- 1College of Life Science, Fujian Normal University, Fuzhou, Fujian, China
- 2Institute of Agricultural Quality Standards and Testing Technology Research & Fujian Key Laboratory of Agro-products Quality and Safety, Fujian Academy of Agricultural Sciences, Fuzhou, Fujian, China
- 3Institute of Food Science and Technology, Fujian Academy of Agricultural Sciences, Fuzhou, Fujian, China
- 4Research Department, Guangdong Yuehai Feed Group Co., LTD, Zhanjiang, Guangdong, China
- 5Department of Gastroenterology, Fujian Provincial Geriatric Hospital, Fuzhou, Fujian, China
Docosahexaenoic acid (DHA) positively influences growth development, feed efficiency, physiological functions, and DHA accumulation in aquatic organisms. Recent mammalian studies demonstrate that DHA in the form of monoglycerides (MGs) is more conducive to facilitating the uptake and impact of DHA bioavailability than triglyceride (TG)-derived DHA. In this study, Schizochytrium-derived monoglyceride as a novel structured lipid form enriched in DHA-MGs was used to replace the fish oil of Litopenaeus vannamei diet and the effects of different DHA-MG levels on the growth performance, lipid composition of muscle and hepatopancreas, and intestinal microbiota of L. vannamei were investigated. The results showed that the diet with a high level of DHA-MGs distinctly increased the final weight, weight gain rate, and specific growth rate of L. vannamei. No notable variation in the biochemical makeup of shrimp muscle was detected among the four diets. The increase in DHA-MGs effectively facilitated the accumulation of DHA in the muscle and hepatopancreas of L. vannamei. In terms of lipidomics analysis, the addition of DHA-MGs significantly influenced the lipid species composition and DHA distribution in shrimp muscle and hepatopancreas. Additionally, the obtained results revealed that the addition of DHA-MGs improved the contents of diglycerides and triglycerides from the neutral lipid category with DHA and DHA located at specific positions on the glycerol backbone for shrimp muscle. Meanwhile, the lipid species from the glycerophospholipid category in shrimp tissues treated with four diets performed different variations. Besides that, the incorporation of DHA-MGs influenced the levels of microbial phylum and genus and the abundance of potential bacterial phenotypes for shrimp midgut. It was noted that the addition of DHA-MGs significantly decreased the abundance of potentially pathogenic bacteria, manifesting that DHA-MGs could readjust the intestinal microecological balance for intestinal health. In conclusion, the use of Schizochytrium-derived DHA-MGs to replace the fish oil in shrimp diet boosted the accumulation of DHA into tissues, modified the levels of lipid species with DHA and DHA located at specific positions on the glycerol backbone, and improved the intestinal health for the growth performance of L. vannamei.
1 Introduction
DHA (C22:6n-3), with its unique structure and multiple double bonds, plays a crucial role in the normal development and function of neural and retinal tissues throughout the human lifespan (Carlson et al., 2013; Gharami et al., 2015). Shrimp is widely recognized as a rich dietary source of DHA. Litopenaeus vannamei, referred to as Pacific white shrimp, is the most important commercial shrimp species extensively cultivated in various nations, including Mexico, Peru, and China (Dugassa and Gaetan, 2018). The annual global production of L. vannamei (>5.0 million tons) provides substantial DHA to consumers, supporting dietary intake requirements. Numerous studies have indicated that the DHA or fish oil content in commercial shrimp feeds significantly influences the DHA concentration found in L. vannamei muscle (Araújo et al., 2019; Kumar et al., 2022). Consequently, augmenting the DHA concentration in L. vannamei muscle has emerged as a prominent subject in aquatic research.
Currently, DHA supplements for shrimp feed are predominantly sourced from fish oil. Approximately 60% of the worldwide fish oil supply is utilized in aquafeeds. Nonetheless, the global fish oil output is insufficient to satisfy the demand for aquatic feeds, and its price has exhibited an increasing trend in recent years, which is attributed to overfishing and environmental contamination (Neylan et al., 2024). Aquatic scientists have dedicated efforts to generate novel DHA-enriched lipid sources from microorganisms to replace marine-derived DHA in feed formulations (Sprague et al., 2017). Among the investigated microorganisms, Schizochytrium sp. is a promising DHA source due to its rapid growth, high yield, and simple fermentation (Carr et al., 2023; Tocher et al., 2019). Critically, substituting fish oil with Schizochytrium sp. oil in shrimp feeds maintains growth performance while significantly enhancing the DHA deposition in shrimp muscle (Kumar et al., 2018; Wang et al., 2017; Xie et al., 2020). Consequently, DHA-rich Schizochytrium sp. oil represents a viable sustainable lipid source to replace the limited and unsustainable fish oil for aquafeeds (Farris et al., 2024).
In terms of the lipid class, the lipid composition of commercial fish oils for aquatic feeds is dominated by TGs (>98% of total lipids). Similarly, Schizochytrium sp. oil, abundant in DHA, also primarily comprises TGs (Shen et al., 2024). The dietary TGs containing DHA are firstly hydrolyzed by lipolytic enzymes to generate free fatty acids (FFAs) and monoglycerides (MGs) in the intestinal tract (Grabner et al., 2017). Following this, FFAs and MGs are absorbed and re-esterified to form TGs, cholesteryl esters (CEs), and glycerophospholipids (GPLs), which are assembled and then secreted by the hepatopancreas and hemolymph to deposit DHA into shrimp muscles (Destaillats et al., 2018). Additionally, recent studies about mammals have proven that the animals treated with DHA-MGs are more conducive to facilitating DHA bioavailability and accumulation in comparison to the animals treated with DHA-TGs (Chevalier and Plourde, 2021; Jin et al., 2020; Li et al., 2025). Previous studies have indicated that DHA positively influences growth development, feed efficiency, physiological functions, and DHA accumulation in aquatic organisms (An et al., 2023; Jiang et al., 2022; Scheuer et al., 2024). To date, limited research has been conducted on DHA-MGs to enhance DHA absorption and accumulation in L. vannamei by substituting the fish oil in shrimp diets. To the best of our knowledge, this study investigated for the first time the effects of DHA-MG supplementation as a substitute for fish oil on the growth performance and DHA deposition in L. vannamei.
Prior research has reported that the lipids of L. vannamei body or tissue can be classified into neutral lipids (NLs), GPLs, sphingolipids (SPs), and so on (Wang et al., 2024). Furthermore, the DHA content and distributional characteristics of a specific lipid class in shrimp are correlated with the DHA levels and lipid sources of aquafeeds (Chen et al., 2024a; López-Marcos et al., 2024). As previously mentioned, the digested FFAs and MGs with different fatty acids are further re-esterified into the specific lipid class of aquatic muscle that is dependent on the hematopoietic enzymes (e.g., lipase, phospholipase) with their natural fatty acid selectivity and positional specificity (Destaillats et al., 2018; Gyamfi et al., 2019). On the other hand, the intestinal tract of aquatic animals is a crucial tissue for trillions of microbial cells known as gut microbiota, which are essential for nutrition absorption, immune response regulation, and homeostasis maintenance (Gao et al., 2023; Zeng et al., 2024). Additionally, the intestinal microbial community and abundance are affected by the aquafeed composition, as it can metabolize the ingested nutrients into microbial biomass and some health-beneficial substances to decrease the risk of diseases (Luan et al., 2023). Recently, researchers mainly focused on the characterization of the intestinal microbiota by altering the shrimp feed composition such as lipid source (Zhang et al., 2014), carbohydrate source (Chen et al., 2021), and protein ratio (Gyan et al., 2022). To investigate the relationship between DHA-MG supplementation and shrimp lipid composition and intestinal microbiota, this study systematically analyzed changes in lipid species and DHA distribution while characterizing shifts in microbial community structure and richness.
This study pioneers the use of dietary DHA-MGs as a partial fish oil substitute, evaluating their effects on growth performance and tissue lipid composition in L. vannamei. Furthermore, the lipidomical characteristics of L. vannamei tissues were deeply analyzed to understand the correlation between DHA-MG addition and DHA distribution. Furthermore, the influences of DHA-MGs on the lipid species and intestinal microbiota of shrimp were revealed to ascertain the suitable DHA-MG addition. The results will facilitate the utilization of an innovative Pacific white shrimp diet including Schizochytrium-derived DHA-MGs to enhance DHA accumulation and alter the intestinal microbiota for shrimp growth.
2 Materials and methods
2.1 Preparation of the experimental diets
Schizochytrium sp. TG was purchased from Fuqing King Dnarmsa Spirulina Co., Ltd. (Fujian, China). The DHA-MG sample was synthesized through the ethanolysis of Schizochytrium sp. TGs (100 g) with Candida antarctica lipase A, as stated in our previous study (He et al., 2019). Following ethanolysis, the DHA-MG product was purified by biphasic extraction method with n-hexane and 90% ethanol. The DHA-MG product, comprising 77.6% DHA and 16.8% DPA, was preserved at -20°C for utilization (He et al., 2019).
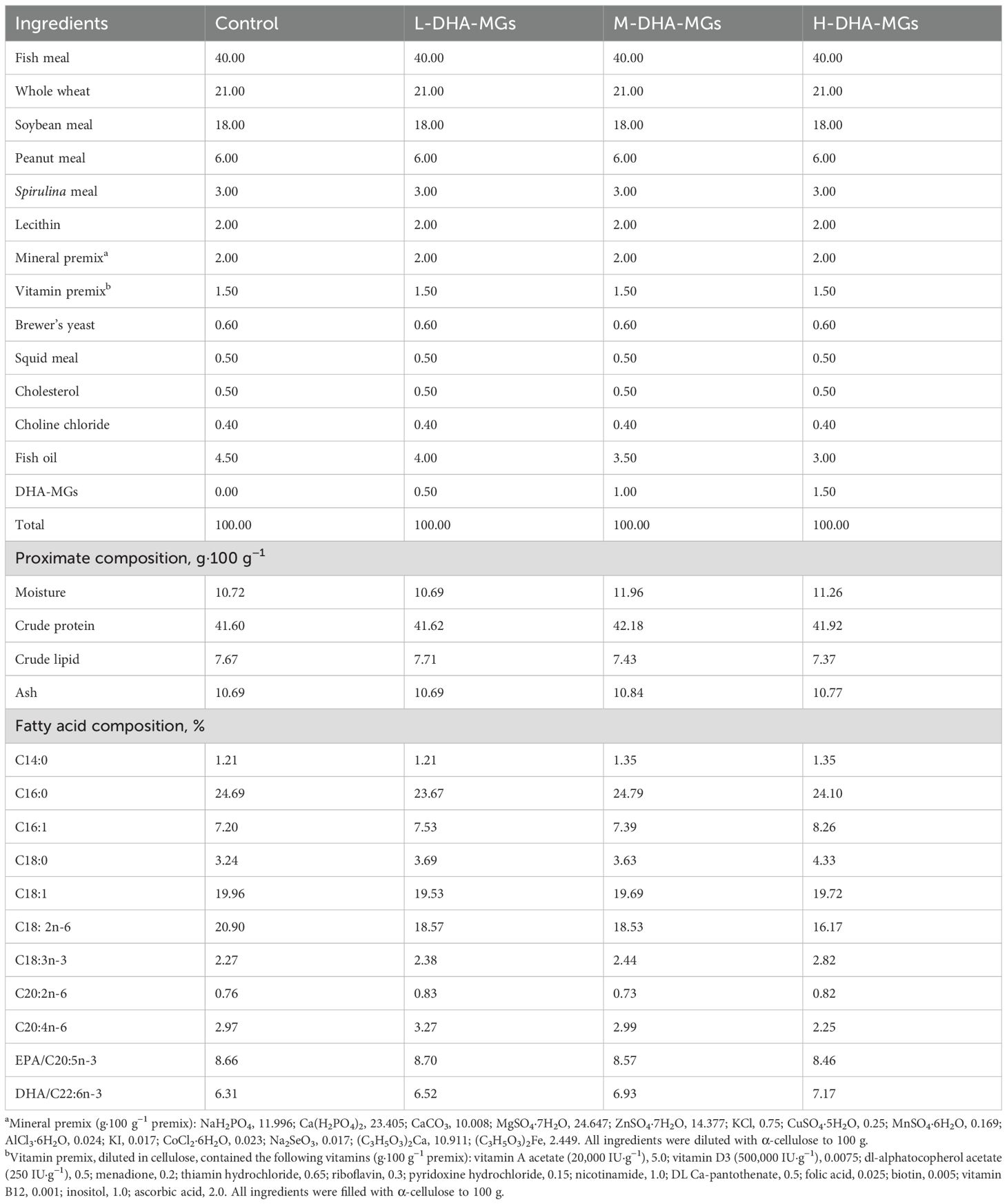
Four isoproteic and isolipidic diets were formulated to contain approximately 41% crude protein and 7.5% crude lipid. The control diet contained only fish oil, while the three experimental diets were supplemented with DHA-MGs to partially replace the fish oil. The abbreviated names of these three experimental diets were L-DHA-MGs (low level of DHA-MGs, 0.5 g·100 g−1), M-DHA-MGs (medium level of DHA-MGs, 1.0 g·100 g−1), and H-DHA-MGs (high level of DHA-MGs, 1.5 g·100 g−1), respectively. The prepared diets were crushed into fine powders through a 120-mesh sieve. All dry ingredients were finely ground and thoroughly mixed before adding different premixed oils. The mixture was dissolved by adding deionized water (200 mL·kg−1 diet) and then wet-extruded into 2.0-mm-diameter pellets using a twin-screw extruder (F-26, SCUT industrial factory, Guangdong, China). The scattered pellets were air-dried at room temperature to approximately 10% moisture and stored at –20°C until use. The ingredients and proximate compositions of the experimental diets are detailed in Table 1.
