- 1Biological Control Research Unit, Center for Natural Sciences and Environmental Research, De La Salle University, Manila, Philippines
- 2Department of Biology, College of Science, De La Salle University, Manila, Philippines
- 3Bureau of Plant Industry - Davao, Davao City, Philippines
- 4College of Agriculture, Sultan Kudarat State University, Sultan Kudarat, Philippines
- 5Center for Medical, Agricultural and Veterinary Entomology, US Department of Agriculture, Agricultural Research Service, Tallahassee, FL, United States
- 6Invasive Insect Biocontrol and Behavior Laboratory, USDA-ARS, Beltsville, MD, United States
Cacao is a promising high-value crop in the Philippines and is considered one of the key players globally in providing the raw materials for chocolate making. However, before the Philippines can take this market opportunity, production constraints from various pests should be addressed. The cacao pod borer (CPB), Conopomorpha cramerella, is one of the major insect pests of cacao in the Philippines, which can cause up to 50% annual loss or even higher if proper management is not employed. Management of CPB heavily relies on chemical control but is becoming unpopular because of its high cost and health risk to people and the environment. A number of efforts have been made to develop sex pheromones to monitor and control CPB. A field bioassay of the new blend of CPB sex pheromones was conducted in southern Philippines to determine its efficiency and identify the appropriate trap height placement in a cacao field, which is important in maximizing the efficiency of the sex pheromone traps for inclusion in the integrated pest management (IPM) program of CPB. Pheromone lures at four different trap heights (1.0 m, 0.5 m above the canopy, 0.0 m or along the canopy, and 0.5 m below the canopy) and a blank lure (control) placed 1.0 m above the canopy were evaluated in terms of male CPB trap catches. Results showed significantly higher male CPB trap catches when installed at least 0.5 m above the canopy while minimal to zero male CPB catches were obtained from the control traps with blank lures, confirming the attractiveness of the new blend of the CPB sex pheromone in the field. This suggests the reliability of the pheromone-based method to monitor and control CPB and as a major component of the IPM program.
1 Introduction
Cacao is a promising high-value crop in the Philippines and was affirmed as such by legislation with provision for funding in Republic Act 7900 (Republic of the Philippines, 2010). Cacao plantations were reported in 1991 to occur in approximately 479,000 farms situated on a total area of 17,300 ha, most of which were backyard or smallholder plantations (Limbo, 2019). The latest statistics have indicated an average of 9.3% per annum growth in the production of cacao in the country from 2017 to 2021 (Philippine Statistics Authority, 2023). Annual production of cocoa beans recorded in 2021 was 10,000 mt, while the land area of production was approximately 31,500 ha.
Cacao growing in the Philippines is recently making a comeback because of increasing global demand for dried, fermented cacao beans. Global consumption of chocolate has been increasing 3% annually with new market demand growing steadily in Central Asia, Eastern Europe, India, and various states of the Russian Federation. In line with this, global cacao production is getting slimmer; thus, there is a need to promote more plantings of cacao. The government is actively promoting more planting of cacao to take the high market opportunity from this commodity. In lieu of the Philippine cacao roadmap (2016–2022) that aimed at a target production of 100,000 mt by the year 2022, cacao farmers would have needed to plant 50 million cacao trees in the last 7 years. Although this was not accomplished, this target can still be achieved since the potential expansion for cacao growing is huge just by utilizing the existing coconut plantations of approximately 2,000,000 ha.
