- 1Department of Emergency Medicine, Medical University of Vienna, Vienna, Austria
- 2PULS – Austrian Cardiac Arrest Awareness Association, Vienna, Austria
- 3Emergency Medical Service Vienna, Vienna, Austria
- 4Department of Critical Care Medicine, VA Pittsburgh Healthcare System, and CRISMA Center, University of Pittsburgh, Pittsburgh, PA, United States
- 5Department of Emergency Medicine, Clinic Ottakring, Vienna Healthcare Group, Vienna, Austria
Background: An increase in regional cerebral oxygen saturation (rSO2) levels during advanced life support in patients with out-of-hospital cardiac arrest (OHCA) is associated with return of spontaneous circulation (ROSC) and can predict neurological outcome. Data from the post-ROSC phase are scarce but may predict clinical outcomes as well.
Methods: For this prospective observational study, we measured rSO2 via near-infrared spectroscopy (NIRS) in patients after ROSC following OHCA in both the pre- and in-hospital setting for up to 72 h. Patients were followed up for their post-ROSC treatment and outcomes. NIRS values were then compared between patients with favorable and non-favorable neurological outcomes, and cutoff values were assessed via receiver operating characteristic (ROC) and Classification and Regression Trees (CART) analyses. In addition, a narrative review on the topic was performed.
Results: Of the 27 included patients, 37% survived hospital discharge, and 26% showed favorable neurological outcomes (CPC 1 or 2). RSO2 was significantly higher in individuals with CPC 1/2 (67 [60–69] % vs. 59 [50–70] %; p = 0.049). This was even more pronounced for initial (= a mean of the first 5 min) NIRS values (70 [65–77] % vs. 57 [49–68] %; p = 0.025) and NIRS values rising in the first 10 min (43% vs. 5% of patients; p = 0.042). A ROC analysis for initial rSO2 showed a significant discriminatory power to predict CPC 1/2 (AUC = 0.789, p = 0.025), and both ROC and CART analyses suggested an optimal cutoff of approximately 62% rSO2.
Conclusion: We identified a potential RSO2 cutoff measured via NIRS in the post-ROSC phase after OHCA to predict favorable neurological outcomes. Initial values and rising trends may be more useful for prognostication than prolonged ICU measurements. These findings are consistent with previous literature and should prompt both larger clinical trials and consideration of this technology by resuscitation societies.
1 Introduction
Near-infrared spectroscopy (NIRS) is used to assess and monitor regional cerebral oxygenation (rSO2) in various settings, including anesthesia, perioperative medicine, and intensive care (1, 2). Near-infrared light emitted from optodes placed on the forehead penetrates underlying tissues and is then reflected back, with varying degrees of absorption depending on the oxygenated status of the molecules. A percentage of tissue oxygen saturation is then calculated. Thus, a lower rSO2 value can serve as a surrogate for reduced frontal cerebral oxygenation and cerebral perfusion, which is otherwise not typically monitored in critical care (1–3). Optimizing cerebral (hypo-)perfusion is a cornerstone of cardiopulmonary resuscitation (CPR) for in- or out-of-hospital cardiac arrest (IHCA / OHCA) treated by basic and advanced life support (BLS / ALS), as prolonged cerebral ischemia leads to dismal neurological outcomes, even if return of spontaneous circulation (ROSC) may have been achieved (4). Therefore, following in the footsteps of end-tidal carbon dioxide (etCO2) (5), rSO2 has been evaluated as a non-invasive prognostication tool during ALS and post-resuscitation care. Higher rSO2 seems to be associated with favorable outcomes during and after CPR, but there is a lot of variation, no consensus on which values predict favorable or unfavorable outcomes, and no clear cutoffs (4, 6–9). Accordingly, current guidelines do not recommend the routine use of NIRS in resuscitation care, but on the other hand, they recognize its potential for future use (5, 10). Furthermore, the most up-to-date consensus on respective science by the International Liaison Committee on Resuscitation (ILCOR) states there are insufficient data for a recommendation for or against the routine use of NIRS (11). Interestingly, an AHA statement on neuroprognostication in comatose cardiac arrest survivors does not mention NIRS (12), whereas a more recent joint statement by the AHA and Neurocritical Care Society on critical care management of patients after cardiac arrest states that NIRS could play a role in post-resuscitation care, and calls for further research (13).
We therefore aimed to investigate: (1) the feasibility of using NIRS during post-resuscitation care, starting directly after ROSC, regardless of whether in pre- or in-hospital setting; and (2) its prognostic value when measured for the subsequent 72 h.
2 Methods
2.1 Study setting and population
For this prospective observational study, patients who received ROSC after OHCA in Vienna, Austria, were included between November 2016 and September 2018. Further inclusion criteria were: ≥ 18 years old, OHCA in the metropolitan area of Vienna, treated by the Emergency Medical Service (EMS) Vienna, and then treated at the Emergency Department (ED) and/or an Intensive Care Unit (ICU) of the Medical University of Vienna. Exclusion criteria were traumatic cardiac arrest, known or suspected pregnancy, and a decision of the team on site that a NIRS measurement was not possible (e.g., due to logistic or space constraints). Patient selection followed a real-world pragmatic approach: all patients seen by the so-called Field Supervisors (FISU; paramedics specially trained in crew resource management and quality management deployed to high-priority missions) of EMS Vienna (14) during the observational period were screened for eligibility to measure NIRS. RSO2 was measured using a NONIN SenSmart® X-100 (Nonin Medical Inc., Plymouth, United States) device (15). One optode was placed on the right forehead of each patient, and one on the left forehead of each patient, and data points were saved every 4 s. The device was lightweight and portable. Inclusion was possible either in the pre- or the in-hospital setting (at the OHCA scene, the ambulance, or the ED—depending on where ROSC occurred) and was continued for up to 72 h post-ROSC (at the intensive care unit—depending on the duration of survival). Gaps between ROSC and the start of NIRS were excluded from any trend analysis. The same device was used for each patient throughout their measurement period, without planned interruptions. The included patients were all treated following the EMS’s and the hospital’s standard operating procedure for post-cardiac arrest care. Any ongoing CPR during transportation was delivered via a mechanical chest compression device. To achieve a comprehensive measurement strategy and maximize compliance, the NIRS device was handled by dedicated personnel, as described previously. In the observational period, this was still a pilot project, which resulted in the presented low patient numbers (14).
Ethical approval for this study (no 1265/2015) was acquired from the Ethical Committee of the Medical University of Vienna, Austria, and informed consent was waived. The study protocol complies with the Declaration of Helsinki.
2.2 Narrative review
To provide a summary of the existing literature on NIRS in post-resuscitation care, we performed a narrative review. Medline/PubMed and Embase were searched using the terms “Near-infrared spectroscopy,” “NIRS,” “cerebral oxygen,” “cerebral oxygenation,” and “rSO2,” in combination with “cardiac arrest,” “resuscitation,” “life support,” “CPR,” “return of spontaneous circulation,” “ROSC,” “intensive care,” “critical care,” and “emergency medicine.” Relevant abstracts were screened, and respective information was extracted from the full-texts.
2.3 Statistical analyses
Data were assessed for normal distribution using a Kolmogorov–Smirnov test. Continuous data are presented as medians and the respective interquartile ranges (IQRs) and were compared among subgroups using the Mann–Whitney U-test. Categorical data are presented as counts and percentages and are compared using the χ2-square test where appropriate. Receiver operating characteristic (ROC) analyses were utilized to assess the discriminatory power of specific cutoff levels and thresholds as potential indicators for clinical decision-making. A classification and regression tree (CART) analysis was conducted to elucidate the importance of NIRS values in a predictive model and to establish a cutoff value. Statistical significance was defined by two-tailed p-values of <0.05. Data analysis was performed using SPSS 22.0 (IBM, United States).