2.2 Experimental shrimp and feeding trial
All experiments received approval from the ethical committee of the Institute of Hydrobiology, Fujian Academy of Agricultural Sciences. The experimental protocols and methods used in this study were performed in accordance with the relevant guidelines and regulations. A total of 500 shrimp were acquired from Fujian Tonghui Aquatic Technology Co., Ltd. (Fuzhou, China) and transferred to a 1,000-L tank in the Marine Biology Research Laboratory (Fujian Academy of Agricultural Sciences). Following a 2-week acclimatization period, 360 healthy L. vannamei (mean initial weight: 5.46 ± 0.30 g) were randomly distributed across 12 tanks (30 shrimp/tank; 300 L seawater, 26 ppt salinity) and assigned equally to four experimental groups (three tanks/group). Throughout the entire cultivation period of 30 days, the shrimp were fed twice daily at 9:00 and 17:00 with 1% of their body weight. Meantime, the daily exchange rate of incoming water was 50% of the water volume. The seawater for all shrimp was controlled at a temperature of 24.0°C ± 2.0°C and a pH of 7.8–8.0.
2.3 Sample collection
At the end of the feeding trials, the shrimp were fasted for 24 h before sampling. Then, all shrimp in each tank were counted and weighed. A total of 12 shrimp from each tank were randomly collected and stored at –20°C before analyses of the proximate composition and fatty acid profile. Three shrimp per tank were dissected to collect hepatopancreas, muscle, and intestinal tissues. The hepatopancreas and muscle samples from each individual were flash-frozen in liquid nitrogen and stored at -80°C for subsequent lipidomic analysis. Intestinal tissue from one randomly selected shrimp per tank underwent DNA extraction using the DNeasy PowerSoil Pro Kit (Qiagen, Hilden, Germany), with extracts stored at -80°C prior to intestinal microbiota community composition analysis.
2.4 Biochemical composition analysis
The moisture, crude protein, crude lipid, ash, and fatty acid composition was respectively determined according to the method of the National Standard of the People’s Republic of China (GB 5009.3-2016, GB 5009.4-2016, GB 5009.5-2016, GB 5009.6-2016, and GB 5009.168-2016). The moisture content was determined by drying the samples to a constant weight at 101°C–105°C. The crude protein contents (N × 6.25) were assayed by using the Kjeldahl method with an automatic Kjeldahl nitrogen analyzer (Kjeltec 8400, FOSS, Hillerød, Denmark). The crude lipid extraction method using a Soxtec System (Soxtec 2055, FOSS, Hillerød, Denmark) was employed to determine the crude lipid content. The ash content of the sample was determined after incineration in a muffle furnace at 550°C ± 25°C for 4 h. The fatty acid profiles were determined by gas chromatography (Agilent GC 7890A, Agilent Technologies, Palo Alto, CA, USA) with flame ionization detection.
2.5 Lipidomics analysis by liquid chromatography/tandem mass spectrometry
The muscle or hepatopancreas (50 mg) in each individual was used to extract total lipid with 280 μL extraction solution (methanol/water = 2:5) and 400 µL MTBE (methyl tert-butyl ether). After centrifugation (13,000 g, 15 min), 350 µL of the supernatant was transferred to a 2.0-mL polypropylene tube and evaporated under a nitrogen stream at 35°C. Subsequently, 100 µL of extraction solution (isopropanol/acetonitrile = 1:1) was used to dissolve the extracted lipids. The dissolved solution was centrifuged at 13,000 g for 10 min, and then the supernatant was subsequently used for lipidomic analysis by a liquid chromatography/tandem mass spectrometry (LC–MS) (Thermo Fisher, UHPLC-Q Exactive HF-X) equipped with an Accucore C30 reversed-phase column (100 mm × 2.1 mm, 2.6 µm, Thermo Fisher, Waltham, MA, USA). The column was maintained at 40°C. The mobile phase was comprised of component A (acetonitrile:water = 50: 50, v/v) containing 0.1% formic acid and 10 mM ammonium acetate and component B (acetonitrile/isopropanol/water = 10: 88: 2, v/v/v) containing 0.02% formic acid and 2 mmol/L ammonium acetate. The mobile phase was pumped with a flow rate of 0.4 mL/min. The injection sample volume was 2 μL, and the temperature of the automatic injector was set at 4°C. The samples were ionized by electrospray ionization (ESI) in negative and positive modes. MS analysis was carried out using the following parameters: ion spray voltage floating (ESI+), + 3,000 V; ion spray voltage floating (ESI–), –3,000 V; sheath gas flow rate, 60 psi; aux gas flow rate, 20 psi; aux gas heater temperature, 370°C; normalized collision energy: 20, 40, and 60 V. The MS spectra were acquired in the m/z range of 200–2,000. Quality control (QC) samples are prepared by mixing equal volumes of extracts from all samples. A QC sample was analyzed after every 10 samples to evaluate the stability of LC–MS during the whole acquisition.
The original data of LC–MS were obtained and pre-processed by the lipid search software (version 4.0.20, Thermo Fisher, Waltham, MA, USA); then, a two-dimensional data matrix containing lipid, lipidIon, category, class, fatty acid chain (fatty acid, FA1, FA2, FA3), calcMz, ionFormula, retention time (RT), and peak intensity was obtained. The relative content of the specified lipid class was calculated in Microsoft Excel 2016 software (Microsoft Corporation, Redmond, WA, USA) by dividing the lipid species peak area by the sum of the peak area of all lipid species detected within the class.
2.6 16s rRNA gene sequencing for intestinal microbiota analysis
At the end of the experiments, the midgut of shrimp was collected to detect the intestinal microbiota by Illumina deep sequencing. For Illumina deep sequencing, the V3–V4 regions of 16s rRNA gene were amplified by the reported PCR method using the primers 341F (5′- CCT ACG GGN GGC WGC AG -3′) and 805R (5′- GAC TAC HVG GGT ATC TAA TCC -3′) with the Illumina adaptor (Illumina, San Diego, CA, USA) sequence and index sequence (Lin et al., 2022). The PCR products were purified by QIAquick PCR Purification kit (Qiagen, Hilden, NRW, Germany), quantified by Qubit 4.0 (Invitrogen, Carlsbad, CA, USA), and then sequenced on an Illumina 300PE MiSeq platform (Sangon Biotech Co., Ltd, Shanghai, China).
After sequencing, the adaptors, barcodes, and primers were firstly removed by cutadapt (version 1.18) and then merged using PEAR (version 0.9.8). Low-quality reads (Q score <20) were removed according to the fqtrim (version 0.94). Chimeras were removed using Usearch (version 11.0.667), and the remaining sequences were clustered into operational taxonomic units (OTUs) at the 3% dissimilarity level. The taxonomies of the bacterial sequences were annotated using Ribosomal Database Project (RDP) classifier (version 2.12) with a confidence threshold of 80%. The α-diversity statistics, including rarefaction curves at a distance of 3%, Good’s coverage, Chao1, the abundance-based coverage estimator (ACE), Shannon, and Simpson were calculated in Mothur (version 1.43.0) for each biomass sample. The β-diversity was analyzed to investigate the differences within and between groups by the principal component analysis (PCA) and principal coordinates analysis (PCoA) based on weighted UniFrac. The weighted UniFrac phylogenetic distance was calculated using QIIME 2 (version 2024). Microbiome profiling was conducted using QIIME 2. Microbial phenotypes were computationally inferred using BugBase, which predicts organism-level traits (e.g., Gram staining, oxygen utilization) from 16S rRNA gene-based ASV tables generated via QIIME 2 with default normalization parameters. PCA, PCoA, Venn diagrams, and bar plots were generated in R (version 3.6.0) by utilizing the vegan (version 2.5–6) package.
2.7 Statistical analysis
Statistical analysis was conducted by utilizing SPSS version 27.0 (SPSS, IBM, Armonk, NY, USA). Continuous variables were expressed as mean ± standard deviation (SD) following verification of normality using the Shapiro–Wilk test. For variables with normal distribution, group differences were analyzed using one-way analysis of variance (ANOVA) followed by Tukey’s HSD post hoc test for multiple comparisons. For variables not conforming to a normal distribution, group comparisons were conducted using the Kruskal–Wallis test, with Dunn’s post hoc test for pairwise comparisons when the statistical significance was indicated. Differences were considered statistically significant at p <0.05.
3 Results
3.1 Growth performance
Following a 30-day treatment period, the growth indices of the shrimp across various diets were assessed, with the results presented in Table 2. According to Table 2, the final weight of shrimp treated with H-DHA-MGs was substantially greater than that of the other groups. Meanwhile, the weight gain rate (WGR) and specific growth rate (SGR) of shrimp consuming the H-DHA-MG diet were significantly higher than those in the control and L-DHA-MGs groups. No significant differences in the hepatopancreas indexes (HI) were observed among the four groups, as given in Table 2. Similarly, there were no statistical differences in the survival rates for the four groups (Table 2).
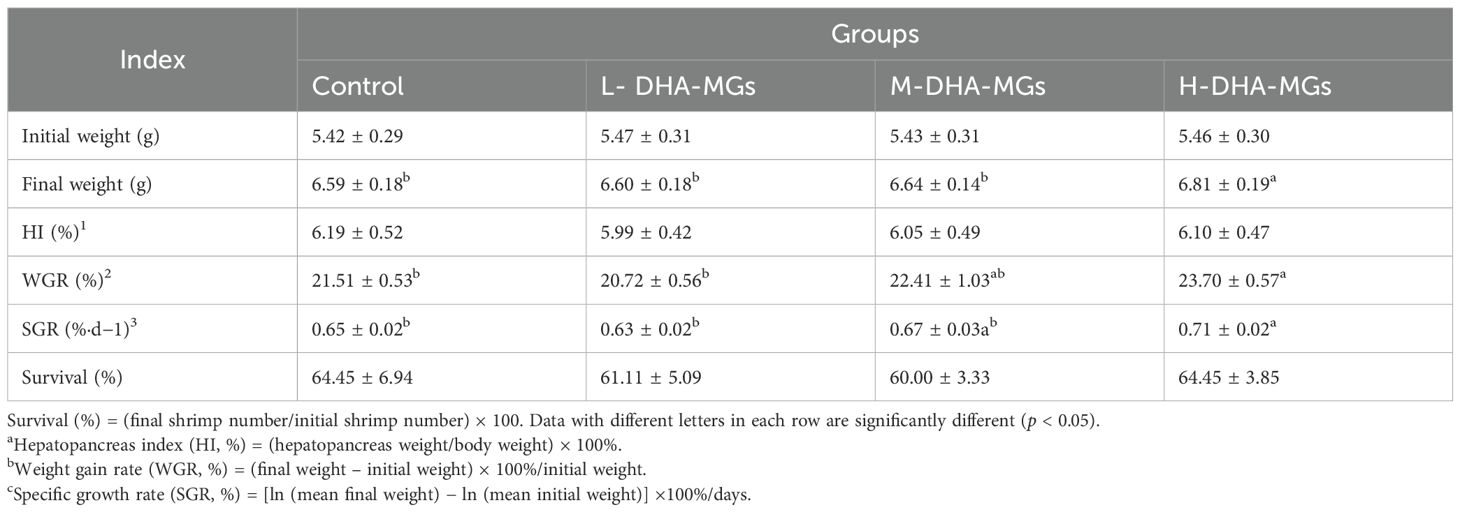
Table 2. Effect of DHA-MGs supplement on the growth performance and survival rate of shrimp fed with experimental diets.
3.2 Biochemical compositions and fatty acid content of shrimp by four diets
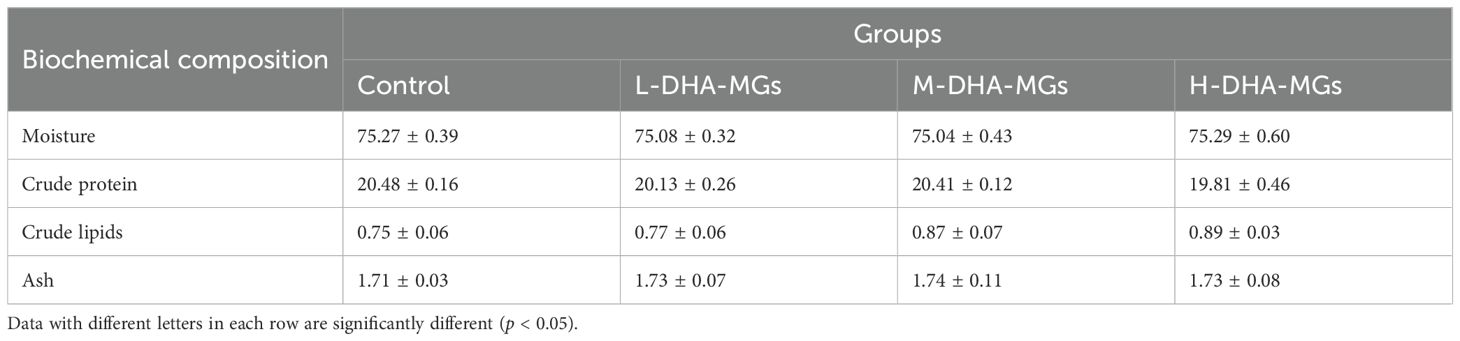
Table 3 presents the biochemical compositions of shrimp muscle for the treated groups. The analysis indicated no significant differences in moisture, crude protein, crude lipid, and ash contents among the shrimp muscles from the four experimental groups as detailed in Table 3. It was noted that the crude lipid of shrimp muscle increased progressively from 0.75% to 0.89% with the increase in DHA-MG addition (Table 3).
