- 1Section of Cardiac Surgery, Department of Cardiac Sciences, Cumming School of Medicine, Libin Cardiovascular Institute of Alberta, University of Calgary, Calgary, AB, Canada
- 2Department of Radiology, Feinberg School of Medicine, Northwestern University, Chicago, IL, United States
- 3Department of Bioengineering, Feinberg School of Medicine, Northwestern University, Chicago, IL, United States
- 4Martha and Richard Melman Family Bicuspid Aortic Valve Program, Division of Cardiothoracic Surgery, Bluhm Cardiovascular Institute, Northwestern University, Chicago, IL, United States
Bicuspid aortic valve (BAV) is a common congenital cardiac malformation affecting 1–2% of people. BAV results from fusion of two adjacent aortic valve cusps, and is associated with dilatation of the aorta, known as bicuspid valve associated aortopathy. Bicuspid valve aortopathy is progressive and associated with catastrophic clinical events, such as aortic dissection and rupture. Therefore, frequent monitoring and early intervention with prophylactic surgical resection of the proximal aorta is often recommended. However, the specific pattern of aortopathy is highly variable among patients, with different segments of the ascending aorta being affected. Individual patient risks are sometimes difficult to predict. Resection strategies are informed by current surgical guidelines which are primarily based on aortic size and growth criteria. These criteria may not optimally reflect the risk of important aortic events. To address these issues in the care of patients with bicuspid valve aortopathy, our translational research group has focused on validating use of novel imaging techniques to establish non-invasive hemodynamic biomarkers for risk-stratifying BAV patients. In this article, we review recent efforts, successes, and ongoing challenges in the development of more precise and individualized surgical approaches for patients with bicuspid aortic valves and associated aortic disease.
Introduction
Bicuspid aortic valve (BAV) is the most common congenital heart defect, affecting 1–2% of the general population (Hoffman and Kaplan, 2002). Abnormality of the aorta is frequently associated with BAV, with thoracic aortic dilation seen in approximately 40% of patients in referral centers (Masri et al., 2016). Consequently, compared to the general population, patients with BAV are at a higher risk for acute aortic emergencies, such as aortic dissection (Januzzi et al., 2004). Given the high morbidity and mortality associated with these emergencies, identifying the optimal timing to intervene, and prevent such events is of paramount importance. However, this is a challenging process as many factors, including patient age, comorbidities, presence or absence of aortic valvular disorders, and family history of BAV, could all affect management.
Over the past three decades, it was perceived that aortopathy associated with BAV, “bicuspid aortopathy,” had a similar pathophysiology to aortic disorders associated with tricuspid aortic valve (TAV) disease. Specifically, it was believed that turbulent or eccentric flow resulting from a narrowed orifice (BAV) led to aortic dilation. However, several ensuing studies demonstrated a strong genetic component for BAV-associated aortopathy in this patient population, which in turn, significantly increases the risk of acute aortic events. These initial findings led to recommendations for more aggressive management approaches, which viewed bicuspid aortopathy in the same light as Marfan's syndrome, thereby advocating for earlier surgical intervention for patients with BAV disease. More recent research, however, has implied that genetic predisposition and hemodynamic irregularities contribute to varying degrees in different subgroups of BAV patients, and the rate of aortic complications is not as high as previously believed (Fedak et al., 2005; Hiratzka et al., 2010; Girdauskas et al., 2011; Itagaki et al., 2015; Sherrah et al., 2016). These recent studies emphasize the importance of identifying the underlying cause of bicuspid aortopathy as it has different therapeutic implications for patients with or without BAV presenting with aortic pathologies.
A few groups have considered the optimal management of BAV-associated aortopathy, and several documents have addressed it, with the first being a multi-societal set of guidelines published in 2010 (Hiratzka et al., 2010). In the more recently published guidelines by the American Heart Association/American College of Cardiology (AHA/ACC) on valvular heart disease, a more conservative set of recommendations were made (Nishimura et al., 2014). Given the significant difference in recommendations, a recent clarification statement was published (Hiratzka et al., 2016). The European Society of Cardiology has also made more conservative recommendations for the management of bicuspid aortopathy (Vahanian et al., 2012; Erbel et al., 2014). In addition, the American Association for Thoracic Surgery (AATS) will be releasing an expert consensus statement in 2017.
Emerging research is considering the genetics and molecular and cellular mechanisms underlying the disease. As Prakash and colleagues elegantly outline, autosomal-dominant transmission of BAV was observed in some 3-generation pedigrees, but there is no single-gene model which clearly explains BAV inheritance. The prevalence of BAV stands nearly 10-fold higher in primary relatives of patients with BAV than in the general population, further supporting the notion that genetics does indeed play an important role (Prakash et al., 2014). To better understand the mechanisms which drive BAV and bicuspid aortopathy, different groups are studying various molecular pathways and genetic foci. Thus far, NOTCH1 remains the only gene which has been implicated for isolated BAV identified using linkage analysis and positional cloning strategies, despite probably being the cause of small proportion of familial cases (Garg et al., 2005; Ellison et al., 2007). These studies are all in their infancy, but continued basic research in this area will undoubtedly shed more light onto the genetic building block of BAV and bicuspid aortopathy.
Bicuspid aortopathy is a very heterogeneous disorder, a feature which has added to the complexity of devising management guidelines. For example, in some instances, despite developing aortopathy, patients can be asymptomatic throughout their life. Moreover, dilation of the aorta may occur in the aortic root, ascending aorta, proximal aortic arch, or a combination of any of these three (Fazel et al., 2008). Moreover, despite ongoing research, it remains to be established if medical therapy is effective in preventing complications for patients with bicuspid aortopathy. Although supportive clinical evidence is still missing, beta-blockers and angiotensin receptor blocking agents are frequently prescribed to protect the BAV-aorta within this patient population (Danyi et al., 2011; Chun et al., 2013; Ziganshin et al., 2015). On the other hand, several groups have studied the risk of developing aneurysmal dilation of the ascending aorta over time in patients with BAV (to a size of 4.0–4.5 cm). It was shown that 20–30% of patients with BAV develop aneurysmal enlargement during 9–25 years of follow up (Michelena et al., 2008, 2014; Tzemos et al., 2008). In fact, in a recent review paper, based on eight independent studies, it was suggested that up to 84% of patients with BAV ultimately develop an aneurysm, and the risk of the aneurysm development was 80-fold higher when compared to the general population (Michelena et al., 2014; Wasfy et al., 2015).
Of clinical significance, dilatation of any or all segments of the aorta is seen in approximately 50% of patients with BAV (Fedak et al., 2005), and ascending aortic aneurysms occur in 1% of BAV patients per year. Although bicuspid aortopathy can manifest in all segments of the aorta, it is more often isolated to the aortic root, ascending aorta, or proximal aortic arch. Most patients will present with maximal dilatation of the tubular mid-ascending aorta, specifically at the greater curvature, with the aortic root and proximal arch being affected to varying degrees. Thus, resection strategies can vary greatly (Fedak et al., 2005; Della Corte et al., 2014a; Fedak and Verma, 2014; Adamo and Braverman, 2015; Moon, 2015; Sundt, 2015). In addition to deciding when to intervene in replacing the aorta in bicuspid aortopathy, assessing what to resect also poses a clinical dilemma (Fedak and Verma, 2014; Sundt, 2015). Bicuspid aortopathy is progressive, increasing the risk of aortic dissection and rupture. To date, these complications have been challenging to predict. Therefore, frequent monitoring and personalized interventions for both timing of surgery and the extent of resection are of paramount importance in preventing these clinical catastrophes and delivering optimal care (Itagaki et al., 2016).
Although different international societies and expert groups have provided unified guidelines regarding optimal management of patients with BAV disease, most surgical recommendations have primarily been based on maximal aortic diameter and growth rate (Nishimura et al., 2014). According to these guidelines, prophylactic replacement of the ascending aorta is performed in roughly 25% of BAV patients within 25 years from the time of diagnosis (Michelena et al., 2011). This has significant implications, as the burden of surgery for BAV patients in the United States exceeds 1 billion dollars per year, and surgical intervention has doubled over the past decade (Opotowsky et al., 2013). It is noted that surgical planning and decision-making for BAV patients is affected by physician bias and historical local practice within institutions, which aren't always consistent and in line with guidelines (Verma et al., 2013; Della Corte et al., 2014b; Girdauskas and Borger, 2014; Michelena et al., 2014; Verma and Siu, 2014; Sundt, 2015; Wasfy et al., 2015). In a recent survey of 100 cardiac surgeons, it was postulated that attitudes on the etiology, inherited aortopathy vs. acquired from hemodynamic stress, rather than proven clinical evidence dictated surgical treatment of BAV aortopathy (Verma et al., 2013). Undeniably, this has complicated widely accepted and universally utilized guidelines, emphasizing the need for more translational and clinical research solely dedicated to BAV patient populations.
Fortunately, over the past 3 years, a concerted effort has been made in understanding the individual variability inherent to BAV disease and the role hemodynamic factors play in its manifestation and progression (Della Corte et al., 2014a; Fedak and Verma, 2014; Girdauskas and Borger, 2014; Martin et al., 2014; Uretsky and Gillam, 2014; Verma and Siu, 2014; Michelena, 2015; Spinale and Bolger, 2015; Itagaki et al., 2016; Sievers et al., 2016). There is a general consensus among experts regarding a critical need in developing personalized risk assessments beyond conventional aortic size and growth criteria, in delivering optimal care to BAV patients. The challenge clinicians face today is a paucity of prognostic models to inform the timing and extent of surgical intervention. To address some of these issues, our translational research group and others have focused on validating use of novel imaging techniques to establish non-invasive hemodynamic biomarkers for risk-stratifying BAV patients. In this review article, we will consider recent efforts, successes, and ongoing challenges in the development of more precise and individualized surgical approaches for patients with BAVs and associated aortic disease.
