- 1Department of Orthopaedic Surgery, Pittsburgh Orthopaedic Spine Research Group (POSR), Pittsburgh, PA, United States
- 2Department of Orthopaedic Surgery, The Bethel Family Musculoskeletal Research Center (BMRC), Pittsburgh, PA, United States
- 3Department of Orthopaedic Surgery, University of Pittsburgh, Pittsburgh Medical Center, PA, United States
- 4Dr. Kiran C. Patel College of Osteopathic Medicine, Nova Southeastern University, Ft. Lauderdale, FL, United States
Introduction: Kyphoplasty is a minimally invasive treatment for osteoporotic vertebral compression fractures (VCFs) aimed at restoring vertebral height and alleviating pain. However, despite its widespread use, kyphoplasty outcomes are typically analyzed without distinguishing between thoracic and lumbar regions. Given the anatomical and biomechanical differences between these regions, it is crucial to compare the efficacy and complication rates of kyphoplasty based on spinal location. This review seeks to evaluate these distinctions and explore their potential clinical implications to inform patient expectations and procedural understanding.
Methods: A comprehensive search strategy was developed using concepts of kyphoplasty, compression fractures, and spinal regions. Resulting literature underwent title, abstract, and full-text screening using the inclusion criteria: (1) Patients underwent kyphoplasty for osteoporotic VCFs of the thoracic and lumbar spine, (2) Clinical, perioperative, or postoperative outcome measures were available, and (3) Outcome measures were subcategorized into thoracic or lumbar spinal regions. Meta-analysis was performed using a random effects model. The ROBINS-I tool was used to evaluate bias.
Results: Twenty-five manuscripts were included in final analysis. Meta-analysis revealed operative time and injected cement volume were 43.07 minutes [95% CI (35.34, 50.79)] and 4.61 ml [95% CI (3.79, 5.43)] in the lumbar spine and 56.42 min [95% CI (41.03, 71.81)] and 4.25 ml [95% CI (3.56, 4.93)] in the thoracic spine, respectively. Likewise, the incidence of cement leakage and subsequent adjacent level fractures were 13% [95% CI (7%, 22%)] and 5% [95% CI (2%, 12%)] in the lumbar spine and 19% [95% CI (10%, 32%)] and 10% [95% CI (2%, 33%)] in the thoracic spine, respectively. Lastly, the post-operative decrease in visual analog scale pain was 5.59 [95% CI (5.23, 5.94)] in the lumbar spine and 5.49 [95% CI (4.79, 6.19)] in the thoracic spine. Across studies, the primary risk of bias was due to confounding variables, resulting in a serious risk of bias in 12 studies (48.0%).
Discussion: Kyphoplasty provides comparable outcomes in thoracic and lumbar VCFs, with no clinically significant differences in pain relief or procedural effectiveness. Despite anatomical differences, the procedure remains safe and effective in both regions, warranting further research on spinal region-specific outcomes.
Introduction
Vertebral fractures affect approximately 1.5 million Americans each year and can result from a variety of causes, including trauma, malignancy, infection, and most commonly, osteoporosis (1, 2). Osteoporotic spinal fractures are particularly prevalent among the elderly population who suffer from diminished bone density, which can be associated with a number of risk factors including female gender, prolonged corticosteroid use, low estrogen levels, tobacco and alcohol abuse, and kidney disease, among others (3–5). With the increasing age of the general population, the incidence of osteoporotic spinal fractures is expected to grow (6, 7). Similarly, the identification of red flag signs such as advanced age, trauma, and corticosteroid use, among others, has enhanced the screening and diagnosis of osteoporotic vertebral compression fractures (OVCFs) within the elderly population (8, 9). These fractures can give rise to a range of symptoms including back pain, kyphotic deformities, and neurological deficits that can result in pervasive loss of mobility and function, social isolation, and increased rate of mortality (7, 10–12). In numerous instances, these fractures have the potential to resolve spontaneously through conservative management or non-invasive therapeutic interventions (13, 14). However, surgical intervention is indicated in a subset of patients contingent on the severity of their symptoms, the extent of spinal deformity, and the presence of neurological impairment (7, 15–19). A variety of clinical and radiological outcomes are used to assess the management of OVCFs, including pain levels, vertebral body height restoration, spinal alignment, incidence of adjacent-level fractures, and surgical complications (18, 20–22). While specific clinical recommendations (i.e., the osteoporotic fracture classification score) have emerged to offer surgeons greater guidance in treatment decision-making (13, 23), the choice still remains heavily reliant on a surgeon's individual judgment, taking into account the unique characteristics of their patient such as age, comorbidities, and frailty, among others.
One minimally invasive surgical technique that has emerged as an effective vertebral augmentation treatment for OVCFs is kyphoplasty (24, 25). Kyphoplasty is a minimally invasive procedure that involves inserting a balloon into the fractured vertebra to restore its height, followed by injection of bone cement to stabilize the fracture and alleviate pain (26). In the evaluation of kyphoplasty for OVCFs, thoracic and lumbar fractures are frequently amalgamated into a single category with their outcomes collectively assessed (27–29). However, key anatomical and biomechanical differences exist between the thoracic and lumbar spine such as facet joint orientation, sagittal alignment, and spinal stability (30–32). These differences may significantly influence the manifestation of symptoms and subsequently affect treatment outcomes. While prior systematic reviews delineate variations in prevalence, biomechanical properties, bone density, blood flow, and subsequent fracture rate between osteoporotic fractures of the lumbar and thoracic regions, they do not provide a comprehensive characterization of the differences in clinical outcomes following kyphoplasty for differing regions of the spine (30). Insights into these differences could establish a more outcomes-based framework to guide surgical decision-making and management in older adults with OVCFs depending on whether the fracture is within the thoracic or lumbar spine. This systematic review and meta-analysis aims to provide an updated evaluation of perioperative, postoperative, and patient-reported outcomes of kyphoplasty for OVCFs, with a focus on differentiating outcomes between thoracic and lumbar regions. This review provides a comprehensive comparison of kyphoplasty outcomes between the thoracic and lumbar spine, assessing whether anatomical and biomechanical differences influence surgical outcomes and highlighting the need for further research.
Methods
Search strategy and data extraction
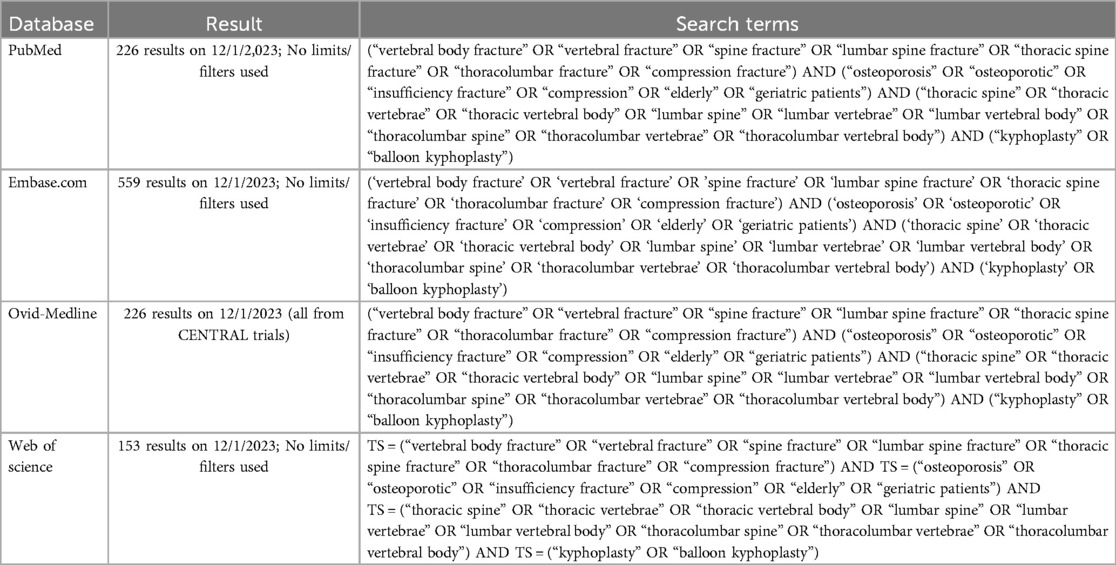
Published literature was searched using key phrases including: “kyphoplasty”, “osteoporotic compression fractures”, and “lumbar and thoracic spine”, with related synonyms. Literature searches were executed in PubMed, Embase.com, Ovid-Medline, and Web of Science from database inception (Table 1). All database searches were completed on November 17, 2023. A total of 1,164 results were retrieved from the database literature search and imported to the Pico Portal systematic review platform (33). Duplicate citations (432) were identified and excluded. Four independent reviewers (M.R.K., C.C., S.L., T.Z.) performed title and abstract screening on the remaining 732 studies, with each manuscript title and abstract screened twice. If two reviewers disagreed on whether a manuscript should advance to full-text screening, a third reviewer (M.R.K.) provided the final decision. Full-text screening was conducted based on the following inclusion criteria: (1) patients undergoing kyphoplasty for osteoporotic compression fractures of the thoracic or lumbar spine, (2) reporting of clinical, patient-reported, perioperative, or postoperative outcome measures, and (3) differentiation of outcomes by spinal region (thoracic vs. lumbar). Any reviews, case reports, conference abstracts, non-English manuscripts, animal studies, expert opinion, editorials, or non-original research were excluded. Data extraction included information regarding study aims, methods, patient population, spinal level at which the compression fracture occurred, surgical and clinical outcomes, and a description of main findings. The variability in reported data across studies posed challenges but did not preclude the feasibility of conducting a meta-analysis. The systematic review was conducted in accordance with PRISMA reporting guidelines.