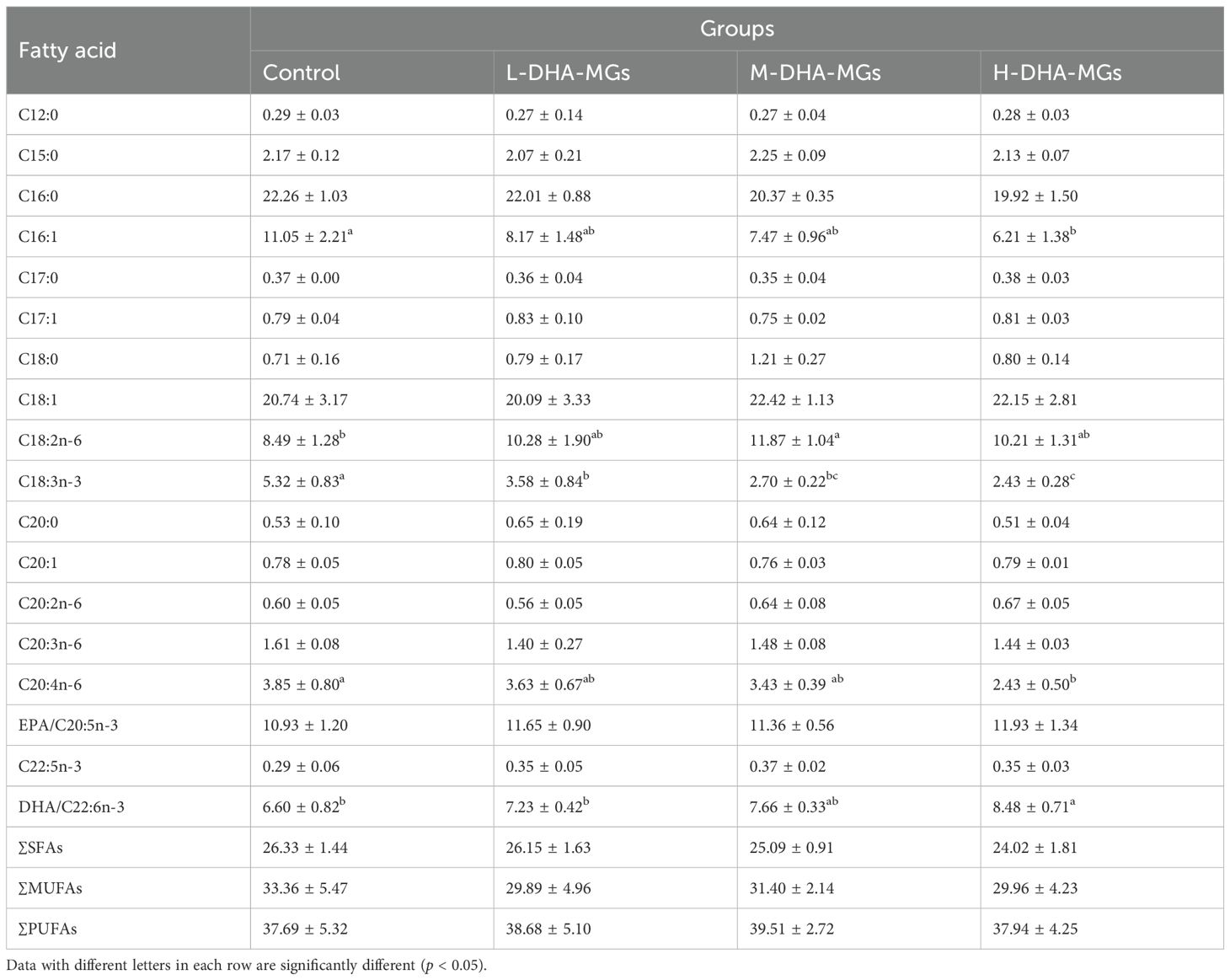
The composition of fatty acids in the muscles of shrimp fed various diets is presented in Table 4. The muscle of shrimp nourished by four distinct diets exhibited significant variations in the concentrations of certain fatty acid species—for instance, the augmentation of DHA-MG addition facilitated the accumulation of DHA in shrimp muscle. The contents of C16:1, C18:3n-3, and C20:4n-6 for shrimp muscle significantly decreased instead with the increased addition of DHA-MGs in the shrimp feeds. The control group shrimp muscles showed the lowest DHA content (6.6%), while the H-DHA-MGs group had the highest (8.48%) (Table 4). This confirms that high DHA-MG levels promote DHA deposition.
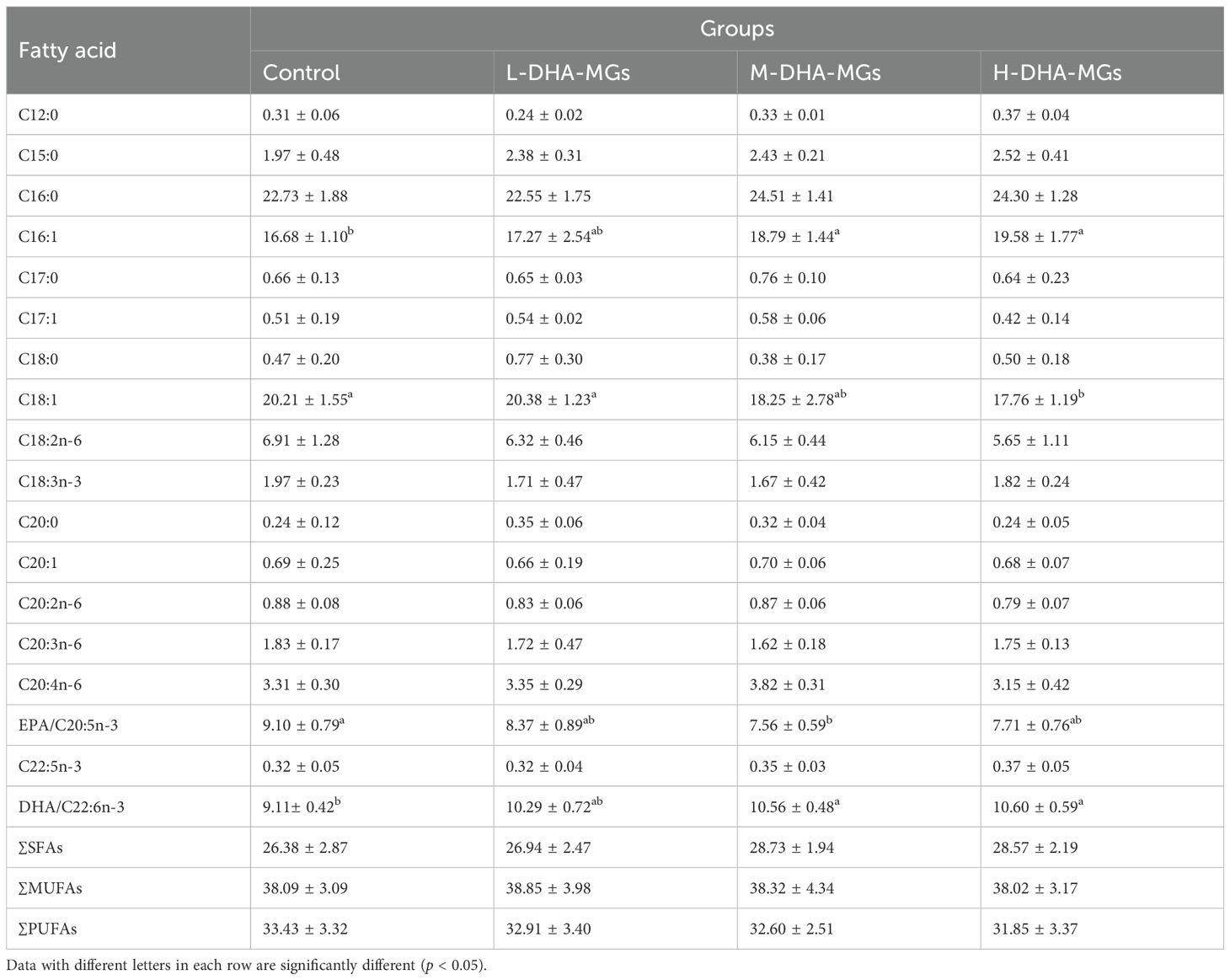
Furthermore, Table 5 presents the fatty acid composition of shrimp hepatopancreas for the four groups. The EPA concentration in shrimp hepatopancreas diminished from 9.10% to around 7.5%, concurrent with an increase in DHA-MGs; simultaneously, the C18:1 and C18:2n-6 contents were also decreased, as shown in Table 5. In a different way, the DHA content of shrimp hepatopancreas remarkably increased from 9.11% to 10.60% with the increase in DHA-MG addition (Table 5). A similar trend was observed in the C16:1 content of shrimp hepatopancreas (Table 5). The results indicated that the addition of DHA-MGs did not facilitate the accumulation of EPA, C18:1, and C18:2n-6 in the hepatopancreas but rather enhanced the deposition of DHA.
3.3 Lipidomics of shrimp tissues by four diets
Lipid profiling revealed seven categories and 53 classes in shrimp muscle and hepatopancreas tissues (Table 6). In terms of the wax lipids (WLs), among the recorded lipid classes, no significant differences were observed in the acyl-(gamma-hydroxy) fatty acid (OAHFA) contents of shrimp muscles or the wax ester (WE) contents of shrimp hepatopancreas for the four diets. It was found that the lowest WE content was obtained by shrimp muscles treated with L-DHA-MGs. The OAHFAs content of shrimp hepatopancreas was distinctly decreased from 0.82% to 0.78% with the increase of DHA-MG addition.
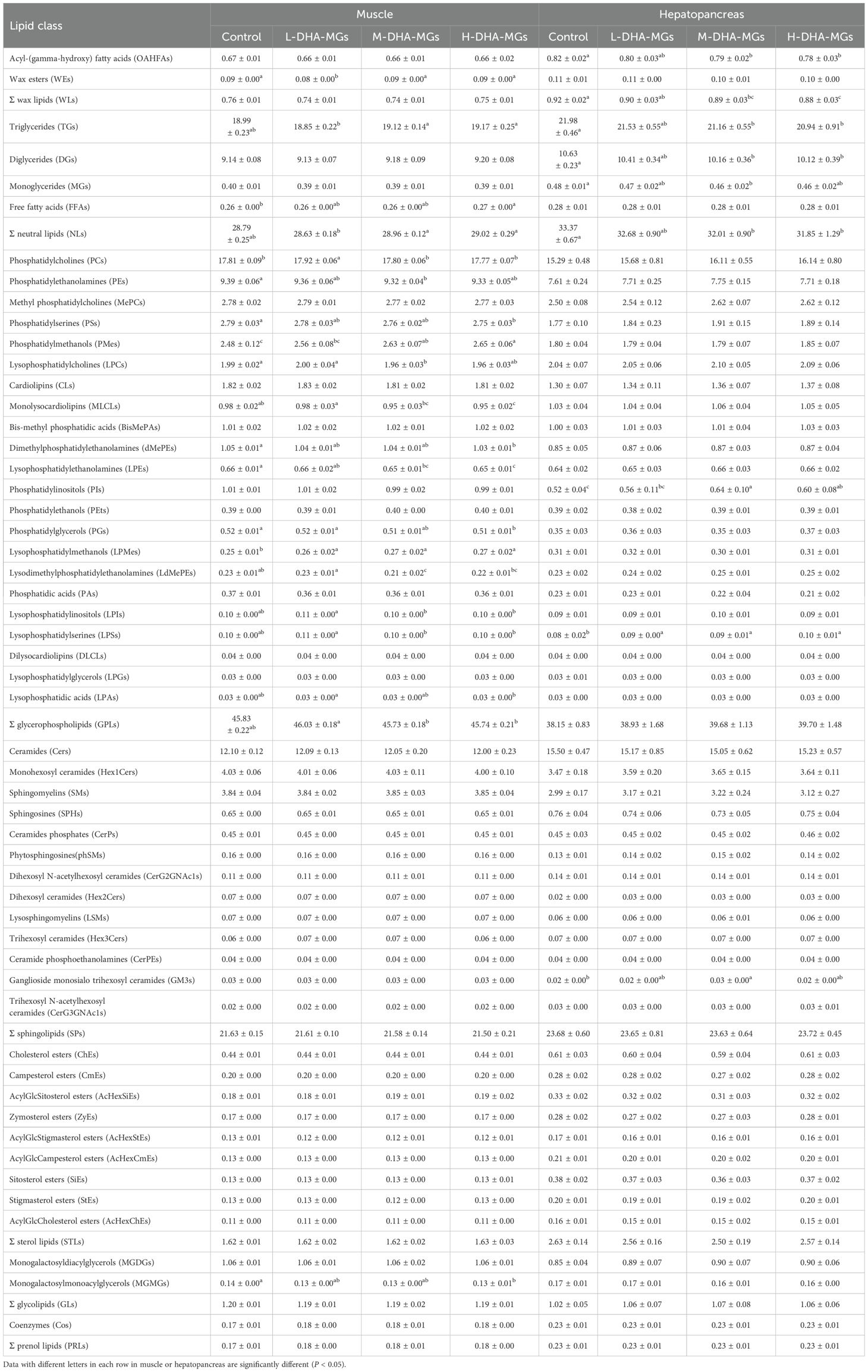
Table 6. Lipid composition (100%) in the muscle and hepatopancreas of four shrimp groups based on lipidomics analysis.
With respect to neutral lipid biosynthesis, the triglyceride (TG) content in shrimp muscle from the M-DHA-MGs and H-DHA-MGs groups was significantly greater than that of the control and L-DHA-MGs groups; concurrently, the highest levels of free fatty acids (FFAs) and total neutral lipids (NLs) in the muscles were observed in shrimp fed the DHA-MG diet. No significant differences in the muscle-derived DGs or MGs content were observed for the four trials. The elevation of DHA-MGs in the shrimp diet progressively reduced the hepatopancreas’ TG concentration as illustrated in Table 6. Comparable changes regarding the contents of DGs and MGs in shrimp hepatopancreas were seen with an increased addition of DHA-MGs. The FFA contents of shrimp hepatopancreas showed no statistical differences among the four groups. The shrimp hepatopancreas exhibited the highest NL content in the control group and the lowest NL content in the H-DHA-MGs group.
Table 6 also states the changes of GPLs and its every class affected by DHA-MGs level. Shrimp muscle for the L-DHA-MG diet gave the highest contents of phosphatidylcholines (PCs, 17.92%), lysophosphatidylcholines (LPCs, 2.00%), lysophosphatidylinositols (LPIs, 0.11%), and lysophosphatidylserines (LPSs, 0.11%) that were significantly higher than those of the resting trials. The lowest phosphatidylethanolamine (PE) and lysodimethylphosphatidylethanolamine (LdMePE) contents of muscle were observed in the M-DHA-MGs group. It was found that the increase in DHA-MG addition led to a significant increment in phosphatidylmethanols (PMes) and lysophosphatidylmethanols (LPMes) contents of shrimp muscle (Table 6). Nevertheless, the distinct decrease in the contents of muscle phosphatidylserines (PSs), monolysocardiolipins (MLCLs), dimethylphosphatidylethanolamines (dMePEs), and phosphatidylglycerols (PGs) was observed as increased in DHA-MG addition. Besides that, no significant differences were found in the resting 10 species of muscle GPLs (e.g., methyl phosphatidylcholines (MePCs), cardiolipins (CLs)) among the four groups. As for shrimp hepatopancreas, the highest phosphatidylinositol (PIs) content was achieved in the M-DHA-MGs group, which was remarkably greater than that of the control group. Additionally, the LPS contents of shrimp hepatopancreas for the control group were significantly lower than those of the DHA-MGs groups. The remaining GPL species of shrimp hepatopancreas exhibited no statistical difference among the four trials.
