- 1Department of Clinical and Experimental Medicine, University of Messina, Messina, Italy
- 2Department of Clinical and Experimental Medicine, University of Catania, Catania, Italy
Zoledronic acid (Zol) is a widely used intravenous aminobisphosphonate to treat both benign and malignant skeletal diseases, and bisphosphonate-related osteonecrosis of the jaw (BRONJ) is a serious side effect whose pathophysiology remains poorly understood. Vascular Endothelial Growth Factor (VEGF) has been recognized to mediate BRONJ in cancer patients undergoing Zol treatment, however data on VEGF are lacking in patients with osteoporosis. Increasing evidences demonstrate that vitamin D influences VEGF levels. The aim of this study was to investigate the influence of Zol on VEGF levels and the possible role for vitamin D on the Zol mediated changes of VEGF concentration in women with postmenopausal osteoporosis. Twenty-eight postmenopausal women with osteoporosis were enrolled and randomized into two groups to receive Zol (5 mg) or placebo. At baseline, at day-3 and day-30 VEGF serum levels were measured; bone turnover markers, 25-hydroxyvitamin D [25(OH)D] and serum calcium were evaluated at baseline. In Zol-treated women, VEGF increased significantly on day-3, and then decreased on day-30. In the Zol-treated women, the percent change of VEGF levels between baseline and day-30 (−18% at day-30 vs. baseline, p = 0.01) was significantly associated with serum 25(OH)D values (r = 0.29, p = 0.028). At a stepwise multiple regression analysis, after correcting for age, BMI, time since menopause, femoral neck BMD, osteocalcin, C-terminal telopeptide of type 1 collagen, and baseline VEGF levels, 25(OH)D levels were independently associated with VEGF change (β = 1.7, SE = 0.71, p = 0.03). For the first time, we detected early modifications of circulating VEGF in postmenopausal women receiving Zol for osteoporosis, identifying a vitamin D-dependent modulation of these changes.
Introduction
Zoledronic acid (Zol) is a powerful intravenous aminobisphosphonate (N-BP) currently used against primary or secondary osteoporosis (1). As shown in a randomized, double-blind, placebo-controlled, multinational study, the HORIZON trial, when compared with the placebo control group, Zol 5 mg administered once a year for up to 3 years was demonstrated to increase bone mineral density (BMD) at different skeletal regions, improve bone metabolism markers and reduce the risk of new morphometric vertebral and hip fractures (2, 3). Zol treatment is commonly safe and well-tolerated, with the most frequent adverse events being transient and reversible (4).
Osteonecrosis of the jaw (ONJ) represents an emergent severe oral disease, with dramatic implications for health related patient's quality of life (5). Since 2003, there has been a surge of interest in the effects of BPs on oral health, and ONJ has been described as a BP related class effect, and the acronym “BRONJ” was introduced to indicate Bisphosphonate-Related Osteonecrosis of Jaw. The American Association of Oral and Maxillofacial Surgeons (AAOMS) defines BRONJ as an exposure of portions of the jawbone in patients who have been exposed to BPs, lasting more than 8 weeks in patients with no history of jaw irradiation (6). Nonetheless, recent studies have clearly shown that also the antiresorptive agent denosumab and some antiangiogenic drugs could similarly induce ONJ (7, 8). Accordingly, the term Medication Related Osteonecrosis of Jaw (MRONJ) has been also acknowledged in medical literature (9, 10). In metastatic cancer patients, the risk of developing BRONJ has been mainly associated with BP potency, high, and cumulative dose as well as intravenous administration (11, 12). BRONJ has been rarely reported in osteoporotic postmenopausal women (13).
In accordance with published data, the angiogenesis suppression may play a role in the pathophysiology of BRONJ, which is still uncertain and under debate (14, 15). Beyond the known antiresorptive, immunomodulating, and direct antitumor actions, BPs are actually considered also antiangiogenic drugs. Particularly, Zol has been proven to decrease VEGF concentrations in patients with metastatic bone cancer (16, 17), and the decrease of serum VEGF levels after intravenous BPs has been proposed as a possible early predictive marker of BRONJ in cancer patients (18).
Postmenopausal women exhibit a higher risk of periodontal diseases due to the fall of estrogen levels, which exert a trophic action on oral cavity (19). Scardina et al. showed a decreased periodontal capillary density, an increased tortuosity and decreased diameter of loops in oral microcirculation in a setting of postmenopausal women (20). Consequently, the oral microcirculatory alterations observed in postmenopause may encourage the adverse effects of some medications, such as Zol, on oral health.
Moreover, conflicting evidences demonstrated that vitamin D can influence VEGF levels: some data support a promoting action, others a suppressive one of vitamin D upon the release of VEGF (21–23). An adequate vitamin D status has been claimed to amplify and maintain over time the effect of BPs, even after their discontinuation (24), and the risk to develop BRONJ has been shown higher in BPs treated patients with concomitant hypovitaminosis (25).
Therefore, it remains still unclear whether a therapeutic dose of Zol for the medical management of osteoporosis in postmenopausal women have anti-angiogenic effects, due to post-dose changes of VEGF, as previously recognized in cancer patients.
The aim of this study was to explore the in vivo effect of Zol on circulating VEGF levels and the possible role for vitamin D on the Zol mediated VEGF perturbancies in a cohort of postmenopausal women treated for osteoporosis.
