- School of Basic Medicine, Qingdao University, Qingdao, China
Purpose: To conduct a systematic review and meta-analysis of the available research on evaluating changes in corneal dendritic cell density (CDCD) and the main subbasal nerve parameters (SNPs) on the ocular surface and assessing the diagnostic performance of in vivo confocal microscopy in patients with dry eye disease.
Methods: A computerized systematic review of literature published in PUBMED, EMBASE, Web of Science, Scopus, and the Cochrane Central Register of Controlled Trials until May 8, 2020 was performed. All statistical analyses were conducted in RevMan V.5.3 software. The weighted mean differences (WMDs) and standardized mean differences (SMDs) with 95% confidence intervals (CI) between dry eye patients and healthy subjects were presented as results.
Results: A total of 11 studies with 755 participants were recruited, and 931 eyes were included in this meta-analysis. However, not all studies reported both CDCD and SNPs. CDCD in the central cornea was higher (WMD = 51.06, 95% CI = 39.42–62.71), while corneal nerve fiber density (CNFD) and corneal nerve fiber length (CNFL) were lower (WMD = −7.96, 95% CI = −12.12 to −3.81; SMD = −2.30, 95%CI = −3.26 to −1.35) in dry eye patients in comparison with the corresponding values in healthy controls (all p < 0.00001).
Conclusion: Taken together, while CNFD and CNFL were lower in dry eye patients, central CDCD showed a significant increase in these patients in comparison with the corresponding values in healthy controls.
Introduction
Dry eye disease (DED) is the most common ocular surface disorder, with hundreds of millions of people affected throughout the world. The latest and authoritative definition of DED was proposed by the Tear Film and Ocular Surface Society Dry Eye Workshop II (TFOS DEWS II) in 2017. The TFOS DEWS II defined DED as a multifactorial disease that is characterized by the loss of homeostasis of the tear film with ocular discomfort symptoms that involves various etiological factors, such as tear film instability, hyperosmolarity, ocular surface inflammation, and neurosensory abnormalities (1). Due to population growth and aging, the prevalence of DED is increasing worldwide, and it currently ranges widely from 5 to 50%, depending on the populations assessed (2). DED seemingly occurs more frequently in Asia than in Western countries (2–4), and it has been reported to occur more frequently in the older population and among women (5–7). Corneal nerve alteration and inflammation both play key roles in DED development (8). However, the mechanisms underlying the discomfort and pain caused by inflammation and the nerve damage in the ocular surface in DED remain unclear.
In vivo confocal microscopy (IVCM) is a well-designed and non-invasive approach that allows for observation of the ocular surface structure in vivo (9). IVCM can be categorized into tandem-scanning confocal microscopy, slit-scanning confocal microscopy, and the newly developed laser-scanning confocal microscopy (10). Using IVCM in clinical assessments, changes in neuromorphic and ocular surface inflammation can be detected and imaged quantitatively (11). The Heidelberg Retinal Tomograph with the Rostock Cornea Module (HRT/RCM) (Heidelberg Engineering, Dossenheim, Germany) is the only commercially available laser-scanning confocal microscope, and is used widely in the diagnosis of DED due to the higher-quality images and the ability to perform serial scanning (10). The differences among previous studies were attributed to the use of various types of IVCM systems. Therefore, in this meta-analysis, we selected studies that used HRT/RCM to evaluate corneal parameters. In comparison with other devices and tests, HRT/RCM allows assessment of the corneal pathology at the cellular level (12). Although changes in the corneal parameters in DED patients have been demonstrated in many studies, conflicting results still exist, especially those pertaining to the density of the subbasal nerve plexus (13). Therefore, this meta-analysis aimed to assess the corneal parameters, mainly the subbasal nerve parameters (SNPs) and corneal dendritic cell density (CDCD), and evaluate the performance of IVCM in diagnosing DED by collecting data from different studies.
Methods
Search Strategy
Databases, including Pubmed, Scopus, EMBASE, Web of Science, and Cochrane Central Register of Controlled Trials, were searched up to May 8, 2020. We developed a search strategy based on Pubmed and made the necessary modifications for each database. The following strategy was used in Pubmed: (dry eye OR dry eye syndrome OR dry eye disease OR xerophthalmia OR xeroma OR keratoconjunctivitis sicca OR Sjögren's Syndrome) AND (in vivo confocal microscopy OR confocal microscopy OR IVCM).
Inclusion and Exclusion Criteria
Inclusion criteria were as follows: (1) at least 10 adults with a definite diagnosis of DED in the test group; (2) a healthy population as the control group; (3) reporting central CDCD and/or at least one corneal nerve parameter (corneal nerve fiber density [CNFD], corneal nerve fiber length [CNFL], corneal nerve branch density [CNBD], or tortuosity coefficient [TC]); (4) using HRT/RCM; and (5) published in English. Studies that met any of the following criteria were excluded: (1) inappropriate types of articles, such as review articles, case reports, editorials, conference papers and abstracts, short surveys, or letters; (2) studies including cases of DED and other ocular disorders simultaneously; (3) studies assessing only animals; (4) studies by the same author (studies with more data, or, in cases involving equal data, the most recently published studies were selected); (5) studies with incomplete raw data; or (6) studies reporting interventions on subjects during trials, such as contact lens wearing, surgery, or anti-inflammatory treatments.
