- 1Experimental Center of Basic Medicine, College of Basic Medical Science, Third Military Medical University, Chongqing, China
- 2Department of Plastic Surgery and Cosmetic Surgery, Daping Hospital, Third Military Medical University, Chongqing, China
Background: Acne is a chronic disorder that affects almost 80% of adolescents and young adults, causing psychological and emotional distress. However, the current treatments for acne are either ineffective or have many side effects. This study was designed to confirm and objectively quantify the effect of a new non-drug combined therapy on acne.
Methods: This study innovatively utilized ultrasound, which enhanced the absorption of aloe vera gel, and soft mask to make a purely physical method without any drugs. In both the treatment group and control group, the number of papules/pustules and the area of hyperpigmented lesions were counted, and a smart mirror intelligent face system was used before and after the combined therapy. Alterations in the skin functional index were recorded and analyzed statistically.
Results: In the treatment group, the combined therapy significantly reduced the number of papules and the area of hyperpigmented lesions and improved skin roughness and local blood circulation. In the control group, there was no obvious improvement over 2 months.
Conclusion: This study suggests that the new non-drug combined therapy significantly improved acne, which provided experimental evidence and treatment guidance for patients with mild to severe acne, especially patients with moderate acne. This new therapy may possibly be an appropriate method for patients who seek topical treatments with mild side effects and low antibiotic resistance rates.
Introduction
Acne is a chronic disorder of pilosebaceous units, which is characterized by comedones, inflammatory papules, pustules, cysts, and nodules, affecting almost 80% of adolescents and young adults (1, 2). Acne lesions can result in post-inflammatory hyperpigmentation (PIH) and scarring (3–5). Inflammation exists throughout the whole cycle of acne lesions, and PIH can cause psychological and emotional distress, leading to a great influence on healthy quality of life in affected individuals (6, 7). The current treatments include topical and systemic medications for moderate to severe acne (8–10). However, their curative effects are limited due to adverse effects and unsatisfactory efficacy. In addition, some medications remain controversial. For example, oral isotretinoin, the current mainstream systemic therapy, has acknowledged adverse reactions, including dizziness, nausea and gastrointestinal reaction. Additionally, its psychological effects and teratogenicity also upset the patients who received the treatment (11, 12). Moreover, the efficacy and evaluation of the combined therapy of photodynamic therapy and minocycline are limited because this treatment was only effective in reducing inflammatory and non-inflammatory lesions without therapeutic effects on PIH and overall skin conditions (13).
In recent years, there have been an increasing number of medicines and cosmetics made from aloe vera gel (AVG), the mucilaginous tissue in the center of the aloe vera leaf (14, 15). Its pharmacological functions include antibacterial, anti-inflammatory effects and wound healing promotion (16–18). A previous study suggested that AVG treatment in combination with tretinoin was effective in reducing non-inflammatory and inflammatory acne lesions. However, its adverse effects were also observed in more than 70% of treated patients, such as scaling, burning, and erythema (19). In addition, it has been reported that the high and low frequencies of ultrasound (US) waves have a role in disaggregating the horny layer and stimulating the absorption of the active compounds (20). This study was designed to evaluate the efficacy of AVG combined with US and external application of soft mask to offer an alternative to patients with mild to severe acne who seek topical treatments with mild side effects and low antibiotic resistance rates.
Materials and Methods
Study Subjects
Sixty-four patients (32 males, 32 females) aged 20–35 years with mild to severe facial acne and PIH were enrolled. All patients were randomly divided into two groups, the control group and treatment group, in this double-blind study. The treatment group contained 40 patients (24 males, 16 females), and the control group contained 24 patients (8 males, 16 females). There was no statistically significant difference in age between the two groups (P > 0.05). In the first week, skin care was carried out once a day. Then, it was changed three times a week for seven weeks. After treatment, the therapeutic efficacy was evaluated.
The inclusion criteria were as follows: (1) Patients who had not received hydroquinone, other depigmenting medication or light treatment at least 1 month before inclusion. (2) Patients with informed consent and active cooperation. (3) Patients without blood system diseases, such as blood clotting disorders. (4) Patients without serious underlying diseases, such as diabetes, hypertension, liver and kidney dysfunction. (5) Patients with normal immune function.
The exclusion criteria were as follows: (1) Patients who were allergic to any tool or drug used in the course of treatment. (2) Patients with incomplete treatment. (3) Patients with serious inflammation. (4) Pregnant women. (5) Patients with mental illness.
Our study was approved by the Ethics Committee of Third Military Medical University and was conducted in conformity with the Declaration of Helsinki. All patients provided written informed consent before enrollment in the study.
Study Contents
The AVG (Perfect, China). The ultrasonic therapeutic apparatus (K-SKIN, 830, China). Powder of soft mask (MedSPA, France). The placebo was composed of a non-drug gel vehicle. In addition, the pimple pin was also used in the study.
Treatment Procedures
In the first week of treatment, the inflammatory mediators of patients were partly cleared up with the pimple pin by an experienced paramedic after cleaning and disinfecting the face with bromogeramine every day, which can avoid the spread of bacterial infection during the treatment effectively. Then, AVG combined with US (20 W, 50 Hz, and 220 V) was applied to the patients' faces for 10–15 min, after which the soft mask (2 mm thickness) was applied to these patients for 20–30 min, and their faces were then cleaned. After the first week, the patients received the combination treatment of US and soft mask three times a week for 7 weeks. The duration of treatment cycle was 8 weeks. The controls only received placebo composed of the non-drug gel vehicle after clearing up with bromogeramine and the pimple pin, and then the faces were cleaned three times a week for 8 weeks.
