- 1Geriatrics Division, Department of Medicine (DIMED), Azienda Ospedale Università di Padova, University of Padova, Padua, Italy
- 2Department of General Psychology (DPG), University of Padova, Padua, Italy
- 3Bluecompanion Ltd, London, United Kingdom
- 4Geriatric Intensive Care Unit, Department of Experimental and Clinical Medicine, University of Firenze, Firenze, Italy
- 5Center for Cognitive Disorders and Dementia - Catanzaro Lido, ASP Catanzaro, Catanzaro, Italy
- 6Scientific Committee of National Association of Third Age Residences (ANASTE) Calabria, Lamezia Terme, Italy
- 7Department of Medical Science, University of Ferrara, Ferrara, Italy
- 8Geriatrics Unit, Department of Clinical and Experimental Medicine, University of Pisa, Pisa, Italy
- 9Acute Geriatric Unit, School of Medicine and Surgery, San Gerardo Hospital, University of Milano-Bicocca, Monza, Italy
- 10Department of Clinical Science, Norway and Diakonhjemmet Hospital, University of Bergen, Oslo, Norway
- 11Unit of Geriatrics, Department of Medicine, Campus Bio-Medico University and Teaching Hospital, Rome, Italy
Objectives: The spread of COVID-19 has undeniably unsettled the social, psychological and emotional life of the entire world population. Particular attention should be paid to older adults with dementia, given their vulnerability to emotional stressors. The aim of this retrospective study is to evaluate the impact of the first wave quarantine related to Covid-19 on psychological and affective well-being of older adults with mild/major neurocognitive disorders and of their caregivers.
Methods: Data on participants' assessment before the quarantine (PREQ) were retrospectively collected. Patients with Mild Cognitive Impairment (MCI) or dementia were recruited from different Centers for Cognitive Decline and Dementia in Italy. During the quarantine, psychological and affective well-being were evaluated by phone through the administrations of scales measuring anxiety and depression (DASS), perceived stress (PSS), coping strategies (COPE) and the caregivers' burden (CBI). The scales' results were compared across participants' PREQ cognitive level (Mini Mental State Examination, MMSE ≥25, 23–24, and ≤ 22) with multiple linear regression models.
Results: The sample included 168 patients (64% women) with a mean age of 79 ± 7 years. After adjusting for potential confounders, more severe cognitive impairment was independently associated with higher DASS and PSS score, and poorer coping strategies (p < 0.05). Cognitive functioning was also inversely associated with CBI.
Conclusions: The impact of the quarantine on the psycho-affective well-being of individuals with MCI and dementia and on caregivers' burden varies according to the PREQ cognitive functioning with more severely impaired patients having worse outcomes.
Key points
• During the pandemic, individuals cognitively more impaired showed more severe depressive and anxious symptoms, compared to those with better cognitive functioning.
• Individuals with greater cognitive impairment showed worse “positive attitude” and “problem orientation” coping strategies, as compared to those with better cognitive functioning.
• Heavier caregiving burden was observed in caregivers of individuals with more severe cognitive impairment.
Introduction
The Italian population, as well as the entire world, is in a delicate historical phase as the spread of novel Coronavirus (COVID-19) is requiring important clinical, social and economic interventions in order to limit escalation of the disease and safeguard individuals' health. Quarantine, social distancing and community containment are the public health measures that have been adopted to isolate people and prevent person-to-person transmission of the disease (1). If, on the one hand, these measures are fundamental to reduce the transmission of COVID-19 and its serious consequences on people's health, on the other hand they may have significant negative sequelae. An increasing number of studies are documenting the impact of COVID-19 itself and of the forced social isolation on psychological well-being (2–4). In this context, it has been recently highlighted the importance of drawing attention also to the psychological consequences of the pandemic on older adults. As at the date worldwide acknowledged, scrupulous consideration should be given to older adults, who represent the section of the population with the highest rate of mortality linked to this virus (5). More specifically, attention should be made to the “frailest among the frail,” the individuals with dementia (6). Different data on the psychological and/or behavioral effects of COVID-19 on individuals with dementia have been reported so far (3, 7). Nonetheless, some questions remain unanswered: do people with Mild Cognitive Impairment (MCI) and dementia experience anxiety and depression because of the COVID-19 pandemic? If so, is there an association between the extent of cognitive impairment and psychological well-being? And how may cognitively impaired older individuals may cope with a sudden and unexpected event, such as COVID-19? The aim of this study is to evaluate the impact of spring 2020 lockdown period in Italy due to COVID-19 on psychological and affective well-being of older adults with different levels of cognitive impairment and of their caregivers. We hypothesized that the COVID-19 quarantine may have had a stronger impact on individuals with worse cognitive performance in terms of affective symptoms and coping strategies and on their caregivers in terms of perceived burden.
