- 1Center for Healthcare Research in Pediatrics, Department of Population Medicine, Harvard Pilgrim Health Care Institute, Boston, MA, United States
- 2Department of Population Medicine, Harvard Medical School, Boston, MA, United States
- 3Broad Institute of MIT and Harvard, Cambridge, MA, United States
- 4Sanford Health Imagenetics, Sioux Falls, SD, United States
- 5Department of Medicine, Brigham and Women’s Hospital, Boston, MA, United States
- 6Ariadne Labs, Boston, MA, United States
- 7Department of Medicine, Harvard Medical School, Boston, MA, United States
- 8National Cancer Institute, Bethesda, MD, United States
- 9Department of Bioethics, National Institutes of Health, Bethesda, MD, United States
- 10Sanford Health Imagenetics, Bemidji, MN, United States
- 11Mayo Clinic Genomics Laboratory, Rochester, MN, United States
- 12Sanford Health Imagenetics, Fargo, ND, United States
- 13Department of Pharmacy Practice, North Dakota State University, Fargo, ND, United States
- 14Department of Pathology, University of Utah School of Medicine, Salt Lake City, UT, United States
- 15ARUP Laboratories, Salt Lake City, UT, United States
- 16Sanford School of Medicine, University of South Dakota, Sioux Falls, SD, United States
Genetic testing has the potential to revolutionize primary care, but few health systems have developed the infrastructure to support precision population medicine applications or attempted to evaluate its impact on patient and provider outcomes. In 2018, Sanford Health, the nation’s largest rural nonprofit health care system, began offering genetic testing to its primary care patients. To date, more than 11,000 patients have participated in the Sanford Chip Program, over 90% of whom have been identified with at least one informative pharmacogenomic variant, and about 1.5% of whom have been identified with a medically actionable predisposition for disease. This manuscript describes the rationale for offering the Sanford Chip, the programs and infrastructure implemented to support it, and evolving plans for research to evaluate its real-world impact.
Introduction
Specialists are increasingly using genetic testing to accelerate diagnoses and improve treatment decisions after patients become sick. However, its true potential may be realized in unselected populations and preventive applications. Pharmacogenomic (PGx) information can be stored until a time of need, thereby avoiding potential life-threatening delays associated with reactive testing (O’Donnell et al., 2014; Relling and Evans, 2015; Weitzel et al., 2017). Patients could additionally be screened for inherited predispositions for conditions with effective preventive and early detection interventions that would otherwise remain unknown until disease onset (Amendola et al., 2015; Olfson et al., 2015; Kalia et al., 2017). Collaborative resources such as the Clinical Pharmacogenetics Implementation Consortium (CPIC), PharmGKB and ClinGen have emerged to develop and aggregate guidelines for genetic information, including its use in unselected populations (Klein et al., 2001; Relling and Klein, 2011; Rehm et al., 2015). Enthusiasm is growing for genomics as a component of precision population medicine, where disease treatment and prevention efforts for all patients are tailored to individuals’ genes, environments, and lifestyles (Goldenberg et al., 2013; Waisbren et al., 2016).
Many commentators anticipate a future in which such uses of genetic testing is standard of care for all patients, including primary care (Collins et al., 2003; Green et al., 2013b; McCarthy et al., 2013; Bell, 2017). Research shows that non-genetic specialist providers are receptive to integrating genetic testing into their practices when appropriately supported (Overby et al., 2014; Raghavan and Vassy, 2014), and that its results often satisfy the informational needs of patients (Roberts et al., 2018). Access to direct-to-consumer genetic testing (Allyse et al., 2018) and third-party services to re-interpret existing genomic data (Wang et al., 2018) is growing. Early projections also suggest that precision population medicine applications may be cost-effective (Bennette et al., 2015; Prince et al., 2017; Zhang et al., 2019). Yet, the challenges of integrating genetics into everyday patient care are daunting. The demands for specialized resources and trained personnel are considerable (Shuldiner et al., 2013; Hoskovec et al., 2018; Ginsburg et al., 2019), as most generalists, advanced practice providers, and nurses have had limited exposure to medical genetics and genomics during their training (Wolyniak et al., 2015; Campion et al., 2019). Few health systems have developed the infrastructure to store genomic data and facilitate clinical decision making (Kho et al., 2013; Williams et al., 2019). Moreover, limited evidence exists about the benefits and risks of population genetic testing, particularly within real-world clinical settings (Carey et al., 2016; Vassy et al., 2017; Manickam et al., 2018; Brothers et al., 2019; Stark et al., 2019). Further data about the impact of genetic testing applied in precision population medicine is critical to justify its implementation at a larger scale (Messner et al., 2017; Shaer et al., 2017).
In 2018, the Sanford Imagenetics Initiative began offering the Sanford Chip, a clinical laboratory-developed test that provides PGx information, as well as optional disease risk information, to adult patients across the Sanford Health system as part of primary care. This manuscript summarizes the environment and processes the Imagenetics Initiative developed to support and evaluate the Sanford Chip Program. The aim of this report is to offer an instructive example for implementing a precision population medicine program that emphasizes genomics (Rehm et al., 2015; National Cancer Institute, 2019; U.S. National Library of Medicine, 2020).
The Sanford Health Imagenetics Initiative
Sanford Health is the largest rural non-profit health system in the United States, serving more than 2 million patients through 46 medical centers, more than 200 clinics and 2,500 providers. Through strategic planning which included primary care providers (PCPs), genetics providers, pharmacists, and health system administrators initiated in 2012, Sanford Health identified that genomics should have an emerging role in the care of its patients. With a vision of the expansive role genetic testing could play in all aspects of medicine, Sanford Health developed an extensive plan for an “Imagenetics” (internal medicine and genetics) Initiative. With help from $125 million in external donations to fund this vision, Sanford Health launched the Imagenetics Initiative in 2014 with the goal of accelerating the implementation of genetic testing into primary care. Key milestones for the Imagenetics Initiative are summarized in Figure 1.
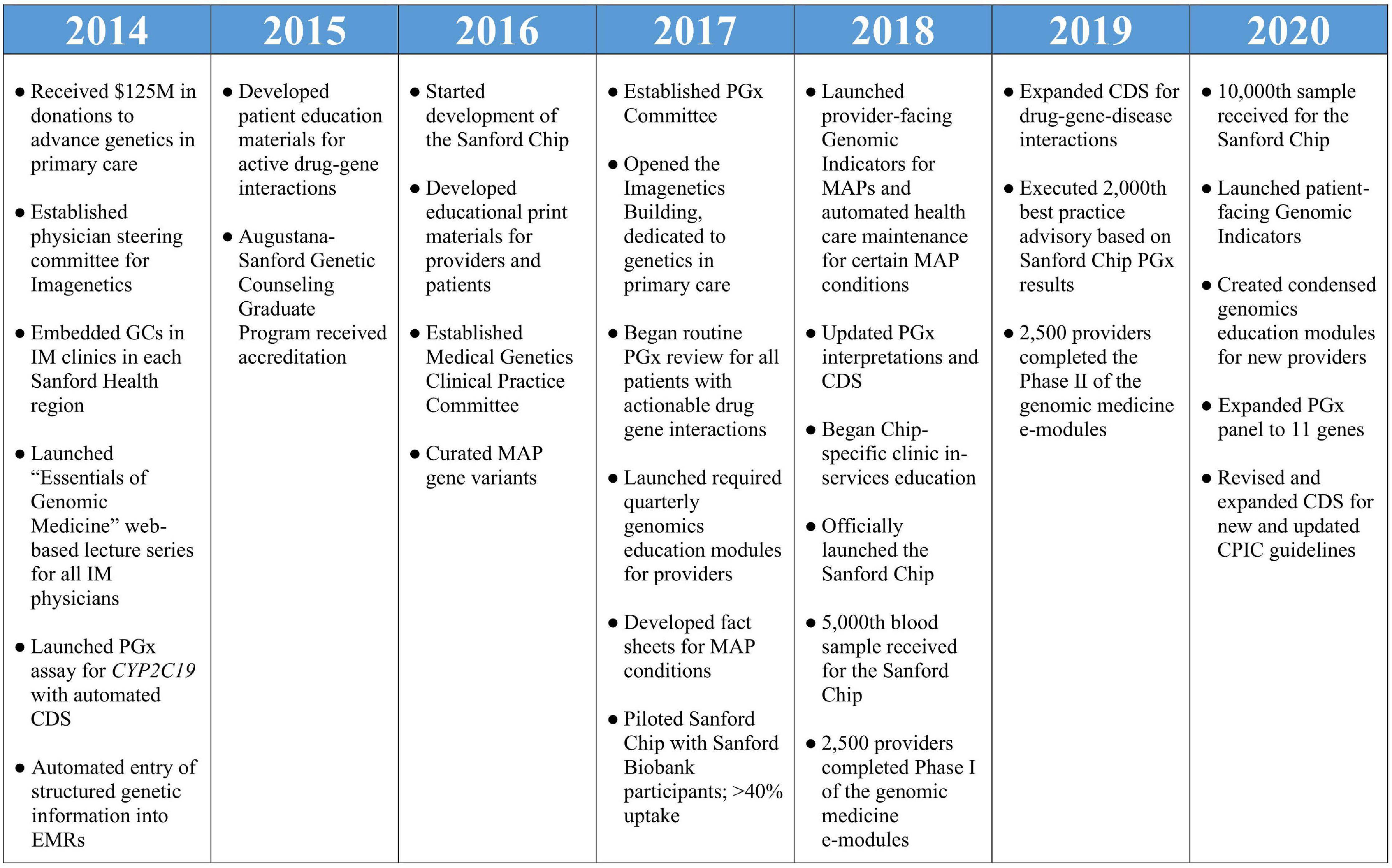
Figure 1. Milestones of the Imagenetics Initiative and Sanford Chip Program. GCs, genetic counselors; IM, internal medicine; PGx, pharmacogenomic; EMRs, electronic medical records; CDS, clinical decision support; MAP, medically actionable predispositions.