Efficacy Endpoints
The number of papules/pustules and the area of hyperpigmented lesions were counted as described previously (21), performed by the same investigator for all patients. Photographs were taken before and after treatment. Skin roughness and red area (an indicator reflecting facial blood circulation) were detected within a quantitative framework by a smart mirror intelligent facial detection system (Langdy, LD6021D, China).
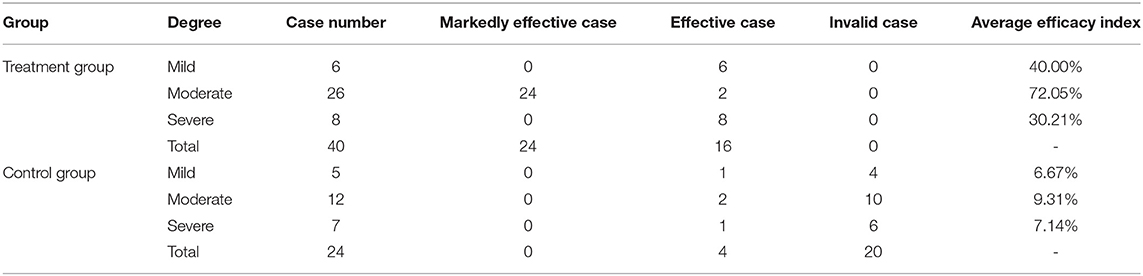
The severity of acne was studied before and after the whole therapeutic session, following more comprehensive evaluation criteria (Table 1) that integrated two systems, including the Investigator's global assessment scale (IGA) for acne severity and the PIH Severity Scale (22). Of note, the number of facial lesions is also very important to assess acne severity quickly. Thus, it was added to the grading score to subdivide the acne severity based upon the modified Pillsbury grading set by the Chinese Medical Association. The acne severity was divided into 4 degrees and 10 grades, and n grades were awarded n scores. After treatment, the severity of acne in the same area was assessed by the same criteria. The score before treatment (sB) and the score after treatment (sA) were recorded and utilized to calculate the efficacy index (EI): EI = (sB-sA)/sB × 100%. The efficacy criteria included four grades: clinical cure (EI > 95%), markedly effective (EI: 60–95%), effective (EI: 20–59%), and invalid (EI < 20%).
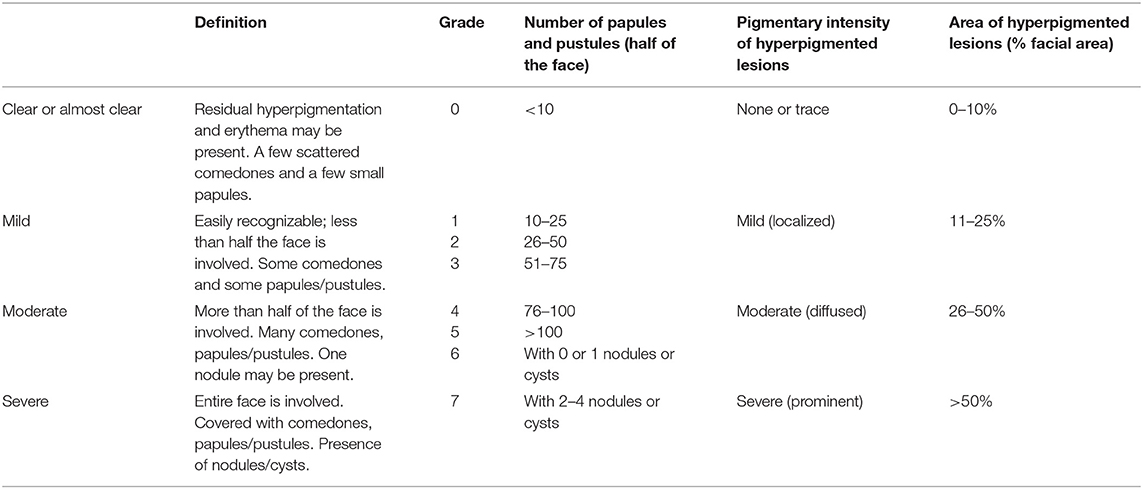
Table 1. Investigator's assessment scale (IGA) for acne severity combined with post-inflammatory hyperpigmentation severity.
Statistical Analysis
The results were analyzed on an intention-to-treat basis, and all statistical analyses were performed with SPSS 24.0 software (SPSS Inc., Chicago, IL, USA). Based on the normal distribution, the data are expressed as the mean ± SD and were analyzed with unpaired t-test between two groups. All statistical tests were two-sided, with a significance level of 0.05.