Data Extraction
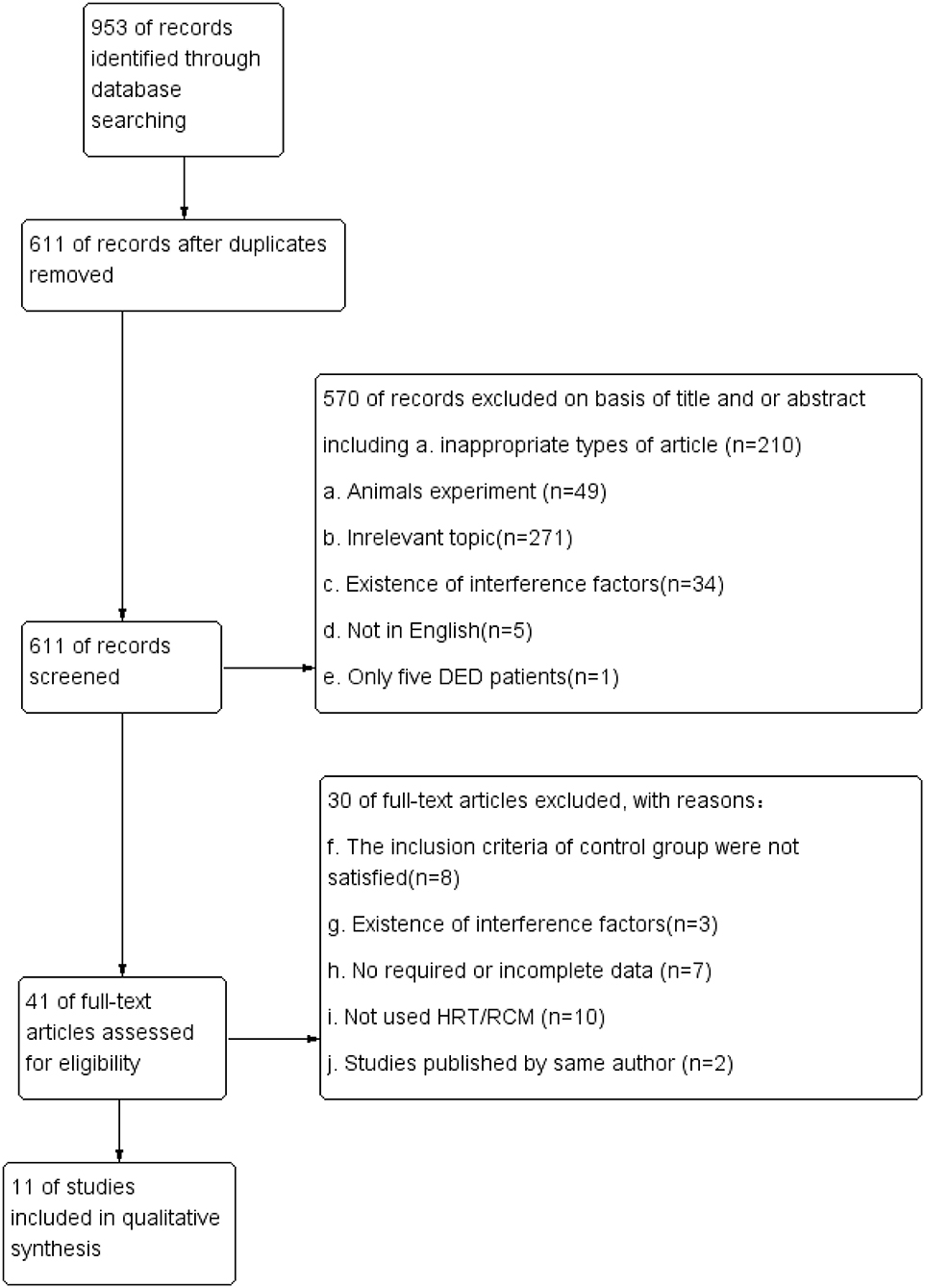
Before the process of screening, all publications searched were exported to Endnote X7. Then, duplicate publications were collated and removed. Two independent reviewers (J.X &Cq.Y) screened eligible titles/abstracts before reading the full article text. Disagreements were resolved via discussion and, if necessary, by consulting a third reviewer (Gh. D). Studies that complied with the inclusion/exclusion criteria were read, and the following information was extracted from the eligible articles: study details (such as the first author's name, year of publication, CDCD, SNPs, and type of IVCM) and patient information (such as mean age, patients' sex, and type of DED). The screening process is summarized in Figure 1, and a-j in the flow diagram describe the screening protocol.
Assessments of Bias Risks
For this study, we assessed these cross-sectional studies using an 11-item checklist recommended by the Agency for Healthcare Research and Quality (14). Article quality was scored as follows: low quality = 0–3, moderate quality = 4–7, and high quality = 8–11. For case-control studies, the Newcastle-Ottawa Scale was used to rate article quality. This scale assesses studies on three parameters, selection, comparability, and exposure, with a maximum score of nine stars. The studies are rated as follows: low quality = 0–5 stars, medium quality = 6–7 stars, and high quality = 8–9 stars (15).
