- 1Institute of Physiology, University Medical Center, Johannes Gutenberg University of Mainz, Mainz, Germany
- 2Department of Neurophysiology, Hamamatsu University School of Medicine, Hamamatsu, Japan
A variety of experimental studies demonstrated that neurotransmitters are an important factor for the development of the central nervous system, affecting neurodevelopmental events like neurogenesis, neuronal migration, programmed cell death, and differentiation. While the role of the classical neurotransmitters glutamate and gamma-aminobutyric acid (GABA) on neuronal development is well established, the aminosulfonic acid taurine has also been considered as possible neuromodulator during early neuronal development. The purpose of the present review article is to summarize the properties of taurine as neuromodulator in detail, focusing on the direct involvement of taurine on various neurodevelopmental events and the regulation of neuronal activity during early developmental epochs. The current knowledge is that taurine lacks a synaptic release mechanism but is released by volume-sensitive organic anion channels and/or a reversal of the taurine transporter. Extracellular taurine affects neurons and neuronal progenitor cells mainly via glycine, GABA(A), and GABA(B) receptors with considerable receptor and subtype-specific affinities. Taurine has been shown to directly influence neurogenesis in vitro as well as neuronal migration in vitro and in vivo. It provides a depolarizing signal for a variety of neuronal population in the immature central nervous system, thereby directly influencing neuronal activity. While in the neocortex, taurine probably enhance neuronal activity, in the immature hippocampus, a tonic taurinergic tone might be necessary to attenuate activity. In summary, taurine must be considered as an essential modulator of neurodevelopmental events, and possible adverse consequences on fetal and/or early postnatal development should be evaluated for pharmacological therapies affecting taurinergic functions.
Introduction
The aminosulfonic acid taurine (2-aminoethanesulfonic acid) is among the most abundant organic molecules in the human body, including the CNS, and has been attributed to a variety of physiological functions (for review Huxtable, 1989; Lambert et al., 2015; Oja and Saransaari, 2015). Taurine is involved in cell volume regulation (Solis et al., 1988; Lambert, 2004), mitochondrial translation (Suzuki et al., 2002), and Ca2+ homeostasis (Chen et al., 2001; El Idrissi, 2008b). Taurine has been suggested to enhance the stability of membranes and directly stabilizes membrane proteins (You and Chang, 1998; Roychoudhury et al., 2013). It modulates inflammation (Marcinkiewicz and Kontny, 2014) and has been shown to reduce apoptosis in a variety of tissues, including the CNS (Taranukhin et al., 2008; Ramos-Mandujano et al., 2014). In addition, taurine possesses an antioxidant effect that, as taurine itself only poorly scavenges radicals (Martincigh et al., 1998), is mediated via indirect effects (Schaffer et al., 2009). In line with this, a variety of studies demonstrated that taurine protects the brain from ischemic or traumatic insults (Sun et al., 2012, 2015; Menzie et al., 2013), including models of perinatal asphyxia (Zhu et al., 2016). Also, taurine is an endogenous agonist of glycine and γ-aminobutyric acid (GABA) receptors (Albrecht and Schousboe, 2005). Therefore, taurine is considered as an endogenous neuromodulator providing an inhibitory effect on the mature CNS. In accordance with this actions, animal studies reported anticonvulsive actions of taurine (El Idrissi et al., 2003; El Idrissi and L’Amoreaux, 2008), which, however, were not completely replicated in humans (reviewed in Oja and Saransaari, 2013). In line with an inhibitory action in the spinal cord, taurine also has as a considerably antinociceptive effect (Pellicer et al., 2007; Terada et al., 2011; Hara et al., 2012). In addition, taurine improves different in vitro correlates of memory formation (Chepkova et al., 2002; del Olmo et al., 2003; Sergeeva et al., 2003) and accordingly augments learning and memory (El Idrissi, 2008a; Neuwirth et al., 2013).
In the immature brain, the taurine concentration is at least 3 times higher than in the adult nervous system, with a considerable downregulation after the first postnatal week in rodents (Huxtable, 1989; Benitez-Diaz et al., 2003). The stimulated taurine release is also significantly larger in immature brains than in adult brains (Oja and Saransaari, 1995). Both observations suggest that taurine may play a particular important role during neuronal development. This suggestion was substantiated by the observations that the development of the visual cortex and the cerebellum was impaired in taurine deficient kitten (Sturman et al., 1985; Palackal et al., 1986). Since these seminal findings of John Sturman, additional studies have been published supporting the hypothesis that taurine is critically involved in a series of neurodevelopmental events. In the following, we like to (i) describe the properties of taurine as neuromodulator in detail and (ii) present recent findings that demonstrate the involvement of taurine on differential neurodevelopmental events.
Taurine Release Mechanisms and Taurine Receptors
For classical neurotransmitter systems, the existence of vesicular transporters, synaptic release mechanisms, and specific receptors has been described. Taurine differs from these substances in some points. First, to our knowledge, no vesicular transports systems for taurine have been identified and taurine release seems to be mainly independent of Ca2+ influx (Kamisaki et al., 1996). The main release pathways for taurine are therefore volume-sensitive organic anion channels (Figure 1A; Qiu et al., 2014; Voss et al., 2014) and/or a reversal of the TauT (SLC6A6; Saransaari and Oja, 2000c). However, in the immature cortex, taurine release seems to occur mainly via volume-sensitive organic anion channels (Figure 1B; Furukawa et al., 2014). Although in the immature nervous system Ca2+-dependent taurine release has been reported (Saransaari and Oja, 1999), later studies demonstrated that this effect is probably secondary to the vesicular release of other neurotransmitters that modulate taurine release (Saransaari and Oja, 2000c). The basal, unstimulated taurine release in the early postnatal CNS has been found to be lower than in the adult CNS (Saransaari and Oja, 2006). However, a variety of stimuli can trigger taurine release in the immature nervous system including volume changes (Oja and Saransaari, 1995), hypoosmotic stimulation (Furukawa et al., 2014), ischemia (Saransaari and Oja, 1999), glutamate, via NMDA, AMPA, and metabotropic receptors (Saransaari and Oja, 1991, 2000a, 2003), and adenosine (Saransaari and Oja, 2000b). In addition, a constitutive taurine release by electrical activity has been observed in the immature neocortex (Figure 1C; Qian et al., 2014).
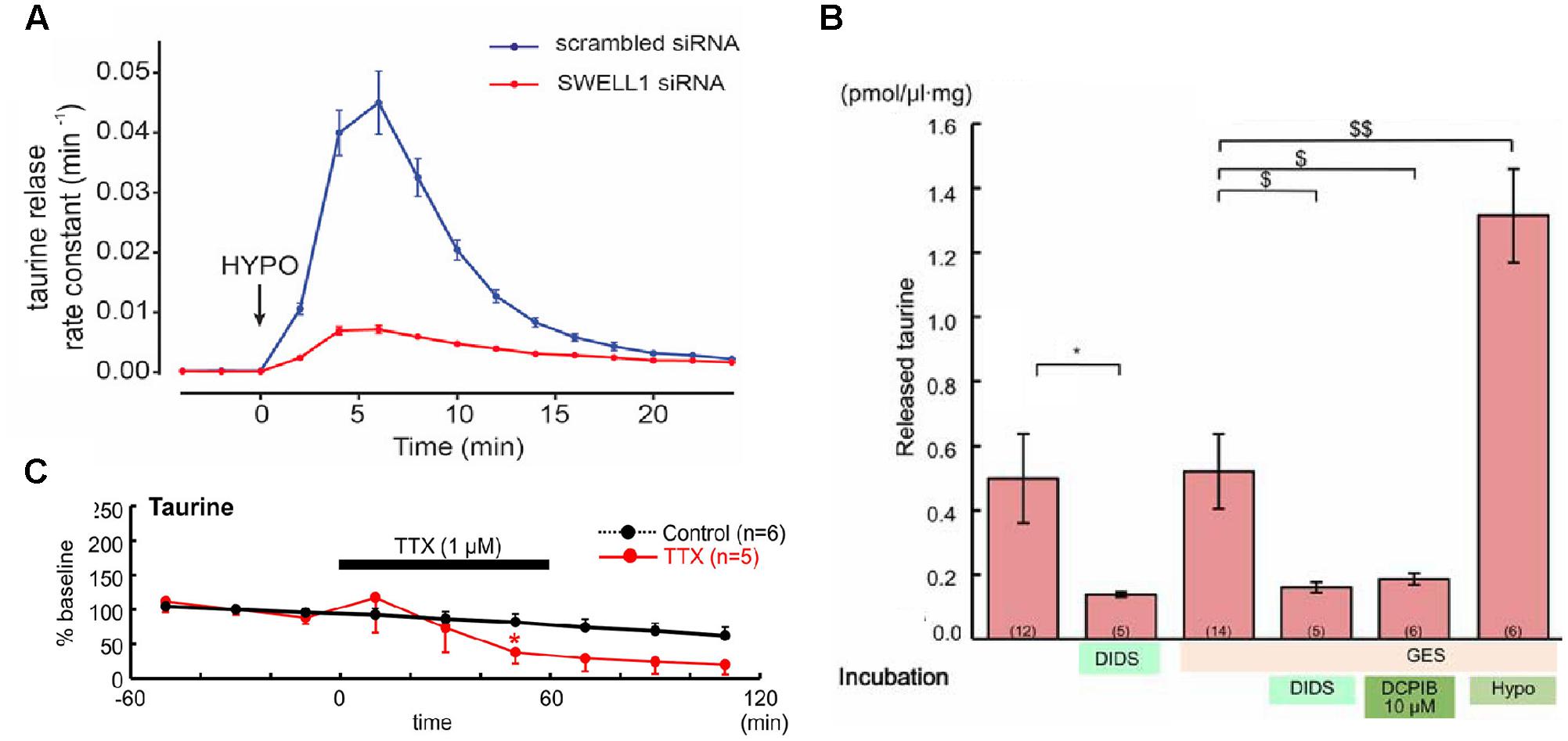
FIGURE 1. Taurine release pathways. (A) The taurine release from HeLa cells after hypoosmotic stimulation was massively attenuated if expression of the volume-regulated anion channel SWELL1 was suppressed (with permission from Qiu et al., 2014). (B) The taurine release from embryonic neocortical slices loaded with 10 mM taurine was not affected by the TauT inhibitor GES, could be blocked the unspecific anion channel blocker DIDS or by DCPIB, a selective blocker of volume-regulated anion channels, and was stimulated by hypoosmotic stimulation (hypo), suggesting that taurine efflux was mainly mediated by volume-regulated anion channels (∗ and $ represent P < 0.05, $$ indicate P < 0.01, with permission from Furukawa et al., 2014). (C) Suppression of electrical activity attenuated the spontaneous taurine release from tangential slices of early postnatal rat neocortex, suggesting the existence of a constitutive, activity-dependent taurine release (modified with permission from Qian et al., 2014).
