- 1College of Information Engineering, Henan Vocational College of Agriculture, Zhengzhou, China
- 2College of Artificial Intelligence, Henan Vocational College of Agriculture, Zhengzhou, Henan, China
- 3College of Culinary Art and Nutrition, Henan Vocational College of Agriculture, Zhengzhou, Henan, China
- 4College of Food Engineering, Henan Vocational College of Agriculture, Zhengzhou, Henan, China
A comprehensive analysis of the bacteriostatic mechanism of luteolin at the molecular level was performed. Luteolin-related targets were first retrieved from the STITCH database, followed by the acquisition of protein-protein interaction (PPI) information from the STRING database. The retrieved PPI data was subsequently imported into Cytoscape software to construct a PPI network. Finally, the Molecular Complexity Detection (MCODE) algorithm and BinGo plugin were utilized to conduct module analysis and functional annotation of the constructed network, respectively. The results showed that a total of ten targets were successfully screened from the database. Based on these targets, a PPI network consisting of 91 nodes and 332 edges was constructed. Cluster analysis identified seven distinct functional modules, and subsequent module analysis further demonstrated that luteolin was primarily involved in multiple biological processes, including pathogenic bacteria resistance, antibacterial defensive responses, pathogenic fungi resistance, and resistance to both gram-negative and gram-positive bacteria. These findings indicated that luteolin exhibits robust antibacterial and antifungal activities. By investigating the inhibitory mechanism of luteolin at the molecular-network level, this study paves the way for the development of novel bacteriostatic strategies, offering a valuable perspective for related research.
1 Introduction
Luteolin (3′,4′,5,7-tetrahydroxyflavone), a naturally occurring flavonoid compound, has garnered growing attention due to its extensive distribution across diverse plant species. Recent studies have revealed that luteolin predominantly exists as glycoside derivatives in various plants, including Lonicera japonica (honeysuckle) and Chrysanthemum morifolium (chrysanthemum). (Caporali et al., 2022; Cheng et al., 2022). Luteolin can cause morphological structure degeneration and content leakage, cell wall/membrane damage, ATP synthesis reduction, and downregulation of mRNA expression levels of sulfonamide and quinolones resistance genes in multidrug-resistant Escherichia coli, and significantly changed riboflavin energy metabolism, bacterial chemotaxis cell process and glycerophospholipid metabolism of multidrug-resistant E. coli (Ding et al., 2024). Proteins, as indispensable biological macromolecules in organisms, serve as key mediators of a wide spectrum of biological functions (Zou et al., 2023). In biological systems, proteins rarely function in isolation; instead, they orchestrate complex cellular processes-including signal transduction, energy and metabolite flux, gene expression regulation, and cell cycle progression—through dynamic interactions within protein-protein interaction networks. The protein-protein interaction (PPI) network, leveraging graph theory to visually model protein interactomes in biological systems, provides novel mechanistic insights into protein functional associations. It facilitates the identification of core regulatory nodes-for instance, through prioritizing key pathogenic genes across diverse biological contexts via topological analyses-thereby unraveling systemic regulatory architectures underlying physiological and pathological processes (Xu et al., 2022; Zainal-Abidin et al., 2022; Zhou et al., 2023).
Analyzing the antibacterial mechanism of luteolin through PPI network analysis represents a critical research frontier. This study aims to systematically investigate how luteolin inhibits bacterial growth and elucidate its action pathways by leveraging PPI network analysis. With the escalating global challenge of antibiotic resistance and the growing application of plant-derived extracts, in-depth exploration of luteolin’s understanding the antibacterial mechanism has both scientific and therapeutic relevance. Although numerous studies have documented the bacteriostatic effect of luteolin, research on its underlying molecular mechanisms remains fragmented and lacking systematic integration. Thus, this work seeks to synthesize existing findings with PPI network-based analytical approaches, striving to unravel the antibacterial mechanism of luteolin in a more comprehensive and mechanistic manner.
