- 1Graduate School of Biomedical Sciences, Icahn School of Medicine at Mount Sinai, New York, NY, United States
- 2Khoury College of Computer Sciences, Northeastern University, Boston, MA, United States
- 3Molecular Pathology Unit, Massachusetts General Hospital, Charlestown, MA, United States
- 4Center for Cancer Research, Massachusetts General Hospital, Charlestown, MA, United States
- 5Department of Pathology, Harvard Medical School, Boston, MA, United States
- 6Broad Institute of Harvard and MIT, Cambridge, MA, United States
- 7Department of Chemistry and Chemical Biology, Harvard University, Cambridge, MA, United States
- 8Winsor School, Boston, MA, United States
Single-cell assays have transformed our ability to model heterogeneity within cell populations. As these assays have advanced in their ability to measure various aspects of molecular processes in cells, computational methods to analyze and meaningfully visualize such data have required matched innovation. Independently, Virtual Reality (VR) has recently emerged as a powerful technology to dynamically explore complex data and shows promise for adaptation to challenges in single-cell data visualization. However, adopting VR for single-cell data visualization has thus far been hindered by expensive prerequisite hardware or advanced data preprocessing skills. To address current shortcomings, we present singlecellVR, a user-friendly web application for visualizing single-cell data, designed for cheap and easily available virtual reality hardware (e.g., Google Cardboard, ∼$8). singlecellVR can visualize data from a variety of sequencing-based technologies including transcriptomic, epigenomic, and proteomic data as well as combinations thereof. Analysis modalities supported include approaches to clustering as well as trajectory inference and visualization of dynamical changes discovered through modelling RNA velocity. We provide a companion software package, scvr to streamline data conversion from the most widely-adopted single-cell analysis tools as well as a growing database of pre-analyzed datasets to which users can contribute.
1 Introduction
Characterization of cell type, while once dominated by pathological description, has over the past decade shifted towards a more quantitative and molecular approach. As such, molecular measurements in single cells have emerged as the centerpiece of the current paradigm of mechanistic biological investigation (Trapnell, 2015). Technological advancements have enabled researchers to measure all aspects of the central dogma of molecular biology at the single-cell level (Stuart and Satija, 2019). Single-cell RNA sequencing (scRNA-seq), a technique that profiles the relative expression of genes in individual cells and single-cell Assay for Transposase Accessible Chromatin using sequencing (scATAC-seq), a technique that surveys genome-wide chromatin accessibility are the most well-established and widely-used of these methods (Buenrostro et al., 2015; Lähnemann et al., 2020). In fact, combined scRNA-seq + scATAC-seq assays are now routine (Perkel, 2021). Additionally, assays to profile DNA methylation (Luo et al., 2018) or protein levels are now maturing and becoming more widely-accessible (Specht et al., 2019; Labib and Kelley, 2020). Most recently, combinations of various data modalities can now routinely be collected in parallel from the same cell (Chen S. et al., 2019; Zhu et al., 2019; Ma et al., 2020; Xing et al., 2020; Swanson et al., 2021).
scRNA-seq experiments generate on the order of millions of sequencing reads that sample the relative expression of approximately 20,000–30,000 transcribed features (e.g., genes) in each cell of the sample. Normalized read counts for each feature can then be compared to discern differences between cells. scATAC-seq samples comprise a larger feature space wherein cells are characterized by the genomic coordinates of chromatin accessible regions and sequence-features derived from these regions (e.g. transcription factor motifs, k-mer frequencies, etc.). Initially performed in dozens to hundreds of cells, these experiments are now performed on the order of millions of cells. With a high dimensional feature space as a result of thousands of features being considered for each cell and large (in cell number) experiments, analysis methods for this data have been required to advance concurrently with the development of these technologies (Chen et al., 2019c; Tian et al., 2019).
With the exception of proof-of-concept methods still too nascent to be widely applied (Chen et al., 2021), omics measurements of single-cells are generally destructive, preventing measurement of a cell at more than a single time point. As a result, most single-cell measurements for studying dynamic processes are of a “snapshot” nature, imposing inherent limitations on the study of such processes from this data (Weinreb et al., 2018). In light of this, transcription rates can be informative of ongoing processes in cells. The recent advent of RNA velocity quantifies and models the ratios of spliced and unspliced RNA (mRNA and pre-mRNA, respectively) such that they indicate the temporal derivative of gene expression patterns and thereby reflect dynamic cellular processes, allowing predictions of past and future cell states (La Manno et al., 2018).
Among others, PCA, t-SNE, and UMAP are dimensional reduction methods that have become common choices for enabling the visualization of high-dimensional single-cell datasets. Dimensionally reduced datasets are plotted such that similar cells cluster together and those with highly differing features are likewise clustered apart. In addition to the visualization and clustering of cells, trajectory inference methods have been proposed to learn a latent topological structure to reconstruct the putative time-ordering (pseudotime) by which cells may progress along a dynamic biological process (Saelens et al., 2018). As single-cell technologies have advanced, techniques to cluster and organize cells based on single-cell assays have advanced alongside them, allowing key insights toward cell type and state characterization. Combined with RNA velocity information, trajectory inference can offer key insights on dynamical changes to cell states. Once in press however, representation of these dimensionally-reduced visualizations is limited to just two or three dimensions. Even using three-dimensional plots from published studies, one cannot dynamically adjust or rotate the visualization to better understand the data from another angle. In addition, cells are typically annotated by features (e.g. time points, cell type or clusters) to investigate stratification along an axis of some biological process. To change the annotations presented in publication, one must often reprocess the raw data, which is time- and skill-intensive, highlighting the need for more dynamical visualization tools. While such current data representations are often limited and static, single-cell omic datasets are information-rich and, in many cases, important biological heterogeneity cannot be easily investigated or visualized outside the scope of the original publication, without spending considerable cost and time to reanalyze the datasets from scratch.
VR visualization methods for single cell data have been recently proposed (Yang et al., 2018; Legetth et al., 2019; Bressan et al., 2021). However, these methods require either expensive hardware or specific data inputs that mandate intermediate to advanced computational skills. Thus, tools and clear protocols are required to enable researchers, especially those who are not able to efficiently reprocess the raw data, to explore the richness of published datasets (or their own unpublished data) through a simple, easy and affordable VR platform. Importantly, this platform must be flexible enough to accept all types of omics data from established and emerging technologies and processing tools currently employed by the single-cell community.
