- 1Division of Congenital Heart Disease, Heart Institute (InCor), São Paulo, Brazil
- 2Division of Pediatric Cardiology, Department of Pediatrics, Leonard M. Miller School of Medicine, University of Miami, Miami, FL, United States
Mechanical circulatory support is an established therapy to support failing hearts as a bridge to transplantation. Although tolerated overall, arrhythmias may occur after ventricular assist device implantation and can complicate patient management. We report on an infant with dilated cardiomyopathy who developed ventricular tachycardia followed by recalcitrant ventricular fibrillation, refractory to comprehensive medical therapy post Berlin Heart EXCOR® (BHE) implant.
Introduction
Heart transplantation remains the definitive treatment for end-stage heart failure (HF) refractory to medical therapy in children, although mortality among pediatric patients on the transplant list is still high given the shortage of donors. With enhanced HF management, the use of ventricular assist devices (VAD) in children has been increasing considerably, with improved device utilization, greater span of indications, and better collaborative outcomes (1). Broad indications of VAD therapy include bridge to transplant, to recovery, for borderline transplant candidates, and as a destination therapy, where VAD allows patients with no transplant perspective to live longer and with an improved quality of life (2). The Berlin Heart EXCOR® (BHE) VAD, developed in 1992, is a paracorporeal cardiac system designed for long-term left, right, or biventricular support, providing assistance for a wide range of patient sizes. It was the first VAD that received approval from the FDA (Federal Drug Administration) for the pediatric population, and its utilization has increased, as there is a lack of miniaturized alternatives for the smallest patients with diseased myocardium or unrepairable anatomy.
The risk of adverse effects during VAD support is substantial, and if they do occur, they carry high morbidity and mortality to date. We sought to describe an infant with recalcitrant ventricular arrhythmia supported with BHE.
Case description
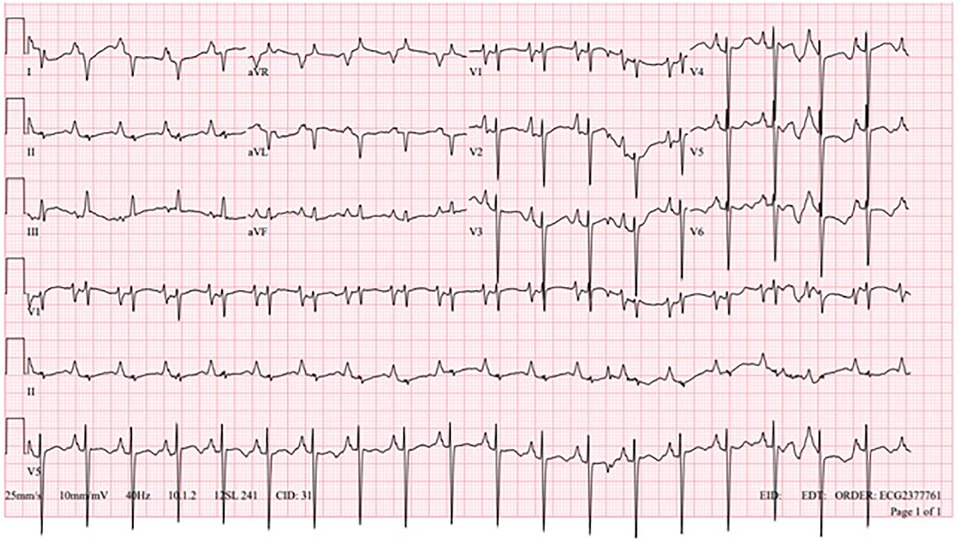
A one-year-old, 6.7 kg, Caucasian male with an atrial septal defect (ASD) and dilated cardiomyopathy (DCM) with stage D advanced HF, with a negative family history of cardiovascular disease, was hospitalized due to acute decompensation. His baseline echocardiogram was remarkable with a 15 mm ASD, moderate right ventricular dilation with preserved ejection fraction (EF), and significant left ventricular dilation with reduced EF = 31% (by Teichholz formula). Cardiac MRI was negative for fibrosis. Basal electrocardiogram (EKG) showed sinus rhythm, biatrial enlargement, right ventricular hypertrophy, and altered baseline repolarization, especially in the left lateral wall (Figure 1).