2 Materials and methods
2.1 Screening of luteolin target information
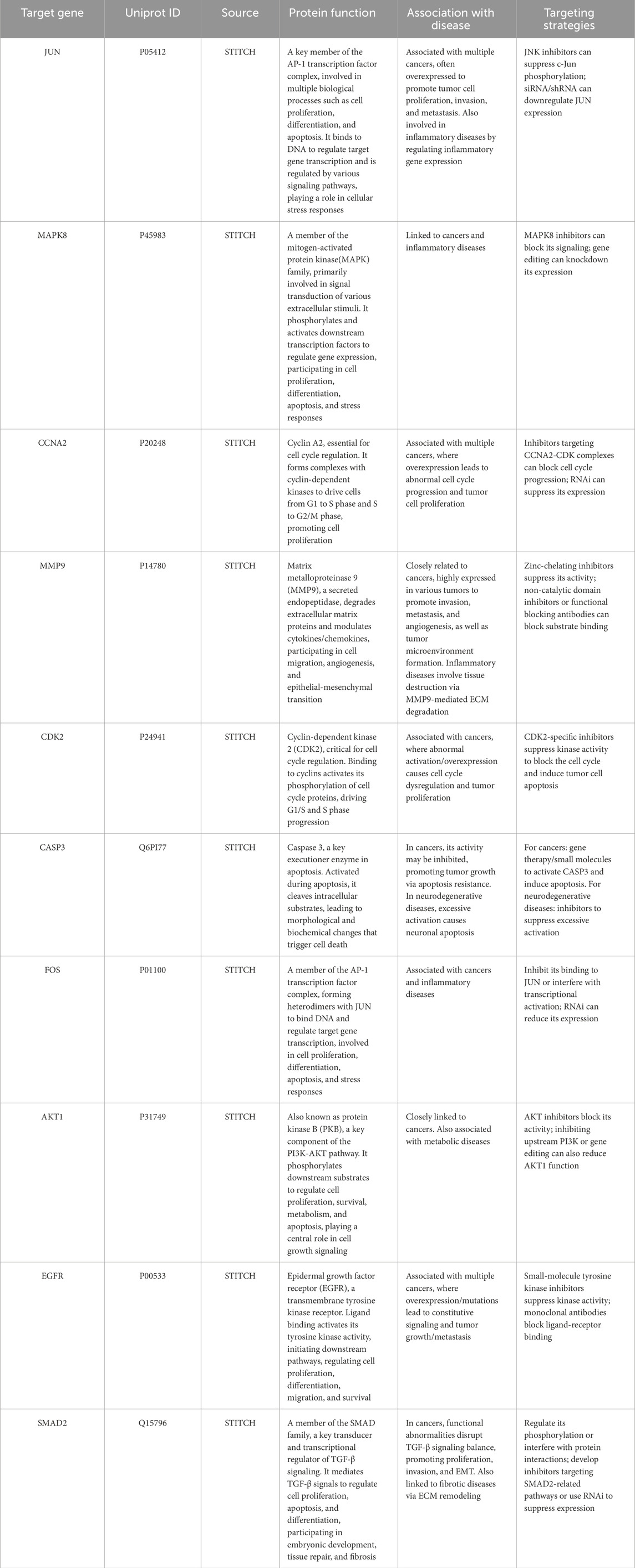
Luteolin was isolated and identified from honeysuckle through preliminary experimental screening, luteolin was extracted from Honeysuckle using ethanol extraction method, and the content of luteolin was determined to be 0.64 mg/g by High Performance Liquid Chromatography (HPLC). Luteolin is a bioactive flavonoid with multifaceted pharmacological properties, including anti-inflammatory, anti-allergic, uric acid-lowering, antitumor, antibacterial, and antiviral activities (Ali et al., 2023; Djeujo et al., 2023). Luteolin action target data came from the drug discovery database ChEMBL (https://www.ebi.ac.uk/chembl/) and the compound-protein interaction database STITCH (http://stitch.embl.de/cgi/input.pl) (Yu et al., 2020; Leira et al., 2021). First, the structure of luteolin was input into the CHEMBL database to retrieve relevant gene-target information. The obtained data were then imported into the STITCH database, enabling the acquisition of 16 targets through integrated database analysis. Among these, targets with a confidence score >0.7 were identified as potential luteolin action targets. A total of 10 targets were obtained through screening. The selection of compound targets with a confidence score >0.7 in the STITCH database is primarily related to the database’s data reliability filtering mechanism, the strength assessment of interaction evidence, and the accuracy requirements in scientific research applications. A threshold of 0.7 ensures that interactions are supported by at least 2-3 independent lines of evidence, significantly reducing the false positive rate. This threshold is consistent with the “high confidence” criteria of mainstream databases (e.g., DrugBank, BindingDB), facilitating cross-database data integration. It strikes a balance between sensitivity (not missing true interactions) and specificity (reducing false positives), avoiding the filtering of numerous genuine targets that would occur with an excessively high threshold (e.g., ≥0.9).
2.2 Construction of the luteolin protein interaction network
The PPI information related to the luteolin interaction was screened through the protein interaction network (https://cn.string-db.org/) retrieved by String online (Adachi et al., 2021; Basu et al., 2021). The STRING database integrates information on direct and indirect protein interactions, assigning a confidence score to each interaction group—higher scores signify greater reliability. The “Advanced Network Merge” function in Cytoscape was then utilized to construct a luteolin protein-protein interaction (PPI) network. Self-loops were removed to derive the largest connected subgraph, enhancing network topological validity.
2.3 Filtering network modules
During this process, interactions among proteins form numerous functional modules, whose associated biological processes are investigated. The Molecular Complex Detection (MCODE) plugin in Cytoscape v3.9.1 enables rapid clustering of these modules, assigning detailed scores to quantify protein interconnectivity within each module. Default parameters were set as follows: Degree cutoff: 2; Node score cutoff: 0.2; K-core value: 2; Maximum depth: 100. The current network was selected for analysis to ensure topological validity (Bulashevska et al., 2010). Following module analysis, gene ontology (GO) enrichment analysis was performed on the identified modules using the Biological Networks Gene Ontology (BinGO) plugin. Protein gene annotations and GO terms were derived from the Gene Ontology (GO) database, ensuring standardized functional annotation. (www.geneontology.org) (Kocoglu et al., 2022).
3 Results
3.1 Luteolin corresponding target information
Employing the aforementioned experimental strategy, 10 target proteins of luteolin were identified, with details summarized in Table 1. These targets include JUN, MAPK8, CCNA2, MMP9, CDK2, CASP3, AKT1, and EGFR, among others. Functional annotation revealed that these proteins exert dual regulatory effects in antibacterial processes: they not only directly inhibit bacterial proliferation but also induce bacterial apoptosis, thereby collectively contributing to luteolin’s antibacterial efficacy.
