- 1Lung Cancer Center, Cancer Center and State Key Laboratory of Biotherapy, West China Hospital of Sichuan University, Chengdu, Sichuan, China
- 2Laboratory of Integrative Medicine, Clinical Research Center for Breast, State Key Laboratory of Biotherapy, West China Hospital, Sichuan University and Collaborative Innovation Center, Chengdu, Sichuan, China
Cancer therapies have made tremendous progress in the last decade, but monotherapy still has apparent limitations and lacks therapeutic efficacy. Thus, the simultaneous administration of multiple drugs has been widely explored and has shown better outcomes. Exosomes, deriving from almost all living cells, are natural nanocarriers designed to deliver drugs to tumor sites. Therefore, combinational antitumor therapies based on exosomes, such as engineered exosomes and different combinations of chemotherapeutic agents, therapeutic nucleic acids, photosensitizers, immunotherapy and phytochemicals, have considerable prospects and potential for clinical translation. Here, we summarize current strategies of cancer combination therapy in exosomes and propose opportunities and challenges in the future.
1 Introduction
Although antitumor therapies have advanced rapidly, cancer remains one of the leading causes of mortality (Sung et al., 2021). With the discovery of non-coding RNAs (ncRNAs) and programmed death receptor 1/programmed death receptor ligand 1 (PD-1/PD-L1), as well as research on their mechanisms, the therapeutic targets of ncRNAs and immunotherapy have started a new chapter of cancer therapy (Bagchi et al., 2021; Kara et al., 2022). However, the degradation of nucleic acids in vivo and the limited benefits of immune checkpoint inhibitors have become new challenges (Dammes and Peer, 2020; Winkle et al., 2021; Yi et al., 2022). In addition, insufficient efficacy, side effects, rapid clearance, and resistance have limited the long-term treatments of conventional chemotherapeutic agents (Gao et al., 2022). The combination of different therapies emerges as the time required and shows considerable promise.
Exosomes (EXO), a subtype of extracellular vesicles (EVs), are generated from various cells and can be detected in body fluids. They contain proteins, lipids, and ncRNAs (miRNAs, lncRNAs, and circRNAs), which determine their properties and possess vital biological or pathologic functions in intercellular communication (Kalluri and LeBleu, 2020; Paskeh et al., 2022). Their low immunogenicity, high plasticity, biocompatibility, and potential for targeted delivery make it a promising carrier in cancer therapy (Kooijmans et al., 2021). Thus, the double-membraned vesicles started to be used to protect and deliver cargos. They successively took therapeutic agents to tumor tissue and then suppressed tumor proliferation (Thakur et al., 2022).
However, rapid clearance of some exosomes in the blood and multidrug resistance of tumors limit the efficacy of exosome-based drug delivery (Huang et al., 2017; Zhang et al., 2019). To further exert the carrier, combinational antitumor strategies of exosomes have been exploited. They have gradually shifted drug-loaded exosomes from single to combinational therapy to cope with heterogeneous cancer cells through synergistic effects. This review summarizes recent advances in exosomes as carriers for combined cancer therapy, including the methods of engineering modification and different combinations of chemotherapeutic agents, therapeutic nucleic acids (TNAs), photosensitizers, immunotherapy and phytochemicals. We also outline the challenges and new strategies, which will provide guidance for future research on multifunctional exosomes.
2 Engineering modification of exosomes
Exosomes derived from cells are capable of targeting recipient cells through the binding of proteins with ligands and present compatibility to parental cells (Xia et al., 2020). For example, dendritic cell (DC) cell-derived exosomes express ICAM-1, which increases exosome uptake by binding to LFA-1 on the surface of activated T cells (Nolte-'t Hoen et al., 2009). However, low drug delivery efficiency, insufficient infiltration and weakened targeting capability in vivo require multiple modifications to solve these problems (Chen et al., 2022). These deficiencies engineering modification of exosomes endows them with not only superior targeted capacity but also longer circulation time and more muscular infiltration (Kooijmans et al., 2016). Here, we present classic methods and recent advances in the engineering modification of exosomes, including direct and indirect ways, such as click chemistry, anchoring peptides, hybrid vesicles, biomimetic EVs, and genetic manipulation (Figure 1; Supplementary Table S1).
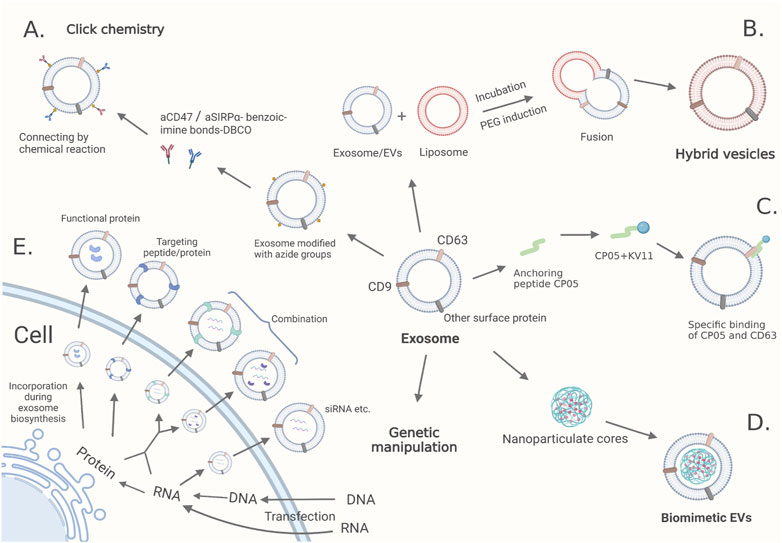
FIGURE 1. The modification methods of engineered exosomes: (A) Click chemistry. Exosomes are modified with azide groups through the intrinsic biosynthesis and metabolic incorporation of phospholipids. Then azide-modified exosomes are conjugated with the dibenzocyclooctynes (DBCO)-modified anti-CD47 antibody (aCD47) and anti-signal regulatory protein alpha (SIRPα) antibody (aSIRPα) linked with pH-sensitive benzoic-imine bonds. (B) Hybrid vesicles. Polyethylene glycol (PEG) and incubation induce efficient fusion between exosomes/EVs and liposomes. (C) Anchoring peptides. Anchoring peptide CP05 linked with KV11 (an anti-angiogenic peptide) bounds specifically to CD63. (D) Biomimetic EVs. Nanoparticulate cores are embedded into the exosomes by electroporation. (E) Genetic manipulation. Transfected nucleic acids or their protein producs are loaded through the intrinsic biosynthesis of exosomes.
