Abstract
Aerobic fitness has been found to play a positive role in brain and cognitive health of children. Yet, many of the neural biomarkers related to aerobic fitness remain unknown. Here, using diffusion tensor imaging, we demonstrated that higher aerobic fitness was related to greater estimates of white matter microstructure in children. Higher fit 9- and 10-year-old children showed greater fractional anisotropy (FA) in sections of the corpus callosum, corona radiata, and superior longitudinal fasciculus, compared to lower fit children. The FA effects were primarily characterized by aerobic fitness differences in radial diffusivity, thereby raising the possibility that estimates of myelination may vary as a function of individual differences in fitness during childhood. White matter structure may be another potential neural mechanism of aerobic fitness that assists in efficient communication between gray matter regions as well as the integration of regions into networks.
INTRODUCTION
Strategies to optimize cognitive and brain development, including physical activity, computerized training games, martial arts, yoga, mindfulness, and school curricula, are in the spotlight (Diamond and Lee, 2011). For example, aerobic fitness plays an important role in the brain health of children (see Chaddock et al., 2011 for a review), especially in terms of brain structure and brain function, including gray matter volumes [e.g., hippocampus (Chaddock et al., 2010a), basal ganglia (Chaddock et al., 2010b)] and functional brain networks (e.g., frontal and parietal cortex; Voss et al., 2011; Chaddock et al., 2012a). These fitness-related differences in brain health are often coupled with performance differences, such that higher fit children have been shown to outperform their lower fit peers on tasks of cognitive control (Chaddock et al., 2010b, 2012a; Voss et al., 2011) and memory (Chaddock et al., 2010a) as well as scholastic achievement tests in the classroom (Coe et al., 2006; Castelli et al., 2007; Trudeau and Shephard, 2008; Chomitz et al., 2009).
Still unknown is the relationship between aerobic fitness and the microstructure of white matter tracts during childhood. The structure of white matter is important for efficient transmission of information between gray matter as well as for the integration of brain areas into structural networks to support cognitive function. During childhood, many white matter tracts increase in estimates of microstructure, in parallel with improvements in cognition (Barnea-Goraly et al., 2005; Muetzel et al., 2008; see Schmithorst and Yuan, 2010, for a review), but there has been little research about individual differences in the structure of these pathways, the plasticity of white matter fibers, or their susceptibility to intervention during development. Given that the size and function of several different gray matter regions differ between higher fit and lower fit children (Hillman et al., 2009; Chaddock et al., 2010a,b; Davis et al., 2011; Pontifex et al., 2011; Voss et al., 2011; Chaddock et al., 2012a), it is possible that white matter microstructure that connects gray matter areas also differs as a function of aerobic fitness. In support of such a hypothesis, higher aerobic fitness levels and participation in physical activity are positively associated with white matter structure in older adults (Colcombe et al., 2006; Marks et al., 2007; Johnson et al., 2012; Voss et al., 2012).
Here, for the first time, we used diffusion tensor imaging (DTI) to investigate the microstructural properties of white matter in higher fit and lower fit 9- and 10-year-old children. We analyzed fractional anisotropy (FA), a general index of white matter microstructure, hypothesized to be higher in tightly bundled, structurally compact fibers with high integrity (Basser, 1995; Beaulieu, 2002; Sen and Basser, 2005; Rykhlevskaia et al., 2008). We also explored specific patterns of diffusivity [radial diffusivity (RD) and axial diffusivity (AD)], hypothesized to reflect potential biological properties of white matter microstructure (Basser, 1995; Pierpaoli and Basser, 1996; Pierpaoli et al., 1996; Song et al., 2002). For example, a reduction in RD is observed in the presence of remyelination, causing RD to often be used as a marker of myelination (Song et al., 2002, 2003, 2005; Nair et al., 2005; Budde et al., 2007; Rykhlevskaia et al., 2008). Additionally, AD is said to be sensitive to changes in axonal fibers including axonal diameter, loss or damage (Song et al., 2003; Budde et al., 2007). By exploring the associations among aerobic fitness and measures of diffusivity (FA, RD, AD), we can investigate microstructural white matter properties influenced by aerobic fitness during child development.
We predicted that higher fit children would show greater estimates of white matter microstructure than lower fit children. Because the structure and function of brain circuits important for cognitive control have been found to be sensitive to aerobic fitness (Bunge and Crone, 2009; see Chaddock et al., 2012b for a review), we focused on white matter tracts that relate to these cognitive skills (Liston et al., 2006; Madsen et al., 2010; Tamnes et al., 2012; Chaddock-Heyman et al., 2013), and that connect frontal, striatal and parietal brain areas. In particular, in this first exploration of the association between aerobic fitness and white matter microstructure in children, we examined a priori white matter tracts (included in Chaddock-Heyman et al., 2013). These tracts included the corpus callosum, which connects the left and right cerebral hemispheres (Olesen et al., 2003; Nagy et al., 2004; Tamnes et al., 2012), the corona radiata which carries ascending and descending information throughout the brain (Olesen et al., 2003; Bendlin et al., 2010; Niogi et al., 2010; Chaddock-Heyman et al., 2013), and the superior longitudinal fasciculus, a tract that provides bidirectional information transfer between the frontal and parietal cortex (Nagy et al., 2004; Chaddock-Heyman et al., 2013). Additionally, we examined the posterior thalamic radiation, nerve fibers connecting the thalamus and caudate nucleus with the cerebral cortex (Olson et al., 2009; Chaddock-Heyman et al., 2013), and the cerebral peduncle, part of the brainstem, which includes nerve tracts carrying motor information to and from the brain to the rest of the body (Olson et al., 2009; Chaddock-Heyman et al., 2013). The present study contributes to our understanding of plasticity of the microstructure of white matter that allows efficient communication between gray matter areas in the brain and integrates the brain into networks important for cognition and academic achievement.
