Abstract
The red seaweed genus Gracilaria is an important source for agar extraction. Gracilaria seaweeds are mostly wild-harvested or commercially cultivated in offshore systems, exposed to fluctuating biotic and abiotic conditions which may affect their growth rates and agar yields. Land-based systems allow for better control over the cultivation conditions but at high infrastructure and operational costs. Here we show that addition of artificial illumination throughout the ambient light period up to 14 hours significantly increased the growth rates of this seaweed. However, adding 4 hours of artificial illumination to an ambient 10-hour light period, for a total of 14 hours did not increase growth rates of Gracilaria cornea grown in a land-based tank system. In addition, we introduced a water inlet spray system that created water turbulence in the growth tanks without the need for a costly air bubbling system. G. cornea grown using the water spray system grew in rates comparable to those grown aided by air bubbling. High quality agar was extracted from the seaweeds. Agar yields were similar in the light addition and the water spray experiments as compared to the controls and remained at values between 14-20% on a dry weight basis. We suggest that artificial light addition should be considered for use in land-based system, even during daytime, after a suitable cost-benefit analysis has been made. We also recommend the use of the water spray method as the preferred low-cost option for maintaining sufficient water turbulence in Gracilaria growth tanks.
Introduction
Agar is an important gelling agent used for many applications in various industries, including the pharmaceutical, food, cosmetics and sciences (El Gamal, 2010; Kammler et al., 2024). The only sources for agars are a limited number of red macroalgae (seaweeds which include seaweeds from the Gelidium, Pterocladiella and Gracilaria genera (Armisen, 1995; Pangestuti and Kim, 2015; Santos and Melo, 2018; Lomartire and Gonçalves, 2023). Currently, a considerable portion of the worldwide supply of these seaweeds comes from collection of wild seaweed stocks. This causes agar supply—for the ever-growing global demand—to fluctuate together with the availability of these wild seaweeds. Water inlet jets were formed by eds (Santos and Melo, 2018). For establishing a constant supply chain of seaweeds for the agar industries, a shift towards mariculture of agarophytic red seaweeds is required. Of all the agar producing seaweeds, the genus Gracilaria has been demonstrated to be the most successful in terms of mariculture (Friedlander et al., 1987; Levy et al., 1990; Friedlander, 2001; Pereira and Yarish, 2008). Different Gracilaria species can grow in almost every ocean or sea in the world, as some species are more adapted to cold temperatures, while others are more tropical. Gracilaria can grow vegetatively and there is no need to establish sophisticated hatchery/nursery facilities for propagation through a sexual life cycle (Bast, 2014). Small cuttings from the tips of the Gracilaria thallus can be treated for epiphyte removal and growth enhancing stimulants and grown in small volumes until planting in the field conditions (Bhagiya et al., 2025). Around 4 million tons of Gracilaria are cultured yearly worldwide (FAO, 2020). Most of the growth is conducted in offshore facilities with the seaweeds tied manually to submerges long lines, baskets, or other floating devices (Bast, 2014; Veeragurunathan et al., 2019; Shah et al., 2021). Growth in offshore conditions leave the seaweeds exposed to constant fluctuations in its abiotic and biotic surroundings. Abiotic changes include diurnal and seasonal changes in ambient light and temperature regimes, as well as other, random events such as water flow rates and turbulence, nutrient availability and water pollution outbreaks. Biotic fluctuations can include the presence of herbivore organisms, pathogenic microorganisms and viruses, and competition with epiphytic or allelopathic algae.
Light regimes and water motion are important factors influencing seaweed growth. Light is of course important as the energy source for photosynthesis (Falkowski and Raven, 2007)Water motion controls the thickness of boundary layers forms around the seaweeds so that in general, faster relative water movement against the seaweed body will result in thinner boundary layers and higher diffusion rates of nutrients and gases into and from the seaweeds (Gonen et al., 1993; Friedlander and Levy, 1995; Hurd, 2000). High nutrient exchange rates are beneficial for rapid seaweed growth.
Offshore commercial growth of seaweed relies solely on ambient sunlight regimes. Daylight length varies between seasons and the light intensity is very influenced by clouds. In-land tank cultivation of seaweeds allows for better control over the light regimes, either through covering seaweed growth tanks or pools with shading nets, or through addition of light sources. However, illumination comes at a financial cost both for the equipment purchase and for the operational cost of electricity. It is thus necessary to investigate the right balance of illumination addition to growth and agar yields.
Land-based systems also allow for control over the water motion around the seaweeds (Gonen et al., 1993; Friedlander and Levy, 1995). Currently water turbulence is created in such systems either by the use of paddle wheels or through rigorous air bubbling (Friedlander and Levy, 1995). Paddle wheels are considered efficient at water displacement, but their mechanical nature makes them prone to cause physical damage to the seaweeds (Friedlander and Levy, 1995). Air bubbling is efficient yet has considerable infrastructure and operational costs (Ben-Ari et al., 2014).
In this work we built a novel land-based tank system for seaweed cultivation. Using this system, we studied the effects of artificial light addition and of a water inlet spray system on the growth and agar yields of G. cornea towards designing a cultivation protocol with increased seaweed yields, decreased operational costs, and thus added commercial value.
