- 1Department of Coastal Studies, East Carolina University, Wanchese, NC, United States
- 2Coastal Studies Institute, Department of Biology, East Carolina University, Wanchese, NC, United States
- 3Integrated Coastal Sciences PhD Program, East Carolina University, Greenville, NC, United States
Environmental changes in the ocean are increasingly affecting commercially important fishery species. The relationship between environmental parameters and the abundance, distribution, phenology and timing of the reproductive cycles in marine organisms though is often not well understood. Our study explores the impact of storms, rainfall, temperature, wind and offshore reproduction on two commercial shrimp populations, brown and white shrimp in North Carolina, using a survey among commercial shrimp fishers and a set of quantitative population models that were developed from a long-term biological dataset. We identify multiple overlaps but also differences between fishers' observations and model outputs in particular for the impacts of wind and storms. We further find that fishers mainly rely on their own experience and other fishers to learn about shrimp abundance and less on information provided by fishery managers. Perceptions of environmental drivers were related to livelihood dependency on the shrimp fishery and fishing experience while knowledge seeking behavior was related to fishing experience, the role fishers play in the fishery, and the number of household members engaged in fishing. We discuss implications for shrimp fishery management and discuss challenges of comparing LEK data collected from fishers with modeling outputs including potential differences in temporal and spatial scales.
1 Introduction
Environmental changes are increasingly affecting commercially important marine species and fisheries globally (Heck et al., 2023; Blasiak et al., 2017). Change in environmental parameters, for example, can affect species’ abundance, distribution, as well as phenology (Sunday et al., 2012; Perry et al., 2005) and can lead to shifts in the timing of reproduction cycles in fishes and invertebrates (Pankhurst and Munday, 2011). These changes are often not well understood and challenge fishery decision-making, which is historically built on the assumption of more static, equilibrium-based fish population dynamics (Szuwalski and Hollowed, 2016; Pinsky et al., 2018).
One option to identify environmental drivers of change in marine species is the use of conventional scientific knowledge (CSK) that is based on data collected according to specific scientific designs and theories (Gaspare et al., 2015; Mackinson, 2001). One example in the context of fisheries is quantitative-empirical modeling that relies on long-term biological survey data (Tuckey et al., 2021; Kohut et al., 2013). These types of models are often based on routine-annual fisheries independent surveys that use a statistically rigorous design such as random stratification that ensures a representative distribution of samples across a relevant environmental parameter such as salinity or depth. These types of data are useful to understand the response of species to environmental conditions and may be used to forecast future scenarios (Bell et al., 2018; Hare et al., 2010). At the same time, empirical models are a simplified representation of a more complex reality and are dependent on underlying assumptions and methods (Ehrlén and Morris, 2015). For example, the survey data used for empirical modeling often comes from discrete-consistent time periods during the year but are used to represent more comprehensive annual-scale trends. Further, annual surveys conducted by management agencies might vary in how well they overlap with harvested populations in space and time (Kohut et al., 2013; Karp et al., 2019). These limitations are important, because fisheries management typically requires information across multiple spatial and temporal scales on an ecosystem-based level for developing salient management strategies (Stephenson et al., 2016; Zeller et al., 2016).
Another type of information to increase the understanding of fisheries dynamics is the integration of local ecological knowledge (LEK) into fishery management, either alone or in combination with empirical models (Silvano and Hallwass 2020; Cook et al., 2014). LEK can be used address some of the limitations of empirical models by providing more comprehensive insights into a fishery or provide essential knowledge on fisheries dynamics in areas where robust data sets to build empirical models are missing (Thornton and Scheer 2012; Silvano and Begossi 2012; Silas et al., 2023). Fishers LEK, defined as the knowledge possessed by fishers about fisheries and their interactions with the local marine environment, is typically generated through fishers’ observations and experience with the local marine environment and its resources over time (Silvano and Valbo-Jørgensen, 2008; Murray et al., 2006; Berkes, 2002). Fishers commonly interact with their local marine environment daily and are able to observe changes in environmental conditions through direct interactions (Morales et al., 2017). In addition, exchange with other fishers can influence LEK as fishers routinely communicate within trusted networks to create a cumulative pool of fishers’ knowledge (Grant and Berkes 2007). Fishers’ LEK thus can provide a detailed understanding of fisheries such as knowledge on fish stocks and population dynamics (Roedel, 1979; Johannes, 1981), historical changes in marine stocks and marine environmental conditions (Johannes et al., 2000), fisheries composition, abundance, and exploitation status (Bender et al., 2014), trophic relationships (Ramires et al., 2015), and historical changes in the marine environment (e.g., turbidity, water temperature) (Rosa et al., 2014; Begossi et al., 2012; Dlamini et al., 2022).
Despite the growing recognition of the importance of LEK, this type of knowledge is rarely applied in fisheries management (Tomasini and Theilade, 2019; Hind, 2015) and mainly promoted in developing countries in instances where data and resources are limited (Berkström et al., 2019). LEK has not commonly been incorporated in fisheries management in developed countries where fisheries management is typically driven by scientific data (Sowman et al., 2014). LEK is also sometimes labeled as ‘anecdotal knowledge’, which could be one of the reasons for its limited uptake in fisheries management in areas that mainly rely on stock assessments and related fishery data (Silvano and Valbo-Jørgensen, 2008). In addition, managers often lack an understanding of how different types of knowledge, such as LEK, might inform fishery management decisions (Heck et al., 2015).
Yet, combining insights from LEK and scientific information can provide a more detailed understanding of a fishery (Hauck et al., 2002; Sowman et al., 2014; Dlamini et al., 2022) and could be used in fishery management decisions, not just in data poor regions, but also in instances when both types of data are available (Ainsworth and Pitcher, 2005). Thus, there is increasingly an interest in integrating LEK with other sources of data to better understand the complexity of fisheries (Berkström et al., 2019). Yet very few studies have actually combined insights from LEK and data driven approaches (Hamilton et al., 2011). Among the few existing studies, examples include comparisons of LEK insights with stock assessment data (Ainsworth and Pitcher, 2005) and species distribution models (Lopes et al., 2019), and combined landings data and underwater visual census data with LEK to assess temporal patterns of exploited fish populations (Azzurro et al., 2019). These studies vary in the level of agreement between the different types of data and highlight challenges of integrating LEK with other sources or data, e.g., shifting baselines for understanding past fishery dynamics. Due to the limited number of studies in this area, more research is needed to explore challenges and opportunities for the integration of LEK with data driven approaches for different aspects and types of fisheries, and to identify in which instances different types of data provide similar insights into fisheries, more complementary information, or if outcomes are divergent.
Our study compares LEK from commercial shrimp fishers with a set of quantitative population models that were developed from a long-term biological dataset to explore environmental drivers of changes in two commercial shrimp populations (NCDMF, 2022; Schlenker et al., 2023; Tuckey et al., 2021; Kimball et al., 2020). By combining two information sources, we identified the main environmental drivers of variations in shrimp populations and identify to what extent insights of LEK and empirical models are similar In addition, we explored how fishers learn about the abundance of shrimp in the area. To investigate these objectives, we explore the following questions: 1) What are the main environmental drivers that affect shrimp populations based on fishers’ knowledge and modeling outputs and how similar are insights derived from LEK and empirical models? 2) How do fishers learn about shrimp abundance? 3) Is there a difference in information seeking behavior and observed environmental drivers among fishers based on their fishing experience, livelihood dependency and their role in the fishery? Our study will provide novel insights into environmental drivers of shrimp populations in North Carolina, where monitoring data of shrimp is limited. In addition, our findings will provide insights on the combined use of modeling data and LEK for identifying environmental drivers of change for an annual fishery and identify to what extent observations of fishers agree or disagree with modeling outputs.
2 Methods
2.1 Study system
Our study explores environmental drivers of change in the shrimp trawl fishery in the Pamlico Sound, North Carolina USA (Figure 1). The Pamlico Sound is the largest lagoonal estuary in the United States and is highly productive for white and brown shrimp. Typically, over half of the annual shrimp landings in North Carolina come from the Pamlico Sound (NCDMF, 2022). About 400 fishers participate in the shrimp fishery annually during recent years (NCDMF, 2022) and the stock is managed by the North Carolina Division of Marine Fisheries (NC DMF). Between 1994 and 2022 landings of shrimp in NC averaged about 7,481,925 pounds per year though they can vary widely from year to year (NCDMF, 2022). The main data available on NC shrimp stocks are commercial landings derived from NC Trip Tickets, a form used by fish dealers to report commercial landings to the state, and two fisheries-independent trawl surveys run by NC DMF.
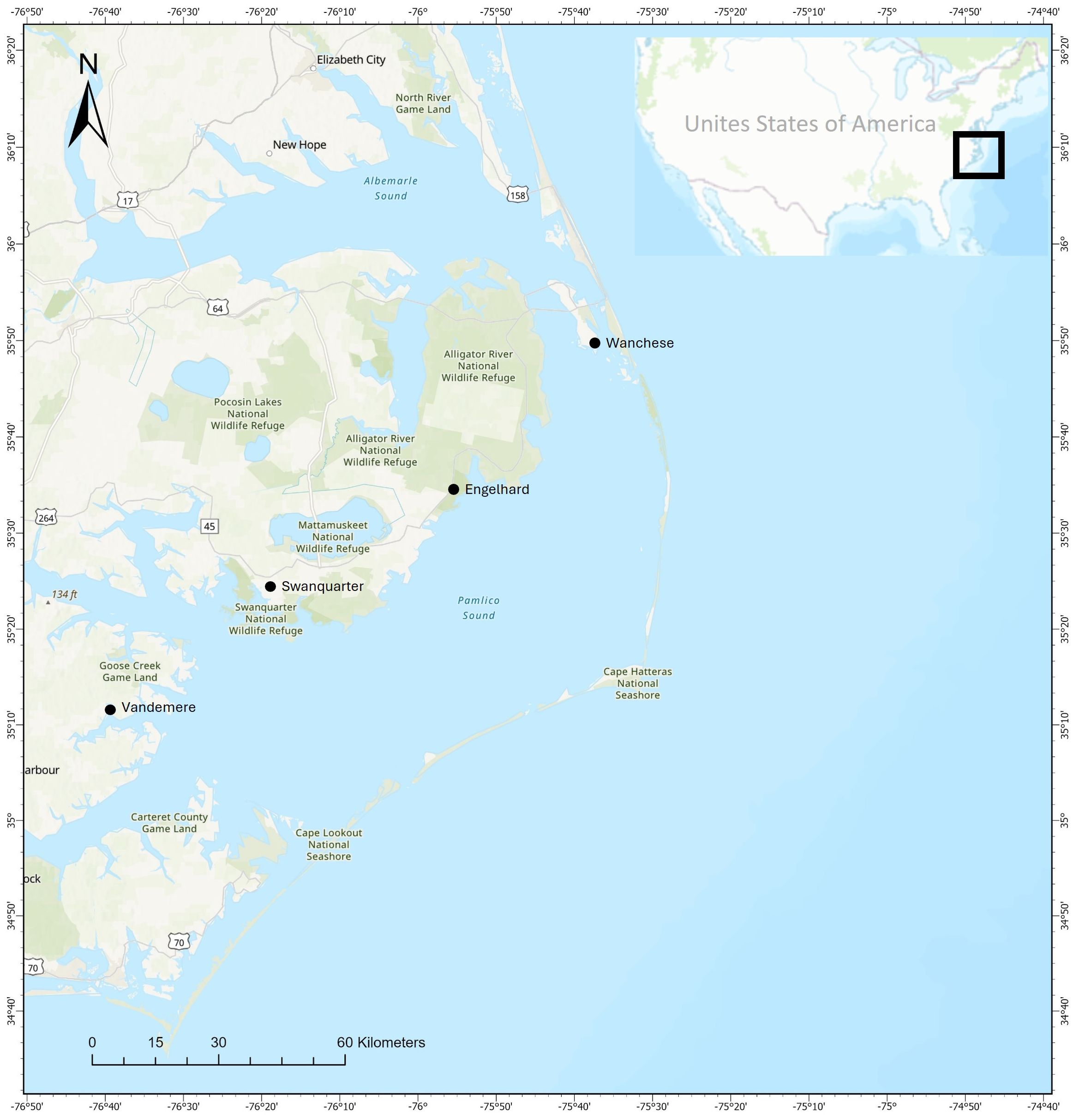
Figure 1. Overview of Pamlico Sound; insert: location within the United States. Basemap from ESRI online.