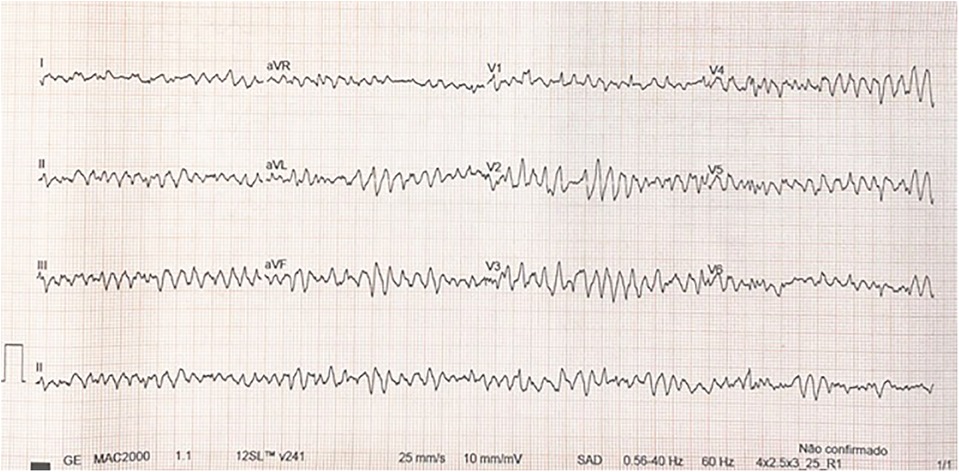
During hospitalization, the patient did not tolerate weaning attempts of inotropic support, requiring extracorporeal membrane oxygenation (ECMO) cannulation due to worsening hemodynamics and overall clinical status. He was decannulated from ECMO after six days. Echocardiogram indicated significant dilation and significant systolic dysfunction of the left ventricle, and significant dilation and mild systolic dysfunction of the right ventricle. Twelve days post decannulation, the patient developed sustained monomorphic VT, with no response to synchronized cardioversion, amiodarone, or beta-blocker therapy. There was VT resolution after lidocaine was associated, but due to escalating inotropic support, ECMO was reinstated. The patient was using milrinone before starting ECMO, and on an optimized dose of beta-blocker; after cannulation, he was maintained with milrinone associated with nitroprusside. Four days after the second ECMO cannulation, he was placed on a BHE biventricular support, 15 ml pumps each. During the BHE implant, there was rupture of a right ventricular apical diverticulum, with extensive sutures necessary for hemostasis. Six days later, the patient developed sustained monomorphic VT (Figure 2), without hemodynamic compromise. EKG demonstrates monomorphic wide QRS complexes with positive concordance in precordial leads and superior axis, and atrioventricular dissociation, suggestive of monomorphic VT.
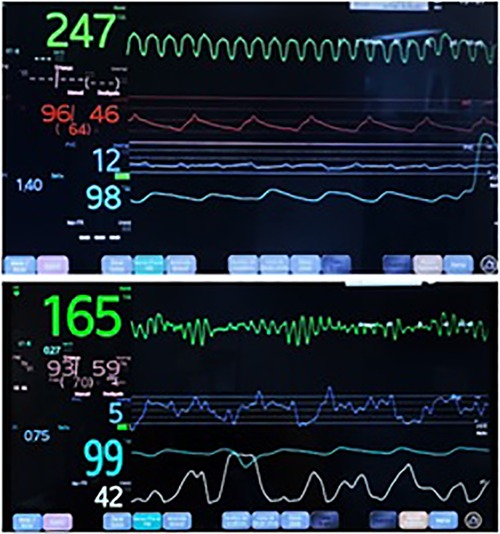
Several antiarrhythmic drugs were attempted (amiodarone, lidocaine, and beta-blockers), as well as synchronized cardioversion, with no response. Although the VT degenerated into ventricular fibrillation (VF) within hours (Figure 3), the patient maintained satisfactory arterial blood pressure and saturation given adequate VAD support (Figure 4).
The patient remained in VF while maintaining reassuring end-organ function and preserved neurological status with full biventricular support with the BHE, and all antiarrhythmic medication was discontinued. The patient underwent heart transplantation 26 days after the start of BHE, remaining on VT/VF for 21 days.
Written informed consent was obtained from the minor’s legal guardian for the publication of any potentially identifiable images or data included in this article.
Discussion
Heart transplantation is the treatment of choice for children with end-stage HF and mechanical circulatory support can be used as a bridge for heart transplantation. The ISHLT registry shows that almost 30% of patients in the pediatric population are on BHE while waiting for heart transplantation (3).
The use of BHE can generate adverse effects such as ventricular arrhythmias after implantation, especially in the early postoperative period (4), as demonstrated with our patient. The patient had no prior documentation of ventricular arrhythmia and his pre-implant MRI was negative for fibrosis. The VT quickly degenerated into ventricular fibrillation and was refractory to medical management.