Pathophysiology and Wavering Guidelines
To develop patient-specific parameters in BAV populations, researchers have considered the pathophysiology of BAV aortopathy. Like other vessels, a normal aortic wall is divided into three layers: intima, media, and adventitia. Elastin fibers, vascular smooth muscles cells, and structural extracellular matrix (ECM) comprise the medial layer. The aortic media regulates tissue biology and biomechanics (Figure 1). Different studies have demonstrated that BAV occurs in conjunction with degeneration of this layer. Bicuspid aortopathy involves medial ECM abnormalities which include (Della Corte et al., 2014b; Itagaki et al., 2016)
1. ECM dysregulation: altered matrix metalloproteinase expression and activity (Thompson and Cockerill, 2006; Ikonomidis et al., 2007, 2012; Wilton et al., 2008; Fedak et al., 2013; Rabkin, 2014)
2. Altered medial ECM architecture: elastin fiber degeneration (de Sa et al., 1999; Bauer et al., 2002; Cotrufo et al., 2003, 2005; Chung et al., 2007; Phillippi et al., 2014)
3. Tissue dysfunction: altered stiffness and biomechanics (Nistri et al., 2002, 2008; Schaefer et al., 2007; Pees and Michel-Behnke, 2012; Oulego-Erroz et al., 2013; Warner et al., 2013; Forsell et al., 2014; Moaref et al., 2014; Petrini et al., 2014)
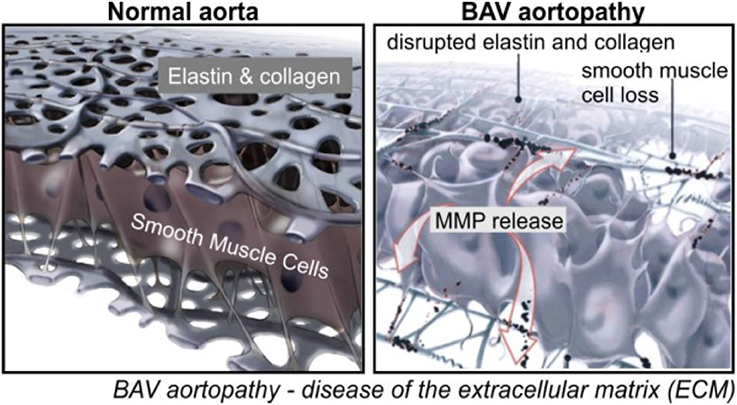
Figure 1. Aortic wall degeneration in BAV (Fedak et al., 2002).
As expected, the presence, severity, and location of these pathologies differs among patients. This poses a significant question: in addition to a possible genetic predisposition to dilatation, do hemodynamic conditions in the aorta contribute to its remodeling in BAV patients?
Unfortunately, the mechanisms which contribute to aortopathy in BAV patients have not been clearly elucidated (Davies et al., 2007; Tadros et al., 2009; Girdauskas et al., 2011; Michelena et al., 2011; Sievers and Sievers, 2011), and it is not known whether genetics leads to aortopathy or if the altered BAV morphology results in isolated diseased areas within the aortic wall secondary to abnormal blood flow from the valve (Figure 2). It is possible that it is a combination of both factors, but a unilateral focus on the genetic component has supported aggressive surgical intervention with respect to the timing and extent of aortic resection. Although previous guidelines and size thresholds for surgical resection were based on algorithms similar to those for patients diagnosed with genetic aortopathies, such as Marfan's syndrome (Bonow et al., 2008), recent clinical data strongly suggest that bicuspid aortopathy is distinct from that of Marfan's (Itagaki et al., 2015). Not surprisingly, clinical approaches in managing BAV aortopathies are highly influenced by different opinions on the varied impact of genetics and hemodynamics on disease progression (Hardikar and Marwick, 2015). Initially, a conservative cut off of 5.5 cm was used in 1998 (Tricoci et al., 2009). In 2010, surgeons were more aggressive, intervening when the aortic diameter was 4.0–4.5 cm (Warnes et al., 2008), but reverted to a more conservative approach of 5.5 cm cut off in 2014 (Svensson et al., 2013; Erbel et al., 2014; Michelena et al., 2015). Throughout this time no clinically and scientifically proven study was reported to support either a conservative or aggressive approach in surgical resection in bicuspid aortopathy (Hardikar and Marwick, 2015). To offer scientifically proven guidelines, which would consistently be used by clinicians, it is of paramount importance to continue the work on the discovery and implementation of novel aortic risk markers (Michelena et al., 2015; Spinale and Bolger, 2015).
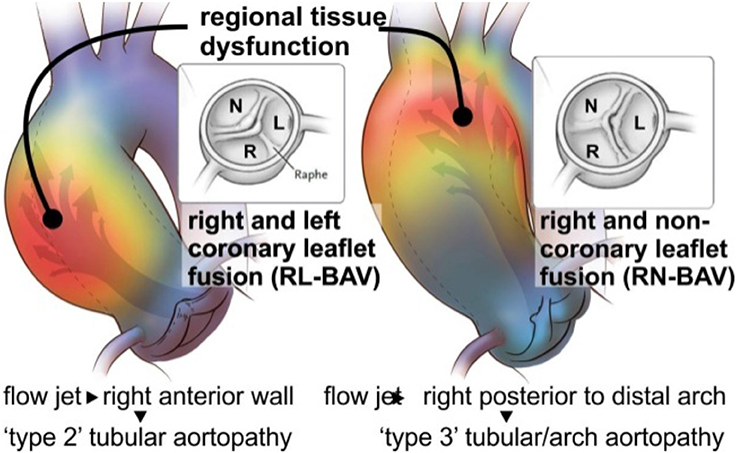
Figure 2. Valve mediated hemodynamics. BAV fusion patterns lay the foundation for changes in aortic outflow and wall shear stress (WSS). Eccentric blood flow from the RL-BAV impinges on regions of dilatation at the tubular ascending aorta wall. Flow from the RN-BAV reflects off the proximal posterior wall and impinges on regions of aortic dilatation within the proximal arch. Adapted with permission (Itagaki et al., 2016).
New Evidence on the Pathophysiology of BAV Aortopathy
Previous work studying the hemodynamic component to BAV-related aortopathy focused on the severity of aortic valve stenosis (AS) or insufficiency (AI) (Tzemos et al., 2008; Girdauskas et al., 2011; Michelena, 2015). It is now believed that these conventional hemodynamic factors alone do not reflect the impact on the aortic wall due to a malformed valve (Girdauskas et al., 2011; Sievers and Sievers, 2011; Atkins and Sucosky, 2014; Della Corte et al., 2014b; Adamo and Braverman, 2015; Michelena et al., 2015; Moon, 2015). These findings imply that the development of bicuspid aortopathy is not primarily driven by a genetic predisposition. Further supporting these results are recent studies by our group and others which have shown that altered aortic flow and valve morphology in BAV patients are related to the expression of the aortopathy phenotype (Kang et al., 2013; Mahadevia et al., 2014; Prakash et al., 2015). As depicted in Figure 2, 4-D flow MRI studies provide strong evidence that valve-mediated local flow dynamics (Barker et al., 2012) and regional differences in wall shear stress (WSS) (Mahadevia et al., 2014) are associated with changes in regional aortic wall histology and proteolytic events (Guzzardi et al., 2015), contributing to unfavorable aortic remodeling. More significantly, these preliminary data can be landmark findings in better understanding valve-mediated hemodynamics' impact on the progression of bicuspid aortopathy. They can also be a platform for clinically-proven justification in utilizing MRI-based biomarkers in risk stratification.
The dearth of prognostic models to assist in the surgical management of BAV patients is the biggest challenge clinicians face today, particularly with regards to the timing and extent of surgical repair. As mentioned, this patient population is often offered aortic resection primarily based on maximal aortic size dimension and the rate at which the aorta expands. It is, however, now understood that measures of aortic size alone are insufficient to dictate treatment algorithms (Della Corte et al., 2012a; Verma et al., 2013; Della Corte, 2014; Michelena et al., 2014; Prakash et al., 2015; Sundt, 2015; Wasfy et al., 2015). Therefore, substantial efforts are currently being made to improve risk prediction for aortic catastrophes in BAV patients (Della Corte et al., 2007, 2012b,c, 2013; Ikonomidis et al., 2013).
Recent work has yielded strong evidence that including measures of downstream valve-mediated hemodynamics into the work-up of BAV patients has a high likelihood to circumvent current prognostic challenges (Davies et al., 2007; Rabkin, 2014; Adamo and Braverman, 2015; Song, 2015). However, conventional diagnostic modalities, such as Doppler echocardiography, 2D phase contrast [PC]-MRI, and CT scan, do not offer the means for a thorough in-vivo assessment of three dimensional blood flow through the aorta, which is necessary to study the role of transvalvular hemodynamics on the downstream forces experienced at the aortic wall and their impact on the progression of aortic dilatation.
Innovative Technology—4D Flow MRI
Recent advances in magnetic resonance imaging have allowed for uncompromised in-vivo assessment of time-resolved 3D blood velocity, using a volumetric technique, referred to as 4D flow MRI (Figure 3). This modality provides an opportunity to quantify complex three dimensional blood flow patterns in-vivo, and has facilitated new insights into sophisticated cardiovascular hemodynamics (Harloff et al., 2009, 2010b; Markl et al., 2010, 2011a; Barker et al., 2012; Mahadevia et al., 2014). In particular, multidimensional 4D flow MRI data, which infers three spatial dimensions describing 3D velocity over time, permits visualization of aortic blood flow, quantification of regional velocity and flow (Markl et al., 2007, 2011b; Frydrychowicz et al., 2008a; Bock et al., 2010), and WSS (Stalder et al., 2008; Barker et al., 2010; Harloff et al., 2010a; Bock et al., 2011; Garcia et al., 2014; van Ooij et al., 2015a).
