- 1Renal Division, Peking University First Hospital, Beijing, China
- 2Peking University Institute of Nephrology, Beijing, China
- 3Key Laboratory of Renal Disease, Ministry of Health of China, Beijing, China
- 4Key Laboratory of Chronic Kidney Disease Prevention and Treatment, Peking University, Ministry of Education, Beijing, China
- 5Research Central Institute, Peking University First Hospital, Beijing, China
Low FCGR3 copy numbers (CNs) has been associated with susceptibility to several systemic autoimmune diseases. However, inconsistent associations were reported and errors caused by shaky methods were suggested to be the major causes. In large scale case control association studies, robust copy number determination method is thus warranted, which was the main focus of the current study. In the present study, FCGR3 CNs of 90 HapMap Asians were firstly checked using four assays including paralog ratio test combined with restriction enzyme digest variant ratio (PRT-REDVR), real-time quantitative (qPCR) using TaqMan assay, real-time qPCR using SYBR Green dye and short tenden repeat (STR). To improve the comparison precision reproductively, the results were compared with those from recently released sequencing data from 1000 genomes project as well as whole-genome tiling BAC array data. The tendencies of inconsistent samples by these methods were also characterized. Refined in-home TaqMan qPCR assay showed the highest correlation with array-CGH results (r = 0.726, p < 0.001) and the highest concordant rate with 1000 genome sequencing data (FCGR3A 91.76%, FCGR3B 85.88%, and FCGR3 81.18%). For samples with copy number variations, comprehensive analysis of multiple methods was required in order to improve detection accuracy. All these method were prone to detect copy number to be higher than that from direct sequencing. All the four PCR based CN determination methods (qPCR using TaqMan probes or SYBR Green, PRT, STR) were prone to higher estimation errors and thus may lead to artificial associations in large-scale case-control association studies. But different to previous reports, we observed that properly refined TaqMan qPCR assay was not inferior to or even more accurate than PRT when using sequencing data as the reference.
Introduction
Fcγ receptors were cellular receptors, which were encoded by FCGR3 locus, for the Fc region of IgG and IgE, and could transmit their signal by tyrosine-based activation (ITAM) or inhibitory motifs (ITIM). Low-affinity activating receptor FcγRIII genes, which encoded FcγRIIIA and FcγRIIIB, were located in 1q23.3 with extensive copy number variation (CNV) (Redon et al., 2006; Nimmerjahn and Ravetch, 2008). Low copy number of FCGR3B had been identified to be associated with a number of systemic autoimmune diseases, such as systemic lupus erythematosus (SLE), rheumatoid arthritis (RA), ANCA-associated vasculitis (AAV), and anti-glomerular basement (anti-GBM disease) disease (Fanciulli et al., 2007; Willcocks et al., 2008; Mamtani et al., 2010; McKinney et al., 2010; Niederer et al., 2010; Zhou et al., 2010, 2011; Molokhia et al., 2011; Chen et al., 2014). However, results for disease associations were not consistent, even for the same phenotype in populations within the same ancestry. Phenotype and genetic heterogeneity might be confounding factors. However, methodology discrepancies were suggested to be the major factor leading to the conflicting results. Certain proportion of errors might lead to false associations, especially when the effect of gene risk was moderate. Thus, in large-scale genetic association studies, easy, economic, robust, and accurate methods of FCGR CN genotyping were prerequisites.
