- 1Hubei University of Chinese Medicine, Wuhan, China
- 2Xiyuan Hospital, China Academy of Chinese Medical Sciences, Beijing, China
- 3Department of Nephrology, Wuhan Hospital of Traditional Chinese Medicine, Wuhan, China
Diabetic kidney disease (DKD), a grave microvascular complication of diabetes, is the primary cause of end-stage renal disease. Despite advances in conventional therapies, their limited efficacy underscores the urgent need for novel, multi-target intervention strategies. Macrophage infiltration and the subsequent chronic microinflammation are central to the pathogenesis of renal injury in DKD. A diverse array of natural bioactive compounds are emerging as promising therapeutic agents, capable of modulating these inflammatory pathways. This review investigates the mechanisms underlying the attenuation of DKD progression by six major classes of natural compounds, such as glycosides, diterpenoids, and alkaloids, among others, through the targeting of macrophage infiltration. Collectively, this synthesis offers a compelling case for developing natural product-based, multiple-target strategies to combat DKD. Collectively, this synthesis builds a compelling case for developing multi-target therapeutic strategies derived from natural products to combat DKD.
1 Introduction
Diabetic kidney disease (DKD), a significant complication of diabetes involving the microvasculature, is the foremost cause of ESRD worldwide (Liu et al., 2024; Zhao et al., 2024). It is predicted that the global prevalence of diabetes will increase from 643 million in 2030 to 783 million by 2045, with up to 40% of these individuals expected to develop kidney disease. This escalating prevalence positions DKD as a major global health challenge, profoundly impacting patient prognosis and imposing a substantial socioeconomic burden (Hill-Briggs et al., 2020; Pan et al., 2022).
At the core of DKD pathogenesis lies chronic renal microinflammation, hallmarked by the infiltration of macrophages into the glomeruli and tubulointerstitium (Wang et al., 2022). Within the diabetic renal microenvironment, these infiltrated macrophages become activated, releasing a cascade of proinflammatory cytokines and chemokines that drive progressive renal tissue damage and functional decline (Liu et al., 2023; Li et al., 2022). Consequently, targeting macrophage-mediated inflammation represents a critical therapeutic strategy for mitigating DKD progression.
According to guidelines from the American Diabetes Association (ADA) and Kidney Disease: Improving Global Outcomes (KDIGO), the standard treatment for DKD combines renin-angiotensin system inhibitors (ACE inhibitors or angiotensin receptor blockers) with cardiorenal protective medications, including sodium-glucose cotransporter-2 inhibitors (SGLT2i), non-steroidal mineralocorticoid receptor antagonists (ns-MRA), and glucagon-like peptide-1 receptor agonists (GLP-1R). However, this strategy provides only partial renal protection and is associated with various adverse effects (Tang et al., 2021; Zhao et al., 2025; de Boer et al., 2022). This highlights a critical unmet clinical need for novel therapeutic strategies. Natural products, many derived from traditional herbal medicines, are emerging as a promising source of multi-target agents capable of modulating these complex inflammatory pathways (Deng et al., 2025; Wang et al., 2024; Chung et al., 2023). This review, therefore, synthesizes current evidence on the mechanisms by which natural products modulate macrophage infiltration and activity in DKD, providing a rationale for their development as adjunctive therapies to prevent or treat this devastating disease.
2 Mechanisms of macrophage infiltration in the intervention of DKD
In adults, the predominant origin of macrophages is from bone marrow hematopoietic stem cells. The development of circulating monocytes that then go on to mature as macrophages is triggered by these progenitors (Chowdhury and Trivedi, 2023). However, certain tissues also harbor self-renewing, embryonically-derived resident macrophage populations (Lazarov et al., 2023). Crucially, the local renal microenvironment dictates macrophage identity and function by inducing tissue-specific gene expression programs that are essential for maintaining homeostasis.
Macrophage infiltration into the kidney is a pivotal early event that drives sustained renal injury in DKD (Jiang et al., 2022). In the context of diabetic conditions, a combination of hyperglycaemia, oxidative stress and advanced glycation end products (AGEs) stimulates renal cells to secrete a range of chemokines and inflammatory mediators. These signals orchestrate the recruitment and subsequent activation of macrophages, thereby initiating and amplifying the local inflammatory cascade (Ansari et al., 2025). Once recruited, these macrophages exhibit remarkable plasticity, polarizing into two distinct functional phenotypes in response to microenvironmental cues, namely, the classically activated (M1) pro-inflammatory and the alternatively activated (M2) anti-inflammatory/reparative subtypes (Wynn and Vannella, 2016). Consequently, inhibiting macrophage infiltration represents a powerful therapeutic approach. This strategy not only limits the expansion of the intrarenal macrophage pool but also modulates the local polarization balance, primarily by reducing the influx of pro-inflammatory monocytes that differentiate into M1-like macrophages. Shifting the balance away from a dominant M1 inflammatory response, which is characterized by citokines such as interleukin (IL)-6 and IL-12, mitigates tissue damage and promotes a microenvironment conducive to M2-mediated repair (Yan et al., 2020). Therefore, a central strategy for modulating the progression of DKD is to target macrophage infiltration.
3 Natural medicines modulate macrophage infiltration
Numerous bioactive constituents derived from Chinese herbal medicine, such as Paeoniflorin, Loganin, Baicalin, Triptolide, Ligustrazine, Curcumin, Hirudin, and Acetylshikonin, demonstrate protective effects against DKD by mitigating renal macrophage infiltration. They orchestrate this process through a multi-targeted mechanism, concurrently suppressing key signaling pathways including TLR4/NF-κB, RAGE/MCP-1, MAPK, PKC/NF-κB, and TGF-β1/Smad, thereby achieving synergistic renoprotection (Table 1; Figure 1).