The sphingolipids (SPs) category was recorded to have 13 classes (e.g., ceramides (Cers), monohexosyl ceramides (Hex1Cers)) in the shrimp muscle and hepatopancreas. The total SP contents of shrimp tissues were around 21.5%–23.72% of crude lipids. The crude lipids of shrimp hepatopancreas by the M-DHA-MG diet had the highest ganglioside monosialo trihexosyl ceramide (GM3) content that was statistically higher than that of the control group. No significant variations in the Cers contents of shrimp muscle and hepatopancreas were observed for the four experiments. Similar phenomena were found in the remaining SP-derived classes of shrimp tissues.
In addition, the obtained lipids of shrimp muscle and hepatopancreas had sterol lipid (STL), glycolipid (GL), and prenol lipid (PRL) categories. It was found that the lowest monogalactosylmonoacylglycerol (MGMG) content of shrimp muscle was determined in the H-DHA-MG diet, which was distinctly lower than that of the control group. Meanwhile, there were no significant differences in the total STL content and every lipid species content of shrimp tissues across the four diets. Similar phenomena were presented in the contents of lipid classes of GL and PRL categories for shrimp tissues (except for muscle MGMGs).
3.4 DHA distribution of lipid species for four diets
The DHA-MG supplement had a positive impact on the DHA accumulation (Tables 4, 5) and lipid specie levels (Table 6) in shrimp tissues. The LC–MS-based lipidomics approach was used to detect the lipid classes containing DHA, with the findings presented in Figure 1. Among the lipid classes including DHA from shrimp muscle, significant differences in the contents of 13 lipid classes containing DHA for the four diets were found in Figure 1A. In these 13 species, TGs and DGs were from the NL category, and the resting species were from the GPL category. The content of muscle DHA-TG species increased significantly with increasing DHA-MG supplementation. Similar trends were observed in BisMePAs, DGs, PEts, PGs, and PMes with DHA, as shown in Figure 1A. The increment in DHA-MG supplement remarkably lowered instead the DHA-LdMePE content of shrimp muscle (Figure 1A). Similarly, the contents of LPA, LPC, LPS, and MLCL with DHA were distinctly decreased. The highest contents of muscle LdMePE, LPC, LPS, MLCL, and PC species with DHA were shown in the L-DHA-MG diet. In terms of the lipid class with DHA in shrimp hepatopancreas, only four lipid species from the PL category including BisMePAs, LPSs, PAs, and PCs with DHA stated significant differences among the treated trials (Figure 1B). The shrimp hepatopancreas from the H-DHA-MG diet attained the highest contents of BisMePA, LPS, and PC with DHA that were remarkably greater than those of the control group (Figure 1B). However, the lowest content of PA species with DHA was recorded in shrimp hepatopancreas treated with the H-DHA-MG diet.
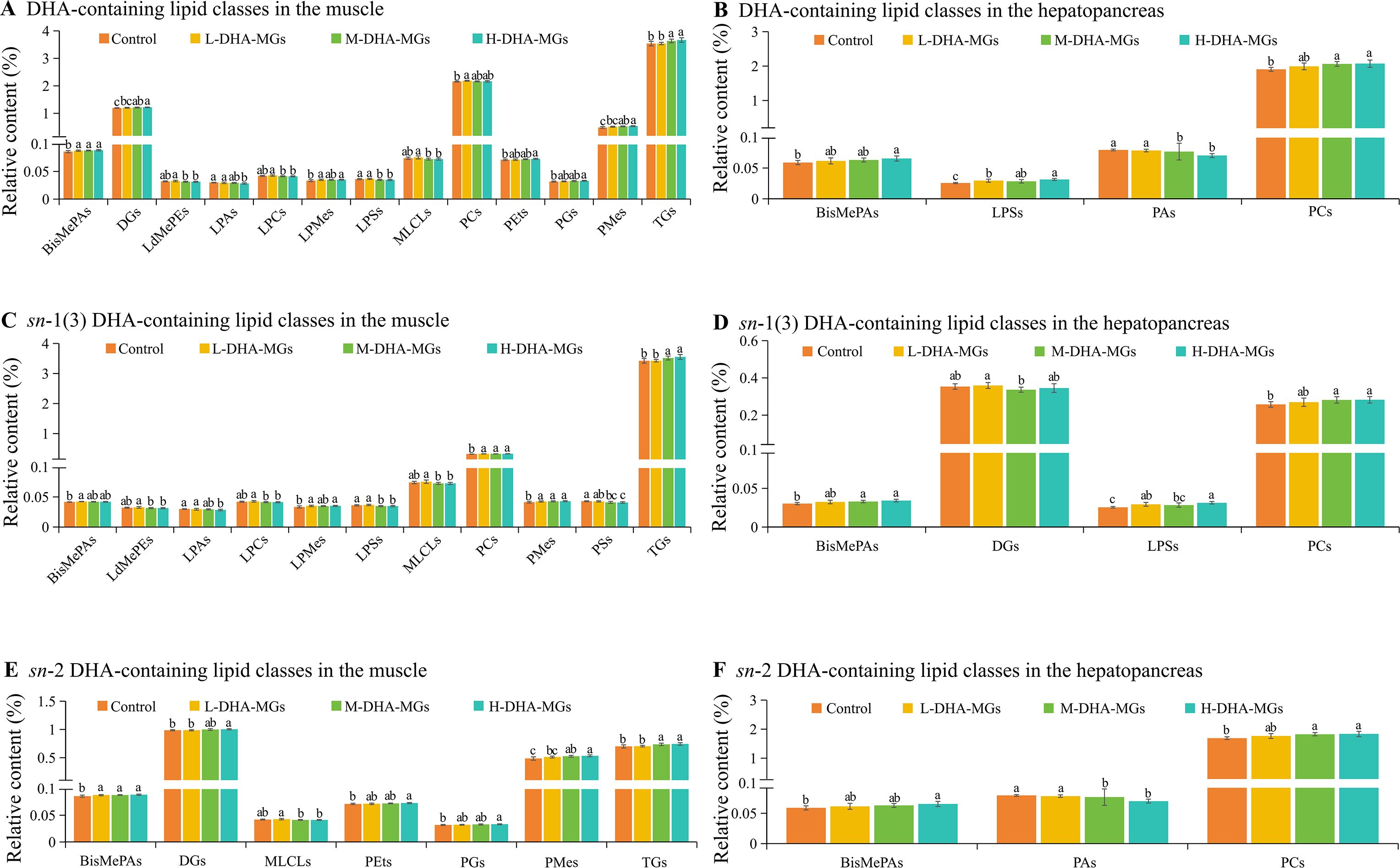
Figure 1. Significant differences in lipid classes with DHA and DHA distribution for muscle and hepatopancreas of four shrimp groups. (A) DHA-containing lipid classes in the muscle, (B) DHA-containing lipid classes in the hepatopancreas, (C) sn-1(3) DHA-containing lipid classes in the muscle, (D) sn-1(3) DHA-containing lipid classes in the hepatopancreas, (E) sn-2 DHA-containing lipid classes in the muscle, and (F) sn-2 DHA-containing lipid classes in the hepatopancreas. The bars with different letters indicate significant difference (p < 0.05).
Moreover, the lipid classes with DHA distributed at the sn-1(3) and 2 positions were determined, and the results are given in Figures 1C–F. It was clear that the crude lipids of shrimp muscle had around 3.5% TGs with DHA distributed at the sn-1(3) position. The data was greater than the values of the remaining lipid species with DHA, as shown in Figure 1C. Interestingly, the H-DHA-MG diet distinctly promoted to DHA accumulation at the sn-1(3) position of TGs in shrimp muscle. Shrimp muscle LPMEs, PCs, and PMes appeared with similar features as expressed in Figure 1C. Nevertheless, the high level of DHA-MGs had negative impacts on the DHA distribution at the sn-1(3) position of muscle LdMePE, LPA, LPC, LPS, MLCL, and PS species. The L-DHA-MG diet tended to deposit DHA at the sn-1 position of muscle BisMePAs (Figure 1C). As for the hepatopancreas, the contents of BisMePAs, LPSs, and PCs with sn-1 DHA were gradually increased with the increment of DHA-MG (Figure 1D). The lowest content of DGs with sn-1 DHA was obtained in the M-DHA-MG diet (Figure 1D).
Furthermore, there were seven muscle-derived lipid species (e.g., BisMePAs, DGs) with sn-2 DHA showing significant differences among the four groups. It was found that the shrimp muscle treated with the H-DHA-MG diet had the highest BisMePA, DG, PEt, PG, PMe, and TG contents that were significantly higher than the ones by the control group (Figure 1E). Besides that, the M-DHA-MG diet was beneficial to accumulating DHA at the sn-2 position of MLCLs. With respect to the sn-2 DHA of lipid species from shrimp hepatopancreas, the contents of BisMePA and PC species with sn-2 DHA were remarkably increased with the increment of DHA-MG; however, the content of PA with sn-2 DHA was reduced accordingly as the DHA-MG addition increased (Figure 1F).
3.5 Intestinal microbiota characteristics of shrimp midgut
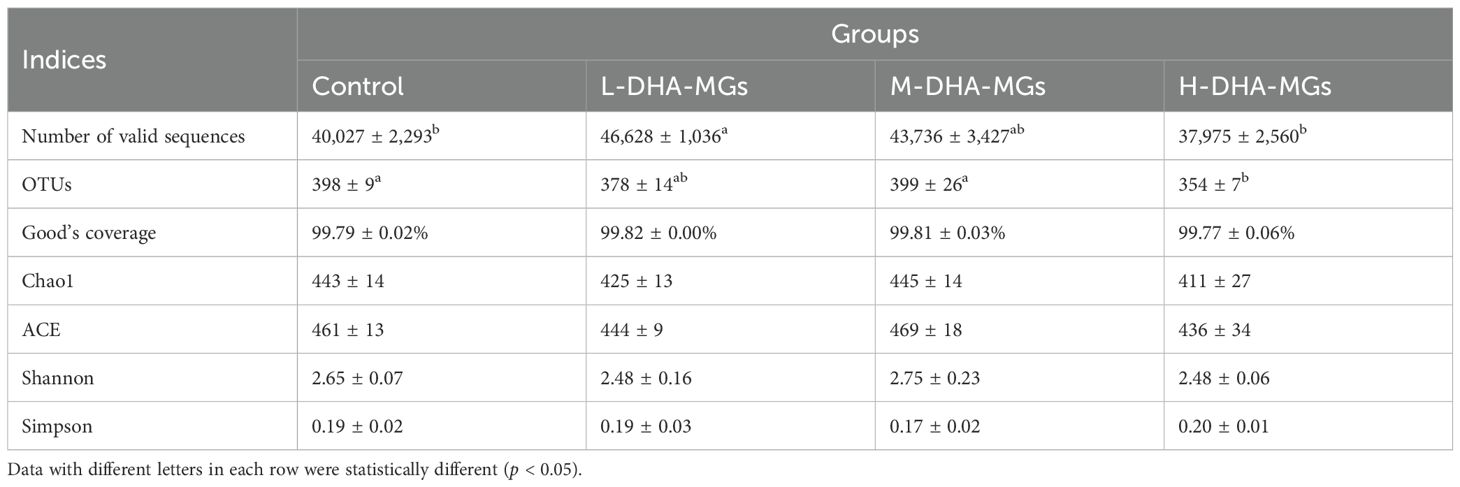
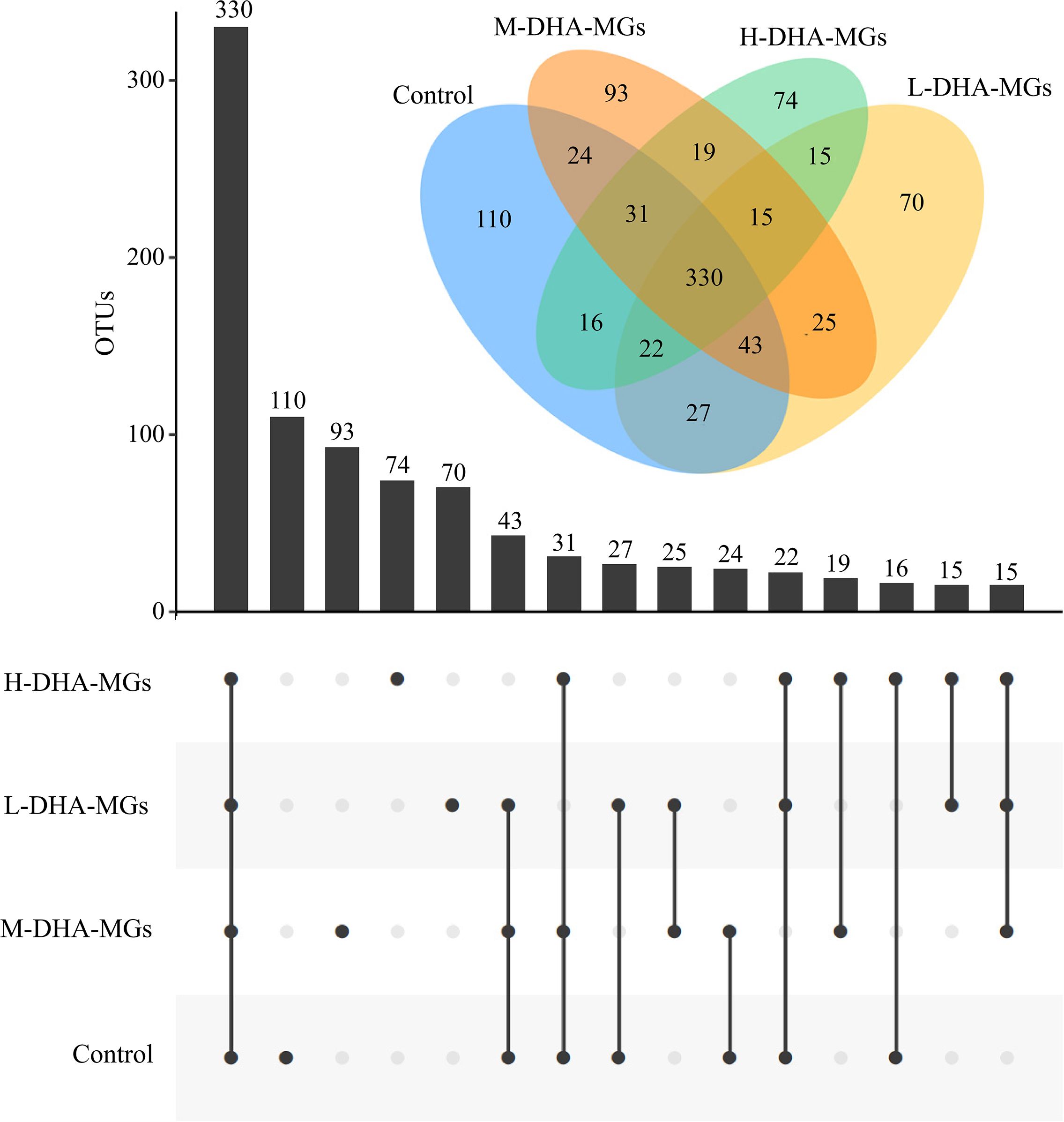
A total of 505,099 valid reads (ranging from 37,216 to 47,669 per sample) were obtained for the four experiments as recorded in Table 7. In addition, the values of Good’s coverage for all samples were above 99% (Table 7), indicating an adequate depth of sequencing. Additionally, the rarefaction curves for the samples from four trials were plateaued in Supplementary Figure S2, revealing that the applied Illumina sequencing technique could record most of the OTUs data for each sample. At 97% sequence identity, the total number of OTUs in all of the studied compartments was 914 (Figure 2). Additionally, the four groups exhibited a similar core bacterial population of 330 OTUs within their intestinal microbiota (Figure 2). The statistical analysis indicated that the common OTUs of the sequenced reads were 54.73% in the control group, 60.33% in the L-DHA-MG group, 56.90% in the M-DHA-MG group, and 63.22% in the H-DHA-MG group. Furthermore, the numbers of unique OTUs in the intestinal microbiota of the control, L-DHA-MG, M-DHA-MG, and H-DHA-MGs group were 110, 70, 93, and 74 with 18.24%, 12.80%, 16.03%, and 14.18% of the total reads, respectively (Figure 2). According to the microbiota α-diversity analysis, no significant differences were found in the richness (Chao1 and ACE) and diversity (Shannon and Simpson) of the intestinal microbiotas among the four groups (Table 7). Regarding β diversity, both PCA and PCoA analyses for the four trials demonstrated a distinct clustering of samples within each experimental group (Supplementary Figure S3), confirming similar response patterns among replicate samples.
