- Department of Statistics, Penn State University, University Park, PA, USA
Autism Spectrum Disorder (ASD) occurs more often among males than females in a 4:1 ratio. Among theories used to explain the causes of ASD, the X chromosome and the Y chromosome theories attribute ASD to the X-linked mutation and the male-limited gene expressions on the Y chromosome, respectively. Despite the rationale of the theory, studies have failed to attribute the sex-biased ratio to the significant linkage or association on the regions of interest on X chromosome. We further study the gender biased ratio by examining the possible interaction effects between two genes in the sex chromosomes. We propose a logistic regression model with mixed effects to detect gene–gene interactions on sex chromosomes. We investigated the power and type I error rates of the approach for a range of minor allele frequencies and varying linkage disequilibrium between markers and QTLs. We also evaluated the robustness of the model to population stratification. We applied the model to a trio-family data set with an ASD affected male child to study gene–gene interactions on sex chromosomes.
1. Introduction
Autism Spectrum Disorders (ASDs) refer to a collection of developmental disabilities in social interaction, communication, and behavior. The prevalence of autism and related ASDs is increasing and about 1% of children need education and social care (Baird et al., 2006). A more recent study estimates that the worldwide median autism spectrum disorder prevalence is 62 out of 10,000 (Elsabbagh et al., 2012).
ASD is four times more common in males than in females (Chakrabarti and Fombonne, 2001). The bias could be in part due to the fact that females are less likely to be diagnosed as ASD at the equivalent level of autistic traits in males (Dworzynski et al., 2012). ASD is also environmental and genetic (Persico and Bourgeron, 2006; Matsuzaki et al., 2012). In Baron-Cohen et al. (2011), they summarized three possible factors that may attribute psychological and physiological changes in the male brain: (a) the masculinizing effect of fetal testosterone; (b) X- and Y-linked theories; (c) autosomal penetrance theory.
Intuitively, the sex chromosomes represent a reasonable starting point in order to find the causes of the gender bias in ASD. In fact, studies found that the X chromosome contains genes that are highly expressed in brain tissues compared to other tissues (Nguyen and Disteche, 2006). This supports the important role of the X chromosome in brain functions, which is also evident from the X-linked mental disabilities. There are also Y-linked male-specific genes expressed in human brains, such as SRY, ZFY, and PCDH11R (Mayer et al., 1998; Durand et al., 2006). The association studies of the genes on X or Y chromosomes with ASD include Serajee and AH (2009), Noor et al. (2010), Chung et al. (2011), Kaya et al. (2012). These studies focused on one of the sex chromosomes but not both at the same time.
For family-based association studies, the transmission/disequilibrium test [TDT, Spielman et al. (1993)], its generalizations such as the sib transmission/disequilibrium test [S-TDT, Spielman and Ewens (1998)], and the family-based association test [FBAT, Horvath et al. (2001)] are standard choices for qualitative data. However, they are not specifically designed for detecting interaction effects in genes. In this article, we look for interaction effects between two genes on sex chromosomes in males. No studies have been conducted to determine attribution of the diseases to gene–gene interactions on the sex chromosomes. We will seek to determine if there are gene–gene interactions on sex chromosomes that drive the gender bias in ASD.
Mixed effects models have been widely adopted in a wide range of disciplines. Mixed effects models use both fixed and random effects. Fixed effect parameters represent the average changes in the response variable, while random effects usually represent the subject-to-subject variability. Examples of the latter include batches in a chemical experiment, classrooms in an education setting, and members in a family. Recently, mixed effects models have received a significant attention in genetic association tests that account for the population stratification and the correlation among the individuals (Zhang et al., 2009; Wang et al., 2011; De Lobel et al., 2012; Zhou and Stephens, 2012). Zhou and Stephens (2012) developed a genome-wide efficient mixed-model association (GEMMA), in which the related polygenic effects are treated as a random effect. De Lobel et al. (2012) introduced a mixed effects model that incorporates gene–gene interactions in autosomal chromosomes. These mixed effects models for the association study, with or without gene–gene interaction, are currently designed for quantitative response variables only.
In this article, we apply a generalized linear mixed effects model to handle dichotomous responses and genetic interaction effects. Generalized mixed effects models (Breslow and Clayton, 1993; McCulloch, 1997) are the extensions of generalized linear models (Nelder and Wedderburn, 1972; McCullagh and Nelder, 1989). Generalized linear models are regression models for different response types and the expected value of the response μi is
where g is an invertible link function, xi is the ith observation for fixed effects, and β is fixed effect coefficients. In a generalized linear mixed effects model with two random effects A and E, the expected response is
For a binary response, the logistic link is defined as g(μi) = log(μi/(1 − μi)) and
Here, μi can be the trait probability for the ith individual. In our study, we treat genetic effects, including an interaction effect, as fixed effects, but we further include unlinked autosomal effects as random effects.
