- 1Sobell Department of Motor Neuroscience and Movement Disorders, Institute of Neurology, University College London, London, United Kingdom
- 2Department of Neuroscience, Shriners Hospitals Pediatric Research Center, Lewis Katz School of Medicine, Temple University, Philadelphia, PA, United States
Editorial on the Research Topic
Gene Therapy for the Central and Peripheral Nervous System
It is with great pleasure that we present the research topic dedicated to Gene Therapy for the Central (CNS) and Peripheral Nervous System (PNS). Gene therapy is at the cutting-edge of techniques utilized to develop novel therapeutics to treat insult(s) to the brain, spinal cord and/or peripheral nerves. Indeed, gene therapy can be applied via many different routes and as such can overcome the many obstacles facing oral and systemic delivery of synthetic drugs, thereby permit greater targeting of neural tissue. With this advantage, gene therapy has the potential to (1) correct disease-causing DNA mutations, (2) eliminate toxic RNA/proteins and/or (3) increase the expression of therapeutic proteins. Gene therapy can ameliorate aspects of debilitating neurological diseases and thus, can provide a platform for functional recovery.
Over the last few years, advances in basic science and technology have enabled enhanced pre-clinical strategies culminating in the emergence of sophisticated treatment options available for patients. The most recent gene therapy success is an FDA and EMA approved antisense oligonucleotide (ASO) that is the first and only treatment option available to treat spinal muscular atrophy. Moreover, ASOs have successfully reduced toxic protein levels in a phase 1/2a clinical trial to treat Huntington's Disease (HD). This treatment option was considered safe and well tolerated and was granted orphan drug designation by the FDA and EMA. These advances offer great hope to patients, their families, clinicians and basic scientists, and emphasizes the potential of gene therapy to treat “the incurable” neurological diseases/disorders.
In 2013, we launched this research topic to provide a platform to continue the discussion and amalgamate recent advances in gene therapy technology and strategies for the treatment of PNS and CNS disorders. In conclusion, we are proud to present an extremely productive discussion consisting of 18 articles in total comprised of eight original articles, five full-length reviews, three mini-reviews and one hypothesis and theory article and represents a world-wide collaboration with submissions received from USA, United Kingdom, Europe, Asia, New Zealand and Australia. This research topic discusses gene therapy strategies to treat neurological disorders including amyotrophic lateral sclerosis (ALS), spinal muscular atrophy (SMA), spinocerebellar ataxia (SCA), neuropathic pain, stroke, peripheral nerve injury and repair.
