- 1NanoBioTech Laboratory, Health Systems Engineering, Department of Natural Sciences, Division of Sciences, Florida Polytechnic University, Lakeland, FL, United States
- 2Morris Lifesciences and Technologies, Nagpur, India
- 3Department of Urology, University of Southern California, Los Angeles, CA, United States
- 4Research and Development, Molekule Inc., Tampa, FL, United States
- 5Applied AI Research Laboratory, Department of Computer Science, University of Houston Victoria, Victoria, TX, United States
The emergence of new SARS-CoV-2 variants made the COVID-19 infection pandemic and/or endemic more severe and life-threatening due to ease of transmission, rapid infection, high mortality, and capacity to neutralize the therapeutic ability of developed vaccines. These consequences raise questions on established COVID-19 infection management strategies based on nano-assisted approaches, including rapid diagnostics, therapeutics, and efficient trapping and virus eradication through stimuli-assisted masks and filters composed of nanosystems. Considering these concerns as motivation, this perspective article highlights the role and aspects of nano-enabled approaches to manage the consequences of the COVID-19 infection pandemic associated with newer SARS-CoV-2 variants of concern and significance generated due to mutations. The controlled high-performance of a nanosystem seems capable of effectively detecting new variables for rapid diagnostics, performing site-specific delivery of a therapeutic agent needed for effective treatment, and developing technologies to purify the air and sanitizing premises. The outcomes of this report project manipulative, multifunctional nanosystems for developing high-performance technologies needed to manage consequences of newer SARS-CoV-2 variants efficiently and effectively through an overall targeted, smart approach.
Introduction: Consequences of Emerging Severe Acute Respiratory Syndrome Coronavirus 2 Variants
The Coronavirus disease (COVID-19) pandemic, as reported by the World Health Organization (WHO), caused by a newly discovered coronavirus classified as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), went from a local issue in China in late 2019 to a global one that will leave waves of repercussions for years to come (Kaushik, 2020; Kaushik et al., 2020) not just social, mental, or economic but also the way the healthcare industry and world would treat a viral infection (Yamamoto et al., 2020; Mahapatra et al., 2020). The SARS-CoV-2 is the seventh and newest addition to the SARS family, noted for their crown-like, glycoprotein structure and ability to bind to cells easily. The bio-informatics in the genetic code hidden within the ribonucleic acid (RNA) chain of the virus itself is one huge obstacle that scientists and medical research professionals have had to overcome when looking for a cure or vaccine. Still, another hurdle in drug advancements is the encapsulation of the RNA within the virus itself (Varahachalam et al., 2021). The outer membrane of SARS-CoV-2 is surrounded by the Spike (S)- protein i.e., glycosylated S proteins (Yamamoto et al., 2020; Panda el al., 2020; Sahoo et al., 2021) that have an affinity to bind to angiotensin-converting enzyme-2 (ACE2) and transmembrane protease serine 2 (TMPRSS2) receptors allowing the virus to deposit its RNA into the cell and start replicating until host cell lysis (Kaushik, 2020; Kaushik et al., 2020; Paliwal et al., 2020).
Over 140 million people have been infected by the COVID-19 pandemic (World Health Organization, 2021). These patients experience no symptoms (asymptomatic) to a wide range of symptoms such as loss of olfactory senses, prolonged respiratory distress, neuro-psychiatric issues, brain damage, and death (Yamamoto et al., 2020). Currently developed vaccines by Pfizer-BioNTech, Moderna, and Janssen (Johnson and Johnson) have been approved by the FDA for emergency use only to control COVID-19 infection caused by the severe infectivity of the original SARS-CoV-2 strain. Despite therapeutic significance, the reported adverse effects of these vaccines are not motivating. Sincere efforts are being made to investigate these side-effects and generate guidelines to decide who should get vaccinated and how this can be accomplished. Besides, these vaccines face a new obstacle of showing efficacy for preventing/contracting COVID-19 from emerging SARS-CoV-2 variants via viral mutations that are spreading infection faster than vaccine development and dispersion.