Studies have shown that cacao is a highly suitable and profitable companion crop of coconut. It can give an additional income of between Php 60,000 and 80,000 from 500 to 600 cacao trees per hectare per year when intercropped with coconut, but before the Philippines can take this market opportunity, production constraints from insect pests and diseases should be addressed. The most important insect pests and diseases of cacao in the Philippines are cacao pod borer (CPB) [Conopomorpha cramerella (Snellen)], cacao mirid bug (CMB) [Helopeltis bakeri Poppius], black pod rot (BPR) caused by Phytopthora palmivora (Butl.), and vascular streak dieback (VSD) caused by Lasiodiplodia theobromae (Pat.) Griffon & Maubl. Both insects and BPR attack the pods while VSD attacks the vascular system of the tree. All of them can lead to significant decreases in production if their populations are not managed. Gavarra (1990) recorded 60%–80% infestation from CPB in Davao. There is no documented record on the possible percent infestation from CMB, but it can cause as high as 90% without control (personal observation, Cacao Demo Farm, Tiaong, Quezon). For BPR, Reinking (1923) recorded a range of infestation from 20% to 30% every year but can reach up to 90% with no control. Alicbusan (1935) observed a similar level of infestation from BPR. In 1990, Tandingan and Ampo reported approximately 49% infestation from VSD in Southern Mindanao (Tandingan et al., 1990).
Collectively, percent infestation from all these pests will surely lead to high annual losses in cacao production in the Philippines, impacting both the industry and the farmers. Farmers usually resort to the use of chemical control, but this is becoming unpopular because of the high cost and health risk to people and the environment. Alternatively, sleeving or bagging of pods with plastic is being used, but this is labor intensive. A new and innovative pest management approach that can incorporate an array of biologically based technologies (BBTs) to underpin effective IPM is needed.
The cacao community stakeholders during the International Cocoa Research Conference identified IPM as one of the components for the proposed Global Cocoa Programme, to improve profitability by increasing productivity, decreasing cost, while maintaining farm viability or stability in times of fluctuating prices. For instance, control strategies for CPB that were tried include pod sleeving (Vanhove et al., 2015); the use of biological control agents such as nematode (Steinernema carpocapsae) (Rosmana et al., 2013), Bacillus thuringiensis toxin (Santoso et al., 2004), and the fungus Beauveria bassiana (Vanhove et al., 2015); and the application of synthetic sex pheromone for mass trapping and mating disruption (Zhang et al., 2008).
Sex-pheromone-based control strategies have been important components of IPM systems owing to their nature as environment friendly regulators of insect behavior (Rizvi et al., 2021). Since they are species-specific, non-target organisms such as natural enemies and pollinators are not harmed. Except for closely related species, even incomplete synthetic pheromone blends usually only attract the target (Cardé and Haynes, 2004). The sex pheromone components of CPB were identified (Beevor et al., 1986) and field tested in Sabah, Malaysia, but were not widely used due to constraints on production. In 2004, new synthetic blends of the sex pheromones were re-evaluated in Malaysia and Indonesia and revealed that lures containing 100 µg placed in a delta trap was the most effective (Zhang et al., 2008). This result was supported by recent field trials in Indonesia where lures containing 100 µg of sex pheromone blend placed in traps 1.0 m above the canopy yielded the highest trap catches (Niogret et al., 2022)
The present study evaluated the potential of sex pheromone as a biological system to monitor and manage the population of CPB in the Philippines. Specifically, this study was conducted to determine the attractiveness of the new blend of CPB sex pheromone, re-evaluate the effect of trap height on the male CPB trap catches, and determine CPB seasonal abundance in southern Philippines.
2 Materials and methods
2.1 Study site
The first field evaluation for the optimization of the trap height was conducted in a 5,200-m2 plot within a 1-ha cacao plantation in Sirib, Calinan, Davao City, Philippines (7.1353° N, 125.4163° E) as the experimental area. The experimental area was divided into 15 subplots measuring 347 m2 per subplot to cover the experimental setup for five treatments with three replications. The field site was selected because of the high pest pressure from CPB. The selected site had 5-year-old cacao plants. The pest management regime in this cacao field site only included plastic sleeving and sanitation. No pesticide application was used since the field is included in the organic farming group in the area. The second field testing using the optimum trap height was performed in the same cacao plantation with a wider experimental area with two sites covering a total area of 9 ha.