Development of the Sanford Chip Program
Testing Platform, Workforce, and Decision Support
A goal of the Imagenetics Initiative was to introduce a low-cost genomic test, the “Sanford Chip,” to enhance preventive care by providing PGx and disease risk information with the strongest evidence for actionability and the potential to improve patient outcomes. The Sanford Chip is offered to patients as an elective service. The laboratory processes for DNA testing, bioinformatic analyses, and reporting are summarized in Figure 2. For all participants enrolled in the Sanford Chip Program, preemptive PGx testing is conducted using the Fluidigm SNP Dynamic Array platform and TaqMan Assay for CYP2D6 copy number assessment. To test for genetic risk factors associated with conditions with proven prevention options (“medically actionable predispositions,” or “MAPs”), the program used Illumina’s Infinium Global Screening Array-24 (GSA). The array captured the entire GWAS catalog as of May 2016 (Buniello et al., 2019), and was customized to include additional markers associated with cardiovascular, oncologic, and neurologic traits for future applications. PGx results are compared to the GSA for concordance. MAPs are confirmed using Sanger sequencing. Testing is performed at the CLIA-certified and CAP-accredited Molecular Genetics Laboratory of Sanford Imagenetics (Sioux Falls, SD). Certified molecular geneticists review quality control metrics for each sample, confirm whether patients have consented for review of MAP variants, and oversee variant interpretation and reporting as applicable. The genes currently examined by the Sanford Chip and their associations with medications and disease are summarized in Supplementary Tables 1,2.
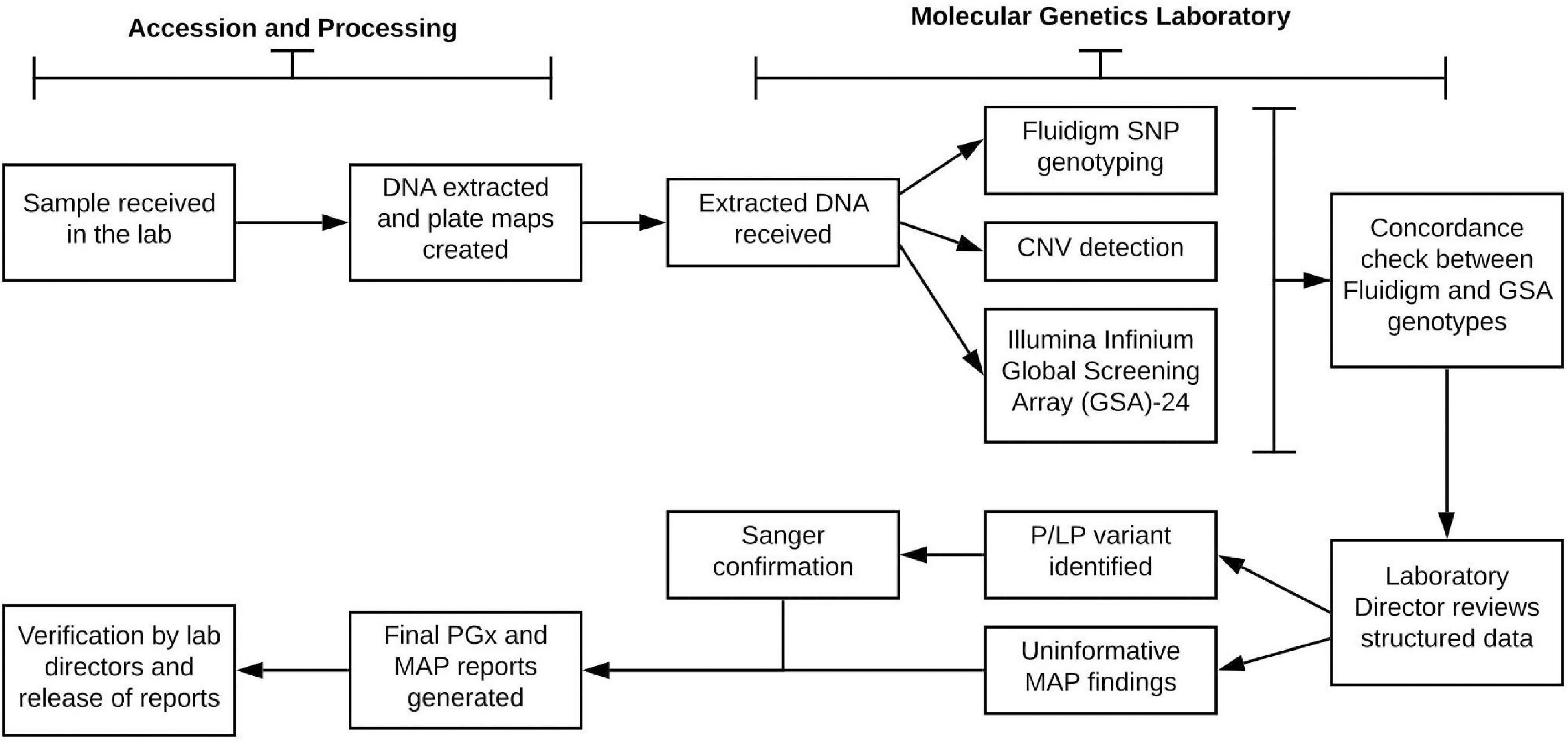
Figure 2. Laboratory process for pharmacogenomic testing and screening for medically actionable predispositions. SNP, single nucleotide polymorphism; P/LP, pathogenic or likely pathogenic; MAP, medically actionable predispositions.
The Imagenetics Initiative developed the technical and clinical expertise to manage the anticipated increased demand for genetic testing system-wide with the roll-out of the Sanford Chip. Sanford Health hired and embedded genetic counselors in primary care clinics, starting with internal medicine clinics in its major markets (Sioux Falls, SD, Fargo, ND, Bismarck, ND, Bemidji, MN) to help ensure patients and providers had the resources to make informed choices about testing and responding to results. This approach has since been expanded to embed genetic counselors in additional primary care clinics, including family medicine and obstetrics and gynecology. To address the shortage of genetic counselors nationally (Hoskovec et al., 2018), Sanford Health partnered with Augustana University to establish the Augustana-Sanford Master of Science in Genetic Counseling Program. This full-time training program, accredited in 2015, has helped develop a pool of qualified professionals who are familiar with the Imagenetics Initiative. The Imagenetics Initiative also leveraged and continues to lean on Sanford Health’s telegenetics expertise to facilitate access for patients who may live far from urban centers. These capabilities are particularly important, given the rural nature of the Sanford Health System.