Results
Comparison of the Average Number of Papules and Hyperpigmented Area Before and After Treatment in the Two Groups
The papules/pustules and pigmentary intensity are the most common symptoms of acne. The results showed that the number of facial papules and hyperpigmented area (%) were 84.50 ± 30.78 and 47.28 ± 19.09 in the treatment group before the combined therapy, respectively. However, the number of facial papules were reduced to 25.53 ± 18.11 (p < 0.01) and the hyperpigmented area (%) was decreased to 16.15 ± 12.25 (p < 0.01) (Figure 1A), after the 2-month combined therapy. The skin conditions were significantly improved with treatment, as the number of facial papules/pustules were significantly decreased, and the area of hyperpigmented lesions was markedly reduced (Figure 2). However, no obvious improvement was observed in the control group after 2-month placebo treatment composed of the non-drug gel vehicle and face cleansing (p > 0.05) (Figure 1B).
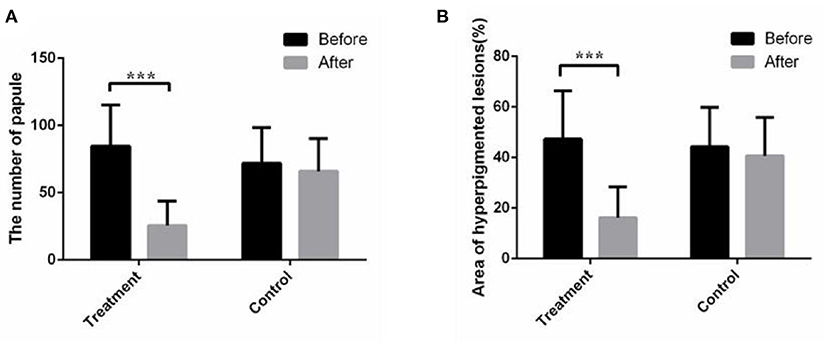
Figure 1. The combined therapy reduced the number of papules and facial hyperpigmented area in the treatment group. (A) Average total number of papules/pustules before and after treatment in the treatment group and control group. (B) Average facial hyperpigmented area (%) before and after treatment in the treatment group and control group. The results are expressed as the mean ± SD. ***p < 0.001.
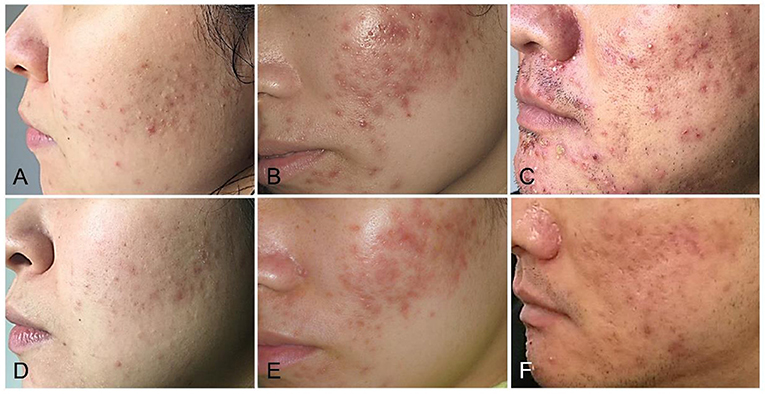
Figure 2. The combined therapy improved the acne condition in patients with mild to severe acne. (A,D) Representative photographs of mild acne patients before and after 2 months of the combined treatment. (B,E) Representative photographs of moderate acne patients before and after 2 months of the combined treatment. (C,F) Representative photographs of severe acne patients before and after 2 months of the combined treatment.
Comparison of Facial Skin Blood Circulation Before and After Treatment in the Two Groups
The distribution of capillaries and the capability of blood circulation are closely related to acne (23). In this study, the proportion of the red area was used to reflect the cutaneous blood circulation detected by intelligent facial detection. The increased red area indicated improved blood circulation. The results showed that the proportion of facial red area increased obviously after 2 months of combined therapy in the treatment group (p < 0.05), while there was no difference in the proportion of facial red area in the control group (Figure 3).
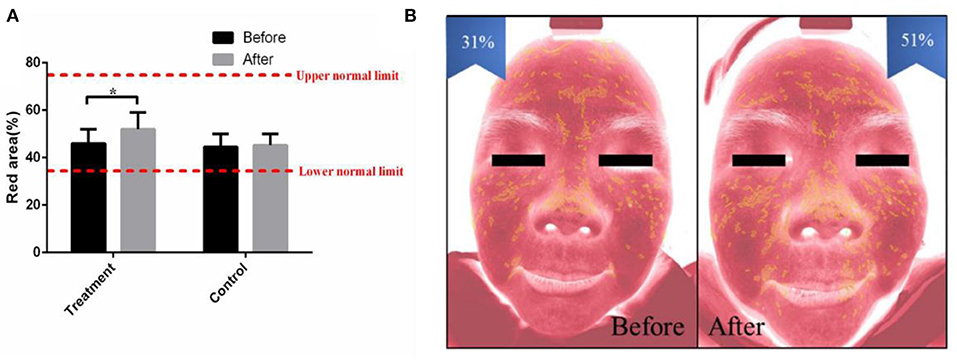
Figure 3. The combined therapy decreased the red area in the treatment group. (A) The proportion of red area before and after treatment in the treatment group and control group. (B) Representative pictures of a 27-year-old female taken by intelligent facial detection in the treatment group, which shows the comparison between the red area values before and after treatment. The results are expressed as the mean ± SD. *p < 0.05.
