- 1School of Clinical Medicine, Chengdu Medical College, Chengdu, China
- 2Department of Stomatology, The First Affiliated Hospital of Chengdu Medical College, Chengdu, China
Periodontitis is a chronic inflammatory disorder that causes irreversible damage to periodontal tissues, such as the alveolar bone, cementum, and periodontal ligament. Clinical treatments for periodontitis include non-surgical and surgical approaches, complemented by the utilization of various biomimetic materials to repair damaged tissues, a process known as periodontal tissue regeneration. Biomimetic materials have demonstrated extensive application prospects in periodontal tissue regeneration owing to their outstanding biocompatibility, controllable mechanical properties, and biomimetic microstructures. This review begins by delineating the cells used in periodontal tissue regeneration and summarizes the applications and research hot spots of commonly employed biomaterials in periodontal tissues. This further highlights the advantages of using biomimetic materials as bone substitutes, barrier membranes, and complex scaffold-manufacturing techniques. In addition, an in-depth discussion is conducted on their biological safety and physical properties. Moreover, this article summarizes the research advancements of extracellular matrix (ECM) materials in periodontal tissues, which hold great promise as highly prospective biomaterials for periodontal tissue regeneration. Finally, this article provides a comprehensive summary of the current limitations and challenges associated with the application of biomaterials in periodontal tissue regeneration. Although biomimetic materials still encounter challenges regarding stability and long-term efficacy during clinical translation, this review offers insights into the basic research and clinical treatment of periodontal regeneration.
1 Introduction
Periodontitis is a highly prevalent chronic inflammatory disorder that mainly affects the alveolar bone, cementum, and periodontal ligament, which are crucial tissues that support the tooth (Peng et al., 2024). Globally, approximately 743 million individuals are affected, with a higher prevalence in low-income groups and older populations (Eke et al., 2020; Papapanou and Susin, 2017). Approximately 42% of adults aged 30 years or older suffer from periodontal disease, and nearly 19% of the global adult population has severe periodontitis (Jain et al., 2024). Chronic periodontitis, characterized by excessive inflammatory responses, can lead to the progressive destruction of tooth-supporting structures, including the gums, periodontal ligament, and alveolar bone, ultimately resulting in tooth loss (Hajishengallis and Chavakis, 2021) (Figure 1). Conventional periodontal treatments, such as scaling, root planing, and open-flap debridement, aim to halt the progression of periodontitis; impaired periodontal tissues and alveolar bone remain largely irrecoverable following these interventions (Liang Y. et al., 2020; Yamada et al., 2022).
Periodontal tissues have diverse cell compositions and individualized tissue structures (Figures 2A,B). The key challenge in periodontal treatment is the restoration of the physiological structure and function of periodontal tissues. Techniques, such as guided tissue regeneration (GTR) and guided bone regeneration (GBR), show promise in repairing periodontal tissues and addressing alveolar bone defects (Abdo et al., 2023; Bottino et al., 2011; Ho et al., 2022). Current clinical approaches include GTR, bone grafting, and application of growth factors. However, these methods often suffer from limitations, such as poor predictability and restricted indications, resulting in unsatisfactory outcomes (Wu et al., 2025). Biomimetic materials can provide cells with a wide range of biochemical and biophysical signals that simulate the extracellular matrix (ECM) of the body. Moreover, these materials possess mechanical adaptability, microstructure connectivity, and intrinsic biological activity, making them an ideal choice for designing living implants for specific applications, covering the fields of tissue engineering and regenerative medicine (Liu et al., 2023). Scaffolds fabricated from biomimetic materials play a critical role in guiding cell proliferation and the formation of new tissue. Single-layer scaffolds, which consist of a uniform material structure, are inadequate for mimicking the complex biological environment of native tissues. In contrast, multi-layer scaffolds constructed from biomimetic materials can more effectively replicate the morphological architecture of tissues and organs, offering distinct advantages in the repair and regeneration of bone cartilage, skin, blood vessels, and various other tissue types (Bertsch et al., 2023). Due to the superior performance of biomimetic materials, a wide range of such materials has been approved for clinical use by the Food and Drug Administration (FDA) in tissue engineering, regeneration, and other biomedical applications (Table 1).