It is regularly stated, that taurine is a partial, low-affinity agonist on GABAA receptors (Albrecht and Schousboe, 2005). However, recent studies demonstrated that the action of taurine depends critically on the subunit composition of these receptors (Figure 2A). GABAA receptors are heteropentameric complexes composed from total 19 subunits (Farrant and Kaila, 2007). For α1 and α2 containing receptors lacking γ2 subunits, taurine seems to be a full agonist, while addition of γ2 subunits to the pentameric complex reduced the amplitude of maximal taurine currents (Kletke et al., 2013). Interestingly, on α2/β1 receptors, GABA acts as superagonist, albeit with a low affinity (Kletke et al., 2013). For all receptors containing α1 or α2 subunits, a rather low taurine affinity above an EC50 value of 10 mM was observed (Dominguez-Perrot et al., 1996; Kletke et al., 2013). On the other hand, GABAA receptors assembled from α4, β2, and δ subunits, which mediate extrasynaptic currents, have a rather high taurine affinity with an EC50 of 57 μM (Ahring et al., 2016) and taurine mediates a larger current than GABA by these receptor subtypes. Accordingly, relatively low taurine concentrations of 10–100 μM mediate a substantial tonic current in thalamic neurons expressing these receptor subtypes (Jia et al., 2008). GABAA receptors containing α6, β2, and δ subunits, which mediate extrasynaptic currents in the cerebellum, express a partially high-binding state with a rather high taurine affinity (EC50 ca. 6 μM, Hadley and Amin, 2007). Taurine is also a partial agonist of the ionotropic GABAC (ρ subunit-containing GABAA) receptor, albeit again with a rather low affinity (EC50 ca. 5 mM, Ochoa-de la Paz et al., 2008). However, ρ subunit-containing hybrid ionotropic GABA receptors (which combine properties of GABAA and GABAC receptors) seem to be more sensitive to taurine and mediate considerable tonic currents at submillimolar taurine concentrations (Chesnoy-Marchais, 2016). An interesting observation is that micromolar taurine concentrations can massively enhance tonic GABAergic currents, which suggest that extrasynaptic GABA and taurine may act synergistically (Ochoa-de la Paz et al., 2008). In summary, these observations indicate that specific subunit compositions of GABAA receptors can assemble high-affinity taurine receptors and indicate that even μM concentrations of taurine can be sufficient to mediate a physiologically relevant activation of ionotropic GABAA receptor subpopulations. Thus, taurine can be considered as a full agonist with a reasonable affinity for distinct, physiologically relevant GABAA receptor subtypes.
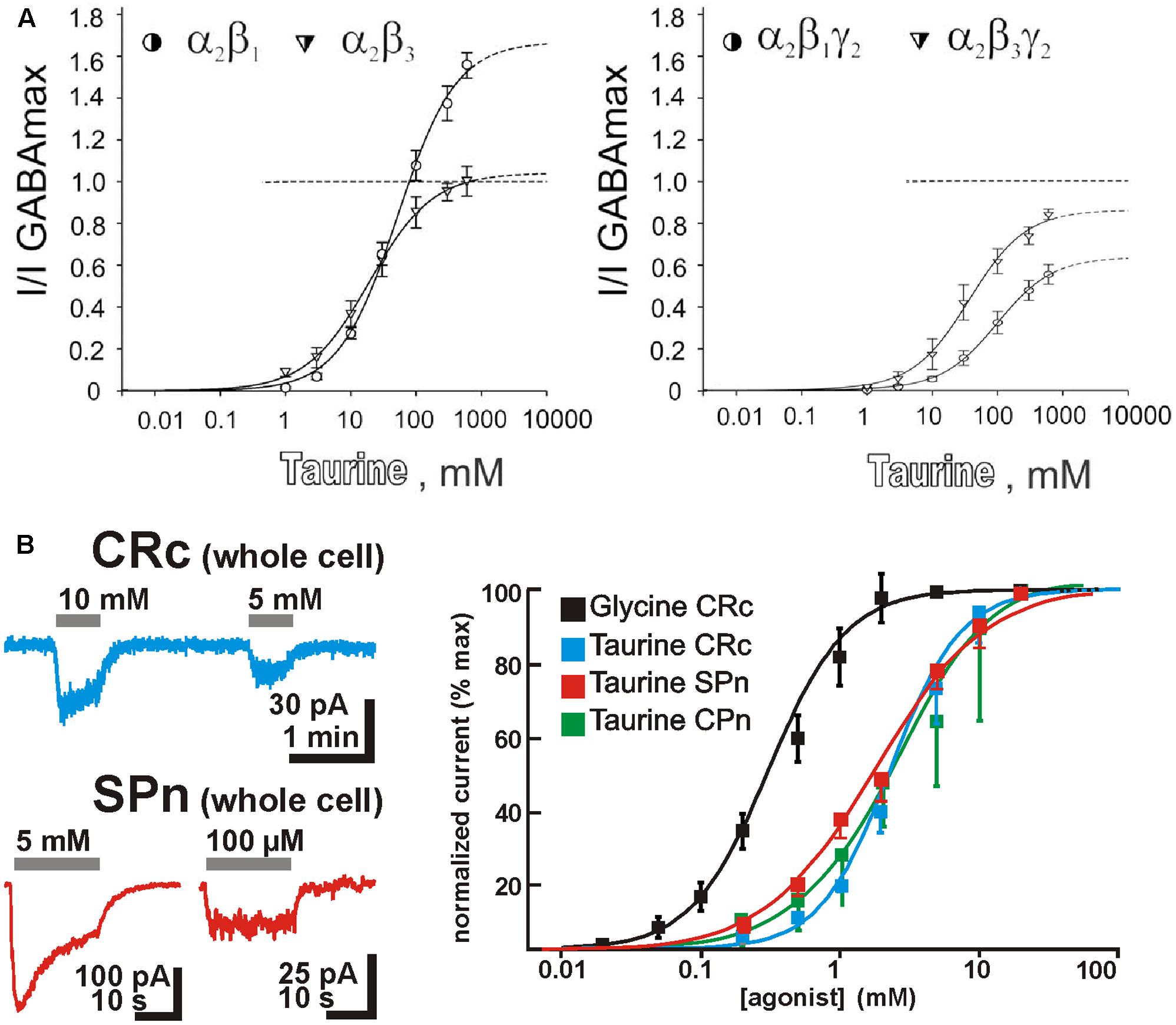
FIGURE 2. Properties of taurine receptors. (A) Taurine affinities and maximal taurine currents critically depend on the subunit composition of GABAA receptors (modified with permission from Kletke et al., 2013). (B) Typical membrane currents and dose–response curves of taurine on glycine receptors for different neuron populations in the immature neocortex revealed that taurine is a low-affinity agonist with comparable affinities in CRc (blue), SP neurons (SPn, red), and neurons from the CP and developing layers (CPn, green) (modified with permission from Kilb et al., 2002, 2008).
In addition to ionotropoic GABAA and GABAC receptors, taurine can also interact with metabotropic GABAB receptors. Baclofen and GABA-replacement experiments suggested that taurine can activate GABAB receptors as a rather high-affinity ligand in the μM range (Kontro and Oja, 1990). In line with this, taurine acts as chemoatractant for migrating neurons via a saclofen-sensitive interaction with GABAB receptors at a concentration of 1 μM (Behar et al., 2001), illustrating the physiological role of GABAB receptors as high-affinity receptors for taurine.
Finally, taurine is also a partial agonist of glycine receptors (Albrecht and Schousboe, 2005). Glycine receptors are composed from four α and one β subunits and form α homomeric or α/β heteromeric receptors (Betz et al., 1999). α1 homomeric receptors have a low taurine affinity with an EC50 of 3.7 mM, while α2 homomeric receptors demonstrate a slightly higher taurine affinity with an EC50 of 2.2 mM (Schmieden et al., 1992). In immature neocortical neurons of the rat, where most probably α2/β heteromeric glycine receptors were expressed, the taurine affinity of these receptors was in the range between 1.1 and 2.4 mM (Figure 2B; Flint et al., 1998; Okabe et al., 2004; Kilb et al., 2008). In summary, these results demonstrate that taurine can be considered as low-affinity agonist for glycine receptors.
In addition to the well-described actions of taurine on GABA and glycine receptors, a few studies also identified NMDA receptors, a ionotropic glutamate receptor subtype, as putative targets of taurine (Suarez and Solis, 2006; Chan et al., 2013). As low as 100 μM taurine is sufficient to augment NMDA-dependent actions on hippocampal fiber volleys, but not postsynaptic NMDA receptors (Suarez and Solis, 2006), suggesting that presynaptic NMDA receptors are potentiated by taurine. In contrast, postsynaptic NMDA receptors seem to be rather inhibited by taurine, although at substantial higher concentration in the mM range (Chan et al., 2014). It was speculated that this postsynaptic inhibition is mediated via an interaction with the allosteric glycine binding site of NMDA receptors (Chan et al., 2014) and depends on NR2B subunits (Chan et al., 2015).
Regarding its nonsynaptic release pathways, taurine can be considered rather as endogenous neuromodulator than as classical neurotransmitter. Thus, it is comparable to other endogenous neuromodulators of GABA receptors, like neurosteroids (Belelli and Lambert, 2005) or the putative endozepine diazepam-binding inhibitor (Christian et al., 2013), of glycine receptors, like Zn2+ (Hirzel et al., 2006), and of NMDA receptors, like D-Serine (Henneberger et al., 2012).
With respect to the observations that different receptors/receptor subtypes have a wide range of affinities for taurine, it is of course essential to know the interstitial taurine concentration in the immature brain. Unfortunately, direct measurements of extracellular taurine concentrations in the immature brain in vivo have not been published. From (i) the taurine concentration of 25 μM measured in the mature CNS under zero-flow conditions by means of microdialysis probes (Molchanova et al., 2004) and (ii) the observation that the total taurine concentration in the immature CNS during the first postnatal week is at least 3 times larger than in the adult CNS (Benitez-Diaz et al., 2003), it can be assumed that the interstitial taurine concentration can reach values above 75 μM. On the other hand, loading experiments suggested that the taurine concentration could be as high as 1 mM in the embryonic neocortex (Furukawa et al., 2014).
Neurotransmitters and Neuronal Activity Influence Corticogenesis
A Short Summary of Neurodevelopmental Events
During development, neurons undergo specific steps of maturation, including neurogenesis, neuronal migration, differentiation, and pruning. The cerebral cortex of vertebrates originates from the two telencephalic vesicles. The earliest neurons generated in the neuroepithelium of this vesicles form the PPL (Angevine and Sidman, 1961). The proliferative zones close to the ventricular surface, termed VZ and SVZ, subsequently continue to generate neurons, which migrate along radial glial processes toward the pial surface and split the PPL into the superficial MZ and the underlying SP. In addition, GABAergic neurons generated in the ganglionic eminences reach the immature neocortex by tangential migration (for review Marín and Rubenstein, 2001). Both MZ and SP are populated by specific neuronal populations, called CRc and SP neurons, respectively (Luhmann et al., 2009; Kirischuk et al., 2014). In between these two transient layers, the CP establishes, which will later develop into layers II to VI of the neocortex (see Bystron et al., 2008 for review). CRc in the MZ play an essential role for cortical lamination and disappear later during development, generating the cell sparse layer I (for review Kirischuk et al., 2014). SP neurons guide the projection of thalamocortical afferents and are transient synaptic targets of these axons before their final targets in layer IV appear (for review Kanold and Luhmann, 2010). The different neuronal populations that build up the mature neocortex later differentiate to form the right connectivity. Subsequently not only a considerable amount of neurons, but also ineffective connections are removed (for review Nikolic et al., 2013). A variety of reports demonstrated that neuronal activity and various neurotransmitters influence cortical development at different levels, ranging from early events as neurogenesis or migration to the establishment of fine-scale neuronal connectivity (for reviews Owens and Kriegstein, 2002; Spitzer, 2006; Wang and Kriegstein, 2009; Luhmann et al., 2016; Khazipov and Milh, 2017).
Immature neuronal activity starts with spontaneous calcium waves that have been observed in mouse neocortical slices already at early embryonic stages in both the VZ (Owens et al., 2000) and the early CP (Corlew et al., 2004). This early in vitro activity was replaced by a distinct set of large-scale network events, beginning with spontaneous and glutamate receptor dependent early network oscillations (Garaschuk et al., 2000), which were followed by giant depolarizing potentials a few days later, characterized by their dependence on depolarizing GABAA receptor-mediated transmission (Allène et al., 2008). In vivo recordings confirmed that spontaneous discontinuous activity occurs already in the early postnatal neocortex (e.g., Khazipov et al., 2004; Hanganu et al., 2006; Yang et al., 2009; Colonnese and Khazipov, 2010). Already at birth local and short network oscillations, termed spindle bursts, in a frequency range of 10–20 Hz occurred, which were a few days later complemented by faster gamma oscillations with a frequency of 30–40 Hz (Khazipov et al., 2013; Yang et al., 2016). This discontinuous activity is generated in the sensory periphery, often independent of physical stimuli, subcortical regions, and in the immature neocortex itself (for review Luhmann et al., 2016; Khazipov and Milh, 2017).