2.1 Click chemistry
Click chemistry refers to rapid, spontaneous and highly efficient reactions between two chemical groups allowing for the incorporation of chemical groups with highly selective reactivity into small molecules or protein modifications without perturbing their biological function and with no membrane damage or undesirable side reactions (Peplow, 2019; Parker and Pratt, 2020). Click chemistry has become an essential tool for constructing biomolecular systems (Goyard et al., 2022). Copper-catalyzed azide alkyne cycloaddition, one of click chemistry, is a reaction between an alkyne and an azide that forms a triazole linkage (Goyard et al., 2022). Functional ligands c(RGDyK), the cyclo (Arg-Gly-Asp-D-Tyr-Lys) peptide, are conjugated onto exosomal surfaces through bio-orthogonal copper-free azide alkyne cyclo-addition and show high affinity to integrin αvβ3 in a mouse model (Tian et al., 2018). Tumor cells express CD47, which can hinder recognition by macrophages via the CD47-signal regulatory protein α (SIRPα) signaling pathway (Liu et al., 2015). Dibenzocyclooctyne (DBCO)-modified antibodies against CD47 and SIRPα are conjugated with exosomes modified with azide groups through pH-sensitive benzoic-imine bonds and released from exosomes in an acidic microenvironment as blocking agents (Nie et al., 2020) (Figure 1A). In addition, click chemistry has also been used in exosome tracing and synchronous imaging monitoring (Xu et al., 2020; Qian et al., 2022). However, applications of click chemistry should be accompanied by intelligent strategies because specific chemical structures are needed as prerequisites.
2.2 Hybrid vesicles
Hybrid vesicles are obtained by fusing exosomes/EVs with liposomes, thereby they have the advantages of both (Figure 1B). On the one hand, polyethylene glycol may trigger the fusion of EVs with functionalized liposomes to create novel biosynthetic hybrid vectors, which provides an effective way to camouflage liposomes (Piffoux et al., 2018). On the other hand, hybrid nanoparticles could load large molecules or exogenous cargo much more easily than EVs, and encapsulated genes can be expressed after endocytosis (Lin et al., 2018; Evers et al., 2022). In fact, with a deep understanding of exosome properties, it will be a pivotal method for us to adjust the proportion of different components in hybrid vesicles to produce superior materials. In addition, new techniques, such as charge-mediated hybrid fusion, open more possibilities of applying hybrid vesicles (Marusic et al., 2022).
2.3 Anchoring peptides
Anchoring peptides can be used as versatile targeting tools with the capacity to bind membrane molecules and antibodies. Through fusion with glycosylphosphatidylinositol anchors, anti-epidermal growth factor receptor (EGFR) nanobodies were strongly enriched in EVs and greatly improved the capacity of targeting tumor cells (Kooijmans et al., 2016). Later, the anchoring peptide CP05 was proven to have the ability to target CD63 on the exosome surface, and some researchers have tried to use CP05-modified exosomes as delivery carriers (Gao et al., 2018). For example, exosomes loaded with KV11 (an anti-angiogenic peptide) through CP05 exhibit better delivery efficiency and suppress neovascularization in retinopathies (Dong et al., 2021) (Figure 1C). Similarly, CP05 was applied to modify 3D-printed scaffolds to improve the grafting efficiency of the engineered exosomes (Zha et al., 2021). Despite promising results in other disease models, CP05 has not been explored in targeting tumor cells, which might be a research direction in the future.
2.4 Biomimetic EVs
Biomimetic EVs are combinations of synthetic nanoparticulate cores and biologically derived membrane coatings, which exhibit excellent pharmacokinetics and biocompatibility (Dehaini et al., 2017) (Figure 1D). For instance, EXO-PMA/Au-BSA@Ce6 nanovehicles, generated from freshly urinary exosomes, are loaded with multifunctionalized PMA/Au-BSA@Ce6 nanoparticles via an instant electroporation strategy, which can bypass the host immune response, prolong circulation time and delay the endocytosis of macrophages (Pan et al., 2020). In the same way, the boron nitride nanotubes loaded with doxorubicin and coated with cell membranes extracted from glioblastoma multiforme cells are enabled to penetrate into glioma tissues and greatly promote cellular internalization and antiproliferation ability as well as extending circulation time (De Pasquale et al., 2020). Given the large number of exosomes needed, taking the membrane structure from other sources into consideration seems feasible.
2.5 Genetic manipulation
Genetic manipulation is an indirect method of modification that requires transfection tools to deliver cargo to parent cells, increase the expression of RNAs or proteins, and finally generate exosomes with ideal targeted molecules or RNAs through a natural biogenesis process (Zhang et al., 2020; Zhang et al., 2021) (Figure 1E). Exosomes expressing cardiac-targeting peptide Lamp2b on the exosomal membrane have been obtained in this way and have been developed as a therapeutic tool for heart disease (Tian et al., 2018). Moreover, engineered EVs as versatile ribonucleoprotein (RNP) delivery vehicles for CRISPR (clustered regularly interspaced short palindromic repeats) genome editing has been explored and created a system capable of delivering RNP targeting multiple loci for multiplex genome editing (Yao et al., 2021). Furthermore, properly combining both modified donor cells and exosomes is a potential choice (Lv et al., 2020). However, it brings worries about introduced ligands interrupting the normal function of other proteins or showing up in undesirable places.
2.6 Other methods
Due to the negatively charged surface, cations can modify EVs by electrostatic interactions, but some cationic materials are cytotoxic and induce lysosome degradation. In addition, metabolic engineering refers to changing parental cells by using a growth medium with specific substances to promote cellular biosynthesis (Zhang et al., 2022). Research on these methods is relatively limited at present.