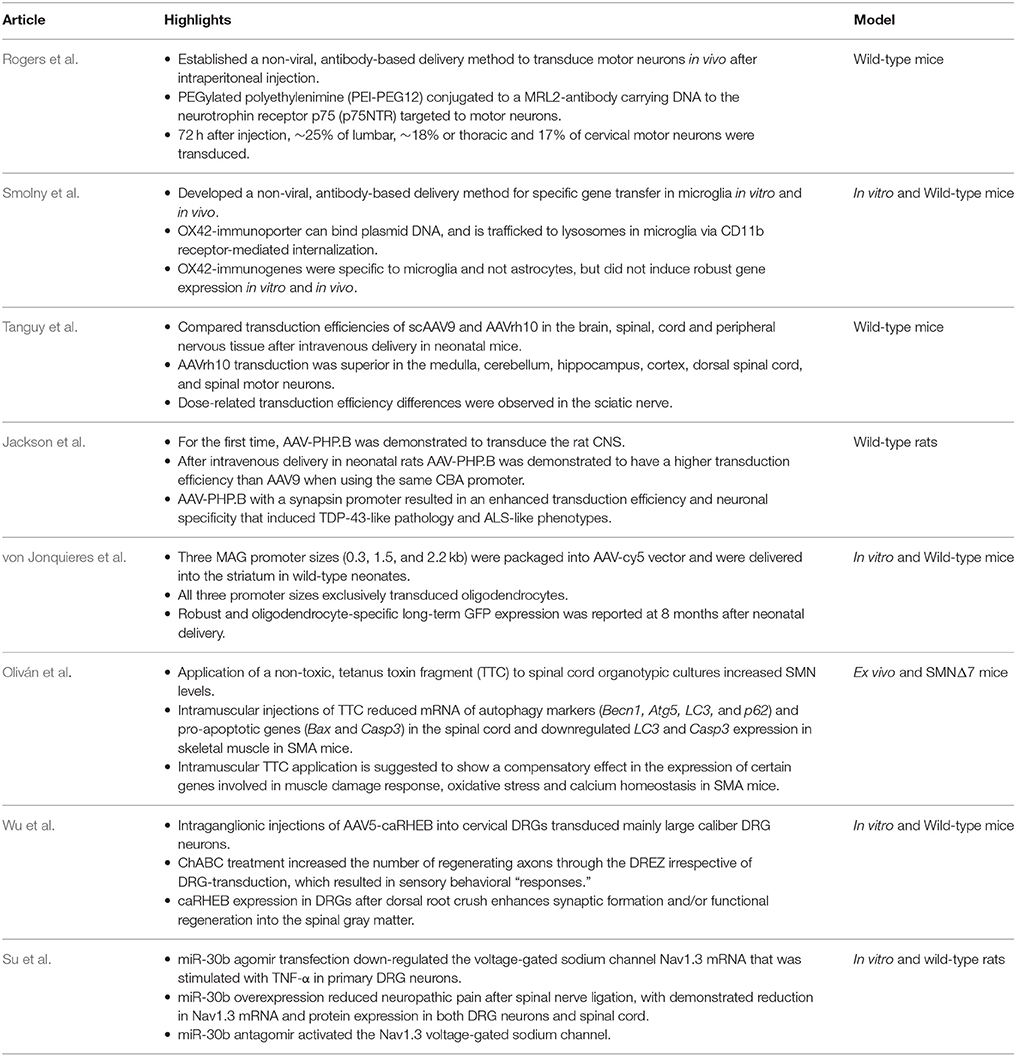
The novel studies presented in this research topic focus on improving transduction efficiency and gene transfer using adeno-associated virus (AAVs) and non-viral methods (Table 1). Tanguy et al compare the transduction efficiency between scAAV9 and AAVrh10 serotypes after systemic delivery. von Jonquieres et al. continue to demonstrate glial-specific transduction in the brain after injecting a chimeric AAV1/2 vector into neonatal striatum, despite using three differently sized MAG promoters. Jackson et al. combined an engineered AAV-serotype with a neuronal-specific promoter to increase transduction efficiency and reduce off-target effects after intravenous delivery. Wu et al. increase the intrinsic growth potential of injured sensory axons using combinatory treatment involving chondroitinase ABC and AAV-mediated constitutively active GTPase Rheb (Wu et al.).
In addition, Smolny et al. present a non-viral, antibody-based delivery method for microglia-specific gene transfer. Rogers et al. describe spinal motor neuron transduction after peripheral delivery of plasmid DNA as a PEGylated polyethylenimine conjugated antibody. Oliván et al. demonstrate that atoxic-tetanus neurotoxin fragment modifies expression of autophagy and pro-apoptotic genes in the spinal cord and skeletal muscle. Su et al. show miRNA-mediated suppression of specific voltage-gated sodium channels can alleviate neuropathic pain.
This research topic also includes a number of full-length and mini-reviews (Table 2). Murlidharan et al. describe the biology of different AAV strains, including their transduction profiles, cellular tropisms and mechanisms of CNS transport, for increased translational application. Parr-Brownlie et al. review lentiviral-vector approaches to enhance transduction efficiency, mediate cell-specificity, restrict gene expression spatially and temporally, and discuss limitations and future prospects. Tan et al. critically analyse the advantages and disadvantages of strategies using non-viral nucleic acids to deliver therapeutic genes by circumventing the immune response and thus, appeasing safety concerns potentially associated with viral-mediated gene therapies. Wagner et al. examine the gene and stem cell pre-clinical therapeutic options that preserve Purkinje cell health to treat SCA, and suggest that RNA interference (RNAi) might have great promise. Finally, Tosolini and Sleigh discuss important considerations learned from the success of a recently FDA- and EMA-approved gene therapy for SMA to develop viable gene therapies and strategies to treat ALS.