Materials and Methods
Study Subjects
This prospective randomized placebo-controlled study considered a cohort of Caucasian postmenopausal women attending the Center for Prevention, Diagnosis and Treatment of Osteoporosis in the Department of Internal Medicine of the University of Messina. Participants were consecutively recruited if affected by at least one prevalent vertebral fracture and with a BMD T-score values indicative of osteopenia or osteoporosis (in accordance with the World Health Organization diagnostic criteria), which were eligible for i.v. therapy with Zol, in accordance with standards of good clinical practice. Exclusion criteria were moderate to severe chronic kidney or liver failure, secondary causes of osteoporosis including hyperthyroidism, hyperparathyroidism, hypercortisolism, or malabsorption, in situ or systemic disease that could have impaired microcirculation such as diabetes mellitus, dyslipidemia, arterial hypertension, connective tissue diseases, autoimmune disorders or oral lichen planus. Patients undergoing treatment for osteoporosis (e.g., BPs, teriparatide, denosumab, selective estrogen receptor modulators, strontium ranelate, and calcitonin) or with a history of prior therapy with these bone agents were also not considered.
Participants were enrolled after a careful clinical check including dental examination with preventive dentistry; women with poor oral hygiene, periodontal diseases, poorly fitting dentures, history of dental diseases, invasive dental procedures, e.g., tooth extractions, or with unhealed open soft tissue lesions in the mouth were not considered for this study.
Fracture risk was assessed by using Fracture Risk Assessment Tool (FRAX), a computer-based algorithm (http://www.shef.ac.uk/FRAX) that estimates the 10-year probability of a major fracture (hip, clinical spine, humerus, or wrist fracture) and the 10-year probability of a hip fracture, as previously reported (26). All the patients were under vitamin D supplementation (cholecalciferol 25,000 IU every 2 weeks) and they took calcium (calcium carbonate 500–1,000 mg daily) when needed to reach the recommended daily calcium intake.
Recruited women were randomized into two groups using a computer-generated randomization schedule in the order in which they were enrolled in the study.
At baseline, 18 participants underwent a single Zol 5 mg/100 mL (Aclasta®) administration at 10.00 a.m. and within 30 min. Ten participants, not receiving Zol served as controls and received i.v. 100 mL saline solution 0.9%.
The study was approved by our institutional research committee Policlinico “G. Martino” Messina (Prot.n. 71/19); it was performed in accordance with the 1964 Declaration of Helsinki and its later amendments for experiments involving humans and uniform requirements for manuscripts submitted to biomedical journals. Signed informed consent was obtained from all the participants.
Biochemical Assays
Serum samples were collected just before Zol administration at baseline, and then at day-3 and day-30 after Zol administration. Serum was separated from the blood corpuscles by centrifugation and stored frozen at −80°C until analyzed.
VEGF-A serum levels were measured at each time-point by Enzyme-Linked Immunosorbent Assay, ELISA (Duoset, R&D Systems Europe Ltd., Abingdon, UK) according to the manufacturer's instructions. The limit of detection of human VEGF-A defined as the analyte concentration resulting in an absorption significantly higher than that of the dilution medium (mean plus two standard deviations) was determined to be 7.9 pg/mL (mean of 6 independent assays).
The inter- and intra-assay coefficient of variation was 4.3 and 6.2%, respectively.
At baseline, levels of Osteocalcin (BGP), Bone-specific alkaline phosphatase (BSAP) as markers of bone formation, and serum C-telopeptide of type 1 collagen (CTX), as marker of bone resorption, 25-hydroxyvitamin D (25(OH)D) and serum calcium were recorded.
BGP was measured by immunoenzymatic assay (Invitrogen Ltd., UK) with an intra-assay coefficient of variance (CV) of 3.1% and inter-assay CV of 3.5%; BSAP was measured by immunoenzymatic assay with the Access Immunoassay System (Beckman Coulter, Fullerton, California) with intra- and interassay CVs of 2.3–3.7% and 4.9–9.8%, respectively; CTX was assessed using the Elecsys 2010 Immunoassay System (Roche, Basel, Switzerland) with intraassay CVs of 1.6% to 3% and interassay CVs of 1.3–4.3%; levels of 25(OH)D were detected by high performance liquid chromatography; serum calcium was measured using standard laboratory techniques.
Bone Mineral Density and Vertebral Fractures
At baseline, a Dual-energy X-ray Absorptiometry (DXA) densitometer (Hologic Discovery) was used to assess BMD at the lumbar spine (L1–L4) and at the femoral neck. The DXA calibration was performed daily following the manufacturer's instruction. Its coefficient of variation (CV) was 0.5%. Vertebral fractures (Vfs) were detected by a conventional spinal radiograph and images were taken from anterior-posterior (AP) and lateral views applied of the thoracic and lumbar spine.
Vfs was defined in accordance to the semiquantitative method proposed by Genant: a fracture was diagnosed if a 20% reduction in the anterior or middle height compared to the posterior height of the same vertebra or the posterior height compared to the superior and anterior adjacent ones was detected (27).
Statistical Analysis
Statistical analyses were performed using MedCalc software (version 10.2.0.0; MedCalc Software, Mariakerke, 173 Belgium). Values were expressed as mean ± SD or median (IQR). The normal distribution of values was verified with the Kolmogorov-Smirnov test. Differences were evaluated using the Student's t-test for paired and unpaired observations, with Wilcoxon test and Mann-Whitney test, and repeated measures analysis of variances. Pearson's correlation coefficient was calculated to evaluate the degree of association between two variables. A multiple regression analysis was performed to determine the influence of one independent variable after correcting for the others. A post-hoc analysis was performed to explore change of VEGF levels in participants who experienced or not an Acute Phase Response (APR) after Zol administration. For all the tests, a P-value of 0.05 or less was considered to indicate statistical significance.
