- 1Institute of Respiratory Diseases, The First Hospital of China Medical University, Shenyang, China
- 2Department of Endoscopy, Shenyang Chest Hospital, Shenyang, China
- 3Department of Laboratory, Shenyang Chest Hospital, Shenyang, China
- 4Institute of Respiratory Diseases, Shenyang Chest Hospital, Shenyang, China
- 5Department of Pulmonary and Critical Care Medicine, Center of Respiratory Medicine, China-Japan Friendship Hospital, Beijing, China
- 6Graduate School of Peking Union Medical College, Chinese Academy of Medical Sciences, Peking Union Medical College, Beijing, China
- 7National Center for Respiratory Medicine, Beijing, China
- 8Institute of Respiratory Medicine, Chinese Academy of Medical Sciences, Beijing, China
- 9National Clinical Research Center for Respiratory Diseases, Beijing, China
Background: The Xpert Mycobacterium tuberculosis/rifampin (MTB/RIF) assay has shown good diagnostic efficacy in brushing and biopsy tissue samples from patients with tracheobronchial tuberculosis (TBTB). However, its diagnostic value in bronchoalveolar lavage fluid (BALF) is still unclear. Therefore, the present retrospective study aimed to evaluate the diagnostic value of the Xpert MTB/RIF assay in BALF.
Methods: The clinical data of 266 patients with suspected TBTB from January 2018 to October 2020 were pooled with complete details of bronchial brush and bronchoalveolar lavage samples. Smears of the bronchial brushings were stained with Auramine O stain to detect acid-fast bacilli (AFB), and BALF samples were used for culturing MTB with the BACTEC MGIT 960 system and the Xpert MTB/RIF assay. The diagnostic performance of these methods was assessed and compared.
Results: A total of 266 patients suspected to have TBTB were enrolled in the final analysis. Of these patients, 179 patients were confirmed to have TBTB and 87 patients were non-TBTB. The sensitivity of the Xpert MTB/RIF assay in BALF (87.2%) was significantly higher than that of the brush smear for AFB (35.2%, p < 0.001). No significant difference was observed between the sensitivities of the Xpert MTB/RIF assay in BALF and MTB culture in BALF (87.2 vs. 84.9%, p = 0.542). The specificities of the Xpert MTB/RIF assay in BALF, MTB culture in BALF, and the bronchial brush smear were 97.7, 97.7, and 98.9%, respectively. The positive predictive value (PPV) and negative predictive value (NPV) of the Xpert MTB/RIF assay in BALF, MTB culture in BALF, and the bronchial brush smear were 98.7 and 78.7%, 98.7 and 75.9%, and 98.4 and 42.6%, respectively. Among the MTB culture-positive patients with TBTB detected by the Xpert assay, 27.0% (20/74) were identified to be resistant to RIF.
Conclusions: The Xpert MTB/RIF assay in BALF enables a rapid and accurate diagnosis of TBTB and identification of RIF resistance, which is crucial for timely and proper treatment. Moreover, in patients with TBTB, BALF could be used as an alternative to bronchial brushing and biopsy tissues for the Xpert MTB/RIF assay.
Introduction
Tuberculosis (TB) is considered to be the ninth leading cause of death worldwide, and it remains one of the major global public health issues (1). Tracheobronchial tuberculosis (TBTB) is a special form of TB and is defined as Mycobacterium tuberculosis (MTB) infection of the tracheobronchial tree with microbial and histopathological evidence, regardless of parenchymal involvement (2). TBTB has been reported in approximately 4.1–54.3% of patients with active pulmonary TB (3, 4). Severe tracheobronchial stenosis caused by TBTB, which affects the patient's quality of life, was detected in 23.3% of patients with TBTB (5). The diagnosis of TBTB is, however, challenging because of its non-specific clinical manifestations, especially in the early stage, and unsatisfactory existing methods for acquiring microbial evidence. Thus, patients with TBTB often face a dilemma of delayed diagnosis and untimely treatment, which result in the occurrence and progression of severe tracheobronchial stenosis (6–8). Therefore, the rapid and accurate diagnosis of TBTB is crucial to minimize or prevent complications.