2. Materials and Methods
2.1. The Model
We introduce a generalized mixed effects model for the association test on sex chromosome in males. Family-based trio data with an affected son will be considered. Assume that two unlinked additive QTLs are associated with the disease. Our model is written as
where pij is the trait probability for the jth individual in the ith family, β0, β1, β2, and β12 are the regression coefficients, Xij and Yij denote the genotypes at two loci of the jth individual in the ith family and they are either 0 or 2, and XYij is the interaction effect of the two. Let Aij be the random effect due to the unlinked autosomal QTLs and Eij be the environmental random effect. The variance - covariance matrix of the two random effects between the jth and the kth individuals in the ith family is given by
where ϕijk is twice of the kinship coefficient between the jth and the kth individual in the ith family. We assume that the random effects follow a Normal distribution with mean 0 and variance Ωijk. The analysis is conducted using an R package pedigreemm (Vazquez et al., 2010). When we consider a model with a binary response variable and random effects, the full maximum likelihood analysis requires a numerical integration technique. In such case, the package pedigreemm uses the Laplace approximation (Tierney and Kadane, 1986). The fixed effects are estimated based on the iterative re-weighted least squares algorithm (Green, 1987). Under the assumption that the estimates follow a Normal distribution, pedigreemm generates the test statistics z = /s.e() and the corresponding p-values (for two-sided test) under the null hypothesis of no association. Our study results are based on these outputs.
2.2. Simulation Study
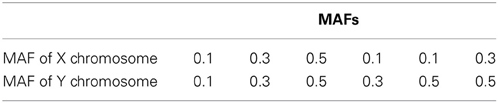
Assume that the two QTLs are unlinked. We generated two markers in linkage disequilibrium (LD) with the two QTLs from D′ = 0 to D' = 1 with an increment of 0.1. For D' = 0, the markers have no LD with QTLs and therefore have no association with the disease. For D' = 1, the markers have complete dependency to QTLs. We assume that QTLs and markers have the same minor allele frequencies (MAFs), which we vary in the simulations at 0.1, 0.3, and 0.5. We set the sample size to be 2000 (1000 families with father and one son in each family) and use σ2a = 0.5 and σ2e = 1.
First, we explain how the correlated random effects within a family are generated. Similarly to De Lobel et al. (2012), the random effects due to unlinked autosomal QTLs are generated as follows:
where AiF and AiO are unlinked autosomal random effects of a father and a son, respectively, in the ith family. This leads to the correlation between AiF and AiO at 0.5 · σ2a.
Second, we need to generate family samples in which the father is unaffected and the son is affected. Initially, we generated a large enough number of samples and selected family samples in which the father is unaffected and the son is affected. The website of the R code for this sampling method is provided in the Supplemental data. The R code generates genotypes with specified MAFs and the two random effects (including Aij as explained above), obtains binary responses based on the trait probabilities, and returns family samples that contain an unaffected father and an affected son.
When the samples are selected under such condition, the sample means of the random effects AiF and AiO can be shifted away from zero. In simulation study, the random effect samples follow a Normal distribution but the two sample means are not equal to zero, which violates the model assumption about the random effect having mean zero. We tested if the discrepancy between sample means and zero affects p-values for testing the significance of interaction effect. We simulated 100 datasets using the parameters in the simulation studies (Tables 1, 2) and found that the discrepancy in means did not affect p-value of the interaction effect (data not shown).
2.2.1. No population stratification
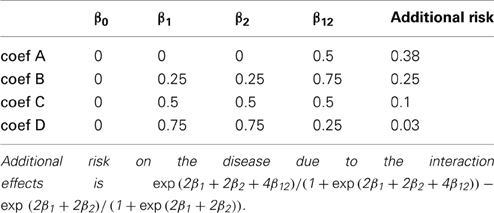
We conducted 24 simulation studies with varying MAFs and regression coefficients β0, β1, β2, and β12. They are summarized in Tables 1, 2, respectively. The regression coefficients in Table 2 represent the following: coef A (β0 = 0, β1 = 0, β2 = 0, and β12= 0.5) has no main effect but an interaction effect, coef B (β0 = 0, β1 = 0.25, β2 = 0.25, and β12= 0.75) has a larger interaction effect, coef C (β0 = 0, β1 = 0.5, β2 = 0.5, and β12= 0.5) has equal main and interaction effects, and coef D (β0 = 0, β1 = 0.75, β2 = 0.75, and β12= 0.25) has larger main effects. We note that the same magnitude of the interaction coefficient does not reflect the same amount of contribution to the disease risk. Despite the fact that coef A and coef C have the same interaction coefficient β12 = 0.5, the increase in risk by adding the interaction effect are about 0.38 and 0.1, respectively. This is due to the fact that the interaction effect occurs on top of the two main effects. When the two main effects are smaller, the increase in disease risk due to the interaction effect becomes larger.
2.2.2. Population stratification
It has been known that population stratification can result in spurious association findings in mixed effects model settings (Abecasis et al., 2000). De Lobel et al. (2012) orthogonalized the genotype scores into within and between-family effects in order to avoid such spurious findings. We study the impact of population stratification on the type I error and power of detecting gene–gene interaction effects using the proposed model.
We consider two populations, each with 1000 individuals. The population stratification can be formulated in three ways in Model (1):
• Different β0: the disease prevalence is different due to other factors
• Different MAFs: MAFs can be different in two populations
• Different β1, β2 and β12: the genotype effects can be different
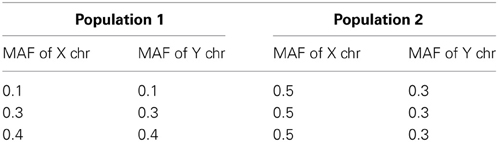
We considered two scenarios: case 1 includes the first two conditions but not the third condition and case 2 includes all three conditions. For case 1, we used β0 = 0 for Population 1 and β0 = 0.2 for Population 2 and the three combinations of MAFs in Table 3. In Population 1, the MAFs of X and Y chromosomes are the same while they are not identical (0.5 and 0.3) in Population 2. The coefficients β1, β2, and β12 are set to be the same for the two populations. For case 2, while keeping the first two conditions the same as those in case 1, we use the regression coefficients from Table 2 for Population 1 and use 0.2 for all the regression coefficients β0, β1, β2, and β12 for Population 2.