2.2 CPB lure and trapping system
CPB lures were polyethylene vials (Just Plastic. Ltd) impregnated with 100 µg of the synthetic new pheromone blends that were obtained from the Invasive Insect Biocontrol and Behavior Laboratory in USDA-ARS, Beltsville, MD, USA. The lures contain a mixture of E,Z,Z- and E,E,Z-4,6,10-hexadecatrienyl acetates and their corresponding alcohols at a ratio of ~ 40:55:4:6 and an equal weight of 2,6-di-tert-butyl-4-methylphenol (BHT) as antioxidant (Vanhove et al., 2015). These lures were shipped to the Philippines in triple-sealed plastic bags. All lures were stored in a freezer until field testing. Deployment of the lures was done using delta traps (Trece Pherocon® VI) with sticky liners.
2.3 Experimental setup
For the assessment of the appropriate trap height, five treatments representing different trap heights were included: Treatment A (trap installed 1.0 m above the canopy), Treatment B (trap installed 0.5 m above the canopy), Treatment C (trap installed 0.0 m or along the canopy), Treatment D (trap installed 0.5 m below the canopy), and Treatment E (control; trap installed 1.0 m above the canopy but with blank CPB lure). The experiment was performed following a randomized complete block design (RCBD) with three replications. The sticky liners were collected and replaced every month for 12 months. The numbers of male CPB trapped in the sticky liners collected every month were counted and recorded separately. Delta traps were rotated every 3 months to avoid bias in the position of the traps (photographs of delta traps used are shown in Figure 1). Average count of male CPB trap catches per month was obtained and compared among treatments. For the second field trial, all traps were installed 0.5 m above the canopy. Collection was done every month for 5 months.