The Xpert MTB/rifampin (MTB/RIF) assay (Cepheid, Sunnyvale, CA, USA) is an integrated semiautomated nucleic acid amplification test designed for rapid detection of the presence of MTB and mutations associated with RIF resistance within 2 h (9). Several previous studies have proved the diagnostic value of bronchoalveolar lavage fluid (BALF) for the Xpert MTB/RIF assay in adults and children with pulmonary TB (10–12). However, because TBTB is a special type of pulmonary TB, the efficacy of its diagnosis by using different methods varies from that for pulmonary TB and even among different subtypes of TBTB because of the different characteristics of exudation, proliferation, and necrosis. Our previous study (13) showed that the sensitivities of the Xpert MTB/RIF assay of bronchial brushing and biopsy samples from TBTB patients were 57.4 and 63.9%, respectively, and the specificity for both types of samples was 100%; this proved that these results were superior to those of sputum smears, bronchial brush smears, and sputum culture. The yield of the Xpert MTB/RIF assay in bronchial brushings was significantly higher for the caseating or ulcerative subtypes than for the fibrostenotic or edematous-hyperemic subtypes (13). Bronchial biopsy and brushing samples may not be available for diagnosing bronchial fibrous stenosis and other conditions; however, in most TBTB cases, BALF is available and is a relatively less invasive and safer method. Hence, it is necessary to evaluate the diagnostic performance of the Xpert MTB/RIF assay in BALF for TBTB patients, and more specifically, to determine the diagnostic performance of the assay for different TBTB bronchoscopic subtypes. Therefore, we conducted this retrospective study to evaluate the efficacy of the Xpert MTB/RIF assay in BALF for the rapid diagnosis of TBTB.
Materials and Methods
Participants
This was a retrospective study conducted in the First Hospital of China Medical University and Shenyang Chest Hospital from Janurary 2018 to October 2020.
A total of 266 suspected TBTB patients were enrolled in the study through the inpatient case registration system in the hospital information system. Inclusion criteria were as follows: (1) inpatient cases suspected to have TBTB who were registered in the hospital information system and were available for follow-up after 6 months and (2) patients who underwent bronchial brush and bronchoalveolar lavage sample collection. Brushing specimens were used for smears for acid-fast bacilli (AFB), while BALF samples were used for MTB culture and the Xpert MTB/RIF assay. Exclusion criteria were as follows: (1) patients without a definite diagnosis after discharge from the hospital and after 6 months of follow-up and (2) patients with obvious pulmonary involvement instead of TBTB alone.
Definition: TBTB was confirmed by the presence of a visible tracheobronchial lesion proximal to a segmental bronchus, with histological evidence from bronchial biopsy and/or microbiological tests (sputum smear or brushing smear or BALF was positive for AFB or MTB culture was positive) (5, 14).
Clinical data of the patients were collected by two authors (YS and QinZ) from the medical databases of the hospitals. The study was approved by the Ethics Committee of China Medical University and Shenyang Chest Hospital. The ethics committee exempted obtaining informed consent from patients because of the retrospective nature of the study. Patient data were deidentified before data access and analysis.
AFB Smear Microscopy
Three bronchial brush smears were sent to the clinical microbiology laboratory for staining directly with rapid Auramine O fluorescent stain (AO stain, Ourchem, Sinopharm Chemical Reagent Co. Ltd., Shanghai, China) for detecting AFB by microscopy (15). Sputum for AFB detection was also collected during the hospitalization. The sample was considered AFB-positive if AFB was detected in at least one bronchial brush or sputum smear microscopy.
BALF
All patients were assessed with an Olympus electronic bronchoscopy system (1T260, Tokyo, Japan). The bronchoscopy findings revealed that TBTB showed highly variable tracheobronchial appearance, and it was classified into seven subtypes according to pathological changes reported by Chung et al. (16): actively caseating, edematous-hyperemic, fibrostenotic, tumorous, granular, ulcerative, and non-specific bronchitis type. Bronchoalveolar lavage was obtained directly from the lesion location. The total amount of 37°C saline, approximately 100 mL, was injected in equal doses, 20 mL in each dose, and finally, the lavage fluid was immediately recovered by vacuum suction (−100~-150 mmHg).
MTB Culture and Drug Sensitivity Test
BALF specimens and sputum were processed using the BACTEC MGIT 960 rapid system (Becton Dickinson, Oxford, UK) for MTB culture (17). If the MTB culture was positive, the drug sensitivity was automatically tested through the BACTEC MGIT 960 SIRE kit (Becton Dickinson) (17).
Xpert MTB/RIF Assay
Five milliliters of BALF specimen were taken and mixed evenly with a sample reagent buffer (SR) (containing sodium hydroxide and isopropanol) in the ratio of 2:1. The mixture was left to stand at room temperature for 15 min and then transferred to the MTB/RIF detection kit (Cepheid, USA). The reaction box was placed in the Gene Xpert instrument (Cepheid) for detection. The Xpert MTB/RIF assay has been designed to detect MTB and also mutations that confer resistance to rifampicin by using three specific primers and five unique molecular probes that target the 81-bp core region of the bacterial RNA polymerase β subunit (rpoB) gene (18–20).
