- 1Liaoning University of Traditional Chinese Medicine, Shenyang, China
- 2Liaoning University of Traditional Chinese Medicine Affiliated Hospital, Shenyang, China
Osteoporosis (OP) is a systemic bone metabolic disorder characterized by a decrease in bone mineral density (BMD) and damage to the trabecular bone microarchitecture. With the increasing global aging population, the incidence of OP has been rising annually, particularly among elderly women, making it a significant public health issue. Traditional diagnostic methods such as dual-energy X-ray absorptiometry (DXA), quantitative computed tomography (QCT), and magnetic resonance imaging (MRI) are effective, but they also have certain limitations. Artificial intelligence (AI) technology is playing an increasingly important role in the management of osteoporosis. Through machine learning (ML), image processing, and data analysis, AI can accurately assess bone density, fracture risk, and other factors, improving the early diagnosis rate of OP and providing strong decision support for clinicians to optimize treatment plans and enhance treatment outcomes. However, it also faces challenges such as AI model interpretability, insufficient diversity in training data, lack of clinical validation, and issues related to privacy protection and ethics. Addressing these problems is crucial for promoting the widespread application of AI technology in this field. As technology continues to advance, AI will become an indispensable part of OP research and clinical applications, driving the development of personalized treatment and precision medicine.
1 Introduction
OP is a systemic bone metabolic disorder characterized by a reduction in BMD and trabecular bone microarchitecture damage (Zhang et al., 2025). This condition typically arises when the rate of bone resorption exceeds that of bone formation, leading to a loss of bone mass, which results in decreased bone strength and an increased risk of fractures (Anam and Insogna, 2021). With the global aging population, the incidence of OP has been increasing annually, becoming a significant public health challenge, particularly among elderly women (Zhang et al., 2024). OP not only severely impacts the quality of life of patients but also places a significant burden on the healthcare system, leading to high medical costs and socioeconomic expenses (Vendrami et al., 2023). OP often presents no obvious symptoms in its early stages, and many patients fail to receive timely diagnosis and treatment before fractures occur, leading to serious health consequences (Surlari et al., 2023). Therefore, early diagnosis and effective treatment are particularly important. The traditional diagnostic methods for OP mainly include DXA, QCT, and MRI (Tse et al., 2021). Although these methods are effective in assessing bone density and identifying changes associated with osteoporosis, they also have certain limitations (Laskey, 1996). Although QCT can provide more precise three-dimensional bone density images, it results in higher radiation exposure compared to DXA (Njeh et al., 1999). Although MRI has a high sensitivity in detecting bone marrow changes, it is costly and has limited accessibility for routine OP screening, which poses certain practical limitations (Bloem et al., 2025). These limitations highlight the urgent need to develop OP early detection tools that are more accessible, accurate, and low-risk.
AI is a key field in computer science, aimed at enabling computer systems to simulate human cognitive processes and intelligent behaviors, granting them human-like thinking abilities (Li et al., 2023). The primary goal is to enhance the system’s intelligence by processing and analyzing multidimensional data, thereby constructing intelligent models capable of prediction and decision-making (Xu et al., 2021). By leveraging the theories from disciplines such as mathematics, logic, computer science, and biology, AI has achieved groundbreaking innovations in the medical field. These technological advancements not only enhance the capabilities of physicians in performing medical tasks but also assist medical technicians in solving many time-consuming and labor-intensive challenges that were previously difficult to address. With the continuous development of ML and deep learning (DL), AI systems are now able to efficiently analyze vast amounts of medical data, including patient records and imaging data, providing profound insights for diagnosis, treatment planning, and patient care (Rana and Bhushan, 2022). In addition, AI-driven medical tools and systems, such as decision support and diagnostic algorithms, assist healthcare professionals in making more accurate and timely clinical decisions (Magrabi et al., 2019). This collaboration not only optimizes healthcare processes but also significantly enhances patient treatment outcomes, holding immense potential to disrupt medical practice, research, and education. It ushers in a new era for precision medicine and data-driven healthcare innovation. In the management of osteoporosis, the application of AI technologies is playing an increasingly important role. Through ML algorithms, imaging processing techniques, and data analysis, AI is capable of accurately assessing bone density, fracture risk, and other factors. These technologies not only improve the early diagnosis rate of OP but also provide strong decision support for clinicians, aiding in the optimization of treatment pathways and enhancing treatment outcomes (Smets et al., 2021). With the continuous advancement of technology, AI is gaining increasing attention in the research and clinical application of osteoporosis, becoming an indispensable part of future OP management. This article aims to review the progress of AI in the diagnosis and risk prediction of osteoporosis. It first reviews the research on AI in OP risk prediction, then explores the application of AI in combination with traditional imaging diagnostic technologies such as X-ray, DXA, CT, QCT, and MRI. Finally, it analyzes the potential, future development directions, and challenges of AI in clinical applications for osteoporosis, in order to provide references for clinical diagnosis, treatment, and research in OP.