The newer variants of the SARS-CoV-2 pose a threat to a rapid global recovery from COVID-19 infection. The ability of SARS-CoV-2 to reproduce, mutate, and spread suggests that new strains of the virus will be ever-present. The new variants of concern and significance of SARS-CoV-2 from the United Kingdom, Brazil, South Africa, and recently double variants investigated in India are believed to spread more rapidly and, in some cases, be more deadly than the original strain of the virus (Abdool Karim and de Oliveira, 2021). Figures 1A,B show a few examples of the new strains and mutations seen globally. All these variants have taken a foothold in the United States since early 2021 (Walensky et al., 2021), and the number of people infected and the efficacy of the current vaccines against the new strains are unknown. For that reason, we believe that the acute necessity for advancements in nanotechnology (Kaushik, 2019) to slow the spread and return the world to a new normal has never been more apparent.
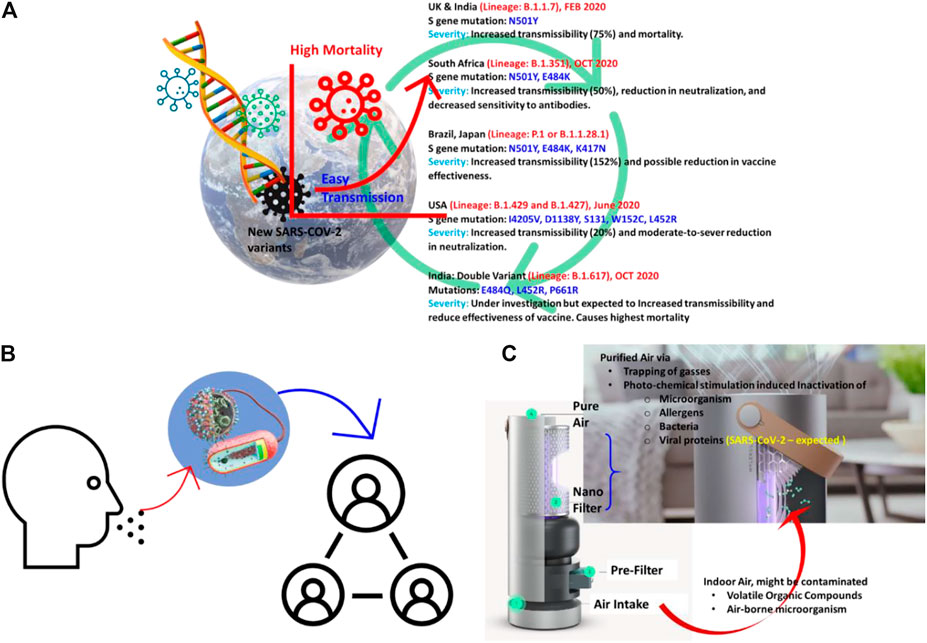
FIGURE 1. Illustration of (A) SARS-COV-2 mutation concerning new variants and their severe effects, (B) transmission of new mutated SARS-CoV-2 in the community, and (C) approach to eradicate virus droplets from the air, especially in indoor air, using an air purifier consisting of a nanoparticle-based filter.
With these severe concerns in mind, this perspective explores the potentials of nanosystems to manage the COVID-19 pandemic/endemic (Phillip, 2021) successfully, where manipulative.
Performance and appearance are vital components. These features involve desired functionality to interact with bioactives, the surface of desired porosity, antiviral and antimicrobial activity, and tunable optical, electrical, magnetic, and molecular properties (Yalcin and Kaushik, 2021; Kaushik, 2020, Kaushik et al., 2020; Paliwal et al., 2020; Ahmadivand et al., 2021). The control and high-performance of smart nanosystems can be useful for 1) efficient surface and membrane systems for trapping and neutralizing the virus, 2) diagnostics unit to detect virus concentration and viral load, and 3) nanotherapeutics to cure COVID-19 infection and manage systems along with post-infection consequences. These nanosystems can also successfully manage the severe consequences of recently investigated newer SARS-CoV-2 variants based on desired performance accomplishments (Kaushik et al., 2020; Paliwal et al., 2020). Besides significant advantages, the challenges and alternative approaches associated with the projected technical field are also discussed in this report.