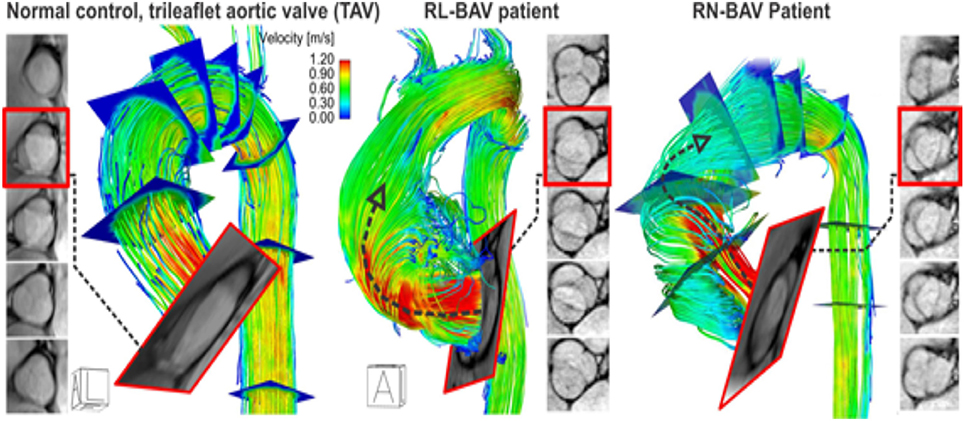
Figure 3. 4D flow MRI in a control and BAV patient with a right-left (RL) fusion pattern and right-non-coronary (RN) fusion pattern. Note that the RL-BAV exerted significant eccentric aortic outflow jet (but not higher velocity, arrow) compared to TAV. The BAV phenotype (RL vs. RN) greatly influences aortic outflow, which in turn, affects aortic segments exposed to elevated WSS (Figure 4). Adapted with permission (Barker et al., 2012; Guzzardi et al., 2015).
Multiple institutions have now shown that 4D flow MRI can be utilized to accurately identify altered flow patterns secondary to BAV, even if aortic stenosis is present (Figures 3, 4). Among the observed hemodynamic changes are eccentric flow patterns, which result in a change of the drag forces at the vessel wall (Figure 4). Despite what is believed to be a multifactorial disease, recent studies have shown WSS to play a major role in bicuspid aortopathy. We now believe that WSW may change local matrix homeostasis, and consequently affect the structure of the ascending aorta (Stalder et al., 2008; Markl et al., 2009; Harloff et al., 2010a; Markl et al., 2010; Bock et al., 2011; Garcia et al., 2014; van Ooij et al., 2015a). In fact, research has shown WSS to affect cell function, implicating its role in the development of aortopathy (den Reijer et al., 2010; Hope et al., 2010a, 2011).
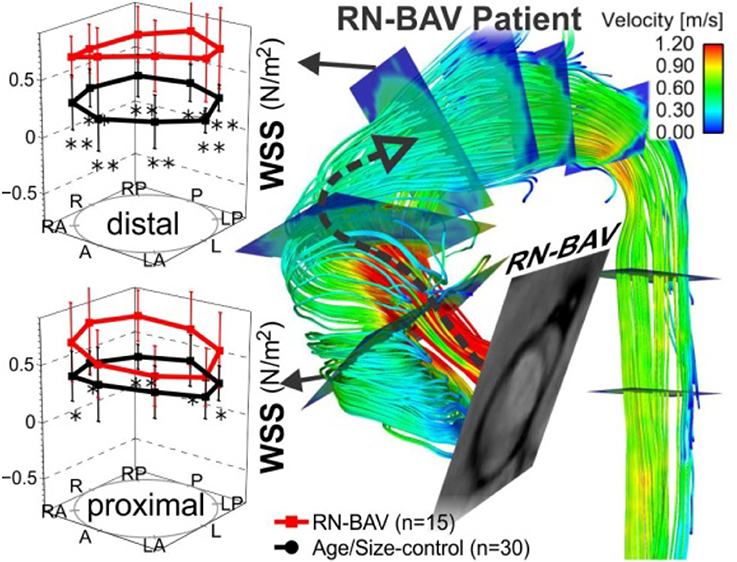
Figure 4. 4D flow & WSS quantification in 15 RN-BAV patients found that 4D flow can detect significant differences (*p < 0.05, **p < 0.001) in aortic WSS distribution compared to 30 TAV controls. (A) Control populations and (B) individual BAV patient. Note the different aortic outflow pattern compared to the RL-BAV patient in Figure 3 (Barker et al., 2012).
Our group has used non-invasive MRI techniques (2D phase contrast MRI) to show BAV-mediated alterations in flow and WSS (Barker et al., 2010). Building on the successes of our initial work, we then employed 4D flow MRI to definitively demonstrate aortic WSS was increased in BAV subjects independent of the degree of stenosis when compared to age and aortic size-matched controls (P < 0.05, Figure 4; Mahadevia et al., 2014). Also of clinical significance, we have shown that regional variation of WSS with the aorta is dependent on aortic valve fusion phenotype (Barker et al., 2012; Mahadevia et al., 2014), and is associated with the diameter of the aorta (Bissell et al., 2013). In one of our recent studies, we considered 30 BAV patients and 30 age-appropriate trileaflet aortic valve (TAV) controls, and showed that altered aortic hemodynamics may be a mechanism by which right and left coronary leaflet (RL-BAV) or right and non-coronary leaflet valve (RN-BAV) fusion influences the expression of aortopathy (Mahadevia et al., 2014).
A significant finding has been the fact that hemodynamic alterations are related to medial wall degeneration (Guzzardi et al., 2015). A pilot study was recently completed, which included both in-vivo 4D flow MRI and aortic tissue resection in 20 BAV patients. The study successfully demonstrated the ability to correlate in-vivo 4D flow derived hemodynamic biomarkers with tissue metrics of bicuspid aortopathy. In this work, 20 BAV patients undergoing aortic resection underwent pre-operative 4D flow MRI to regionally map WSS and had histologic examination of their resected tissue samples. Samples obtained from regions of both elevated and normal WSS within the same patient were paired, and compared for medial elastin degeneration by histology and ECM dysregulation by protein expression. As depicted in Figures 6, 7, regions of increased WSS showed greater medial elastin degradation compared to adjacent segments with normal WSS. Moreover, multiplex protein analyses of ECM regulatory molecules revealed an increase in TGFβ-1, MMP-1, MMP-2, MMP-3, and TIMP-1 in increased WSS areas, suggesting ECM dysregulation in regions of elevated WSS. In a much larger prospective cohort, the aim will be to more comprehensively characterize aortic tissues resected from segments of abnormal WSS, further clarifying the impact of altered blood flow.
The Promise of Personalized Medicine for BAV Patients
The pilot study demonstrated the potential utility of 4D flow MRI to identify areas with more advanced aortopathy in patients. Future work will focus on these significant findings, with the objectives being the discovery and validation of key hemodynamic imaging biomarkers. This should include refining aortic MRI protocols, creating “maps” or “atlases” of normal age and gender-matched imaging biomarkers of aortic hemodynamics in both BAV and TAV patients, and pushing current boundaries by carrying out a clinico-pathologic correlation study in BAV and TAV patients with aortopathy to establish imaging biomarkers predictive of aortic tissue pathology and dysfunction.
A limitation of 4D flow MRI is the acquisition time needed and its low blood-tissue contrast, which has proven a challenge for its translation to routine clinical use. Ongoing efforts to decrease exam time with the use of accelerated imaging strategies such as radial undersampling, k-t approaches, or compressed sensing are rapidly becoming available for the clinic (Baltes et al., 2005; Lustig et al., 2007; Moftakhar et al., 2007). This should hopefully result in reduced acquisition times for the assessment of valve dynamics and time-resolved aortic 3D geometry. Moreover, these advances should improve 3D segmentation of various parts of the aorta for precise assessment of hemodynamic imaging biomarkers, such as WSS and flow displacement, and amalgamate the analysis of aortic valve morphology (RL-, RN-BAV, etc.), geometry (orifice area), and dynamics (opening angle).
Additionally, the pilot study considered whether aortic WSS, ECM architecture and protein expression, and non-traditional hemodynamic parameters can affect regional aortic tissue function. However, it is still not known which factors associated with valve-mediated hemodynamics are most sensitive in predicting the development and progression of aortopathy. For instance, WSS gradient (WSSG) and oscillatory shear index (OSI) are also known to promote remodeling (Hope et al., 2010a; Bissell et al., 2013). Moreover, previous work studying WSS was based on two-dimensional imaging planes manually placed in the thoracic aorta (Frydrychowicz et al., 2008b, 2009; Stalder et al., 2008; Markl et al., 2010, 2011c, 2013; Harloff et al., 2010a; Hope et al., 2010b; Barker et al., 2012; Burk et al., 2012; Potters et al., 2014), and was hence, limited in calculating imaging biomarkers along the entire length of the aorta. A future goal would be to develop a comprehensive data analysis protocol.
In a recent study, an algorithm was developed to compute volumetric 3D WSS along the entire surface of the aorta (Figure 5; Potters et al., 2014; van Ooij et al., 2015a). Test–retest experiments for systolic WSS demonstrated excellent accuracy, with a 9% coefficient of variance, and a 6% inter-observer error (van Ooij et al., 2015b). We also now know that elevated WSS could be seen on the outer curvature of the ascending aorta in 13 BAV patients, with a significant correlation to peak systolic velocity (Cibis et al., 2015). Future work will focus on extending the methodology to incorporate additional imaging biomarkers implicated in vessel wall remodeling, such as WSSG and OSI.