3 Combinational antitumor strategies
When monotherapy is not enough to suppress the proliferation of cancer cells or drug resistance appears, combination therapy shows more efficacy. However, few drugs could reach the tumor site after systemic administration. To achieve a high concentration of drugs at the local site, nanocarriers are modified to aggregate drugs in the tumor environment. Codelivery nanocarriers have become a newly emerging strategy to magnify synergistic effects and minimize adverse reactions (Al Bostami et al., 2022). We summarize current combinational antitumor plans based on exosomes and propose some views about future challenges and development (Figure 2; Supplementary Table S2).
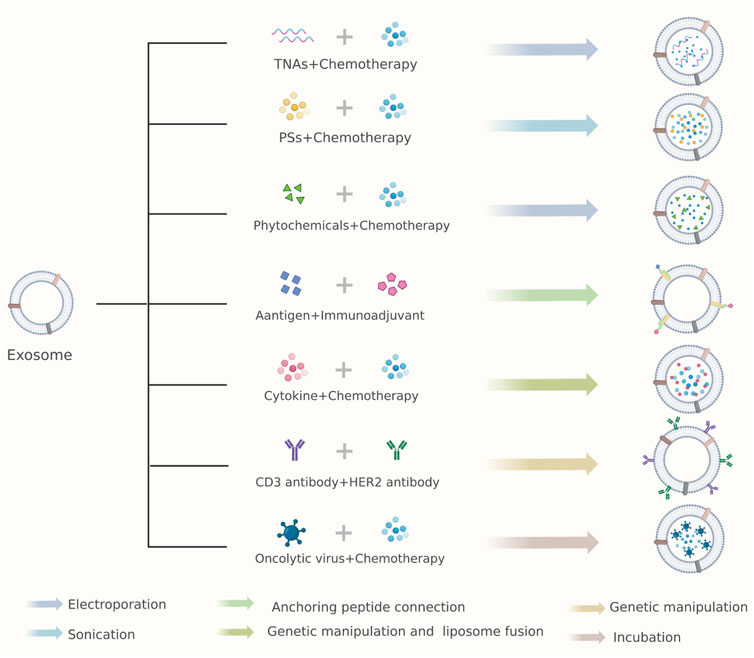
FIGURE 2. Combinational antitumor strategies of exosomes as drug carriers. Arrows with different colors represent various drug loading methods. TNAs, therapeutic nucleic acids; PSs, photosensitizers; Her2, human epidermal growth factor receptor 2.
3.1 TNAs combined with chemotherapeutic agents
Many efforts have been made to illustrate the role of ncRNAs in cancer (Slack and Chinnaiyan, 2019). Numerous studies have identified specific miRNA or lncRNA participating in the development of tumor proliferation, metastasis and resistance (Wang et al., 2019). Thus, codelivery of TNAs and classical chemotherapeutic agents by exosomes is growing into an emerging plan. A recent study designed target-specific exosomes to deliver miR-21 inhibitors (miR-21i) and 5-fluorouracil (5-FU) into 5-FU-resistant HCT-116 cells, a colorectal cancer cell line with highly expressed miR-21 (Liang et al., 2020). They fused Her2 (human epidermal growth factor receptor 2) with LAMP2 (lysosome-associated membrane proteins 2), and the fusion protein expressed on the surface of exosomes facilitated cellular uptake. 5-FU and miR-21i were packaged into exosomes through electroporation by mixing an appropriate concentration. Then, the engineered exosomes were proven safe and effective both in vivo and in vitro. Another study utilized A15-EXOs to deliver cholesterol-modified miRNA (miR-159) and doxorubicin to triple-negative breast cancer (TNBC), which exhibits potent antitumor activity and precisely targeted delivery (Gong et al., 2019). In addition, a recent report identified lncRNA PGM5 antisense RNA 1 (PGM5-AS1) as a prognostic indicator correlated with better OS in colorectal cancer patients treated with oxaliplatin, and exosomes loaded with PGM5-AS1 and oxaliplatin indeed inhibited the proliferation, metastasis, and acquired resistance to oxaliplatin of colon cancer cells (Hui et al., 2022).
Engineered exosomes indeed increase the concentration of therapeutic agents in local tumors. Exosomes loaded with TNAs and chemotherapeutic drugs make it feasible for clinical translation. However, electroporation widely used in loading drugs can induce the aggregation of nucleic acids, which might cause unknown effects (Jeyaram et al., 2020). In addition, the efficiency of this approach remains to be improved. An appropriate proportion of every agent, and a suitable electroporation solution system or parameters will be one of the future research directions (Liang et al., 2020).
3.2 Chemotherapy-photodynamic therapy
Photodynamic therapy (PDT) is a cancer treatment that excites photosensitizers in tumor tissues by specific wavelengths of light, resulting in the generation of reactive oxygen species (ROS) and leading to cell death (Choi et al., 2022). A new combination strategy of exosomes appeared recently, which armed exosome with CD47 and ferroptosis inducer erastin (ER) on the surface together with the core of the photosensitizer Bengal (RB) (Du et al., 2021). Upon irradiation with the laser, ER and RB synergistically induced cell death in the tumor region without apparent toxicity. Cellular glutathione (GSH) can be oxidized and subsequently reduced to participate in antioxidant activity, restricting PDT, which relies on ROS. Moreover, glutamine is an essential source of nutrition for tumor cells and GSH synthesis (Altman et al., 2016). Therefore, glutamine metabolism inhibitors and PDT could be used simultaneously. Zhu et al. designed exosomes carrying aggregation-induced emission luminogens (AIEgens) and proton pump inhibitors (PPIs) to combine enhanced type-1 photodynamic therapy with tumor glutamine starvation therapy, which showed a significant antitumor effect (Zhu et al., 2022). Apart from these, the superiority of exosomes combined with PDT in real-time fluorescence imaging is visible (Pan et al., 2020; Qian et al., 2022).