Results
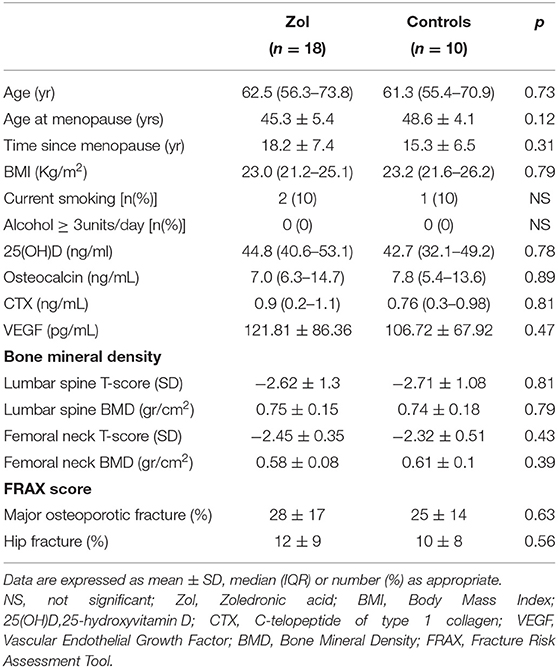
Baseline clinical features and main laboratory data of the participants are shown in Table 1. Particularly, at baseline, no significant differences in any of the investigated variables were observed between both groups of women.
In the overall population (n = 28), serum VEGF values were positively associated with femoral BMD (r = 0.42; p < 0.001) (Figure 1), and this correlation was maintained even after adjusting for age and BMI (r = 0.58; p = 0.01).
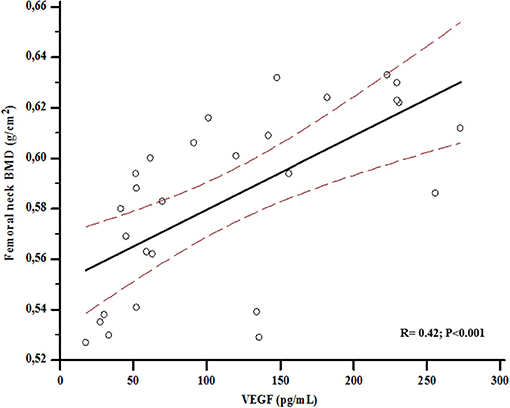
Figure 1. Association between bone mineral density (BMD) at femoral neck and Vascular Endothelial Growth Factor (VEGF) levels at baseline.
At baseline, VEGF values were associated with BGP levels (r = 0.57; p < 0.001) (Figure 2), and serum BGP positively correlated with femoral neck BMD (r = 0.33; p = 0.003) (Figure 3).
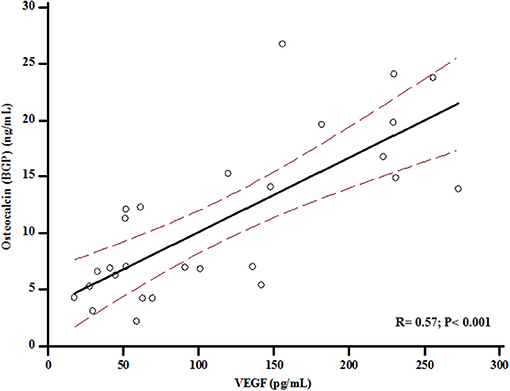
Figure 2. Association between levels of Osteocalcin and Vascular Endothelial Growth Factor (VEGF) at baseline.
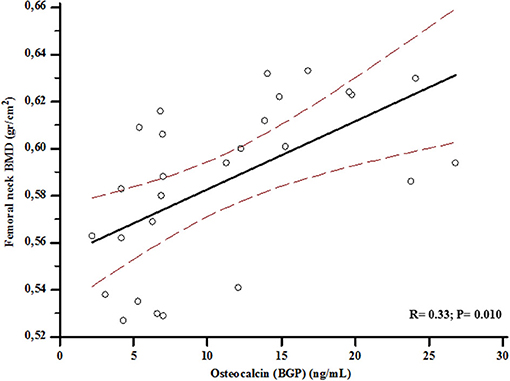
Figure 3. Association between levels of osteocalcin (BGP) and bone mineral density (BMD) at femoral neck at baseline.
In the 18 participants who received Zol, median VEGF levels significantly increased at day-3 to a value of 138.83 ± 100.89 pg/mL (+13% vs. baseline, p = 0.04), and then decreased at day-30 to a value of 102.43 ± 72.21 pg/mL (−26% vs. day-3, p = 0.002 and −18% vs. baseline, p = 0.01) (Figure 4). Conversely, controls did not show any significant changes over time (114.61 ± 68.13 and 110.72 ± 71.12 pg/mL at day-3 and day-30, respectively; pall > 0.5 vs. baseline). In the Zol-treated women, the percent change of VEGF levels between baseline and day-30 was significantly associated with serum 25(OH)D values (r = 0.29, p = 0.028) (Figure 5). Change of VEGF at day-3 was also associated with CTX levels (r = 0.77, p = 0.003). At a stepwise multiple regression analysis, 25(OH)D levels were the only predictor of VEGF change, after correcting for age, BMI, time since menopause, femoral neck BMD, BGP, CTX and baseline levels of VEGF (β = 1.7, SE = 0.71, p = 0.03). A post-hoc analysis showed that the rise of VEGF at day-3 was higher in the 3 participants who suffered APR in comparison with the other women receiving Zol who did not suffer APR [153.85 ± 126.5 vs. 135.15 ± 100.1 (+13%, p = 0.3)], although not reaching a statistical significance.