Risk of bias assessment
Risk of bias was evaluated using the Risk Of Bias In Non-randomized Studies of Interventions (ROBINS-I) tool (34). Two reviewers (T.Z., S.L.) assessed the risk of bias as definitely low, moderate, serious, or critical using a series of signaling questions across seven domains including bias due to confounding, bias in selection of participants into the study, bias in classification of interventions, bias due to deviation from intended interventions, bias due to missing data, bias in measurement of outcomes, and bias in selection of reported result. A third reviewer (M.R.K.) facilitated consensus in cases where the two primary reviewers disagreed on the assessment of bias within a domain.
Statistical analysis
A meta-analysis was conducted for continuous outcome measures with data across individual studies converted to means and standard deviations using approximation techniques. If a study did not explicitly report the change between preoperative and postoperative values for a variable of interest, it was calculated using the provided mean and standard deviation distributions before and after surgery. If only the mean and range were provided for a specific outcome measure, the standard deviation was approximated by dividing the range by four. Likewise, if only the median and range were reported, the mean and standard deviation were approximated using the method described by Hozo et al. (35). Lastly, if only the mean was reported, the standard deviation was conservatively estimated as half of the mean. A meta-analysis of proportions was performed to calculate pooled rates for cement leakage and subsequent adjacent fracture with a forest plot created to visualize the results. For all meta-analyses, an effect size was computed for each study and a pooled effect size with 95% confidence intervals was calculated to summarize across studies. Heterogeneity among studies was evaluated using the Higgins I² statistic, with values exceeding 50% indicating substantial heterogeneity. A random effects model was employed across all meta-analyses as a more conservative approach, accounting for both intra-study and inter-study variability (36, 37). Similarly, a meta-analysis was conducted only for outcomes examined in at least five studies, as smaller sample sizes may produce unreliable estimates (36, 37). All statistical analyses were conducted using R and RStudio (Posit Software, PBC, Boston, MA) with the meta and metafor packages.
Results
Overview of included studies
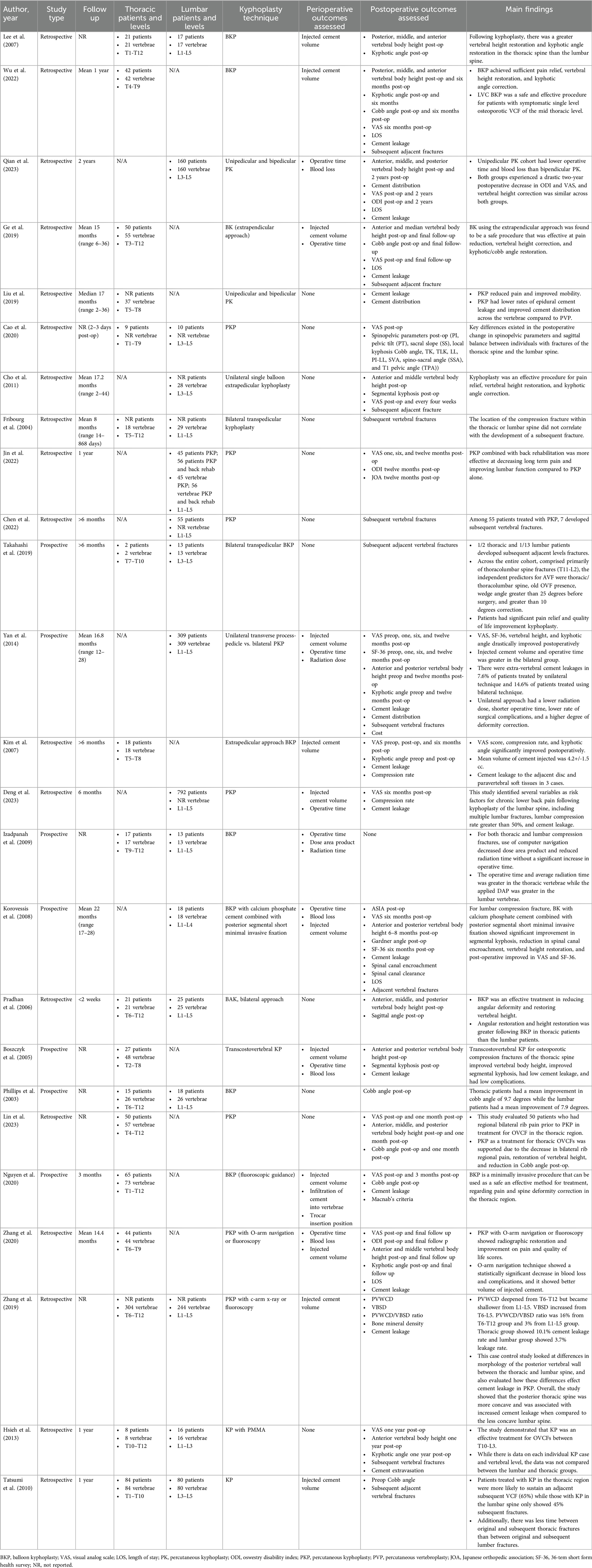
A total of 1,164 results were initially retrieved from the database literature search and yielded 25 manuscripts after screening (Figure 1). All included manuscripts were published between 2003 and 2023 and included both retrospective (N = 18, 92%) and prospective (N = 7, 28%) studies. Of these manuscripts, 7 (28.0%) reported on kyphoplasty outcomes for lumbar fracture patient cohorts, 8 (32.0%) reported on kyphoplasty outcomes for thoracic fracture patient cohorts, and 10 (40.0%) reported on kyphoplasty outcomes for lumbar and thoracic patient cohorts. An overview of all included studies is provided in Table 2.
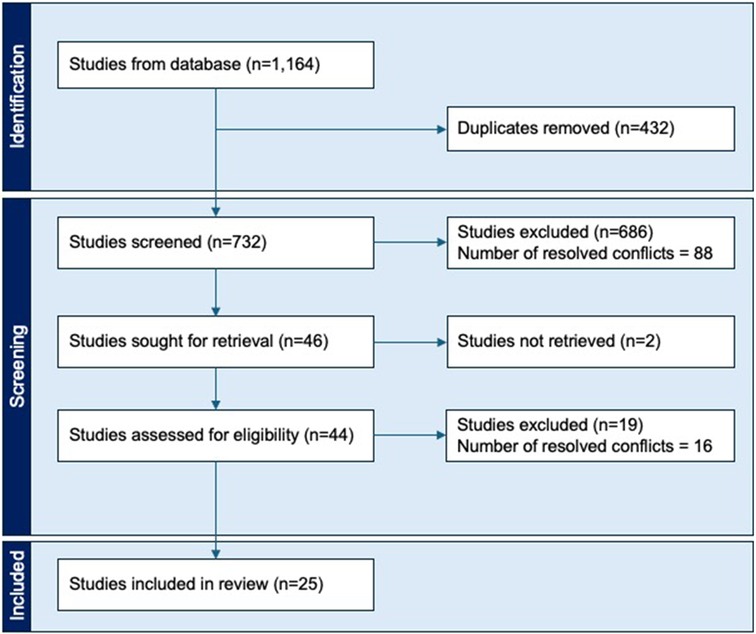
Figure 1. Flow diagram depicting the selection process for studies included within this systematic review.