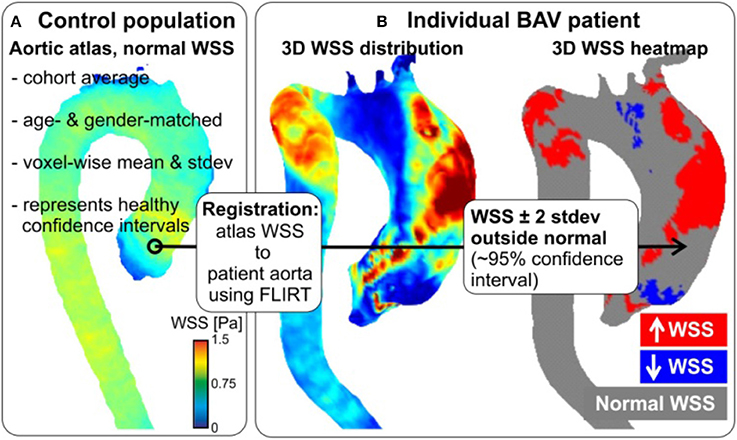
Figure 5. Patient-specific WSS heat maps. Healthy atlases are registered to BAV data with tools developed for brain mapping. Maps can be reliably generated, even in patients with complex aortic geometries (as here).
Prior research on creating “heat maps” or “atlases” utilized a 3D WSS mapping technique which allowed for compact visualization of hemodynamic parameters studies across multiple subjects. This mapping, however, is limited in that it does not detect where “abnormal” values exist. Therefore, a database of healthy volunteer 4D flow MRI exams was created to produce an aortic “atlas” which established regional confidence intervals for normal physiologic WSS throughout the aorta (Figure 5; van Ooij et al., 2015a). Linear intra- and inter-modal brain image registration (FLIRT, Linear Image Registration Tool, FMRIB, Oxford; Jenkinson and Smith, 2001) were utilized to co-register aortic 3D WSS of 10 TAV patients with no aortic stenosis, but present aortic dilatation, and TAV patients with aortic stenosis, but no aortic dilatation, with the atlas. The dilatation cohort had significantly lower WSS on 7% of the ascending aorta, whereas the stenosis cohort showed significantly higher WSS on 34% of the ascending aorta surface (van Ooij et al., 2015a,c). A future research goal is to build on these efforts to construct age and gender matched atlases of the imaging biomarkers in a large population.
In summary, patient-specific WSS can be reliably computed and co-registered to a healthy control atlas, representing the normal ranges of physiologic WSS. These normal WSS atlases can subsequently be employed to create “heat maps” which represent regions of abnormally high or low WSS in a patient in question (Figures 5B, 6; van Ooij et al., 2015c). This heat map provides a foundation for thorough, yet succinct, assessment to detect regions, and segments of the aortic wall which are exposed to abnormal hemodynamics on an individual, patient-specific, basis.
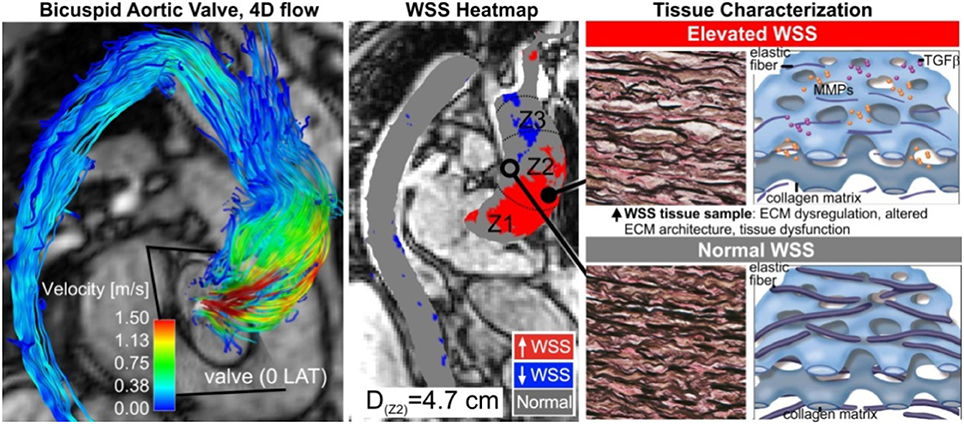
Figure 6. Tissue histopathology. Aortic wall regions are exposed to elevated WSS (middle, red region), due to eccentric transvalvular BAV flow (left). This manifests in the expression of abnormal tissue metrics of aortopathy (right). Adapted with permission (Guzzardi et al., 2015).
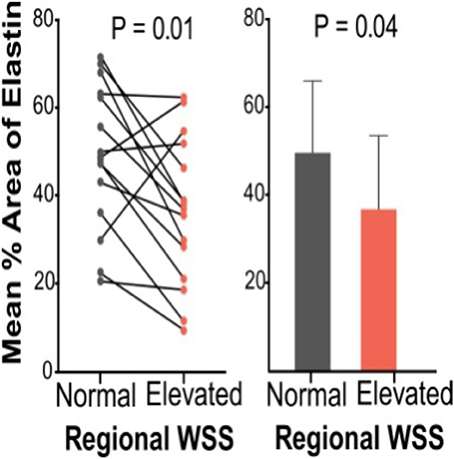
Figure 7. Elastin content from Verhoeff–Van Gieson elastin staining for patient pairs and cumulative group means for aortic wall subjected to normal and elevated WSS (Guzzardi et al., 2015).
Imaging Hemodynamic Biomarkers and Aortic Wall Pathology
A major component of personalized medicine is the potential utility of non-invasive biomarkers in diagnosing and prognosticating clinical risk and outcomes. The same holds true for BAV and bicuspid aortopathy; much current investigation is aimed at identifying such markers to improve the management of this patient population. For instance, Della Corte's group recently published a study which shows that the ratio of circulating Transforming Growth Factor Beta-1 to soluble endoglin is an early biomarker for bicuspid aortopathy. Although systemic biomarkers remain to be more fully validated, such investigations provide a foundation for future work in this area (Forte et al., 2017).
The focus of this review, however, is the role of novel non-invasive imaging hemodynamic biomarkers. As mentioned above, to date, most research on the hemodynamic hypothesis for bicuspid aortopathy has focused on aortic valve function, namely the severity of aortic stenosis or insufficiency (Tzemos et al., 2008; Girdauskas et al., 2012; Michelena, 2015). These factors alone, however, do not fully reflect the hemodynamic burden exacted on the aortic wall, secondary to the malformed aortic valve. Moreover, despite not being completely well-understood, there are inherent differences in the development of BAV vs. TAV aortopathy. For instance, the associated aortopathy in most patients with TAV is thought to be a result of aortic valve stenosis and altered post-valve hemodynamics. On the other hand, a strong genetic predisposition has been suggested to contribute to aortopathy in BAV patients. Interestingly, a recent study showed that TAV aortopathy was associated with more severe histologic abnormalities compared to BAV aortopathy, especially when stratified by diameter (Heng et al., 2015). Properly assessing the diagnostic value of novel imaging biomarkers requires further work in this area to gain a better appreciation of the differences in the development of BAV vs. TAV aortopathy.
As alluded to above, recently, a correlation between WSS and regional aortic tissue remodeling in BAV patients was established (Guzzardi et al., 2015). It was concluded that elastin content and structure was significantly disrupted in areas of high WSS with a change in the expression of specific MMPs and TGF-beta. There was also an observed trend toward differences in the elastic modulus and tissue stiffness using biaxial testing. Although further clarification is required, these data promise an opportunity for utilizing valve-mediated hemodynamics as non-invasive biomarkers of aortopathy susceptibility and progression.
Despite making great strides in advancing our understanding of BAV aortopathy and its clinical implications, work has so far considered only one hemodynamic biomarker, systolic WSS, within a small sample size. Future research will need to focus on identifying the hemodynamic metric most predictive of disease severity, as well as elucidating the role other clinical factors play, in a larger population sample size. To determine the differential impact and magnitude of valve-mediated hemodynamics as compared to genetic predisposition and other non-hemodynamic factors in BAV patients, future work should compare our results to a purely hemodynamic-mediated aortopathy reference group. Finally, the pilot study was not powered to assess difference in MMP-2 expression, thus ongoing and future studies can consider MMP-2 activity, histopathology, and biomechanics in larger samples sizes.
Conclusion
Bicuspid aortic valve is the most common congenital cardiac defect. Multiple studies provide strong evidence for the clinical significance of this disease, especially as how it relates to pathologic abnormalities of the aorta. Many studies have also shown the devastating sequelae of aortic complications in patients with BAVs. All of these impose a highly unfavorable health and economic burden on patients and society-at-large. To better understand the etiology and pathophysiology of bicuspid aortopathy, numerous investigators have undertaken studying different aspects of the disease. These efforts have successfully elucidated critical mechanisms and factors influencing disease development and progression in bicuspid aortopathy. To an extent, the motivation for this work has been the objective of defining a uniform, safe, and evidence-based set of guidelines for the medical and surgical management of these patients. Despite key advances, more research is needed. To this end, our group and others have focused on discovering and identifying novel, non-invasive histopathologic, and hemodynamic biomarkers which could potentially play a key role in further improving the care of patients with bicuspid aortopathy. By leveraging basic and translational research techniques, novel imaging modalities, and perhaps systemic biomarkers, improved risk prediction may result in more individualized treatment options and optimal management strategies.