Materials and Methods
Study Design
This study is part of the GeroCovid protocol, a multi-purpose, multi-setting and multicenter initiative (8). GeroCovid involves individuals aged ≥ 60 years, prospectively or retrospectively observed since March 1st, 2020. Data are collected in multiple investigational sites in Italy and Norway, and recorded in a de-identified clinical e-Registry. This study (GeroCovid “GeroCovid dementia—psychological health cohort”) involves 10 Italian Centers for Cognitive Decline and Dementia (CDCDs) and considers three phases: before (PREQ, January–February), during (DQ, March-May) and after (POSTQ, July–December) quarantine. As for PREQ and POSTQ data were collected retrospectively and during the follow-up visits, respectively (see Figure 1 for schematic representation of the study). All the records were thus collected from January to December 2020. The study was approved by the BIO-CAMPUS Ethic Committee, University of Rome—Prot. Number: 22.5(20).20 OSS ComEt-UCBM. Each center, moreover, had the approval of its own Ethic Committee.
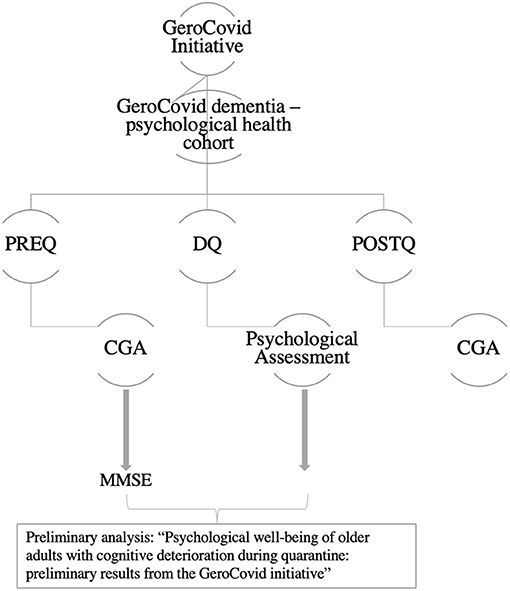
Figure 1. Schematic representation of study design. PREQ, evaluation before quarantine; DQ, evaluation during quarantine; POSTQ, evaluation after quarantine; CGA, comprehensive geriatric assessment; MMSE, Mini Mental State Examination.
Participants
Individuals with MCI or dementia were recruited from Italian CDCDs, according the following inclusion criteria: (1) last routine cognitive evaluation between January and March 2020 and next follow-up visit expected into 6–9 months; (2) Diagnosis of Alzheimer's Disease (AD) or other dementia, or diagnosis of MCI. Exclusion criteria were the following: (1) inability to undergo psychometrics tests for any reason; or, (2) history of psychiatric illness, according to clinical anamnesis. An initial number of 496 individuals meeting these criteria were originally contacted; of them, 260 agreed to participate to the study, although 10 did not complete the questionnaires. The total sample is thus composed of 250 participants.
Procedure
For the purpose of this study, we considered participants' sociodemographic data (age, sex, years of education, cohabiting status), information on risk behaviors (smoking and alcohol consumption, Yes/No), medical history (including diagnosis of MCI and dementia, depressive mood and other coexisting chronic diseases), and drug treatments. The following comorbidities were considered: cardiovascular diseases (ischemic heart diseases, heart failure), atrial fibrillation, hypertension, cerebrovascular diseases, diabetes mellitus, dyslipidemia, thyroid dysfunctions, gastrointestinal diseases, cancer, osteoporosis, osteoarthritis, rheumatologic diseases, chronic obstructive pulmonary disease, hematologic disorders, chronic kidney disease, Parkinson's disease, vision deficits, hearing deficits. The total number of chronic diseases, calculated as the sum of the above-mentioned conditions, was used as comorbidity indicators.
A comprehensive geriatric assessment was performed in the PREQ and POSTQ phases. For this preliminary study, among the PREQ evaluations we considered cognitive performance through the Mini-Mental State Examination [MMSE (9)], nutritional status through the Mini-Nutritional Assessment [MNA (10)], and functional status through the Activities of Daily Living [ADL (11)] and Instrumental ADL scales [IADL (12)]. Concerning participants' social environment, we considered the presence of a formal or informal caregiver, and the number of informal visits received on average by each participant before the quarantine period.
