- 1Department of Molecular & Cell Biology, University of California, Berkeley, Berkeley, CA, United States
- 2Helen Wills Neuroscience Institute, University of California, Berkeley, Berkeley, CA, United States
Microglia are the resident, innate immune cells of the central nervous system (CNS) and are critical in managing CNS injuries and infections. Microglia also maintain CNS homeostasis by influencing neuronal development, viability, and function. However, aberrant microglial activity and phenotypes are associated with CNS pathology, including autism spectrum disorder (ASD). Thus, improving our knowledge of microglial regulation could provide insights into the maintenance of CNS homeostasis as well as the prevention and treatment of ASD. Control of microglial activity is in part overseen by small, lipid-derived molecules known as endogenous cannabinoids (endocannabinoids). Endocannabinoids are one component of the endocannabinoid system (ECS), which also includes the enzymes that metabolize these ligands, in addition to cannabinoid receptor 1 (CB1) and 2 (CB2). Interestingly, increased ECS signaling leads to an anti-inflammatory, neuroprotective phenotype in microglia. Here, we review the literature and propose that ECS signaling represents a largely untapped area for understanding microglial biology and its relationship to ASD, with special attention paid to issues surrounding the use of recreational cannabis (marijuana). We also discuss major questions within the field and suggest directions for future research.
Introduction
Microglia represent a self-sustaining population of cells that originates from the yolk sac and colonizes the brain in utero (Alliot et al., 1999; Ginhoux et al., 2010; Bruttger et al., 2015; Askew et al., 2017; Huang et al., 2018). Microglia are the resident immune cells of the central nervous system (CNS) and thus are the first line of defense against CNS infection and injury. For example, they phagocytize debris and pathogens as well as initiate neuroinflammatory responses through release of cytokines such as interleukin-1 (IL-1), IL-6, and tumor necrosis factor alpha (TNF-α) (Yang et al., 2010; Janda et al., 2018). They also recruit natural killer cells, macrophages, and lymphocytes to sites of infection and injury (Yang et al., 2010). Moreover, microglia influence the health of their local environment through release of neurotrophic and neurotoxic factors (Nakajima et al., 2001; Srinivasan et al., 2004; Parkhurst et al., 2013).
In addition to the aforementioned roles, microglia carry out other functions essential for CNS homeostasis (Saijo and Glass, 2011; Butovsky and Weiner, 2018; Lenz and Nelson, 2018). Specifically, these cells oversee neurogenesis by both phagocytizing and directing the migration of newborn neurons (Sierra et al., 2010; Ribeiro Xavier et al., 2015). Microglia also regulate neuronal connections by engulfing excessive synaptic structures through use of the classical complement cascade (Stevens et al., 2007). Lastly, microglia modulate neuronal plasticity via release of neurotrophins such as brain-derived neurotrophic factor (BDNF) (Parkhurst et al., 2013). Early wiring of the brain requires tight control of these processes and therefore microglia critically impact CNS development (Paolicelli et al., 2011; Bialas and Stevens, 2013; Shigemoto-Mogami et al., 2014).
Due to their wide-ranging contributions to CNS homeostasis and development, microglia displaying irregular activity and/or phenotypes can lead to disorders of the CNS (Salter and Stevens, 2017; Butovsky and Weiner, 2018). In this review, we focus on microglial dysfunction as it relates to autism spectrum disorder and associated conditions. We also discuss the role of the endogenous cannabinoid system in modulating microglial involvement in these disorders.
Microglia and Autism Spectrum Disorder
The Autism and Developmental Disabilities Monitoring Network estimates the current prevalence of autism spectrum disorder (ASD) to be 1 in 59 among children in the United States (Baio et al., 2018). ASD denotes a collection of heterogeneous neurodevelopmental disorders defined by (1) repetitive, restricted behaviors and interests and (2) abnormalities in socio-communication (American Psychiatry Association, 2013). Thus, ASD is an umbrella term and it encompasses several disorders including autism, Asperger’s syndrome, pervasive developmental disorder not otherwise specified, and childhood disintegrative disorder (American Psychiatry Association, 2013). Conditions frequently comorbid with ASD include intellectual disability, attention-deficit/hyperactivity disorder, epilepsy, perturbed sleep patterns, aggression, anxiety, and altered sensory perception (Leitner, 2014; Srivastava and Schwartz, 2014; Fakhoury, 2015; Park et al., 2016; Postorino et al., 2016). These associated conditions can vary in severity and be more or less common within patient subsets. Finally, due to the lack of available therapeutics for ASD, there is a continuous, pressing need for investigation into the causes and progression of the disorder.
Given their role in CNS development and neuroinflammation, microglia are poised to influence the pathogenesis of ASD (Edmonson et al., 2016; Salter and Stevens, 2017; Lenz and Nelson, 2018). Evidence for microglial involvement in ASD comes from both post-mortem- and positron-emission tomography (PET)-imaging studies which show increased neuroinflammation and numbers of activated microglia in brains of ASD patients (Vargas et al., 2005; Morgan et al., 2010; Suzuki et al., 2013). More recently, a large-scale analysis of transcriptomic datasets from post-mortem cerebral cortex has revealed a distinct microglial signature in ASD brains (Gandal et al., 2018). This is concordant with previous observations of microglial activation-related gene enrichment in ASD brain-derived gene networks (Voineagu et al., 2011).