Up to date, multiple assays for FCGR CN genotyping had been developed, including real-time quantitative PCR (qPCR), paralog ratio test combined with restriction enzyme digest variant ratio (PRT-REDVR), multiples ligation-dependent probe amplification (MLPA), short tenden repeat (STR), comparative genomic hybridization (CGH), and direct sequencing (Aitman et al., 2006; Fanciulli et al., 2007; Breunis et al., 2008, 2009; Willcocks et al., 2008; Mamtani et al., 2010). But due to high similarity and CN complexity in the region, no single method or single test was suggested to be ideal in fastness, accuracy and economy. Though sequencing data was the gold standard, it was not suitable for large scale case-control study considering the expenses. Our aim was to evaluate the efficacy of the widely used methods and find a robust assay with a low error rate for further large-scale case control genetic association study. In the current study, we determined the copy numbers for 90 HapMap Chinese Han from Beijing (CHB) and Japanese from Tokyo (JPT) individuals using four copy number assays: PRT-REDVR, TaqMan qPCR, SYBR Green qPCR, and STR (Fanciulli et al., 2007; Hollox et al., 2009; Zhou et al., 2010), as these methods were more widely taken and economical for large-scale association. We compared our results of each method with the copy numbers from established open database including the whole-genome tiling BAC array data and the 1000 genome sequencing data.
Notably, thanks to the 1000 Genomes Project phase 3, an integrated map of structural variation in 2504 human genomes which was constructed in 2015. It presented the most comprehensive set of human structural variants to date. As an integrated resource for disease and population genetic studies, it represented an invaluable resource for the construction and analysis of personalized genomes. In terms of method comparison, it was also of special importance, as it revealed CN variations at individual instead of pooling level (Sudmant et al., 2015). Thus, to improve the comparison efficiency, the sequencing data from 1000G samples represented as the gold standard available compared to previous reports replying on CGH data. In this way, we were aiming to find a method in FCGR3 copy number detection which was the fastest and cheapest without scarifying accuracy.
Materials and Methods
Subjects
The study population was based on the HapMap Phase II Han Chinese individuals from Beijing (CHB, n = 45) and Japanese individuals from Tokyo (JPT, n = 45). Genomic DNA was purchased from the Coriell Cell Repositories (Catalog ID: HAPMAPPT02). This study was approved by the Medical Ethics Committee of Peking University First Hospital.
Real-Time Quantitative PCR (qPCR)
TaqMan qPCR System
FCGR3B and FCGR3B copy number (CN), determined by Quantitative PCR (qPCR), was performed on an Applied Biosystems 7500 (Foster City, CA, USA) as previously reported (Zhou et al., 2010). Primers and TaqMan probes specifically amplify the target gene were designed to avoid paralogous or allelic sequence variants. Coagulation factor V gene (F5) was included as an internal control for copy number. The qPCRs was in 50 ul reaction system including 80 ng genomic DNA, 5ul 10 × PCR buffer, 4 ul 2.5 mM deoxyribonucleoside triphosphate, 15 pmol/each forward primers, 15 pmol/each reverse primers, 5 pmol/each TaqMan probes, 1.5 U Taq polymerase (Takara, Dalian, China), and 26.7ul ddH2O. Cycling conditions were 94°C for 10 min, 40 cycles with 10 s denaturation step at 94°C, followed by 62°C (FCGR3B or FCGR3B) annealing step for 45 s and a 72°C extension step for 10 s (Zhou et al., 2010). The target gene and the control gene were amplified in the same tube and each test was run in triplicate. The standard curves to test the efficiency of the assay were run using independent genomic DNA (Zhou et al., 2010).
SYBR Green qPCR System
FCGR3B copy number assay was also carried out by SYBR Green qPCR system, performed on an Applied Biosystems 7500 (Foster City, CA, USA) and analyzed by the relative standard curve method. The primers, qPCR reactions, and calculation were referred to previous reports (Aitman et al., 2006).
Paralog Ratio Test (PRT)
The primer pair FCGR3A/FCGR3B co-amplified in FCGR3A and FCGR3B. The other primer pair, FCGR3/c18, amplified a sequence of the same length in both target genes and a third region in Chromosome 18 (Niederer et al., 2010). The amplification of 5–10 ng of genomic DNA in PCR reaction was performed using Phusion Hot Start High Fidelity polymerase (Finnzymes) with the HF buffer. The cycling conditions were 98°C for 60 s, then 30 cycles at 98°C for 10 s, annealing at 65°C (for FCGR3A/FCGR3B) or 60°C (for FCGR3/c18) for 20 s and elongation at 72°C for 30 s (for FCGR3A/FCGR3B) or 5 s (for FCGR3/c18), followed by 72°C for 7 min to reduce single stranded DNA products (Niederer et al., 2010). The PCR products were added to Hi-DiTM Formamide with the fluorescent GeneScanTM 500 LIZTM Size Standard (Applied Biosystems) and the following analysis were performed on ABI 3730 × l DNA Analyzer/Sequencer (Applied Biosystems). Peak areas corresponding to the two products were recorded for HEX-labeled products using GeneMapper 4.0 software (Applied Biosystems).