Capsulated in membrane structures, such as exosomes and cellular membranes, provide the second chance to use drugs with low water solubility, adverse pharmacokinetics or biodistribution. Meanwhile, it puts forward the idea of adding antimetabolites to combined regimens based on exosomes. Studies of other agents acting inside tumor cells will unceasingly process to find more strategies for cancer therapy.
3.3 Immunotherapy in combinational therapy
Engineered exosomes are widely used in antitumor immunity. Bearing abundant signature proteins from their parental cells, DC-derived exosomes (DEXs) have been used as new vaccines (Naseri et al., 2020). Zuo et al. equipped DEX with a hepatocellular carcinoma (HCC)-targeting peptide-P47, an immunoadjuvant high mobility group nucleosome-binding protein 1 (HMGN1) and an α-fetoprotein epitope (Zuo et al., 2022). This vaccine system could recruit and activate DCs in tumor sites, triggering antigen-specific immune responses and inducing tumor-killing effects in orthotopic HCC mice. In the same way, α-lactalbumin (α-LA)-modified exosomes delivered the immunogenic cell death inducers human neutrophil elastase (ELANE) and Hiltonol (TLR3 agonist) to TNBC and resulted in enhanced CD8 T-cell responses in a mouse xenograft model and patient-derived tumor organoids (Huang et al., 2022). In addition, exosomes painted with a dual antibody targeting CD3 T cells and breast cancer-associated HER2 receptors can recruit not only T cells but also induce specific killing of HER2-positive breast cancer cells (Huang et al., 2022). Lung cancer cell-derived EVs could bring oncolytic viruses (OVs) and chemotherapy drugs such as paclitaxel to tumor tissue, leading to enhanced antitumor effects in nude mice (Shi et al., 2020). Exosomes as delivery carriers provides a new method to augment the effects of immunizing agents and turn cold tumors hot.
In addition, exosome delivery of small interfering RNAs (siRNAs) targeting key immune-related genes is another successful exploration of antitumor immunity. Immune checkpoint blockade induces responses well in some cancer types, whereas it presents dismal effects in glioblastoma (GBM) because of the blood‒brain barrier (BBB) and suppressive microenvironment in GBM (Lai et al., 2022). C(RGDyK)-conjugated EVs smoothly bring PD-L1 siRNA to GBM and improve the immunosuppressive microenvironment after short-burst radiation, which provides a scheme for immune checkpoint therapy in brain tumors (Tian et al., 2022). Similarly, codelivery of oxaliplatin and siRNA against galectin-9 triggers ICD and reverses immunosuppression caused by M2 tumor-associated macrophages in pancreatic ductal adenocarcinoma (Zhou et al., 2021).
Given their targeting and penetrating ability, exosomes also demonstrate powerful advantages in metastatic cancer. Lv et al. fused CD47-expressing exosomes with thermosensitive liposomes to develop a nano delivery system loaded with granulocyte-macrophage colony-stimulating factor (GM-CSF) and docetaxel, which could promote the repolarization of macrophages toward M1 and show more significant therapeutic benefits than either agent alone in metastatic peritoneal carcinoma (Lv et al., 2020; Shi et al., 2020; Garofalo et al., 2019).
3.4 Phytochemicals in combinational therapy
The antitumor effects of phytochemicals have been confirmed (Kola et al., 2022). Due to their low toxicity and multiple modulation pathways, phytochemicals have become a new option for cancer therapy (Gao et al., 2022). Curcumin, a plant polyphenolic compound, can suppress tumor growth through various mechanisms (Bhattacharyya et al., 2007; Sanidad et al., 2019). However, like most drugs, it cannot cross the BBB and lacks tumor-targeting ability. RGE-modified exosomes loaded with superparamagnetic iron oxide nanoparticles (SPIONs) and curcumin could cross the BBB smoothly, target glioma accurately, and markedly improve the antitumor effects (Jia et al., 2018). Phytochemical-based combinational regimens are limited by the volume of exosomes for multiple drugs, but hybrid vesicles or a combination of other therapies could provide potential solutions.
4 Discussion
Currently, the trend of combinational therapy is overwhelming because of poor therapeutic effects of single agent and drug resistance. Although several nanomaterials have been applied in codelivery, cytotoxicity and low permeability restrict their application (Chen et al., 2021).Exosomes as delivery carriers possess many advantages, such as high efficacy, low toxicity, low immunogenicity, low off-target risks, bioavailability and stability. Delivery by exosomes increases the efficiency of cancer treatments and reduces unwanted drug aggregation. Exosomes can penetrate biotic barriers and deep tissues to target tumors, which endows them with significant advantages in cancer therapy, especially in metastatic carcinoma. Despite the great potential of exosome-based therapies, unscalable production of exosomes remains a challenge to translate these therapies to clinical practice. First, a standard protocol and commercial production of exosomes are needed. Given that large amounts of exosomes are needed, numerous technologies are under study to increase production (Debbi et al., 2022; Seo et al., 2022). Some alternative sources are also being explored, such as exosomes extracted from milk and plants (Badawy et al., 2018; Dad et al., 2021; El-Kattawy et al., 2021; Zhong et al., 2021). The digestive tract can absorb them, but the concentration of the contents reaching the treatment site is another question to consider. Second, how to optimize loading efficiency has always been a problem. Since passive diffusion is the primary mechanism of the present methods, such as incubation, freeze—thaw cycling, sonication, and electroporation, the loading efficiency is unsatisfactory. The interesting idea that the self-assembly of exogenous siRNAs could be encapsulated into secretory exosomes in the liver and delivered to the targeted tissue attracted our attention (Fu et al., 2021). Although it is an innovation of active loading, whether it can be artificially controlled and improve the loading efficiency needs more evidence. Finally, more strategies remain to be developed to provide more superior choices. For example, the delivery of CRISPR—Cas by exosome has made some progress, and if self-assembly of CRISPR in vivo could be realized, it will becomes a powerful tool for gene therapy (Wan et al., 2022).