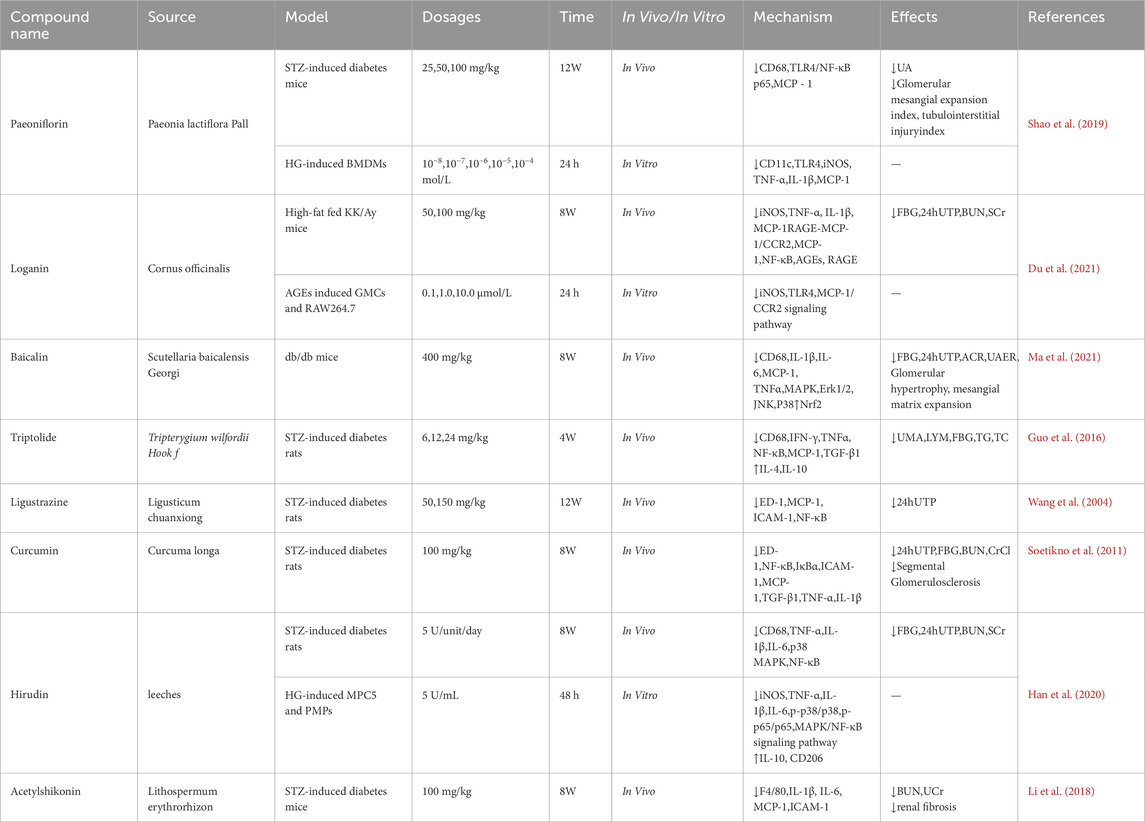
Table 1. The mechanism of natural bioactive compounds attenuating renal macrophage infiltration in diabetic kidney disease.
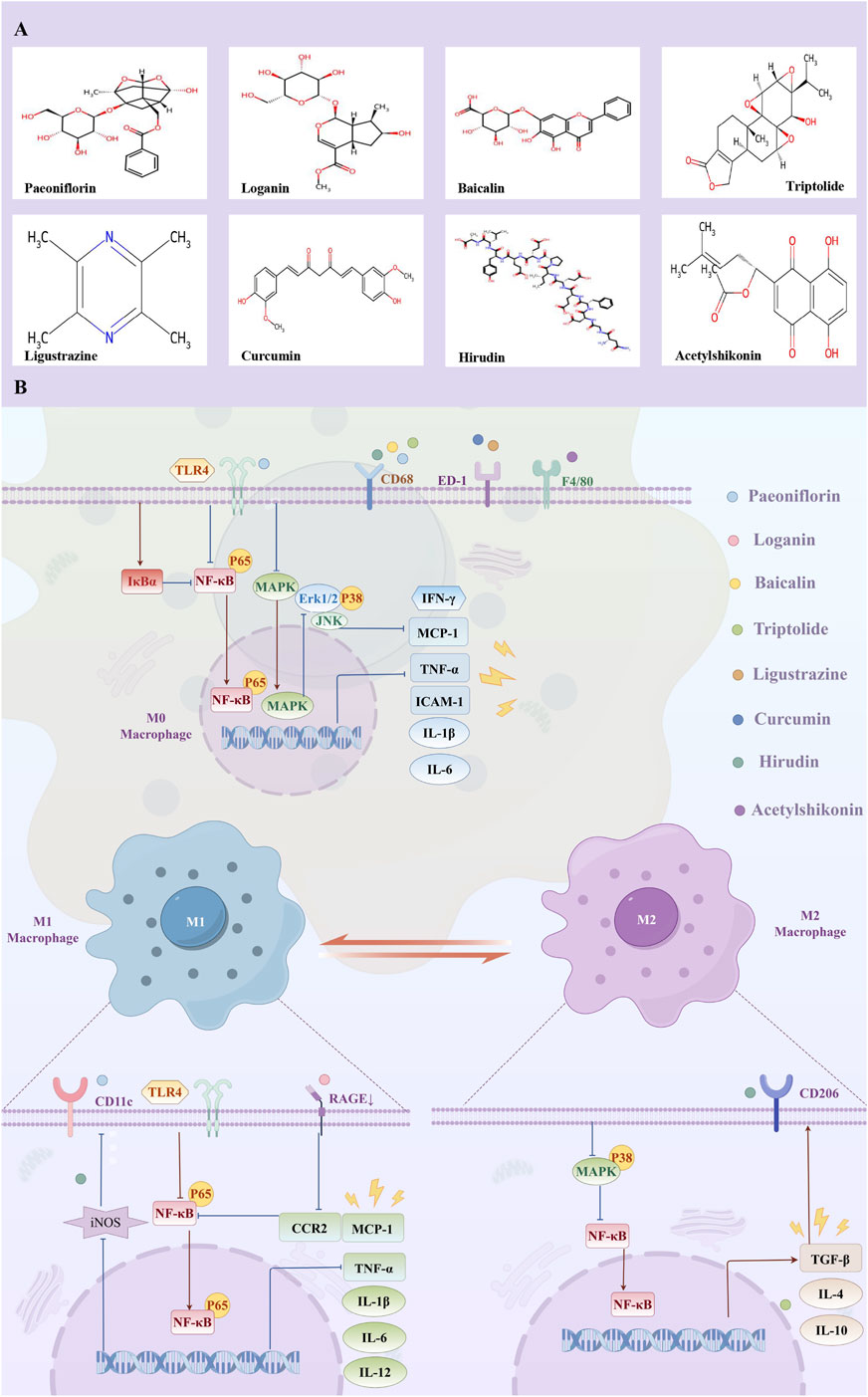
Figure 1. (A) Molecular structural formula of natural products; (B) The mechanism of natural bioactive compounds attenuating renal macrophage infiltration in diabetic kidney disease.