Author Contributions
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
Funding
Research reported in this publication was supported by the National Heart, Lung, and Blood Institute of the National Institutes of Health under Award Number R01HL133504. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
Adamo, L., and Braverman, A. C. (2015). Surgical threshold for bicuspid aortic valve aneurysm: a case for individual decision-makin. Heart 101, 1361–1367. doi: 10.1136/heartjnl-2014-306601
Atkins, S. K., and Sucosky, P. (2014). Etiology of bicuspid aortic valve disease: focus on hemodynamics. World J. Cardiol. 6, 1227–1233. doi: 10.4330/wjc.v6.i12.1227
Baltes, C., Kozerke, S., Hansen, M. S., Pruessmann, K. P., Tsao, J., and Boesiger, P. (2005). Accelerating cine phase-contrast flow measurements using k-t BLAST and k-t SENSE. Magn. Reson. Med. 54, 1430–1438. doi: 10.1002/mrm.20730
Barker, A. J., Lanning, C., and Shandas, R. (2010). Quantification of hemodynamic wall shear stress in patients with bicuspid aortic valve using phase-contrast MRI. Ann. Biomed. Eng. 38, 788–800. doi: 10.1007/s10439-009-9854-3
Barker, A. J., Markl, M., Burk, J., Lorenz, R., Bock, J., Bauer, S., et al. (2012). Bicuspid aortic valve is associated with altered wall shear stress in the ascending aorta. Circ. Cardiovasc. Imaging 5, 457–466. doi: 10.1161/CIRCIMAGING.112.973370
Bauer, M., Pasic, M., Meyer, R., Goetze, N., Bauer, U., Siniawski, H., et al. (2002). Morphometric analysis of aortic media in patients with bicuspid and tricuspid aortic valve. Ann. Thorac. Surg. 74, 58–62. doi: 10.1016/S0003-4975(02)03650-0
Bissell, M. M., Hess, A. T., Biasiolli, L., Glaze, S. J., Loudon, M., Pitcher, A., et al. (2013). Aortic dilation in bicuspid aortic valve disease: flow pattern is a major contributor and differs with valve fusion type. Circ. Cardiovasc. Imaging. 6, 499–507. doi: 10.1161/CIRCIMAGING.113.000528
Bock, J., Frydrychowicz, A., Lorenz, R., Hirtler, D., Barker, A. J., Johnson, K. M., et al. (2011). In vivo noninvasive 4D pressure difference mapping in the human aorta: phantom comparison and application in healthy volunteers and patients. Magn. Reson. Med. 66, 1079–1088. doi: 10.1002/mrm.22907
Bock, J., Frydrychowicz, A., Stalder, A. F., Bley, T. A., Burkhardt, H., Hennig, J., et al. (2010). 4D phase contrast MRI at 3 T: effect of standard and blood-pool contrast agents on SNR, PC-MRA, and blood flow visualization. Magn. Reson. Med. 63, 330–338. doi: 10.1002/mrm.22199
Bonow, R. O., Carabello, B. A., Chatterjee, K., de Leon, A. C., Faxon, D. P., Freed, M. D., et al. (2008). 2008 Focused update incorporated into the ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1998 Guidelines for the Management of Patients With Valvular Heart Disease): endorsed by the Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Circulation 118, e523–e661. doi: 10.1016/j.jacc.2008.05.007
Burk, J., Blanke, P., Stankovic, Z., Barker, A., Russe, M., Geiger, J., et al. (2012). Evaluation of 3D blood flow patterns and wall shear stress in the normal and dilated thoracic aorta using flow-sensitive 4D CMR. J. Cardiovasc. Magn. Reson. 14:84. doi: 10.1186/1532-429X-14-84
Chun, A. S., Elefteriades, J. A., and Mukherjee, S. K. (2013). Medical treatment for thoracic aortic aneurysm - much more work to be done. Prog. Cardiovasc. Dis. 56, 103–108. doi: 10.1016/j.pcad.2013.05.008
Chung, A. W., Au Yeung, K., Sandor, G. G., Judge, D. P., Dietz, H. C., and van Breemen, C. (2007). Loss of elastic fiber integrity and reduction of vascular smooth muscle contraction resulting from the upregulated activities of matrix metalloproteinase-2 and -9 in the thoracic aortic aneurysm in Marfan syndrome. Circ. Res. 101, 512–522. doi: 10.1161/CIRCRESAHA.107.157776
Cibis, M., Jarvis, K., Markl, M., Rose, M., Rigsby, C., Barker, A. J., et al. (2015). The effect of resolution on viscous dissipation measured with 4D flow MRI in patients with Fontan circulation: evaluation using computational fluid dynamics. J. Biomech. 48, 2984–2989. doi: 10.1016/j.jbiomech.2015.07.039
Cotrufo, M., Della Corte, A., De Santo, L. S., De Feo, M., Covino, F. E., and Dialetto, G. (2003). Asymmetric medial degeneration of the ascending aorta in aortic valve disease: a pilot study of surgical management. J. Heart Valve Dis. 12, 127–133; discussion 134–125.
Cotrufo, M., Della Corte, A., De Santo, L. S., Quarto, C., De Feo, M., Romano, G., et al. (2005). Different patterns of extracellular matrix protein expression in the convexity and the concavity of the dilated aorta with bicuspid aortic valve: preliminary results. J. Thorac. Cardiovasc. Surg. 130, 504–511. doi: 10.1016/j.jtcvs.2005.01.016
Danyi, P., Elefteriades, J. A., and Jovin, I. S. (2011). Medical therapy of thoracic aortic aneurysms: are we there yet? Circulation 124, 1469–1476. doi: 10.1161/CIRCULATIONAHA.110.006486
Davies, R. R., Kaple, R. K., Mandapati, D., Gallo, A., Botta, D. M., Elefteriades, J. A., et al. (2007). Natural history of ascending aortic aneurysms in the setting of an unreplaced bicuspid aortic valve. Ann. Thorac. Surg. 83, 1338–1344. doi: 10.1016/j.athoracsur.2006.10.074
Della Corte, A. (2014). The conundrum of aortic dissection in patients with bicuspid aortic valve: the tissue, the mechanics and the mathematics. Eur. J. Cardiothorac. Surg. 48, 150–151. doi: 10.1093/ejcts/ezu418
Della Corte, A., Bancone, C., Buonocore, M., Dialetto, G., Covino, F. E., Manduca, S., et al. (2013). Pattern of ascending aortic dimensions predicts the growth rate of the aorta in patients with bicuspid aortic valve. JACC Cardiovasc. Imaging 6, 1301–1310. doi: 10.1016/j.jcmg.2013.07.009
Della Corte, A., Bancone, C., Conti, C. A., Votta, E., Redaelli, A., Del Viscovo, L., et al. (2012b). Restricted cusp motion in right-left type of bicuspid aortic valves: a new risk marker for aortopathy. J. Thorac. Cardiovasc Surg. 144, 360–369, 369.e1. doi: 10.1016/j.jtcvs.2011.10.014
Della Corte, A., Bancone, C., Dialetto, G., Covino, F. E., Manduca, S., D'Oria, V., et al. (2014a). Towards an individualized approach to bicuspid aortopathy: different valve types have unique determinants of aortic dilatation. Eur. J. Cardiothorac. Surg. 45, e118–e124; discussion e124. doi: 10.1093/ejcts/ezt601
Della Corte, A., Bancone, C., Quarto, C., Dialetto, G., Covino, F. E., Scardone, M., et al. (2007). Predictors of ascending aortic dilatation with bicuspid aortic valve: a wide spectrum of disease expression. Eur. J. Cardiothorac. Surg. 31, 397–404; discussion 404–405. doi: 10.1016/j.ejcts.2006.12.006
Della Corte, A., Body, S. C., Booher, A. M., Schaefers, H. J., Milewski, R. K., Michelena, H. I., et al. (2014b). Surgical treatment of bicuspid aortic valve disease: knowledge gaps and research perspectives. J. Thoracic Cardiovascular. Surg. 147, 1749–1757. doi: 10.1016/j.jtcvs.2014.01.021
Della Corte, A., Buonocore, M., and Del Viscovo, L. (2012c). Rationale and methods for quantifying ascending aortic flow eccentricity: back to the underlying mechanism? J. Magn. Reson. Imaging 36, 505–506; author reply 507. doi: 10.1002/jmri.23656
Della Corte, A., De Santo, L. S., and Forte, A. (2012a). Missing link between aortic wall pathology and aortic diameter: methodological bias or worrisome finding? Eur. J. Cardiothorac. Surg. 42, 195–196; author reply 196. doi: 10.1093/ejcts/ezs006
den Reijer, P. M., Sallee, D., van der Velden, P., Zaaijer, E. R., Parks, W. J., Ramamurthy, S., et al. (2010). Hemodynamic predictors of aortic dilatation in bicuspid aortic valve by velocity-encoded cardiovascular magnetic resonance. J. Cardiovasc. Magn. Reson. 12:4. doi: 10.1186/1532-429X-12-4
de Sa, M., Moshkovitz, Y., Butany, J., and David, T. E. (1999). Histologic abnormalities of the ascending aorta and pulmonary trunk in patients with bicuspid aortic valve disease: clinical relevance to the ross procedure. J. Thorac. Cardiovasc. Surg. 118, 588–594.