These different patterns of neuronal activity play important roles for various physiological processes during neuronal development. Proliferation in the VZ is directly influenced by spontaneous calcium waves and spontaneous activity projected from the sensory periphery (Weissman et al., 2004; Bonetti and Surace, 2010). Spontaneous rhythmic intracellular calcium waves control neuronal migration (Komuro and Kumada, 2005). In addition, the rate of apoptosis as programmed cell death, which is an essential factor structuring the neocortical circuits, also directly depends on electrical activity (for review Blanquie et al., 2017a). And finally, spontaneous neuronal activity influences growth and differentiation of neuronal dendrites and axonal projections (for reviews Yamamoto and López-Bendito, 2012; Luhmann et al., 2016) and contributes to the formation of topographic maps (for reviews Hanganu-Opatz, 2010; Assali et al., 2014; Luhmann and Khazipov, 2017). While for many of these activity patterns, a vesicular release of GABA and glutamate is essential, recent studies identified taurine as a factor that contribute to GABAergic effects on neuronal development and also directly controls activity levels in the immature CNS as follows.
Taurine Affects Corticogenesis
The neurogenesis in the VZ and SVZ is influenced by GABAA receptors (LoTurco et al., 1995). Activation of GABA or glycine receptors also directly controls neuronal migration (for review Luhmann et al., 2015). Activation of GABAA receptors induces apoptosis in CRc (Blanquie et al., 2017b), but promotes neuronal differentiation and synaptogenesis in principal cells and interneurons (Maric et al., 2001; Meier et al., 2003; see Wang and Kriegstein, 2009 for review). However, it should be noted in this respect that none of the mentioned studies unequivocally identified GABA as the endogenous neurotransmitter mediating these actions, but only demonstrated that GABAA receptors are required. In fact, it has been shown that (i) neuronal migration is influenced by GABAA receptors even in the absence of synaptic neurotransmitter release (Manent et al., 2005) and (ii) tonic GABAA receptor mediated currents in embryonic neurons are unaffected by a massive GABA depletion, but sensitive to conditions enhancing extracellular taurine levels (Furukawa et al., 2014), allowing the speculation that taurine may contribute as endogenous agonist to the reported effects. With respect to the GABAergic and glycinergic actions, it must be considered that both neurotransmitters mediate a depolarizing action in the immature CNS, due to a high expression rate of the chloride loader NKCC1 and a low expression of the chloride extruder KCC2 (Yamada et al., 2004; Watanabe and Fukuda, 2015). Taurine is directly involved in this process, as it mediates an inhibition of KCC2 function via phosphorylation in immature neurons, thus maintaining a depolarizing action of GABAA or glycine receptors (Inoue et al., 2012). In the following, we will summarize the action of taurine on distinct neuronal subpopulations and discuss how this interaction can interfere with neuronal development.
Action of Taurine on Identified Neuronal Populations in the Developing Brain
Neural Stem Cells
In the SVZ of the cortex, the activation of GABAA receptors significantly reduced neurogenesis (LoTurco et al., 1995; Haydar et al., 2000). The observation that pharmacological inhibition of GABAA receptors enhances the proliferation (LoTurco et al., 1995) indicates that an intrinsic agonist of GABAA receptors controls neurogenesis. In the VZ, however, activation of GABAA receptors increases proliferation (Haydar et al., 2000), probably by preventing neuroblast from exiting the proliferative cycle toward G0 phase. Since the proliferation of neuroblasts in the postnatal SVZ is limited by a nonsynaptic release of GABAergic agonists (Liu et al., 2005), it is intriguing to speculate that taurine contributes to these effects. Neuroepithelial stem cells directly contact cerebrospinal fluid (Lehtinen and Walsh, 2011; Lehtinen et al., 2011; Chau et al., 2015), which at least during early developmental stages subsequently to neural tube closing is consisting mainly of amniotic fluid, in which taurine is accumulated from maternal blood (Sturman et al., 1977; Sturman, 1981).
Recently it has been demonstrated that 10 mM taurine indeed increases the proliferation of mice embryonic progenitor cells in vitro (Hernandez-Benitez et al., 2010; Figure 3A). Similar effects were also found for human neuronal precursor cells, where proliferation is also enhanced by millimolar amounts of taurine, albeit it is not clear whether this process requires GABAA or glycine receptors (Hernandez-Benitez et al., 2013). For hippocampal progenitors, it has also been demonstrated that 100 μM taurine augment proliferation (Shivaraj et al., 2012). In the mouse cochlea, millimolar taurine concentrations enhance proliferation of stem cells, but augment only the number of glutamatergic neurons, while the number of GABAergic neurons descending from cochlear stem cells is decreased (Wang et al., 2015). These results suggest that taurine can also provide a signal that determines the composition of neuronal elements within a circuitry. A similar observation was also made in the retina, where 100 μM taurine acting via glycine receptors stimulate the differentiation of retinal progenitor cells toward rod photoreceptors (Altshuler et al., 1993; Young and Cepko, 2004).
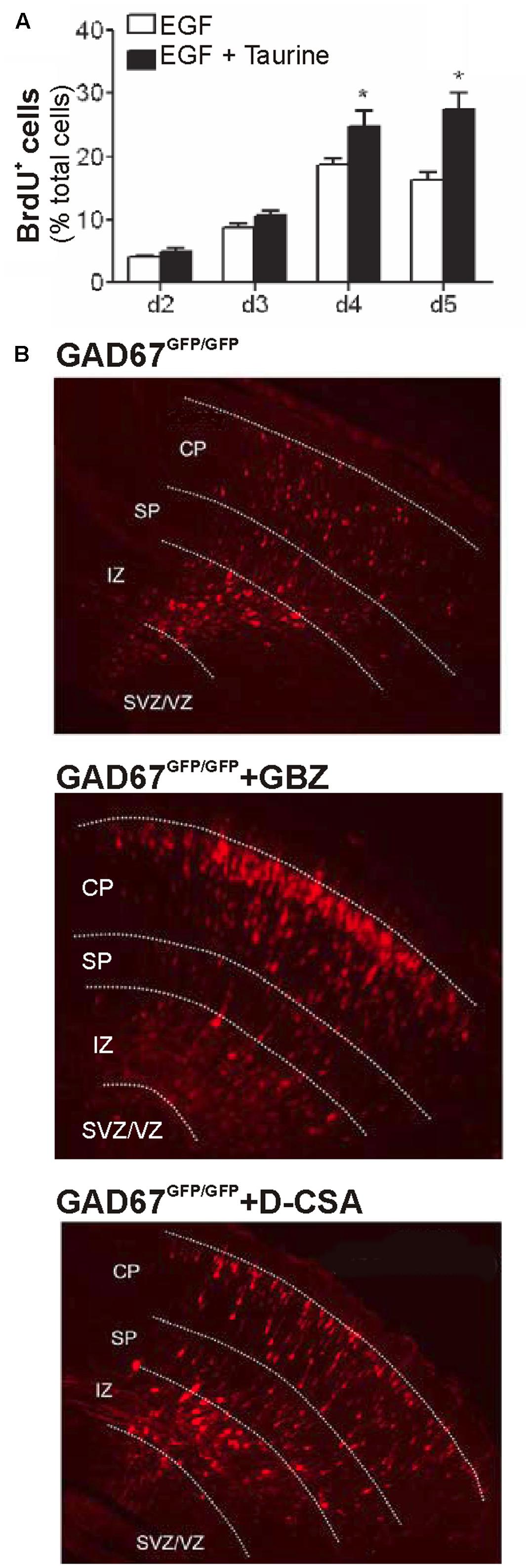
FIGURE 3. Effect of taurine on proliferation and migration. (A) Taurine (10 mM) enhances the fraction of BrdU positive neurons in mouse neurospheres after 4–5 days in culture, indicating that taurine promotes proliferation (∗ indicate P < 0.05, with permission from Hernandez-Benitez et al., 2010). (B) Radial migration of RFP-labeled neurons in the substantially GABA-depleted GAD67-GFP mice can be enhanced by inhibition of GABAA receptors with gabazine (middle image) and by inhibition of maternal taurine synthesis with D-cysteinate (D-CSA, lower image), indicating that taurine acting of GABAA receptors modulate radial migration in vivo (modified with permission from Furukawa et al., 2014).
Intriguingly, electrophysiological experiments failed to reveal taurine-induced membrane currents in cells of the VZ (Flint et al., 1998), suggesting that these cells probably did not express taurine-sensitive glycine receptors. However, as from this publication, it is not clear whether the taurine experiments were performed in the presence of GABAergic antagonists, taurine probably affect neuroblasts exclusively via GABAA receptors functionally expressed in VZ neuroblasts (LoTurco et al., 1995; Ma and Barker, 1995; Tochitani et al., 2010). On the other hand, it should also be considered that activation of GABAA and/or glycine receptors can also influence proliferation indirectly, e.g., via interactions with bFGF or brain-derived neurotrophic factor (BDNF) release (Berninger et al., 1995; Antonopoulos et al., 1997).
Interestingly, taurine can also enhance adult neurogenesis under both in vitro and in vivo conditions (Hernandez-Benitez et al., 2012; Ramos-Mandujano et al., 2014; Gebara et al., 2015). This enhanced adult neurogenesis may underlie the beneficial effect of chronic taurine administration on various learning paradigms in adult mice (El Idrissi, 2008a; Neuwirth et al., 2013; Kim et al., 2014), although it should be noted that taurine can also directly affect in vitro correlates of memory formation (Chepkova et al., 2002; del Olmo et al., 2003; Sergeeva et al., 2003).
Migrating Neurons
Already Sturman et al. (1985) suggested that in taurine deficient kitten neuronal migration in the cerebellum was hampered. In the visual cortex of these animals, clear indications for massively impaired migration were observed (Palackal et al., 1986). Taurine induced small inward currents in putatively migrating neurons of the rat cortical IZ at E19 via an activation of glycine receptors (Flint et al., 1998), whereas the taurinergic currents in clearly identified radially migrating neurons of the mouse neocortex were exclusively mediated via GABAA receptors (Furukawa et al., 2014). In addition, it has been shown that GABAA receptors (Barker et al., 1998; Heck et al., 2007), GABAB receptors (Behar et al., 2001), GABAC receptors (Denter et al., 2010), and glycine receptors (Nimmervoll et al., 2011), all possible targets of taurine, affect neuronal migration. Regarding the GABAB receptors, Behar et al. (2001) directly demonstrated that taurine acts as potent chemoatractant for migrating neurons via an interaction with GABAB receptors.
In dissociated cultures of cerebellar granule cells, taurine depletion indeed attenuated neuronal migration (Maar et al., 1995), which replicates the in vivo observation of Sturman et al. (1985) in the cerebellum of taurine deficient kitten. Recent in vivo studies by Furukawa et al. (2014) utilized GABA-deficient mice to specify the role of taurine for radial migration. Interestingly, they observed that radial migration was not significantly affected in a homozygous GAD-67 mouse, in which the GABA content was reduced to 12.7% of the wild-type level. However, inhibition of GABAA receptors accelerated radial migration in this GAD-67 deficient mouse to a similar extent as in wild-type animals (Furukawa et al., 2014; Figure 3B), suggesting that GABA is not required as endogenous ligand for the GABAA receptors regulating migration. This suggestion was substantiated by the findings that (i) the tonic currents in migrating neurons were unaffected by GABA depletion but (ii) enhanced in the presence of the TauT blocker GES, and (iii) attenuated after blockade of taurine synthesis with D-cystein (Furukawa et al., 2014; Figure 3B). And in line with these in vitro experiments, indicating that taurine is probably the most relevant endogenous agonist of extrasynaptic GABAA receptors, depletion of taurine by maternal D-cystein administration accelerated migration in both wild-type and homozygous GAD-67 mice in vivo (Furukawa et al., 2014). In summary, these experiments provide mechanistic evidences that taurine is indeed a major endogenous modulator of radial migration.