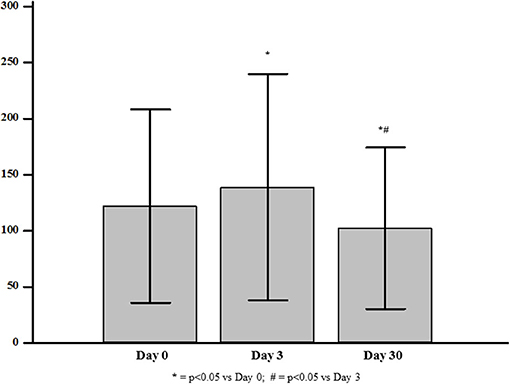
Figure 4. Levels of Vascular Endothelial Growth Factor (VEGF) at baseline and at day-3 and day-30 after zoledronic acid administration.
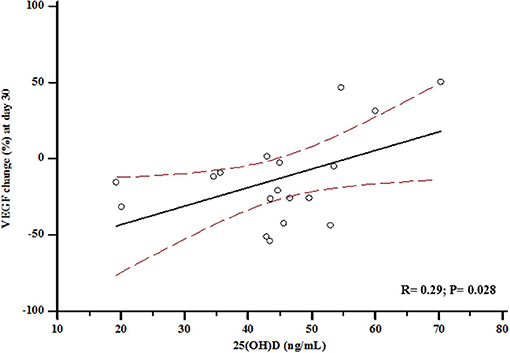
Figure 5. Association between vitamin D status at baseline and percent change of Vascular Endothelial Growth Factor (VEGF) at day-30.
Discussion
Zol is the most potent and long-acting N-BP used for the treatment of osteoporosis, Paget's disease, myeloma, and cancers to reduce adverse skeletal related events (1). Change of VEGF levels have been previously reported in cancer patients receiving Zol (17), and decreased VEGF values have been also associated with the BRONJ risk (18).
To the best of our knowledge, this is the first prospective cohort study investigating the early serum changes of VEGF levels in high fracture risk postmenopausal women receiving Zol. Particularly, VEGF levels significantly raised soon after 3 days from Zol administration, and then decreased after 30 days to a value below the baseline one.
The raise of VEGF at day-3 may fall in the window of the frequent occurring APR after i.v. N-BP administration.
APR is a common side effect observed in 10 to 50% of patients after the first Zol dose. It consists of a systemic immune reaction induced by the stimulation of circulating γδ T cells and the subsequent release of several inflammatory cytokines as interleukin 6 (IL-6), tumor necrosis factor-β (TNF-β) and interferon-γ (IFN-γ) (28, 29). Related symptoms occur within 24–36 h and usually disappear in about 3 days (30).
VEGF is a specific endothelial cell mitogen and a strong vascular permeability factor; thus, it may be considered as a proinflammatory cytokine. Consequently, in patients receiving Zol, the early increase of VEGF levels could be a biochemical mark of VEGF involvement in the cytokine storm leading to APR (31). Accordingly, at a post-hoc analysis, the rise of VEGF at day-3 was 13% higher in the 3 participants who suffered APR in comparison with the other women receiving Zol who did not experienced APR, although without significant difference.
On the other hand, the decrease of VEGF levels after 30 days could be determined by the well-known Zol related suppression of bone turnover and Zol activity on endothelial cells. Osteoclasts, osteoblasts, osteocytes and endothelial cells are sources of VEGF (32). In the bone tissue, VEGF stimulates angiogenesis, osteoblast differentiation, and finally bone formation and bone healing. Furthermore, VEGF has been shown to promote osteoclast differentiation and survival (33). Accordingly, VEGF could orchestrate the crosstalk between endothelial, osteoblastic, and hematopoietic cells (34). Data on VEGF levels in osteoporotic patients are conflicting, and circulating VEGF could be influenced by treatment (35).
In all the participants of this study, at baseline, we observed a significant positive association between VEGF and both BGP and femoral BMD. These findings have not been previously described and add new insights about the possible VEGF key role in bone health; moreover, they are consistent with previous studies showing significantly lower VEGF levels in women with postmenopausal osteoporosis (36). Therefore, the significant association of VEGF with BGP we observed could suggest a potential role of VEGF as a marker of bone formation. The mechanical loading has been reported to enhance osteocyte production of various cytokines, including VEGF, regulating bone homeostasis: this could explain its association with BMD at femoral neck, showed in Figure 1 (37).
Several lines of evidence based on both in vitro and in vivo studies (17, 18, 38, 39) suggest the anti-angiogenic and anti-tumor VEGF-related Zol effect. However, few researches involved humans and mostly cancer patients. In 30 patients suffering from solid tumors with bone metastases, treated with a single infusion of Zol 4 mg, Santini et al. reported a significant reduction of VEGF levels after 2, 7, and 21 days, with the largest significance (p < 0.001) on day 21 (40). Moreover, low intermittent doses of Zol (1 mg every 7 days for 4 times followed by 4 mg every 28 days for 3 times), produced significant reductions of the circulating VEGF early after 7 days in cancer patients with bone metastasis from solid tumors. Moreover, after 84 days and a 16 mg cumulative Zol dose, VEGF levels tended to rise, although remaining significantly lower in comparison with baseline (40). Remarkably, even though the different dose and schedule, the reduction of serum VEGF levels observed in osteoporotic postmenopausal women in our study was of a smaller magnitude than that one seen in the oncology setting.