Engineered exosomes have been developed for dual/multiple drug codelivery, which can improve pharmacokinetic behavior and tumor accumulation capacity as well as achieve tumor site-targeted delivery. Drugs based on exosomes, like zofin for COVID-19, have been approved by FDA for clinical trials, but their applications in cancer focus on diagnosis or safty assessment now (Mitrani et al., 2021). There is no doubt that exosomes transform conventional disease treatment into a new era of cell-free therapy and are promising for realizing clinical translation.
5 Conclusion and perspectives
This review has summarized the recent advances in exosomes as carriers for combinational antitumor therapies, including the methods of engineering modification and different combinations of chemotherapeutic agents, therapeutic nucleic acids, photosensitizers, immunotherapy and phytochemicals. Despite the progressive advances, there is still a gap between experimental research and clinical application. Further studies are quite essential to explore the properties of various exosomes for further application. In addition, it is necessary to further clarify how to select appropriate loading methods and route of administration to promote loading efficiency and absorption of cancer cells for different target tissue and drug’s chemical composition. Exosomes play an important role as carriers in combination antitumor therapy, but the choice of combination therapy strategy is critical because not all combinations enhance antitumor effects and combination strategies might increase the risk of adverse reactions. For clinical application, completely pure exosomes, without any biological/biochemical contaminations, have not been obtained, so how to further purify the acquired exosomes is an important step in achieving clinical transformation. Although there are many challenges in the emerging field, multifunctional exosomes have shown significant effects in a variety of cancer animal models, suggesting great potential for future clinical transformation.
Author contributions
GX and ZX write this review and have contributed equally to this work and share first authorship. FL is the corresponding author.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2022.1107329/full#supplementary-material
References
Al Bostami, R. D., Abuwatfa, W. H., and Husseini, G. A. (2022). Recent advances in nanoparticle-based Co-delivery systems for cancer therapy. Nanomater. (Basel) 12 (15), 2672. doi:10.3390/nano12152672
Altman, B. J., Stine, Z. E., and Dang, C. V. (2016). From krebs to clinic: Glutamine metabolism to cancer therapy. Nat. Rev. Cancer 16 (10), 619–634. doi:10.1038/nrc.2016.71
Badawy, A. A., El-Magd, M. A., and AlSadrah, S. A. (2018). Therapeutic effect of camel milk and its exosomes on MCF7 cells in vitro and in vivo. Integr. Cancer Ther. 17 (4), 1235–1246. doi:10.1177/1534735418786000
Bagchi, S., Yuan, R., and Engleman, E. G. (2021). Immune checkpoint inhibitors for the treatment of cancer: Clinical impact and mechanisms of response and resistance. Annu. Rev. Pathol. 16, 223–249. doi:10.1146/annurev-pathol-042020-042741
Bhattacharyya, S., Mandal, D., Sen, G. S., Pal, S., Banerjee, S., Lahiry, L., et al. (2007). Tumor-induced oxidative stress perturbs nuclear factor-kappaB activity-augmenting tumor necrosis factor-alpha-mediated T-cell death: Protection by curcumin. Cancer Res. 67 (1), 362–370. doi:10.1158/0008-5472.CAN-06-2583
Chen, J., Tan, Q., Yang, Z., and Jin, Y. (2022). Engineered extracellular vesicles: Potentials in cancer combination therapy. J. Nanobiotechnology 20 (1), 132. doi:10.1186/s12951-022-01330-y
Chen, L., Hong, W., Ren, W., Xu, T., Qian, Z., and He, Z. (2021). Recent progress in targeted delivery vectors based on biomimetic nanoparticles. Signal Transduct. Target Ther. 6 (1), 225. doi:10.1038/s41392-021-00631-2
Choi, J., Sun, I. C., Sook Hwang, H., Yeol Yoon, H., and Kim, K. (2022). Light-triggered photodynamic nanomedicines for overcoming localized therapeutic efficacy in cancer treatment. Adv. Drug Deliv. Rev. 186, 114344. doi:10.1016/j.addr.2022.114344
Dad, H. A., Gu, T. W., Zhu, A. Q., Huang, L. Q., and Peng, L. H. (2021). Plant exosome-like nanovesicles: Emerging therapeutics and drug delivery nanoplatforms. Mol. Ther. 29 (1), 13–31. doi:10.1016/j.ymthe.2020.11.030
Dammes, N., and Peer, D. (2020). Paving the road for RNA therapeutics. Trends Pharmacol. Sci. 41 (10), 755–775. doi:10.1016/j.tips.2020.08.004
De Pasquale, D., Marino, A., Tapeinos, C., Pucci, C., Rocchiccioli, S., Michelucci, E., et al. (2020). Homotypic targeting and drug delivery in glioblastoma cells through cell membrane-coated boron nitride nanotubes. Mater Des. 192, 108742. doi:10.1016/j.matdes.2020.108742
Debbi, L., Guo, S., Safina, D., and Levenberg, S. (2022). Boosting extracellular vesicle secretion. Biotechnol. Adv. 59, 107983. doi:10.1016/j.biotechadv.2022.107983