Investigation of Heterogeneity
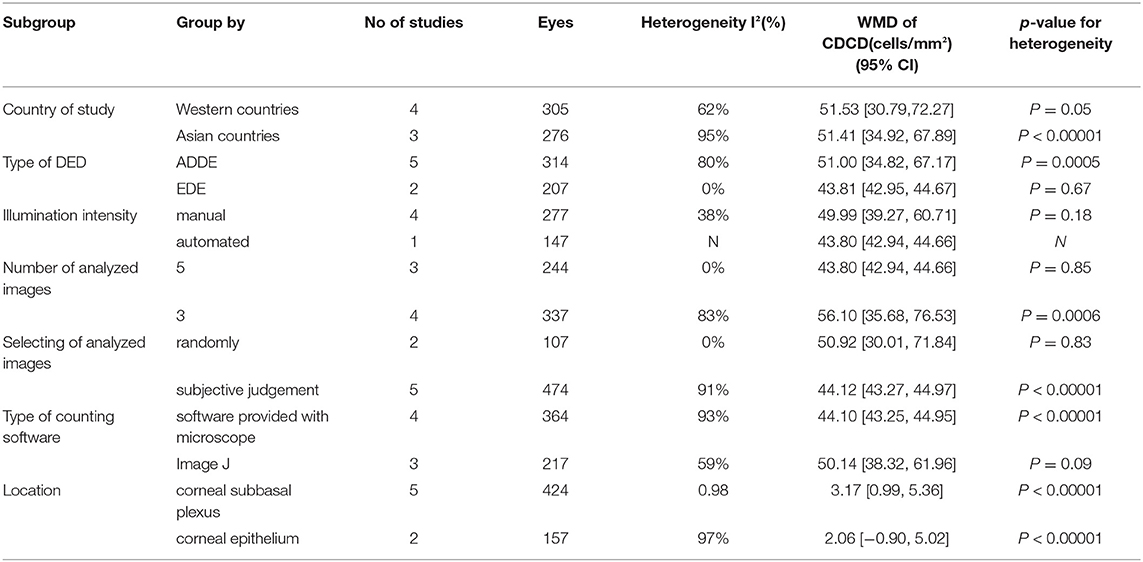
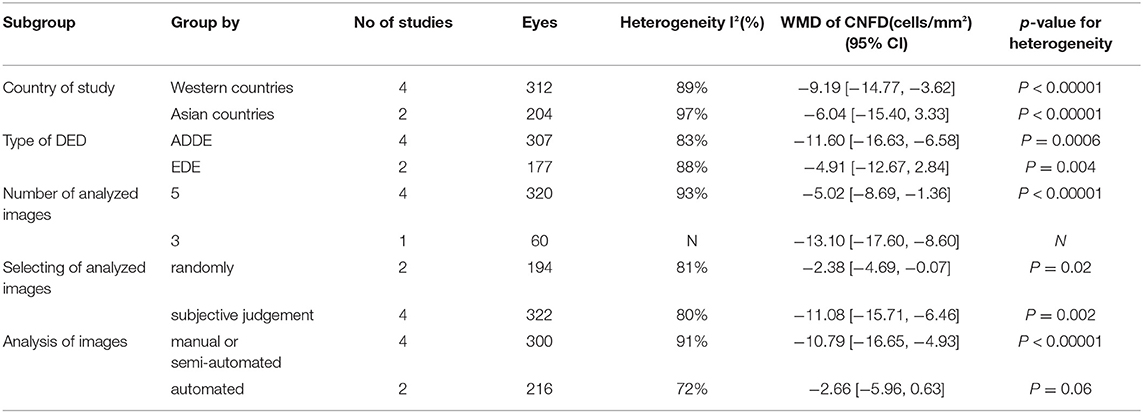
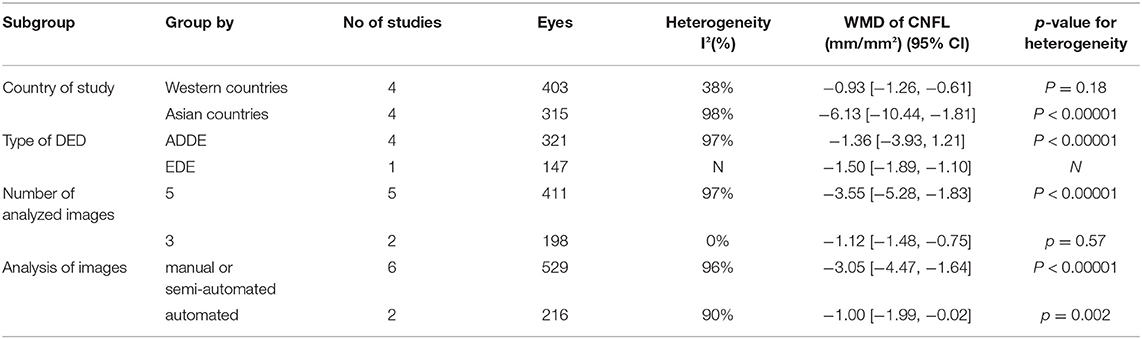
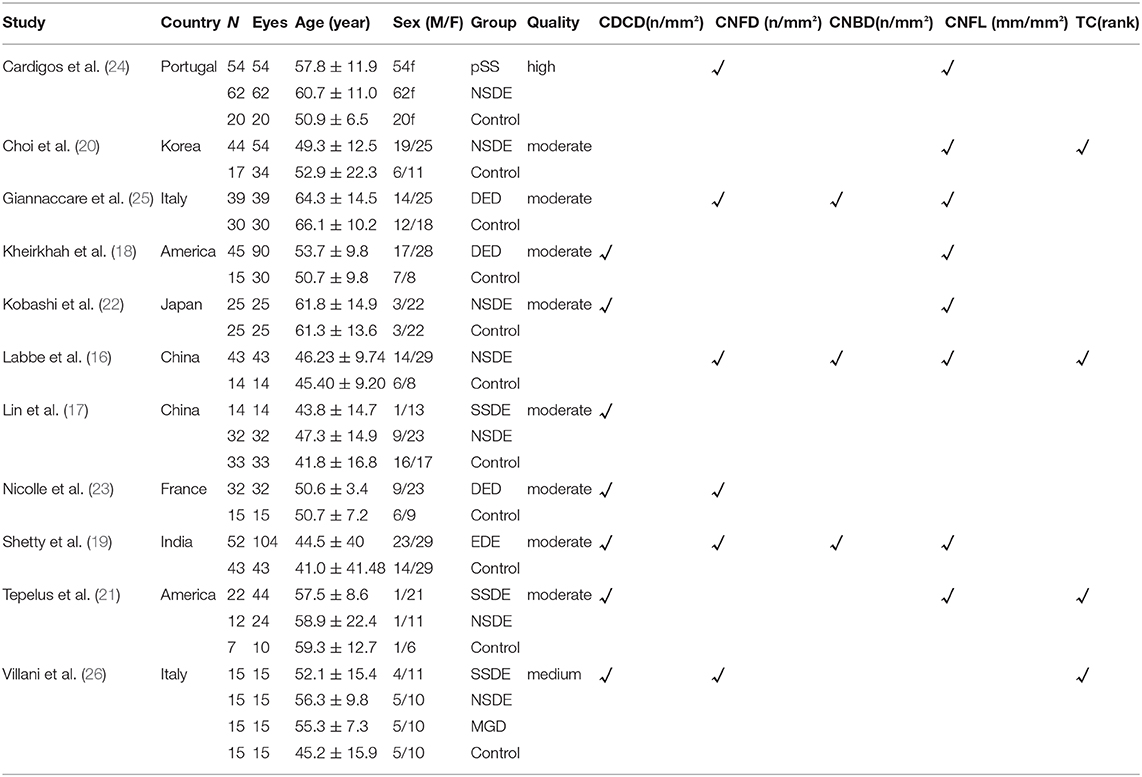
Due to the substantial heterogeneity among the studies, subgroup analysis was conducted to investigate heterogeneity as follows: country of research, type of DED, and IVCM images acquisition and analysis (post-hoc analyses that were not pre-planned). Based on the references included, more details were shown in Tables 1–3.