Perioperative outcomes
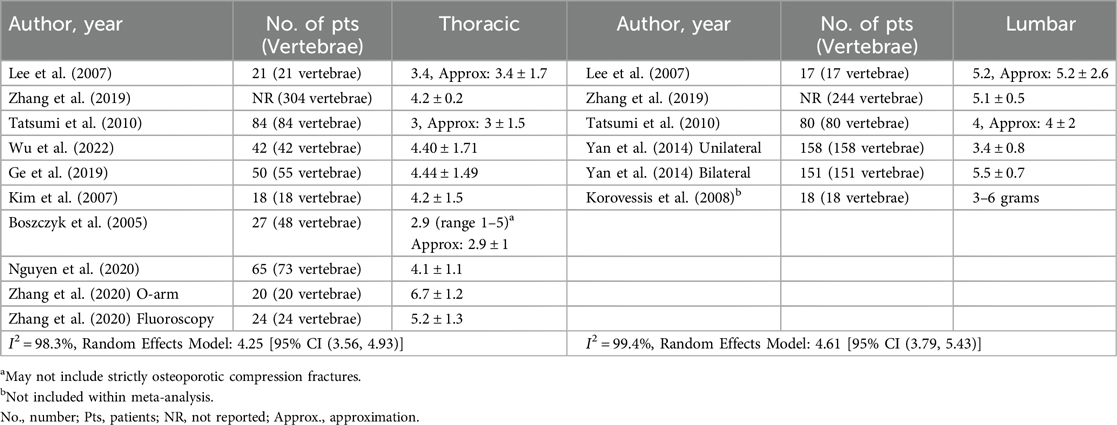
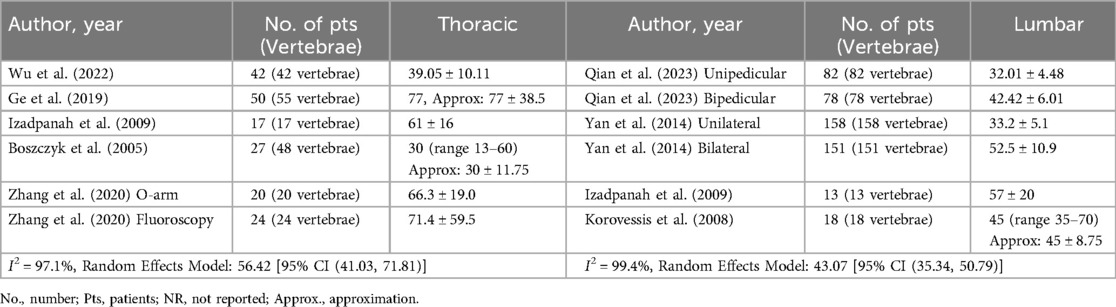
The most commonly reported perioperative outcomes included injected cement volume (9 thoracic studies, 5 lumbar studies) and operative time (5 thoracic studies, 4 lumbar studies). Meta-analysis using a random effects model revealed an injected cement volume of 4.25 ml [95% CI (3.56, 4.93)] in the thoracic spine and 4.61 ml [95% CI (3.79, 5.43)] in the lumbar spine (Table 3). Likewise, meta-analysis using a random effects model found operative time in the thoracic spine to be 56.42 min [95% CI (41.03, 71.81)] and 43.07 min [95% CI (35.34, 50.79)] in the lumbar spine (Table 4). Other common perioperative outcomes reported included blood loss (1 thoracic study, 2 lumbar studies) and length of stay (3 thoracic studies, 2 lumbar studies). Across thoracic cohorts, the observed blood loss ranged from 6.2 to 7.9 ml and the lumbar studies showed a range of 5.46 to 50.0 ml (Supplementary Table S1). The length of stay ranged from 3.48 to 7.3 days following thoracic procedures and 2.0 to 4.9 days following lumbar procedures (Supplementary Table S2).
Postoperative outcomes
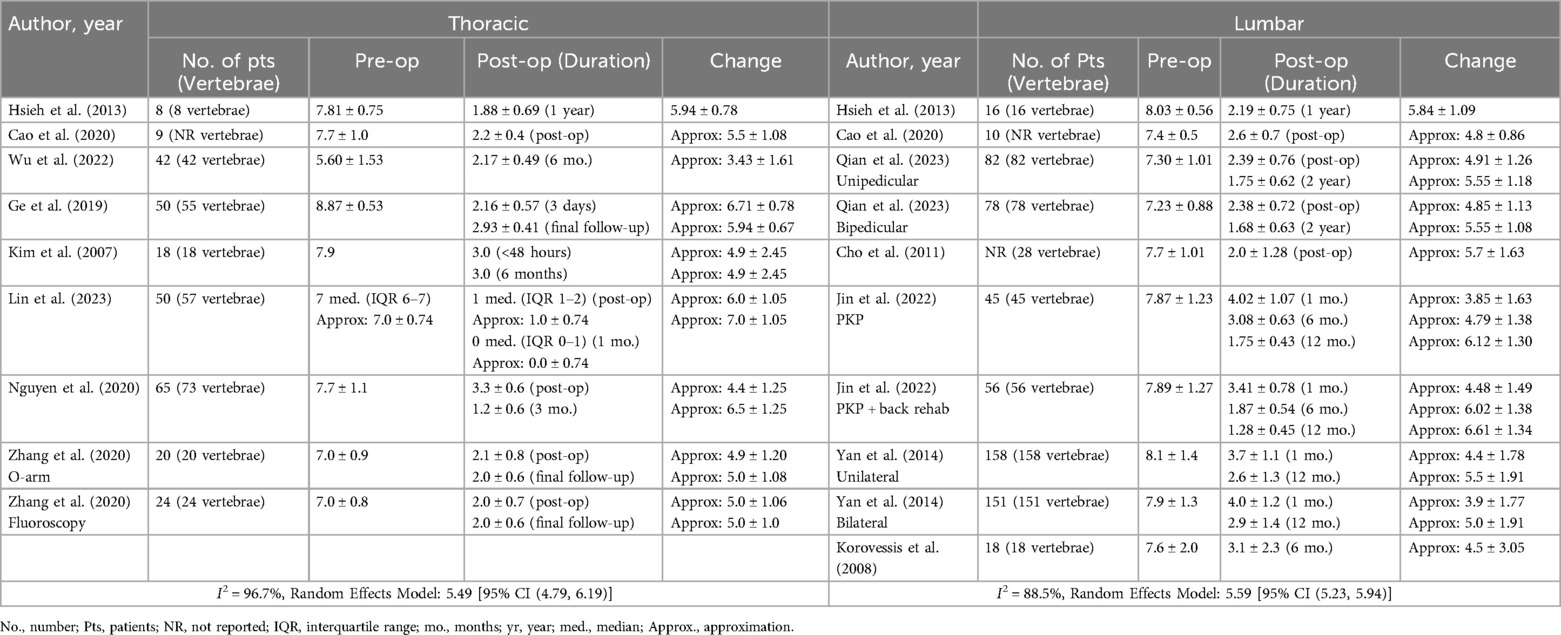
The most commonly reported postoperative outcomes included VAS (8 thoracic studies, 7 lumbar studies), kyphotic angle (5 thoracic studies, 4 lumbar studies), cobb angle (7 thoracic studies, 3 lumbar studies), anterior vertebral body height (8 thoracic studies, 7 lumbar studies), middle vertebral body height (6 thoracic studies, 3 lumbar studies), and posterior vertebral body height (5 thoracic studies, 5 lumbar studies). Using the VAS score from the last postoperative visit recorded in each study, meta-analysis using a random effects model revealed a decrease in VAS score of 5.49 [95% CI (4.79, 6.19)] following thoracic kyphoplasty and a decrease of 5.59 [95% CI (5.23, 5.94)] following lumbar kyphoplasty (Table 5).