Statistical Analysis
IBM SPSS version 20.0 software (SPSS Inc., Chicago, IL, USA) was used for the statistical analysis. The sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of the different diagnostic methods were calculated with two-sided 95% confidence intervals (CIs). The diagnostic performance of the different diagnostic methods for various bronchial subtypes was compared with chi-square tests. A P-value of <0.05 was considered to be statistically significant.
Results
Patient Characteristics
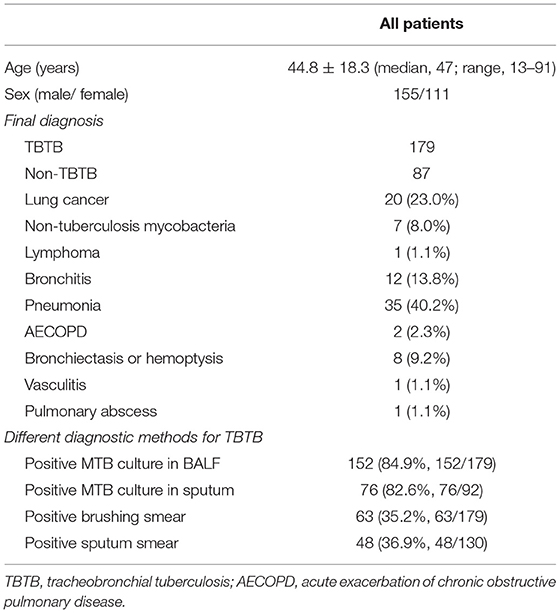
A total of 266 patients (155 males and 111 females) were included in the final analysis. Of these 266 patients, 179 patients were diagnosed to have TBTB. The age of the patients ranged from 13 to 91 years (mean age: 44.8 years) (Table 1). The diagnostic yield of the different diagnostic methods is shown in Table 1. The details of the remaining 87 patients with non-TBTB are also provided in Table 1. The diagnostic yield of the Xpert MTB/RIF assay in different specimens obtained from TBTB patients is shown in Supplementary Table. As Table 1 shown, 87 cases were found to be other diagnosis among people suspected of having TBTB since in the differential diagnosis.
Among the patients of TBTB, as for one of the initial treatment options in our patient, totally 89 patients underwent therapeutic bronchoscopy. Under bronchoscopy, if there was caseating necrosis or granular nodules or mucosal congestion and edema, cryotherapy via bronchoscope was performed; while if there was fibrous stenosis, balloon dilatation via bronchoscope was performed. None of the patients in this study received corticosteroid therapy for TBTB.
Diagnostic Efficacy of Conventional Techniques and the Xpert MTB/RIF Assay in BALF for TBTB
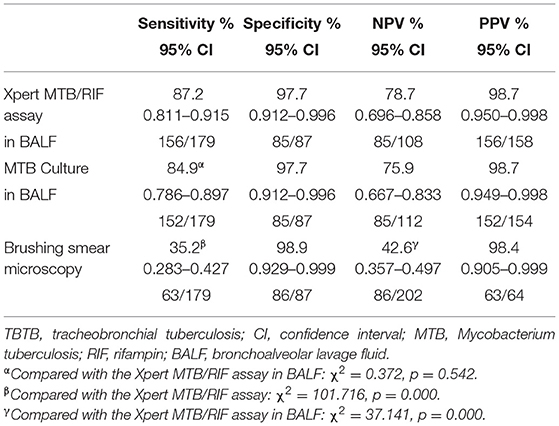
The diagnostic performance of the conventional techniques and the Xpert MTB/RIF assay in BALF for TBTB is shown in Table 2. Bronchial brush smear showed AFB positivity in 35.2% (63/179, 95% CI 0.283–0.427) of TBTB patients, while MTB culture in BALF specimens was positive in 84.9% (152/179, 95% CI 0.786–0.897) of TBTB patients (Table 2). The sensitivity and specificity of the Xpert MTB/RIF assay in BALF to detect TBTB were 87.2% (156/179, 95% CI 0.811–0.915) and 97.7% (85/87, 95% CI 0.912−0.996), respectively (Table 2). The sensitivity and NPV of the Xpert MTB/RIF assay in BALF were significantly higher than those of bronchial brush smear microscopy (χ2 = 101.716, p < 0.001; χ2 = 37.141, p < 0.001) (Table 2). No difference in sensitivity was observed between the Xpert MTB/RIF assay and MTB culture in BALF (χ2 = 0.372, p = 0.542) (Table 2).