At the time of this writing, three non-peer-reviewed methods employing VR technology that produce two- and three-dimensional visualizations of single-cell data have recently been reported. CellexalVR enables the visualization of standard scRNA-seq data though requires users to preprocess their data through scripting (Legetth et al., 2019). Unfortunately, this tool also requires expensive and dedicated VR hardware to operate. Another recent method for visualizing single-cell data in VR is Theia (Bressan et al., 2021), which has been designed with a focus on the exploration of spatial datasets for both RNA and protein measurements. Similar to CellexalVR, expensive computing power and VR hardware required to use Theia creates a barrier to entry. An alternative to these high-performance methods for VR visualization of single-cell data is starmap (Yang et al., 2018), which allows the use of inexpensive cardboard visor hardware. However, starmap lacks the advanced portability of outputs from commonly-used scRNA-seq analysis tools and limits cell annotation to clustering results of transcriptomic data. Of note, there are currently no peer-reviewed tools available for the visualization of single-cell data in VR illustrating the novelty in this area of research. To overcome the limitations of these existing methods as well as build on their qualities and initial progress, we present singlecellVR, an interactive web application, which implements a flexible, innovative visualization for various modalities of single-cell data built on VR technology. singlecellVR supports clustering, trajectory inference and abstract graph analysis for transcriptomic as well as epigenomic and proteomic single cell data. Importantly, singlecellVR supports visualization of cell dynamics as described by RNA velocity, a recent milestone in the sequence-based analysis of single cells (La Manno et al., 2018; Bergen et al., 2020). singlecellVR is a browser-contained, free, and open-access tool. Notably, we have developed a one-command conversion tool, scvr to directly prepare the results of commonly-used single-cell analysis tools for visualization using singlecellVR.
2 Results
2.1 SinglecellVR User Experience and Overview
SinglecellVR is an easy-to-use web platform and database that can be operated from inexpensive, cardboard visor hardware that costs as little as ∼$8 and is available online from popular vendors including Google and Amazon. The webpage, available at http://www.singlecellvr.com enables users to explore several preloaded datasets or upload their own datasets for VR visualization. Visualization can be done either on a personal computer or smartphone. To facilitate the transition between the personal computer browser view and the phone-enabled VR visor (VR mode), we have implemented an easy way to transition between these two visualizations as described in the next sections. In VR mode an interactive visualization is presented to the user, allowing them to manipulate and visualize single-cell data using an array of annotations through the cardboard visor. Additionally, singlecellVR features the ability to receive as inputs, the standard output files of commonly-used tools for standard single-cell analysis: Seurat (Hao et al., 2021), Scanpy (along with EpiScanpy) (Wolf et al., 2018; Danese et al., 2019), STREAM (Chen et al., 2019a), PAGA (Wolf et al., 2019), and scVelo (Bergen et al., 2020). A companion package, scvr enables the conversion of these standard outputs to VR-compatible objects in a single command.
In the sections below, we will describe the basis for this VR visualization platform as well as provide descriptive examples of the visualization that can be performed using singlecellVR. We will compare singlecellVR to existing methods and describe its unique advantages that build on the early progress of single-cell data visualization in VR. We include a detailed protocol and quick-start guide that describe how the web platform enables researchers to explore their own data and dually functions as a database for preformatted datasets that can be explored immediately in VR (Supplementary Note 1).
2.2 VR Database and scvr Preprocessing Tool
SinglecellVR provides a growing database of several datasets processed for VR visualization. Initialization and future growth of this database is enabled, scale-free through the streamlined scvr utility. As shown in Figure 1, to use singlecellVR, the user may select a precomputed dataset or convert their data from commonly used single-cell workflows. This conversion can be easily accomplished by using scvr, a simple one-line command tool for performing data conversion and produces a simple zipped .json file with all the information required for visualizing cells and their annotations in VR. Additionally, datasets for which RNA velocity information has been calculated may be submitted directly for visualization of velocity in VR without prior conversion (Supplementary Note 2; Supplementary Notebook 4).

FIGURE 1. An overview of the singlecellVR user experience. Top, grey: The outputs of a standard 2-dimensional scRNA-seq analysis. Middle and bottom, purple: a step-by-step overview of the singlecellVR workflow: 1 Schematic of flexible data conversion. One command to install (via the Python pip package manager) and one command to convert the data to be VR-compatible. 2.Webpage for uploading and exploring VR data. 3 VR mode visualization using a cheap smartphone enabled headset.
Conversion from the standard output of any single-cell analysis tool to this format would normally pose a significant methodological roadblock to most users, especially non-computational biologists. To bridge this gap, scvr parses and converts the outputs of Scanpy, EpiScanpy, Seurat, PAGA, and STREAM (respectively .loom, .h5ad and .pkl) and creates the required zipped .json file (Supplementary Note 2). This file contains the 3-D coordinates of cells in a specified space (e.g. UMAP, LLE, etc.), cell annotations (e.g. FACS-sorting labels, clustering solutions, sampling time or pseudotime, etc.), and feature quantification (gene expression levels, transcription factor deviation, etc.). It also contains the graph structure (the coordinates of nodes and edges) obtained from supported trajectory inference methods. Users interested in visualizing scRNA-seq dynamics using RNA velocity generated from spliced and unspliced read counts can likewise prepare this information for visualization in singlecellVR using the scvr companion utility. Users can follow established workflows for obtaining these insights from the raw read file inputs as well as make use of the tutorials available at the singlecellVR GitHub Repository (Section 5; Supplementary Notebook 4).
Importantly, scvr has been made available as a Python pip package to streamline its installation and can convert a processed dataset for VR visualization with a simple command. To install, one can simply open their command line utility and run: “pip install scvr.” Once installation is completed, the user can navigate to https://github.com/pinellolab/singlecellvr, to copy and customize the example commands provided to execute the one-step process for converting their data to a VR-compatible format. In addition to the documentation of scvr we have filmed a short video tutorial found on the homepage of singlecellVR to further assist less experienced users in preparing their data for visualization.
To showcase the functionality and generalizability of scvr across data types, we have preprocessed a collection of 17 published datasets, which includes both scRNA-seq as well as scATAC-seq and single cell proteomic data and made them available for immediate VR visualization. Taken together we believe this step addresses a key limitation of previously-developed VR tools mentioned above, and a formal comparison is presented in Section 2.5 (Yang et al., 2018; Legetth et al., 2019; Bressan et al., 2021).
Excitingly, given the small footprint of the files obtained with scvr, we are offering users the ability to easily submit their processed data to the singlecellVR GitHub Repository (see Supplementary Figure S1) to make the tool a general resource for the field. In this way, we hope to even further extend the ability of biologists to visualize once static datasets and easily generate new hypotheses through manipulation of a large number of rich datasets. Therefore, we envision that our website will function as a repository for VR visualization data of single cell biological annotations.
2.3 A Simple, Cloud-Based Web Tool for VR Visualization
SinglecellVR is available as a webapp at http://www.singlecellvr.com. This website enables users to explore several preloaded datasets or upload their own datasets for VR visualization. To build singlecellVR we have adopted recent web technologies, Dash by Plotly and A-FRAME, a recently-developed JavaScript framework for VR/AR. This allowed us to create a tool that is portable and does not require any installation. The website can be reached through any web browser and browser compatibility was tested against Google Chrome, Apple Safari, and Mozilla Firefox. Visualization can be done either on a personal computer or smartphone (both Android and Apple smartphones).