Altered synaptic density is observed in post-mortem ASD brain tissue (Hutsler and Zhang, 2010) and ASD mouse models (Comery et al., 1997; Tang et al., 2014; Wang et al., 2017). These alterations are presumably due to deficits in developmental synaptic pruning (Hansel, 2019). Indeed, current thinking posits that microglia can exert control over the progression of ASD through synaptic pruning dysregulation (Di Marco et al., 2016; Lenz and Nelson, 2018). This hypothesis is supported by the finding that inhibiting microglial autophagy leads to increased synaptic density and reduced sociability in mice (Kim et al., 2017). Moreover, mice with loss of microglia-enriched fractalkine receptor CX3C-chemokine receptor 1 (CX3CR1) display impaired synaptic pruning and reduced social interactions (Zhan et al., 2014). Additional support for the involvement of microglia in ASD pathogenesis comes from studies on mouse models of Rett syndrome (RTT), a syndromic form of ASD caused by mutations in the gene encoding methyl-CpG binding protein 2 (MECP2) (Lombardi et al., 2015). In one model of RTT, neuronal, but not microglial, loss of Mecp2 leads to excessive synaptic engulfment by microglia in later stages of the disease (Schafer et al., 2016). This suggests that neuronal loss of MECP2 is sufficient to induce aberrant microglial activity and it is consistent with the observation that deletion of Mecp2 using a Cx3cr1-Cre line does not produce RTT-like symptoms (Wolf et al., 2017). Furthermore, Mecp2-null microglia produce toxic levels of glutamate that damage post-synaptic structures in vitro (Maezawa and Jin, 2010). Still, due to the phenotypic and genetic heterogeneity of ASD, it remains to be seen if these findings in RTT models are representative of autism etiology in general.
Finally, children born to mothers who experience infections or autoimmune disease during their pregnancies are more likely to develop ASD (Jiang et al., 2016; Careaga et al., 2017). This phenomenon, known as maternal immune activation (MIA), has been phenocopied in rodent models (Shi et al., 2003; Patterson, 2011; Careaga et al., 2017; Salter and Stevens, 2017). While embryonic microglia may mediate the neuroinflammatory consequences of MIA (Salter and Stevens, 2017), how this underlies ASD remains unclear.
Considered together, the aforementioned findings implicate microglia as targets for the treatment of ASD. Due to its anti-inflammatory effects, the endogenous cannabinoid (endocannabinoid) system represents a promising tool for modulating microglial involvement in ASD. We next provide a brief overview of the endocannabinoid system and then summarize the evidence linking microglial-endocannabinoid signaling to ASD, with attention paid to issues surrounding the use of recreational cannabis.
The Architecture of the Endocannabinoid System
The endocannabinoid system (ECS) exerts control over microglial activity and therefore shows promise for treating CNS dysfunction (Benito et al., 2008; Stella, 2009; Lisboa et al., 2016). The ECS consists of three major components: (1) small, lipid-derived endocannabinoids (eCBs), (2) the enzymes responsible for synthesizing and degrading eCBs, and (3) the metabotropic receptors that recognize eCBs (Lutz et al., 2015). The most well-known eCBs in the brain are N-arachidonoylethanolamine (AEA or anandamide) and 2-arachidonoylglycerol (2-AG) (Lutz et al., 2015; Parsons and Hurd, 2015). In response to increased cytoplasmic calcium, 2-AG and AEA are synthesized on demand from lipid precursors by the enzymes diacylglycerol lipase (DAGL) and N-acyl-phosphatidylethanolamine phospholipase D (NAPE-PLD), respectively (Alger and Kim, 2011; Lutz et al., 2015; Parsons and Hurd, 2015). The enzyme primarily responsible for degrading 2-AG is monoacylglycerol lipase (MAGL), whereas AEA is catabolized by fatty acid amide hydrolase (FAAH). In the CNS, these components are expressed in neurons, microglia, astrocytes, and oligodendrocytes (Figure 1; Stella, 2009; Lutz et al., 2015; Ilyasov et al., 2018). The two main receptors for eCBs include cannabinoid receptors 1 (CB1) and 2 (CB2), both of which are G protein-coupled (Parsons and Hurd, 2015). Finally, while CB1 is enriched in neurons, CB2 expression is primarily restricted to microglia (Stella, 2009).
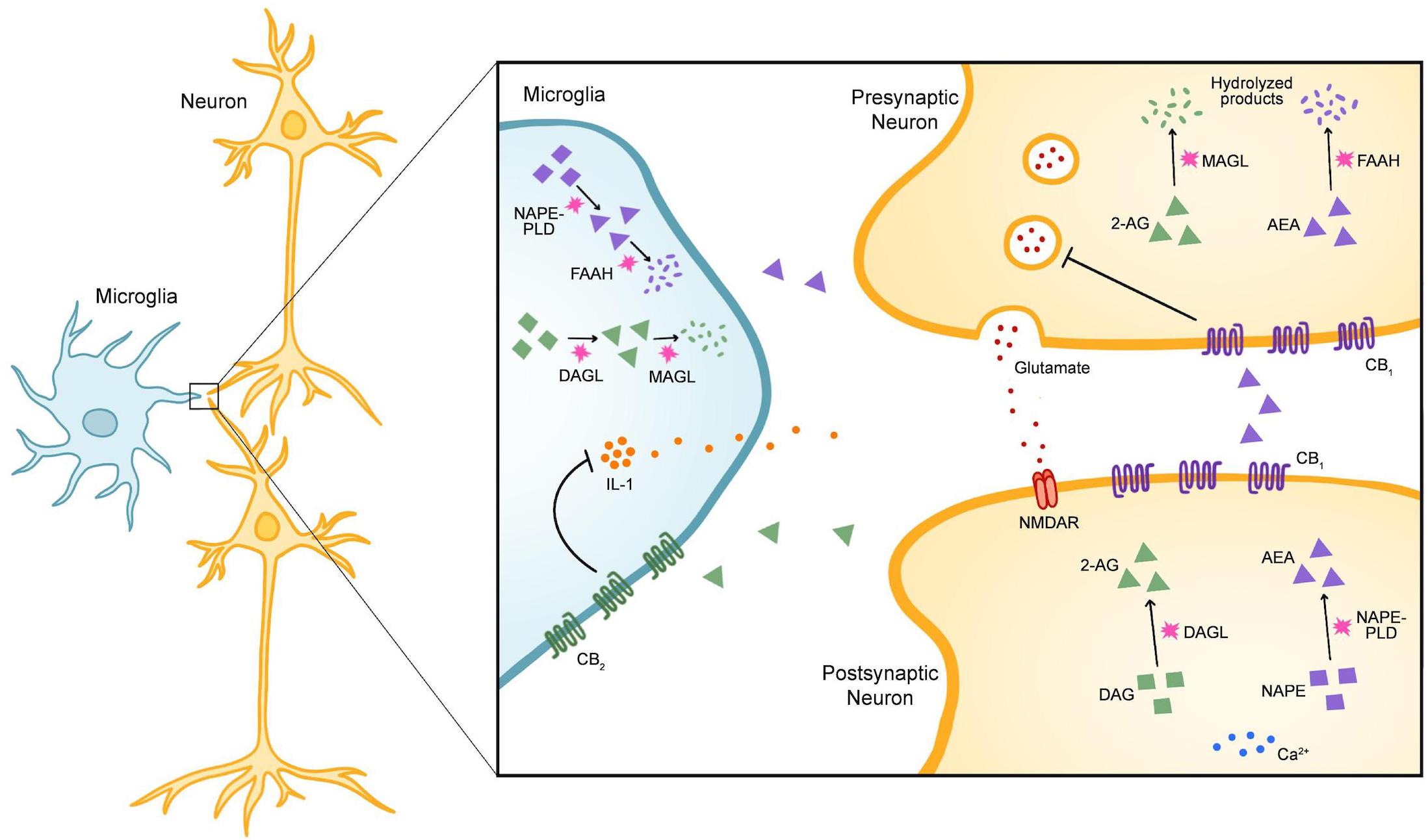