Statistical Analysis
Review Manager V5.3 (RevMan V.5.3) was used for the meta-analysis. We collected the data for continuous variables; the mean, standard deviation, and sample size were extracted from each study. Weighted mean differences (WMDs) with 95% CI values for continuous variable outcomes were calculated for CDCD and CNFD. However, for CNFL, one set of data (16) was about 1,000 times larger than the others. Measurement methods and units of measurement were checked, and no substantial differences were found. We adopted standardized mean differences (SMDs) because of the greatly different data for CNFL. In some studies, the CNFD was defined as total length of corneal nerve fiber (mm/mm2), whereas in other studies, it was defined as the number of corneal nerve fibers (n/mm2). In order to facilitate comparison, the total corneal nerve length (mm/mm2) was considered as CNFL. Meanwhile, the sum of corneal nerves within a frame, in units of n/mm2, was considered as CNFD. Heterogeneity of the results of the different studies was tested using the I2 value. If I2 > 50% and p < 0.05, significant heterogeneity was indicated statistically. A fixed-effect model was used if I2 < 50%. Conversely, a random-effects model was applied for significant heterogeneity. Because of the limited number of included studies, bias analysis was not performed.
Results
Characteristics of the Eligible Studies
After the screening process, a total of 10 cross-sectional studies (17–25) and one case-control study (26) were included. The 11 studies assessed a total of 755 participants, and 931 eyes met our criteria and were included. The corneal parameters reported by the included studies are shown in Table 4. In the eligible studies, DED patients and healthy controls were matched for age.
Corneal Dendritic Cell Density
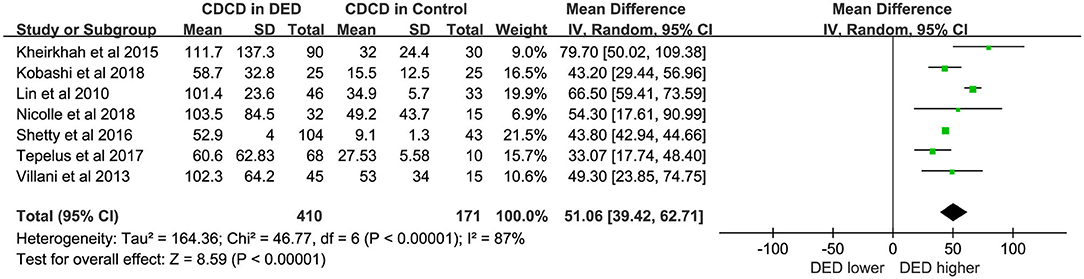
Seven studies (17–19, 21–23, 26) with a total of 581 eyes (DED,410; Controls,171) were included in this meta-analysis. As reported by previous studies (18, 19), corneal dendritic cells were identified as bright single dendritic structures with cell bodies. The CDCD in DED patients was significantly higher than that in the controls (WMD = 51.06, 95% CI 39.42–62.71, p < 0.00001, I2 = 87%). Further details were provided in Figure 2.
Corneal Nerve Fiber Parameters
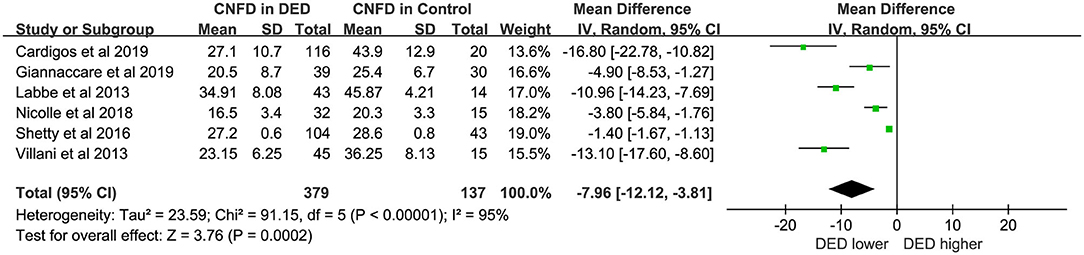
Six studies (16, 19, 23–26) with 516 eyes (DED, 379; Controls, 137) involved examinations of CNFD using HRT/RCM. The difference in CNFD between DED patients and controls showed statistical significance. The DED group showed lower CNFD than controls (WMD = −7.96, 95% CI −12.12 to −3.81, p < 0.00001, I2 = 95%). For additional details, see Figure 3.
