
Submit your research
Start your submission and get more impact for your research by publishing with us.

Start your submission and get more impact for your research by publishing with us.

Ready to publish? Check out our author guidelines for everything you need to know about submission, from choosing a journal and section to preparing your manuscript.

Reviewing a manuscript? See our editorial guidelines for everything you need to know about Frontiers’ peer review process.

Our efficient and rigorous peer review means you’ll get a decision on your manuscript in just 77 days.

Article processing charges (APCs) apply to articles that are accepted for publication by our external editors, following rigorous peer review.

Frontiers in Pharmacology now partners with the International Federation of Associations of Pharmaceutical Physicians and Pharmaceutical Medicine; a collaboration aiming to advance high-quality pharmacological research.
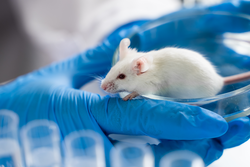
Last chance to submit a paper on the 4R principals in ethnopharmacology: Reduce, Refine, Replace and Responsibility using animal models.

Frontiers in Pharmacology now partners with the South African Society for Basic and Clinical Pharmacology. A partnership that aims to promote high quality research and foster collaborations within the pharmacological field.

Explore this trending review on herbal and dietary supplements for depression.

Shape the future of your field by proposing your own Research Topic. Submit a brief idea explaining its relevance, the challenge it tackles, and the community it serves. We particularly welcome cross-disciplinary ideas with real-world impact.

Fundisa African Academy of Medicines Development
Cape Town, South Africa
Specialty Chief Editor
Drugs Outcomes Research and Policies

The University of Queensland
Brisbane, Australia
Associate Editor
Drugs Outcomes Research and Policies

Faculty of Pharmacy, University of Lisbon
Lisbon, Portugal
Associate Editor
Drugs Outcomes Research and Policies

University of Nigeria, Nsukka
Nsukka, Nigeria
Associate Editor
Drugs Outcomes Research and Policies