Comparison of Diagnostic Consistency of the Different Diagnostic Techniques
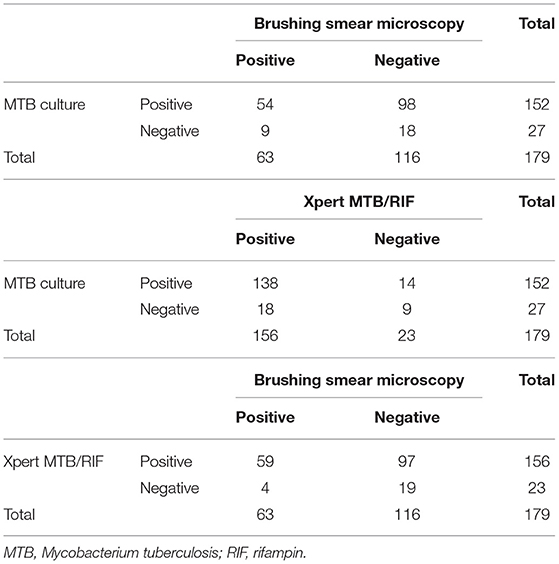
The diagnostic consistencies of the three different diagnostic techniques were compared (Table 3). The bronchial brush smear was not consistent with MTB culture in BALF (Kappa coefficient = 0.009, p = 0.826) and with the Xpert MTB/RIF assay in BALF (Kappa coefficient = 0.075, p = 0.055). However, the MTB culture and the Xpert MTB/RIF assay showed a fair agreement in BALF samples (Kappa coefficient = 0.257, p = 0.001). These findings revealed that the Xpert MTB/RIF assay in BALF, MTB culture in BALF, and bronchial brush smear showed a certain complementarity in their diagnostic performance for TBTB patients.
Diagnostic Yield of the Xpert MTB/RIF Assay According to Bronchoscopic TBTB Subtypes
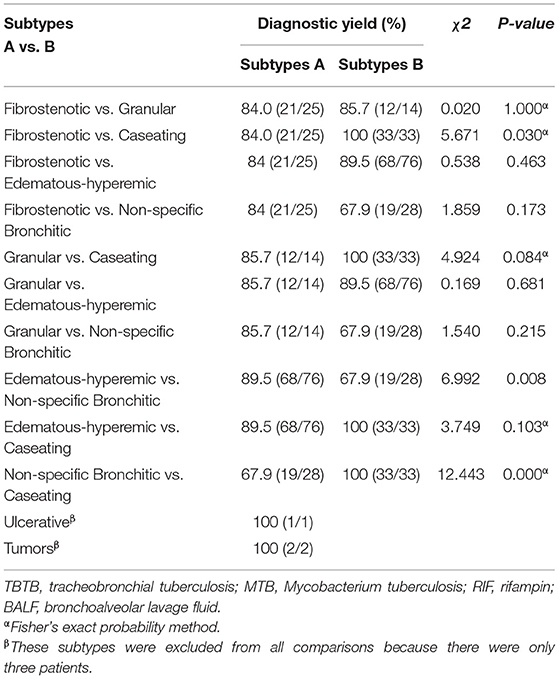
The diagnostic yield of the Xpert MTB/RIF assay in different bronchoscopic TBTB subtypes were as follows: 100% (33/33) in an actively caseating subtype, 85.7% (12/14) in a granular subtype, 84% (21/25) in a fibrostenotic subtype, 89.5% (68/76) in an edematous-hyperemic subtype, and 67.9% (19/28) in a non-specific bronchitic subtype. The diagnostic performances of the Xpert MTB/RIF assay in BALF were compared for different bronchoscopic subtypes (Table 4). The tumorous and ulcerative subtypes were not included because our sample contained only three cases of these subtypes. The diagnostic rate of the Xpert MTB/RIF assay in BALF for the actively caseating subtype was significantly higher than that for the fibrostenotic subtype (χ2 = 5.671, p = 0.030) (Table 4). Moreover, the diagnostic rate of the Xpert MTB/RIF assay in BALF for the non-specific bronchitic subtype was significantly lower than that for the actively caseating subtype (χ2 = 12.443, p < 0.001) or for the edematous-hyperemic subtype (χ2 = 6.992, p = 0.008) (Table 4).