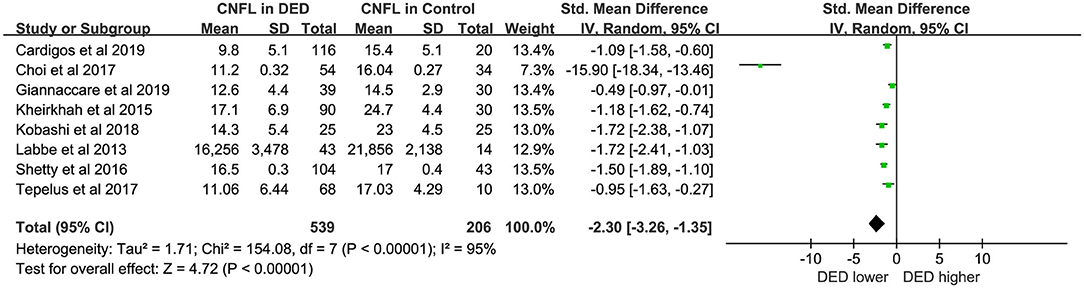
Eight studies (16, 19, 23–25) with a total of 745 eyes (DED, 539; Controls, 206) were included in the meta-analysis for CNFL. The CNFL in DED was marginally lower than that in healthy controls (SMD = −2.30, 95% CI −3.26 to −1.35, p < 0.00001, I2 = 95%). The detailed results can be found in Figure 4.
Subgroup Analysis
Due to the high heterogeneity among studies, we performed the following subgroup analyses:
(1) Type of DED: Patients were divided into those with aqueous-deficient dry eye (ADDE) and evaporative dry eye (EDE).
(2) Country: Patients were divided into those from Western and Asian countries.
(3) IVCM images acquisition and analysis: Based on various conditions and different parameters, we did several subgroup analyses.
In assessments based on the type of DED, the high heterogeneity in subgroups for CNFD and CNFL persisted. In subgroup analyses stratified by country, Western countries showed lower heterogeneity than Asian countries for CDCD (I2 = 62%, p = 0.05 vs. I2 = 95%, p < 0.00001), CNFD (I2 = 91%, p < 0.00001 vs. I2 = 97%, p < 0.00001) and CNFL (I2 = 38%, p = 0.18 vs. I2 = 98%, p < 0.00001). However, the overall model showed no significant difference between subgroups in CDCD (p = 0.99) and CNFD (p = 0.57) without heterogeneity (I2 = 0%). Conversely, there was a subgroup difference in CNFL (I2 = 81.9%, p = 0.02). The details were shown in Tables 1–3.
For the results of IVCM images' acquisition and analysis, manual illumination intensity might be one of source of heterogeneity (I2=38%, p = 0.18). For analyzing CNFD of images, fully automated analysis might have lower heterogeneity (I2 = 72%, p = 0.06). Of note, due to the small amount of literature, most of the heterogeneity was not reduced. Thus, the results with low heterogeneity seemed unreliable.
Quality Assessment
The results of the quality assessment were shown in Table 4. Among the studies included, most of the studies were moderate quality, although some unsure risk of bias still exists. In this regard, we performed subgroup analysis. Further details were shown in Tables 1–3.
Discussion
DED is a typical multifactorial disease with a complex pathophysiology (8). Due to the peculiarity of the cornea, IVCM allows operators to observe corneal nerves and the immune condition in DED and other ocular conditions directly via a non-invasive, quantitative approach. In recent years, growing concern about inflammation and nerve damage has made it important to identify new biomarkers in DED.
Corneal Dendritic Cell Density
The overall results showed significantly increased CDCD in the central corneal region of DED patients (WMD = 51.06, P < 0.00001). However, there was substantial heterogeneity (I2 > 50%). Therefore, we adopted a random effect model, with subgroup analyses performed to explain heterogeneity. For subgroup analyses, the findings indicated no significant difference (p = 0.99) without heterogeneity (I2 = 0%) between subgroups in different countries. Further details were shown in Figure 2 and Table 1. The nationality of patients cannot be considered a source of heterogeneity. Moreover, another study (26) enrolled DED patients, including those with primary Sjögren's syndrome (pSS), non-Sjögren's syndrome dry eye (NSDE), and meibomian gland diseases (MGDs). Thus, the data for the same control groups were used because MGD belongs to EDE, while pSS and NSDE belong to ADDE. However, as previously reported (18), CDCD in ADDE was significantly higher than that in EDE (p = 0.001). In some studies (21, 27), a significant increase was observed in CDCD in Sjögren's syndrome dry eye (SSDE) compared to NSDE. Dendritic cells (DCs) play a key role in pSS (28). Other possible factors may contribute to the strong heterogeneity, such as the definition of DCs, diagnostic criteria of DED, and sex ratio of the subjects. However, due to differences in classifications, we could not evaluate these factors.
In the pathogenesis of dry eye, DCs play an important role in inducing the activation of T cells (29), thus triggering an inflammatory cascade reaction. All the CDCD data included in this study pertained to the central cornea. In one study (17), the data for the center and periphery of the cornea were reported simultaneously. To maintain consistency, we selected the data from the central area of the cornea. The density of corneal epithelial DCs in the periphery and the limbus are reported to be higher than those in the central cornea (30, 31). Animal experiments also confirmed this statement (32). Epithelial DCs are mainly located near the subbasal nerve plexus (11).