In women with postmenopausal osteoporosis, only one elegant study by Ishtiaq et al. evaluated the VEGF change in vivo and in vitro after N-BPs. Alendronate (Aln), administered at a weekly dose of 70 mg, produced a VEGF decline after 3 and 6 months, reaching a statistical significance after 12 months (p = 0.02). Consistently, Aln and Zol, in the culture medium of two osteoblastic cell lines, significantly reduced the VEGF production, confirming that N-BP related in vivo modification of this cytokine should be attributed to a direct effect on bone cells (21).
The VEGF reduction obtained in vitro with Zol was more pronounced than that one with Aln and it occurred at a lower dose, in accordance with higher Zol potency; surprisingly, the addition to the medium in osteoblastic cell lines of calcitriol, the active vitamin D metabolite, reverted the N-BP induced suppression of VEGF release (21).
An inadequate vitamin D status could even amplify the VEGF-mediated suppression of angiogenesis induced by N-BPs, and particularly by Zol; due to this pathophysiological mechanism, an increased prevalence of BRONJ in patients with vitamin D deficiency has been suggested by some researches (18, 25, 41, 42).
Our findings are consistent with in vitro data by Ishtiaq et al. (21), although the significance of the effect of N-BPs on the production of pro-angiogenic factors to in vivo pharmacology remain uncertain; our data showed that the serum 25(OH)D concentration, indicative of vitamin D status, was significantly associated with the VEGF change in postmenopausal women receiving Zol. In other terms, enrolled women, which already had normal vitamin D levels at baseline, showed a smaller reduction of VEGF serum levels at the end of the observation period the more the vitamin D levels were higher.
Among its extraskeletal effects (43–46), vitamin D has been proven to influence VEGF levels, although its relationship with this cytokine is still under debate and divergent data exist in the literature. Vitamin D has been suggested to enhance angiogenic factors in several in vitro ed in vivo studies (47–49). Particularly, vitamin D boosts the expression of VEGF in the cell culture of human umbilical vein endothelial cells (HUVECs) (44, 50, 51), and a direct overexpression of VEGF trough vitamin D receptor (VDR) activation by calcitriol has been reported in vascular smooth muscle cells (VSMCs) (44). In a recent study which used an animal model of pre-eclampsia, vitamin D deficiency lowered the placental protein levels of pro-angiogenic proteins VEGF; a low dose vitamin D supplementation beginning from pre-pregnancy and continuing through pregnancy normalized the levels of VEGF (47). On the other hand, a down-regulation of VEGF by 1,25(OH)2D and its metabolites has been reported in vitro (52), and consistently a vitamin D-induced suppression of VEGF levels has been observed in breast cancer women treated with tamoxifen (22) and in chronic spontaneous urticaria (23).
Emami et al. demonstrated also that vitamin D might decrease pro-angiogenic factors such as visfatin and in turn VEGF in ulcerative colitis patients with low 25(OH)D levels (53). Additionally, in breast cancer xenograft tumors, 1,25(OH)2D3 could induce apoptosis of VEGF sprouting endothelial cells, thereby reducing the blood vessel density modulating pathologic angiogenesis (54, 55).
The current knowledge about vitamin D and angiogenesis still remain not fully clarified and further focused researches are needed.
We must recognize this study has limitations including the small sample size, consisting of only women in postmenopausal age and the observation period not long enough to capture Zol-induced VEGF modifications in the long term. This study is not focused on BRONJ; thus, the observed changes of VEGF levels after Zol administration may have uncertain significance, and their pathophysiological role requires additional investigations. Thus, further studies, focused on osteoporotic postmenopausal women, with a large sample size and with a long follow-up, are needed to confirm our data and to address the association with BRONJ. Additionally, measuring other angiogenic factors could lead to a more in-depth profile of the effect of Zol on angiogenesis.
At the same time, our findings improve the knowledge of cytokine modifications after N-BP administration, suggesting a role for vitamin D on the Zol mediated VEGF perturbancies.
In conclusion, in osteoporotic postmenopausal women, Zol administration at a dose of 5 mg provoked early modifications of circulating VEGF and vitamin D modulated this VEGF change.
Data Availability Statement
The datasets generated during and/or analyzed for this study are available from the corresponding author on reasonable request.
Ethics Statement
The studies involving human participants were reviewed and approved by Institutional Research Committee Policlinico G. Martino Messina (Prot.n. 71/19). The patients/participants provided their written informed consent to participate in this study.
Author Contributions
FB, AC, and NM designed the study. ARS, AC, and FB analyzed data and wrote the manuscript. AG, SL, and FC had full access to all the data in the study and take responsibility for the integrity and the accuracy of the data analysis. All authors reviewed the manuscript.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
The content of the manuscript have previously appeared online in a preprint (doi: 10.21203/rs.3.rs-530052/v1).