Due to heterogeneity in measuring and reporting methods across studies, the findings for kyphotic angle, cobb angle, and anterior, middle, and posterior vertebral body heights were solely reported from studies that directly compared outcomes between thoracic and lumbar procedures. For kyphotic angle correction, Lee et al. reported a reduction of 8.6% in thoracic kyphotic angle compared to a 6.9% reduction in lumbar kyphotic angle (38). Hsieh et al. observed mean reductions of 18.30° ± 5.45° in thoracic kyphotic angle and 19.07° ± 5.53° in lumbar kyphotic angle (39). Regarding Cobb angles, Cao et al. reported a reduction in thoracic vertebrae from 25.1° ± 14.2° preoperatively to 22.0° ± 12.95° postoperatively, while lumbar vertebrae showed a slight increase from 27.5° ± 21.8° to 30.96° ± 21.6° (40). Phillips et al. reported a reduction in thoracic Cobb angle by 9.7° (range 0–29) and in lumbar Cobb angle by 7.9° (range 0–23) (41).
Following kyphoplasty, Lee et al. reported an increase of 25%, 44%, and 6% in anterior, middle, and posterior vertebral body height in the thoracic spine, respectively, and an increase of 13%, 33%, and 3% in anterior, middle, and posterior vertebral body height in the lumbar spine (38). Similarly, Pradhan et al. observed an increase of 27%, 52%, and 3% in anterior, middle, and posterior vertebral body height in the thoracic spine following kyphoplasty, respectively, and an increase of 1%, 34%, and 11% in anterior, middle, and posterior vertebral body height in the lumbar spine (42). One year following the kyphoplasty procedure, Hsieh et al. reported a 5.64 ± 4.46 mm increase in anterior vertebral body height within the thoracic spine, and a 5.85 ± 7.82 mm increase in anterior vertebral body height within the lumbar spine (39).
Complications
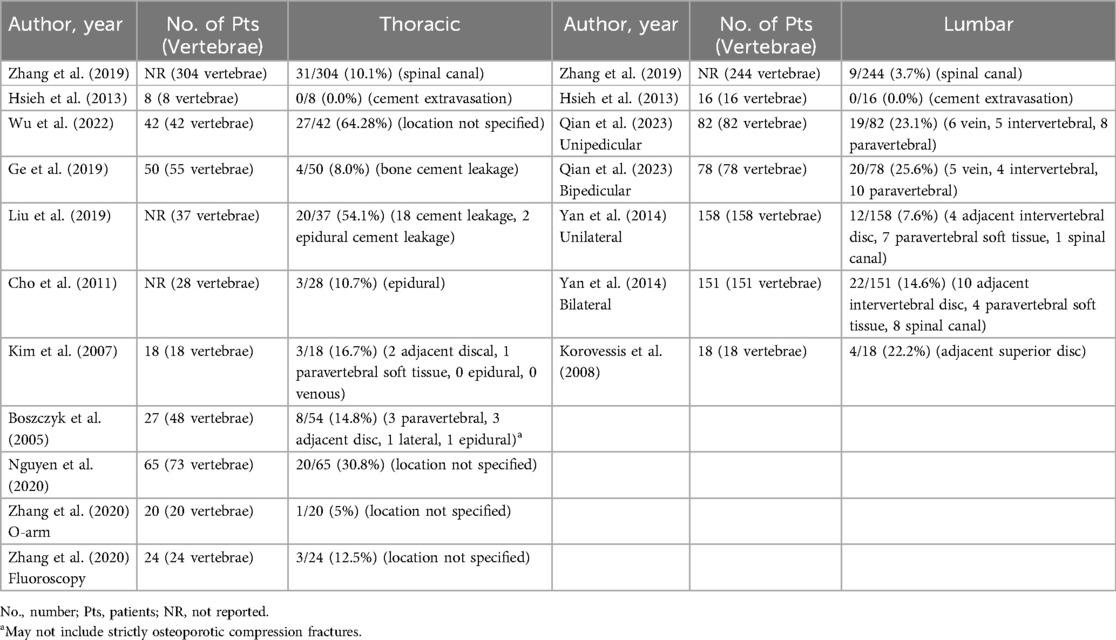
Amongst the studies included, cement leakage (9 thoracic studies, 4 lumbar studies) and subsequent fractures (7 thoracic studies, 7 lumbar studies) were the most described complications (Tables 6, 7). In thoracic procedures, the pooled proportion of cases with cement leakage was 19% [95% CI (10%, 32%)] using a random-effects model, with significant heterogeneity observed (I2 = 89%) (Figure 2A). The fixed-effects model estimated a slightly higher proportion of 21% [95% CI (18%, 25%)]. For lumbar procedures, the pooled proportion of cement leakage was 13% [95% CI (7%, 22%)], with substantial heterogeneity (I2 = 85%). The fixed-effects model produced a comparable estimate of 14% [95% CI (12%, 17%)] (Figure 2B).
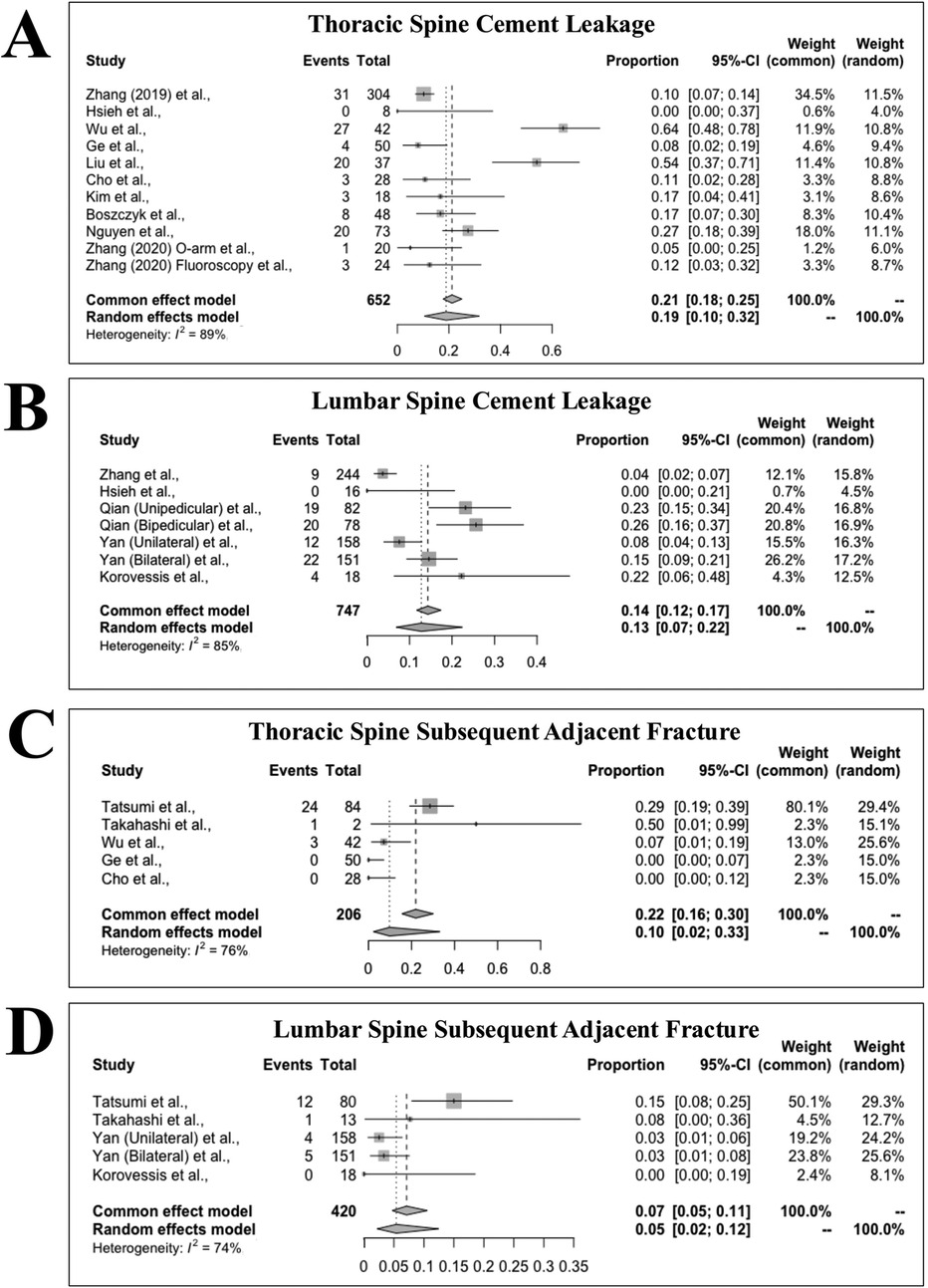
Figure 2. Forest plot analysis illustrating the proportion of cement leakage following kyphoplasty in the thoracic spine (A) and the lumbar spine (B). Forest plot analysis illustrating the proportion of subsequent adjacent fracture following kyphoplasty in the thoracic spine (C) and the lumbar spine (D).