Materials and methods
Growth system
The seaweed growth system was composed of six 130 liter flat bottomed polypropylene tanks (70 cm L· 50 cm W·40 cm H), fed continuously with fresh filtered (1 μm) seawater. The water exchange rate was about 5 tank volumes per hour. Constant water motion in the tanks were achieved either solely by water inlet jets (water spray) or by aeration coupled with water spray. Seawater was supplied to the tanks through a 40 cm long, 30 mm diameter, PVC tube, located in the bottom of the tanks. The tube was perforated (3 mm hole diameter) every 2 cm. This created 18 water jets which constantly mixed the water in the tank. In parallel, other tanks were supplied with optional aeration through a set of two 16 mm diameter tubes perforated every 2 cm, which were laid across the length of the growth tanks, thus forming air bubbles which constantly stirred the water in the tanks. Water height levels were determined with a 35 cm long standpipe. Preliminary studies have shown that the Gracilaria seaweeds did not grow well under direct sunlight in our growth systems (Light intensities up to 2000 μmol photons m-2 s-2 during summer months) so the tanks were placed under a shading net which lowered ambient sunlight by 40%. Three growth tanks were equipped with LED panels which provided artificial light when necessary (Figure 1A). Water temperatures were continuously monitored throughout each growth period.
Figure 1
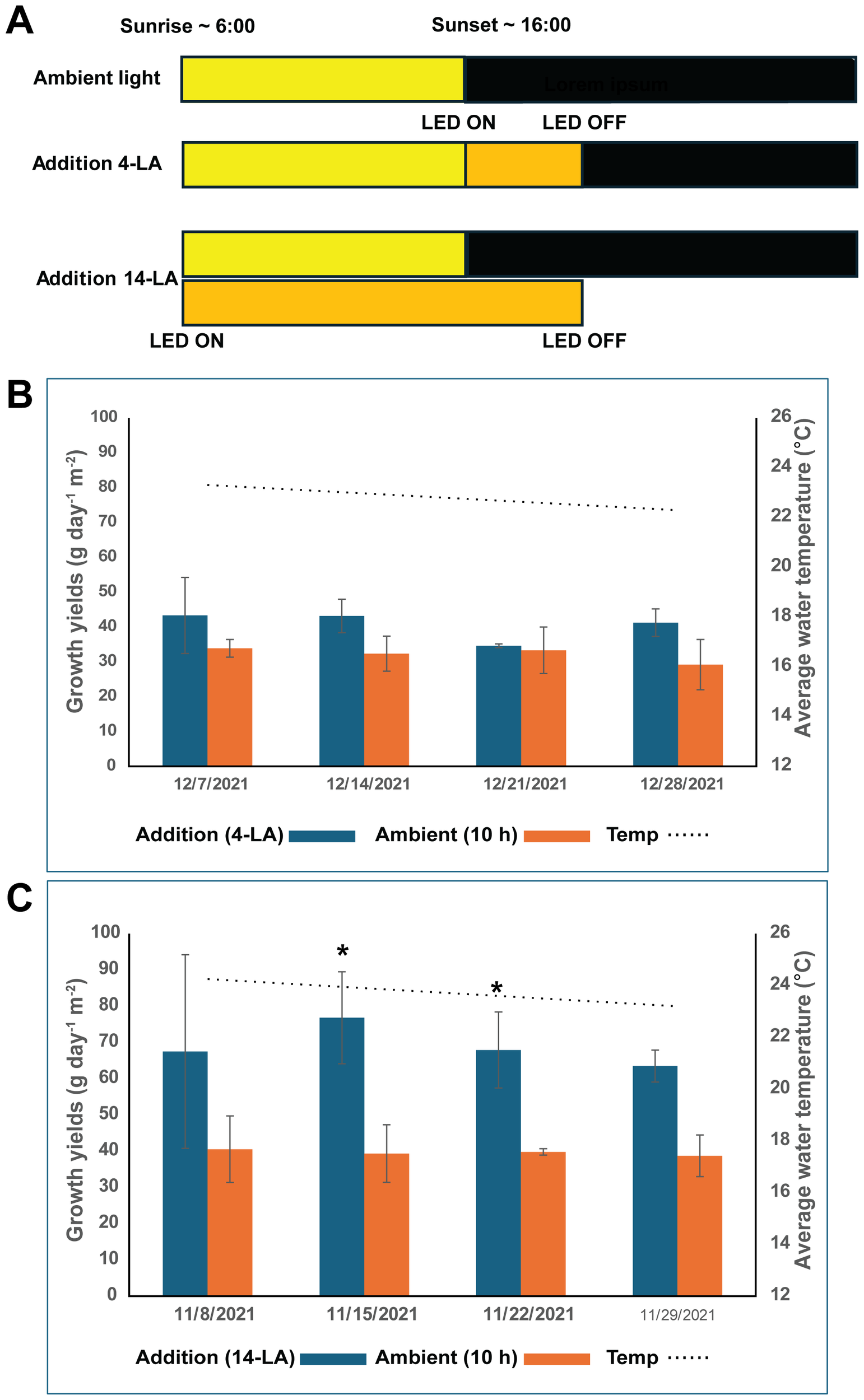
Light addition experiments. (A) light addition scheme of a 24-hour period. Yellow bars represent natural sunlight, orange bars represent artificial LED illumination, Black bars represent darkness. The points where the LEDs were turned on and off are indicated. (B) Growth yields of G. cornea with 4 hours light addition compared to ambient conditions. Water temperature is also indicated, (C) 14 hours l light addition compared to ambient conditions. Water temperature is also indicated. * marks a statistically significant (p < 0.05) difference between light addition and ambient. Error bars represent standard deviation with n=3.
