Abstract
Objective:
Gestational alloimmune liver disease is a rare and serious condition caused by a maternal-fetal alloimmune disorder. There are not many studies about the antenatal treatment (IVIG infusion) of affected fetuses as the diagnosis is generally made postnatally. The possibility of an early diagnosis by means of ultrasonography and a gynecologist's assesment can provide prompt treatment of this disease.
Case report:
We report the case of 38-year-old pregnant woman referred to our centre in view of severe fetal hydrops seen by ultrasound at 31 weeks + 1 day gestation. A male infant was born and subsequently died after developing liver failure. Postmortem examination revealed the presence of diffuse hepatic fibrosis in the absence of hemosiderin deposits and no extrahepatic siderosis. Immunohistochemical analysis was also performed which showed diffuse hepatocyte positivity for the terminal complement complex (C5b-C9) confirming the suspicion of GALD.
Methods:
A comprehensive literature search published from 2000 to 2022 was conducted on PubMed and Scopus. Paper selection was performed following the PRISMA guidelines. Fifteen retrospective studies were identified and selected.
Results:
A total of 15 manuscripts describing 26 cases were finally included in our research. Twenty-two fetuses/newborns with suspected GALD were studied, of which 11 had a confirmed histopathological diagnosis of GALD. Prenatal diagnosis of gestational alloimmune liver disease is difficult because ultrasound findings may be absent or nonspecific. Only one case report described fetal hydrops similar to our clinical case. As highlighted by the current case, in fetuses presenting with hydrops, once the most common etiologies have been excluded, hepatobiliary complications and liver failure caused by GALD should be considered
Conclusions:
Global knowledge of this disorder and its wide spectrum of presentations may help to increase the number of cases that are diagnosed early and accurately. The recurrence rate of an infant being affected with GALD in another pregnancy is more that 90%. Recurrence however can be prevented by treatment with IVIG during pregnancy. This highlights the importance of having obstetricians and pediatricians familiar with gestational alloimmune liver disease.
Introduction
Gestational alloimmune liver disease is a rare and serious condition caused by a maternal-fetal alloimmune disorder, which occurs antenatally and leads to severe fetal liver failure with possible neonatal hemochromatosis (1).
Several studies showed that prenatal treatment using IVIG infusion in women at risk of recurrence of gestational alloimmune disease can prevent the disease in subsequent pregnancies. However, there are not many studies about the antenatal treatment of affected fetuses as the diagnosis is generally made postnatally (2).
Therefore, the possibility of an early diagnosis by means of ultrasonography and a gynecologist's assesment can provide prompt treatment of this disease (3).
Case presentation
A 38-year-old woman, primigravida at 31 weeks + 1 day gestation, was referred to our centre with severe fetal hydrops seen by ultrasound. The patient reported a reduction of fetal movements since the week before, on a background of an otherwise normal pregnancy. On admission in our unit, fetal ultrasound scan showed significant cephalic edema and bilateral thoracic effusion.
The patient's medical history was unremarkable. She did not report risk factors and chronic medications. There was no familial history of congenital defects.
The routine pregnancy investigations were performed and showed: positive Rhesus blood, normal blood tests, negative serological profile, normal urine tests and negative RT PCR Covid19.
The first and second trimester assessments of pregnancy were normal.
On admission the serological investigations and autoimmunity profile showed negative results. A fetal cardiotocograph trace displayed reduced variability. Spontaneous premature rupture of amnio-chorionic membranes occurred a few hours later with stained meconium. Decision for delivery by emergency cesarean section was taken.
A male infant was born with a birth weight of 2,500 g (>97°p). The 1- and 5-minute Apgar scores were 1 and 3, respectively. The pH at birth was 7,31. The infant was admitted to the intensive care unit for further observation and care. On admission he was hypotonic, unresponsive and diffusely edematous.