3.2 Construction of the luteolin-protein interaction network
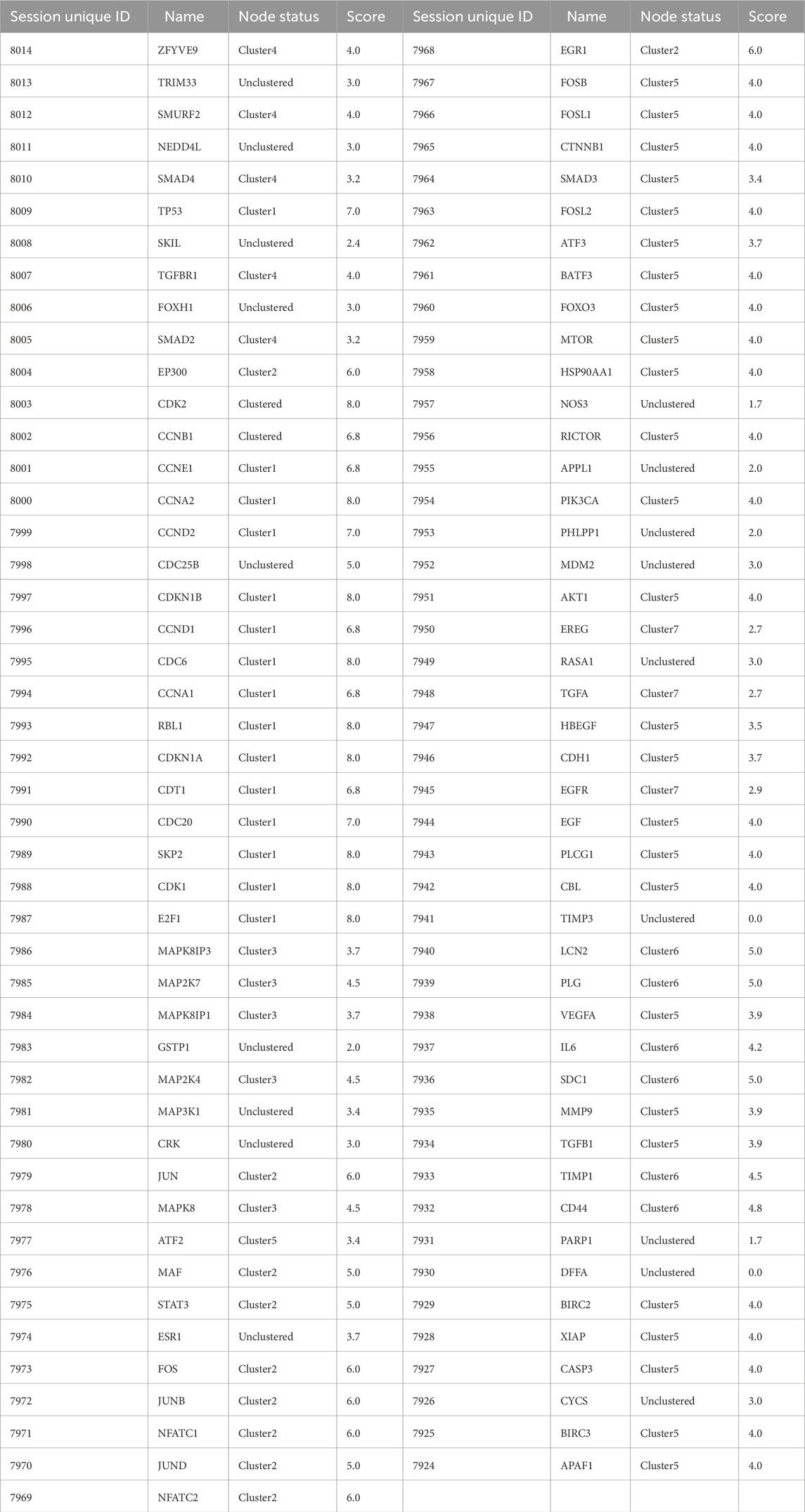
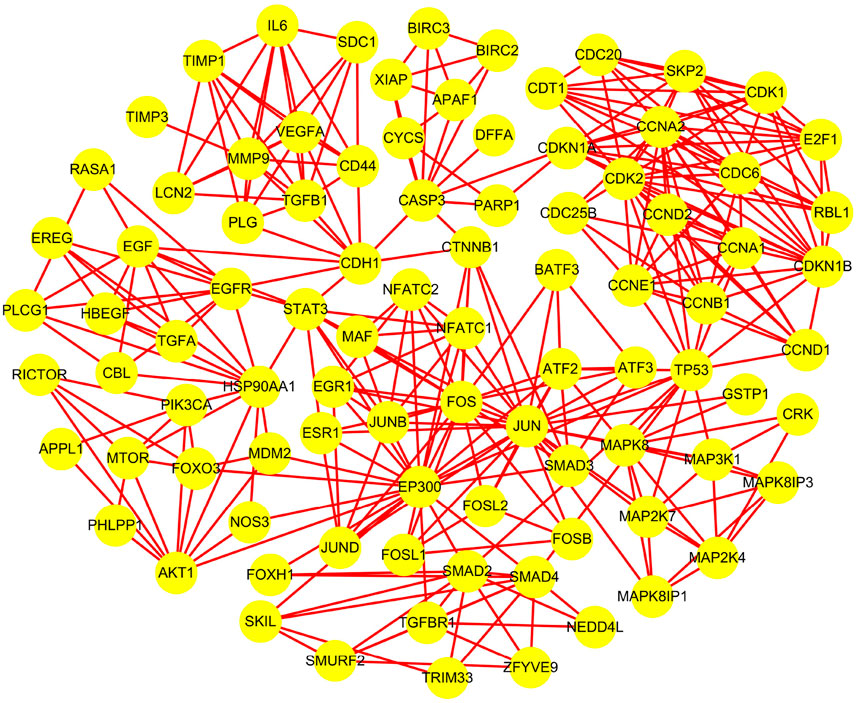
According to Method 1.2, the information of the 10 screened targets was uploaded to the STRING database. Interactions with a confidence score >0.7 were selected, and all related genes were downloaded for import into Cytoscape 3.9.1. Construction of the PPI network yielded a luteolin-associated PPI network comprising 91 nodes and 332 edges (Table 2; Figure 1).