DQ evaluations were carried-out by means of telephonic interviews to the patients, and included:
- Depression Anxiety Stress Scales-21 [DASS-21 (13)] composed by a set of three self-report scales designed to measure the emotional states of depression, anxiety and stress. Each of the three DASS-21 scales contains 7 items, divided into subscales with similar content. Cut-off scores for depression, anxiety, and stress were 10, 8, and 15, respectively (14). The depression scale (including the items 3, 5, 10, 13, 16, 17, and 21) assesses dysphoria, hopelessness, devaluation of life, self-deprecation, lack of interest/involvement, anhedonia and inertia. The anxiety scale (including the items 2, 4, 7, 9, 15, 19, and 20) assesses autonomic arousal, skeletal muscle effects, situational anxiety, and subjective experience of anxious affect. The stress scale is sensitive to levels of chronic non-specific arousal and included the items 1, 6, 8, 11, 12, 14, and 18). It assesses difficulty relaxing, nervous arousal, and being easily upset/agitated, irritable/over-reactive and impatient. Scores for depression, anxiety and stress are calculated by summing the scores for the relevant items, then multiplied by two. The cut-offs used to detect the presence of symptoms of depression
- Perceived Stress Scale [PSS-10; (15)] the most frequently used psychological measure to assess perceptions of stress. The degree to which the situations in a person's life are rated as stressful are evaluated by 10 items constructed to capture the level at which respondents perceive their lives as unpredictable, uncontrollable, or overloaded. The scale also contains a series of direct questions about current levels of perceived stress. The PSS was designed to be used in samples drawn from the general population with an educational level at least equal to lower middle school. The items and the response alternatives are easy to understand: for each item, respondents are asked to indicate how often they felt a certain way in the last month (“0 = Never,” “4 = Very often”). The PSS scores are obtained by reverse-scoring the responses to the four positively formulated items (items 4, 5, 7, and 8), then adding together the scores for each and every item. A short 4-item scale can be obtained using questions 2, 4, 5, and 10 of the 10 items in the PSS scale.
- Coping Orientation to Problems Experienced [COPE (16)] a multidimensional coping inventory to assess the different ways in which people respond to stress. Five scales (of four items each) measure conceptually distinct aspects of problem-focused coping (active coping, planning, suppression of competing activities, restraint coping, seeking of instrumental social support); five scales measure aspects of what might be viewed as emotion-focused coping (seeking of emotional social support, positive reinterpretation, acceptance, denial, turning to religion); and three scales measuring coping responses that arguably are less useful (focus on and venting of emotions, behavioral disengagement, mental disengagement).
The subscales are calculated as follows: the subscales “social support” (indicated by the sum of items 1, 10, 15, 18, 25), “positive attitude” (indicated by the sum of items 2, 6, 12, 16, 23, 24), “orientation to problem” (indicated by the sum of items 3, 5, 9,13, 20), and “transcendent orientation” (indicated by the sum of items 8, 11, 14, 19).
Finally, telephonic interviews were also performed to patients' caregivers to evaluate their burden through the Caregiver Burden Inventory [CBI; (17)] a 24-item self-report questionnaire for assessing the burden of caregivers caring for people with chronic disease. The items are rated on a 5-point Likert scale from 0 “Never” to 4 “Nearly always.” The questions cover 5 dimensions of caregiver burden: objective burden; time-dependence, referring to time demands for assistance; psychological burden, understood as the caregiver's feelings of exclusion from expectations and opportunities; physical burden, which describes the caregiver's feelings of fatigue and health problems; social burden, which describes the caregiver's feelings of role conflict; and emotional burden, which describes the caregiver's feelings of shame or embarrassment caused by the patient. Time spent for assistance, social involvement, physical involvement, and relational involvement are represented, respectively by the sum of the items from 1 to 5, from 6 to 10, 11 to 14, and 15 to 19.
The presence of a caregiver or appointed legal guardian (e.g., a support administrator) was always required during telephone interviews in order to limit potential biases due to patients' cognitive impairment and their ability to answer questions (18). All participants (or their caregivers or guardians) gave informed consent to their involvement to the study.
Statistical Analyses
Descriptive characteristics of the sample are expressed as means ± standard deviations or as count (%), as appropriate. Participants were categorized according to their PREQ MMSE value (≥25, 23–24, and ≤ 22), and the comparison of the sociodemographic and health-related characteristics between such PREQ MMSE groups was performed through the ANOVA.
In order to test the hypothesis that individuals with worse cognitive performance (and their caregivers) may have been more strongly impacted by the COVID-19 quarantine, we first compared the DASS, PSS, COPE, and CBI scores (as continuous variables) between the three PREQ MMSE groups. To take into account the effect of potential confounders (i.e. age, sex, education, social environment, depressive mood, use of antipsychotics, and number of chronic diseases) in the association between PREQ MMSE with psychological well-being (depression, anxiety, stress), coping strategies, and caregiver burden, we run multivariable linear regressions. As independent variable, we considered PREQ MMSE either as continuous or categorical variable, in order to evaluate possible dose-response relationships. As dependent variables, we considered total DASS and PSS scores, the subscales “social support” (indicated by the sum of items 1, 10, 15, 18, 25), “positive attitude” (indicated by the sum of items 2, 6, 12, 16, 23, 24), “orientation to problem” (indicated by the sum of items 3, 5, 9,13, 20), and “transcendent orientation” (indicated by the sum of items 8, 11, 14, 19) of COPE, and the total CBI score and its subscales (time spent for assistance—sum of items 1 to 5, social involvement—sum of items 6 to 10, physical involvement—sum of items 11 to 14, relational involvement—sum of items 15 to 20) all as continuous variables.