Cajal–Retzius Cells
In CRc, taurine-induced inward currents were mediated by glycine receptors with a low affinity (EC50 = 2.4 mM, Kilb et al., 2002; Figure 2B). In line with the high intracellular Cl- concentration in these cells (Achilles et al., 2007), activation of glycine receptors mediate a membrane depolarization in CRc (Kilb et al., 2002). Qian et al. (2014) were able to demonstrate that a strong electrical stimulation indeed stimulates the release of taurine and GABA, while glycine was not released. Voltage sensitive dye imaging revealed that such strong electrical stimulation induced a rapidly propagating wave of depolarization in the MZ of tangential slices. This propagating activity was insensitive to glutamate receptor blockade, but was partially attenuated by the application of either GABAergic or glycinergic antagonist, while application of both GABAergic and glycinergic antagonists completely abolished this activity (Qian et al., 2014). Because microdialysis experiments performed in this study demonstrate that glycine was not released (Qian et al., 2014), these results suggest a contribution of taurine. Since this propagating activity was observed at the pial surface of tangential slices in the MZ, most probably CRc contribute to this activity. Accordingly, whole-cell recordings from CRc revealed that these electrical stimuli induced a strong inward current that was also insensitive to glutamatergic antagonists, only partially reduced by GABAergic and glycinergic antagonists and completely blocked if GABAergic and glycinergic antagonists were combined (Qian et al., 2014). Since taurine was not localized in the presynaptic structures and glycine was not released after electrical stimulation (Qian et al., 2014), these results suggest that, although a substantial portion of this tangentially propagating activity transients is mediated by synaptic release of GABA, taurine acting on GABAA and glycine receptors in CRc also considerably contributed to the propagation of activity in the MZ in a neuromodulatory fashion.
Neurons in the Cortical Plate or Developing Cortical Layers
Taurine activates cortical neurons via glycine receptors at a rather low affinity with an EC50 of about 1 mM (Flint et al., 1998; Okabe et al., 2004; Figure 2B). While no evidences for a synaptic activation of glycine receptor were found, inhibition of taurine transport with GES, hypoosmotic stimuli, and strong electrical stimulation evoke a putative nonsynaptic release of taurine, which activated glycine and probably also GABAA receptors (Flint et al., 1998; Furukawa et al., 2014; Qian et al., 2014). Also in cortical neurons, the activation of glycine receptors mediates an excitatory effect, as concluded from taurine-induced Ca2+ transients and the fact that focal glycine application enhances the frequency of GABAergic PSCs (Flint et al., 1998). Sava et al. (2014) were able to replicate these findings and found that a prolonged application of 300 μM taurine induced a tonic inward current in putative projection neurons of the CP, which was in about 50% of the cells associated with a massive increase in GABAergic PSCs (Figure 4A). Pharmacological experiments revealed that about 80% of the tonic taurine-induced currents in these neurons are mediated via glycine, and about 20% via GABAA receptors (Sava et al., 2014). The taurine-induced GABAergic responses are excitatory (Figure 4B) and are abolished in the presence of the glycinergic antagonist strychnine. As similar GABAergic PSCs could also be evoked by tonic application of glycine (Sava et al., 2014), it is concluded that glycine receptors on the presynaptic cells are essential to generate the GABAergic PSCs. Further analysis of the presynaptic GABAergic interneurons, which were visually identified in GAD67-GFP transgenic animals (Tamamaki et al., 2003), demonstrated that taurine induces a strychnine-sensitive inward current (Figure 4C), which was associated with a massive increase in action potential discharges in the majority of GABAergic interneurons (Figure 4D; Sava et al., 2014). This observation demonstrates that taurine indeed excites GABAergic interneurons. Further experiments revealed that these taurine-induced GABAergic PSCs increase the frequency of action potential discharges in putative pyramidal neurons (Sava et al., 2014). In summary, these studies demonstrated that taurine has an excitatory net effect in immature neuronal circuits in situ and thus most probably contribute network activity in the developing neocortex. However, it will be necessary to determine whether taurine contributes to excitation also in vivo, since recent experiments demonstrated depolarizing, but inhibitory effects of GABAA receptors in the developing neocortex under in vivo conditions (Kirmse et al., 2015).
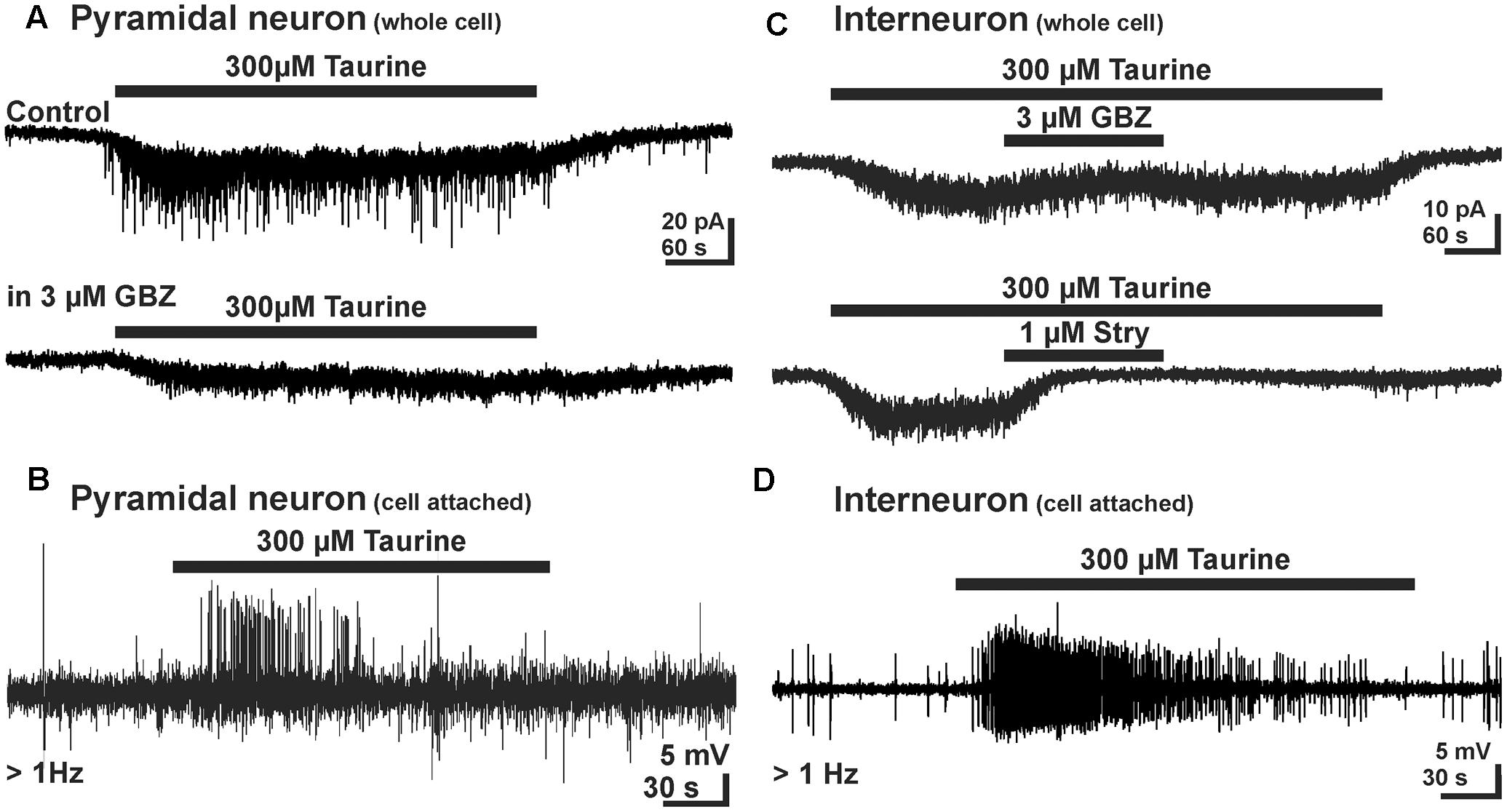
FIGURE 4. Effect of taurine on GABAergic networks in early postnatal mouse neocortex. (A) In pyramidal neurons taurine induced a tonic inward current and increased the frequency of GBZ-sensitive GABAergic PSCs. (B) Cell-attached recordings demonstrating that GABAergic PSCs enhance action potential frequency, suggesting that the taurine-induced GABAergic PSCs are excitatory. (C) In GABAergic interneurons taurine induced an inward-current that was relatively insensitive to GBZ, but suppressed by strychnine, indicating that taurine acts mainly via glycine receptors in this cell type. (D) Cell-attached recordings from GABAergic interneurons demonstrate that the taurine-induced inward current enhances action potential frequency, suggesting that the taurine is an excitatory neuromodulator in immature interneurons (with permission from Sava et al., 2014).
Subplate Neurons
For SP neurons, it was also demonstrated that taurine activates glycine receptors with a low affinity (EC50 = 1.7 mM; Kilb et al., 2008). While higher taurine concentrations evoke desensitizing responses, 100 μM taurine induced a tonic inward current in this cell type (Figure 2B). Also in SP neurons, the activation of glycine receptors mediates a depolarizing action (Kilb et al., 2008). Further analyses revealed that tonic taurinergic currents, although evoking only small subthreshold depolarizations, substantially lower the action potential threshold, thus demonstrating an obvious excitatory effect on SP neurons. Inhibition of the taurine transport with GES as well as hypotonic stimulation induced inward currents, which suggests that also in the SP neurons taurine can be an endogenous agonist of glycine receptors (Kilb et al., 2008). In summary, these experiments indicate that taurine can act as neuromodulator in the SP. As a fraction of SP neurons are GABAergic, it was also investigated whether SP neurons contribute to the taurine-induced GABAergic PSCs in pyramidal neurons (see section “Neurons in the Cortical Plate or Developing Cortical Layers”). However, these experiments revealed that ablation of the SP does not significantly reduce the frequency of taurine-induced GABAergic PSCs, demonstrating that GABAergic projections from SP to pyramidal neurons do not considerably contribute to the taurine-induced activity in the CP (Sava et al., 2014). On the other hand, ambient taurine level could be highest in the embryonic SP (Furukawa et al., 2014). Since the SP is a crucial element for structural and functional development of the neocortex (for review Kanold and Luhmann, 2010) and is essentially involved in the regulation of excitability in the developing neocortex (Dupont et al., 2006; Hanganu et al., 2009), further experiments are needed to evaluate whether SP neurons are the particularly important target for the neurodevelopmental effects of taurine.