Ellison, J. W., Yagubyan, M., Majumdar, R., Sarkar, G., Bolander, M. E., Atkinson, E. J., et al. (2007). Evidence of genetic locus heterogeneity for familial bicuspid aortic valve. J. Surg. Res. 142, 28–31. doi: 10.1016/j.jss.2006.04.040
Erbel, R., Aboyans, V., Boileau, C., Bossone, E., Bartolomeo, R. D., Eggebrecht, H., et al. (2014). 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the ESC. Eur. Heart J. 35, 2873–2926. doi: 10.1093/eurheartj/ehu281
Fazel, S. S., Mallidi, H. R., Lee, R. S., Sheehan, M. P., Liang, D., Fleischman, D., et al. (2008). The aortopathy of bicuspid aortic valve disease has distinctive patterns and usually involves the transverse aortic arch. J. Thorac. Cardiovasc. Surg. 135, 901–907, 907.e1–2. doi: 10.1016/j.jtcvs.2008.01.022
Fedak, P. W., David, T. E., Borger, M., Verma, S., Butany, J., and Weisel, R. D. (2005). Bicuspid aortic valve disease: recent insights in pathophysiology and treatment. Expert Rev. Cardiovasc. Ther. 3, 295–308. doi: 10.1586/14779072.3.2.295
Fedak, P. W., de Sa, M. P., Verma, S., Nili, N., Kazemian, P., Butany, J., et al. (2013). Vascular matrix remodeling in patients with bicuspid aortic valve malformations: implications for aortic dilatation. J. Thorac. Cardiovasc. Surg. 126, 797–806. doi: 10.1016/S0022-5223(03)00398-2
Fedak, P. W., and Verma, S. (2014). Bicuspid aortopathy and the development of individualized resection strategies. J. Thorac. Cardiovasc. Surg. 148, 2080–2081. doi: 10.1016/j.jtcvs.2014.09.059
Fedak, P. W., Verma, S., David, T. E., Leask, R. L., Weisel, R. D., and Butany, J. (2002). Clinical and pathophysiological implications of a bicuspid aortic valve. Circulation 106, 900–904. doi: 10.1161/01.CIR.0000027905.26586.E8
Forsell, C., Bjorck, H. M., Eriksson, P., Franco-Cereceda, A., and Gasser, T. C. (2014). Biomechanical properties of the thoracic aneurysmal wall: differences between bicuspid aortic valve and tricuspid aortic valve patients. Ann. Thorac. Surg. 98, 65–71. doi: 10.1016/j.athoracsur.2014.04.042
Forte, A., Bancone, C., Cobellis, G., Buonocore, M., Santarpino, G., Fischlein, T. J. M., et al. (2017). A possible early biomarker for bicuspid aortopathy: circulating transforming growth factor beta-1 to soluble endoglin ratio. Circ. Res. 120, 1800–1811. doi: 10.1161/CIRCRESAHA.117.310833
Frydrychowicz, A., Arnold, R., Harloff, A., Schlensak, C., Hennig, J., Langer, M., et al. (2008a). In vivo 3-dimensional flow connectivity mapping after extracardiac total cavopulmonary connection. Circulation 118, E16–E17. doi: 10.1161/CIRCULATIONAHA.107.761304
Frydrychowicz, A., Berger, A., Russe, M. F., Stalder, A. F., Harloff, A., Dittrich, S., et al. (2008b). Time-resolved magnetic resonance angiography and flow-sensitive 4-dimensional magnetic resonance imaging at 3 Tesla for blood flow and wall shear stress analysis. J. Thorac. Cardiovasc. Surg. 136, 400–407. doi: 10.1016/j.jtcvs.2008.02.062
Frydrychowicz, A., Stalder, A. F., Russe, M. F., Bock, J., Bauer, S., Harloff, A., et al. (2009). Three-dimensional analysis of segmental wall shear stress in the aorta by flow-sensitive four-dimensional-MRI. J. Magn. Reson. Imaging 30, 77–84. doi: 10.1002/jmri.21790
Garcia, J., Markl, M., Schnell, S., Allen, B., Entezari, P., Mahadevia, R., et al. (2014). Evaluation of aortic stenosis severity using 4D flow jet shear layer detection for the measurement of valve effective orifice area. Magn Reson Imaging 32, 891–898. doi: 10.1016/j.mri.2014.04.017
Garg, V., Muth, A. N., Ransom, J. F., Schluterman, M. K., Barnes, R., King, I. N., et al. (2005). Mutations in NOTCH1 cause aortic valve disease. Nature 437, 270–274. doi: 10.1038/nature03940
Girdauskas, E., and Borger, M. A. (2014). Surgical threshold for bicuspid aortic valveassociated aortopathy: does the phenotype matter? JACC Cardiovasc Imaging 7:318. doi: 10.1016/j.jcmg.2013.12.012
Girdauskas, E., Borger, M. A., Secknus, M. A., Girdauskas, G., and Kuntze, T. (2011). Is aortopathy in bicuspid aortic valve disease a congenital defect or a result of abnormal hemodyanimcs? A critical reappraisal of a one-sided argument. Eur. J. Cardiothorac. Surg. 39, 809–814. doi: 10.1016/j.ejcts.2011.01.001
Girdauskas, E., Disha, K., Raisin, H. H., Secknus, M. A., Borger, M. A., and Kuntze, T. (2012). Risk of late aortic events after an isolated aortic valve replacement for bicuspid aortic valve stenosis with concomitant ascending aortic dilation. Eur. J. Cardiothorac. Surg. 42, 832–837; discussion 837–838. doi: 10.1093/ejcts/ezs137
Guzzardi, D. G., Barker, A. J., van Ooij, P., Malaisrie, S. C., Puthumana, J. J., Belke, D. D., et al. (2015). Valve-related hemodynamics mediate human bicuspid aortopathy: insights from wall shear stress mapping. J. Am. Coll. Cardiol. 66, 892–900. doi: 10.1016/j.jacc.2015.06.1310
Hardikar, A. A., and Marwick, T. H. (2015). The natural history of guidelines: the case of aortopathy related to bicuspid aortic valves. Int. J. Cardiol. 199, 150–153. doi: 10.1016/j.ijcard.2015.06.059
Harloff, A., Nussbaumer, A., Bauer, S., Stalder, A. F., Frydrychowicz, A., Weiller, C., et al. (2010a). In vivo assessment of wall shear stress in the atherosclerotic aorta using flow-sensitive 4D MRI. Magn. Reson. Med. 63, 1529–1536. doi: 10.1002/mrm.22383
Harloff, A., Simon, J., Brendecke, S., Assefa, D., Helbing, T., Frydrychowicz, A., et al. (2010b). Complex plaques in the proximal descending aorta: an underestimated embolic source of stroke. Stroke 41, 1145–1150. doi: 10.1161/STROKEAHA.109.577775
Harloff, A., Strecker, C., Dudler, P., Nussbaumer, A., Frydrychowicz, A., Olschewski, M., et al. (2009). Retrograde embolism from the descending aorta: visualization by multidirectional 3D velocity mapping in cryptogenic stroke. Stroke 40, 1505–1508. doi: 10.1161/STROKEAHA.108.530030
Heng, E., Stone, J. R., Kim, J. B., Lee, H., MacGillivray, T. E., and Sundt, T. M. (2015). Comparative histology of aortic dilatation associated with bileaflet versus trileaflet aortic valves. Ann. Thorac. Surg. 100, 2095–2101; discussion 2101. doi: 10.1016/j.athoracsur.2015.05.105
Hiratzka, L. F., Bakris, G. L., Beckman, J. A., Bersin, R. M., Carr, V. F., Casey, D. E., et al. (2010). ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM guidelines for the diagnosis and management of patients with Thoracic Aortic Disease. Circulation 121, e266–e369. doi: 10.1161/CIR.0b013e3181d4739e
Hiratzka, L. F., Creager, M. A., Isselbacher, E. M., Svensson, L. G., Nishimura, R. A., Bonow, R. O., et al. (2016). Surgery for aortic dilatation in patients with bicuspid aortic valves: a statement of clarification from the american college of cardiology/american heart association task force on clinical practice guidelines. J. Am. Coll. Cardiol. 67, 724–731. doi: 10.1016/j.jacc.2015.11.006
Hoffman, J., and Kaplan, S. (2002). The incidence of congenital heart disease. J. Am. Coll. Cardiol. 39, 1890–1900. doi: 10.1016/S0735-1097(02)01886-7
Hope, M. D., Hope, T. A., Crook, S. E., Ordovas, K. G., Urbania, T. H., Alley, M. T., et al. (2011). 4D flow CMR in assessment of valve-related ascending aortic disease. JACC. Cardiovasc. Imaging 4, 781–787. doi: 10.1016/j.jcmg.2011.05.004
Hope, M. D., Hope, T. A., Meadows, A. K., Ordovas, K. G., Urbania, T. H., Alley, M. T., et al. (2010a). Bicuspid aortic valve: four-dimensional MR evaluation of ascending aortic systolic flow patterns. Radiology 255, 53–61. doi: 10.1148/radiol.09091437
Hope, M. D., Hope, T. A., Urbania, T. H., and Higgins, C. B. (2010b). Four-dimensional flow magnetic resonance imaging with wall shear stress analysis before and after repair of aortopulmonary fistula. Circ. Cardiovasc. Imaging 3, 766–768. doi: 10.1161/CIRCIMAGING.110.957712
Ikonomidis, J. S., Ivey, C. R., Wheeler, J. B., Akerman, A. W., Rice, A., Patel, R. K., et al. (2013). Plasma biomarkers for distinguishing etiologic subtypes of thoracic aortic aneurysm disease. J. Thorac. Cardiovasc. Surg. 145, 1326–1333. doi: 10.1016/j.jtcvs.2012.12.027
Ikonomidis, J. S., Jones, J. A., Barbour, J. R., Stroud, R. E., Clark, L. L., Kaplan, B. S., et al. (2007). Expression of matrix metalloproteinases and endogenous inhibitors within ascending aortic aneurysms of patients with bicuspid or tricuspid aortic valves. J. Thorac. Cardiovasc. Surg. 133, 1028–1036. doi: 10.1016/j.jtcvs.2006.10.083
Ikonomidis, J. S., Ruddy, J. M., Benton, S. M., Arroyo, J., Brinsa, T. A., Stroud, R. E., et al. (2012). Aortic dilatation with bicuspid aortic valves: cusp fusion correlates to matrix metalloproteinases and inhibitors. Ann. Thorac. Surg. 93, 457–463. doi: 10.1016/j.athoracsur.2011.09.057
Itagaki, S., Chiang, Y., and Tang, G. H. (2016). Why does the bicuspid aortic valve keep eluding us? Cardiol. Rev. 24, 119–130. doi: 10.1097/CRD.0000000000000053
Itagaki, S., Chikwe, J. P., Chiang, Y. P., Egorova, N. N., and Adams, D. H. (2015). Long term risk for aortic complications after aortic valve replacement in patients with bicuspid aortic valve versus Marfan syndorme. J. Am. Coll. Cardiol. 65, 2363–2369. doi: 10.1016/j.jacc.2015.03.575
Januzzi, J. L., Isselbacher, E. M., Fattori, R., Cooper, J. V., Smith, D. E., Fang, J., et al. (2004). Characterizing the young patient with aortic dissection: results from the International Registry of Aortic Dissection (IRAD). J. Am. Coll. Cardiol. 43, 665–669. doi: 10.1016/j.jacc.2003.08.054
Jenkinson, M., and Smith, S. (2001). A global optimisation method for robust affine registration of brain images. Med. Image Anal. 5, 143–156. doi: 10.1016/S1361-8415(01)00036-6
Kang, J. W., Song, H. G., Yang, D. H., Baek, S., Kim, D. H., Song, J. M., et al. (2013). Association between bicuspid aortic valve phenotype and patterns of valvular dysfunction and bicuspid aortopathy: comprehensive evaluation using MDCT and echocardiography. JACC. Cardiovasc. Imaging 6, 150–161. doi: 10.1016/j.jcmg.2012.11.007
Lustig, M., Donoho, D., and Pauly, J. M. (2007). Sparse MRI: the application of compressed sensing for rapid MR imaging. Magn. Reson. Med. 58, 1182–1195. doi: 10.1002/mrm.21391
Mahadevia, R., Barker, A. J., Schnell, S., Entezari, P., Kansal, P., Fedak, P. W., et al. (2014). Bicuspid aortic cusp fusion morphology alters aortic three-dimensional outflow patterns, wall shear stress, and expression of aortopathy. Circulation 129, 673–682. doi: 10.1161/CIRCULATIONAHA.113.003026
Markl, M., Bauer, S., Bock, J., Stalder, A., Frydrychowicz, A., and Harloff, A. (2009). “Wall shear stress in normal & atherosclerotic carotid arteries,” in Proceedings: 21st Annual International Conference on MR Angiography (Lansing, MI), 68.