Results
Our sample included 250 individuals (62% women) with a mean age of 79.6 ± 6.7 years and a PREQ MMSE of 23.1 ± 2.8. The most frequent cognitive disorders in our sample were MCI (23.2%), mild AD (30.8%), and mild vascular dementia (21.6%). Comparing the characteristics of participants by cognitive performance (Table 1), we found that those with lower pre-quarantine MMSE were more likely to be older, women, to have lower educational level and functional status, with the need for a caregiver.
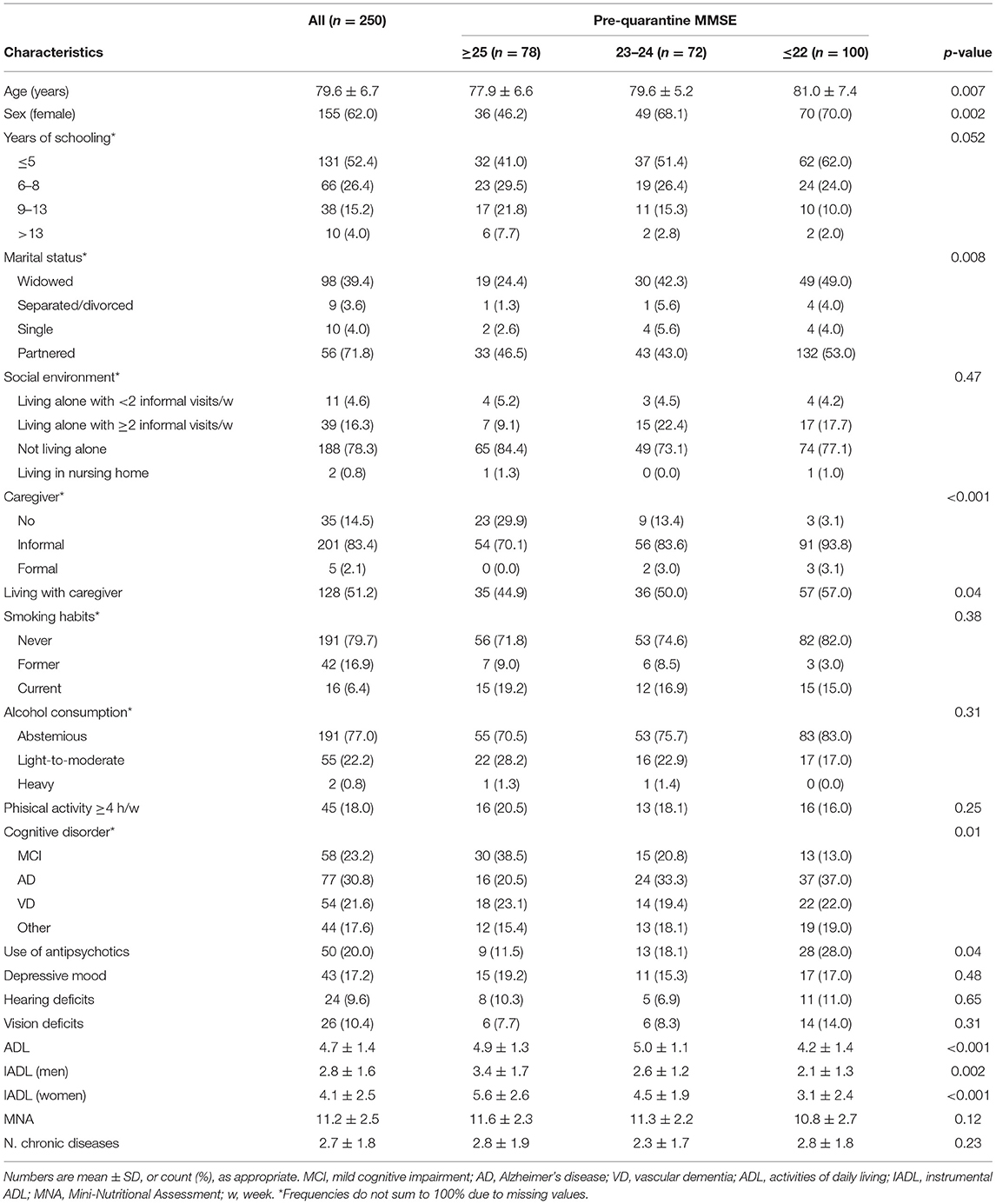
Table 1. Sociodemographic and health-related characteristics of participants by pre-quarantine cognitive functioning.
Symptoms of anxiety, depression, and psychological stress (DASS) were observed in 28.8, 48, and 24.8% of the sample, respectively (for cut-off scores see the Methods section). As shown in Table 1, individuals cognitively more impaired (MMSE ≤ 22) showed higher total DASS and PSS scores, compared both to individuals with MMSE ≥25 and 22 < MMSE <25. As for the COPE scale, individuals with greater cognitive impairment showed worse “positive attitude” and “problem orientation” coping strategies, as compared to those with higher MMSE scores. As expected, higher caregiving burden, in particular as for time spent for assistance (items 1–5) was observed in caregivers of individuals with more severe cognitive impairment (Table 2).