Clinical decision support (CDS) was developed for Sanford Chip findings using provider and patient Genomic Indicators. Genomic Indicators are functions specific to the EPIC electronic medical records (EMRs) system that facilitate the use of genetic information, including storage of PGx information and findings about MAPs (Caraballo et al., 2020). Indicators appear in the patient summary section of the EMR as well as within the patient’s MyChart record.
Pharmacogenomic Testing
Details about the development of Sanford Health’s PGx program have been published previously (Petry et al., 2019), and points relevant to the Sanford Chip Program are briefly re-stated here. The Imagenetics Initiative formed a system-wide interdisciplinary Pharmacogenomics Committee (PGx Committee) to oversee the PGx program, including prioritization of drug-gene relationships and development of guidance for using PGx information to inform drug selection and dosing decisions. The PGx committee also established a process for developing CDS to ensure that PGx information would be integrated into patients’ EMRs, and that the information would be available at the time of prescribing.
Based on PGx information, automated CDS for 63 drug-gene interactions and 3 drug-gene-disease interactions informs providers that patients may benefit from alterations to medication orders. Examples of the CDS developed for voriconazole orders for patients with CYP2C19 rapid metabolizer genotypes is presented in the Supplementary Image 1. Automated CDS alerts were also developed for drug-gene-disease interactions that may be informed by genetic screening. For instance, CDS notifies providers to avoid medications that may prolong the QT interval in patients with long QT syndrome (Priori et al., 2013; Al-Khatib et al., 2018). CDS is evaluated annually by PGx pharmacists with specialty-specific clinicians and updated to match CPIC® or other consensus guidelines. Patient-facing Genomic Indicators are also sent to MyChart and include a short summary of the PGx findings and uninformative or disease risk MAP findings in plain language, and encourages patients to contact Imagenetics specialists for more information. Notably, CDS was considered for instances when medication orders were consistent with CPIC® guidelines, but ultimately omitted to avoid “alert fatigue” (Shojania et al., 2010; Slight et al., 2013; Bryant et al., 2014; Hinderer et al., 2017; Tolley et al., 2018).
Screening for Medically Actionable Predispositions
The Imagenetics Initiative simultaneously developed approaches to screen patients for MAPs. A Medical Genetics Clinical Practice Committee (CPC) was established to include a group of medical professionals including physicians, genetic counselors, and pharmacists to develop practice guidelines for the Sanford Health system. The Medical Genetics CPC opted to screen for variants in genes recommended by the American College of Medical Genetics and Genomics (ACMG) for secondary findings disclosure (Green et al., 2013a; Kalia et al., 2017). The committee recognized that the ACMG list of genes was not intended to guide or endorse population genetic screening (ACMG Board of Directors, 2019). Nevertheless, it felt that the deliberations about the actionability of these genes (Green et al., 2012) and development of resources to inform clinical responses for unselected populations (Clinical Genome Resource, 2020) represented a consensus about genes for which disclosure would have the best likelihood of leading to clinical benefits.
The Medical Genetics CPC has considered expanding the gene list to screen for additional genes and conditions. The ACMG list of genes was intended to be a “minimal list” for secondary findings disclosure (Green et al., 2013a), and programs that screen biobank participants for MAPs, such as the Geisinger MyCode initiative, currently screen more genes associated with conditions (Carey et al., 2016). Given considerable concern about population genetic screening (Prince et al., 2017; ACMG Board of Directors, 2019; Brothers et al., 2019), Sanford Health has opted for a conservative approach. However, the Medical Genetics CPC reviews the list twice annually to discuss whether changes should be made, including adding or removing conditions and genes.
The laboratory identifies and reports variants on the Sanford Chip that it classifies as pathogenic or likely pathogenic (Richards et al., 2015). Genomic Indicators for MAP findings are automatically integrated into the EMRs of Sanford Chip recipients. Their use automatically modifies recommended surveillance according to best practice guidelines.
Preparing Providers and Patients for the Sanford Chip Program
A physician steering committee with representatives from internal medicine, family medicine, pediatrics, and obstetrics designed an educational program for both health care providers and for administrators of the Sanford Health System. Prior to the launch of the Sanford Chip test, a combination of live and recorded lectures was made available to all providers. From 2017 to 2019, more than 2,500 Sanford physicians and advanced practice providers were required to complete quarterly continuing medical education focused on genetic medicine, with an emphasis on preparing providers to be able to respond to Sanford Chip requests and results. The modules were also offered to all Sanford Health administrators. Details about the content and impact of this educational program are the focus of ongoing research and forthcoming publications. Since the launch of the Sanford Chip, ongoing educational efforts have included in-services, recorded modules and internal and external websites which host repositories for educational resources for health care teams and patients.
The Patient Experience
The Imagenetics Initiative began to offer the Sanford Chip to patients in 2018. An overview of the current and ongoing process for inviting, testing, and reporting Sanford Chip results is summarized in Figure 3. Adult patients are eligible to receive the Sanford Chip if they receive primary care (internal medicine, family medicine, or obstetrics/gynecology) within the Sanford Health System and have a Sanford MyChart account through which they can receive communications and access laboratory findings. The most common way eligible patients receive the Sanford Chip is by responding to invitations sent through MyChart. Links that are embedded in the invitations direct patients to a secure web-based platform where they provide clinical consent for testing. During the process, there is an option to request contact with a laboratory genetic counselor via the medium of their choice (phone, secure MyChart message). Patients who provide clinical consent report their personal and family histories of disease on questionnaires. Patients’ PCPs approve the request to receive the Sanford Chip. In less than 20 cases the patient’s PCP did not approve, and the order was forwarded to a dedicated physician in each region for further consideration and approval, if appropriate. Patients then have a blood draw at any Sanford laboratory for genetic screening. To date, more than 11,000 patients have participated in the Sanford Chip Program, 62% of whom are female and 38% of whom are male. Participating women and men are 50 and 58 years old on average, respectively, and have an average of 9.4 and 8.9 medications on their medication lists, respectively. Patients who participate in the Sanford Chip Program are slightly older than Sanford Health patients overall (average age of women: 48; average age of men: 52) and taking more medications (average medications for men: 7.3; average medications for women: 7.8). Over 90% of participants have been identified with at least one informative pharmacogenomic variant (Supplementary Table 3). Approximately 1.5% of participants who agreed to screening for MAPs have been identified with an autosomal dominant disease predisposition, as summarized in Table 1. Another 1.6% and 0.8% of individuals have been identified with variants in MUTYH and ATP7B, respectively. These individuals received genetic counseling regarding their carrier status and were offered full sequencing to confirm whether they are homozygous or compound heterozygous for pathogenic variants. The process for inviting, consenting, and ultimately communicating results is periodically reviewed by Sanford legal, compliance and privacy offices.