MATERIALS AND METHODS
PARTICIPANTS
Preadolescent 9- and 10-year-old children were recruited from East-Central Illinois. Children were screened for several factors that influence physical activity participation or cognitive function. To begin, the Kaufman Brief Intelligence Test (K-BIT; Kaufman and Kaufman, 1990) was administered to each child to obtain a composite intelligence quotient (IQ) score including both crystallized and fluid intelligence measures. Participants were excluded if their scores were more than 1 standard deviation below the mean (85%). Next, a guardian of the child completed the attention-deficit hyperactivity disorder (ADHD) Rating Scale IV (DuPaul et al., 1998) to screen for the presence of attentional disorders. Participants were excluded if they scored above the 85th percentile. Pubertal timing was also assessed using a modified Tanner Staging System (self-report, with schematics; Taylor et al., 2001) with all participants at or below a score of 2 on a 5-point scale of developmental stages. In addition, socioeconomic status (SES) was determined by creating a trichotomous index based on three variables: participation in a free or reduced-price meal program at school, the highest level of education obtained by the child’s mother and father, and the number of parents who worked full-time (Birnbaum et al., 2002).
Furthermore, eligible participants were required to (1) qualify as higher fit or lower fit (see Aerobic Fitness Assessment), (2) demonstrate right handedness (as measured by the Edinburgh Handedness Questionnaire; Oldfield, 1971), (3) report no adverse health conditions, physical incapacities, or neurological disorders, (4) report no use of medications that influenced central nervous system function, (5) successfully complete a mock MRI session to screen for claustrophobia in an MRI machine, and (6) sign an informed assent approved by the University of Illinois at Urbana-Champaign. A legal guardian also provided written informed consent in accordance with the Institutional Review Board of the University of Illinois at Urbana-Champaign.
Fifty-two participants were initially eligible for the present study (after exclusions due to K-BIT scores, ADHD, pubertal timing, VO2max criteria, etc). Of this sample, 32 children completed the white matter imaging component of the MRI session. Eight children were excluded from analysis due to visible motion on the reconstructed data and/or lack of whole-brain coverage during acquisition. Twenty-four children were included in the analysis, including 12 higher fit participants (seven boys, five girls) and 12 lower fit participants (eight boys, four girls). All participants were compensated $10/h for the demographic and fitness testing and $20 for participation in the MRI session.
AEROBIC FITNESS ASSESSMENT
The aerobic fitness level of each child was determined by measuring maximal oxygen uptake (VO2max) using a computerized indirect calorimetry system (ParvoMedics True Max 2400) during a modified Balke protocol (American College of Sports Medicine, 2006). Specifically, participants ran on a motor-driven treadmill at a constant speed with increases in grade increments of 2.5% every 2 min until volitional exhaustion. Averages for oxygen uptake (VO2) and respiratory exchange ratio (RER; the ratio between carbon dioxide and oxygen) were assessed every 30 s. In addition, heart rate was measured throughout the fitness test [using a Polar heart rate monitor (Polar WearLink®;+ 31, Polar Electro, Finland)], and ratings of perceived exertion were assessed every 2 min using the children’s OMNI scale (Utter et al., 2002).
VO2max was defined when oxygen consumption remained at a steady state despite an increase in workload. Relative peak oxygen consumption was based upon maximal effort as evidenced by (1) a plateau in oxygen consumption corresponding to an increase of <2 mL/kg/min despite an increase in workload, (2) a peak heart rate >185 beats per minute (American College of Sports Medicine, 2006) accompanied by a heart rate plateau (i.e., an increase in work rate without a concomitant increase in heart rate, Freedson and Goodman, 1993), (3) RER >1.0 (Bar-Or, 1983), and/or (4) ratings on the children’s OMNI scale of perceived exertion >8 (Utter et al., 2002). Relative peak oxygen consumption was expressed in mL/kg/min.
Aerobic fitness group assignments (i.e., higher fit and lower fit) were based on whether a child’s VO2max value fell above the 70th percentile or below the 30th percentile according to normative data provided by Shvartz and Reibold (1990). Children who did not qualify as higher fit or lower fit were excluded from further participation.
MAGNETIC RESONANCE IMAGING ACQUISITION
Diffusion-weighted images were acquired on a Siemens Allegra 3T head-only scanner with repetition time (TR) = 4.8 s, echo time (TE) = 100.4 ms, and 3.44 mm2 in-plane resolution with 4 mm slice thickness. To obtain whole-head coverage, 32 slices were collected parallel to the anterior-posterior commissure plane with no interslice gap. Four T2-weighted images (b-value = 0 s/mm2) and one 30-direction diffusion-weighted echo planar imaging scan (b-value = 1000 s/mm2) were collected.
Diffusion in the brain can be modeled by a diffusion tensor/diffusion ellipsoid. FA is calculated from the three eigenvalues (λ1, λ2, λ3) of the diffusion tensor and represents the anisotropy (directional dependence) of diffusion (Basser, 1995; Beaulieu, 2002; Sen and Basser, 2005), independently of the rate of diffusion. FA ranges from 0 to 1, with higher values reflecting increased directionality of diffusion (i.e., water traveling more parallel to a tract compared to perpendicularly). In a region with free diffusion, the FA value is 0 and the diffusion is isotropic. If the diffusion is more in one direction, i.e., anisotropic diffusion, the FA value approaches 1. RD is the average of the second and third eigenvalues (λ2, λ3), reflective of diffusivity perpendicular to the major axis of the tensor (Basser, 1995; Pierpaoli and Basser, 1996; Pierpaoli et al., 1996; Song et al., 2002). AD is the diffusion along the principal diffusion eigenvalue (λ1) of the ellipsoid.
DIFFUSION DATA ANALYSIS
Image analyses and tensor calculations were performed using FSL 5.0.1 (FMRIB Software Library). First, each participant’s data were passed through an automated pipeline consisting of (1) motion and eddy current correction, (2) removal of non-brain tissue using the Brain Extraction Tool (Smith, 2002), and (3) local fitting of the diffusion tensor model at each voxel using FMRIB’s Diffusion Toolbox v2.0 (FDT)1. The products of the multi-step pipeline included FA and AD images; RD maps were calculated as the mean of the second and third eigenvalues (Song et al., 2002).
Next, diffusion data were processed using TBSS v1.2 (Tract-Based Spatial Statistics; Smith et al., 2006). Each participant’s FA data were aligned into the 1 mm × 1 mm × 1 mm standard Montreal Neurological Institute (MNI152) space via the FMRIB58_FA template using the FMRIB’s Non-linear Registration Tool (Andersson et al., 2007a,b), and a mean diffusion image was created. The mean FA image was then thinned to create an average skeleton representing the centers of the tracts shared by all participants, and the skeleton was thresholded at FA > 0.20. Each participant’s aligned FA data were projected onto the skeleton, taking on the FA value from the local center of the nearest relevant tract. RD and AD skeletons for each participant were formed in a similar manner by projecting the analogous data onto the mean skeleton.