Figure 1 (A) Empty delta trap with sticky liner, (B) delta trap with a pheromone lure showing male CPB trap catch, and (C) delta trap placed 1.0 m above the tree canopy.
2.4 Data analysis
Data were analyzed using IBM SPSS Statistics for Windows, version 20 (IBM Corp. Armonk, NY, USA). For the evaluation of the effect of trap height placement, insect count data from trap catches were log transformed and analyzed by ANOVA followed by post-hoc Tukey–Kramer test to determine differences among treatments.
3 Results
The average trap catches of male CPB per month from the treatments with varying trap height are shown in Figure 2. The male CPB trap catches were recorded the highest in the trap installed 0.5 m above the canopy but not significantly different from 1.0 m above the canopy for all the monthly sampling dates (Figure 2) and overall trap catches per height (Table 1). The average trap catches for the different treatments were recorded in the following order: 0.5 m above the canopy, 1.0 m above the canopy, 0.5 m below the canopy, and 0.0 m or along the canopy. No male CPB was recovered in the control treatment except in February, August, and September 2018 but only one individual in one of the traps was captured. Additionally, the monthly male CPB trap catches showed the seasonal abundance pattern of CPB in the field. Two peaks of population were recorded (Figure 2). The highest peak was recorded in October and a second but lower peak was recorded in April.
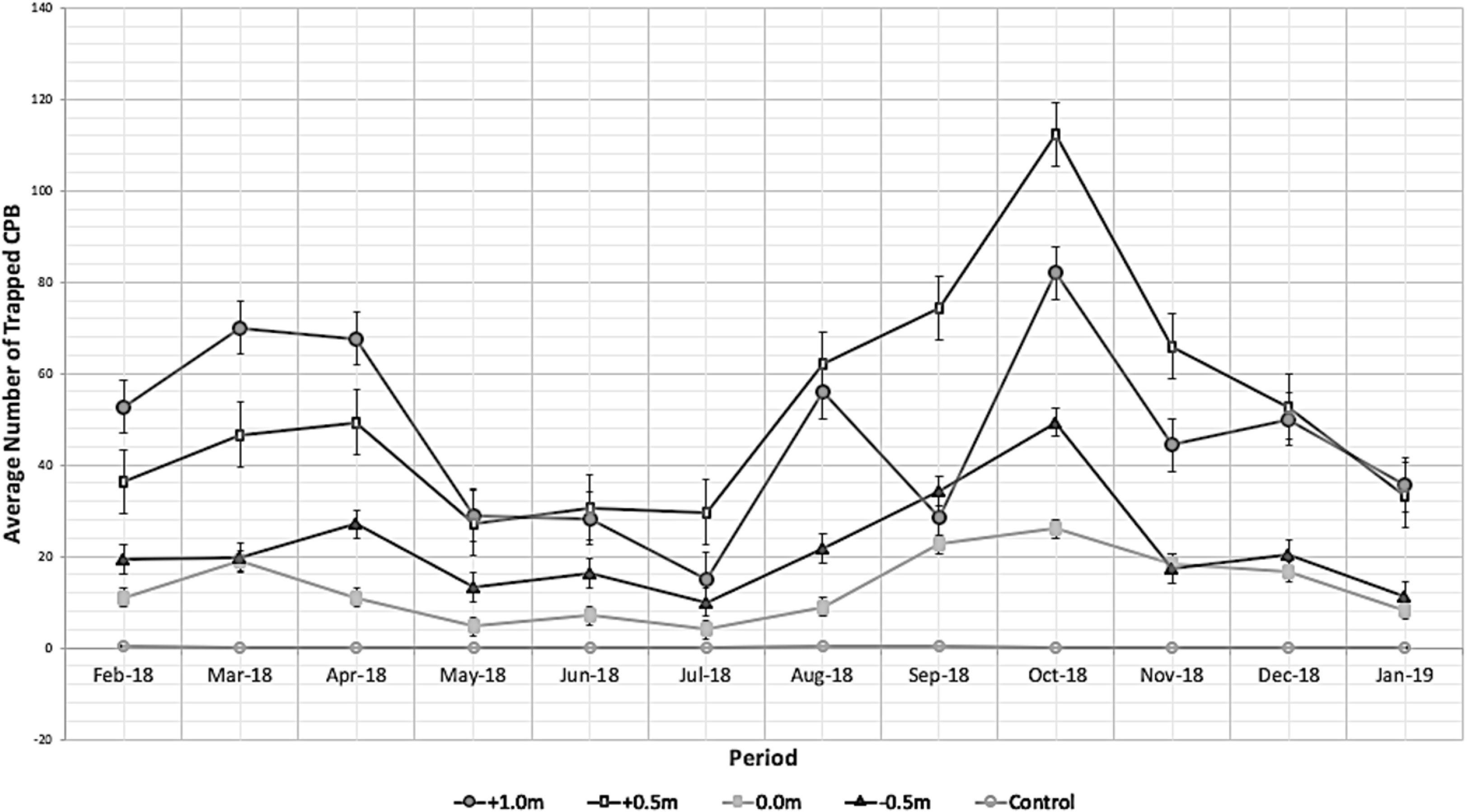
Figure 2 Cacao pod borer (CPB) average trap catches for 12 months at different heights, Sirib, Calinan, Davao City, Philippines, 2018–2019 (Amalin et al., 2020).
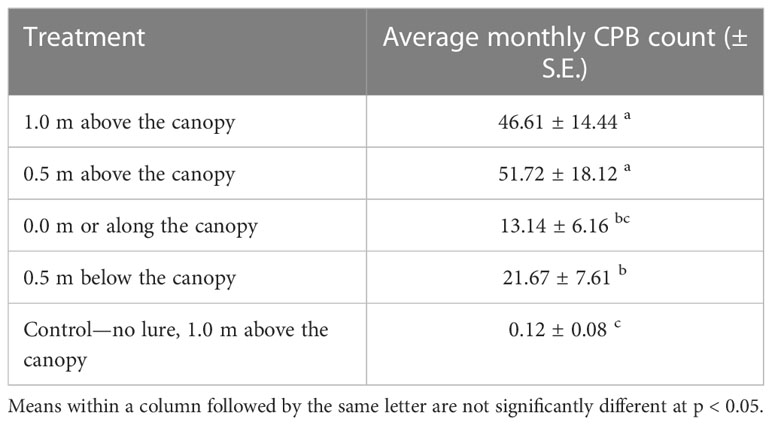
Table 1 Overall average cacao pod borer (CPB) trap catches for the different treatments, Sirib, Calinan, Davao City, Philippines, 2018–2019 (Amalin et al., 2020).