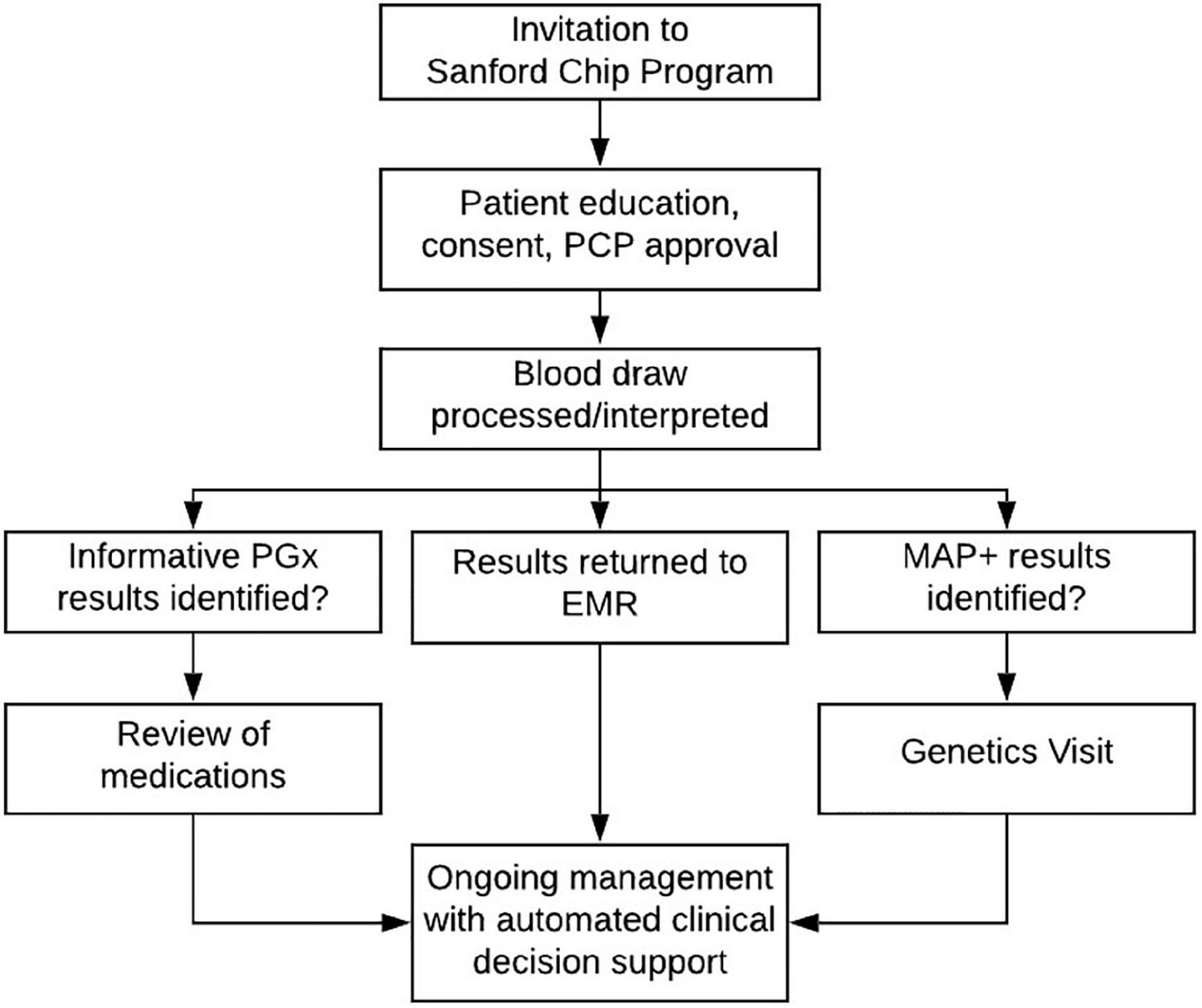
Figure 3. Process for testing, return of results, and ongoing patient management. EMR, electronic medical record; PGx, pharmacogenomic results; MAP, findings about medically actionable predispositions; CDS, clinical decision support.
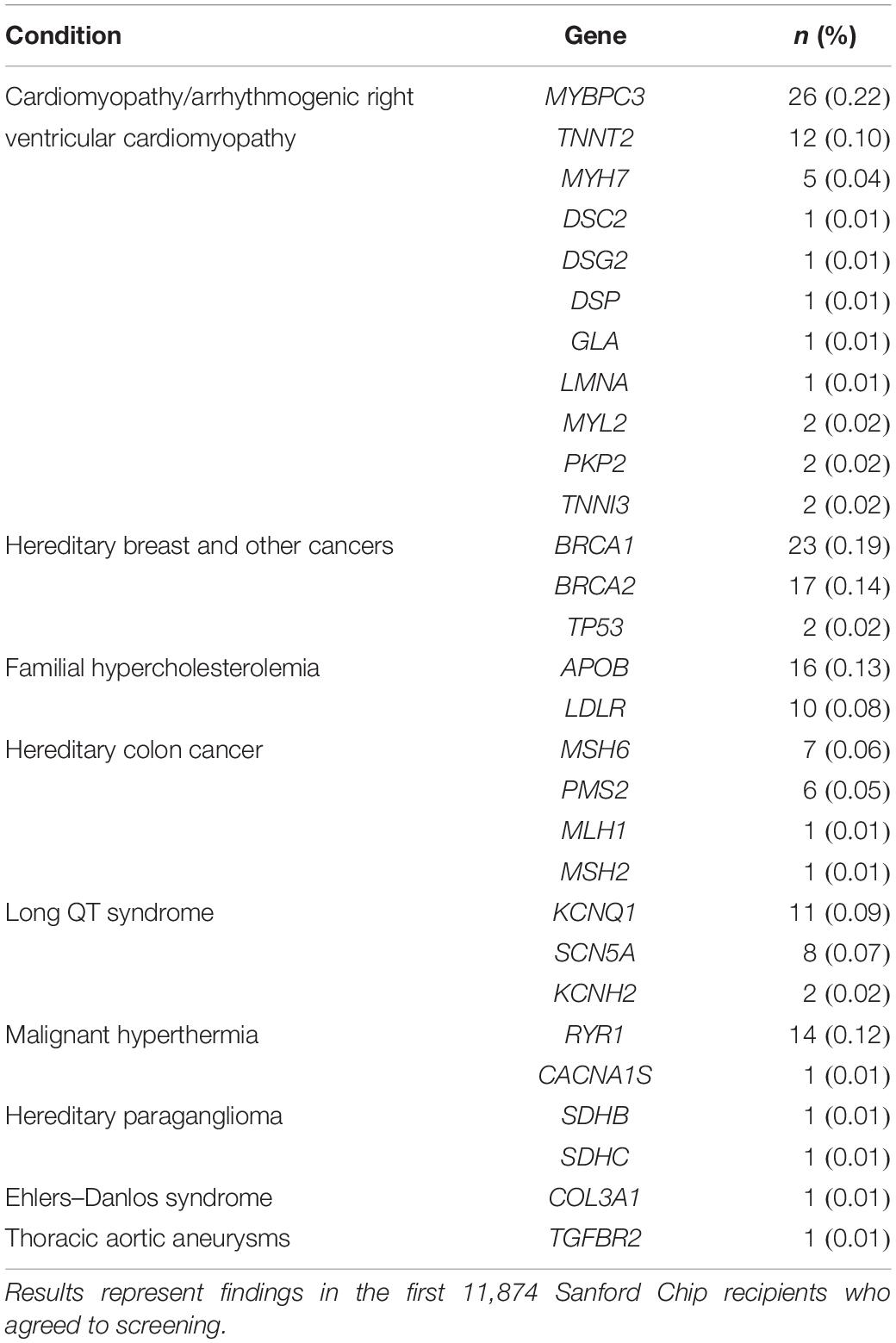
Table 1. Frequency of pathogenic and likely pathogenic variants in genes screened for medically actionable predispositions (MAPs).
Return of Sanford Chip Results
Sanford Chip results are automatically placed into the EMRs via a Health Level Seven interface to facilitate CDS. Simultaneously, automated alerts inform laboratory genetic counselors that results are ready for provider and patient notification. Final reports are sent to ordering providers’ queues for review. Upon release of structured data into the EMR, a message is sent to an in-basket leading to a complete retrospective review of drug-gene variants by a clinical pharmacist. Clinical pharmacists review every patient’s PGx results, medication lists, and clinical profiles, and provide recommendations for alterations, if needed, to PCPs through a clinical note within the patient’s medical record. PCPs have discretion about whether to discuss PGx results with the patient or to change existing medications. Future prescribing of medications affected by a patient’s PGx results is supported by programmed CDS.
When MAPs are identified, laboratory genetic counselors call the ordering providers’ offices and discuss the findings with the providers or their staff. The laboratory genetic counselors then contact patients to review the results and offer clinical appointments with genetic specialists to discuss the findings more thoroughly. Reports are released to patients via their MyChart portal no later than 14 days after reports are drafted. At subsequent clinical appointments with a specialist, comprehensive family histories are collected and reviewed. Genetic counselors discuss the variants and the associated conditions as well as any health management or screening recommendations, along with implications for other family members. At the end of the patient visit, the genetic counselor reviews and initiates additional referrals and follow-up genetic testing as appropriate.
Patients who are not identified with reportable variants in the MAP gene list are informed via MyChart that they have “uninformative results” regarding disease risks. The emphasis on results being “uninformative” rather than “negative” is conventional for genetic testing in the absence of a known familial variant, to minimize the risk that patients will interpret a lack of findings to mean that they have no pathogenic variants for a genetic disorder (Uhlmann et al., 2009). These risks may be even greater for array-based genetic screening approaches that screen for pre-specified list of pathogenic variants, as is currently used for the Sanford Chip Program, given that many causal variants are unique to individuals or families (Alfares et al., 2018). Ordering providers have the option to release the uninformative results, with or without standardized verbiage that emphasizes these limitations of the Sanford Chip to identify disease risks, to the patient portal or allow the system to release the results after 14 days. Follow-up of uninformative results is done at the ordering providers’ discretion.