2.3. Application to Autism Study
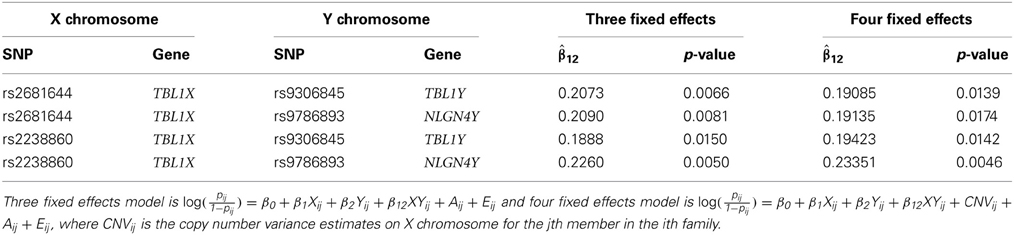
We obtained the parent-offsprings trios data from dbGaP at http://www/ncbi.nlm.nih.gov/gap through dbGaP accession number phs000267.v1.p1. As the interest is on the interaction of genetic variables on the X and Y chromosomes, we selected families who have an affected son. We have a total of 2216 individuals in 1108 families. We focused on 90 and 2 SNPs on the X and Y chromosomes, respectively. The ninety SNPs on the X chromosome are in PTCHD1, TBL1X, and NLGN3, which are candidate genes for autism spectrum disorder (Noor et al., 2010; Chung et al., 2011; Kaya et al., 2012). The two SNPs on Y chromosome are based on Serajee and AH (2009). Out of the six SNPs in Serajee and AH (2009), only three (rs9306845, rs9786893, and rs16980459) are available in dbGaP data. Also, the two genotypes rs9786893 and rs16980459 are identical and hence only two are used in the study. In total 180 tests were conducted. We excluded families whose SNP is missing, and thus the number of families we used for testing varies. The smallest number of families we used is 1077.
3. Results
3.1. Simulation Study
3.1.1. No population stratification
The type I error and power of detecting interaction effects for varying LD, MAFs and regression coefficients are summarized in Figure 1. The MAFs in the top row of Figure 1 are equal in both markers while MAFs are different in the bottom row. Each plot includes the results of four different regression coefficients listed in Table 2 varying D′ from 0 to 1.
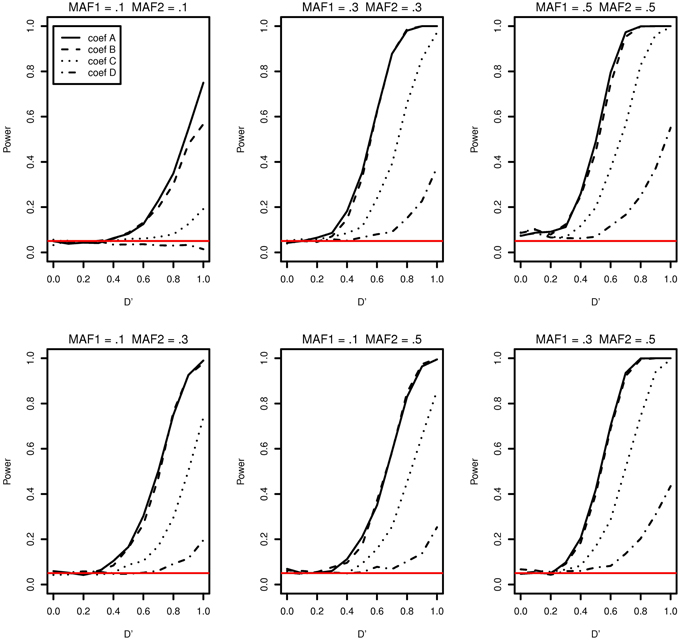
Figure 1. Type I error and power of detecting interaction effects with different MAFs and four regression coefficients. For all four β0 = 0, coef A: β1 = 0, β2 = 0, and β12 = 0.5, coef B: β1 = 0.25, β2 = 0.25, and β12 = 0.75, coef C: β1 = 0.5, β2 = 0.5, and β12 = 0.5, coef D: β1 = 0.75, β2 = 0.75, and β12 = 0.25, and σ2a = 0.5 and σ2e = 1, when the sample size is 2000 (1000 families). The red horizontal line indicates the 0.05 significance level.
The power of detecting interaction effects is significantly affected by the MAF values. This can be best shown by comparing the first and the last plots in the top row. When MAFs are 0.1 on two markers, the maximum power to detect the interaction effect is 0.75 for coef A when LD is equal to 1. Meanwhile, when MAFs are both 0.5, the power is about 0.8 for coef A when LD is larger than 0.6. The power is associated with the number of nonzero interaction genotypes. The expected number of nonzero genotypes in the interaction is only 20 when both MAFs are 0.1, which is 1% of the sample size 2000. This may not be large enough number to be able to detect the interaction effects at the power level 0.8.