In a study reported by Almond et al, of 204 children using pediatric VAD, 75% survived to 12 months post-VAD, with 64% transplanted, 6% recovered, and 5% still alive on the device at the time of the publication (5). In a recent systematic review by Rohde et al., the authors described that, among patients supported by BHE, the overall mortality rate was 26.1%, while survival to transplant was 62.9%, and 8.3% were weaned off VAD (6).
Ventricular arrhythmias represent a common event in the adult population after implantation of a long-term VAD, especially in the early postoperative period, and being equally prevalent in left ventricular and biventricular support (7). Ventricular arrhythmias are better tolerated in this population over a period of hours to days (4), especially if the right ventricle is also supported mechanically, or if it has preserved function (8), however, there is an increased risk of intracardiac thrombus formation and cardiac output compromise. Synchronized electrical cardioversion may be occasionally necessary (9), but is not always successful.
The majority of arrhythmia triggering mechanisms (75%) are associated with pre-existing substrates such as myocardial fibrosis and ischemia. However, other factors may contribute to the development of ventricular arrhythmias in this patient population. Examples include apical insertion of the inflow cannula that may cause ventricular scarring, suction mechanism related to the device in situations of hypovolemia or high ventricular assistance, QTc interval prolongation in the early postoperative period due to ventricular unloading, and use of inotropic drugs during stabilization phases (7, 8).
Kyle WB et al. described a 71% incidence of arrhythmias in patients with VADs, with ventricular tachycardia (VT) being the most common, affecting 78% of the patients, with the majority presenting non-sustained VT (10). Only one of the 32 patients had documented sustained VT. Patients supported with BHE were excluded from this study. In the BHE post-market approval surveillance study from the Advanced Cardiac Therapies Improving Outcomes Network (ACTION) registry, the incidence of cardiac arrhythmia was extremely low, with only 1 event in 72 patients (0.5 per 100 patient-months) (11), lower than that reported in the Berlin Heart Study Group (21 events in 320 patients, at 4.91 events per 100 patient-months) (5, 12, 13). No information was available regarding the type of arrhythmia.
In another study, out of 320 children implanted with BHE VAD from 2007 to 2016 from the Investigational Device Exemption/Post-Approval study database by Wilkens SJ et al., treatable ventricular arrhythmias were identified in 26 patients (8%), of which 53% were sustained ventricular arrhythmia (14), occurring at a median of 9 days after implant. Outcomes of arrhythmia treatment were not reported, but 53% required defibrillation or cardioversion and 41% required drug treatment or cardioversion. Independent factors for ventricular arrhythmias identified in this cohort were ECMO pre-implant and older age.
Although uncommon, the fortuity of biventricular assist support allowed the patient to remain stable while waiting for transplantation. Weighing the risk/benefit ratio (and toxicity) of antiarrhythmic medications, the patient was kept with no antiarrhythmic therapy, awake and extubated, while on VF. Further studies are required to explore the benefit of additional interventions and risks of controlling arrhythmias in children with biVAD waiting for heart transplant.
Conclusions
Ventricular tachycardia and fibrillation can occur after mechanical circulatory device implantation and be refractory to medical therapy, but biventricular ventricular assist devices can provide support until transplantation.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Ethics statement
Ethical approval was not required for the study involving human samples in accordance with the local legislation and institutional requirements. Written informed consent for participation in this study was provided by the participants’ legal guardians/next of kin. Written informed consent was obtained from the minor(s)' legal guardian/next of kin for the publication of any potentially identifiable images or data included in this article.