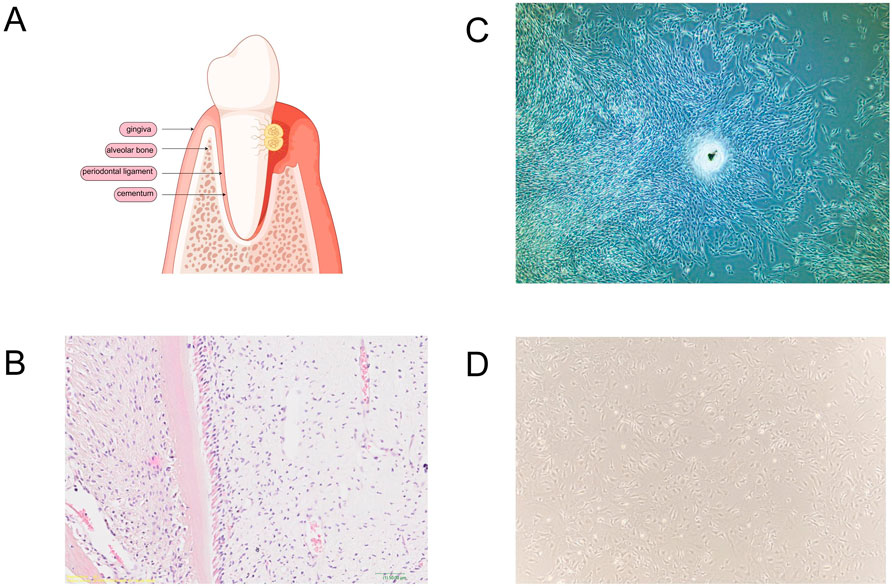
Figure 2. Periodontal tissues and cells suitable for periodontal tissue regeneration. (A) Tissue destruction of periodontal tissue and periodontitis. (B) Periodontal tissues of SD rats were stained with hematoxylin and eosin (H&E) for histological examination. (C) Dental follicle stem cells from beagle dogs. (D) Bone marrow-derived mesenchymal stem cells from C57 mice.
With the application of tissue engineering technology in organ regeneration, biomimetic materials have been widely used for repairing tissue defects. Research into biomimetic materials holds the potential for developing new diagnostic and therapeutic strategies, enhancing the regeneration of damaged periodontal tissues, and providing avenues for restoring periodontal structure and function. This study reviewed eligible publications, identified based on inclusion/exclusion criteria, over a range of 29 years (from 1997 to 2025). This manuscript enumerates the cells utilized in periodontal regeneration research and presents a comprehensive review of the applications of biomaterials in periodontal regeneration. This study provides a theoretical foundation for basic research on periodontal tissue regeneration, thereby facilitating its clinical applications.