Figure 1. The Components of the Endogenous Cannabinoid System in Microglia and Neurons. In the central nervous system (CNS), the endogenous cannabinoids (eCBs) N-arachidonoylethanolamine (AEA) and 2-arachidonoylglycerol (2-AG) are the most widely-recognized ligands of the endogenous cannabinoid (endocannabinoid) system (ECS). The two main receptors for eCBs are cannabinoid receptor 1 (CB1) and cannabinoid receptor 2 (CB2), both of which are G-protein coupled. Within the CNS, eCB signaling is classically understood to modulate synaptic activity. In the example given here, release of glutamate from presynaptic neurons activates N-methyl-D-aspartate receptors (NMDARs) in postsynaptic neurons. In response to increased cytoplasmic calcium, the enzyme diacylglycerol lipase (DAGL) catalyzes the synthesis of 2-AG from diacylgylcerol (DAG) and N-acyl-phosphatidylethanolamine phospholipase D (NAPE-PLD) catalyzes the synthesis of AEA from the precursor N-acylphosphatidylethanolamine (NAPE). After 2-AG and AEA are released into the synaptic cleft, they stimulate CB1 receptors on presynaptic neurons and inhibit further neurotransmitter release. 2-AG is mainly degraded by the enzyme monoacylglycerol lipase (MAGL) whereas AEA is degraded by fatty-acid amide hydrolase (FAAH). While DAGL, NAPE-PLD, MAGL, and FAAH are expressed broadly throughout the CNS, CB1 is enriched in neurons and CB2 is enriched in microglia. Stimulation of CB2 leads to a protective phenotype in microglia that is characterized by a reduction in the release of pro-inflammatory cytokines such as interleukin-1 (IL-1).
Acute consumption of Cannabis sativa (marijuana) yields wide-ranging effects on memory, cognition, appetite, and mood in both humans and rodents (Curran et al., 2016; Kendall and Yudowski, 2016). These effects result from action of the phytocannabinoid (or plant-derived cannabinoid) Δ 9-tetrahydrocannabinol (THC) on CB1 within the brain (Panagis et al., 2014; Curran et al., 2016; Kendall and Yudowski, 2016). Yet, long-term consequences of cannabis use have been poorly studied, especially with regards to microglia and their impact on neuronal circuitry. Notably, increased eCB signaling is associated with an anti-inflammatory, protective phenotype in microglia (Benito et al., 2008; Stella, 2009; Lisboa et al., 2016). For example, pharmacological inhibition of FAAH decreases microglial activation marker expression, cytokine production, and synaptic plasticity deficits, in the hippocampi of aged rats (Murphy et al., 2012). Additionally, stimulation of CB2 inhibits microglial activation and increases striatal neuron survival and motor coordination in a model of Huntington’s disease excitotoxicity (Palazuelos et al., 2009). Moreover, exposure to anti-inflammatory cytokines increases eCB production and CB2 expression in microglia (Mecha et al., 2015). These findings cast the ECS as an attractive target for influencing microglial activity (Dhopeshwarkar and Mackie, 2014; Lisboa et al., 2016; Cassano et al., 2017; Donvito et al., 2018). However, the consequences of manipulating eCB signaling on ASD risk and pathogenesis are largely uncharacterized. The increasing legality and use of cannabis currently seen throughout the world therefore requires a better understanding of eCB signaling in microglia as it relates to ASD.
Microglial-Endocannabinoid Signaling and ASD
Cannabis Use and ASD Risk
Approximately 2.5–5% of people between the ages of 15–64 years old consume cannabis, making it the most popular illicit drug in the world (Gunn et al., 2016). THC readily crosses the fetal-placental barrier (Wu et al., 2011) and is also secreted in breast milk (Perez-Reyes and Wall, 1982). As of now, there is no strong link between prenatal cannabis use and an increased risk of ASD in offspring. Prenatal exposure to cannabis does correlate with negative outcomes in child development, including growth restriction (Zuckerman et al., 1989; El Marroun et al., 2009; Gunn et al., 2016) and decreased cognitive performance (Richardson et al., 2002; Goldschmidt et al., 2004; Gunn et al., 2016). Still, few studies have been carried out on this topic and these observations are not always reproducible (Wu et al., 2011; El Marroun et al., 2018). Since rates of cannabis use in both pregnant and non-pregnant women are steadily increasing (Brown et al., 2017), there is an unmet need for clarifying the relationship between prenatal cannabis exposure and CNS development. Thus, future clinical and pre-clinical investigations should focus on elucidating the long-lasting effects of prenatal cannabis exposure and if these effects are linked to ASD pathogenesis. Such studies should be paired with efforts to determine if prenatal cannabis exposure impacts microglial synaptic pruning and thereby neurodevelopment in general. These experiments could also take place in the context of pathogen-induced MIA to better recapitulate environmental risks for ASD.