3 Results
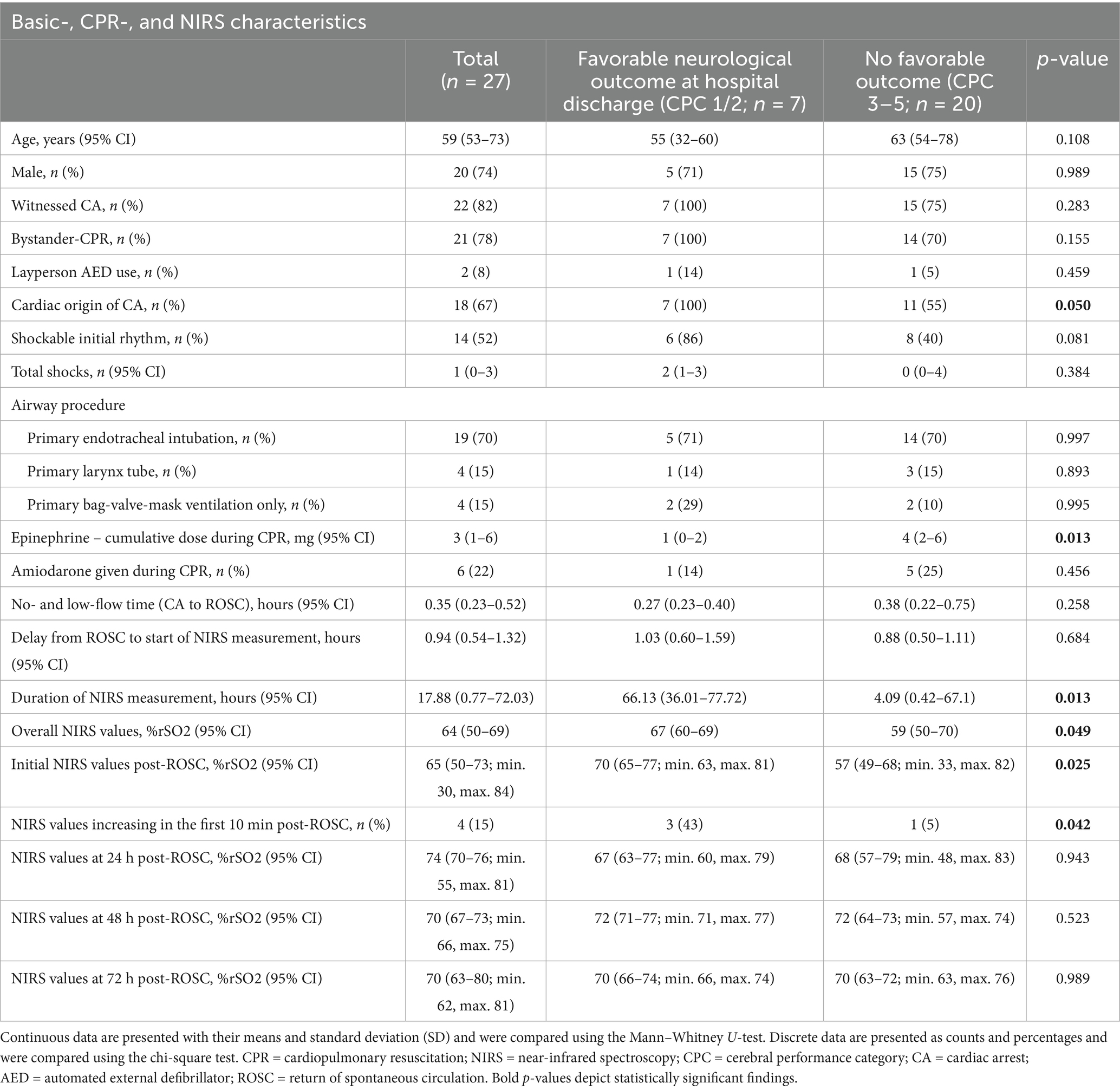
We included 27 patients with OHCA who had been treated with standard ALS, reached sustained ROSC, and then received NIRS measurements. None regained consciousness immediately after ROSC. No extracorporeal CPR (eCPR) patients were included. In 10 patients, the NIRS measurement was started in the pre-hospital setting, and in 17 patients in the in-hospital setting after ED admission (ROSC after ED admission). There were no re-arrests during the observational period. Ten patients (37% of all included) survived to hospital discharge, and seven of them (26% of all included or 70% of the survivors to hospital discharge) had favorable neurological outcomes (CPC 1 or 2; whereas six patients reached CPC 1, one patient CPC 2, two patients CPC 3, and one patient CPC 4). Modified Rankin Scale (mRS) at hospital discharge was excellent (1.5 ± 0.5) in those reaching a CPC of 1 or 2. Further patient characteristics are shown in Table 1, and Supplementary Table S1 shows the comparison between pre-hospital and in-hospital measurements.
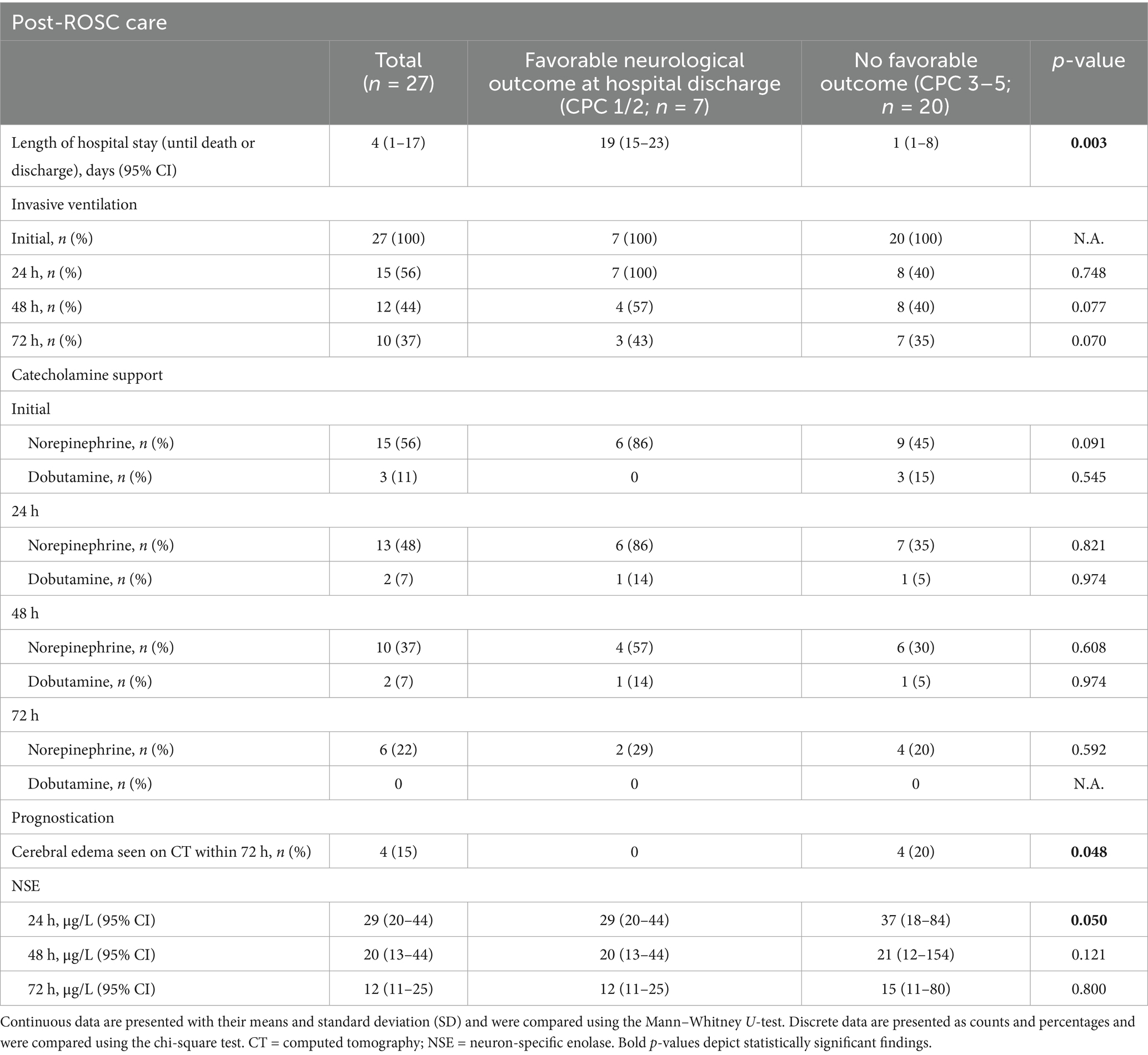
The overall length of hospital stay (until death or discharge) was 4 (1–17) days. In the subgroup of patients who survived to hospital discharge, it was 20 (14–37) days. Standard post-resuscitation care was applied to all patients, and, if applicable, reversible causes of cardiac arrest were treated (e.g., PCI for a presumed cardiac origin was performed in 67% of cases). All patients received targeted temperature management (active cooling, 32–34°C for 24 h) and were mechanically ventilated (pressure-controlled or pressure-regulated volume controlled). See Table 2 for more information about post-ROSC care, including catecholamine support and prognostication, and Supplementary Table S2 for details on ventilation settings and blood gas analyses.