2 The application of AI in OP risk prediction
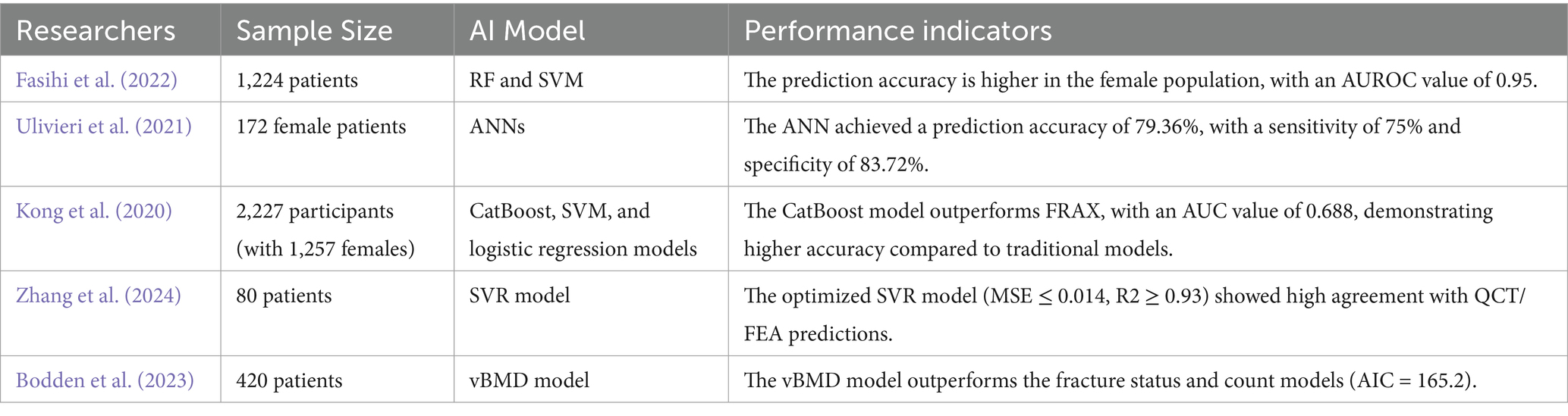
Accurate prediction of osteoporotic fracture risk is crucial for early intervention and reducing fracture incidence. With the continuous advancement of AI technology, many studies have begun to utilize AI models for fracture risk prediction (Table 1). Compared to traditional prediction methods, AI-based OP risk prediction can analyze multi-dimensional data, improving the accuracy of risk assessment. This significantly enhances prediction precision, providing a more reliable basis for early clinical intervention. In a study involving 1,224 men and women, Fasihi L et al. employed various ML algorithms, such as random forests (RF) and support vector machines (SVM), to predict the risk of osteoporosis. The results showed that certain algorithms outperformed traditional methods, particularly in female patients, where the AUROC value reached 0.95, demonstrating extremely high predictive accuracy (Fasihi et al., 2022). Ulivieri FM et al. evaluated an AI-based model for predicting fragility fractures, the Bone Strain Index (BSI), by analyzing spinal X-ray and DXA data from 172 female patients using artificial neural networks (ANNs). The study selected five variables: age, age at menopause, BMI, femoral total bone mineral content (FTot BMC), and femoral total bone strain index (FTot BSI). The results demonstrated that the ANN model achieved an accuracy of 79.36%, a sensitivity of 75%, and a specificity of 83.72% (Ulivieri et al., 2021). These studies provide strong support for the prediction of OP and its associated fracture risks, especially demonstrating great potential for application in female patients.
Kong et al. (2020) developed a ML-based fracture risk prediction model using CatBoost, SVM, and logistic regression models, and validated it in the prospective Ansung cohort. The research results show that the CatBoost model performs the best in predicting hip fragility fractures, with an AUC value of 0.688, significantly outperforming both the FRAX tool (0.663) and traditional models. Zhang et al. (2023) developed a ML model based on QCT images to predict proximal femoral strength. Femoral strength was calculated using finite element analysis (QCT/FEA), and 50 predictive variables were extracted. The SVR model, optimized through feature selection and dimensionality reduction, performed the best (MSE ≤ 0.014, R2 ≥ 0.93). The prediction results were in high agreement with QCT/FEA, demonstrating the potential of this model in clinical assessments. Bodden et al. (2023) predicted vertebral fragility fractures (VF) by using a convolutional neural network (CNN) framework to automatically extract volumetric BMD (vBMD) from routine CT images. The study included 420 patients and analyzed the relationship between vBMD and fractures. The results indicated that patients with low vBMD had a higher risk of fractures across all segments of the spine, particularly in the L1-5 region. Compared to traditional prediction models based on fracture status and count, the vBMD model performed better (Akaike’s information criteria, AIC = 165.2). These studies also demonstrated the accuracy and application potential of AI in predicting OP fractures, particularly in personalized prediction through the extraction of features such as bone mineral density from imaging data. In conclusion, compared to traditional prediction methods, AI models can handle more dimensions of data and provide more precise risk assessments. In the future, with further development and optimization of technology, the application of AI in OP risk prediction is expected to provide more reliable evidence for early clinical intervention, helping to reduce fracture incidence and improve patients’ quality of life.