3.1 Glycoside compounds
The monoterpene glycoside Paeoniflorin (PF), which is isolated from various Paeonia species, is characterized by diverse pharmacological activities. Anti-allergic, antioxidant, and anti-inflammatory activities are among its documented range of effects (Cao et al., 2023). This compound has shown great promise as a treatment for various diseases, including arthritis, psoriasis, lupus and diabetes (Hong et al., 2022). Preclinical evidence strongly supports the utility of PF as a renoprotective compound in DKD (Zhang et al., 2023). Nuclear factor-κB (NF-κB) is a widely distributed nuclear transcription factor that regulates the expression of immune-related genes and participates in innate and adaptive immunity, mediating inflammatory responses (Yu et al., 2020). Toll-like receptors (TLRs) play a pivotal role in the innate immune response, recognising both pathogen- and damage-associated molecular patterns (de Melo et al., 2022). TLRs activate NF-κB transcription through both MyD88-dependent and MyD88-independent pathways, leading to the release of multiple proinflammatory cytokines including tumor necrosis factor alpha (TNF-α), IL-6, and monocyte chemoattractant protein-1 (MCP-1). Among these, the TLR4 signaling pathway plays a pivotal role in the inflammatory response and progressive fibrosis associated with kidney disease (Fock and Parnova, 2021). This activation, through both MyD88-dependent and independent pathways, converges on the transcription factor NF-κB, triggering the expression of proinflammatory cytokines (e.g., TNF-α, IL-6) and the potent chemokine MCP-1. Both experimental TLR4 knockout and clinical observations have confirmed a direct correlation between TLR4 expression, macrophage infiltration, and the severity of renal injury in DKD (Devaraj et al., 2011; Lin et al., 2012). By specifically inhibiting the TLR4/NF-κB p65 signaling axis, PF effectively suppresses this inflammatory cascade, thereby blocking macrophage migration into renal tissue and attenuating the progression of DKD (Shao et al., 2019).
Loganin is the most abundant cycloarene-type triterpene glycoside in Cornus officinalis (Zhang et al., 2022). Through its mediation of anti-inflammation, combating of oxidation, and inhibition of fibrosis, it exhibits broad biological activity against various kidney injuries, both acute and chronic (Jiang et al., 2012). Notably, strychnine exhibits renoprotective effects in early-stage DKD (Kong et al., 2023). AGEs are a pathogenic factor in DKD, causing kidney damage through multiple mechanisms (Yuan et al., 2017). AGEs drive renal injury by binding to their receptor (RAGE), which activates downstream signaling cascades, including NF-κB and Protein Kinase C (PKC). The transcription of MCP-1 is subsequently upregulated by this (Nebb et al., 2022). As a key chemokine, MCP-1 orchestrates the recruitment of macrophages to the kidney via its receptor, CC chemokine receptor 2 (CCR2), thus initiating and amplifying the local inflammatory response (He et al., 2023). Investigations into loganin’s mechanism of action highlight its role in inhibiting the RAGE–MCP-1/CCR2 axis as a key mechanism for reducing macrophage infiltration and alleviating renal injury in DKD models. This was substantiated by an in vivo study where an 8-week, low-dose treatment significantly improved renal pathology (Du et al., 2021).
Baicalin (BAI) is one of the most abundant flavonoid compounds found in the root of Scutellaria baicalensis Georgi (Wen et al., 2023). Modern pharmacological research indicates that BAI has a number of biological properties, such as antitumour, antibacterial, antioxidant and anti-inflammatory effects. This suggests its potential for treating various diseases (Li et al., 2025). In the field of DKD, BAI has been shown to have effective renal protective properties owing to its significant anti-inflammatory and antioxidant effects (Ren et al., 2023). The efficacy of BAI was demonstrated in a trial involving early-stage DKD patients, where it significantly improved markers of renal function, including 24-h urinary protein (UPT) and urinary albumin excretion rate (UAER), by reducing oxidative stress and modulating immune responsiveness (Yang et al., 2019). The mitogen-activated protein kinase (MAPK) signaling pathway is a central regulator of inflammatory responses. Its dysregulated activation prompts the secretion of inflammatory mediators and the infiltration of immune cells into tissues (Moon and Ro, 2021). BAI directly inhibits the phosphorylation of key proteins in the MAPK pathway—extracellular signal-related kinases one and 2 (Erk1/2), C-jun N-terminal kinase (JNK), and p38, thereby blocking downstream inflammatory signaling. Concurrently, it downregulates MCP-1 expression, reducing macrophage chemotaxis (Ma et al., 2021). Concurrently, BAI engages the antioxidant response by activating the nuclear factor E2-related factor 2 (Nrf2) pathway, a master regulator of endogenous antioxidant defenses. By suppressing MAPK-driven inflammation while bolstering Nrf2-mediated cytoprotection, this dual-pronged mechanism synergistically inhibits macrophage infiltration and ameliorates renal damage, as evidenced by reduced albuminuria in DKD animal models (Ma et al., 2021).
3.2 Diterpenoid compounds
Triptolide (TP), a diterpene lactone derived from the roots of Tripterygium wilfordii Hook. f. has been widely used in the management of inflammatory and immune-mediated diseases due to its significant anti-inflammatory and immunosuppressive properties (Song et al., 2023). It is currently regarded as one of the most exciting bioactive compounds in the shift from traditional to modern medicine (Gao et al., 2021). Recent research demonstrates that TP has a positive effect on multiple kidney diseases, characterised by a significant reduction in proteinuria (He et al., 2015; Zhou et al., 2016; Lv et al., 2023). In DKD, the immunopathology is driven in part by a skewed T-helper (Th) cell balance, favoring pro-inflammatory Th1 and pro-fibrotic Th2 responses (Wu et al., 2011). This inflammatory milieu promotes the NF-κB-dependent upregulation of MCP-1, the principal chemokine orchestrating the kidney’s recruitment of monocytes/macrophages. Local injury is amplified and fibrosis is driven by these recruited immune cells through mechanisms that include the induction of ransforming growth factor β1 (TGF-β1) (Tanase et al., 2022). Preclinical studies in a rat model of DKD confirm that TP mitigates renal injury precisely by targeting this axis: it rebalances the Th1/Th2 ratio, suppresses NF-κB activation, and consequently, inhibits the infiltration of pathogenic monocytes/macrophages (Guo et al., 2016).
Despite its therapeutic promise, the clinical translation of TP is severely hampered by significant systemic toxicity and poor aqueous solubility. To overcome these limitations, next-generation strategies are being pursued. These include the development of structurally optimized analogues with improved safety profiles, such as (5R)-5-Hydroxytriptolide (LLDT-8) and Minnelide, alongside novel drug delivery systems. Such advancements seek to improve the therapeutic index of triptolide-based compounds, with the potential to unlock their use for treating DKD and other inflammatory disorders (Tong et al., 2021).