NIRS was measured up to 72 h post-ROSC (see Supplementary Figure 1 for details of the measurement duration). The median duration of NIRS was 17.88 (0.77–72.03) h (all patients). Cerebral oxygenation was significantly higher in individuals reaching a favorable neurological outcome (67 [60–69] % vs. 59 (50–70) %; p = 0.049). Between patients with a CPC of 3/4 and 5, there was no significant difference in rSO2 (Supplementary Table S3). When evaluating various points of time during the measurement period, this difference was even stronger for initial (= a mean of the first 5 min) NIRS values (70 [65–77] % for CPC 1/2 vs. 57 (49–68) % for CPC 3–5; p = 0.025) and NIRS values rising within the first 10 min (43% of patients for CPC 1/2 vs. 5% of patients for CPC 3–5; p = 0.042). No significant difference was detectable beyond 24 h. See Table 1 for an overview of the NIRS values and respective comparisons.
A ROC analysis of the rSO2 values during the whole measurement period to predict a favorable neurological outcome yielded no significant result. However, an additional ROC analysis of the initial NIRS values (Figure 1) showed their significant discriminatory power to predict a favorable neurological outcome (AUC = 0.789, p = 0.025). An rSO2 cutoff of 62.5% predicted a CPC of 1 or 2 with a sensitivity of 100% and a specificity of 65%. Accordingly, a CART analysis for the prognostic value of the initial rSO2 resulted in an optimal cutoff value of 62% to be highly predictive of a favorable neurological outcome, with an overall correct classification rate of 74.1% (Figure 2).
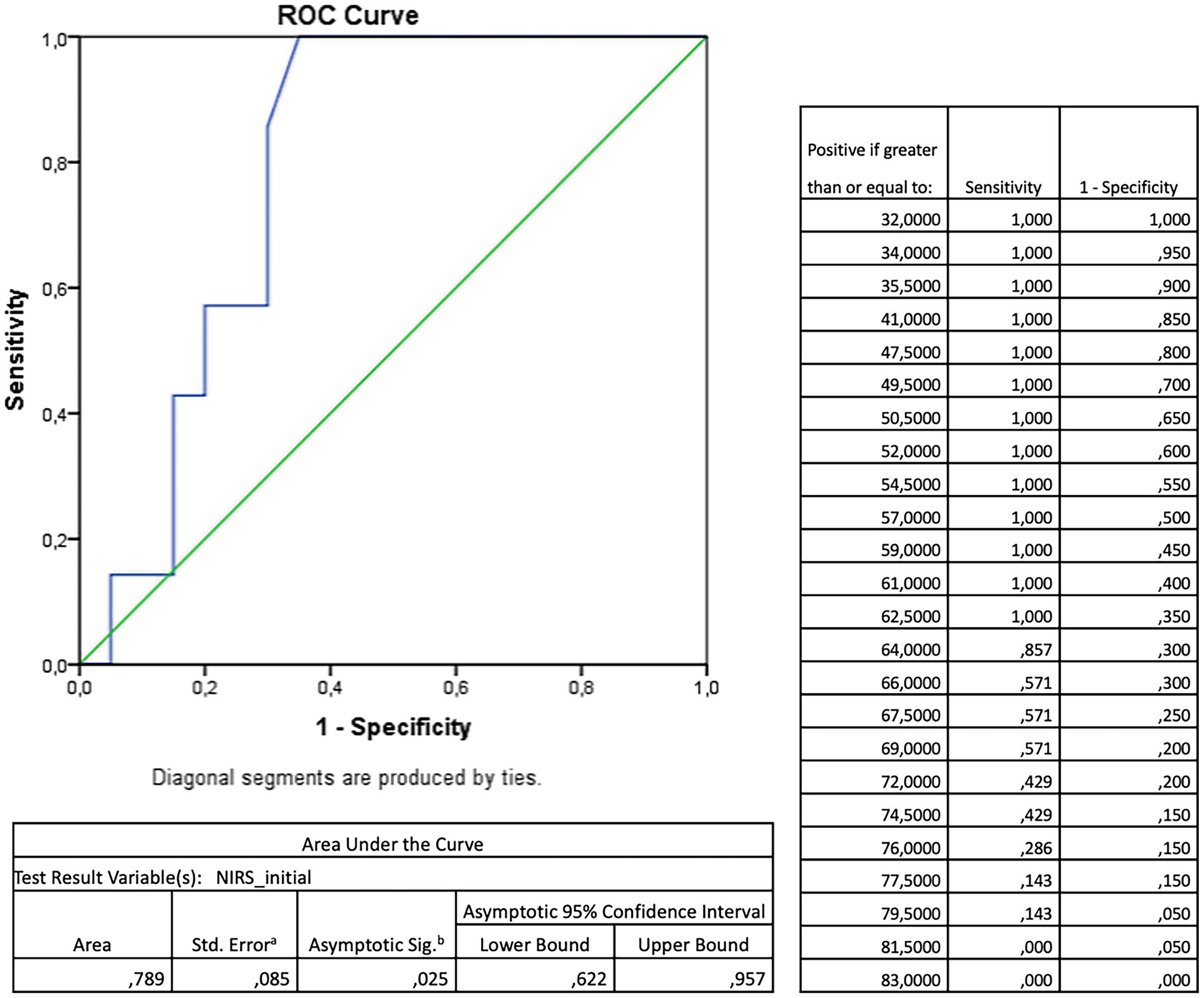
Figure 1. Receiver operating characteristic (ROC)—analysis for the prognostic value of the initial (= a mean of the first five minutes) cerebral oxygenation measured by near-infrared spectroscopy (NIRS) towards favourable neurological outcome measured by cerebral performance category (CPC) 1 or 2 after return of spontaneous circulation after out-of-hospital cardiac arrest.
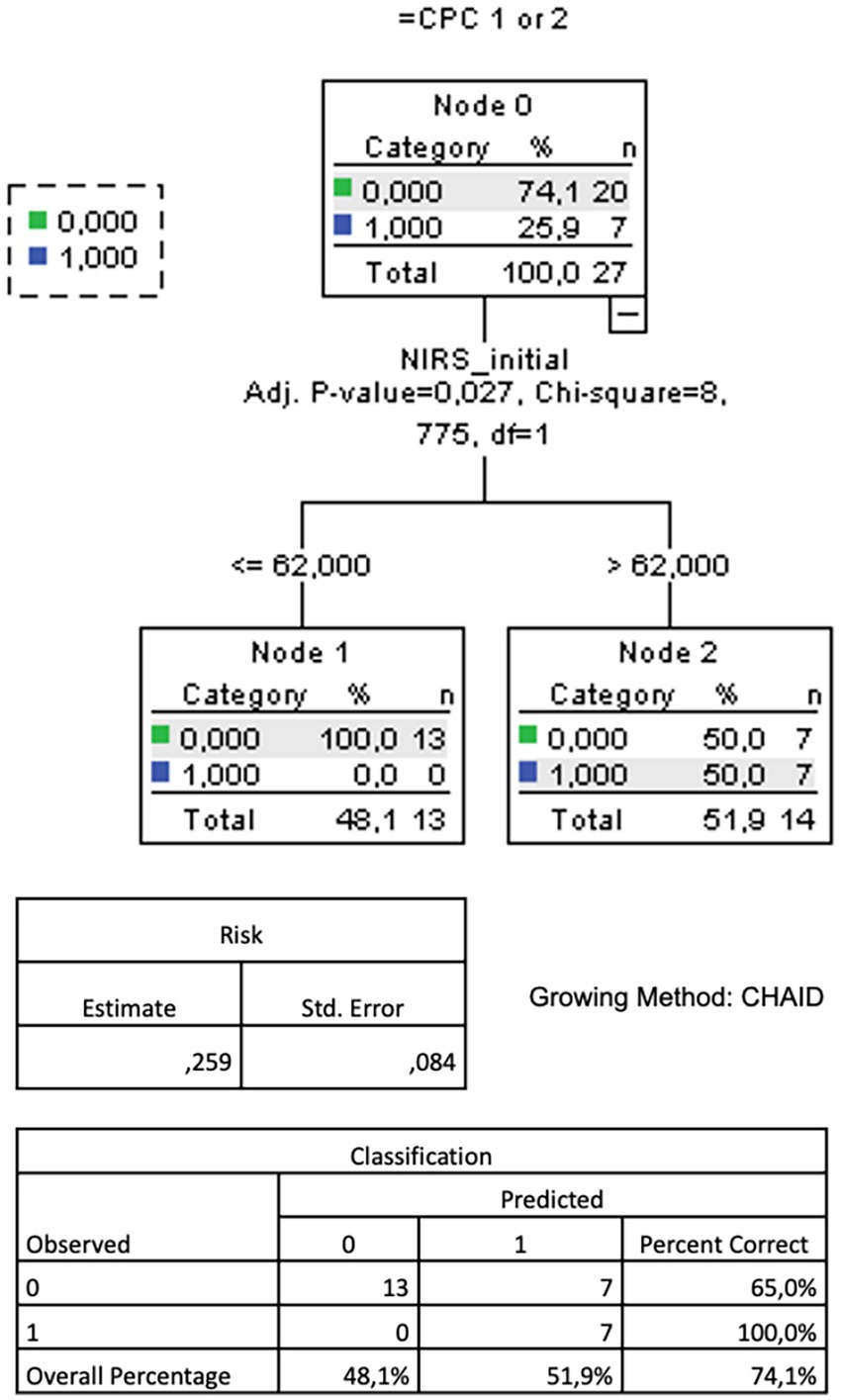
Figure 2. CART analysis (growing method CHAID) for the prognostic value of the initial (= a mean of the first five minutes) cerebral oxygenation measured by near-infrared spectroscopy (NIRS) for favourable neurological outcome measured by cerebral performance category (CPC) 1 or 2 after return of spontaneous circulation after out-of-hospital cardiac arrest. CART = classification and regression tree. CHAID = Chi-square Automated Interaction Detection.