Cannabinoid Signaling as a Target for ASD Treatment
Pre-clinical evidence supporting the role of ECS signaling in ASD comes from research on rodent models of MIA and neuroinflammation. For instance, in response to the innate immunostimulant polyinosinic:polycytidylic acid [poly(I:C)], MIA-based production of IL-17 induces abnormal cortical development and ASD-like sociability deficits in mouse offspring (Choi et al., 2016). Interestingly, administration of AEA decreases IL-17 production and increases expression of the anti-inflammatory cytokine IL-10 in a mouse model of immune hypersensitivity (Jackson et al., 2014). Treatment with the innate immunostimulant lipopolysaccharide (LPS) during adolescence leads to increased AEA tone and FAAH activity in the amygdala, as well as decreased sociability, in mice (Doenni et al., 2016). These alterations are attenuated with administration of an FAAH inhibitor (Doenni et al., 2016). Still, the contribution of microglia to either the initiation or resolution of these neuroinflammatory effects is unknown and should be the focus of future endeavors.
Cannabidiol (CBD), the second major phytocannabinoid in cannabis (Atakan, 2012), has gained attention as a possible treatment for ASD (Salgado and Castellanos, 2018). Indeed, three clinical reports have recently established that CBD alleviates major symptoms associated with ASD, including seizures, sleeplessness, and anxiety, in children (Barchel et al., 2018; Aran et al., 2019; Bar-Lev Schleider et al., 2019). In addition, because CBD possess a weak affinity for CB1 and CB2, it has no psychoactive effects and may even prevent some of the harmful consequences of THC (Zuardi et al., 2012; Morales et al., 2017; Mouro et al., 2019). Lastly, a commercially available, oral CBD extract (Epidiolex) has recently gained FDA-approval for treatment of drug-resistant epilepsy (Sekar and Pack, 2019), which can be an additional burden faced by ASD patients (Sansa et al., 2011; Kokoszka et al., 2017; Long et al., 2019). Nevertheless, because exposure to other cannabinoids negatively affects the development of the adolescent brain in rats (Cha et al., 2006; Schneider and Koch, 2007; Quinn et al., 2008), parents and physicians should practice extreme caution when recommending the use of cannabinoids to treat ASD (Atakan, 2012).
Subsequent work in this field must emphasize replicating the usefulness of ECS signaling in ASD via paradigms that include larger and more diverse populations. If these results hold, it will be important to establish if abatement of ASD symptoms is due to eCB signaling in microglia. For example, CBD blocks microglial activation (Martin-Moreno et al., 2011) and neuroinflammation (Elliott et al., 2018; Maroon and Bost, 2018), both of which are linked to seizure susceptibility (Rana and Musto, 2018; Zhao et al., 2018). Consequently, it will be beneficial to establish if CBD-based reduction of epilepsy in ASD patients is reliant on microglial-based mechanisms. Utilizing mice with microglia-specific loss of ECS components in combination with ASD-relevant mouse models could shed light on this area.
Conclusion
Microglia are indispensable orchestrators of CNS development and homeostasis and are therefore likely involved in the pathogenesis of ASD. Microglial activity can be modulated by eCB signaling, which makes the ECS a potentially forceful tool in the prevention and management of CNS dysfunction. Future work must focus on detailing the mechanisms by which altered eCB signaling in microglia yields protective and detrimental effects in the CNS, particularly as it relates to the effects of chronic cannabis use. Answering these questions could provide improved therapeutics for ASD and its associated conditions.
Author Contributions
All authors wrote the manuscript. KT and DA designed the figure.
Funding
This work was supported by a National Institutes of Health (NIH) postdoctoral training grant (T32AI100829-06A1 to DA) and an NIH research project grant (1R01HD092093 to KS).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors would like to thank Dr. Marissa Co for providing invaluable comments in this manuscript.
References
Alger, B. E., and Kim, J. (2011). Supply and demand for endocannabinoids. Trends Neurosci. 34, 304–315. doi: 10.1016/j.tins.2011.03.003
Alliot, F., Godin, I., and Pessac, B. (1999). Microglia derive from progenitors, originating from the yolk sac, and which proliferate in the brain. Brain Res. Dev. Brain Res. 117, 145–152. doi: 10.1016/s0165-3806(99)00113-3
American Psychiatry Association, (2013). Diagnostic and Statistical Manual of Mental Disorders: DSM-5, 5th Edn. Arlington, VA: American Psychiatry Association.
Aran, A., Cassuto, H., Lubotzky, A., Wattad, N., and Hazan, E. (2019). Brief report: cannabidiol-rich cannabis in children with autism spectrum disorder and severe behavioral problems-a retrospective feasibility study. J. Autism Dev. Disord. 49, 1284–1288. doi: 10.1007/s10803-018-3808-2
Askew, K., Li, K., Olmos-Alonso, A., Garcia-Moreno, F., Liang, Y., Richardson, P., et al. (2017). Coupled proliferation and apoptosis maintain the rapid turnover of microglia in the adult brain. Cell Rep. 18, 391–405. doi: 10.1016/j.celrep.2016.12.041
Atakan, Z. (2012). Cannabis, a complex plant: different compounds and different effects on individuals. Ther. Adv. Psychopharmacol. 2, 241–254. doi: 10.1177/2045125312457586
Baio, J., Wiggins, L., Christensen, D. L., Maenner, M. J., Daniels, J., Warren, Z., et al. (2018). Prevalence of autism spectrum disorder among children aged 8 years - autism and developmental disabilities monitoring network, 11 sites, United States, 2014. MMWR Surveill. Summ. 67, 1–23. doi: 10.15585/mmwr.ss6706a1
Barchel, D., Stolar, O., De-Haan, T., Ziv-Baran, T., Saban, N., Fuchs, D. O., et al. (2018). Oral cannabidiol use in children with autism spectrum disorder to treat related symptoms and Co-morbidities. Front. Pharmacol. 9:1521. doi: 10.3389/fphar.2018.01521
Bar-Lev Schleider, L., Mechoulam, R., Saban, N., Meiri, G., and Novack, V. (2019). Real life experience of medical cannabis treatment in autism: analysis of safety and efficacy. Sci. Rep. 9:200. doi: 10.1038/s41598-018-37570-y
Benito, C., Tolon, R. M., Pazos, M. R., Nunez, E., Castillo, A. I., and Romero, J. (2008). Cannabinoid CB2 receptors in human brain inflammation. Br. J. Pharmacol. 153, 277–285.