Author contributions
DS: Writing – original draft, Writing – review & editing. SL: Writing – original draft, Writing – review & editing. TT: Writing – original draft, Writing – review & editing. IZ: Writing – original draft, Writing – review & editing. AS: Writing – original draft, Writing – review & editing. DM: Writing – original draft, Writing – review & editing. EA: Writing – original draft, Writing – review & editing. SM: Writing – original draft, Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Shugh SB, Riggs KW, Morales DLS. Mechanical circulatory support in children: past, present and future. Transl Pediatr. (2019) 8(4):269–77. doi: 10.21037/tp.2019.07.14
2. Tunuguntla H, Conway J, Villa C, Rapoport A, Jeewa A. Destination-therapy ventricular assist device in children: “the future is now”. Can J Cardiol. (2020) 36(2):216–22. doi: 10.1016/j.cjca.2019.10.033
3. Edelson JB, Huang Y, Griffis H, Huang J, Mascio CE, Chen JM, et al. The influence of mechanical circulatory support on post-transplant outcomes in pediatric patients: a multicenter study from the international society for heart and lung transplantation (ISHLT) registry. J Heart Lung Transplant. (2021) 40(11):1443–53. doi: 10.1016/j.healun.2021.06.003
4. Shirazi JT, Lopshire JC, Gradus-Pizlo I, Hadi MA, Wozniak TC, Malik AS. Ventricular arrhythmias in patients with implanted ventricular assist devices: a contemporary review. Europace. (2013) 15(1):11–7. doi: 10.1093/europace/eus364
5. Almond CS, Morales DL, Blackstone EH, Turrentine MW, Imamura M, Massicotte MP, et al. Berlin heart EXCOR pediatric ventricular assist device for bridge to heart transplantation in US children. Circulation. (2013) 127(16):1702–11. doi: 10.1161/CIRCULATIONAHA.112.000685
6. Rohde S, Antonides CFJ, Dalinghaus M, Muslem R, Bogers AJJC. Clinical outcomes of paediatric patients supported by the Berlin heart EXCOR: a systematic review. Eur J Cardiothorac Surg. (2019) 56(5):830–9. doi: 10.1093/ejcts/ezz092
7. Nakahara S, Chien C, Gelow J, Dalouk K, Henrikson CA, Mudd J, et al. Ventricular arrhythmias after left ventricular assist device. Circ Arrhythm Electrophysiol. (2013) 6(3):648–54. doi: 10.1161/CIRCEP.113.000113
8. Busch MC, Haap M, Kristen A, Haas CS. Asymptomatic sustained ventricular fibrillation in a patient with left ventricular assist device. Ann Emerg Med. (2011) 57(1):25–8. doi: 10.1016/j.annemergmed.2010.05.023
9. Arkles JS, Marchlinski F. When should the electrophysiologist be involved in managing patients with ventricular assist devices and ventricular arrhythmias? J Innov Card Rhythm Manag. (2019) 10(4):3605–10. doi: 10.19102/icrm.2019.100407
10. Kyle WB, Decker J, Macicek SL, Valdes SO, Morales D, Hong B, et al. Arrhythmias in children with ventricular assist devices. Cardiol Young. (2015) 25(2):255–60. doi: 10.1017/S1047951113002126
11. Zafar F, Conway J, Bleiweis MS, Al-Aklabi M, Ameduri R, Barnes A, et al. ACTION learning network investigators. Berlin heart EXCOR and ACTION post-approval surveillance study report. J Heart Lung Transplant. (2021) 40(4):251–9. doi: 10.1016/j.healun.2021.01.010
12. Jaquiss RD, Humpl T, Canter CE, Morales DL, Rosenthal DN, Fraser CD Jr. Postapproval outcomes: the Berlin heart EXCOR pediatric in North America. ASAIO J. (2017) 63(2):193–7. doi: 10.1097/MAT.0000000000000454
13. Fraser CD Jr, Jaquiss RD, Rosenthal DN, Humpl T, Canter CE, Blackstone EH, et al. Berlin heart study investigators. Prospective trial of a pediatric ventricular assist device. N Engl J Med. (2012) 367(6):532–41. doi: 10.1056/NEJMoa1014164
Keywords: pediatrics, cardiology, arrhythmias, tachycardia, heart failure, Berlin Heart EXCOR®
Citation: Silva DLM, Lemouche SO, Takahashi TY, Zanon IdC, Siqueira A, Machado D, Azeka E and de Melo SL (2024) Case Report: Sustained ventricular arrhythmia in a child supported by a Berlin heart EXCOR ventricular assist device. Front. Transplant. 3:1302060. doi: 10.3389/frtra.2024.1302060
Received: 26 September 2023; Accepted: 7 February 2024;
Published: 5 March 2024.
Edited by:
Sebastian Michel, LMU Munich University Hospital, GermanyReviewed by:
Erik Strauss, University of Maryland, United StatesTakashi Kido, Osaka University, Japan
© 2024 Silva, Lemouche, Takahashi, Zanon, Siqueira, Machado, Azeka and de Melo. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Stephanie Ondracek Lemouche c3RlcGhhbmllLmxlbW91Y2hlQGhjLmZtLnVzcC5icg==
 Diego Lineker Marquetto Silva
Diego Lineker Marquetto Silva Stephanie Ondracek Lemouche
Stephanie Ondracek Lemouche Tatiane Yukari Takahashi1
Tatiane Yukari Takahashi1 Desiree Machado
Desiree Machado Estela Azeka
Estela Azeka