His laboratory tests showed thrombocytopenia, abnormal synthetic liver function (hypoalbuminemia and coagulopathy), increased liver enzymes and increased ferritin. Multiple transfusions of concentrated blood cells, platelets, and plasma were administered.
Metabolic and inherited diseases associated with early onset neonatal liver failure were ruled out. Abdominal ultrasound showed regular liver morphology and size, with smooth profile. Diffuse hyperechogenicity of parenchyma was seen with no evidence of focal lesions. Moderate intra-abdominal effusion was present in the perisplenic and perihepatic areas, as well as among the bowel loops. An echocardiogram showed no pericardial effusion and a slight increase in pulmonary pressure.
In light of a suspected diagnosis of acute liver failure (NALF), immunoglobulins IV were administered and an exanguinotransfusion was performed.
Unfortunately, the patient's condition deteriorated with the onset of early renal failure unresponsive to treatment and the appearance of multiple cerebral hemorrhagic areas documented on ultrasound and later on MRI. The patient died on the 10th day of postnatal life after progressive multiple organ failure.
The postmortem exam, which was formally allowed by parents, revealed the presence of diffuse hepatic fibrosis in the absence of hemosiderin deposits and no extrahepatic siderosis. Immunohistochemical analysis was also performed and showed diffuse hepatocyte positivity for the terminal complement complex (C5b-C9).
This last analysis supports the suspected diagnosis of gestational alloimmune liver disease even in the absence of visceral hemosiderin deposits.