Efficient Trapping and Eradication of Newer Severe Acute Respiratory Syndrome Coronavirus 2 Variants
The application of nanotechnology to combat newer SARS-CoV-2 variants may increase because effective and efficient surface sanitation has been demonstrated using nanomaterials of improved and tunable antibacterial/antiviral properties. The COVID-19 infection spreads mainly through air molecules; but can rest on various surfaces for hours and can still infect an individual. Considering the airborne transmission mode as a primary concern, it becomes essential to trap the virus-containing droplets, particulates, and aerosol in indoor air using an efficient and effective technology. In this direction, air purifiers are emerging as an alternative technological solution where a filter composition, filtration process, and understanding of airflow dynamics are crucial factors. For example, the air purifier of Molekule IncTM purifies indoor air based on photochemical oxidation processes where the role of a photosensitive nanoparticle is critical (Kaushik et al., 2020) [12] (Figure 1C). This technology has received FDA approval and successfully eradicates viruses, bacteria, allergens, volatile organic compounds, and particulates from indoor air.
New ways of sanitizing surfaces have emerged as a potential approach to eradicate SARS-CoV-2. For example, among nanosystems such as copper, silver, and others, have potential uses as antiviral surface coatings (Talebian et al., 2020; Kaushik et al., 2020) have potential as antivirals. The use of titanium dioxide (TiO2) nanoparticle-based photocatalysts are able to neutralize the virus. The photocatalyst features of TiO2 have been observed to perform better in antiviral/antimicrobial settings with a higher surface area (Sadique et al., 2021). As such, multiple methods of forming and using these devices have been documented. These include, hydrothermal processes, chemical vapor deposition, and a newer method called up-scalable, pulsed-pressure, metal-organic chemical vapor deposition (pp-MOCVD) (Lee et al., 2013). The pp-MOCVD process has been demonstrated to create surfaces with larger surface areas though the process is less proven than epoxy and paint-based TiO2 adhesion solutions (Krumdieck et al., 2019).
Regarding the photostimulation approach, UV-C light has been demonstrated to eliminate coronavirus droplets in the air (Seyer and Sanlidag, 2020). Radiative exposure at a wavelength of 222 nm with an intensity of 2 mJ/cm2 can inactivate more than 99.9% of SARS-CoV-2 protein (Buonanno et al., 2020). Both TiO2 photocatalysts and UV-C sanitation lights are not dependent on molecular structure and work chemically as opposed to a biologically, so there is no reason to suspect that newer strains of COVID-19 infection will exhibit increased resistance to them unless these are demonstrated in future work to be more resistant to UV light or temperature. However, UV-C is known to affect biological function, so a careful approach to design advanced systems is required.
One of the most effective and simple solutions to reduce the spread of COVID-19 is face masks/shields that reduce the transmission of virus caused aerofluid dyanamix i.e., droplets, aerosols, airborne, and particulates containing SARS-CoV-2. Masks, however, vary significantly in design according to dimensions, materials (antiviral/antibacterial/oxidant or a combination of these), and effectiveness (capability of trapping and eradication) (Sadique et al., 2021). In this direction, the N95 masks have been demonstrated to be the most effective at reducing the spread of COVID-19, as they can reduce escaping particles (size 0.3 micron) by 95% and incoming particles by 70%. However, they are challenging to produce in the quantities needed and only work well for a short time. Masks designed to incorporate nanotechnology could be more effective and reusable than existing mask designs (Zangmeister et al., 2020).
Given that masks are a physical technology that blocks viral transmission, standard masks will work just as well for the newer variants as they do on the existing strains of the virus (Kaushik et al., 2020). Masks built in part by or utilizing nanotechnology are also unlikely to be affected because most methods proposed in nanotechnology-based masks are based on chemical reactions that make it difficult for biological structures to pass through or survive the masks. This function is not explicitly targeted for COVID-19. Recently, the development of multifunctional masks containing stimulation responsive and antiviral nanoparticles have been reported to trap and eradicate SARS-CoV-2 efficiently (Kaushik et al., 2020; Yalcin and Kaushik, 2021). However, the development of such masks for a larger population can be of concern. For example, the safety for the wearer and of end of life disposition of nanoparticle masks should be considered carefully as discussed by the recent recall of some graphene containing masks.