RIF Resistance in TBTB Patients Based on the Xpert MTB/RIF Assay
Of the Xpert MTB/RIF-positive TBTB patients, 74 patients were also confirmed by the MGIT 960 system for drug susceptibility test. Twenty of the 74 patients with TBTB (27.0%) were found to be resistant to RIF, which was not significantly different from the result obtained for a traditional drug susceptibility test (25.7% (19/74); χ2 = 0.035, p = 0.852). The Xpert MTB/RIF assay and the traditional drug sensitivity test showed high consistency in detecting whether TBTB patients were resistant to RIF (Kappa coefficient = 0.965, p < 0.001).
Discussion
The diagnosis and treatment of TBTB are often delayed, resulting in the development of bronchial stenosis. Therefore, a timely and accurate diagnosis of TBTB is critical. Bronchoalveolar lavage is a less invasive and technically easy method for diagnosis. In our present study, we found that the diagnostic efficacy of the Xpert MTB/RIF assay in BALF was similar to that of the MTB culture in BALF but much faster for the initiation of the treatment.
To date, sputum smear examination for AFB is considered to be a popular and rapid bacteriological examination method for diagnosing TBTB. However, it is worth noting that more than one-third of TBTB patients have negative sputum smears (21, 22), which is similar to the result of our present study (the negative smear rate was 63.1%). The bronchial brush smear also did not show satisfactory results; the detection rate was 35.2% for TBTB, similar to that reported in previous studies (23), which indicated the limited improvement in diagnosis efficacy. Although MTB culture is considered as the gold standard for diagnosing TB, it usually takes 2–8 weeks to produce results, while the Xpert MTB/RIF assay is less time-consuming (24). This retrospective study showed that the sensitivity of the Xpert MTB/RIF assay did not differ from that of MTB culture in BALF for diagnosing TBTB. The sensitivity and specificity of the GeneXpert system for diagnosing TB in an intermediate burden city were reported to be 80 and 98%, respectively (25). The PPV and NPV of the test were 92.3 and 95.1%, respectively. Therefore, the Xpert MTB/RIF assay can accurately detect MTB in BALF and prevent complications of bronchial stenosis more rapidly by enabling early proper treatment.
In our previous study (13), the Xpert MTB/RIF assay was performed on samples of bronchial brush and biopsy tissues from TBTB patients. The sensitivity of the Xpert MTB/RIF assay in BALF for TBTB patients was 87.2%, which was higher than that for bronchial brush samples (57.4%; χ2 = 24.822, p = 0.000), while the sensitivity of the Xpert MTB/RIF assay in BALF for TBTB was higher than that of the Xpert MTB/RIF assay in biopsy tissue in our previous study (63.9%, χ2 = 16.097, p = 0.000) (Supplementary Table). The comparison of the sensitivity, specificity, NPV, and PPV showed that the Xpert system had a more satisfactory “all-round” performance. The results obtained from different studies based on different diagnostic tests on different bronchoscopic samples, however, seem to be controversial. The comparison of AFB positivity showed that bronchial aspirate/bronchoalveolar lavage produced the highest diagnostic yield (51.5%) as compared to endobronchial biopsy (37.3%), transbronchial lung biopsy (45.5%), and transbronchial needle aspiration (33%) (26). For patients with pleural TB, where it is difficult to obtain sputum even after induction in some patients, bronchial lavage AFB smear, culture, and the Xpert assay were positive in 9.5, 17.9, and 26.2% patients, respectively (27). Bronchial lavage provided an immediate diagnosis in 22 (26.2%) patients (27). Bronchial lavage, although not a surrogate to pleural biopsy, offers an additional approach to the early diagnosis of pleural TB in patients unable to produce sputum. In addition to being a diagnostic tool, this method also has epidemiological significance in containing TB epidemic (27). When non-neoplastic and neoplastic bronchopulmonary lesions were pooled together, bronchial brushing showed good sensitivity (80.9%) and specificity (85.7%) as compared to bronchial washing, which had sensitivity and specificity of 47.6 and 71.4%, respectively, for the final pulmonary cytology (28). Thus, the diagnostic efficacy of the Xpert assay in different bronchoscopic samples needs to be evaluated in large-scale samples and for different TB groups. The difference in the diagnostic yield of the Xpert assay in BALF and bronchoscopic brush or biopsies might be due to the presence of necrosis or inflammatory cells that are not representative of the pathological condition. Bronchial brushing and biopsy are relatively invasive procedures, in which bleeding is a common complication. However, bronchoalveolar lavage is a procedure with a low risk of injury and bleeding to patients compared with biopsy procedure. Combined with its satisfactory “all round” performance, the Xpert assay in BALF might be a good choice for diagnosing patients suspected to have TBTB. In spite of the above advantages, what should be stressed is that among the 266 suspected TBTB patients, 87 cases were found to be other diagnosis among people suspected of having TBTB since in the differential diagnosis. Thus, differentiating from other possibilities is necessary for patients suspecting TBTB, and for this purpose, bronchial brushing and biopsy would be necessary for the confirmed diagnosis, especially when malignancies could not be excluded. In the clinical setting that TBTB is highly suspected, especially for the subtype with relatively low diagnostic yield by conventional methods, Xpert assay in BALF could be served as an alternative to bronchial brushing and biopsy.