REGION-OF-INTEREST ANALYSIS
Diffusion values (FA, RD, AD) were calculated for each participant within a priori ROIs, created from the JHU ICBM-DTI-81 white matter labels atlas2 (Mori et al., 2005; Wakana et al., 2007; Hua et al., 2008). Tract ROIs were created in the genu, body and splenium of the corpus callosum, and the left and right corona radiata, superior longitudinal fasciculus, posterior thalamic radiation, and cerebral peduncle (Figure 1). An FSL command, fslmaths, was used to create each ROI (e.g., fslmaths JHUAtlas –uthr 16 –thr 16 RCerebralPeduncle).
FIGURE 1
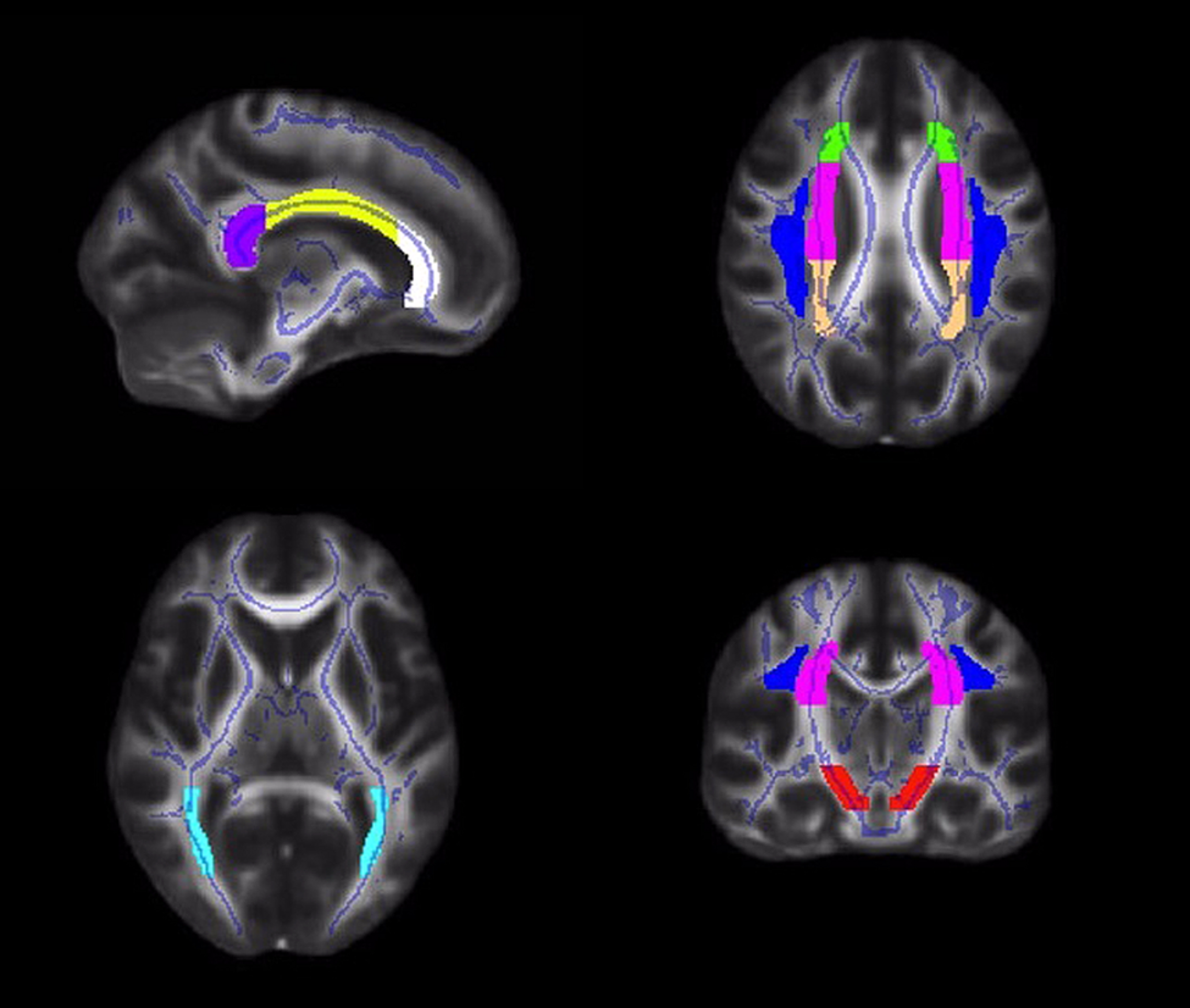
Illustrations of the white matter tract ROIs in the corpus callosum (white = genu; yellow = body; purple = splenium), corona radiata (anterior = green; superior = pink; posterior = peach), superior longitudinal fasciculus (blue), posterior thalamic radiation (light blue), and cerebral peduncle (red).
STATISTICAL ANALYSES
Our primary hypothesis was that higher fit children would show greater white matter microstructure in all tracts of interest, as indexed by FA, compared to lower fit children. First, because there were significant and robust correlations between hemispheres for each tract (all p > 0.8, p < 0.0001), we averaged FA values across left and right hemispheres. We then performed a multivariate analysis of variance (MANOVA) to examine associations between aerobic fitness group (lower fit, higher fit) and FA in all tract ROIs (genu, body, and splenium of the corpus callosum, average corona radiata, average superior longitudinal fasciculus, average posterior thalamic radiation, average cerebral peduncle). Given a significant multivariate effect, univariate ANOVAs were conducted to examine specific differences between higher fit and lower fit children in FA for each tract of interest. For those tracts showing significant differences in FA (p < 0.05) between groups, we conducted secondary analyses on RD and AD to better understand the underlying biological properties of overall FA differences in white matter microstructure (Burzynska et al., 2010; Johnson et al., 2012).