3.3 Alkaloids
Ligustrazine is an efficacious alkaloid monomer extracted from the Chinese medicinal herb Ligusticum chuanxiong (Mu et al., 2023). This compound exerts its pharmacological effects through a multi-target mechanism, including improving microcirculatory disorders, inhibiting aldose reductase activity, providing antioxidant and anti-inflammatory effects, inhibiting fibrosis, and regulating autophagy. It demonstrates significant renal protective efficacy in the treatment of DKD (Gong et al., 2023). The clinical efficacy of Ligustrazine is substantiated by a metanalysis of 25 randomized controlled trials involving 1,645 patients. This analysis demonstrated that Ligustrazine significantly improves key markers of renal function, including blood urea nitrogen (BUN) and serum creatinine (SCr), while markedly reducing UTP and UAER. These data establish Ligustrazine as an effective therapy for preserving renal function and mitigating proteinuria in patients with DKD (Wang et al., 2012). The renoprotective mechanism of Ligustrazine is primarily attributed to its ability to suppress tubulointerstitial inflammation, a core pathological feature of DKD driven by macrophage infiltration (Liu S. et al., 2024). Intercellular adhesion molecule-1 (ICAM-1) is a key molecule mediating the adhesion of macrophages to vascular endothelium. Under physiological conditions, it exhibits basal low expression. However, its expression is increased in response to inflammatory stimuli (Ohga et al., 2007). Research indicates that anti-ICAM-1 antibodies effectively inhibit macrophage infiltration into DKD tissues by blocking the function of this molecule. This finding confirms the pivotal role of ICAM-1 in promoting macrophage recruitment in DKD (Wang et al., 2025). Ligustrazine directly targets the upstream signaling cascade governing ICAM-1 expression. Specifically, it inhibits the Protein Kinase C (PKC)/NF-κB signaling axis, which in turn prevents NF-κB-dependent transcription of the ICAM-172 gene. By downregulating this key adhesion molecule, Ligustrazine effectively diminishes macrophage infiltration into the renal tubulointerstitium, thus alleviating local inflammation and injury (Wang et al., 2004).
3.4 Polyphenolic compounds
Turmeric plants are widely cultivated and recognized throughout Southeast Asia, with their rhizomes commonly used as a spice and yellow food coloring (Yu et al., 2019; Chen et al., 2024). Curcumin (CUR) is a polyphenolic compound extracted from Curcuma longa L. as its core active ingredient. With its significant antioxidant and anti-inflammatory properties, coupled with favorable pharmacological safety characteristics, it demonstrates considerable therapeutic potential in the management of chronic inflammation, tumors, metabolic disorders and neurological diseases (ALTamimi et al., 2021). Its utility in DKD is substantiated by a meta-analysis of randomized controlled trials, which showed that CUR supplementation not only reduces serum creatinine but also concurrently improves key metabolic and cardiovascular risk factors, including total cholesterol, blood pressure, and fasting blood glucose (Jie et al., 2021). Mechanistically, CUR’s renoprotective effects are largely attributed to its potent inhibition of the NF-κB signaling pathway, a central hub for inflammation and fibrosis in the kidney (Liu et al., 2021). In the quiescent state, NF-κB is sequestered in the cytoplasm by its inhibitor, IκB. Within the diabetic milieu, however, pathogenic stimuli trigger the degradation of IκB, liberating NF-κB for nuclear translocation. Subsequent phosphorylation of its p65 subunit is a critical step that unleashes its transcriptional activity, driving the expression of pro-inflammatory and pro-fibrotic genes (Capece et al., 2022; Wang et al., 2010). Curcumin intervenes at critical nodes within this cascade to suppress the expression of key NF-κB target genes that mediate macrophage infiltration (e.g., ICAM-1, MCP-1) and fibrosis (TGF-β1). By inhibiting this inflammatory-fibrotic axis, CUR effectively reduces immune cell infiltration and extracellular matrix deposition, thereby attenuating the advancement progression of DKD (Soetikno et al., 2011).
3.5 Peptide compounds
Hirudin, a polypeptide derived from medicinal leeches, is the most potent natural inhibitor of thrombin known (Tian et al., 2024). While renowned for its clinical use as an anticoagulant, Hirudin’s therapeutic activities extend to anti-inflammatory and anti-fibrotic effects, which are highly relevant to DKD pathology (Liu S-J. et al., 2024). Recent findings have highlighted that thrombin plays a pivotal role in kidney disease and inflammatory regulation. For instance, nanoparticle-specific inhibition of thrombin activity can mitigate ischemia-reperfusion-induced kidney injury and improve renal function (DiSilvestro, 2000). Inflammation and thrombin exhibit bidirectional regulation, jointly driving disease progression. Concurrently, DKD represents a significant microvascular complication of diabetes, with its pathological essence being a chronic inflammatory state (Wang et al., 2023). A central feature of DKD is podocyte injury, which compromises the integrity of the glomerular filtration barrier and leads to proteinuria (Chen et al., 2024). This damage is driven by the pathological activation of intracellular signaling cascades, including the p38 MAPK and NF-κB pathways, which promote apoptosis and the secretion of inflammatory cytokines (Ural et al., 2023; Gong et al., 2021; Cui et al., 2019). Hirudin directly counteracts this pathology. Preclinical evidence demonstrates that Hirudin can suppress macrophage infiltration and proinflammatory cytokine expression through p38 MAPK/NF-κB-dependent mechanisms, thereby alleviating hyperglycemia-induced foot cell apoptosis and inflammatory responses. The functional consequence of this targeted mechanism is a marked improvement in renal function and a significant decrease in proteinuria in DKD animal models (Han et al., 2020).
3.6 Naphthoquinone compounds
Acetylshikonin, a naphthoquinone compound isolated from the plant Lithospermum erythrorhizon, exhibits pleiotropic biological activities such as antitumor, antimicrobial, and antioxidant effects (Lin et al., 2023a). Its therapeutic potential has been established for cardiovascular diseases, sexual dysfunction, and cancer (Lin et al., 2023b). Notably, Acetylshikonin exerts broad beneficial effects on diabetes and its complications, highlighting its promising potential as a novel treatment for DKD. Activation of the TGF-β1/Smad signaling pathway is closely associated with the fibrotic process in kidney disease. TGF-β1 not only promotes mesangial cell proliferation, mesangial matrix expansion and glomerulosclerosis, but also makes a significant contribution to tubulointerstitial fibrosis, making it a key pathway in the progression of DKD (Wang et al., 2021). Downstream, the Smad protein family, especially Smad2, Smad3, and Smad7, plays a pivotal role in the pathological regulation of DKD. Studies indicate that in STZ-induced DKD models, conditional knockout of Smad2 in fibroblasts using the fibroblast-specific protein-1 (FSP1) promoter significantly reduces renal fibrosis (Loeffler et al., 2018). Additionally, knockout of Smad3 markedly ameliorates pathological features including glomerular basement membrane thickening, extracellular matrix accumulation and proteinuria (Wang et al., 2021). Early renal inflammation and hypertrophy are crucial preliminary stages in the progression of renal fibrosis (Tian et al., 2015). Research in STZ-induced diabetic mouse models has shown that Acetylshikonin significantly alleviates inflammatory responses and fibrotic lesions in the kidneys (Li et al., 2018). Specifically, it inhibits macrophage infiltration and downregulates the expression of inflammatory cytokines IL-1β, IL-6, MCP-1, and the adhesion molecule ICAM-1, thereby effectively reducing local renal inflammation. Trichrome staining further confirmed that Acetylshikonin significantly inhibits renal fibrosis. At the signaling level, Acetylshikonin acts by reducing TGF-β1 levels and Smad2/3 phosphorylation, concurrently restoring Smad7 expression. In summary, Acetylshikonin prevents kidney inflammation and slows down the development of fibrosis in DKD by blocking the TGF-β1/Smad pathway. This results in fewer macrophages infiltrating the kidneys and lower expression of pro-inflammatory factors (Li et al., 2018).