Comparison of Facial Skin Roughness Before and After Treatment in the Two Groups
Facial skin roughness is based on the percentage of texture thickness, surface protruding area and pore distribution area, which indicates the current facial smoothness (24). Intelligent facial detection showed that the average facial skin roughness (%) of the treatment group was 56.20 ± 5.50 before receiving the combined therapy. After 2 months of combined therapy, the average facial skin roughness (%) was increased to 65.12 ± 6.20 (p < 0.05) (Figure 4A). After the combined treatment, the skin surface became soft and smooth, and the uncomfortableness induced by rough skin was markedly eased (Figure 4B). The facial skin roughness of the control group was similar to that 2 months prior (Figure 4A).
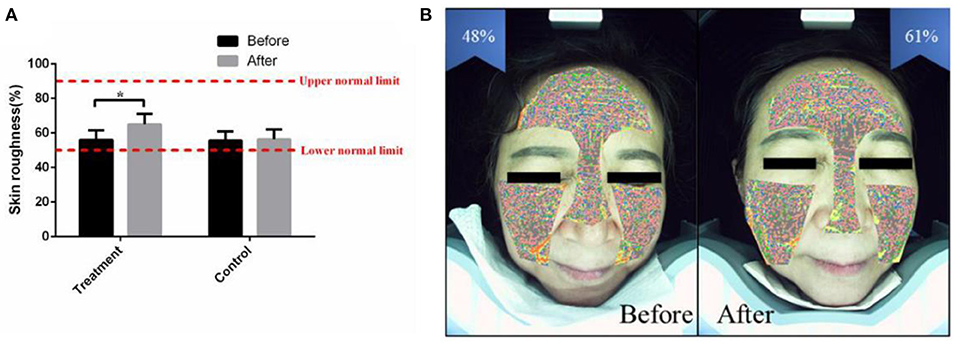
Figure 4. The combined therapy rescued the skin roughness in the treatment group. (A) The average value of facial skin roughness (%) before and after treatment in the treatment group and control group. (B) Representative pictures of a 32-year-old female taken by intelligent facial detection in the treatment group, which shows the comparison between the values of facial skin roughness before and after treatment. The results are expressed as the mean ± SD. *p < 0.05.
Efficacy Evaluation After Treatment in Two Groups
In this study, the skin conditions of 24 patients in the control group and 40 patients in the treatment group before and after treatment were classified and evaluated after 2 months of treatment. The therapeutic efficacy data are shown in Table 2.
The results showed that in the treatment group, the effective rate was 40.00%, and the marked effective rate was 60.00% after 2 months of combined therapy. In the control group, the effective rate was only 16.67%, and the invalid rate was 83.33% (Figure 5A). Of note, the markedly effective rate of the patients with moderate acne in the treatment group was 92.31% (Figure 5B). In addition, the values of the average efficacy index of the patients with mild, moderate and severe acne were 40.00, 72.05, and 30.21%, respectively (Table 2). Of note, the patients with moderate facial acne had the highest average efficacy index, indicating that the combined therapy had its best effect on these patients.
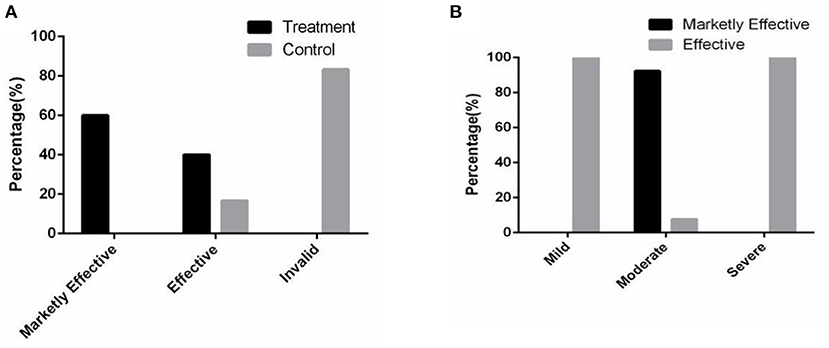
Figure 5. The combined therapy significantly promoted the skin condition in the treatment group. (A) The proportion distribution of the total efficacy in two groups. (B) The proportion distribution of the efficacy in the treatment group.
Safety Evaluation in the Course of Treatment in the Treatment Group-
In this study, no serious side effects occurred during the whole treatment cycle in the treatment group and the control group. In the treatment group, nine patients had mild local reactions, such as itching, local inflammation, and scattered small abscesses. Two patients complained of mild pain at the treatment site. One patient had a small area of erythema in the treatment site, and one patient had a slight increase in facial pigmentation (Figure 6). However, these symptoms were tolerable and had been improved at the end of the treatment. There were no side effects, such as dry skin, chapped skin, gastrointestinal reaction, dizziness, or nausea, which were observed during the process of traditional acne treatment. Due to the properties of pure physical therapy, the possibility of drug resistance, and teratogenicity in the future may be very low.