Brown and white shrimp have a complex annual lifecycle in which adults reproduce in ocean waters and juveniles and sub adults grow in estuarine waters, including the Pamlico Sound, before emigrating back to the ocean (NCDMF, 2022). Abundance of shrimp can vary dramatically from one year to the next in the Pamlico Sound, and this variation is reflected in the commercial catch (Schlenker et al., 2023). In multiple systems in the southeast U.S., penaeid shrimp abundance has been shown to be highly dependent on environmental conditions and shrimp populations in this region are responding rapidly to climate change (Schlenker et al., 2023; Tuckey et al., 2021; Kimball et al., 2020). While white and brown shrimp have a similar life cycle, they differ in reproductive and juvenile recruitment timing by several months, which means that these two species are subjected to different environmental drivers and their abundance cycles are not synchronous (Schlenker et al., 2023). Further, white shrimp are thought to be more sensitive to winter temperatures than brown shrimp in this region (Morley et al., 2017).
2.2 Fishers LEK
A questionnaire-based survey was distributed to North Carolina shrimp fishers who fish in the Pamlico Sound between November 2020 and May 2022 using a random sampling approach. Before distributing the full survey, we piloted the survey with a subset of five local fishers. The survey was exempt by ECU IRB UMCIRB 20-002189. In total, we distributed 275 surveys with a margin of error of 3.3% with a 95% confidence interval as the total number of commercial shrimp fishers is about 400. Due to University restrictions as a result of the COVID-19 pandemic, two hundred surveys were left at fish houses, where commercial fishers unload and report their catch, in multiple commercial fishing ports including the communities of Wanchese, Engelhard, Swan Quarter, and Vandemere, North Carolina between November 2020 and May 2021. Surveys were left with a provided pre-paid return envelope and a QR code for an online response option. After University restrictions were lifted, fishers were approached in person at six shrimp trawling ports along the coast of North Carolina using an intercept approach. The fishing ports included the communities of Oriental, Bayboro, Beaufort, Sneads Ferry, Belhaven, North Carolina and Newport News, Virginia. Shrimp fishers had the choice of completing the questionnaire and returning it on sight or returning it via a pre-paid return envelope. In total 50 surveys were returned including 15 online surveys and 35 paper-based surveys. Fifteen online surveys and 23 paper-based surveys were returned during the first phase based on questionnaire drop-offs in fish houses, with a return rate of 19%. During the intercept survey, 12 out of 75 surveys were returned, with a response rate of 16%.
The survey included questions about observed shrimp abundance and environmental drivers that might affect the shrimp population. We first asked about the observed trend in the historic abundance of white and brown shrimp on a scale from decreasing (-1), stable/no trend (0) or increasing (1). The next section determined fishers’ knowledge about the impact of multiple environmental variables on shrimp abundance. These variables included the abundance of reproductive adult shrimp, winds, temperature, rainfall patterns, and major storm events. The response data were coded as binary variables, where the environmental driver had an effect (1) or not (0). In addition, we included an open-ended question for each environmental variable asking about the details about the environmental driver and its impact on shrimp abundance. The survey also included an “other” option for potential drivers that we did not include. In addition, the questionnaire included questions on fishing patterns including the role that respondents have in the fishery (boat owner, captain or other), years being active in the shrimp fishery, number of different fisheries that respondents participated in ranging from 1–3 or more, how fishers learn about shrimp populations including their own experience, family and friends, other fishers, fishery managers or fishery blogs and online information. The last section focused on socio-demographic information including year of birth, livelihood dependency on the shrimp fishery (no dependency =0 to very high dependency =4), and how many household members participate in fishing (see Supplementary S2 for the questionnaire).
Survey data was analyzed in IBM SPSS 29 using descriptive statistics, Chi-square analysis, and Spearman rank correlation analysis to analyze correlations method of learning about the fishery with fishing patterns, livelihood dependency, if fishing was the main source of income (0=no, 1=yes) and number of household members engaged in fishing. For correlation analysis, role of the fishery was coded as boat captain =1 and others = 0. Qualitative data in open-ended questions were analyzed in excel. The data was used to create a Sankey diagram to illustrate the connections between environmental drivers and changes in the shrimp populations. The strengths of the Sankey connections indicate the number of respondents who mentioned this driver.
2.2 Shrimp population modeling
To compare LEK with empirically modeled shrimp fishery dynamics, we used output by Schlenker et al. (2023). Specifically, Schlenker et al. (2023) used regression-based modeling of annual distribution and abundance data of white and brown shrimp in the Pamlico Sound, North Carolina. The goal of that study was to determine the group of environmental variables that affect annual abundance of shrimp in this system. Notably, the modeling conducted by Schlenker et al. (2023) was based on fisheries-independent catch and environmental data.
Multiple shrimp life stages (juvenile and adult, depending on species) were modeled separately in Schlenker et al. (2023) using a generalized linear modeling (GLM) framework. Specifically, for brown shrimp we used the empirical model that explained interannual variation of the juvenile stage abundance within nursery habitats of the Pamlico Sound ecosystem. This model was based on a trawl survey called Program 120 that is conducted annually by the NC DMF and targets juvenile stage organisms in tidal creeks. The Program 120 dataset goes back to 1971, but we used data from 1986 to 2019 to match available climate data. Catches of juvenile brown shrimp in this survey have been shown to effectively track interannual changes in commercial landings occurring later in the same year, and thus effectively represents year class strength prior to any fisheries harvest (NCDMF 2022). The juvenile brown shrimp model also contained predictor variables that were either the same as those asked about in the LEK questionnaire of fishers (e.g., temperature) or were related variables (e.g., North Atlantic Oscillation in the empirical models and storm events in the fisher questionnaire; Table 1). The empirical model of juvenile brown shrimp explained 81% of the deviance (analogous to an r2 value) in annual abundance.
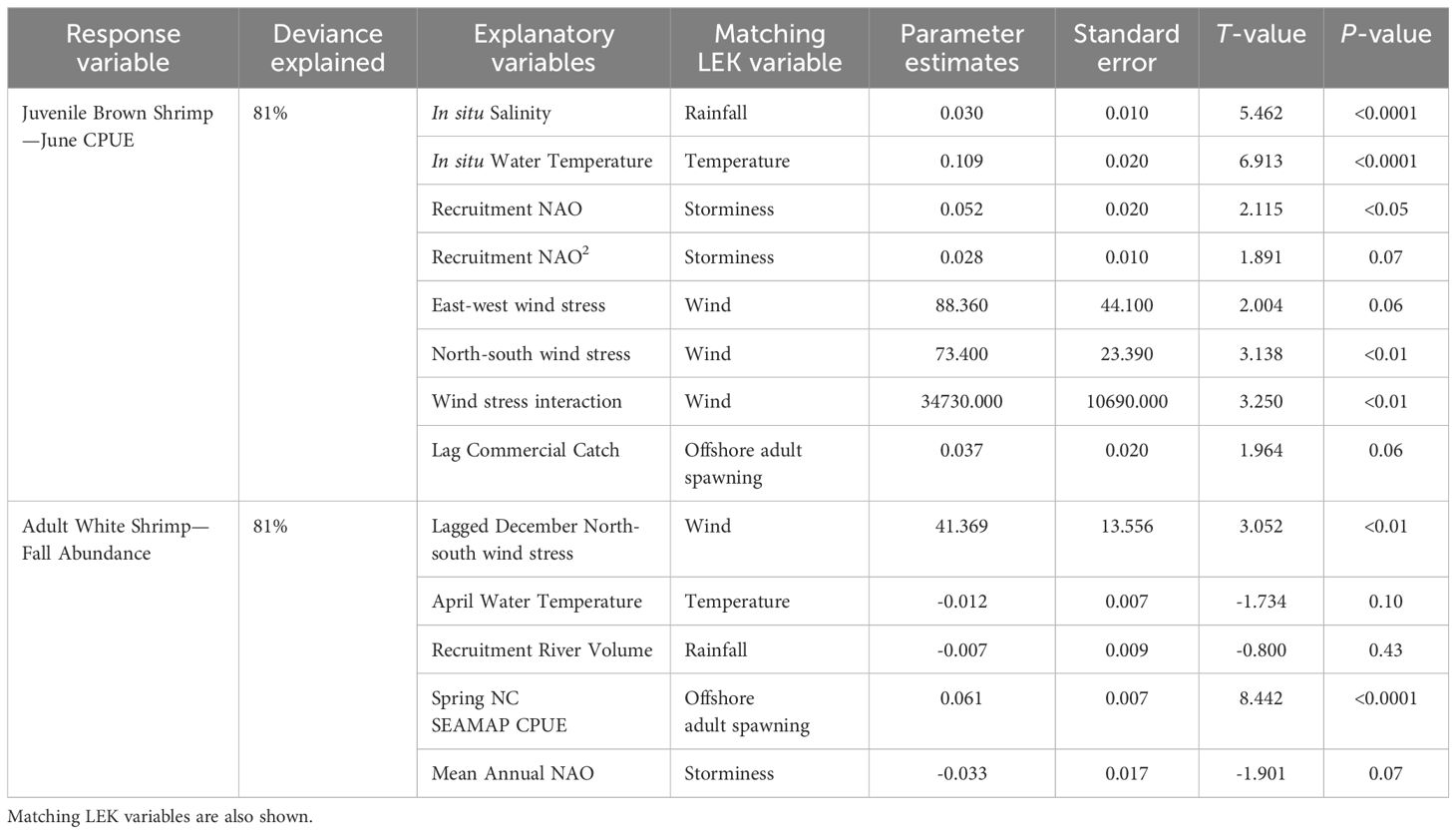
Table 1. Variables included for best fit generalized linear models predicting the annual mean catch per unit effort of juvenile brown shrimp and the modeled abundance of adult white shrimp.
For white shrimp there was only one empirical model option, which was of the adult life stage during the fall season, and thus the data are collected contemporaneously with fisheries harvest (Schlenker et al., 2023). Annual abundance of adult white shrimp was based on another annual trawl survey conducted by the NC DMF (Program 195), which originated in 1987 and takes place throughout the Pamlico Sound and the adjacent rivers that empty into it. The original white shrimp model explained 75% of the deviance in annual fall abundance, but contained only two variables, the reproductive biomass of adult white shrimp and December north wind stress. Therefore, for the present study, where we wanted to directly compare model predictions to fisher’s knowledge, we modified the model in Schlenker et al. (2023), by adding additional predictor variables to directly compare with LEK results. The variables asked about in our survey of fishers, but not included in the original white shrimp model, were temperature, rainfall, and storm events. To incorporate these additional variables, we used the same procedures outlined in Schlenker et al. (2023). Specifically, multiple GLM models were fitted for adult white shrimp that included different combinations and interactions of the predictor variable set that were ecologically relevant (1987-2019). These model variations were evaluated using Akaike’s Information Criterion corrected for small sample size (AICc) (Burnham and Anderson, 2002; Berg et al) and the proportion of deviance explained was calculated using the modEva package (Márcia Barbosa et al., 2013). The selected white shrimp model that compares environmental drivers to LEK explains 81% of the deviance in annual abundance (Table 1).
For both juvenile brown shrimp and adult white shrimp we calculated the relative importance of each predictor variable to the empirical model by running all possible sub-model combinations of the final predictor variable set. For each of these sub-models the AICc scores were calculated and the Akaike weights were summed for each variable (Burnham and Anderson, 2002). The relative variable importance was then adjusted to a 0 to 1 scale in order to compare with LEK results (see below).
2.3 Comparison of LEK with empirical modeling approaches
We compared LEK from commercial fishers with empirical modeling outcomes for shrimp historic abundance trends (1987-2019). Annual abundance from the empirical models was based on generalized additive models (GAMs) of NC DMF trawl survey data in the Pamlico Sound (Program 195 survey), as published in Schlenker et al. (2023). Briefly, GAMs were used to represent annual spatial variation in shrimp density across the Pamlico Sound, which allows interpolation between sampling stations of the Program 195 survey. The result is a gridded annual map of shrimp density that can be summed to get an annual value of total abundance (Schlenker et al., 2023). For the present analysis, those annual abundance estimates were rescaled to reflect deviations from the historic mean (i.e., Z-scores: subtracted long-term mean abundance and divided by standard deviation).