Within each plot, it is apparent that the power increases as the additional risk due to the interaction effect increases (see the last column in Table 2). The power of detecting interaction effects is generally 0.8 or larger when D' is 1 in most of the cases, excluding coef D in all plots and all regression coefficients combinations in MAF1 = 0.1 and MAF2 = 0.1. The proposed model detects interaction effects at a power greater than 0.8 (D' = 1) when their risk is as low as 0.1 with MAFs larger than 0.1.
The power in general increases as D' increases. However, there are two exceptions; first, when coef D (MAF1 = 0.1 and MAF2 = 0.1) and when D' is less than 0.4 (MAF1 = 0.5 and MAF2 = 0.5). In the former, both the number of nonzero genotypes in interaction effect and the additional risk due to interaction effect are not large enough to be detected by the proposed model. When the sample size is 5000, the decreasing trend in power disappears, although the power is still as low as 0.1 at D' = 1 (See Supplementary Data: Supplemental Figure 1). In the latter, the type I errors in the third plot (MAF1 = 0.5 and MAF2 = 0.5) in Figure 1 are 0.073, 0.086, 0.088, 0.075 for coef A to coef D, respectively, and they are larger than expected. The 95% confidence intervals are (0.057, 0.089), (0.068,0.104), (0.070, 0.106), and (0.058, 0.092), respectively. These slightly inflated type I errors do not seem to appear when the sample size is 5000, in which type I errors are 0.054, 0.064, 0.078, and 0.063, respectively. The type I error for coef C is the only one that is statistically significantly larger than expected. The 95% confidence interval is (0.061,0.0950). We suspect that these two exceptional trends are due to the lack of information by relatively small sample size.
Testing interaction effects is reliable using the proposed model excluding some cases when the additional risk due to interaction effects are insignificant or when the number of nonzero interaction genotypes is quite small. On the other hand, we found that the variance estimates, and in Equation (2), are heavily biased and close to zero in the simulation studies. Consequently, the fixed effect estimates are identical or close to those of a logistic regression model in the simulation study settings. While the generalized linear mixed effects model is computationally more demanding, it can accommodate more general family structures.
As an example, we compared the type I error and the power of the two models in Figure 2 when there are a father and four male siblings in a family. Figure 2 contains only two cases coef A and coef D for a demonstration. There is no significant differences in power between two models for coef D (gray curves). The two black plots in Figure 2 are well separated and this shows that the generalized linear mixed effects model performs better in detecting interaction effects for coef A for all MAFs. The discrepancy in power for coef A at D' = 1 is as high as 0.080 in MAF1 = 0.3 and MAF2 = 0.3. In addition, the generalized linear mixed effects model is more suitable to incorporate the genetic correlation among family members even if it is computationally more expensive.
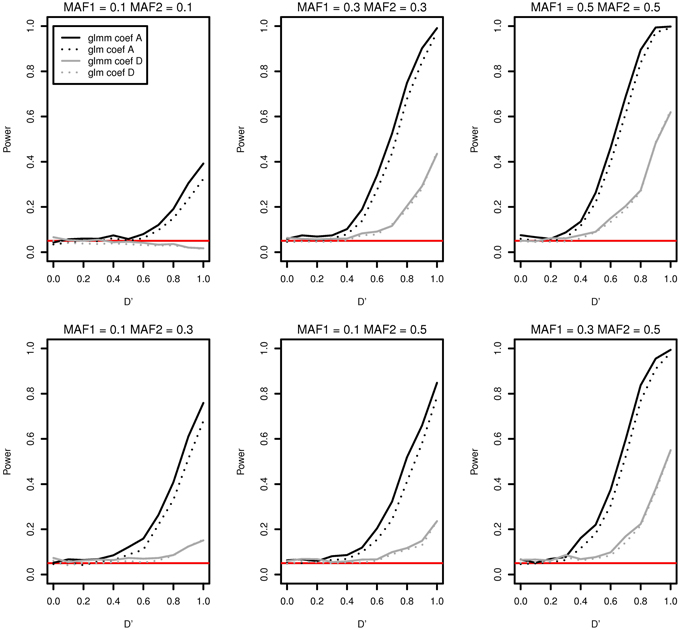
Figure 2. Type I error and power of detecting interaction effects with different MAFs using a generalized linear mixed effects model (solid) and a generalized linear model (dotted) when samples with five family members are randomly generated. Two regression coefficients are β0 = 0, coef A: β1 = 0, β2 = 0, and β12 = 0.5 (shown in black), coef D: β1 = 0.75, β2 = 0.75, and β12 = 0.25 (shown in gray), σ2a = 10 and σ2e = 0.01, and the sample size is 2000 (400 families). The red horizontal line indicates the 0.05 significance level.
Breslow and Clayton (1993) illustrated that generalized mixed effects model estimates are biased when applied to clustered binary data, and therefore we anticipated that the model estimates would be biased. Table 4 shows the median interaction effect estimates and the 95% confidence intervals at D' = 0, 0.5, and 1 when the two MAFs are equal to each other. As expected, the estimated parameters are underestimated in all cases. Regardless, the power of detecting interaction effects is larger than 0.8 at D' ≥ 0.8 excluding when coef D and when MAFs are both 0.1. See Figure 1. Based on the performance of detecting interaction effects, the proposed model is suitable to detect interaction effects in the simulation study settings.