RESULTS
PARTICIPANT DEMOGRAPHICS
Demographic and fitness data are provided in Table 1. No differences in age, gender, IQ, ADHD, pubertal timing, or SES existed between aerobic fitness groups (t’s < 1.4, p’s > 0.19). We confirmed that higher fit children had higher relative VO2max scores than lower fit children [t (22) = 8.0, p < 0.001], as expected with our recruitment of extreme aerobic fitness groups.
Table 1
| Variable | Lower fit | Higher fit |
|---|---|---|
| N | 12 (8 boys) | 12 (7 boys) |
| Age (years) | 10.1 (0.7) | 9.9 (0.6) |
| VO2max (mL/kg/min) | 35.7 (6.0)* | 52.1 (3.9)* |
| VO2max percentile (%) | 14.8 (21.8)* | 82.7 (3.6)* |
| Height (inches) | 141.1 (9.1) | 141.5 (5.9) |
| Weight (pounds) | 42.5 (10.1)* | 33.5 (6.0)* |
| K-BITa composite score (IQ) | 110.6 (15.0) | 114.8 (6.8) |
| K-BITa crystallized score (vocabulary) | 106.4 (12.5) | 108.1 (5.5) |
| K-BITa fluid score (matrices) | 112.2 (17.0) | 118.7 (8.8) |
| ADHDb | 7.6 (5.5) | 6.8 (4.2) |
| Tannerc | 1.8 (0.4) | 1.5 (0.5) |
| SESd (median) | 3.0 (0.0) | 2.7 (0.7) |
Participant mean demographic and fitness data (SD) by aerobic fitness group.
Kaufman Brief Intelligence Test (Kaufman and Kaufman, 1990).
bScores on the ADHD Rating Scale V (DuPaul et al., 1998).
cPubertal timing assessed using a modified Tanner Staging System (Taylor et al., 2001).
dSocioeconomic Status was determined by the creation of a trichotomous index based on three variables: child participation in a free or reduced-price lunch program at school, the highest level of education obtained by the child’s mother and father, and the number of parents who worked full-time (Birnbaum et al., 2002).
*Significantly different at p < 0.001.
AEROBIC FITNESS AND WHITE MATTER MICROSTRUCTURE
The overall multivariate test indicated a significant effect of aerobic fitness on white matter microstructure [F(9,14) = 4.40, p = 0.007]. Next, univariate ANOVAs were performed to identify the specific dependent variables that contributed to the overall effect.
Higher fit children showed greater FA in the body of the corpus callosum [F(1,22) = 5.27, p = 0.032], bilateral superior corona radiata [F(1,22) = 6.18, p = 0.021], and bilateral superior longitudinal fasciculus [F(1,22) = 5.71, p = 0.026; Table 2]. No aerobic fitness differences were found in FA of the genu of the corpus callosum [F(1,22) = 1.51, p = 0.23], splenium of the corpus callosum [F(1,22) = 2.09, p = 0.16], anterior corona radiata [F(1,22) = 0.34, p = 0.57], posterior corona radiata [F(1,22) = 2.37, p = 0.08], posterior thalamic radiation [F(1,22) = 1.11, p = 0.30] or cerebral peduncle [F (1,22) = 0.23, p = 0.64; Table 2].
Table 2
| Lower fit (M, SD) | Higher fit (M, SD) | |
|---|---|---|
| FA corpus callosum genu | 0.5277 (0.04) | 0.5600 (0.08) |
| FA corpus callosum body | 0.4125 (0.03)* | 0.4510 (0.05)* |
| FA corpus callosum splenium | 0.5644 (0.02) | 0.5789 (0.02) |
| FA anterior corona radiata | 0.3627 (0.03) | 0.3763 (0.07) |
| FA superior corona radiata | 0.3441 (0.02)* | 0.3749 (0.04)* |
| FA posterior corona radiata | 0.3149 (0.02) | 0.3292 (0.02) |
| FA superior longitudinal fasciculus | 0.3135 (0.01)* | 0.3301 (0.02)* |
| FA posterior thalamic radiation | 0.3971 (0.02) | 0.4076 (0.03) |
| FA cerebral peduncle | 0.4792 (0.07) | 0.4684 (0.04) |
| RD corpus callosum body | 1.000 (0.70) | 0.900 (0.16) |
| RD superior corona radiata | 0.854 (0.05)* | 0.781 (0.10)* |
| RD superior longitudinal fasciculus | 0.928 (0.04)* | 0.871 (0.07)* |
| AD Corpus callosum body | 1.914 (0.12) | 1.87967 (0.18) |
| AD superior corona radiata | 1.486 (0.08) | 1.425 (0.11) |
| AD superior longitudinal fasciculus | 1.459 (0.06) | 1.413 (0.09) |
FA, RD, and AD (mean, standard deviation) in white matter tracts of interest in lower fit and higher fit children.
RD and AD values are reported in μm2/ms. *Significant aerobic fitness group difference, p < 0.05.
Secondary analyses on these significant regions showed that group differences in FA in the body of the corpus callosum were not driven by significant differences in RD or AD (F’s < 2.3, p’s > 0.1). Fitness differences in FA in the superior corona radiata were driven by significant differences in RD [F(1,22) = 4.97, p = 0.036]. Additionally, aerobic fitness differences in the superior longitudinal fasciculus were characterized by group differences in RD [F(1,22) = 6.61, p = 0.017]. There were no aerobic fitness group differences in AD in any white matter tracts. Table 2 provides means and standard deviations for the DTI measures of white matter microstructure for higher fit and lower fit children.
DISCUSSION
The present study is the first to demonstrate that aerobic fitness is positively related to the microstructure of white matter fiber tracts in the brain during childhood. Higher fit 9- and 10-year-old children (>70th percentile VO2max) showed greater FA in sections of the corpus callosum, corona radiata, and superior longitudinal fasciculus, compared to lower fit children (<30th percentile VO2max). An exploration of the components of the diffusion tensor showed that the relationships between aerobic fitness and FA were primarily characterized by fitness group differences in RD. Because axonal caliber and thickness of the myelin sheath determine conduction velocity (Paus, 2010), our results raise the possibility that children with higher fitness levels may have faster neural conduction between brain regions important for cognitive control. White matter microstructure may be another potential neural mechanism of aerobic fitness that assists in efficient communication between gray matter and the integration of regions into networks to support cognitive function and academic achievement.