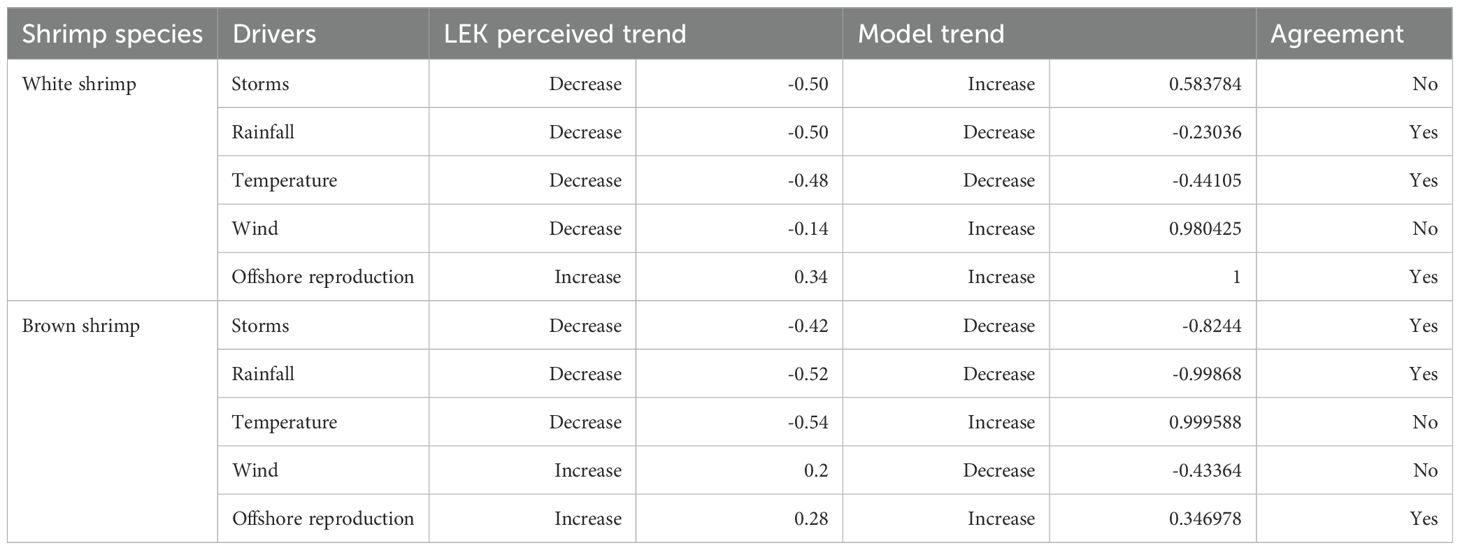
The present study also compared environmental drivers of shrimp abundance from the empirical modeling results with LEK using five matched environmental variables for both brown and white shrimp (Table 1). Although our LEK survey questions were designed with the intent to compare them to the empirical models in Schlenker et al., the timing of both studies’ progression did not allow for a perfect match between the variables examined and explanation for these variables between the different approaches is warranted (Table 1). Winds were included in our analyses, because they act as a proxy for surface ocean currents and estuarine water circulation, which can impact larval transport and recruitment of shrimp. Further, average wind intensity over time can be associated with broader seasonal weather patterns. Winds in the empirical model were included as a cumulative daily wind intensity in the north-south direction during December for white shrimp, which is thought to influence the distribution of reproductive adults in the ocean off North Carolina. For brown shrimp winds were included as both the north-south and east-west intensity during their recruitment period (January – April). Winds in the LEK approach simply asked fishers if winds were important in driving annual abundance. For temperature, empirical models used water temperature during the time of sampling for brown shrimp, while for white shrimp, water temperature during the period of reproduction in April was used. For LEK, the questionnaire did not specify air versus water temperature or season. This approach seemed justified, because in estuaries and coastal areas there is a tight coupling between air and water temperatures in the southeast U.S (Morley et al., 2017). To compare the importance of rainfall in the LEK results, empirical models of brown shrimp used salinity during the time of sampling, which was appropriate because rainfall in this system is related to the magnitude of subsequent streamflow and salinity in the Pamlico Sound (Durham et al., 2019). For white shrimp the influence of rainfall was included using stream gauge flow data in the watershed during the recruitment period (May – July), because salinity data were not available in white shrimp nursery habitats. Finally, the North Atlantic Oscillation (NAO) is a large-scale climate mode in this region, where a negative phase is linked to greater precipitation and more storm activity in the western Atlantic (Hurrell, 1995; Ezer and Atkinson, 2014). Therefore, the NAO variable from empirical models was matched with storm events in the LEK results.
Fishers’ observations of drivers of change was assessed as the percentage of respondents that selected a particular driver as important. The percentage of respondents for each variable, which ranged from 0-100% of respondents, was then converted to 0–1 scale by dividing the percent of respondents that selected a specific driver by 100. This score was compared to our index of relative importance for each predictor variable in the empirical models.
In situ Water Temperature and Salinity refer to the temperature and salinity measured by the corresponding survey, whereas April Temperature refers to mean April (period of reproduction for white shrimp) water temperature measured by three local NOAA buoys. Commercial Catch refers to the standardized catch of shrimp (weight) within Pamlico Sound. NC SEAMAP CPUE refers to the log catch per unit effort of shrimp on the North Carolina coast caught during the Southeast Area Monitoring and Assessment Program trawl survey. Recruitment is the species-specific time period during which post-larval shrimp recruit to Pamlico Sound.
3 Results
3.1 Socio-demographics of shrimp fishers included in our study
Most respondents were boat owners (62%) followed by vessel captains (32%), with some respondents falling into both categories (Table 2). 30% of respondents had other roles in the fishery including crew members, fish processors, or fish business owners or managers. The mean age of fishers was 49 years (21 minimum, 93 maximum). 30% of respondents had participated in the shrimp fishery for less than 10 years, 38% between 10–20 years and 32% of respondents have participated in the NC shrimp fishery for more than 20 years. For most respondents (63.3%), fishing was the main source of income, and most participants fished for both brown (92.0%) and white (93.9%) shrimp. In addition, 73% of fishers in this study participated in more than one fishery, although 68% of respondents indicated a high or very high dependency on the shrimp fishery for their livelihood. The mean number of family members working in the shrimp fishery was 3 (minimum 0, maximum 22).
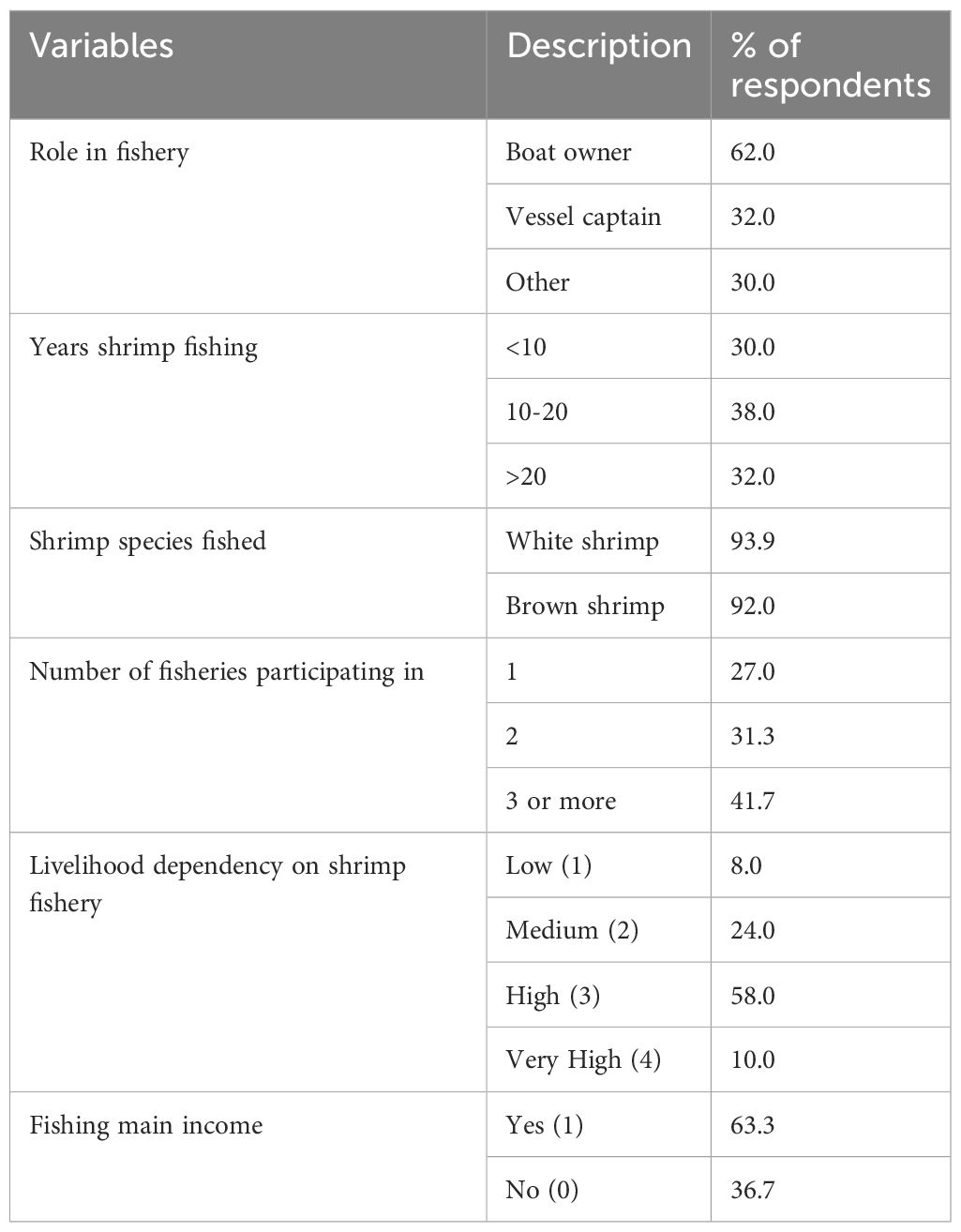
Table 2. North Carolina fishers’ socio-demographics and role in the fishery (N=50) based on a survey-questionnaire, including percentage of respondents that fit into specific categories.
3.2 Trend in the abundance of white and brown shrimp
The empirical model demonstrated variation in the annual abundance of shrimp over 33 years and was marked with periodic high abundance years in both species. For white shrimp there was a trend towards an increase in abundance over time, with the most recent seven years being above the historic average (Figure 2A). Fishers shared similar observations as almost half the respondents (47.7%) indicated an increase in white shrimp abundance over time and only 9.1% indicated a decreasing trend (Figure 2B). For brown shrimp, the empirical model showed little evidence of a trend in abundance since the mid-1990s. Similarly, fishers’ observations indicated that the population seemed to be stable without a clear trend (56.8%).
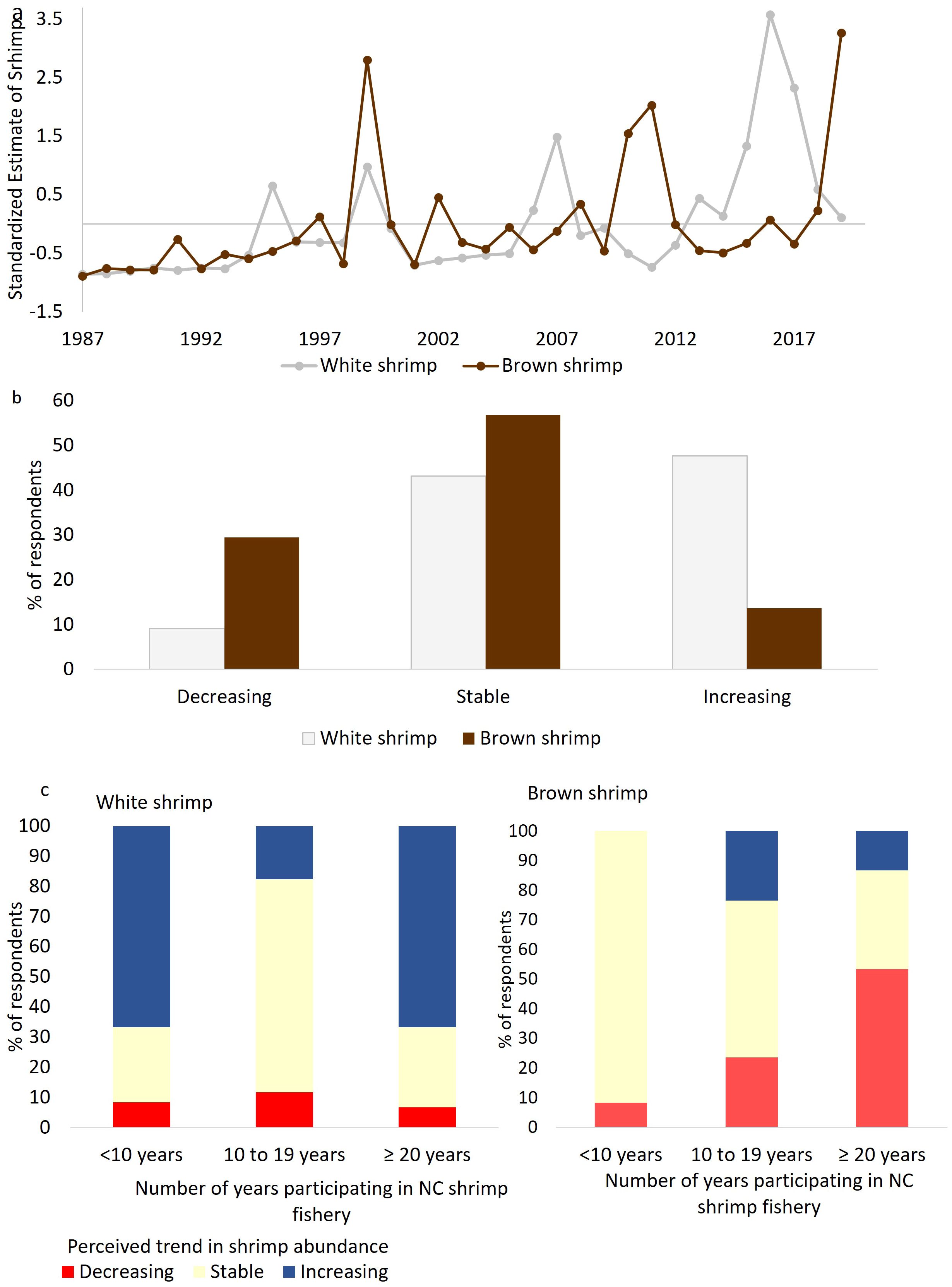
Figure 2. Changes in the annual abundance of white and brown shrimp in Pamlico Sound over time based on (A) empirically modeled abundance (B) fishers’ perceived trend in abundance over time, and (C) perceived trend in abundance over time by years participating in the shrimp fishery.
