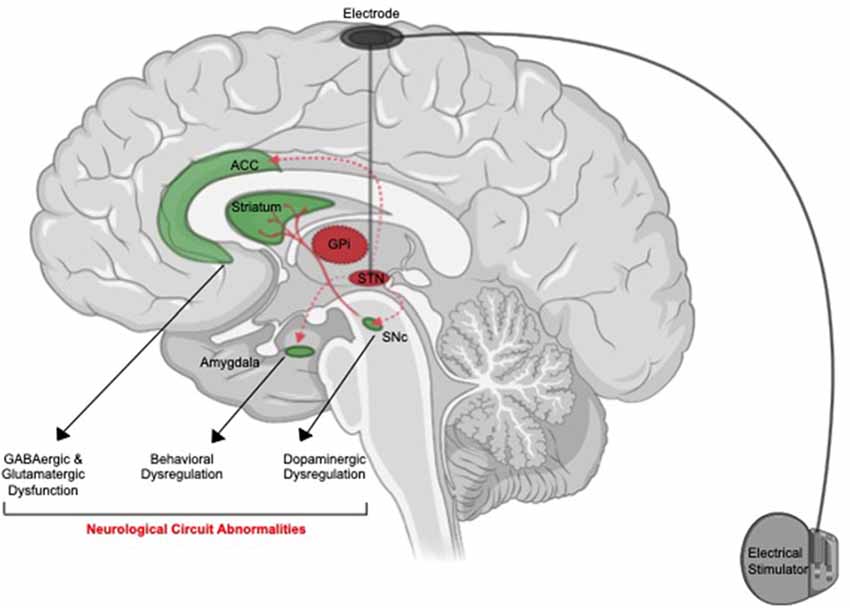
Figure 1. Putative neuroanatomical mechanisms of suicide associated with deep brain stimulation (DBS). Off-target stimulation of brain regions connected with anterior cingulate cortex (ACC, amygdala) or adjacent to (SNc) the suicide risk in patients who received subthalamic nucleus (STN) might cause induction of neurological pathways associated with suicide. Activation of the ACC might cause a disturbance in GABAergic and glutamatergic signaling. Activation of the amygdala might result in behavioral abnormalities. Activation of SNc and dorsal striatum circuit can result in dopaminergic dysfunction. These neurobiological changes might translate into elevated psychosocial distress and potentiate suicidal ideation (SI) and suicidal behavior (SB).
Specifically, STN stimulation can modulate dopamine release from the substantia nigra pars compacta (SNc) in animal studies (Shimo and Wichmann, 2009). In light of the critical involvement of dysfunctional dopaminergic signaling in PD patients, DBS might further disrupt the dopaminergic system, potentiating SI or SB. Mechanistically, since dopamine has been shown to drive impulse control disorders (Ahlskog, 2011), the dysregulated release of this neurotransmitter might consequently increase the risk of SI (Costanza et al., 2014). In fact, dysregulated activation of dopamine signaling, particularly in the dorsal striatum (the main target of SNc dopaminergic projections), has been linked to suicide (Fitzgerald et al., 2017). Consistent with this report, a recent study has demonstrated a correlation between reduced dopamine transporter availability and increased hopelessness scores in all bilateral striatal areas of subjects with major depressive disorders (MDD; Pettorruso et al., 2020). Last but not least, it is also worth noting that a rapid post-DBS reduction in dopamine therapy might cause a general hypodopaminergic state in the brain and the unmasking of depression and SI in PD patients (Berardelli et al., 2019).
Another important brain region that is affected by DBS at the STN is the amygdala, which regulates emotions and aggressive behaviors (Accolla et al., 2016). Since abnormalities in functions and structure of this brain region have been associated with SA in subjects with neuropsychiatric diseases (Spoletini et al., 2011; Wang et al., 2020), possibly off-target stimulation of the amygdala might drive suicide risk in DBS subjects. Increased functional connectivity at this neuroanatomical region has been linked to increased SI and SA in patients with MDD (Kang et al., 2017; Alarcón et al., 2019). Association between the self and death in an exploratory study of the neurobiological origins of SI has also been attributed to amygdala activation (Wei et al., 2018; Ballard et al., 2020). Mechanistically, activation of the amygdala can exacerbate dopaminergic dysfunction (Lai and Chang, 2019) and consequently potentiating suicide risk as described above. Enhanced amygdala connectivity has also been linked to excessive glutamatergic-induced neuro-excitotoxicity and increased suicide risk (Sequeira et al., 2009; Cabrera et al., 2019; Ousdal et al., 2019).
It was also hypothesized that DBS-induced modulation of connections between the STN and the prefrontal cortex areas and mid-brain serotonin neurons may be implicated in mood-related changes and subsequent increased suicide risk (Temel et al., 2005).
Similarly, STN connectivity with frontal cortical regions such as the anterior cingulate cortex (ACC) is highly relevant in the context of suicide (Brunenberg et al., 2012). The ACC is a complex cortical region that modulates emotional behaviors and abnormalities in its function are the basis of the development of several mood disorders (Drevets et al., 2008). In suicidal subjects, ACC functional activity was enhanced (Minzenberg et al., 2016). ACC activation also results in increases in glutamate/GABA expression (Zhao et al., 2018; Lewis et al., 2020). As a result of this, bystander-stimulation of the ACC during DBS therapy might drive these neurotransmitter disturbances, which have been linked to the increased risk for suicide (Sequeira et al., 2009; Cabrera et al., 2019).
Despite these speculative mechanisms, it is worth noting that contradictory findings have indicated a reduction in functional connectivity at the amygdala (Johnston et al., 2017; Wang et al., 2020) and ACC (Huber et al., 2020) in patients with SI/SA. Therefore, definitive proofs for the involvement of the amygdala and/or ACC off-targeting in the development of DBS-associated suicidality have yet to be formally demonstrated.
Aberrant Immunological Activation
The speculative involvement of the immune system in suicide associated with DBS stems from emerging evidence that links neuroinflammatory responses, orchestrated by microglia and macrophages, to both of these phenomena (Figure 2).
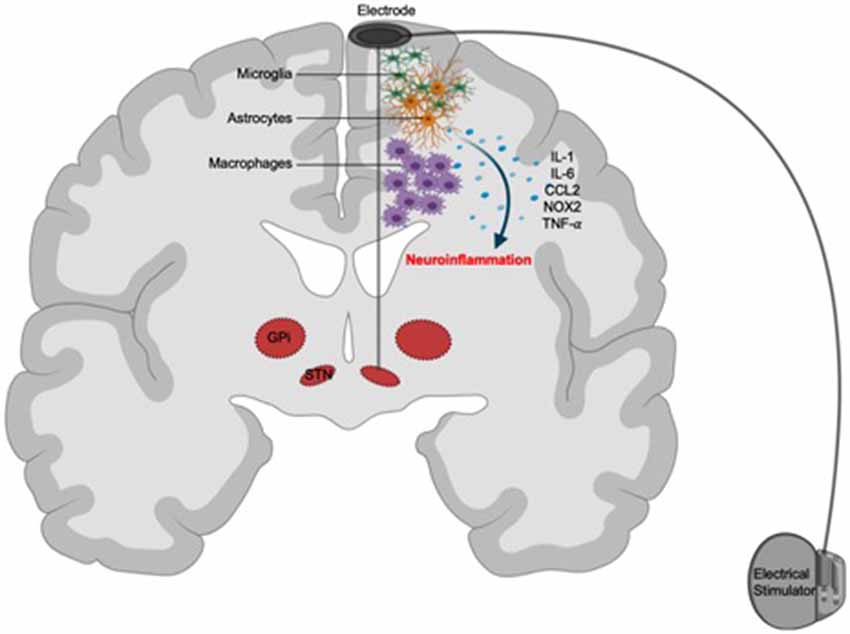
Figure 2. Putative immunological mechanisms of suicide associated with DBS. Surgical implantation of neuroelectrodes might cause immunoreactivity against these devices. Focal activation of recruited peripheral macrophages and brain resident microglia and astrocytes (glial scarring) in response to the electrodes triggers the release of inflammatory mediators, such as IL-1, IL-6, CCL2, NOX2, and TNF-α, resulting in neuroinflammation and subsequent aggravation of psychosocial risk factors for suicide.
Microglia mature from a yolk sac progenitor and take residence in the nervous tissues while macrophages originate from the bone marrow and could be recruited to other tissues in various pathological contexts (Thion and Garel, 2020). Despite their ontological differences, the primary function of these innate immune cells is to patrol the body during homeostatic development to support tissue debris clearance (Butovsky and Weiner, 2018; Kierdorf et al., 2019; Tay et al., 2019). However, these cells are also able to respond to pathogens, noxious environmental insults, and surgical implants. Upon recognition of these foreign stimuli, microglia and macrophages elicit a robust inflammatory reaction, characterized by the release of cytokines and chemokines to amplify and sustain this response. While the intended function of this reactive inflammation is to protect the body from invading micro-organisms, its maladaptation could result in unresolved tissue damage and ensuing pathology.