Markl, M., Brendecke, S. M., Simon, J., Barker, A. J., Weiller, C., and Harloff, A. (2013). Co-registration of the distribution of wall shear stress and 140 complex plaques of the aorta. Magn. Reson. Imaging 31, 1156–1162. doi: 10.1016/j.mri.2013.05.001
Markl, M., Geiger, J., Arnold, R., Stroh, A., Damjanovic, D., Foll, D., et al. (2011a). Comprehensive 4-dimensional magnetic resonance flow analysis after successful heart transplantation resolvescontroversial intraoperative findings and reveals complex hemodynamic alterations. Circulation 123, e381–e383. doi: 10.1161/CIRCULATIONAHA.110.979971
Markl, M., Geiger, J., Kilner, P. J., Foll, D., Stiller, B., Beyersdorf, F., et al. (2011b). Time-resolved three-dimensional magnetic resonance velocity mapping of cardiovascular flow paths in volunteers and patients with Fontan circulation. Eur. J. Cardiothorac. Surg. 39, 206–212. doi: 10.1016/j.ejcts.2010.05.026
Markl, M., Harloff, A., Bley, T. A., Zaitsev, M., Jung, B., Weigang, E., et al. (2007). Time-resolved 3D MR velocity mapping at 3T: improved navigator-gated assessment of vascular anatomy and blood flow. J. Magn. Reson. Imaging 25, 824–831. doi: 10.1002/jmri.20871
Markl, M., Wallis, W., and Harloff, A. (2011c). Reproducibility of flow and wall shear stress analysis using flow-sensitive four-dimensional MRI. J. Magn. Reson. Imaging 33, 988–994. doi: 10.1002/jmri.22519
Markl, M., Wegent, F., Zech, T., Bauer, S., Strecker, C., Schumacher, M., et al. (2010). In vivo wall shear stress distribution in the carotid artery: effect of bifurcation geometry, internal carotid artery stenosis, and recanalization therapy. Circ. Cardiovasc. Imaging 3, 647–655. doi: 10.1161/CIRCIMAGING.110.958504
Martin, M., Alonso-Montes, C., Florez, J. P., Pichel, I. A., Rozado, J., Andía, J. B., et al. (2014). Bicuspid aortic valve syndrome: a heterogeneous and still unknown condition. Int. J. Cardiol. 177:1105. doi: 10.1016/j.ijcard.2014.09.132
Masri, A., Kalahasti, V., Alkharabsheh, S., Svensson, L. G., Sabik, J. F., Roselli, E. E., et al. (2016). Characteristics and long-term outcomes of contemporary patients with bicuspid aortic valves. J. Thorac. Cardiovasc. Surg. 151, 1650–1659. doi: 10.1016/j.jtcvs.2015.12.019
Michelena, H. I., Desjardins, V. A., Avierinos, J. F., Russo, A., Nkomo, V. T., Sundt, T. M., et al. (2008). Natural history of asymptomatic patients with normally functioning or minimally dysfunctional bicuspid aortic valve in the community. Circulation 117, 2776–2784. doi: 10.1161/CIRCULATIONAHA.107.740878
Michelena, H. I., Prakash, S. K., Della Corte, A., Bissell, M. M., Anavekar, N., Mathieu, P., et al. (2014). Bicuspid aortic valve: identifying knowledge gaps and rising to the challenge from the International Bicuspid Aortic Valve Consortium (BAVCon). Circulation 129, 2691–2704. doi: 10.1161/CIRCULATIONAHA.113.007851
Michelena, H. I. (2015). The bicuspid aortic valve aortopathy mystery continues: are we that mediocre? Trends Cardiovasc. Med. 25, 452–455. doi: 10.1016/j.tcm.2014.12.016
Michelena, H. I., Della Corte, A., Prakash, S. K., Milewicz, D. M., Evangelista, A., and Enriquez-Sarano, M. (2015). Bicuspid aortic valve aortopathy in adults: incidence, etiology, and clinical significance. Int. J. Cardiol. 201, 400–407. doi: 10.1016/j.ijcard.2015.08.106
Michelena, H. I., Khanna, A. D., Mahoney, D., Margaryan, E., Topilsky, Y., Suri, R. M., et al. (2011). Incidence of aortic complications in patients with bicuspid aortic valves. JAMA 306, 1104–1112. doi: 10.1001/jama.2011.1286
Moaref, A., Khavanin, M., and Shekarforoush, S. (2014). Aortic distensibility in bicuspid aortic valve patients with normal aortic diameter. Ther. Adv. Cardiovasc. Dis. 8, 128–132. doi: 10.1177/1753944714531062
Moftakhar, R., Aagaard-Kienitz, B., Johnson, K., Turski, P. A., Turk, A. S., Niemann, D. B., et al. (2007). Noninvasive measurement of intra-aneurysmal pressure and flow pattern using phase contrast with vastly undersampled isotropic projection imaging. AJNR Am. J. Neuroradiol. 28, 1710–1714. doi: 10.3174/ajnr.A0648
Moon, M. R. (2015). Extent of distal resection for bicuspid aortopathy: Is surgical experience a factor? J. Thorac. Cardiovasc. Surg. 150, 125–126. doi: 10.1016/j.jtcvs.2015.04.004
Nishimura, R. A., Otto, C. M., Bonow, R. O., Carabello, B. A., Erwin, J. P. III, Guyton, R. A., et al. (2014). 2014 AHA/ACC Guideline for the Management of Patients with Valvular Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 129, 2440–2492. doi: 10.1161/CIR.0000000000000029
Nistri, S., Grande-Allen, J., Noale, M., Basso, C., Siviero, P., Maggi, S., et al. (2008). Aortic elasticity and size in bicuspid aortic valve syndrome. Eur. Heart J. 29, 472–479. doi: 10.1093/eurheartj/ehm528
Nistri, S., Sorbo, M. D., Basso, C., and Thiene, G. (2002). Bicuspid aortic valve: abnormal aortic elastic properties. J. Heart Valve Dis. 11, 369–373; discussion 373–364.