Once the users have uploaded their data to singlecellVR, they have the option to view and explore the data in 3-D directly in their web browser or to quickly jettison the data to their mobile device for visualization in a VR headset (Figure 2 and Supplementary Figure S2). A key challenge associated with developing a method for visualization of single-cell data is transporting data that is typically processed in desktop settings to the smartphone-based VR visualization. In fact, we predict that in most cases, users will prefer to upload their data through a computer in which they may have run their analyses. To overcome this challenge and enable a seamless transition to a smartphone for VR view, our website dynamically generates a QR code that enables users to open the VR view on their phone to view data uploaded through a personal computer. This mixed approach is particularly useful because, as mentioned before, most users are not processing single-cell data analysis from a phone nor would they keep the files on a mobile device.

FIGURE 2. Step-by-step protocol for data processing and using singlecellVR. Step 1 Single-cell data can be generated using a variety of technologies or downloaded from online repositories*. Data can then be preprocessed and prepared for use (most often as a feature matrix) with common single-cell analysis tools**. Step 2 Pre-processed data can be analyzed using common single-cell analysis tools (listed here). Step 3 Users can process their data for use with singlecellVR from any of the standard outputs created by analysis tools listed in Step 2. listed at the top in a single command. Step 4 Users can select from pre-processed data or upload their own data (Step 4b) and scan the dynamically generated QR code with their phone to begin the VR visualization (Step 4c). Step 5 Users can use the QR code on the website to transfer their data to their phone for use with simple hardware. * and ** are explained in the Section 5 section, Step 1.
2.4 Supported Tools and Analysis
2.4.1 Visualizing Single-Cell Clustering Solutions in VR
As previously mentioned, Scanpy and Seurat are two commonly-used tools for performing cell clustering as well as differential expression analysis. Here we demonstrate the utility of singlecellVR to visualize the common outputs of these tools, showcasing both the clustering solutions as well as differentially expressed genes or other technical or biological features that are visualized easily through the VR interface (Figure 3). A key advantage of our tool is the ability to supply multiple annotations to cells to visualize various attributes of the measured data, for example based on a biological query of interest or experimental design. This may include stratification by cluster identity, time points, tissues, or FACS-based labels. In Figure 3, we demonstrate the ability to select visualizations by various cluster identifications, which are user-customizable. With the advent of cross-experiment integration methods that can integrate not only multiple scRNA-seq experiments but experiments across modalities of single-cell data collection, this flexible labelling strategy should enable the user in the future to visualize even the most novel and complicated experiments in rich detail.
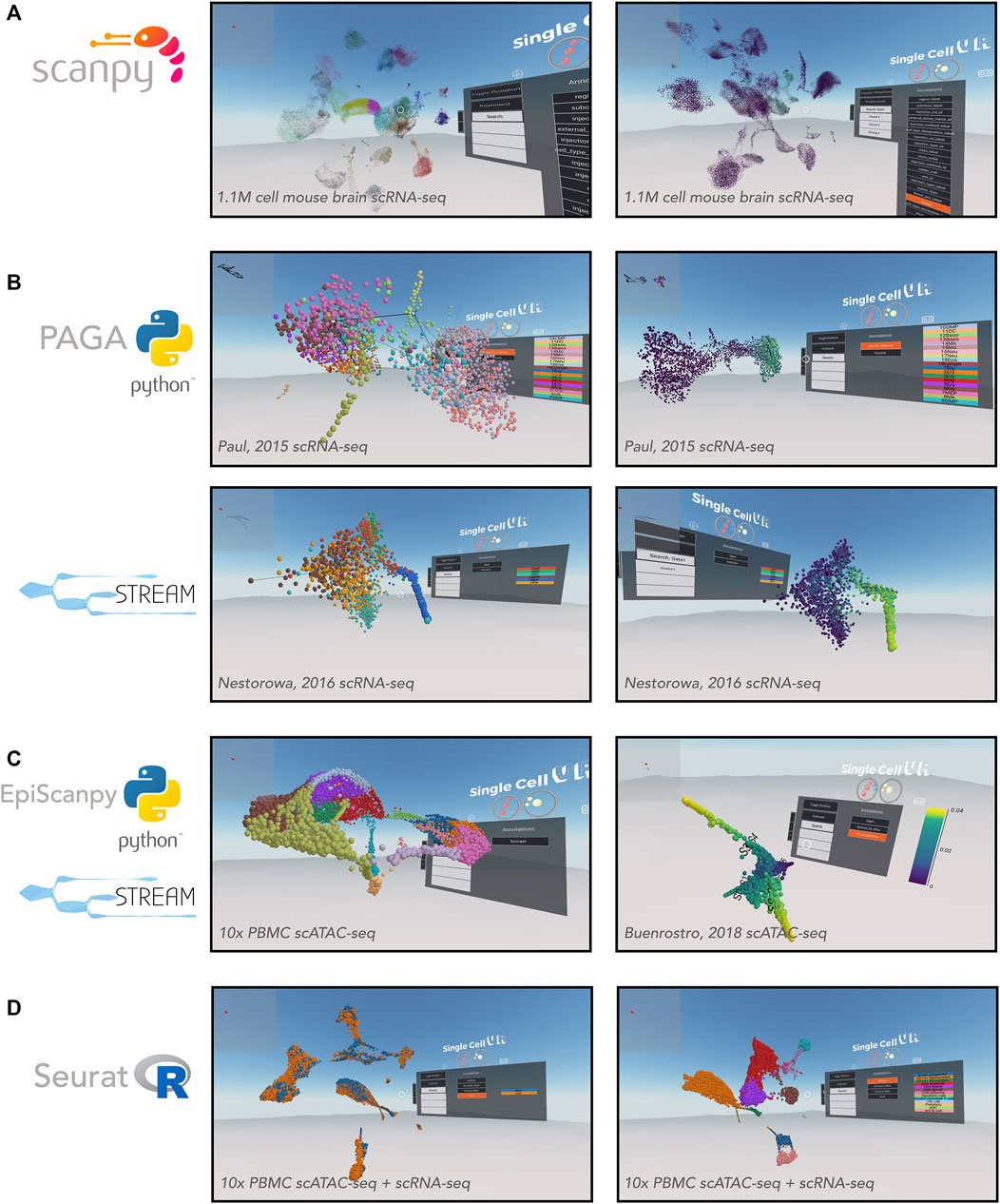
FIGURE 3. VR visualization of single-cell processed datasets profiled by different technologies and analyzed by various computational tools. (A) Scanpy offers solutions for clustering single-cell data. Shown is a UMAP of the Allen Brain Atlas mouse brain scRNA-seq dataset from Yao (2020) and processed by Scanpy. Leiden clustering solution (left) and expression of Gad1 (right). (B) Trajectory inference applications. PAGA offers a partition-based graph abstraction to uncover potential trajectories (edges) between group of cells (nodes) (top-left) relative gene expression (e.g., Klf1, top-right), amongst other annotations. The PAGA-analyzed dataset shown here is from Paul, et al. (2015). STREAM offers the visualization of developmental trajectories, which can be visualized by cell identity (bottom-left) or by relative gene expression (e.g., Gata1, bottom-right), amongst other annotations. The STREAM-analyzed dataset shown here is from Nestorowa, et al. (2016). (C) Epigenomic applications. EpiScanpy enables the clustering and visualization of scATAC-seq data (left). PBMC (healthy donor) 10,000 cells dataset analyzed by EpiScanpy and with colors corresponding to clustering solutions (Louvain clustering). STREAM was used to perform trajectory inference on th scATAC-seq dataset Buenrostro et al. (2018) (right). (D) Seurat offers solutions for clustering single-cell data as well as integrating datasets across experiments. Shown is a Seurat-integrated scRNA-seq and scATAC-seq PBMC dataset from 10x Genomics, colored by technology (left) and cell type (right).