Seaweed growth
Gracilaria cornea seaweeds are grown vegetatively year-round in mariculture systems at the National Center for Mariculture (NCM), Eilat, Israel, where they are used mainly as a food source for sea urchins. Thes are tropical seaweeds originally brought from Jamaica (Levy and Friedlander, 1994) but acclimated to grow vegetatively in the NCM. No sexual reproductive tissues appear in this cultivar and its assumed to be sterile, thus its life stage is unclear. Growth experiments were conducted in short term sessions of 4–5 weeks. At the beginning of each growth session, fresh, healthy-looking Gracilaria cornea seaweeds were collected. Epiphytes, if any, were gently removed by hand. The stocking density was 1 kg per square meter, or 350 grams per tank in our setup. Nutrient addition was done in weekly batches; at the start of each week ammonium and phosphate were added to a final concentration of 1 and 0.1 mM. Water flow was halted for 2 hours to allow the seaweeds to absorb the nutrients (Friedlander, 2001). The nutrient content of the fresh seaweeds was not measured. At the end of every week, seaweeds from each tank were harvested, allowed to drip dry for at least 10 minutes and then weighed. Excess seaweed material was harvested, and the seaweed were re-stocked at the original density. Daily growth yields were calculated using this formula:
Where W is the weight (grams), at the time (in days) of measurement (tn) or at the beginning of the experiment (time zero, t0). S is the surface area of the growth tanks in m2.
Experimental setup
Two distinct sets of experiments were made: Light addition and Water movement. Light addition experiments were conducted during autumn and winter months (November-December 2021), when daylight period is relatively short in Eilat—9–10 hours, as compared to 13–14 hours in the summer. Two light addition regimes were tested, and the growth rates of the seaweed were compared to G. cornea seaweeds grown without light addition. In all setups the seaweeds were exposed to ambient light regimes, which were 10 hours day light with peak intensity of ~900 μmol photons m-2 s-2 at noon (under the shading net). Artificial light was supplied by using 100W, 6000K LED arrays, which provided extra 500 μmol photons m-2 s-2 for each tank (Figure 1A). In the light addition first setup (14-LA), artificial illumination was provided throughout the daylight period, from sunrise and up to 4 hours after sunset, for a total of 14 hours, in parallel to the ambient light, which amounts to a daily quota addition of about 8.82 mol photons per tank. In the second setup (4-LA), artificial illumination was provided only after sunset for four hours, for a daily quota addition of about 2.52 mol photons per tank. Water movement in these experiments was achieved through air bubbling from the bottom of the tanks. All light regime treatments were conducted in triplicates.
Water motion experiments were performed during autumn, winter and summer times (October 2021, January-February 2022 and June-July 2022, respectively). Two methods for maintaining turbulent water motion in the seaweed growth tanks were compared, to test their effect on G. cornea growth and agar yields. In the first method (“water spray”), water movement was achieved only by the inlet water pipe; seawater was supplied through a perforated 2.5 cm PVC tube placed in the bottom of the tank. In this manner the water sprayed into the tanks, thus causing water turbulence (Figure 2A, top). For the second method (“bubbling”) water method was achieved through atmospheric bubbling which was supplied by an electric air pump to perforated pipes placed in the bottom of the tanks, coupled with the water spray. The air bubbles and spray constantly mixed the water and the seaweeds in the tank (Figure 2A, bottom). All water motion treatments were conducted in triplicates.