Genetic investigations were also performed: the karyotype was normal, Array CGH showed a duplication of maternal origin on the short arm of chromosome 10. No pathogenetic significance can currently be attributed to the latter chromosomal finding. No pathogenic variants of the Neu1 gene have been identified at WES but only of the variant of unknown significance of the NPC1 gene (c.2257G > A), which is present in heterozygotes.
Materials and methods
A systematic review of literature was performed using Scopus (www.scopus.com, accessed on 19.07.2022) and PubMed (www.ncbi.nlm.nih.gov, accessed on 19.07.2022) databases in order to answer the clinical question. The search terms were: “GALD”, “Gestational Alloimmune Liver Disease”, “pregnancy AND GALD”, “pregnancy AND Gestational Alloimmune Liver Disease”. Articles describing one or more cases of GALD, published from January 2000 to July 2022, were identified from the above databases. Only papers written in English were included in our research. Studies were deemed eligible for inclusion in the present review if they described at least one case of GALD. All duplicate studies were excluded. Selected articles were independently reviewed by two authors (A.N. and S.Z.). The whole process is described in Table 1 and shows the list of included and excluded articles and the reason for exclusion.
Table 1
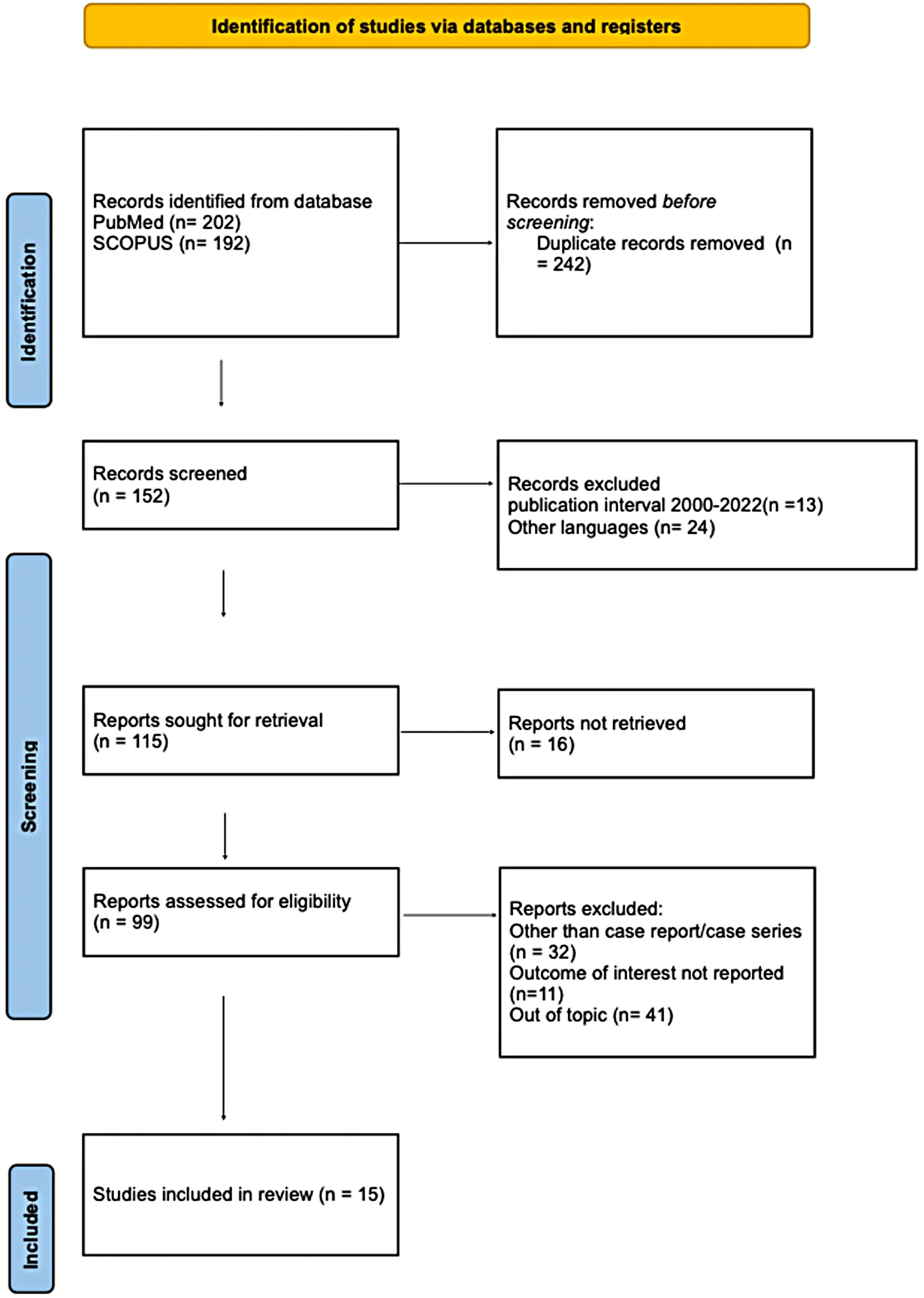 |
Prisma flow chart.
From: Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. doi: 10.1136/bmj.n71. For more information, visit: http://www.prisma-statement.org/.
We collected the following information: maternal age, parity, gestational age, ultrasound findings, clinical presentation, fetal/neonatal outcome, treatment, microsocopic diagnosis, maternal comorbidities. All this data is listed in Table 2.
