- 1Department of Stomatology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
- 2School of Stomatology, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
- 3Hubei Province Key Laboratory of Oral and Maxillofacial Development and Regeneration, Wuhan, China
Oral-maxillofacial bone defects complicated by tumors, infections, or other bone diseases pose a significant clinical challenge. Traditional tissue-engineered bone substitute still has limitations regarding its three elements that resulting in unsatisfactory regeneration capability. Smart materials are a cutting-edge type of functional materials that can sense and respond to a wide range of environmental conditions or stimuli, including optical, electrical, magnetic, mechanical, thermal, and chemical signals. According to the type of stimulus to which the materials respond, they can be classified into externally stimulated materials and internally stimulated materials. This review, based on the latest advances in smart materials for bone defect repair, summarizes the different stimulus-responsive strategies of smart materials and the materials under each strategy. It also discusses the classic biomedical applications of these materials in the repair of oral-maxillofacial bone injuries in recent studies, compares the advantages and disadvantages of different strategies, and discusses the current challenges and future prospects of smart materials.
1 Introduction
Bone defects in the oral and maxillofacial region are common clinical challenges, severely affecting patients’ masticatory function, facial appearance, and quality of life. During the process of repairing these bone defects, a variety of biomaterials are widely used.
Bone tissue engineering integrates biomaterial scaffolds, cells, and bioactive factors to construct biomimetic structures to enhance bone regeneration (Koons et al., 2020). The application of additive manufacturing technology and topographical, chemical, and/or biochemical modifications has continuously enhanced the osteogenic activity of bone tissue engineering (Zhang et al., 2019b; 2020). Recently, researchers gradually recognized that the repair process of bone defects is not a static and one-stage process (Li et al., 2024). Bone regeneration and remodeling are long-term dynamic processes. Therefore, there is a need to develop responsive biomaterials that can synchronize the interactions between the material and the surrounding tissues in both space and time. Meanwhile, challenge pathological conditions, such as bacterial infection, chronic inflammation, and disorders affecting systemic metabolism, raised the difficulties of local regenerative capability of the defect area, which requests more complex approaches to simultaneously cope with adverse metabolic conditions and stimulate tissue regeneration (Monfoulet et al., 2014; Tao et al., 2020).
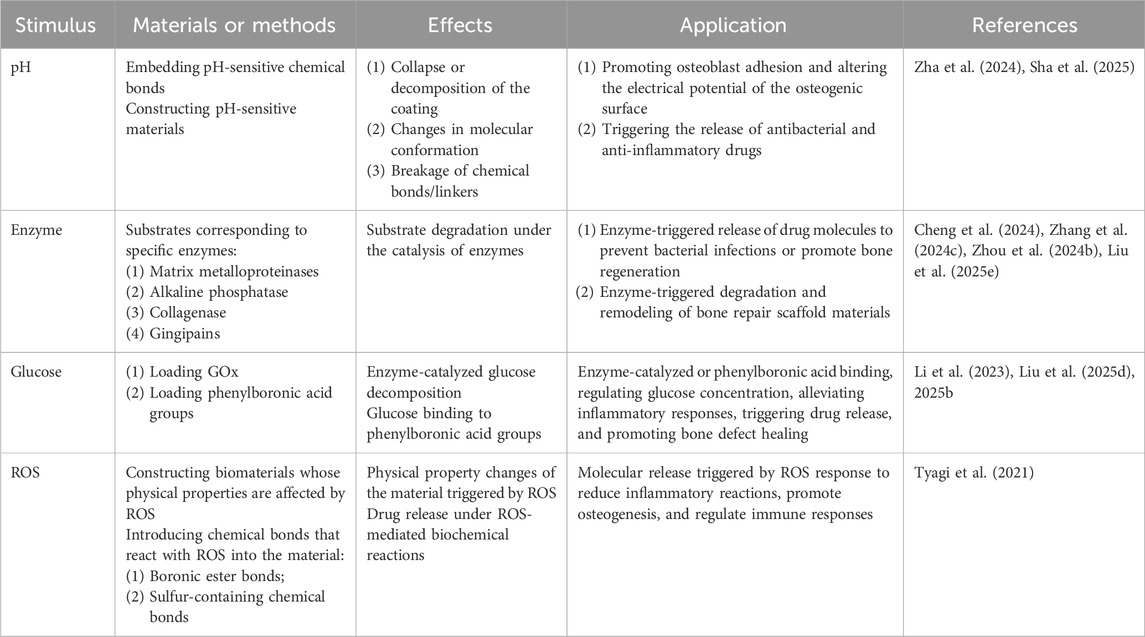
In this context, smart materials, also known as responsive biomaterials, have emerged. These smart materials retain the basic framework of traditional materials but have been endowed with the ability to sense and respond to environmental changes through innovative approaches such as the introduction of functional groups, the incorporation of electromagnetic materials, the reconfiguration of material structures, and the embedding of sensors (Wei et al., 2022). This dynamic responsiveness enables smart materials to better adapt to the complex microenvironmental changes during bone defect repair, thereby enhancing the effectiveness and efficiency of tissue regeneration (Table 1). The types of stimuli that smart materials can respond to can be divided into external stimuli (such as light irradiation, electric and magnetic fields, ultrasound, and appropriate mechanical stimulation) and internal stimuli (such as excess reactive oxygen species (ROS), slight acidity, endogenous electric fields, specific ion concentrations, secreted enzymes, or specific immune environments) (Figure 1).
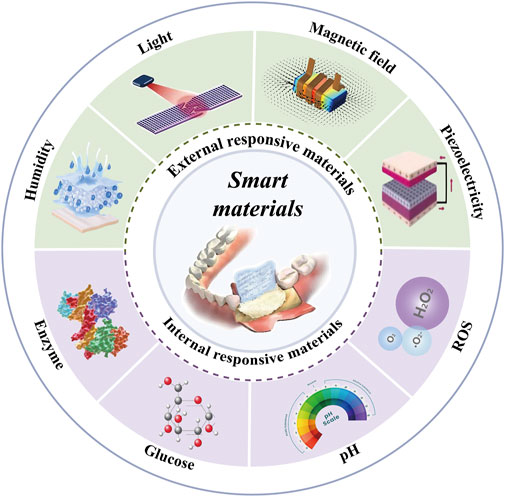
Figure 1. Mechanisms and Stimuli Factors of Smart Materials. Abbreviations: ROS, Reactive Oxygen Species; pH, potential of hydrogen.
In this review, we summarized the different stimulus-responsive strategies, including smart materials under external and internal stimulus-responsive strategies, and elaborated on the classic biomedical applications for oral and maxillofacial bone injury repair in recent studies. We also compared the advantages and disadvantages of different strategies and discussed the current challenges and future prospects of these new biomaterials. This knowledge may help to construct multifunctional biomaterials in the future to meet the needs of oral-maxillofacial bone defects repair in different environments.
2 External stimuli
External stimuli such as light, magnetic fields, electrical stimulation, and appropriate mechanical stimulation can generate heat or promote the adhesion, proliferation, and differentiation of osteoblasts within scaffolds, thereby facilitating bone therapy and regeneration. In this section, we elaborate on various external stimulus-responsive strategies (Table 2).
2.1 Photothermal
Photothermal stimulation refers to the process of achieving regulatory effects by converting light energy into thermal energy (Liu A. et al., 2025). The core components of photothermal responsive materials comprise: (1) Photothermal conversion components (e.g., graphene, black phosphorus, and gold nanoparticles), which efficiently absorb specific wavelengths of light, thereby inducing transformations such as material deformation, drug release, or bioactivity activation (Huang et al., 2017; Chen et al., 2019; Bisoyi and Li, 2022); (2) Bone repair matrices (e.g., polylactic acid, polycaprolactone, hydrogels, or β-tricalcium phosphate), which provide mechanical support and facilitate the delivery of osteogenic factors (such as magnesium ions) (Zhang D. et al., 2024; Liu A. et al., 2025).
Commonly utilized light sources encompass near-infrared (NIR) and ultraviolet (UV) radiation. NIR is widely utilized for photothermal effects due to its exceptional tissue penetration depth, enabling energy delivery to deep tissues. This capability is attributed to minimal absorption by hemoglobin and water molecules, ensuring limited energy attenuation during therapeutic applications (Zhang J. et al., 2024). Under photothermal stimulation, scaffolds exhibit a shape memory effect: heating above the transition temperature softens the material, allowing it to conform to irregular bone defects; subsequent cooling solidifies the scaffold into the desired geometry (Wang H. et al., 2025; 2025c). Concurrently, photothermally triggered temperature elevation induces material expansion or cleavage of chemical bonds, enabling precise release of anti-inflammatory drugs, growth factors, or other bioactive agents (Zhang D. et al., 2024). In photothermal-activated systems, mild heating (42 °C) upregulates heat shock protein HSP70 and activates the MAPK/ERK osteogenic pathway. This process further promotes the release of mineralizing ions (e.g., Ca2+, PO43-), thereby accelerating tissue mineralization (Zhu et al., 2024).