4 Discussion and literature review
Our three key findings were that (1) an early NIRS measurement after ROSC is feasible, regardless of whether in the pre- or in-hospital setting, (2) rS02 levels after ROSC can predict favorable neurological outcomes, and (3) early measurements and the trajectory of measurements appear to be more important than prolonged measurements over time.
As a complete novelty, we measured NIRS right from the time point when ROSC occurred, no matter whether still in the pre-hospital or already in the in-hospital setting. This approach was feasible.
Neither of the respective guiding documents by ILCOR, the AHA, or the ERC currently suggests the use of NIRS during or after cardiac arrest. However, it seems that until now, insufficient attention has been paid to the growing body of literature on it, as summative publications on post-resuscitation care often either include no or only singular NIRS studies, or studies on specific patient groups. Of note, most evidence is derived from or about rSO2 measurements during CPR and not a longer observational period after ROSC. This may have led to a somewhat incomplete picture of the technology’s prognostication possibilities (11, 16–22). Recently, a statement by the AHA and Neurocritical Care Society on critical care management of patients after cardiac arrest mentions NIRS to potentially contribute to a more individualized monitoring and treatment approach in the future, and suggests further research (13). While a body of evidence on NIRS during CPR is available, a consensus on cutoffs or target values has yet to be reached (4, 8, 18, 23–31). The situation is even more scarce and heterogeneous for post-ROSC measurements, and there are no robust data on a comprehensive and continuous rSO2 assessment during and after CPR (32).
4.1 Pre-hospital post-ROSC NIRS
A pattern of increasing rSO2 during CPR has been shown to be associated with ROSC and survival in the pre-hospital setting (8, 26, 27, 29, 30). Directly after ROSC, some patients suffer from repeated hemodynamic instability and/or re-arrests. Recognizing such states quickly is a challenge, especially in the pre-hospital setting, where usually no invasive blood pressure measurement is available, and (re-)assessing patients during transport might be challenging. NIRS, however, does not need pulsatile flow and can serve as additional monitoring to timely detect patient deterioration or re-arrest (3, 6, 29, 33–37), as well as guidance for interventions (38–40). Data on pre-hospital rSO2 measurements and their association with (neuro-)prognostication are even more scarce than in-hospital ones.
4.2 NIRS in post-resuscitation intensive care
ICU treatment after ROSC is increasingly focused on achieving favorable neurological outcomes and improving the quality of life of cardiac arrest survivors, rather than merely ensuring survival to hospital discharge. Post-cardiac arrest brain injury, the leading cause of mortality after CPR, can range from no or mild impairment to permanent disability or brainstem death, rendering neuromonitoring and prognostication increasingly important. A multimodal approach is applied, aiming at optimal personalized care (12, 21, 41–44). For instance, non-invasive neuromonitoring is increasingly being used in ICUs, either for primary brain injury or patients at risk of a secondary one. Apart from the features discussed above for the pre-hospital phase, here NIRS plays a part in a multimodal approach (13, 45), is non-invasive, does not require additional skills (46), and can identify cardiopulmonary instability (40) or monitor brain oxygen delivery and metabolism (47, 48). Furthermore, it seems more practical for widespread use than invasive techniques such as intracranial pressure monitoring (49). However, evidence on the clinical effects of rSO2 monitoring is still uncertain (50). There have been reports on rSO2 measurements during targeted temperature management after cardiac arrest, suggesting an rSO2 decrease after the onset of hypothermia and values normalizing again afterward (9, 51–56). In the complicated multi-organ post-cardiac arrest syndrome, cerebral autoregulation and blood pressure dynamics play an important part, opening a chance of preventing further cerebral injury, as well as promoting recovery (20). For instance, arterial pressure goals could be monitored via NIRS or special respective indices which could then serve as a surrogate for (impaired) cerebral autoregulation (7, 13, 49, 57–59).
4.3 Ranges and cutoffs
Post-ROSC rSO2 usually lies between 50 and 70%, with noticeable upwards trends the longer the measurement is continued (of note, there are no robust data on >72 h post-ROSC) (9, 48, 51, 56). This is in accordance with our results. However, single studies also report lower values of approximately 40% (55, 60). Despite reports of patients with low rSO2 values during CPR still reaching favorable outcomes (61), both available literature on post-ROSC NIRS and our study strongly suggest higher mean or median rSO2 values over a certain period of time (mostly up to 24, 48, or 72 h) to be associated with a favorable neurological outcome, albeit not always reaching statistical significance. Reported rSO2 values in the respective outcome subgroups are quite heterogeneous and range approximately 60–70% for CPC1/2 and 50–66% for CPC 3–5. However, there are overlaps that can make exact classifications into rSO2-groups difficult (6, 51–54, 56, 62–64). There have been reports of rSO2 differences between neurological outcome groups, “wearying off” or diminishing after a certain amount of time (48, 65)—an effect that we also observed in our cohort. Explanatory hypotheses for such dynamics include primary cerebral metabolic suppression after ROSC leading to a low cerebral oxygen extraction fraction, an oxygen supply/demand mismatch, cerebral ischemia due to instable hemodynamics, hypocapnia, small vessel caliber reduction, or impaired microvascular perfusion for lower-, and (reactive) hyperemia or hyperperfusion with increased cerebral blood flow for higher rSO2 values (45). Although prior reports have described a decrease in both cerebral metabolic rate of oxygen (CMRO₂) and oxygen extraction fraction (OEF) 24–72 h after cardiac arrest, suggesting a preserved global flow–metabolism coupling (42), our observation of described dynamics in rSO₂ may reflect regional and functional heterogeneities that are not captured by global metrics alone: First, cerebral blood flow (CBF) following cardiac arrest is often regionally heterogeneous, with imaging studies revealing coexisting zones of hyperemia, hypoperfusion, and no-flow, particularly in metabolically vulnerable regions such as watershed zones or the hippocampus (66). Second, impaired cerebrovascular autoregulation in the subacute phase may lead to inadequate regional perfusion in response to systemic changes (e.g., hypotension or vasopressor withdrawal), even when overall CMRO₂ is reduced (67). Third, NIRS-derived rSO₂ is susceptible to influences beyond pure flow–metabolism dynamics, including microvascular dysfunction, capillary flow heterogeneity, hemoglobin changes, or mitochondrial impairment limiting oxygen utilization despite adequate perfusion (67, 68). We could observe that rising rSO2 values in the first 10 min after ROSC led to a higher percentage of patients performing neurologically well—a more beneficial response to reperfusion and a faster cerebral recovery from hypoxia could be the reason, rendering NIRS a potential marker for individual cerebral reconstitution after ROSC.