4 Conclusion and perspectives
This review synthesizes existing evidence indicating that multiple natural active ingredients converge on the core pathogenic mechanism in DKD, namely, macrophage-mediated inflammation. Although current standard therapies, such as renin-angiotensin system inhibitors, SGLT2 inhibitors, and GLP-1 receptor agonists, provide cardiorenal protection through hemodynamic and metabolic regulation, a substantial residual risk of disease progression persists, primarily driven by ongoing inflammatory and fibrotic processes. The natural compounds discussed herein inhibit macrophage infiltration and downregulate the expression of key inflammatory mediators such as MCP-1 and ICAM-1, thereby intervening in pathological pathways that act synergistically with existing treatments. This offers a promising combinatorial strategy to disrupt the inflammatory-fibrotic axis driving DKD progression.
However, translational challenges remain, as most efficacy data are derived from preclinical models, and issues such as low bioavailability and dose-limiting toxicity hinder clinical application. To realize this synergistic potential, innovation in drug delivery systems and rigorous clinical trials are essential to validate the efficacy and safety of these natural compounds as adjuvants to standard therapy. The ultimate goal is to harness the complementary mechanisms of both conventional and natural agents to achieve superior therapeutic outcomes. Ultimately, this will fulfill their therapeutic value as a multi-target treatment strategy for DKD.
Author contributions
YY: Writing – original draft, Conceptualization. WW: Conceptualization, Writing – original draft. YC: Writing – original draft, Software. KZ: Writing – review and editing, Methodology. TZ: Methodology, Writing – review and editing. XS: Data curation, Writing – review and editing. JY: Data curation, Writing – review and editing. PL: Funding acquisition, Writing – review and editing. RY: Conceptualization, Writing – review and editing. GW: Conceptualization, Writing – review and editing, Supervision.
Funding
The authors declare that financial support was received for the research and/or publication of this article. This study was supported by the National Natural Science Foundation of China (No. 82274489 and 82575000), Beijing Natural Science Foundation (No. 7232326).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Altamimi J. Z., AlFaris N. A., Al-Farga A. M., Alshammari G. M., BinMowyna M. N., Yahya M. A. (2021). Curcumin reverses diabetic nephropathy in streptozotocin-induced diabetes in rats by inhibition of PKCβ/p66Shc axis and activation of FOXO-3a. J. Nutr. Biochem. 87, 108515. doi:10.1016/j.jnutbio.2020.108515
Ansari Z., Chaurasia A., Neha N., Sharma N., Bachheti R. K., Gupta P. C. (2025). Exploring inflammatory and fibrotic mechanisms driving diabetic nephropathy progression. Cytokine Growth Factor Rev. 84, 120–134. doi:10.1016/j.cytogfr.2025.05.007
Cao Y., Xiong J., Guan X., Yin S., Chen J., Yuan S., et al. (2023). Paeoniflorin suppresses kidney inflammation by regulating macrophage polarization via KLF4-mediated mitophagy. Phytomed Int. J. Phytother. Phytopharm. 116, 154901. doi:10.1016/j.phymed.2023.154901
Capece D., Verzella D., Flati I., Arboretto P., Cornice J., Franzoso G. (2022). NF-κB: blending metabolism, immunity, and inflammation. Trends Immunol. 43, 757–775. doi:10.1016/j.it.2022.07.004
Chen Y., Chen M., Zhu W., Zhang Y., Liu P., Li P. (2024). Morroniside attenuates podocytes lipid deposition in diabetic nephropathy: a network pharmacology, molecular docking and experimental validation study. Int. Immunopharmacol. 138, 112560. doi:10.1016/j.intimp.2024.112560
Chen Y., Tao J., He Y., Hou X., Fang J., Huang J., et al. (2024). Curcumin targets CXCL16-mediated podocyte injury and lipid accumulation in diabetic kidney disease treatment. Arch. Pharmacal Res. 47, 924–939. doi:10.1007/s12272-024-01521-1
Chowdhury S., Trivedi A. K. (2023). Origin, production and molecular determinants of macrophages for their therapeutic targeting. Cell Biol. Int. 47, 15–29. doi:10.1002/cbin.11914
Chung J. Y.-F., Lan H.-Y., Tang P. M.-K. (2023). New insights into traditional Chinese medicine in treatment of diabetic nephropathy. Integr. Med. Nephrol. Androl. 10, e00026. doi:10.1097/IMNA-D-22-00026
Cui F.-Q., Tang L., Gao Y.-B., Wang Y.-F., Meng Y., Shen C., et al. (2019). Effect of baoshenfang formula on podocyte injury via inhibiting the NOX-4/ROS/p38 pathway in diabetic nephropathy. J. Diabetes Res. 2019, 2981705. doi:10.1155/2019/2981705
de Boer I. H., K hunti K., Sadusky T., Tuttle K. R., Neumiller J. J., Rhee C. M., et al. (2022). Diabetes management in chronic kidney disease: a consensus report by the american diabetes association (ADA) and kidney disease: improving global outcomes (KDIGO). Diabetes Care 45, 3075–3090. doi:10.2337/dci22-0027
de Melo T. R., de Souza K. S. C., Ururahy M. A. G., Bortolin R. H., Bezerra J. F., de Oliveira Galvão M. F., et al. (2022). Toll-like receptor inflammatory Cascade and the development of diabetic kidney disease in children and adolescents with type 1 diabetes. J. Paediatr. Child. Health 58, 996–1000. doi:10.1111/jpc.15884
Deng Y., Zhu H., Xing J., Gao J., Duan J., Liu P., et al. (2025). The role of natural products in improving lipid metabolism disorder-induced mitochondrial dysfunction of diabetic kidney disease. Front. Physiol. 16, 1624077. doi:10.3389/fphys.2025.1624077