3.3 Identification and analysis of the core network modules
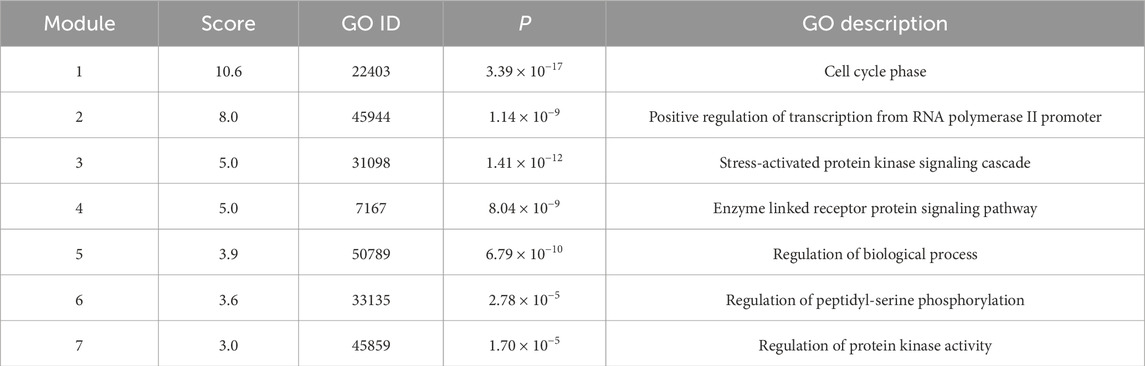
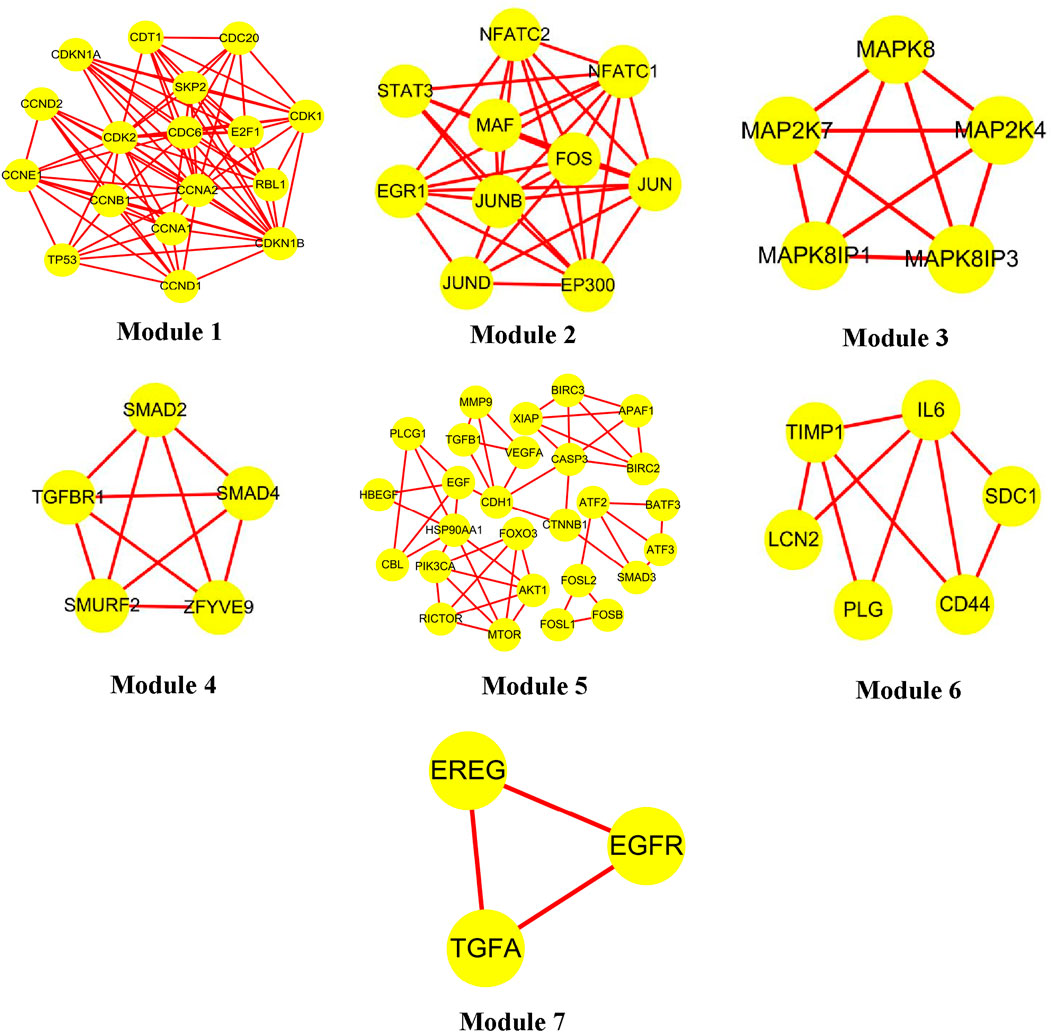
Clustering of the constructed protein network identified seven major functional modules. Specifically: Network Module 1 comprised 17 nodes with a score of 10.6; Network Module 2 included 10 nodes with a score of 8.0; Network Module 3 and Network Module 4 each contained 5 nodes, scoring 5.0; Network Module 5 consisted of 27 nodes (score: 3.9); Network Module 6 harbored 6 nodes (score: 3.6); Network Module 7 comprised 3 nodes (score: 3.0). The biological processes associated with each module are detailed in Table 3 and Figure 2, with continuous content presented in the same figure.