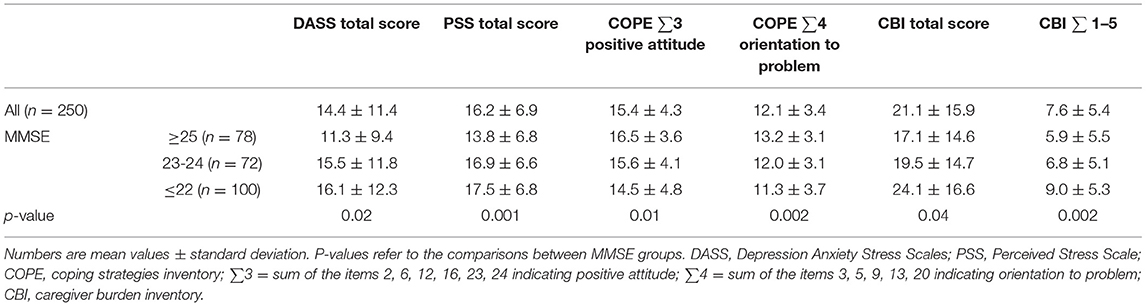
Table 2. Psychological well-being and coping strategies scales in the total sample and by pre-quarantine cognitive functioning.
The linear regression models confirmed that lower cognitive functioning was independently associated with a stronger negative psychological and affective reaction to quarantine, as well as with a poorer implementation of coping strategies, and with higher caregiving burden (Table 3). Specifically, higher stress was reported by caregivers in association with a reduction of the time dedicated to themselves, a greater sense of failure of hopes and expectations, and physical involvement (Items 1–5, 6–10, and 11–14 of CBI, respectively). No substantial differences were observed when testing the association between PREQ MMSE and other COPE and CBI subscales (data not shown).
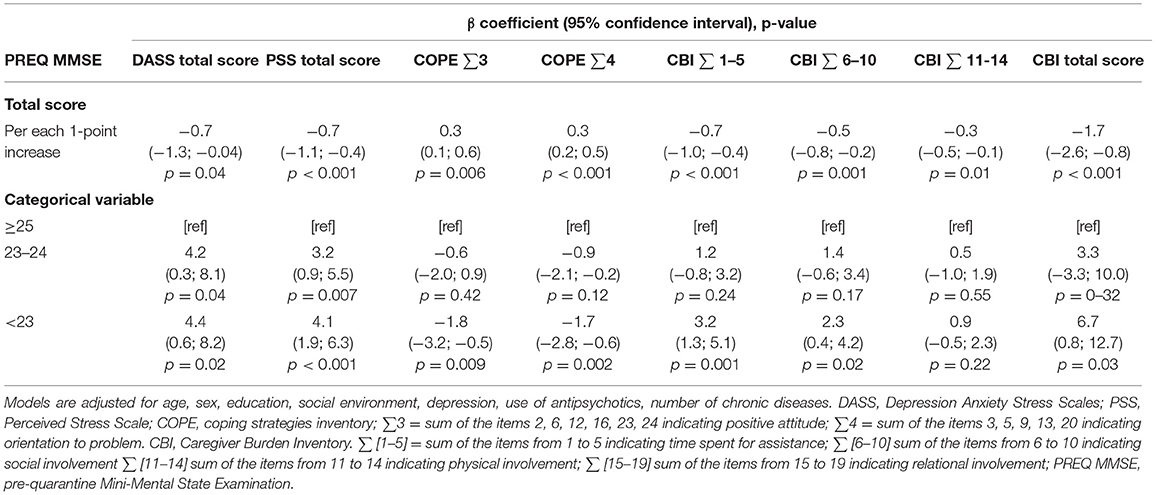
Table 3. Linear regression models on the association between pre-quarantine MMSE and patients' psychological well-being and caregivers' burden during quarantine.
Discussion
This study showed that, in individuals with MCI and dementia, the more severe the cognitive impairment, the higher the depression and anxiety experienced during the first wave of quarantine due to COVID-19. This evidence suggests that, despite the potential lack of awareness on the pandemic, individuals with dementia did perceive distress during the quarantine period. In particular, they showed higher scores in those items investigating psycho-somatic symptoms (“I feel my mouth dry” and “I feel nervous”). These symptoms are reported to represent psychological distress expressed through physical disturbances by individuals unable to express their emotions due to genetic and environmental factors (19). What is more, this finding is also corroborated by an increasing and challenging literature attesting that somatic disorders in individuals with dementia are related to outcomes and quality of life (20). Interestingly, among the most stressful events in life (i.e., spouse or relative death illness/surgical interventions, or problems with the family), people with dementia also reported as strongly demanding and tense “change in environment” (21), which exactly is what happen with Covid-19 occurrence. Similarly, Giebel et al. (22) found that social care and support services changes and closures altered the typical physical and “communal” environment negatively impacting on psychological well-being of people with dementia.