Corneal Nerve Parameters
In short, CNFD and CNFL were lower in DED patients compared with healthy controls (WMD = −7.96, p < 0.00001; SMD = −2.30, p < 0.00001). And the forest plots of CNFL and CNFD both showed statistically great heterogeneity between studies (Figures 3, 4). However, in the subgroup analysis of different types of DED, the source of this heterogeneity remained elusive (Tables 2, 3). In subgroup analyses, we hypothesized that the pathogenesis of DED patients in Asian countries may be more complicated than that in Western countries. Moreover, in the present study, the exclusion criteria did not include contact lens wearing. Thus, we could not rule out the possibility of DED caused by contact lens wearing. A previous study revealed that wearing contact lenses led to activation and increase in CDCD, as well as a decrease in the subbasal nerve density, indicating that contact lens wear has an impact on the outcome of DED (33).
Only four studies (16, 20, 21, 26) reported corneal nerve TC, and three of those (16, 19, 25) reported CNBD. Therefore, we did not generate the forest plot. Most of the nerve endings that innervate the cornea are located in the SNPs (34). There is no universally agreed definition for corneal nerve parameters, which is one of reasons why changes in corneal nerve parameters varied between different studies. However, there are conflicting results for the difference in CNFD between DED and controls. Some articles (16, 35) have reported a reduction of CNFD in DED, while others reported no difference in SND (36, 37) or a significant increase in corneal nerve density in DED (37, 38). In addition, some studies defined the total length of the nerve fibers per square millimeter as nerve density (18, 20), while others (25, 39, 40) considered the total number of nerves per square millimeter as CNFD. This may be another reason for the conflicting findings. Moreover, variations in nerve density might affect the periods and severity of DED.
IVCM Images Acquisition and Analysis
For IVCM images acquisition and analysis, although the confocal microscope used in included studies was HRTS, the operating and examination procedure of most studies was subjective. We performed a general quality assessment for the included studies, which indicated IVCM examination might be a major source of risks of bias. The operator selection, image capture, and image analysis were different in different studies. Moreover, the software used to quantify the corneal parameters was not the same. It was reported that CNFL analyzed by manual analysis software (CCMetrics) was higher than using semiautomated or automated software (NeuronJ and ACCMetrics). ACCMetrics was more time-efficient and could provide objective results, since it could distinguish nerve fibers with adjacent pixels by fully automated algorithm (41, 42). Of note, one study (43) has developed a quality evaluation form for the examination of corneal nerve parameters, which is meaningful for future studies. However, the images should be selected randomly to minimize subjective bias. Also, a set of standardized operating procedures should be developed with a unified scanning depth range, a fixed position, and other identical image settings. It might be valuable for future research.
Limitations and Future Directions
The present study has some limitations that should be considered. There was considerable heterogeneity in the included studies, such as methods for measuring parameters, examined location for the cornea, and ethnic variation. It is probably one factor of the risk of bias in assessment of corneal parameters between dry eye patients and healthy subjects. And for some corneal parameters (e.g., branch density, fiber area), we could not conduct a meta-analysis because of incomplete reporting of data. This meta-analysis might be meaningful in assessing corneal pathology in DED and future relevant research. The quantification of CDCD and corneal nerve parameters by IVCM might be valuable for early diagnosis of dry eye, predicting the severity of dry eye, and contributing to clinical evaluation of anti-inflammatory drug efficacy.
Conclusion
In summary, the CDCD and SNPs in DED could be examined by IVCM. However, there is still a lack of gold standard criteria for the definitions of parameters and a complete, objective assessment system. In general, CNFD and CNFL were reduced while CDCD showed a significant increase in DED patients. Moreover, IVCM could provide objective markers for diagnosing DED but was not suitable for indicating the subtype of DED.
Data Availability Statement
The original contributions generated for this study are included in the article/supplementary material, further inquiries can be directed to the corresponding author/s.
Author Contributions
JX and PC designed the program, searched and reviewed the studies, and were in charge of the manuscript. CY and YL assessed the studies, extracted the data, and wrote the part of the manuscript. SH extracted the data and wrote the part of the manuscript. GD directed the project, contributed to the discussion, reviewed, and edited the manuscript. GD had full access to all the data in the study and had final responsibility for the decision to submit for publication. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by the National Natural Science Foundation of China (81670829 and 81970782).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We would like to thank Editage (www.editage.com) for English language editing.