Bialas, A. R., and Stevens, B. (2013). TGF-beta signaling regulates neuronal C1q expression and developmental synaptic refinement. Nat. Neurosci. 16, 1773–1782. doi: 10.1038/nn.3560
Brown, Q. L., Sarvet, A. L., Shmulewitz, D., Martins, S. S., Wall, M. M., and Hasin, D. S. (2017). Trends in marijuana use among pregnant and nonpregnant reproductive-aged women, 2002-2014. JAMA 317, 207–209.
Bruttger, J., Karram, K., Wortge, S., Regen, T., Marini, F., Hoppmann, N., et al. (2015). Genetic cell ablation reveals clusters of local self-renewing microglia in the mammalian central nervous system. Immunity 43, 92–106. doi: 10.1016/j.immuni.2015.06.012
Butovsky, O., and Weiner, H. L. (2018). Microglial signatures and their role in health and disease. Nat. Rev. Neurosci. 19, 622–635. doi: 10.1038/s41583-018-0057-5
Careaga, M., Murai, T., and Bauman, M. D. (2017). Maternal immune activation and autism spectrum disorder: from rodents to nonhuman and human primates. Biol. Psychiatry 81, 391–401. doi: 10.1016/j.biopsych.2016.10.020
Cassano, T., Calcagnini, S., Pace, L., De Marco, F., Romano, A., and Gaetani, S. (2017). Cannabinoid receptor 2 signaling in neurodegenerative disorders: from pathogenesis to a promising therapeutic target. Front. Neurosci. 11:30. doi: 10.3389/fnins.2017.00030
Cha, Y. M., White, A. M., Kuhn, C. M., Wilson, W. A., and Swartzwelder, H. S. (2006). Differential effects of delta9-THC on learning in adolescent and adult rats. Pharmacol. Biochem. Behav. 83, 448–455. doi: 10.1016/j.pbb.2006.03.006
Choi, G. B., Yim, Y. S., Wong, H., Kim, S., Kim, H., Kim, S. V., et al. (2016). The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science 351, 933–939. doi: 10.1126/science.aad0314
Comery, T. A., Harris, J. B., Willems, P. J., Oostra, B. A., Irwin, S. A., Weiler, I. J., et al. (1997). Abnormal dendritic spines in fragile X knockout mice: maturation and pruning deficits. Proc. Natl. Acad. Sci. U.S.A. 94, 5401–5404. doi: 10.1073/pnas.94.10.5401
Curran, H. V., Freeman, T. P., Mokrysz, C., Lewis, D. A., Morgan, C. J., and Parsons, L. H. (2016). Keep off the grass? Cannabis, cognition and addiction. Nat. Rev. Neurosci. 17, 293–306. doi: 10.1038/nrn.2016.28
Dhopeshwarkar, A., and Mackie, K. (2014). CB2 Cannabinoid receptors as a therapeutic target-what does the future hold? Mol. Pharmacol. 86, 430–437. doi: 10.1124/mol.114.094649
Di Marco, B., Bonaccorso, C. M., Aloisi, E., D’Antoni, S., and Catania, M. V. (2016). Neuro-Inflammatory mechanisms in developmental disorders associated with intellectual disability and autism spectrum disorder: a neuro- immune perspective. CNS Neurol. Disord. Drug Targets 15, 448–463. doi: 10.2174/1871527315666160321105039
Doenni, V. M., Gray, J. M., Song, C. M., Patel, S., Hill, M. N., and Pittman, Q. J. (2016). Deficient adolescent social behavior following early-life inflammation is ameliorated by augmentation of anandamide signaling. Brain Behav. Immun. 58, 237–247. doi: 10.1016/j.bbi.2016.07.152
Donvito, G., Nass, S. R., Wilkerson, J. L., Curry, Z. A., Schurman, L. D., Kinsey, S. G., et al. (2018). The endogenous cannabinoid system: a budding source of targets for treating inflammatory and neuropathic pain. Neuropsychopharmacology 43, 52–79. doi: 10.1038/npp.2017.204
Edmonson, C. A., Ziats, M. N., and Rennert, O. M. (2016). A non-inflammatory role for microglia in autism spectrum disorders. Front. Neurol. 7:9. doi: 10.3389/fneur.2016.00009
El Marroun, H., Brown, Q. L., Lund, I. O., Coleman-Cowger, V. H., Loree, A. M., Chawla, D., et al. (2018). An epidemiological, developmental and clinical overview of cannabis use during pregnancy. Prev. Med. 116, 1–5. doi: 10.1016/j.ypmed.2018.08.036
El Marroun, H., Tiemeier, H., Steegers, E. A., Jaddoe, V. W., Hofman, A., Verhulst, F. C., et al. (2009). Intrauterine cannabis exposure affects fetal growth trajectories: the generation R study. J. Am. Acad. Child Adolesc. Psychiatry 48, 1173–1181. doi: 10.1097/CHI.0b013e3181bfa8ee
Elliott, D. M., Singh, N., Nagarkatti, M., and Nagarkatti, P. S. (2018). Cannabidiol attenuates experimental autoimmune encephalomyelitis model of multiple sclerosis through induction of myeloid-derived suppressor cells. Front. Immunol. 9:1782. doi: 10.3389/fimmu.2018.01782
Fakhoury, M. (2015). Autistic spectrum disorders: a review of clinical features, theories and diagnosis. Int. J. Dev. Neurosci. 43, 70–77. doi: 10.1016/j.ijdevneu.2015.04.003
Gandal, M. J., Haney, J. R., Parikshak, N. N., Leppa, V., Ramaswami, G., Hartl, C., et al. (2018). Shared molecular neuropathology across major psychiatric disorders parallels polygenic overlap. Science 359, 693–697. doi: 10.1126/science.aad6469