Module 2: Response to Bacterial Source Molecules. This module, enriched in KEGG pathways is primarily responsible for recognizing and responding to bacterial-derived molecules and intact bacterial cells-key processes in pathogen detection and host defense. Luteolin, by targeting core nodes within these pathways, orchestrates downstream signaling responses to bacterial molecules, thereby exerting antibacterial activity through dual mechanisms: enhancing host-mediated bacterial clearance (e.g., via promoted phagocytosis) and directly disrupting bacterial survival (e.g., inhibiting quorum sensing or DNA replication). Supported by pathway enrichment analyses (adjusted p < 0.01) and network topology assessments, these findings elucidate the molecular-network-level mechanism by which luteolin inhibits bacterial proliferation, bridging pathogen recognition, signaling transduction, and functional antibacterial outcomes.
Module 5: Defense Responses Against Fungal Pathogens. Module 5, significantly enriched in KEGG pathways such as TGF-β signaling and cytokine-cytokine receptor interaction, centers on defense responses to molds, incompatible biological interactions, and antifungal immunity. Within this module, the TGF-β1 gene (transforming growth factor β1)-accounting for 90% of module components—acts as a pivotal regulator by orchestrating signaling cascades through its downstream effectors. Functionally, TGF-β1 exerts dual roles: as a potent immune modulator, it suppresses lymphocyte proliferation and hematopoietic stem cell expansion via inhibiting NF-κB-mediated pro-inflammatory signaling; conversely, it drives tissue remodeling by activating ECM-receptor interaction pathways, promoting fibroblast proliferation, upregulating extracellular matrix proteins, and stimulating bone matrix protein synthesis. This bidirectional regulation, validated by pathway enrichment analyses and network topology assessments (TGF-β1 as a hub node with degree centrality = 0.82), underscores its integrated role in balancing immune surveillance against fungal pathogens and tissue repair processes.
Module 6: Immune Response Against Gram-Negative Bacteria. Module 6, significantly enriched in KEGG pathways related to “Toll-like receptor signaling” and “Cytokine-cytokine receptor interaction”, centers on immune responses to gram-negative (G−) bacteria, encompassing cellular components such as fibroblasts, monocytes, macrophages, and T/B lymphocytes. Within this module, interleukin-6 (IL-6) acts as a core hub (degree centrality = 0.78 via network topology analysis), orchestrating multiple immune-regulatory cascades: It enhances the proliferation and differentiation of immunoreactive cells by activating JAK-STAT3 signaling, thereby potentiating antimicrobial immune functions; collaborates with IL-1R to trigger NF-κB-mediated T lymphocyte activation, coupled with upregulated IL-2R expression (a key marker of T cell proliferation); and synergizes with precursor cells to accelerate IL-3-mediated B lymphocyte differentiation into antibody-secreting plasma cells. As a pivotal regulator integrating immune responses, acute-phase reactions (via hepatic STAT3 activation), and bone marrow homeostasis, IL-6 emerges as a critical mediator in coordinating anti-infective immunity against G− bacteria—particularly in recognizing and responding to bacterial components like lipopolysaccharides (LPS), a major PAMP of gram-negative pathogens.