Microglial reactivity in DBS has been observed in animal studies. For instance, STN implantation of stainless-steel electrodes triggered a localized microglial activation and sustained memory impairment (Hirshler et al., 2010). Similarly, cortical implants also resulted in chronic gliosis of microglial and astrocytic origins (Griffith and Humphrey, 2006; Rosskothen-Kuhl et al., 2018). Consistent with these observations, another study revealed a more elaborated spatial and temporal neuroinflammatory cascade, characterized by neuronal and progenitor cell loss, axonal and myelin reassembly, microglia and astrocyte reactivity, and pericyte deficiency-driven blood-brain barrier disruption, both acutely and chronically around implanted devices (Wellman et al., 2019). Interestingly, the density of neural tissues and the implanted probes as well as the size and fixation method of the implants play an important role in determining the extent of microglia and astrocyte reaction with regions with high-density probes exhibiting more pronounced neuroinflammation (Thelin et al., 2011; Lind et al., 2012) while the numbers of implanted devices did not appear to affect glial cell scarring (Lind et al., 2013). Furthermore, infiltrating macrophages have recently been shown to be the primary orchestrators of neuroinflammatory reaction to cortical implanted electrodes (Ravikumar et al., 2014). In this study, the authors highlight the hallmark accumulation of infiltrating macrophages at the implant sites and the positive correlation between their density and neuronal damage. Therefore, macrophages and microglia might cooperate to induce a biphasic neuroinflammatory and degenerative response to a chronically implanted device (Potter et al., 2012; Giordano et al., 2020). Corroborating evidence from studies on post-mortem brain tissues in humans also confirmed these preclinical findings. In a pediatric status dystonicus case study, evidence of gliosis and multinucleated giant cells (macrophage aggregates) was observed along the trajectories of the implanted electrodes (Kronenbuerger et al., 2015). Larger case series in PD patients also revealed extensive gliosis and macrophage accumulation in the majority of subjects undergoing DBS (Vedam-Mai et al., 2018). Notably, gliosis is associated with septicemia in deceased subjects (Kronenbuerger et al., 2015) but does not correlate with the duration of the DBS regimen (Vedam-Mai et al., 2018). Along with studies in animals, these findings suggest that neuroinflammation occurs during DBS and might represent a reaction against implanted neuroelectrodes, rather than against the DBS treatment itself (Amorim et al., 2015; Hadar et al., 2017; Lopez-Cuina et al., 2018).
Similar to DBS, microglia and macrophages have been implicated in a stress-diathesis paradigm of SI and SB (Baharikhoob and Kolla, 2020). It is postulated that suicide results from a complex interplay between psychosocial and biological stressors during which biological stimuli, i.e., macrophage and microglial mediated-neuroinflammation, act to aggravate the non-biological determinants of suicide. In support of this hypothesis, microgliosis was observed in post-mortem brain tissues of suicide subjects (Steiner et al., 2008; Schnieder et al., 2014; Torres-Platas et al., 2014; Cabrera et al., 2019). Markers of macrophage-mediated systemic inflammation, such as IL-1, IL-6, and TNF-α (Steiner et al., 2013), as well as those of localized microglia-derived neuroinflammatory responses, such as IL-6 and NOX2, are elevated in suicide subjects (Ganança et al., 2016; Schiavone et al., 2016). Furthermore, suicide is associated with neuroinflammatory signatures in selective brain regions, suggesting widespread inflammation is not necessary for the precipitation of SB (Serafini et al., 2020). Interestingly, in depressed suicides, microglia expression of quinolinic acid was elevated (Steiner et al., 2011). Macrophage infiltration was also documented in the brain tissues of these subjects, evident by increased expression of the surface protein CD45 and the soluble chemokine CCL2 (Brisch et al., 2017). Last but not the least, nonsteroidal anti-inflammatory drugs that could effectively curb neuroinflammation can reduce SI in humans (Lehrer and Rheinstein, 2019), further supporting the role of glial-cell-mediated neuroinflammation in the potentiation of suicide risk.
Mechanistically, these microglia and macrophage-derived neuroinflammatory mediators are thought to interfere with homeostatic serotonergic and glutamatergic transmission by the metabolic pathway of indoleamine 2,3-dioxygenase (IDO; Suzuki et al., 2019; Baharikhoob and Kolla, 2020; Serafini et al., 2020). In this prominent hypothesis on the inflammatory origin of suicide, augmented inflammatory milieu in the specific brain regions that are produced by microglia can activate IDO-dependent catabolism of tryptophan to generate kynurenine. This metabolite in turn promotes the development of neurotoxic microglia, which can produce quinolinic acid and consequentially trigger glutamatergic/NMDA-receptor-dependent neurotoxicity. Alternatively, tryptophan degradation could also result in reduced serotonin production, which has been implicated in mood disorders and suicide (Pompili et al., 2017).
Given the collective implications of neuroinflammation elicited by macrophages and microglia in both DBS and suicide, it is plausible to envision that suicide associated with DBS might result from unregulated regional reactivity of these cells to the implanted device. Notably, innate immune cells in these subjects might also be more susceptible to eliciting dysregulated inflammation due to their underlying neurological conditions, potentiating the risk for psychosocial stress-driven suicide.
Nevertheless, both experimental and clinical data to formally support this hypothesis are currently lacking. Due to the lack of validated animal models of suicide and mood disorder, future studies that utilize microglial PET tracers (Cavaliere et al., 2020) in patients with DBS would be required to provide a definitive answer to this topic of immense clinical and scientific interest.
Clinical Implications
The selection of the candidate patient to undergo DBS is the most effective means of avoiding psychiatric complications, including suicidality development, following operation (Lang and Widner, 2002; Rodriguez et al., 2007; Pollak, 2013; Boel et al., 2016). In this regard, the presence of a multidisciplinary team consisting of a neurologist, a psychiatrist, and a psychologist is of critical importance for a comprehensive evaluation of the patient’s suitability for DBS treatment. Eligibility guidelines for DBS include patients diagnosed with extrapyramidal diseases that are either refractory to conventional medications (i.e., inconsistent or marginal responses to medical treatments that result in fluctuating motor symptoms and/or dyskinesia) or chronically/severely suffering from medication-related side effects. Besides these inclusion criteria, disabling and active psychiatric disturbances, history of SI/SB, past diagnoses of unipolar/bipolar affective or psychotic disorders represent contraindications to DBS. Furthermore, even in the absence of major psychiatric disturbances, attention must be paid to symptoms of emotional lability, apathy, impulsivity, and irritability/anger. In these situations, the patients must be fully treated for such minor psychiatric issues to proceed with DBS operation, if DBS treatment is considered necessary. It’s also worth noting that the physicians should inform the patients about the potential development of psychiatric disturbance after DBS surgery even if they had no previous history of these symptoms. Other exclusion criteria for DBS include the presence of moderate/severe cognitive impairment as well as other medical comorbidities. DBS therapy is also not recommended when insufficient motivation or unrealistic expectations are expressed by the patient or their family members. The patients should be fully aware that DBS treatment might require series of surgical and medication adjustments and thus, patience and significant time commitment are required. Lastly, the patients should be realistic about the efficacy of this treatment as it is not the ultimate cure for extrapyramidal diseases and only results in meaningful therapeutic outcomes (Lang and Widner, 2002; Rodriguez et al., 2007; Pollak, 2013; Boel et al., 2016).
After the DBS, a consistent post-operative follow-up with a multidisciplinary team of physicians, particularly in the first few years after DBS surgery, is recommended (Lang and Widner, 2002; Rodriguez et al., 2007; Pollak, 2013; Boel et al., 2016). During such routines, detailed clinical interviews with the patients and their caregivers should be conducted. To assess the progression of extrapyramidal diseases and various parameters the patients’ psychiatric/psychological conditions, standardized assessment scales, including the United Parkinson’s Disease Rating Scale (UPDRS; Poewe, 2009), the Montgomery–Åsberg Depression Rating Scale (MADRS; Williams and Kobak, 2008), the Columbia-Suicide Severity Rating Scale (C-SSRS; Posner et al., 2011), the Ardouin Scale of Behavioral in Parkinson’s Disease (ASBPD; Rieu et al., 2015) for psychiatric assessment related to hypodopaminergic (as apathy) and hyperdopaminergic (as dysregulated impulse control) and non-motor fluctuations, and the Austin CEP Interview for expectation assessment (adapted from the field of surgical epilepsy; Wilson et al., 1999), could be employed. Additionally, since increased or de novo cognitive impairment after DBS surgery could influence psychiatric complications, comprehensive neurocognitive assessment should also be included as an integral part of postoperative follow-up. Collectively, the findings from these assessments provide a comprehensive snapshot of the post-operative state of the patients, which could be compared with their pre-operative status so that appropriate interventions could be devised.