Restriction Enzyme Digest Variant Ratio (REDVR)
In the present study, we performed two REDVR assays in order to distinguish the variants from FCGR3B and FCGR3B (C733 Arginine > Stop) and to distinguish human neutrophil antigens (HNA) HNA1a and HNA1b on FCGR3B (C147T). The primer sequences were referred to previously published article (Hollox et al., 2009). We amplified the two regions in duplex using the same conditions as the PRT described above with some modification. Copy number calls were estimated based on mean ratios of the product and the reference standard for experimental calibration. A maximum likelihood approach was used to analyze the PRT copy number calls in combination with the REDVR analysis.
Short Tandem Repeat (STR)
The primers for STR could be referred to previously report (Hollox et al., 2009). The PCR condition was based on PRT with some modifications, that was, 26 cycles for amplification and 2 μl for capillary electrophoresis analysis. During amplification, the wrong replication of Taq DNA polymerase would generate slippage peaks in MSAT1 amplification. The 2 bp larger real peak would add to the area under the slippage peak in case that the two peaks failed to overlap. It was difficult to distinguish a slippage peak from a real peak which were 2 bp. Given that the amount of slippage peak was dependent on the proportional to variant length, we corrected a length dependent factor to real peaks which coinciding with slippage peaks. If it showed more than two peaks, the peak which had the second smallest area was used to divide the area of each peak. If it showed more than four peaks, the peak with the third smallest area was used to divide the area of each peak. In this way, we determined the value for copy numbers after slippage correction.
Results
Respective Comparisons among All the 4 PCR Based CN Determination Assays
In the present study, we performed 4 methods with 3 repeats (TaqMan qPCR, PRT-REDVR, SYBR Green qPCR, and STR) to estimate the copy numbers for FCGR3 locus in CHB (n = 45) and JPT (n = 45) populations (detailed copy number results for FCGR3A, FCGR3B, and FCGR3 by each method was shown in Table S1).
We next analyzed the concordant rate for FCGR3A, FCGR3B, and FCGR3 with two or three assays (TaqMan qPCR, PRT, REDVR, SYBR Green qPCR).
In two assay concordant analysis, TaqMan qPCR showed the highest concordant rate with other methods. The concordant rates were 77.78 ~ 91.11% (91.11% for FCGR3A, 84.44% for FCGR3B, and 77.78% for FCGR3) between TaqMan qPCR and PRT-REDVR, 74.44% for FCGR3B between TaqMan qPCR and SYBR Green, and 76.67% for FCGR3 between TaqMan qPCR and STR. SYBR Green (74.44% with TaqMan qPCR and 65.56% with PRT-REDVR for FCGR3B) and STR (76.67% with TaqMan qPCR and 61.11% with PRT-REDVR for FCGR3) showed the lowest concordant rate with other methods. In three assay analysis, the concordant rates were much poorer, 62.22% for FCGR3B (TaqMan qPCR, PRT-REDVR and SYBR Green) and 60.00% for FCGR3 (TaqMan qPCR, PRT-REDVR and STR). This result was consistent with previous observations (Haridan et al., 2015).
Not only TaqMan and SYBR Green were qPCR based methods, PRT, REDVR, and STR were also partly qPCR based in methodology. They might skew toward same mistakes in copy number detection. In general, even if these methods showed good concordant rate with each other, it was hard to determine which one was the most suitable method in FCGR3A, FCGR3B, and FCGR3 copy number detection. In order to solve the problem, all the results by these methods (TaqMan qPCR, PRT-REDVR, and STR) should be tested with a “gold standard” in paired samples.