The analysis of different patterns of diffusivity allows us to speculate about the biological properties of white matter structure that may differ as a function of aerobic fitness in children. First, our data suggest that higher fit children have greater FA, or more tightly bundled and structurally compact fibers, in the corpus callosum, specifically in the body of the white matter tract. This effect in children supports and extends the association between aerobic fitness and the body of the corpus callosum in an aging population (Johnson et al., 2012). Whereas the fitness-FA association in the corpus callosum in older adults was driven by negative fitness-RD associations (Johnson et al., 2012), we did not observe aerobic fitness group differences in RD or AD in this tract in the child sample. Thus, it is possible that the increased estimate of fiber integrity in this tract was not solely a function of greater myelination (RD) or axonal fiber diameter (AD). Other tissue differences may have played a role, such as increased neurotrophic factors or increased vascularization, two cellular effects of exercise which are observed in rodents that could influence white matter (Ding et al., 2006). Regardless of the biological mechanism, because the corpus callosum connects the left and right cerebral hemispheres and facilitates interhemispheric communication and the exchange of cognitive, motor and sensory information between the hemispheres (Gazzaniga, 1995), structurally compact fibers may help lead to superior cognitive performance in higher fit children, as suggested in previous work (Olesen et al., 2003; Nagy et al., 2004; Tamnes et al., 2012).
We also demonstrate significant effects of aerobic fitness on FA in the superior corona radiata and superior longitudinal fasciculus, driven by associations with RD. We suggest that aerobic fitness during childhood may influence fiber structural integrity and fiber alignment in these tracts perhaps via increased myelination (reductions in RD; Song et al., 2003, 2005; Sun et al., 2006, 2008). To link RD to myelination, Song et al. (2005) used an experimental model of demyelination and remyelination in a mouse brain and demonstrated that RD increased with demyelination and subsequently decreased with the progression of remyelination. The RD changes were found to be specific to changes in myelin integrity, and distinct from axonal injury (Song et al., 2005). Like the corpus callosum, no aerobic fitness differences were found in terms of AD in the superior corona radiata or superior longitudinal fasciculus, which suggests that axonal diameter, loss or damage may not relate to individual differences in fitness (Song et al., 2003; Budde et al., 2007). This is not unexpected given that axonal information is not influenced by myelination (Song et al., 2003; Budde et al., 2007). Because the corona radiata has ascending and descending tracts from the cerebral cortex that integrate information throughout the brain, and cognitively, the tract is known to play a role in processing speed, cognitive control and memory (Olesen et al., 2003; Bendlin et al., 2010; Niogi et al., 2010; Chaddock-Heyman et al., 2013), it is important to investigate its plasticity. Similarly, as the superior longitudinal fasciculus provides bidirectional information transfer between the frontal and parietal cortex (Petrides and Pandya, 1984; Schmahmann and Pandya, 2006), and these connections have been found to correlate with performance on tasks of attentional and interference control (Chaddock-Heyman et al., 2013), and working memory in children (Nagy et al., 2004), our data have promising implications for the influence of aerobic fitness on brain health during childhood.
Whereas we demonstrate that aerobic fitness is associated with white matter microstructure across a number of tracts throughout the brain, the results also demonstrate some specificity. The fitness groups did not differ in the structure of the posterior thalamic radiation or cerebral peduncle, two tracts involved in motor function. Given our recent report of an association between interference control and the microstructure of the posterior thalamic radiation and cerebral peduncle (Chaddock-Heyman et al., 2013), and our reports that higher fit children show less interference RT than lower fit peers (Chaddock et al., 2010b), it is surprising that higher fit children did not have greater estimates of structure in these lower level motor tracts. However, some research suggests that cognitive gains with physical activity and fitness are specific to tasks that require increased cognitive control (Kramer et al., 1999; Colcombe and Kramer, 2003; Colcombe et al., 2004; Chaddock et al., 2011), so it is possible that aerobic fitness would not influence lower level motor tracts, as observed herein.
This study provides a first step in identifying the relationship between aerobic fitness and white matter microstructure during childhood, but the results should be evaluated in the context of their limitations. Studies with larger sample sizes, whole-brain exploratory analyses, a physical activity intervention, and a number of cognitive tasks should be conducted to confirm the complex relationship among physical activity, aerobic fitness, white matter structure, and cognition. Because we approached the study with a priori hypotheses, additional investigations should employ whole-brain analyses corrected for multiple comparisons. Also, it is important to remember that DTI does not measure tissue parameters (e.g., fiber integrity, myelination) directly, but measures the displacement of water molecules. Thus, indices of underlying microstructural properties can only be inferred from this displacement. Furthermore, because there are many axons within a voxel and TBSS only selects one axon per voxel, a number of different tissue parameters can result in differences in FA, RD, and AD.
These results arrive at an important time, as children become increasingly unfit and sedentary, and educators reduce or eliminate opportunities for physical activity during the school day in favor of academic topics (Centers for Disease Control and Prevention, 2010). In fact, white matter microstructure in the corona radiata has been found to relate to mathematics performance in the classroom (van Eimeren et al., 2008), which raises the possibility that the removal of fitness training opportunities during the school day may unintentionally have deleterious effects on white matter tracts, potentially reducing scholastic achievement. In fact, our study raises the possibility that greater white matter integrity, perhaps via greater myelination, is one pathway by which higher fit children excel in cognitive and academic performance compared to their less fit peers. Hopefully these findings will reinforce the importance of aerobic fitness during development and lead to additional physical activity opportunities in and out of the school environment.
Statements
Acknowledgments
Funding was provided by grants from the National Institute on Aging at the National Institute of Health to Arthur Kramer (RO1 AG25032 and R37 AG025667) and the National Institute of Child Health and Human Development (HD 055352) to Charles Hillman. Thank you to Holly Tracy and Nancy Dodge for their help with data collection.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
1.^http://www.fmrib.ox.ac.uk/fsl/fdt
2.^http://www.fmrib.ox.ac.uk/fsl/data/atlas-descriptions.html#wm
References
1
American College of Sports Medicine. (2006). ACSM’s Guidelines for Exercise Testing and Prescription7th Edn. New York, NY: Lippincott Williams & Wilkins.