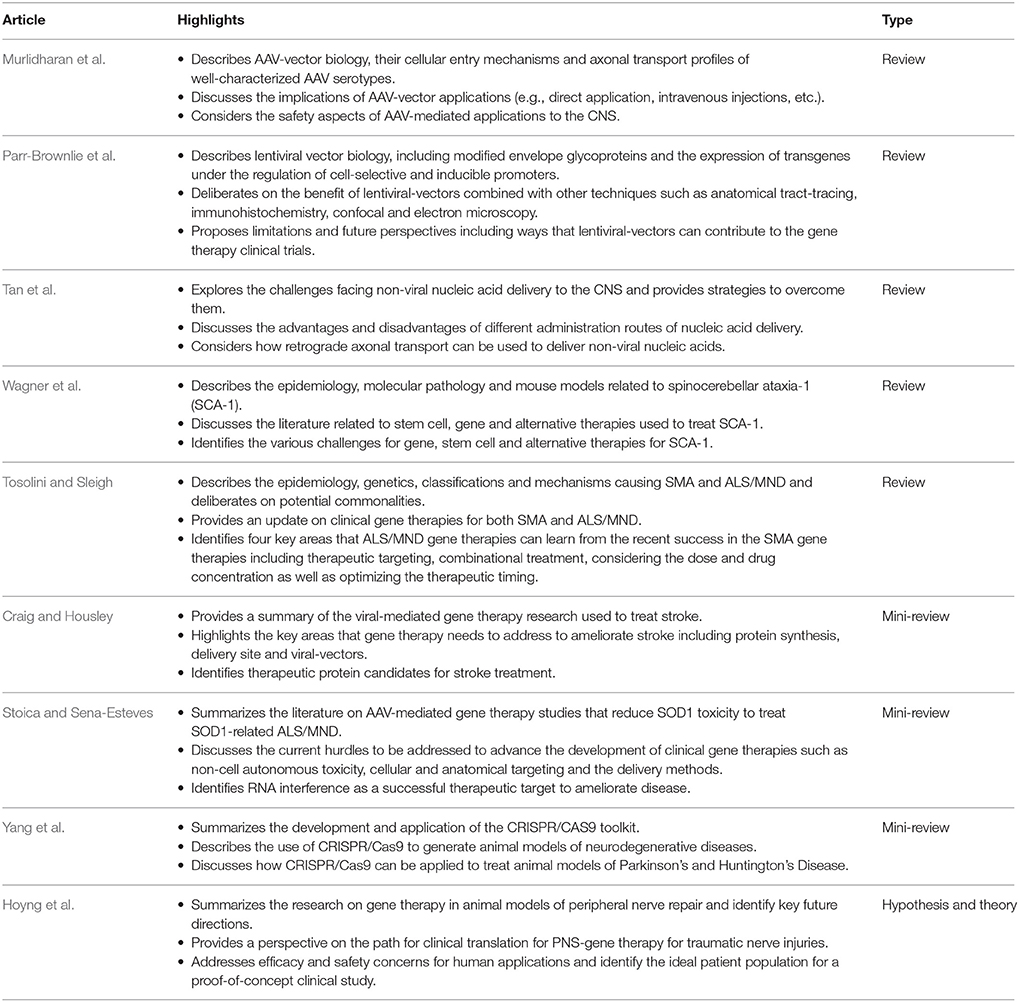
Table 2. Highlights from the review, mini-review and hypothesis and theory articles published in this research topic.
For the mini-reviews, Yang et al. discuss the “hot topic” of CRISPR/Cas9 gene editing and how these tools can be applied to various research models and the development of treatments for neurodegenerative disease, such as HD and Parkinson's disease. Craig and Housley focus on gene therapy approaches for stroke and discuss injury mechanisms, appropriate timings for therapeutic intervention and deliberate on candidate therapeutic proteins as therapeutic options. Stoica and Sena-Esteves deliver a succinct mini-review of AAV-mediated SOD1-ALS amelioration strategies and describe the hurdles to overcome for CNS gene delivery.
The Hypothesis and theory submission by Hoyng et al. summarize the state-of-research for peripheral nerve repair, identify future targets and provide a translational perspective on PNS gene therapy.
This research forum describes many important characteristics of diverse gene therapy applications for the development of tangible treatment options for different CNS and PNS disorders. Indeed, the advances in gene therapy strategies discussed within this research topic give hope that treatment options for many incurable CNS and PNS disorders are closer to becoming a viable clinical option.
Author Contribution
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
Funding
AT holds a postdoctoral position supported by a Wellcome Trust Senior Investigator Award [107116/Z/15/Z] to Giampietro Schiavo (Institute of Neurology, University College London). This work was supported by National Institute of Health (R01NS103481), Shriners Hospitals for Pediatric Research (SHC-84050) and DOD (SC140089) to GS.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Keywords: gene therapy, CNS, PNS, AAV, lentivirus, non-viral vectors, neurons, glia
Citation: Tosolini AP and Smith GM (2018) Editorial: Gene Therapy for the Central and Peripheral Nervous System. Front. Mol. Neurosci. 11:54. doi: 10.3389/fnmol.2018.00054
Received: 14 January 2018; Accepted: 07 February 2018;
Published: 22 February 2018.
Edited by:
Hermona Soreq, Hebrew University of Jerusalem, IsraelReviewed by:
Shai Berlin, Technion Israel Institute of Technology, IsraelCopyright © 2018 Tosolini and Smith. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Andrew P. Tosolini, YS50b3NvbGluaUB1Y2wuYWMudWs=
George M. Smith, Z2VvcmdlLnNtaXRoQHRlbXBsZS5lZHU=
 Andrew P. Tosolini
Andrew P. Tosolini George M. Smith
George M. Smith