Correlation with the Whole-Genome Tiling BAC Array Data
We used whole-genome tiling BAC array data from the Wellcome Trust Sanger Institute in paired samples as reference, which was more widely used in previous method reports (Redon et al., 2006). As shown in Figure 1, all of TaqMan qPCR, PRT and STR results showed a significant correlation with array-CGH results (p < 0.001). Amongst, TaqMan qPCR assay showed the highest correlation with array-CGH results with r = 0.726 (p < 0.001). There was also a significant correlation between PRT (r = 0.667, p < 0.001), STR (r = 0.677, p < 0.001) with the array-CGH results (Figure 1). It also indicated that all the four methods had good performance for CN calculation in tendency.
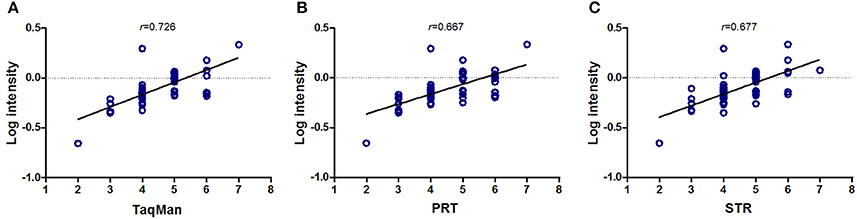
Figure 1. Correlation analysis of copy number measurements with array-CGH. TaqMan qPCR (A, r = 0.726), PRT-REDVR (B, r = 0.667) and STR (C, r = 0.677) were significant correlated with array-CGH results (p < 0.001).
Correlation with the 1000 Genome Sequencing Data
However, copy numbers were integers instead of linear ones. We conducted a concordant analysis between our TaqMan qPCR, PRT, REDVR, STR, SYBR Green qPCR results of FCGR3A, FCGR3B, FCGR3 and the sequencing data from the 1000 genome phase-3-structural-variant-dataset in paired samples (Sudmant et al., 2015) (Figure 2). A significantly increased inconsistency was observed for every method.
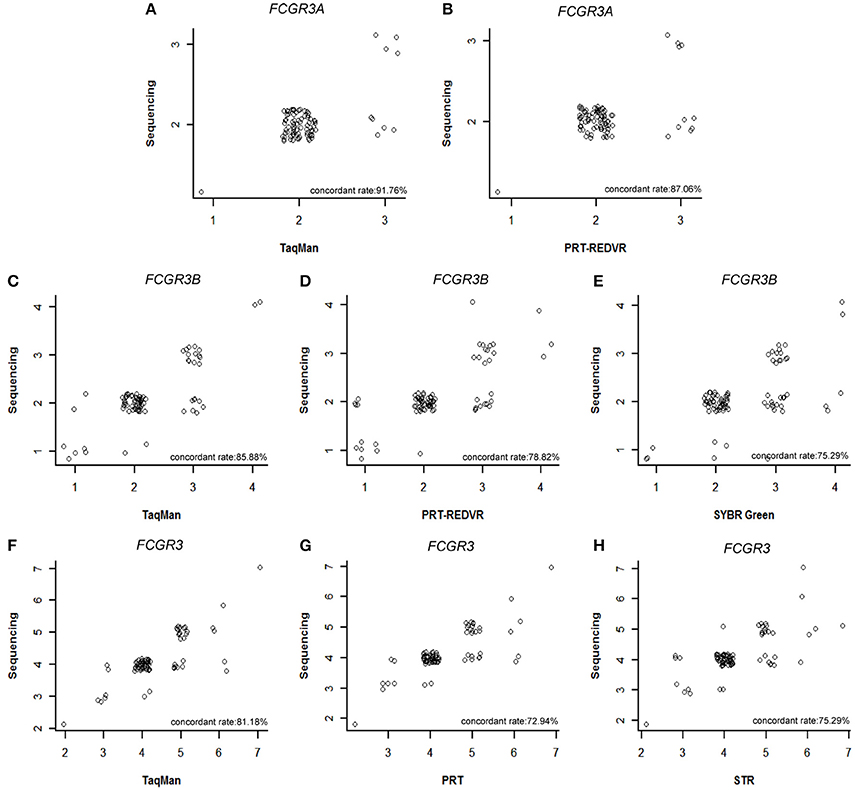
Figure 2. Concordant analysis of CNV measurement with 1000 genome sequencing data. Concordant rate between 1000 genome sequencing data and PRT-REDVR of FCGR3A (A), 1000 genome sequencing data and TaqMan qPCR of FCGR3A (B), 1000 genome sequencing data and PRT-REDVR of FCGR3B (C), 1000 genome sequencing data and SYBR qPCR of FCGR3B (D), 1000 genome sequencing data and TaqMan qPCR of FCGR3B (E), 1000 genome sequencing data and PRT of FCGR3 (F), 1000 genome sequencing data and STR of FCGR3 (G), 1000 genome sequencing data and TaqMan qPCR of FCGR3 (H).