Efficient Detection of Newer Severe Acute Respiratory Syndrome Coronavirus 2 Variants
One way to reduce the spread of COVID-19 is via rapid detection, contact tracing and monitoring, and isolation/quarantine. To achieve this, a large volume of tests that can deliver accurate information quickly are needed. Rapid diagnostic methods of detecting SARS-CoV-2 selectively, either qualitatively or quantitively, are well-established and have proven to help control virus transmission (Kaushik et al., 2020; Ahmadivand et al., 2021; Morales-Narváezand Dincer, 2020). The gold standard currently used is real-time polymerase chain reaction (RT-PCR) to deliver accurate information but it is time-consuming (Giovannini et al., 2021; Rahimi et al., 2021). In contrast, other methods such as the rapid antigen testing can be done quickly but can only detect a large count of COVID-19 antigens from the concentrations in the range of 1484 copies/mL to 780 × 106 copies/mL (Fenollar et al., 2021). Recently, antibody-based immunosensing approaches, including electrical, optical, capacitance, and magneto-plasmonic techniques, have efficiently detected SARS-CoV-2 protein and allowed rapid detection of COVID-19 viral protein at pM to fM levels in a small volume of sample (Kaushik et al., 2020; Jain et al., 2021). High-performance optoelectronic nanosystems have been introduced in the above discussed diagnostic approaches to achieve better performance and develop a miniaturized system suitable for point-of-care (POC) testing. The POC diagnostics of the COVID-19 pandemic, supported by the internet of medical things (IoMT) are highly recommended to successfully manage this pandemic (Kaushik et al., 2020; Jain et al., 2021). The IoMT approach will enable rapid bioinformatics sharing with medical centers for timely analysis to make timely therapeutic decisions. However, as these systems need to be tested using real-world samples, systematic validation is a remaining challenge before recommending these diagnostics techniques for clinical application.
Regarding newer SARS-CoV-2 variant detection, the virus structure associated with a mutated form is overall, very similar to the original SARS-CoV-2 protein. Thus, the currently available testing systems are expected to be effective for efficient diagnostics of the COVID-19 pandemic. However, cross-validation has been suggested by health agencies. Besides, new types of biomarkers such as the CRISPR system (Rahimi et al., 2021) can be a choice to detect new SARS-CoV-2 selectively.
Personalized Nanomedicine to Manage Newer Severe Acute Respiratory Syndrome Coronavirus 2 Variants
Personalized nanomedicine is emerging as the future therapy for the prevention and treatment of viral diseases. Due to the acceptability and high performance of advanced nanomedicine, a targeted viral infection can be treated and managed efficiently. This is due to the site-specific delivery of an antiviral cargo of high efficacy improving health and making real-time evaluation possible (Kaushik et al., 2018a; Kaushik et al., 2018b; Kaushik, 2020; Varahachalam et al., 2021). The sustainability of nanoparticles in a biological system (up to 100 days) is critical in detecting and monitoring a targeted disease and related symptoms. For example, if microarray-equipped nanoparticles are introduced into a patient’s body that can perform the role of a monitoring assay system at varying times during the extent of stay in the biological system before excretion, constant monitoring and detection of the virus would be possible. This approach would allow medical professionals to successfully administer personalized healthcare regimens to their patients who ultimately become infected. The microarray nanoparticles would not require expensive or extensive lab equipment in the delivery system, thus allowing widespread and constant surveillance of the global healthcare system. This approach is emerging essential as nanoparticles are known to interact with biomolecules inside the body while circulating and how this affects a nanoparticle’s specificity toward the targeted analyte. Thus, the to understand the toxicokinetic behavior of nanoparticles in the body, especially for long-term exposure, reliable in vivo models should be explored. Utilizing this modern approach to medicine connected with the IoMT and artificial intelligence (AI), the world's preparedness to handle this type of biological invasion could effectively eradicate this virus and future strains of the virus (Varahachalam et al., 2021; Kaushik, 2020).
A multi-drug loaded, time-release nanotechnology approach to recognize the different biomarkers of SARS-CoV-2 and the appropriate materials to deliver as antiviral treatment to the affected area can be utilized to treat the symptoms associated with COVID-19, as well as the virus itself. This multifaceted approach has shown viability in the treatment of HIV and in penetrating the blood-brain barrier (BBB) (Varahachalam et al., 2021; Kaushik, 2020) without adverse effects, offering possibilities in treating the brain damage associated with the viral infection. With lethal variants of the SARS-CoV-2 continually emerging, the development of this technology to treat this virus and its long-term effects is the most promising approach in antiviral care.