Evaluating Outcomes: Imagenetics Metrics
Given the robust genomics infrastructure that was established, the Imagenetics Initiative and Sanford Chip Program also provide a real-world setting to generate evidence about its impact on providers and patients (Khoury et al., 2018; Murray et al., 2019). In 2019, Sanford Health began a collaboration with investigators at the Harvard Medical School to launch the Imagenetics Medical/Economic Impact and Reactions to the Sanford Chip Study (METRICS). This research collaboration has focused its initial work on four key aspects of this precision population medicine program: provider preparedness, PGx testing, medically actionable findings, and uninformative MAP findings.
Discussion
With the Sanford Chip, the Imagenetics Initiative implemented one of the first genetics-focused precision population medicine programs that is fully integrated into a health system. Nearly five years prior to offering the Sanford Chip, Sanford Health’s Imagenetics Initiative began developing the plans and infrastructure to support genetic testing in primary care settings. This work included creating the appropriate governance structure, recruiting appropriate personnel, expanding the laboratory and clinical capacity, creating decision support, and ensuring providers were educated and supported. The approach we have summarized here provides a real-world example for implementing genetic testing to inform preventive care in the future, and illuminates the complex planning involved in launching such an enterprise.
The infrastructure that the Imagenetics Initiative created to prepare for the Sanford Chip required a significant financial and strategic commitment from Sanford Health. The support provided to the Sanford Chip Program allowed the Imagenetics Initiative to establish a laboratory and informatics pipeline appropriate for genetic testing at a large scale and create education programs and CDS to support health care providers without specialized training in genetics. Additional investments support retrospective review of pharmacologic information of all Sanford Chip recipients and robust follow-up with patients who are identified with MAPs. The prices charged to payers (primarily patients) to receive the Sanford Chip, are unlikely to cover the costs to develop the program, but current projections about preemptive pharmacogenomic testing and genetic screening show encouraging evidence about their cost effectiveness from a health sector and societal perspective (Plumpton et al., 2019; Zhang et al., 2019; Dong et al., 2020). Nevertheless, health systems that expect precision population medicine programs to be revenue-neutral from the start are likely to find it challenging to ensure providers are maximizing the potential benefits of genetic information for patients (Dzau et al., 2016; Gaff et al., 2017; Levy et al., 2019).
The Sanford experience also provides an instructive example for training and supporting health care providers for precision population medicine system-wide. The Imagenetics Initiative developed ongoing educational curricula tailored to the needs of both generalist and specialist providers. It also developed automated CDS that informs providers about how the results from PGx and genetic screening can inform medical decision making at the time the information is needed. Two aspects of Sanford’s provider education approach merit particular emphasis. First, genetics education over a 2-year period was mandatory for all physicians and advanced practice providers. While other health systems have implemented or capitalized on large-scale provider education efforts for genetics (Murray, 2014; Rubanovich et al., 2018; Crellin et al., 2019), few have had the institutional commitment to require them over long periods of time. Second, the Sanford-Augustana genetic counseling training program not only helps address a national shortage of genetic counselors, but its graduates who take positions at Sanford Health are already familiar with the Sanford patient population and the Sanford Chip.
The challenges of implementing genetics-focused precision population medicine programs are exacerbated by a dearth of guidelines for managing healthy patients with genetic predispositions who have no personal or family history of disease. The current landscape may encourage management practices that may overtreat patients (Woolever, 2008; Caulfield et al., 2013; Diamandis, 2015). Uncertainties about the downstream impact of genetic screening have made payers reluctant to cover genetic screening (Phillips et al., 2014). In order for the field to realize the benefits of genetic screening, it will be important to establish clinical guidelines which facilitate consistent management of asymptomatic patients.
The inclusion of a dedicated research component (METRICS) is another notable aspect of Sanford’s precision population medicine program. While the evidence base for genomic medicine from clinical research is growing rapidly, there is a need to complement these efforts with real-world evidence that follows clinical implementation (Sherman et al., 2016; Berger et al., 2017). METRICS will explore the impact of integrating genetic testing into general clinical practice in an environment that has successfully developed the infrastructure and processes to support it. Moreover, the research will collect patient-reported outcomes that are sometimes omitted from evaluations of precision medicine innovations (Glasgow et al., 2018).
The current approach used to implement the Sanford Chip Program has limitations. PGx testing and genetic screening of healthy patients is voluntary and not standard of care (U.S. Food and Drug Administration, 2018; ACMG Board of Directors, 2019). Experiences about and data from the Sanford Chip Program will help address knowledge gaps about the impact of genomic testing in rural populations (Mapes et al., 2020). However, the areas served by Sanford Health are primarily white, and the work pursued by Sanford Health may provide limited insight to address well-recognized issues about health disparities and underrepresentation of marginalized communities that genomic testing may exacerbate (Landry and Rehm, 2018; Kim and Sarkar, 2019; Martin et al., 2019; Roberts et al., 2019). The array used for genetic screening is less sensitive for identifying pathogenic genetic variants than approaches such as exome or genome sequencing, as noted previously, necessitating a strong educational program to explain the limitations. As the applications for genomics in medicine grow, so should educational content in medical training to address such limitations.
Despite its limitations, the decision to use array technology to launch the Sanford Chip Program provided a lower cost platform for which the infrastructure for supporting a precision population genomic medicine approach could be implemented. The infrastructure that Sanford Health developed positions it to shift to more testing approaches such as exome or genome sequencing in the future. Moreover, the Sanford Chip Program provides the ability for patients to obtain genomic testing in a medical setting that they might otherwise seek from direct-to-consumer options that may omit the expertise to guide interpretation and ongoing management of genetic testing results. The health system chose to pursue a clinical approach to address this patient demand and provide genetic information in a medically responsible manner.
Future versions of the Sanford Chip may include polygenic risk predictions for common, complex conditions, additional PGx information, more comprehensive coverage of MAP genes, or other disease-gene relationships deemed appropriate for clinical return in healthy populations. Looking ahead, we envision an environment where the inclusion of genomics in the care of patients is standard and contributes to more precise risk assessment and management. The Sanford Chip Program is an important step in achieving this vision and will inform the future of genomic medicine not only in the Sanford Health system, but at health systems worldwide.
Members of The Imagenetics Metrics Team
Sanford Health: Jordan Baye, Megan Bell, Kristen Deberg, Benjamin Forred, Colette Free, Catherine Hajek, Joel Van Heukelom, Ashley Hopp, Allison Hutchinson, Ryne Lees, Jennifer Leonhard, Amanda Massmann, Michelle Moore, Amelia Mroch, Natasha Petry, Dylan Platt, Erin Royer, April Schultz, Murat Sincan, Bethany Tucker, and Elizabeth Wheeler. Harvard Pilgrim Health Care Institute: Kurt Christensen, Lauren Galbraith, Jessica LeBlanc, Ryan Walsh, and Emilie Zoltick. Brigham and Women’s Hospital: Robert Green, Charlene Preys, and Carrie Zawatsky. Mayo Clinic: Lisa Mullineaux. National Institutes of Health: Leila Jamal.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics Statement
The studies involving human participants were reviewed and approved by the Sanford Health Institutional Review Board, including a waiver of informed consent to analyze de-identified, aggregated data.
Author Contributions
MB, AH, JRL, MM, LM, NP, DP, SS, AS, BT, JV, EW, and CH contributed to the conception and design of the Imagenetics Initiative and have done clinical and educational work at Sanford Health. KC, CZ, LG, RCG, JLL, LJ, and EZ helped guide analysis of these clinical and educational interventions. The provider preparedness efforts have been led by AH, DP, and CH, with team members JLL, LJ, AS, EW, NP, EZ, LG, DP, KC, and CH contributing to the design of the study and data analysis. Clinical and research efforts regarding medically actionable predispositions have been led by LM, BT, and MM, with team members EZ, MM, BT, KC, LG, CH, JV, CZ, AS, and JRL contributing to the design of the study. Clinical and research efforts regarding pharmacogenomic results have been led by AS, with team members EZ, JV, KC, LG, CH, and NP contributing to the design of the study. Clinical and research efforts regarding uninformative findings have been led by MB, with team members DP, JLL, EZ, KC, LG, CZ, BT, JRL, and CH contributing to the design of the study. All authors revised the manuscript, and approved the final submitted version.