References
1. Reid IR, Green JR, Lyles KW, Reid DM, Trechsel U, Hosking DJ, et al. Zoledronate. Bone. (2020) 137:115390. doi: 10.1016/j.bone.2020.115390
2. Black DM, Delmas PD, Eastell R, Reid IR, Boonen S, Cauley JA, et al. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med. (2007) 356:1809–22. doi: 10.1056/NEJMoa067312
3. Black DM, Reid IR, Boonen S, Bucci-Rechtweg C, Cauley JA, Cosman F, et al. The effect of 3 versus 6 years of zoledronic acid treatment of osteoporosis: a randomized extension to the HORIZON-Pivotal Fracture Trial (PFT). J Bone Miner Res. (2012) 27:243–54. doi: 10.1002/jbmr.1494
4. Adami S, Bhalla AK, Dorizzi R, Montesanti F, Rosini S, Salvagno G, et al. The acute-phase response after bisphosphonate administration. Calcif Tissue Int. (1987) 41:326–31. doi: 10.1007/BF02556671
5. Sato T, Kusumoto J, Takeda D, Kishimoto M, Kashin M, Furudoi S, et al. Which symptoms negatively affect the oral health-related quality of life in patients with osteonecrosis of the jaw? Oral Surg Oral Med Oral Pathol Oral Radiol. (2020) 130:175–80. doi: 10.1016/j.oooo.2020.03.051
6. Ruggiero SL. Guidelines for the diagnosis of bisphosphonate-related osteonecrosis of the jaw (BRONJ). Clin Cases Miner Bone Metab. (2007) 4:37–42.
7. Malan J, Ettinger K, Naumann E, Beirne OR. The relationship of denosumab pharmacology and osteonecrosis of the jaws. Oral Surg Oral Med Oral Pathol Oral Radiol. (2012) 114:671–6. doi: 10.1016/j.oooo.2012.08.439
8. Ghidini G, Manfredi M, Giovannacci I, Mergoni G, Sarraj A, Mureddu M, et al. Medication-related osteonecrosis of the jaw: risk factors in patients under biphosphonate versus patients under antiresorptive-antiangiogenic drugs. Minerva Dent Oral Sci. (2017) 66:135–40. doi: 10.23736/S0026-4970.17.04056-0
9. Fusco V, Santini D, Armento G, Tonini G, Campisi G. Osteonecrosis of jaw beyond antiresorptive (Bone-Targeted) agents: new horizons in oncology. Expert Opin Drug Saf. (2016) 15:925–35. doi: 10.1080/14740338.2016.1177021
10. Oteri G, Campisi G, Panzarella V, Morreale I, Nucera R, Di Fede O, et al. Could the combined administration of bone antiresorptive drug, taxanes, and corticosteroids worsen medication related osteonecrosis of the jaws in cancer patients? Biomed Res Int. (2018) 2018:1–6. doi: 10.1155/2018/4021952
11. Khosla S, Burr D, Cauley J, Dempster DW, Ebeling PR, Felsenberg D, et al. Bisphosphonate-associated osteonecrosis of the jaw: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. (2007) 22:1479–91. doi: 10.1359/jbmr.0707onj
12. Bamias A, Kastritis E, Bamia C, Moulopoulos LA, Melakopoulos I, Bozas G, et al. Osteonecrosis of the jaw in cancer after treatment with bisphosphonates: incidence and risk factors. JCO. (2005) 23:8580–7. doi: 10.1200/JCO.2005.02.8670
13. Rizzoli R, Reginster J-Y, Boonen S, Bréart G, Diez-Perez A, Felsenberg D, et al. Adverse reactions and drug–drug interactions in the management of women with postmenopausal osteoporosis. Calcif Tissue Int. (2011) 89:91–104. doi: 10.1007/s00223-011-9499-8
14. Reid IR, Cornish J. Epidemiology and pathogenesis of osteonecrosis of the jaw. Nat Rev Rheumatol. (2012) 8:90–6. doi: 10.1038/nrrheum.2011.181
15. Estilo CL, Fornier M, Farooki A, Carlson D, Bohle G, Huryn JM. Osteonecrosis of the jaw related to bevacizumab. JCO. (2008) 26:4037–8. doi: 10.1200/JCO.2007.15.5424
16. Wood J, Bonjean K, Ruetz S, Bellahcène A, Devy L, Foidart JM, et al. Novel antiangiogenic effects of the bisphosphonate compound zoledronic acid. J Pharmacol Exp Ther. (2002) 302:1055–61. doi: 10.1124/jpet.102.035295
17. Santini D, Vincenzi B, Dicuonzo G, Avvisati G, Massacesi C, Battistoni F, et al. Zoledronic acid induces significant and long-lasting modifications of circulating angiogenic factors in cancer patients. Clin Cancer Res. (2003) 9:2893–7.