In addition to flexibility for visualizing complex experimental setups, singlecellVR is able to visualize large experiments. To demonstrate this utility, we first processed (using Scanpy and scvr) and visualized on singlecellVR, scRNA-seq data from the Chan-Zuckerberg Biohub Tabula Muris project, a dataset consisting of 44,949 cells and 20 tissues from seven mice (Schaum et al., 2018). In Supplementary Figure S3A, clustering analyses of this dataset are projected into VR, colored by mouse tissue (left) and Louvain cluster identity (right). With a quick rendering time (<1 s) for the Tabula Muris dataset, we next explored the realm of visualization for a modern, large atlas-scale dataset (>1 M cells). Using Scanpy and scvr, we successfully processed and visualized on our website, cells from the Allen Brain Institute that capture cortical and hippocampal development inside the mouse brain (Figure 3A) (Yao, 2020). This dataset consists of 1,093,785 cells and is among the largest scRNA-seq datasets created, to date. Visualization of this dataset in a dynamic VR setting creates the opportunity for more in-depth study of sub-sections of the data, which is particularly valuable for such a large dataset. While the datasets visualized in this manuscript were obtained in their pre-processed state, we have created IPython notebook tutorials for integrating datasets from multiple transcriptomic experiments as is performed in Seurat; these may be accessed in the associated GitHub repository.
2.4.2 Visualizing Single-Cell Trajectory Inference Results in VR
Single-cell measurements are particularly useful for capturing cross-section snapshots of a biological process. With dense cell sampling, one can often observe transient cell states that exist between two, more stable states. However, without an intrinsic understanding of the process being studied, it may be difficult to order these cells along a time axis of a biological process. To enable ordering cells by transcriptional (or epigenomic) states, pseudotemporal ordering, based on trajectory inference and machine learning algorithms has become a useful technique for the single-cell field. Trajectory inference, like clustering, describes a high-dimensional biological process and being limited to a two/three-dimensional static visualization on paper, with a limited selection of genes or annotations is not ideal. Thus, we intend for our tool to leverage the richness of these datasets and make their general usefulness to the field more widespread. We therefore wanted to extend our VR visualization to the results of common trajectory inference tools (Figure 3B). singlecellVR supports two trajectory inference tools: PAGA, a partition-based graph abstraction trajectory inference method and STREAM, a method based on principal graphs (Albergante et al., 2020) that recovers a tree like structure to summarize developmental trajectories and to visualize the relative densities of cell populations along each branch.
To showcase the ability of singlecellVR to visualize trajectory inference results, we reprocessed a popular myeloid and erythroid differentiation dataset (Paul et al., 2015), performing trajectory inference using PAGA. PAGA is designed specifically to preserve relative cell topology in constructing the trajectory along a pseudotime axis. In the depiction of the PAGA-generated trajectory, nodes (gray) correspond to cell groups, and edges (black lines between nodes) connecting the groups quantify their connectivity and confidence (thickness) (Figure 3B, top). To showcase the VR output of STREAM we reprocessed a popular mouse blood dataset (Nestorowa et al., 2016). In STREAM, a set of smooth curves, termed principal graph, are fitted to the data and each curve represents a developmental branch. Within singlecellVR, we are able to easily explore these trajectories and observe qualitatively, the distribution of cells along each branch in the UMAP space (Figure 3B, bottom). The branches of these trajectories are represented by the curves that cut through the cells.
SinglecellVR and scvr also support processing and visualizing single-cell epigenomic data. To demonstrate this functionality, we first used the EpiScanpy workflow to cluster a scATAC-seq dataset from 10x Genomics containing 10,000 cell PBMC (healthy donor) (Figure 3C, left). Next we reprocessed with STREAM a scATAC-seq dataset profiling human hematopoiesis (Buenrostro et al., 2018) (Figure 3C, right). In addition, we extend singlecellVR to single-cell quantitative proteomics data. To this end we reprocessed data from SCoPE2, a recent assay to quantitate proteins in single cells using mass spectrometry (Specht et al., 2019). We performed trajectory inference using STREAM on one SCoPE2 dataset profiling the transition from monocytes to macrophages in the absence of polarizing cytokines. Our analysis revealed a bifurcated branch structure as cells progress towards macrophage phenotypes (Supplementary Figure S3B). Importantly, such bifurcation is not readily visualized in previous reports in two dimensions. Finally, we took advantage of the recent advances in the multi-omics field, using Seurat to integrate and co-embed PBMC cells profiled by scRNA-seq and scATAC-seq by 10x Genomics (Figure 3D).
2.4.3 Visualizing RNA Velocity Analysis in VR
Having successfully applied the singlecellVR framework to the visualization of trajectory inference analyses for multiple modalities of single-cell data, we sought to extend the framework further to visualize dynamical changes at single-cell resolution by way of RNA velocity. We first demonstrated this on a popular endocrine pancreas dataset (Figure 4), which has been previously employed to demonstrate the utility of visualizing dynamic processes using velocity.
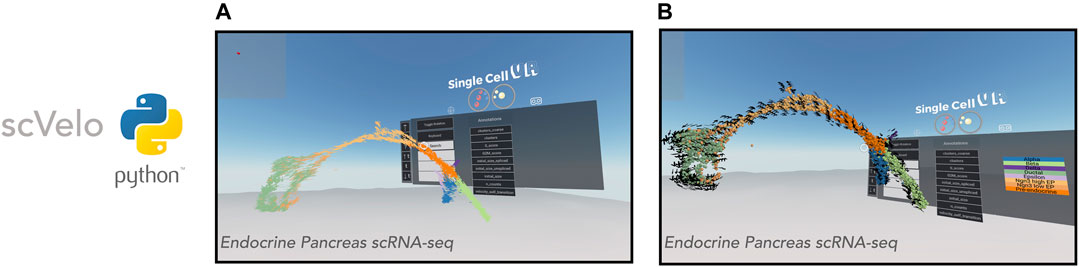
FIGURE 4. VR visualization of single-cell datasets with RNA Velocity. scVelo enables efficient analysis of the RNA velocity attributes of single-cell data. Shown is a 3-D UMAP of an endocrine pancreas dataset (Bastidas-Ponce et al., 2019). (A): Cells are displayed as their corresponding 3-D velocity vectors and colored according to cluster annotation. (B): Cells are displayed as 3-D orbs surrounded by a corresponding grid of velocity vectors. Cells are colored according to cluster annotation.