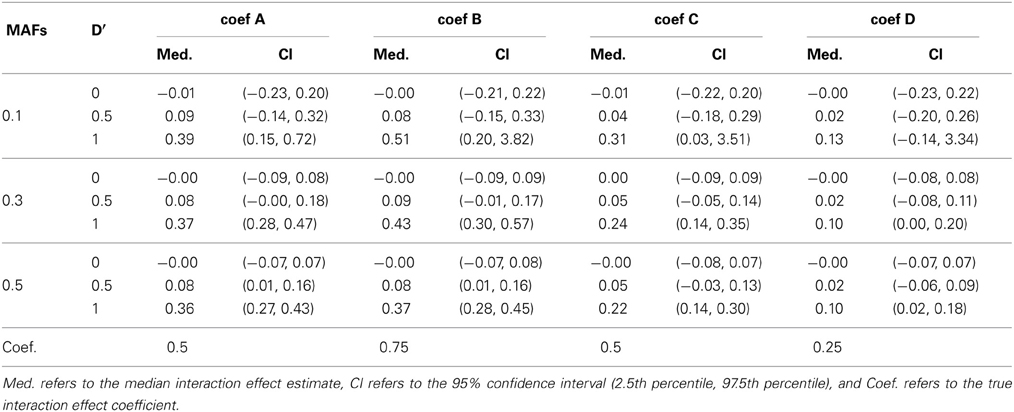
Table 4. The median and 95% confidence interval of the interaction coefficient estimates at D' = 0, 0.5, and 1 when the two MAFs are the same.
3.1.2. Population stratification
The simulation study results of type I error and power of detecting interaction effects when population stratification is present are shown in Figure 3. The plots in the top row correspond to case 1 and the ones in the bottom correspond to case 2. In both cases, the type I error rates are consistent around 0.05 and no spurious false positives are found. In both case 1 and case 2, the increase in power is mainly affected by the increase in MAFs: the larger MAFs the better power among the plots in both top and bottom rows. As explained earlier, this is related to the expected numbers of nonzero interaction genotypes. They are 160 [(0.1 × 0.1 + 0.5 × 0.3)/2 × 2000], 240, and 310, for the first, the second and the third columns, respectively in Figure 3.
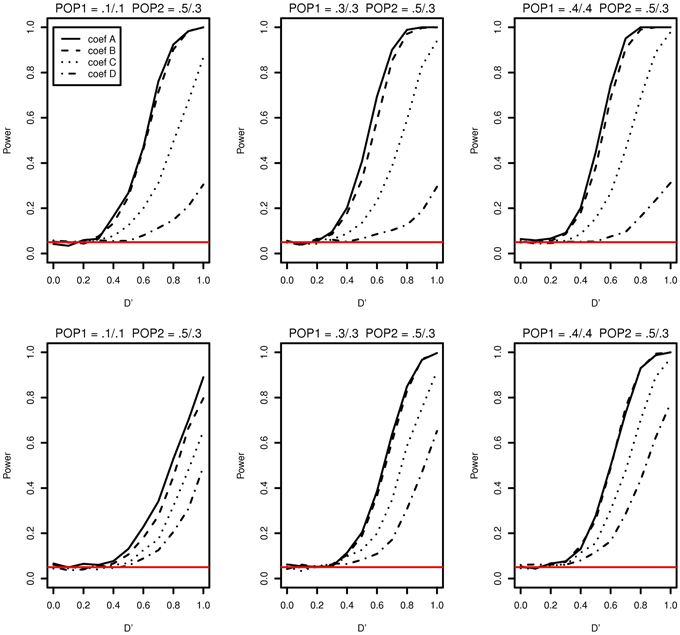
Figure 3. Type I error and power of detecting interaction effects with different MAFs when population stratification is present with the equal sample size 1000 (500 families) in two populations. Plots in the top row: β0 = 0 for Population 1 and β0 = 0.2 for Population 2 sharing the same regression coefficients coef A: β1 = 0, β2 = 0, and β12 = 0.5, coef B: β1 = 0.25, β2 = 0.25, and β12 = 0.75, coef C: β1 = 0.5, β2 = 0.5, and β12 = 0.5, coef D: β1 = 0.75, β2 = 0.75, and β12 = 0.25, and σ2a = 0.5 and σ2e = 1. Plots in the bottom row: β0 = 0 for Population 1 with the regression coefficients as above and β0 = β1 = β2 = β12 = 0.2 for Population 2, and σ2a = 0.5 and σ2e = 1 for both populations. The red horizontal line indicates the 0.05 significance level.
If it were not known that there is population stratification, the first simulation setting (MAF1 = 0.1 and MAF2 = 0.1 in Population 1 and MAF1 = 0.5 and MAF2 = 0.3 in Population 2) can be considered as MAF1 = 0.3 and MAF2 = 0.2 with no population stratification. And this is equivalent to MAF1 = 0.2 and MAF2 = 0.3. Any loss in the power due to population stratification in case 1 can be revealed by comparing the power with the same MAFs with no population stratification. The type I error and the power of the first plot in Figure 3 can be compared with those in the first in the bottom (MAF1 = 0.1 and MAF2 = 0.3) and the middle one in the top (MAF1 = 0.3 and MAF2 = 0.3) in Figure 1. The power lies in the middle of the increasing trend as MAFs increase. Likewise, the middle plot in the top row in Figure 3, considered equivalent to MAF1 = 0.3 and MAF2 = 0.4, can be compared to the middle on the top (MAF1 = 0.3 and MAF2 = 0.3) and the last (MAF1 = 0.3 and MAF2 = 0.5) in Figure 1. This also does not indicate any loss in power due to population stratification with different MAFs in two populations.