2 Stem cells used in periodontal tissue regeneration
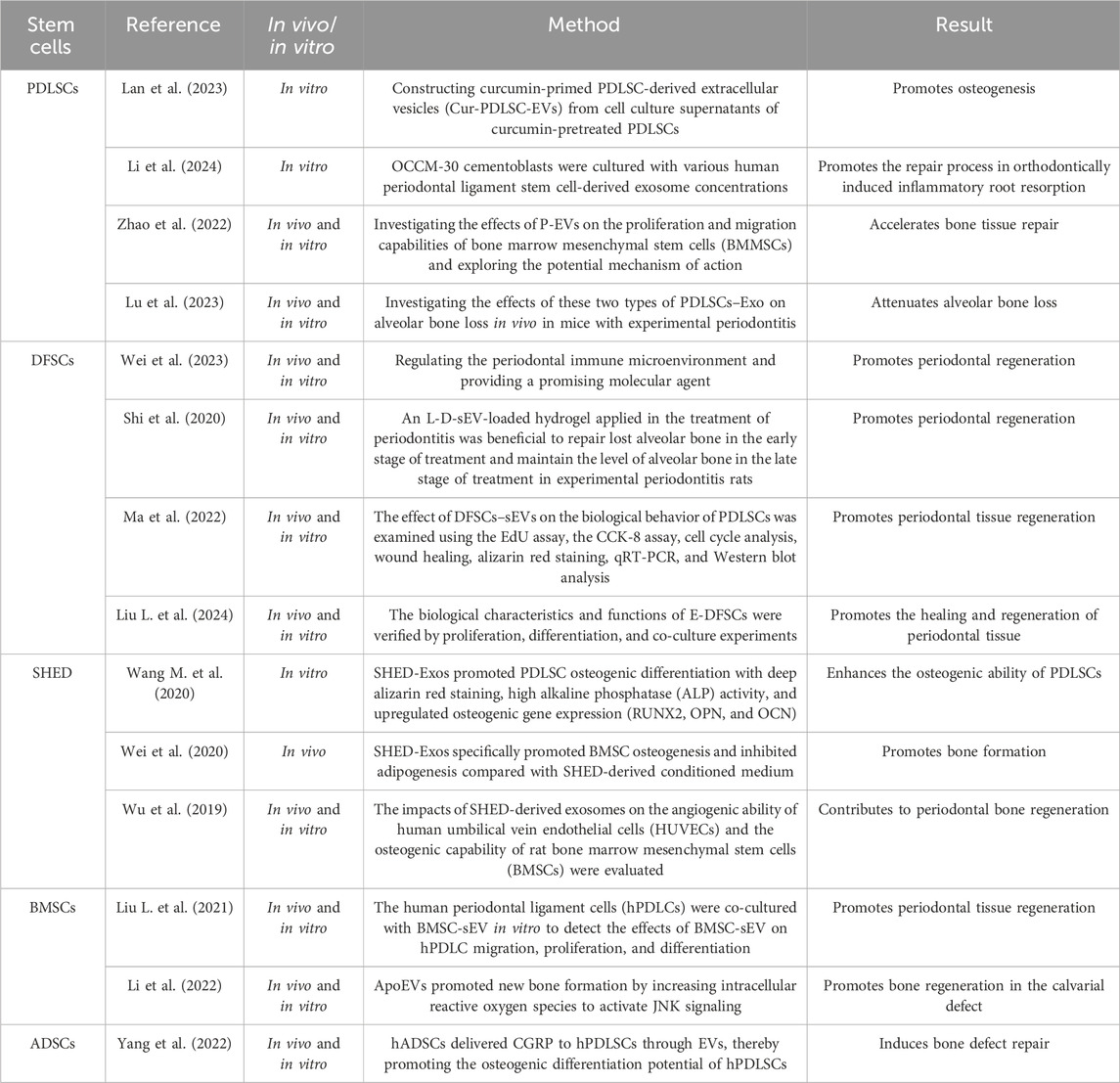
Tissue engineering involves three key components: cells, scaffolds, and bioactive factors. Cells that secrete bioactive factors play critical roles in tissue repair and regeneration. Various dental-derived stem cells have been investigated for their potential in periodontal tissue regeneration, along with cells from other tissue sources, which are also of great significance in this field. Numerous types of stem cells have been investigated in animal models to assess their potential for periodontal tissue regeneration. This section focuses on the role of these cells in periodontal tissue regeneration (Table 2).
2.1 Periodontal ligament stem cells
A distinct population of multipotent postnatal stem cells resides in the periodontal ligament, where they contribute to tissue homeostasis and regeneration. Periodontal ligament stem cells (PDLSCs) were initially isolated using a range of scientifically validated techniques and comprehensively characterized by Seo et al. (2004) and Trubiani et al. (2005). PDLSCs are somatic stem cells with the potential to differentiate into multiple cell types and undergo robust clonal self-renewal (Coura et al., 2008; Dapeng et al., 2014; Li et al., 2014).