Opotowsky, A. R., Perlstein, T., Landzberg, M. J., Colan, S. D., O'Gara, P. T., Body, S. C., et al. (2013). A shifting approach to management of the thoracic aorta in bicuspid aortic valve. J. Thorac. Cardiovasc. Surg. 146, 339–346. doi: 10.1016/j.jtcvs.2012.10.028
Oulego-Erroz, I., Alonso-Quintela, P., Mora-Matilla, M., Gautreaux Minaya, S., and Lapena-Lopez de Armentia, S. (2013). Ascending aorta elasticity in children with isolated bicuspid aortic valve. Int. J. Cardiol. 168, 1143–1146. doi: 10.1016/j.ijcard.2012.11.080
Pees, C., and Michel-Behnke, I. (2012). Morphology of the bicuspid aortic valve and elasticity of the adjacent aorta in children. Am. J. Cardiol. 110, 1354–1360. doi: 10.1016/j.amjcard.2012.06.043
Petrini, J., Jenner, J., Rickenlund, A., Eriksson, P., Franco-Cereceda, A., Caidahl, K., et al. (2014). Elastic properties of the descending aorta in patients with a bicuspid or tricuspid aortic valve and aortic valvular disease. J. Am. Soc. Echocardiogr. 27, 393–404. doi: 10.1016/j.echo.2013.12.013
Phillippi, J. A., Green, B. R., Eskay, M. A., Kotlarczyk, M. P., Hill, M. R., Robertson, A. M., et al. (2014). Mechanism of aortic medial matrix remodeling is distinct in patients with bicuspid aortic valve. J. Thorac. Cardiovasc. Surg. 147, 1056–1064. doi: 10.1016/j.jtcvs.2013.04.028
Potters, W. V., van Ooij, P., Marquering, H., Vanbavel, E., and Nederveen, A. J. (2014). Volumetric arterial wall shear stress calculation based on cine phase contrast MRI. J. Magn. Reson. Imaging. 41, 505–516. doi: 10.1002/jmri.24560
Prakash, A., Adlakha, H., Rabideau, N., Hass, C. J., Morris, S. A., Geva, T., et al. (2015). Segmental aortic stiffness in children and young adults with connective tissue disorders: relationships with age, aortic size, rate of dilation, and surgical root replacement. Circulation 132, 595–602. doi: 10.1161/CIRCULATIONAHA.114.014934
Prakash, S. K., Bossé, Y., Muehlschlegel, J. D., Michelena, H. I., Limongelli, G., Della Corte, A., et al. (2014). A roadmap to investigate the genetic basis of bicuspid aortic valve and its complications insights from the International BAVCon (Bicuspid Aortic Valve Consortium). J. Am. Coll. Cardiol. 64, 832–839. doi: 10.1016/j.jacc.2014.04.073
Rabkin, S. W. (2014). Differential expression of MMP-2, MMP-9 and TIMP proteins in thoracic aortic aneurysm - comparison with and without bicuspid aortic valve: a meta-analysis. Vasa 43, 433–442. doi: 10.1024/0301-1526/a000390
Schaefer, B. M., Lewin, M. B., Stout, K. K., Byers, P. H., and Otto, C. M. (2007). Usefulness of bicuspid aortic valve phenotype to predict elastic properties of the ascending aorta. Am. J. Cardiol. 99, 686–690. doi: 10.1016/j.amjcard.2006.09.118
Sherrah, A. G., Andvik, S., van der Linde, D., Davies, L., Bannon, P. G., Padang, R., et al. (2016). Nonsyndromic thoracic aortic aneurysm and dissection: outcomes with Marfan syndrome versus bicuspid aortic valve aneurysm. J. Am. Coll. Cardiol. 67, 618–626. doi: 10.1016/j.jacc.2015.11.039
Sievers, H. H., and Sievers, H. L. (2011). Aortopathy in bicuspid aortic valve disease - genes or hemodynamics? or Scylla and Charybdis? Eur. J. Cardiothorac. Surg. 39, 803–804. doi: 10.1016/j.ejcts.2011.02.007
Sievers, H. H., Stierle, U., Hachmann, R. M., and Charitos, E. I. (2016). New insights in the association between bicuspid aortic valve phenotype, aortic configuration and valve haemodynamics. Eur. J. Cardiothorac. Surg. 49, 439–446. doi: 10.1093/ejcts/ezv087
Song, J. K. (2015). Bicuspid aortic valve: unresolved issues and role of imaging specialists. J. Cardiovasc. Ultrasound. 23, 1–7. doi: 10.4250/jcu.2015.23.1.1
Spinale, F. G., and Bolger, A. F. (2015). Fate versus flow: wall shear stress in the aortopathy associated with bicuspid aortic valves. J. Am. Coll. Cardiol. 66, 901–904. doi: 10.1016/j.jacc.2015.07.002
Stalder, A. F., Russe, M. F., Frydrychowicz, A., Bock, J., Hennig, J., and Markl, M. (2008). Quantitative 2D and 3D phase contrast MRI: optimized analysis of blood flow and vessel wall parameters. Magn. Reson. Med. 60, 1218–1231. doi: 10.1002/mrm.21778
Sundt, T. M. (2015). Aortic replacement in the setting of bicuspid aortic valve: how big? How much? J. Thorac. Cardiovasc. Surg. 149, S2–S9. doi: 10.1016/j.jtcvs.2014.07.069
Svensson, L. G., Adams, D. H., Bonow, R. O., Kouchoukos, N. T., Miller, D. C., O'Gara, P. T., et al. (2013). Aortic valve and ascending aorta guidelines for management and quality measures. Ann. Thorac. Surg. 95, S1–S66. doi: 10.1016/j.athoracsur.2012.12.027
Tadros, T. M., Klein, M. D., and Shapira, O. M. (2009). Ascending aortic dilatation associated with bicuspid aortic valve: pathophysiology, molecular biology, and clinical implications. Circulation 119, 880–890. doi: 10.1161/CIRCULATIONAHA.108.795401
Thompson, M., and Cockerill, G. (2006). Matrix metalloproteinase-2: the forgotten enzyme in aneurysm pathogenesis. Ann. N. Y. Acad. Sci. 1085, 170–174. doi: 10.1196/annals.1383.034
Tricoci, P., Allen, J. M., Kramer, J. M., Califf, R. M., and Smith, S. C. Jr. (2009). Scientific evidence underlying the ACC/AHA clinical practice guidelines. JAMA 301, 831–841. doi: 10.1001/jama.2009.205
Tzemos, N., Therrien, J., Yip, J., Thanassoulis, G., Tremblay, S., Jamorski, M. T., et al. (2008). Outcomes in adults with bicuspid aortic valves. JAMA 300, 1317–1325. doi: 10.1001/jama.300.11.1317
Uretsky, S., and Gillam, L. D. (2014). Nature versus nurture in bicuspid aortic valve aortopathy: more evidence that altered hemodynamics may play a role. Circulation 129, 622-624. doi: 10.1161/CIRCULATIONAHA.113.007282
Vahanian, A., Alfieri, O., Andreotti, F., Antunes, M. J., Barón-Esquivias, G., Baumgartner, H., et al. (2012). Guidelines on the management of valvular heart disease (version 2012): the Joint Task Force on the Management of Valvular Heart Disease of the European Society of Cardiology and the European Association for Cardiothoracic Surgery. Eur. J. Cardiothoracic Surg. 42, S1–S44. doi: 10.1093/eurheartj/ehs109
van Ooij, P., Potters, W. V., Collins, J., Carr, M., Carr, J., Malaisrie, S. C., et al. (2015c). Characterization of abnormal wall shear stress using 4D flow MRI in human bicuspid aortopathy. Ann. Biomed. Eng. 43, 1385–1397. doi: 10.1007/s10439-014-1092-7
van Ooij, P., Potters, W. V., Nederveen, A. J., Allen, B. D., Collins, J., Carr, J., et al. (2015a). A methodology to detect abnormal relative wall shear stress on the full surface of the thoracic aorta using four-dimensional flow MRI. Magn. Reson. Med. 73, 1216–1227. doi: 10.1002/mrm.25224
van Ooij, P., Powell, A. L., Potters, W. V., Carr, J. C., Markl, M., and Barker, A. J. (2015b). Reproducibility and interobserver variability of systolic blood flow velocity and 3D wall shear stress derived from 4D flow MRI in the healthy aorta. J. Magn. Reson. Imaging 43, 236–248. doi: 10.1002/jmri.24959
Verma, S., and Siu, S. C. (2014). Aortic dilatation in patients with bicuspid aortic valve. N. Engl. J. Med. 370, 1920–1929. doi: 10.1056/NEJMra1207059
Verma, S., Yanagawa, B., Kalra, S., Ruel, M., Peterson, M. D., Yamashita, M. H., et al. (2013). Knowledge, attitudes, and practice patterns in surgical management of bicuspid aortopathy: a survey of 100 cardiac surgeons. J. Thorac. Cardiovasc. Surg. 146, 1033–1040. doi: 10.1016/j.jtcvs.2013.06.037
Warner, P. J., Al-Quthami, A., Brooks, E. L., Kelley-Hedgepeth, A., Patvardhan, E., Kuvin, J. T., et al. (2013). Augmentation index and aortic stiffness in bicuspid aortic valve patients with non-dilated proximal aortas. BMC Cardiovasc. Disord. 13:19. doi: 10.1186/1471-2261-13-19
Warnes, C. A., Williams, R. G., Bashore, T. M., Child, J. S., Connolly, H. M., Dearani, J. A., et al. (2008). ACC/AHA 2008 Guidelines for the Management of Adults with Congenital Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (writing committee to develop guidelines on the management of adults with congenital heart disease). Circulation 118, e714–e833. doi: 10.1161/CIRCULATIONAHA.108.190811
Wasfy, J. H., Armstrong, K., Milford, C. E., and Sundt, T. M. (2015). Bicuspid aortic disease and decision making under uncertainty: the limitations of clinical guidelines. Int. J. Cardiol. 181, 169–171. doi: 10.1016/j.ijcard.2014.12.020
Wilton, E., Bland, M., Thompson, M., and Jahangiri, M. (2008). Matrix metalloproteinase expression in the ascending aorta and aortic valve. Interact. Cardiovasc. Thorac. Surg. 7, 37–40. doi: 10.1510/icvts.2007.163311
Keywords: bicuspid aortic valve, aortopathy, MRI, biomarkers discovery, precision medicine
Citation: Fatehi Hassanabad A, Barker AJ, Guzzardi D, Markl M, Malaisrie C, McCarthy PM and Fedak PWM (2017) Evolution of Precision Medicine and Surgical Strategies for Bicuspid Aortic Valve-Associated Aortopathy. Front. Physiol. 8:475. doi: 10.3389/fphys.2017.00475
Received: 31 March 2017; Accepted: 21 June 2017;
Published: 10 July 2017.
Edited by:
Alessandro Della Corte, Department of Cardiothoracic Sciences - Second University of Naples, Monaldi Hospital, ItalyReviewed by:
Amy Banes-Berceli, Oakland University, United StatesDan Predescu, Rush University, United States
Copyright © 2017 Fatehi Hassanabad, Barker, Guzzardi, Markl, Malaisrie, McCarthy and Fedak. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Paul W. M. Fedak, cGF1bC5mZWRha0BnbWFpbC5jb20=
 Ali Fatehi Hassanabad
Ali Fatehi Hassanabad Alex J. Barker2
Alex J. Barker2 Michael Markl
Michael Markl Paul W. M. Fedak
Paul W. M. Fedak