2
AnderssonJ. R.JenkinsonM.SmithS. (2007a). TR07JA1: Non-Linear Optimisation.Available at: http://www.fmrib.ox.ac.uk/analysis/techrep (accessed March 1 2014).
3
AnderssonJ. R.JenkinsonM.SmithS. (2007b). TR07JA2: Non-Linear Registration, Aka Spatial Normalisation. Available at: www.fmrib.ox.ac.uk/analysis/techrep (accessed March 1 2014).
4
Barnea-GoralyN.MenonV.EckertM.TammL.BammerR.KarchemskiyA.et al (2005). White matter development during childhood and adolescence: a cross-sectional diffusion tensor imaging study.Cereb. Cortex151848–1854. 10.1093/cercor/bhi062
5
Bar-OrO. (1983). Pediatric Sports Medicine For The Practitioner: From Physiologic Principles to Clinical Applications. New York, NY: Springer. 10.1007/978-1-4612-5593-2
6
BasserP. J. (1995). Inferring microstructural features and the physiological state of tissues from diffusion-weighted images.NMR Biomed.8333–344. 10.1002/nbm.1940080707
7
BeaulieuC. (2002). The basis of anisotropic water diffusion in the nervous system: a technical review.NMR Biomed.15435–455. 10.1002/nbm.782
8
BendlinB. B.FitzgeraldM. E.RiesM. L.XulG.KastmanE. K.ThielB. W.et al (2010). White matter in aging and cognition: a cross-sectional study of microstructure in adults aged eighteen to eighty-three.Dev. Neuropsychol.35257–277. 10.1080/87565641003696775
9
BirnbaumA. S.LytleL. A.MurrayD. M.StoryM.PerryC. L.BoutelleK. N. (2002). Survey development for assessing correlates of young adolescents’ eating.Am. J. Health Behav.26284–295. 10.5993/AJHB.26.4.5
10
BuddeM. D.KimJ. H.LiangH. F.SchmidtR. E.RussellJ. H.CrossA. H.et al (2007). Toward accurate diagnosis of white matter pathology using diffusion tensor imaging.Magn. Reson. Med.57688–695. 10.1002/mrm.21200
11
BungeS. A.CroneE. A. (2009). “Neural correlates of the development of cognitivecontrol,” inNeuroimaging in Developmental Clinical NeuroscienceedsRumseyJ.ErnstM. (Cambridge: Cambridge University Press) 22–37. 10.1017/CBO9780511757402.005
12
BurzynskaA. Z.PreuschhofC.BäckmanL.NybergL.LiS. C.LindenbergerU.et al (2010). Age-related differences in white matter microstructure: region-specific patterns of diffusivity.Neuroimage492104–2112. 10.1016/j.neuroimage.2009.09.041
13
CastelliD. M.HillmanC. H.BuckS. M.ErwinH. E. (2007). Physical fitness and academic achievement in third- and fifth-grade students.J. Sport Exerc. Psychol.29239–252.
14
Centers for Disease Control and Prevention. (2010). The Association Between School-Based Physical Activity, Including Physical Education, and Academic Performance.Available at: http://www.cdc.gov/healthyyouth/health_and_academics/pdf/pape_executive_summary.pdf (accessed March 1 2014).
15
ChaddockL.EricksonK. I.PrakashR. S.KimJ. S.VossM. W.VanPatterM.et al (2010a). A neuroimaging investigation of the association between aerobic fitness, hippocampal volume and memory performance in preadolescent children.Brain Res.1358172–183. 10.1016/j.brainres.2010.08.049
16
ChaddockL.EricksonK. I.PrakashR. S.VanPatterM.VossM. V.PontifexM. B.et al (2010b). Basal ganglia volume is associated with aerobic fitness in preadolescent children.Dev. Neurosci.32249–256. 10.1159/000316648
17
ChaddockL.EricksonK. I.PrakashR. S.VossM. V.VanPatterM.PontifexM. B.et al (2012a). A functional MRI investigation of the association between childhood aerobic fitness and neurocognitive control.Biol. Psychol.89260–268. 10.1016/j.biopsycho.2011.10.017
18
ChaddockL.VossM. W.KramerA. F. (2012b). Physical activity and fitness effects on cognition and brain health in children and older adults.Kinesiol. Rev.137–45.
19
ChaddockL.PontifexM.HillmanC. H.KramerA. F. (2011). A review of the relation of aerobic fitness and physical activity to brain structure and function in children.J. Int. Neuropsychol. Soc.17975–985. 10.1017/S1355617711000567
20
Chaddock-HeymanL.EricksonK. I.VossM. W.PowersJ. P.KnechtA. M.PontifexM. B.et al (2013). White matter microstructure is associated with cognitive control in children.Biol. Psychol.94109–115. 10.1016/j.biopsycho.2013.05.008
21
ChomitzV. R.SliningM. M.McGowanR. J.MitchellS. E.DawsonG. F.HackerK. A. (2009). Is there a relationship between physical fitness and academic achievement? Positive results from public school children in the northeastern United States.J. Sch. Health7930–37. 10.1111/j.1746-1561.2008.00371.x
22
CoeD. P.PivarnikJ. M.WomackC. J.ReevesM. J.MalinaR. M. (2006). Effect of physical education and activity levels on academic achievement in children.Med. Sci. Sports Exerc.381515–1519. 10.1249/01.mss.0000227537.13175.1b
23
ColcombeS. J.EricksonK. I.ScalfP. E.KimJ. S.PrakashR.McAuleyE.et al (2006). Aerobic exercise training increases brain volume in aging humans.J. Gerontol. A Biol. Sci. Med. Sci.611166–1170. 10.1093/gerona/61.11.1166
24
ColcombeS. J.KramerA. F. (2003). Fitness effects on the cognitive function of older adults: a meta-analytic study.Psychol. Sci.14125–130. 10.1111/1467-9280.t01-1-01430
25
ColcombeS. J.KramerA. F.EricksonK. I.ScalfP.McAuleyE.CohenN. J.et al (2004). Cardiovascular fitness, cortical plasticity, and aging.Proc. Natl. Acad. Sci. U.S.A.1013316–3321. 10.1073/pnas.0400266101
26
DavisC. L.TomporowskiP. D.McDowellJ. E.AustinB. P.MillerP. H.YanasakN. E.et al (2011). Exercise improves executive function and achievement and alters brain activation in overweight children: a randomized, controlled trial.Health Psychol.3091–98. 10.1037/a0021766
27
DiamondA.LeeK. (2011). Interventions shown to aid executive function development in children 4–12 years old.Science333959–964. 10.1126/science.1204529
28
DingY. H.LiJ.ZhouY.RafolsJ. A.ClarkJ. C.DingY. (2006). Cerebral angiogenesis and expression of angiogenic factors in aging rats after exercise.Curr. Neurovasc. Res.315–23. 10.2174/156720206775541787
29
DuPaulG. J.PowerT. J.AnastopoulosA.ReidR. (1998). ADHD Rating Scale – IV: Checklists, Norms, And Clinical Interpretation.New York, NY: Guilford Press.