Funding
The Imagenetics Initiative and Sanford Chip Program, and this work was supported Sanford Health. KC was also supported by NIH grant K01-HG009173, and RCG and CZ were also supported by NIH grant R01-HL143295.
Conflict of Interest
RCG has received compensation for advising the following companies: AIA, Grail, Humanity, Kneed Media, Plumcare, UnitedHealth, Verily, VibrentHealth, Wamberg; and is co-founder of Genome Medical, Inc., a technology and services company providing genetics expertise to patients, providers, employers and care systems.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors thank Charlene Preys and Ally Hempel for their assistance.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fgene.2021.626845/full#supplementary-material
Supplementary Image 1 | Examples of clinical decision support alerts for patients with CYP2C19 rapid metabolizer status. The top example is the alert that activates for an adult patient, while the bottom example is the alert that activates for a pediatric patient.
Supplementary Table 1 | Summary of pharmacogenomic variants and medically actionable predispositions that are targeted by the Sanford Chip.
Supplementary Table 2 | Summary of medically actionable predispositions that are targeted by the Sanford Chip.
Supplementary Table 3 | Frequency of phenotypes/genotypes in pharmacogenomic genes. Results represent findings from the first 10,723 patients who enrolled in the Sanford Chip program. Counts vary by gene depending on the genes included on the Sanford Chip at the time patients received testing. Some genes from Supplementary Table 1 are omitted because they were only recently added to the Sanford Chip.
References
ACMG Board of Directors (2019). The use of ACMG secondary findings recommendations for general population screening: a policy statement of the American College of Medical Genetics and Genomics (ACMG). Genet. Med. 21, 1467–1468. doi: 10.1038/s41436-019-0502-5
Alfares, A., Aloraini, T., Subaie, L. A., Alissa, A., Qudsi, A. A., Alahmad, A., et al. (2018). Whole-genome sequencing offers additional but limited clinical utility compared with reanalysis of whole-exome sequencing. Genet. Med. 20, 1328–1333. doi: 10.1038/gim.2018.41
Al-Khatib, S. M., Stevenson, W. G., Ackerman, M. J., Bryant, W. J., Callans, D. J., Curtis, A. B., et al. (2018). 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J. Am. Coll. Cardiol. 72, e91–e220. doi: 10.1016/j.jacc.2017.10.054
Allyse, M. A., Robinson, D. H., Ferber, M. J., and Sharp, R. R. (2018). Direct-to-consumer testing 2.0: emerging models of direct-to-consumer genetic testing. Mayo Clin. Proc. 93, 113–120. doi: 10.1016/j.mayocp.2017.11.001
Amendola, L. M., Dorschner, M. O., Robertson, P. D., Salama, J. S., Hart, R., Shirts, B. H., et al. (2015). Actionable exomic incidental findings in 6503 participants: challenges of variant classification. Genome Res. 25, 305–315. doi: 10.1101/gr.183483.114
Bell, M. (2017). Where personalized medicine, patient engagement, and primary care collide. S. D. Med. Spec No, 34–36.
Bennette, C. S., Gallego, C. J., Burke, W., Jarvik, G. P., and Veenstra, D. L. (2015). The cost-effectiveness of returning incidental findings from next-generation genomic sequencing. Genet. Med. 17, 587–595. doi: 10.1038/gim.2014.156
Berger, M. L., Sox, H., Willke, R. J., Brixner, D. L., Eichler, H. G., Goettsch, W., et al. (2017). Good practices for real-world data studies of treatment and/or comparative effectiveness: recommendations from the Joint ISPOR-ISPE Special Task Force on real-world evidence in Health Care decision making. Value Health 20, 1003–1008. doi: 10.1016/j.jval.2017.08.3019
Brothers, K. B., Vassy, J. L., and Green, R. C. (2019). Reconciling opportunistic and population screening in clinical genomics. Mayo Clin. Proc. 94, 103–109. doi: 10.1016/j.mayocp.2018.08.028
Bryant, A. D., Fletcher, G. S., and Payne, T. H. (2014). Drug interaction alert override rates in the Meaningful Use era: no evidence of progress. Appl. Clin. Inform. 5, 802–813. doi: 10.4338/ACI-2013-12-RA-0103
Buniello, A., MacArthur, J. A. L., Cerezo, M., Harris, L. W., Hayhurst, J., Malangone, C., et al. (2019). The NHGRI-EBI GWAS Catalog of published genome-wide association studies, targeted arrays and summary statistics 2019. Nucleic Acids Res. 47, D1005–D1012. doi: 10.1093/nar/gky1120
Campion, M., Goldgar, C., Hopkin, R. J., Prows, C. A., and Dasgupta, S. (2019). Genomic education for the next generation of health-care providers. Genet. Med. 21, 2422–2430. doi: 10.1038/s41436-019-0548-4
Caraballo, P. J., Sutton, J. A., Giri, J., Wright, J. A., Nicholson, W. T., Kullo, I. J., et al. (2020). Integrating pharmacogenomics into the electronic health record by implementing genomic indicators. J. Am. Med. Inform Assoc. 27, 154–158. doi: 10.1093/jamia/ocz177
Carey, D. J., Fetterolf, S. N., Davis, F. D., Faucett, W. A., Kirchner, H. L., Mirshahi, U., et al. (2016). The Geisinger MyCode community health initiative: an electronic health record-linked biobank for precision medicine research. Genet. Med. 18, 906–913. doi: 10.1038/gim.2015.187
Caulfield, T., Evans, J., McGuire, A., McCabe, C., Bubela, T., Cook-Deegan, R., et al. (2013). Reflections on the cost of “low-cost” whole genome sequencing: framing the health policy debate. PLoS Biol. 11:e1001699. doi: 10.1371/journal.pbio.1001699
Clinical Genome Resource (2020). ClinGen Actionability Reports [Online]. Available online at: https://actionability.clinicalgenome.org/ac/ (accessed April 12, 2020).
Collins, F. S., Green, E. D., Guttmacher, A. E., and Guyer, M. S. (2003). A vision for the future of genomics research. Nature 422, 835–847.
Crellin, E., McClaren, B., Nisselle, A., Best, S., Gaff, C., and Metcalfe, S. (2019). Preparing medical specialists to practice genomic medicine: education an essential part of a broader strategy. Front. Genet. 10:789. doi: 10.3389/fgene.2019.00789
Diamandis, E. P. (2015). The hundred person wellness project and Google’s baseline study: medical revolution or unnecessary and potentially harmful over-testing? BMC Med. 13:5. doi: 10.1186/s12916-014-0239-6
Dong, O. M., Wheeler, S. B., Cruden, G., Lee, C. R., Voora, D., Dusetzina, S. B., et al. (2020). Cost-effectiveness of multigene pharmacogenetic testing in patients with acute coronary syndrome after percutaneous coronary intervention. Value Health 23, 61–73. doi: 10.1016/j.jval.2019.08.002
Dzau, V. J., Ginsburg, G. S., Chopra, A., Goldman, D., Green, E. D., Leonard, D. G. B., et al. (2016). Realizing the Full Potential of Precision Medicine in Health and Health Care: A Vital Direction for Health and Health Care. Washington, DC: National Academy of Medicine.
Gaff, C. L., Winship, I. M., Forrest, S. M., Hansen, D. P., Clark, J., Waring, P. M., et al. (2017). Preparing for genomic medicine: a real world demonstration of health system change. NPJ Genom Med. 2:16. doi: 10.1038/s41525-017-0017-4
Ginsburg, G. S., Horowitz, C. R., and Orlando, L. A. (2019). What will it take to implement genomics in practice? Lessons from the IGNITE network. Per. Med. 16, 259–261. doi: 10.2217/pme-2019-0021
Glasgow, R. E., Kwan, B. M., and Matlock, D. D. (2018). Realizing the full potential of precision health: the need to include patient-reported health behavior, mental health, social determinants, and patient preferences data. J. Clin. Transl. Sci. 2, 183–185. doi: 10.1017/cts.2018.31
Goldenberg, A. J., Dodson, D. S., Davis, M. M., and Tarini, B. A. (2013). Parents’ interest in whole-genome sequencing of newborns. Genet. Med. 16, 78–84. doi: 10.1038/gim.2013.76
Green, R. C., Berg, J. S., Berry, G. T., Biesecker, L. G., Dimmock, D. P., Evans, J. P., et al. (2012). Exploring concordance and discordance for return of incidental findings from clinical sequencing. Genet. Med. 14, 405–410. doi: 10.1038/gim.2012.21
Green, R. C., Berg, J. S., Grody, W. W., Kalia, S. S., Korf, B. R., Martin, C. L., et al. (2013a). ACMG recommendations for reporting of incidental findings in clinical exome and genome sequencing. Genet. Med. 15, 565–574. doi: 10.1038/gim.2013.73
Green, R. C., Rehm, H. L., and Kohane, I. S. (2013b). “Clinical genome sequencing,” in Genomic and Personalized Medicine, 2 Edn, eds G. S. Ginsberg and H. F. Willard (San Diego, CA: Academic Press), 102–122.