3 The application of AI in OP diagnosis
Many studies have shown that the integration of artificial intelligence with X-ray, DXA, CT, QCT, and MRI provides more precise and intelligent solutions for the diagnosis of OP. AI can automatically extract key features from images, improve diagnostic efficiency, reduce errors, and assist in early risk identification, thereby enabling personalized treatment plans. For details, refer to Table 2.
3.1 AI in combination with X-ray and DXA
In the diagnosis of osteoporosis, X-ray and DXA are commonly used diagnostic methods. X-ray helps detect fractures or bone abnormalities by evaluating changes in bone density, but its sensitivity and specificity have limitations in early diagnosis. In contrast, DXA is considered the gold standard for OP diagnosis (Morgan and Prater, 2017). DXA can accurately measure BMD and, by comparing it to standard values, determine whether a patient is at risk for OP or osteopenia. However, the availability of DXA equipment is limited, and in many countries (Kanis et al., 2021), there is a significant shortage of DXA machines. The emergence of AI technology provides a new solution for the diagnosis of OP. By combining AI with X-ray and DXA, AI can automatically analyze images, extract key bone density features, and detect subtle changes, improving the accuracy and speed of diagnosis. AI can also integrate with other health data to provide more comprehensive diagnoses and personalized treatment recommendations. Especially in resource-limited areas, it can supplement the shortage of DXA equipment and expand the coverage of screening.
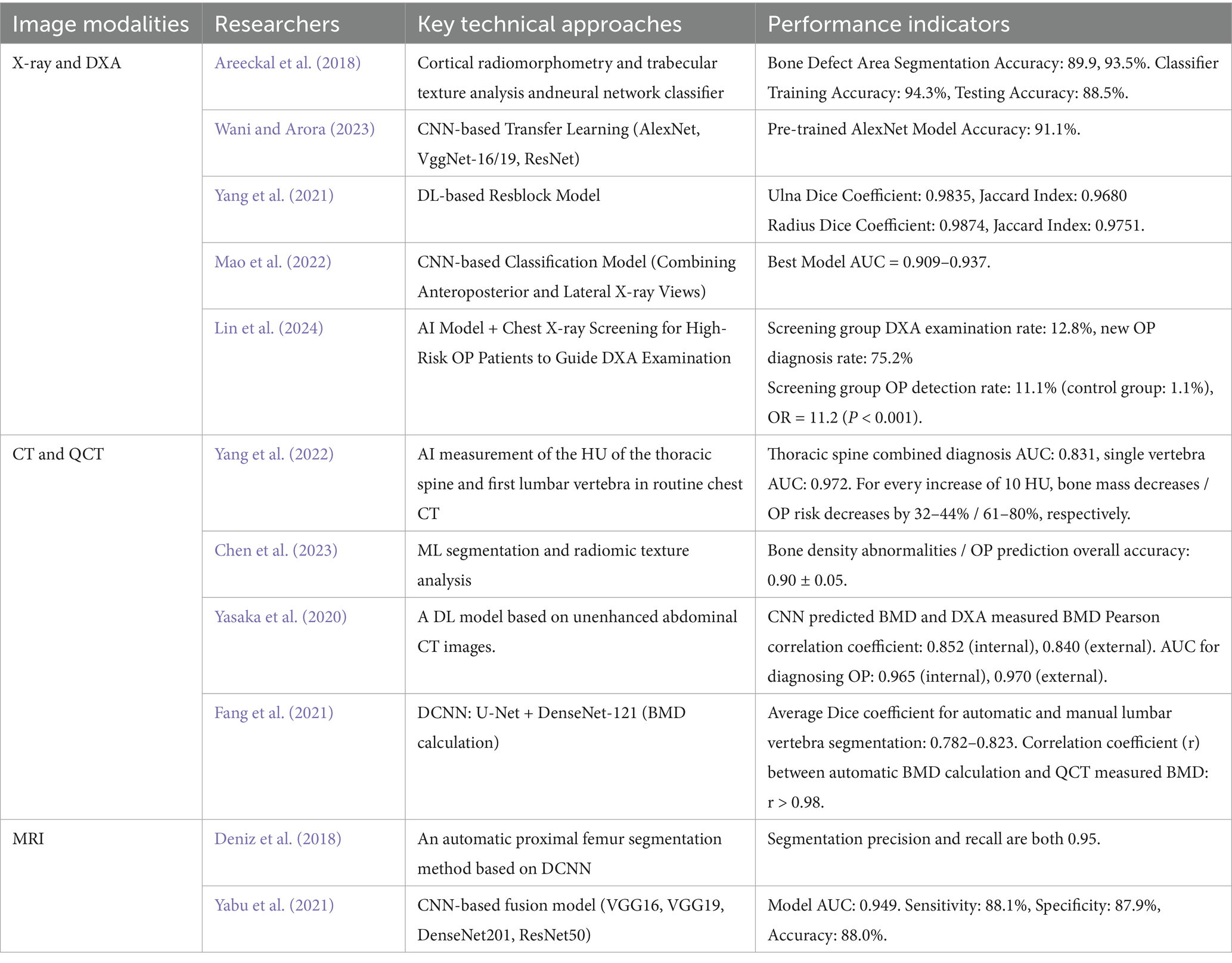
Areeckal et al. (2018) proposed a low-cost automated tool for the early diagnosis of OP using wrist and hand X-ray images. The tool combines cortical radiomorphometry and trabecular texture analysis, utilizing a neural network classifier to distinguish between healthy and low bone mass subjects. Experimental results showed that the segmentation method achieved accuracy rates of 89.9 and 93.5% in detecting bone loss regions. The classifier achieved training and testing accuracy rates of 94.3 and 88.5%, respectively. This technology provides an effective tool for low-cost, large-scale screening of OP risk. It is especially suitable for developing countries or regions with limited equipment and resources. Wani and Arora (2023) proposed a CNN-based method for detecting OP using X-ray images. The study used a dataset of 381 knee X-ray images and employed transfer learning techniques with pre-trained models like AlexNet, VggNet-16, ResNet, and VggNet-19 for classification. The results showed that the pre-trained AlexNet model achieved an accuracy of 91.1%, significantly higher