30
FreedsonP. S.GoodmanT. L. (1993). “Measurement of oxygen consumption,” inPediatric Laboratory Exercise Testing: Clinical Guidelinesed.RowlandT. W. (Champaign, IL: Human Kinetics) 91–113.
31
GazzanigaM. S. (1995). Principles of human brain organization derived from split-brain studies.Neuron14217–228. 10.1016/0896-6273(95)90280-5
32
HillmanC. H.BuckS. M.ThemansonJ. R.PontifexM. B.CastelliD. M. (2009). Aerobic fitness and cognitive development: event-related brain potential and task performance of executive control in preadolescent children.Dev. Psychol.45114–129. 10.1037/a0014437
33
HuaK.ZhangJ.WakanaS.JiangH.LiX.ReichD. S.et al (2008). Tract probability maps in stereotaxic spaces: analyses of white matter anatomy and tract-specific quantifcation.Neuroimage39336–347. 10.1016/j.neuroimage.2007.07.053
34
JohnsonN. F.KimC.ClaseyJ. L.BaileyA.GoldB. T. (2012). Cardiorespiratory fitness is positively correlated with cerebral white matter integrity in healthy seniors.Neuroimage591514–1523. 10.1016/j.neuroimage.2011.08.032
35
KaufmanA. S.KaufmanN. L. (1990). Kaufman Brief Intelligence Test.Circle Pines: AGS.
36
KramerA. F.HahnS.CohenN.BanichM.McAuleyE.HarrisonC.et al (1999). Aging, fitness, and neurocognitive function.Nature400418–419. 10.1038/22682
37
ListonC.WattsR.TottenhamN.DavidsonM. C.NiogiS.UlugA. M.et al (2006). Fronto-striatal microstructure modulates efficient recruitment of cognitive control.Cereb. Cortex16553–560. 10.1093/cercor/bhj003
38
MadsenK. S.BaareW. F.VestergaardM.SkimmingeA.EjersboL. R.RamsøyT. Z.et al (2010). Response inhibition is associated with white matter microstructure in children.Neuropsychologia48854–862. 10.1016/j.neuropsychologia.2009.11.001
39
MarksB. L.MaddenD. J.BucurB.ProvenzaleJ. M.WhiteL. E.CabezaR.et al (2007). Role of aerobic fitness and aging on cerebral white matter integrity.Ann. N. Y. Acad. Sci.1097171–174. 10.1196/annals.1379.022
40
MoriS.WakanaS.Van ZijlP. C. M. (2005). MRI Atlas of Human White Matter.Amsterdam: Elsevier.
41
MuetzelR. L.CollinsP. F.MuellerB. A.SchisselA. M.LimK. O.LucianaM. (2008). The development of corpus callosum microstructure and associations with bimanual task performance in healthy adolescents.Neuroimage391918–1925. 10.1016/j.neuroimage.2007.10.018
42
NagyZ.WesterbergH.KlingbergT. (2004). Maturation of white matter is associated with the development of cognitive functions during childhood.J. Cogn. Neurosci.161227–1233. 10.1162/0898929041920441
43
NairG.TanahashiY.LowH. P.Billings-GagliardiS.SchwartzW. J.DuongT. Q. (2005). Myelination and long diffusion times alter diffusion-tensor-imaging contrast in myelin-deficient shiverer mice.Neuroimage28165–174. 10.1016/j.neuroimage.2005.05.049
44
NiogiS.MukherjeeP.GhajarJ.McCandlissB. D. (2010). Individual differences in distinct components of attention are linked to anatomical variations in distinct white matter tracts.Front. Neuroanat.4:2. 10.3389/neuro.05.002.2010
45
OldfieldR. C. (1971). The assessment and analysis of handedness: the Edinburgh inventory.Neuropsychologia997–113. 10.1016/0028-3932(71)90067-4
46
OlesenP.NagyZ.WesterbergH.KlingbergT. (2003). Combined analysis of DTI and fMRI data reveals a joint maturation of white and grey matter in a fronto-parietal network.Cogn. Brain Res.1848–57. 10.1016/j.cogbrainres.2003.09.003
47
OlsonE. A.CollinsP. F.HooperC. J.MuetzelR.LimK. O.LucianaM. (2009). White matter integrity predicts delay discounting behavior in 9- to 23-year-olds: a diffusion tensor imaging study.J. Cogn. Neurosci.211406–1421. 10.1162/jocn.2009.21107
48
PausT. (2010). Growth of white matter in the adolescent brain: myelin or axon?Brain Cogn.7226–35. 10.1016/j.bandc.2009.06.002
49
PetridesM.PandyaD. N. (1984). Projections to the frontal cortex from the posterior parietal region in the rhesus monkey.J. Comp. Neurol.228105–116. 10.1002/cne.902280110
50
PierpaoliC.BasserP. J. (1996). Toward a quantitative assessment of diffusion anisotropy.Magn. Reson. Med.36893–906. 10.1002/mrm.1910360612
51
PierpaoliC.JezzardP.BasserP. J.BarnettA.Di ChiroG. (1996). Diffusion tensor MR imaging of the human brain.Radiology201637–648. 10.1148/radiology.201.3.8939209
52
PontifexM. B.RaineL. B.JohnsonC. R.ChaddockL.VossM. W.CohenN. J.et al (2011). Cardiorespiratory fitness and the flexible modulation of cognitive control in preadolescent children.J. Cogn. Neurosci.231332–1345. 10.1162/jocn.2010.21528
53
RykhlevskaiaE.GrattonG.FabianiM. (2008). Combining structural and functional neuroimaging data for studying brain connectivity: a review.Psychophysiology45173–187. 10.1111/j.1469-8986.2007.00621.x
54
SchmahmannJ. D.PandyaD. N. (2006). Fiber Pathways of The Brain.New York: Oxford University Press. 10.1093/acprof:oso/9780195104233.001.0001
55
SchmithorstV. J.YuanW. (2010). White matter development during adolescence as shown by diffusion MRI.Brain Cogn.7216–25. 10.1016/j.bandc.2009.06.005
56
SenP. N.BasserP. J. (2005). A model for diffusion in white matter in the brain.Biophys. J.892927–2938. 10.1529/biophysj.105.063016
57
ShvartzE.ReiboldR. C. (1990). Aerobic fitness norms for males and females aged 6 to 75 years: a review.Aviat. Space Environ. Med.613–11.