Summary
Taurine can be considered as an important neurodevelopmental modulator (Figure 5). Taurine-mediated currents were identified on most major neuronal populations in the immature neocortex (Kilb, 2017). Due to its depolarizing effect on most investigated neuronal populations in the neocortex, taurine can directly induce intracellular Ca2+ transients, which are causal for many neurodevelopmental events like migration or differentiation (Spitzer et al., 2000; Komuro and Kumada, 2005). In addition, taurine increases neuronal excitability in most neocortical neuron types due to this depolarization. In accordance with this single-cell effects, it was shown that taurine increase the network activity in the immature CP in vitro (Sava et al., 2014). Taurine can therefore also indirectly impact neuronal development via these effects on activity patterns (for review Wang and Kriegstein, 2009; Luhmann et al., 2016). In the immature hippocampus, it has also be shown that nonsynaptic taurine can modulate neural excitability, dose dependently in both excitatory and inhibitory directions (Chen et al., 2014), due to a consistent depolarizing effect in combination with a dominant shunting effect at higher taurine concentrations. Selective inhibition of glycine receptors with strychnine induces in these immature hippocampal preparations epileptiform activity (Chen et al., 2014), indicating that taurine, as main endogenous agonist of glycine receptors in the immature CNS, most probably contribute to inhibitory net effects in the immature hippocampus. The next important step in the evaluation of taurinergic actions on the activity of immature networks would therefore be to unravel the direct effects of taurine under in vivo conditions, since in this situation activation of GABAA receptors mediates depolarizing but inhibitory effects in the developing neocortex (Kirmse et al., 2015). Given that in vitro taurine mediates mainly excitatory network effects in the neocortex, but an inhibitory net action in the hippocampus, it would be interesting to uncover the effect of taurine on the excitability in other regions of the CNS, like thalamus, basal ganglia, hypothalamic nuclei, amygdala, and the spinal cord. The essential role of taurine for neuronal development entails that any interference with the taurinergic system during pregnancy or early childhood increases the risk for developmental disorders. Accordingly, the adverse effect of the antiepileptic drug vigabatrin, which severely impaired neuronal development (Manent et al., 2007), has already been correlated to a disturbed taurine homeostasis under these conditions (Jammoul et al., 2009).
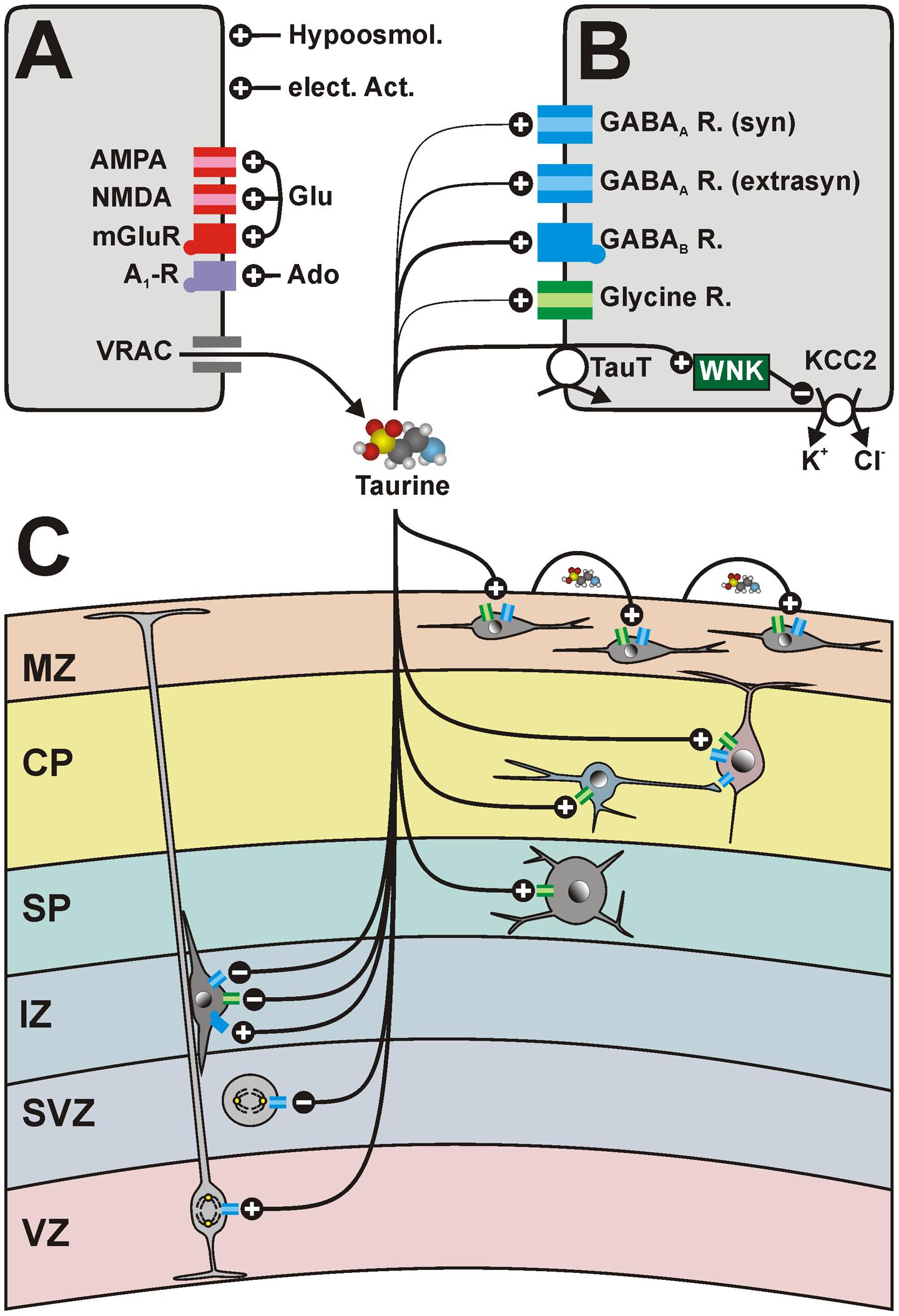
FIGURE 5. Schematic diagram summarizing the effects of taurine on the immature neocortex. (A) Taurine release is mediated mainly by volume-regulated anion channels (VRAC). The release of taurine is activated by hypoosmotic conditions, electrical activity and via glutamate (Glu), and adenosine (Ado) receptors. (B) Taurine mediates its effects via low-affinity binding to glycine receptors (green symbols) or GABAA receptors (blue symbols) with subunit compositions typical for synaptic receptors. While the taurine affinity to putatively extrasynaptic GABAA receptors is moderate, taurine is a high-affinity ligand for GABAB receptors. In addition, the intracellular taurine concentration, regulated by the TauT, suppresses the function of the Cl- extruder KCC2 via activation of the WNK pathway, thus maintaining depolarizing taurinergic membrane responses. (C) Putative effect of taurine on different cell populations in the developing neocortex. Taurine promotes proliferation in the VZ, but attenuates proliferation in the SVZ. It stimulates chemotaxis via GABAB receptors and suppresses radial migration via GABAA and glycine receptors. Taurine depolarizes SP neurons, pyramidal cell and GABAergic interneurons in the CP, as well as CRc in the MZ via activation of GABAA and/or glycine receptors. The taurinergic depolarization of GABAergic interneurons is in vitro sufficient to generate GABAergic network activity transmitted to pyramidal cells. CRc participate to propagating activity in the MZ mediated by activity-dependent taurine release. See text for details.
Author Contributions
WK and AF drafted, wrote, and revised the text. WK and AF approved the final version of the manuscript and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
WK received support from the Deutsche Forschungsgemeinschaft (DFG-Grant KI 835/2-3). AF received support from Japan Society for the Promotion of Science (15H05872, 17H04025, and 17K19682).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
WK and AF thank all colleagues, who dedicated their work to a better understanding of the physiological role of taurine. We apologize that we were not able to cite all publications that were related to this issue due to space limitations. We thank our coworkers and the funding agencies, especially the Deutsche Forschungsgemeinschaft and the Ministry of Education, Culture, Sports, Science, and Technology of Japan for continuous support.
Abbreviations
BDNF, brain derived neurotrophic factor; bFGF, basic fibroblast growth factor; CNS, central nervous system; CP, cortical plate; CRc, Cajal–Retzius cells; GABA, gamma-aminobutyric acid; IZ, intermediate zone; MZ, marginal zone; PPL, primordial plexiform layer; PSCs, postsynaptic currents; SP, subplate; SVZ, subventricular zone; TauT, taurine transporter; VZ, ventricular zone.
References
Achilles, K., Okabe, A., Ikeda, M., Shimizu-Okabe, C., Yamada, J., Fukuda, A., et al. (2007). Kinetic properties of Cl- uptake mediated by Na+-dependent K+-2Cl- cotransport in immature rat neocortical neurons. J. Neurosci. 27, 8616–8627. doi: 10.1523/JNEUROSCI.5041-06.2007
Ahring, P. K., Bang, L. H., Jensen, M. L., Strobaek, D., Hartiadi, L. Y., Chebib, M., et al. (2016). A pharmacological assessment of agonists and modulators at α4β2γ2 and α4β2δ GABAA receptors: The challenge in comparing apples with oranges. Pharmacol. Res. 111, 563–576. doi: 10.1016/j.phrs.2016.05.014
Albrecht, J., and Schousboe, A. (2005). Taurine interaction with neurotransmitter receptors in the CNS: an update. Neurochem. Res. 30, 1615–1621. doi: 10.1007/s11064-005-8986-6
Allène, C., Cattani, A., Ackman, J. B., Bonifazi, P., Aniksztejn, L., Ben-Ari, Y., et al. (2008). Sequential generation of two distinct synapse-driven network patterns in developing neocortex. J. Neurosci. 28, 12851–12863. doi: 10.1523/JNEUROSCI.3733-08.2008
Altshuler, D., LoTurco, J. J., Rush, J., and Cepko, C. (1993). Taurine promotes the differentiation of a vertebrate retinal cell type in vitro. Development 119, 1317–1328.
Angevine, J. B., and Sidman, R. L. (1961). Autoradiographic study of cell migration during histogenesis of cerebral cortex in the mouse. Nature 192, 766–768. doi: 10.1038/192766b0
Antonopoulos, J., Pappas, I. S., and Parnavelas, J. G. (1997). Activation of the GABAA receptor inhibits the proliferative effects of bFGF in cortical progenitor cells. Eur. J. Neurosci. 9, 291–298. doi: 10.1111/j.1460-9568.1997.tb01399.x
Assali, A., Gaspar, P., and Rebsam, A. (2014). Activity dependent mechanisms of visual map formation–from retinal waves to molecular regulators. Semin. Cell. Dev. Biol. 35, 136–146. doi: 10.1016/j.semcdb.2014.08.008
Barker, J. L., Behar, T., Li, Y. X., Liu, Q. Y., Ma, W., Maric, D., et al. (1998). GABAergic cells and signals in CNS development. Perspect. Dev. Neurobiol. 5, 305–322.
Behar, T. N., Smith, S. V., Kennedy, R. T., McKenzie, J. M., Maric, I., and Barker, A. L. (2001). GABAB receptors mediate motility signals for migrating embryonic cortical cells. Cereb. Cortex 11, 744–753. doi: 10.1093/cercor/11.8.744
Belelli, D., and Lambert, J. J. (2005). Neurosteroids: endogenous regulators of the GABA(A) receptor. Nat. Rev. Neurosci. 6, 565–575. doi: 10.1038/nrn1703
Benitez-Diaz, P., Miranda-Contreras, L., Mendoza-Briceno, R. V., Pena-Contreras, Z., and Palacios-Pru, E. (2003). Prenatal and postnatal contents of amino acid neurotransmitters in mouse parietal cortex. Dev. Neurosci. 25, 366–374. doi: 10.1159/000073514
Berninger, B., Marty, S., Zafra, F., Berzaghi, M. D., Thoenen, H., and Lindholm, D. (1995). GABAergic stimulation switches from enhancing to repressing BDNF expression in rat hippocampal-neurons during maturation in-vitro. Development 121, 2327–2335.