Ginhoux, F., Greter, M., Leboeuf, M., Nandi, S., See, P., Gokhan, S., et al. (2010). Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science 330, 841–845. doi: 10.1126/science.1194637
Goldschmidt, L., Richardson, G. A., Cornelius, M. D., and Day, N. L. (2004). Prenatal marijuana and alcohol exposure and academic achievement at age 10. Neurotoxicol. Teratol. 26, 521–532. doi: 10.1016/j.ntt.2004.04.003
Gunn, J. K., Rosales, C. B., Center, K. E., Nunez, A., Gibson, S. J., Christ, C., et al. (2016). Prenatal exposure to cannabis and maternal and child health outcomes: a systematic review and meta-analysis. BMJ Open 6:e009986. doi: 10.1136/bmjopen-2015-009986
Hansel, C. (2019). Deregulation of synaptic plasticity in autism. Neurosci. Lett. 688, 58–61. doi: 10.1016/j.neulet.2018.02.003
Huang, Y., Xu, Z., Xiong, S., Sun, F., Qin, G., Hu, G., et al. (2018). Repopulated microglia are solely derived from the proliferation of residual microglia after acute depletion. Nat. Neurosci. 21, 530–540. doi: 10.1038/s41593-018-0090-8
Hutsler, J. J., and Zhang, H. (2010). Increased dendritic spine densities on cortical projection neurons in autism spectrum disorders. Brain Res. 1309, 83–94. doi: 10.1016/j.brainres.2009.09.120
Ilyasov, A. A., Milligan, C. E., Pharr, E. P., and Howlett, A. C. (2018). The endocannabinoid system and oligodendrocytes in health and disease. Front. Neurosci. 12:733. doi: 10.3389/fnins.2018.00733
Jackson, A. R., Nagarkatti, P., and Nagarkatti, M. (2014). Anandamide attenuates Th-17 cell-mediated delayed-type hypersensitivity response by triggering IL-10 production and consequent microRNA induction. PLoS One 9:e93954. doi: 10.1371/journal.pone.0093954
Janda, E., Boi, L., and Carta, A. R. (2018). Microglial phagocytosis and its regulation: a therapeutic target in Parkinson’s disease? Front. Mol. Neurosci. 11:144. doi: 10.3389/fnmol.2018.00144
Jiang, H. Y., Xu, L. L., Shao, L., Xia, R. M., Yu, Z. H., Ling, Z. X., et al. (2016). Maternal infection during pregnancy and risk of autism spectrum disorders: a systematic review and meta-analysis. Brain Behav. Immun. 58, 165–172. doi: 10.1016/j.bbi.2016.06.005
Kendall, D. A., and Yudowski, G. A. (2016). Cannabinoid receptors in the central nervous system: their signaling and roles in disease. Front. Cell Neurosci. 10:294. doi: 10.3389/fncel.2016.00294
Kim, H. J., Cho, M. H., Shim, W. H., Kim, J. K., Jeon, E. Y., Kim, D. H., et al. (2017). Deficient autophagy in microglia impairs synaptic pruning and causes social behavioral defects. Mol. Psychiatry 22, 1576–1584. doi: 10.1038/mp.2016.103
Kokoszka, M. A., McGoldrick, P. E., La Vega-Talbott, M., Raynes, H., Palmese, C. A., Wolf, S. M., et al. (2017). Epilepsy surgery in patients with autism. J. Neurosurg. Pediatr. 19, 196–207. doi: 10.3171/2016.7.PEDS1651
Leitner, Y. (2014). The co-occurrence of autism and attention deficit hyperactivity disorder in children - what do we know? Front. Hum. Neurosci. 8:268. doi: 10.3389/fnhum.2014.00268
Lenz, K. M., and Nelson, L. H. (2018). Microglia and beyond: innate immune cells as regulators of brain development and behavioral function. Front. Immunol. 9:698. doi: 10.3389/fimmu.2018.00698
Lisboa, S. F., Gomes, F. V., Guimaraes, F. S., and Campos, A. C. (2016). Microglial cells as a link between cannabinoids and the immune hypothesis of psychiatric disorders. Front. Neurol. 7:5. doi: 10.3389/fneur.2016.00005
Lombardi, L. M., Baker, S. A., and Zoghbi, H. Y. (2015). MECP2 disorders: from the clinic to mice and back. J. Clin. Invest. 125, 2914–2923. doi: 10.1172/JCI78167
Long, S., Zhou, H., Li, S., Wang, T., Ma, Y., Li, C., et al. (2019). The clinical and genetic features of co-occurring epilepsy and autism spectrum disorder in chinese children. Front. Neurol. 10:505. doi: 10.3389/fneur.2019.00505
Lutz, B., Marsicano, G., Maldonado, R., and Hillard, C. J. (2015). The endocannabinoid system in guarding against fear, anxiety and stress. Nat. Rev. Neurosci. 16, 705–718. doi: 10.1038/nrn4036
Maezawa, I., and Jin, L. W. (2010). Rett syndrome microglia damage dendrites and synapses by the elevated release of glutamate. J. Neurosci. 30, 5346–5356. doi: 10.1523/JNEUROSCI.5966-09.2010
Maroon, J., and Bost, J. (2018). Review of the neurological benefits of phytocannabinoids. Surg. Neurol. Int. 9:91. doi: 10.4103/sni.sni_45_18
Martin-Moreno, A. M., Reigada, D., Ramirez, B. G., Mechoulam, R., Innamorato, N., Cuadrado, A., et al. (2011). Cannabidiol and other cannabinoids reduce microglial activation in vitro and in vivo: relevance to Alzheimer’s disease. Mol. Pharmacol. 79, 964–973. doi: 10.1124/mol.111.071290