Figure 2
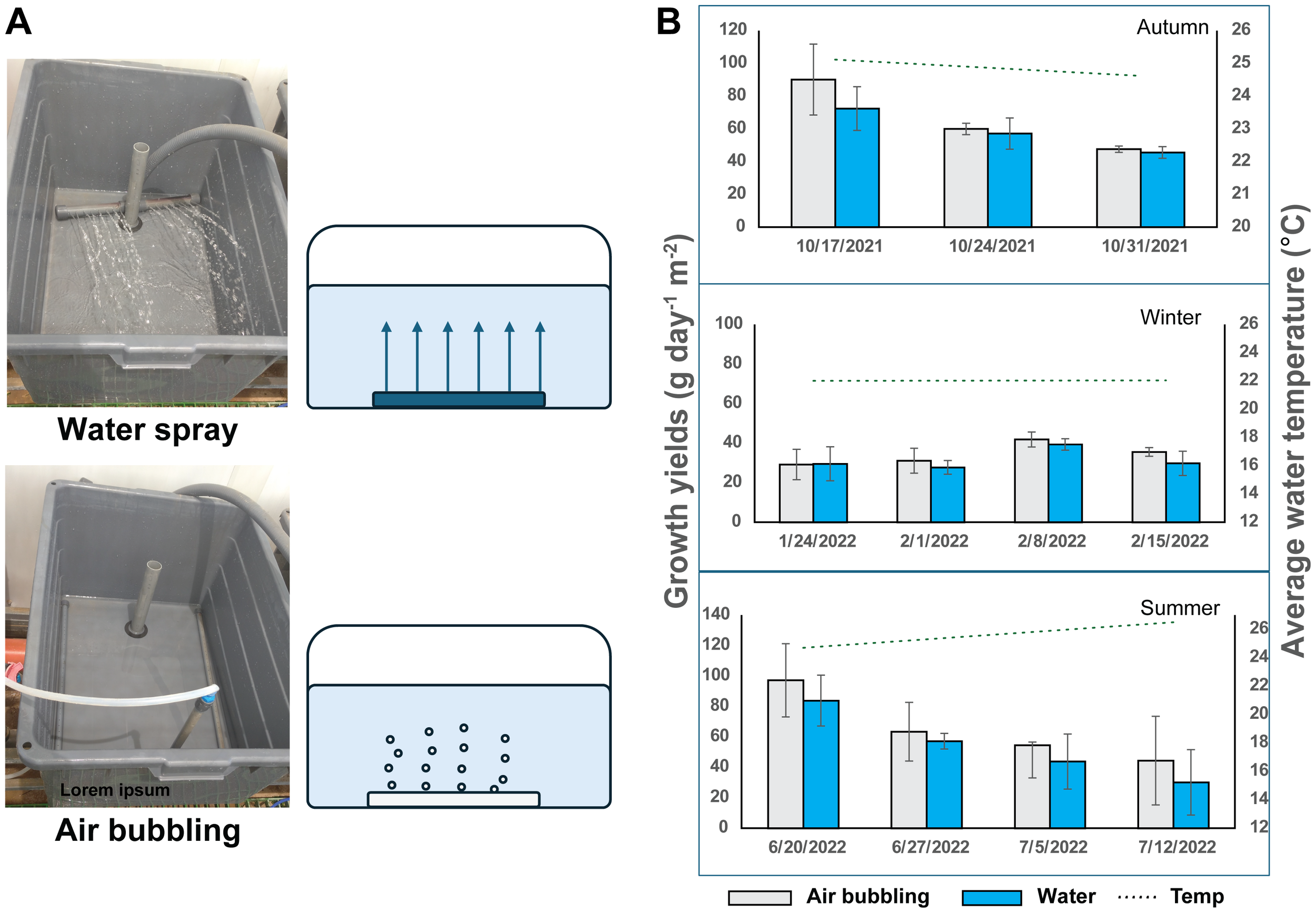
Water motion experiments. (A) water motion schemes. Top – the water spray method where the inlet water are sprayed into the tank from a perforated tube in the bottom of the tank. Bottom – the air bubbling system where water motion is caused by air bubbles pumped into the tank through a perforated tube in the bottom of the tank. (B) Growth yields of G. cornea grown with the water spray or air bubbling method, in three seasons. Water temperature is also indicated. Error bars represent standard deviation with n=3.
Agar gel properties
At the beginning of the experiment the quality of the agar extracted from G. cornea was assessed and compared to a commercial agar: BD® Bacto™ Agar (Becton, Dickinson and Company, Sparks, MD, USA). 1.5% gels were made by dissolving 1.5 g agar in 100 ml boiling distilled water. Gelling temperatures (Tg) measurements were conducted as follows: First, 3 ml of boiling agar solution were poured into transparent glass 5 ml tubes. Then, digital thermometers with a long probe were inserted into the tubes and the temperatures were constantly monitored. As the solution gradually cooled the thermometer probe was removed and re-inserted every minute. The gelling point temperature was determined as the temperature where the gel stabilized to a degree where a distinct funnel-like hole remained in the gel once the probe was taken out. For gel melting temperature (Tm) measurements glass tubes with 3 ml 1.5% gels were prepared and allowed to gel completely. A small, round weight was inserted into each tube on top of the gels. The tubes were then transferred to a water bath at room temperature and the water bath heat was turned on was turned on to war, the gels. The temperature inside the gels was constantly measured using a digital thermometer inserted into a designated gel-containing tube. The melting point was determined as the temperature in which the gels became liquid, allowing the round weights to rapidly sink to the bottom of the tube. Gel strength was measured using a homemade system devised in our lab (Shauli and Salomon, 2025). Briefly, gel strength was determined as the weight required by a flat, round, tip (4 cm2) connected to a glass cylinder filled with water to break the surface of the agar gels.
Agar yields
For agar extraction the seaweeds were sundried and ground to <1 mm particles. The dry seaweeds were cooked in hot distilled water (temperature ~ 95°C) for 2 hours, at a seaweed to water ratio of 1:50 (w/v). The hot mixture was then squeeze-filtered through a 25 mm nylon mesh to remove solids. The filtrates were poured into deep rectangular plastic trays, allowed to cool briefly and then frozen overnight at -20°C. The frozen slabs were allowed to thaw on plastic nets until most of the water dripped away and only a thick gel remained. Excess water was removed through dehydration at 60°C and the resulting crisp agar films were ground to a powder and weighed. Agar yields were calculated as the percentage of agar powder weight from the original dry weight of the seaweeds. All measurements were done in triplicates.
Statistics
Due to the small data sets in all experiments (n=3) the non-parametric Mann-Whitney U test was to evaluate significance between each sample pair, (Two-tail, α<0.05).