Support of Artificial Intelligence and Machine Learning to Detect Severe Acute Respiratory Syndrome Coronavirus 2 Variants
AI and machine learning (ML) data science technologies have been introduced to get more insight into the COVID-19 pandemic (Kaushik et al., 2020). The study of the spread of newer SARS-CoV-2 variants and updated contact tracing methods is of utmost importance to better manage the pandemic. However, researchers and experts have not fully realized the potential highly complex pattern of SARS-CoV-2 replication. The risk of mortality in patients infected with the newer SARS-CoV-2 variants is currently a major concern. On April 6, 2021, researchers at Stanford University identified the first known case of a newer SARS-CoV-2 variant with two mutations (double mutation) in the United States which was first reported in India on March 2020 (Crist, 2021). Among the factors that the WHO has considered for the operational definition for designating a variant of concern (VOCs) are as follows (Challen et al., 2021):
1) Increased transmissibility or detrimental change in the COVID-19 epidemiology.
2) Increase in virulence or change in clinical presentation.
3) Decreased effectiveness of social measures and public health or available diagnostics, vaccines, and therapeutics.
According to a recent study (Malone et al., 2020), AI can be used to predict blueprints for designing universal vaccines against SARS-CoV-2 that contain a sufficiently broad repertoire of T-cell epitopes capable of providing coverage and protection across the global population. Researchers are looking for ways to stay a step ahead of these variants with the help of AI/ML. The advanced artificial neural network can potentially analyze virus mutations and create a vaccine to match them. A protein sequence is put into a deep neural network model, it can quickly identify whether the input is correct for vaccine subunits (Yang et al., 2021). There is also a need for highly accurate vaccine boosters which are effective and compatible with different ethnicities and races of humans. This opens various aspects to AI and ML researchers to perform predictive analytics, ML modeling, and other AI-assisted computer models to speed up and provide more accessible genetic instructions in vaccine development.
Discussion, Challenges and Viewpoints
The possible projection of nano-biotechnology approaches for the successful management of COVID-19 infection originated due to newer SARS-CoV-2 variants and is illustrated in Figure 2. The new variants of SARS-CoV-2 due to mutation(s) in the virus are associated with various regional and genomic conditions and are emerging as a major concern as they facilitate COVID-19 infection with high mortality. It has been demonstrated that available patented technology, rapid detection systems, and optimized therapies serve as an efficient approach to managing COVID-19 infection in a personalized manner. However, a careful and critical review of available diagnostics and therapeutic approaches is suggested by various health experts. In this direction, nano-enabled approaches need to be updated with reference to a targeted challenge.

FIGURE 2. Projected nano-biotechnology-assisted approaches to manage the severe effects of COVID-19 infection pandemic/endemic or both caused by newer SARS-CoV-2 variants.
Besides, an important point is the inhalation of nanoparticles if used in access during the fabrication of masks and coatings on surfaces. Another scenario, in the case of nanomedicine and nano-assisted targeted delivery aspects, is that the selected nanoparticle may affect the human cellular function that may lead to an unintended disorder. In both the cases i.e., inhalation and nano-bio interface, the patient may have severe side effect associated with respiratory system, neurological disorder, organ dysfunction, etc. Thus, it is crucial to explore the bio-safety aspects of a nanosystem prior to adopting it to develop systems for fighting against SARS-CoV-2 infection and transmission (or COVID-19 pandemic).
As newer SARS-CoV-2 variants transmit rapidly through the aerosol and droplet mechanisms. This raises the urgency of developing effective nano-enabled systems that can trap and eradicate new variants of virus using external stimulation (e.g., photochemical oxidation) and efficient membranes suitable to develop masks and filters of air purification systems. An efficient and high-performance SARS-CoV-2 diagnostic sensor that can be rapidly re-calibrated to address the concerns of newer virus strains is also needed. For increasing the selectivity, considerable attention should be paid to developing biomarkers specific to newer variants of SARS-CoV-2. These biomarkers will be helpful in developing early stage selective diagnostics and evaluating the efficacy of a given therapy. Since COVID-19 infection due to newer SARS-CoV-2 variants can be of higher mortality, this raises the need to re-evaluate the efficacy of optimized therapies and developed vaccines. In case of infection, a nanomedicine consisting of therapeutic agents of higher efficacy must be developed for site-specific delivery with long-acting therapeutic performance. The COVID-19 infection behaves differently among people due to variation in geographical region, race, gender, and age. Therefore, optimizing personalized approaches for detection and treatment is of immediate need and can be achieved by developing selective markers while introducing nanotechnology for optimal performance. Although developed vaccines are effective, but not as expected due to varying virus strains. Thus, engineered vaccine design is urgently required to make them selective and effective against new variants.