As for the phylum level, the four groups attained similar structure and composition of the intestinal microbiome. Meantime, the recorded intestinal microbiome mainly consisted of Proteobacteria, Bacteroidetes, Verrucomicrobia, and Planctomycetes with 44.31%, 38.92%, 13.67%, and 1.36% of the sequenced reads, respectively (Figure 3A). The phylum Proteobacteria constituted 54.02% of the M-DHA-MG group, a proportion significantly higher than that observed in the resting groups (Supplementary Table S1). Conversely, the shrimp subjected to M-DHA-MGs exhibited the lowest proportion of Bacteroidetes (27.49%) among the sequenced reads. In addition, the added DHA-MGs had no significant effect on the relative abundance of Verrucomicrobia for shrimp midgut. Moreover, at the genus level, Flavobacterium, Celeribacter, Haloferula, unclassified_Rhodobacteraceae, and Hanstruepera were the main microorganisms in the four groups (Figure 3B). The dietary supplementation of DHA-MGs significantly reduced the abundance of Flavobacterium in the shrimp midgut compared to the control group, as given in Supplementary Table S2. Additionally, the proportion of Celeribacter in the control group was significantly lower than that in the other groups (Supplementary Table S2).
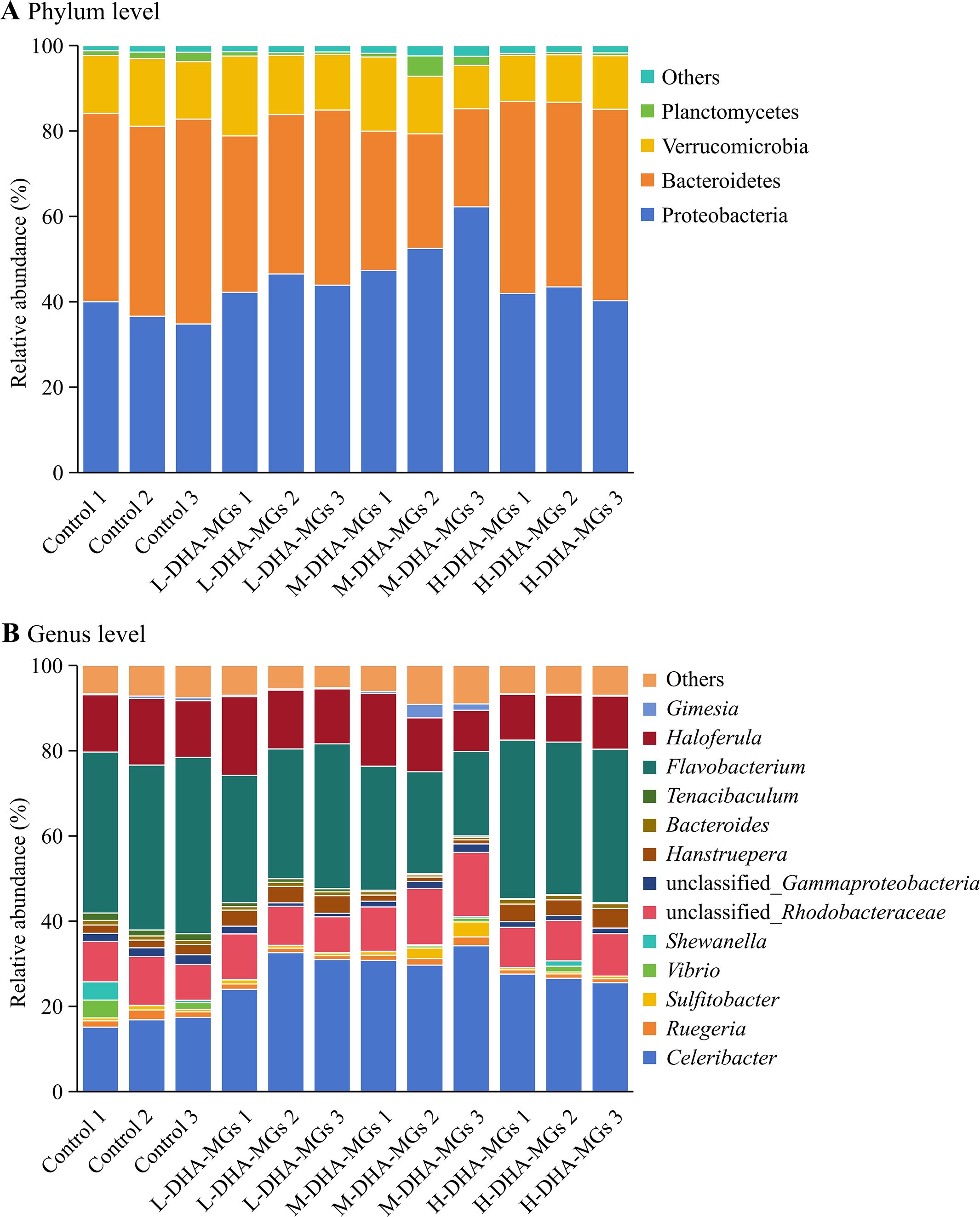
Figure 3. Composition of shrimp midgut microbiota at the phylum (A) and genus (B) levels in the four groups.
Additionally, Figure 4 illustrated the variations in the amounts of nine phenotypic bacteria within the shrimp midgut microbiota among the four diets. The results indicated that the incorporation of DHA-MGs had a negative effect on the aerobic bacteria in the shrimp intestine (Figure 4A). The shrimp intestines exhibited the lowest abundance of anaerobic microorganisms in the M-DHA-MG diet and the highest abundance in the H-DHA-MG diet (Figure 4B). The abundance of facultatively anaerobic microorganisms in the DHA-MG diet was greater than that of the control group (Figure 4C). The shrimp midguts treated with the M-DHA-MG and H-DHA-MG diets achieved a higher abundance of gram-negative microorganisms compared with the control and L-DHA-MG diets (Figure 4D), while greater abundances of midgut gram-positive microorganisms were obtained in the M-DHA-MG and H-DHA-MG diets (Figure 4E). The greater abundances of midgut microorganisms that contain mobile elements (Figure 4F), are oxidative stress tolerant (Figure 4G), and that form biofilms (Figure 4H) were presented in the M-DHA-MG diet in comparison to the other groups. The abundance of midgut potentially pathogenic microorganisms was distinctly decreased when the shrimp were fed diets supplemented with DHA-MGs.
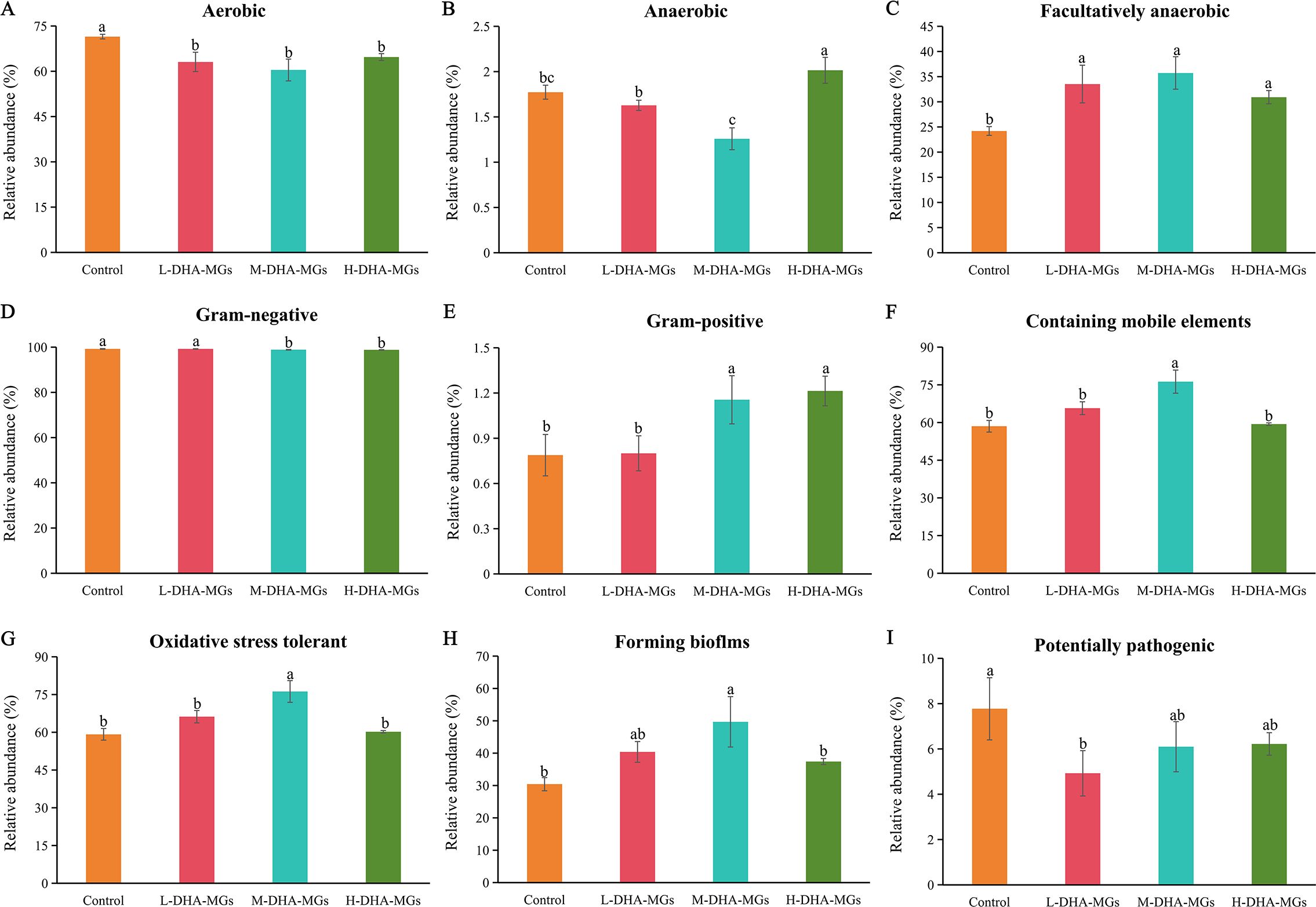
Figure 4. Nine potential phenotype analyses of shrimp midgut microbiota in the four groups. (A) Aerobic, (B) Anaerobic, (C) Facultatively anaerobic, (D) Gram-negative, (E) Gram-positive, (F) Containing mobile elements, (G) Oxidative stress tolerant, (H) Forming biofilms, and (I) Potentially pathogenic. The bars with different letters indicate significantly different (p < 0.05).
4 Discussion
4.1 DHA-MGs promoted the growth performance of L. vannamei
DHA, recognized as an essential component of aquatic feeds, enhances the growth performance of farmed aquatic species (An et al., 2023; Kumar et al., 2022). Recent studies indicate that MGs enriched in DHA may enhance the emulsification of digested DHA-MGs in the intestinal tract, hence facilitating the absorption and accumulation of DHA into the tissues of certain mammalian species (Chevalier and Plourde, 2021; Jin et al., 2020). In this study, shrimp (L. vannamei) fed the H-DHA-MG diet for 30 days achieved the highest final weight (6.81 ± 0.19 g), along with superior WGR and SGR among all dietary groups (Table 2). Nevertheless, no significant differences in HI values and survival rates were observed among the four groups. In agreement with the results of our study, previous reports also demonstrated that supplementation with DHA improved the overall growth performance of L. vannamei juveniles, while survival remained the same (Araújo et al., 2019). Our partial substitution of fish oil with DHA-MGs may have had a limited effect on the growth performance of the shrimp, resulting in no statistical differences in survival rates among the four groups. The enhanced growth performance of L. vannamei fed high levels of DHA-MG may stem from these novel structured lipids. The two hydroxyl radicals on their glycerol backbone confer enhanced hydrophilicity, enabling a more effective absorption of nutrients from the formulated diet and thereby improving growth (Beltrame et al., 2023; Jin et al., 2020). Liu et al. (2024) found that juvenile shrimp fed with MGs could exhibit a significant improvement in WGR, SGR, and FC by promoting lipase activity, augmenting the expression of triglyceride and fatty acid decomposition-related genes, and lowering the levels of plasma triglycerides. Similar results were observed in other aquaculture animals such as zebrafish (Wang et al., 2022), Atlantic salmon (Oxley et al., 2007), and brown trout (Li and Olsen, 2017).