Psychiatric post-operative care involves both psychopharmacological adjustments and psychotherapeutic interventions, such as the recommended cognitive-behavioral therapies (CBT). Psychotherapeutic interventions, which are particularly relevant in the context of suicide prevention (Costanza et al., 2020k, 2021), could also address other specific features of PD patients receiving DBS, such as frustration due to “disproportionate or unrealistic expectations” related to DBS (Wrench et al., 2004), and the occurrence of a scenario named “burden of normality” (often detected in patients whose chronic and disabling disease could be rapidly and drastically improved; thus paradoxically exposing them to difficulties in restoring normal life; Wilson et al., 2001, 2004; Gilbert, 2012). In this regard, the two constructs of demoralization and Meaning in Life (MiL) have recently received clinical attention as psychotherapeutic targets for these PD patients, similarly to those who experienced a severe somatic disease and underwent a disjointed “before” and an “after” existential experience (Costanza et al., 2019, 2020c,f). These two constructs are intimately associated with each other as loss of MiL is one of the constituting components of demoralization (Chytas et al., 2019; Costanza et al., 2020e). Furthermore, MiL and demoralization are two important resilience and risk factors, respectively, for the development of suicidality (Costanza et al., 2020b,g,h). Other promising psychotherapeutic targets include the two dimensions of Interpersonal Theory of Suicide (IPTS), such as the feeling of “perceived burdensomeness” and “thwarted belongingness” (Baertschi et al., 2017, 2018a,b). The interpersonal nature of IPTS, in particular, can address psychoeducational needs for both patients and their family members/care-givers (Costanza et al., 2018).
Besides these interventions, strategies aiming at correcting neuroanatomical and neuroimmunological dysfunctions could be devised to address the possible involvement of these pathways in the development of suicidality in patients receiving DBS. For instance, given the acute reduction in dopamine therapy in DBS patients after undergoing this treatment, dosing could be adjusted accordingly to avoid undesirable effects of a DBS induced-hypodopaminergic state (Berardelli et al., 2019). Targeting ACC functional connectivity defects that are associated with suicide might also be specifically achieved with the emerging use of ketamine and esketamine (Chen et al., 2019a, b). Additionally, strategies to suppress neuroinflammation have also been attempted. Specifically, experimental studies have revolved around the development of surgical innovations to mitigate the response of the immune system to the DBS implants. As such, different materials, surface structure, shapes, size, density, and fixation methods of the implant devices might be critical to minimize microglial reactivity (Thelin et al., 2011; Lind et al., 2012, 2013; Eles et al., 2017; Golabchi et al., 2019). Furthermore, adjunct treatment with anti-inflammatory agents systemically (Yuan et al., 2019) or locally properties (Zhong and Bellamkonda, 2007; Gutowski et al., 2015; Liu et al., 2017) also presents an attractive therapeutic roadmap.
Conclusion
In summary, while it remains elusive whether and to what extent neuroanatomical and/or immunological mechanisms contribute to suicide associated with DBS, this phenomenon represents an unmet medical need. Besides the novel therapeutic insights stemming from these mechanistic postulates and in line with the current shift in healthcare toward predictive/precision medicine rather than secondary preventative methods (Amerio et al., 2020), more consensual and multi-disciplinary guidelines (including neurosurgical, psychiatric, and psychological interventions) both for DBS candidate selection and for post-operative follow-up of patients undergoing DBS are of critical importance.
Data Availability Statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
Author Contributions
AC, MR, AAm, AAg, and FZ contributed to the conception of the work, researched the literature, and drafted the primary manuscript. GS, MA, GB, IB, and MP contributed to the conception of the work, carefully revised the manuscript, and provided the intellectual impetus. KDN supervised all steps of the work, revised, finalized, and edited the final version of the manuscript. All authors contributed to the article and approved the submitted version.
Conflict of Interest
KN is the scientific founder of Tranquis Therapeutics, a neuroimmunology company that develops immunotherapies for neurological diseases.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We are deeply grateful to Prof. Luc Mallet and his team (Institut du Cerveau, Hôpital Pitié-Salpêtrière, Paris, France) for bringing us, thanks to the vast experience of their center, closer to this topic.
References
Accolla, E. A., Herrojo Ruiz, M., Horn, A., Schneider, G. H., Schmitz-Hübsch, T., Draganski, B., et al. (2016). Brain networks modulated by subthalamic nucleus deep brain stimulation. Brain 139, 2503–2515. doi: 10.1093/brain/aww182
Ahlskog, J. E. (2011). Pathological behaviors provoked by dopamine agonist therapy of Parkinson’s disease. Physiol. Behav. 104, 168–172. doi: 10.1016/j.physbeh.2011.04.055
Alarcón, G., Sauder, M., Teoh, J. Y., Forbes, E. E., and Quevedo, K. (2019). Amygdala functional connectivity during self-face processing in depressed adolescents with recent suicide attempt. J. Am. Acad. Child Adolesc. Psychiatry 58, 221–231. doi: 10.1016/j.jaac.2018.06.036
Amerio, A., Sibilla, F., Pescini, R., Ciprandi, R., Casciaro, R., Grimaldi Filiolo, P., et al. (2020). Mental health and cystic fibrosis: time to move from secondary prevention to predictive medicine. Pediatr. Pulmonol. 55, 2204–2206. doi: 10.1002/ppul.24928
Amorim, B. O., Covolan, L., Ferreira, E., Brito, J. G., Nunes, D. P., de Morais, D. G., et al. (2015). Deep brain stimulation induces antiapoptotic and anti-inflammatory effects in epileptic rats. J. Neuroinflammation 12:162. doi: 10.1186/s12974-015-0384-7
Appleby, B. S., Duggan, P. S., Regenberg, A., and Rabins, P. V. (2007). Psychiatric and neuropsychiatric adverse events associated with deep brain stimulation: a meta-analysis of ten years’ experience. Mov. Disord. 22, 1722–1728. doi: 10.1002/mds.21551
Au, K. L. K., Wong, J. K., Tsuboi, T., Eisinger, R. S., Moore, K., Lemos Melo Lobo Jofili Lopes, J., et al. (2020). Globus pallidus internus (GPi) deep brain stimulation for Parkinson’s disease: expert review and commentary. Neurol. Ther. [Epub ahead of print]. doi: 10.1007/s40120-020-00220-5
Baertschi, M., Costanza, A., Richard-Lepouriel, H., Pompili, M., Sarasin, F., Weber, K., et al. (2017). The application of the interpersonal-psychological theory of suicide to a sample of Swiss patients attending a psychiatric emergency department for a non-lethal suicidal event. J. Affect. Disord. 210, 323–331. doi: 10.1016/j.jad.2016.12.049
Baertschi, M., Costanza, A., Canuto, A., and Weber, K. (2018a). The dimensionality of suicidal ideation and its clinical implications. Int. J. Methods Psychiatr. Res. 28:e1755. doi: 10.1002/mpr.1755
Baertschi, M., Costanza, A., Canuto, A., and Weber, K. (2018b). The function of personality in suicidal ideation from the perspective of the interpersonal-psychological theory of suicide. Int. J. Environ. Res. Public Health 15:636. doi: 10.3390/ijerph15040636
Baharikhoob, P., and Kolla, N. J. (2020). Microglial dysregulation and suicide: a stress-diathesis perspective. Front. Psychiatry 11:781. doi: 10.3389/fpsyt.2020.00781
Balash, Y., Merims, D., and Giladi, N. (2007). Suicidal thoughts in patients with Parkinson’s disease treated by deep brain stimulation of the subthalamic nuclei: two case reports and review of the literature. Acta Neuropsychiatr. 19, 208–210. doi: 10.1111/j.1601-5215.2007.00177.x
Ballard, E. D., Gilbert, J. R., Fields, J. S., Nugent, A. C., and Zarate, C. A. Jr. (2020). Network changes in insula and amygdala connectivity accompany implicit suicidal associations. Front. Psychiatry 11:577628. doi: 10.3389/fpsyt.2020.577628