Visualization of RNA velocity using singlecellVR has two modes. In the default mode, each cell is represented by an arrow where the magnitude and direction of the arrow denote the velocity of that cell (Figure 4A). For larger datasets, cells may be represented as spheres while a surrounding grid system of arrows denotes the predicted trajectory of a given cell (Figure 4B). This is particularly helpful for interpreting the overall direction of cells in various clustering regions or subsets of a given trajectory. In either mode, the arrows are animated to gravitate towards the direction of the corresponding cell trajectory. Latent time, t is a parameter of the velocity calculation for a given cell. To aid in user comprehension of observed velocity, the speed and distance of the animated velocity vector may be calibrated on the fly during the VR experience through adjustment of the t parameter using the floating VR assistance menu. These results taken together with the visualizations of clustering analyses as well as trajectory inference analyses indicate that singlecellVR is a robust, generalizable tool across multiple modalities of single-cell analysis.
2.4.4 Creating Reproducible Visualizations
To enable singlecellVR users to create visualizations that can be reproduced upon sharing, we have included a feature in the VR interface, which captures absolute x, y, and z coordinates such that one may navigate to an identical position with precision. In line with this, we have also included pitch, yaw, and roll descriptions of the camera angle view. These position descriptions of the VR viewpoint can be captured and shared as part of the visualization. Viewpoint descriptions may also be toggled on or off (Supplementary Figure S4).
2.5 Comparison of singlecellVR to Existing Methods
As mentioned above, there are currently three unpublished reports of VR tools created to visualize single-cell data: CellexalVR (Legetth et al., 2019), starmap (Yang et al., 2018), and Theia (Bressan et al., 2021). In this section, we compare these tools to singlecellVR on two axes: 1) ease of use and 2) overall performance for visualization and analysis in VR.
2.5.1 Ease of Use
Competing Tools
Both CellexalVR and Theia require or recommend HTC Vive or HTC Vive Pro VR hardware (∼$500–1,000), an Intel Core i7 processor (∼$300) or better, an NVIDIA GTX1080 or NVIDIA GeForce RTX 3080/3090 (∼$1,500–3,000), 16–32 GB RAM (∼$50–150) and a solid-state hard drive (SSD) (1 TB SSD recommended for CellexalVR) (∼$50–100). Altogether, this equipment requires a minimum investment of roughly $,2300–$4,550. These are computational equipment that most biologists will not have at their disposal within their lab, likely limiting use of this tool to more computationally-focused labs.
CellexalVR requires software and boilerplate-level to pre-process the data in preparation for VR visualization is required and therefore requires the user to perform scripting to prepare data for downstream use with the VR visualization. While Theia has provided a convenient python script to convert AnnData objects, their software is not open-source, hindering further community contribution.
A contrasting alternative to CellexalVR and Theia is Starmap, which is compatible with low-cost hardware such as Google Cardboard. However, Starmap takes as input comma-separated values containing information of the three-dimensional coordinates of cells in the visualization as well as annotations (e.g., cluster ID), and up to 12 features per cell. This file must be prepared entirely by the user without assistance from the Starmap platform, limiting the audience of this tool to experienced computational biologists.
singlecellVR
The single-command companion package for data preparation, scvr described above enables users to visualize their own precomputed data directly from the outputs of commonly-used single-cell RNA-seq analysis tools. Currently supported tools include Scanpy, EpiScanpy, Seurat, PAGA, STREAM, and scVelo. singlecellVR is the only tool of the three discussed (CellexalVR, Theia, and Starmap) that features a QR code to quickly transport the VR data visualization to another device.
2.5.2 VR Performance and Analysis Capabilities
Competing Tools
CellexalVR proposes a versatile, user-friendly visualization for standard scRNA-seq workflow outputs and demonstrates comparable utility on scATAC-seq data. Theia offers a similarly high-performance visualization of single-cell data. Theia’s key distinguishing contribution is it’s visualization of spatial transcriptomic single-cell datasets.
Starmap is only demonstrated on scRNA-seq data and lacks the ability to visualize analyses beyond clustering (such as trajectory inference or an illustration of velocity). Further, Starmap is only capable of displaying up to 12 features for a given cell, limiting the throughput with which users may analyze their data.
singlecellVR
In contrast to existing methods, singlecellVR offers both a high-performance visualization with in-depth analysis and the ability to visualize all modalities of data at scale, while at the same time offering a software that is compatible with low-cost hardware and requires minimal computational abilities. These advances, which build on the progress made by these initial methods create a tool, which offers a low-cost alternative to existing tools with virtually zero barrier to entry, while maintaining high-performance VR visualizations.
3 Discussion
The amount of publicly available scRNA-seq data has exploded in recent years. With new assays to capture chromatin accessibility, DNA methylation and protein levels in single cells, we predict a second wave of dataset generation. Each of these datasets is extremely high-dimensional and thus, rich with latent information about a given biological sample. Ideally, biologists would be able to explore this treasure-trove of data from any angle and make hypotheses assisted by in silico analysis at little to no time cost. Often however, experimental biologists lack the advanced computational skills and/or time required to reprocess and reanalyze raw data from published experiments to gain an understanding of the data from their desired angle of interest. Additionally, biologists who wish to thoroughly explore data prior to publication may rely on a computational specialist who is less connected to the biological problem of interest, introducing a disconnect in hypothesis-driven experimental turnover.
While once primarily reserved for entertainment, VR has found utility in both industrial and academic applications. In this manuscript we present a protocol for visualizing single-cell data in VR. This protocol is based on singlecellVR, a VR-based visualization platform for single cell data and discusses its innovations and differences with existing methods. Importantly, we provide a simple mechanism to prepare results from commonly-used single-cell analysis tools for VR visualization with a single command to considerably increase accessibility (see Section 5). With this added utility, we seek to empower non-computational biologists to explore their data and employ rapid hypothesis testing that could not be made from the traditional static representations typical of communication in a scientific report on paper or a computer screen.
We anticipate that VR will become increasingly useful as a research and education tool and that the construction of software libraries will aid such advancements. VR has also recently found application in other sources of biological data, including single-neuron morphological imaging data (Wang et al., 2019), three-dimensional confocal microscopy data for fluorescent molecule localization (i.e., fluorophore-tagged proteins) within cells (Stefani et al., 2018), and three-dimensional single-molecule localization super-resolution microscopy (Spark et al., 2020). Our scalable and flexible VR visualization framework is not limited to scRNA-seq and it can be also easily adapted to other single-cell assays and tools that already support epigenomic data and/or single-cell proteomic data (EpiScanpy (Danese et al., 2019), Seurat (Stuart et al., 2019), and STREAM (Chen et al., 2019a)). Finally, we extend our framework to computational methods that derive the RNA velocity of single cells for visualization in VR (La Manno et al., 2018; Bergen et al., 2020). With the recent advances in spatially-resolved transcriptomics (Welch et al., 2019) and corresponding analysis methods (Hao et al., 2021; Miller et al., 2021), visualization of such data has already been extended to a VR framework (Bressan et al., 2021). We believe this new sort of three-dimensional VR will also become especially useful once made available to the general research community via inexpensive hardware and facile data preprocessing and preparation for VR visualization. As software to analyze single cells reach their maturity, one could imagine the incorporation of such visualizations into more clinically translatable settings, such as medical devices.