The time points or durations of the measurements vary in the literature. While it seems to have become standard to not only measure at single points in time anymore (e.g., at ROSC and at hospital admission) and to rather use means or medians up until a certain time period after ROSC, the optimal approach is still unknown. It remains debatable whether either overall means/medians or dynamics (e.g., from a baseline or nadirs) are more beneficial (9, 32, 69). Rarely, only mortality, but not neurological function, has been assessed, showing a trend toward lower rSO2 values in non-survivors (48, 51, 70).
Naturally, a clear rSO2 cutoff to discriminate favorable vs. unfavorable outcome groups (or also other more secondary outcomes) would be a milestone in modern post-resuscitation care for both the pre-hospital (in addition to etCO2) and the in-hospital setting (in addition to invasive blood pressure monitoring). Accordingly, several receiver operating curve (ROC) analyses have in the past been conducted, with mixed results ranging from thresholds of 40% (area under the curve [AUC] 0.9, 81% sensitivity, 96% specificity) (64), 49% (AUC 0.8, 100% specificity, 45% sensitivity) (62), and 50% (AUC 0.8, 70% specificity, 86% sensitivity) (52), up to 55% (AUC 0.6, 52% specificity, 62% sensitivity) (9). When combined with NSE values, one study reported an optimal cutoff of 83% (AUC 0.9, 100% specificity, 76% sensitivity) (63). Other previous data showed no association of rSO2 and NSE (7). Since NSE should be assessed dynamically, correlating single NSE values with rSO2 levels has limited value.
Interestingly, we could not find a good overall rSO2 cutoff for a favorable neurological outcome in our cohort (whereas it was probably underpowered for this), but rather a threshold of 63% for the mean rSO2 over the first 5 min after ROSC (AUC = 0.8, 65% specificity, 100% sensitivity). With a high correct classification rate, this cutoff could be confirmed by CART analysis. This fits with the above-discussed finding of the beneficial effect of rSO2 values rising in the first 10 min after ROSC and underscores the hypothesis that values over such a threshold could mean a better post-ROSC individual adaptation. A comprehensive NIRS measurement in the pre- and in-hospital setting—wherever ROSC occurs—is a condition to “catch” these probably short-lived dynamics.
4.4 Knowledge gaps and outlook
Even though the described reports, including ours at hand, on NIRS in the post-ROSC phase have provided more insights into the topic, certain knowledge gaps persist:
• Is a unilateral NIRS measurement sufficient? Previous reports suggest so if there is no sign of unilateral pathology (71), and one instead of two optodes per patient would be more cost-efficient.
• What is the optimal mode of measurement/monitoring (means/medians, deltas, trends, nadirs, etc.)? Are there different optimal modes for different outcomes (e.g., initial values and dynamics after ROSC for neurological prognostication, such as in our study)?
• The relation between rSO2 and etCO2 should be further researched for its combined and supplemental prognostication potential, not only during but also after ROSC (72, 73).
• Do certain interventions (e.g., medication or ventilation details), as well as certain hemodynamic conditions (e.g., hemodynamically relevant dysrhythmia) in critical and intensive care, have an impact on rSO2? (23, 24)
• Is higher rSO2 necessarily directly associated with improved or preserved neurological function (also on a molecular level), or is it rather a surrogate for interventions, care, or inter-patient characteristics? (7)
• What is an rSO2 cutoff that is feasible for clinical use while providing sufficient sensitivity and specificity for a favorable neurological outcome?
• Is predicting an unfavorable neurological outcome clinically more relevant in post-ROSC ICU patients than a favorable outcome?
• Does a NIRS measurement in clinical use directly or indirectly impact more imminent clinical outcomes than neurological performance, such as, for instance, the duration and invasiveness of ventilation, episodes of hemodynamic deterioration, etc.?
In the future, not only should these knowledge gaps be addressed in large trials providing sensible endpoints with adequate power, but respective opinion-leading associations and societies should also acknowledge the growing body of literature on NIRS in resuscitation medicine as well. As one piece of the prognostication mosaic, NIRS does indeed show undeniable potential for the pre- and in-hospital setting. However, especially for its use by EMS crews, technical issues must be addressed first—ideally, NIRS would be integrated into already-existing ALS monitor-defibrillator devices.
Another interesting novel possibility to integrate such a multimodal approach into clinical practice is using artificial intelligence (74), which would probably profit from additional monitoring data without adding to the clinicians’ actual plate of work. It is imaginable that a linear approach (9) could then give real-time rSO2 trends combined with other prognostication factors.
5 Limitations
This pilot study is purely hypothesis-generating. One limitation is that we only report median values for the right and left optodes, which is, however, a common practice in previous literature. Another limitation is that the assessed data are not very new at the time of publication (this was due to organizational and resource reasons). Furthermore, due to resource limitations, we only assessed neurological outcome at hospital discharge and not at further timepoints afterward. We only used a convenience sample size, thus probably rendering our results underpowered and missing various effects (especially the patient group surviving >24 h was small). However, we did manage to obtain statistically significant results in some domains despite the low patient numbers in the subgroups, underscoring the reported effect. Moreover, patient selection was not controlled but followed a pragmatic real-life approach of FISUs, starting NIRS measurement whenever they were on scene and the patients were eligible. Furthermore, our subgroup with poor outcomes also showed a generally unfavorable prognostic profile (see Table 1), and even though these differences did not reach statistical significance, this was probably due to the low subgroup patient numbers. Of note, we did not have any information on rhythmology at the emergency department level or various interventions performed in the ICU, which potentially had impacted NIRS values during the observational period. We also did not have enough information on somatosensory evoked potentials, as they were either not assessed in our patients or the respective data were not available. Finally, we did not include eCPR patients, which could potentially impact our results. Our findings are of a hypothesis-generating nature only.
Regarding the narrative review section of this publication, the search strategy was designed without he involvement of an information specialist. Additionally, neither a scoping nor a systematic review was conducted, and no risk of bias assessments were performed, which limits the value of the summarized information.
6 Conclusion
We identified a potential RSO2 cutoff measured via NIRS in the post-ROSC phase after OHCA to predict a favorable neurological outcome. Initial values and rising dynamics could be more useful for prognostication than a prolonged measurement in the ICU. These findings fit into previous literature and should trigger larger respective trials on the one hand, and the technology being addressed by resuscitation societies on the other hand.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The studies involving humans were approved by Ethics Committee of the Medical University of Vienna, Austria. The studies were conducted in accordance with the local legislation and institutional requirements. The ethics committee/institutional review board waived the requirement of written informed consent for participation from the participants or the participants’ legal guardians/next of kin because it was not deemed necessary by the EC due to patients being unconscious at the time of NIRS measurement.
Author contributions
SS: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. AK: Data curation, Methodology, Validation, Writing – review & editing. FE: Data curation, Methodology, Validation, Writing – review & editing. CS: Data curation, Methodology, Validation, Writing – review & editing. FM: Data curation, Methodology, Validation, Writing – review & editing. PA: Project administration, Resources, Validation, Writing – review & editing. MaG: Investigation, Project administration, Resources, Validation, Writing – review & editing. MiG: Project administration, Resources, Supervision, Validation, Writing – review & editing. DG: Investigation, Project administration, Resources, Supervision, Validation, Writing – review & editing. AZ: Project administration, Resources, Supervision, Writing – review & editing. AS: Data curation, Investigation, Validation, Writing – review & editing. WS: Investigation, Project administration, Supervision, Validation, Writing – review & editing. MH: Investigation, Project administration, Supervision, Validation, Writing – review & editing. HL: Investigation, Project administration, Supervision, Validation, Writing – review & editing. MK: Project administration, Resources, Supervision, Validation, Writing – review & editing. TU: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Writing – original draft, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This research received a grant (no. 02/2022) by the Austrian association for medical and general intensive care and emergency medicine (ÖGIAIN-Österreichische Gesellschaft für Internistische und Allgemeine Intensivmedizin und Notfallmedizin). The ÖGIAIN was not involved in the study design, data analysis, or publication.