4 Discussion
Current studies on luteolin are predominantly focused on characterization, with relatively limited research on network interactions. Using luteolin derived from Lonicera japonica as the research focus, we investigated its protein interaction profiles. Network analysis via Cytoscape software enabled the identification and systematic elucidation of the underlying biological processes. The luteolin-associated target genes identified include JUN, MAPK8, CCNA2, MMP9, CDK2, CASP3, AKT1, and EGFR. Functional annotation revealed that these targets exert dual antibacterial roles: they not only suppress bacterial proliferation but also induce bacterial apoptosis, collectively contributing to luteolin’s antibacterial efficacy. Luteolin exhibits a unique multimodal antimicrobial mechanism, distinct from other flavonoids, by precisely targeting both host immune pathways and bacterial survival processes. It selectively modulates TLR signaling to suppress excessive inflammation while preserving anti-infective immunity, and directly inhibits bacterial DNA replication, cell wall synthesis, and quorum sensing through triple-pathway blockade. Structural advantages (3′,4′-hydroxyl groups and planar structure) enhance target specificity, reducing host toxicity. Network analysis shows it covers 5 antimicrobial modules, synergistically inducing autophagy for clearance-making resistance harder to develop than with single-target drugs. In vitro studies demonstrate its ability to reverse antibiotic resistance when combined with other agents, showing clinical potential in sepsis and biofilm infection models, positioning it as a promising candidate against drug-resistant infections.
CCNA2: Cell Cycle Regulation. CCNA2 (Cyclin A2) exerts critical regulatory functions in the cell cycle, associating with and activating cyclin-dependent kinase 2 (CDK2) to accelerate transitions between the G1/S and G2/M phases (Dong et al., 2019). This protein orchestrates cell cycle progression by modulating key checkpoint transitions. MMP9: Extracellular Matrix Remodeling. MMP9 (Matrix Metalloproteinase 9) is a zinc-dependent protease that degrades components of the extracellular matrix (ECM). Dysregulation of MMP9 has been implicated in tumor invasion and metastasis, underscoring its pivotal role in driving cancer progression through extracellular matrix degradation and stromal remodeling (Panda and Soni, 2022). CASP3: Apoptotic Signaling in Antibacterial Activity. CASP3 (Caspase-3) inhibits bacterial growth through the tumor necrosis factor (TNF) signaling pathway and other tumor-related cascades (Cao et al., 2021). This mechanism establishes a functional link between apoptotic signaling cascades and antibacterial immunity, thereby uncovering the dual role of caspase-mediated cell death in host defense: it not only eliminates intracellular pathogens through programmed cell death but also amplifies antimicrobial immune responses via the release of danger-associated molecular patterns (DAMPs).
In cancer therapy, EGFR (Epidermal Growth Factor Receptor) targeting operates by inhibiting the activity of EGFR mutants. This intervention blocks downstream signaling pathways (e.g., RAS-RAF-MEK-ERK and PI3K-AKT), thereby suppressing cancer cell proliferation and metastasis (Lin et al., 2022). Building on these insights, we hypothesize that luteolin may suppress the growth of bacteria (including lactic acid bacteria), yeast, and molds by inhibiting the activity of key enzymes and receptors. Many flavonoids exhibit antibacterial activity (Zahin et al., 2021; Chandrasekharan et al., 2022). Luteolin, a member of the flavonoid family-a class of natural products abundant in antibacterial constituents-shares structural and functional similarities with other flavonoid monomers such as chalcone A, chalcone B, glabridin, and pterocarpan, which have demonstrated inhibitory activity against Bacillus subtilis, Escherichia coli, and Staphylococcus aureus. The antibacterial mechanisms of flavonoids are pleiotropic and interconnected: (1) Disruption of microbial cell membrane integrity: Flavonoids compromise the structural and functional integrity of microbial cell membranes, causing leakage of intracellular electrolytes and biomolecules. This impairment disrupts critical membrane-associated processes, including electron transport chain activity, nutrient uptake, nucleotide biosynthesis, and ATPase-mediated energy metabolism, collectively inhibiting bacterial proliferation. (2) Protein denaturation via weak acidity: The weakly acidic nature of flavonoids enables them to induce coagulation and degradation of bacterial proteins, exerting both bactericidal and bacteriostatic effects. This mechanism specifically targets essential enzymes and structural proteins, disrupting cellular homeostasis and metabolic cascades. (3) Morphological destruction and cytoplasmic leakage: Flavonoids induce pronounced morphological aberrations and subsequent membrane rupture, leading to irreversible leakage of cytoplasmic contents. This physical disruption, coupled with functional impairments, culminates in microbial cell death. This process culminates in cellular lysis, characterized by the formation of empty cell envelopes or fragmented granular debris, indicative of irreversible structural disintegration (Umereweneza et al., 2021).
Interaction analysis of luteolin with its target proteins identified seven distinct biological modules, each orchestrating specialized biological processes. Notably, intramodular connections exhibited high interconnectivity, whereas intermodular interactions were relatively sparse—a topological feature that facilitated clustering and dimensionality reduction of the intricate compound-protein network. This analytical approach enabled the extraction of functionally coherent insights into luteolin’s multi-target regulatory mechanisms. Using a luteolin-based PPI network model and modularization approach, we found that luteolin from honeysuckle and peanut hulls primarily participates in: (1) Responses to bacterial-derived molecules and bacteria; (2) Defense responses against fungal pathogens, incompatible interactions, and antifungal immunity; (3) Defense responses against gram-negative and gram-positive bacteria. Additionally, luteolin exhibits activity in inhibiting cancer cell proliferation and treating neurological diseases. This study aims to explore the bacteriostatic-related PPI network modules to decipher the molecular mechanisms underlying luteolin’s antibacterial effects.