18. Vincenzi B, Napolitano A, Zoccoli A, Iuliani M, Pantano F, Papapietro N, et al. Serum VEGF levels as predictive marker of bisphosphonate-related osteonecrosis of the jaw. J Hematol Oncol. (2012) 5:56. doi: 10.1186/1756-8722-5-56
19. Meurman JH, Tarkkila L, Tiitinen A. The menopause and oral health. Maturitas. (2009) 63:56–62. doi: 10.1016/j.maturitas.2009.02.009
20. Scardina GA, Messina P. Oral microcirculation in post-menopause: a possible correlation with periodontitis: oral microcirculation in post-menopause. Gerodontology. (2012) 29:E1045–51. doi: 10.1111/j.1741-2358.2011.00608.x
21. Ishtiaq S, Edwards S, Sankaralingam A, Evans BAJ, Elford C, Frost ML, et al. The effect of nitrogen containing bisphosphonates, zoledronate and alendronate, on the production of pro-angiogenic factors by osteoblastic cells. Cytokine. (2015) 71:154–60. doi: 10.1016/j.cyto.2014.10.025
22. Shahvegharasl Z, Pirouzpanah S, Mahboob SA, Montazeri V, Adili A, Asvadi I, et al. Effects of cholecalciferol supplementation on serum angiogenic biomarkers in breast cancer patients treated with tamoxifen: a controlled randomized clinical trial. Nutrition. (2020) 72:110656. doi: 10.1016/j.nut.2019.110656
23. Zhao J-W, Ping J-D, Wang Y-F, Liu X-N, Li N, Hu Z-L, et al. Vitamin D suppress the production of vascular endothelial growth factor in mast cell by inhibiting PI3K/Akt/p38 MAPK/HIF-1α pathway in chronic spontaneous urticaria. Clin Immunol. (2020) 215:108444. doi: 10.1016/j.clim.2020.108444
24. Catalano A, Bellone F, Santoro D, Schwarz P, Gaudio A, Basile G, et al. Vitamin D boosts alendronate tail effect on bone mineral density in postmenopausal women with osteoporosis. Nutrients. (2021) 13:1878. doi: 10.3390/nu13061878
25. Ardine M, Generali D, Donadio M, Bonardi S, Scoletta M, Vandone AM, et al. Could the long-term persistence of low serum calcium levels and high serum parathyroid hormone levels during bisphosphonate treatment predispose metastatic breast cancer patients to undergo osteonecrosis of the jaw? Ann Oncol. (2006) 17:1336–7. doi: 10.1093/annonc/mdl045
26. Catalano A, Morabito N, Basile G, Fusco S, Castagna G, Reitano F, et al. Fracture risk assessment in postmenopausal women referred to an italian center for osteoporosis: a single day experience in Messina. Clin Cases Miner Bone Metab. (2013) 10:191–4.
27. Genant HK, Wu CY, van Kuijk C, Nevitt MC. Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res. (2009) 8:1137–48. doi: 10.1002/jbmr.5650080915
28. Rossini M, Adami S, Viapiana O, Ortolani R, Vella A, Fracassi E, et al. Circulating γδ T cells and the risk of acute-phase response after zoledronic acid administration. J Bone Miner Res. (2012) 27:227–30. doi: 10.1002/jbmr.521
29. Catalano A, Morabito N, Atteritano M, Basile G, Cucinotta D, Lasco A. Vitamin D reduces musculoskeletal pain after infusion of zoledronic acid for postmenopausal osteoporosis. Calcif Tissue Int. (2012) 90:279–85. doi: 10.1007/s00223-012-9577-6
30. Reid IR, Gamble GD, Mesenbrink P, Lakatos P, Black DM. Characterization of and risk factors for the acute-phase response after zoledronic acid. J Clin Endocrinol Metab. (2010) 95:4380–7. doi: 10.1210/jc.2010-0597
31. Hetland ML, Christensen IJ, Lottenburger T, Johansen JS, Svendsen MN, Hørslev-Petersen K, et al. Circulating VEGF as a biological marker in patients with rheumatoid arthritis? Preanalytical and biological variability in healthy persons and in patients. Dis Markers. (2008) 24:1–10. doi: 10.1155/2008/707864
32. Misso G, Porru M, Stoppacciaro A, Castellano M, De Cicco F, Leonetti C, et al. Evaluation of the in vitro and in vivo antiangiogenic effects of denosumab and zoledronic acid. Cancer Biol Ther. (2012) 13:1491–500. doi: 10.4161/cbt.22274
33. Aldridge SE, Lennard TWJ, Williams JR, Birch MA. Vascular endothelial growth factor receptors in osteoclast differentiation and function. Biochem Biophys Res Commun. (2005) 335:793–8. doi: 10.1016/j.bbrc.2005.07.145
34. Byrne AM, Bouchier-Hayes DJ, Harmey JH. Angiogenic and cell survival functions of vascular endothelial growth factor (VEGF). J Cellular Mol Med. (2005) 9:777–94. doi: 10.1111/j.1582-4934.2005.tb00379.x
35. Pepe J, Cipriani C, Cantatore FP, Fabbri A, Pola E, Vinicola V, et al. The effect of parathyroid hormone (1–84) treatment on serum bone morphogenetic protein 4 and vascular endothelial growth factor in postmenopausal women with established osteoporosis. J Endocrinol Invest. (2017) 40:663–7. doi: 10.1007/s40618-017-0636-8
36. Senel K, Baykal T, Seferoglu B, Altas EU, Baygutalp F, Ugur M, et al. Circulating vascular endothelial growth factor concentrations in patients with postmenopausal osteoporosis. Aoms. (2013) 4:709–12. doi: 10.5114/aoms.2013.36896
37. Yan Y, Wang L, Ge L, Pathak JL. Osteocyte-mediated translation of mechanical stimuli to cellular signaling and its role in bone and non-bone-related clinical complications. Curr Osteoporos Rep. (2020) 18:67–80. doi: 10.1007/s11914-020-00564-9