Our results are, therefore, in line with other studies: as recently reviewed by Sepulveda-Loyola et al. (23) in their meta-analysis, several consequences on mental health occur along with pandemics, such as depression, emotional disturbances, stress, deflection of mood, irritability and insomnia. Alarmingly, Yip et al. (24) also showed that these disorders are associated with higher suicide rates during pandemics, particularly in older adult populations.
In our study, individuals with more severe cognitive impairment were also found to have poorer coping strategies than those with higher cognitive performance. One can argue that this finding is not strictly linked to the pandemic or to the current cognitive status of participants (25), and unfortunately, we had not detailed information on psycho-affective distress level before quarantine. Yet the association remained significant even after adjusting for the presence of PREQ depression. Therefore, it is possible that poorer and less efficient coping strategies exposed the individuals to higher social distancing-associated distress (26). On the whole, subjects with more severe cognitive impairment, in spite of a limited awareness of pandemic-related issues, are not protected from the deleterious psychological effects of COVID-19, as also confirmed by Boutoleau-Bretonnière et al. (27). It is well-known that older adults are at increased risk of being socially isolated compared to younger adults under normal conditions (28). Similarly, people living with cognitive impairment are particularly subjected to the effects of social isolation that negatively impact their cognitive and affective well-being (29). The pandemic seems thus to have done nothing but worsen an already existing framework of frailty typical of this population (30, 31). Interestingly, in our study this happens in the most cognitively impaired individuals that might be considered as already so compromised that social isolation cannot hit them furtherly. Instead, social and environmental stimuli appear to be still important even in the more advanced stages of dementia. This result stresses the importance of supporting individuals with dementia through cognitive stimulation trainings and not just with pharmacological treatments (32).
As expected, these consequences did not seem to affect only patients, but also their caregivers. Indeed, also in the context of COVID-19 quarantine, we found that the greater the cognitive impairment of the patients, the heavier the burden of their caregivers. Our results confirm the other few studies available that suggest an increase of global caregiver burden during the COVID-19 (33–35). Our study, moreover, adds some novelty to the current literature by highlighting that the main burden experienced is related to psychological affliction, more than physical or time-dependent assistance. Our results only partially are in accordance with those reported by Cohen et al. (author?) (36), according to whom family members' main concern was, for severe dementia cases, fear of absence of the paid caregiver during the pandemic. For mild cases, instead, caregivers mainly reported fear of spreading the disease while assisting their relatives with instrumental activities.
Limitations and Strength
Some limitations should be acknowledged. First of all, the scales used for the psycho-affective evaluation are not validated for the remote administration. However, although originally not designed in this format, these tools have been previously administered remotely (37). Secondary, the psycho-affective profile of patients with cognitive deterioration before the pandemic is not known, we only have reference population data. However, as stated in the Methods section, this is a preliminary study and a more complete, longitudinal one (Gerocovid initiative) will show, at least, cognitive change (measured by MMSE) before and after the pandemic. Similarly, pre-pandemic anamnestic and clinical data were collected retrospectively. Finally, our results should be considered as limited to mild and moderate dementia and not generalizable to individuals with more severe cognitive impairment. Conversely, the research topic is timely and brings novelty to the COVID-19 literature. Different aspects of psychological well-being are considered, including the precious, though sometimes neglected, caregivers' point of view.
Conclusions
This study found that during the Covid-19 pandemic, people with neurocognitive impairments seem to experience, psycho-affective disorders, which vary according to their pre-quarantine cognitive functioning. In particular, patients with more severe cognitive impairment psychologically seem to have suffered the most of the effects of the pandemic, as well as their caregivers. Our study points out the role of poor and dysfunctional coping strategies adopted by individuals with MCI and dementia to explain the distress related to Covid-19 pandemic. Limited positive attitude and orientation to problem behaviors in facing the pandemic further contribute to the stress response. In conclusions, physicians and health care professionals caring for people with neurocognitive disorders should be aware that cognitive impairment does not prevent from the negative effects of the pandemic on emotional and affective distress. Attention should be given to the psychological well-being of individuals with MCI or dementia, and of their caregivers.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics Statement
The studies involving human participants were reviewed and approved by BIO-CAMPUS Ethic Committee, University of Rome – Prot. Number: 22.5(20).20 OSS Com-Et-UCBM. The patients/participants provided their written informed consent to participate in this study.
Author Contributions
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
Conflict of Interest
SS is shareholder and GZ is shareholder and employee of Bluecompanion Ltd (London, UK).
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
We are thankful to all the colleagues who contributed to the data collection, and to all the study participants. We thank Gilda Borselli for her precious support for the organization of the GeroCovid initiative.