Hinderer, M., Boeker, M., Wagner, S. A., Lablans, M., Newe, S., Hülsemann, J. L., et al. (2017). Integrating clinical decision support systems for pharmacogenomic testing into clinical routine - a scoping review of designs of user-system interactions in recent system development. BMC Med. Inform. Decis. Mak. 17:81. doi: 10.1186/s12911-017-0480-y
Hoskovec, J. M., Bennett, R. L., Carey, M. E., DaVanzo, J. E., Dougherty, M., Hahn, S. E., et al. (2018). Projecting the supply and demand for certified genetic counselors: a workforce study. J. Genet. Couns. 27, 16–20. doi: 10.1007/s10897-017-0158-8
Kalia, S. S., Adelman, K., Bale, S. J., Chung, W. K., Eng, C., Evans, J. P., et al. (2017). Recommendations for reporting of secondary findings in clinical exome and genome sequencing, 2016 update (ACMG SF v2.0): a policy statement of the American College of Medical Genetics and Genomics. Genet. Med. 19, 249–255. doi: 10.1038/gim.2016.190
Kho, A. N., Rasmussen, L. V., Connolly, J. J., Peissig, P. L., Starren, J., Hakonarson, H., et al. (2013). Practical challenges in integrating genomic data into the electronic health record. Genet. Med. 15, 772–778. doi: 10.1038/gim.2013.131
Khoury, M. J., Feero, W. G., Chambers, D. A., Brody, L. C., Aziz, N., Green, R. C., et al. (2018). A collaborative translational research framework for evaluating and implementing the appropriate use of human genome sequencing to improve health. PLoS Med. 15:e1002631. doi: 10.1371/journal.pmed.1002631
Kim, I. E. Jr., and Sarkar, I. N. (2019). Racial representation disparity of population-level genomic sequencing efforts. Stud. Health Technol. Inform. 264, 974–978. doi: 10.3233/shti190369
Klein, T. E., Chang, J. T., Cho, M. K., Easton, K. L., Fergerson, R., Hewett, M., et al. (2001). Integrating genotype and phenotype information: an overview of the PharmGKB project. Pharmacogenetics research network and knowledge base. Pharmacogenomics J. 1, 167–170. doi: 10.1038/sj.tpj.6500035
Landry, L. G., and Rehm, H. L. (2018). Association of racial/ethnic categories with the ability of genetic tests to detect a cause of cardiomyopathy. JAMA Cardiol. 3, 341–345. doi: 10.1001/jamacardio.2017.5333
Levy, K. D., Blake, K., Fletcher-Hoppe, C., Franciosi, J., Goto, D., Hicks, J. K., et al. (2019). Opportunities to implement a sustainable genomic medicine program: lessons learned from the IGNITE Network. Genet. Med. 21, 743–747. doi: 10.1038/s41436-018-0080-y
Manickam, K., Buchanan, A. H., Schwartz, M. B., Hallquist, M. L. G., Williams, J. L., Rahm, A. K., et al. (2018). Exome sequencing–based screening for brca1/2 expected pathogenic variants among adult biobank participants. JAMA Netw. Open 1:e182140. doi: 10.1001/jamanetworkopen.2018.2140
Mapes, B. M., Foster, C. S., Kusnoor, S. V., Epelbaum, M. I., AuYoung, M., Jenkins, G., et al. (2020). Diversity and inclusion for the All of Us research program: a scoping review. PLoS One 15:e0234962. doi: 10.1371/journal.pone.0234962
Martin, A. R., Kanai, M., Kamatani, Y., Okada, Y., Neale, B. M., and Daly, M. J. (2019). Clinical use of current polygenic risk scores may exacerbate health disparities. Nat. Genet. 51, 584–591. doi: 10.1038/s41588-019-0379-x
McCarthy, J. J., McLeod, H. L., and Ginsburg, G. S. (2013). Genomic medicine: a decade of successes, challenges, and opportunities. Sci. Transl. Med. 5:189sr184. doi: 10.1126/scitranslmed.3005785
Messner, D. A., Koay, P., Al Naber, J., Cook-Deegan, R., Majumder, M., Javitt, G., et al. (2017). Barriers to clinical adoption of next-generation sequencing: a policy Delphi panel’s solutions. Per. Med. 14, 339–354. doi: 10.2217/pme-2016-0104
Murray, M. F. (2014). Educating physicians in the era of genomic medicine. Genome Med. 6:45. doi: 10.1186/gm564
Murray, M. F., Evans, J. P., and Khoury, M. J. (2019). DNA-based population screening: potential suitability and important knowledge gaps. JAMA 323, 307–308. doi: 10.1001/jama.2019.18640
National Cancer Institute (2019). NCI Dictionary of Genetics Terms [Online]. Available online at: https://www.cancer.gov/publications/dictionaries/genetics-dictionary (accessed November 15, 2019)
O’Donnell, P. H., Danahey, K., Jacobs, M., Wadhwa, N. R., Yuen, S., Bush, A., et al. (2014). Adoption of a clinical pharmacogenomics implementation program during outpatient care–initial results of the University of Chicago “1,200 Patients Project”. Am. J. Med. Genet. C Semin. Med. Genet. 166c, 68–75. doi: 10.1002/ajmg.c.31385
Olfson, E., Cottrell, C. E., Davidson, N. O., Gurnett, C. A., Heusel, J. W., Stitziel, N. O., et al. (2015). Identification of medically actionable secondary findings in the 1000 Genomes. PLoS One 10:e0135193. doi: 10.1371/journal.pone.0135193
Overby, C. L., Erwin, A. L., Abul-Husn, N. S., Ellis, S. B., Scott, S. A., Obeng, A. O., et al. (2014). Physician attitudes toward adopting genome-guided prescribing through clinical decision support. J. Pers. Med. 4, 35–49. doi: 10.3390/jpm4010035
Petry, N., Baye, J., Aifaoui, A., Wilke, R. A., Lupu, R. A., Savageau, J., et al. (2019). Implementation of wide-scale pharmacogenetic testing in primary care. Pharmacogenomics 20, 903–913. doi: 10.2217/pgs-2019-0043
Phillips, K. A., Trosman, J. R., Kelley, R. K., Pletcher, M. J., Douglas, M. P., and Weldon, C. B. (2014). Genomic sequencing: assessing the health care system, policy, and big-data implications. Health Aff. (Millwood). 33, 1246–1253. doi: 10.1377/hlthaff.2014.0020
Plumpton, C. O., Pirmohamed, M., and Hughes, D. A. (2019). Cost-effectiveness of panel tests for multiple pharmacogenes associated with adverse drug reactions: an evaluation framework. Clin. Pharmacol. Ther. 105, 1429–1438. doi: 10.1002/cpt.1312
Prince, A. E., Cadigan, R. J., Henderson, G. E., Evans, J. P., Adams, M., Coker-Schwimmer, E., et al. (2017). Is there evidence that we should screen the general population for Lynch syndrome with genetic testing? A systematic review. Pharmgenomics Pers. Med. 10, 49–60. doi: 10.2147/pgpm.s123808
Priori, S. G., Wilde, A. A., Horie, M., Cho, Y., Behr, E. R., Berul, C., et al. (2013). HRS/EHRA/APHRS expert consensus statement on the diagnosis and management of patients with inherited primary arrhythmia syndromes: document endorsed by HRS, EHRA, and APHRS in May 2013 and by ACCF, AHA, PACES, and AEPC in June 2013. Heart Rhythm. 10, 1932–1963. doi: 10.1016/j.hrthm.2013.05.014
Raghavan, S., and Vassy, J. L. (2014). Do physicians think genomic medicine will be useful for patient care? Per. Med. 11, 424–433. doi: 10.2217/pme.14.25
Rehm, H. L., Berg, J. S., Brooks, L. D., Bustamante, C. D., Evans, J. P., Landrum, M. J., et al. (2015). ClinGen - the clinical genome resource. N. Engl. J. Med. 372, 2235–2242. doi: 10.1056/NEJMsr1406261