Such proposed approaches are time-consuming as a lot of scientific studies are required followed up by a plan of clinical trials. Giving preference to infection severity, the role of AI/ML and IoMT becomes crucial (Jain et al., 2021; Kumar et al., 2021). The AI/ML will undoubtedly help predict the accuracy of models expected to perform in the desired manner. Moreover, the assistance of IoMT will be helpful to introduce telehealth/telemedicine for timely therapy decisions and the sharing of bioinformatics with medical centers to establish a correlation between viral infection progression and human health (Jain et al., 2021; Kumar et al., 2021).
For managing the COVID-19 pandemic more effectively, this perspective article highlights the role of high-performance nanotechnology with manipulative features to manage the consequences of newer SARS-CoV-2 variants efficiently and in a timely manner. The capabilities of nano-assisted protective systems, current testing systems, and therapeutics are critically reviewed in this report. The authors believe that re-evaluation and recalibration is required to confirm the efficiency of these developed systems. Besides, considerable efforts should be made to test the efficacy of the investigated biomarkers regarding new variants. The investigation of new biomarkers, such as CRISPR, is also required to develop more efficient systems to manage the COVID-19 pandemic/endemic or both.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Author Contributions
All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
Conflict of Interest
JD is employed with Molekule Inc., United States and KM is employed with Morris Lifesciences and Technologies, Nagpur, Maharashtra, India.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The handling Editor declared a past co-authorship with the authors AK and JD.
Acknowledgments
The authors acknowledge their respective departments and institutions for providing facilities and support.
References
Abdool Karim, S. S., and de Oliveira, T. (2021). New SARS-CoV-2 Variants - Clinical, Public Health, and Vaccine Implications. N. Engl. J. Med. 384, 1866–1868. doi:10.1056/NEJMc2100362
Ahmadivand, A., Gerislioglu, B., Ramezani, Z., Kaushik, A., Manickam, P., and Ghoreishi, S. A. (2021). Functionalized Terahertz Plasmonic Metasensors: Femtomolar-Level Detection of SARS-CoV-2 Spike Proteins. Biosens. Bioelectron. 177, 112971. doi:10.1016/j.bios.2021.112971
Buonanno, M., Welch, D., Shuryak, I., and Brenner, D. J. (2020). Far-UVC Light (222 Nm) Efficiently and Safely Inactivates Airborne Human Coronaviruses. Sci. Rep. 10, 10285. doi:10.1038/s41598-020-67211-2
Challen, R., Brooks-Pollock, E., Read, J. M., Dyson, L., Tsaneva-Atanasova, K., and Danon, L. (2021). Risk of Mortality in Patients Infected with SARS-CoV-2 Variant of Concern 202012/1: Matched Cohort Study. BMJ 372, n579. doi:10.1136/bmj.n579
Crist, C. (2021). New Double Mutation Variant from India Found in US. WebMD. Available at: https://www.webmd.com/vaccines/covid-19-vaccine/news/20210405/new-double-mutation-variant-from-india-found-in-us (Accessed April 18, 2021). New York City, New York.