Mecha, M., Feliu, A., Carrillo-Salinas, F. J., Rueda-Zubiaurre, A., Ortega-Gutierrez, S., de Sola, R. G., et al. (2015). Endocannabinoids drive the acquisition of an alternative phenotype in microglia. Brain Behav. Immun. 49, 233–245. doi: 10.1016/j.bbi.2015.06.002
Morales, P., Hurst, D. P., and Reggio, P. H. (2017). Molecular targets of the phytocannabinoids: a complex picture. Prog. Chem. Org. Nat. Prod. 103, 103–131. doi: 10.1007/978-3-319-45541-9_4
Morgan, J. T., Chana, G., Pardo, C. A., Achim, C., Semendeferi, K., Buckwalter, J., et al. (2010). Microglial activation and increased microglial density observed in the dorsolateral prefrontal cortex in autism. Biol. Psychiatry 68, 368–376. doi: 10.1016/j.biopsych.2010.05.024
Mouro, F. M., Miranda-Lourenco, C., Sebastiao, A. M., and Diogenes, M. J. (2019). From cannabinoids and neurosteroids to statins and the ketogenic diet: new therapeutic avenues in rett syndrome? Front. Neurosci. 13:680. doi: 10.3389/fnins.2019.00680
Murphy, N., Cowley, T. R., Blau, C. W., Dempsey, C. N., Noonan, J., Gowran, A., et al. (2012). The fatty acid amide hydrolase inhibitor URB597 exerts anti-inflammatory effects in hippocampus of aged rats and restores an age-related deficit in long-term potentiation. J. Neuroinflammation 9:79. doi: 10.1186/1742-2094-9-79
Nakajima, K., Honda, S., Tohyama, Y., Imai, Y., Kohsaka, S., and Kurihara, T. (2001). Neurotrophin secretion from cultured microglia. J. Neurosci. Res. 65, 322–331. doi: 10.1002/jnr.1157
Palazuelos, J., Aguado, T., Pazos, M. R., Julien, B., Carrasco, C., Resel, E., et al. (2009). Microglial CB2 cannabinoid receptors are neuroprotective in Huntington’s disease excitotoxicity. Brain 132, 3152–3164. doi: 10.1093/brain/awp239
Panagis, G., Mackey, B., and Vlachou, S. (2014). Cannabinoid regulation of brain reward processing with an emphasis on the role of CB1 receptors: a step back into the future. Front. Psychiatry 5:92. doi: 10.3389/fpsyt.2014.00092
Paolicelli, R. C., Bolasco, G., Pagani, F., Maggi, L., Scianni, M., Panzanelli, P., et al. (2011). Synaptic pruning by microglia is necessary for normal brain development. Science 333, 1456–1458. doi: 10.1126/science.1202529
Park, H. R., Lee, J. M., Moon, H. E., Lee, D. S., Kim, B. N., Kim, J., et al. (2016). A short review on the current understanding of autism spectrum disorders. Exp. Neurobiol. 25, 1–13. doi: 10.5607/en.2016.25.1.1
Parkhurst, C. N., Yang, G., Ninan, I., Savas, J. N., Yates, J. R. III, Lafaille, J. J., et al. (2013). Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell 155, 1596–1609. doi: 10.1016/j.cell.2013.11.030
Parsons, L. H., and Hurd, Y. L. (2015). Endocannabinoid signalling in reward and addiction. Nat. Rev. Neurosci. 16, 579–594. doi: 10.1038/nrn4004
Patterson, P. H. (2011). Maternal infection and immune involvement in autism. Trends Mol. Med. 17, 389–394. doi: 10.1016/j.molmed.2011.03.001
Perez-Reyes, M., and Wall, M. E. (1982). Presence of delta9-tetrahydrocannabinol in human milk. N. Engl. J. Med. 307, 819–820. doi: 10.1056/nejm198209233071311
Postorino, V., Fatta, L. M., Sanges, V., Giovagnoli, G., De Peppo, L., Vicari, S., et al. (2016). Intellectual disability in autism spectrum disorder: investigation of prevalence in an italian sample of children and adolescents. Res. Dev. Disabil. 48, 193–201. doi: 10.1016/j.ridd.2015.10.020
Quinn, H. R., Matsumoto, I., Callaghan, P. D., Long, L. E., Arnold, J. C., Gunasekaran, N., et al. (2008). Adolescent rats find repeated Delta(9)-THC less aversive than adult rats but display greater residual cognitive deficits and changes in hippocampal protein expression following exposure. Neuropsychopharmacology 33, 1113–1126. doi: 10.1038/sj.npp.1301475
Rana, A., and Musto, A. E. (2018). The role of inflammation in the development of epilepsy. J. Neuroinflammation 15:144. doi: 10.1186/s12974-018-1192-7
Ribeiro Xavier, A. L., Kress, B. T., Goldman, S. A., Lacerda de Menezes, J. R., and Nedergaard, M. (2015). A distinct population of microglia supports adult neurogenesis in the subventricular zone. J. Neurosci. 35, 11848–11861. doi: 10.1523/JNEUROSCI.1217-15.2015
Richardson, G. A., Ryan, C., Willford, J., Day, N. L., and Goldschmidt, L. (2002). Prenatal alcohol and marijuana exposure: effects on neuropsychological outcomes at 10 years. Neurotoxicol. Teratol. 24, 309–320. doi: 10.1016/s0892-0362(02)00193-9
Saijo, K., and Glass, C. K. (2011). Microglial cell origin and phenotypes in health and disease. Nat. Rev. Immunol. 11, 775–787. doi: 10.1038/nri3086
Salgado, C. A., and Castellanos, D. (2018). Autism spectrum disorder and cannabidiol: have we seen this movie before? Glob. Pediatr. Health 5:2333794X18815412.