than the 79% accuracy when no pre-trained network was used. Yang et al. (2021) proposed a DL-based residual block (Resblock) model for the automatic segmentation of the ulna and radius in dual-energy X-ray images. The study used data from 360 subjects and evaluated the model performance through five-fold cross-validation. The results showed that the proposed model outperformed traditional methods in segmentation accuracy, with average Dice coefficients of 0.9835 and 0.9874 for the ulna and radius, respectively, and Jaccard indices of 0.9680 and 0.9751. Mao et al. (2022) proposed a screening method based on CNNs for detecting OP and osteopenia through lumbar spine X-rays. The study collected data from 6,908 participants, using DXA-measured BMD values as the reference standard. Three types of CNN models were developed for classification, and the impact of including clinical covariates (such as age, gender, and BMI) on diagnostic performance was explored. The results showed that the model based on antero-posterior and lateral X-rays performed best in diagnosing osteoporosis, with an AUC ranging from 0.909 to 0.937. Lin et al. (2024) conducted a randomized controlled trial to evaluate the effectiveness of an AI model in identifying high-risk OP patients through chest X-rays and performing DXA screening. The study included 40,658 participants, of whom 12.1% were identified as high risk. In the screening group, 12.8% underwent DXA, and 75.2% were diagnosed with new osteoporosis. The OP detection rate in the screening group was significantly higher than in the control group (11.1% vs. 1.1%), with an odds ratio of 11.2 (p < 0.001). The study highlights the clinical value of combining chest X-ray imaging with AI technology in the early diagnosis of OP. AI technology automates the analysis of X-ray images, helping to detect osteoporosis more quickly and accurately, thereby reducing diagnosis time and improving screening efficiency. This method holds great potential, especially in resource-limited areas.
In conclusion, the combination of AI with X-ray and DXA provides a more intelligent and precise solution for the diagnosis of OP, significantly improving diagnostic accuracy. Through DL, AI can precisely segment and classify OP regions, enhancing early detection accuracy. However, AI still relies on high-quality equipment and diverse datasets, and the limitations of current datasets may affect diagnostic outcomes across different populations. Additionally, challenges remain in data acquisition, model interpretability, and infrastructure development. As AI technology continues to advance, it is expected to play an increasingly important role in OP diagnosis, improving patient care and treatment outcomes.