Acknowledgments
We thank all personnel involved in acquiring the study data, especially Gerhard Ruzicka, Juergen Novotny, and the members of the Field Supervisor Unit (FISU), and the research car of the Emergency Medical Service Vienna, in particular Rene Adler, Harald Bendl, Philipp Gonzo, Ronald Kopta, Michael Mareda, Henrik Maszar, Marcel Pfaffenlehner, Julian Raming, Bernhard Saxinger, Bertram Schadler, Martin Thalhammer, Harald Glueck, Joerg Holzinger, Matthias Kießlinger, Manfred Meixner, Andreas Schindler, Georg Schrattenbacher, and Stefan Schauer.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Generative AI statement
The author(s) declare that no Gen AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2025.1590908/full#supplementary-material
References
1. Ali, J, Cody, J, Maldonado, Y, and Ramakrishna, H. Near-infrared spectroscopy (NIRS) for cerebral and tissue oximetry: analysis of evolving applications. J Cardiothorac Vasc Anesth. (2022) 36:2758–66. doi: 10.1053/j.jvca.2021.07.015
2. Moerman, A, and Wouters, P. Near-infrared spectroscopy (NIRS) monitoring in contemporary anesthesia and critical care. Acta Anaesthesiol Belg. (2010) 61:185–94.
3. Marin, T, and Moore, J. Understanding near-infrared spectroscopy: an update. Crit Care Nurs Clin North Am. (2024) 36:41–50. doi: 10.1016/j.cnc.2023.08.001
4. Huppert, EL, and Parnia, S. Cerebral oximetry: a developing tool for monitoring cerebral oxygenation during cardiopulmonary resuscitation. Ann N Y Acad Sci. (2022) 1509:12–22. doi: 10.1111/nyas.14706
5. Soar, J, Böttiger, BW, Carli, P, Couper, K, Deakin, CD, Djärv, T, et al. European resuscitation council guidelines 2021: adult advanced life support. Resuscitation. (2021) 161:115–51. doi: 10.1016/j.resuscitation.2021.02.010
6. Lanks, C, Kim, CB, and Rossiter, HB. A “NIRS” death experience: a reduction in cortical oxygenation by time-resolved near-infrared spectroscopy preceding cardiac arrest. J Clin Monit Comput. (2018) 32:683–6. doi: 10.1007/s10877-017-0061-8
7. Jakkula, P, Hästbacka, J, Reinikainen, M, Pettilä, V, Loisa, P, Tiainen, M, et al. Near-infrared spectroscopy after out-of-hospital cardiac arrest. Crit Care. (2019) 23:171. doi: 10.1186/s13054-019-2428-3
8. Schnaubelt, S, Sulzgruber, P, Menger, J, Skhirtladze-Dworschak, K, Sterz, F, and Dworschak, M. Regional cerebral oxygen saturation during cardiopulmonary resuscitation as a predictor of return of spontaneous circulation and favourable neurological outcome—a review of the current literature. Resuscitation. (2018) 125:39–47. doi: 10.1016/j.resuscitation.2018.01.028
9. Genbrugge, C, Eertmans, W, Meex, I, Van Kerrebroeck, M, Daems, N, Creemers, A, et al. What is the value of regional cerebral saturation in post-cardiac arrest patients? A prospective observational study. Crit Care Lond Engl. (2016) 20:327. doi: 10.1186/s13054-016-1509-9
10. Nolan, JP, Sandroni, C, Böttiger, BW, Cariou, A, Cronberg, T, Friberg, H, et al. European resuscitation council and European Society of Intensive Care Medicine Guidelines 2021: post-resuscitation care. Resuscitation. (2021) 161:220–69. doi: 10.1016/j.resuscitation.2021.02.012
11. Berg, KM, Bray, JE, Ng, K-C, Liley, HG, Greif, R, Carlson, JN, et al. 2023 international consensus on cardiopulmonary resuscitation and emergency cardiovascular care science with treatment recommendations: summary from the basic life support; advanced life support; pediatric life support; neonatal life support; education, implementation, and teams; and first aid task forces. Circulation. (2023) 148:e187–280. doi: 10.1161/CIR.0000000000001179
12. Geocadin, RG, Callaway, CW, Fink, EL, Golan, E, Greer, DM, Ko, NU, et al. Standards for studies of neurological prognostication in comatose survivors of cardiac arrest: a scientific statement from the American Heart Association. Circulation. (2019) 140:e517–42. doi: 10.1161/CIR.0000000000000702
13. Hirsch, KG, Abella, BS, Amorim, E, Bader, MK, Barletta, JF, Berg, K, et al. Critical Care Management of Patients after Cardiac Arrest: a scientific statement from the American Heart Association and Neurocritical care society. Neurocrit Care. (2024) 40:1–37. doi: 10.1007/s12028-023-01871-6
14. Mueller, M, Losert, H, Sterz, F, Gelbenegger, G, Girsa, M, Gatterbauer, M, et al. Prehospital emergency medicine research by additional teams on scene – concepts and lessons learned. Resusc Plus. (2023) 16:100494. doi: 10.1016/j.resplu.2023.100494
15. SenSmart X-100. Nonin (n.d.). https://www.nonin.com/support/x100/ (accessed March 1, 2022).
16. Sandroni, C, D’Arrigo, S, Cacciola, S, Hoedemaekers, CWE, Kamps, MJA, Oddo, M, et al. Prediction of poor neurological outcome in comatose survivors of cardiac arrest: a systematic review. Intensive Care Med. (2020) 46:1803–51. doi: 10.1007/s00134-020-06198-w
17. Sandroni, C, D’Arrigo, S, and Nolan, JP. Prognostication after cardiac arrest. Crit Care. (2018) 22:150. doi: 10.1186/s13054-018-2060-7
18. Kool, M, Atkins, DL, Van de Voorde, P, Maconochie, IK, and Scholefield, BRPLS ILCOR Task Force. Focused echocardiography, end-tidal carbon dioxide, arterial blood pressure or near-infrared spectroscopy monitoring during paediatric cardiopulmonary resuscitation: a scoping review. Resusc Plus. (2021) 6:100109. doi: 10.1016/j.resplu.2021.100109
19. Soloperto, R, Magni, F, Farinella, A, Bogossian, EG, Peluso, L, De Luca, N, et al. A comparison of prognostic factors in a large cohort of in-hospital and out-of-hospital cardiac arrest patients. Life Basel Switz. (2024) 14:403. doi: 10.3390/life14030403
20. Gonzalez, D, Dahiya, G, Mutirangura, P, Ergando, T, Mello, G, Singh, R, et al. Post cardiac arrest Care in the Cardiac Intensive Care Unit. Curr Cardiol Rep. (2024) 26:35–49. doi: 10.1007/s11886-023-02015-0
21. Park, JS, Kim, EY, You, Y, Min, JH, Jeong, W, Ahn, HJ, et al. Combination strategy for prognostication in patients undergoing post-resuscitation care after cardiac arrest. Sci Rep. (2023) 13:21880. doi: 10.1038/s41598-023-49345-1
22. Kang, JK, Darby, Z, Bleck, TP, Whitman, GJR, Kim, BS, and Cho, S-M. Post-cardiac arrest Care in Adult Patients after Extracorporeal Cardiopulmonary Resuscitation. Crit Care Med. (2024) 52:483–94. doi: 10.1097/CCM.0000000000006102
23. Putzer, G, Martini, J, Spraider, P, Hornung, R, Pinggera, D, Abram, J, et al. Effects of different adrenaline doses on cerebral oxygenation and cerebral metabolism during cardiopulmonary resuscitation in pigs. Resuscitation. (2020) 156:223–9. doi: 10.1016/j.resuscitation.2020.06.024
24. Nosrati, R, Lin, S, Mohindra, R, Ramadeen, A, Toronov, V, and Dorian, P. Study of the effects of epinephrine on cerebral oxygenation and metabolism during cardiac arrest and resuscitation by hyperspectral near-infrared spectroscopy. Crit Care Med. (2019) 47:e349–57. doi: 10.1097/CCM.0000000000003640
25. Reagan, EM, Nguyen, RT, Ravishankar, ST, Chabra, V, Fuentes, B, Spiegel, R, et al. Monitoring the relationship between changes in cerebral oxygenation and electroencephalography patterns during cardiopulmonary resuscitation: a feasibility study. Crit Care Med. (2018) 46:757–63. doi: 10.1097/CCM.0000000000003014