2.2 Dental follicle stem cells
Dental follicle stem cells (DFSCs), originating from the neural crest and located in the dental follicle tissue of the tooth germ, serve as direct precursors to periodontal tissues (Figure 2C) (Zhang et al., 2019). These multipotent stem cells differentiate into osteoblasts. Initially identified by Morsczeck et al. (2005), DFSCs are precursor cells for periodontal tissues. They can develop into the periodontal ligament, cementum, and alveolar bone under specific conditions during the late stage of tooth development (Liu L. et al., 2024; Shi et al., 2020).
2.3 Stem cells from human exfoliated deciduous teeth
Stem cells from human exfoliated deciduous teeth (SHED) are a highly proliferative and clonogenic cell population capable of differentiating into various cell types, including neural cells, adipocytes, and odontoblasts (Yan et al., 2021). Miura first discovered that exfoliated human deciduous teeth contain these multipotent stem cells (Miura et al., 2003). SHED-derived exosomes combined with β-tricalcium phosphate (TCP) promote alveolar bone regeneration by enhancing angiogenesis and osteogenesis (Wu et al., 2019). Owing to their multipotent nature, similar to that of mesenchymal stem cells (MSCs), SHED hold significant promise in regenerative medicine (Mohd Nor et al., 2023).
2.4 Bone mesenchymal stem cells
Multipotent stem cells reside in both the adipose tissue and bone marrow, with bone mesenchymal stem cells (BMSCs) being predominant (Figure 2D) (Luo P. et al., 2023). These adult stem cells exhibit multi-differentiation, self-renewal, migration, and colonization abilities (Hade et al., 2021). As initiator cells, BMSCs promote periodontal tissue regeneration and possess immunoregulatory properties crucial for the periodontal inflammatory response (Song et al., 2007; Zhang et al., 2018). In dental applications, BMSCs have demonstrated significant efficacy in periodontal regeneration (Kawaguchi et al., 2004).
2.5 Adipose-derived stem cells
Adipose-derived stem cells (ADSCs) are a population of multipotent stromal cells extracted from the SVF of subcutaneous adipose tissue. These cells exhibit a self-renewal capacity and the potential to differentiate into diverse cell lineages, making them attractive candidates for tissue repair and regeneration (Gou et al., 2024; Sándor et al., 2014; Si et al., 2019). ADSCs exert their therapeutic effects by releasing paracrine factors and extracellular vesicles (EVs) that modulate the local microenvironment and stimulate endogenous regenerative processes (Zhang et al., 2020). Numerous studies have reported the successful application of ADSCs in the regeneration of the alveolar bone, a critical component of the oral and maxillofacial complex (Lemaitre et al., 2017; Yang et al., 2022; Zhang et al., 2025).