3.2 The combination of AI with CT and QCT
CT and QCT also play important roles in the diagnosis of osteoporosis. CT helps detect fractures and changes in bone structure through high-resolution scans, but due to its higher radiation dose, it is generally not used as a routine screening tool (Liebl et al., 2013; Huda et al., 2002). QCT, on the other hand, can accurately quantify bone density, making it particularly suitable for assessing areas like the spine and femur (Chiba et al., 2022). Compared to traditional DXA, QCT offers higher precision and spatial resolution, distinguishing between cortical bone and trabecular bone density, thereby providing a more comprehensive analysis of bone quality (Brunnquell et al., 2021). However, the high radiation dose and cost of CT and QCT remain their limitations. Combining CT and QCT with AI can significantly enhance the sensitivity, specificity, and accuracy of OP diagnostic tools, addressing some of the shortcomings of traditional methods. Yang et al. (2022) explored the use of AI to measure the attenuation values (HU) of the thoracic and first lumbar vertebrae in routine chest CT images for OP screening. The study found that with age, especially in postmenopausal women, the CT attenuation values of the thoracic and first lumbar vertebrae decreased, and these values were significantly correlated with BMD. The attenuation values demonstrated high predictive and diagnostic efficacy. For every 10 HU increase in CT values, the risk of osteopenia or OP decreased by 32–44% and 61–80%, respectively. The combined diagnostic efficacy of all thoracic vertebrae was higher than that of a single vertebra, with AUC values of 0.831 and 0.972, respectively. This study suggests that AI-assisted chest CT can effectively screen high-risk populations and reduce the incidence of fractures. Chen et al.’s study developed a bone density screening tool combining ML segmentation and radiomic texture analysis, using chest low-dose CT (LDCT) for bone density prediction. By analyzing 197 patients, the study demonstrated the high accuracy of the automatic segmentation model and two-level classifier in detecting abnormal bone density and osteoporosis, with an overall prediction accuracy of 0.90 ± 0.05 (Chen et al., 2023). A study investigated the prediction of lumbar vertebral BMD using a DL model based on unenhanced abdominal CT images. The results showed that the BMD predicted by the CNN model was highly correlated with the BMD measured by DXA, with Pearson correlation coefficients of 0.852 and 0.840, respectively. The model demonstrated excellent performance in diagnosing osteoporosis, with AUCs of 0.965 and 0.970 for the internal and external validation datasets, respectively (Yasaka et al., 2020). This study shows that the CNN model in abdominal CT images can accurately reflect the BMD measured by DXA, providing a new approach for non-invasive BMD assessment.
Fang et al. developed an automated method based on deep CNN (DCNN) for vertebral segmentation and BMD calculation in CT images. The study used data from 1,449 patients, with U-Net for vertebral segmentation and DenseNet-121 for BMD calculation. The results showed good correlation between automated and manual segmentation, with average Dice coefficients for lumbar vertebrae of 0.823, 0.786, and 0.782. Additionally, the BMD calculated by the automated method showed high correlation with the BMD measured by QCT (r > 0.98). This method enables the automatic identification of osteoporosis, osteopenia, and normal bone mineral density, providing support for clinical OP screening (Fang et al., 2021). In conclusion, AI combined with CT and QCT offers significant advantages in the screening and diagnosis of osteoporosis, such as improved diagnostic accuracy, automated segmentation and classification, and more precise bone quality analysis. However, the high radiation dose and equipment costs of CT and QCT limit their widespread adoption. Additionally, the AI models suffer from insufficient diversity in their training datasets, and clinical validation is still limited, affecting their application across different populations. Therefore, despite the great potential of AI in imaging diagnostics, issues related to data diversity and interpretability must be addressed in order to achieve broader clinical applications.
3.3 The combination of AI and MRI
MRI has high tissue contrast and is commonly used for soft tissue imaging (Bruno et al., 2019). Compared to X-ray and CT, MRI is more sensitive in detecting early changes in bone marrow, which helps in predicting fracture risk (Zhang et al., 2021). When combined with artificial intelligence (AI), MRI can automatically analyze subtle changes in the images, not only improving diagnostic accuracy but also optimizing image processing, such as noise reduction and image enhancement. The integration of AI with MRI represents a significant advancement in medical imaging, making diagnoses more accurate and efficient, particularly in the early detection of bone diseases. The study by Deniz et al. proposed an automatic proximal femur segmentation method based on DCNN to enhance the clinical application of MRI in bone quality measurement and fracture risk assessment. The study used an MRI dataset from 86 subjects, and after CNN training and four-fold cross-validation, the segmentation results achieved a high Dice similarity coefficient of 0.95, with both precision and recall at 0.95 (Deniz et al., 2018). This suggests that the CNN method can effectively improve the accuracy of bone measurement, aiding in the clinical management of osteoporosis. Yabu et al. developed a CNN-based method for detecting fresh osteoporotic vertebral fractures (OVF), using 1,624 T1-weighted MR images from 814 patients for training and validation. By integrating VGG16, VGG19, DenseNet201, and ResNet50, the model achieved a ROC curve AUC of 0.949, with sensitivity, specificity, and accuracy of 88.1, 87.9, and 88.0%, respectively (Yabu et al., 2021). The study demonstrates the potential of CNN in accurately detecting fresh osteoporotic vertebral fractures, significantly aiding early diagnosis and intervention. Using multiple CNN architectures also enhances the model’s robustness and performance. Overall, MRI combined with AI holds potential in bone quality assessment and fracture risk prediction, but existing studies have small sample sizes, and the generalizability of the results has yet to be validated. Therefore, the clinical application of MRI in OP and fracture-related diseases still requires further validation through large-scale, multi-center clinical trials to confirm its effectiveness and feasibility.