References
1. Craig J, Nichols K, Akpek E, Caffery B, Dua H, Joo C, et al. TFOS DEWS II definition and classification report. Ocular Surf. (2017) 15:276–83. doi: 10.1016/j.jtos.2017.05.008
2. Stapleton F, Alves M, Bunya V, Jalbert I, Lekhanont K, Malet F, et al. TFOS DEWS II epidemiology report. Ocular Surf. (2017) 15:334–65. doi: 10.1016/j.jtos.2017.05.003
3. Hyon J, Yang H, Han S. Association between dry eye disease and psychological stress among paramedical workers in Korea. Sci Rep. (2019) 9:3783. doi: 10.1038/s41598-019-40539-0
4. Siffel C, Hennies N, Joseph C, Lascano V, Horvat P, Scheider M, et al. Burden of dry eye disease in Germany: a retrospective observational study using German claims data. Acta Ophthalmol (Copenh). (2020) 98:e504–12. doi: 10.1111/aos.14300
5. Jie Y, Xu L, Wu Y, Jonas J. Prevalence of dry eye among adult Chinese in the Beijing Eye Study. Eye. (2009) 23:688–93. doi: 10.1038/sj.eye.6703101
6. Han SB, Hyon JY, Woo SJ, Lee JJ, Kim TH, Kim KW. Prevalence of dry eye disease in an elderly korean population. Arch Ophthalmol. (2011) 129:633–8. doi: 10.1001/archophthalmol.2011.78
7. Mai ELC, Lin C-C, Lian I, Liao R, Chen M, Chang C. Population-based study on the epidemiology of dry eye disease and its association with presbyopia and other risk factors. Int Ophthalmol. (2019) 39:2731–9. doi: 10.1007/s10792-019-01117-5
8. Bron AJ, de Paiva CS, Chauhan SK, Bonini S, Gabison EE, Jain S, et al. TFOS DEWS II pathophysiology report. Ocular Surf. (2017) 15:438–510. doi: 10.1016/j.jtos.2017.05.011
9. Zhivov A, Stachs O, Kraak R, Stave J, Guthoff RF. In vivo confocal microscopy of the ocular surface. Ocular Surf. (2006) 4:81–93. doi: 10.1016/S1542-0124(12)70030-7
10. Cruzat A, Qazi Y, Hamrah P. In vivo confocal microscopy of corneal nerves in health and disease. Ocular Surf. (2017) 15:15–47. doi: 10.1016/j.jtos.2016.09.004
11. Cruzat A, Witkin D, Baniasadi N, Zheng L, Ciolino J, Jurkunas U, et al. Inflammation and the nervous system: the connection in the cornea in patients with infectious keratitis. Invest Ophthalmol Vis Sci. (2011) 52:5136–43. doi: 10.1167/iovs.10-7048
12. Lee OL, Tepelus TC, Huang J, Irvine AG, Irvine C, Chiu GB, et al. Evaluation of the corneal epithelium in non-Sjogren's and Sjogren's dry eyes: an in vivo confocal microscopy study using HRT III RCM. BMC Ophthalmol. (2018) 18:309. doi: 10.1186/s12886-018-0971-3
13. Belmonte C, Nichols JJ, Cox SM, Brock JA, Begley CG, Bereiter DA, et al. TFOS DEWS II pain and sensation report. Ocular Surf. (2017) 15:404–37. doi: 10.1016/j.jtos.2017.05.002
14. Rostom A, Dubé C, Cranney A, Saloojee N, Sy R, Garritty C, et al. Celiac Disease. Rockville (MD): Agency for Healthcare Research and Quality (US) (2004). (Evidence Reports/Technology Assessments, No. 104.) Appendix D. Quality Assessment Forms. Available online at: https://www.ncbi.nlm.nih.gov/books/NBK35156/
15. Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. (2010) 25:603–5. doi: 10.1007/s10654-010-9491-z
16. Labbe A, Liang QF, Wang ZQ, Zhang Y, Xu L, Baudouin C, et al. Corneal nerve structure and function in patients with non-Sjogren dry eye: clinical correlations. Invest Ophthalmol Vis Sci. (2013) 54:5144–50. doi: 10.1167/iovs.13-12370
17. Lin H, Li W, Dong N, Chen WS, Liu J, Chen LL, et al. Changes in corneal epithelial layer inflammatory cells in aqueous tear-deficient dry eye. Invest Ophthalmol Vis Sci. (2010) 51:122–8. doi: 10.1167/iovs.09-3629
18. Kheirkhah A, Saboo US, Abud TB, Dohlman TH, Arnoldner MA, Hamrah P, et al. Reduced corneal endothelial cell density in patients with dry eye disease. Am J Ophthalmol. (2015) 159:1022–6.e1022. doi: 10.1016/j.ajo.2015.03.011
19. Shetty R, Sethu S, Deshmukh R, Deshpande K, Ghosh A, Agrawal A, et al. Corneal dendritic cell density is associated with subbasal nerve plexus features, ocular surface disease index, and serum vitamin D in evaporative dry eye disease. Biomed Res Int. (2016) 2016:4369750. doi: 10.1155/2016/4369750
20. Choi EY, Kang HG, Lee CH, Yeo A, Noh HM, Gu N, et al. Langerhans cells prevent subbasal nerve damage and upregulate neurotrophic factors in dry eye disease. PLoS ONE. (2017) 12:e0176153. doi: 10.1371/journal.pone.0176153
21. Tepelus TC, Chiu GB, Huang JY, Huang P, Sadda SR, Irvine J, et al. Correlation between corneal innervation and inflammation evaluated with confocal microscopy and symptomatology in patients with dry eye syndromes: a preliminary study. Graefes Arch Clin Exp Ophthalmol. (2017) 255:1771–8. doi: 10.1007/s00417-017-3680-3
22. Kobashi H, Kamiya K, Sambe T, Nakagawa R. Factors influencing subjective symptoms in dry eye disease. Int J Ophthalmol. (2018) 11:1926–31. doi: 10.18240/ijo.2018.12.08
23. Nicolle P, Liang H, Reboussin E, Rabut G, Warcoin E, Brignole-Baudouin F, et al. Proinflammatory markers, chemokines, and enkephalin in patients suffering from dry eye disease. Int J Mol Sci. (2018) 19:14. doi: 10.3390/ijms19041221
24. Cardigos J, Barcelos F, Carvalho H, Hipólito D, Crisóstomo S, Vaz-Patto J, et al. Tear meniscus and corneal sub-basal nerve plexus assessment in primary Sjögren syndrome and Sicca syndrome patients. Cornea. (2019) 38:221–8. doi: 10.1097/ICO.0000000000001800