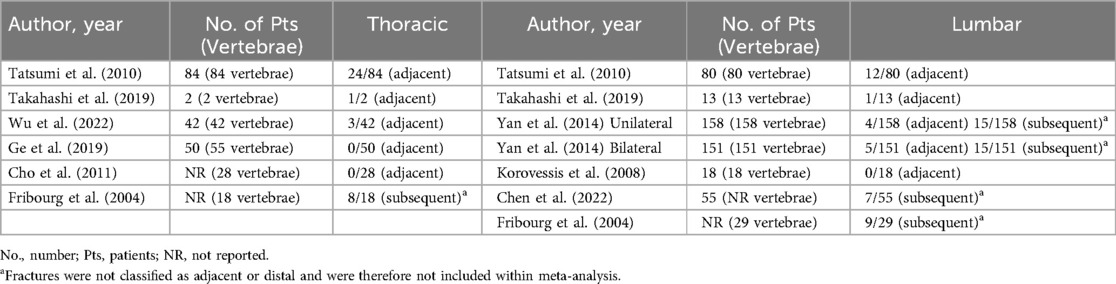
Regarding subsequent adjacent fractures, the pooled proportion for thoracic procedures was 10% [95% CI (2%, 33%)] under a random-effects model, with heterogeneity of I2 = 76%. The fixed-effects model estimated a higher proportion of 22% [95% CI (16%, 30%)] (Figure 2C). For lumbar procedures, the pooled proportion of adjacent fractures was 5% [95% CI (2%, 12%)] using a random-effects model, with I2 = 74%. The fixed-effects model yielded a slightly higher proportion of 7% [95% CI (5%, 11%)] (Figure 2D).
Risk of bias
Across all included studies, the primary risk of bias pertained to the risk of bias due to confounding factors. This resulted in a serious risk of bias in 12 studies (48.0%) and moderate risk of bias in 13 studies (52.0%) (Supplementary Figure S1).
Discussion
Kyphoplasty is a widely used surgical technique for the treatment of OVCFs in the thoracic and lumbar spine offering a safe option for fragile osteoporotic patients. This procedure is particularly advantageous because of its rapid onset of analgesia, ability to promote early mobilization without the need for bracing, and the ability to treat multiple levels simultaneously (6). Thoracic and lumbar OVCFs are often analyzed together in clinical reports assessing kyphoplasty based outcome measures such as pain relief, radiographic improvement, and complication rates, potentially overlooking how the distinct anatomical and biomechanical differences between these regions may influence surgical and patient outcomes. For example, the thoracic vertebra have smaller, steeper pedicles, a narrower anterior border, and less angulation between the pedicle and vertebra compared to the lumbar region (43). These anatomical differences can make certain procedural steps, such as pedicle cannulation, balloon inflation, and cement distribution, more challenging. To our knowledge, this is the first systematic review to evaluate how such differences between the thoracic and lumbar spine may impact perioperative and postoperative outcomes in patients undergoing kyphoplasty for the treatment of OVCFs.
One of the primary outcomes we assessed in our study was post-procedural pain relief, as pain is one of the major indications for kyphoplasty. Approximately one third of patients with OVCFs experience chronic pain at the site of the fracture, which often results in limitations to their daily activities and lifestyle (44). Kyphoplasty frequently results in a significant pain reduction, and Mooney et al. demonstrated that, regardless of vertebral height improvement, the majority of patients who undergo kyphoplasty for vertebral compression fractures experience pain relief and enhanced quality of life (45). For example, Li et al. investigated kyphoplasty treatment of OVCFs at the mid thoracic vertebrae and found that the VAS pain score of patients decreased significantly from 8.34 ± 1.19 preoperatively to 2.49 ± 1.01 postoperatively (27). Likewise, Qian et al. investigated the treatment of lumbar vertebral compression fractures and reported a significant reduction in VAS scores from 7.30 ± 1.01 preoperatively to 2.39 ± 0.76 postoperatively in the unipedicular group and from 7.23 ± 0.88 preoperatively to 2.38 ± 0.72 postoperatively in the bipedicular group (46). Our study found similar pain relief, as measured through VAS, between patients undergoing kyphoplasty for OVCFs of the thoracic and the lumbar spine. Specifically, both groups showed significant improvement in pain levels from pre-procedural to post-procedural with no notable difference between the thoracic and lumbar groups in terms of pain relief.
The prevalence of kyphoplasty related surgical complications was another key variable examined in our study was. Cement leakage into the paravertebral soft tissues, veins, intradiscal space, or intraspinal canal is a common complication of kyphoplasty and can lead to severe complications such as neurological deficits, shortness of breath, wound infections, and adjacent segment fractures (47, 48). Prior studies have reported the overall incidence of cement extravasation to be around 10% (49, 50). Cement extravasation occurs through pathways created by structural irregularities, including blood vessels, vertebral shell damage, fracture clefts, or improper instrument placement (51, 52). Performing kyphoplasty earlier after an OVCF has previously been shown to lead to better vertebral height restoration, reduced kyphotic deformity, and a lower incidence of cement leakage (53). Our review highlighted a higher mean rate of cement leakage in the thoracic spine compared to the lumbar spine. Although the substantial overlap in confidence intervals suggests that this difference is not clinically significant and that the risk of this complication is comparable between anatomical locations, the higher rate of cement leakage in the thoracic spine may be attributed to anatomical challenges, such as posterior wall morphology, which might complicate instrument placement or surgical view (54). Similarly, we investigated the prevalence of subsequent adjacent level fractures. The redistribution of biomechanical stress after kyphoplasty may place increased strain on adjacent vertebrae, increasing the risk for adjacent-level fractures that can worsen spinal deformity, exacerbate pain, and contribute to functional decline (55). Subsequent fractures are typically subacute or late complications, making them difficult to predict. According to a recent meta-analysis, they can occur between 7 and 52 months after percutaneous kyphoplasty (56). More than 50% of subsequent fractures occur adjacent to the treated vertebrae, reigniting the ongoing debate over whether kyphoplasty increases the incidence of subsequent vertebral fractures. Prior studies have estimated the incidence of adjacent-level fractures following vertebral augmentation to range between 7%–29% (57). Several risk factors have been identified for the development of an adjacent-level fracture, including age, vertebral body height loss, cement leakage, and vertebral angles, among others (55, 57–59). Within our study, the pooled proportion of adjacent-level fractures following kyphoplasty was higher in the thoracic spine compared to the lumbar spine. While the overlap within confidence intervals suggests this different may not be clinically significant, anatomical differences impacting biomechanical stress distribution in the thoracic spine may further increase its susceptibility to adjacent-level fractures. Further research is warranted to explore potential reasons for a higher likelihood of subsequent adjacent level fractures and cement leakage following kyphoplasty of the thoracic spine.
As larger volumes are thought to be associated with improved vertebral body height restoration and kyphotic angulation correction, we also investigated differences in the volume of cement injected during kyphoplasty (60). Prior studies have suggested that larger cement volumes may be a significant predictor of pain relief following the procedure, with volumes exceeding 4.5cc recommended for optimal outcomes (61). Given the anatomical differences between the thoracic and lumbar vertebrae, it was hypothesized that less cement would be required for thoracic vertebrae. While meta-analysis did reveal a decreased volume of cement injected during kyphoplasty of the thoracic spine compared to the lumbar spine, this difference was minimal and likely indicates that surgeons are using relatively similar volumes of cement regardless of the vertebral region being treated.