Benabid, A. L., Chabardes, S., Mitrofanis, J., and Pollak, P. (2009). Deep brain stimulation of the subthalamic nucleus for the treatment of Parkinson’s disease. Lancet Neurol. 8, 67–81. doi: 10.1016/S1474-4422(08)70291-6
Berardelli, I., Belvisi, D., Nardella, A., Falcone, G., Lamis, D. A., Fabbrini, G., et al. (2019). Suicide in Parkinson’s disease: a systematic review. CNS Neurol. Disord. Drug Targets 18, 466–477. doi: 10.2174/1871527318666190703093345
Bernal-Pacheco, O., Oyama, G., Foote, K. D., Dai, Y. E., Wu, S. S., Jacobson, C. E. IV., et al. (2013). Taking a better history for behavioral issues pre- and post-deep brain stimulation: issues missed by standardized scales. Neuromodulation 16, 35–39; discussion 39–40. doi: 10.1111/j.1525-1403.2012.00477.x
Berney, A., Vingerhoets, F., Perrin, A., Guex, P., Villemure, J. G., Burkhard, P. R., et al. (2002). Effect on mood of subthalamic DBS for Parkinson’s disease: a consecutive series of 24 patients. Neurology 59, 1427–1429. doi: 10.1212/01.wnl.0000032756.14298.18
Boel, J. A., Odekerken, V. J., Geurtsen, G. J., Schmand, B. A., Cath, D. C., Figee, M., et al. (2016). Psychiatric and social outcome after deep brain stimulation for advanced Parkinson’s disease. Mov. Disord. 31, 409–413. doi: 10.1002/mds.26468
Børretzen, M. N., Bjerknes, S., Sæhle, T., Skjelland, M., Skogseid, I. M., Toft, M., et al. (2014). Long-term follow-up of thalamic deep brain stimulation for essential tremor-patient satisfaction and mortality. BMC Neurol. 14:120. doi: 10.1186/1471-2377-14-120
Brisch, R., Steiner, J., Mawrin, C., Krzyżanowska, M., Jankowski, Z., and Gos, T. (2017). Microglia in the dorsal raphe nucleus plays a potential role in both suicide facilitation and prevention in affective disorders. Eur. Arch. Psychiatry Clin. Neurosci. 267, 403–415. doi: 10.1007/s00406-017-0774-1
Brunenberg, E. J., Moeskops, P., Backes, W. H., Pollo, C., Cammoun, L., Vilanova, A., et al. (2012). Structural and resting state functional connectivity of the subthalamic nucleus: identification of motor STN parts and the hyperdirect pathway. PLoS One 7:e39061. doi: 10.1371/journal.pone.0039061
Buhmann, C., Huckhagel, T., Engel, K., Gulberti, A., Hidding, U., Poetter-Nerger, M., et al. (2017). Adverse events in deep brain stimulation: a retrospective long-term analysis of neurological, psychiatric and other occurrences. PLoS One 12:e0178984. doi: 10.1371/journal.pone.0178984
Burkhard, P. R., Vingerhoets, F. J., Berney, A., Bogousslavsky, J., Villemure, J. G., and Ghika, J. (2004). Suicide after successful deep brain stimulation for movement disorders. Neurology 63, 2170–2172. doi: 10.1212/01.wnl.0000145603.48221.b5
Butovsky, O., and Weiner, H. L. (2018). Microglial signatures and their role in health and disease. Nat. Rev. Neurosci. 9, 622–635. doi: 10.1038/s41583-018-0057-5
Cabrera, B., Monroy-Jaramillo, N., Fries, G. R., Mendoza-Morales, R. C., García-Dolores, F., Mendoza-Larios, A., et al. (2019). Brain gene expression pattern of subjects with completed suicide and comorbid substance use disorder. Mol. Neuropsychiatry 5, 60–73. doi: 10.1159/000493940
Cavaliere, C., Tramontano, L., Fiorenza, D., Alfano, V., Aiello, M., and Salvatore, M. (2020). Gliosis and neurodegenerative diseases: the role of PET and MR imaging. Front. Cell. Neurosci. 14:75. doi: 10.3389/fncel.2020.00075
Chen, M.-H., Cheng, C.-M., Gueorguieva, R., Lin, W.-C., Li, C.-T., Hong, C.-J., et al. (2019a). Maintenance of antidepressant and antisuicidal effects by D-cycloserine among patients with treatment-resistant depression who responded to low-dose ketamine infusion: a double-blind randomized placebo-control study. Neuropsychopharmacology 44, 2112–2118. doi: 10.1038/s41386-019-0480-y
Chen, M.-H., Lin, W.-C., Tu, P.-C., Li, C.-T., Bai, Y.-M., Tsai, S.-J., et al. (2019b). Antidepressant and antisuicidal effects of ketamine on the functional connectivity of prefrontal cortex-related circuits in treatment-resistant depression: a double-blind, placebo-controlled, randomized, longitudinal resting fMRI study. J. Affect. Disord. 259, 15–20. doi: 10.1016/j.jad.2019.08.022
Chiken, S., and Nambu, A. (2016). Mechanism of deep brain stimulation: inhibition, excitation, or disruption? Neuroscientist 22, 313–322. doi: 10.1177/1073858415581986
Chytas, V., Costanza, A., Piguet, V., Cedraschi, C., and Bondolfi, G. (2019). Demoralization and meaning in life in suicidal ideation: a role for patients suffering from chronic pain? Rev. Med. Suisse 15, 1282–1285.
Combs, H. L., Folley, B. S., Berry, D. T., Segerstrom, S. C., Han, D. Y., Anderson-Mooney, A. J., et al. (2015). Cognition and depression following deep brain stimulation of the subthalamic nucleus and globus pallidus pars internus in Parkinson’s disease: a meta-analysis. Neuropsychol. Rev. 25, 439–454. doi: 10.1007/s11065-015-9302-0
Costanza, A., Ambrosetti, J., Wyss, K., Bondolfi, G., Sarasin, F., and Khan, R. (2018). Prevention of suicide at emergency room: from the “Interpersonal theory of suicide” to the connectedness. Rev. Med. Suisse 14, 335–338.
Costanza, A., Amerio, A., Aguglia, A., Escelsior, A., Serafini, G., Berardelli, I., et al. (2020a). When sick brain and hopelessness meet: some aspects of suicide in the neurological patient. CNS Neurol. Disord. Drug Targets 19, 257–263. doi: 10.2174/1871527319666200611130804
Costanza, A., Amerio, A., Aguglia, A., Serafini, G., and Amore, M. (2020b). Meaning in life and demoralization constructs in light of the interpersonal theory of suicide: a trans-theoretical hypothesis for a cross-sectional study. Psychol. Res. Behav. Manag. 13, 855–858. doi: 10.2147/PRBM.S279829
Costanza, A., Amerio, A., Odone, A., Baertschi, M., Richard-Lepouriel, H., Weber, K., et al. (2020c). Suicide prevention from a public health perspective. What makes life meaningful? The opinion of some suicidal patients. Acta Biomed. 91, 128–134. doi: 10.23750/abm.v91i3-S.9417
Costanza, A., Amerio, A., Radomska, M., Ambrosetti, J., Di Marco, S., Prelati, M., et al. (2020d). Suicide assessment of the elderly with physical illness in the emergency department. Front. Psychiatry 11:558974. doi: 10.3389/fpsyt.2020.558974
Costanza, A., Baertschi, M., Richard-Lepouriel, H., Weber, K., Berardelli, I., Pompili, M., et al. (2020e). Demoralization and its relationship with depression and hopelessness in suicidal patients attending an emergency department. Int. J. Environ. Res. Public Health 17:2232. doi: 10.3390/ijerph17072232
Costanza, A., Baertschi, M., Richard-Lepouriel, H., Weber, K., Pompili, M., and Canuto, A. (2020f). The presence and the search constructs of meaning in life in suicidal patients attending a psychiatric emergency department. Front. Psychiatry 11:327. doi: 10.3389/fpsyt.2020.00327
Costanza, A., Chytas, V., Mazzola, V., Piguet, V., Desmeules, J., Bondolfi, G., et al. (2020g). The role of demoralization and meaning in life (DEMiL) in influencing suicidal ideation among patients affected by chronic pain: protocol of a single-center, observational, case-control study. JMIR Res. Protoc. 9:e24882. doi: 10.2196/24882
Costanza, A., Di Marco, S., Burroni, M., Corasaniti, F., Santinon, P., Prelati, M., et al. (2020h). Meaning in life and demoralization: a mental-health reading perspective of suicide in the time of COVID-19. Acta Biomed. 91:e2020163. doi: 10.23750/abm.v91i4.10515
Costanza, A., Mazzola, V., Radomska, M., Amerio, A., Aguglia, A., Prada, P., et al. (2020i). Who consults an adult psychiatric emergency department? Pertinence of admissions and opportunities for telepsychiatry. Medicina 56:295. doi: 10.3390/medicina56060295
Costanza, A., Rothen, S., Achab, S., Thorens, G., Baertschi, M., Weber, K., et al. (2020j). Impulsivity and impulsivity-related endophenotypes in suicidal patients with substance use disorders: an exploratory study. Int. J. Mental Health Addict. [Epub ahead of print].doi: 10.1007/s11469-020-00259-3
Costanza, A., Zenga, F., Rudà, R., Amerio, A., Aguglia, A., Serafini, G., et al. (2020k). Suicidality in patients with brain tumors: a brief literature review with clinical exemplar. Medicina 56:725. doi: 10.3390/medicina56120725
Costanza, A., Baertschi, M., Weber, K., and Canuto, A. (2015). Neurological diseases and suicide: from neurobiology to hopelessness. Rev. Med. Suisse 11461, 402–405.