Results
Effect of light addition on G. cornea
The effect of artificial, white LED light addition on the growth rates of G. cornea were tested. Addition of illumination for only 4 hours during dark hours for a total light period of 14 hours (LA-4) resulted only in a small increase in weekly growth rates across four weeks of growth, that was not statistically significant (Figure 1B). In contrast, additional illumination for 14 hours, during both ambient daytime and 4 hours after sunset, almost doubled the weekly growth rates of the seaweeds compared to seaweeds grown under the ambient light regime (Figure 1C).
Effect of water motion method on G. cornea growth rates
Keeping rapid water movement around the seaweed body is important for efficient nutrient uptake and seaweed growth. Here we compared two methods of achieving water motion in the seaweed growth tanks. Water turbulence in the growth tanks was created by air bubbling from the bottom of the tanks, or through water jets from the bottom of the tank. Three separate growth sessions were made—in the autumn, winter and summer—and the growth rates of G. cornea were compared. In all three seasons similar growth rates were measured in the water spray and in the air bubbling methods. No statistically significant differences were observed (Figure 2B). The results also provided us with information regarding optimal growth temperatures for G. cornea in our system. Highest growth rates were recorded when the water temperature was around 24°C in the autumn (Figure 2B, top) and 22°C in the summer (Figure 2B, bottom). Once the temperature dropped in the autumn or rose in the summer the growth rates started to decrease. Addition of illumination during autumn helped raise the growth rates despite lower temperatures (compare Figures 1B, 2B).
Agar content of G. cornea grown under different conditions
Agar is the main industrial extract of Gracilaria seaweeds, the effect of illumination regimes and water movement system on its abundance and quality were analyzed. The quality of the agar extracted from G. cornea was on par with the commercial values with a melting and gelling points at 92.9 ± 2.2°C and 31.2 ± 0.8°C, respectively, compared to 91.7 ± 2.1°C and 33.2 ± 0.7°C for the Bacto™ agar. Gel strength was measured to be 307.8 ± 5.5 g cm-2 for the G. cornea agar, compared to 300.3 ± 15.8 g cm-2 for the Bacto™ agar.
There was no significant difference in the agar yields of G. cornea between the LA-14 and ambient conditions (Figure 3) on a seaweed weight basis. Nonetheless, the total agar produced per square meter is expected to be higher in the LA-14 conditions, due to the higher total biomass accumulation (Figure 1C). Interestingly, seaweeds that were grown with water mixing through the water spray method had higher agar yields than those with the traditional air bubbling setup (Figure 3).
Figure 3
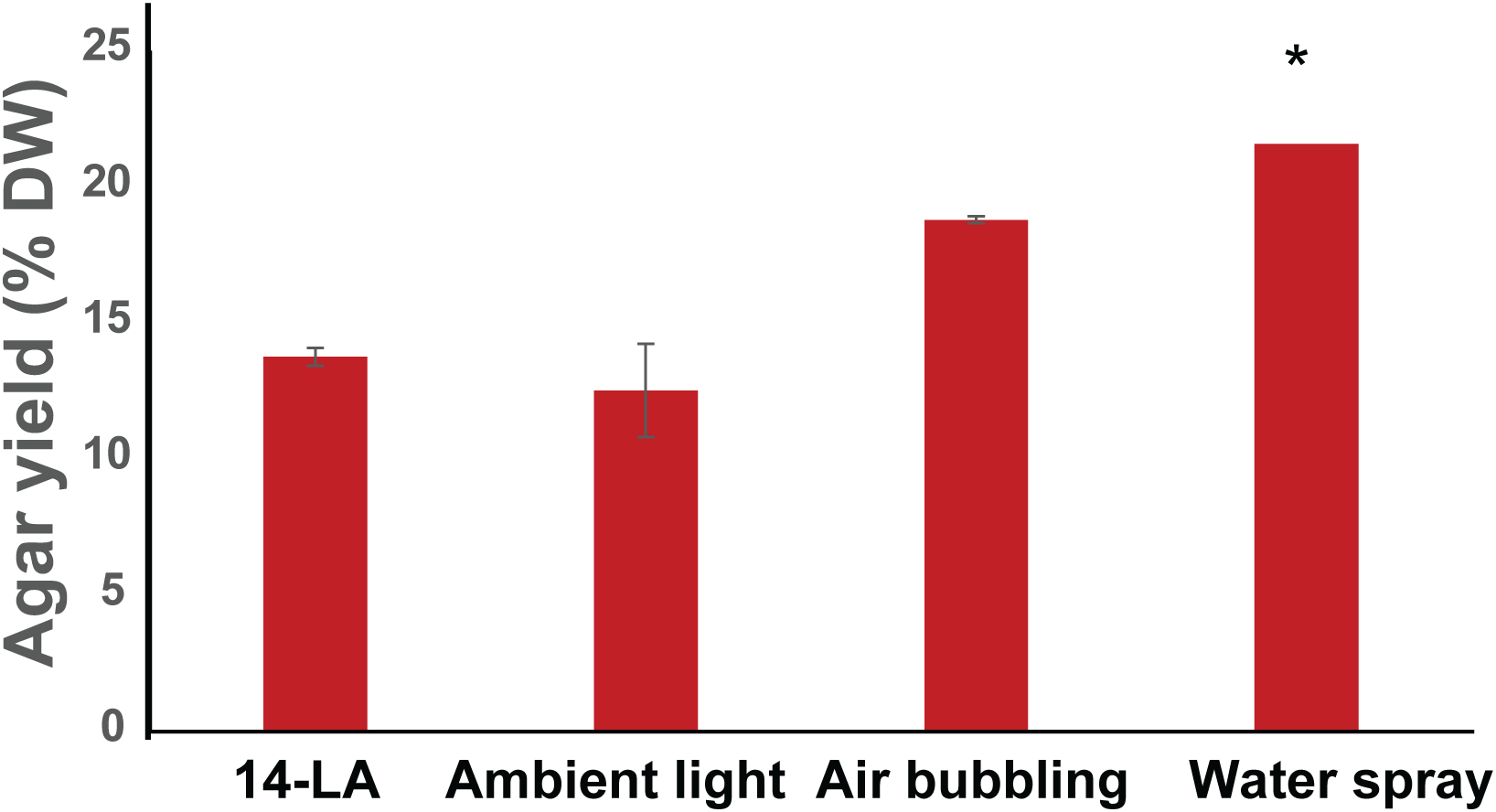
Agar yields of Gracilaria cornea. Seaweeds from the light addition and the water motion experiments were harvested, sundried and underwent agar extraction. The results present the percent yield of the agar on a seaweed dry weight basis. * marks a statistically significant (p < 0.05) difference between light addition and ambient. Error bars represent standard deviation with n=3.