4 Conclusion
This manuscript presents singlecellVR, a scalable web platform for the VR visualization of single-cell data and its associated preprocessing software, scvr, which streamlines the results of commonly used single-cell workflows for visualization in VR. singlecellVR enables any researcher to easily visualize single-cell data in VR. The platform is user-friendly, requires no advanced technical skills or dedicated hardware. Importantly, we have curated and preprocessed several recent single-cell datasets from key studies across various modalities of data generation and analysis approaches, providing the scientific community with an important resource from which they may readily explore and extract biological insight.
4.1 Key Points
• singlecellVR is a web platform that enables quick and easy visualization of single-cell data in virtual reality. This is highlighted by a database of pre-loaded datasets ready for exploration at a single click or via a QR code to quickly jettison the visualization to a smartphone enabled VR visor.
• scvr is a companion package to easily convert standard outputs of common single-cell tools in a single command
• singlecellVR is made for use with cheap and easily-available VR hardware such as Google Cardboard (∼$8).
• singlecellVR can visualize both clustering solutions as well as trajectory inference models of single-cell data for transcriptomic, epigenomic, and proteomic data as well as multi-modally integrated datasets. Additionally, singlecellVR offers a three-dimensional VR visualization of RNA velocity dynamics.
5 Materials and Methods
5.1 Single-Cell Data Preparation
All datasets were processed using Scanpy (version 1.5.1, RRID:SCR_018139), AnnData (version 0.7.6, RRID:SCR_018209), EpiScanpy (version 0.1.8), Seurat (version 3.1.5, RRID:SCR_007322), PAGA (part of Scanpy, version 1.5.1, RRID:SCR_018139), STREAM (version 1.0), and scVelo (version 0.2.3, RRID:SCR_018168) following their documentations. Jupyter notebooks to reproduce data processing are available at https://github.com/pinellolab/singlecellvr. Analyses were performed on a 2019 MacBook Pro (2.4 GHz Intel Core i9, 16 GB RAM).
5.2 Preparation of Processed Data for Visualization in VR
The preprocessing package, scvr generates a series of .json files containing the spatial coordinates representative of cell embeddings in 3D embedding (e.g. PCA, UMAP, etc.) and information including labels and features (e.g., gene expression, TF motif deviation, etc.). These .json files are zipped upon output from scvr into a single file that can be easily uploaded to singlecellVR for visualization.
5.3 SinglecellVR Webapp Construction
To build singlecellVR, we used A-FRAME (version 1.2.0), Dash by Plotly (version 1.13.3).
Data Availability Statement
The source code and the supporting data for this study are available online on GitHub at https://github.com/pinellolab/singlecellvr. The preprocessing package, scvr is included within that repository https://pypi.org/project/scvr/. The documentation for scvr is available here: https://github.com/pinellolab/singlecellvr. Video tutorials for learning about and running visualization experiments with singlecellVR (and using scvr to prepare the data) are available on YouTube, here: https://www.youtube.com/playlist?list=PLXqLNtGqlbeMaAuiBStnBzUNE6a-ULYx8. All the analyses in this article can be reproduced using the Jupyter notebooks available at https://github.com/pinellolab/singlecellvr. Additionally, we have provided a wiki within the same repository for a more detailed guide to reproducing results from the paper as they pertain to the supplementary materials.
Author Contributions
Authors DFS, HC, MEV, and QQ contributed equally to this publication. DFS, HC, MEV, and LP conceived this project and designed the experiment, which was begun at the 2019 HackSeq, at the University of British Columbia where input from collaborators mentioned in the acknowledgements was received. DFS, HC, MEV, and RDC processed the data hosted in the database as well as produced the VR image demonstrations of singlecellVR shown in this manuscript. DFS led the development of the virtual reality framework. HC led the development of Dash-based website and the preprocessing module, scvr. QQ contributed to extending the VR tool with velocity and new API. QZ contributed to extending the VR tool to incorporate single-cell protein analysis. MEV led the preparation of the manuscript. All authors performed user-testing of the software. LP supervised the development of this work and provided guidance. All authors wrote and approved the final manuscript.
Funding
This project has been made possible in part by grant number 2019-202669 from the Chan Zuckerberg Foundation. LP is also partially supported by the National Human Genome Research Institute (NHGRI) Career Development Award (R00HG008399) and Genomic Innovator Award (R35HG010717). MEV is supported by the National Cancer Institute (NCI) Ruth L. Kirschstein NRSA Individual Predoctoral Fellowship (1F31CA257625-01).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
We would like to acknowledge the organizers and participants of HackSeq19 (University of British Columbia), where this project began. We would like to especially acknowledge those HackSeq19 participants that contributed to the development of this project: Michelle Crown, Alexander Dungate, David Lin, Terry Lin, and Sepand Dyanatkar Motaghed. Additionally, we would like to thank Ashley Browne, Salma Ibrahim, and Kate McCurley for their contributions to the construction of the singlecellVR database through the Winsor Science Internship Program.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fgene.2021.764170/full#supplementary-material
Supplementary Figure 1 | Instructions for contributing VR-processed data to the singlecellVR data repository. Users can contribute to the growing repository of VR datasets by submitting a pull request to our GitHub repository: https://github.com/pinellolab/singlecellvr. To do so, first fork and clone the repository (steps 1 and 2, above). Next, add your data (step 3). Finally, create a pull request (step 4) to be submitted for approval. Once approved, your data will be incorporated into the growing repository of VR datasets. It is necessary to add the “VR Dataset” flag (purple, already added to the sidebar) to the pull request. In addition, we ask users to describe the data, methods used and available annotations (e.g. genes, timepoints, clusters labels etc.) in the commit message or comment section of the pull request. Note: for velocity results, files >50 MB are too large to be shared through GitHub and must be shared via other channels. However, coordination of this sharing may proceed through GitHub as shown in this figure. For more, see Supplementary Note 2 and Supplementary Notebook 4.
Supplementary Figure 2 | Tips for using the VR interface. (A) It is not required but one can easily connect a keyboard with your smartphone using a Bluetooth-enabled keyboard (a small portable keyboard can be purchased from Amazon for ∼$10). However, you can still use a normal computer with your browser and explore using your mouse and keyboard, the three-dimensional transcriptional space with cells, trajectories and graph abstractions. The full set of interactive keyboard functionalities are detailed above. (B) There are several similar versions of cardboard VR adapters available for ∼$8. Many VR headsets such as Google Cardboard have a single button that allows a user to click the screen of their phone while immersed in a virtual reality experience. By holding down this button, users without a keyboard may move forward in the direction of their gaze. You can also simply use your computer screen to do initial exploration of the data in 2-D. (C) Users may navigate the VR visualization via a combination of gaze controls and keyboard inputs. A circle, centered in the user’s field of view indicates the direction that a user will move through the virtual space and also acts as the appendage through which the user will interact with objects in the visualization. Additionally, users may select the “keyboard” button on the menu to render a virtual keyboard. Cardboard users may use this keyboard to search for available features to render on the display. The “Enter/Return” key on the virtual keyboard clears the current search. Subsequently selecting the “keyboard” button will hide the keyboard from view.