Among the bronchoscopic subtypes, the actively caseating subtype occurs in the early stage of disease progression (16), and its diagnosis rate with the Xpert MTB/RIF assay in BALF was higher than that for the fibrostenotic subtype in our study, which is commonly considered to be the later stage of the disease (16). Previous studies have also shown that the actively caseating, edematous-hyperemic, and fibrostenotic subtypes have different degrees of bronchial lumen stenosis (16) and the worst prognosis (29). Our study showed that the diagnostic rate of the Xpert MTB/RIF assay in BALF for the actively caseating or edematous-hyperemic subtype was higher than that for the non-specific bronchitic subtype; this finding indicating the potential influence of different specific characteristics of different bronchoscopic subtypes on the diagnostic yield of Xpert MTB/RIF assay in BALF. For the earlier and more active stage, the diagnosis rate would be higher, while for the later and more fibrostenotic state, the diagnosis rate might be lower. And our finding further demonstrates the superior diagnostic value of the Xpert MTB/RIF assay in BALF for TBTB patients with bronchial lumen stenosis. Another merit of the Xpert MTB/RIF assay is that it could rapidly detect whether TBTB patients were resistant to RIF. Consistent with related reports, the Xpert MTB/RIF assay showed a high agreement with traditional drug sensitivity tests in our study (30–32).
The present study has several limitations. First, this was a retrospective study based on electronic medical records as the source of patient data. Therefore, more prospective studies are needed. Second, most TBTB cases were concomitant TBTB in pulmonary TB patients. However, patients with obvious pulmonary involvement instead of TBTB alone were excluded in order to evaluate the diagnostic efficacy of Xpert MTB/RIF assay in BALF. Thus, the sample size was small, especially for the special subtypes of ulcerative and tumor. The corresponding results of TBTB patients with tumor subtype and ulcer subtype were not analyzed due to the extremely small sample size. Third, the sensitivity of the Xpert MTB/RIF assay in BALF, brushing tissues, and biopsy tissues from TBTB patients could not be directly compared.
In conclusion, for patients suspected to have TBTB, the Xpert MTB/RIF assay in BALF is a more sensitive method to detect MTB than the conventional methods of bronchial brush smear. BALF could be considered as an alternative to biopsy tissue and brushings for the Xpert MTB/RIF assay for TBTB patients. The Xpert MTB/RIF assay in BALF showed a satisfactory “all round” performance, which could enable an early appropriate treatment.
Data Availability Statement
The original contributions generated for this study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author/s.
Ethics Statement
The study was approved by the Ethics Committee of China Medical University and Shenyang Chest Hospital. The ethics committee exempted obtaining informed consent from patients because of the retrospective nature of the study.
Author Contributions
GH conceived the study. GH and YL designed the study. QingZ and CL conducted bronchial brush and bronchoalveolar lavage. YS and QingZ wrote the manuscript. GH, YL, QingZ, CL, YS, QinZ, HZ, and YF contributed to data collection and data analysis. All authors read and approved the final manuscript.