Discussion
Improving cultivation techniques for agar producing seaweed species is of great importance for a sustainable global production of agar (Santos and Melo, 2018). Cultivation of Gracilaria sp. in land-based systems can allow for better control over ambient parameters such as light regimes and water turbulence, as compared to growth in offshore conditions where the seaweeds are exposed to fluctuating natural conditions.
Since seaweeds are photosynthetic organisms and rely on light energy for their growth it is important to measure the effect of added light on their performance and define its parameters. In our setup it was apparent that adding only 4 extra hours of illumination at medium intensities (500 μmol photons m-2 s-2) to the naturally occurring 10-hour daylight regime was not enough to significantly increase the growth rates of G. cornea. On the other hand, the addition of light throughout the entire daytime coupled with extra 4 hours of illumination did increase growth rates in a significant matter. This implies that although G. cornea under our ambient condition was exposed to irradiance of around 900 μmol photons m-2 s-2 during daytime, it was not yet saturated with light, and thus the addition of another 500 μmol photons m-2 s-2 of illumination increased its productivity. This is of special interest since current literature suggests that Gracilaria sp., including G. cornea, is already usually light-saturated at much lower intensities around ~365 μmol photons m-2 s-1 (Reviewed in Salomon, 2025). This can perhaps be attributed to the different experimental photosynthetic rates measurements setups performed in the different reviewed reports and needs to be studied in more detail. It is also interesting that exposure to full sunlight was harmful to the seaweeds, and thus a shading net was required, whereas adding the artificial illumination was helpful. This means that to increase productivity we had to partially shade the sunlight while simultaneously adding more light. Several reasons for this paradox may be made. First, it is possible that the harm for the seaweeds results from the UV spectrum of the light, which is not found in the LED illumination systems. UV exposure may create cellular damage, especially in long exposures. In addition, the direct impact of the sun light on the growth tanks may cause them to heat. In our system this does not seem to be the problem since water temperatures remained stable throughout the day due to sufficient water replacement rates. Finally, it is perhaps possible that the maximal photosynthetic rates of G. cornea in our cultivation systems occur at light intensities around 1,400 μmol photons m-2 s-2. Thus, illumination with only 900 μmol photons m-2 s-2 (under the shading net) or with direct sunlight (~2000 μmol photons m-2 s-2may result in decreased photosynthetic rates and limited growth.
No effect on agar yields (percentage of the seaweed weight) was observed even in the 14 hour light addition protocol as compared to the ambient conditions, and some increase was observed in the water spray system compared to the bubbling treatment (Figure 3). There is a notable difference in the agar yields between the G. cornea seaweeds grown for the light addition experiment and the water movement experiment, this is perhaps because of the different seasons in which the seaweeds were cultivated, but the exact reason for this difference is unknown to us. Seasonal variance of agar yield was previously reported for G. cornea grown in the Yucatan cost, Mexico, where it averaged 31.6% during the cold season and 21.4% during the rainy season (Freile-Pelegrín and Robledo, 1997). These values are higher than compared to our findings, however these can be explained perhaps by the many differences in growth habitat between the Bay of Mexico and in our open land-based system.
Although the increase in seaweed growth as a result of 14 hours of light addition is encouraging, it is up to each seaweed farmer to closely examine the cost versus benefit of this approach. In our systems we calculated that the operational cost of the extra 14 hours of 100W illumination (not including the price of the equipment) was around 0.24 $ US per day. The additional biomass was estimated to be around 30 g day-1 m-2. This implies that illumination costs for 1 Kg of extra seaweed (fresh weight) will be roughly 8 $ US. Using a conservative estimation of 10% dry mass (from the wet weight of the seaweed) and maximum 20% agar yields (on a dry weight basis) we can calculate that the 1 Kg fresh Gracilaria cornea will yield 20 grams of agar. So, for 1 Kg of agar and investment of around 400 $ US in electricity is required. This is currently not a financially viable option since the retail price of 1 Kg of agar can reach 580 $ US per Kg and up, depending on grade (taken online from Merck website1). Nevertheless, in cases where the costs of electricity are lower, or the electricity is produced locally (by solar panels for example), adding artificial illumination should be considered, even when there is apparently sufficient ambient light.