25. Giannaccare G, Pellegrini M, Sebastiani S, Moscardelli F, Versura P, Campos EC. In vivo confocal microscopy morphometric analysis of corneal subbasal nerve plexus in dry eye disease using newly developed fully automated system. Graefes Arch Clin Exp Ophthalmol. (2019) 257:583–9. doi: 10.1007/s00417-018-04225-7
26. Villani E, Magnani F, Viola F, Santaniello A, Scorza R, Nucci P, et al. In vivo confocal evaluation of the ocular surface morpho-functional unit in dry eye. Optom Vis Sci. (2013) 90:576–86. doi: 10.1097/OPX.0b013e318294c184
27. Machetta F, Fea AM, Actis AG, de Sanctis U, Dalmasso P, Grignolo FM. In vivo confocal microscopic evaluation of corneal langerhans cells in dry eye patients. Open Ophthalmol J. (2014) 8:51–9. doi: 10.2174/1874364101408010051
28. Hillen M, Ververs F, Kruize A, Van Roon J. Dendritic cells, T-cells and epithelial cells: a crucial interplay in immunopathology of primary Sjögren's syndrome. Expert Rev Clin Immunol. (2014) 10:521–31. doi: 10.1586/1744666X.2014.878650
29. Maruoka S, Inaba M, Ogata N. Activation of dendritic cells in dry eye mouse model. Invest Ophthalmol Vis Sci. (2018) 59:3269–77. doi: 10.1167/iovs.17-22550
30. Alzahrani Y, Colorado L, Pritchard N, Efron N. Longitudinal changes in Langerhans cell density of the cornea and conjunctiva in contact lens-induced dry eye. Clin Exp Optom. (2017) 100:33–40. doi: 10.1111/cxo.12399
31. Mobeen R, Stapleton F, Chao C, Madigan M, Briggs N, Golebiowski B. Corneal epithelial dendritic cell density in the healthy human cornea: a meta-analysis of in-vivo confocal microscopy data. Ocular Surf. (2019) 17:753–62. doi: 10.1016/j.jtos.2019.07.001
32. Hamrah P, Huq S, Liu Y, Zhang Q, Dana M. Corneal immunity is mediated by heterogeneous population of antigen-presenting cells. J Leukoc Biol. (2003) 74:172–8. doi: 10.1189/jlb.1102544
33. Liu Q, Xu Z, Xu Y, Zhang J, Li Y, Xia J, et al. Changes in corneal dendritic cell and sub-basal nerve in long-term contact lens wearers with dry eye. Eye Contact Lens. (2020) 40:238–44. doi: 10.1097/ICL.0000000000000691
34. Vaishnav YJ, Rucker SA, Saharia K, McNamara NA. Rapid, automated mosaicking of the human corneal subbasal nerve plexus. Biomed Tech. (2017) 62:609–13. doi: 10.1515/bmt-2016-0148
35. Erdélyi B, Kraak R, Zhivov A, Guthoff R, Németh J. In vivo confocal laser scanning microscopy of the cornea in dry eye. Graefes Arch Clin Exp Ophthalmol. (2007) 245:39–44. doi: 10.1007/s00417-006-0375-6
36. Tuominen ISJ, Konttinen YT, Vesaluoma MH, Moilanen JAO, Helinto M, Tervo TMT. Corneal innervation and morphology in primary Sjogren's syndrome. Invest Ophthalmol Vis Sci. (2003) 44:2545–9. doi: 10.1167/iovs.02-1260
37. Hosal BM, Ornek N, Zilelioglu G, Elhan AH. Morphology of corneal nerves and corneal sensation in dry eye: a preliminary study. Eye. (2005) 19:1276–9. doi: 10.1038/sj.eye.6701760
38. Zhang M, Chen J, Luo L, Xiao Q, Sun M, Liu Z. Altered corneal nerves in aqueous tear deficiency viewed by in vivo confocal microscopy. Cornea. (2005) 24:818–24. doi: 10.1097/01.ico.0000154402.01710.95
39. Malik RA, Kallinikos P, Abbott CA, van Schie CHM, Morgan P, Efron N, et al. Corneal confocal microscopy: a non-invasive surrogate of nerve fibre damage and repair in diabetic patients. Diabetologia. (2003) 46:683–8. doi: 10.1007/s00125-003-1086-8
40. Khamar P, Nair AP, Shetty R, Vaidya T, Subramani M, Ponnalagu M, et al. Dysregulated Tear fluid nociception-associated factors, corneal dendritic cell density, and vitamin D levels in evaporative dry eye. Invest Ophthalmol Vis Sci. (2019) 60:2532–42. doi: 10.1167/iovs.19-26914
41. Dehghani C, Pritchard N, Edwards K, Russell AW, Malik RA, Efron N. Fully automated, semiautomated, and manual morphometric analysis of corneal subbasal nerve plexus in individuals with and without diabetes. Cornea. (2014) 33:696–702. doi: 10.1097/ICO.0000000000000152
42. Schaldemose EL, Fontain FI, Karlsson P, Nyengaard JR. Improved sampling and analysis of images in corneal confocal microscopy. J Microsc. (2017) 268:3–12. doi: 10.1111/jmi.12581
Keywords: dry eye, dendritic cell density, subbasal nerve parameters, in vivo confocal microscopy, meta-analaysis
Citation: Xu J, Chen P, Yu C, Liu Y, Hu S and Di G (2021) In vivo Confocal Microscopic Evaluation of Corneal Dendritic Cell Density and Subbasal Nerve Parameters in Dry Eye Patients: A Systematic Review and Meta-analysis. Front. Med. 8:578233. doi: 10.3389/fmed.2021.578233
Received: 01 July 2020; Accepted: 12 March 2021;
Published: 07 April 2021.
Edited by:
Jorge L. Alio, Miguel Hernández University, SpainReviewed by:
Rayaz A. Malik, Weill Cornell Medicine- Qatar, QatarGiuseppe Giannaccare, University of Magna Graecia, Italy
Copyright © 2021 Xu, Chen, Yu, Liu, Hu and Di. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Guohu Di, Z3VvaHVfZGlAMTYzLmNvbQ==
†These authors have contributed equally to this work
 Jing Xu
Jing Xu Peng Chen
Peng Chen Chaoqun Yu
Chaoqun Yu Yaning Liu
Yaning Liu Shaohua Hu
Shaohua Hu Guohu Di
Guohu Di