Devaraj S., Tobias P., Jialal I. (2011). Knockout of toll-like receptor-4 attenuates the pro-inflammatory state of diabetes. Cytokine 55, 441–445. doi:10.1016/j.cyto.2011.03.023
DiSilvestro R. A. (2000). Zinc in relation to diabetes and oxidative disease. J. Nutr. 130, 1509S–1511S. doi:10.1093/jn/130.5.1509S
Du Q., Fu Y.-X., Shu A.-M., Lv X., Chen Y.-P., Gao Y.-Y., et al. (2021). Loganin alleviates macrophage infiltration and activation by inhibiting the MCP-1/CCR2 axis in diabetic nephropathy. Life Sci. 272, 118808. doi:10.1016/j.lfs.2020.118808
Fock E. M., Parnova R. G. (2021). Protective effect of mitochondria-targeted antioxidants against inflammatory response to lipopolysaccharide challenge: a review. Pharmaceutics 13, 144. doi:10.3390/pharmaceutics13020144
Gao J., Zhang Y., Liu X., Wu X., Huang L., Gao W. (2021). Triptolide: pharmacological spectrum, biosynthesis, chemical synthesis and derivatives. Theranostics 11, 7199–7221. doi:10.7150/thno.57745
Gong P., Wang P., Pi S., Guo Y., Pei S., Yang W., et al. (2021). Proanthocyanidins protect against cadmium-induced diabetic nephropathy through p38 MAPK and Keap1/Nrf2 signaling pathways. Front. Pharmacol. 12, 801048. doi:10.3389/fphar.2021.801048
Gong L., Wang R., Wang X., Liu J., Han Z., Li Q., et al. (2023). Research progress of natural active compounds on improving podocyte function to reduce proteinuria in diabetic kidney disease. Ren. Fail 45, 2290930. doi:10.1080/0886022X.2023.2290930
Guo H., Pan C., Chang B., Wu X., Guo J., Zhou Y., et al. (2016). Triptolide improves diabetic nephropathy by regulating Th cell balance and macrophage infiltration in rat models of diabetic nephropathy. Exp. Clin. Endocrinol. Diabetes Off. J. Ger. Soc. Endocrinol. And. Ger. Diabetes Assoc. 124, 389–398. doi:10.1055/s-0042-106083
Han J., Pang X., Zhang Y., Peng Z., Shi X., Xing Y. (2020). Hirudin protects against kidney damage in Streptozotocin-Induced diabetic nephropathy rats by inhibiting inflammation via P38 MAPK/NF-κB pathway. Drug Des. Dev. Ther. 14, 3223–3234. doi:10.2147/DDDT.S257613
He L., Peng X., Liu G., Tang C., Liu H., Liu F., et al. (2015). Anti-inflammatory effects of triptolide on IgA nephropathy in rats. Immunopharmacol. Immunotoxicol. 37, 421–427. doi:10.3109/08923973.2015.1080265
He S., Yao L., Li J. (2023). Role of MCP-1/CCR2 axis in renal fibrosis: mechanisms and therapeutic targeting. Med. Baltim. 102, e35613. doi:10.1097/MD.0000000000035613
Hill-Briggs F., Adler N. E., Berkowitz S. A., Chin M. H., Gary-Webb T. L., Navas-Acien A., et al. (2020). Social determinants of health and diabetes: a scientific review. Diabetes Care 44, 258–279. doi:10.2337/dci20-0053
Hong H., Lu X., Wu C., Chen J., Chen C., Zhang J., et al. (2022). A review for the pharmacological effects of paeoniflorin in the nervous system. Front. Pharmacol. 13, 898955. doi:10.3389/fphar.2022.898955
Jiang W.-L., Zhang S.-P., Hou J., Zhu H.-B. (2012). Effect of loganin on experimental diabetic nephropathy. Phytomed Int. J. Phytother. Phytopharm. 19, 217–222. doi:10.1016/j.phymed.2011.08.064
Jiang W.-J., Xu C.-T., Du C.-L., Dong J.-H., Xu S.-B., Hu B.-F., et al. (2022). Tubular epithelial cell-to-macrophage communication forms a negative feedback loop via extracellular vesicle transfer to promote renal inflammation and apoptosis in diabetic nephropathy. Theranostics 12, 324–339. doi:10.7150/thno.63735
Jie Z., Chao M., Jun A., Wei S., LiFeng M. (2021). Effect of curcumin on diabetic kidney disease: a systematic review and meta-analysis of randomized, double-blind, placebo-controlled clinical trials. Evid-Based Complement. Altern. Med. Ecam 2021, 6109406. doi:10.1155/2021/6109406
Kong X., Zhao Y., Wang X., Yu Y., Meng Y., Yan G., et al. (2023). Loganin reduces diabetic kidney injury by inhibiting the activation of NLRP3 inflammasome-mediated pyroptosis. Chem. Biol. Interact. 382, 110640. doi:10.1016/j.cbi.2023.110640
Lazarov T., Juarez-Carreño S., Cox N., Geissmann F. (2023). Physiology and diseases of tissue-resident macrophages. Nature 618, 698–707. doi:10.1038/s41586-023-06002-x
Li Z., Hong Z., Peng Z., Zhao Y., Shao R. (2018). Acetylshikonin from Zicao ameliorates renal dysfunction and fibrosis in diabetic mice by inhibiting TGF-β1/Smad pathway. Hum. Cell 31, 199–209. doi:10.1007/s13577-017-0192-8
Li H.-D., You Y.-K., Shao B.-Y., Wu W.-F., Wang Y.-F., Guo J.-B., et al. (2022). Roles and crosstalks of macrophages in diabetic nephropathy. Front. Immunol. 13, 1015142. doi:10.3389/fimmu.2022.1015142
Li X., Xu R., Zhang D., Cai J., Zhou H., Song T., et al. (2025). Baicalin: a potential therapeutic agent for acute kidney injury and renal fibrosis. Front. Pharmacol. 16, 1511083. doi:10.3389/fphar.2025.1511083
Lin M., Yiu W. H., Wu H. J., Chan L. Y. Y., Leung J. C. K., Au W. S., et al. (2012). Toll-like receptor 4 promotes tubular inflammation in diabetic nephropathy. J. Am. Soc. Nephrol. JASN 23, 86–102. doi:10.1681/ASN.2010111210
Lin S.-S., Chang T.-M., Wei A. I.-C., Lee C.-W., Lin Z.-C., Chiang Y.-C., et al. (2023a). Acetylshikonin induces necroptosis via the RIPK1/RIPK3-dependent pathway in lung cancer. Aging (Albany NY) 15, 14900–14914. doi:10.18632/aging.25316
Lin S.-S., Chang T.-M., Wei A. I.-C., Lee C.-W., Lin Z.-C., Chiang Y.-C., et al. (2023b). Acetylshikonin induces necroptosis via the RIPK1/RIPK3-dependent pathway in lung cancer. Aging (Milano) 15, 14900–14914. doi:10.18632/aging.205316
Liu D., Chen X., He W., Lu M., Li Q., Zhang S., et al. (2024). Update on the pathogenesis, diagnosis, and treatment of diabetic tubulopathy. Integr. Med. Nephrol. Androl. 11, e23. doi:10.1097/IMNA-D-23-00029