Recent nutritional studies have revealed that an optimal n-3/n-6 PUFA ratio in shrimp feed can improve the growth performance, nutrient utilization, and health of aquatic animals (Dong et al., 2023; Jin et al., 2019). This work indicates that the incorporation of DHA-MGs progressively elevated the n-3/n-6 PUFA ratio in the shrimp diet (Table 1), potentially enhancing the growth performance of L. vannamei. Thus, the results in Table 2 manifested that the high level of DHA-MGs to replace the fish oil was beneficial for shrimp growth.
4.2 DHA-MGs boosted the accumulation of DHA into shrimp tissues
The addition of DHA-MGs did not significantly alter the biochemical composition of shrimp muscle, as shown in Table 3. However, the highest crude lipid content was examined in the H-DHA-MG diet (Table 3). Wang et al. (2020) demonstrated that the shrimp diet supplemented with monolaurate distinctly improved the activities of lipase and protease in the intestine and promoted the accumulation of lipid in the whole body. Similar trends were found in the other aquatic species fed with monoglycerides with short/medium-chain fatty acids (Shin et al., 2023; Tang et al., 2023; Tran et al., 2020) and oleic acid (Li and Olsen, 2017). The high level of DHA-MGs might drive the intestinal tract to secrete some vital digestive enzymes (e.g., lipase) and re-esterify the absorbed DHA-MGs to generate a series of lipids (e.g., NLs, GPLs) into the muscle. Furthermore, DHA-MGs facilitated the accumulation of DHA in shrimp muscle (Table 4) and enhanced the nutritional value of shrimp muscle. The possible reasons were that (1) DHA-MGs, as a good emulsion, could alter the nutritional quality to meet the shrimp requirements (Li and Olsen, 2017; Oxley et al., 2007; Wang et al., 2022), (2) DHA-MGs might enhance the activities of digestive enzymes for shrimp and upregulate some key genes (e.g., dehydrogenase and elongase) to deposit the digested DHA into the muscle (Chen et al., 2024b; Santigosa et al., 2024). Consequently, the findings in Tables 3 and 4 indicate that DHA-MGs may serve as a novel lipid source to partially substitute fish oil for the lipid and DHA accumulation in shrimp.
The hepatopancreas is a crucial organ in shrimp for regulating lipid metabolism (Phonsiri et al., 2024; Vogt, 2019). Previous works have demonstrated that aquatic feeds containing different fatty acid compositions affected the lipid uptake and storage of shrimp hepatopancreas (Figure 5), leading to the distinct disparity in the fatty acid profile of shrimp tissues (Araújo et al., 2019; Zhang et al., 2022). The data presented in Table 5 indicates that the incorporation of DHA-MGs as a substitute for fish oil effectively increased the DHA concentration in shrimp hepatopancreas from 9.11% to 10.6%. The likely explanation is that DHA-MG was preferentially reconstructed to generate the desired lipids (e.g., NLs, GPLs) by the key enzymes of Kennedy pathway and fatty acid metabolism (Destaillats et al., 2018; Hishikawa et al., 2017), and then the lipids containing DHA were deposited into the hepatopancreas.
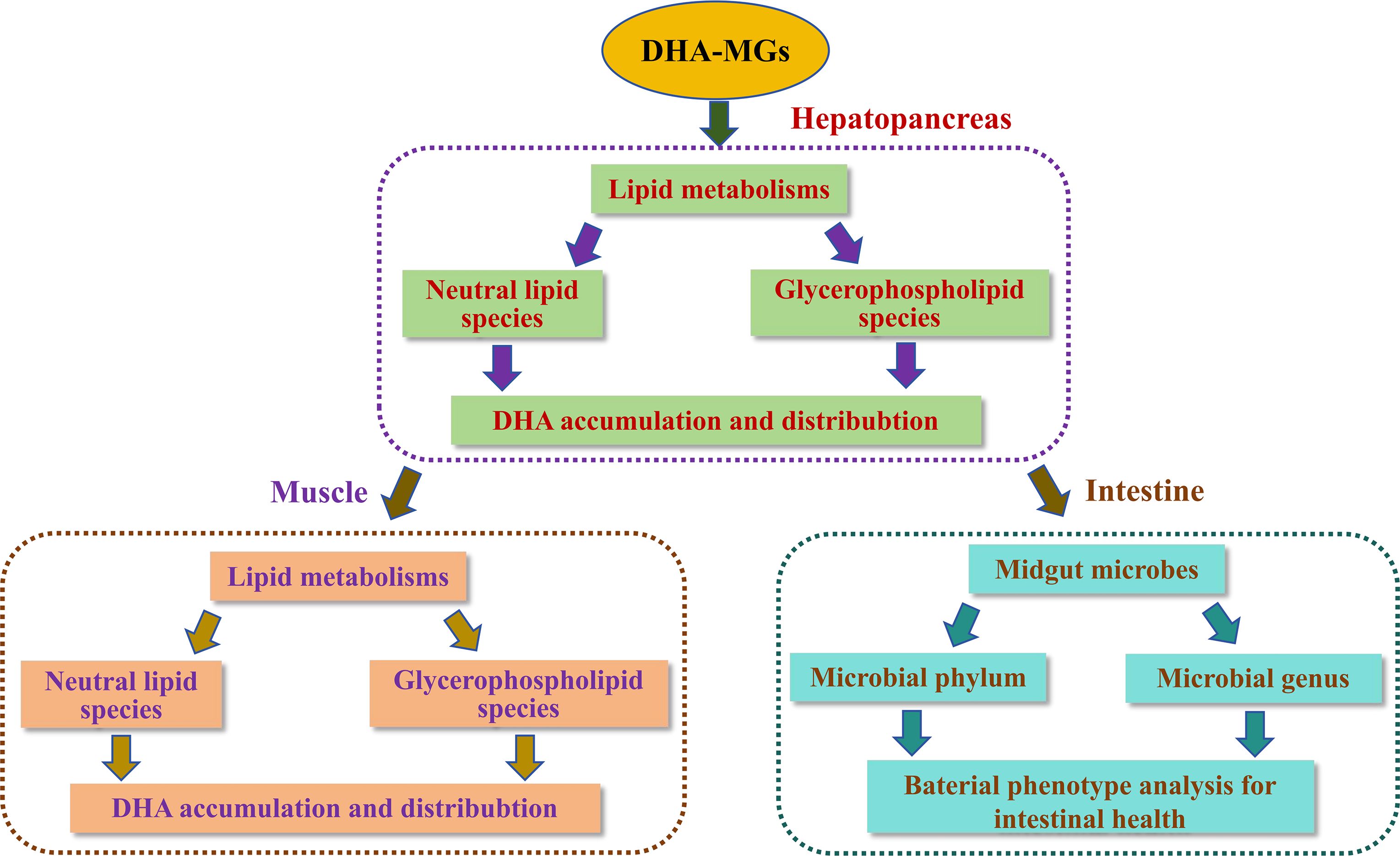
Figure 5. Potential mechanism to regulate the DHA accumulation and distribution of lipid metabolisms for shrimp muscle and hepatopancreas and modify the healthy intestinal microecological balance.
4.3 DHA-MGs modified the level of lipid species and DHA distribution
LC–MS technique, as a useful analytical tool, had been widely employed to detect the lipid classes of shrimp (Sun et al., 2020; Tu et al., 2022; Wang et al., 2024; Yu et al., 2020; Zhu et al., 2022). Sun et al. (2020) chose this analytic technique to examine the lipidomics of shrimp and stated that the shrimp lipids were mainly categorized as NLs (e.g., TGs, DGs), GPLs (e.g., PCs, PEs), SPs (e.g., Cers, Hex1Cers), and so on. In this work, the lipids of the muscle and hepatopancreas of the shrimp treated with the four diets were also classified into these mentioned lipid categories (Table 6). Among the seven lipid categories, GPLs exhibited the highest concentrations in both shrimp muscle and hepatopancreas, a finding which aligns with previous reports (Sun et al., 2020; Tu et al., 2022; Wang et al., 2024; Yu et al., 2020; Zhu et al., 2022). Dietary supplementation with different levels of DHA-monoglycerides (DHA-MGs) significantly influenced the lipid composition across categories (Table 6)—for instance, the highest TG, PC, and PE contents in shrimp muscle were observed in the H-DHA-MG, L-DHA-MG, and control groups, respectively. These findings suggest that partial fish oil replacement with DHA-MGs may modulate key enzymatic activities (e.g., diglyceride acyltransferase (DGAT), lysophosphatidic acid acyltransferase (LPAAT)) in lipid metabolism (Burri et al., 2012; Körbes et al., 2016; Xiang et al., 2024), consequently altering specific lipid species profiles (Figure 5). Furthermore, the shrimp muscle and hepatopancreas showed differences in the content of designated lipid species (Table 6)—for example, increasing the dietary DHA-MG levels resulted in the elevation of the TG content in muscle tissue with a reduction in hepatopancreatic TG accumulation. The M-DHA-MG group showed the lowest PI concentration in the hepatopancreas; nevertheless, no significant changes in muscle PI content were noted among the four diets. The possible explanation is that particular shrimp tissues may contain intrinsic catalytic enzymes that regulate the lipid metabolism levels for the manufacture of specific lipid classes (Figure 5) (Wu et al., 2023).
It was well known that DHA, as a fatty acyl group, was commonly distributed in the FFAs and/or lipid species with glycerol backbone (Li et al., 2025). This work also identified DHA in these lipid species, as illustrated in Figure 1 and Supplementary Figure S1. Notably, DHA-MG supplementation exerted tissue-divergent regulatory effects on the concentrations of lipid classes with DHA among the four experimental groups. In hepatopancreas, DHA-MG supplementation selectively altered four lipidic classes with DHA from the GPL category, while in muscle tissue DHA-MGs predominantly modulated TGs and DGs with DHA (NLs) and 11 lipidic classes with DHA (e.g., PCs, PGs, etc.) from the GPL category. The phenomena can be elucidated by the fact that various tissues of aquatic animals often possess inherent lipid metabolism that selectively re-esterify ingested DHA-MGs to produce lipid classes with DHA (Weil et al., 2013; Xu et al., 2020).
Moreover, the DHA-MG supplement had positive impacts on the DHA distribution of NL and GPL categories with glycerol backbone of shrimp muscle and hepatopancreas, as stated in Figures 1C–F. It was clear that the contents of some lipid classes (e.g., TGs, PMEs) with sn-1(3) and sn-2 DHA for shrimp tissues were increased with increasing DHA-MG. Nevertheless, an opposite trend was noted in several lipid species (e.g., LPAs, LPCs) with sn-1(3) and sn-2 DHA. Pioneering works have demonstrated that the key enzymes (e.g., diglyceride acyltransferase (DGAT), lysophosphatidic acid acyltransferase (LPAAT)) of lipid pathways perform substrate specificity toward DHA for the biosynthesis of particular lipid classes (Klińska-Bąchor et al., 2024; Körbes et al., 2016; Nobusawa et al., 2017; Wayne et al., 2021; Xiang et al., 2024). Thus, the results in Figures 1C–F indicated that the digested DHA-MGs might be reconstructed by the lipase/phospholipase of lipid metabolism to drive the DHA rearrangement toward the sn-1(3) and 2 positions (Figure 5). To understand the potential mechanism, it is necessary to determine the regulatory factors and metabolites by multi-omics technology in the future.
4.4 DHA-MGs adjusted the abundance of midgut microbe
It has been reported that altering the aquatic feed composition played an essential role in the colonization and proliferation of symbiotic microorganisms in the gut (Gao et al., 2023; Luan et al., 2023; Qiao et al., 2017). The gut microbiota of aquatic animals plays critical roles in nutrition, development, immunity, and resistance against invasive pathogens (Holt et al., 2021; Zeng et al., 2024). In the present work, the dominant phyla of midgut microbe in each group were Proteobacteria, Bacteroidetes, Verrucomicrobia, and Planctomycetes, a finding which was consistent with that of previous studies (Fan et al., 2019; Huang et al., 2018; Zheng et al., 2017). Moreover, dietary DHA-MGs influenced the abundances of these phyla in the shrimp midgut. Shrimp subjected to M-DHA-MGs had the greatest abundances of Proteobacteria and Planctomycetes, while this group recorded the lowest abundance of Bacteroidetes. These results indicated that the appropriate intake of DHA-MGs might selectively promote the growth of Proteobacteria and Planctomycetes while suppressing Bacteroidetes proliferation. Moreover, it was interesting to find that the abundance of Verrucomicrobia (13.67%) for all groups was greater than the ones reported by Huang et al. (2018) and Fan et al. (2019), likely due to the differences in the farming environment with the specific microorganisms (Kim et al., 2021). Conversely, the shrimp midgut for H-DHA-MGs exhibited the lowest abundance of Verrucomicrobia, implying that the high DHA level of shrimp feed may be detrimental to the growth of bacteria from Verrucomicrobia. Liu et al. (2024) asserted that the appropriate addition of MGs could maintain the stability of the intestinal microbiota to improve the abundance of beneficial bacteria via upregulating intestinal antimicrobial peptide-related genes and tumor necrosis factor-alpha levels. Some previous works about the intestinal microbiota of shrimp affected by the DHA addition had reported similar results as well (Liang et al., 2022; Zhang et al., 2022, 2014).