Salter, M. W., and Stevens, B. (2017). Microglia emerge as central players in brain disease. Nat. Med. 23, 1018–1027. doi: 10.1038/nm.4397
Sansa, G., Carlson, C., Doyle, W., Weiner, H. L., Bluvstein, J., Barr, W., et al. (2011). Medically refractory epilepsy in autism. Epilepsia 52, 1071–1075. doi: 10.1111/j.1528-1167.2011.03069.x
Schafer, D. P., Heller, C. T., Gunner, G., Heller, M., Gordon, C., Hammond, T., et al. (2016). Microglia contribute to circuit defects in Mecp2 null mice independent of microglia-specific loss of Mecp2 expression. eLife 5:e15224. doi: 10.7554/eLife.15224
Schneider, M., and Koch, M. (2007). The effect of chronic peripubertal cannabinoid treatment on deficient object recognition memory in rats after neonatal mPFC lesion. Eur. Neuropsychopharmacol. 17, 180–186. doi: 10.1016/j.euroneuro.2006.03.009
Sekar, K., and Pack, A. (2019). Epidiolex as adjunct therapy for treatment of refractory epilepsy: a comprehensive review with a focus on adverse effects. F1000Res 8:234. doi: 10.12688/f1000research.16515.1
Shi, L., Fatemi, S. H., Sidwell, R. W., and Patterson, P. H. (2003). Maternal influenza infection causes marked behavioral and pharmacological changes in the offspring. J. Neurosci. 23, 297–302. doi: 10.1523/jneurosci.23-01-00297.2003
Shigemoto-Mogami, Y., Hoshikawa, K., Goldman, J. E., Sekino, Y., and Sato, K. (2014). Microglia enhance neurogenesis and oligodendrogenesis in the early postnatal subventricular zone. J. Neurosci. 34, 2231–2243. doi: 10.1523/JNEUROSCI.1619-13.2014
Sierra, A., Encinas, J. M., Deudero, J. J., Chancey, J. H., Enikolopov, G., Overstreet-Wadiche, L. S., et al. (2010). Microglia shape adult hippocampal neurogenesis through apoptosis-coupled phagocytosis. Cell Stem Cell 7, 483–495. doi: 10.1016/j.stem.2010.08.014
Srinivasan, B., Roque, C. H., Hempstead, B. L., Al-Ubaidi, M. R., and Roque, R. S. (2004). Microglia-derived pronerve growth factor promotes photoreceptor cell death via p75 neurotrophin receptor. J. Biol. Chem. 279, 41839–41845. doi: 10.1074/jbc.m402872200
Srivastava, A. K., and Schwartz, C. E. (2014). Intellectual disability and autism spectrum disorders: causal genes and molecular mechanisms. Neurosci. Biobehav. Rev. 46(Pt 2), 161–174. doi: 10.1016/j.neubiorev.2014.02.015
Stella, N. (2009). Endocannabinoid signaling in microglial cells. Neuropharmacology 56(Suppl. 1), 244–253. doi: 10.1016/j.neuropharm.2008.07.037
Stevens, B., Allen, N. J., Vazquez, L. E., Howell, G. R., Christopherson, K. S., Nouri, N., et al. (2007). The classical complement cascade mediates CNS synapse elimination. Cell 131, 1164–1178. doi: 10.1016/j.cell.2007.10.036
Suzuki, K., Sugihara, G., Ouchi, Y., Nakamura, K., Futatsubashi, M., Takebayashi, K., et al. (2013). Microglial activation in young adults with autism spectrum disorder. JAMA Psychiatry 70, 49–58.
Tang, G., Gudsnuk, K., Kuo, S. H., Cotrina, M. L., Rosoklija, G., Sosunov, A., et al. (2014). Loss of mTOR-dependent macroautophagy causes autistic-like synaptic pruning deficits. Neuron 83, 1131–1143. doi: 10.1016/j.neuron.2014.07.040
Vargas, D. L., Nascimbene, C., Krishnan, C., Zimmerman, A. W., and Pardo, C. A. (2005). Neuroglial activation and neuroinflammation in the brain of patients with autism. Ann. Neurol. 57, 67–81. doi: 10.1002/ana.20315
Voineagu, I., Wang, X., Johnston, P., Lowe, J. K., Tian, Y., Horvath, S., et al. (2011). Transcriptomic analysis of autistic brain reveals convergent molecular pathology. Nature 474, 380–384. doi: 10.1038/nature10110
Wang, M., Li, H., Takumi, T., Qiu, Z., Xu, X., Yu, X., et al. (2017). Distinct defects in spine formation or pruning in two gene duplication mouse models of autism. Neurosci. Bull. 33, 143–152. doi: 10.1007/s12264-017-0111-8
Wolf, Y., Boura-Halfon, S., Cortese, N., Haimon, Z., Sar Shalom, H., Kuperman, Y., et al. (2017). Brown-adipose-tissue macrophages control tissue innervation and homeostatic energy expenditure. Nat. Immunol. 18, 665–674. doi: 10.1038/ni.3746
Wu, C. S., Jew, C. P., and Lu, H. C. (2011). Lasting impacts of prenatal cannabis exposure and the role of endogenous cannabinoids in the developing brain. Future Neurol. 6, 459–480. doi: 10.2217/fnl.11.27
Yang, I., Han, S. J., Kaur, G., Crane, C., and Parsa, A. T. (2010). The role of microglia in central nervous system immunity and glioma immunology. J. Clin. Neurosci. 17, 6–10. doi: 10.1016/j.jocn.2009.05.006
Zhan, Y., Paolicelli, R. C., Sforazzini, F., Weinhard, L., Bolasco, G., Pagani, F., et al. (2014). Deficient neuron-microglia signaling results in impaired functional brain connectivity and social behavior. Nat. Neurosci. 17, 400–406. doi: 10.1038/nn.3641
Zhao, H., Zhu, C., and Huang, D. (2018). Microglial activation: an important process in the onset of epilepsy. Am. J. Transl. Res. 10, 2877–2889.
Zuardi, A. W., Crippa, J. A., Hallak, J. E., Bhattacharyya, S., Atakan, Z., Martin-Santos, R., et al. (2012). A critical review of the antipsychotic effects of cannabidiol: 30 years of a translational investigation. Curr. Pharm. Des. 18, 5131–5140. doi: 10.2174/138161212802884681
Keywords: microglia, endocannabinoids, autism, neuroinflammation, neurodevelopment
Citation: Araujo DJ, Tjoa K and Saijo K (2019) The Endocannabinoid System as a Window Into Microglial Biology and Its Relationship to Autism. Front. Cell. Neurosci. 13:424. doi: 10.3389/fncel.2019.00424
Received: 13 June 2019; Accepted: 03 September 2019;
Published: 18 September 2019.
Edited by:
Yu Tang, Central South University, ChinaReviewed by:
Carmen Guaza, Cajal Institute (CSIC), SpainNagarkatti Prakash, University of South Carolina, United States
Raphael Mechoulam, The Hebrew University of Jerusalem, Israel
Copyright © 2019 Araujo, Tjoa and Saijo. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Kaoru Saijo, a3NhaWpvQGJlcmtlbGV5LmVkdQ==
 Daniel John Araujo1
Daniel John Araujo1 Karensa Tjoa
Karensa Tjoa Kaoru Saijo
Kaoru Saijo