Fenollar, F., Bouam, A., Ballouche, M., Fuster, L., Prudent, E., Colson, P., et al. (2021). Evaluation of the Panbio COVID-19 Rapid Antigen Detection Test Device for the Screening of Patients with COVID-19. J. Clin. Microbiol. 59, e02589-e02620. doi:10.1128/JCM.02589-20
Giovannini, G., Haick, H., and Garoli, D. (2021). Detecting COVID-19 from Breath: A Game Changer for a Big Challenge. ACS Sens. 6, 1408–1417. doi:10.1021/acssensors.1c00312
Jain, S., Nehra, M., Kumar, R., Dilbaghi, N., Hu, T., Kumar, S., et al. (2021). Internet of Medical Things (IoMT)-Integrated Biosensors for point-of-care Testing of Infectious Diseases. Biosens. Bioelectron. 179, 113074. doi:10.1016/j.bios.2021.113074
Kaushik, A. (2019). Biomedical Nanotechnology Related Grand Challenges and Perspectives. Front. Nanotechnol. 1, 1. doi:10.3389/fnano.2019.00001
Kaushik, A., Jayant, R. D., Bhardwaj, V., and Nair, M. (2018a). Personalized Nanomedicine for CNS Diseases. Drug Discov. Today 23, 1007–1015. doi:10.1016/j.drudis.2017.11.010
Kaushik, A., Jayant, R. D., and Nair, M. (2018b). Nanomedicine for neuroHIV/AIDS Management. Nanomedicine 13, 669–673. doi:10.2217/nnm-2018-0005
Kaushik, A. K., Dhau, J. S., Gohel, H., Mishra, Y. K., Kateb, B., Kim, N.-Y., et al. (2020). Electrochemical SARS-CoV-2 Sensing at point-of-care and Artificial Intelligence for Intelligent COVID-19 Management. ACS Appl. Bio Mater. 3 (11), 7306–7325. doi:10.1021/acsabm.0c01004
Kaushik, A. (2020). Manipulative Magnetic Nanomedicine: the Future of COVID-19 Pandemic/endemic Therapy. Expert Opin. Drug Deliv., 1–4. doi:10.1080/17425247.2021.1860938
Krumdieck, S. P., Boichot, R., Gorthy, R., Land, J. G., Lay, S., Gardecka, A. J., et al. (2019). Nanostructured TiO2 Anatase-Rutile-Carbon Solid Coating with Visible Light Antimicrobial Activity. Sci. Rep. 9, 1883. doi:10.1038/s41598-018-38291-y
Kumar, S., Nehra, M., Khurana, S., Dilbaghi, N., Kumar, V., Kaushik, A., et al. (2021). Aspects of point-of-care Diagnostics for Personalized Health Wellness. Ijn 16, 383–402. doi:10.2147/IJN.S267212
Lee, D., Krumdieck, S., and Talwar, S. D. (2013). Scale-up Design for Industrial Development of a PP-MOCVD Coating System. Surf. Coat. Technol. 230, 39–45. doi:10.1016/j.surfcoat.2013.06.064
Mahapatra, S. R., Sahoo, S., Dehury, B., Raina, V., Patro, S., Misra, N., et al. (2020). Designing an Efficient Multi-Epitope Vaccine Displaying Interactions with Diverse HLA Molecules for an Efficient Humoral and Cellular Immune Response to Prevent COVID-19 Infection. Expert Rev. Vaccin. 19 (9), 871–885. doi:10.1080/14760584.2020.1811091
Malone, B., Simovski, B., Moliné, C., Cheng, J., Gheorghe, M., Fontenelle, H., et al. (2020). Artificial Intelligence Predicts the Immunogenic Landscape of SARS-CoV-2 Leading to Universal Blueprints for Vaccine Designs. Sci. Rep. 10, 22375. doi:10.1038/s41598-020-78758-5
Morales-Narváez, E., and Dincer, C. (2020). The Impact of Biosensing in a Pandemic Outbreak: COVID-19. Biosens. Bioelectron. 163, 112274. doi:10.1016/j.bios.2020.112274
Paliwal, P., Sargolzaei, S., Bhardwaj, S. K., Bhardwaj, V., Dixit, C., and Kaushik, A. (2020). Grand Challenges in Bio-Nanotechnology to Manage the COVID-19 Pandemic. Front. Nanotechnol. 2:571284. doi:10.3389/fnano.2020.571284
Panda, P. K., Arul, M. N., Patel, P., Verma, S. K., Luo, W., Rubahn, H.-G., et al. (2020). Structure-based Drug Designing and Immunoinformatics Approach for SARS-CoV-2. Sci. Adv. 6 (28), eabb8097. doi:10.1126/sciadv.abb8097