The application of photothermal stimulation is highly controllable; NIR can be exactly delivered to the defect site to achieve therapeutic effect. Moreover, it is non-invasive, non-toxic, and possesses high biosecurity. The photothermal effect itself has the ability to regulate the immune microenvironment, promoting the polarization of macrophages from pro-inflammatory M1 to anti-inflammatory M2, reducing inflammation and enhancing angiogenesis (Zhang D. et al., 2024; Zhu et al., 2024). In addition, UV (wavelength 254–365 nm) can also induce the formation of a three-dimensional network structure between material molecules, serving as a stimulus for photo-responsive materials. Ding et al. developed a photo-responsive hydrogel for bone tissue formation, which is composed of a photo-cross-linkable polymer solution, a photo-initiator, and a UV absorber. Upon UV irradiation, the polymer undergoes photo-crosslinking to form a solid filler that can carry human bone marrow mesenchymal stem cells, facilitating osteogenic differentiation (Ding et al., 2022). Similarly, Hu et al. achieved the modification of hyaluronic acid with methacrylic anhydride, enabling the hyaluronic acid to undergo photo-crosslinking under UV irradiation to form a stable three-dimensional network structure. Based on this structure, an antibacterial agent was loaded, making it an injectable material with both oral-maxillofacial defect repair and antibacterial functions (Hu et al., 2024).
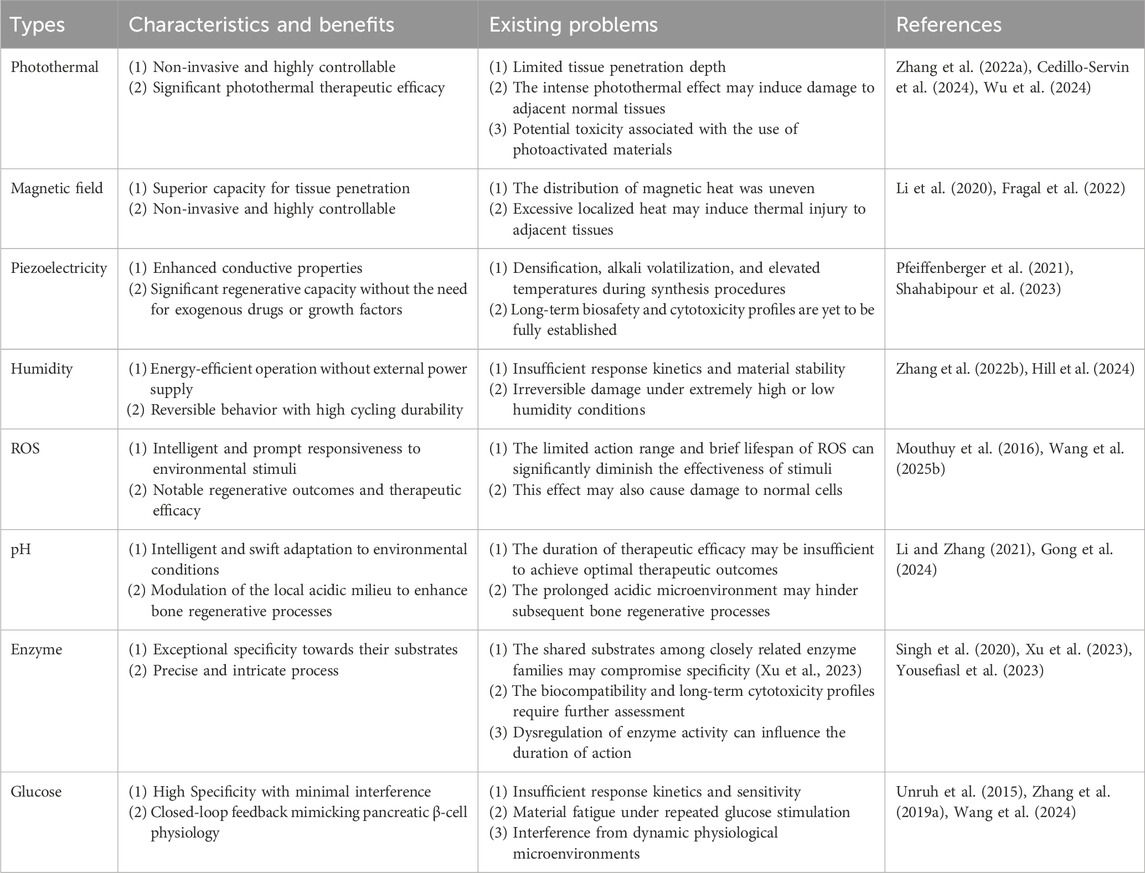
In addition to the encouraging achievements of the aforementioned photo-responsive biomaterials, there are still unresolved issues. NIR has low penetration efficiency in deep tissues, which affects the photothermal conversion effect in maxillofacial scaffold defects, thereby ultimately hindering the regeneration of deep tissues in vivo (Chen et al., 2020).
2.2 Magnetic field
Magnetic field (MF) is a non-invasive stimulation, which excels in high tissue penetration, less toxic side effects and high controllability. External static magnetic fields (SMFs) have direct biological effects on cells and promote osteogenesis of mesenchymal stem cells (MSCs) by affecting cell metabolism and signaling (Yan et al., 2025). The enhanced osteogenesis is thought to be associated with MF-induced opening/closing of ion channels, cytoskeleton remodeling, cellular membrane potential elevation of the stimulated osteoblasts. Also, the biological effects of MF also act on pathogens. Wu et al. reported the disruption of bacterial biofilms by integrating magnetic nanoparticles (MNPs) into tricalcium phosphate scaffolds under the action of SMFs, which led to the effective control of infection (Wu et al., 2024).
The construction of magnetic-responsive materials is usually based on MNPs, dominated by Fe3O4, Fe2O3, and FeO. Chen et al. has designed a magnetic-responsive composite coating by loading γ-Fe2O3 nanoparticles onto TiO2 nanoporous arrays, which promotes cell proliferation and accelerates osteogenesis under SMFs (Chen et al., 2024). Some studies have combined magnetic silica nanoparticles with MSCs to prepare magneto-mechanical-bioengineered MSCs, which can activate the YAP/β-catenin signaling pathway under SMF to promote osteogenesis, mineralization, and angiogenesis, while decease bone resorption and rebalancing bone metabolism (Yu C. et al., 2023).
In addition, magnetic materials improve the mechanical strength of scaffolds. One study significantly enhanced the mechanical properties of hydrogel by introducing Fe3O4 nanoparticles and tannic acid (Zou et al., 2023). Magnetic materials can be used for remote drug-controlled release with the help of MF. A study has developed a double crosslinked magnetic hydrogel for remote controlled pulsatile release of parathyroid hormone by MNPs, which can mimic the clinical mode of drug delivery (Long et al., 2023).
Magnetic responsive materials have significantly enhanced the functions of bone implant materials, but their biosafety issues need to be noted. Degradation of magnetic materials in vivo products may be cytotoxic. MNPs may release metal ions (Co2+, Fe3+) after degradation in vivo, and long-term accumulation can easily induce cytotoxicity or inflammatory reactions (Liu W. et al., 2023). Therefore, biodegradable magnetic phases should be developed, or the risk of ion leakage should be reduced through surface functionalization such as coating with stem cell membranes (Wu et al., 2024). On the other hand, magnetic fields have tissue penetration capabilities, energy attenuation is significant with increasing depth (Shen et al., 2023). Therefore, for deep bone defects, multi-level amplification strategies can be combined to enhance local magnetic field strength (Cedillo-Servin et al., 2024).
2.3 Humidity
Humidity-responsive smart materials detect environmental humidity changes and generate controllable physical or chemical responses. These responses—such as swelling, contraction, degradation, or drug release—promote bone tissue regeneration. The mechanism relies on material hydration/dehydration via water molecule adsorption/desorption, primarily driven by physical interactions (e.g., hydrogen bonding, van der Waals forces) between the material and water (Shuai et al., 2022; Du et al., 2023; Mao et al., 2024; Yang et al., 2024).
Many synthetic polymers exhibit humidity responsiveness. The molecular chains of thermoplastic polyurethane contain numerous amino (N–H) and carbonyl (C=O) groups. Upon exposure to moisture, water molecules form hydrogen bonds with these functional groups, leading to hydration-induced deformation. This property renders thermoplastic polyurethane an ideal candidate for bone defect repair in minimally invasive surgery (Zhang Y. et al., 2019). Moreover, incorporating naturally humidity-responsive polymers into synthetic polymers enhances composite properties while maintaining structural stability. For instance, amorphous calcium-magnesium pyrophosphate possesses substantial free volume and active sites, enabling rapid hydration-driven expansion. Cassava starch contains abundant hydroxyl groups that form water-absorbing hydrogen bonds. Amorphous calcium-magnesium pyrophosphate/cassava starch composite scaffolds exhibit rapid humidity response: their swelling increases porosity, promote cell/cytokine attachment, while the expansion rate matches bone growth, thereby supporting in vivo tissue regeneration (Yang et al., 2024). Similarly, silk fibroin protein extracted from silkworm cocoons was applied. Silk fibroin comprises disordered hydrophilic (amorphous) regions and crystallizable hydrophobic blocks (β-crystal regions). The water-soluble hydrophilic regions confer elasticity and toughness, enabling hydration-driven shape memory. MgO particles were incorporated to modulate degradation rate, enhancing the material’s adaptability to in vivo bone tissue regeneration (Mao et al., 2024).