Using the optimum trap height obtained from the first field trial, pheromone activity evaluation in the 9-ha cacao field likewise showed attractiveness of the sex pheromone lure as compared to the blank lures. Male CPB trap catches during the 5-month trapping period also affirm the seasonal abundance observed during the first field trial; the highest trap catch was observed in April 2021, which eventually decreased until July (Figure 3).
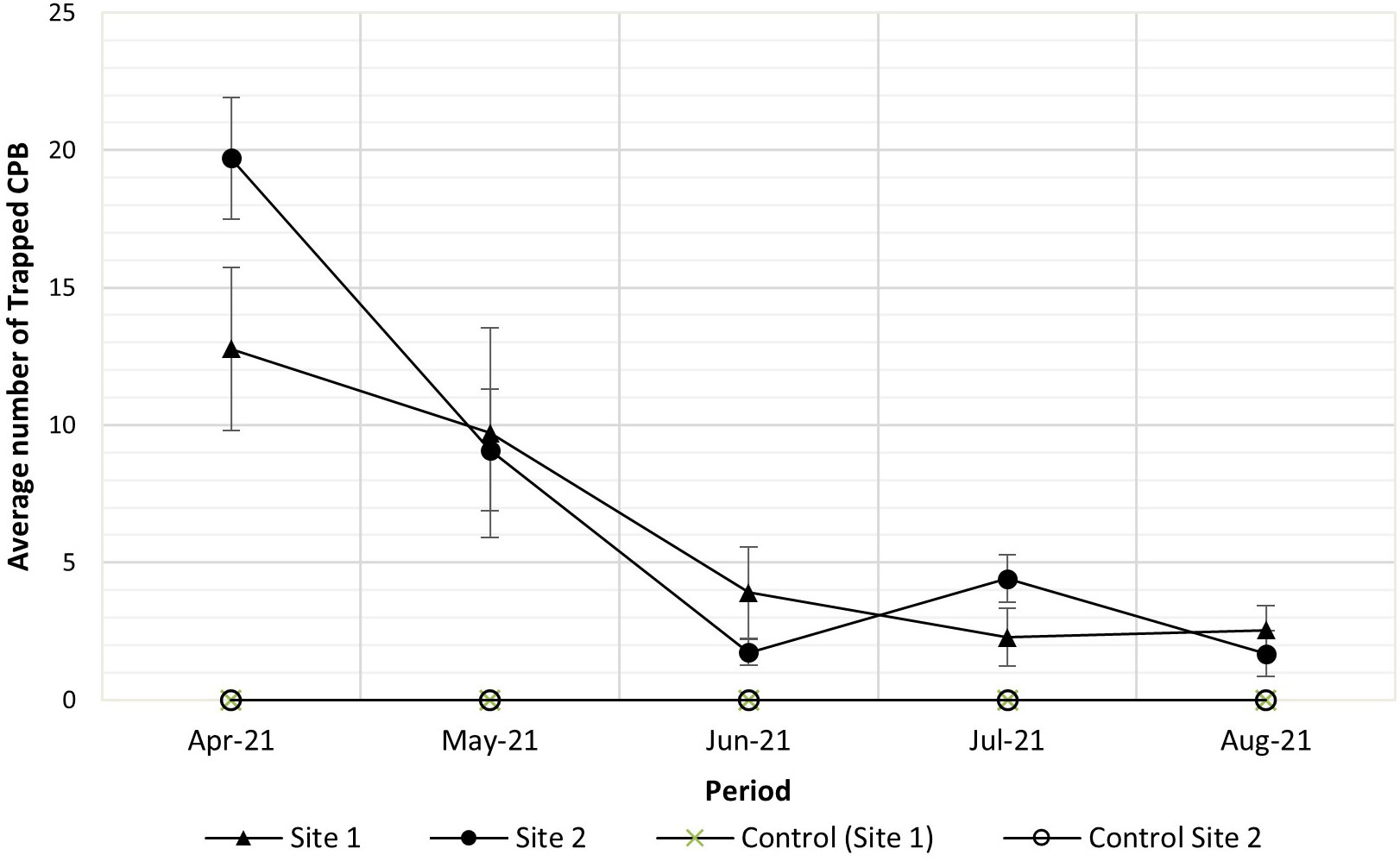
Figure 3 Cacao pod borer (CPB) average trap catches for 5 months (March–July 2021) using traps placed 0.5 m above the tree canopy, Sirib, Calinan, Davao City, Philippines.