4 Summary and outlook
AI technology plays a crucial role in the diagnosis of osteoporosis. Through ML, DL, and image processing, AI can handle vast amounts of medical data, enabling precise bone density evaluation and fracture risk prediction, thus assisting doctors in making informed decisions. Research shows that AI, combined with traditional imaging technologies such as X-rays, DXA, CT, and MRI, can significantly improve diagnostic accuracy, particularly in early screening and risk assessment, where it demonstrates tremendous potential. However, the practical clinical application of AI still faces several challenges that need to be addressed. First, the issue of AI model interpretability remains unresolved. Despite significant achievements of DL in medical image analysis, its “black box” nature limits the acceptance and trust of clinical practitioners (Dhar et al., 2023). Future research should focus on improving model transparency by using interpretability techniques to help doctors understand the rationale behind model decisions, thereby better integrating AI into the clinical decision-making process. Secondly, the lack of diversity in training data is a major bottleneck restricting the development of AI. Most existing studies are based on data from a single region, ethnicity, or age group, which may result in the model performing unevenly across different populations (Shams et al., 2025). More diverse and global training data are needed in the future to ensure that AI can be applied effectively across different groups. In addition, the lack of clinical validation also hinders the widespread adoption of AI technology. While laboratory data show excellent performance, the complexity of real-world clinical environments presents additional challenges for AI models (van de Sande et al., 2024). Prospective clinical trials must be conducted to validate the actual impact of AI models on patient treatment outcomes, ensuring their feasibility in real-world settings. In addition, the ethical issues and data security concerns of AI must be given high attention. Ensuring patient data privacy, avoiding biases in AI model decisions, and maintaining ethical standards in the use of AI are all important issues that cannot be overlooked in the future application of AI. Finally, as AI continues to advance in the diagnosis and treatment of osteoporosis, optimizing personalized treatment plans will be key to future development. By integrating multimodal data, AI can tailor personalized treatment plans for different patients, thus improving treatment outcomes and patients’ quality of life.
Author contributions
WZ: Writing – review & editing, Writing – original draft. DT: Writing – review & editing. SK: Writing – review & editing, Funding acquisition. KL: Conceptualization, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the Liaoning Province Science and Technology Plan Joint Program (2023JH2/101700222).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that Gen AI was used in the creation of this manuscript. This article has been edited for grammar using GPT-4.1 mini.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Anam, A. K., and Insogna, K. (2021). Update on osteoporosis screening and management. Med. Clin. North Am. 105, 1117–1134. doi: 10.1016/j.mcna.2021.05.016
Areeckal, A. S., Jayasheelan, N., Kamath, J., Zawadynski, S., Kocher, M., and David, S. S. (2018). Early diagnosis of osteoporosis using radiogrammetry and texture analysis from hand and wrist radiographs in Indian population. Osteoporos. Int. 29, 665–673. doi: 10.1007/s00198-017-4328-1
Bloem, H. L., Vande Berg, B., Cardoso, F. N., and Omoumi, P. (2025). “General principles of MRI of the bone marrow” in Musculoskeletal Imaging. eds. T. Pope, J. L. Bloem, W. B. Morrison, D. J. Wilson, and L. White (Cham: Springer Nature Switzerland), 1–22.
Bodden, J., Dieckmeyer, M., Sollmann, N., Burian, E., Rühling, S., Löffler, M. T., et al. (2023). Incidental vertebral fracture prediction using neuronal network-based automatic spine segmentation and volumetric bone mineral density extraction from routine clinical CT scans. Front. Endocrinol. (Lausanne) 14:1207949. doi: 10.3389/fendo.2023.1207949
Brunnquell, C. L., Winsor, C., Aaltonen, H. L., and Telfer, S. (2021). Sources of error in bone mineral density estimates from quantitative CT. Eur. J. Radiol. 144:110001. doi: 10.1016/j.ejrad.2021.110001
Bruno, F., Arrigoni, F., Mariani, S., Splendiani, A., Di Cesare, E., Masciocchi, C., et al. (2019). Advanced magnetic resonance imaging (MRI) of soft tissue tumors: techniques and applications. Radiol. Med. 124, 243–252. doi: 10.1007/s11547-019-01035-7
Chen, Y. C., Li, Y. T., Kuo, P. C., Cheng, S. J., Chung, Y. H., Kuo, D. P., et al. (2023). Automatic segmentation and radiomic texture analysis for osteoporosis screening using chest low-dose computed tomography. Eur. Radiol. 33, 5097–5106. doi: 10.1007/s00330-023-09421-6
Chiba, K., Osaki, M., and Ito, M. (2022). “Assessment of osteoporosis by QCT, HR-pQCT, and MRI” in Osteoporotic fracture and systemic skeletal disorders: Mechanism, assessment, and treatment. eds. H. E. Takahashi, D. B. Burr, and N. Yamamoto (Singapore: Springer Singapore), 177–185.