For single method precision, TaqMan qPCR assay still showed the highest concordant rate with the sequencing results for FCGR3A (91.76%), FCGR3B (85.88%), and FCGR3 (81.18%). PRT-REDVER showed the lowest concordant rate for FCGR3 (72.94%). The concordant rate was also low in SYBR Green qPCR assay for FCGR3B (75.29%) and STR assay for FCGR3 (75.29%).
Error was observed in almost every copy number detection assay (Figure 2). Although there was 0–5% error rate for wrong classification of varied copy numbers, it was strange that the highest error rate was observed in FCGR3A and FCGR3B 2-copy (8.24–20.00% error rate) and FCGR3 4-copy (14.12–17.65%). For 2-copy detection, TaqMan showed the lowest error rate 8.24% for FCGR3A, 11.76% for FCGR3B, and 14.12% for FCGR3. PRT showed a higher error rate in 2-copy detection, 12.94% for FCGR3A, 16.47% for FCGR3B, and 17.65% for FCGR3. The SYBR Green showed the highest error rate with 20.00% in FCGR3B 2-copy detection. This observation suggested that erroneous copy number determination might be increased due to wrong counting of the diploid nature of the genome.
And in all methods, the wrong detections tended to be higher estimation (Figure 3). 2-copy for FCGR3A, 1-copy and 2-copy for FCGR3B, and 3-copy, 4-copy, and 5-copy for FCGR3 were skewing toward higher estimated in all the utilized methods. FCGR3A showed that wrong detected samples by TaqMan qPCR and PRT-REDVR were all estimated to be higher. For FCGR3B and FCGR3 wrong detected samples, both higher and lower estimations were observed in TaqMan qPCR, PRT, REDVR, and STR, in which higher estimation took up the majority. 100% SYBR Green wrong detected samples were higher estimated. There was a tendency that samples with lower (2-copy for FCGR3A, 1-copy, 2-copy, 3-copy for FCGR3B, and 2-copy, 3-copy, 4-copy, and 5-copy for FCGR3) copy number were prone to higher estimation. Samples with higher copy numbers (4-copy for FCGR3B, 6-copy and 7-copy for FCGR3) were tended to be lower estimated.