Dehaini, D., Wei, X., Fang, R. H., Masson, S., Angsantikul, P., Luk, B. T., et al. (2017). Erythrocyte-platelet hybrid membrane coating for enhanced nanoparticle functionalization. Adv. Mater 29 (16), 1606209. doi:10.1002/adma.201606209
Dong, X., Lei, Y., Yu, Z., Wang, T., Liu, Y., Han, G., et al. (2021). Exosome-mediated delivery of an anti-angiogenic peptide inhibits pathological retinal angiogenesis. Theranostics 11 (11), 5107–5126. doi:10.7150/thno.54755
Du, J., Wan, Z., Wang, C., Lu, F., Wei, M., Wang, D., et al. (2021). Designer exosomes for targeted and efficient ferroptosis induction in cancer via chemo-photodynamic therapy. Theranostics 11 (17), 8185–8196. doi:10.7150/thno.59121
El-Kattawy, A. M., Algezawy, O., Alfaifi, M. Y., Noseer, E. A., Hawsawi, Y. M., Alzahrani, O. R., et al. (2021). Therapeutic potential of camel milk exosomes against HepaRG cells with potent apoptotic, anti-inflammatory, and anti-angiogenesis effects for colostrum exosomes. Biomed. Pharmacother. 143, 112220. doi:10.1016/j.biopha.2021.112220
Evers, M. J. W., van de Wakker, S. I., de Groot, E. M., de Jong, O. G., Gitz-Francois, J. J. J., Seinen, C. S., et al. (2022). Functional siRNA delivery by extracellular vesicle-liposome hybrid nanoparticles. Adv. Healthc. Mater 11 (5), e2101202. doi:10.1002/adhm.202101202
Fu, Z., Zhang, X., Zhou, X., Ur-Rehman, U., Yu, M., Liang, H., et al. (2021). In vivo self-assembled small RNAs as a new generation of RNAi therapeutics. Cell. Res. 31 (6), 631–648. doi:10.1038/s41422-021-00491-z
Gao, Q., Feng, J., Liu, W., Wen, C., Wu, Y., Liao, Q., et al. (2022). Opportunities and challenges for co-delivery nanomedicines based on combination of phytochemicals with chemotherapeutic drugs in cancer treatment. Adv. Drug Deliv. Rev. 188, 114445. doi:10.1016/j.addr.2022.114445
Gao, X., Ran, N., Dong, X., Zuo, B., Yang, R., Zhou, Q., et al. (2018). Anchor peptide captures, targets, and loads exosomes of diverse origins for diagnostics and therapy. Sci. Transl. Med. 10 (444), eaat0195. doi:10.1126/scitranslmed.aat0195
Garofalo, M., Villa, A., Rizzi, N., Kuryk, L., Rinner, B., Cerullo, V., et al. (2019). Extracellular vesicles enhance the targeted delivery of immunogenic oncolytic adenovirus and paclitaxel in immunocompetent mice. J. Control Release 294, 165–175. doi:10.1016/j.jconrel.2018.12.022
Gong, C., Tian, J., Wang, Z., Gao, Y., Wu, X., Ding, X., et al. (2019). Functional exosome-mediated co-delivery of doxorubicin and hydrophobically modified microRNA 159 for triple-negative breast cancer therapy. J. Nanobiotechnology 17 (1), 93. doi:10.1186/s12951-019-0526-7
Goyard, D., Ortiz, A. M., Boturyn, D., and Renaudet, O. (2022). Multivalent glycocyclopeptides: Conjugation methods and biological applications. Chem. Soc. Rev. 51 (20), 8756–8783. doi:10.1039/d2cs00640e
Huang, L., Rong, Y., Tang, X., Yi, K., Qi, P., Hou, J., et al. (2022). Engineered exosomes as an in situ DC-primed vaccine to boost antitumor immunity in breast cancer. Mol. Cancer 21 (1), 45. doi:10.1186/s12943-022-01515-x
Huang, W., Chen, L., Kang, L., Jin, M., Sun, P., Xin, X., et al. (2017). Nanomedicine-based combination anticancer therapy between nucleic acids and small-molecular drugs. Adv. Drug Deliv. Rev. 115, 82–97. doi:10.1016/j.addr.2017.06.004
Hui, B., Lu, C., Wang, J., Xu, Y., Yang, Y., Ji, H., et al. (2022). Engineered exosomes for co-delivery of PGM5-AS1 and oxaliplatin to reverse drug resistance in colon cancer. J. Cell. Physiol. 237 (1), 911–933. doi:10.1002/jcp.30566
Jeyaram, A., Lamichhane, T. N., Wang, S., Zou, L., Dahal, E., Kronstadt, S. M., et al. (2020). Enhanced loading of functional miRNA cargo via pH gradient modification of extracellular vesicles. Mol. Ther. 28 (3), 975–985. doi:10.1016/j.ymthe.2019.12.007
Jia, G., Han, Y., An, Y., Ding, Y., He, C., Wang, X., et al. (2018). NRP-1 targeted and cargo-loaded exosomes facilitate simultaneous imaging and therapy of glioma in vitro and in vivo. Biomaterials 178, 302–316. doi:10.1016/j.biomaterials.2018.06.029
Kalluri, R., and LeBleu, V. S. (2020). The biology, function, and biomedical applications of exosomes. Science 367 (6478), eaau6977. doi:10.1126/science.aau6977
Kara, G., Calin, G. A., and Ozpolat, B. (2022). RNAi-based therapeutics and tumor targeted delivery in cancer. Adv. Drug Deliv. Rev. 182, 114113. doi:10.1016/j.addr.2022.114113
Kola, P., Metowogo, K., Manjula, S. N., Katawa, G., Elkhenany, H., Mruthunjaya, K. M., et al. (2022). Ethnopharmacological evaluation of antioxidant, anti-angiogenic, and anti-inflammatory activity of some traditional medicinal plants used for treatment of cancer in Togo/Africa. J. Ethnopharmacol. 283, 114673. doi:10.1016/j.jep.2021.114673
Kooijmans, S. A. A., de Jong, O. G., and Schiffelers, R. M. (2021). Exploring interactions between extracellular vesicles and cells for innovative drug delivery system design. Adv. Drug Deliv. Rev. 173, 252–278. doi:10.1016/j.addr.2021.03.017
Kooijmans, S. A. A., Fliervoet, L. A. L., van der Meel, R., Fens, M., Heijnen, H. F. G., van Bergen En Henegouwen, P. M. P., et al. (2016). PEGylated and targeted extracellular vesicles display enhanced cell specificity and circulation time. J. Control Release 224, 77–85. doi:10.1016/j.jconrel.2016.01.009