Humidity-responsive materials hold significant potential for bone regeneration, yet several challenges require improvement. While current materials enable programmable multi-stage deformations at varying humidity levels, their performance often be negatively affected in extreme humidity environment (Du et al., 2023). Also, existing systems exhibit constrained stiffness, compromising their usage in load-bearing bone defects. Last but not least, maintaining structural integrity and stable volume post-hydration is critical.
2.4 Piezoelectricity
Piezoelectric materials refer to certain materials that are associated with mechanical stress and the generation of electrical charges on surfaces. Generally, a piezoelectric material will generate an induced charge internally after being mechanically stressed, triggering a positive piezoelectric effect. If an electric field is subsequently applied to this material, it will cause a geometric strain, resulting in an inverse piezoelectric effect (Uchino, 2017).
Interestingly, natural bone defect healing progress, coordinately regulated by chemical, physical, and electrical signals (Pfeiffenberger et al., 2021), intrinsically leverages piezoelectricity: collagen polarization under stress generates a net negative surface charge. This attracts calcium ions into osteoblasts via voltage-gated channels, facilitating mineralization (Ahn and Grodzinsky, 2009; Khare et al., 2020). By exploiting bone’s sensitivity to these piezoelectric signals, which modulate metabolism and osteogenesis, piezoelectric materials demonstrate strong potential in bone tissue engineering (Nain et al., 2024).
Researchers explore piezoelectric materials for the complex oral-maxillofacial environment, with injectable/moldable hydrogels incorporating piezoelectric nanoparticles emerging as a central focus (Zhu et al., 2020). These composites offer excellent biocompatibility, tunable mechanics, and efficient localized electroactivity, enhancing bone repair. Innovatively, Zhou et al. incorporated dynamically covalently crosslinked piezoelectric nanoparticles into a hydrogel. This design improved material performance and significantly accelerated bone healing in vivo. Mechanistic studies revealed the hydrogel promotes intracellular calcium influx, continuously activating PI3K/Akt and MAPK/ERK osteogenic pathways to drive bone marrow mesenchymal stem cell differentiation (Zhou S. et al., 2024).
Successful bone repair requires inflammation control, demanding piezoelectric materials that synergistically regulate the inflammatory microenvironment. Wu et al. developed a BaTiO3/PDA@HA hydrogel scaffold that provides electro-immunomodulation via bioactive interfaces, promoting reparative M2 macrophage polarization via PI3K/Akt signaling to create a pro-regenerative niche (Wu et al., 2023). For inflamed environments, Ricotti et al. designed a BaTiO3/graphene oxide hydrogel system; its synergistic effects directly drive new bone formation in oral-maxillofacial inflammation, offering novel strategies for severe inflammatory bone defects (Ricotti et al., 2024).
Piezoelectric material applications are rapidly expanding, particularly in periosteal engineering where electrical stimulation enhances bone repair (Liu J. et al., 2023; Liu H. et al., 2023; Yue et al., 2025). Recognizing the periosteum’s critical role in early bone formation, its protection/utilization is now a key bone defect strategy (Shahabipour et al., 2023). Yue et al. developed a PVDF piezoelectric periosteal scaffold with curcumin-loaded Mg-MOF, synergistically promoting nerve repair, angiogenesis, and inflammation regulation (Yue et al., 2025). Separately, Liu et al.'s TiO2@PVDF nanofiber membrane (0.3 wt% TiO2) demonstrated markedly enhanced cell adhesion/proliferation via high surface potential, while electromechanical stimulation robustly induced early alkaline phosphatase activity–confirming electrical properties’ essential role in osteogenesis initiation (Liu J. et al., 2023).
Piezoelectric materials for oral-maxillofacial bone repair have evolved from single-component exploration to designing mechanism-driven multifunctional composites (hydrogels, fiber membranes, coated scaffolds). Future efforts must address long-term stability and precise electrical control in physiological environments, establish standardized performance comparisons, and translate findings into clinical solutions for large and infected defects.
3 Internal stimulation
Pathological progression closely links to altered physical/biochemical microenvironmental cues. These can act as intrinsic triggers for specific materials, triggering structural transformations that elicit biological effects on surrounding tissues. Highlighting distinctions between endogenous and exogenous stimulus strategies, this section focuses on recent advances in internal stimulus-responsive implants (Table 3).
3.1 pH
The microenvironmental pH critically regulates tissue-engineered bone regeneration by modulating protein adsorption on artificial bone surfaces, osteogenesis-related cellular behaviors, bone matrix secretion/maturation, biomineralization, and inflammatory responses with vascular remodeling in bone defects (Liu et al., 2016; Hao et al., 2017).
Microenvironmental pH critically regulates MSCs and osteoblast proliferation (Hao et al., 2017). Acidic pretreatment (pH 6.8) enhances stem cell marker expression while improving viability and proliferation (Hazehara-Kunitomo et al., 2019). While an alkaline environment (pH 8.0–8.4) promotes initial proliferation in pre-osteoblasts, alkali-treated titanium surfaces inducing local pH elevation cause cell alkalosis and inhibit human bone marrow mesenchymal stem cell proliferation (Li et al., 2014; Galow et al., 2017). Notably, both acidic (pH 6.3/6.7) and highly alkaline (pH 8.5) conditions significantly suppress human bone marrow mesenchymal stem cell proliferation by accelerating cellular senescence, whereas physiological (pH 7.0/7.4) and mildly alkaline (pH 8.0) microenvironments optimally support cell survival and proliferation (Fliefel et al., 2016).
Oral-maxillofacial bone defects form a local acidic microenvironment due to reduced blood supply, anaerobic metabolism, and lactic acid accumulation from hematoma, infection, and inflammation (Hazehara-Kunitomo et al., 2019). Tissue-engineered bone exhibits reduced pH buffering from limited vascular ingrowth, inflammatory responses, and confined cell space, leading to acidic metabolite accumulation and heightened cellular sensitivity to pH fluctuations (Monfoulet et al., 2014; Pj and Carmeliet, 2016).
Construction strategies for pH-responsive materials comprise three main categories: functional group design, dynamic bond incorporation, and use of self-assembling peptides/nanozymes (Sha et al., 2025). Anionic polymers (e.g., polyacrylic acid) swell with cations, while cationic chitosan dissolves in acidic environments—suitable for infected bone defects (Lin et al., 2018). Dynamic bonds (e.g., hydrazone, Schiff base) enable self-healing and responses to pH/temperature (Li Z. et al., 2022). Self-assembling peptides (e.g., histidine-rich) form nanofiber gels at pH 6.0. Nanozymes like sulfur quantum dots exhibit acidic peroxidase-like activity (sterilization) and neutral catalase-like activity (bone repair) (Li Z. et al., 2022; Liu et al., 2025c).
Current pH-responsive bone repair materials primarily comprise smart polymers (e.g., polyacrylic acid, chitosan, polyline) or nanocomposite structures (e.g., Metallo phenolic networks, peptide self-assembly systems) (Bonchev and Bogovska-Gigova, 2025; Sha et al., 2025). These materials adapt to local pH changes by reversibly altering physical/chemical properties—such as swelling/contraction, degradation modulation, and drug release—enabling targeted antimicrobial delivery in the acidic microenvironment (pH 5.5–6.8) of infected oral-maxillofacial defects (Lin et al., 2025; Ma et al., 2025). During early inflammation, accelerated acidic degradation releases antimicrobial agents; as pH rises to physiological levels, degradation slows to provide sustained scaffolding for bone regeneration (Li Z. et al., 2022).
pH-responsive bone repair materials face technical limitations. Current materials require >0.5 pH unit changes for activation—insufficient for mild infections with only 0.2–0.3 unit differences (Tapponi et al., 2025). For example, metal phenolic networks coatings trigger drug release only at pH < 6.0, delaying response to early mild infections (pH 6.5–7.0) (Ali et al., 2023).
3.2 Enzymes
Enzyme-responsive smart materials for bone repair are a class of biomaterials that can specifically recognize changes in enzyme activity in the bone injury microenvironment and trigger their own functions (such as drug release, structural transformation, or signal transduction) accordingly (Liu X. et al., 2025).
The core principle of enzyme-responsive materials is enzyme-catalyzed reactions, and their design relies on enzyme-sensitive chemical bonds and enzyme-catalyzed signal transduction. The former involves embedding chemical bonds in the material that can be hydrolyzed or modified by specific enzymes (such as phosphate ester bonds, peptide bonds),thereby enabling the material to be triggered by specific enzymes and undergo conformational changes (Zhang M. et al., 2024; Zhou X. et al., 2024). Alkaline phosphatase, matrix metalloproteinase, collagenase, and gingipains can all serve as target enzymes (Liu et al., 2021; Liu X. et al., 2025; Xu et al., 2023; Zhou Y. et al., 2023; Zhou X. et al., 2024; Zhang M. et al., 2024).