References
1. Wilder-Smith A, Freedman DO. Isolation, quarantine, social distancing and community containment: pivotal role for old-style public health measures in the novel coronavirus (2019-nCoV) outbreak. J Travel Med. (2020) 27:taaa020. doi: 10.1093/jtm/taaa020
2. Pancani L, Marinucci M, Aureli N, Riva P. Forced social isolation and mental health: a study on 1,006 Italians under COVID-19 lockdown. Front Psychol. (2021) 12:1540. doi: 10.3389/fpsyg.2021.663799
3. Cagnin A, Di Lorenzo R, Marra C, Bonanni L, Cupidi C, Laganà V, et al. Behavioral and psychological effects of coronavirus disease-19 quarantine in patients with dementia. Front Psychiatry. (2020) 11:916. doi: 10.3389/fpsyt.2020.578015
4. Orrù G, Ciacchini R, Gemignani A, Conversano C. Psychological intervention measures during the COVID-19 pandemic. Clin Neuropsychiatry. (2020) 17:76–9.
5. Yanez ND, Weiss NS, Romand JA, Treggiari MM. COVID-19 mortality risk for older men and women. BMC Public Health. (2020) 20:1–7. doi: 10.1186/s12889-020-09826-8
6. Devita M, Bordignon A, Sergi G, Coin A. The psychological and cognitive impact of Covid-19 on individuals with neurocognitive impairments: research topics and remote intervention proposals. Aging Clin Exp Res. (2020) 33:733–6. doi: 10.1007/s40520-020-01637-6
7. Simonetti A, Pais C, Jones M, Cipriani MC, Janiri D, Monti L, et al. Neuropsychiatric symptoms in elderly with dementia during COVID-19 pandemic: definition, treatment, future directions. Front Psychiatry. (2020) 11:579842. doi: 10.3389/fpsyt.2020.579842
8. Trevisan C, Del Signore S, Fumagalli S, Gareri P, Malara A, Mossello E, et al. Assessing the impact of COVID-19 on the health of geriatric patients: The European GeroCovid Observational Study. Eur J Intern Med. (2021) 87:29–35.
9. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatric Res. (1975) 12:189–98.
10. Rubenstein LZ, Harker JO, Salva A, Guigoz Y, Vellas B. Screening for undernutrition in geriatric practice: developing the short-form Mini Nutritional Assessment (MNA-SF). J Geront. (2001) 56A:M366–77. doi: 10.1093/gerona/56.6.M366
11. Katz S, Downs TD, Cash HR, Grotz RC. Progress in development of the index of ADL. Gerontologist. (1970) 10:20–30. doi: 10.1093/geront/10.1_Part_1.20
12. Lawton MP, Brody EM. Assessment of older people: self-maintaining and instrumental activities of daily living. Gerontologist. (1969) 9:179–86. doi: 10.1093/geront/9.3_Part_1.179
13. Bottesi G, Ghisi M, Altoè G, Conforti E, Melli G, Sica C. The Italian version of the Depression Anxiety Stress Scales-21: factor structure and psychometric properties on community and clinical samples. Comprehens Psychiatry. (2015) 60:170–81. doi: 10.1016/j.comppsych.2015.04.005
14. Lovibond SH, Lovibond PF. Manual for the Depression Anxiety and Stress Scales. 2nd ed. Sydney, NSW: Psychology Foundation (1995).
15. Cohen S, Williamson G. Perceived stress in a probability sample of the United States. In: Spacapan S, Oskamp S, editors. The Social Psychology of Health. Newbury Park, CA: Sage Publishers (1988).
16. Carver CS. You want to measure coping but your protocol'too long: consider the brief cope. Int J Behav Med. (1997) 4:92. doi: 10.1207/s15327558ijbm0401_6