The possible loss in the power when the main and interaction effects on the disease are different in two populations can be discovered by comparing powers in the top and ones in the bottom in Figure 3. In the top row of Figure 3, the main and interaction effects in two populations vary simultaneously as in Table 2, and in the bottom row, we replaced the regression coefficients to β0 = β1 = β2 = β12 = 0.2 in Population 2. In Population 2, the additional risk caused by the interaction effect is 0.1271, which is smaller than those in coef A and coef B and larger than coef C and coef D in Table 2. This is reflected in a decrease in power for coef A, coef B, and an increase in power for coef D, when we compare plots in the top and the ones in the bottom in Figure 3. For coef C, the change in power due to the different genetic effects seems to be affected by MAFs: the power decreases in the first plot in the bottom but stays constant in the second and the third plots, relative to the ones in the top in Figure 3.
In both case 1 and case 2, there is no spurious interaction effect detected. The impact of population stratification on the power of detecting the interaction effect using Model (1) has not found in the simulation study in neither case 1 nor case 2.
3.2. Application to Autism Study
Our interest lies in the presence of the interaction between two SNPs and the statistical test will be focused only on the interaction effects. The chance of falsely rejecting the null hypothesis (type I error) becomes larger when we conduct multiple statistical tests simultaneously for a given significance level α. The Bonferroni correction is a simple but conservative approach to correct this. Instead, we used a permutation test in order to find an adjusted type I error. In general, all the variables are permuted together to generate null datasets. In our case, however, family members share the same value in Y chromosome. In other words, the father and the son in a family have the same genotype on Y chromosome. Due to this constraints, we permute the two genetic variables separately. The fathers' genotype scores are permuted and the offsprings are given the same value of the father's. After 1000 repetitions, we found the adjusted significance level α' = 0.001 such that less than or equal to 50 (out of 1000) repetitions include at least one statistically significant interaction effect. Using α' = 0.001, we found no statistically significant interaction effect. The two SNPs on X chromosome, rs2681644 and rs2238860 that have the smallest p-values are shown on Table 5. Both are located on the intron region of TBL1X.
An advantage of regression models over a contingency table is the capability to include additional variables of information flexibly. We added an additional information on copy number variation (CNV) on X chromosome to the logistic regression mixed effects model. The additional variable CNV is obtained after processing the raw CNVs using an R package DNAcopy version 1.30.0 (Olshen et al., 2004). In this study, the variable CNV on the genes of our interest do not statistically significantly affect on the autism risk. The regression coefficients of interaction when CNV is included are given in the last two columns in Table 5.
4. Discussion
We applied a logistic regression model with mixed effects to detect gene–gene interactions on the sex chromosomes in trio data. Especially, only males who have both X and Y chromosomes are included for the study. In our study, we used binary response and explanatory variables and associated the potential correlation among family members using random effects. From the simulation studies, we find that the proposed model detected interaction effects at a power greater than 0.8 (D' = 1) when the risk due to them is as low as 0.1 and MAFs are larger than 0.1. It is robust to population stratification and there is no increase in type I error rate.
Family-based association study data consist of families in which at least a member is affected. Logistic regression models were studied for such data using conditional likelihood on all other possible cases that the specific number of family members are affected. This is referred to as ascertainment adjustment (Burton et al., 2000). In our case, a father is considered as a control and a son as a case in a family. Regarding this as one control and one case study in a family, referred to as 1-1 matched, the conditional likelihood estimate can be obtained by setting the intercept equal to 0, the new variables defined as X*i = XiO − XiF, and all the response variable set to 1 (Hosmer and Lemeshow, 1989, Chapter 7). A brief explanation on the background is provided in the Supplemental data. The type I error and power of the 1-1 matched logistic model are presented in Figure 4 for the MAFs and regression coefficients used in the no population stratification simulation study. The 1-1 matched logistic model performs slightly better in detecting the existing interaction effects when both MAF1 and MAF2 are 0.1. However, the logistic regression model with mixed effects performs better in power in all remaining cases. We suspect that the reason is in that the 1-1 matched logistic regression model infers the interaction effect of the population while the logistic model with mixed effect infers the interaction effects within the samples.