Lai, J. J., Chau, Z. L., Chen, S. Y., Hill, J. J., Korpany, K. V., Liang, N. W., et al. (2022). Exosome processing and characterization approaches for research and technology development. Adv. Sci. (Weinh) 9 (15), e2103222. doi:10.1002/advs.202103222
Liang, G., Zhu, Y., Ali, D. J., Tian, T., Xu, H., Si, K., et al. (2020). Engineered exosomes for targeted co-delivery of miR-21 inhibitor and chemotherapeutics to reverse drug resistance in colon cancer. J. Nanobiotechnology 18 (1), 10. doi:10.1186/s12951-019-0563-2
Lin, Y., Wu, J., Gu, W., Huang, Y., Tong, Z., Huang, L., et al. (2018). Exosome-liposome hybrid nanoparticles deliver CRISPR/Cas9 system in MSCs. Adv. Sci. (Weinh) 5 (4), 1700611. doi:10.1002/advs.201700611
Liu, X., Pu, Y., Cron, K., Deng, L., Kline, J., Frazier, W. A., et al. (2015). CD47 blockade triggers T cell-mediated destruction of immunogenic tumors. Nat. Med. 21 (10), 1209–1215. doi:10.1038/nm.3931
Lv, Q., Cheng, L., Lu, Y., Zhang, X., Wang, Y., Deng, J., et al. (2020). Thermosensitive exosome-liposome hybrid nanoparticle-mediated chemoimmunotherapy for improved treatment of metastatic peritoneal cancer. Adv. Sci. (Weinh) 7 (18), 2000515. doi:10.1002/advs.202000515
Marusic, N., Otrin, L., Rauchhaus, J., Zhao, Z., Kyrilis, F. L., Hamdi, F., et al. (2022). Constructing artificial respiratory chain in polymer compartments: Insights into the interplay between bo 3 oxidase and the membrane. Proc. Natl. Acad. Sci. U. S. A. 119 (20), 15006–15017. doi:10.1073/pnas.1919306117
Mitrani, M. I., Bellio, M. A., Meglin, A., Khan, A., Xu, X., Haskell, G., et al. (2021). Treatment of a COVID-19 long hauler with an amniotic fluid-derived extracellular vesicle biologic. Respir. Med. Case Rep. 34, 101502. doi:10.1016/j.rmcr.2021.101502
Naseri, M., Bozorgmehr, M., Zoller, M., Ranaei Pirmardan, E., and Madjd, Z. (2020). Tumor-derived exosomes: The next generation of promising cell-free vaccines in cancer immunotherapy. Oncoimmunology 9 (1), 1779991. doi:10.1080/2162402X.2020.1779991
Nie, W., Wu, G., Zhang, J., Huang, L. L., Ding, J., Jiang, A., et al. (2020). Responsive exosome nano-bioconjugates for synergistic cancer therapy. Angew. Chem. Int. Ed. Engl. 59 (5), 2018–2022. doi:10.1002/anie.201912524
Nolte-'t Hoen, E. N., Buschow, S. I., Anderton, S. M., Stoorvogel, W., and Wauben, M. H. (2009). Activated T cells recruit exosomes secreted by dendritic cells via LFA-1. Blood 113 (9), 1977–1981. doi:10.1182/blood-2008-08-174094
Pan, S., Pei, L., Zhang, A., Zhang, Y., Zhang, C., Huang, M., et al. (2020). Passion fruit-like exosome-PMA/Au-BSA@Ce6 nanovehicles for real-time fluorescence imaging and enhanced targeted photodynamic therapy with deep penetration and superior retention behavior in tumor. Biomaterials 230, 119606. doi:10.1016/j.biomaterials.2019.119606
Parker, C. G., and Pratt, M. R. (2020). Click chemistry in proteomic investigations. Cell. 180 (4), 605–632. doi:10.1016/j.cell.2020.01.025
Paskeh, M. D. A., Entezari, M., Mirzaei, S., Zabolian, A., Saleki, H., Naghdi, M. J., et al. (2022). Emerging role of exosomes in cancer progression and tumor microenvironment remodeling. J. Hematol. Oncol. 15 (1), 83. doi:10.1186/s13045-022-01305-4
Peplow, M. (2019). Click chemistry targets antibody-drug conjugates for the clinic. Nat. Biotechnol. 37 (8), 835–837. doi:10.1038/d41587-019-00017-4
Piffoux, M., Silva, A. K. A., Wilhelm, C., Gazeau, F., and Tareste, D. (2018). Modification of extracellular vesicles by fusion with liposomes for the design of personalized biogenic drug delivery systems. ACS Nano 12 (7), 6830–6842. doi:10.1021/acsnano.8b02053
Qian, R., Jing, B., Jiang, D., Gai, Y., Zhu, Z., Huang, X., et al. (2022). Multi-antitumor therapy and synchronous imaging monitoring based on exosome. Eur. J. Nucl. Med. Mol. Imaging 49 (8), 2668–2681. doi:10.1007/s00259-022-05696-x
Sanidad, K. Z., Sukamtoh, E., Xiao, H., McClements, D. J., and Zhang, G. (2019). Curcumin: Recent advances in the development of strategies to improve oral bioavailability. Annu. Rev. Food Sci. Technol. 10, 597–617. doi:10.1146/annurev-food-032818-121738
Seo, N., Nakamura, J., Kaneda, T., Tateno, H., Shimoda, A., Ichiki, T., et al. (2022). Distinguishing functional exosomes and other extracellular vesicles as a nucleic acid cargo by the anion-exchange method. J. Extracell. Vesicles 11 (3), e12205. doi:10.1002/jev2.12205
Shi, X., Cheng, Q., Hou, T., Han, M., Smbatyan, G., Lang, J. E., et al. (2020). Genetically engineered cell-derived nanoparticles for targeted breast cancer immunotherapy. Mol. Ther. 28 (2), 536–547. doi:10.1016/j.ymthe.2019.11.020
Slack, F. J., and Chinnaiyan, A. M. (2019). The role of non-coding RNAs in oncology. Cell. 179 (5), 1033–1055. doi:10.1016/j.cell.2019.10.017
Sung, H., Ferlay, J., Siegel, R. L., Laversanne, M., Soerjomataram, I., Jemal, A., et al. (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71 (3), 209–249. doi:10.3322/caac.21660