Additional analysis revealed significant differences in the perceived trend of shrimp abundance based on the time (<10, 10-19, or ≥20 years) fishers have been part of the NC shrimp fishery for both brown (χ2 = 11.832, df=4, p=0.019) and white shrimp (χ2 = 10.313, df=4, p=0.035). 67% of shrimp fishers that have been fishing for more than 20 years for shrimp in North Carolina and 67% of fishers having participated less than 10 years in the fishery observed an increase in white shrimp. On the other hand, 92% of fishers that have been less than 10 years in the shrimp fishery observed a stable trend for brown shrimp whereas 53% of fishers that spent more than 20 years fishing for shrimp in NC observed a declining trend in brown shrimp.
3.3 Drivers of changes in shrimp abundance
Fishers’ LEK identified similar environmental drivers of change between brown and white shrimp (Figure 3). Fishers consistently identified the abundance of the adult-reproductive population as driving increases in abundance. While rainfall and storms were frequently implicated as contributing to decreases in abundance. Temperature was frequently mentioned as a driver for both increases and decreases in abundance of both species. Finally, winds were not indicated as an important environmental driver compared to other variables, except for increases in brown shrimp.
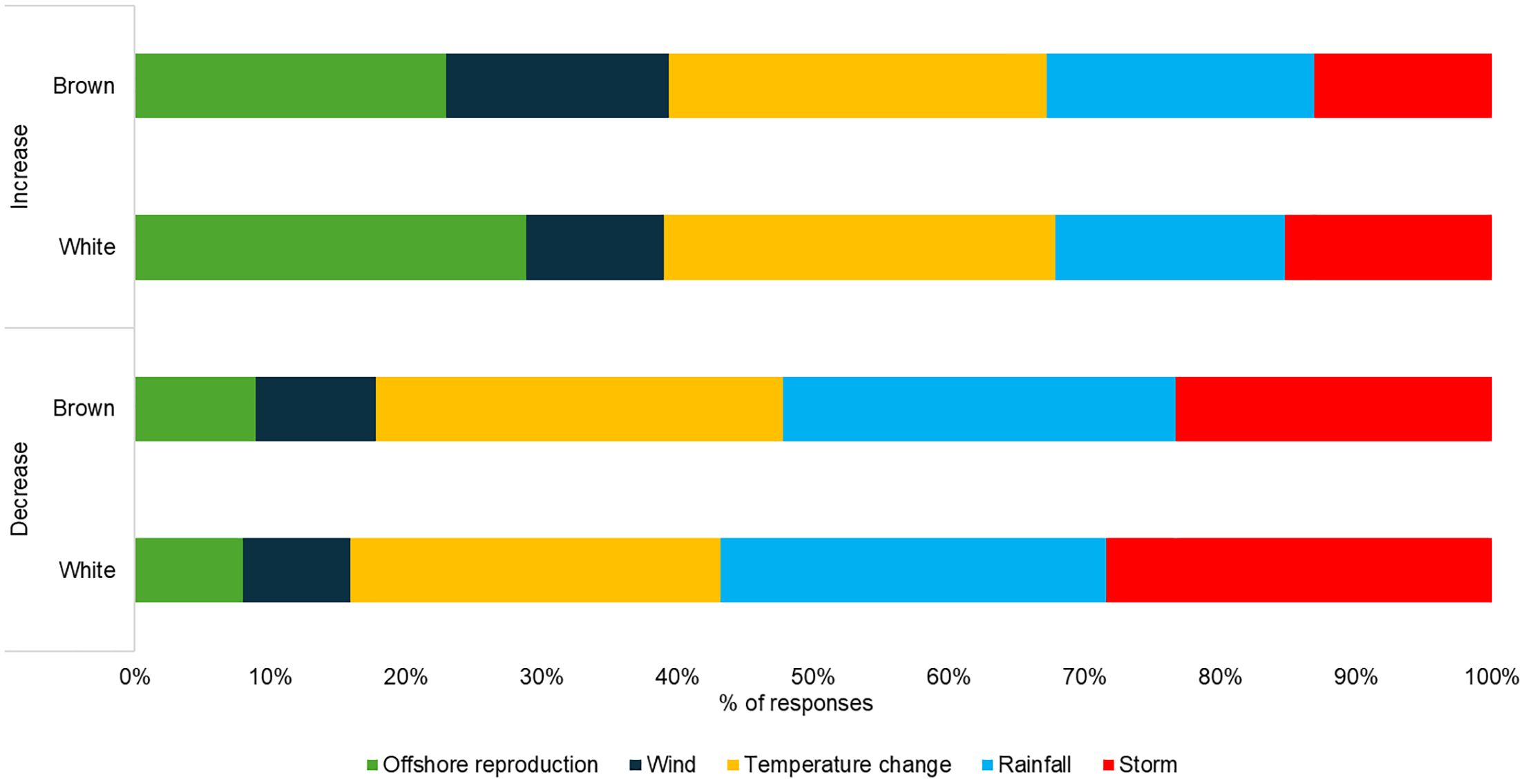
Figure 3. Drivers of change in the abundance of white and brown shrimp based on LEK (% of all responses).
The comparison of environmental drivers from empirical models and fishers’ observations showed that agreement differed among variables and between species. We find that the largest discrepancies in observed drivers and modeling outputs were related to the influence of storms on white shrimp populations, as storms were seen by fishers to mainly reduce white shrimp abundance while the model outputs indicated a positive effect on white shrimp populations. Thus, while both approaches indicated that storms were an important variable, the direction of importance was only in agreement between LEK and empirical modeling approaches for brown shrimp, where storms had a negative impact on abundance. In addition, the influence of temperature on brown shrimp was different across the two types of data as fishers observed temperature changes to have a negative effect on brown shrimp populations whereas the model showed a positive relationship with temperature (Figure 4, Table 3).
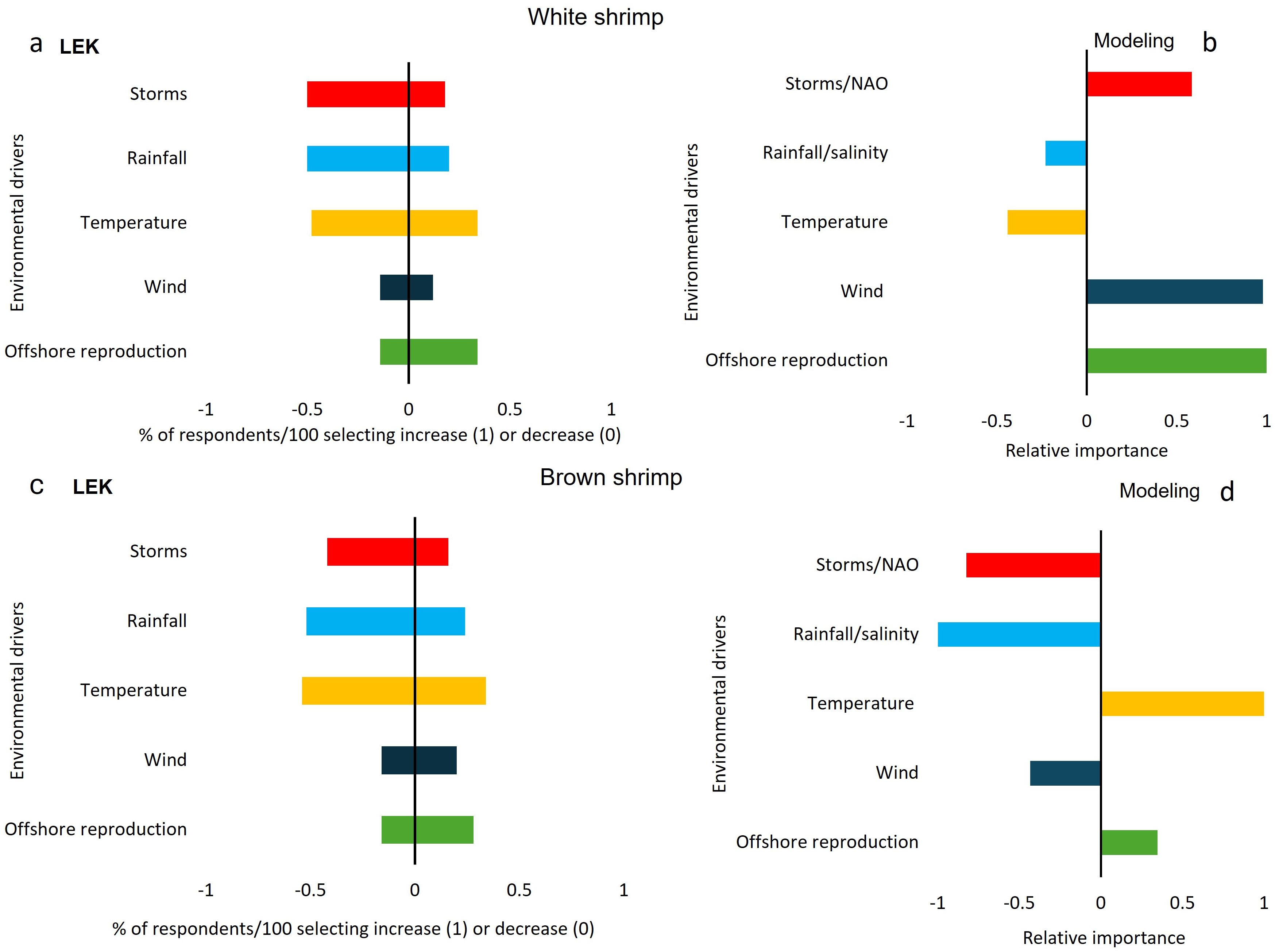
Figure 4. Drivers of change in white shrimp abundance based on LEK where values indicate proportion of respondents that selected each driver (a) and based on empirical models where values indicate the relative importance of a given variable (b). Drivers of change in brown shrimp abundance where values indicate proportion of respondents that selected each driver (c) and based on empirical models where values indicate importance of a given variable (d). The direction (+/-) of a response indicates if a variable had a positive or negative effect on shrimp abundance.
For both species, the two data sets agreed on the negative impact of rainfall on shrimp abundance, although the magnitude of the effect was not as great for empirical modeling of white shrimp compared to LEK (Figure 4). Results of LEK data for temperature were ambiguous, with similar amounts of respondents choosing negative and positive impacts, while empirical modeling showed a moderate negative relationship with temperature for white shrimp and a strongly positive relationship for brown shrimp.