17. Novak M, Guest C. Application of a multidimensional caregiver burden inventory. Gerontol. (1989) 29:798–803.
18. Savla J, Roberto KA, Blieszner R, McCann BR, Hoyt E, Knight AL. Dementia caregiving during the “stay-at-home” phase of COVID-19 pandemic. J Gerontol Ser B. (2021) 76:e241–5. doi: 10.1093/geronb/gbaa129
19. Karukivi M, Saarijärvi S. Development of alexithymic personality features. World J Psychiatry. (2014) 4:91. doi: 10.5498/wjp.v4.i4.91
20. Mukku SSR, Desai G, Chaturvedi SK. Depression and somatic symptoms in dementia: a narrative review. J Geriatr Mental Health. (2020) 7:11. doi: 10.4103/jgmh.jgmh_4_20
21. Fountoulakis KN, Pavlidis I, Tsolaki M. Life events and dementia: what is the nature of their relationship? Psychiatry Res. (2011) 190:156–8. doi: 10.1016/j.psychres.2011.05.011
22. Giebel C, Cannon J, Hanna K, Butchard S, Eley R, Gaughan A, et al. Impact of COVID-19 related social support service closures on people with dementia and unpaid carers: a qualitative study. Aging Ment Health. (2021) 25:1281–8. doi: 10.1080/13607863.2020.1822292
23. Sepúlveda-Loyola W, Rodríguez-Sánchez I, Pérez-Rodríguez P, Ganz F, Torralba R, Oliveira DV, et al. Impact of social isolation due to COVID-19 on health in older people: mental and physical effects and recommendations. J Nutr Health Aging. (2020) 24:364–5. doi: 10.1007/s12603-020-1500-7
24. Yip PS, Cheung YT, Chau PH, Law YW. The impact of epidemic outbreak. Crisis. (2010) 31:86–92. doi: 10.1027/0227-5910/a000015
25. Dröes RM, Van Der Roest HG, Van Mierlo L, Meiland FJ. Memory problems in dementia: adaptation and coping strategies and psychosocial treatments. Expert Rev Neurother. (2011) 11:1769–82. doi: 10.1586/ern.11.167
26. Kessler RC. Stress, social status, psychological distress. J Health Soc Behav. (1979) 20:259–72. doi: 10.2307/2136450
27. Boutoleau-Bretonnière C, Pouclet-Courtemanche H, Gillet A, Bernard A, Deruet AL, Gouraud I, et al. The effects of confinement on neuropsychiatric symptoms in Alzheimer's disease during the COVID-19 crisis. J Alzheimer's Dis. (2020) 76:41–7. doi: 10.3233/JAD-200604
28. Courtin E, Knapp M. Social isolation, loneliness and health in old age: a scoping review. Health Soc Care Commun. (2017) 25:799–812. doi: 10.1111/hsc.12311
29. Greenwood N, Mezey G, Smith R. Social exclusion in adult informal carers: a systematic narrative review of the experiences of informal carers of people with dementia and mental illness. Maturitas. (2018) 112:39–45. doi: 10.1016/j.maturitas.2018.03.011
30. dos Santos Azevedo LV, Calandri IL, Slachevsky A, Graviotto HG, Santos Vieira MC, Andrade CBD, et al. Impact of social isolation on people with dementia and their family caregivers. J Alzheimer's Dis. (2021) 81:607–17. doi: 10.3233/JAD-201580
31. Gorenko JA, Moran C, Flynn M, Dobson K, Konnert C. Social isolation and psychological distress among older adults related to COVID-19: a narrative review of remotely-delivered interventions and recommendations. J Appl Gerontol. (2021) 40:3–13. doi: 10.1177/0733464820958550
32. Devita M, Masina F, Mapelli D, Anselmi P, Sergi G, Coin A. Acetylcholinesterase inhibitors and cognitive stimulation, combined and alone, in treating individuals with mild Alzheimer's disease. Aging Clin Exp Res. (2021) 1–7. [Epub ahead of print].
33. Cohen SA, Kunicki ZJ, Drohan MM, Greaney ML. Exploring changes in caregiver burden and caregiving intensity due to COVID-19. Gerontol Geriatr Med. (2021) 7:2333721421999279. doi: 10.1177/2333721421999279
34. Canevelli M, Valletta M, Blasi MT, Remoli G, Sarti G, Nuti F, et al. Facing dementia during the COVID-19 outbreak. J Am Geriatr Soc. (2020) 68:1673–6. doi: 10.1111/jgs.16644
35. Park SS. Caregivers' mental health and somatic symptoms during COVID-19. J Gerontol Ser B. (2021) 76:e235–40. doi: 10.1093/geronb/gbaa121
36. Cohen G, Russo MJ, Campos JA, Allegri RF. Living with dementia: increased level of caregiver stress in times of COVID-19. Int Psychogeriatr. (2020) 32:1377–81.
Keywords: COVID-19, dementia, psychological well-being, older adults, distress
Citation: Coin A, Devita M, Trevisan C, Biasin F, Terziotti C, Signore SD, Fumagalli S, Gareri P, Malara A, Mossello E, Volpato S, Monzani F, Bellelli G, Zia G, Ranhoff AH and Antonelli Incalzi R (2021) Psychological Well-Being of Older Adults With Cognitive Deterioration During Quarantine: Preliminary Results From the GeroCovid Initiative. Front. Med. 8:715294. doi: 10.3389/fmed.2021.715294
Received: 26 May 2021; Accepted: 25 August 2021;
Published: 22 September 2021.
Edited by:
Marcia G. Ory, Texas A&M University, United StatesReviewed by:
Lindsay M. K. Wallace, University of Cambridge, United KingdomNathalie Barth, Université Jean Monnet, France
Copyright © 2021 Coin, Devita, Trevisan, Biasin, Terziotti, Signore, Fumagalli, Gareri, Malara, Mossello, Volpato, Monzani, Bellelli, Zia, Ranhoff and Antonelli Incalzi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Maria Devita, bWFyaWEuZGV2aXRhQHVuaXBkLml0
 Alessandra Coin
Alessandra Coin Maria Devita
Maria Devita Caterina Trevisan1
Caterina Trevisan1 Susanna Del Signore
Susanna Del Signore Pietro Gareri
Pietro Gareri Alba Malara
Alba Malara Enrico Mossello
Enrico Mossello Giuseppe Bellelli
Giuseppe Bellelli Gianluca Zia
Gianluca Zia Raffaele Antonelli Incalzi
Raffaele Antonelli Incalzi