Costanza, A., D’Orta, I., Perroud, N., Burkhardt, S., Malafosse, A., Mangin, P., et al. (2014). Neurobiology of suicide: do biomarkers exist? Int. J. Legal Med. 128, 73–82. doi: 10.1007/s00414-013-0835-6
Costanza, A., Prelati, M., and Pompili, M. (2019). The meaning in life in suicidal patients: the presence and the search for constructs. a systematic review. Medicina 55:465. doi: 10.3390/medicina55080465
Costanza, A., Radomska, M., Zenga, F., Amerio, A., Aguglia, A., Serafini, G., et al. (2021). Severe suicidality in athletes with chronic traumatic encephalopathy: a case series and overview on putative ethiopathogenetic mechanisms. Int. J. Environ. Res. Public Health 18:876. doi: 10.3390/ijerph18030876
Denys, D., Feenstra, M., and Schuurman, R. (2012). Deep Brain Stimulation—A New Frontier in Psychiatry. Berlin Heidelberg: Springer.
Deuschl, G., Schüpbach, M., Knudsen, K., Pinsker, M. O., Cornu, P., Rau, J., et al. (2013). Stimulation of the subthalamic nucleus at an earlier disease stage of Parkinson’s disease: concept and standards of the EARLYSTIM-study. Parkinsonism Relat. Disord. 19, 56–61. doi: 10.1016/j.parkreldis.2012.07.004
Doshi, P. K., Chhaya, N., and Bhatt, M. H. (2002). Depression leading to attempted suicide after bilateral subthalamic nucleus stimulation for Parkinson’s disease. Mov. Disord. 17, 1084–1085. doi: 10.1002/mds.10198
Dostrovsky, J. O., Levy, R., Wu, J. P., Hutchison, W. D., Tasker, R. R., and Lozano, A. M. (2000). Microstimulation-induced inhibition of neuronal firing in human globus pallidus. J. Neurophysiol. 84, 570–574. doi: 10.1152/jn.2000.84.1.570
Drevets, W. C., Savitz, J., and Trimble, M. (2008). The subgenual anterior cingulate cortex in mood disorders. CNS Spectr. 13, 663–668. doi: 10.1017/s1092852900013754
Du, J., Liu, X., Zhou, X., Wang, H., Zhou, W., Jiang, J., et al. (2020). Parkinson’s disease-related risk of suicide and effect of deep brain stimulation: meta-analysis. Parkinsons Dis. 2020:8091963. doi: 10.1155/2020/8091963
Eles, J. R., Vazquez, A. L., Snyder, N. R., Lagenaur, C., Murphy, M. C., Kozai, T. D., et al. (2017). Neuroadhesive L1 coating attenuates acute microglial attachment to neural electrodes as revealed by live two-photon microscopy. Biomaterials 113, 279–292. doi: 10.1016/j.biomaterials.2016.10.054
Fitzgerald, M. L., Kassir, S. A., Underwood, M. D., Bakalian, M. J., Mann, J. J., and Arango, V. (2017). Dysregulation of striatal dopamine receptor binding in suicide. Neuropsychopharmacology 42, 974–982. doi: 10.1038/npp.2016.124
Foncke, E. M., Schuurman, P. R., and Speelman, J. D. (2006). Suicide after deep brain stimulation of the internal globus pallidus for dystonia. Neurology 66, 142–143. doi: 10.1212/01.wnl.0000191328.05752.e2
Funkiewiez, A., Ardouin, C., Caputo, E., Krack, P., Fraix, V., Klinger, H., et al. (2004). Long term effects of bilateral subthalamic nucleus stimulation on cognitive function, mood and behaviour in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 75, 834–839. doi: 10.1136/jnnp.2002.009803
Ganança, L., Oquendo, M. A., Tyrka, A. R., Cisneros-Trujillo, S., Mann, J. J., and Sublette, M. E. (2016). The role of cytokines in the pathophysiology of suicidal behavior. Psychoneuroendocrinology 63, 296–310. doi: 10.3390/cells10030504
Giannini, G., Francois, M., Lhommée, E., Polosan, M., Schmitt, E., Fraix, V., et al. (2019). Suicide and suicide attempts after subthalamic nucleus stimulation in Parkinson disease. Neurology 93, e97–e105. doi: 10.1212/WNL.0000000000007665
Gilbert, F. (2012). The burden of normality: from ‘chronically ill’ to ‘symptom free’. New ethical challenges for deep brain stimulation post-operative treatment. J. Med. Ethics 38, 408–412. doi: 10.1136/medethics-2011-100044
Giordano, F., Caporalini, C., Peraio, S., Mongardi, L., Buccoliero, A. M., Cavallo, M. A., et al. (2020). Post-mortem histopathology of a pediatric brain after bilateral DBS of GPI for status dystonicus: case report and review of the literature. Childs Nerv. Syst. 36, 1845–1851. doi: 10.1007/s00381-020-04761-w
Golabchi, A., Wu, B., Cao, B., Bettinger, C. J., and Cui, X. T. (2019). Zwitterionic polymer/polydopamine coating reduce acute inflammatory tissue responses to neural implants. Biomaterials 155:112096. doi: 10.1016/j.biomaterials.2019.119519
Griffith, R. W., and Humphrey, D. R. (2006). Long-term gliosis around chronically implanted platinum electrodes in the Rhesus macaque motor cortex. Neurosci. Lett. 406, 81–86. doi: 10.1016/j.neulet.2006.07.018
Gutowski, S. M., Shoemaker, J. T., Templeman, K. L., Wei, Y., Latour, R. A. Jr., Bellamkonda, R. V., et al. (2015). Protease-degradable PEG-maleimide coating with on-demand release of IL-1Ra to improve tissue response to neural electrodes. Biomaterials 44, 55–70. doi: 10.1016/j.biomaterials.2014.12.009
Hadar, R., Dong, L., Del-Valle-Anton, L., Guneykaya, D., Voget, M., Edemann-Callesen, H., et al. (2017). Deep brain stimulation during early adolescence prevents microglial alterations in a model of maternal immune activation. Brain Behav. Immun. 63, 71–80. doi: 10.1016/j.bbi.2016.12.003
Hawton, K., and van Heeringen, K. (2009). Suicide. Lancet 373, 1372–1381. doi: 10.1016/S0140-6736(09)60372-X
Herrington, T. M., Cheng, J. J., and Eskandar, E. N. (2016). Mechanisms of deep brain stimulation. J. Neurophysiol. 115, 19–38. doi: 10.1152/jn.00281.2015
Herzog, J., Volkmann, J., Krack, P., Kopper, F., Pötter, M., Lorenz, D., et al. (2003). Two-year follow-up of subthalamic deep brain stimulation in Parkinson’s disease. Mov. Disord. 18:1332. doi: 10.1002/mds.10518
Hirshler, Y. K., Polat, U., and Biegon, A. (2010). Intracranial electrode implantation produces regional neuroinflammation and memory deficits in rats. Exp. Neurol. 222, 42–50. doi: 10.1016/j.expneurol.2009.12.006
Houeto, J. L., Mesnage, V., Mallet, L., Pillon, B., Gargiulo, M., du Moncel, S. T., et al. (2002). Behavioural disorders, Parkinson’s disease and subthalamic stimulation. J. Neurol. Neurosurg. Psychiatry 72, 701–707. doi: 10.1136/jnnp.72.6.701
Huber, R. S., McGlade, E. C., Legarreta, M., Subramaniam, P., Renshaw, P. F., and Yurgelun-Todd, D. A. (2020). Cingulate white matter volume and associated cognitive and behavioral impulsivity in Veterans with a history of suicide behavior. J. Affect. Disord. 281, 117–124. doi: 10.1016/j.jad.2020.11.126
Jakobs, M., Fomenko, A., Lozano, A. M., and Kiening, K. L. (2019). Cellular, molecular and clinical mechanisms of action of deep brain stimulation-a systematic review on established indications and outlook on future developments. EMBO Mol. Med. 11:e9575. doi: 10.15252/emmm.201809575
Johnston, J. A. Y., Wang, F., Liu, J., Blond, B. N., Wallace, A., Liu, J., et al. (2017). Multimodal neuroimaging of frontolimbic structure and function associated with suicide attempts in adolescents and young adults with bipolar disorder. Am. J. Psychiatry 174, 667–675. doi: 10.1176/appi.ajp.2016.15050652
Kang, S.-G., Na, K.-S., Choi, J.-W., Kim, J.-H., Son, Y.-D., and Lee, Y. J. (2017). Resting-state functional connectivity of the amygdala in suicide attempters with major depressive disorder. Prog. Neuropsychopharmacol. Biol. Psychiatry 77, 222–227. doi: 10.1016/j.pnpbp.2017.04.029
Kierdorf, K., Masuda, T., Jordão, M. J. C., and Prinz, M. (2019). Macrophages at CNS interfaces: ontogeny and function in health and disease. Nat. Rev. Neurosci. 20, 547–562. doi: 10.1038/s41583-019-0201-x
Kleiner-Fisman, G., Herzog, J., Fisman, D. N., Tamma, F., Lyons, K. E., Pahwa, R., et al. (2006). Subthalamic nucleus deep brain stimulation: summary and meta-analysis of outcomes. Mov. Disord. 21, S290–S304. doi: 10.1002/mds.20962
Krack, P., Batir, A., Van Blercom, N., Chabardes, S., Fraix, V., Ardouin, C., et al. (2003). Five-year follow-up of bilateral stimulation of the subthalamic nucleus in advanced Parkinson’s disease. N. Engl. J. Med. 349, 1925–1934. doi: 10.1056/NEJMoa035275
Kronenbuerger, M., Nolte, K. W., Coenen, V. A., Burgunder, J.-M., Krauss, J. K., and Weis, J. (2015). Brain alterations with deep brain stimulation: new insight from a neuropathological case series. Mov. Disord. 30, 1125–1130. doi: 10.1002/mds.26247
Lai, C.-W., and Chang, C.-H. (2019). Adaptive anxious states and down-regulation of dopamine activity under amygdala activation in rats. Behav. Brain Res. 361, 1–6. doi: 10.1016/j.bbr.2018.12.049
Lang, A. E., and Widner, H. (2002). Deep brain stimulation for Parkinson’s disease: patient selection and evaluation. Mov. Disord. 17, S94–S101. doi: 10.1002/mds.10149
Lehrer, S., and Rheinstein, P. H. (2019). Nonsteroidal anti-inflammatory drugs (NSAIDs) reduce suicidal ideation and depression. Discov. Med. 28, 205–212.