The latter, that is, enzyme-catalyzed signal transduction, refers to the conversion of pathological signals into chemical changes recognizable by materials. These nanozymes, which have catalase, superoxide dismutase, and glutathione peroxidase enzyme-like properties, effectively reprogram the microenvironment of the mandible and treat mandibular osteoradionecrosis (Cheng et al., 2024). Compared with traditional bone repair materials, enzyme-responsive materials have the advantage that they can release drugs only in areas with high enzyme expression, thereby avoiding systemic toxicity. Enzyme activity is positively correlated with the degree of pathology, and the material can automatically adjust the release amount accordingly, avoiding excessive drug damage (Zhou X. et al., 2024).
Enzyme-responsive smart materials face clinical translation challenges primarily concerning enzyme stability. The complex in vivo environment can deactivate immobilized enzymes, necessitating protective strategies like nanoencapsulation (Singh et al., 2020). For materials with multiple enzymes, optimizing sequential reactions and cascade regulation remains critical. Future integration with artificial intelligence and machine learning offers potential to predict enzyme-material interactions and optimize kinetics (Kroll et al., 2023; Wang et al., 2025d).
3.3 Glucose
Long-term hyperglycemia can weaken the immune system of patients, leading to exacerbation of oral-maxillofacial inflammation, reduced bone repair capacity, severe loss of alveolar bone mass, poor osseointegration of dental implants, and poor repair of oral-maxillofacial bone defects (Wu et al., 2015).
The design of glucose-responsive smart materials is primarily based on two main strategies: glucose oxidase (GOx)-based system, phenylboronic acid-based system (Li et al., 2023). GOx, primarily found in human red blood cells, renal tubules, and hepatocytes, specifically catalyzes the conversion of β-D-glucose into gluconic acid and hydrogen peroxide (H2O2). Based on this principle, glucose-responsive 3D-printed scaffolds can be engineered (Liu et al., 2025b). Phenylboronic acid, as a Lewis acid containing a boron atom, has the core characteristic of being able to specifically and reversibly bind with the vicinal diol group in glucose molecules (Morariu, 2023). This unique glucose responsiveness makes it an ideal molecular tool for constructing smart delivery systems (Wang et al., 2024; Liu S. et al., 2025). Elevated blood glucose prompts phenylboronic acid groups to bind glucose preferentially. This disrupts boronate ester crosslinks, loosening the hydrogel structure. Conversely, low glucose reduces this binding, stabilizing crosslinks and maintaining a compact hydrogel to slow substance release (Wang et al., 2024).
It is worth noting that the bone tissue of patients with type 2 diabetes often has dysfunction in resistance to deformation and fracture. This decline in mechanical properties makes fracture healing even more difficult. Therefore, good mechanical properties are crucial for providing a stable microenvironment for bone tissue regeneration (Jiang et al., 2022).
Smart materials for bone repair based on GOx and phenylboronic acid groups have shown significant potential in the repair of diabetic bone defects. Although their response mechanisms are different, both can achieve dynamic regulation of the pathological microenvironment of bone defects.
3.4 ROS
Excessive ROS increase the apoptosis rate of osteoblasts, damage stem cell function, accelerate osteoclast differentiation and bone resorption, exacerbate inflammatory responses and vascular damage, thereby delay the healing process of bone defects (Tao et al., 2020; Ren et al., 2022).
Traditional non-degradable materials may release toxic ions in an ROS environment or hinder the ingrowth of new tissue, further delaying healing and creating a vicious cycle (Wang et al., 2025b). Whereas, the therapeutic goal of ROS-responsive materials is not simply to reduce ROS levels, but to regulate ROS levels within an appropriate range (Tyagi et al., 2021; Ren et al., 2022). Low concentrations of ROS can activate pathways such as MAPK/ERK, promoting the expression of osteogenic markers, and driving the osteogenic differentiation of MSCs. Therefore, excessive clearance of ROS may block osteogenic differentiation signals and delay bone defect healing (Mouthuy et al., 2016; Tyagi et al., 2021; Zhang Q. et al., 2024).
Interestingly, under controlled conditions, ROS can exert strong antibacterial effects (Xu et al., 2025). Recently, an emerging dynamic therapy has emerged that treats deep hypoxic infected bone defects by increasing ROS levels. This implant generates sulfate radicals (·SO4−) and ·OH in a hypoxic environment, killing bacteria through lipid peroxidation and ferroptosis mechanisms. However, the critical concentration of ROS that promotes osteogenesis still needs further investigation (Wang et al., 2025b).
ROS-responsive materials are primarily engineered around three core mechanisms: chemical bond cleavage, physicochemical property transformation, and bioactive regulation. The chemical bond cleavage strategy, utilizing ROS-sensitive bonds (e.g., thioketal, selenium-selenium bond, phenylboronic ester) that oxidatively break under high ROS levels to trigger material degradation or payload release, represents the most prevalent design approach (Li X. et al., 2022; Zhang Q. et al., 2024). Notably, bond sensitivity varies, necessitating selection based on the application context, such as chronic inflammation (Yu et al., 2022). The physicochemical property transformation mechanism exploits ROS-induced alterations in material state or surface characteristics, primarily categorized into hydrophobicity-to-hydrophilicity transitions (solubility switching) for controlled release and surface charge reversal to enhance cellular uptake (Zhou J. et al., 2023). Bioactive regulation extends beyond delivery, directly targeting the pathological microenvironment by incorporating ROS-scavenging antioxidants or nanozymes, or by modulating ROS levels to influence cell behavior and promote tissue repair (Yin et al., 2021; Peng et al., 2024; Zhang Q. et al., 2024).
To address complex pathological environments like infected bone defects, multi-mechanistic integration, representing a cutting-edge approach, combines strategies such as photothermal effects to accelerate ROS-sensitive bond cleavage or enzyme-responsiveness for dual-signal triggered release (Tian et al., 2022; Yu P. et al., 2023).
However, significant challenges remain: current ROS-responsive systems often exhibit linear response mechanisms, ill-suited to fluctuating ROS levels characteristic of sites like bone defects. The inherent background concentration, activity, and fluctuations of endogenous ROS often render single-mechanism responses insufficient, underscoring the urgent need for strategies responsive to multiple signals or featuring intelligent feedback control (Chen et al., 2023; Zhang Q. et al., 2024).
4 Conclusion
Here, we reviewed the research progress of smart materials in the field of maxillofacial bone reconstruction, discusses the characteristics of smart materials and their applications, and analyzes in detail the current status and prospects of the application of external stimulus and internal stimulus-responsive smart materials. By categorizing the types of stimuli to which smart materials respond, this paper discusses in depth the application scenarios of each type of stimuli in clinical practice and looks forward to the future development direction (Table 4). Despite promising functionalities, smart stimuli-responsive systems are still in the preclinical exploration phase, requesting intensified research efforts to bridge the gap toward clinical adoption.
Firstly, comprehensive biosafety assessments of these materials and their bioactive degradation byproducts are imperative. Secondly, given the prolonged regeneration timeline for bone tissue defects—particularly segmental defects requiring months of healing—coupled with lifelong remodeling processes, the long-term structural integrity and sustained responsiveness of smart bone substitutes must be rigorously validated through extended in vivo studies. Furthermore, osseous tissues exhibit distinct physical and mechanical characteristics compared to other clinical targets—particularly in terms of load-bearing capacity and mineralization dynamics—necessitating tailored optimization of external stimulus parameters (e.g., intensity, duration, frequency) prior to clinical deployment. Notably, internal stimuli entail patient-specific modulation due to inter-individual variability and dynamic pathophysiological progression, significantly complicating their control precision (Figure 2).
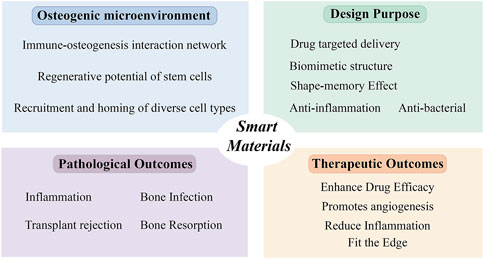
Figure 2. Design strategies and mechanisms of smart materials in the treatment of oral-maxillofacial bone defects.