Deniz, C. M., Xiang, S., Hallyburton, R. S., Welbeck, A., Babb, J. S., Honig, S., et al. (2018). Segmentation of the proximal femur from MR images using deep convolutional neural networks. Sci. Rep. 8:16485. doi: 10.1038/s41598-018-34817-6
Dhar, T., Dey, N., Borra, S., and Sherratt, R. S. (2023). Challenges of deep learning in medical image analysis—improving explainability and trust. IEEE Trans. Technol. Soc. 4, 68–75. doi: 10.1109/TTS.2023.3234203
Fang, Y., Li, W., Chen, X., Chen, K., Kang, H., Yu, P., et al. (2021). Opportunistic osteoporosis screening in multi-detector CT images using deep convolutional neural networks. Eur. Radiol. 31, 1831–1842. doi: 10.1007/s00330-020-07312-8
Fasihi, L., Tartibian, B., Eslami, R., and Fasihi, H. (2022). Artificial intelligence used to diagnose osteoporosis from risk factors in clinical data and proposing sports protocols. Sci. Rep. 12:18330. doi: 10.1038/s41598-022-23184-y
Huda, W., Ravenel, J. G., and Scalzetti, E. M. (2002). How do radiographic techniques affect image quality and patient doses in CT? Semin. Ultrasound CT MR 23, 411–422. doi: 10.1016/s0887-2171(02)90012-0
Kanis, J. A., Norton, N., Harvey, N. C., Jacobson, T., Johansson, H., Lorentzon, M., et al. (2021). SCOPE 2021: a new scorecard for osteoporosis in Europe. Arch. Osteoporos. 16:82. doi: 10.1007/s11657-020-00871-9
Kong, S. H., Ahn, D., Kim, B. R., Srinivasan, K., Ram, S., Kim, H., et al. (2020). A novel fracture prediction model using machine learning in a community-based cohort. JBMR Plus 4:e10337. doi: 10.1002/jbm4.10337
Laskey, M. A. (1996). Dual-energy X-ray absorptiometry and body composition. Nutrition 12, 45–51. doi: 10.1016/0899-9007(95)00017-8
Li, Z., Wang, L., Wu, X., Jiang, J., Qiang, W., Xie, H., et al. (2023). Artificial intelligence in ophthalmology: the path to the real-world clinic. Cell Rep. Med. 4:101095. doi: 10.1016/j.xcrm.2023.101095
Liebl, H., Baum, T., Karampinos, D. C., Patsch, J., Malecki, A., Schaff, F., et al. (2013). Emerging research on bone health using high-resolution CT and MRI. Curr. Radiol. Rep. 2:31. doi: 10.1007/s40134-013-0031-y
Lin, C., Tsai, D. J., Wang, C. C., Chao, Y. P., Huang, J. W., Lin, C. S., et al. (2024). Osteoporotic precise screening using chest radiography and artificial neural network: the OPSCAN randomized controlled trial. Radiology 311:e231937. doi: 10.1148/radiol.231937
Magrabi, F., Ammenwerth, E., McNair, J. B., De Keizer, N. F., Hyppönen, H., Nykänen, P., et al. (2019). Artificial intelligence in clinical decision support: challenges for evaluating AI and practical implications. Yearb. Med. Inform. 28, 128–134. doi: 10.1055/s-0039-1677903
Mao, L., Xia, Z., Pan, L., Chen, J., Liu, X., Li, Z., et al. (2022). Deep learning for screening primary osteopenia and osteoporosis using spine radiographs and patient clinical covariates in a Chinese population. Front. Endocrinol. (Lausanne) 13:971877. doi: 10.3389/fendo.2022.971877
Morgan, S. L., and Prater, G. L. (2017). Quality in dual-energy X-ray absorptiometry scans. Bone 104, 13–28. doi: 10.1016/j.bone.2017.01.033
Njeh, C. F., Fuerst, T., Hans, D., Blake, G. M., and Genant, H. K. (1999). Radiation exposure in bone mineral density assessment. Appl. Radiat. Isot. 50, 215–236. doi: 10.1016/s0969-8043(98)00026-8
Rana, M., and Bhushan, M. (2022). Machine learning and deep learning approach for medical image analysis: diagnosis to detection. Multimed. Tools Appl. 1, 1–39. doi: 10.1007/s11042-022-14305-w
Shams, R. A., Zowghi, D., and Bano, M. (2025). AI and the quest for diversity and inclusion: a systematic literature review. AI Ethics 5, 411–438. doi: 10.1007/s43681-023-00362-w
Smets, J., Shevroja, E., Hügle, T., Leslie, W. D., and Hans, D. (2021). Machine learning solutions for osteoporosis-a review. J. Bone Miner. Res. 36, 833–851. doi: 10.1002/jbmr.4292