Lewis, C. P., Port, J. D., Blacker, C. J., Sonmez, A. I., Seewoo, B. J., Leffler, J. M., et al. (2020). Altered anterior cingulate glutamatergic metabolism in depressed adolescents with current suicidal ideation. Transl. Psychiatry 10:119. doi: 10.1038/s41398-020-0792-z
Lezcano, E., Gómez-Esteban, J. C., Tijero, B., Bilbao, G., Lambarri, I., Rodriguez, O., et al. (2016). Long-term impact on quality of life of subthalamic nucleus stimulation in Parkinson’s disease. J. Neurol. 263, 895–905. doi: 10.1007/s00415-016-8077-4
Lhommée, E., Klinger, H., Thobois, S., Schmitt, E., Ardouin, C., Bichon, A., et al. (2012). Subthalamic stimulation in Parkinson’s disease: restoring the balance of motivated behaviours. Brain 135, 1463–1477. doi: 10.1093/brain/aws078
Lhommée, E., Wojtecki, L., Czernecki, V., Witt, K., Maier, F., Tonder, L., et al. (2018). Behavioural outcomes of subthalamic stimulation and medical therapy versus medical therapy alone for Parkinson’s disease with early motor complications (EARLYSTIM trial): secondary analysis of an open-label randomised trial. Lancet Neurol. 17, 223–231. doi: 10.1016/S1474-4422(18)30035-8
Li, W., Abbas, M. M., Acharyya, S., Ng, H. L., Tay, K. Y., Au, W. L., et al. (2018). Suicide in Parkinson’s disease. Mov. Disord. Clin. Pract. 5, 177–182. doi: 10.1002/mdc3.12599
Limousin, P., Krack, P., Pollak, P., Benazzouz, A., Ardouin, C., Hoffmann, D., et al. (1998). Electrical stimulation of the subthalamic nucleus in advanced Parkinson’s disease. N. Engl. J. Med. 339, 1105–1111. doi: 10.1056/NEJM199810153391603
Lind, G., Gällentoft, L., Danielsen, N., Schouenborg, J., and Pettersson, L. M. (2012). Multiple implants do not aggravate the tissue reaction in rat brain. PLoS One 7:e47509. doi: 10.1371/journal.pone.0047509
Lind, G., Linsmeier, C. E., and Schouenborg, J. (2013). The density difference between tissue and neural probes is a key factor for glial scarring. Sci. Rep. 3:2942. doi: 10.1038/srep02942
Liu, B., Kim, E., Meggo, A., Gandhi, S., Luo, H., Kallakuri, S., et al. (2017). Enhanced biocompatibility of neural probes by integrating microstructures and delivering anti-inflammatory agents via microfluidic channels. J. Neural Eng. 14:026008. doi: 10.1088/1741-2552/aa52dc
Liu, Y., Li, W., Tan, C., Liu, X., Wang, X., Gui, Y., et al. (2014). Meta-analysis comparing deep brain stimulation of the globus pallidus and subthalamic nucleus to treat advanced Parkinson disease. J. Neurosurg. 121, 709–718. doi: 10.3171/2014.4.JNS131711
Lopez-Cuina, M., Fernagut, P. O., Canron, M. H., Vital, A., Lannes, B., De Paula, A. M., et al. (2018). Deep brain stimulation does not enhance neuroinflammation in multiple system atrophy. Neurobiol. Dis. 118, 155–160. doi: 10.1016/j.nbd.2018.07.016
Minzenberg, M. J., Lesh, T. A., Niendam, T. A., Cheng, Y., and Carter, C. S. (2016). Conflict-related anterior cingulate functional connectivity is associated with past suicidal ideation and behavior in recent-onset psychotic major mood disorders. J. Neuropsychiatry Clin. Neurosci. 28, 299–305. doi: 10.1176/appi.neuropsych.15120422
Moro, E., Lozano, A. M., Pollak, P., Agid, Y., Rehncrona, S., Volkmann, J., et al. (2010). Long-term results of a multicenter study on subthalamic and pallidal stimulation in Parkinson’s disease. Mov. Disord. 25, 578–586. doi: 10.1002/mds.22735
Munhoz, R. P., Cerasa, A., and Okun, M. S. (2014). Surgical treatment of dyskinesia in Parkinson’s disease. Front Neurol. 5:65. doi: 10.3389/fneur.2014.00065
Negida, A., Elminawy, M., El Ashal, G., Essam, A., Eysa, A., and Abd Elalem Aziz, M. (2018). Subthalamic and pallidal deep brain stimulation for Parkinson’s disease. Cureus 10:e2232. doi: 10.7759/cureus.2232
Odekerken, V. J., Boel, J. A., Schmand, B. A., de Haan, R. J., Figee, M., van den Munckhof, P., et al. (2016). GPi vs. STN deep brain stimulation for Parkinson disease: three-year follow-up. Neurology 86, 755–761. doi: 10.1212/WNL.0000000000002401
Ostertag, L., Golay, P., Dorogi, Y., Brovelli, S., Bertran, M., Cromec, I., et al. (2019). The implementation and first insights of the French-speaking Swiss programme for monitoring self-harm. Swiss Med. Wkly. 149:w20016. doi: 10.4414/smw.2019.20016
Ousdal, O. T., Milde, A. M., Craven, A. R., Ersland, L., Endestad, T., Melinder, A., et al. (2019). Prefrontal glutamate levels predict altered amygdala-prefrontal connectivity in traumatized youths. Psychol. Med. 49, 1822–1830. doi: 10.1017/S0033291718002519
Pettorruso, M., d’Andrea, G., Martinotti, G., Cocciolillo, F., Miuli, A., Di Muzio, I., et al. (2020). Hopelessness, dissociative symptoms, and suicide risk in major depressive disorder: clinical and biological correlates. Brain Sci. 10:E519. doi: 10.3390/brainsci10080519
Poewe, W. (2009). Clinical measures of progression in Parkinson’s disease. Mov. Disord. 24, S671–S676. doi: 10.1002/mds.22600
Pollak, P. (2013). Deep brain stimulation for Parkinson’s disease—patient selection. Handb. Clin. Neurol. 116, 97–105. doi: 10.1016/B978-0-444-53497-2.00009-7
Pollak, P., Benabid, A. L., Gross, C., Gao, D. M., Laurent, A., Benazzouz, A., et al. (1993). Effects of the stimulation of the subthalamic nucleus in Parkinson disease. Rev. Neurol. 149, 175–176.