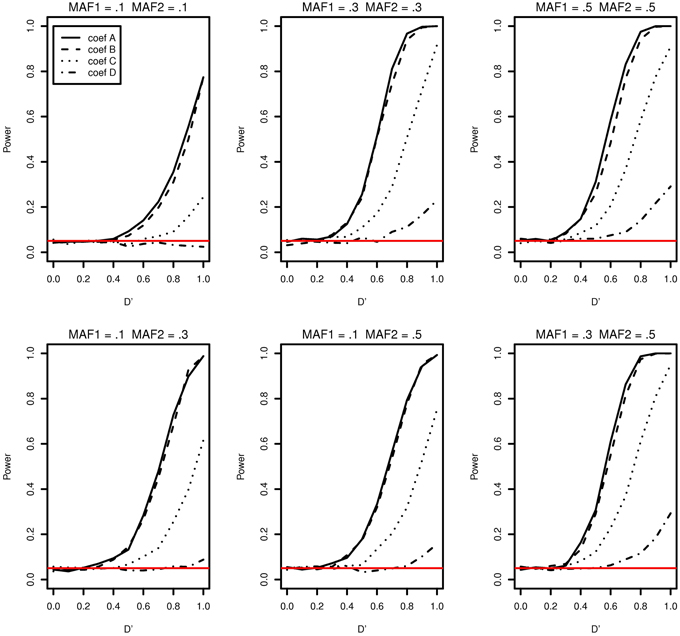
Figure 4. Type I error and power of detecting interaction effects with different MAFs and four regression coefficients using an 1-1 matched logistic regression model with no population stratification. For all four β0 = 0, coef A: β1 = 0, β2 = 0, and β12 = 0.5, coef B: β1 = 0.25, β2 = 0.25, and β12 = 0.75, coef C: β1 = 0.5, β2 = 0.5, and β12 = 0.5, coef D: β1 = 0.75, β2 = 0.75, and β12 = 0.25, and σ2a = 0.5 and σ2e = 1, and the sample size is 2000 (1000 families). The red horizontal line indicates the 0.05 significance level.
While the proposed model works well for detecting interaction effects and performs better in power compared to the 1-1 matched logistic regression model in the most of our simulation settings, the model underestimates the interaction effects. Therefore, the logistic regression model with mixed effects is not appropriate when the interest is in the parameter estimates or the true disease risks due to interaction effects.
Author Contributions
Yeonok Lee, Debashis Ghosh, and Yu Zhang designed the project. Yeonok Lee performed the data analysis and wrote the paper; Debashis Ghosh and Yu Zhang read and approved the final manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
Funding
The project was supported by grants NIH CA129102 and R01 HG004718. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. Publication of this article was supported by NIH R01HG004718.
Supplementary Material
The Supplementary Material for this article can be found online at: http://www.frontiersin.org/journal/10.3389/fgene.2013.00239/abstract
References
Abecasis, G., Cardon, L., and Cookson, W. (2000). A general test of association for quantitative traits in nuclear families. Am. J. Hum. Genet. 66, 279–292. doi: 10.1086/302698
Baird, G., Simonoff, E., Pickles, A., Chandler, S., Loucas, T., Meldrum, D., et al. (2006). Prevalence of disorders of the autism spectrum in a population cohort of children in South Thames: the Special Needs and Autism Project (SNAP). Lancet 368, 210–215. doi: 10.1016/S0140-6736(06)69041-7
Baron-Cohen, S., Lombardo, M. V., Auyeung, B., Ashwin, E., Chakrabarti, B., and Knickmeyer, R. (2011). Why are autism spectrum conditions more prevalent in males? PLoS Biol. 9:e1001081. doi: 10.1371/journal.pbio.1001081
Breslow, N. E., and Clayton, D. G. (1993). Approximate inference in generalized linear mixed models. J. Am. Stat. Assoc. 88, 9–25.
Burton, P. R., Palmer, L. J., Jacobs, K., Keen, K. J., Olson, J. M., and Elston, R. C. (2000). Ascertainment adjustment: Where does it take us? Am. J. Hum. Genet. 67, 1505–1514. doi: 10.1086/316899
Chakrabarti, S., and Fombonne, E. (2001). Pervasive developmental disorders in preschool children. JAMA 285, 3093–3099. doi: 10.1001/jama.285.24.3093
Chung, R.-H., Ma, D., Wang, K., Hedges, D., Jaworski, J., Gilbert, J., et al. (2011). An X chromosome-wide association study in autism families identifies TBL1X as a novel autism spectrum disorder candidate gene in males. Mol. Autism 2:18. doi: 10.1186/2040-2392-2-18
De Lobel, L., Thijs, L., Kouznetsova, T., Staessen, J. A., and Van Steen, K. (2012). A family-based association test to detect gene-gene interactions in the presence of linkage. Eur. J. Hum. Genet. 20, 973–980. doi: 10.1038/ejhg.2012.45
Durand, C. M., Kappeler, C., Betancur, C., Delorme, R., Quach, H., Goubran-Botros, H., et al. (2006). Expression and genetic variability of PCDH11Y, a gene specific to Homo sapiens and candidate for susceptibility to psychiatric disorders. Am. J. Med. Genet. B Neuropsychiatr. Genet. 141B, 67–70. doi: 10.1002/ajmg.b.30229
Dworzynski, K., Ronald, A., Bolton, P., and Happé F. (2012). How different are girls and boys above and below the diagnostic threshold for autism spectrum disorders? J. Am. Acad. Child Psy. 51, 788–797. doi: 10.1016/j.jaac.2012.05.018
Elsabbagh, M., Divan, G., Koh, Y.-J., Kim, Y. S., Kauchali, S., Marcín, C., et al. (2012). Global prevalence of autism and other pervasive developmental disorders. Autism Res. 5, 160–179. doi: 10.1002/aur.239
Green, P. J. (1987). Penalized likelihood for general semi-parametric regression models. Int. Stat. Rev. 55, 245–259. doi: 10.2307/1403404
Horvath, S., Xu, X., and Laird, N. M. (2001). The family based association test method: strategies for studying general genotype–phenotype associations. Eur. J. Hum. Genet. 9, 301–306. doi: 10.1038/sj.ejhg.5200625
Hosmer, D. W., and Lemeshow, S. (1989). Applied Logistic Regression. New York, NY: A Wiley-Interscience publication.