Table 2
| Author | Title | Maternal age | Parity | EG | Ultrasound findings | Clinical presentation | Fetal/neonatal outcome | IG Therapy during pregnancy | Histological/immunohistochemical diagnosis | Maternal comorbidities |
|---|---|---|---|---|---|---|---|---|---|---|
| Moorhead R. | Successful pregnancy outcomes following intravenous immunoglobulin treatment in a woman with a previous fetal death in utero due to gestational alloimmune liver disease: A case report | 32 | >1 | 34 + 2 |
| Unknow | Fetal death | No | Yes | Low-grade squamous intraepithelial cervical lesion |
| Sarker M. | Intravenous immunoglobulin induced pancytopenia while preventing development of gestational alloimmune liver disease: A case report | 33 | 0 | 21 + 0 |
| Pulmonary hemaorrage | Neonatal death | No | No | Neonatal demise secondary to GALD |
| Sciard C. | Prenatal imaging features suggestive of liver gestational allo immune disease | Unknown | / | / | Case 1: - IUGR
| Case 4: splenomegaly | Case 1: fetal death Case 2: neonatal death Case 3: TOP Case 4: alive | Only in one case | Yes | Unknown |
| Concejo Iglesias P. | Usefulness of dual gradient-echo MR imaging for the prenatal | 34 | 0 | 27 + 2 |
| Unknown | Fetal death | Yes | Yes | No |
| Mariano da Rocha C.R. | Neonatal Liver Failure and Congenital Cirrhosis due to Gestational Alloimmune Liver Disease: A Case Report and Literature Review | Unknown | / | 31 + 4 |
|
| Neonatal death | No | Yes | No |
| Casas-Alba D. | Broadening the spectrum of neonatal hemochromatosis | 30 | / | 39 + 6 | / |
| Alive | Yes (not in pregnancy) | No | No |
| Yeh P.J. | Efficacy of Intravenous Immunoglobulin/Exchange Transfusion Therapy on Gestational Alloimmune Liver Disease | Unknown | 0 | 37 + 3 | / | Twin A: - Ascites
| Alive | Yes (not in pregnancy) | Yes | ANA+Anti-DNA+↑ IgG level ↓ C3 level |
| Anastasio H.B. | Gestational Alloimmune Liver Disease.A Devastating Condition Preventable With Maternal Intravenous Immunoglobulin | 30 | 0 | 29 |
|
| Neonatal death | No | No | No |
| Tsunoda T. | Neonatal liver failure owing to gestational alloimmune liver disease without iron overload | 26 | 0 | 34 |
|
| Alive (liver transplantation) | Yes (not in pregnancy) | Yes | No |
| Clarke NE. | Fulminant liver failure in a neonate | 25 | 0 | 27 + 6 |
|
| Neonatal death | No | Yes | No |
| Midorikawa H. | Disparate clinical findings in monochorionic twins with neonatal hemochromatosis | Unknown | / | 36 | Twin A: Oligohydramnios Twin B: Normal | Twin A: - Liver failure
| Alive | Yes only for twin A (not in pregnancy) | No | No |
| Flores-Torres J. | PIGA Mutations Can Mimic Neonatal Hemochromatosis | 22 | 0 | 37 |
|
| Alive | Yes (not in pregnancy) | No | No |
| Hutchings G. | Plasmapheresis as an Alternative to High-Dose Intravenous Immunoglobulin in the Prevention of Gestational Alloimmune Liver Disease | 27 | >1 | / | 1st pregnancy: - IUGR i 2nd pregnancy: terminated (diagnosis of sickle cell) 3rd pregnancy: - IUGR
| 1st:liver failure (liver transplantation) 5th: -Hepatic failure 6th: -Transaminitis
| 1st: neonatal death 2nd: terminated 3th: fetal death 4th: alive 5th: neonatal death 6th: alive | Only 6th case | Yes (only 1stcase) | Sicke cell trait |
| Iskandar A.T.P. | Neonatal hemochromatosis attributed to gestational alloimmune liver disease treated with IVIG and exchange transfusion therapy: an evidence-based case report | 29 | 0 | 33 |
|
| Alive | Yes (not in pregnancy) + plasmapheresis | No | No |
| Mc Adams R.M. | Ileal atresia and multiple jejunal perforations in a premature neonate with gestational alloimmune liver disease | / | / | 32 + 3 |
|
| Alive | Yes (not in pregnancy) | No | No |
Data of systematic review.
Twelve articles were not included because they did not report the outcome of interest and did not report any information about the pregnancy.
The main risk of bias of this work is that all papers selected in the literature are case reports and case series.
We use the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to elaborate this review and the literature quality has been evaluated through the use of the CARE (Case Report) check list.
Due to the rarity of this occurrence, we presented data in a descriptive manner.