4 Discussion
This study showed that the new blend of the synthetic CPB sex pheromones was effective in attracting male CPB in the Philippines. In both field trials, traps baited with sex pheromones attracted large numbers of male CPB while traps with blank lures baited minimal numbers, which can be considered as transient visit and not attraction, since there was no load of pheromone blend in the control lure. Similar attractiveness was observed in Sabah and peninsular Malaysia and in Sumatra and Sulawesi, Indonesia (Zhang et al., 2008; Vanhove et al., 2015; Niogret et al., 2022). The high male CPB trap catches in at least 0.5 m above the canopy confirm that the mating flight behavior of CPB is similar to other moth species. Mating in most moth species occurs when the alluring scent of a female causes a male moth to fly in search of a potential partner and usually mating flight for moth species occurs at dusk (Browne et al., 1969; Kan et al., 2002).
Our result suggests that 0.5 m above the canopy could be the most appropriate placement of the pheromone traps in the cacao field. This reaffirms the earliest report on optimum height placement that has been used in different field trials that followed (Ho et al., 1987). This variation in trap catch as affected by trap position was also supported by the behavior of CPB in the field. It was observed that female CPB prefers to rest higher in the canopy (Niogret et al., 2019), which might result in a higher concentration of sex pheromones above the canopy. As a result, male CPB may have adapted to fly above the canopy in search of the females for mating.
The seasonal abundance pattern of CPB in the field using the monthly male CPB trap catches showed two peaks of population (Figure 2). This abundance pattern coincided with the two harvest seasons per year of cacao. CPB preferred to oviposit on a 3-month-old pod (Ali et al., 2005), which usually occurs from October to November and April to May during the fruit development phase of the cacao plant in the primary peak season and the secondary peak season, respectively. The seasonal abundance was also observed in the second larger field trial in which the highest trap catches were observed in April, which decreased until July. However, average CPB trap catches were lower than those observed during the same period in 2018, which may due to differences in environmental conditions, among other factors.
Weather patterns can affect the behavior of insects. Rainfall, in particular, has been found to be one of two major disturbances that can affect CPB behavior during the quiescent period (Niogret et al., 2019). Based on the modified Coronas classification that groups regions into four climatic types based on average rainfall distribution, the Davao region is classified under Type IV (Iguchi et al., 2018). This climate type is characterized by an even distribution of rainfall throughout the year. This suggests, therefore, that the observed seasonal abundance in the CPB population is likely due to the harvest season rather than rainfall distribution. Nevertheless, further research is necessary to fully elucidate the effects of environmental factors on the behavior of CPB in the field.
The seasonal abundance pattern of male CPB obtained from this study suggests the potential of the sex pheromone trap as a monitoring tool for CPB, which can be the basis for pest management response to CPB. In addition, this trapping system can also significantly contribute in controlling insect population due to the relatively high trap catch of male CPB in the field. A female CPB can produce approximately 60 to 200 eggs on cacao pods, which eventually develop into larvae that burrow into and feed on the pulp (Azhar and Long, 1996; Santoso et al., 2004). Trapping limits the number of mating pairs, which will effectively reduce subsequent populations. Large-scale mass trapping studies (>200 ha) showed that synthetic sex pheromones significantly reduced pod damage due to CPB infestation (Beevor et al., 1993). A follow-up study is underway to verify these results and also to determine the optimum number of traps for 1 ha of cacao farm.