On the other hand, dietary DHA-MGs had distinct effects on the abundance of bacteria genus in the shrimp midgut. DHA-MGs significantly reduced the abundance of Flavobacterium and improved the proportion of Celeribacter (Figure 3B), suggesting that DHA-MGs could readjust to establish a new healthy midgut microbiome balance for promoting shrimp growth (Costantini et al., 2017; Yu et al., 2021). Furthermore, the findings of the bacterial phenotype study (Figure 4) indicated that the addition of DHA-MGs significantly influenced the number of microbes with varying biological characteristics (Figure 5). The addition of DHA-MGs was observed to enhance the abundance of gram-positive microbes while concurrently reducing the abundance of aerobic and gram-negative microorganisms. This was the first time to report the phenomenon concerning the abundance of shrimp gram-positive bacteria influenced by the addition of DHA-MGs. Furthermore, the M-DHA-MG diet effectively reduced the abundance of anaerobic microorganisms while enhancing the prevalence of facultatively anaerobic microorganisms and bacteria possessing mobile elements, oxidative stress tolerance, and biofilm-forming capabilities. The increased abundance of these four bacterial phenotypes indicates that moderate dietary supplementation with DHA-MGs can enhance the resilience of shrimp intestinal microbiota to diverse environmental challenges (Fu et al., 2024). Moreover, the addition of DHA-MGs markedly diminished the abundance of potentially pathogenic microorganisms (Figure 4I), indicating that DHA-MGs may serve as a potential bacteriostat to regulate the gut microbiota and inhibit the proliferation of pathogenic bacteria, thereby enhancing growth performance (Fu et al., 2021). To the best of our knowledge, this work was the first to elucidate the details between intestinal microbiota and DHA-MG addition, and the potential mechanism remained unclear. Further research is required to elucidate the interaction between the digestion of DHA-MGs and midgut bacteria in order to produce innovative shrimp feeds incorporating DHA-MGs, thereby demonstrating the mechanisms that influence changes in the microbial community structure of the shrimp midgut.
5 Conclusion
In summary, this work showed that the high level of DHA-MGs from Schizochytrium sp. to partially replace fish oil was conducive to the growth performance and DHA accumulation of L. vannamei. Furthermore, the incorporation of DHA-MGs significantly influenced the levels of particular lipid classes and their distribution of DHA. Additionally, it might favor intestinal health by regulating intestinal microbial community composition. Taken together, Schizochytrium-derived DHA-MGs could be the potential novel candidate to replace fish oil in shrimp diet in view of the growth performance, DHA accumulation and distribution, and intestinal microbiota.
Data availability statement
The sequences obtained in this study had been deposited in NCBI’s Sequence Read Archive (SRA) database under the BioProject accession number PRJNA113647.
Ethics statement
The animal study was approved by The ethics committee of the Institute of Hydrobiology, Fujian Academy of Agricultural Sciences. The study was conducted in accordance with the local legislation and institutional requirements.
Author contributions
WH: Conceptualization, Data curation, Funding acquisition, Investigation, Methodology, Writing – original draft. YS: Conceptualization, Resources, Supervision, Writing – original draft. XW: Conceptualization, Data curation, Investigation, Methodology, Writing – original draft. DL: Conceptualization, Resources, Supervision, Writing – original draft. TL: Data curation, Investigation, Writing – original draft. KC: Data curation, Investigation, Resources, Writing – original draft. QL: Data curation, Investigation, Methodology, Writing – original draft. BT: Data curation, Investigation, Writing – original draft. YZ: Formal Analysis, Funding acquisition, Writing – review & editing. YH: Conceptualization, Data curation, Funding acquisition, Investigation, Project administration, Supervision, Writing – original draft, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the National Natural Science Foundation of Fujian Province (Grant No. 2023J01192), the Special Scientific Research Funds for Public Scientific Research Institution of Fujian (Grant No. 2023R1020002), the Science and Technology Project of Fujian Academy of Agricultural Sciences (Grant No. CXTD2021015-3), the Engineering Research Center of Marine Biopharmaceutical Resource, Fujian Province University, Xiamen Medical College (Grant No. MNPR-2022002), and the Open Bidding for Selecting the Best Candidates Technology Project of Fuzhou Ocean Research Institute (Grant No. 2024F16).
Conflict of interest
Author KC was employed by the company Guangdong Yuehai Feed Group Co., LTD.
The remianing authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmars.2025.1588954/full#supplementary-material
References
An W., Xu J., Chen F., Ma Y., Su Z., Guang J., et al. (2023). Effects of dietary n-3 LC-PUFA levels on the growth, immunity, and lipid metabolism of freshwater carnivorous teleost largemouth bass (Micropterus salmoides) juveniles. Aquacult. Rep. 32, 101704. doi: 10.1016/j.aqrep.2023.101704
Araújo B. C., Mata-Sotres J. A., Viana M. T., Tinajero A., and Braga A. (2019). Fish oil-free diets for Pacific white shrimp Litopenaeus vannamei: the effects of DHA-EPA supplementation on juvenile growth performance and muscle fatty acid profile. Aquaculture 511, 734276. doi: 10.1016/j.aquaculture.2019.734276
Beltrame G., Ahonen E., Damerau A., Gudmundsson H. G., Haraldsson G. G., and Linderborg K. M. (2023). Lipid structure influences the digestion and oxidation behavior of docosahexaenoic and eicosapentaenoic acids in the simulated digestion system. J. Agric. Food Chem. 71, 10087–10096. doi: 10.1021/acs.jafc.3c02207
Burri L., Hoem N., Banni S., and Berge K. (2012). Marine omega-3 phospholipids: metabolism and biological activities. Int. J. Mol. Sci. 13, 15401–15419. doi: 10.3390/ijms131115401
Carlson S. J., Fallon E. M., Kalish B. T., Gura K. M., and Puder M. (2013). The role of the ω-3 fatty acid DHA in the human life cycle. J. Parenter. Enteral Nutr. 37, 15–22. doi: 10.1177/0148607112467821
Carr L., Glencross B., and Santigosa E. (2023). The importance of essential fatty acids and their ratios in aquafeeds to enhance salmonid production, welfare, and human health. Front. Anim. Sci. 4. doi: 10.3389/fanim.2023.1147081
Chen Y., Chi S., Zhang S., Dong X., Yang Q., Liu H., et al. (2021). Replacement of fish meal with Methanotroph (Methylococcus capsulatus, Bath) bacteria meal in the diets of Pacific white shrimp (Litopenaeus vannamei). Aquaculture 541, 736801. doi: 10.1016/j.aquaculture.2021.736801
Chen Z., Kong Y., Huang Z., Zheng X., Zheng Z., Yao D., et al. (2024b). Exogenous alpha-linolenic acid and Vibrio parahaemolyticus induce EPA and DHA levels mediated by delta-6 desaturase to enhance shrimp immunity. Int. J. Biol. Macromol. 257, 128583. doi: 10.1016/j.ijbiomac.2023.128583
Chen C., Tian H., Liu X., Dai Y., Wen X., Zhao H., et al. (2024a). Effects of dietary lipid levels on growth, lipid metabolism, fatty acid composition and antioxidant capacity of juvenile greasyback shrimp (Metapenaeus ensis). Aquacul. Rep. 36, 102146. doi: 10.1016/j.aqrep.2024.102146
Chevalier L. and Plourde M. (2021). Comparison of pharmacokinetics of omega-3 fatty acid supplements in monoacylglycerol or ethyl ester in humans: a randomized controlled trial. Eur. J. Clin. Nutr. 75, 680–688. doi: 10.1038/s41430-020-00767-4
Costantini L., Molinari R., Farinon B., and Merendino N. (2017). Impact of omega-3 fatty acids on the gut microbiota. Int. J. Mol. Sci. 18, 2645. doi: 10.3390/ijms18122645
Destaillats F., Oliveira M., Bastic Schmid V., Masserey-Elmelegy I., Giuffrida F., Thakkar S. K., et al. (2018). Comparison of the incorporation of DHA in circulatory and neural tissue when provided as triacylglycerol (TAG), monoacylglycerol (MAG) or phospholipids (PL) provides new insight into fatty acid bioavailability. Nutrients 10, 620. doi: 10.3390/nu10050620
Dong Y., Wei Y., Wang L., Song K., Zhang C., Lu K., et al. (2023). Dietary n-3/n-6 polyunsaturated fatty acid ratio modulates growth performance in spotted seabass (Lateolabrax maculatus) through regulating lipid metabolism, hepatic antioxidant capacity and intestinal health. Anim. Nutr. 14, 20–31. doi: 10.1016/j.aninu.2023.04.005
Dugassa H. and Gaetan D. G. (2018). Biology of white leg shrimp, Penaeus vannamei: review. World J. Fish Mar. Sci. 10, 5–17. doi: 10.5829/idosi.wjfms.2018.05.17
Fan L., Wang Z., Chen M., Qu Y., Li J., Zhou A., et al. (2019). Microbiota comparison of Pacific white shrimp intestine and sediment at freshwater and marine cultured environment. Sci. Total Environ. 657, 1194–1204. doi: 10.1016/j.scitotenv.2018.12.069
Farris N. W., Willora F. P., Blaauw D. H., Gupta S., Santigosa E., Carr I., et al. (2024). Progressive substitution of fish oil with Schizochytrium-derived algal oil in the diet of Atlantic salmon (Salmo salar) parr subjected to winter signal period. Aquacul. Rep. 36, 102130. doi: 10.1016/j.aqrep.2024.102130
Fu Z., Lin Z., Huang K., Li Z., Luo Z., Han F., et al. (2024). Dinotefuran exposure alters biochemical, metabolomic, gut microbiome, and growth responses in decapoda pacific white shrimp Penaeus vannamei. J. Hazard. Mater. 469, 133930. doi: 10.1016/j.jhazmat.2024.133930
Fu Y., Wang Y., Gao H., Li D., Jiang R., Ge L., et al. (2021). Associations among dietary omega-3 polyunsaturated fatty acids, the gut microbiota, and intestinal immunity. Mediators Inflamm. 2021, 8879227. doi: 10.1155/2021/8879227
Gao Q., Liu P., Li Y., Song D., Long W., Wang Z., et al. (2023). Gut microbiota, host genetics and phenotypes in aquatic animals: a review. Aquacul. Rep. 31, 101648. doi: 10.1016/j.aqrep.2023.101648
Gharami K., Das M., and Das S. (2015). Essential role of docosahexaenoic acid towards development of a smarter brain. Neurochem. Int. 89, 51–62. doi: 10.1016/j.neuint.2015.08.014
Grabner G. F., Zimmermann R., Schicho R., and Taschler U. (2017). Monoglyceride lipase as a drug target: at the crossroads of arachidonic acid metabolism and endocannabinoid signaling. Pharmacol. Ther. 175, 35–46. doi: 10.1016/j.pharmthera.2017.02.033
Gyamfi D., Ofori Awuah E., and Owusu S. (2019). “Lipid metabolism: an overview,” in The Molecular Nutrition of Fats. Ed. Patel V. B. (Academic Press, New York, NY), 17–32. doi: 10.1016/B978-0-12-811297-7.00002-0
Gyan R. W., Yang Q., Tan B., Dong X., Chi S., Liu H., et al. (2022). Effects of replacing fish meal with distillers’ dried grains with solubles on the growth performance and gut microbiota in juvenile Pacific whiteleg shrimp Litopenaeus vannamei. N. Am. J. Aquacul. 84, 191–205. doi: 10.1002/naaq.10236
He Y., Wang X., Zhang Y., Guo Z., Jiang Y., and Chen F. (2019). Enzymatic ethanolysis subjected to Schizochytrium biomass: sequential processing for DHA enrichment and biodiesel production. Energy Convers. Manage. 184, 159–171. doi: 10.1016/j.enconman.2019.01.051
Hishikawa D., Valentine W. J., Iizuka-Hishikawa Y., Shindou H., and Shimizu T. (2017). Metabolism and functions of docosahexaenoic acid-containing membrane glycerophospholipids. FEBS Lett. 591, 2730–2744. doi: 10.1002/1873-3468.12825
Holt C. C., Bass D., Stentiford G. D., and van der Giezen M. (2021). Understanding the role of the shrimp gut microbiome in health and disease. J. Invertebr. Pathol. 186, 107387. doi: 10.1016/j.jip.2020.107387
Huang F., Pan L., Song M., Tian C., and Gao S. (2018). Microbiota assemblages of water, sediment, and intestine and their associations with environmental factors and shrimp physiological health. Appl. Microbiol. Biotechnol. 102, 8585–8598. doi: 10.1007/s00253-018-9229-5
Jiang X., Pan K., Yang Y., Shu-Chien A. C., and Wu X. (2022). Dietary DHA oil supplementation promotes ovarian development and astaxanthin deposition during the ovarian maturation of Chinese mitten crab Eriocheir sinensis. Aquacult. Nutr. 2022, 9997317. doi: 10.1155/2022/9997317
Jin J., Jin Q., Wang X., and Akoh C. C. (2020). High Sn-2 docosahexaenoic acid lipids for brain benefits, and their enzymatic syntheses: a review. Engineering 6, 424–431. doi: 10.1016/j.eng.2020.02.009
Jin M., Lu Y., Pan T., Zhu T., Yuan Y., Sun P., et al. (2019). Effects of dietary n-3 LC-PUFA/n-6 C18 PUFA ratio on growth, feed utilization, fatty acid composition and lipid metabolism related gene expression in black seabream, Acanthopagrus schlegelii. Aquaculture 500, 521–531. doi: 10.1016/j.aquaculture.2018.10.056