Author contributions
YY: Visualization, Writing – original draft, Writing – review and editing. ZL: Writing – review and editing. XQ: Writing – review and editing. KS: Funding acquisition, Writing – review and editing. LX: Funding acquisition, Writing – review and editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This review was supported by Natural Science Foundation of Hubei Province for General Project, Grant Numbers: 2025AFB748, 2023AFB765; Key projects of Tongji Hospital Fund, Grant Number: 2024A23; School of Stomatology Teaching Research Project, Grant Number: 2024KQ-JY202; Key Research and Development Program of Ningxia Hui Autonomous Region, Grant Number: 2022BEG03160.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Ahn, A. C., and Grodzinsky, A. J. (2009). Relevance of collagen piezoelectricity to “Wolff’s Law”: a critical review. Med. Eng. and Phys. 31, 733–741. doi:10.1016/j.medengphy.2009.02.006
Ali, A., Javed, R., Farhangi, S., Shah, T., Ullah, S., ul Ain, N., et al. (2023). Metal phenolic networks (MPNs)-based pH-sensitive stimulus responsive nanosystems for drug delivery in tumor microenvironment. J. Drug Deliv. Sci. Technol. 84, 104536. doi:10.1016/j.jddst.2023.104536
Aljohani, A., and Desai, S. (2018). 3D printing of porous scaffolds for medical applications. AJEAS 11, 1076–1085. doi:10.3844/ajeassp.2018.1076.1085
Aversa, R., Apicella, A., Tamburrino, F., and Petrescu, F. I. T. (2018). Mechanically stimulated osteoblast cells growth. AJEAS 11, 1023–1036. doi:10.3844/ajeassp.2018.1023.1036
Bansal, M., Dravid, A., Aqrawe, Z., Montgomery, J., Wu, Z., and Svirskis, D. (2020). Conducting polymer hydrogels for electrically responsive drug delivery. J. Control. Release 328, 192–209. doi:10.1016/j.jconrel.2020.08.051
Bisoyi, H. K., and Li, Q. (2022). Liquid crystals: Versatile self-Organized smart Soft materials. Chem. Rev. 122, 4887–4926. doi:10.1021/acs.chemrev.1c00761
Bonchev, A., and Bogovska-Gigova, R. (2025). Alkasites in restorative dentistry: a review of their performance and properties. J. Dent. 160, 105916. doi:10.1016/j.jdent.2025.105916
Cedillo-Servin, G., Dahri, O., Meneses, J., van Duijn, J., Moon, H., Sage, F., et al. (2024). 3D printed Magneto-active Microfiber scaffolds for remote stimulation and Guided organization of 3D in vitro Skeletal Muscle Models. Small 20, 2307178. doi:10.1002/smll.202307178
Chen, L., Dong, Y., Tang, C.-Y., Zhong, L., Law, W.-C., Tsui, G. C. P., et al. (2019). Development of direct-Laser-Printable light-powered nanocomposites. ACS Appl. Mat. Interfaces 11, 19541–19553. doi:10.1021/acsami.9b05871
Chen, G., Cao, Y., Tang, Y., Yang, X., Liu, Y., Huang, D., et al. (2020). Advanced near-infrared light for Monitoring and modulating the Spatiotemporal dynamics of cell functions in Living systems. Adv. Sci. 7, 1903783. doi:10.1002/advs.201903783
Chen, W., Zhang, H., Zhou, Q., Zhou, F., Zhang, Q., and Su, J. (2023). Smart Hydrogels for Bone reconstruction via modulating the microenvironment. Research 6, 2501354. doi:10.34133/research.0089
Chen, H., Tan, X., Fu, J., Xi, H., Xue, P., Rogachev, A. V., et al. (2024). γ-Fe2O3/Polydopamine/TiO2 nano-porous array composite coating (FPTCC) to modulate antibacterial, osteogenesis, and osseointegration through photothermal-magnetic response. Mater. and Des. 248, 113516. doi:10.1016/j.matdes.2024.113516
Chen, T., Zhang, Z., Zhou, X., Zhang, Z., Liu, J., Gong, Q., et al. (2025). Fe3O4@PDA nanoparticle-Doped smart hydrogel scaffold for Osteochondral defect repair by synergistical stimulation. Adv. Funct. Mater. n/a 35, 2501354. doi:10.1002/adfm.202501354
Cheng, Z., Wang, Y., Lin, H., Chen, Z., Qin, R., Wang, T., et al. (2024). Engineering dual active sites and defect structure in nanozymes to reprogram Jawbone microenvironment for osteoradionecrosis therapy. Adv. Sci. (Weinh) 12, 2413215. doi:10.1002/advs.202413215
Ding, A., Lee, S. J., Ayyagari, S., Tang, R., Huynh, C. T., and Alsberg, E. (2022). 4D biofabrication via instantly generated graded hydrogel scaffolds. Bioact. Mater. 7, 324–332. doi:10.1016/j.bioactmat.2021.05.021
Du, B., Wang, X., Xia, Y., Wu, Y., Wu, B., and Huang, S. (2023). Hygroscopic tunable Multishape memory effect in Cellulosic Macromolecular networks with a Supramolecular Mesophase. ACS Macro Lett. 12, 835–840. doi:10.1021/acsmacrolett.3c00239
Fliefel, R., Popov, C., Tröltzsch, M., Kühnisch, J., Ehrenfeld, M., and Otto, S. (2016). Mesenchymal stem cell proliferation and mineralization but not osteogenic differentiation are strongly affected by extracellular pH. J. Craniomaxillofac Surg. 44, 715–724. doi:10.1016/j.jcms.2016.03.003
Fragal, E. H., Fragal, V. H., Silva, E. P., Paulino, A. T., da Silva Filho, E. C., Mauricio, M. R., et al. (2022). Magnetic-responsive polysaccharide hydrogels as smart biomaterials: Synthesis, properties, and biomedical applications. Carbohydr. Polym. 292, 119665. doi:10.1016/j.carbpol.2022.119665
Galow, A.-M., Rebl, A., Koczan, D., Bonk, S. M., Baumann, W., and Gimsa, J. (2017). Increased osteoblast viability at alkaline pH in vitro provides a new perspective on bone regeneration. Biochem. Biophysics Rep. 10, 17–25. doi:10.1016/j.bbrep.2017.02.001
Gong, S., Lang, S., Wang, Y., Li, X., Tian, A., Ma, J., et al. (2024). pH-responsive mesoporous silica nanoparticles loaded with Naringin for targeted osteoclast inhibition and bone regeneration. Int. J. Nanomedicine 19, 6337–6358. doi:10.2147/IJN.S456545
Hao, Z., Song, Z., Huang, J., Huang, K., Panetta, A., Gu, Z., et al. (2017). The scaffold microenvironment for stem cell based bone tissue engineering. Biomater. Sci. 5, 1382–1392. doi:10.1039/c7bm00146k
Hazehara-Kunitomo, Y., Hara, E. S., Ono, M., Aung, K. T., Komi, K., Pham, H. T., et al. (2019). Acidic pre-Conditioning enhances the stem cell Phenotype of human bone marrow stem/Progenitor cells. Int. J. Mol. Sci. 20, 1097. doi:10.3390/ijms20051097
Hill, C., Altgen, M., Penttila, P., and Rautkari, L. (2024). Review: interaction of water vapour with wood and other hygro-responsive materials. J. Mat. Sci. 59, 7595–7635. doi:10.1007/s10853-024-09636-y
Hu, Z., Lv, X., Zhang, H., Zhuang, S., Zheng, K., Zhou, T., et al. (2024). An injectable gel based on photo-cross-linkable hyaluronic acid and mesoporous bioactive glass nanoparticles for periodontitis treatment. Int. J. Biol. Macromol. 257, 128596. doi:10.1016/j.ijbiomac.2023.128596
Huang, X., Zhang, W., Guan, G., Song, G., Zou, R., and Hu, J. (2017). Design and functionalization of the NIR-responsive photothermal Semiconductor nanomaterials for cancer Theranostics. Acc. Chem. Res. 50, 2529–2538. doi:10.1021/acs.accounts.7b00294
Jiang, Y., Li, W., and Bao, C. (2022). A novel glucose-sensitive scaffold accelerates osteogenesis in diabetic conditions. BioMed Res. Int. 2022, 4133562. doi:10.1155/2022/4133562
Khare, D., Basu, B., and Dubey, A. K. (2020). Electrical stimulation and piezoelectric biomaterials for bone tissue engineering applications. Biomaterials 258, 120280. doi:10.1016/j.biomaterials.2020.120280
Koons, G. L., Diba, M., and Mikos, A. G. (2020). Materials design for bone-tissue engineering. Nat. Rev. Mater 5, 584–603. doi:10.1038/s41578-020-0204-2
Krishna, A. S., Radhakumary, C., and Sreenivasan, K. (2015). Calcium ion modulates protein release from chitosan-hyaluronic acid poly electrolyte gel. Polym. Eng. and Sci. 55, 2089–2097. doi:10.1002/pen.24050