5 Conclusion
Results from this study showed the suitability of the use of pheromone as a component of the pest management program for CPB. The significantly higher male CPB trap catches in the trap with lures loaded with the new CPB pheromone blend compared to the minimal to zero catches in control traps with blank lures showed the attractiveness of the CPB sex pheromone in the field. Determination of the correct height of the CPB trapping tool of at least 0.5 m above the canopy is valuable in maximizing the efficiency of the sex pheromone traps in monitoring and controlling CPB. Additionally, the data on seasonal abundance showed the importance of this pheromone-based method in monitoring and potentially for mass trapping of CPB. These data will help farmers in deciding when would be the proper time to deploy other management strategies. Incorporation with other strategies will allow a more sustainable and efficient IPM system. Further research on the determination of the length of time of the efficacy of the lures in the field and the optimum number of traps in a 1-ha cacao field is needed to maximize or capitalize on the inclusion of the pheromone-based method in the IPM program for CPB in the Philippines.
Data availability statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding author.
Author contributions
All authors listed have made a substantial, direct, and intellectual contribution to the work, and approved it for publication.
Funding
This study was funded by the Department of Science and Technology - Philippine Council for Agriculture, Aquatic, and Natural Resources Research and Development (DOST-PCAARRD), Los Baños, Laguna, Philippines, under the cacao pest management program (biologically based approaches).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Ali N. M., Hassan S. T. S. (2005). Distribution of cocoa pod borer (CPB) Conopomarpha cramerella (Snellen) (Lepidoptera: gracillariidae) egg population with respect to the pod phenology. Pertanika J. Trop. Agric. Sci. 28(1), 49–57.
Alicbusan L. (1935). Beneficial effects on diseased cacao trees of removing infected parts and disinfecting the wounds. Philippine Agr. 23 (10), 891–903.
Amalin D., Arecelo M., Alamrinez B. J., Castillo R., Zhang A. (2020). Biological activity evaluation of the sex pheromone of cocoa pod borer, Conopomorpha cramerella snellen. ACIAR Proc. Ser. 149, 7–10.
Azhar I., Long G. E. (1996). Effect of cocoa pod age on egg distribution and egg parasitism of the cocoa pod borer in Malaysia. Entomol. Experiment. Applicata (Netherlands) 81 (1), 81–89. doi: 10.1111/j.1570-7458.1996.tb02017.x
Beevor P. S., Cork A., Hall D. R., Nesbitt B. F., Day R. K., Mumford J. D. (1986). Components of female sex pheromone of cocoa pod borer moth, Conopomorpha cramerella. J. Chem. Ecol. 12, 1–23. doi: 10.1007/BF01045587
Beevor P. S., Mumford J. D., Shah S., Day R. K., Hall D. R. (1993). Observations on pheromone-baited mass trapping for control of cocoa pod borer, Conopomorpha cramerella, in Sabah, East Malaysia. Crop Protection 12, 134–140. doi: 10.1016/0261-2194(93)90140-E
Browne L. B., Hoo C. F. S., Gerwen A. C. M. V., Sherwell I. R. (1969). Mating flight behaviour in three species of oncopera moths (Lepidoptera: Hepialidae). Aust. J. Entomol 8, 168–172. doi: 10.1111/j.1440-6055.1969.tb00753.x
Cardé R. T., Haynes K. F. (2004). “Structure of the pheromone communication channel in moths,” in Advances in Insect Chemical Ecology (Cambridge: Cambridge University Press), 283–332. doi: 10.1017/CBO9780511542664.009
Gavarra M. R. (1990). Laboratory screening of some insecticides for the control of the cacao pod borer, Conopomorpha cramerella (Snellen). Philippine Entomol. (Philippines) 8(2), 731–736.
Ho S. H., Beevor P. S., Mumford J. D. (1987). “A practical approach to the control of the cocoa pod borer moth using synthetic sex pheromone in an integrated system,” in Management of the cocoa pod borer (Kuala Lumpur, Malaysia: Malaysia Plant Protection Society), 53–68.
Iguchi J. A., Seposo X. T., Honda Y. (2018). Meteorological factors affecting dengue incidence in Davao, Philippines. BMC Public Health 18, 629. doi: 10.1186/s12889-018-5532-4
Kan E., Kitajima H., Hidaka T., Nakashima T., Sato T. (2002). Dusk mating flight in the swift moth, Endoclita excrescens (Butler) (Lepidoptera: hepialidae). Appl. Entomol. Zool. 37, 147–153. doi: 10.1303/aez.2003.147
Limbo R. (2019) Policy support and processes for institutionalizing organic agriculture (OA) in the Philippines, some evidences and challenges. Available at: http://ap.fftc.agnet.org/ap_db.php?id=981&print=1 (Accessed May 6, 2020).