Funding
This research was supported by the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (No. 2020-PT320-001), and National Natural Science Foundation of China (No. 81900040, 81700041).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2021.682107/full#supplementary-material
References
2. Shim Y. Endobronchial tuberculosis. Respirology (Carlton, Vic.). (1996) 1:95–106. doi: 10.1111/j.1440-1843.1996.tb00017.x
3. Lee J, Park S, Lee D, Shin D, Yang S, Yoo B. Endobronchial tuberculosis. Clinical and bronchoscopic features in 121 cases. Chest. (1992) 102:990–4. doi: 10.1378/chest.102.4.990
4. Jung S, Park H, Kim J, Kim S. Incidence and clinical predictors of endobronchial tuberculosis in patients with pulmonary tuberculosis. Respirology (Carlton, Vic.). (2015) 20:488–95. doi: 10.1111/resp.12474
5. Su Z, Cheng Y, Wu Z, Zhang P, Chen W, Zhou Z, et al. Incidence and predictors of tracheobronchial tuberculosis in pulmonary tuberculosis: a multicentre, large-scale and prospective study in Southern China. Respiration. (2019) 97:153–9. doi: 10.1159/000492335
6. Siow W, Lee P. Tracheobronchial tuberculosis: a clinical review. J Thorac Dis. (2017) 9:E71–7. doi: 10.21037/jtd.2017.01.49
7. Faisal M, Harun H, Hassan T, Ban A, Chotirmall S, Abdul Rahaman J. Treatment of multiple-level tracheobronchial stenosis secondary to endobronchial tuberculosis using bronchoscopic balloon dilatation with topical mitomycin-C. BMC Pulmonary Med. (2016) 16:53. doi: 10.1186/s12890-016-0209-1
8. Albert R, Petty T. Endobronchial tuberculosis progressing to bronchial stenosis. Fiberoptic bronchoscopic manifestations. Chest. (1976) 70:537–9. doi: 10.1378/chest.70.4.537
9. Kay A, González Fernández L, Takwoingi Y, Eisenhut M, Detjen AK, Steingart KR, et al. Xpert MTB/RIF and Xpert MTB/RIF Ultra assays for active tuberculosis and rifampicin resistance in children. Cochrane Database Syst Rev. (2020) 8:CD013359. doi: 10.1002/14651858.CD013359.pub2
10. Pan X, Yang S, Deighton M, Qu Y, Hong L, Su F. A comprehensive evaluation of Xpert MTB/RIF assay with bronchoalveolar lavage fluid as a single test or combined with conventional assays for diagnosis of pulmonary tuberculosis in china: a two-center prospective study. Front Microbiol. (2018) 9:444. doi: 10.3389/fmicb.2018.00444
11. Lu Y, Zhu Y, Shen N, Tian L, Sun Z. Evaluating the diagnostic accuracy of the Xpert MTB/RIF assay on bronchoalveolar lavage fluid: a retrospective study. Int J Infect Dis. (2018) 71:14–9. doi: 10.1016/j.ijid.2018.01.030
12. Saini I, Mukherjee A, Gautam H, Singla M, Jat KR, Lodha R, et al. Diagnostic yield of Xpert MTB/RIF in bronchoalveolar lavage in children with probable pulmonary tuberculosis. Indian Pediatr. (2018) 55:1062–5. doi: 10.1007/s13312-018-1443-9
13. Zhang Q, Zhang Q, Sun B, Liu C, Su AN, Wang XH, et al. GeneXpert MTB/RIF for rapid diagnosis and rifampin resistance detection of endobronchial tuberculosis. Respirology (Carlton, Vic.). (2018) 23:950–5. doi: 10.1111/resp.13316
14. Cheng L, Gu Y, Gui X, Fang Y, Wang H, Sha W. Diagnostic value of virtual bronchoscopic navigation in the bronchial tuberculosis induced central airway stenosis. Infect Dis Ther. (2020) 9:165–74. doi: 10.1007/s40121-020-00283-9
15. Hendry C, Dionne K, Hedgepeth A, Carroll K, Parrish N. Evaluation of a rapid fluorescent staining method for detection of mycobacteria in clinical specimens. J Clin Microbiol. (2009) 47:1206–8. doi: 10.1128/JCM.02097-08
16. Chung H, Lee J. Bronchoscopic assessment of the evolution of endobronchial tuberculosis. Chest. (2000) 117:385–92. doi: 10.1378/chest.117.2.385
17. He Y, Gong Z, Zhao X, Zhang D, Zhang Z. Mycobacterium tuberculosis comprehensive determination of and from targeted capture sequencing. Front Cell Infect Microbiol. (2020) 10:449. doi: 10.3389/fcimb.2020.00449
18. Sánchez-Cabral O, Santillán-Díaz C, Flores-Bello Á, Herrera-Ortega MI, Sandoval-Gutiérrez JL, Santillán-Doherty P, et al. GeneXpert MTB/RIF assay with transbronchial lung cryobiopsy for Mycobacterium tuberculosis diagnosis. Ann Transl Med. (2020) 8:351. doi: 10.21037/atm.2020.02.100