Liu X., Zhang X., Cai X., Dong J., Chi Y., Chi Z., et al. (2021). Effects of curcumin on high glucose-induced epithelial-to-mesenchymal transition in renal tubular epithelial cells through the TLR4-NF-κB signaling pathway. Diabetes Metab. Syndr. Obes. Targets Ther. 14, 929–940. doi:10.2147/DMSO.S296990
Liu Y., Xu K., Xiang Y., Ma B., Li H., Li Y., et al. (2023). Role of MCP-1 as an inflammatory biomarker in nephropathy. Front. Immunol. 14, 1303076. doi:10.3389/fimmu.2023.1303076
Liu S., Han S., Wang C., Chen H., Xu Q., Feng S., et al. (2024a). MAPK1 Mediates MAM Disruption and mitochondrial dysfunction in diabetic kidney disease via the PACS-2-Dependent Mechanism. Int. J. Biol. Sci. 20, 569–584. doi:10.7150/ijbs.89291
Liu S.-J., Cao Y.-L., Zhang C. (2024b). Hirudin in the treatment of chronic kidney disease. Molecules 29, 1029. doi:10.3390/molecules29051029
Loeffler I., Liebisch M., Allert S., Kunisch E., Kinne R. W., Wolf G. (2018). FSP1-specific SMAD2 knockout in renal tubular, endothelial, and interstitial cells reduces fibrosis and epithelial-to-mesenchymal transition in murine STZ-induced diabetic nephropathy. Cell Tissue Res. 372, 115–133. doi:10.1007/s00441-017-2754-1
Lv C., Cheng T., Zhang B., Sun K., Lu K. (2023). Triptolide protects against podocyte injury in diabetic nephropathy by activating the Nrf2/HO-1 pathway and inhibiting the NLRP3 inflammasome pathway. Ren. Fail 45, 2165103. doi:10.1080/0886022X.2023.2165103
Ma L., Wu F., Shao Q., Chen G., Xu L., Lu F. (2021). Baicalin alleviates oxidative stress and inflammation in diabetic nephropathy via Nrf2 and MAPK signaling pathway. Drug Des. Dev. Ther. 15, 3207–3221. doi:10.2147/DDDT.S319260
Moon H., Ro S. W. (2021). MAPK/ERK signaling pathway in hepatocellular carcinoma. Cancers 13, 3026. doi:10.3390/cancers13123026
Mu Q., Yao K., Syeda M. Z., Zhang M., Cheng Q., Zhang Y., et al. (2023). Ligustrazine nanoparticle hitchhiking on neutrophils for enhanced therapy of cerebral ischemia-reperfusion injury. Adv. Sci. weinh Baden-Wurtt Ger. 10, e2301348. doi:10.1002/advs.202301348
Nebbioso M., Lambiase A., Armentano M., Tucciarone G., Sacchetti M., Greco A., et al. (2022). Diabetic retinopathy, oxidative stress, and sirtuins: an in depth look in enzymatic patterns and new therapeutic horizons. Surv. Ophthalmol. 67, 168–183. doi:10.1016/j.survophthal.2021.04.003
Ohga S., Shikata K., Yozai K., Okada S., Ogawa D., Usui H., et al. (2007). Thiazolidinedione ameliorates renal injury in experimental diabetic rats through anti-inflammatory effects mediated by inhibition of NF-kappaB activation. Am. J. Physiol. Ren. Physiol. 292, F1141–F1150. doi:10.1152/ajprenal.00288.2005
Pan S., Li Z., Wang Y., Liang L., Liu F., Qiao Y., et al. (2022). A comprehensive weighted gene co-expression network analysis uncovers potential targets in diabetic kidney disease. J. Transl. Int. Med. 10, 359–368. doi:10.2478/jtim-2022-0053
Ren G., Jiao P., Yan Y., Ma X., Qin G. (2023). Baicalin exerts a protective effect in diabetic nephropathy by repressing inflammation and oxidative stress through the SphK1/S1P/NF-κB signaling pathway. Diabetes Metab. Syndr. Obes. Targets Ther. 16, 1193–1205. doi:10.2147/DMSO.S407177
Shao Y.-X., Gong Q., Qi X.-M., Wang K., Wu Y.-G. (2019). Paeoniflorin ameliorates macrophage infiltration and activation by inhibiting the TLR4 signaling pathway in diabetic nephropathy. Front. Pharmacol. 10, 566. doi:10.3389/fphar.2019.00566
Soetikno V., Sari F. R., Veeraveedu P. T., Thandavarayan R. A., Harima M., Sukumaran V., et al. (2011). Curcumin ameliorates macrophage infiltration by inhibiting NF-κB activation and proinflammatory cytokines in streptozotocin induced-diabetic nephropathy. Nutr. Metab. 8, 35. doi:10.1186/1743-7075-8-35
Song J., He G.-N., Dai L. (2023). A comprehensive review on celastrol, triptolide and triptonide: insights on their pharmacological activity, toxicity, combination therapy, new dosage form and novel drug delivery routes. Biomed. Pharmacother. = Biomed. Pharmacother. 162, 114705. doi:10.1016/j.biopha.2023.114705
Tanase D. M., Gosav E. M., Anton M. I., Floria M., Seritean Isac P. N., Hurjui L. L., et al. (2022). Oxidative stress and NRF2/KEAP1/ARE pathway in diabetic kidney disease (DKD): new perspectives. Biomolecules 12, 1227. doi:10.3390/biom12091227
Tang G., Li S., Zhang C., Chen H., Wang N., Feng Y. (2021). Clinical efficacies, underlying mechanisms and molecular targets of chinese medicines for diabetic nephropathy treatment and management. Acta Pharm. Sin. B (11), 2749–2767. doi:10.1016/j.apsb.2020.12.020
Tian F., Yi X., Yang F., Chen Y., Zhu W., Liu P., et al. (2024). Research progress on the treatment of diabetic nephropathy with leech and its active ingredients. Front. Endocrinol. (Lausanne) 15, 1296843. doi:10.3389/fendo.2024.1296843
Tian W., Lei H., Guan R., Xu Y., Li H., Wang L., et al. (2015). Icariside II ameliorates diabetic nephropathy in streptozotocin-induced diabetic rats. Drug Des. Dev. Ther. 9, 5147–5157. doi:10.2147/DDDT.S90060
Tong L., Zhao Q., Datan E., Lin G.-Q., Minn I., Pomper M. G., et al. (2021). Triptolide: reflections on two decades of research and prospects for the future. Nat. Prod. Rep. 38, 843–860. doi:10.1039/d0np00054j
Ural C., Celik A., Ozbal S., Guneli E., Arslan S., Ergur B. U., et al. (2023). The renoprotective effects of taurine against diabetic nephropathy via the p38 MAPK and TGF-β/Smad2/3 signaling pathways. Amino Acids 55, 1665–1677. doi:10.1007/s00726-023-03342-w