Betz, H., Kuhse, J., Schmieden, V., Laube, B., Kirsch, J., and Harvey, R. J. (1999). Structure and functions of inhibitory and excitatory glycine receptors. Ann. N. Y. Acad. Sci. 868, 667–676. doi: 10.1111/j.1749-6632.1999.tb11343.x
Blanquie, O., Kilb, W., Sinning, A., and Luhmann, H. J. (2017a). Homeostatic interplay between electrical activity and neuronal apoptosis in the developing neocortex. Neuroscience 358, 190–200. doi: 10.1016/j.neuroscience.2017.06.030
Blanquie, O., Liebmann, L., Hubner, C. A., Luhmann, H. J., and Sinning, A. (2017b). NKCC1-mediated GABAergic signaling promotes postnatal cell death in neocortical Cajal-Retzius cells. Cereb. Cortex 27, 1644–1659. doi: 10.1093/cercor/bhw004
Bonetti, C., and Surace, E. M. (2010). Mouse embryonic retina delivers information controlling cortical neurogenesis. PLOS ONE 5:e15211. doi: 10.1371/journal.pone.0015211
Bystron, I., Blakemore, C., and Rakic, P. (2008). Development of the human cerebral cortex: boulder committee revisited. Nat. Rev. Neurosci. 9, 110–122. doi: 10.1038/nrn2252
Chan, C. Y., Singh, I., Magnuson, H., Zohaib, M., Bakshi, K. P., Le Francois, B., et al. (2015). Taurine targets the GluN2b-containing NMDA receptor subtype. Adv. Exp. Med. Biol. 803, 531–544. doi: 10.1007/978-3-319-15126-7_\43
Chan, C. Y., Sun, H. S., Shah, S. M., Agovic, M. S., Friedman, E., and Banerjee, S. P. (2014). Modes of direct modulation by taurine of the glutamate NMDA receptor in rat cortex. Eur. J. Pharmacol. 728, 167–175. doi: 10.1016/j.ejphar.2014.01.025
Chan, C. Y., Sun, H. S., Shah, S. M., Agovic, M. S., Ho, I., Friedman, E., et al. (2013). Direct interaction of taurine with the NMDA glutamate receptor subtype via multiple mechanisms. Adv. Exp. Med. Biol. 775, 45–52. doi: 10.1007/978-1-4614-6130-2_\4
Chau, K. F., Springel, M. W., Broadbelt, K. G., Park, H. Y., Topal, S., Lun, M. P., et al. (2015). Progressive differentiation and instructive capacities of amniotic fluid and cerebrospinal fluid proteomes following neural tube closure. Dev. Cell. 35, 789–802. doi: 10.1016/j.devcel.2015.11.015
Chen, R. Q., Okabe, A., Sun, H. Y., Sharopov, S., Hanganu-Opatz, I. L., Kolbaev, S. N., et al. (2014). Activation of glycine receptors modulates spontaneous epileptiform activity in the immature rat hippocampus. J. Physiol. 592, 2153–2168. doi: 10.1113/jphysiol.2014.271700
Chen, W. Q., Jin, H., Nguyen, M., Carr, J., Lee, Y. J., Hsu, C. C., et al. (2001). Role of taurine in regulation of intracellular calcium level and neuroprotective function in cultured neurons. J. Neurosci. Res. 66, 612–619. doi: 10.1002/jnr.10027
Chepkova, A. N., Doreulee, N., Yanovsky, Y., Mukhopadhyay, D., Haas, H. L., and Sergeeva, O. A. (2002). Long-lasting enhancement of corticostriatal neurotransmission by taurine. Eur. J. Neurosci. 16, 1523–1530. doi: 10.1046/j.1460-9568.2002.02223.x
Chesnoy-Marchais, D. (2016). Persistent GABAA/C responses to gabazine, taurine and beta-alanine in rat hypoglossal motoneurons. Neuroscience 330, 191–204. doi: 10.1016/j.neuroscience.2016.05.048
Christian, C. A., Herbert, A. G., Holt, R. L., Peng, K., Sherwood, K. D., Pangratz-Fuehrer, S., et al. (2013). Endogenous positive allosteric modulation of GABA(A) receptors by diazepam binding inhibitor. Neuron 78, 1063–1074. doi: 10.1016/j.neuron.2013.04.026
Colonnese, M. T., and Khazipov, R. (2010). ”Slow activity transients” in infant rat visual cortex: a spreading synchronous oscillation patterned by retinal waves. J. Neurosci. 30, 4325–4337. doi: 10.1523/JNEUROSCI.4995-09.2010
Corlew, R., Bosma, M. M., and Moody, W. J. (2004). Spontaneous, synchronous electrical activity in neonatal mouse cortical neurons. J. Physiol. 560, 377–390. doi: 10.1113/jphysiol.2004.071621
del Olmo, N., Handler, A., Alvarez, L., Bustamante, J., del Rio, R. M., and Solis, J. M. (2003). Taurine-induced synaptic potentiation and the late phase of longterm potentiation are related mechanistically. Neuropharmacology 44, 26–39. doi: 10.1016/S0028-3908(02)00310-6
Denter, D. G., Heck, N., Riedemann, T., White, R., Kilb, W., and Luhmann, H. J. (2010). GABA(C) receptors are functionally expressed in the intermediate zone and regulate radial migration in the embryonic mouse neocortex. Neuroscience 167, 124–134. doi: 10.1016/j.neuroscience.2010.01.049
Dominguez-Perrot, C., Feltz, P., and Poulter, M. O. (1996). Recombinant GABA(A) receptor desensitization: the role of the gamma(2) subunit and its physiological significance. J. Physiol. 497, 145–159. doi: 10.1113/jphysiol.1996.sp021756
Dupont, E., Hanganu, I. L., Kilb, W., Hirsch, S., and Luhmann, H. J. (2006). Rapid developmental switch in the mechanisms driving early cortical columnar networks. Nature 439, 79–83. doi: 10.1038/nature04264
El Idrissi, A. (2008a). Taurine improves learning and retention in aged mice. Neurosci. Lett. 436, 19–22. doi: 10.1016/j.neulet.2008.02.070
El Idrissi, A. (2008b). Taurine increases mitochondrial buffering of calcium: role in neuroprotection. Amino Acids 34, 321–328.
El Idrissi, A., and L’Amoreaux, W. J. (2008). Selective resistance of taurine-fed mice to isoniazide-potentiated seizures: in vivo functional test for the activity of glutamic acid decarboxylase. Neuroscience 156, 693–699. doi: 10.1016/j.neuroscience.2008.07.055
El Idrissi, A., Messing, J., Scalia, J., and Trenkner, E. (2003). Prevention of epileptic seizures by taurine. Adv. Exp. Med. Biol. 526, 515–525. doi: 10.1007/978-1-4615-0077-3_\62
Farrant, M., and Kaila, K. (2007). The cellular, molecular and ionic basis of GABA(A) receptor signalling. Prog. Brain Res. 160, 59–87. doi: 10.1016/S0079-6123(06)60005-8
Flint, A. C., Liu, X. L., and Kriegstein, A. R. (1998). Nonsynaptic glycine receptor activation during early neocortical development. Neuron 20, 43–53. doi: 10.1016/S0896-6273(00)80433-X
Furukawa, T., Yamada, J., Akita, T., Matsushima, Y., Yanagawa, Y., and Fukuda, A. (2014). Roles of taurine-mediated tonic GABA(A) receptor activation in the radial migration of neurons in the fetal mouse cerebral cortex. Front. Cell. Neurosci. 8:88. doi: 10.3389/fncel.2014.00088
Garaschuk, O., Linn, J., Eilers, J., and Konnerth, A. (2000). Large-scale oscillatory calcium waves in the immature cortex. Nat. Neurosci. 3, 452–459. doi: 10.1038/74823
Gebara, E., Udry, F., Sultan, S., and Toni, N. (2015). Taurine increases hippocampal neurogenesis in aging mice. Stem Cell Res. 14, 369–379. doi: 10.1016/j.scr.2015.04.001
Hadley, S. H., and Amin, J. (2007). Rat α6β2δ GABAA receptors exhibit two distinct and separable agonist affinities. J. Physiol. 581, 1001–1018. doi: 10.1113/jphysiol.2007.132886
Hanganu, I. L., Ben-Ari, Y., and Khazipov, R. (2006). Retinal waves trigger spindle bursts in the neonatal rat visual cortex. J. Neurosci. 26, 6728–6736. doi: 10.1523/JNEUROSCI.0752-06.2006
Hanganu, I. L., Okabe, A., Lessmann, V., and Luhmann, H. J. (2009). Cellular mechanisms of subplate-driven and cholinergic input-dependent network activity in the neonatal rat somatosensory cortex. Cereb. Cortex 19, 89–105. doi: 10.1093/cercor/bhn061
Hanganu-Opatz, I. L. (2010). Between molecules and experience: role of early patterns of coordinated activity for the development of cortical maps and sensory abilities. Brain Res. Rev. 64, 160–176. doi: 10.1016/j.brainresrev.2010.03.005
Hara, K., Nakamura, M., Haranishi, Y., Terada, T., Kataoka, K., and Sata, T. (2012). Antinociceptive effect of intrathecal administration of hypotaurine in rat models of inflammatory and neuropathic pain. Amino Acids 43, 397–404. doi: 10.1007/s00726-011-1094-9
Haydar, T. F., Wang, F., Schwartz, M. L., and Rakic, P. (2000). Differential modulation of proliferation in the neocortical ventricular and subventricular zones. J. Neurosci. 20, 5764–5774.
Heck, N., Kilb, W., Reiprich, P., Kubota, H., Furukawa, T., Fukuda, A., et al. (2007). GABA-A receptors regulate neocortical neuronal migration in vitro and in vivo. Cereb. Cortex 17, 138–148. doi: 10.1093/cercor/bhj135
Henneberger, C., Bard, L., and Rusakov, D. A. (2012). D-Serine: a key to synaptic plasticity? Int. J. Biochem. Cell. Biol. 44, 587–590. doi: 10.1016/j.biocel.2012.01.005
Hernandez-Benitez, R., Pasantes-Morales, H., Saldana, I. T., and Ramos-Mandujano, G. (2010). Taurine stimulates proliferation of mice embryonic cultured neural progenitor cells. J. Neurosci. Res. 88, 1673–1681. doi: 10.1002/jnr.22328
Hernandez-Benitez, R., Ramos-Mandujano, G., and Pasantes-Morales, H. (2012). Taurine stimulates proliferation and promotes neurogenesis of mouse adult cultured neural stem/progenitor cells. Stem Cell Res. 9, 24–34. doi: 10.1016/j.scr.2012.02.004
Hernandez-Benitez, R., Vangipurarn, S. D., Ramos-Mandujano, G., Lyman, W. D., and Pasantes-Morales, H. (2013). Taurine enhances the growth of neural precursors derived from fetal human brain and promotes neuronal specification. Develop. Neurosci. 35, 40–49. doi: 10.1159/000346900
Hirzel, K., Müller, U., Latal, A. T., Hülsmann, S., Grudzinska, J., Seeliger, M. W., et al. (2006). Hyperekplexia phenotype of glycine receptor alpha1 subunit mutant mice identifies Zn2+ as an essential endogenous modulator of glycinergic neurotransmission. Neuron 52, 679–690. doi: 10.1016/j.neuron.2006.09.035
Huxtable, R. J. (1989). Taurine in the central nervous-system and the mammalian actions of taurine. Prog. Neurobiol. 32, 471–533. doi: 10.1016/0301-0082(89)90019-1
Inoue, K., Furukawa, T., Kumada, T., Yamada, J., Wang, T. Y., Inoue, R., et al. (2012). Taurine inhibits K+-Cl- cotransporter KCC2 to regulate embryonic Cl- homeostasis via with-no-lysine (WNK) protein kinase signaling pathway. J. Biol. Chem. 287, 20839–20850. doi: 10.1074/jbc.M111.319418
Jammoul, F., Wang, Q. P., Nabbout, R., Coriat, C., Duboc, A., Simonutti, M., et al. (2009). Taurine deficiency is a cause of vigabatrin-induced retinal phototoxicity. Ann. Neurol. 65, 98–107. doi: 10.1002/ana.21526
Jia, F., Yue, M., Chandra, D., Keramidas, A., Goldstein, P. A., Homanics, G. E., et al. (2008). Taurine is a potent activator of extrasynaptic GABA(A) receptors in the thalamus. J. Neurosci. 28, 106–115. doi: 10.1523/JNEUROSCI.3996-07.2008
Kamisaki, Y., Wada, K., Nakamoto, K., and Itoh, T. (1996). Release of taurine and its effects on release of neurotransmitter amino acids in rat cerebral cortex. Adv. Exp. Med. Biol. 403, 445–454. doi: 10.1007/978-1-4899-0182-8_\48
Kanold, P. O., and Luhmann, H. J. (2010). The subplate and early cortical circuits. Annu. Rev. Neurosci. 33, 23–48. doi: 10.1146/annurev-neuro-060909-153244
Khazipov, R., and Milh, M. (2017). Early patterns of activity in the developing cortex: focus on the sensorimotor system. Semin. Cell. Dev. Biol. doi: 10.1016/j.semcdb.2017.09.014 [Epub ahead of print].