Operative time is another critical consideration in kyphoplasty, especially when treating thoracic OVCFs. The presence of ribs in the thoracic spine may increase the technical difficulty of the procedure due to challenges in balloon implantation, as well as the increased risk of pedicle fractures and neural dysfunction (43). Likewise, the thoracic spine has smaller pedicle size and increased kyphosis which leads to difficulty in needle placement and cannulation (62). We hypothesized that thoracic kyphoplasty would result in longer operative times compared to lumbar procedures. Previously reported operative times for lumbar kyphoplasty are between 52.5 and 120 min (29, 63–66). Our meta-analysis revealed a 13 min increase in operative time for kyphoplasty of the thoracic spine vs. the lumbar spine. Prolonged operative time elevates a patient's risk for infection, anesthesia-related complications, excessive blood loss, and increased surgical costs, among other potential complications. Nevertheless, further research is needed to better understand this difference in operative time and determine whether it has clinically significant impacts on the predominantly frail and elderly patients undergoing this operative procedure.
Our study highlights several key similarities in outcomes between thoracic and lumbar OVCFs treated with kyphoplasty, including cement volume injected, VAS pain scores, cement leakage rates, and the incidence of subsequent adjacent fractures. Despite the anatomical challenges associated with the thoracic spine, our findings support that kyphoplasty can be performed safely and effectively in both regions with comparable outcomes. Nevertheless, this review draws attention to longer operative times and a higher incidence of cement leakage and adjacent-level fractures in the thoracic spine. While these differences may not be clinically significant, they underscore the need for larger, more controlled prospective studies to further explore potential variations in outcomes based on fracture location. Likewise, these findings provide an outcomes-based framework for surgeons to inform surgical decision-making and optimize treatment strategies for elderly patients with OVCFs depending on the spinal region affected.
Limitations
This review was not registered. Although we conducted a thorough and systematic assessment of the literature across multiple databases using all key terms and search combinations deemed relevant, there remains a possibility that studies offering additional insights were unintentionally omitted. Similarly, any insights that could have been gained from reviews, case reports, conference abstracts, non-English manuscripts, animal studies, expert opinions, editorials, or non-original research were not captured due to the exclusion criteria employed in this review. The authors noted substantial variability in sample sizes, kyphoplasty techniques, and the demographic and clinical profiles of research participants across studies that may limit the generalizability of the findings. The most prevalent source of bias across studies arose from confounding variables. Most studies included in this review did not directly compare the outcomes of kyphoplasty performed on the thoracic and lumbar spine within the same study. Consequently, cross-study comparisons are inherently influenced by variations in surgeons, surgical techniques, patient populations, and hospital resources, which can impact the interpretation of results. Likewise, several studies included in this review did not clearly distinguish whether their patient populations consisted of individuals with acute and/or chronic OVCFs, representing another potential confounding variable. Lastly, to conduct meta-analysis, we converted the reported data from various studies to means and standard deviations using established statistical approximation methods whenever these metrics were not provided directly. As these techniques assumed an underlying normal distribution, the resulting estimates may be imprecise when that assumption is violated, potentially influencing our pooled outcomes. Nevertheless, all approximation used in analysis are documented in Tables 3–5. Future research on the effectiveness of different kyphoplasty techniques for osteoporotic fractures should incorporate spinal region-specific stratification, distinguishing between thoracic and lumbar fractures while evaluating their respective outcome measures. This direct approach would offer valuable insights into regional differences in outcomes within a controlled cohort, enhancing the understanding of treatment efficacy.
Conclusion
This systematic review and meta-analysis provided a comprehensive comparison of perioperative and postoperative outcomes of kyphoplasty between thoracic and lumbar regions of the spine, addressing a critical gap in the literature. Our findings suggest that despite the anatomic and biomechanical differences between the spinal regions, key outcome measures such as pain relief, cement leakage rates, and subsequent adjacent fracture rates remain comparable. While the literature suggests that kyphoplasty in the thoracic region poses technical challenges, including increased operative time and difficult deformity correction, the overall safety and success of the procedure remains consistent across the two spinal regions. The comparable outcomes support a broader adoption of kyphoplasty in the thoracic spine, challenging the prevailing notion that the procedure is more suitable for the lumbar spine. Future studies investigating kyphoplasty should subcategorize patient cohorts by operative spinal region to directly evaluate the impact of varying anatomical and biomechanical factors in the success of the kyphoplasty procedure between the thoracic and lumbar spine regions.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Author contributions
MK: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Visualization, Writing – original draft, Writing – review & editing. RR: Conceptualization, Investigation, Methodology, Supervision, Writing – original draft, Writing – review & editing. CG: Conceptualization, Investigation, Supervision, Writing – original draft, Writing – review & editing. TE: Writing – original draft, Writing – review & editing. CCi: Data curation, Investigation, Writing – review & editing. CCo: Conceptualization, Investigation, Methodology, Supervision, Writing – review & editing. TZ: Data curation, Investigation, Writing – review & editing. SL: Data curation, Investigation, Writing – review & editing. JC: Investigation, Writing – original draft, Writing – review & editing. MS: Investigation, Supervision, Writing – review & editing. RW: Investigation, Supervision, Writing – review & editing. JL: Investigation, Supervision, Writing – review & editing. VS: Conceptualization, Investigation, Supervision, Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmscd.2025.1597288/full#supplementary-material
References
1. Issa K, Diebo BG, Faloon M, Naziri Q, Pourtaheri S, Paulino CB, et al. The epidemiology of vertebral osteomyelitis in the United States from 1998 to 2013. Clin Spine Surg. (2018) 31(2):E102–e8. doi: 10.1097/BSD.0000000000000597
2. Alexandru D, So W. Evaluation and management of vertebral compression fractures. Perm J. (2012) 16(4):46–51. doi: 10.7812/TPP/12-037
3. Ralston SH, Fraser J. Diagnosis and management of osteoporosis. Practitioner. (2015) 259(1788):15–92.26882774