Results
We described our clinical case and then we performed a literature review with MEDLINE (PubMed) and Scopus. We analyzed 15 manuscripts describing 26 cases. A summary of the characteristics is presented in Table 2.
Twenty-two fetuses/newborns with suspected GALD were studied. Of these, 11 had a histopathological diagnosis of GALD. In the other cases GALD diagnosis was made based on suggestive clinical findings and imaging assessments. The mean maternal age is 28,8 years; however, this information is reported in only 10 cases.
Gestational age at presentation ranges from a minimum of 21 + 0 weeks to a maximum of 39 + 6 gestational weeks. Six women were nulliparous. Only three women had comorbidities; one woman was a carrier of sickle cell trait (4), another had a previous diagnosis of LSIL (5) while the third presented with an elevated serum immunoglobulin G level, a low complement level, positive antinuclear antibody and positive anti-double-strand DNA antibody (6). However, none of these comorbidities have been described as an association with the development of GALD in pregnancy.
The clinical presentation is extremely heterogeneous. It was only in three case reports that the patient reported reduced fetal movement, as was in our case.
Only ten infants with diagnosis of GALD survived. Eight of them underwent post-natal intra-venous immunoglobulin therapy (IVIG), One case had IVIG combined with plasmapheresis (8), while two babies underwent a liver transplant (4, 9).
Regarding the ultrasound findings, the review showed that the most representative parameter was fetal growth restriction which was present in 13 fetuses (59%) followed by oligohydramnios present in 10 out of 22 fetuses (45%); fetal ascites (6/22, 27%) and pericardial effusion (3/22, 14%). Placental edema (3/22, 14%) was rare. Only one case described fetal hydrops as was the presentation in our case, but this case developed additional ultrasonographic signs such as severe placental edema, inferior vena cava dilatation and an irregular and nodular liver surface (10); A single case reported the following: splenomegaly, echogenic bowel and echogenic kidneys (11).
Discussion
Gestational alloimmune liver disease (GALD) is a maternal-fetal alloimmune disorder involving the fetal liver, often resulting in neonatal liver failure (12, 13).
The damage starts antenatally when the fetus receives the maternal IgG, between the 17th and 22nd week of gestation. This period has been suggested to be the time of liver injury onset in affected fetuses (14, 15).
GALD can present anytime from 18 weeks gestation to 3 months post-delivery (16).
The incidence of GALD seems to be very rare on the basis of the cases reported in literature.
So far, There is no gold diagnostic tool for GALD. Despite the most common presentation benign neonatal hemochromatosis, extrahepatic siderosis is not found in up to 40% of cases of gestational alloimmune hepatopathy, as it was revealed in our case, demonstrating the spectrum of disease phenotypes (1).
Immunohistochemical staining for C5b-9 complex, the neoantigen created by the action of complement, is pathognomonic for gestational alloimmune liver disease (17).
However, if the condition is not suspected, immunostaining may not be performed and the diagnosis can be easily missed.
Detecting liver failure due to GALD can be challenging, especially because no serologic tests are available. Consequently, the diagnosis of GALD is made postnatally. Suspicion for this complication in pregnancy should be raised if there's a maternal history of multiple fetal deaths or a previous sibling with neonatal liver failure (7).
Prenatal diagnosis of gestational alloimmune liver disease is difficult because ultrasound findings may be absent or nonspecific. There is usually a history of intrauterine growth restriction, oligohydramnios and prematurity (18).
As highlighted by the current case, in fetuses presenting with hydrops, once the most common etiologies have been excluded, hepatobiliary complications and liver failure caused by GALD should be taken into account (19–23).
Conclusions
Global knowledge of this disorder and its wide spectrum of presentations may help to increase the number of cases that are diagnosed in a timely manner.
In addition, once a woman has delivered an infant with GALD, the probability that the next pregnancy will be affected is greater than 90%. Recurrence can however be prevented by treatment with IVIG during pregnancy (1, 24).