Khazipov, R., Minlebaev, M., and Valeeva, G. (2013). Early gamma oscillations. Neuroscience 250, 240–252. doi: 10.1016/j.neuroscience.2013.07.019
Khazipov, R., Sirota, A., Leinekugel, X., Holmes, G. L., Ben-Ari, Y., and Buzsáki, G. (2004). Early motor activity drives spindle bursts in the developing somatosensory cortex. Nature 432, 758–761. doi: 10.1038/nature03132
Kilb, W. (2017). Putative role of taurine as neurotransmitter during perinatal cortical development. Adv. Exp. Med. Biol. 975, 281–292. doi: 10.1007/978-94-024-1079-2_\25
Kilb, W., Hanganu, I. L., Okabe, A., Sava, B. A., Shimizu-Okabe, C., Fukuda, A., et al. (2008). Glycine receptors mediate excitation of subplate neurons in neonatal rat cerebral cortex. J. Neurophysiol. 100, 698–707. doi: 10.1152/jn.00657.2007
Kilb, W., Ikeda, M., Uchida, K., Okabe, A., Fukuda, A., and Luhmann, H. J. (2002). Depolarizing glycine responses in Cajal-Retzius cells of neonatal rat cerebral cortex. Neuroscience 112, 299–307. doi: 10.1016/S0306-4522(02)00071-4
Kim, H. Y., Kim, H. V., Yoon, J. H., Kang, B. R., Cho, S. M., Lee, S., et al. (2014). Taurine in drinking water recovers learning and memory in the adult APP/PS1 mouse model of Alzheimer’s disease. Sci. Rep. 4:7467. doi: 10.1038/srep07467
Kirischuk, S., Luhmann, H. J., and Kilb, W. (2014). Cajal-Retzius cells: update on structural and functional properties of these mystic neurons that bridged the 20th century. Neuroscience 275, 33–46. doi: 10.1016/j.neuroscience.2014.06.009
Kirmse, K., Kummer, M., Kovalchuk, Y., Witte, O. W., Garaschuk, O., and Holthoff, K. (2015). GABA depolarizes immature neurons and inhibits network activity in the neonatal neocortex in vivo. Nat. Commun. 6:7750. doi: 10.1038/ncomms8750
Kletke, O., Gisselmann, G., May, A., Hatt, H., and Sergeeva, O. A. (2013). Partial agonism of taurine at gamma-containing native and recombinant GABA(A) receptors. PLOS ONE 8:e61733. doi: 10.1371/journal.pone.0061733
Komuro, H., and Kumada, T. (2005). Ca2+ transients control CNS neuronal migration. Cell Calcium 37, 387–393. doi: 10.1016/j.ceca.2005.01.006
Kontro, P., and Oja, S. S. (1990). Interactions of taurine with GABA-B binding-sites in mouse-brain. Neuropharmacology 29, 243–247. doi: 10.1016/0028-3908(90)90008-F
Lambert, I. H. (2004). Regulation of the cellular content of the organic osmolyte taurine in mammalian cells. Neurochem. Res. 29, 27–63. doi: 10.1023/B:NERE.0000010433.08577.96
Lambert, I. H., Kristensen, D. M., Holm, J. B., and Mortensen, O. H. (2015). Physiological role of taurine - from organism to organelle. Acta Physiol. 213, 191–212. doi: 10.1111/apha.12365
Lehtinen, M. K., and Walsh, C. A. (2011). Neurogenesis at the brain-cerebrospinal fluid interface. Ann. Rev. Cell. Dev. Biol. 27, 653–679. doi: 10.1146/annurev-cellbio-092910-154026
Lehtinen, M. K., Zappaterra, M. W., Chen, X., Yang, Y. J., Hill, A. D., Lun, M., et al. (2011). The cerebrospinal fluid provides a proliferative niche for neural progenitor cells. Neuron 69, 893–905. doi: 10.1016/j.neuron.2011.01.023
Liu, X., Wang, Q., Haydar, T. F., and Bordey, A. (2005). Nonsynaptic GABA signaling in postnatal subventricular zone controls proliferation of GFAP-expressing progenitors. Nat. Neurosci. 8, 1179–1187. doi: 10.1038/nn1522
LoTurco, J. J., Owens, D. F., Heath, M. J., Davis, M. B., and Kriegstein, A. R. (1995). GABA and glutamate depolarize cortical progenitor cells and inhibit DNA synthesis. Neuron 15, 1287–1298. doi: 10.1016/0896-6273(95)90008-X
Luhmann, H. J., Fukuda, A., and Kilb, W. (2015). Control of cortical neuronal migration by glutamate and GABA. Front. Cell. Neurosci. 9:4. doi: 10.3389/fncel.2015.00004
Luhmann, H. J., and Khazipov, R. (2017). Neuronal activity patterns in the developing barrel cortex. Neuroscience doi: 10.1016/j.neuroscience.2017.05.025 [Epub ahead of print].
Luhmann, H. J., Kilb, W., and Hanganu-Opatz, I. L. (2009). Subplate cells: amplifiers of neuronal activity in the developing cerebral cortex. Front. Neuroanat. 3:19. doi: 10.3389/neuro.05.019.2009
Luhmann, H. J., Sinning, A., Yang, J. W., Reyes-Puerta, V., Stuttgen, M. C., Kirischuk, S., et al. (2016). Spontaneous neuronal activity in developing neocortical networks: from single cells to large-scale interactions. Front. Neural. Circuits 10:40. doi: 10.3389/fncir.2016.00040
Ma, W., and Barker, J. L. (1995). Complementary expressions of transcripts encoding GAD(67) and GABA(A) receptor alpha-4, beta-1, and gamma-1 subunits in the proliferative zone of the embryonic rat central-nervous-system. J. Neurosci. 15, 2547–2560.
Maar, T., Moran, J., Schousboe, A., and Pasantes-Morales, H. (1995). Taurine deficiency in dissociated mouse cerebellar cultures affects neuronal migration. Int. J. Dev. Neurosci. 13, 491–502. doi: 10.1016/0736-5748(95)00068-R
Manent, J. B., Demarque, M., Jorquera, I., Pellegrino, C., Ben-Ari, Y., Aniksztejn, L., et al. (2005). A noncanonical release of GABA and glutamate modulates neuronal migration. J. Neurosci. 25, 4755–4765. doi: 10.1523/JNEUROSCI.0553-05.2005
Manent, J. B., Jorquera, I., Mazzucchelli, I., Depaulis, A., Perucca, E., Ben-Ari, Y., et al. (2007). Fetal exposure to GABA-acting antiepileptic drugs generates hippocampal and cortical dysplasias. Epilepsia 48, 684–693. doi: 10.1111/j.1528-1167.2007.01056.x
Marcinkiewicz, J., and Kontny, E. (2014). Taurine and inflammatory diseases. Amino Acids 46, 7–20. doi: 10.1007/s00726-012-1361-4
Maric, D., Liu, Q. Y., Maric, I., Chaudry, S., Chang, Y. H., Smith, S. V., et al. (2001). GABA expression dominates neuronal lineage progression in the embryonic rat neocortex and facilitates neurite outgrowth via GABAA autoreceptor/Cl- channels. J. Neurosci. 21, 2343–2360.
Marín, O., and Rubenstein, J. L. (2001). A long, remarkable journey: tangential migration in the telencephalon. Nat. Rev. Neurosci. 2, 780–790. doi: 10.1038/35097509
Martincigh, B. S., Mundoma, C., and Simoji, R. H. (1998). Antioxidant chemistry: hypotaurine-taurine oxidation by chlorite. J. Physical. Chem. A 102, 9838–9846. doi: 10.1021/jp982575c
Meier, J., Akyeli, J., Kirischuk, S., and Grantyn, R. (2003). GABA(A) receptor activity and PKC control inhibitory synaptogenesis in CNS tissue slices. Mol. Cell. Neurosci. 23, 600–613. doi: 10.1016/S1044-7431(03)00079-4
Menzie, J., Prentice, H., and Wu, J. Y. (2013). Neuroprotective mechanisms of taurine against ischemic stroke. Brain Sci. 3, 877–907. doi: 10.3390/brainsci3020877
Molchanova, S., Oja, S. S., and Saransaari, P. (2004). Characteristics of basal taurine release in the rat striatum measured by microdialysis. Amino Acids 27, 261–268. doi: 10.1007/s00726-004-0139-8
Neuwirth, L. S., Volpe, N. P., and El Idrissi, A. (2013). Taurine effects on emotional learning and memory in aged mice: neurochemical alterations and differentiation in auditory cued fear and context conditioning. Adv. Exp. Med. Biol. 775, 195–214. doi: 10.1007/978-1-4614-6130-2_\17
Nikolic, M., Gardner, H. A. R., and Tucker, K. L. (2013). Postnatal neuronal apoptosis in the cerebral cortex: physiological and pathophysiological mechanisms. Neuroscience 254, 369–378. doi: 10.1016/j.neuroscience.2013.09.035
Nimmervoll, B., Denter, D. G., Sava, I., Kilb, W., and Luhmann, H. J. (2011). Glycine receptors influence radial migration in the embryonic mouse neocortex. Neuroreport 22, 509–513. doi: 10.1097/WNR.0b013e328348aafe
Ochoa-de la Paz, L. D., Martinez-Davila, I. A., Miledi, R., and Martinez-Torres, A. (2008). Modulation of human GABA rho 1 receptors by taurine. Neurosci. Res. 61, 302–308. doi: 10.1016/j.neures.2008.03.009
Oja, S. S., and Saransaari, P. (1995). Chloride ions, potassium stimulation and release of endogenous taurine from cerebral cortical slices from 3 day old and 3 month old mice. Neurochem. Int. 27, 313–318. doi: 10.1016/0197-0186(95)00012-W
Oja, S. S., and Saransaari, P. (2013). Taurine and epilepsy. Epilepsy Res. 104, 187–194. doi: 10.1016/j.eplepsyres.2013.01.010
Oja, S. S., and Saransaari, P. (2015). Open questions concerning taurine with emphasis on the brain. Adv. Exp. Med. Biol. 803, 409–413. doi: 10.1007/978-3-319-15126-7_\31
Okabe, A., Kilb, W., Shimizu-Okabe, C., Hanganu, I. L., Fukuda, A., and Luhmann, H. J. (2004). Homogenous glycine receptor expression in cortical plate neurons and Cajal-Retzius cells of neonatal rat cerebral cortex. Neuroscience 123, 715–724. doi: 10.1016/j.neuroscience.2003.10.014
Owens, D. F., Flint, A. C., Dammerman, R. S., and Kriegstein, A. R. (2000). Calcium dynamics of neocortical ventricular zone cells. Dev. Neurosci. 22, 25–33. doi: 10.1159/000017424
Palackal, T., Moretz, R., Wisniewski, H., and Sturman, J. (1986). Abnormal visual cortex development in the kitten associated with maternal dietary taurine deprivation. J. Neurosci. Res. 15, 223–239. doi: 10.1002/jnr.490150212
Pellicer, F., Lopez-Avila, A., Coffeen, U., Ortega-Legaspi, J. M., and del Angel, R. (2007). Taurine in the anterior cingulate cortex diminishes neuropathic nociception: A possible interaction with the glycine(A) receptor. Eur. J. Pain 11, 444–451. doi: 10.1016/j.ejpain.2006.06.003
Qian, T. Z., Chen, R. Q., Nakamura, M., Furukawa, T., Kumada, T., Akita, T., et al. (2014). Activity-dependent endogenous taurine release facilitates excitatory neurotransmission in the neocortical marginal zone of neonatal rats. Front. Cell. Neurosci. 8:33. doi: 10.3389/fncel.2014.00033
Qiu, Z. Z., Dubin, A. E., Mathur, J., Tu, B., Reddy, K., Miraglia, L. J., et al. (2014). SWELL1, a plasma membrane protein, is an essential component of volume-regulated anion channel. Cell 157, 447–458. doi: 10.1016/j.cell.2014.03.024
Ramos-Mandujano, G., Hernandez-Benitez, R., and Pasantes-Morales, H. (2014). Multiple mechanisms mediate the taurine-induced proliferation of neural stem/progenitor cells from the subventricular zone of the adult mouse. Stem Cell Res. 12, 690–702. doi: 10.1016/j.scr.2014.02.009
Roychoudhury, A., Bieker, A., Haussinger, D., and Oesterhelt, F. (2013). Membrane protein stability depends on the concentration of compatible solutes - a single molecule force spectroscopic study. Biol. Chem. 394, 1465–1474. doi: 10.1515/hsz-2013-0173
Saransaari, P., and Oja, S. S. (1991). Excitatory amino acids evoke taurine release from cerebral cortex slices from adult and developing mice. Neuroscience 45, 451–459. doi: 10.1016/0306-4522(91)90240-O
Saransaari, P., and Oja, S. S. (1999). Characteristics of ischemia-induced taurine release in the developing mouse hippocampus. Neuroscience 94, 949–954. doi: 10.1016/S0306-4522(99)00384-X
Saransaari, P., and Oja, S. S. (2000a). Involvement of metabotropic glutamate receptors in ischemia-induced taurine release in the developing and adult hippocampus. Neurochem. Res. 25, 1067–1072.