Relling, M. V., and Evans, W. E. (2015). Pharmacogenomics in the clinic. Nature 526, 343–350. doi: 10.1038/nature15817
Relling, M. V., and Klein, T. E. (2011). CPIC: clinical pharmacogenetics implementation consortium of the pharmacogenomics research network. Clin. Pharmacol. Ther. 89, 464–467. doi: 10.1038/clpt.2010.279
Richards, S., Aziz, N., Bale, S., Bick, D., Das, S., Gastier-Foster, J., et al. (2015). Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of medical genetics and genomics and the association for molecular pathology. Genet. Med. 17, 405–423. doi: 10.1038/gim.2015.30
Roberts, J. S., Robinson, J. O., Diamond, P. M., Bharadwaj, A., Christensen, K. D., Lee, K. B., et al. (2018). Patient understanding of, satisfaction with, and perceived utility of whole-genome sequencing: findings from the MedSeq project. Genet. Med. 20, 1069–1076. doi: 10.1038/gim.2017.223
Roberts, M. C., Mensah, G. A., and Khoury, M. J. (2019). Leveraging implementation science to address health disparities in genomic medicine: examples from the field. Ethn. Dis. 29, 187–192. doi: 10.18865/ed.29.S1.187
Rubanovich, C. K., Cheung, C., Mandel, J., and Bloss, C. S. (2018). Physician preparedness for big genomic data: a review of genomic medicine education initiatives in the United States. Hum. Mol. Genet. 27, R250–R258. doi: 10.1093/hmg/ddy170
Shaer, O., Nov, O., Westendorf, L., and Ball, M. (2017). Communicating personal genomic information to non-experts: a new frontier for human-computer interaction. Found. Trends Hum. Comput. Interact. 11, 1–62. doi: 10.1561/1100000067
Sherman, R. E., Anderson, S. A., Dal Pan, G. J., Gray, G. W., Gross, T., Hunter, N. L., et al. (2016). Real-world evidence - what is it and what can it tell us? N. Engl. J. Med. 375, 2293–2297. doi: 10.1056/NEJMsb1609216
Shojania, K. G., Jennings, A., Mayhew, A., Ramsay, C., Eccles, M., and Grimshaw, J. (2010). Effect of point-of-care computer reminders on physician behaviour: a systematic review. CMAJ 182, E216–E225. doi: 10.1503/cmaj.090578
Shuldiner, A. R., Relling, M. V., Peterson, J. F., Hicks, J. K., Freimuth, R. R., Sadee, W., et al. (2013). The pharmacogenomics research network translational pharmacogenetics program: overcoming challenges of real-world implementation. Clin. Pharmacol. Ther. 94, 207–210. doi: 10.1038/clpt.2013.59
Slight, S. P., Seger, D. L., Nanji, K. C., Cho, I., Maniam, N., Dykes, P. C., et al. (2013). Are we heeding the warning signs? Examining providers’ overrides of computerized drug-drug interaction alerts in primary care. PLoS One 8:e85071. doi: 10.1371/journal.pone.0085071
Stark, Z., Dolman, L., Manolio, T. A., Ozenberger, B., Hill, S. L., Caulfied, M. J., et al. (2019). Integrating genomics into healthcare: a global responsibility. Am. J. Hum. Genet. 104, 13–20. doi: 10.1016/j.ajhg.2018.11.014
Tolley, C. L., Slight, S. P., Husband, A. K., Watson, N., and Bates, D. W. (2018). Improving medication-related clinical decision support. Am. J. Health Syst. Pharm. 75, 239–246. doi: 10.2146/ajhp160830
U.S. Food and Drug Administration (2018). The FDA Warns Against the Use of Many Genetic Tests with Unapproved Claims to Predict Patient Response to Specific Medications: FDA Safety Communication [Online]. Available online at: https://www.fda.gov/medical-devices/safety-communications/fda-warns-against-use-many-genetic-tests-unapproved-claims-predict-patient-response-specific#actions (accessed July 6, 2020)
U.S. National Library of Medicine (2020). Home page. Genetics Home Reference [Online]. Bethesda, MD: The Library.
Uhlmann, W. R., Schuette, J. L., and Yashar, B. M. (2009). A Guide to Genetic Counseling. Hoboken, NJ: Wiley-Blackwell.
Vassy, J. L., Christensen, K. D., Schonman, E. F., Blout, C. L., Robinson, J. O., Krier, J. B., et al. (2017). The impact of whole genome sequencing on the primary care and outcomes of healthy adult patients: a pilot randomized trial. Ann. Intern. Med. 167, 159–169. doi: 10.7326/M17-0188
Waisbren, S. E., Weipert, C. M., Walsh, R. C., Petty, C. R., and Green, R. C. (2016). Psychosocial factors influencing parental interest in genomic sequencing of newborns. Pediatrics 137(Suppl. 1) S30–S35. doi: 10.1542/peds.2015-3731G
Wang, C., Cahill, T. J., Parlato, A., Wertz, B., Zhong, Q., Cunningham, T. N., et al. (2018). Consumer use and response to online third-party raw DNA interpretation services. Mol. Genet. Genomic Med. 6, 35–43. doi: 10.1002/mgg3.340
Weitzel, K. W., Cavallari, L. H., and Lesko, L. J. (2017). Preemptive panel-based pharmacogenetic testing: the time is now. Pharm. Res. 34, 1551–1555. doi: 10.1007/s11095-017-2163-x
Williams, M. S., Taylor, C. O., Walton, N. A., Goehringer, S. R., Aronson, S., Freimuth, R. R., et al. (2019). Genomic information for clinicians in the electronic health record: lessons learned From the Clinical Genome resource project and the electronic medical records and genomics network. Front. Genet. 10:1059. doi: 10.3389/fgene.2019.01059
Wolyniak, M. J., Bemis, L. T., and Prunuske, A. J. (2015). Improving medical students’ knowledge of genetic disease: a review of current and emerging pedagogical practices. Adv. Med. Educ. Pract. 6, 597–607. doi: 10.2147/amep.s73644
Woolever, D. R. (2008). The art and science of clinical decision making. Fam. Pract. Manag. 15, 31–36.
Keywords: pharmacogenomic testing, genetic counseling, decision support systems – clinical, genetic testing, primary health care
Citation: Christensen KD, Bell M, Zawatsky CLB, Galbraith LN, Green RC, Hutchinson AM, Jamal L, LeBlanc JL, Leonhard JR, Moore M, Mullineaux L, Petry N, Platt DM, Shaaban S, Schultz A, Tucker BD, Van Heukelom J, Wheeler E, Zoltick ES and Hajek C (2021) Precision Population Medicine in Primary Care: The Sanford Chip Experience. Front. Genet. 12:626845. doi: 10.3389/fgene.2021.626845
Received: 06 November 2020; Accepted: 11 February 2021;
Published: 12 March 2021.
Edited by:
Suzette J. Bielinski, Mayo Clinic, United StatesReviewed by:
Chiara Fabbri, King’s College London, United KingdomPeter Hulick, NorthShore University Health System, United States
Copyright © 2021 Christensen, Bell, Zawatsky, Galbraith, Green, Hutchinson, Jamal, LeBlanc, Leonhard, Moore, Mullineaux, Petry, Platt, Shaaban, Schultz, Tucker, Van Heukelom, Wheeler, Zoltick and Hajek. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Catherine Hajek, Q2F0aGVyaW5lLmhhamVrQHNhbmZvcmRoZWFsdGgub3Jn
 Kurt D. Christensen
Kurt D. Christensen Megan Bell4
Megan Bell4 Carrie L. B. Zawatsky
Carrie L. B. Zawatsky Jessica L. LeBlanc
Jessica L. LeBlanc Natasha Petry
Natasha Petry Dylan M. Platt
Dylan M. Platt Sherin Shaaban
Sherin Shaaban April Schultz
April Schultz Bethany D. Tucker
Bethany D. Tucker Joel Van Heukelom
Joel Van Heukelom Emilie S. Zoltick
Emilie S. Zoltick