Surlari, Z., Budală, D. G., Lupu, C. I., Stelea, C. G., Butnaru, O. M., and Luchian, I. (2023). Current Progress and challenges of using artificial intelligence in clinical dentistry-a narrative review. J. Clin. Med. 12:7378. doi: 10.3390/jcm12237378
Tse, J. J., Smith, A. C. J., Kuczynski, M. T., Kaketsis, D. A., and Manske, S. L. (2021). Advancements in osteoporosis imaging, screening, and study of disease etiology. Curr. Osteoporos. Rep. 19, 532–541. doi: 10.1007/s11914-021-00699-3
Ulivieri, F. M., Rinaudo, L., Piodi, L. P., Messina, C., Sconfienza, L. M., Sardanelli, F., et al. (2021). Bone strain index as a predictor of further vertebral fracture in osteoporotic women: An artificial intelligence-based analysis. PLoS One 16:e0245967. doi: 10.1371/journal.pone.0245967
Van de Sande, D., Chung, E. F. F., Oosterhoff, J., van Bommel, J., Gommers, D., and van Genderen, M. E. (2024). To warrant clinical adoption AI models require a multi-faceted implementation evaluation. NPJ Digit. Med. 7:58. doi: 10.1038/s41746-024-01064-1
Vendrami, C., Gatineau, G., Shevroja, E., Gonzalez Rodriguez, E., Lamy, O., and Hans, D. (2023). Does artificial intelligence have a role in osteoporosis management? Rev. Med. Suisse 19, 752–755. doi: 10.53738/revmed.2023.19.823.752
Wani, I. M., and Arora, S. (2023). Osteoporosis diagnosis in knee X-rays by transfer learning based on convolution neural network. Multimed. Tools Appl. 82, 14193–14217. doi: 10.1007/s11042-022-13911-y
Xu, Y., Liu, X., Cao, X., Huang, C., Liu, E., Qian, S., et al. (2021). Artificial intelligence: a powerful paradigm for scientific research. Innovation (Camb) 2:100179. doi: 10.1016/j.xinn.2021.100179
Yabu, A., Hoshino, M., Tabuchi, H., Takahashi, S., Masumoto, H., Akada, M., et al. (2021). Using artificial intelligence to diagnose fresh osteoporotic vertebral fractures on magnetic resonance images. Spine J. 21, 1652–1658. doi: 10.1016/j.spinee.2021.03.006
Yang, J., Liao, M., Wang, Y., Chen, L., He, L., Ji, Y., et al. (2022). Opportunistic osteoporosis screening using chest CT with artificial intelligence. Osteoporos. Int. 33, 2547–2561. doi: 10.1007/s00198-022-06491-y
Yang, F., Weng, X., Miao, Y., Wu, Y., Xie, H., and Lei, P. (2021). Deep learning approach for automatic segmentation of ulna and radius in dual-energy X-ray imaging. Insights Imaging 12:191. doi: 10.1186/s13244-021-01137-9
Yasaka, K., Akai, H., Kunimatsu, A., Kiryu, S., and Abe, O. (2020). Prediction of bone mineral density from computed tomography: application of deep learning with a convolutional neural network. Eur. Radiol. 30, 3549–3557. doi: 10.1007/s00330-020-06677-0
Zhang, M., Gong, H., and Zhang, M. (2023). Prediction of femoral strength of elderly men based on quantitative computed tomography images using machine learning. J. Orthop. Res. 41, 170–182. doi: 10.1002/jor.25334
Zhang, Y., Ma, M., Tian, C., Liu, J., Huang, X., Duan, Z., et al. (2024). Current status and dilemmas of osteoporosis screening tools: a narrative review. Clin Nutr ESPEN. 64, 207–214. doi: 10.1016/j.clnesp.2024.10.001
Zhang, T., Wu, J., Chen, Y. C., Wu, X., Lu, L., and Mao, C. (2021). Magnetic resonance imaging has better accuracy in detecting new-onset rib fractures as compared to computed tomography. Med. Sci. Monit. 27:e928463. doi: 10.12659/msm.928463
Keywords: osteoporosis, artificial intelligence, machine learning, fracture risk, diagnosis
Citation: Zhang W, Tai D, Kang S and Li K (2025) The application progress of artificial intelligence in osteoporosis diagnosis. Front. Artif. Intell. 8:1699762. doi: 10.3389/frai.2025.1699762
Edited by:
Shilong Su, Peking University Third Hospital, ChinaReviewed by:
Xinzhou Huang, The 3201 Hospital Affiliated to the Medical School of Xi’an Jiaotong University, ChinaCopyright © 2025 Zhang, Tai, Kang and Li. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Keda Li, a29kYXI3NzdAMTYzLmNvbQ==
 Wenwei Zhang
Wenwei Zhang Dongxu Tai2
Dongxu Tai2