Kroll, A., Ranjan, S., Engqvist, M. K. M., and Lercher, M. J. (2023). A general model to predict small molecule substrates of enzymes based on machine and deep learning. Nat. Commun. 14, 2787. doi:10.1038/s41467-023-38347-2
Lendlein, A., and Gould, O. E. C. (2019). Reprogrammable recovery and actuation behaviour of shape-memory polymers. Nat. Rev. Mater 4, 116–133. doi:10.1038/s41578-018-0078-8
Li, P., and Zhang, X. (2021). Research progress on the effects of microenvironment acid-base level in tissue-engineered bone regeneration. Int. J. STOMATOLOGY 48, 64–70. doi:10.7518/gjkq.2021005
Li, J., Wang, G., Wang, D., Wu, Q., Jiang, X., and Liu, X. (2014). Alkali-treated titanium selectively regulating biological behaviors of bacteria, cancer cells and mesenchymal stem cells. J. Colloid Interface Sci. 436, 160–170. doi:10.1016/j.jcis.2014.08.053
Li, S., Wei, C., and Lv, Y. (2020). Preparation and application of magnetic responsive materials in bone tissue engineering. Curr. Stem Cell Res. Ther. 15, 428–440. doi:10.2174/1574888X15666200101122505
Li, N., Guo, R., and Zhang, Z. J. (2021). Bioink Formulations for bone tissue regeneration. Front. Bioeng. Biotechnol. 9, 630488. doi:10.3389/fbioe.2021.630488
Li, X., Xu, K., He, Y., Tao, B., Li, K., Lin, C., et al. (2022a). ROS-responsive hydrogel coating modified titanium promotes vascularization and osteointegration of bone defects by orchestrating immunomodulation. Biomaterials 287, 121683. doi:10.1016/j.biomaterials.2022.121683
Li, Z., Zhu, Y., and Matson, J. B. (2022b). pH-responsive self-assembling peptide-based biomaterials: Designs and applications. ACS Appl. Bio Mat. 5, 4635–4651. doi:10.1021/acsabm.2c00188
Li, Y., Feng, G., Liu, J., Yang, T., Hou, R., Liu, J., et al. (2023). Progress in glucose-sensitive hydrogels for biomedical applications. Macromol. Chem. Phys. 224, 2300257. doi:10.1002/macp.202300257
Li, N., Wang, J., Feng, G., Liu, Y., Shi, Y., Wang, Y., et al. (2024). Advances in biomaterials for oral-maxillofacial bone regeneration: spotlight on periodontal and alveolar bone strategies. Regen. Biomater. 11, rbae078. doi:10.1093/rb/rbae078
Lin, J.-H., Feng, F., Yu, M.-C., Wang, C.-H., and Chang, P.-C. (2018). Modulation of periodontitis progression using pH-responsive nanosphere encapsulating metronidazole or N-phenacylthialzolium bromide. J. Periodontal Res. 53, 22–28. doi:10.1111/jre.12481
Lin, M., Stehle, Y., Chen, L., Yang, M., Zeng, K., Wang, C., et al. (2025). A 3D-printed chitosan-based pH-responsive dual functional scaffold for osteomyelitis: synergistic antibacterial and osteogenic treatment. Carbohydr. Polym. 366, 123866. doi:10.1016/j.carbpol.2025.123866
Liu, W., Wang, T., Yang, C., Darvell, B. W., Wu, J., Lin, K., et al. (2016). Alkaline biodegradable implants for osteoporotic bone defects--importance of microenvironment pH. Osteoporos. Int. 27, 93–104. doi:10.1007/s00198-015-3217-8
Liu, S., Wang, Y.-N., Ma, B., Shao, J., Liu, H., and Ge, S. (2021). Gingipain-responsive Thermosensitive hydrogel loaded with SDF-1 facilitates in situ periodontal tissue regeneration. ACS Appl. Mater Interfaces 13, 36880–36893. doi:10.1021/acsami.1c08855
Liu, H., Shi, Y., Zhu, Y., Wu, P., Deng, Z., Dong, Q., et al. (2023a). Bioinspired piezoelectric periosteum to Augment bone regeneration via synergistic immunomodulation and osteogenesis. ACS Appl. Mat. Interfaces 15, 12273–12293. doi:10.1021/acsami.2c19767
Liu, J., Cheng, Y., Wang, H., Yang, D., Liu, C., Dou, W., et al. (2023b). Regulation of TiO2 @PVDF piezoelectric nanofiber membranes on osteogenic differentiation of mesenchymal stem cells. Nano Energy 115, 108742. doi:10.1016/j.nanoen.2023.108742
Liu, W., Zhao, H., Zhang, C., Xu, S., Zhang, F., Wei, L., et al. (2023c). In situ activation of flexible magnetoelectric membrane enhances bone defect repair. Nat. Commun. 14, 4091. doi:10.1038/s41467-023-39744-3
Liu, A., Wang, C., Deng, S., Zhang, S., Zhao, Z., Xiao, H., et al. (2025a). Application of light-responsive nanomaterials in bone tissue engineering. Pharmaceutics 17, 98. doi:10.3390/pharmaceutics17010098
Liu, J., Luo, D., Fu, X., Yang, T., Hou, R., Li, P., et al. (2025b). Preparation, Characterization, and in vitro osteogenic properties of a novel glucose-sensitive 3D-printed scaffold containing Metformin based on enzymatic cascade reaction. J. Polym. Sci. 63, 595–609. doi:10.1002/pol.20240561
Liu, J., Wei, J., Xiao, S., Yuan, L., Liu, H., Zuo, Y., et al. (2025c). Multienzyme-activity sulfur quantum dot Nanozyme-Mediated cascade reactions in Whole-stage Symptomatic therapy of infected bone defects. ACS Nano 19, 6858–6875. doi:10.1021/acsnano.4c12343
Liu, S., Lu, M., Zhang, M., Sun, X., Luo, B., and Wu, Y. (2025d). Multifaceted Catalytic glucose Depletion and reactive oxygen species-scavenging Nanoenzyme composite hydrogel for facilitating diabetic bone regeneration. ACS Nano 19, 2677–2694. doi:10.1021/acsnano.4c14596
Liu, X., Schreiber, A. C., Astudillo Potes, M. D., Dashtdar, B., Hamouda, A. M., Rezaei, A., et al. (2025e). Bone enzyme-responsive biodegradable poly(propylene fumarate) and polycaprolactone Polyphosphoester Dendrimer cross-Linked via Click Chemistry for bone tissue engineering. Biomacromolecules 26, 835–847. doi:10.1021/acs.biomac.4c00999
Long, J., Wang, Y., Lu, M., Etxeberria, A. E., Zhou, Y., Gu, P., et al. (2023). Dual-cross-linked magnetic hydrogel with programmed release of parathyroid hormone promotes bone healing. ACS Appl. Mat. Interfaces 15, 35815–35831. doi:10.1021/acsami.3c03047
Lu, Y., Song, J., Lv, Y., Heng, B. C., Xu, M., He, Y., et al. (2025). An Osteoconductive Janus hydrogel with Full Barrier protection and adaptable degradation properties for Superior bone regeneration. Adv. Sci. n/a, e06736. doi:10.1002/advs.202506736
Ma, M., Zou, F., Abudureheman, B., Han, F., Xu, G., Xie, Y., et al. (2023). Magnetic Microcarriers with Accurate Localization and proliferation of mesenchymal stem cell for Cartilage defects repairing. ACS Nano 17, 6373–6386. doi:10.1021/acsnano.2c10995
Ma, G., Xu, K., Yu, L., and Haag, R. (2025). pH-responsive Polyglycerol Nanogels for periodontitis treatment through antibacterial and pro-angiogenesis action. Angew. Chem. Int. Ed. 64, e202418882. doi:10.1002/anie.202418882
Mao, Z., Bi, X., Yu, C., Chen, L., Shen, J., Huang, Y., et al. (2024). Mechanically robust and personalized silk fibroin-magnesium composite scaffolds with water-responsive shape-memory for irregular bone regeneration. Nat. Commun. 15, 4160. doi:10.1038/s41467-024-48417-8
Monfoulet, L.-E., Becquart, P., Marchat, D., Vandamme, K., Bourguignon, M., Pacard, E., et al. (2014). The pH in the microenvironment of human mesenchymal stem cells is a critical factor for optimal osteogenesis in tissue-engineered constructs. Tissue Eng. Part A 20, 1827–1840. doi:10.1089/ten.TEA.2013.0500
Morariu, S. (2023). Advances in the design of phenylboronic acid-based glucose-sensitive hydrogels. Polymers 15, 582. doi:10.3390/polym15030582
Mouthuy, P.-A., Snelling, S. J. B., Dakin, S. G., Milković, L., Gašparović, A. Č., Carr, A. J., et al. (2016). Biocompatibility of implantable materials: an oxidative stress viewpoint. Biomaterials 109, 55–68. doi:10.1016/j.biomaterials.2016.09.010
Nain, A., Chakraborty, S., Barman, S. R., Gavit, P., Indrakumar, S., Agrawal, A., et al. (2024). Progress in the development of piezoelectric biomaterials for tissue remodeling. Biomaterials 307, 122528. doi:10.1016/j.biomaterials.2024.122528