Pompili, M., Gentile, G., Scassellati, C., Bonvicini, C., Innamorati, M., Erbuto, D., et al. (2017). Genetic association analysis of serotonin and signal transduction pathways in suicide attempters from an Italian sample of psychiatric patients. Neurosci. Lett. 656, 94–102. doi: 10.1016/j.neulet.2017.07.020
Porat, O., Cohen, O. S., Schwartz, R., and Hassin-Baer, S. (2009). Association of preoperative symptom profile with psychiatric symptoms following subthalamic nucleus stimulation in patients with Parkinson’s disease. J. Neuropsychiatry Clin. Neurosci. 21, 398–405. doi: 10.1176/jnp.2009.21.4.398
Posner, K., Brown, G. K., Stanley, B., Brent, D. A., Yershova, K. V., Oquendo, M. A., et al. (2011). The columbia-suicide severity rating scale: initial validity and internal consistency findings from three multisite studies with adolescents and adults. Am. J. Psychiatry 168, 1266–1277. doi: 10.1176/appi.ajp.2011.10111704
Potter, K. A., Buck, A. C., Self, W. K., and Capadona, J. R. (2012). Stab injury and device implantation within the brain results in inversely multiphasic neuroinflammatory and neurodegenerative responses. J. Neural Eng. 9:046020. doi: 10.1088/1741-2560/9/4/046020
Ravikumar, M., Sunil, S., Black, J., Barkauskas, D. S., Haung, A. Y., Miller, R. H., et al. (2014). The roles of blood-derived macrophages and resident microglia in the neuroinflammatory response to implanted intracortical microelectrodes. Biomaterials 35, 8049–8064. doi: 10.1016/j.biomaterials.2014.05.084
Rieu, I., Martinez-Martin, P., Pereira, B., De Chazeron, I., Verhagen Metman, L., Jahanshahi, M., et al. (2015). International validation of a behavioral scale in Parkinson’s disease without dementia. Mov. Disord. 30, 705–713. doi: 10.1002/mds.26223
Rocha, S., Monteiro, A., Linhares, P., Chamadoira, C., Basto, M. A., Reis, C., et al. (2014). Long-term mortality analysis in Parkinson’s disease treated with deep brain stimulation. Parkinsons Dis. 2014:717041. doi: 10.1155/2014/717041
Rodrigues, A. M., Rosas, M. J., Gago, M. F., Sousa, C., Fonseca, R., Linhares, P., et al. (2010). Suicide attempts after subthalamic nucleus stimulation for Parkinson’s disease. Eur. Neurol. 63, 176–179. doi: 10.1159/000289097
Rodriguez, R. L., Fernandez, H. H., Haq, I., and Okun, M. S. (2007). Pearls in patient selection for deep brain stimulation. Neurologist 13, 253–260. doi: 10.1097/NRL.0b013e318095a4d5
Rosskothen-Kuhl, N., Hildebrandt, H., Birkenhäger, R., and Illing, R. B. (2018). Astrocyte hypertrophy and microglia activation in the rat auditory midbrain is induced by electrical intracochlear stimulation. Front. Cell. Neurosci. 12:43. doi: 10.3389/fncel.2018.00043
Schiavone, S., Neri, M., Mhillaj, E., Morgese, M. G., Cantatore, S., Bove, M., et al. (2016). The NADPH oxidase NOX2 as a novel biomarker for suicide: evidence from human post mortem brain samples. Transl. Psychiatry 5:e813. doi: 10.1038/tp.2016.76
Schnieder, T. P., Trencevska, I., Rosoklija, G., Stankov, A., Mann, J. J., Smiley, J., et al. (2014). Microglia of prefrontal white matter in suicide. J. Neuropathol. Exp. Neurol. 73, 880–890. doi: 10.1097/NEN.0000000000000107
Schuepbach, W. M. M., Rau, J., Knudsen, K., Volkmann, J., Krack, P., Timmermann, L., et al. (2013). Neurostimulation for Parkinson’s disease with early motor complications. N. Engl. J. Med. 368, 610–622. doi: 10.1056/NEJMoa1205158
Sequeira, A., Mamdani, F., Ernst, C., Vawter, M. P., Bunney, W. E., Lebel, V., et al. (2009). Global brain gene expression analysis links glutamatergic and GABAergic alterations to suicide and major depression. PLoS One 4:e6585. doi: 10.1371/journal.pone.0006585
Serafini, G., Parisi, V. M., Aguglia, A., Amerio, A., Sampogna, G., Fiorillo, A., et al. (2020). A specific inflammatory profile underlying suicide risk? Systematic review of the main literature findings. Int. J. Environ. Res. Public Health 17:2393. doi: 10.3390/ijerph17072393
Shepard, M. D., Perepezko, K., Broen, M. P. G., Hinkle, J. T., Butala, A., Mills, K. A., et al. (2019). Suicide in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 90, 822–829. doi: 10.1136/jnnp-2018-319815
Shimo, Y., and Wichmann, T. (2009). Neuronal activity in the subthalamic nucleus modulates the release of dopamine in the monkey striatum. Eur. J. Neurosci. 29, 104–113. doi: 10.1111/j.1460-9568.2008.06565.x
Smeding, H. M., Speelman, J. D., Koning-Haanstra, M., Schuurman, P. R., Nijssen, P., van Laar, T., et al. (2006). Neuropsychological effects of bilateral STN stimulation in Parkinson disease: a controlled study. Neurology 66, 1830–1836. doi: 10.1212/01.wnl.0000234881.77830.66
Soulas, T., Gurruchaga, J.-M., Palfi, S., Cesaro, P., Nguyen, J.-P., and Fénelon, G. (2008). Attempted and completed suicides after subthalamic nucleus stimulation for Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 79, 952–954. doi: 10.1136/jnnp.2007.130583
Soulas, T., Sultan, S., Gurruchaga, J. M., Palfi, S., and Fénelon, G. (2011). Depression and coping as predictors of change after deep brain stimulation in Parkinson’s disease. World Neurosurg. 75, 525–532. doi: 10.1016/j.wneu.2010.06.015
Spoletini, I., Piras, F., Fagioli, S., Rubino, I. A., Martinotti, G., Siracusano, A., et al. (2011). Suicidal attempts and increased right amygdala volume in schizophrenia. Schizophr. Res. 125, 30–40. doi: 10.1016/j.schres.2010.08.023
St George, R. J., Carlson-Kuhta, P., King, L. A., Burchiel, K. J., and Horak, F. B. (2015). Compensatory stepping in Parkinson’s disease is still a problem after deep brain stimulation randomized to STN or GPi. J. Neurophysiol. 114, 1417–1423. doi: 10.1152/jn.01052.2014
Steiner, J., Bielau, H., Brisch, R., Danos, P., Ullrich, O., Mawrin, C., et al. (2008). Immunological aspects in the neurobiology of suicide: elevated microglial density in schizophrenia and depression is associated with suicide. J. Psychiatr. Res. 42, 151–157. doi: 10.1016/j.jpsychires.2006.10.013
Steiner, J., Gos, T., Bogerts, B., Bielau, H., Drexhage, H. A., and Bernstein, H. G. (2013). Possible impact of microglial cells and the monocyte-macrophage system on suicidal behavior. CNS Neurol. Disord. Drug Targets 12, 971–979. doi: 10.2174/18715273113129990099
Steiner, J., Walter, M., Gos, T., Guillemin, G. J., Bernstein, H. G., Sarnyai, Z., et al. (2011). Severe depression is associated with increased microglial quinolinic acid in subregions of the anterior cingulate gyrus: evidence for an immune-modulated glutamatergic neurotransmission? J. Neuroinflammation 8:94. doi: 10.1186/1742-2094-8-94
Strutt, A. M., Simpson, R., Jankovic, J., and York, M. K. (2012). Changes in cognitive-emotional and physiological symptoms of depression following STN-DBS for the treatment of Parkinson’s disease. Eur. J. Neurol. 19, 121–127. doi: 10.1111/j.1468-1331.2011.03447.x
Suzuki, H., Ohgidani, M., Kuwano, N., Chrétien, F., Lorin de la Grandmaison, G., Onaya, M., et al. (2019). Suicide and microglia: recent findings and future perspectives based on human studies. Front. Cell. Neurosci. 13:31. doi: 10.3389/fncel.2019.00031
Tay, T. L., Carrier, M., and Tremblay, M. È. (2019). Physiology of microglia. Adv. Exp. Med. Biol. 1175, 129–148. doi: 10.1007/978-981-13-9913-8_6
Temel, Y., Blokland, A., Steinbusch, H. W., and Visser-Vandewalle, V. (2005). The functional role of the subthalamic nucleus in cognitive and limbic circuits. Prog. Neurobiol. 76, 393–413. doi: 10.1016/j.pneurobio.2005.09.005