Thakur, A., Parra, D. C., Motallebnejad, P., Brocchi, M., and Chen, H. J. (2022). Exosomes: Small vesicles with big roles in cancer, vaccine development, and therapeutics. Bioact. Mater 10, 281–294. doi:10.1016/j.bioactmat.2021.08.029
Tian, T., Liang, R., Erel-Akbaba, G., Saad, L., Obeid, P. J., Gao, J., et al. (2022). Immune checkpoint inhibition in GBM primed with radiation by engineered extracellular vesicles. ACS Nano 16 (2), 1940–1953. doi:10.1021/acsnano.1c05505
Tian, T., Zhang, H. X., He, C. P., Fan, S., Zhu, Y. L., Qi, C., et al. (2018). Surface functionalized exosomes as targeted drug delivery vehicles for cerebral ischemia therapy. Biomaterials 150, 137–149. doi:10.1016/j.biomaterials.2017.10.012
Wan, T., Zhong, J., Pan, Q., Zhou, T., Ping, Y., and Liu, X. (2022). Exosome-mediated delivery of Cas9 ribonucleoprotein complexes for tissue-specific gene therapy of liver diseases. Sci. Adv. 8 (37), eabp9435. doi:10.1126/sciadv.abp9435
Wang, J., Zhu, S., Meng, N., He, Y., Lu, R., and Yan, G. R. (2019). ncRNA-encoded peptides or proteins and cancer. Mol. Ther. 27 (10), 1718–1725. doi:10.1016/j.ymthe.2019.09.001
Winkle, M., El-Daly, S. M., Fabbri, M., and Calin, G. A. (2021). Noncoding RNA therapeutics - challenges and potential solutions. Nat. Rev. Drug Discov. 20 (8), 629–651. doi:10.1038/s41573-021-00219-z
Xia, Y., Rao, L., Yao, H., Wang, Z., Ning, P., and Chen, X. (2020). Engineering macrophages for cancer immunotherapy and drug delivery. Adv. Mater 32 (40), e2002054. doi:10.1002/adma.202002054
Xu, L., Faruqu, F. N., Liam-Or, R., Abu Abed, O., Li, D., Venner, K., et al. (2020). Design of experiment (DoE)-driven in vitro and in vivo uptake studies of exosomes for pancreatic cancer delivery enabled by copper-free click chemistry-based labelling. J. Extracell. Vesicles 9 (1), 1779458. doi:10.1080/20013078.2020.1779458
Yao, X., Lyu, P., Yoo, K., Yadav, M. K., Singh, R., Atala, A., et al. (2021). Engineered extracellular vesicles as versatile ribonucleoprotein delivery vehicles for efficient and safe CRISPR genome editing. J. Extracell. Vesicles 10 (5), e12076. doi:10.1002/jev2.12076
Yi, M., Zheng, X., Niu, M., Zhu, S., Ge, H., and Wu, K. (2022). Combination strategies with PD-1/PD-L1 blockade: Current advances and future directions. Mol. Cancer 21 (1), 28. doi:10.1186/s12943-021-01489-2
Zha, Y., Li, Y., Lin, T., Chen, J., Zhang, S., and Wang, J. (2021). Progenitor cell-derived exosomes endowed with VEGF plasmids enhance osteogenic induction and vascular remodeling in large segmental bone defects. Theranostics 11 (1), 397–409. doi:10.7150/thno.50741
Zhang, F., Guo, J., Zhang, Z., Duan, M., Wang, G., Qian, Y., et al. (2022). Application of engineered extracellular vesicles for targeted tumor therapy. J. Biomed. Sci. 29 (1), 14. doi:10.1186/s12929-022-00798-y
Zhang, M., Zang, X., Wang, M., Li, Z., Qiao, M., Hu, H., et al. (2019). Exosome-based nanocarriers as bio-inspired and versatile vehicles for drug delivery: Recent advances and challenges. J. Mater Chem. B 7 (15), 2421–2433. doi:10.1039/c9tb00170k
Zhang, Q., Zhang, H., Ning, T., Liu, D., Deng, T., Liu, R., et al. (2020). Exosome-delivered c-met siRNA could reverse chemoresistance to cisplatin in gastric cancer. Int. J. Nanomedicine 15, 2323–2335. doi:10.2147/IJN.S231214
Zhang, X., Zhang, H., Gu, J., Zhang, J., Shi, H., Qian, H., et al. (2021). Engineered extracellular vesicles for cancer therapy. Adv. Mater 33 (14), e2005709. doi:10.1002/adma.202005709
Zhong, J., Xia, B., Shan, S., Zheng, A., Zhang, S., Chen, J., et al. (2021). High-quality milk exosomes as oral drug delivery system. Biomaterials 277, 121126. doi:10.1016/j.biomaterials.2021.121126
Zhou, W., Zhou, Y., Chen, X., Ning, T., Chen, H., Guo, Q., et al. (2021). Pancreatic cancer-targeting exosomes for enhancing immunotherapy and reprogramming tumor microenvironment. Biomaterials 268, 120546. doi:10.1016/j.biomaterials.2020.120546
Zhu, D., Zhang, T., Li, Y., Huang, C., Suo, M., Xia, L., et al. (2022). Tumor-derived exosomes co-delivering aggregation-induced emission luminogens and proton pump inhibitors for tumor glutamine starvation therapy and enhanced type-I photodynamic therapy. Biomaterials 283, 121462. doi:10.1016/j.biomaterials.2022.121462
Keywords: exosome, engineered exosomes, drug carriers, cancer, combinational therapy
Citation: Xiao G, Xu Z and Luo F (2023) Combinational antitumor strategies of exosomes as drug carriers: Mini review. Front. Pharmacol. 13:1107329. doi: 10.3389/fphar.2022.1107329
Received: 24 November 2022; Accepted: 28 December 2022;
Published: 20 January 2023.
Edited by:
Mohammed Abu El-Magd, Kafrelsheikh University, EgyptReviewed by:
Hoda Elkhenany, Alexandria University, EgyptElSayed Fatouh, Kafrelsheikh University, Egypt
Copyright © 2023 Xiao, Xu and Luo. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Feng Luo, huaxiluofeng@sina.com
†These authors have contributed equally to this work and share first authorship
 Guixiu Xiao
Guixiu Xiao Zihan Xu
Zihan Xu Feng Luo1*
Feng Luo1*