Discussion
To the best of our knowledge, the therapy that combines grease removal and AVG importing by ultrasonic and soft mask is the first non-drug treatment of acne. The current treatments of acne usually utilize oral and/or topical medications, which are mainly antibiotics and retinoids (8, 25). However, it was widely reported that retinoids treatment led to highly frequent adverse irritation, such as erythema, dryness, stinging, and burning (9, 26). Besides, although skin lesions subside after acne treatment, inflammatory pigmentation easily develops in the later stage of inflammation due to the photosensitivity of these drugs. It extends the time to repair skin significantly and affects the appearance of patients, leading to a great psychological burden to patients. Moreover, many other adverse reactions, including dizziness, nausea, and gastrointestinal reaction, excruciate acne patients in the course of treatment (27, 28). In terms of the application of antibiotics, its extensive use has inevitably resulted in an alarming rise in Propionibacterium acne resistance, thus obviously weakened effects (29). Our observations demonstrated that the new combined therapy had a very significant anti-inflammatory effect, pigmentation reduction and post-inflammatory acne recovery with fewer adverse reactions than previous treatments. In addition, it improved the skin conditions by replenishing cutaneous moisture and smoothing the skin.
At present, researchers have not yet reached a consensus on the severity scale and therapeutic efficacy of acne vulgaris globally. Because IGA for acne severity does not involve facial pigmentation or evaluate the acne condition quantitatively, the PIH severity scale and the number of facial lesions were incorporated into IGA (22). The new evaluation system we proposed is more comprehensive and practical.
Acne is caused by chronic inflammatory lesions of pilosebaceous units. Hyperkeratosis and sebum blocking pores are the earliest changes (30). If irritable free fatty acids are not cleared, they will continue to stimulate hair follicles, leading to facial hyperpigmentation in post-inflammatory acne. Thus, the inflammatory papules and pustules were treated with the pimple pin first to reduce the sustained skin damage caused by the accumulation of sebum. In addition, the repatency of pilosebaceous ducts will benefit AVG permeation. Although the pimple pin may damage part of the skin tissue, slight injury to human skin has no significant effect. Of note, before ultilizing the pimple pin, bromogeramine was used to disinfect the skin without irritation, avoiding the possible infection during the treatment.
As a traditional natural medicine, AVG possesses a variety of biological effects, including anti-inflammatory, antioxidative, and antipigmentation effects, which are beneficial for acne treatment and repair of skin lesions (31, 32). The neutral polysaccharides, aloe mannan and acemannan separated from the gel part of aloe leaves have been identified to have anti-inflammatory and immunosuppressive effects by reducing the adhesion of leukocytes and the level of TNF-α and ameliorating the capability of self-repair by inhibiting thromboxane (an inhibitor of lesion repair) (33–35). The oxidation reaction of skin lesions is also one of the main reasons that prolongs the process of lesion repair. Glutathione peroxide activity, superoxide dismutase enzymes, and phenolic antioxidants were found to be present in AVG, which may be responsible for the antioxidant effects (36). AVG can inhibit tyrosinase and dihydroxyphenylalanine (DOPA), a priming agent in the enzymatic conversion of tyrosine into melanin that reduces pigmentation (37). The muco-polysaccharides along with amino acids and zinc found in aloe vera are capable of maintaining the integrity and moisture of skin, reducing erythema and helping prevent skin ulcers (38, 39). In addition, studies have also shown that AVG is capable of increasing the flexibility of skin and reducing its fragility (40). Furthermore, as a natural substance, AVG is easier to obtain and has fewer side effects than chemical medicines.
To enhance the absorption effect, AVG was imported into the epidermis by ultrasound. The physical and chemical properties of ultrasound were identified as useful for drug depolymerization and lowering viscosity (41). In addition, it was also found that ultrasound could stimulate the decomposition of the stratum corneum and promote the convective motion of intraepidermal molecules (42). Thus, ultrasound can enhance the absorption of drugs, nutrients, and cream. Moreover, the mechanical action and thermal effect of ultrasound are useful to promote cutaneous blood circulation and increase the permeability of the cell membrane, thereby enhancing tissue metabolism and regeneration capability (43). Meanwhile, the microcirculation and lymphatic circulation of skin will be accelerated by ultrasonic irradiation, which improves the absorption and removal of necrotic tissue. Because of the skin's barrier function, the absorption rate of topical medicine is not sufficient. Thus, based on the features of ultrasound, AVG penetrates percutaneously into the lesions more easily to enhance the skin's self-repair capabilities. In addition, there are several effective mechanisms in the treatment of topical lesions, including inhibition of bacterial colonization, subsequent tissue formation, neovascularization, and macrophage stimulation for fibroblast proliferation (44). According to previous reports, various ultrasound frequencies, intensities and treatment durations will result in various effects. The most common ultrasound treatment of inflammatory conditions is low-intensity, high-frequency and pulsed mode for minimizing thermal effects, whereas, the optimal protocol of ultrasound-assisted repair of skin lesions will be further explored in future studies (45).
A moist membrane can be formed on the skin surface by using a soft mask, thereby protecting skin from the external environment and achieving transient softening and fat gettering. In addition, the soft mask has a role in pore cleansing, anti-inflammation and moisture-oil balance. Therefore, using soft mask is not only for a more effective therapeutic effect but also for the prevention of acne regeneration.
It is not the first time to apply AVG or US in the researches related with acne treatment. However, their therapeutic effects were not satisfactory. A study has confirmed that the cream containing propolis, tea tree oil and AVG was effective in reducing acne, however, the role of which was restricted to the decreased number of papules/pustules and erythema without the evaluation of the pigmentation improvement and the overall acne condition (46). In addition, the use of US contributed to the antibacterial effects of lysozyme-shelled microbubbles on inhibiting the inflammation of acne, but the results of which were limited and lacked the comparisons of other important clinical features associated with acne treatment (47). Of note, it has been widely recognized that personal lifestyle and diet affect the development of acne. For example, sleep duration and quality have a reciprocal relationship with acne occurrence and severity (6). A lot of evidence indicated that high glycemic load diet could exacerbate acne (48). Thus, during the treatment, our patients were encouraged to avoid greasy and high glucose diet, wash their faces regularly, maintain a regular schedule, etc., In the absence of these possible influence factors, it is more conducive to assess the curative effect of the combine therapy.