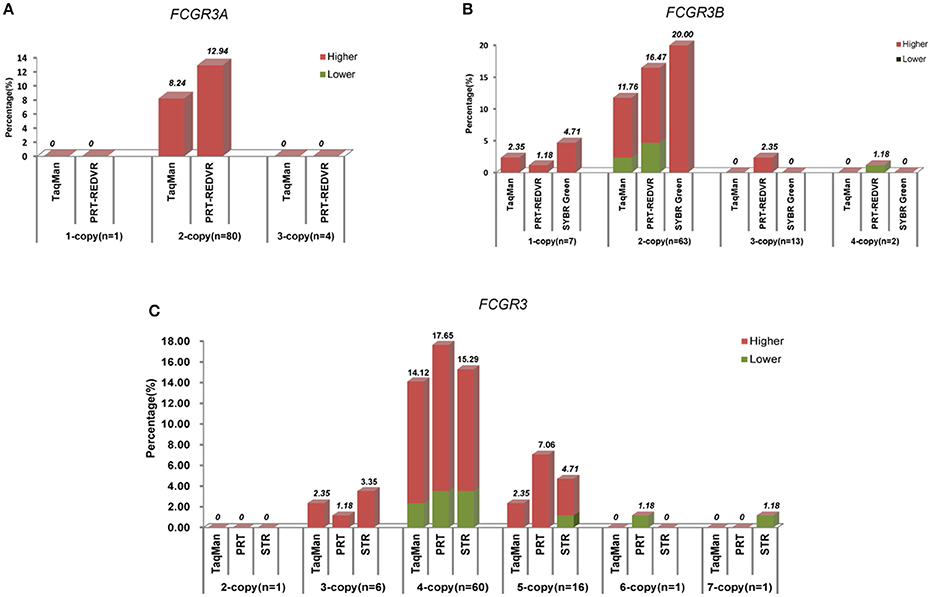
Figure 3. Characteristics of wrong detected copy numbers. The percentage (%) of wrong detected copy number by every measurement for each copy of FCGR3A (A), FCGR3B (B), and FCGR3 (C) was labeled above the column. If the copy number were detected higher than the sequencing data, the column would be colored red. If the copy number were detected lower than the sequencing data, the column would be colored green.
Discussion
In the present study, after the comparison of PRT-REDVR, TaqMan qPCR, SYBR Green qPCR, and STR, we found that TaqMan qPCR was the statistically supported method which could detect the copy number more accurately at FCGR3 region in the HapMap CHB and JPT population in our institute. qPCR was widely used and criticized by its veracity and reliability as it could potentially introduce false positive calls. In our research, this problem might be involved in our SYBR Green qPCR assay. SYBR Green qPCR showed the lowest concordant rate and correlation with results from database in FCGR3B detection. Therefore, we refined the method of TaqMan qPCR in FCGR3A, FCGR3B, and FCGR3 copy number determination. And TaqMan qPCR showed the highest concordant rate with 1000 genome sequencing data and correlation with the array-CGH results. Our data showed that TaqMan qPCR assay might be an option for future high throughput case-control study.
We used two standard populations from the HapMap project, 45 CHB and 45 JPT individuals. Previous studies by other groups also defined the copy number of HapMap CHB and JPT populations. We also compared the previously reported results with 1000 genome sequencing data and array-CGH result in paired samples (Hollox et al., 2009). The correlation between previously reported data and array-CGH results was 0.618 (p < 0.001) and the concordant rate was 85.88% for FCGR3A, 78.82% for FCGR3B and 70.59% for FCGR3 comparing with 1000 genome sequencing data. The differences in copy number detection in one sample by the four methods (Taqman, SYBR Green, PRT-REDVR, and STR) individually could reflect either measurement error or real copy number heterogeneity between repeats. Meanwhile, the integration of multiple methods could improve the accuracy. The integrated copy number FCGR3 showed 89.4% consistency with the 1000 genome sequencing data which was higher than using PRT (72.94%), STR (75.29%), and Taqman (81.18%) alone. The integrated copy number FCGR3 also showed higher (0.932) correlation with the whole-genome tiling BAC array data than using PRT (0.667), STR (0.677), and Taqman (0.726) alone. Comparing with the result of single method, the integration by several methods would surely improve the accuracy. Our PRT and STR results were successfully replicated the previous reported data which also based on these two methods. However, TaqMan qPCR refined by our laboratory showed better performance than PRT. Our finding suggested that before case-control association studies, methodologies should be confirmed with standard population. That is to detect certain gene copy number using multiple methods in the standard population and followed by comparing the results with array-CGH results or 1000 genome sequencing data for methodology evaluation. Array-CGH reflects copy numbers based on relative dosage signal and the 1000 genome sequencing data precisely presented every copy number for each sample from a certain locus. Array-CGH had been utilized for detection evaluation by other groups (Hollox et al., 2009). However, we proposed that 1000 genome sequencing data would be a better option and array-CGH could be an implement in methodology evaluation. In this way, any laboratory could find a suitable method for copy number detection with minimum error rate. And method refinement would further improve the accuracy of detection.