38. Guise TA. Antitumor effects of bisphosphonates: promising preclinical evidence. Cancer Treat Rev. (2008) 34:S19–24. doi: 10.1016/j.ctrv.2008.03.006
39. Lipton A. Emerging role of bisphosphonates in the clinic—antitumor activity and prevention of metastasis to bone. Cancer Treat Rev. (2008) 34:S25–30. doi: 10.1016/j.ctrv.2008.03.008
40. Santini D, Vincenzi B, Galluzzo S, Battistoni F, Rocci L, Venditti O, et al. Repeated intermittent low-dose therapy with zoledronic acid induces an early, sustained, and long-lasting decrease of peripheral vascular endothelial growth factor levels in cancer patients. Clin Cancer Res. (2007) 13:4482–6. doi: 10.1158/1078-0432.CCR-07-0551
41. Hokugo A, Christensen R, Chung EM, Sung EC, Felsenfeld AL, Sayre JW, et al. Increased prevalence of bisphosphonate-related osteonecrosis of the jaw with vitamin D deficiency in rats. J Bone Miner Res. (2010) 25:1337–49. doi: 10.1002/jbmr.23
42. Bedogni A, Saia G, Bettini G, Tronchet A, Totola A, Bedogni G, et al. Osteomalacia: the missing link in the pathogenesis of bisphosphonate-related osteonecrosis of the jaws? Oncologist. (2012) 17:1114–9. doi: 10.1634/theoncologist.2012-0141
43. Jamali N, Song Y, Sorenson CM, Sheibani N. 1,25(OH) [[sb]]2[[/s]] D [[sb]]3[[/s]] regulates the proangiogenic activity of pericyte through VDR-mediated modulation of VEGF production and signaling of VEGF and PDGF receptors. FASEB BioAdvances. (2019) 1:415–34. doi: 10.1096/fba.2018-00067
44. Cardus A, Panizo S, Encinas M, Dolcet X, Gallego C, Aldea M, et al. 1,25-Dihydroxyvitamin D3 regulates VEGF production through a vitamin D response element in the VEGF promoter. Atherosclerosis. (2009) 204:85–9. doi: 10.1016/j.atherosclerosis.2008.08.020
45. Saponaro F, Marcocci C, Zucchi R. Vitamin D status and cardiovascular outcome. J Endocrinol Invest. (2019) 42:1285–90. doi: 10.1007/s40618-019-01057-y
46. Lemke D, Klement RJ, Schweiger F, Schweiger B, Spitz J. Vitamin D resistance as a possible cause of autoimmune diseases: a hypothesis confirmed by a therapeutic high-dose vitamin D protocol. Front Immunol. (2021) 12:655739. doi: 10.3389/fimmu.2021.655739
47. Nema J, Sundrani D, Joshi S. Prenatal vitamin D supplementation reduces blood pressure and improves placental angiogenesis in an animal model of preeclampsia. Food Funct. (2020) 11:10413–22. doi: 10.1039/D0FO01782E
48. Purswani JM, Gala P, Dwarkanath P, Larkin HM, Kurpad A, Mehta S. The role of vitamin D in pre-eclampsia: a systematic review. BMC Pregnancy Childbirth. (2017) 17:231. doi: 10.1186/s12884-017-1408-3
49. Nema J, Sundrani D, Joshi S. Role of vitamin D in influencing angiogenesis in preeclampsia. Hypertens Pregnancy. (2019) 38:201–7. doi: 10.1080/10641955.2019.1647231
50. Grundmann M, Haidar M, Placzko S, Niendorf R, Darashchonak N, Hubel CA, et al. Vitamin D improves the angiogenic properties of endothelial progenitor cells. Am J Physiol. (2012) 303:C954–62. doi: 10.1152/ajpcell.00030.2012
51. Zhong W, Gu B, Gu Y, Groome LJ, Sun J, Wang Y. Activation of vitamin D receptor promotes VEGF and CuZn-SOD expression in endothelial cells. J Steroid Biochem Mol Biol. (2014) 140:56–62. doi: 10.1016/j.jsbmb.2013.11.017
52. Nakagawa K, Sasaki Y, Kato S, Kubodera N, Okano T. 22-Oxa-1α,25-Dihydroxyvitamin D 3 inhibits metastasis and angiogenesis in lung cancer. Carcinogenesis. (2005) 26:1044–54. doi: 10.1093/carcin/bgi049
53. Emami MR, Sharifi A, Yaseri M, Derakhshanian H, Hosseinzadeh-Attar MJ. Vitamin D suppresses proangiogenic factors in patients with ulcerative colitis: a randomized double blind placebo controlled clinical trial. Complement Ther Clin Pract. (2020) 39:101086. doi: 10.1016/j.ctcp.2020.101086
54. Wu X, Hu W, Lu L, Zhao Y, Zhou Y, Xiao Z, et al. Repurposing vitamin D for treatment of human malignancies via targeting tumor microenvironment. Acta Pharmaceutica Sinica B. (2019) 9:203–19. doi: 10.1016/j.apsb.2018.09.002
Keywords: bisphosphonate, zoledronic acid, vascular endothelial growth factor, osteonecrosis, postmenopausal, osteoporosis, vitamin D
Citation: Bellone F, Catalano A, Sottile AR, Gaudio A, Loddo S, Corica F and Morabito N (2021) Early Changes of VEGF Levels After Zoledronic Acid in Women With Postmenopausal Osteoporosis: A Potential Role of Vitamin D. Front. Med. 8:748438. doi: 10.3389/fmed.2021.748438
Received: 27 July 2021; Accepted: 15 October 2021;
Published: 18 November 2021.
Edited by:
Federica Saponaro, University of Pisa, ItalyReviewed by:
Cristiana Cipriani, Sapienza University of Rome, ItalyStefano Frara, Vita-Salute San Raffaele University, Italy
Copyright © 2021 Bellone, Catalano, Sottile, Gaudio, Loddo, Corica and Morabito. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Federica Bellone, ZmJlbGxvbmVAdW5pbWUuaXQ=; orcid.org/0000-0002-0467-4921
 Federica Bellone
Federica Bellone Antonino Catalano
Antonino Catalano Angelo Ruggero Sottile1
Angelo Ruggero Sottile1