In our study, the combined therapy lasted 2 months followed by a 1-month follow-up period, during which it was observed that the hyperpigmentation condition was further improved in the treatment group, indicating that the 2-month combined therapy conduced to post-inflammatory recovery. However, a limitation of the study is the short-term follow-up period, which should be extended from 1 to 3 months even longer to collect more long-term results and assess the possible recurrence rate.
In summary, the non-drug acne therapy we proposed, which combined grease removal and AVG importation by ultrasonication and soft mask, can accelerate the recovery of post-inflammatory acne. The therapeutic effect is the most prominent for patients with moderate acne. As a non-drug therapy, it has more advantages than traditional therapies, such as more obvious effects, easier operation, and safer experience. Despite the relatively long treatment cycle of the combined therapy, fewer side effects are assured due to its properties of pure physiotherapy. Most people may be more willing to accept safer treatment without sequelae than to replace the current damage with other losses. Therefore, this new treatment is worthy of further promotion and application.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics Statement
The studies involving human participants were reviewed and approved by the Ethics Committee of Army Medical University. The patients/participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author Contributions
HZ carried out the experiments, collected and analyzed the data, and drafted the manuscript. WZ, XS, and XL contributed to acquisition and data analysis. HL and YL designed the experiments, supervised the project, and revised the manuscript. All authors contributed to the article and approved the submitted version.
Funding
This study was supported by Chongqing Normal University Postgraduate's Research and Innovation Project (No. CYS20367).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Abad-Casintahan F, Chow SK, Goh CL, Kubba R, Hayashi N, Noppakun N, et al. Frequency and characteristics of acne-related post-inflammatory hyperpigmentation. J Dermatol. (2016) 43:826–8. doi: 10.1111/1346-8138.13263
2. Connolly D, Vu HL, Mariwalla K, Saedi N. Acne scarring-pathogenesis, evaluation, and treatment options. J Clin Aesthet Dermatol. (2017) 10:12–23. [Epub ahead of print].
3. Yin NC, McMichael AJ. Acne in patients with skin of color: practical management. Am J Clin Dermatol. (2014) 15:7–16. doi: 10.1007/s40257-013-0049-1
4. Mu YZ, Jiang L, Yang H. The efficacy of fractional ablative carbon dioxide laser combined with other therapies in acne scars. Dermatol Ther. (2019) 32:e13084. doi: 10.1111/dth.13084
5. Moradi Tuchayi S, Makrantonaki E, Ganceviciene R, Dessinioti C, Feldman SR, Zoubouli CC. Acne vulgaris. Nat Rev Dis Primers. (2015) 1:15029. doi: 10.1038/nrdp.2015.29
6. Dreno B, Bagatin E, Blume-Peytavi U, Rocha M, Gollnick H. Female type of adult acne: physiological and psychological considerations and management. J Dtsch Dermatol Ges. (2018) 16:1185–94. doi: 10.1111/ddg.13664
7. Davern J, O'Donnell AT. Stigma predicts health-related quality of life impairment, psychological distress, and somatic symptoms in acne sufferers. PLoS ONE. (2018) 13:e0205009. doi: 10.1371/journal.pone.0205009
8. Fox L, Csongradi C, Aucamp M, Plessis J, Gerber M. Treatment modalities for acne. Molecules. (2016) 21:1063. doi: 10.3390/molecules21081063