19. Allahyartorkaman M, Mirsaeidi M, Hamzehloo G, Amini S, Zakiloo M, Nasiri M. Low diagnostic accuracy of Xpert MTB/RIF assay for extrapulmonary tuberculosis: a multicenter surveillance. Sci Rep. (2019) 9:18515. doi: 10.1038/s41598-019-55112-y
20. Wang F, Liu K, Peng J, Luo Y, Tang G, Lin Q, et al. Combination of Xpert MTB/RIF and TBAg/PHA ratio for prompt diagnosis of active tuberculosis: a two-center prospective cohort study. Front Med. (2020) 7:119. doi: 10.3389/fmed.2020.00119
21. Sahin F, Yildiz P. Characteristics of endobronchial tuberculosis patients with negative sputum acid-fast bacillus. J Thorac Dis. (2013) 5:764–70. doi: 10.3978/j.issn.2072-1439.2013.12.15
22. Qingliang X, Jianxin W. Investigation of endobronchial tuberculosis diagnoses in 22 cases. Eur J Med Res. (2010) 15:309–13. doi: 10.1186/2047-783X-15-7-309
23. Um S, Yoon Y, Lee S, Yim JJ, Yoo CG, Chung HS, et al. Predictors of persistent airway stenosis in patients with endobronchial tuberculosis. Int J Tuberc Lung Dis. (2008) 12:57–62. doi: 10.1258/ijsa.2007.007095
24. Horne D, Kohli M, Zifodya J, Schiller I, Dendukuri N, Tollefson D, et al. Xpert MTB/RIF and Xpert MTB/RIF Ultra for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev. (2019) 6:CD009593. doi: 10.1002/14651858.CD009593.pub4
25. To K, Kam K, Chan D, Yip WH, Chan KP, Lo R, et al. Utility of GeneXpert in analysis of bronchoalveolar lavage samples from patients with suspected tuberculosis in an intermediate-burden setting. J Infect. (2018) 77:296–301. doi: 10.1016/j.jinf.2018.06.011
26. Kumar R, Singh M, Gupta N, Goel N. Bronchoscopy in immediate diagnosis of smear negative tuberculosis. Pneumonol Alergol Pol. (2014) 82:410–4. doi: 10.5603/PiAP.2014.0053
27. Sumalani K, Akhter N, Ahmed M, Chawla D, Rizvi N. Diagnostic implications of bronchial lavage in patients with pleural tuberculosis. Adv Respir Med. (2020) 88:389–93. doi: 10.5603/ARM.a2020.0149
28. Choudhury M, Singh S, Agarwal S. Efficacy of bronchial brush cytology and bronchial washings in diagnosis of non neoplastic and neoplastic bronchopulmonary lesions. Turk Patoloji Derg. (2012) 28:142–6. doi: 10.5146/tjpath.2012.01113
29. Mondoni M, Repossi A, Carlucci P, Centanni S, Sotgiu G. Bronchoscopic techniques in the management of patients with tuberculosis. Int J Infect Dis. (2017) 64:27–37. doi: 10.1016/j.ijid.2017.08.008
30. Han Y, Xiao N, Huang S, Qin M, Che N, Liu Z. The application of Xpert Mycobacterium tuberculosis/Rifampicin, quantitative polymerase chain reaction and high resolution melting curve in the diagnosis of superficial lymph node TB. Curr Pharm Biotechnol. (2019) 20:1044–54. doi: 10.2174/1389201020666190716104131
31. Patel J, Upadhyay M, Kundnani V, Merchant Z, Jain S, Kire N. Diagnostic efficacy, sensitivity, and specificity of Xpert MTB/RIF assay for spinal tuberculosis and rifampicin resistance. Spine. (2020) 45:163–9. doi: 10.1097/BRS.0000000000003225
Keywords: Xpert MTB/RIF, tracheobronchial tuberculosis, bronchoalveolar lavage, diagnosis, Mycobacterium tuberculosis
Citation: Sun Y, Zhang Q, Zhang Q, Liu C, Zhang H, Fu Y, Liu Y and Hou G (2021) Diagnostic Efficacy of Xpert MTB/RIF Assay in Bronchoalveolar Lavage Fluid for Tracheobronchial Tuberculosis: A Retrospective Analysis. Front. Med. 8:682107. doi: 10.3389/fmed.2021.682107
Received: 17 March 2021; Accepted: 12 July 2021;
Published: 18 August 2021.
Edited by:
Piero Valentini, Università Cattolica del Sacro Cuore, ItalyReviewed by:
Jesica Mazza-Stalder, Centre Hospitalier Universitaire Vaudois (CHUV), SwitzerlandQiao Liu, Nanjing Medical University, China
Copyright © 2021 Sun, Zhang, Zhang, Liu, Zhang, Fu, Liu and Hou. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Gang Hou, aG91Z2FuZ2NtdUAxNjMuY29t; Yongyu Liu, TDI0MzE1NTE2QHNpbmEuY29t
†These authors have contributed equally to this work
 Yue Sun1†
Yue Sun1† Qing Zhang
Qing Zhang Qin Zhang
Qin Zhang Gang Hou
Gang Hou