26. Sakai, T, Hirose, T, Shiozaki, T, Takagawa, R, Ohnishi, M, Hayashida, S, et al. Pre-hospital portable monitoring of cerebral regional oxygen saturation (rSO2) by ambulance personnel during cardiopulmonary resuscitation: a prospective observational analysis of 87 cases in Osaka city, Japan. Resusc Plus. (2021) 6:100093. doi: 10.1016/j.resplu.2021.100093
27. Genbrugge, C, De Deyne, C, Eertmans, W, Anseeuw, K, Voet, D, Mertens, I, et al. Cerebral saturation in cardiac arrest patients measured with near-infrared technology during pre-hospital advanced life support. Results from Copernicus I cohort study. Resuscitation. (2018) 129:107–13. doi: 10.1016/j.resuscitation.2018.03.031
28. Kawaguchi, T, Tsukuda, J, Onoe, R, Morisawa, K, Yoshida, T, Hayashi, K, et al. Association between regional cerebral oxygen saturation and outcome of patients with out-of-hospital cardiac arrest: an observational study. Resusc Plus. (2023) 13:100343. doi: 10.1016/j.resplu.2022.100343
29. Takegawa, R, Hayashida, K, Rolston, DM, Li, T, Miyara, SJ, Ohnishi, M, et al. Near-infrared spectroscopy assessments of regional cerebral oxygen saturation for the prediction of clinical outcomes in patients with cardiac arrest: a review of clinical impact, evolution, and future directions. Front Med. (2020) 7:587930. doi: 10.3389/fmed.2020.587930
30. Sanfilippo, F, Murabito, P, Messina, A, Dezio, V, Busalacchi, D, Ristagno, G, et al. Cerebral regional oxygen saturation during cardiopulmonary resuscitation and return of spontaneous circulation: a systematic review and meta-analysis. Resuscitation. (2021) 159:19–27. doi: 10.1016/j.resuscitation.2020.12.002
31. Takegawa, R, Shiozaki, T, Ogawa, Y, Hirose, T, Mori, N, Ohnishi, M, et al. Usefulness of cerebral rSO2 monitoring during CPR to predict the probability of return of spontaneous circulation. Resuscitation. (2019) 139:201–7. doi: 10.1016/j.resuscitation.2019.04.015
32. Schnaubelt, S, and Uray, T. The need for comprehensive NIRS-measurement from on-scene treatment to post-ROSC phase. Resuscitation. (2019) 144:191–2. doi: 10.1016/j.resuscitation.2019.07.038
33. Meex, I, De Deyne, C, Dens, J, Scheyltjens, S, Lathouwers, K, Boer, W, et al. Feasibility of absolute cerebral tissue oxygen saturation during cardiopulmonary resuscitation. Crit Care Lond Engl. (2013) 17:R36. doi: 10.1186/cc12546
34. Schewe, J-C, Thudium, MO, Kappler, J, Steinhagen, F, Eichhorn, L, Erdfelder, F, et al. Monitoring of cerebral oxygen saturation during resuscitation in out-of-hospital cardiac arrest: a feasibility study in a physician staffed emergency medical system. Scand J Trauma Resusc Emerg Med. (2014) 22:58. doi: 10.1186/s13049-014-0058-y
35. Asim, K, Gokhan, E, Ozlem, B, Ozcan, Y, Deniz, O, Kamil, K, et al. Near infrared spectrophotometry (cerebral oximetry) in predicting the return of spontaneous circulation in out-of-hospital cardiac arrest. Am J Emerg Med. (2014) 32:14–7. doi: 10.1016/j.ajem.2013.09.010
36. Parker, J, Walenta, T, and Turner-Nelson, K. Near-infrared spectroscopy in transport with a patient in multi-factorial shock. Air Med J. (2019) 38:235–8. doi: 10.1016/j.amj.2019.03.005
37. Raschdorf, K, Mohseni, A, Hogle, K, Cheung, A, So, K, Manouchehri, N, et al. Evaluation of transcutaneous near-infrared spectroscopy for early detection of cardiac arrest in an animal model. Sci Rep. (2023) 13:4537. doi: 10.1038/s41598-023-31637-1
38. Nissen, P, Brassard, P, Jørgensen, TB, and Secher, NH. Phenylephrine but not ephedrine reduces frontal lobe oxygenation following anesthesia-induced hypotension. Neurocrit Care. (2010) 12:17–23. doi: 10.1007/s12028-009-9313-x
39. Meng, L, Cannesson, M, Alexander, BS, Yu, Z, Kain, ZN, Cerussi, AE, et al. Effect of phenylephrine and ephedrine bolus treatment on cerebral oxygenation in anaesthetized patients. Br J Anaesth. (2011) 107:209–17. doi: 10.1093/bja/aer150
40. Balakrishnan, B, Dasgupta, M, Gajewski, K, Hoffmann, RG, Simpson, PM, Havens, PL, et al. Low near infrared spectroscopic somatic oxygen saturation at admission is associated with need for lifesaving interventions among unplanned admissions to the pediatric intensive care unit. J Clin Monit Comput. (2018) 32:89–96. doi: 10.1007/s10877-017-0007-1
41. Perkins, GD, Callaway, CW, Haywood, K, Neumar, RW, Lilja, G, Rowland, MJ, et al. Brain injury after cardiac arrest. Lancet. (2021) 398:1269–78. doi: 10.1016/S0140-6736(21)00953-3
42. Sandroni, C, Cronberg, T, and Sekhon, M. Brain injury after cardiac arrest: pathophysiology, treatment, and prognosis. Intensive Care Med. (2021) 47:1393–414. doi: 10.1007/s00134-021-06548-2
43. Coppler, PJ, and Elmer, J. Optimizing cerebral oxygen delivery after cardiac arrest: a role for neuromonitoring. Resuscitation. (2021) 169:220–2. doi: 10.1016/j.resuscitation.2021.10.043
44. Katzenschlager, S, Popp, E, Wnent, J, Weigand, MA, and Gräsner, J-T. Developments in post-resuscitation Care for out-of-Hospital Cardiac Arrests in adults-a narrative review. J Clin Med. (2023) 12:3009. doi: 10.3390/jcm12083009
45. Kinoshita, K, Sakurai, A, and Ihara, S. The pitfalls of bedside regional cerebral oxygen saturation in the early stage of post cardiac arrest. Scand J Trauma Resusc Emerg Med. (2015) 23:95. doi: 10.1186/s13049-015-0173-4
46. Robba, C, Battaglini, D, Rasulo, F, Lobo, FA, and Matta, B. The importance of monitoring cerebral oxygenation in non brain injured patients. J Clin Monit Comput. (2023) 37:943–9. doi: 10.1007/s10877-023-01002-8
47. Bhatia, A, and Gupta, AK. Neuromonitoring in the intensive care unit. II. Cerebral oxygenation monitoring and microdialysis. Intensive Care Med. (2007) 33:1322–8. doi: 10.1007/s00134-007-0660-9
48. Ahn, A, Yang, J, Inigo-Santiago, L, and Parnia, S. A feasibility study of cerebral oximetry monitoring during the post-resuscitation period in comatose patients following cardiac arrest. Resuscitation. (2014) 85:522–6. doi: 10.1016/j.resuscitation.2013.12.007
49. Skrifvars, MB, and Åneman, A. Near-infrared spectroscopy “under pressure” as a post-cardiac arrest monitoring technique of cerebral autoregulation. Resuscitation. (2020) 152:203–4. doi: 10.1016/j.resuscitation.2020.04.020
50. Hansen, ML, Hyttel-Sørensen, S, Jakobsen, JC, Gluud, C, Kooi, EMW, Mintzer, J, et al. Cerebral near-infrared spectroscopy monitoring (NIRS) in children and adults: a systematic review with meta-analysis. Pediatr Res. (2022) 96:856–67. doi: 10.1038/s41390-022-01995-z
51. Meex, I, Dens, J, Jans, F, Boer, W, Vanhengel, K, Vundelinckx, G, et al. Cerebral tissue oxygen saturation during therapeutic hypothermia in post-cardiac arrest patients. Resuscitation. (2013) 84:788–93. doi: 10.1016/j.resuscitation.2013.01.003
52. Storm, C, Leithner, C, Krannich, A, Wutzler, A, Ploner, CJ, Trenkmann, L, et al. Regional cerebral oxygen saturation after cardiac arrest in 60 patients--a prospective outcome study. Resuscitation. (2014) 85:1037–41. doi: 10.1016/j.resuscitation.2014.04.021
53. Bouglé, A, Daviaud, F, Bougouin, W, Rodrigues, A, Geri, G, Morichau-Beauchant, T, et al. Determinants and significance of cerebral oximetry after cardiac arrest: a prospective cohort study. Resuscitation. (2016) 99:1–6. doi: 10.1016/j.resuscitation.2015.11.011