Saransaari, P., and Oja, S. S. (2000b). Modulation of the ischemia-induced taurine release by adenosine receptors in the developing and adult mouse hippocampus. Neuroscience 97, 425–430.
Saransaari, P., and Oja, S. S. (2000c). Taurine and neural cell damage. Amino Acids 19, 509–526. doi: 10.1007/s007260070003
Saransaari, P., and Oja, S. S. (2003). Characterization of N-methyl-D-aspartate-evoked taurine release in the developing and adult mouse hippocampus. Amino Acids 24, 213–221.
Saransaari, P., and Oja, S. S. (2006). Characteristics of taurine release in slices from adult and developing mouse brain stem. Amino Acids 31, 35–43. doi: 10.1007/s00726-006-0290-5
Sava, B. A., Chen, R. Q., Sun, H. Y., Luhmann, H. J., and Kilb, W. (2014). Taurine activates GABA(A)ergic networks in the neocortex of immature mice. Front. Cell. Neurosci. 8:26. doi: 10.3389/fncel.2014.00026
Schaffer, S. W., Azuma, J., and Mozaffari, M. (2009). Role of antioxidant activity of taurine in diabetes. Can. J. Physiol. Pharmacol. 87, 91–99. doi: 10.1139/Y08-110
Schmieden, V., Kuhse, J., and Betz, H. (1992). Agonist pharmacology of neonatal and adult glycine receptor alpha subunits: identification of amino acid residues involved in taurine activation. EMBO J. 11, 2025–2032.
Sergeeva, O. A., Chepkova, A. N., Doreulee, N., Eriksson, K. S., Poelchen, W., Monnighoff, I., et al. (2003). Taurine-induced long-lasting enhancement of synaptic transmission in mice: role of transporters. J. Physiol. 550, 911–919. doi: 10.1113/jphysiol.2003.045864
Shivaraj, M. C., Marcy, G., Low, G. L., Ryu, J. R., Zhao, X. F., Rosales, F. J., et al. (2012). Taurine induces proliferation of neural stem cells and synapse development in the developing mouse brain. PLOS ONE 7:e42935. doi: 10.1371/journal.pone.0042935
Solis, J. M., Herranz, A. S., Herreras, O., Lerma, J., and Martin, D. R. (1988). Does taurine act as an osmoregulatory substance in the rat brain? Neurosci. Lett. 91, 53–58.
Spitzer, N. C. (2006). Electrical activity in early neuronal development. Nature 444, 707–712. doi: 10.1038/nature05300
Spitzer, N. C., Lautermilch, N. J., Smith, R. D., and Gomez, T. M. (2000). Coding of neuronal differentiation by calcium transients. Bioessays 22, 811–817. doi: 10.1002/1521-1878(200009)22:9<811::AID-BIES6>3.0.CO;2-G
Sturman, J. A. (1981). Origin of taurine in developing rat brain. Brain Res. 254, 111–128. doi: 10.1016/0165-3806(81)90063-8
Sturman, J. A., Moretz, R. C., French, J. H., and Wisniewski, H. M. (1985). Postnatal taurine deficiency in the kitten results in a persistence of the cerebellar external granule cell layer: correction by taurine feeding. J. Neurosci. Res. 13, 521–528. doi: 10.1002/jnr.490130407
Sturman, J. A., Rassin, D. K., and Gaull, G. E. (1977). Taurine in developing rat brain: maternal-fetal transfer of [35 S] taurine and its fate in the neonate. J. Neurochem. 28, 31–39. doi: 10.1111/j.1471-4159.1977.tb07705.x
Suarez, L. M., and Solis, J. M. (2006). Taurine potentiates presynaptic NMDA receptors in hippocampal Schaffer collateral axons. Eur. J. Neurosci. 24, 405–418. doi: 10.1111/j.1460-9568.2006.04911.x
Sun, M., Zhao, Y. M., Gu, Y., and Xu, C. (2012). Therapeutic window of taurine against experimental stroke in rats. Translat. Res. 160, 223–229. doi: 10.1016/j.trsl.2012.02.007
Sun, M., Zhao, Y. M., Gu, Y., and Zhang, Y. Z. (2015). Protective effects of taurine against closed head injury in rats. J. Neurotrauma 32, 66–74. doi: 10.1089/neu.2012.2432
Suzuki, T., Suzuki, T., Wada, T., Saigo, K., and Watanabe, K. (2002). Taurine as a constituent of mitochondrial tRNAs: new insights into the functions of taurine and human mitochondrial diseases. EMBO J. 21, 6581–6589. doi: 10.1093/emboj/cdf656
Tamamaki, N., Yanagawa, Y., Tomioka, R., Miyazaki, J., Obata, K., and Kaneko, T. (2003). Green fluorescent protein expression and colocalization with calretinin, parvalbumin, and somatostatin in the GAD67-GFP knock-in mouse. J. Comp. Neurol. 467, 60–79. doi: 10.1002/cne.10905
Taranukhin, A. G., Taranukhina, E. Y., Saransaari, P., Djatchkova, I. M., Pelto-Huikko, M., and Oja, S. S. (2008). Taurine reduces caspase-8 and caspase-9 expression induced by ischemia in the mouse hypothalamic nuclei. Amino Acids 34, 169–174. doi: 10.1007/s00726-006-0405-z
Terada, T., Hara, K., Haranishi, Y., and Sata, T. (2011). Antinociceptive effect of intrathecal administration of taurine in rat models of neuropathic pain. Can. J. Anesth. 58, 630–637. doi: 10.1007/s12630-011-9504-8
Tochitani, S., Sakata-Haga, H., and Fukui, Y. (2010). Embryonic exposure to ethanol disturbs regulation of mitotic spindle orientation via GABAA receptors in neural progenitors in ventricular zone of developing neocortex. Neurosci. Lett 472, 128–132. doi: 10.1016/j.neulet.2010.01.071
Voss, F. K., Ullrich, F., Munch, J., Lazarow, K., Lutter, D., Mah, N., et al. (2014). Identification of LRRC8 heteromers as an essential component of the volume-regulated anion channel VRAC. Science 344, 634–638. doi: 10.1126/science.1252826
Wang, D. D., and Kriegstein, A. R. (2009). Defining the role of GABA in cortical development. J. Physiol. 587, 1873–1879. doi: 10.1113/jphysiol.2008.167635
Wang, Q., Zhu, G. H., Xie, D. H., Wu, W. J., and Hu, P. (2015). taurine enhances excitability of mouse cochlear neural stem cells by selectively promoting differentiation of glutamatergic neurons over GABAergic neurons. Neurochem. Res. 40, 924–931. doi: 10.1007/s11064-015-1546-9
Watanabe, M., and Fukuda, A. (2015). Development and regulation of chloride homeostasis in the central nervous system. Front. Cell. Neurosci. 9:371. doi: 10.3389/fncel.2015.00371
Weissman, T. A., Riquelme, P. A., Ivic, L., Flint, A. C., and Kriegstein, A. R. (2004). Calcium waves propagate through radial glial cells and modulate proliferation in the developing neocortex. Neuron 43, 647–661. doi: 10.1016/j.neuron.2004.08.015
Yamada, J., Okabe, A., Toyoda, H., Kilb, W., Luhmann, H. J., and Fukuda, A. (2004). Cl- uptake promoting depolarizing GABA actions in immature rat neocortical neurones is mediated by NKCC1. J. Physiol. 557, 829–841. doi: 10.1113/jphysiol.2004.062471
Yamamoto, N., and López-Bendito, G. (2012). Shaping brain connections through spontaneous neural activity. Eur. J. Neurosci. 35, 1595–1604. doi: 10.1111/j.1460-9568.2012.08101.x
Yang, J. W., Hanganu-Opatz, I. L., Sun, J. J., and Luhmann, H. J. (2009). Three patterns of oscillatory activity differentially synchronize developing neocortical networks in vivo. J. Neurosci. 29, 9011–9025. doi: 10.1523/JNEUROSCI.5646-08.2009
Yang, J. W., Reyes-Puerta, V., Kilb, W., and Luhmann, H. J. (2016). Spindle bursts in neonatal rat cerebral cortex. Neural. Plast. 2016:3467832. doi: 10.1155/2016/3467832
You, J. S., and Chang, K. J. (1998). Taurine protects the liver against lipid peroxidation and membrane disintegration during rat hepatocarcinogenesis. Adv. Exp. Med. Biol. 442, 105–112. doi: 10.1007/978-1-4899-0117-0_14
Young, T. L., and Cepko, C. L. (2004). A role for ligand-gated ion channels in rod photoreceptor development. Neuron 41, 867–879. doi: 10.1016/S0896-6273(04)00141-2
Keywords: cerebral cortex, subplate, Cajal–Retzius cells, GABA receptors, glycine receptors, migration, rodent, review
Citation: Kilb W and Fukuda A (2017) Taurine as an Essential Neuromodulator during Perinatal Cortical Development. Front. Cell. Neurosci. 11:328. doi: 10.3389/fncel.2017.00328
Received: 01 September 2017; Accepted: 04 October 2017;
Published: 24 October 2017.
Edited by:
Enrico Cherubini, Scuola Internazionale Superiore di Studi Avanzati (SISSA), ItalyReviewed by:
Susumu Ito, Kokushikan University, JapanRustem Khazipov, Institut National de la Santé et de la Recherche Médicale, France
Copyright © 2017 Kilb and Fukuda. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Werner Kilb, d2tpbGJAdW5pLW1haW56LmRl
†These authors have contributed equally to this work.
 Werner Kilb
Werner Kilb Atsuo Fukuda
Atsuo Fukuda