58
SmithS. M. (2002). Fast robust automated brain extraction.Hum. Brain Mapp.17143–155. 10.1002/hbm.10062
59
SmithS. M.JenkinsonM.Johansen-BergH.RueckertD.NicholsT. E.MackayC.et al (2006). Tract-based spatial statistics: voxelwise analysis of multi-subject diffusion data.Neuroimage311487–1505. 10.1016/j.neuroimage.2006.02.024
60
SongS. K.SunS. W.JuW. K.LinS. J.CrossA. H.NeufeldA. H. (2003). Diffusion tensor imaging detects and differentiates axon and myelin degeneration in mouse optic nerve after retinal ischemia.Neuroimage201714–1722. 10.1016/j.neuroimage.2003.07.005
61
SongS. K.SunS. W.RamsbottomM. J.ChangC.RussellJ.CrossA. H. (2002). Dysmyelination revealed through MRI as increased radial (but unchanged axial) diffusion of water.Neuroimage171429–1436. 10.1006/nimg.2002.1267
62
SongS. K.YoshinoJ.LeT. Q.LinS. J.SunS. W.CrossA. H.et al (2005). Demyelination increases radial diffusivity in corpus callosum of mouse brain.Neuroimage26132–140. 10.1016/j.neuroimage.2005.01.028
63
SunS. W.LiangH. F.CrossA. H.SongS. K. (2008). Evolving Wallerian degeneration after transient retinal ischemia in mice characterized by diffusion tensor imaging.Neuroimage401–10. 10.1016/j.neuroimage.2007.11.049
64
SunS. W.LiangH. F.LeT. Q.ArmstrongR. C.CrossA. H.SongS. K. (2006). Differential sensitivity of in vivo and ex vivo diffusion tensor imaging to evolving optic nerve injury in mice with retinal ischemia.Neuroimage321195–1204. 10.1016/j.neuroimage.2006.04.212
65
TamnesC. K.FjellA. M.WestlyeL. T.OstbyY.WalhovdK. B. (2012). Becoming consistent: developmental reductions in intraindividual variability in reaction time are related to white matter integrity.J. Neurosci.32972–892. 10.1523/JNEUROSCI.4779-11.2012
66
TaylorS. J. C.WhincupP. H.HindmarshP. C.LampeF.OdokiK.CookD. G. (2001). Performance of a new pubertal self-assessment questionnaire: a preliminary study.Paediatr. Perinat. Epidemiol.1588–94. 10.1046/j.1365-3016.2001.00317.x
67
TrudeauF.ShephardR. J. (2008). Physical education, school physical activity, school sports and academic performance.Int. J. Behav. Nutr. Phys. Act.510. 10.1186/1479-5868-5-10
68
UtterA. C.RobertsonR. J.NiemanD. C.KangJ. (2002). Children’s OMNI scale of perceived exertion: walking/running evaluation.Med. Sci. Sporst Exerc.34139–144. 10.1097/00005768-200201000-00021
69
van EimerenL.NiogiS. N.McCandlissB. D.HollowayI. D.AnsariD. (2008). White matter microstructures underlying mathematical abilities in children.Neuroreport191117–1121. 10.1097/WNR.0b013e328307f5c1
70
VossM. W.ChaddockL.KimJ. S.VanPatterM.PontifexM. B.RaineL. B.et al (2011). Aerobic fitness is associated with greater efficiency of the network underlying cognitive control in preadolescent children.Neuroscience199166–176. 10.1016/j.neuroscience.2011.10.009
71
VossM. W.HeoS.PrakashR. S.EricksonK. I.AlvesH.ChaddockL.et al (2012). The influence of aerobic fitness on cerebral white matter integrity and cognitive function in older adults: results of a one-year exercise intervention.Hum. Brain Mapp.342972–2985. 10.1002/hbm.22119
72
WakanaS.CaprihanA.PanzenboeckM. M.FallonJ. H.PerryM.GollubR. L.et al (2007). Reproducibility of quantitative tractography methods applied to cerebral white matter.Neuroimage36630–644. 10.1016/j.neuroimage.2007.02.049
Summary
Keywords
cardiorespiratory fitness, development, diffusion tensor imaging, fiber tracts, microstructure
Citation
Chaddock-Heyman L, Erickson KI, Holtrop JL, Voss MW, Pontifex MB, Raine LB, Hillman CH and Kramer AF (2014) Aerobic fitness is associated with greater white matter integrity in children. Front. Hum. Neurosci. 8:584. doi: 10.3389/fnhum.2014.00584
Received
02 April 2014
Accepted
14 July 2014
Published
19 August 2014
Volume
8 - 2014
Edited by
Francesco Di Russo, University of Rome “Foro Italico”, Italy
Reviewed by
Giancarlo Zito, San Giovanni Calibita Fatebenefratelli Hospital, Italy; Tomas Paus, University of Toronto, Canada
Copyright
© 2014 Chaddock-Heyman, Erickson, Holtrop, Voss, Pontifex, Raine, Hillman and Kramer.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Laura Chaddock-Heyman, Department of Psychology and Beckman Institute for Advanced Science and Technology, University of Illinois at Urbana-Champaign, 405 North Mathews Avenue, Urbana, IL 61801, USA e-mail: lchaddo2@illinois.edu
This article was submitted to the journal Frontiers in Human Neuroscience.
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.