9. Oge' LK, Broussard A, Marshall MD. Acne vulgaris: diagnosis and treatment. Am Fam Physician. (2019) 100:475–84.
10. Barbieri JS, Spaccarelli N, Margolis DJ, James WD. Approaches to limit systemic antibiotic use in acne: systemic alternatives, emerging topical therapies, dietary modification, and laser and light-based treatments. J Am Acad Dermatol. (2019) 80:538–49. doi: 10.1016/j.jaad.2018.09.055
11. Erdemir U, Okan G, Gungor S, Tekin B, Yildiz SO, Yildiz E. The oral adverse effects of isotretinoin treatment in acne vulgaris patients: a prospective, case-control study. Niger J Clin Pract. (2017) 20:860–6. doi: 10.4103/1119-3077.183248
12. Vallerand IA, Lewinson RT, Farris MS, Sibley CD, Ramien ML, Bulloch AGM, et al. Efficacy and adverse events of oral isotretinoin for acne: a systematic review. Br J Dermatol. (2018) 178:76–85. doi: 10.1111/bjd.15668
13. Xu X, Zheng Y, Zhao Z, Zhang X, Liu P, Li C. Efficacy of photodynamic therapy combined with minocycline for treatment of moderate to severe facial acne vulgaris and influence on quality of life. Medicine. (2017) 96:e9366. doi: 10.1097/MD.0000000000009366
14. Grindlay D, Reynolds T. The aloe vera phenomenon: a review of the properties and modern uses of the leaf parenchyma gel. J Ethnopharmacol. (1986) 16:117–51. doi: 10.1016/0378-8741(86)90085-1
15. Triantafyllidi A, Xanthos T, Papalois A, Triantafillidis JK. Herbal and plant therapy in patients with inflammatory bowel disease. Ann Gastroenterol. (2015) 28:210–20. doi: 10.1155/2015/197167
16. Jain S, Rathod N, Nagi R, Sur J, Laheji A, Gupta N. Antibacterial effect of aloe vera gel against oral pathogens: an in-vitro study. J Clin Diagn Res. (2016) 10:ZC41–4. doi: 10.7860/JCDR/2016/21450.8890
17. Reuter J, Jocher A, Stump J, Grossjohann B, Franke G, Schempp CM. Investigation of the anti-inflammatory potential of aloe vera gel (97.5%) in the ultraviolet erythema test. Skin Pharmacol Physiol. (2008) 21:106–10. doi: 10.1159/000114871
18. Norman G, Christie J, Liu Z, Westby MJ, Jefferies JM, Hudson T, et al. Antiseptics for burns. Cochrane Database Syst Rev. (2017) 7:CD011821. doi: 10.1002/14651858.CD011821.pub2
19. Hajheydari Z, Saeedi M, Morteza-Semnani K, Soltani A. Effect of aloe vera topical gel combined with tretinoin in treatment of mild and moderate acne vulgaris: a randomized, double-blind, prospective trial. J Dermatolog Treat. (2014) 25:123–9. doi: 10.3109/09546634.2013.768328
20. Juhász M, Korta D, Mesinkovska NA. A review of the use of ultrasound for skin tightening, body contouring, and cellulite reduction in dermatology. Dermatolog Surg. (2018) 44:949–63. doi: 10.1097/DSS.0000000000001551
21. Lee HJ, Kim JY, Park KD, Lee WJ. Randomized controlled double-blind study of a cleanser composed of 5-aminolevulinic acid and peptides on mild and moderate acne vulgaris. J Cosmet Dermatol. (2020) 19:1745–50. doi: 10.1111/jocd.13232
22. Callender VD, Young CM, Kindred C, Taylor SC. Efficacy and safety of clindamycin phosphate 1.2% and tretinoin 0.025% gel for the treatment of acne and acne-induced post-inflammatory hyperpigmentation in patients with skin of color. J Clin Aesthet Dermatol. (2012) 5:25–32.
23. Knutsen-Larson S, Dawson AL, Dunnick CA, Dellavalle RP. Acne vulgaris: pathogenesis, treatment, and needs assessment. Dermatol Clin. (2012) 30:99–106, viii–ix. doi: 10.1016/j.det.2011.09.001
24. Kislevitz M, Lu KB, Wamsley C, Hoopman J, Kenkel J, Akgul Y. Novel use of non-invasive devices and microbiopsies to assess facial skin rejuvenation following laser treatment. Lasers Surg Med. (2020) 52:822–30. doi: 10.1002/lsm.23233
25. Stangeland KZ, Huldt-Nystrøm T, Li X, Danielsen K. Treatment of acne. Tidsskr Nor Laegeforen. (2019) 139. doi: 10.4045/tidsskr.18.0946
26. Khalil S, Bardawil T, Stephan C, Darwiche N, Abbas O, Kibbi AG, et al. Retinoids: a Journey from the molecular structures and mechanisms of action to clinical uses in dermatology and adverse effects. J Dermatolog Treat. (2017) 28:684–96. doi: 10.1080/09546634.2017.1309349
27. Reilly BK, Ritsema TS. Managing nonteratogenic adverse reactions to isotretinoin treatment for acne vulgaris. JAAPA. (2015) 28:34–9. doi: 10.1097/01.JAA.0000459815.24908.02
28. Sevimli Dikicier B. Topical treatment of acne vulgaris: efficiency, side effects, and adherence rate. J Int Med Res. (2019) 47:2987–92. doi: 10.1177/0300060519847367
29. Bonati LM, Dover JS. Treating acne with topical antibiotics: current obstacles and the introduction of topical minocycline as a new treatment option. J Drugs Dermatol. (2019) 18:240–4.