Kim P. S., Shin N. R., Lee J. B., Kim M. S., Whon T. W., Hyum D. W., et al. (2021). Host habitat is the major determinant of the gut microbiome of fish. Microbiome 9, 166. doi: 10.1186/s40168-021-01113-x
Klińska-Bąchor S., Demski K., Gong Y., and Banaś A. (2024). Biochemical characterization of acyl-CoA:diacylglycerol acyltransferase2 from the diatom Phaeodactylum tricornutum and its potential effect on LC-PUFAs biosynthesis in planta. BMC Plant Biol. 24, 309. doi: 10.1186/s12870-024-05014-7
Körbes A. P., Kulcheski F. R., Margis R., Margis-Pinheiro M., and Turchetto-Zolet A. C. (2016). Molecular evolution of the lysophosphatidic acid acyltransferase (LPAAT) gene family. Mol. Phylogenet. Evol. 96, 55–69. doi: 10.1016/j.ympev.2015.12.001
Kumar N., Chandan N. K., Gupta S. K., Bhushan S., and Patole P. B. (2022). Omega-3 fatty acids effectively modulate growth performance, immune response, and disease resistance in fish against multiple stresses. Aquaculture 547, 737506. doi: 10.1016/j.aquaculture.2021.737506
Kumar V., Habte-Tsion H., Allen K. M., Bowman B. A., Thompson K. R., El-Haraon E., et al. (2018). Replacement of fish oil with Schizochytrium meal and its impact on the growth and lipid metabolism of Pacific white shrimp (Litopenaeus vannamei). Aquacul. Nutr. 24, 1769–1781. doi: 10.1111/anu.12816
Li F., Ning Y., Zhang Y., Huang H., Yuan Q., Wang X., et al. (2025). Positional distribution of DHA in triacylglycerols: natural sources, synthetic routes, and nutritional properties. Crit. Rev. Food Sci. Nutr. 1–19. doi: 10.1080/10408398.2025.2479071
Li K. and Olsen R. E. (2017). Metabolism of sn-1(3)-monoacylglycerol and sn-2-monoacylglycerol in caecal enterocytes and hepatocytes of brown trout (Salmo trutta). Lipids 52, 61–71. doi: 10.1007/s11745-016-4215-0
Liang X., Luo X., Lin H., Han F., Qin J. G., Chen L., et al. (2022). Growth, health, and gut microbiota of female pacific white shrimp, Litopenaeus vannamei broodstock fed different phospholipid sources. Antioxidants 11, 1143. doi: 10.3390/antiox11061143
Lin Q., Yang L., Han L., Wang Z., Luo M., Zhu D., et al. (2022). Effects of soy hull polysaccharide on dyslipidemia and pathoglycemia in rats induced by a high-fat-high-sucrose diet. Food Sci. Hum. Wellness 11, 49–57. doi: 10.1016/j.fshw.2021.07.006
Liu R., Ding Y., Jing F., Chen Z., Su C., and Pan L. (2024). Effects of dietary glycerol monolaurate on growth and digestive performance, lipid metabolism, immune defense and gut microbiota of shrimp (Penaeus vannamei). Fish Shellfish Immunol. 151, 109666. doi: 10.1016/j.fsi.2024.109666
López-Marcos S., Escobedo-Fregoso C., Palacios E., Kraffe E., Arredondo-Espinoza R., Barajas-Sandoval D., et al. (2024). Muscle transcriptional response and fatty acid profile of Pacific white shrimp Litopenaeus vannamei fed dietary fish and canola oil: insights into growth performance discrepancies. Aquacul. Int. 32, 8479–8500. doi: 10.1007/s10499-024-01575-4
Luan Y., Li M., Zhou W., Yao Y., Yang Y., Zhang Z., et al. (2023). The fish microbiota: research progress and potential applications. Engineering 29, 137–146. doi: 10.1016/j.eng.2022.12.011
Neylan K. A., Johnson R. B., Barrows F. T., Marancik D. P., Hamilton S. L., and Gardner L. D. (2024). Evaluating a microalga (Schizochytrium sp.) as an alternative to fish oil in fish-free feeds for sablefish (Anoplopoma fimbria). Aquaculture 578, 740000. doi: 10.1016/j.aquaculture.2023.740000
Nobusawa K., Hori K., Mori H., Kurokawa K., and Ohta H. (2017). Differently localized lysophosphatidic acid acyltransferases crucial for triacylglycerol biosynthesis in the oleaginous alga Nannochloropsis. Plant J. 90, 547–559. doi: 10.1111/tpj.13512
Oxley A., Jutfelt F., Sundell K., and Olsen R. E. (2007). Sn-2-monoacylglycerol, not glycerol, is preferentially utilised for triacylglycerol and phosphatidylcholine biosynthesis in Atlantic salmon (Salmo salar L.) intestine. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 146, 115–123. doi: 10.1016/j.cbpb.2006.09.007
Phonsiri K., Mavichak R., Panserat S., and Boonanuntanasarn S. (2024). Differential responses of hepatopancreas transcriptome between fast and slow growth in giant freshwater prawns (Macrobrachium rosenbergii) fed a plant-based diet. Sci. Rep. 14, 4957. doi: 10.1038/s41598-024-54349-6
Qiao F., Liu Y. K., Sun Y. H., Wang X. D., Chen K., Li Y. T., et al. (2017). Influence of different dietary carbohydrate sources on the growth and intestinal microbiota of Litopenaeus vannamei at low salinity. Aquacul. Nutr. 23, 444–452. doi: 10.1111/anu.12412
Santigosa E., Lemos D., Coelho R. T. I., and Carr I. (2024). Omega-3 EPA and DHA from algal oil improve pacific white shrimp zootechnical performance and nutritional quality without changing sensory quality. Aquacult. Res. 2024, 7543478. doi: 10.1155/2024/7543478
Scheuer F., Owatari M. S., Brasil E. M., Sterzelecki F. C., Wagner R., Xavier A. C., et al. (2024). Dietary enrichment with fish oil improved n–3 LC-PUFA profile in aquacultured Sardinella brasiliensis fillet. J. Food Compos. Anal. 127, 105978. doi: 10.1016/j.jfca.2024.105978
Shen L., Li F., Jiang C., Cao X., Jin J., Wang X., et al. (2024). Comparative analysis of DHA positional distribution and triacylglycerol molecular species in algal oil (Schizochytrium sp.) from different oil processing. Food Biosci. 58, 103634. doi: 10.1016/j.fbio.2024.103634
Shin J., Feyaerts J., Hasanthi M., Lee C., Shin D., Lee Y., et al. (2023). Evaluation of a mixture of short-chain and medium-chain fatty acid glycerides as a dietary supplement in diets for Pacific white shrimp (Litopenaeus vannamei). Aquacul. Int. 31, 1483–1498. doi: 10.1007/s10499-022-01037-9
Sprague M., Betancor M. B., and Tocher D. R. (2017). Microbial and genetically engineered oils as replacements for fish oil in aquaculture feeds. Biotechnol. Lett. 39, 1599–1609. doi: 10.1007/s10529-017-2402-6
Sun H., Song Y., Zhang H., Zhang X., Liu Y., Wang X., et al. (2020). Characterization of lipid composition in the muscle tissue of four shrimp species commonly consumed in China by UPLC–Triple TOF–MS/MS. LWT 128, 109469. doi: 10.1016/j.lwt.2020.109469
Tang Y., Zhang Z., Zhang L., He Y., Xu N., Fang W., et al. (2023). Effects of supplemental glycerol monolaurate on growth, hepatic lipid metabolism, antioxidant capacity and mitochondrial function of large yellow croaker (Larimichthys crocea) fed diets with high soybean oil level. Aquanculture 564, 739024. doi: 10.1016/j.aquaculture.2022.739024
Tocher D. R., Betancor M. B., Sprague M., Olsen R. E., and Napier J. A. (2019). Omega-3 long-chain polyunsaturated fatty acids, EPA and DHA: bridging the gap between supply and demand. Nutrients 11, 89. doi: 10.3390/nu11010089
Tran N. T., Li Z., Wang S., Zheng H., Aweya J. J., Wen X., et al. (2020). Progress and perspectives of short-chain fatty acids in aquaculture. Rev. Aquacul. 12, 283–298. doi: 10.1111/raq.12317
Tu C., Qi X., Shui S., Lin H., Benjakul S., and Zhang B. (2022). Investigation of the changes in lipid profiles induced by hydroxyl radicals in whiteleg shrimp (Litopenaeus vannamei) muscle using LC/MS-based lipidomics analysis. Food Chem. 369, 130925. doi: 10.1016/j.foodchem.2021.130925
Vogt G. (2019). Functional cytology of the hepatopancreas of decapod crustaceans. J. Morphol. 280, 1405–1444. doi: 10.1002/jmor.21040
Wang Y., Abdullah, Zhang C., Li Y., Zhang H., Wang J., et al. (2020). Effects of dietary glycerol monolaurate on the growth performance, digestive enzymes, body composition and non-specific immune response of white shrimp (Litopenaeus vannamei). Aquacul. Rep. 18, 100535. doi: 10.1016/j.aqrep.2020.100535
Wang Y., Li M., Filer K., Xue Y., Ai Q., and Mai K. (2017). Replacement of fish oil with a DHA-rich Schizochytrium meal on growth performance, activities of digestive enzyme and fatty acid profile of Pacific white shrimp (Litopenaeus vannamei) larvae. Aquacul. Nutr. 23, 1113–1120. doi: 10.1111/anu.12479
Wang L., Zang M., Cheng X., Wang S., Zhao X., Zhao B., et al. (2024). Evaluation of changes in the lipid profiles of dried shrimps (Penaeus vannamei) during accelerated storage based on chemical and lipidomics analysis. LWT 191, 115564. doi: 10.1016/j.lwt.2023.115564
Wang C., Zhang C., Yu H., Zan Z., Li J., Li P., et al. (2022). Glycerol monolaurate and triglycerol monolaurate alleviated high-fat diet induced lipid accumulation and damage of liver in zebrafish (Danio rerio). Aquaculture 561, 738616. doi: 10.1016/j.aquaculture.2022.738616
Wayne L. L., Gachotte D. J., Graupner P. R., Adelfinskaya Y., McCaskill D. G., Metz J. G., et al. (2021). Plant and algal lysophosphatidic acid acyltransferases increase docosahexaenoic acid accumulation at the sn-2 position of triacylglycerol in transgenic Arabidopsis seed oil. PloS One 16, e0256625. doi: 10.1371/journal.pone.0256625
Weil C., Lefèvre F., and Bugeon J. (2013). Characteristics and metabolism of different adipose tissues in fish. Rev. Fish Biol. Fisheries 23, 157–173. doi: 10.1007/s11160-012-9288-0
Wu S., Tan J., Zhang H., Hou D., and He J. (2023). Tissue-specific mechanisms of fat metabolism that focus on insulin actions. J. Adv. Res. 53, 187–198. doi: 10.1016/j.jare.2022.12.009
Xiang X., Ji R., Han S., Xu X., Zhu S., Li Y., et al. (2024). Differences in diacylglycerol acyltransferases expression patterns and regulation cause distinct hepatic triglyceride deposition in fish. Commun. Biol. 7, 480. doi: 10.1038/s42003-024-06022-x
Xie S., Wei D., Tan B., Liu Y., Tian L., and Niu J. (2020). Schizochytrium limacinum supplementation in a low fish-meal diet improved immune response and intestinal health of juvenile. Penaeus monodon. Front. Physiol. 11. doi: 10.3389/fphys.2020.00613
Xu H., Meng X., Jia L., Wei Y., Sun B., and Liang M. (2020). Tissue distribution of transcription for 29 lipid metabolism-related genes in Takifugu rubripes, a marine teleost storing lipid predominantly in liver. Fish Physiol. Biochem. 46, 1603–1619. doi: 10.1007/s10695-020-00815-7
Yu H., Fang C., Li P., Wu M., and Shen S. (2021). The relevance of DHA with modulating of host-gut microbiome signatures alterations and repairing of lipids metabolism shifts. Eur. J. Pharmacol. 895, 173885. doi: 10.1016/j.ejphar.2021.173885
Yu Z., Li D., Yin F., Zhao Q., Liu Z., Song L., et al. (2020). Lipid profiles in by-products and muscles of three shrimp species (Penaeus monodon, Penaeus vannamei, and Penaeus chinensis). Eur. J. Lipid Sci. Technol. 122, 1900309. doi: 10.1002/ejlt.201900309
Zeng S., He J., and Huang Z. (2024). The intestine microbiota of shrimp and its impact on cultivation. Appl. Microbiol. Biotechnol. 108, 362. doi: 10.1007/s00253-024-13213-3
Zhang H., Liu M., Shao R., Zhang J., Zuo R., Tan B., et al. (2022). The effects of different lipid sources on the growth, intestinal health, and lipid metabolism of the Pacific white shrimp (Litopenaeus vannamei). Aquanculture 548, 737655. doi: 10.1016/j.aquaculture.2021.737655
Zhang M., Sun Y., Chen K., Yu N., Zhou Z., Chen L., et al. (2014). Characterization of the intestinal microbiota in Pacific white shrimp, Litopenaeus vannamei, fed diets with different lipid sources. Aquaculture 434, 449–455. doi: 10.1016/j.aquaculture.2014.09.008
Zheng Y., Yu M., Liu J., Qiao Y., Wang L., Li Z., et al. (2017). Bacterial community associated with healthy and diseased Pacific white shrimp (Litopenaeus vannamei) larvae and rearing water across different growth stages. Front. Microbiol. 8. doi: 10.3389/fmicb.2017.01362
Keywords: Litopenaeus vannamei, docosahexaenoic acid, monoglycerides, lipidomics, intestinal microbiota
Citation: Huang W, Song Y, Wang X, Lin D, Luo T, Cheng K, Li Q, Tian B, Zheng Y and He Y (2025) Replacement of fish oil with Schizochytrium-derived monoglycerides enriched in docosahexaenoic acid on the growth performance, lipid composition, and intestinal microbiota of Pacific white shrimp (Litopenaeus vannamei). Front. Mar. Sci. 12:1588954. doi: 10.3389/fmars.2025.1588954
Received: 06 March 2025; Accepted: 11 July 2025;
Published: 05 August 2025.
Edited by:
Gladstone Sagada, Victory Farms, KenyaReviewed by:
Sofia Priyadarsani Das, National Taiwan Ocean University, TaiwanYuwen Dong, University of Pennsylvania, United States
Ioannis A Giantsis, Aristotle University of Thessaloniki, Greece
Amit Ranjan, Tamil Nadu Fisheries University, India
Federico Moroni, Spanish National Research Council (CSIC), Spain
Guoxiang Chi, Xiamen Medical College, China
Copyright © 2025 Huang, Song, Wang, Lin, Luo, Cheng, Li, Tian, Zheng and He. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yongbiao Zheng, eW9uZ2JpYW96aGVuZ0Bmam51LmVkdS5jbg==; Yongjin He, eW9uZ2ppbmhlQGZqbnUuZWR1LmNu
 Wei Huang
Wei Huang Yongkang Song2
Yongkang Song2 Xiaonan Wang
Xiaonan Wang Baoyu Tian
Baoyu Tian Yongjin He
Yongjin He