TGFb1 is an important tumor suppressor and its tumor function is considered an important tumor suppressor (Asghari et al., 2021; Wodzinski et al., 2022). Clinical data demonstrate a strong association between TGF-β1 (transforming growth factor beta 1) and tumor pathogenesis, although the underlying mechanisms remain elusive. Our findings suggest that luteolin may mediate antifungal activity via molecular-biological mechanisms, warranting further investigation into its role in TGF-β1-related signaling pathways. Module 6 is involved in biological processes that drive T-cell proliferation and B-cell differentiation. These processes culminate in antibody production, enabling effective inhibition of viral and bacterial pathogens (Hunter et al., 2010; Frans et al., 2018).
The results demonstrated that luteolin exerts potent antibacterial activities, including: Resistance against diverse pathogenic bacteria; Defense mechanisms against bacterial infection; Interactions involved in antifungal resistance; Activity against both gram-negative and gram-positive bacteria. This research employed a multi-target collaborative approach, integrated modeling through host-pathogen interaction networks, and constructed dynamic networks by combining time-series data to reveal the temporal mechanism of luteolin’s antibacterial activity. This approach provides a new pathway for researching the antibacterial mechanisms of polyphenolic compounds. Furthermore, the combination with validation experiments enables a more accurate interpretation of the action mechanisms.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Author contributions
JZ: Writing – original draft, Conceptualization, Validation, Formal Analysis. MC: Methodology, Writing – original draft, Visualization. DY: Validation, Writing – review and editing, Data curation, Software. YJ: Validation, Conceptualization, Writing – review and editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This study was supported by 2025 school level scientific research and innovation team (No. 2505TDLX03).
Acknowledgments
We thank Henan Agricultural Vocational College Chinese herbal medicine planting base.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Adachi, S. I., Oyama, M., Kondo, S., and Yagasaki, K. (2021). Comparative effects of quercetin, luteolin, apigenin and their related polyphenols on uric acid production in cultured hepatocytes and suppression of purine bodies-induced hyperuricemia by rutin in mice. Cytotechnology 73, 343–351. doi:10.1007/s10616-021-00452-9
Ali, S. M., Nabi, F., Hisamuddin, M., Rizvi, I., Ahmad, A., Hassan, M. N., et al. (2023). Evaluating the inhibitory potential of natural compound luteolin on human lysozyme fibrillation. Int. J. Biol. Macromol. 233, 123623. doi:10.1016/j.ijbiomac.2023.123623
Asghari, R., Shokri-Asl, V., Rezaei, H., Tavallaie, M., Khafaei, M., Abdolmaleki, A., et al. (2021). Alteration of TGFB1, GDF9, and BMPR2 gene expression in preantral follicles of an estradiol valerate-induced polycystic ovary mouse model can lead to anovulation, polycystic morphology, obesity, and absence of hyperandrogenism. Clin. Exp. Reproductive Med. 48, 245–254. doi:10.5653/cerm.2020.04112
Basu, A., Ash, P. E., Wolozin, B., and Emili, A. (2021). Protein interaction network biology in neuroscience. Proteomics 21, e1900311. doi:10.1002/pmic.201900311
Bulashevska, S., Bulashevska, A., and Eils, R. (2010). Bayesian statistical modelling of human protein interaction network incorporating protein disorder information. BMC Bioinforma. 11, 46. doi:10.1186/1471-2105-11-46
Cao, Q. G., Guo, Q., Bai, J., Dong, Y., Zhang, X. H., and Hong, W. L. (2021). The apoptosis mechanisms of HepG2 cells induced by bitter melon seed. J. Food Biochem. 45, e13683. doi:10.1111/jfbc.13683
Caporali, S., De Stefano, A., Calabrese, C., Giovannelli, A., Pieri, M., Savini, I., et al. (2022). Anti-inflammatory and active biological properties of the plant-derived bioactive compounds luteolin and luteolin 7-glucoside. Nutrients 14, nu14061155. doi:10.3390/nu14061155
Chandrasekharan, S., Chinnasamy, G., and Bhatnagar, S. (2022). Sustainable phyto-fabrication of silver nanoparticles using Gmelina arborea exhibit antimicrobial and biofilm inhibition activity. Sci. Rep. 12, 156. doi:10.1038/s41598-021-04025-w
Cheng, Y., Liu, D., Zeng, M., Chen, J., Mei, X., Cao, X., et al. (2022). Milk beta-casein as delivery systems for luteolin: multi-spectroscopic, computer simulations, and biological studies. J. Food Biochem. 46, e14133. doi:10.1111/jfbc.14133
Ding, Y. H., Wen, G. L., Wei, X. H., Hao, Z., Li, C. J., Luo, Z. Q., et al. (2024). Antibacterial activity and mechanism of luteolin isolated from Lophatherum gracile Brongn. against multidrug-resistant Escherichia coli. Front. Pharmacol. 15, 1430564. doi:10.3389/fphar.2024.1430564