Water motion is especially important for seaweed growth. Rapid relative water movement helps decrease the thickness of the naturally occurring water boundary layer surrounding the seaweed surfaces. This allows for faster diffusion and better nutrient and gas exchange (Gonen et al., 1993; Friedlander and Levy, 1995; Hurd, 2000). Currently, literature regarding land-based seaweed growth systems describes the use of air bubbling or paddle wheels as the means for maintaining the water and the seaweeds in motion. However, a previous report calculated that the costs of air pumping can account for a substantial percentage of the operational costs of seaweed tanks (Ben-Ari et al., 2014). Here we suggest the use of water jets as an alternative. Our results indicate that there were no negative effects on growth rates or agar yields on Gracilaria cornea seaweeds grown using the water spray method as compared to the traditional bubbling method. This eliminates the need for capital investment in the air pumps and tubing setup as well as the operational costs involved in air bubbling. We suggest that the water flow rates provided by the inlet water jets were enough to allow for sufficient diffusion and nutrient uptake rates for the seaweeds growth, at least compared to the air bubbling mode. We believe that there are some other disadvantages of the bubbling system which are not found in the water spray method. In the air bubbling system, it must be considered that in many cases the bubbles also induce the movement of the seaweed together with the water, thus reducing the relative water flow rates against the seaweed body, air mixing increases the risks of oxidative reactions which may harm the seaweed (Friedlander and Levy, 1995). Enriching the water with air may also cause high oxygen levels which may interfere with the photosynthetic reaction.
Several factors still need to be determined, especially regarding upscaling the water spray system. We suggest the use of long shallow tanks, up to 40 cm high, like in our system, where the water inflow pipe can be easily located in its bottom, while providing sufficient water turbulence. This should especially be feasible in U or V shaped tanks where the water spray inlet tube is located in the middle. The long tanks can be fed by a single, long, water pipe ranging the entire length of the tank, or by a series of shorter pipes, this will depend on the inlet water pressure and needs to be determined in situ. In such a system, if for some reason air bubbling is required, the water spray inlet pipe can serve as an air tube when connected to an air compressor, while water inlet can be made by an alternative pipe, from the top of the tank. The location of the water jet head inside the tank is of potential importance due to the resistance of the tank water column against the water jets, which may require additional water pressure in the inlet flow tubing. In addition, the water spray tubes may entangle or interfere with the seaweeds, although this was not observed in our setup, which serves as another reason to keep them the water spray tubes as close to the bottom of the tank as possible.
In conclusion, we demonstrated that addition of artificial LED illumination, even during daytime, for 14 hours enhanced G. cornea daily growth rates. Importantly, the water spray method is introduced as a low cost yet efficient alternative for maintaining good water flow and seaweed growth rates and agar yields in land-based cultivation systems. The combination of higher growth rates coupled with decreased expenses on electricity demand for air compressors for air bubbling should result in more increased net values for the cultivated Gracilaria seaweeds and their agar. This may drive the global agar industry toward increased mariculture practices and less reliance on depleting natural seaweed stocks, and ensuring a constant, sustainable supply of agar and other seaweed extracts.
Statements
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Author contributions
LS: Data curation, Methodology, Investigation, Writing – review & editing, Formal Analysis. ES: Supervision, Software, Methodology, Conceptualization, Investigation, Writing – original draft, Funding acquisition, Visualization, Data curation, Formal Analysis, Writing – review & editing, Project administration.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. Funding was received from the Israeli Ministry for Agriculture and Food Security. Grant number 30-01-0003.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Footnotes
1.^ https://www.sigmaaldrich.com/ accessed May 27, 2025
References
1
Armisen R. (1995). World-wide use and importance of Gracilaria. J. Appl. Phycol3, 231–245. doi: 10.1007/BF00003998
2
Bast F. (2014). “An illustrated review on cultivation and life history of agronomically important seaplants,” in Seaweed: Mineral Composition, Nutritional and Antioxidant Benefits and Agricultural Uses. Ed. PominV. H. (Nova Publishers, New York), 39–70.
3
Ben-Ari T. Neori A. Ben-Ezra D. Shauli L. Odintsov V. Shpigel M. (2014). Management of Ulva lactuca as a biofilter of mariculture effluents in IMTA system. Aquaculture434, 493–498. doi: 10.1016/j.aquaculture.2014.08.034
4
Bhagiya B. K. Yadav D. S. Mantri V. A. (2025). Explant priming with bio-effectors improves regeneration by 6-benzylaminopurine treatment while adventitious lateral shoot development by kinetin in marine red alga Gracilaria dura (Rhodophyta). Discover Oceans2, 10. doi: 10.1007/s44289-025-00048-7
5
El Gamal A. A. (2010). Biological importance of marine algae. Saudi Pharm. J.18, 1–25. doi: 10.1016/j.jsps.2009.12.001
6
Falkowski P. G. Raven J. A. (2007). Aquatic photosynthesis. 2nd Edn (New Jersey: Princeton University Press).