Differences between the approaches occurred for wind, which was a strong predictor for an increase in white shrimp abundance and had a moderate negative effect for brown shrimp in empirical models but was not frequently indicated to affect abundance according to fisher observations (Figure 4, Table 3). Finally, the abundance of reproducing adult shrimp in offshore areas was indicated as having a positive effect for both species and methods. However, for white shrimp, empirical modeling indicated this variable as being the most important, while it was only of moderate importance for LEK (Figure 4).
Qualitative responses to open-ended questions provided some additional insights into fishers’ observations (Figure 5). Fishers mentioned that fluctuations in temperature cause spatial and temporal changes in shrimp distribution. For example, temperature changes were seen as causing shrimp to move out to sea faster and shifting the shrimp season from October/November to late fall until February. In addition, fishers note spatial changes in shrimp abundance and the fact that shrimp can now be caught further north than in previous years as shrimp migrate north with higher temperatures. Comments further mentioned that an increase in temperature increases the abundance of white shrimp but reduces the abundance of brown shrimp. As for rain, fishers mention that rain contributes to pollution through runoff but also changes salinity in near-shore areas that leads to shrimp migration away from areas with too much freshwater. A similar effect was mentioned for storms, that increased rainfall leads to greater freshwater runoff and subsequently reduced shrimp abundance in estuaries.
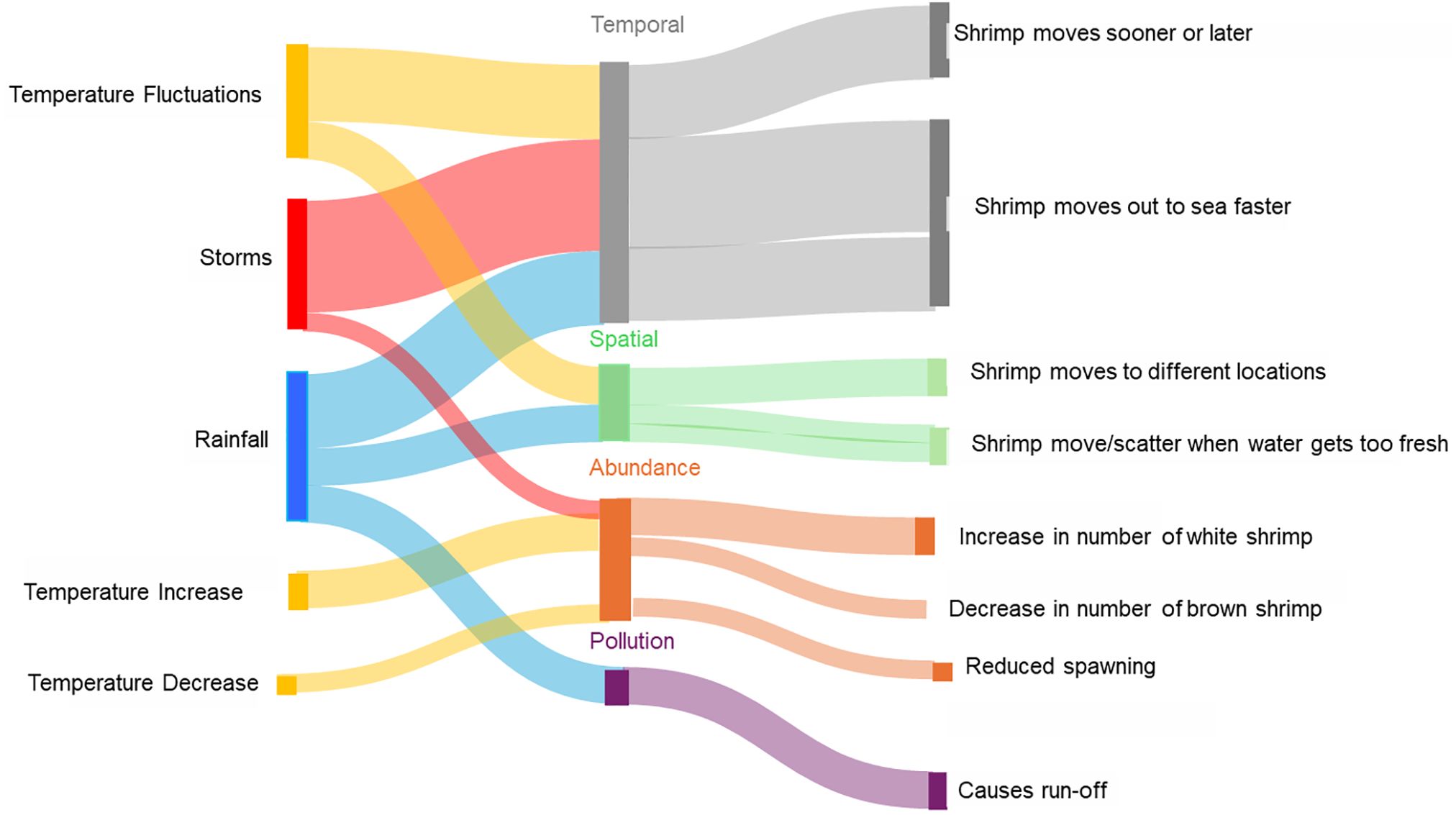
Figure 5. Sankey diagram based on fishers’ description of environmental drivers and their impacts on shrimp populations in terms of temporal and spatial changes in the distribution of shrimp and in perceived abundance.
3.4 Difference in fishers’ observations by fishing experience and livelihood dependency
We found that fishing experience in terms of the number of years fishing correlated significantly and positively with linking temperature and rain as a driver for an increase in brown shrimp and temperature and storms as an increase in white shrimp populations (Table 4). While these results match modeling outputs for temperature as a driver for an increase in brown shrimp and storms for white shrimp, the positive influence of rainfall on brown shrimp populations and temperature on white shrimp did not match findings in the model. Livelihood dependency was significantly and positively correlated with the influence of offshore spawning on brown and white shrimp populations, which matched modeling outputs. Livelihood dependency was also the only variable that correlated significantly with drivers for a decline in shrimp population, in particular storms as a driver for a decline in brown shrimp and temperature for white shrimp, which both matched modeling outputs. Age was not significantly related to the selection of any environmental driver.
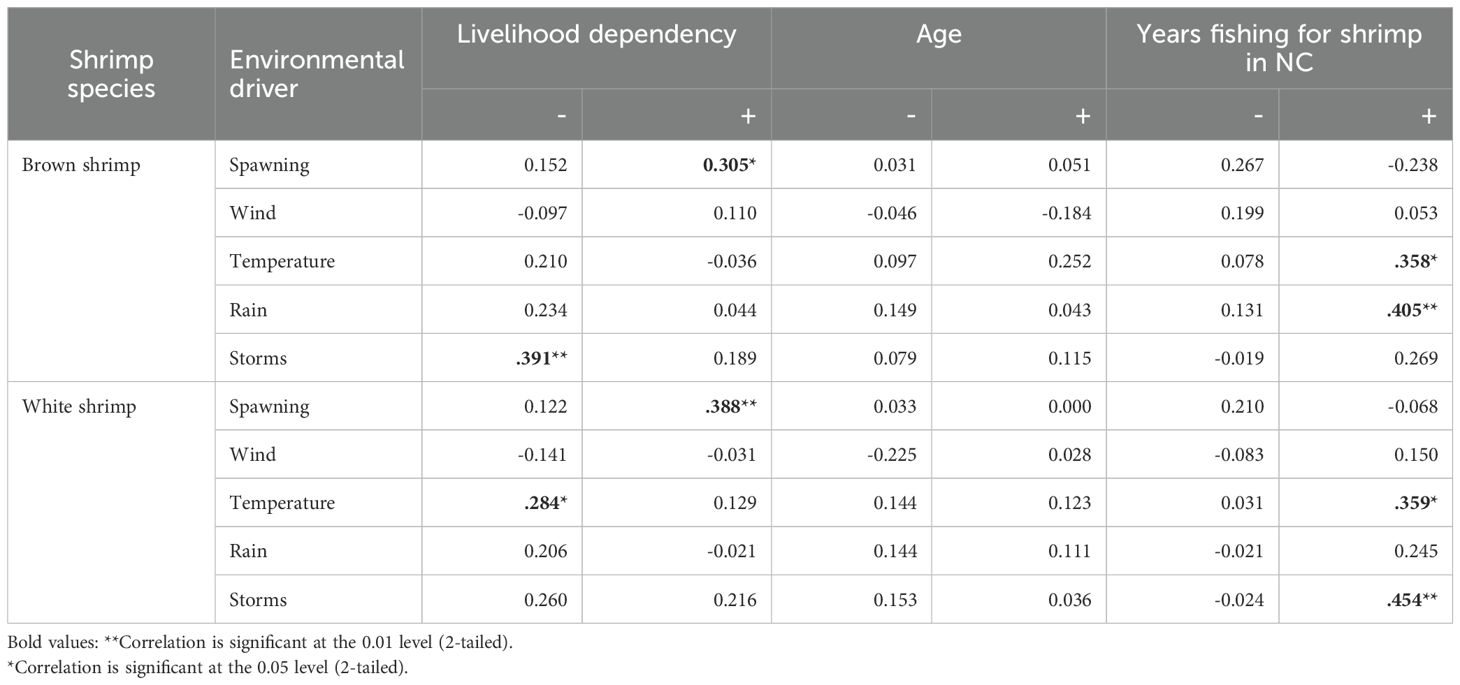
Table 4. Correlation between fishers’ observations of drivers of change and fishing patterns (Spearman correlation coefficients).
3.5 Knowledge acquisition about shrimp
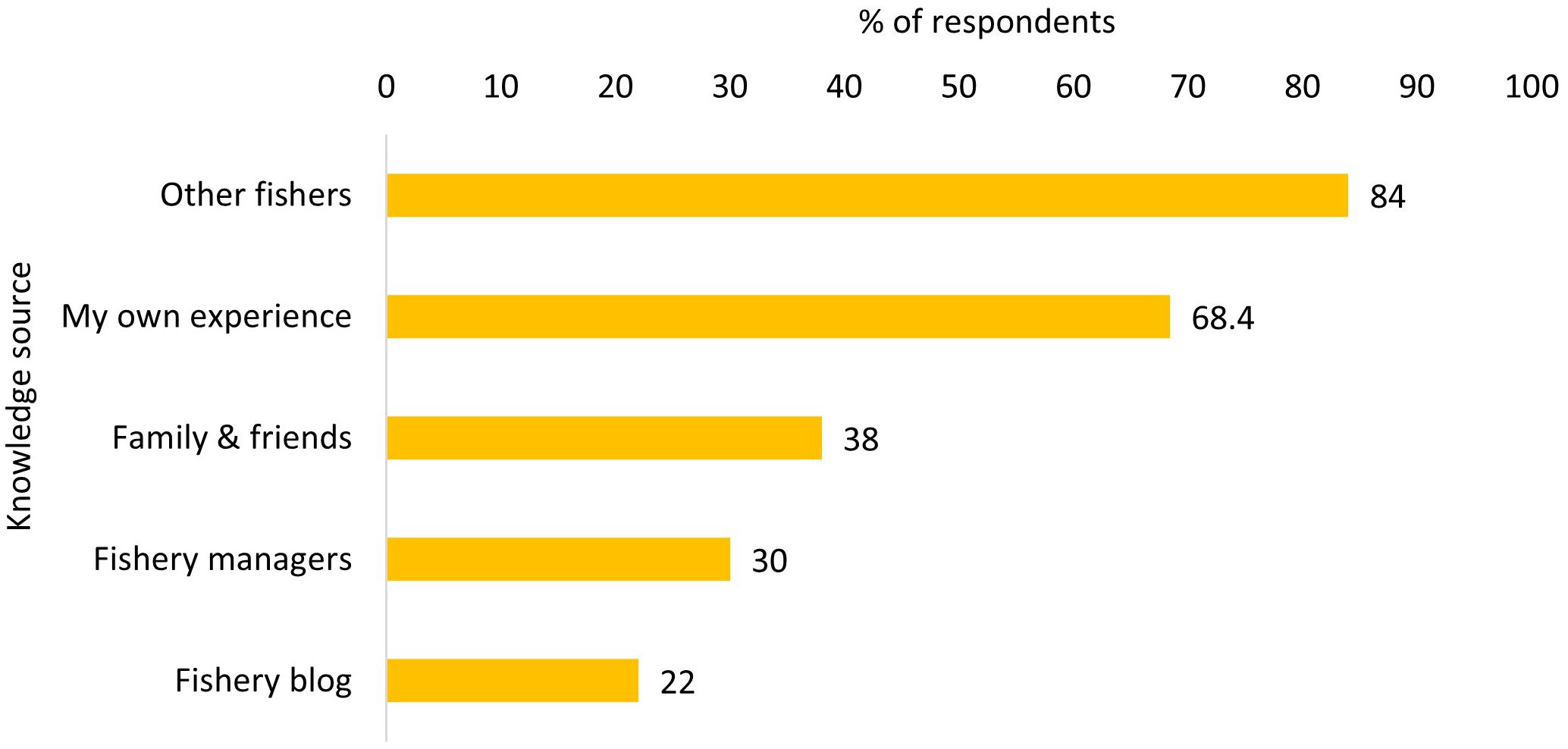
Most fishers in our study learned about shrimp through conversations with other fishers (84%) followed by their own previous experience (68.4%) (Figure 6). Fishery managers (30%) and fishery related blogs (22%) were less frequently selected as knowledge sources.