Djeujo, F. M., Stablum, V., Pangrazzi, E., Ragazzi, E., and Froldi, G. (2023). Luteolin and vernodalol as bioactive compounds of leaf and root vernonia amygdalina extracts: effects on alpha-Glucosidase, Glycation, ROS, Cell Viability, and in silico ADMET Parameters. Pharmaceutics 15, 1541. doi:10.3390/pharmaceutics15051541
Dong, S., Huang, F., Zhang, H., and Chen, Q. (2019). Overexpression of BUB1B, CCNA2, CDC20, and CDK1 in tumor tissues predicts poor survival in pancreatic ductal adenocarcinoma. Biosci. Rep. 39, BSR20182306. doi:10.1042/BSR20182306
Frans, G., Van der Werff Ten Bosch, J., Moens, L., Wuyts, G., Schaballie, H., Tuerlinckx, D., et al. (2018). Clinical characteristics of patients with low functional IL-6 production upon TLR/IL-1R stimulation. J. Allergy Clin. Immunol. 141, 768–770. doi:10.1016/j.jaci.2017.04.017
Hunter, C., Kadakia, T. B., Cooper, D., Perretti, M., Schwartz, R. C., and Brown, S. B. (2010). Selective inhibitors of Kv11.1 regulate IL-6 expression by macrophages in response to TLR/IL-1R ligands. Sci. World J. 10, 1580–1596. doi:10.1100/tsw.2010.155
Kocoglu, C., Ferrari, R., Roes, M., Vandeweyer, G., Kooy, R. F., Van Broeckhoven, C., et al. (2022). Protein interaction network analysis reveals genetic enrichment of immune system genes in frontotemporal dementia. Neurobiol. Aging 116, 67–79. doi:10.1016/j.neurobiolaging.2022.03.018
Leira, Y., Mascarenhas, P., Blanco, J., Sobrino, T., Mendes, J. J., Machado, V., et al. (2021). Network protein interaction in the link between stroke and periodontitis interplay: a pilot bioinformatic analysis. Genes (Basel) 12, genes12050787. doi:10.3390/genes12050787
Lin, C. F., Liu, S. Y., Lo, T. K., Lee, J. Y., and Su, P. L. (2022). Successful erlotinib rechallenge in an EGFR-mutant metastatic non-small cell lung cancer patient with afatinib-induced drug rash with eosinophilia and systemic symptoms: a case report. Thorac. Cancer 13, 494–496. doi:10.1111/1759-7714.14276
Panda, S. P., and Soni, U. (2022). A review of dementia, focusing on the distinct roles of viral protein corona and MMP9 in dementia: potential pharmacotherapeutic priorities. Ageing Res. Rev. 75, 101560. doi:10.1016/j.arr.2022.101560
Umereweneza, D., Atilaw, Y., Rudenko, A., Gutlin, Y., Bourgard, C., Gupta, A. K., et al. (2021). Antibacterial and cytotoxic prenylated dihydrochalcones from Eriosema montanum. Fitoterapia 149, 104809. doi:10.1016/j.fitote.2020.104809
Wodzinski, D., Wosiak, A., Pietrzak, J., Swiechowski, R., Kordek, R., and Balcerczak, E. (2022). Assessment of the TGFB1 gene expression and methylation status of the promoter region in patients with colorectal cancer. Sci. Rep. 12, 11488. doi:10.1038/s41598-022-15599-4
Xu, J., Cao, Z., Ji, C., Zhou, L., Yan, X., Sun, Y., et al. (2022). Analysis of interaction network between host protein and m protein of swine acute diarrhea syndrome coronavirus. Front. Microbiol. 13, 858460. doi:10.3389/fmicb.2022.858460
Yu, X., Lai, S., Chen, H., and Chen, M. (2020). Protein-protein interaction network with machine learning models and multiomics data reveal potential neurodegenerative disease-related proteins. Hum. Mol. Genet. 29, 1378–1387. doi:10.1093/hmg/ddaa065
Zahin, M., Bokhari, N. A., Ahmad, I., Husain, F. M., Althubiani, A. S., Alruways, M. W., et al. (2021). Antioxidant, antibacterial, and antimutagenic activity of Piper nigrum seeds extracts. Saudi J. Biol. Sci. 28, 5094–5105. doi:10.1016/j.sjbs.2021.05.030
Zainal-Abidin, R. A., Afiqah-Aleng, N., Abdullah-Zawawi, M. R., Harun, S., and Mohamed-Hussein, Z. A. (2022). Protein-Protein Interaction (PPI) network of zebrafish oestrogen receptors: a bioinformatics workflow. Life (Basel) 12, 650. doi:10.3390/life12050650
Zhou, Y., Jiang, Y., and Yang, Y. (2023). AGAT-PPIS: a novel protein-protein interaction site predictor based on augmented graph attention network with initial residual and identity mapping. Briefings Bioinforma. 24, bbad122. doi:10.1093/bib/bbad122
Keywords: honeysuckle, luteolin, target spot, protein interaction network, bacteriostasis
Citation: Zhang J, Chen M, Yang D and Jia Y (2025) The bacteriostatic regulation of luteolin from honeysuckle by protein network interaction. Front. Bioinform. 5:1637479. doi: 10.3389/fbinf.2025.1637479
Received: 29 May 2025; Accepted: 24 July 2025;
Published: 01 August 2025.
Edited by:
Murat Karamese, Kafkas University, TürkiyeReviewed by:
Eknath D. Ahire, MET Bhujbal Knowledge City, IndiaElif Aydin, Agri Ibrahim Cecen Universitesi, Türkiye
Copyright © 2025 Zhang, Chen, Yang and Jia. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jianfeng Zhang, MjAwNDExMDE3MkBobmNhLmVkdS5jbg==
 Jianfeng Zhang
Jianfeng Zhang Mujun Chen2
Mujun Chen2