Wang C., Wang Y., Zhang H., Liu B., Qiu X. (2021). Zishen qingre tongluo formula improves renal fatty acid oxidation and alleviated fibrosis via the regulation of the TGF-β1/Smad3 signaling pathway in hyperuricemic nephrology rats. Biomed. Res. Int. 2021, 2793823. doi:10.1155/2021/2793823
Wang G., Wu S., Xu W., Jin H., Zhu Z., Li Z., et al. (2010). Geniposide inhibits high glucose-induced cell adhesion through the NF-kappaB signaling pathway in human umbilical vein endothelial cells. Acta Pharmacol. Sin. 31, 953–962. doi:10.1038/aps.2010.83
Wang B., Ni Q., Wang X., Lin L. (2012). Meta-analysis of the clinical effect of ligustrazine on diabetic nephropathy. Am. J. Chin. Med. 40, 25–37. doi:10.1142/S0192415X12500036
Wang J., Zhang R., Wu C., Wang L., Liu P., Li P. (2024). Exploring potential targets for natural product therapy of DN: the role of SUMOylation. Front. Pharmacol. 15, 1432724. doi:10.3389/fphar.2024.1432724
Wang J., Zhang R., Wu C., Wang L., Liu P., Li P. (2024). Exploring potential targets for natural product therapy of DN: the role of SUMOylation. Front. Pharmacol. 15, 1432724. doi:10.3389/fphar.2024.1432724
Wang L., Wang H.-L., Liu T.-T., Lan H.-Y. (2021). TGF-beta as a master regulator of diabetic nephropathy. Int. J. Mol. Sci. 22, 7881. doi:10.3390/ijms22157881
Wang X., Zhao J., Li Y., Rao J., Xu G. (2022). Epigenetics and endoplasmic reticulum in podocytopathy during diabetic nephropathy progression. Front. Immunol. 13, 1090989. doi:10.3389/fimmu.2022.1090989
Wang Y., Cheng G., Wang G., Zhou X., Ma M., Wang J. (2025). Predictive and diagnostic value of MCP-1, MIF, and ICAM-1 in type-2 diabetes mellitus patients with diabetic kidney disease. Pak J. Med. Sci. 41, 1943–1948. doi:10.12669/pjms.41.7.12283
Wang Y., Sui Z., Wang M., Liu P. (2023). Natural products in attenuating renal inflammation via inhibiting the NLRP3 inflammasome in diabetic kidney disease. Front. Immunol. 14, 1196016. doi:10.3389/fimmu.2023.1196016
Wen Y., Wang Y., Zhao C., Zhao B., Wang J. (2023). The pharmacological efficacy of baicalin in inflammatory diseases. Int. J. Mol. Sci. 24, 9317. doi:10.3390/ijms24119317
Wu C.-C., Sytwu H.-K., Lu K.-C., Lin Y.-F. (2011). Role of T cells in type 2 diabetic nephropathy. Exp. Diabetes Res. 2011, 514738. doi:10.1155/2011/514738
Wynn T. A., Vannella K. M. (2016). Macrophages in tissue repair, regeneration, and fibrosis. Immunity 44, 450–462. doi:10.1016/j.immuni.2016.02.015
Yan Y., Tan R., Liu P., Li J., Zhong X., Liao Y., et al. (2020). Oridonin alleviates IRI-induced kidney injury by inhibiting inflammatory response of macrophages via AKT-related pathways. Med. Sci. Monit. 26, e921114–e921114-11. doi:10.12659/MSM.921114
Yang M., Kan L., Wu L., Zhu Y., Wang Q. (2019). Effect of baicalin on renal function in patients with diabetic nephropathy and its therapeutic mechanism. Exp. Ther. Med. 17, 2071–2076. doi:10.3892/etm.2019.7181
Yu Y., Sun J., Wang R., Liu J., Wang P., Wang C. (2019). Curcumin management of myocardial fibrosis and its mechanisms of action: a review. Am. J. Chin. Med. 47, 1675–1710. doi:10.1142/S0192415X19500861
Yu H., Lin L., Zhang Z., Zhang H., Hu H. (2020). Targeting NF-κB pathway for the therapy of diseases: mechanism and clinical study. Signal Transduct. Target Ther. 5, 209. doi:10.1038/s41392-020-00312-6
Yuan Y., Sun H., Sun Z. (2017). Advanced glycation end products (AGEs) increase renal lipid accumulation: a pathogenic factor of diabetic nephropathy (DN). Lipids Health Dis. 16, 126. doi:10.1186/s12944-017-0522-6
Zhang F., Yan Y., Zhang J., Li L., Wang Y.-W., Xia C.-Y., et al. (2022). Phytochemistry, synthesis, analytical methods, pharmacological activity, and pharmacokinetics of loganin: a comprehensive review. Phytother. Res. PTR 36, 2272–2299. doi:10.1002/ptr.7347
Zhang X.-E., Pang Y.-B., Bo Q., Hu S.-Y., Xiang J.-Y., Yang Z.-R., et al. (2023). Protective effect of paeoniflorin in diabetic nephropathy: a preclinical systematic review revealing the mechanism of action. PLOS One 18, e0282275. doi:10.1371/journal.pone.0282275
Zhao H., Li Z., Yan M., Ma L., Dong X., Li X., et al. (2024). Irbesartan ameliorates diabetic kidney injury in db/db mice by restoring circadian rhythm and cell cycle. J. Transl. Int. Med. 12, 157–169. doi:10.2478/jtim-2022-0049
Zhao M., Cao Y., Ma L. (2025). New insights in the treatment of DKD: recent advances and future prospects. BMC Nephrol. 26, 72. doi:10.1186/s12882-025-03953-3
Keywords: diabetic kidney disease, macrophage infiltration, inflammation, active compounds, Chinese herbal medicine
Citation: Yan Y, Wang W, Chen Y, Zhao K, Zhan T, Song X, Yang J, Liu P, Yu R and Wang G (2025) Potential pathways for natural active ingredients to intervene in diabetic kidney disease: targeting macrophage infiltration. Front. Physiol. 16:1723762. doi: 10.3389/fphys.2025.1723762
Received: 13 October 2025; Accepted: 10 November 2025;
Published: 24 November 2025.
Edited by:
Komuraiah Myakala, Georgetown University Medical Center, United StatesReviewed by:
Vishal Dubey, Sundyota Numandis Probioceuticals Pvt. Ltd., IndiaCopyright © 2025 Yan, Wang, Chen, Zhao, Zhan, Song, Yang, Liu, Yu and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Peng Liu, ZHJsaXVwZW5nQHNpbmEuY24=; Renhuan Yu, dGV6aG9uZ2V5dUB2aXAuc2luYS5jb20=; Gang Wang, MTMyNzc5NTgyMDVAMTYzLmNvbQ==
†These authors share first authorship
 Yan Yan
Yan Yan Wenru Wang
Wenru Wang Yao Chen2†
Yao Chen2† Keqin Zhao
Keqin Zhao Peng Liu
Peng Liu Renhuan Yu
Renhuan Yu