Correlation analysis revealed that the number of years engaged in the shrimp fishery, the role in the fishery, and the number of family members that are also working in the shrimp fishery influenced how knowledge was gained in the fishery (Table 5). Fishers that have been engaged in the shrimp fishery for longer and those with more family members that are also working in the shrimp fishery relied more on family and friends and less on fishery managers compared to participants that engaged in shrimp fishing for a shorter period of time and those that have fewer family members engaged in the shrimp fishery (Table 5). Finally, boat captains relied more on conversations with other fishers than non-boat captains to learn about the shrimp fishery. Livelihood dependency and the number of fisheries that participants engaged in did not correlate significantly with any knowledge acquisition source.
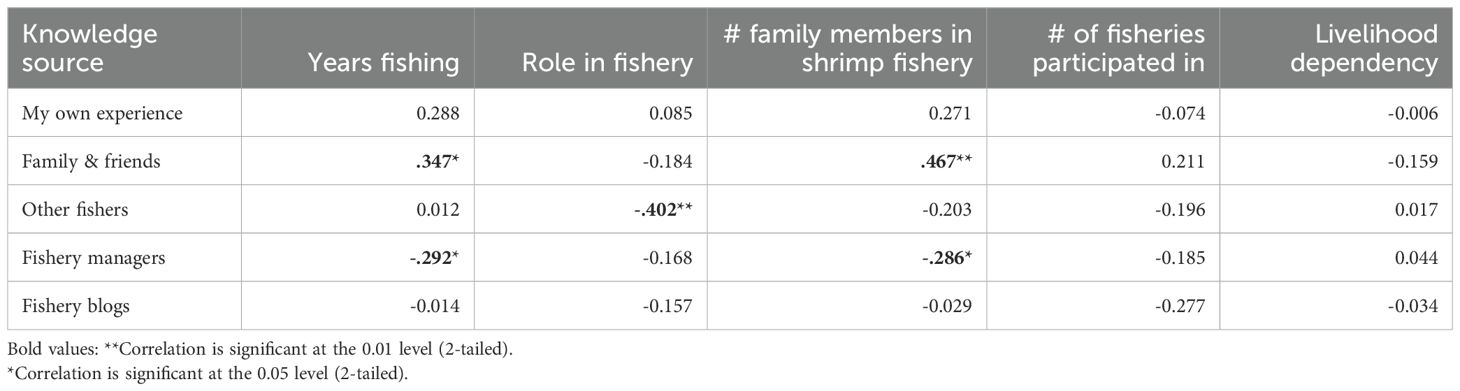
Table 5. Correlation coefficients between knowledge sources and characteristics of fisher respondents.
4 Discussion
Penaeid shrimp are highly sensitive to interannual variation in climate, due to their annual life cycle and reliance on distinct habitat types during recruitment, growth, and reproduction. The importance of environmental variables for shrimp was revealed in both the empirical models and the local ecological knowledge (LEK) from commercial shrimp fishers. The overall level of correspondence between empirical modeling results and LEK in our study was very similar between the two species, although there were differences in where matches and mismatches occurred among the variables examined.
LEK and empirical models showed similar findings for historic trends in shrimp abundance for both species. The most common response of fishers was that white shrimp were increasing in abundance, while few indicated they were decreasing. Similarly, the empirical modeling approach indicated white shrimp abundances were greater than the average for the timeseries from 2013 to 2019 (when our time series ended) in Pamlico Sound. Indeed, commercial landings of white shrimp in North Carolina have increased over time and were particularly high between 2016 and 2019 (NCDMF, 2022). The trend of increasing numbers of white shrimp is reflected across multiple states within the southeast U.S. and indicates that this species is responding positively to climate change (Kimball et al., 2020; Tuckey et al., 2021; Schlenker et al., 2023). Conversely, brown shrimp annual landings have varied greatly over time, but there has been no overall trend (NCDMF, 2022). This matches our empirical modeling and LEK results, where the majority of respondents indicated stable brown shrimp populations and estimated abundances of brown shrimp over the last portion of the timeseries were not significantly different than the timeseries mean. This broad correspondence in abundance trends for both species suggests that, even though LEK and empirical modeling are based on very different information, they can converge and can be comparable, even in the context of highly variable species such as shrimp.
The environmental variables considered to be the most important for shrimp populations based on fishers’ LEK were remarkably similar for brown and white shrimp. Fishers generally attributed negative effects on shrimp with high levels of rainfall and storms. Part of this interpretation appeared to reflect access to shrimp for fishing. For example, a common response by fishers was that high rainfall and storms push shrimp out of the estuaries sooner during the year, which leads to them dispersing into the ocean where they may occur at lower densities. Although, others implicated heavy rainfall to diminish water quality. In other systems, white and brown shrimp have also been shown to be more abundant when salinities are higher, presumably occurring when freshwater input and precipitation has been lower (Kimball et al., 2023).
Our empirical modeling results supported the LEK for storms and rainfall, with one exception. Specifically, there was a mismatch for white shrimp and storms. This disagreement between approaches may be due to the way that storms were included in the empirical modeling, namely the NAO climate index. The NAO is most often attributed to storminess during winter months, which corresponds to the brown shrimp reproductive season (Williams, 1955; Rogers et al., 1993; Visbeck et al., 2001; Chartrand and Pausata, 2020). White shrimp reproduce during spring and summer, and the relationship of this species with the NAO might simply be due to the amount of annual rainfall that occurs during different climate phases (Wenner et al., 2005; Williams, 1955). In contrast, fisher’s knowledge of the effects of storms is based on more precise cause-and-effect events, such as comparing catch rates before and after a tropical storm. The NAO index thus might not be an effective indicator for storms with white shrimp empirical modeling. In consideration of future research, the LEK makes clear that storms are important for shrimp population dynamics and the behavior of fishers, including where fishing is conducted. Thus, our LEK results are indicating an important area for further analysis of how storms impact shrimp population-fishery dynamics, particularly when considering how climate change is expected to intensify severe weather.
Temperature was an important variable for LEK and empirical modeling. An examination of the information provided by LEK can help explain why fishers chose temperature as having a positive and negative impact on shrimp abundance. The more detailed responses given by fishers indicated that temperature has an important impact on the spatial distribution of shrimp and on phenological patterns (e.g., annual timing of emigration from estuaries to the ocean). Further, fishers might have interpreted the temperature effect at varying scales. For example, one response suggested that increased temperatures are leading to an overall shift in shrimp to the north, which is increasing abundance in the northern North Carolina border region with Virginia. Still, others indicated that increased temperature is reducing shrimp abundance, because shrimp have a shorter residence time in the estuary. Some of the variation that we see in shrimp abundance could be due to variation in phenology, which is a challenge that has recently been recognized by management agencies (Olmos et al., 2023; Kohut et al., 2013).
The variables wind and offshore spawning were distinct among our suite of environmental drivers, because their effects are temporally displaced from the harvest. For instance, our empirical modeling used the abundance of reproductively mature white shrimp offshore during spring to predict abundance of catch occurring many months later (Schlenker et al., 2023). Similarly, wind patterns during the previous winter were used to predict white shrimp abundance the following year, because winds at this time probably indicate the severity of winter experienced by adult shrimp that will spawn the following year. LEK did not frequently indicate these two variables as affecting shrimp abundance, even though they are important variables in the empirical modeling. Thus, it might be more difficult for fishers to associate variation in catch to conditions occurring months earlier, compared to observations of environmental dynamics during the harvest season. We conclude that being able to examine lagged environmental drivers is a strength of empirical models, while LEK might attribute annual variation to more proximal drivers.
Our study further finds that the length of time spent in the shrimp fishery was related to perceived trends in the fishery, which was also found in previous studies on LEK (Shephard et al., 2021). As these observations matched our modeling data, our study suggests that LEK can provide valuable insights into long-term trends that are also observed using different types of information. In addition, we find that years spent in the fishery were also related to the selection of specific environmental drivers, in particular temperature, rainfall and storms. Thus, experience in a fishery might enable fishers to detect the impacts of environmental drivers that might change more slowly, such as temperature, or are more episodic, such as storms.
We found that most fishers in our study learned about shrimp abundance through other fishers and from their own previous experience. This finding is different from previous studies that found that the sharing of knowledge and information among fishers is uncommon (Ames, 2004) due to the competitive nature of fishing and a lack of trust among commercial fishers (Calderwood et al., 2021; Haskell et al., 2019). The literature on information sharing, however, is mixed and recent studies have found a that commercial fishers are more willing to engage in information sharing networks among fishers with similar fishing success (Turner et al., 2014). In addition, previous studies found that sharing information about resource dynamics can enhance fishing productivity in local, trusted networks among fishers (Barnes et al., 2017). As shrimp abundance varies in response to diverse environmental drivers, fishers in our study might rely on each other to better understand these fluctuations. Another reason for information sharing within this community might be that shrimp are an annual “crop” and overfishing is not considered a likely scenario, because a strong year class can directly follow a poor one (NCDMF, 2022). Further, the number of commercial fisher participants, and annual shrimp fishing trips, has been steadily declining in North Carolina (NCDMF, 2022), which might contribute to reduced competition among current participants. Our findings showed that there were differences in knowledge acquisition strategies based on the role that fishers played. Specifically, boat captains relied less on information provided by other fishers than non-boat captains. One explanation might be that boat captains are often hesitant to share local ecological knowledge with other fishers due to the competitive nature of fishing (Barnes et al., 2017). Boat captains thus might be less willing to discuss the fishery with other fishers compared to boat owners, crew members and other members of the fishery.
Information exchange among fishery managers and fishers can help improve management practices and regulations that enhance both social and environmental outcomes (Young et al., 2016). Yet, in our study, less than one third of fishers appeared to engage with fishery managers to learn about dynamics in the shrimp fishery. One explanation for these low numbers might be that accessible information from management is not necessarily formatted in a way that is applicable to local fishers (Nguyen et al., 2019). Another reason might be a lack of trust among fishers towards fishery managers as trust provided seems to shape information exchange in fisheries (Wade et al., 2023). In particular, fishers who have been part of the shrimp fishery for a long period of time relied more on family and friends and less on information provided by fishery managers. Thus, this group of fishers might have different relationships with fishery managers compared to fishers who just entered the shrimp fishery. Indeed, the North Carolina shrimp fishery has a long history of regulatory changes, that often has restricted access to historic fishing areas and increased gear regulation. A study by Wade et al. (2023) found that trust in information might be independent from trust in the actors (i.e., fishery managers) that provide information to boat captains and non-captains. Thus, further investigation into the reasons why shrimp fishers might not seek information provided by fishery managers in annual crop fisheries could provide further insights. Specifically, whether a lack of trust in fishery managers or the information provided by them is the main reason for a lack of communication among the two groups. A more participatory approach for the shrimp fishery could also enhance information exchange as more frequent and reciprocal interactions between different actors over time might increase the perceived trustworthiness of information (Grabner-Kräuter and Bitter, 2015) and potentially also in terms of the trustworthiness of the actor providing the information.
In terms of limitations, we found that comparing outputs of empirical models and LEK data can be challenging. Fishers’ knowledge is accumulated over many years and covers all seasons of the fishery. Empirical modeling parameters, on the other hand, cover different temporal scales, e.g., in the case of storms, which can lead to differences in the two data sets. At the same time, fishers’ observations might mainly happen during the harvest season, while modeling data can detect the influence of environmental drivers on shrimp populations outside of this range and detect lagged environmental drivers. In addition, we faced challenges due to the COVID-19 pandemic, which prohibited face-to-face survey administration during the first months of the survey distribution. Leaving the surveys at fish houses provided a good alternative with similar response rates for both the face-to-face administration and the survey at fish houses.