It is very important for obstetricians and pediatricians to be familiar with gestational alloimmune liver disease.
Statements
Data availability statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
Ethics statement
Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author contributions
SZ and AN: contributed to the design and implementation of the research and to the analysis of the results. SZ and AN: carried out data collection, processed data and wrote the manuscript. LD, EV and NP: were involved in planning and supervised the work. All authors contributed to the article and approved the submitted version.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1.
AnastasioHBGrundyMBirsnerMLBlakemoreKJ. Gestational alloimmune liver disease: a devastating condition preventable with maternal intravenous immunoglobulin. Obstet Gynecol. (2016) 128(5):1092–94. 10.1097/AOG.0000000000001569
2.
BaruteauJHeissatSBrouéPCollardeau-FrachonSBouvierRet alTransient neonatal liver disease after maternal antenatal intravenous ig infusions in gestational alloimmune liver disease associated with neonatal haemochromatosis. J Pediatr Gastroenterol Nutr. (2014) 59(5):629–35. 10.1097/MPG.0000000000000514
3.
CheeYYWongSCMWongMSR. Heterogeneous presentation of neonatal hemochromatosis in dichorionic twins. AJP Rep. (2018) 8(4):e332–4. 10.1055/s-0038-1675335
4.
HutchingsGWilliamsOSokalEWittingtonPF. Plasmapheresis as an alternative to high-dose intravenous immunoglobulin in the prevention of gestational alloimmune liver disease. Fetal Diagn Ther. (2013) 34(3):180–3. 10.1159/000351283
5.
MoorheadRDeanJBrenneckeS. Successful pregnancy outcomes following intravenous immunoglobulin treatment in a woman with a previous fetal death in utero due to gestational alloimmune liver disease: a case report. Case Rep Womens Health. (2022) 35:e00419. 10.1016/j.crwh.2022.e00419
6.
YehPJHuangSFChiangMCWangCJLaiMW. Efficacy of intravenous immunoglobulin/exchange transfusion therapy on gestational alloimmune liver disease. Front Pediatr. (2021) 9:680730. 10.3389/fped.2021.680730
7.
McAdamsRM. Ileal atresia and multiple jejunal perforations in a premature neonate with gestational alloimmune liver disease. J Pediatr Surg Case Rep. (2017) 19:48–51. 10.1016/j.epsc.2017.02.013
8.
Iskandar A.PJamarinVMulyanaK. Neonatal hemochromatosis attributed to gestational alloimmune liver disease treated with intravenous immunoglobulin and exchange transfusion therapy: an evidence-based case report. PI. (2021) 61(6):350. 10.14238/pi61.6.2021.350-5
9.
TsunodaTInuiAKawamotoMSogoTKomatsuHKasaharaMet alNeonatal liver failure owing to gestational alloimmune liver disease without iron overload. Hepatol Res. (2015) 45(5):601–5. 10.1111/hepr.12381
10.
Concejo IglesiasPLiébana de RojasCBartolomé MateosARisco MontemayorBRasero PonferradaMGalindoA. Usefulness of dual gradient-echo MR imaging for the prenatal diagnosis of gestational alloimmune disease. J Obstet Gynaecol. (2021) 41(6):981–3. 10.1080/01443615.2020.1798907
11.
ClarkeNEGilbyDSavoiaHOliverMRRogersonS. Fulminant liver failure in a neonate. J Paediatr Child Health. (2016) 52(3):338–41. 10.1111/jpc.13090
12.
TaylorSAKellySAlonsoEMWhitingtonPF. The effects of gestational alloimmune liver disease on fetal and infant morbidity and mortality. J Pediatr. (2018) 196:123–8.e1. 10.1016/j.jpeds.2017.12.054
13.
TaylorSAWhitingtonPF. Neonatal acute liver failure. Liver Transpl. (2016) 22:677–85. 10.1002/lt.24433
14.