Thelin, J., Jörntell, H., Psouni, E., Garwicz, M., Schouenborg, J., Danielsen, N., et al. (2011). Implant size and fixation mode strongly influence tissue reactions in the CNS. PLoS One 6:e16267. doi: 10.1371/journal.pone.0016267
Thion, M. S., and Garel, S. (2020). Microglial ontogeny, diversity and neurodevelopmental functions. Curr. Opin. Genet. Dev. 65, 186–194. doi: 10.1016/j.gde.2020.06.013
Thobois, S., Mertens, P., Guenot, M., Hermier, M., Mollion, H., Bouvard, M., et al. (2002). Subthalamic nucleus stimulation in Parkinson’s disease: clinical evaluation of 18 patients. J. Neurol. 5, 529–534. doi: 10.1007/s004150200059
Torres-Platas, S. G., Cruceanu, C., Chen, G. G., Turecki, G., and Mechawar, N. (2014). Evidence for increased microglial priming and macrophage recruitment in the dorsal anterior cingulate white matter of depressed suicides. Brain Behav. Immun. 2, 50–59. doi: 10.1016/j.bbi.2014.05.007
Tsuboi, T., Lemos Melo Lobo Jofili Lopes, J., Moore, K., Patel, B., Legacy, J., Ratajska, A. M., et al. (2020). Long-term clinical outcomes of bilateral GPi deep brain stimulation in advanced Parkinson’s disease: 5 years and beyond. J. Neurosurg. [Epub ahead of print]. doi: 10.3171/2020.6.JNS20617
Turecki, G., Ernst, C., Jollant, F., Labonté, B., and Mechawar, N. (2012). The neurodevelopmental origins of suicidal behavior. Trends Neurosci. 35, 14–23. doi: 10.1016/j.tins.2011.11.008
Umemura, A., Oka, Y., Yamamoto, K., Okita, K., Matsukawa, N., and Yamada, K. (2011). Complications of subthalamic nucleus stimulation in Parkinson’s disease. Neurol. Med. Chir. 51, 749–755. doi: 10.2176/nmc.51.749
van Heeringen, K., and Mann, J. J. (2014). The neurobiology of suicide. Lancet Psychiatry 1, 63–72. doi: 10.1016/S2215-0366(14)70220-2
Vedam-Mai, V., Rodgers, C., Gureck, A., Vincent, M., Ippolito, G., Elkouzi, A., et al. (2018). Deep Brain Stimulation associated gliosis: a post-mortem study. Parkinsonism Relat. Disord. 54, 51–55. doi: 10.1016/j.parkreldis.2018.04.009
Voon, V., Krack, P., Lang, A. E., Lozano, A. M., Dujardin, K., Schüpbach, M., et al. (2008). A multicentre study on suicide outcomes following subthalamic stimulation for Parkinson’s disease. Brain 13, 2720–2728. doi: 10.1093/brain/awn214
Wang, L., Zhao, Y., Edmiston, E. K., Womer, F. Y., Zhang, R., Zhao, P., et al. (2020). Structural and functional abnormities of amygdala and prefrontal cortex in major depressive disorder with suicide attempts. Front. Psychiatry 10:923. doi: 10.3389/fpsyt.2019.00923
Wei, S., Chang, M., Zhang, R., Jiang, X., Wang, F., and Tang, Y. (2018). Amygdala functional connectivity in female patients with major depressive disorder with and without suicidal ideation. Ann. Gen. Psychiatry 17:37. doi: 10.1186/s12991-018-0208-0
Weintraub, D., Duda, J. E., Carlson, K., Luo, P., Sagher, O., Stern, M., et al. (2013). Suicide ideation and behaviours after STN and GPi DBS surgery for Parkinson’s disease: results from a randomised, controlled trial. J. Neurol. Neurosurg. Psychiatry 84, 1113–1118. doi: 10.1136/jnnp-2012-304396
Wellman, S. M., Li, L., Yaxiaer, Y., McNamara, I., and Kozai, T. D. Y. (2019). Revealing spatial and temporal patterns of cell death, glial proliferation and blood-brain barrier dysfunction around implanted intracortical neural interfaces. Front. Neurosci. 13:493. doi: 10.3389/fnins.2019.00493
Williams, J., and Kobak, K. (2008). Development and reliability of a structured interview guide for the Montgomery-Åsberg Depression Rating Scale (SIGMA). Br. J. Psychiatry 192, 52–58. doi: 10.1192/bjp.bp.106.032532
Wilson, S. J., Bladin, P. F., and Saling, M. M. (2004). Paradoxical results in the cure of chronic illness: the “burden of normality” as exemplified following seizure surgery. Epilepsy Behav. 5, 13–21. doi: 10.1016/j.yebeh.2003.11.013
Wilson, S., Bladin, P., and Saling, M. (2001). The “burden of normality”: concepts of adjustment after surgery for seizures. J. Neurol. Neurosurg. Psychiatry 70, 649–656. doi: 10.1136/jnnp.70.5.649
Wilson, S. J., Saling, M., Lawrence, J. A., and Bladin, P. F. (1999). Outcome of temporal lobectomy: expectations and the prediction of perceived success. Epilepsy Res. 36, 1–14. doi: 10.1016/s0920-1211(99)00016-9
Witt, K., Daniels, C., Reiff, J., Krack, P., Volkmann, J., Pinsker, M. O., et al. (2008). Neuropsychological and psychiatric changes after deep brain stimulation for Parkinson’s disease: a randomised, multicentre study. Lancet Neurol. 7, 605–614. doi: 10.1016/S1474-4422(08)70114-5
Wrench, J., Wilson, S. J., and Bladin, P. F. (2004). Mood disturbance before and after seizure surgery: a comparison of temporal and extratemporal resections. Epilepsia 45, 534–543. doi: 10.1111/j.0013-9580.2004.48803.x
Xu, Y., Yang, B., Zhou, C., Gu, M., Long, J., Wang, F., et al. (2021). Suicide and suicide attempts after subthalamic nucleus stimulation in Parkinson’s disease: a systematic review and meta-analysis. Neurol. Sci. 42, 267–274. doi: 10.1007/s10072-020-04555-7
Yuan, Y., Wu, C., and Ling, E. A. (2019). Heterogeneity of microglia phenotypes: developmental, functional and some therapeutic considerations. Curr. Pharm. Des. 25, 2375–2393. doi: 10.2174/1381612825666190722114248
Zarzycki, M. Z., and Domitrz, I. (2020). Stimulation-induced side effects after deep brain stimulation—a systematic review. Acta Neuropsychiatr. 32, 57–64. doi: 10.1017/neu.2019.35
Zhang, J., Li, J., Chen, F., Liu, X., Jiang, C., Hu, X., et al. (2020). STN versus GPi deep brain stimulation for dyskinesia improvement in advanced Parkinson’s disease: a meta-analysis of randomized controlled trials. Clin. Neurol. Neurosurg. 201:106450. doi: 10.1016/j.clineuro.2020.106450
Zhao, J., Verwer, R. W. H., Gao, S.-F., Qi, X.-R., Lucassen, P. J., Kessels, H. W., et al. (2018). Prefrontal alterations in GABAergic and glutamatergic gene expression in relation to depression and suicide. J. Psychiatr. Res. 102, 261–274. doi: 10.1016/j.jpsychires.2018.04.020
Keywords: deep brain stimulation, suicide, neuroinflammation, extrapyramidal diseases,suicidal ideation (SI), suicidal behavior (SB), suicide attempt (SA), Parkinson’s disease
Citation: Costanza A, Radomska M, Bondolfi G, Zenga F, Amerio A, Aguglia A, Serafini G, Amore M, Berardelli I, Pompili M and Nguyen KD (2021) Suicidality Associated With Deep Brain Stimulation in Extrapyramidal Diseases: A Critical Review and Hypotheses on Neuroanatomical and Neuroimmune Mechanisms. Front. Integr. Neurosci. 15:632249. doi: 10.3389/fnint.2021.632249
Received: 22 November 2020; Accepted: 15 March 2021;
Published: 08 April 2021.
Edited by:
Elizabeth B. Torres, The State University of New Jersey, United StatesReviewed by:
Xinglong Yang, The First Affiliated Hospital of Kunming Medical University, ChinaJennifer R. Stapleton-Kotloski, Wake Forest School of Medicine, United States
Copyright © 2021 Costanza, Radomska, Bondolfi, Zenga, Amerio, Aguglia, Serafini, Amore, Berardelli, Pompili and Nguyen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Alessandra Costanza,YWxlc3NhbmRyYS5jb3N0YW56YUB1bmlnZS5jaA==
 Alessandra Costanza
Alessandra Costanza Michalina Radomska
Michalina Radomska Guido Bondolfi
Guido Bondolfi Francesco Zenga
Francesco Zenga Andrea Amerio
Andrea Amerio Andrea Aguglia
Andrea Aguglia Gianluca Serafini
Gianluca Serafini Mario Amore
Mario Amore Isabella Berardelli
Isabella Berardelli Maurizio Pompili
Maurizio Pompili Khoa D. Nguyen
Khoa D. Nguyen