However, although long-term fisheries-independent data is the gold standard when it comes to building empirical models, this type of data does have limitations. For example, fisheries independent surveys are typically conducted at static timepoints. The Program 195 survey conducts their sampling in June and September of each year regardless of the environmental conditions, meaning that their surveys may not always be optimally timed to encounter peak shrimp abundances. In contrast, fishers will typically pursue shrimp until their densities decrease enough that it no longer makes economic sense to continue fishing. Additionally, although our empirical models using environmental variables to predict abundance explained large amounts of variation in the data for both brown and white shrimp (81% for both species), we are limited to assessing the relationships between variables that have been measured over long time series in close proximity to the Pamlico Sound. While some fishers highlighted the importance of water quality in their comments, we were not able to examine the relationship between water quality and shrimp abundance due to a lack of long-term data sets in the region that collect that type of data.
5 Conclusions
Fisheries independent data, which was used for our empirical modeling, is typically collected with the same methods, including survey timing, each year. While this format of data collection is important for examining time series of data, challenges occur when there are shifts in the ecosystem that have direct impacts on the timing of migrations in and out of the survey area. Indeed, the LEK results suggest that shrimp populations might vary in how they overlap with the fisheries independent survey conducted in the Pamlico Sound, based on variability in temperature, rainfall and storms. Further, based on our survey findings, at least some of the fishers recognize that climate change is shifting the dynamics of the shrimp fishery in this region, which is supported by fisheries independent data and the expansion of the shrimp fishery (Schlenker et al., 2023; Tuckey et al., 2021; Kimball et al., 2020). With climate change having the potential to rapidly alter temperature and salinity regimes in estuaries, it will be important to consider how shifts in the population dynamics of marine organisms might impact routine biological monitoring. Specific to North Carolina, NC DMF is currently (as of 2025) considering ways to modify the Program 195 survey of Pamlico Sound, which was used for our empirical modeling (pers. comm. NC DMF staff, May 2025). A major consideration in redesigning a survey is to determine how best to align the timing of sampling with the changing phenology of resource species like penaeid shrimp, which was suggested by both our LEK and empirical modeling results. Further, from an analysis perspective, our results suggest the importance of using environmental covariates when examining abundance trends from routine survey data, such as Program 195. For example, years with higher temperatures in the spring and summer, or rainfall events during the fall, can lead to a more rapid life cycle or phenology of penaeid shrimp.
Our findings indicate the value of both fishers’ LEK and empirical model outputs for fisheries management and to support policy decisions (Bevilacqua et al., 2016; Lopes et al., 2019). While empirical modeling outputs provided critical insights into lagged environmental drivers, LEK provided more insights into environmental drivers that might fall out of the temporal range of model input parameters. Therefore, while both LEK and CSK can be critical to gain deeper insights into the environmental drivers of a fishery, it is also essential to consider the strengths and limitations of both types of data. Additional studies that compare LEK with empirical modeling are needed. Some of the differences in our comparison study reveal challenges with this type of research. For example, some variables might be considered at multiple spatial or temporal scales with LEK, while empirical modeling is very precise in how variables are defined. Often empirical modeling approaches are constrained in scope, based on the availability of appropriate environmental data to explain population dynamics. Therefore, future research making such comparisons should consider these constraints when collecting LEK data to best match it with empirical modeling results. At the same time, the collection of LEK should allow for opportunities to reveal new observations from fishers that might inform future population modeling efforts and data collection efforts. Some of these new efforts could involve cooperative data collection between researchers and fishers, which might improve communication between the fishery and managers (Johnson and van Densen, 2007).
Our LEK and empirical modeling analysis of the shrimp fishery has demonstrated that the dynamics of this socio-ecological system are complex and changing over time. It will be important to monitor these changes within the context of the many potential contributing factors, including environmental forces, changes in the fished populations (i.e., shifting penaeid shrimp species), and the characteristics of the fishing community. Such efforts are common at the federal level (Monaco et al., 2021) and have also been suggested for the state of North Carolina, where social and ecological indicators have consistently changed over multiple decades (North Carolina Collaboratory, 2025). Further, enhanced communication and coordination between stakeholders in the shrimp fishery and management might be especially important in North Carolina. The commercial shrimp fishery in this region has recently been challenged by the State Legislature, with an attempt to remove shrimp trawling from estuarine areas in North Carolina over concerns about potential impacts to other fisheries (North Carolina Collaboratory, 2025). Effective interactions between fishery stakeholders and managers or researchers can better inform science-based management decisions, which are critical when different stakeholder groups in fisheries are in opposition (Goethel et al., 2023).
Data availability statement
The raw data supporting the modeling outputs of this article will be made available by the authors, without undue reservation.
Ethics statement
The study involving humans was approved by the University and Medical Center Institutional Review Board, East Carolina University UMCIRB 20-002189.
Author contributions
NH: Conceptualization, Data curation, Formal Analysis, Funding acquisition, Methodology, Project administration, Supervision, Visualization, Writing – original draft, Writing – review & editing. LS: Data curation, Formal Analysis, Methodology, Validation, Visualization, Writing – original draft, Writing – review & editing. SF: Data curation, Writing – review & editing, Investigation, Methodology, Visualization, Formal Analysis, Writing – original draft. JM: Conceptualization, Funding acquisition, Investigation, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. LS received support from North Carolina Sea Grant (Project Number: NCSG-RM-02).
Acknowledgments
We thank Chris Stewart and Jason Rock from NCDMF for helpful advice regarding the development of the questionnaire.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmars.2025.1589076/full#supplementary-material
References
Ainsworth C. H. and Pitcher T. J. (2005). “Using local ecological knowledge in ecosystem models,” in Fisheries assessment and management in data-limited situations (Alaska Sea Grant College Program, University of Alaska Fairbanks, Fairbanks, Alaska, USA), 289–304.
Ames T. (2004). “Putting fishers’ knowledge to work,” in Fishers’ Knowledge in Fisheries Science and Management. Eds. Haggan N., Neis B., and Baird I. G. (Paris, France: UNESCO Publishing), 353–363.
Azzurro E., Sbragaglia V., Cerri J., Bariche M., Bolognini L., Souissi J. B., et al. (2019). Climate change, biological invasions, and the shifting distribution of Mediterranean fishes: A large-scale survey based on local ecological knowledge. Global Change Biol. 25, 2779–2792. doi: 10.1111/gcb.14670
Barnes M. L., Arita S., Kalberg K., and Leung P. (2017). When does it pay to cooperate? Strategic information exchange in the harvest of common-pool fishery resources. Ecol. Economics 131, 1–11. doi: 10.1016/j.ecolecon.2016.08.005
Begossi A., Lopes P., and Silvano R. (2012). Co-management of reef fisheries of the snapper-grouper complex in a human ecological context in Brazil. Global Prog. ecosystem-based fisheries Manage., 1–22. doi: 10.4027/gpebfm.2012.018
Bell R. J., Wood A., Hare J., Richardson D., Manderson J., and Miller T. (2018). Rebuilding in the face of climate change. Can. J. Fisheries Aquat. Sci. 75, 1405–1414. doi: 10.1139/cjfas-2017-0085
Bender M. G., MaChado G. R., de Azevedo Silva P. J., Floeter S. R., Monteiro-Netto C., Luiz O. J., et al. (2014). Local ecological knowledge and scientific data reveal overexploitation by multigear artisanal fisheries in the Southwestern Atlantic. PloS One 9, e110332. doi: 10.1371/journal.pone.0110332
Berkes F. (2002). Back to the future: ecosystem dynamics and local knowledge. Understanding Transformation in Human and Natural Systems.
Berkström C., Papadopoulos M., Jiddawi N. S., and Nordlund L. M. (2019). Fishers’ local ecological knowledge (LEK) on connectivity and seascape managemen. Front. Mar. Sci. 6, 130. doi: 10.3389/fmars.2019.00130
Bevilacqua A. H. V., Carvalho A. R., Angelini R., and Christensen V. (2016). More than anecdotes: fishers’ ecological knowledge can fill gaps for ecosystem modeling. PloS One 11, e0155655. doi: 10.1371/journal.pone.0155655
Blasiak R., Spijkers J., Tokunaga K., Pittman J., Yagi N., and Österblom H. (2017). Climate change and marine fisheries: Least developed countries top global index of vulnerability. PloS One 12, e0179632. doi: 10.1371/journal.pone.0179632
Burnham K. P. and Anderson D. R. (2002). Model selection and multimodel inference: a practical information-theoretic approach (New York: Spnnger-Veflag, New York).
Calderwood J., Pedreschi D., and Reid D. G. (2021). Technical and tactical measures to reduce unwanted catches in mixed fisheries: do the opinions of irish fishers align with management advice? Mar. Policy 123, 104290. doi: 10.1016/j.marpol.2020.104290
Chartrand J. and Pausata F. S. R. (2020). Impacts of the North Atlantic Oscillation on winter precipitations and storm track variability in southeast Canada and the northeast United States. Weather Climate Dynamics 1, 731–744. doi: 10.5194/wcd-1-731-2020
Cook C. N., Wardell-Johnson G., Carter R. W., and Hockings M. (2014). How accurate is the local ecological knowledge of protected area practitioners? Ecol. Soc. 19 (2).
Dlamini V., Samson M., Curtis C., and O’Brien G. (2022). Co-opting small-scale fishers’ knowledge with scientific data to improve understanding of fish community structures in the Lower Komati River, South Africa. Mar. Freshw. Res. 74, 111–124. doi: 10.1071/mf22169
Durham C. L., Eggleston D. B., and Nail A. J. (2019). “Process-based statistical models predict dynamic estuarine salinity,” in Lagoon Environments Around the World—A Scientific Perspective, (IntechOpen).
Ehrlén J. and Morris W. F. (2015). Predicting changes in the distribution and abundance of species under environmental change. Ecol. Lett. 18, 303–314. doi: 10.1111/ele.12410
Ezer T. and Atkinson L. P. (2014). Accelerated flooding along the US East Coast: On the impact of sea-level rise, tides, storms, the Gulf Stream, and the North Atlantic Oscillations. Earth’s Future 2, 362–382. doi: 10.1002/2014EF000252
Gaspare L., Bryceson I., and Kulindwa K. (2015). Complementarity of fishers’ traditional ecological knowledge and conventional science: Contributions to the management of groupers (Epinephelinae) fisheries around Mafia Island, Tanzania. Ocean Coast. Manage. 114, 88–101. doi: 10.1016/j.ocecoaman.2015.06.011
Goethel D. R., Omori K. L., Punt AndréE., Lynch P. D., Berger A. M., de Moor C. L., et al. (2023). Oceans of plenty? Challenges, advancements, and future directions for the provision of evidence-based fisheries management advice. Rev. Fish Biol. Fisheries 33, 375–410. doi: 10.1007/s11160-022-09726-7
Grabner-Kräuter S. and Bitter S. (2015). Trust in online social networks: A multifaceted perspective. Forum Soc. economics. doi: 10.1080/07360932.2013.781517
Grant S. and Berkes F. (2007). Fisher knowledge as expert system: A case from the longline fishery of Grenada, the Eastern Caribbean. Fisheries Research 84 (2), 162–170.
Hamilton R., de Mitcheson Y. S., and Aguilar-Perera A. (2011). “The role of local ecological knowledge in the conservation and management of reef fish spawning aggregations,” in Reef fish spawning aggregations: biology, research and management (Dordrecht: Springer Netherlands), 331–369.
Hare J. A., Alexander M. A., Fogarty M. J., Williams E. H., and Scott J. D. (2010). Forecasting the dynamics of a coastal fishery species using a coupled climate–population model. Ecol. Appl. 20, 452–464. doi: 10.1890/08-1863.1
Haskell N., Mamula A., and Collier T. C. (2019). Competition or cooperation? Peer effects in the Pacific coast groundfish fishery. Land Economics 95, 258–278. doi: 10.3368/le.95.2.258
Hauck M., Sowman M., Russell E., Clark B. M., Harris J. M., Venter A., et al. (2002). Perceptions of subsistence and informal fishers in South Africa regarding the management of living marine resources. Afr. J. Mar. Sci. 24, 463–474. doi: 10.2989/025776102784528556
Heck N., Beck M. W., Reguero B., Pfliegner K., Ricker M., and Prütz R. (2023). Global climate change risk to fisheries–A multi-risk assessment. Mar. Policy 148, 105404. doi: 10.1016/j.marpol.2022.105404
Heck N., Stedman R. C., and Gaden M. (2015). The integration of social science information into Great Lakes fishery management: Opportunities and challenges. Fisheries Res. 167, 30–37. doi: 10.1016/j.fishres.2015.01.008
Hind E. J. (2015). A review of the past, the present, and the future of fishers’ knowledge research: a challenge to established fisheries science. ICES J. Mar. Sci. 72, 341–358. doi: 10.1093/icesjms/fsu169
Hurrell J. W. (1995). Decadal trends in the North Atlantic Oscillation: Regional temperatures and precipitation. Science 269, 676–679. doi: 10.1126/science.269.5224.676
Johannes R. E. (1981). Working with fishermen to improve coastal tropical fisheries and resource management. Bull. Mar. Sci. 31, 673–680.