Nicosia, A., Salamone, M., Costa, S., Ragusa, M. A., and Ghersi, G. (2023). Mimicking molecular pathways in the design of smart hydrogels for the design of Vascularized engineered tissues. Int. J. Mol. Sci. 24, 12314. doi:10.3390/ijms241512314
Peng, C., Wang, G., Li, J., Wang, Y., Shu, Z., Tang, M., et al. (2024). Ros-responsive and scavenging bifunctional hydrogel enables co-delivery of anti-inflammatory agent and osteogenetic nanoparticle for periodontitis treatment. Mater. and Des. 239, 112777. doi:10.1016/j.matdes.2024.112777
Pfeiffenberger, M., Damerau, A., Lang, A., Buttgereit, F., Hoff, P., and Gaber, T. (2021). Fracture healing research—Shift towards in vitro Modeling? Biomedicines 9, 748. doi:10.3390/biomedicines9070748
Pj, S. N. van G., G, C., and Carmeliet, G. (2016). Targeting the hypoxic response in bone tissue engineering: a balance between supply and consumption to improve bone regeneration. Mol. Cell. Endocrinol. 432, 96–105. doi:10.1016/j.mce.2015.12.024
Ren, X., Liu, H., Wu, X., Weng, W., Wang, X., and Su, J. (2022). Reactive oxygen species (ROS)-Responsive biomaterials for the treatment of bone-related diseases. Front. Bioeng. Biotechnol. 9, 820468. doi:10.3389/fbioe.2021.820468
Ricotti, L., Cafarelli, A., Manferdini, C., Trucco, D., Vannozzi, L., Gabusi, E., et al. (2024). Ultrasound stimulation of piezoelectric nanocomposite hydrogels Boosts Chondrogenic differentiation in vitro, in both a Normal and inflammatory Milieu. ACS Nano 18, 2047–2065. doi:10.1021/acsnano.3c08738
Roldan, L., Montoya, C., Solanki, V., Cai, K. Q., Yang, M., Correa, S., et al. (2023). A novel injectable piezoelectric hydrogel for periodontal disease treatment. ACS Appl. Mat. Interfaces 15, 43441–43454. doi:10.1021/acsami.3c08336
Sha, Z., Wu, Y., Zheng, Y., Yang, K., Gong, X., Xuan, L., et al. (2025). Advances in pH-responsive drug delivery systems for periodontitis treatment. Drug Deliv. 32, 2522109. doi:10.1080/10717544.2025.2522109
Shahabipour, F., Satta, S., Mahmoodi, M., Sun, A., de Barros, N. R., Li, S., et al. (2023). Engineering organ-on-a-chip systems to model viral infections. Biofabrication 15, 022001. doi:10.1088/1758-5090/ac6538
Shang, J., Le, X., Zhang, J., Chen, T., and Theato, P. (2019). Trends in polymeric shape memory hydrogels and hydrogel actuators. Polym. Chem. 10, 1036–1055. doi:10.1039/c8py01286e
Shen, J., He, R., He, J., Liao, L., Huang, Y., Min, S., et al. (2023). Oscillating magnetic field induced bone injury repair by using drug-free Micromotors. Adv. Sci. n/a, e03254. doi:10.1002/advs.202503254
Shuai, C., Wang, Z., Peng, S., Shuai, Y., Chen, Y., Zeng, D., et al. (2022). Water-responsive shape memory thermoplastic polyurethane scaffolds triggered at body temperature for bone defect repair. Mat. Chem. Front. 6, 1456–1469. doi:10.1039/D1QM01635K
Singh, N., Dhanya, B. S., and Verma, M. L. (2020). Nano-immobilized biocatalysts and their potential biotechnological applications in bioenergy production. Mater. Sci. Energy Technol. 3, 808–824. doi:10.1016/j.mset.2020.09.006
Tao, H., Ge, G., Liang, X., Zhang, W., Sun, H., Li, M., et al. (2020). ROS signaling cascades: dual regulations for osteoclast and osteoblast. Acta Biochim. Biophys. Sin. (Shanghai) 52, 1055–1062. doi:10.1093/abbs/gmaa098
Tapponi, S., Yusuf, A., Alsaafin, F., and Hussain, Z. (2025). Breaking barriers with pH-responsive nanocarriers: a new frontier in precision oncology. Int. J. Pharm. 682, 125931. doi:10.1016/j.ijpharm.2025.125931
Tian, Y., Li, Y., Liu, J., Lin, Y., Jiao, J., Chen, B., et al. (2022). Photothermal therapy with regulated Nrf2/NF-κB signaling pathway for treating bacteria-induced periodontitis. Bioact. Mater 9, 428–445. doi:10.1016/j.bioactmat.2021.07.033
Tyagi, N., Gambhir, K., Kumar, S., Gangenahalli, G., and Verma, Y. K. (2021). Interplay of reactive oxygen species (ROS) and tissue engineering: a review on clinical aspects of ROS-responsive biomaterials. J. Mater Sci. 56, 16790–16823. doi:10.1007/s10853-021-06338-7
Uchino, K. (2017). Chapter 1 - the Development of piezoelectric materials and the new perspective, in Advanced Piezoelectric Materials (Second Edition), Editor K. Uchino Second Edition (New Delhi, India: Woodhead Publishing), 1–92. doi:10.1016/B978-0-08-102135-4.00001-1
Unruh, R. M., Roberts, J. R., Nichols, S. P., Gamsey, S., Wisniewski, N. A., and McShane, M. J. (2015). Preclinical evaluation of poly(HEMA-co-acrylamide) hydrogels encapsulating glucose oxidase and Palladium Benzoporphyrin as Fully implantable glucose sensors. J. Diabetes Sci. Technol. 9, 985–992. doi:10.1177/1932296815590439
Wang, L., Yang, H., Zhang, C., Zhang, Y., He, Y., Liu, Y., et al. (2024). A blood glucose fluctuation-responsive delivery system promotes bone regeneration and the repair function of Smpd3-reprogrammed BMSC-derived exosomes. Int. J. Oral Sci. 16, 65–15. doi:10.1038/s41368-024-00328-6
Wang, H., Zhang, J., Li, Z., Liu, J., Chang, H., Jia, S., et al. (2025a). NIR-programmable 3D-printed shape-memory scaffold with dual-thermal responsiveness for precision bone regeneration and bone tumor management. J. Nanobiotechnology 23, 300. doi:10.1186/s12951-025-03375-1
Wang, Z., Huang, Y., He, S., Li, M., Gong, J., Cheng, L., et al. (2025b). Oxygen-independent sulfate radical and Fe2+-modified implants for Fast sterilization and osseointegration of infectious bone defects. ACS Nano 19, 18804–18823. doi:10.1021/acsnano.5c04147
Wang, Z., Ma, D., Liu, J., Xu, S., Qiu, F., Hu, L., et al. (2025c). 4D printing polymeric biomaterials for adaptive tissue regeneration. Bioact. Mater 48, 370–399. doi:10.1016/j.bioactmat.2025.01.033
Wang, Z., Xie, D., Wu, D., Luo, X., Wang, S., Li, Y., et al. (2025d). Robust enzyme discovery and engineering with deep learning using CataPro. Nat. Commun. 16, 2736. doi:10.1038/s41467-025-58038-4
Wei, H., Cui, J., Lin, K., Xie, J., and Wang, X. (2022). Recent advances in smart stimuli-responsive biomaterials for bone therapeutics and regeneration. Bone Res. 10, 17–19. doi:10.1038/s41413-021-00180-y
Wu, Y.-Y., Xiao, E., and Graves, D. T. (2015). Diabetes mellitus related bone metabolism and periodontal disease. Int. J. Oral Sci. 7, 63–72. doi:10.1038/ijos.2015.2
Wu, P., Shen, L., Liu, H.-F., Zou, X.-H., Zhao, J., Huang, Y., et al. (2023). The marriage of immunomodulatory, angiogenic, and osteogenic capabilities in a piezoelectric hydrogel tissue engineering scaffold for military medicine. Mil. Med. Res. 10, 35. doi:10.1186/s40779-023-00469-5
Wu, H., Chen, C., Li, J., Yu, D., Wu, X., Huang, H., et al. (2024). Engineered Magneto-piezoelectric nanoparticles-enhanced scaffolds disrupt biofilms and activate oxidative Phosphorylation in Icam1+ macrophages for infectious bone defect regeneration. ACS Nano 18, 35575–35594. doi:10.1021/acsnano.4c13562
Xu, A., Zhuang, C., Xu, S., He, F., Xie, L., Yang, X., et al. (2018). Optimized bone regeneration in Calvarial bone defect based on Biodegradation-Tailoring dual-shell Biphasic bioactive Ceramic Microspheres. Sci. Rep. 8, 3385. doi:10.1038/s41598-018-21778-z
Xu, Y., Luo, Y., Weng, Z., Xu, H., Zhang, W., Li, Q., et al. (2023). Microenvironment-responsive metal-phenolic nanozyme release Platform with antibacterial, ROS scavenging, and osteogenesis for periodontitis. ACS Nano 17, 18732–18746. doi:10.1021/acsnano.3c01940
Xu, J., Zhou, F., Cao, L., Wang, H., Hu, J., Qiu, S., et al. (2025). ROS-balancing-engineered bio-heterojunction hydrogel accelerated the infected bone regeneration based on Sono-chemo dynamic therapy. Bioact. Mater. 52, 440–459. doi:10.1016/j.bioactmat.2025.05.031