Supplemental Figure 3 | (A) Rendering the single-cell virtual reality visualization. Scanpy offers tools for clustering, which can be visualized using singlecellVR. Cells can be visualized and colored by various annotations. Shown: mouse tissue type, (left) or their cluster ID (right). The Scanpy-analyzed dataset shown here is from the Chan Zuckerberg Initiative’s Tabula Muris dataset (Schaum et al., 2018). (B) STREAM-processed single-cell proteomics data from SCoPE2 (Specht et al., 2019). These visualizations are an example of an advantage gained by trajectory analysis and three-dimensional visualization. 3-D UMAP plots (ordered left to right, top to bottom) generated by STREAM, respectively colored by pseudotime progression, cell type (orange: monocyte, blue: macrophage), expression of Safb, and expression of Pfn1.
Supplementary Figure 4 | Camera coordinates and angle descriptions enable reproducible visualizations. Shown is a UMAP of the Allen Brain Atlas mouse brain scRNA-seq dataset Yao, et al., 2020 (Yao, 2020) and processed by Scanpy colored by the Leiden clustering solution. Close-ups of the coordinates as well as the toggle for displaying coordinates are shown.
References
Albergante, L., Mirkes, E., Bac, J., Chen, H., Martin, A., Faure, L., et al. (2020). Robust and Scalable Learning of Complex Intrinsic Dataset Geometry via ElPiGraph. Entropy 22, 296. doi:10.3390/e22030296
Bastidas-Ponce, A., Tritschler, S., Dony, L., Scheibner, K., Tarquis-Medina, M., Salinno, C., et al. (2019). Massive Single-Cell mRNA Profiling Reveals a Detailed Roadmap for Pancreatic Endocrinogenesis. Development 146. doi:10.1242/DEV.173849
Bergen, V., Lange, M., Peidli, S., Wolf, F. A., and Theis, F. J. (2020). Generalizing RNA Velocity to Transient Cell States through Dynamical Modeling. Nat. Biotechnol. 38, 1408–1414. doi:10.1038/s41587-020-0591-3
Bressan, D., Mulvey, C. M., Qosaj, F., Becker, R., Grimaldi, F., Coffey, S., et al. (2021). Exploration and Analysis of Molecularly Annotated, 3D Models of Breast Cancer at Single-Cell Resolution Using Virtual Reality. bioRxiv 06, 448342. doi:10.1101/2021.06.28.448342
Buenrostro, J. D., Corces, M. R., Lareau, C. A., Wu, B., Schep, A. N., Aryee, M. J., et al. (2018). Integrated Single-Cell Analysis Maps the Continuous Regulatory Landscape of Human Hematopoietic Differentiation. Cell 173, 1535–1548. doi:10.1016/j.cell.2018.03.074
Buenrostro, J. D., Wu, B., Litzenburger, U. M., Ruff, D., Gonzales, M. L., Snyder, M. P., et al. (2015). Single-cell Chromatin Accessibility Reveals Principles of Regulatory Variation. Nature 523, 486–490. doi:10.1038/nature14590
Chen, H., Albergante, L., Hsu, J. Y., Lareau, C. A., Lo Bosco, G., Guan, J., et al. (2019a). Single-cell Trajectories Reconstruction, Exploration and Mapping of Omics Data with STREAM. Nat. Commun. 10. doi:10.1038/s41467-019-09670-4
Chen, H., Lareau, C., Andreani, T., Vinyard, M. E., Garcia, S. P., Clement, K., et al. (2019c). Assessment of Computational Methods for the Analysis of Single-Cell ATAC-Seq Data. Genome Biol. 20. doi:10.1186/s13059-019-1854-5
Chen, S., Lake, B. B., and Zhang, K. (2019d). High-throughput Sequencing of the Transcriptome and Chromatin Accessibility in the Same Cell. Nat. Biotechnol. 37, 1452–1457. doi:10.1038/s41587-019-0290-0
Chen, W., Guillaume-Gentil, O., Dainese, R., Rainer, P. Y., Zachara, M., Gäbelein, C. G., et al. (2021). Genome-wide Molecular Recording Using Live-Seq. bioRxiv 03, 436752. doi:10.1101/2021.03.24.436752
Danese, A., Richter, M. L., Fischer, D. S., Theis, F. J., and Colomé-Tatché, M. (2019). EpiScanpy: Integrated Single-Cell Epigenomic Analysis. bioRxiv. doi:10.1101/648097
Hao, Y., Hao, S., Andersen-Nissen, E., Mauck, W. M., Zheng, S., Butler, A., et al. (2021). Integrated Analysis of Multimodal Single-Cell Data. Cell 184, 3573–3587. doi:10.1016/J.CELL.2021.04.048
La Manno, G., Soldatov, R., Zeisel, A., Braun, E., Hochgerner, H., Petukhov, V., et al. (2018). RNA Velocity of Single Cells. Nature 560, 494–498. doi:10.1038/s41586-018-0414-6
Labib, M., and Kelley, S. O. (2020). Single-cell Analysis Targeting the Proteome. Nat. Rev. Chem. 4, 143–158. doi:10.1038/s41570-020-0162-7
Lähnemann, D., Köster, J., Szczurek, E., McCarthy, D. J., Hicks, S. C., Robinson, M. D., et al. (2020). Eleven Grand Challenges in Single-Cell Data Science. Genome Biol. doi:10.1186/s13059-020-1926-6
Legetth, O., Rodhe, J., Lang, S., Dhapola, P., Pålsson, J., Wallergård, M., et al. (2019). CellexalVR: A Virtual Reality Platform to Visualise and Analyse Single-Cell Data. bioRxiv. doi:10.1101/329102
Luo, C., Hajkova, P., and Ecker, J. R. (2018). Dynamic DNA Methylation: In the Right Place at the Right Time. Science 361, 1336–1340. doi:10.1126/science.aat6806
Ma, S., Zhang, B., LaFave, L. M., Earl, A. S., Chiang, Z., Hu, Y., et al. (2020). Chromatin Potential Identified by Shared Single-Cell Profiling of RNA and Chromatin. Cell 183, 1103–1116. doi:10.1016/J.CELL.2020.09.056
Miller, B. F., Bambah-Mukku, D., Dulac, C., Zhuang, X., and Fan, J. (2021). Characterizing Spatial Gene Expression Heterogeneity in Spatially Resolved Single-Cell Transcriptomic Data with Nonuniform Cellular Densities. Genome Res. 271288, 120. doi:10.1101/GR.271288.120