Our study aims to find a better way to determine FCGR3 genomic copy number in Asia populations. Nevertheless, TaqMan qPCR, PRT-REDVR, STR, SYBR Green qPCR were prone to detect a higher copy number. Therefore, in order to improve the accuracy in copy number determination in samples with high copy numbers, a combination of two or more methods was required. Our refined TaqMan qPCR alone was statistically supported as an option in FCGR3 copy number determination for future case-control association studies. Advantages include small amount of DNA, high-throughput analysis, low running cost, and feasibility of equipment. However, this method could potentially introduce false positive results and DNA integrity should also be warranted. We hoped such endeavor will benefit future case-control studies with regard to complex CNVs, particularly for disease associations with genetic variations of FcγRs.
Author Contributions
XZ and HZ designed the research; XZ, DB, PH, JL and YQ performed the research; YQ analyzed the data; and YQ and XZ wrote the paper.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This work was supported by grants from the National Key Research and Development Program of China (2016YFC0904100); National Key Technology R&D Program (2013BAI09B14); Natural Science Foundation for Innovation Research Group of China (81321064); Natural Science Foundation for Excellent Young Scientists (813220065); National Science Foundation of China (Grant No. 81270795; 81470945; 81570629); Capital of Clinical Characteristics and the Applied Research Fund (Z141107002514037); Beijing Municipal Science and Technology for the Outstanding Ph.D. Program (20121000110); Beijing Natural Science Foundation (Grant No. 7131016; 7152148) and Chinese Society of Nephrology (15020030591). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fgene.2016.00220/full#supplementary-material
References
Aitman, T. J., Dong, R., Vyse, T. J., Norsworthy, P. J., Johnson, M. D., Smith, J., et al. (2006). Copy number polymorphism in Fcgr3 predisposes to glomerulonephritis in rats and humans. Nature 439, 851–855. doi: 10.1038/nature04489
Breunis, W. B., van Mirre, E., Bruin, M., Geissler, J., de Boer, M., Peters, M., et al. (2008). Copy number variation of the activating FCGR2C gene predisposes to idiopathic thrombocytopenic purpura. Blood 111, 1029–1038. doi: 10.1182/blood-2007-03-079913
Breunis, W. B., van Mirre, E., Geissler, J., Laddach, N., Wolbink, G., van der Schoot, E., et al. (2009). Copy number variation at the FCGR locus includes FCGR3A, FCGR2C and FCGR3B but not FCGR2A and FCGR2B. Hum. Mutat. 30, E640–E650. doi: 10.1002/humu.20997
Chen, J. Y., Wang, C. M., Chang, S. W., Cheng, C. H., Wu, Y. J., Lin, J. C., et al. (2014). Association of FCGR3A and FCGR3B copy number variations with systemic lupus erythematosus and rheumatoid arthritis in Taiwanese patients. Arthrit. Rheumatol. 66, 3113–3121. doi: 10.1002/art.38813
Fanciulli, M., Norsworthy, P. J., Petretto, E., Dong, R., Harper, L., Kamesh, L., et al. (2007). FCGR3B copy number variation is associated with susceptibility to systemic, but not organ-specific, autoimmunity. Nat. Genet. 39, 721–723. doi: 10.1038/ng2046