Yan, J., Chen, H., Pan, Y., Yan, Y., Tang, S., Zhou, Q., et al. (2025). Magnetic labeling of physically tunable hydrogel-induced mesenchymal stem cell spheroids with IONPs for MRI tracking and bone regeneration. Nano Today 61, 102620. doi:10.1016/j.nantod.2024.102620
Yang, M., Cai, X., Wang, C., Li, P., Chen, S., Liu, C., et al. (2024). Humidity-responsive amorphous calcium–magnesium pyrophosphate/cassava starch scaffold for enhanced Neurovascular bone regeneration. ACS Appl. Mat. Interfaces 16, 35964–35984. doi:10.1021/acsami.4c03204
Yin, Y., Yang, J., Pan, Y., Gao, Y., Huang, L., Luan, X., et al. (2021). Mesopore to Macropore transformation of metal–Organic framework for drug delivery in inflammatory Bowel disease. Adv. Healthc. Mater. 10, 2000973. doi:10.1002/adhm.202000973
Yousefiasl, S., Zare, I., Ghovvati, M., and Ghomi, M. (2023). “Enzyme-responsive materials: properties, design, and applications,” in Stimuli-responsive materials for biomedical applications (American Chemical Society), 203–229. doi:10.1021/bk-2023-1436.ch009
Yu, M., Wang, D., Zhong, D., Xie, W., and Luo, J. (2022). Adropin carried by reactive oxygen species-responsive nanocapsules ameliorates renal lipid toxicity in diabetic mice. ACS Appl. Mat. Interfaces 14, 37330–37344. doi:10.1021/acsami.2c06957
Yu, C., Yang, W., Yang, L., Ye, L., Sun, R., Gu, T., et al. (2023a). Synergistic effect of magneto-mechanical bioengineered stem cells and magnetic field to alleviate Osteoporosis. ACS Appl. Mat. Interfaces 15, 19976–19988. doi:10.1021/acsami.3c01139
Yu, P., Li, Y., Sun, H., Zhang, H., Kang, H., Wang, P., et al. (2023b). Mimicking Antioxidases and hyaluronan Synthase: a Zwitterionic nanozyme for photothermal therapy of Osteoarthritis. Adv. Mater 35, e2303299. doi:10.1002/adma.202303299
Yuan, T., Tan, M., Xu, Y., Xiao, Q., Wang, H., Wu, C., et al. (2023). All-in-one smart dressing for simultaneous angiogenesis and neural regeneration. J. Nanobiotechnology 21, 38. doi:10.1186/s12951-023-01787-5
Yue, X., Sun, X., Li, Z., Wang, T., Kong, W., Li, S., et al. (2025). Biomimetic piezoelectric periosteum-bone integrated implant promotes bone defect repair by remodeling osteogenic microenvironment. Adv. Funct. Mater. n/a, 2423492. doi:10.1002/adfm.202423492
Zha, K., Hu, W., Xiong, Y., Zhang, S., Tan, M., Bu, P., et al. (2024). Nanoarchitecture-integrated hydrogel Boosts angiogenesis–osteogenesis–Neurogenesis Tripling for infected bone fracture healing. Adv. Sci. 11, 2406439. doi:10.1002/advs.202406439
Zhang, J., Sun, Y., Li, X., and Xu, J. (2019a). Fabrication of porous NiMn2O4 nanosheet arrays on nickel foam as an advanced sensor material for non-enzymatic glucose detection. Sci. Rep. 9, 18121. doi:10.1038/s41598-019-54746-2
Zhang, J., Zhou, W., Wang, H., Lin, K., and Chen, F. (2019b). 3D-printed surface promoting osteogenic differentiation and angiogenetic factor expression of BMSCs on Ti6Al4V implants and early osseointegration in vivo. J. Mater. Sci. and Technol. 35, 336–343. doi:10.1016/j.jmst.2018.09.063
Zhang, Y., Hu, J., Zhao, X., Xie, R., Qin, T., and Ji, F. (2019c). Mechanically robust shape memory polyurethane nanocomposites for minimally invasive bone repair. ACS Appl. Bio Mat. 2, 1056–1065. doi:10.1021/acsabm.8b00655
Zhang, K., Li, Y., and Yang, Y. (2020). Influence of the low voltage pulsed magnetic field on the columnar-to-equiaxed transition during directional solidification of superalloy K4169. J. Mater. Sci. and Technol. 48, 9–17. doi:10.1016/j.jmst.2020.02.009
Zhang, Y., Li, C., Zhang, W., Deng, J., Nie, Y., Du, X., et al. (2022a). 3D-printed NIR-responsive shape memory polyurethane/magnesium scaffolds with tight-contact for robust bone regeneration. Bioact. Mater. 16, 218–231. doi:10.1016/j.bioactmat.2021.12.032
Zhang, Y., Zhang, C., Wang, R., Tan, W., Gu, Y., Yu, X., et al. (2022b). Development and challenges of smart actuators based on water-responsive materials. Soft Matter 18, 5725–5741. doi:10.1039/d2sm00519k
Zhang, D., Xu, D., Huang, Q., Luo, L., Bian, H., Chen, D., et al. (2024a). Nanomaterial-based STING inhibition for accelerating bone defect repair via photothermal-responsive hydrogel delivery. Nano Res. 18, 94907481. doi:10.26599/NR.2025.94907481
Zhang, J., Zhuang, Y., Sheng, R., Tomás, H., Rodrigues, J., Yuan, G., et al. (2024b). Smart stimuli-responsive strategies for titanium implant functionalization in bone regeneration and therapeutics. Mater. Horizons 11, 12–36. doi:10.1039/D3MH01260C
Zhang, M., Yu, T., Li, J., Yan, H., Lyu, L., Yu, Y., et al. (2024c). Matrix metalloproteinase-responsive hydrogel with on-Demand release of Phosphatidylserine promotes bone regeneration through immunomodulation. Adv. Sci. 11, 2306924. doi:10.1002/advs.202306924
Zhang, Q., Chen, W., Li, G., Ma, Z., Zhu, M., Gao, Q., et al. (2024d). A factor-free hydrogel with ROS scavenging and responsive degradation for enhanced diabetic bone healing. Small 20, 2306389. doi:10.1002/smll.202306389
Zhou, J., Fang, C., Rong, C., Luo, T., Liu, J., and Zhang, K. (2023a). Reactive oxygen species-sensitive materials: a promising strategy for regulating inflammation and favoring tissue regeneration. Smart Mater. Med. 4, 427–446. doi:10.1016/j.smaim.2023.01.004
Zhou, Y., Liu, J., Xue, P., and Zhang, J. (2023b). Collagenase-responsive hydrogel loaded with GSK2606414 nanoparticles for periodontitis treatment through inhibiting inflammation-induced expression of PERK of periodontal Ligament stem cells. Pharmaceutics 15, 2503. doi:10.3390/pharmaceutics15102503
Zhou, S., Xiao, C., Fan, L., Yang, J., Ge, R., Cai, M., et al. (2024a). Injectable ultrasound-powered bone-adhesive nanocomposite hydrogel for electrically accelerated irregular bone defect healing. J. Nanobiotechnology 22, 54. doi:10.1186/s12951-024-02320-y
Zhou, X., Jiang, J., Dang, J., Wang, Y., Hu, R., Shen, C., et al. (2024b). Intelligent Supramolecular modification for implants: endogenous regulation of bone defect repair in Osteoporosis. Adv. Mater. 36, 2406227. doi:10.1002/adma.202406227
Zhu, P., Chen, Y., and Shi, J. (2020). Piezocatalytic tumor therapy by ultrasound-triggered and BaTiO3-Mediated piezoelectricity. Adv. Mater. 32, 2001976. doi:10.1002/adma.202001976
Zhu, C., He, M., Wang, J., Huang, Y., Deng, W., Liu, L., et al. (2024). Bioactivated polyetheretherketone scaffold able to generate mild heat for promoting bone regeneration via activating MAPK/ERK signaling pathway. J. Mater. Sci. and Technol. 190, 188–199. doi:10.1016/j.jmst.2023.12.033
Keywords: smart materials, stimuli-responsive materials, bone tissue engineering, oral-maxillofacial bone, bone repair and regeneration
Citation: Yu Y, Liu Z, Qin X, Song K and Xu L (2025) Smart materials: innovative strategies for oral-maxillofacial bone defects repair. Front. Bioeng. Biotechnol. 13:1629292. doi: 10.3389/fbioe.2025.1629292
Received: 15 May 2025; Accepted: 11 August 2025;
Published: 26 August 2025.
Edited by:
Qianju Wu, Xiamen Stomatological Hospital, ChinaReviewed by:
Zhennan Deng, Wenzhou Medical University, ChinaZuodong Zhao, University Hospitals Leuven, Belgium
Copyright © 2025 Yu, Liu, Qin, Song and Xu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ke Song, c29uZ2tlX2NvY29AMTYzLmNvbQ==; Lianyi Xu, bHl4dV90akB0amgudGptdS5lZHUuY24=
 Yilin Yu
Yilin Yu Zhenyuan Liu2
Zhenyuan Liu2 Ke Song
Ke Song Lianyi Xu
Lianyi Xu