7
FAO (2020). The State of World Fisheries and Aquaculture 2020. (Rome, Italy: FAO). doi: 10.4060/ca9229en
8
Freile-Pelegrín Y. Robledo D. (1997). Effects of season on the agar content and chemical characteristics of Gracilaria cornea from Yucatán, México. Botanica Marina40, 285-290. doi: 10.1515/botm.1997.40.1-6.285
9
Friedlander M. (2001). Inorganic nutrition in pond cultivated Gracilaria conferta (Rhodophyta): Nitrogen, phosphate and sulfate. J. Appl. Phycol13, 279–286. doi: 10.1023/A:1011139329415
10
Friedlander M. Levy I. (1995). Cultivation of Gracilaria in outdoor tanks and ponds. J. Appl. Phycol7, 315–324. doi: 10.1007/BF00004005
11
Friedlander M. Shalev R. Ganor T. Strimling S. Ben-Amotz A. Klar H. et al . (1987). Seasonal fluctuations of growth rate and chemical composition of Gracilaria cf. conferta in outdoor culture in Israel. Hydrobiologia151, 501–507. doi: 10.1007/BF00046174
12
Gonen Y. Kimmel E. Friedlander M. (1993). Effect of relative water motion on photosynthetic rate of red alga Gracilaria conferta. Hydrobiologia260, 493–498. doi: 10.1007/BF00049061
13
Hurd C. L. (2000). Water motion, marine macroalgal physiology, and production. J. Phycol36, 453–472. doi: 10.1046/j.1529-8817.2000.99139.x
14
Kammler S. Malvis Romero A. Burkhardt C. Baruth L. Antranikian G. Liese A. et al . (2024). Macroalgae valorization for the production of polymers, chemicals, and energy. Biomass Bioenergy183, 107105. doi: 10.1016/j.biombioe.2024.107105
15
Levy I. Beer S. Friedlander M. (1990). Growth, photosynthesis and agar in wild-type strains of Gracilaria verrucosa and G. conferta (Gracilariales, Rhodophyta), as a strain selection experiment. Hydrobiologia204, 381–387. doi: 10.1007/BF00040260
16
Levy I. Friedlander M. (1994). Seasonal growth activity of local and foreign gracilarioid strains in Israel. J. Appl. Phycol6, 447–454. doi: 10.1007/BF02182397
17
Lomartire S. Gonçalves A. M. M. (2023). Algal Phycocolloids: Bioactivities and pharmaceutical applications. Mar. Drugs21, 384. doi: 10.3390/md21070384
18
Pangestuti R. Kim S. (2015). “An overview of phycocolloids: The principal commercial seaweed extracts,” in Marine Algae Extracts. Eds. KimS. K.ChojnackaK. (New Jersey, USA: Panggesuty). doi: 10.1002/9783527679577.ch19
19
Pereira R. Yarish C. (2008). “Mass production of marine macroalgae,” in Encyclopedia of Ecology. Eds. JørgensenS. E.FathB. D. (London, UK: Academic Press), 2236–2247. doi: 10.1016/B978-008045405-4.00066-5
20
Salomon E. (2025). “Photosynthetic traits of commercially important red seaweed,” in Biotechnological Interventions to Aid Commercial Seaweed Farming. Eds. RathoreM. S.MantriV. A. (Springer Nature Singapore, Singapore), 239–256. doi: 10.1007/978-981-97-9427-0_11
21
Santos R. Melo R. A. (2018). Global shortage of technical agars: back to basics (resource management). J. Appl. Phycol30, 2463–2473. doi: 10.1007/s10811-018-1425-2
22
Shah Y. Yadav A. Kumar M. A. Kavale M. G. Prasad K. Mantri et al . (2021). ‘Proof of concept’ of how tube-net diameter affects growth and agar content in industrially important farmed red seaweed Gracilaria dura. J. Appl. Phycol33, 2349–2358. doi: 10.1007/s10811-021-02443-x
23
Shauli L. Salomon E. (2025). A Simple method for measuring agar gel strength. Phycology5, 6. doi: 10.3390/phycology5010006
24
Veeragurunathan V. Prasad K. Malar Vizhi J. Singh N. Meena R. Mantri V. A. (2019). Gracilaria debilis cultivation, agar characterization and economics: bringing new species in the ambit of commercial farming in India. J. Appl. Phycol31, 2609–2621. doi: 10.1007/s10811-019-01775-z
Summary
Keywords
Gracilaria , agar, mariculture, land based cultivation, seaweed, water spray, cultivation
Citation
Shauli L and Salomon E (2025) Effects of light addition and water inlet spray on growth rates and agar yields of the red seaweed Gracilaria cornea grown in a land based maricultural system. Front. Mar. Sci. 12:1640877. doi: 10.3389/fmars.2025.1640877
Received
04 June 2025
Accepted
05 August 2025
Published
22 August 2025
Volume
12 - 2025
Edited by
Thomas Wichard, Friedrich Schiller University Jena, Germany
Reviewed by
Daniel Robledo, National Polytechnic Institute of Mexico (CINVESTAV), Mexico
Zujaila Nohemy Qui Minet, Université de Bretagne Occidentale, France
Updates
Copyright
© 2025 Shauli and Salomon.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Eitan Salomon, eitansol@ocean.org.il
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.