Niogret J., Ekayanti A., Ingram K., Lambert S., Kendra P. E., Alborn H., et al. (2019). Development and behavioral ecology of Conopomorpha cramerella (Lepidoptera: gracillariidae). flen 102, 382–387. doi: 10.1653/024.102.0214
Niogret J., Ekayanti A., Zhang A. (2022). Sex pheromone of cocoa pod borer, Conopomorpha cramerella: field activity evaluation of pheromone formulations in an Indonesia plantation. Insects 13. doi: 10.3390/insects13080663
Philippine Statistics Authority. (2023). Crops Statistics of the Philippines, 2017-2021. Available at: https://psa.gov.ph/content/crops-statistics-philippines-national-and-regional (Accessed April 16, 2023).
Reinking O. A. (1923). Comparative study of Phytophthora faberi on coconut and cacao in the Philippines. J. Agric. Res. 25, 267–284.
Republic of the Philippines (2010) High value crops development act. Available at: https://www.senate.gov.ph/lisdata/96778118!.pdf (Accessed May 6, 2020).
Rizvi S. A. H., George J., Reddy G. V. P., Zeng X., Guerrero A. (2021). Latest developments in insect sex pheromone research and its application in agricultural pest management. Insects 12, 484. doi: 10.3390/insects12060484
Rosmana A., Shepard M., Hebbar P., Mustari A. (2013). Control of cocoa pod borer and phytophthora pod rot using degradable plastic pod sleeves and a nematode, Steinernema carpocapsae. Indones. J. Agric. Sci. 11, 41. doi: 10.21082/ijas.v11n2.2010.41-47
Santoso D., Chaidamsari T., Wiryadiputra S., de Maagd R. A. (2004). Activity of Bacillus thruringiensis toxins against cocoa pod borer larvae. Pest Manage. Sci. 60, 735–738. doi: 10.1002/ps.927
Tandingan I. C., Ampo A. E., Southern M. U (1990). Survey of VSD (Vascular streak dieback) of cacao in southern Mindanao [Philippines] and its control using different pruning frequencies. USM CA Res. J. (Philippines) 1 (2), 114–119.
Vanhove W., Vanhoudt N., Bhanu K. R. M., Abubeker S., Feng Y., Yu M., et al. (2015). Geometric isomers of sex pheromone components do not affect attractancy of Conopomorpha cramerella in cocoa plantations. J. Appl. Entomol. 139, 660–668. doi: 10.1111/jen.12212
Keywords: cacao pod borer, sex pheromone, integrated pest management, field evaluation, Philippines
Citation: Amalin DM, Arcelo M, Almarinez BJM, Castillo RC, Legaspi JC, Santos KLT, Tavera MAA, Janairo JIB and Zhang A (2023) Field evaluation of the sex pheromone of the cacao pod borer (Conopomorpha cramerella Snellen) in the Philippines. Front. Agron. 5:1165299. doi: 10.3389/fagro.2023.1165299
Received: 13 February 2023; Accepted: 28 April 2023;
Published: 19 May 2023.
Edited by:
Anamika Sharma, Virginia Tech, United StatesReviewed by:
Jozsef Kiss, Szent István University, HungaryMirza Abdul Qayyum, Muhammad Nawaz Shareef University of Agriculture, Pakistan
Copyright © 2023 Amalin, Arcelo, Almarinez, Castillo, Legaspi, Santos, Tavera, Janairo and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Divina M. Amalin, ZGl2aW5hLmFtYWxpbkBkbHN1LmVkdS5waA==
 Divina M. Amalin
Divina M. Amalin Mercedes Arcelo3
Mercedes Arcelo3 Jesusa C. Legaspi
Jesusa C. Legaspi Kris Lord T. Santos
Kris Lord T. Santos Jose Isagani B. Janairo
Jose Isagani B. Janairo