PanXKellySMelin-AldanaHMalladiPWhitingtonPF. Novel mechanism of fetal hepatocyte injury in congenital alloimmune hepatitis involves the terminal complement cascade. Hepatology. (2010) 51(6):2061–68. 10.1002/hep.23581
15.
HeissatSCollardeau-FrachonSBaruteauJDubrucEBouvierRFabreM. Neonatal hemochromatosis: diagnostic work-up based on a series of 56 cases of fetal death and neonatal liver failure. J Pediatr. (2015) 166:66–73. 10.1016/j.jpeds.2014.09.030
16.
FeldmanAGWhitingtonPF. Neonatal hemochromatosis. J Clin Exp Hepatol. (2013) 3(4):313–20. 10.1016/j.jceh.2013.10.004
17.
DubrucENadaudBRuchelliEHeissatSBaruteauJBrouéPet alRelevance of C5b9 immunostaining in the diagnosis of neonatal hemochromatosis. Pediatr Res. (2017) 81(5):712–21. 10.1038/pr.2017.8
18.
SciardCCollardeau-FrachonSAtallahACombourieuDMassardierJHeissatSet alPrenatal imaging features suggestive of liver gestational allo immune disease. J Gynecol Obstet Hum Reprod. (2019) 48(1):61–4. 10.1016/j.jogoh.2018.11.005
19.
SarkerMDeBoltCStrongN. Intravenous immunoglobulin induced pancytopenia while preventing development of gestational alloimmune liver disease: a case report. Case Rep Womens Health. (2022) 35:e00422. 10.1016/j.crwh.2022.e00422
20.
Roos Mariano da RochaCRostirola GuedesRKielingCORossato AdamiMCerskiCTGonçalves VieiraSM. Neonatal liver failure and congenital cirrhosis due to gestational alloimmune liver disease: a case report and literature review. Case Rep Pediatr. (2017) 2017:7432859. 10.1155/2017/7432859
21.
Casas-AlbaDClotetJInarejosEJJouCFonsCMoleraC. Broadening the spectrum of neonatal hemochromatosis. J Matern Fetal Neonatal Med. (2020) 33(6):1024–6. 10.1080/14767058.2018.1506442
22.
MidorikawaHMizuochiTOkadaJIHisanoT. Disparate clinical findings in monochorionic twins with neonatal hemochromatosis. Pediatr Int. (2017) 59(11):1215–6. 10.1111/ped.13421
23.
Flores-TorresJCarverJDSanchez-ValleA. Piga mutations can mimic neonatal hemochromatosis. Pediatrics. (2021) 147(3):e20200918. 10.1542/peds.2020-0918
24.
WhitingtonPKellySTaylorSNóbregaSSchreiberRSokalEet alAntenatal treatment with intravenous immunoglobulin to prevent gestational alloimmune liver disease: comparative effectiveness of 14-week versus 18-week initiation. Fetal Diagn Ther. (2018) 43(3):218–25. 10.1159/000477616
Summary
Keywords
GALD, hydrop, immunoglobuline therapy, ultrasound, pregnancy
Citation
Zermano S, Novak A, Vogrig E, Parisi N and Driul L (2023) GALD: new diagnostic tip for early diagnosis - a case report and literature review. Front. Reprod. Health 5:1077304. doi: 10.3389/frph.2023.1077304
Received
22 October 2022
Accepted
24 April 2023
Published
11 May 2023
Volume
5 - 2023
Edited by
Aleksandar K. Stanic, University of Wisconsin-Madison, United States
Reviewed by
Michal Silber, University of Pennsylvania, United States Anastasia Vatopoulou, University of Ioannina, Greece
Updates
Copyright
© 2023 Zermano, Novak, Vogrig, Parisi and Driul.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Lorenza Driul lorenza.driul@uniud.it
†These authors have contributed equally to this work
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.