4. Do HM, Doan HT-N, Dinh SN. Practical evaluation of risk factors in patients with osteoporosis-induced thoracic and lumbar vertebral compression fractures requiring surgery. Surg Pract Sci. (2024) 19:100260. doi: 10.1016/j.sipas.2024.100260
5. Wilson-Barnes SL, Lanham-New SA, Lambert H. Modifiable risk factors for bone health & fragility fractures. Best Pract Res Clin Rheumatol. (2022) 36(3):101758. doi: 10.1016/j.berh.2022.101758
6. Prost S, Pesenti S, Fuentes S, Tropiano P, Blondel B. Treatment of osteoporotic vertebral fractures. Orthop Traumatol Surg Res. (2021) 107(1 Supplement):102779. doi: 10.1016/j.otsr.2020.102779
7. Rajasekaran S, Kanna RM, Schnake KJ, Vaccaro AR, Schroeder GD, Sadiqi S, et al. Osteoporotic thoracolumbar fractures-how are they different?-classification and treatment algorithm. J Orthop Trauma. (2017) 31(Suppl 4):S49–s56. doi: 10.1097/BOT.0000000000000949
8. Maselli F, Palladino M, Barbari V, Storari L, Rossettini G, Testa M. The diagnostic value of red flags in thoracolumbar pain: a systematic review. Disabil Rehabil. (2022) 44(8):1190–206. doi: 10.1080/09638288.2020.1804626
9. Han CS, Hancock MJ, Downie A, Jarvik JG, Koes BW, Machado GC, et al. Red flags to screen for vertebral fracture in people presenting with low back pain. Cochrane Database Syst Rev. (2023) 8(8):Cd014461. doi: 10.1002/14651858.CD014461.pub2
10. Lee HM, Park SY, Lee SH, Suh SW, Hong JY. Comparative analysis of clinical outcomes in patients with osteoporotic vertebral compression fractures (OVCFs): conservative treatment versus balloon kyphoplasty. Spine J. (2012) 12(11):998–1005. doi: 10.1016/j.spinee.2012.08.024
11. Lau E, Ong K, Kurtz S, Schmier J, Edidin A. Mortality following the diagnosis of a vertebral compression fracture in the medicare population. J Bone Joint Surg Am. (2008) 90(7):1479–86. doi: 10.2106/JBJS.G.00675
12. Suzuki N, Ogikubo O, Hansson T. The prognosis for pain, disability, activities of daily living and quality of life after an acute osteoporotic vertebral body fracture: its relation to fracture level, type of fracture and grade of fracture deformation. Eur Spine J. (2009) 18(1):77–88. doi: 10.1007/s00586-008-0847-y
13. Spiegl U, Bork H, Grüninger S, Maus U, Osterhoff G, Scheyerer MJ, et al. Osteoporotic fractures of the thoracic and lumbar vertebrae: diagnosis and conservative treatment. Dtsch Arztebl Int. (2021) 118(40):670–7. doi: 10.3238/arztebl.m2021.0295
14. Alimy AR, Anastasilakis AD, Carey JJ, D'Oronzo S, Naciu AM, Paccou J, et al. Conservative treatments in the management of acute painful vertebral compression fractures: a systematic review and network meta-analysis. JAMA Netw Open. (2024) 7(9):e2432041. doi: 10.1001/jamanetworkopen.2024.32041
15. Watanabe K, Katsumi K, Ohashi M, Shibuya Y, Hirano T, Endo N, et al. Surgical outcomes of spinal fusion for osteoporotic vertebral fracture in the thoracolumbar spine: comprehensive evaluations of 5 typical surgical fusion techniques. J Orthop Sci. (2019) 24(6):1020–6. doi: 10.1016/j.jos.2019.07.018
16. Jang HD, Kim EH, Lee JC, Choi SW, Kim HS, Cha JS, et al. Management of osteoporotic vertebral fracture: review update 2022. Asian Spine J. (2022) 16(6):934–46. doi: 10.31616/asj.2022.0441
17. Gutierrez-Gonzalez R, Royuela A, Zamarron A. Vertebral compression fractures: pain relief, progression and new fracture rate comparing vertebral augmentation with brace. BMC Musculoskelet Disord. (2023) 24(1):898. doi: 10.1186/s12891-023-07041-1
18. Häckel S, Renggli AA, Albers CE, Benneker LM, Deml MC, Bigdon SF, et al. How to measure the outcome in the surgical treatment of vertebral compression fractures? A systematic literature review of highly cited level-I studies. BMC Musculoskelet Disord. (2021) 22(1):579. doi: 10.1186/s12891-021-04305-6
19. Hoyt D, Urits I, Orhurhu V, Orhurhu MS, Callan J, Powell J, et al. Current concepts in the management of vertebral compression fractures. Curr Pain Headache Rep. (2020) 24(5):16. doi: 10.1007/s11916-020-00849-9
20. Madassery S. Vertebral compression fractures: evaluation and management. Semin Intervent Radiol. (2020) 37(2):214–9. doi: 10.1055/s-0040-1709208
21. Schoenfeld AJ, Bono CM. Measuring spine fracture outcomes: common scales and checklists. Injury. (2011) 42(3):265–70. doi: 10.1016/j.injury.2010.11.040
22. Kumar K, Nguyen R, Bishop S. A comparative analysis of the results of vertebroplasty and kyphoplasty in osteoporotic vertebral compression fractures. Oper Neurosurg. (2010) 67(3):ons171-ons88. doi: 10.1227/01.NEU.0000380936.00143.11
23. Blattert TR, Schnake KJ, Gonschorek O, Gercek E, Hartmann F, Katscher S, et al. Nonsurgical and surgical management of osteoporotic vertebral body fractures: recommendations of the spine section of the German society for orthopaedics and trauma (DGOU). Global Spine J. (2018) 8(2 Suppl):50s–5. doi: 10.1177/2192568217745823
24. Boss S, Srivastava V, Anitescu M. Vertebroplasty and kyphoplasty. Phys Med Rehabil Clin N Am. (2022) 33(2):425–53. doi: 10.1016/j.pmr.2022.01.008
25. Filippiadis DK, Marcia S, Masala S, Deschamps F, Kelekis A. Percutaneous vertebroplasty and kyphoplasty: current Status, new developments and old controversies. Cardiovasc Intervent Radiol. (2017) 40(12):1815–23. doi: 10.1007/s00270-017-1779-x
26. McGirt MJ, Parker SL, Wolinsky J-P, Witham TF, Bydon A, Gokaslan ZL. Vertebroplasty and kyphoplasty for the treatment of vertebral compression fractures: an evidenced-based review of the literature. Spine J. (2009) 9(6):501–8. doi: 10.1016/j.spinee.2009.01.003
27. Li Y, Feng X, Pan J, Yang M, Li L, Su Q, et al. Percutaneous vertebroplasty versus kyphoplasty for thoracolumbar osteoporotic vertebral compression fractures in patients with distant lumbosacral pain. Pain Physician. (2021) 24(3):E349–e56.33988957
28. Liu JT, Liao WJ, Tan WC, Lee JK, Liu CH, Chen YH, et al. Balloon kyphoplasty versus vertebroplasty for treatment of osteoporotic vertebral compression fracture: a prospective, comparative, and randomized clinical study. Osteoporos Int. (2010) 21(2):359–64. doi: 10.1007/s00198-009-0952-8
29. Chen L, Yang H, Tang T. Unilateral versus bilateral balloon kyphoplasty for multilevel osteoporotic vertebral compression fractures: a prospective study. Spine (Phila Pa 1976). (2011) 36(7):534–40. doi: 10.1097/BRS.0b013e3181f99d70
30. Spiegl UJ, Scheyerer MJ, Osterhoff G, Grüninger S, Schnake KJ. Osteoporotic mid-thoracic vertebral body fractures: what are the differences compared to fractures of the lumbar spine?-a systematic review. Eur J Trauma Emerg Surg. (2022) 48(3):1639–47. doi: 10.1007/s00068-021-01792-z
31. Stott B, Driscoll M. Biomechanical evaluation of the thoracolumbar spine comparing healthy and irregular thoracic and lumbar curvatures. Comput Biol Med. (2023) 160:106982. doi: 10.1016/j.compbiomed.2023.106982
32. Iyer S, Christiansen BA, Roberts BJ, Valentine MJ, Manoharan RK, Bouxsein ML. A biomechanical model for estimating loads on thoracic and lumbar vertebrae. Clin Biomech (Bristol). (2010) 25(9):853–8. doi: 10.1016/j.clinbiomech.2010.06.010
33. Agai E. Pico Portal (2021). Available online at: https://picoportal.org/ (Accessed November 17, 2023).
34. Sterne J, Higgins J. Risk of Bias in Non-Randomized Studies—of Interventions (ROBINS-I). Ottawa: Cochrane Methods Bias (2024). Available online at: https://methods.cochrane.org/bias/risk-bias-non-randomized-studies-interventions (Accessed November 17, 2023).
35. Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. (2005) 5(1):13. doi: 10.1186/1471-2288-5-13
36. Guolo A, Varin C. Random-effects meta-analysis: the number of studies matters. Stat Methods Med Res. (2017) 26(3):1500–18. doi: 10.1177/0962280215583568
37. Jackson D, Turner R. Power analysis for random-effects meta-analysis. Res Synth Methods. (2017) 8(3):290–302. doi: 10.1002/jrsm.1240
38. Lee TO, Jo DJ, Kim SM. Outcome and efficacy of height gain and sagittal alignment after kyphoplasty of osteoporotic vertebral compression fractures. J Korean Neurosurg Soc. (2007) 42(4):271–5. doi: 10.3340/jkns.2007.42.4.271
39. Hsieh JY, Wu CD, Wang TM, Chen HY, Farn CJ, Chen PQ. Reduction of the domino effect in osteoporotic vertebral compression fractures through short-segment fixation with intravertebral expandable pillars compared to percutaneous kyphoplasty: a case control study. BMC Musculoskelet Disord. (2013) 14(75). doi: 10.1186/1471-2474-14-75
40. Cao Z, Wang G, Hui W, Liu B, Liu Z, Sun J. Percutaneous kyphoplasty for osteoporotic vertebral compression fractures improves spino-pelvic alignment and global sagittal balance maximally in the thoracolumbar region. PLoS One. (2020) 15(1):e0228341. doi: 10.1371/journal.pone.0228341
41. Phillips FM, Ho E, Campbell-Hupp M, McNally T, Todd Wetzel F, Gupta P. Early radiographic and clinical results of balloon kyphoplasty for the treatment of osteoporotic vertebral compression fractures. Spine (Phila Pa 1976). (2003) 28(19):2260–5. discussion 5–7. doi: 10.1097/01.BRS.0000085092.84097.7B
42. Pradhan BB, Bae HW, Kropf MA, Patel VV, Delamarter RB. Kyphoplasty reduction of osteoporotic vertebral compression fractures: correction of local kyphosis versus overall sagittal alignment. Spine (Phila Pa 1976). (2006) 31(4):435–41. doi: 10.1097/01.brs.0000200036.08679.1e
43. Ge J, Cheng X, Li P, Yang H, Zou J. The clinical effect of kyphoplasty using the extrapedicular approach in the treatment of thoracic osteoporotic vertebral compression fracture. World Neurosurg. (2019) 131:e284–e9. doi: 10.1016/j.wneu.2019.07.133
44. Harrop JS, Prpa B, Reinhardt MK, Lieberman I. Primary and secondary osteoporosis' Incidence of subsequent vertebral compression fractures after kyphoplasty. Spine (Phila Pa 1976). (2004) 29(19):2120–5. doi: 10.1097/01.brs.0000141176.63158.8e
45. Mooney JH, Amburgy J, Self M, Agee BS, Schoel L, Pritchard PR, et al. Vertebral height restoration following kyphoplasty. J Spine Surg. (2019) 5(2):194–200. doi: 10.21037/jss.2019.04.02
46. Qian Y, Li Y, Shen G, Zhong X, Tang C, He S. Comparison of unipedicular and bipedicular kyphoplasty for treating acute osteoporotic vertebral compression fractures in the lower lumbar spine: a retrospective study. BMC Musculoskelet Disord. (2023) 24(1):410. doi: 10.1186/s12891-023-06545-0
47. Semaan H, Obri T, Bazerbashi M, Paull D, Liu X, Sarrouj M, et al. Clinical outcome and subsequent sequelae of cement extravasation after percutaneous kyphoplasty and vertebroplasty: a comparative review. Acta Radiol. (2018) 59(7):861–8. doi: 10.1177/0284185117732599
48. Rose LD, Bateman G, Ahmed A. Clinical significance of cement leakage in kyphoplasty and vertebroplasty: a systematic review. Eur Spine J. (2024) 33(4):1484–9. doi: 10.1007/s00586-023-08026-3
49. Hulme PA, Krebs J, Ferguson SJ, Berlemann U. Vertebroplasty and kyphoplasty: a systematic review of 69 clinical studies. Spine (Phila Pa 1976). (2006) 31(17):1983–2001. doi: 10.1097/01.brs.0000229254.89952.6b
50. Taylor RS, Taylor RJ, Fritzell P. Balloon kyphoplasty and vertebroplasty for vertebral compression fractures: a comparative systematic review of efficacy and safety. Spine (Phila Pa 1976). (2006) 31(23):2747–55. doi: 10.1097/01.brs.0000244639.71656.7d
51. Baroud G, Crookshank M, Bohner M. High-viscosity cement significantly enhances uniformity of cement filling in vertebroplasty: an experimental model and study on cement leakage. Spine (Phila Pa 1976). (2006) 31(22):2562–8. doi: 10.1097/01.brs.0000240695.58651.62
52. Truumees E, Hilibrand A, Vaccaro AR. Percutaneous vertebral augmentation. Spine J. (2004) 4(2):218–29. doi: 10.1016/j.spinee.2003.08.029
53. Li Z, Chen J, Li D, Yang J, Qin J, Guan Y. Clinical efficacy and safety of kyphoplasty for the treatment of osteoporotic vertebral compression fractures at different surgical timings based on the theory of “dynamic-static integration”. J Tradit Chin Med Sci. (2024) 11(1):86–92.
54. Zhang S, Wang GJ, Wang Q, Yang J, Xu S, Yang CH. A mysterious risk factor for bone cement leakage into the spinal canal through the batson vein during percutaneous kyphoplasty: a case control study. BMC Musculoskelet Disord. (2019) 20(1):423. doi: 10.1186/s12891-019-2807-6
55. Shin HK, Park JH, Lee IG, Park JH, Park JH, Cho Y. A study on the relationship between the rate of vertebral body height loss before balloon kyphoplasty and early adjacent vertebral fracture. J Back Musculoskelet Rehabil. (2021) 34(4):649–56. doi: 10.3233/BMR-200121
56. Ding JK, Zhao B, Zhai YF. Subsequent fractures after vertebroplasty in osteoporotic vertebral fractures: a meta-analysis. Neurosurg Rev. (2022) 45(3):2349–59. doi: 10.1007/s10143-022-01755-x
57. Zhang T, Wang Y, Zhang P, Xue F, Zhang D, Jiang B. What are the risk factors for adjacent vertebral fracture after vertebral augmentation? A meta-analysis of published studies. Global Spine J. (2020) 12(1):130–41. doi: 10.1177/2192568220978223
58. Morozumi M, Matsubara Y, Muramoto A, Morita Y, Ando K, Kobayashi K, et al. A study of risk factors for early-onset adjacent vertebral fractures after kyphoplasty. Global Spine J. (2019) 10(1):13–20. doi: 10.1177/2192568219834899
59. Ko BS, Cho KJ, Park JW. Early adjacent vertebral fractures after balloon kyphoplasty for osteoporotic vertebral compression fractures. Asian Spine J. (2019) 13(2):210–5. doi: 10.31616/asj.2018.0224
60. Adida S, Taori S, Wong VR, Tang A, Sefcik RK, Zhang X, et al. Analysis of injected cement volume and clinical outcomes following balloon-assisted kyphoplasty in a series of 368 patients. Clin Neurol Neurosurg. (2024) 243:108367. doi: 10.1016/j.clineuro.2024.108367
61. Röder C, Boszczyk B, Perler G, Aghayev E, Külling F, Maestretti G. Cement volume is the most important modifiable predictor for pain relief in BKP: results from SWISSspine, a nationwide registry. Eur Spine J. (2013) 22(10):2241–8. doi: 10.1007/s00586-013-2869-3
62. Kim HS, Kim SW, Ju CI. Balloon kyphoplasty through extrapedicular approach in the treatment of middle thoracic osteoporotic compression fracture: t5-T8 level. J Korean Neurosurg Soc. (2007) 42(5):363–6. doi: 10.3340/jkns.2007.42.5.363
63. Chen C, Wei H, Zhang W, Gu Y, Tang G, Dong R, et al. Comparative study of kyphoplasty for chronic painful osteoporotic vertebral compression fractures via unipedicular versus bipedicular approach. J Spinal Disord Tech. (2011) 24(7):E62–5. doi: 10.1097/BSD.0b013e318228f470
64. Rebolledo BJ, Gladnick BP, Unnanuntana A, Nguyen JT, Kepler CK, Lane JM. Comparison of unipedicular and bipedicular balloon kyphoplasty for the treatment of osteoporotic vertebral compression fractures: a prospective randomised study. Bone Joint J. (2013) 95-B(3):401–6. doi: 10.1302/0301-620X.95B3.29819
65. Yan L, Jiang R, He B, Liu T, Hao D. A comparison between unilateral transverse process-pedicle and bilateral puncture techniques in percutaneous kyphoplasty. Spine (Phila Pa 1976). (2014) 39(26 Spec No.):B19–26. doi: 10.1097/BRS.0000000000000493
Keywords: kyphoplasty, osteoporotic compression fractures, thoracic compression fractures, lumbar compression fractures, clinical outcomes
Citation: Kann MR, Ramanathan R, Gonzalez C Jr, Edwards T, Cisneros C, Como CJ, Zou T, Lutes S, Chagas J, Spitnale MJ, Wawrose RA, Lee JY and Sharma V (2025) A comparison of clinical outcomes following kyphoplasty for osteoporotic compression fractures of the thoracic and lumbar spine: a systematic review and meta-analysis. Front. Musculoskelet. Disord. 3:1597288. doi: 10.3389/fmscd.2025.1597288
Received: 20 March 2025; Accepted: 15 July 2025;
Published: 18 August 2025.
Edited by:
Maryse Fortin, Concordia University, CanadaReviewed by:
Filippo Maselli, Sapienza University of Rome, ItalyDaryl Pinion Fields, University of Wisconsin-Madison, United States
Copyright: © 2025 Kann, Ramanathan, Gonzalez, Edwards, Cisneros, Como, Zou, Lutes, Chagas, Spitnale, Wawrose, Lee and Sharma. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Vivek Sharma, c2hhcm1hdjdAdXBtYy5lZHU=
 Michael R. Kann
Michael R. Kann Rahul Ramanathan
Rahul Ramanathan Christopher Gonzalez Jr1,2,3
Christopher Gonzalez Jr1,2,3 Timothy Edwards
Timothy Edwards