Johannes R. E., Freeman M. M. R., and Hamilton R. J. (2000). Ignore fishers’ knowledge and miss the boat. Fish Fisheries 1, 257–271. doi: 10.1046/j.1467-2979.2000.00019.x
Johnson T. R. and van Densen W. L. T. (2007). Benefits and organization of cooperative research for fisheries management. ICES J. Mar. Sci. 64, 834–840. doi: 10.1093/icesjms/fsm014
Karp M. A., Peterson J. O., Lynch P. D., Griffis R. B., Adams C. F., Arnold W. S., et al. (2019). Accounting for shifting distributions and changing productivity in the development of scientific advice for fishery management. ICES J. Mar. Sci. 76, 1305–1315. doi: 10.1093/icesjms/fsz048
Kimball M. E., Allen D. M., Kenny P. D., and Ogburn-Matthews V. (2020). Decadal-scale changes in subtidal nekton assemblages in a warm-temperate estuary. Estuaries Coasts 43, 927–939. doi: 10.1007/s12237-019-00692-1
Kimball M. E., Pfirrmann B. W., Allen D. M., Ogburn-Matthews V., and Kenny P. D. (2023). Intertidal creek pool nekton assemblages: Long-term patterns in diversity and abundance in a warm-temperate estuary. Estuaries Coasts 46 (3), 860–877.
Kohut J., Palamara L., Curchitser E., Manderson J., DiDomenico G., and Oliver M. J. (2013). “Toward dynamic marine spatial planning tools: An example with butterfish assessment in the mid-atlantic bight,” in 2013 MTS/IEEE OCEANS-Bergen, (Bergen, Norway: IEEE), 10–14 June 2013.
Lopes P. F. M., Verba JúliaT., Begossi A., and Pennino M. G. (2019). Predicting species distribution from fishers’ local ecological knowledge: a new alternative for data-poor management. Can. J. Fisheries Aquat. Sci. 76, 1423–1431. doi: 10.1139/cjfas-2018-0148
Mackinson S. (2001). Integrating local and scientific knowledge: an example in fisheries science. Environ. Manage. 27, 533–545. doi: 10.1007/s0026702366
Márcia Barbosa A., Real R., Muñoz A-R., and Brown J. A. (2013). New measures for assessing model equilibrium and prediction mismatch in species distribution models. Diversity Distributions 19, 1333–1338. doi: 10.1111/ddi.12100
Monaco M. E., Spooner E., Oakes S. A., Harvey C. J., and Kelble C. R. (2021). Introduction to the NOAA Integrated Ecosystem Assessment Program: Advancing Ecosystem Based Management. Coast. Manag. 49 (1), 1–8. doi: 10.1080/08920753.2021.1846109
Morales E. M. Q., Lepofsky D., and Berkes F. (2017). Ethnobiology and fisheries: Learning from the past for the present. J. Ethnobiol. 37 (3), 369–379.
Morley J. W., Batt R. D., and Pinsky M. L. (2017). Marine assemblages respond rapidly to winter climate variability. Global Change Biol. 23, 2590–2601. doi: 10.1111/gcb.13578
Murray G., Neis B., and Johnsen J. P. (2006). Lessons learned from reconstructing interactions between local ecological knowledge, fisheries science, and fisheries management in the commercial fisheries of Newfoundland and Labrador, Canada. Human Ecology 34 (4), 549–571.
NCDMF, North Carolina Division of Marine Fisheries (2022). North Carolina Shrimp Fishery Management Plan, Amendment 2 (Morehead City, NC: North Carolina Division of Marine Fisheries).
Nguyen V. M., Young N., Corriveau M., Hinch S. G., and Cooke S. J. (2019). What is “usable” knowledge? Perceived barriers for integrating new knowledge into management of an iconic Canadian fishery. Can. J. Fisheries Aquat. Sci. 76, 463–474. doi: 10.1139/cjfas-2017-0305
North Carolina Collaboratory (2025). The status of North Carolina’s marine fisheries and coastal habitats – with recommendations to enhance marine public trust resources and management outcomes.
Olmos M., Ianelli J., Ciannelli L., Spies I., McGilliard C. R., and Thorson J. T. (2023). Estimating climate-driven phenology shifts and survey availability using fishery-dependent data. Prog. Oceanography 215, 103035. doi: 10.1016/j.pocean.2023.103035
Pankhurst N. W. and Munday P. L. (2011). Effects of climate change on fish reproduction and early life history stages. Mar. Freshw. Res. 62, 1015–1026. doi: 10.1071/MF10269
Perry A. L., Low P. J., Ellis J. R., and Reynolds J. D. (2005). Climate change and distribution shifts in marine fishes. Science 308, 1912–1915. doi: 10.1126/science.1111322
Pinsky M. L., Reygondeau G., Caddell R., Palacios-Abrantes J., Spijkers J., and Cheung W. W. L. (2018). Preparing ocean governance for species on the move. Science 360 (6394), 1189–1191. doi: 10.1126/science.aat2360
Ramires M., Clauzet M., Barrella W., Rotundo M. M., Silvano R. A. M., and Begossi A. (2015). Fishers’ knowledge about fish trophic interactions in the southeastern Brazilian coast. J. ethnobiology ethnomedicine 11, 1–11. doi: 10.1186/s13002-015-0012-8
Roedel P. M. (1979). Scientific assistance to Caribbean fisheries. Gulf Carib. Fish. Instit 31, 66–69.
Rogers B. D., Shaw R. F., Herke W. H., and Blanchet R.H. (1993). Recruitment of postlarval and juvenile brown shrimp (Penaeus aztecus Ives) from offshore to estuarine waters of the northwestern Gulf of Mexico. Estuarine Coast. Shelf Sci. 36, 377–394. doi: 10.1006/ecss.1993.1023
Rosa R., Carvalho A. R., and Angelini R. (2014). Integrating fishermen knowledge and scientific analysis to assess changes in fish diversity and food web structure. Ocean Coast. Manage. 102, 258–268. doi: 10.1016/j.ocecoaman.2014.10.004
Schlenker L. S., Stewart C., Rock J., Heck N., and Morley J. W. (2023). Environmental and climate variability drive population size of annual penaeid shrimp in a large lagoonal estuary. PloS One 18, e0285498. doi: 10.1371/journal.pone.0285498
Shephard S., Ryan D., O’Reilly P., and Roche W. (2021). Using local ecological knowledge to inform semi-quantitative fishery surveillance indicators: an example in marine recreational angling. ICES J. Mar. Sci. 78, 3805–3816. doi: 10.1093/icesjms/fsab234
Silas M. O., Semba M. L., Mgeleka S. S., Van Well L., Linderholm H. W., and Gullström M.. (2023). Using fishers’ local ecological knowledge for management of small-scale fisheries in data-poor regions: Comparing seasonal interview and field observation records in East Africa. Fisheries Research 264, 106721.
Silvano R. A. M. and Valbo-Jørgensen J. (2008). Beyond fishermen’s tales: contributions of fishers’ local ecological knowledge to fish ecology and fisheries management. Environment Dev. sustainability 10, 657–675. doi: 10.1007/s10668-008-9149-0
Silvano R. A. and Begossi A. (2012). Fishermen's local ecological knowledge on Southeastern Brazilian coastal fishes: contributions to research, conservation, and management. Neotrop. Ichthyol. 10, 133–147.
Silvano R. A. M. and Hallwass G. (2020). Participatory research with fishers to improve knowledge on small-scale fisheries in tropical rivers. Sustainability 12 (11), 4487.
Sowman M., Sunde J., Raemaekers S., and Schultz O. (2014). Fishing for equality: Policy for poverty alleviation for South Africa’s small-scale fisheries. Mar. Policy 46, 31–42. doi: 10.1016/j.marpol.2013.12.005
Stephenson R. L., Paul S., Pastoors M. A., Kraan M., Holm P., Wiber M., et al. (2016). Integrating fishers’ knowledge research in science and management. ICES J. Mar. Sci. 73, 1459–1465. doi: 10.1093/icesjms/fsw025
Sunday J. M., Bates A. E., and Dulvy N. K. (2012). Thermal tolerance and the global redistribution of animals. Nat. Climate Change 2, 686–690. doi: 10.1038/nclimate1539
Szuwalski C. S. and Hollowed A. B. (2016). Climate change and non-stationary population processes in fisheries management. ICES J. Mar. Sci. 73, 1297–1305. doi: 10.1093/icesjms/fsv229
Thornton T. F. and Scheer A. M. (2012). Collaborative engagement of local and traditional knowledge and science in marine environments: a review. Ecol. Soc 17 (3).
Tomasini S. and Theilade I. (2019). Local ecological knowledge indicators for wild plant management: autonomous local monitoring in Prespa, Albania. Ecol. Indic. 101, 1064–1076. doi: 10.1016/j.ecolind.2019.01.076
Tuckey T. D., Swinford J. L., Fabrizio M. C., Small H. J., and Shields J. D. (2021). Penaeid shrimp in Chesapeake Bay: population growth and black gill disease syndrome. Mar. Coast. Fisheries 13, 159–173. doi: 10.1002/mcf2.10143
Turner R. A., Polunin N. V. C., and Stead S. M. (2014). Social networks and fishers’ behavior: exploring the links between information flow and fishing success in the Northumberland lobster fishery. Ecol. Soc. 19. doi: 10.5751/es-06456-190238
Visbeck M. H., Hurrell J. W., Polvani L., and Cullen H. M. (2001). The North Atlantic Oscillation: past, present, and future. Proc. Natl. Acad. Sci. 98, 12876–12877. doi: 10.1073/pnas.231391598
Wade E., Alexander S. M., Gerkey D., and Biedenweg K. (2023). Exploring the Relationship Between Fishing Actors and Network Prominence in information-sharing Networks in Jamaican small-scale Fisheries. Hum. Ecol. 51, 877–889. doi: 10.1007/s10745-023-00444-7
Wenner E. L., Knott D. M., Barans C. A., Wilde S., Blanton J. O., and Amft J. (2005). Key factors influencing transport of white shrimp (Litopenaeus setiferus) post-larvae into the Ossabaw Sound system, Georgia, USA. Fisheries Oceanography 14, 175–194. doi: 10.1111/j.1365-2419.2005.00328.x
Williams A. B. (1955). A contribution to the life histories of commercial shrimps (Penaeidae) in North Carolina. Bull. Mar. Sci. 5, 116–146.
Young N., Nguyen V. M., Corriveau M., Cooke S. J., and Hinch S. G. (2016). Knowledge users’ perspectives and advice on how to improve knowledge exchange and mobilization in the case of a co-managed fishery. Environ. Sci. Policy 66, 170–178. doi: 10.1016/j.envsci.2016.09.002
Keywords: fisheries, environmental drivers, shrimp abundance, fishers’ observations, LEK, empirical modeling
Citation: Heck N, Schlenker LS, Farquhar SD and Morley JW (2025) Environmental drivers of shrimp abundance: comparing insights from local ecological knowledge and empirical modeling. Front. Mar. Sci. 12:1589076. doi: 10.3389/fmars.2025.1589076
Received: 06 March 2025; Accepted: 25 July 2025;
Published: 18 August 2025.
Edited by:
Lorenzo Mari, Polytechnic University of Milan, ItalyReviewed by:
Ulisses Miranda Azeiteiro, University of Aveiro, PortugalDareen Almojil, Nuwat for Environmental Research and Education, Bahrain
Ualerson Peixoto, Marine Research Institute (IMAR), Portugal
Copyright © 2025 Heck, Schlenker, Farquhar and Morley. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Nadine Heck, aGVja24xOUBlY3UuZWR1
†Present address: Lela S. Schlenker, Avantgride Power, Portland, OR, United States
 Nadine Heck
Nadine Heck Lela S. Schlenker
Lela S. Schlenker Samantha D. Farquhar
Samantha D. Farquhar James W. Morley
James W. Morley