Kaya, N., Colak, D., Albakheet, A., Al-Owain, M., Abu-Dheim, N., Al-Younes, B., et al. (2012). A novel X-linked disorder with developmental delay and autistic features. Ann. Neurol. 71, 498–508. doi: 10.1002/ana.22673
Matsuzaki, H., Iwata, K., Manabe, T., and Mori, N. (2012). Triggers for autism, genetic and environmental factors. J. Central Nervous Syst. Dis. 4, 27–36. doi: 10.4137/JCNSD.S9058
Mayer, A., Lahr, G., Swaab, D. F., Pilgrim, C., and Reisert, I. (1998). The Y-chromosomal genes SRY and ZFY are transcribed in adult human brain. Neurogenetics 1, 281–288. doi: 10.1007/s100480050042
McCullagh, P., and Nelder, J. (1989). Generalized Linear Models. Monographs on statistics and applied probability. Boca Raton, FL: Chapman and Hall.
McCulloch, C. E. (1997). Maximum likelihood algorithms for generalized linear mixed models. J. Am. Stat. Assoc. 92, 162–170. doi: 10.1080/01621459.1997.10473613
Nelder, J. A., and Wedderburn, R. W. M. (1972). Generalized Linear Models. J. R. Stat. Soc. Rer. A-G 135, 370–384. doi: 10.2307/2344614
Nguyen, D. K., and Disteche, C. M. (2006). High expression of the mammalian X chromosome in brain. Brain Res. 1126, 46–49. doi: 10.1016/j.brainres.2006.08.053
Noor, A., Whibley, A., Marshall, C. R., Gianakopoulos, P. J., Piton, A., Carson, A. R., et al. (2010). Disruption at the PTCHD1 locus on Xp22.11 in autism spectrum disorder and intellectual disability. Sci. Trans. Med. 2:49ra68. doi: 10.1126/scitranslmed.3001267
Olshen, A. B., Venkatraman, E. S., Lucito, R., and Wigler, M. (2004). Circular binary segmentation for the analysis of array-based DNA copy number data. Biostatistics 5, 557–572. doi: 10.1093/biostatistics/kxh008
Persico, A. M., and Bourgeron, T. (2006). Searching for ways out of the autism maze: genetic, epigenetic and environmental clues. Trends Neurosci. 29, 349–358. doi: 10.1016/j.tins.2006.05.010
Serajee, F. J. and AH, M. H. (2009). Association of Y chromosome haplotypes with autism. J. Child Neurol. 24, 1258–1261. doi: 10.1177/0883073809333530
Spielman, R. S., and Ewens, W. J. (1998). A sibship test for linkage in the presence of association: the sib transmission/disequilibrium test. Am. J. Hum. Genet. 62, 450–458. doi: 10.1086/301714
Spielman, R. S., McGinnis, R. E., and Ewens, W. J. (1993). Transmission test for linkage disequilibrium: the insulin gene region and insulin-dependent diabetes mellitus (IDDM). Am. J. Hum. Genet. 52, 506–516.
Tierney, L., and Kadane, J. B. (1986). Accurate approximations for posterior moments and marginal densities. J. Am. Stat. Assoc. 81, 82–86. doi: 10.1080/01621459.1986.10478240
Vazquez, A. I., Bates, D. M., Rosa, G. J. M., Gianola, D., and Weigel, K. A. (2010). Technical note: an R package for fitting generalized linear mixed models in animal breeding. J. Anim. Sci. 88, 497–504. doi: 10.2527/jas.2009-1952
Wang, L., Jia, P., Wolfinger, R. D., Chen, X., Grayson, B. L., Aune, T. M., et al. (2011). An efficient hierarchical generalized linear mixed model for pathway analysis of genome-wide association studies. Bioinformatics 27, 686–692. doi: 10.1093/bioinformatics/btq728
Zhang, L., Martin, E. R., Morris, R. W., and Li, Y.-J. (2009). Association test for X-linked QTL in family-based designs. Am. J. Hum. Genet. 84, 431–444. doi: 10.1016/j.ajhg.2009.02.010
Keywords: binary traits, gene–gene interaction, generalized linear mixed effect model, logistic model, trio data, sex chromosomes
Citation: Lee Y, Ghosh D and Zhang Y (2013) Association testing to detect gene–gene interactions on sex chromosomes in trio data. Front. Genet. 4:239. doi: 10.3389/fgene.2013.00239
Received: 31 July 2013; Accepted: 24 October 2013;
Published online: 13 November 2013.
Edited by:
Xuefeng Wang, Harvard University, USACopyright © 2013 Lee, Ghosh and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Debashis Ghosh and Yu Zhang, Department of Statistics, Penn State University, University Park, PA 16802, USA e-mail:Z2hvc2hkQHBzdS5lZHU=;eXV6aGFuZ0BzdGF0LnBzdS5lZHU=
 Yeonok Lee
Yeonok Lee Debashis Ghosh
Debashis Ghosh Yu Zhang
Yu Zhang