30. Bergler-Czop B, Hadasik K, Brzezinska-Wcislo L. Acne inversa: difficulties in diagnostics and therapy. Postepy Dermatol Alergol. (2015) 32:296–301. doi: 10.5114/pdia.2014.44012
31. Cui Y, Ye Q, Wang H, Li Y, Yao W, Qian H. Hepatoprotective potential of aloe vera polysaccharides against chronic alcohol-induced hepatotoxicity in mice. J Sci Food Agric. (2014) 94:1764–71. doi: 10.1002/jsfa.6489
32. Liu C, Cui Y, Pi F, Cheng Y, Guo Y, Qian H. Extraction, purification, structural characteristics, biological activities and pharmacological applications of acemannan, a polysaccharide from aloe vera: a review. Molecules. (2019) 24:1554. doi: 10.3390/molecules24081554
33. Hes M, Dziedzic K, Gorecka D, Jedrusek-Golińska A, Gujska E. Aloe vera (l.) webb.: natural sources of antioxidants - a review. Plant Foods Hum Nutr. (2019) 74:255–65. doi: 10.1007/s11130-019-00747-5
34. Gavriil L, Detopoulou M, Petsini F, Antonopoulou S, Fragopoulou E. Consumption of plant extract supplement reduces platelet activating factor-induced platelet aggregation and increases platelet activating factor catabolism: a randomised, double-blind and placebo-controlled trial. Br J Nutr. (2019) 121:982–91. doi: 10.1017/S0007114519000308
35. Ashouri F, Beyranvand F, Beigi Boroujeni N, Tavafi M, Sheikhian A, Mohammad Varzi A, et al. Macrophage polarization in wound healing: role of aloe vera/chitosan nanohydrogel. Drug Deliv Transl Res. (2019) 9:1027–42. doi: 10.1007/s13346-019-00643-0
36. Sumi FA, Sikder B, Rahman MM, Lubna SR, Ulla A, Hossain MH, et al. Phenolic content analysis of aloe vera gel and evaluation of the effect of aloe gel supplementation on oxidative stress and fibrosis in isoprenaline-administered cardiac damage in rats. Prev Nutr Food Sci. (2019) 24:254–64. doi: 10.3746/pnf.2019.24.3.254
37. Gupta SD, Masakapalli SK. Mushroom tyrosinase inhibition activity of aloe vera l. Gel from different germplasms. Chin J Nat Med. (2013) 11:616–20. doi: 10.1016/S1875-5364(13)60071-0
38. Chrapusta E, Kaminski A, Duchnik K, Bober B, Adamski M, Bialczyk J. Mycosporine-like amino acids: potential health and beauty ingredients. Marine Drugs. (2017) 15:326. doi: 10.3390/md15100326
39. Zhang J, Xu X, Rao NV, Argyle B, McCoard L, Rusho WJ, et al. Novel sulfated polysaccharides disrupt cathelicidins, inhibit rage and reduce cutaneous inflammation in a mouse model of rosacea. PLoS ONE. (2011) 6:e16658. doi: 10.1371/journal.pone.0016658
40. Pereira GG, Guterres SS, Balducci AG, Colombo P, Sonvico F. Polymeric films loaded with vitamin e and aloe vera for topical application in the treatment of burn wounds. Biomed Res Int. (2014) 2014:641590. doi: 10.1155/2014/641590
41. Mirzapour-Kouhdasht A, Sabzipour F, Taghizadeh MS, Moosavi-Nasab M. Physicochemical, rheological, and molecular characterization of colloidal gelatin produced from common carp by-products using microwave and ultrasound-assisted extraction. J Texture Stud. (2019) 50:416–25. doi: 10.1111/jtxs.12408
42. Seah BC, Teo BM. Recent advances in ultrasound-based transdermal drug delivery. Int J Nanomed. (2018) 13:7749–63. doi: 10.2147/IJN.S174759
43. Tiwari SB, Pai RM, Udupa N. Influence of ultrasound on the percutaneous absorption of ketorolac tromethamine in vitro across rat skin. Drug Deliv. (2008) 11:47–51. doi: 10.1080/10717540490265261
44. White J, Ivins N, Wilkes A, Carolan-Rees G, Harding KG. Non-contact low-frequency ultrasound therapy compared with uk standard of care for venous leg ulcers: a single-centre, assessor-blinded, randomised controlled trial. Int Wound J. (2016) 13:833–42. doi: 10.1111/iwj.12389
45. Kwan RL, Lu S, Choi HM, Kloth LC, Cheing GL. Efficacy of biophysical energies on healing of diabetic skin wounds in cell studies and animal experimental models: a systematic review. Int J Mol Sci. (2019) 20:368. doi: 10.3390/ijms20020368
46. Mazzarello V, Donadu MG, Ferrari M, Piga G, Usai D, Zanetti S, et al. Treatment of acne with a combination of propolis, tea tree oil, and Aloe vera compared to erythromycin cream: two double-blind investigations. Clin Pharmacol. (2018) 10:175–81. doi: 10.2147/CPAA.S180474
47. Liao AH, Hung CR, Lin CF, Lin YC, Chen HK. Treatment effects of lysozyme-shelled microbubbles and ultrasound in inflammatory skin disease. Sci Rep. (2017) 7:41325. doi: 10.1038/srep41325
Keywords: acne (acne vulgaris), non-drug therapy, aloe vera gel, ultrasound, soft mask, hyperpigmentation
Citation: Zhong H, Li X, Zhang W, Shen X, Lu Y and Li H (2021) Efficacy of a New Non-drug Acne Therapy: Aloe Vera Gel Combined With Ultrasound and Soft Mask for the Treatment of Mild to Severe Facial Acne. Front. Med. 8:662640. doi: 10.3389/fmed.2021.662640
Received: 01 February 2021; Accepted: 29 April 2021;
Published: 21 May 2021.
Edited by:
Hans Christian Ring, Bispebjerg Hospital, DenmarkReviewed by:
Unni Krishna S. R. L. Samavedam, Incyte Corporation, United StatesIrina Khamaganova, Pirogov Russian National Research Medical University, Russia
Copyright © 2021 Zhong, Li, Zhang, Shen, Lu and Li. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hongli Li, bGlob25nbGlAdG1tdS5lZHUuY24=; Yuangang Lu, c2tpbjUxNUAxNjMuY29t
 Hongyu Zhong1
Hongyu Zhong1 Hongli Li
Hongli Li