Phillips, N. (2021). The Coronavirus Is Here to Stay - Here's what that Means. Nature 590, 382–384. doi:10.1038/d41586-021-00396-2
Rahimi, H., Salehiabar, M., Barsbay, M., Ghaffarlou, M., Kavetskyy, T., Sharafi, A., et al. (2021). CRISPR Systems for COVID-19 Diagnosis. ACS Sens. 6, 1430–1445. doi:10.1021/acssensors.0c02312
Sadique, M. A., Yadav, S., Ranjan, P., Verma, S., Salammal, S. T., Khan, M. A., et al. (2021). High-performance Antiviral Nano-Systems as a Shield to Inhibit Viral Infections: SARS-CoV-2 as a Model Case Study. J. Mater. Chem. B. doi:10.1039/D1TB00472G
Sahoo, S., Mahapatra, S. R., Parida, B. K., Rath, S., Dehury, B., Raina, V., et al. (2021). DBCOVP: a Database of Coronavirus Virulent Glycoproteins. Comput. Biol. Med. 129, 104131. doi:10.1016/j.compbiomed.2020.104131
Seyer, A., and Sanlidag, T. (2020). Solar Ultraviolet Radiation Sensitivity of SARS-CoV-2. The Lancet Microbe 1 (1), e8–e9. doi:10.1016/S2666-5247(20)30013-6
Talebian, S., Wallace, G. G., Schroeder, A., Stellacci, F., and Conde, J. (2020). Nanotechnology-based Disinfectants and Sensors for SARS-CoV-2. Nat. Nanotechnol. 15 (8), 618–621. doi:10.1038/s41565-020-0751-0
Varahachalam, S. P., Lahooti, B., Chamaneh, M., Bagchi, S., Chhibber, T., Morris, K., et al. (2021). Nanomedicine for the SARS-CoV-2: State-Of-The-Art and Future Prospects. Ijn 16, 539–560. doi:10.2147/IJN.S283686
Walensky, R. P., Walke, H. T., and Fauci, A. S. (2021). SARS-CoV-2 Variants of Concern in the United States-Challenges and Opportunities. JAMA 325 (11), 1037–1038. doi:10.1001/jama.2021.2294
World Health Organization (2021). WHO Coronavirus (COVID-19) Dashboard. Available at: https://covid19.who.int (Accessed April 18, 2021).
Yalcin, H. C., and Kaushik, A. (2021). Support of Intelligent Emergent Materials to Combat COVID-19 Pandemic. Emergent Mater. 4, 1–2. doi:10.1007/s42247-021-00189-3
Yamamoto, V., Bolanos, J. F., Fiallos, J., Strand, S. E., Morris, K., Shahrokhinia, S., et al. (2020). COVID-19: Review of a 21st Century Pandemic from Etiology to Neuro-Psychiatric Implications. JAD 77, 459–504. doi:10.3233/JAD-200831
Yang, Z., Bogdan, P., and Nazarian, S. (2021). An In Silico Deep Learning Approach to Multi-Epitope Vaccine Design: a SARS-CoV-2 Case Study. Sci. Rep. 11, 3238. doi:10.1038/s41598-021-81749-9
Keywords: COVID-19 pandemic, SARS-COV-2 variants, nanosystems, biosensors, nanomedicine, artificial intelligence, intelligent healthcare, personalized COVID-19 management
Citation: Gage A, Brunson K, Morris K, Wallen SL, Dhau J, Gohel H and Kaushik A (2021) Perspectives of Manipulative and High-Performance Nanosystems to Manage Consequences of Emerging New Severe Acute Respiratory Syndrome Coronavirus 2 Variants. Front. Nanotechnol. 3:700888. doi: 10.3389/fnano.2021.700888
Received: 27 April 2021; Accepted: 31 May 2021;
Published: 17 June 2021.
Edited by:
Yogendra Kumar Mishra, University of Southern Denmark, DenmarkReviewed by:
Suresh K. Verma, KIIT University, IndiaAnkit Kumar Dubey, Indian Institute of Technology Madras, India
Copyright © 2021 Gage, Brunson, Morris, Wallen, Dhau, Gohel and Kaushik. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ajeet Kaushik, YWplZXQubnBsQGdtYWlsLmNvbQ==
†These authors have contributed equally to this work
 Alexander Gage
Alexander Gage Kaitlyn Brunson1†
Kaitlyn Brunson1† Kevin Morris
Kevin Morris Scott L. Wallen
Scott L. Wallen Jaspreet Dhau
Jaspreet Dhau Hardik Gohel
Hardik Gohel Ajeet Kaushik
Ajeet Kaushik