Haridan, U. S., Mokhtar, U., Machado, L. R., Abdul Aziz, A. T., Shueb, R. H., Zaid, M., et al. (2015). A comparison of assays for accurate copy number measurement of the low-affinity Fc gamma receptor genes FCGR3A and FCGR3B. PLoS ONE 10:e0116791. doi: 10.1371/journal.pone.0116791
Hollox, E. J., Detering, J. C., and Dehnugara, T. (2009). An integrated approach for measuring copy number variation at the FCGR3 (CD16) locus. Hum. Mutat. 30, 477–484. doi: 10.1002/humu.20911
Mamtani, M., Anaya, J. M., He, W., and Ahuja, S. K. (2010). Association of copy number variation in the FCGR3B gene with risk of autoimmune diseases. Genes Immun. 11, 155–160. doi: 10.1038/gene.2009.71
McKinney, C., Fanciulli, M., Merriman, M. E., Phipps-Green, A., Alizadeh, B. Z., Koeleman, B. P., et al. (2010). Association of variation in Fcγ receptor 3B gene copy number with rheumatoid arthritis in Caucasian samples. Ann. Rheum. Dis. 69, 1711–1716. doi: 10.1136/ard.2009.123588
Molokhia, M., Fanciulli, M., Petretto, E., Patrick, A. L., McKeigue, P., Roberts, A. L., et al. (2011). FCGR3B copy number variation is associated with systemic lupus erythematosus risk in Afro-Caribbeans. Rheumatology 50, 1206–1210. doi: 10.1093/rheumatology/keq456
Niederer, H. A., Willcocks, L. C., Rayner, T. F., Yang, W., Lau, Y. L., Williams, T. N., et al. (2010). Copy number, linkage disequilibrium and disease association in the FCGR locus. Hum. Mol. Genet. 19, 3282–3294. doi: 10.1093/hmg/ddq216
Nimmerjahn, F., and Ravetch, J. V. (2008). Fcγ receptors as regulators of immune responses. Nat. Rev. Immunol. 8, 34–47. doi: 10.1038/nri2206
Redon, R., Ishikawa, S., Fitch, K. R., Feuk, L., Perry, G. H., Andrews, T. D., et al. (2006). Global variation in copy number in the human genome. Nature 444, 444–454. doi: 10.1038/nature05329
Sudmant, P. H., Rausch, T., Gardner, E. J., Handsaker, R. E., Abyzov, A., Huddleston, J., et al. (2015). An integrated map of structural variation in 2,504 human genomes. Nature 526, 75–81. doi: 10.1038/nature15394
Willcocks, L. C., Lyons, P. A., Clatworthy, M. R., Robinson, J. I., Yang, W., Newland, S. A., et al. (2008). Copy number of FCGR3B, which is associated with systemic lupus erythematosus, correlates with protein expression and immune complex uptake. J. Exp. Med. 205, 1573–1582. doi: 10.1084/jem.20072413
Zhou, X. J., Lv, J. C., Bu, D. F., Yu, L., Yang, Y. R., Zhao, J., et al. (2010). Copy number variation of FCGR3A rather than FCGR3B and FCGR2B is associated with susceptibility to anti-GBM disease. Int. Immunol. 22, 45–51. doi: 10.1093/intimm/dxp113
Keywords: copy number, FCGR3, TaqMan qPCR assay, HapMap CHB, PRT
Citation: Qi Y-y, Zhou X-j, Bu D-f, Hou P, Lv J-c and Zhang H (2016) Comparison of Multiple Methods for Determination of FCGR3A/B Genomic Copy Numbers in HapMap Asian Populations with Two Public Databases. Front. Genet. 7:220. doi: 10.3389/fgene.2016.00220
Received: 11 September 2016; Accepted: 12 December 2016;
Published: 26 December 2016.
Edited by:
Mogens Fenger, Senior Consultant, DenmarkReviewed by:
Jing Li, Case Western Reserve University, USAYi Zhang, University of Chinese Academy of Sciences, China
Copyright © 2016 Qi, Zhou, Bu, Hou, Lv and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hong Zhang, hongzh@bjmu.edu.cn
†These authors have contributed equally to this work.
 Yuan-yuan Qi
Yuan-yuan Qi Xu-jie Zhou
Xu-jie Zhou Ding-fang Bu5
Ding-fang Bu5 Hong Zhang
Hong Zhang