54. Sarıtaş, A, Çinleti, BA, Zincircioğlu, Ç, Uzun, U, Köse, I, and Şenoğlu, N. Effect of regional cerebral oximetry to estimate neurologic prognostic outcomes in patients administered targeted temperature management. Am J Emerg Med. (2018) 36:2236–41. doi: 10.1016/j.ajem.2018.04.016
55. Nakatani, Y, Nakayama, T, Nishiyama, K, and Takahashi, Y. Effect of target temperature management at 32-34 °C in cardiac arrest patients considering assessment by regional cerebral oxygen saturation: a multicenter retrospective cohort study. Resuscitation. (2018) 126:185–90. doi: 10.1016/j.resuscitation.2018.02.007
56. Sakurai, A, Ihara, S, Tagami, R, Yamaguchi, J, Sugita, A, Kuwana, T, et al. Parameters influencing brain oxygen measurement by regional oxygen saturation in Postcardiac arrest patients with targeted temperature management. Ther Hypothermia Temp Manag. (2020) 10:71–5. doi: 10.1089/ther.2019.0032
57. Ameloot, K, De Deyne, C, Eertmans, W, Ferdinande, B, Dupont, M, Palmers, P-J, et al. Early goal-directed haemodynamic optimization of cerebral oxygenation in comatose survivors after cardiac arrest: the Neuroprotect post-cardiac arrest trial. Eur Heart J. (2019) 40:1804–14. doi: 10.1093/eurheartj/ehz120
58. Kirschen, MP, Majmudar, T, Beaulieu, F, Burnett, R, Shaik, M, Morgan, RW, et al. Deviations from NIRS-derived optimal blood pressure are associated with worse outcomes after pediatric cardiac arrest. Resuscitation. (2021) 168:110–8. doi: 10.1016/j.resuscitation.2021.09.023
59. Hoiland, RL, Robba, C, Menon, DK, Citerio, G, Sandroni, C, and Sekhon, MS. Clinical targeting of the cerebral oxygen cascade to improve brain oxygenation in patients with hypoxic-ischaemic brain injury after cardiac arrest. Intensive Care Med. (2023) 49:1062–78. doi: 10.1007/s00134-023-07165-x
60. Ibrahim, AW, Trammell, AR, Austin, H, Barbour, K, Onuorah, E, House, D, et al. Cerebral oximetry as a real-time monitoring tool to assess quality of in-hospital cardiopulmonary resuscitation and post cardiac arrest care. J Am Heart Assoc. (2015) 4:e001859. doi: 10.1161/JAHA.115.001859
61. Storm, C, Wutzler, A, Trenkmann, L, Krannich, A, von Rheinbarben, S, Luckenbach, F, et al. Good neurological outcome despite very low regional cerebral oxygen saturation during resuscitation--a prospective preclinical trial in 29 patients. Scand J Trauma Resusc Emerg Med. (2016) 24:43. doi: 10.1186/s13049-016-0234-3
62. Kwon, WY, Jung, YS, Suh, GJ, Kim, T, Kwak, H, Kim, T, et al. Regional cerebral oxygen saturation in cardiac arrest survivors undergoing targeted temperature management 36 °C versus 33 °C: a randomized clinical trial. Resuscitation. (2021) 167:362–71. doi: 10.1016/j.resuscitation.2021.07.026
63. Sun, H, Zhu, J, Ma, L, Lu, J, Liu, L, and Zhou, B. Changes of regional saturation of cerebral oxygenation and neuron specific enolase in patients after cardiopulmonary resuscitation. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. (2021) 33:1094–8. doi: 10.3760/cma.j.cn121430-20210115-00067
64. Nishiyama, K, Ito, N, Orita, T, Hayashida, K, Arimoto, H, Beppu, S, et al. Regional cerebral oxygen saturation monitoring for predicting interventional outcomes in patients following out-of-hospital cardiac arrest of presumed cardiac cause: a prospective, observational, multicentre study. Resuscitation. (2015) 96:135–41. doi: 10.1016/j.resuscitation.2015.07.049
65. Tran, LN, Patel, J, Yang, J, O’Neill, C, Yin, D, Nguyen, R, et al. The association between post-cardiac arrest cerebral oxygenation and survival with favorable neurological outcomes: a multicenter study. Resuscitation. (2020) 154:85–92. doi: 10.1016/j.resuscitation.2020.06.006
66. Iordanova, B, Li, L, Clark, RSB, and Manole, MD. Alterations in cerebral blood flow after resuscitation from cardiac arrest. Front Pediatr. (2017) 5:174. doi: 10.3389/fped.2017.00174
67. van den Brule, JMD, van der Hoeven, JG, and Hoedemaekers, CWE. Cerebral perfusion and cerebral autoregulation after cardiac arrest. Biomed Res Int. (2018) 2018:1–5. doi: 10.1155/2018/4143636
68. Nakashima, R, Hifumi, T, Kawakita, K, Okazaki, T, Egawa, S, Inoue, A, et al. Critical care management focused on optimizing brain function after cardiac arrest. Circ J. (2017) 81:427–39. doi: 10.1253/circj.CJ-16-1006
69. Takegawa, R, Hayashida, K, Yin, T, Choudhary, RC, Miyara, SJ, Khalili, H, et al. Real-time brain monitoring by near-infrared spectroscopy predicts neurological outcome after cardiac arrest and resuscitation in rats: a proof of concept study of a novel prognostic measure after cardiac arrest. J Clin Med. (2021) 11:131. doi: 10.3390/jcm11010131
70. Inal, MT, Memiş, D, Yıldırım, I, Uğur, H, Erkaymaz, A, and Turan, FN. The prognostic value of cerebral oxygen saturation measurement for assessing prognosis after cardiopulmonary resuscitation. Rev Bras Anestesiol. (2017) 67:355–61. doi: 10.1016/j.bjan.2016.07.008
71. Hazenberg, L, Aries, M, Beqiri, E, Mess, WH, van Mook, W, Delnoij, T, et al. Are NIRS-derived cerebral autoregulation and ABPopt values different between hemispheres in hypoxic-ischemic brain injury patients following cardiac arrest? J Clin Monit Comput. (2023) 37:1427–30. doi: 10.1007/s10877-023-01008-2
72. Yagi, T, Shinozaki, K, Okuma, Y, Yin, T, Nishikimi, M, Kiguchi, T, et al. Assessment of cerebral blood oxygenation by near-infrared spectroscopy before and after resuscitation in a rat asphyxia cardiac arrest model. Adv Exp Med Biol. (2021) 1269:311–5. doi: 10.1007/978-3-030-48238-1_49
73. Eastwood, GM, Tanaka, A, and Bellomo, R. Cerebral oxygenation in mechanically ventilated early cardiac arrest survivors: the impact of hypercapnia. Resuscitation. (2016) 102:11–6. doi: 10.1016/j.resuscitation.2016.02.009
Keywords: cerebral oxygenation, near-infrared spectroscopy, NIRS, cardiac arrest, cardiopulmonary resuscitation, CPR, prognostication
Citation: Schnaubelt S, Kornfehl A, Eibensteiner F, Schriefl C, Mayr FB, Aigner P, Gatterbauer M, Girsa M, Grassmann D, Zajicek A, Spiel A, Schreiber W, Holzer M, Losert H, Krammel M and Uray T (2025) Comprehensive pre- and in-hospital near-infrared-spectroscopy (NIRS) monitoring after return of spontaneous circulation predicts neurological outcome following out-of-hospital cardiac arrest: a prospective observational study and literature review. Front. Med. 12:1590908. doi: 10.3389/fmed.2025.1590908
Edited by:
Alberto Zani, Vita-Salute San Raffaele University, ItalyReviewed by:
Simone Maria Zerbi, ASST Lariana, ItalyJae Hwan Kim, Chung-Ang University Hospital, Republic of Korea
Copyright © 2025 Schnaubelt, Kornfehl, Eibensteiner, Schriefl, Mayr, Aigner, Gatterbauer, Girsa, Grassmann, Zajicek, Spiel, Schreiber, Holzer, Losert, Krammel and Uray. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sebastian Schnaubelt, c2ViYXN0aWFuLnNjaG5hdWJlbHRAbWVkdW5pd2llbi5hYy5hdA==
†These authors have contributed equally to this work
 Sebastian Schnaubelt
Sebastian Schnaubelt Andrea Kornfehl
Andrea Kornfehl Felix Eibensteiner
Felix Eibensteiner Christoph Schriefl
Christoph Schriefl Florian B. Mayr4
Florian B. Mayr4 Alexander Spiel
Alexander Spiel Michael Holzer
Michael Holzer Heidrun Losert
Heidrun Losert Mario Krammel
Mario Krammel