Nestorowa, S., Hamey, F. K., Pijuan Sala, B., Diamanti, E., Shepherd, M., Laurenti, E., et al. (2016). A Single-Cell Resolution Map of Mouse Hematopoietic Stem and Progenitor Cell Differentiation. Blood 128, e20–e31. doi:10.1182/blood-2016-05-716480
Paul, F., Arkin, Y. a., Giladi, A., Jaitin, D. A., Kenigsberg, E., Keren-Shaul, H., et al. (2015). Transcriptional Heterogeneity and Lineage Commitment in Myeloid Progenitors. Cell 163, 1663–1677. doi:10.1016/j.cell.2015.11.013
Perkel, J. M. (2021). Single-cell Analysis Enters the Multiomics Age. Nature 595, 614–616. doi:10.1038/D41586-021-01994-W
Saelens, W., Cannoodt, R., Todorov, H., and Saeys, Y. (2018). A Comparison of Single-Cell Trajectory Inference Methods: towards More Accurate and Robust Tools. bioRxiv. doi:10.1101/276907
Schaum, N., Karkanias, J., Neff, N. F., May, A. P., Quake, S. R., Wyss-Coray, T., et al. (2018). Single-cell Transcriptomics of 20 Mouse Organs Creates a Tabula Muris. Nature 562, 367–372. doi:10.1038/s41586-018-0590-4
Spark, A., Kitching, A., Esteban-Ferrer, D., Handa, A., Carr, A. R., Needham, L.-M., et al. (2020). vLUME: 3D Virtual Reality for Single-Molecule Localization Microscopy. Nat. Methods 17, 1097–1099. doi:10.1038/s41592-020-0962-1
Specht, H., Emmott, E., Petelski, A. A., Huffman, R. G., Perlman, D. H., Serra, M., et al. (2019). Single-cell Proteomic and Transcriptomic Analysis of Macrophage Heterogeneity. bioRxiv. doi:10.1101/665307
Stefani, C., Lacy-Hulbert, A., and Skillman, T. (2018). ConfocalVR: Immersive Visualization for Confocal Microscopy. J. Mol. Biol. 430, 4028–4035. doi:10.1016/J.JMB.2018.06.035
Stuart, T., Butler, A., Hoffman, P., Hafemeister, C., Papalexi, E., Mauck, W. M., et al. (2019). Comprehensive Integration of Single-Cell Data. Cell 177, 1888–1902. doi:10.1016/j.cell.2019.05.031
Stuart, T., and Satija, R. (2019). Integrative Single-Cell Analysis. Nat. Rev. Genet. 20, 257–272. doi:10.1038/s41576-019-0093-7
Swanson, E., Lord, C., Reading, J., Heubeck, A. T., Genge, P. C., Thomson, Z., et al. (2021). Simultaneous Trimodal Single-Cell Measurement of Transcripts, Epitopes, and Chromatin Accessibility Using tea-seq. Elife 10. doi:10.7554/ELIFE.63632
Tian, L., Dong, X., Freytag, S., Lê Cao, K.-A., Su, S., JalalAbadi, A., et al. (2019). Benchmarking Single Cell RNA-Sequencing Analysis Pipelines Using Mixture Control Experiments. Nat. Methods 16, 479–487. doi:10.1038/s41592-019-0425-8
Trapnell, C. (2015). Defining Cell Types and States with Single-Cell Genomics. Genome Res. 25, 1491–1498. doi:10.1101/gr.190595.115
Wang, Y., Li, Q., Liu, L., Zhou, Z., Ruan, Z., Kong, L., et al. (2019). TeraVR Empowers Precise Reconstruction of Complete 3-D Neuronal Morphology in the Whole Brain. Nat. Commun. 10, 1–9. doi:10.1038/s41467-019-11443-y
Weinreb, C., Wolock, S., Tusi, B. K., Socolovsky, M., and Klein, A. M. (2018). Fundamental Limits on Dynamic Inference from Single-Cell Snapshots. Proc. Natl. Acad. Sci. USA 115, E2467–E2476. doi:10.1073/pnas.1714723115
Welch, J. D., Kozareva, V., Ferreira, A., Vanderburg, C., Martin, C., and Macosko, E. Z. (2019). Single-Cell Multi-Omic Integration Compares and Contrasts Features of Brain Cell Identity. Cell 177, 1873–1887. doi:10.1016/j.cell.2019.05.006
Wolf, F. A., Angerer, P., and Theis, F. J. (2018). SCANPY: Large-Scale Single-Cell Gene Expression Data Analysis. Genome Biol. 19. doi:10.1186/s13059-017-1382-0
Wolf, F. A., Hamey, F. K., Plass, M., Solana, J., Dahlin, J. S., Göttgens, B., et al. (2019). PAGA: Graph Abstraction Reconciles Clustering with Trajectory Inference through a Topology Preserving Map of Single Cells. Genome Biol. 20. doi:10.1186/s13059-019-1663-x
Xing, Q. R., Farran, C. A. E., Zeng, Y. Y., Yi, Y., Warrier, T., Gautam, P., et al. (2020). Parallel Bimodal Single-Cell Sequencing of Transcriptome and Chromatin Accessibility. Genome Res. 30, 1027–1039. doi:10.1101/GR.257840.119
Yang, A., Yao, Y., Li, J., and Ho, J. W. K. (2018). Starmap: Immersive Visualisation of Single Cell Data Using Smartphone-Enabled Virtual Reality. bioRxiv. doi:10.1101/324855
Yao, Z., Nguyen, T. N., van Velthoven, C. T. J., Goldy, J., Sedeno-Cortes, A. E., Baftizadeh, F., et al. (2020). A Taxonomy of Transcriptomic Cell Types across the Isocortex and Hippocampal Formation. bioRxiv. doi:10.1101/2020.03.30.015214
Keywords: single-cell, scRNA-seq, scATAC-seq, virtual reality, VR, data visualization, clustering, trajectory inference
Citation: Stein DF, Chen H, Vinyard ME, Qin Q, Combs RD, Zhang Q and Pinello L (2021) singlecellVR: Interactive Visualization of Single-Cell Data in Virtual Reality. Front. Genet. 12:764170. doi: 10.3389/fgene.2021.764170
Received: 25 August 2021; Accepted: 27 September 2021;
Published: 28 October 2021.
Edited by:
Guangchuang Yu, Southern Medical University, ChinaReviewed by:
Dake Zhang, Beihang University, ChinaIndu Khatri, Leiden University Medical Center, Netherlands
Copyright © 2021 Stein, Chen, Vinyard, Qin, Combs, Zhang and Pinello. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Luca Pinello, bHBpbmVsbG9AbWdoLmhhcnZhcmQuZWR1
†These authors have contributed equally to this work
 David F. Stein
David F. Stein Huidong Chen3,4,5,6†
Huidong Chen3,4,5,6† Michael E. Vinyard
Michael E. Vinyard Qian Qin
Qian Qin Rebecca D. Combs
Rebecca D. Combs Qian Zhang
Qian Zhang Luca Pinello
Luca Pinello