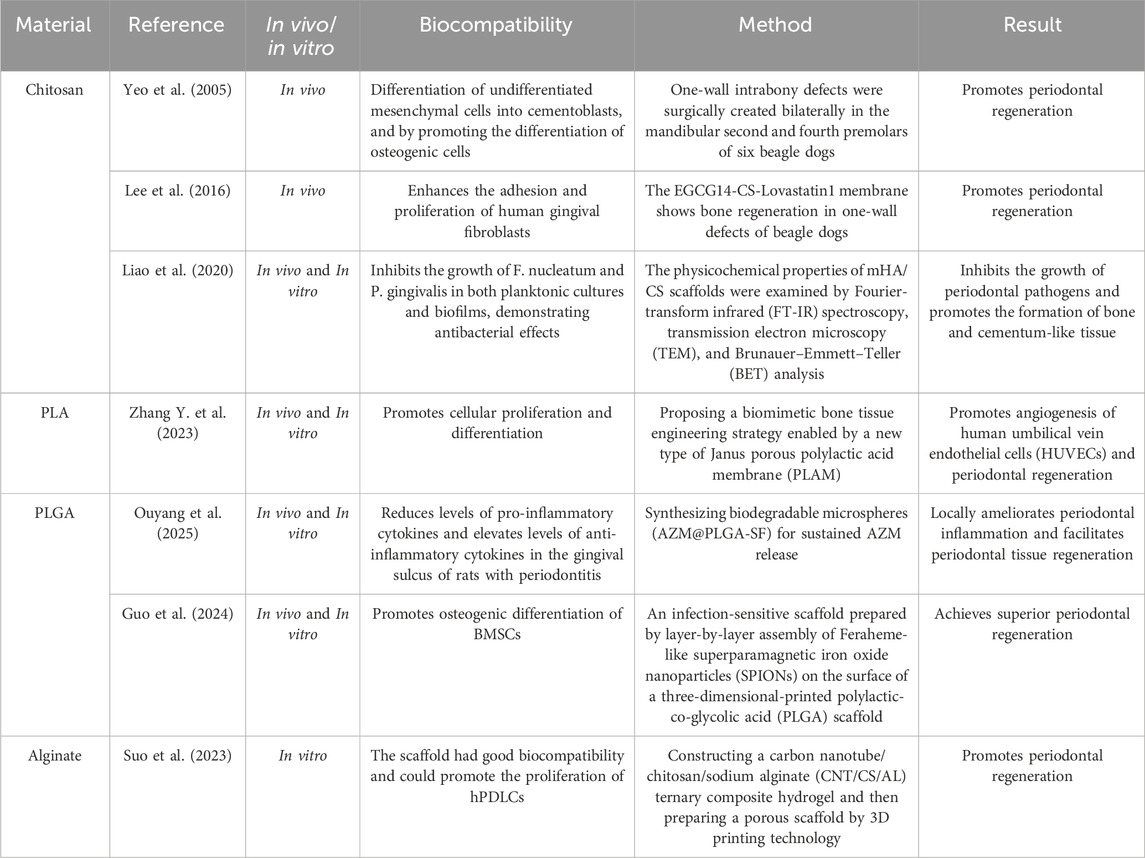
3 Biomimetic materials used in periodontal tissue regeneration
Bioactive nanomaterials have significantly transformed the field of bone tissue regeneration, effectively addressing the limitations of autogenous and allogeneic grafts (Figure 3). Engineered scaffolds and nanocomposites with high biocompatibility, such as calcium phosphate (CaP) composites, thermosensitive hydrogels, and hydroxyapatite, play crucial roles in facilitating bone repair (Hao et al., 2024). The micro and nanoscale characteristics of natural bone, including porosity, surface topography, and fiber alignment, necessitate reevaluation in order to design scaffolds that effectively stimulate tissue growth (Scott et al., 2013). An interconnected 3D pore architecture is essential for bone tissue scaffolds as it facilitates adequate cell accommodation, the migration of osteoprogenitor cells and immune cells, vascularization, and innervation. The optimal size range for micropores to promote cellular infiltration and attachment within bone tissues is generally considered to be between 50 and 150 µm (Yan et al., 2024). In contrast, macropores ranging from 100 to 600 µm provide enhanced integration with host bone tissue while supporting vascularization and new bone formation (Zhu et al., 2020).
3.1 CaP biomaterials
CaP-based materials have garnered considerable interest in implant dentistry because of their superior osteoconductivity and bone-regenerative capabilities (Huang et al., 2017; Yang et al., 2011). Their chemical and structural resemblance to natural bone makes them ideal for repairing bone defects (Mohd Pu’ad et al., 2019). Following alkali treatment, the scaffolds were immersed in a pre-established solution to allow coating formation. The presence of nanostructured F/CaP coating led to a marked upregulation of osteogenic genes and attenuated bacterial growth. While human periodontal ligament stem cells (hPDLSCs) on the scaffolds with 500 μm strand spacing showed high expression of osteogenic markers (runt-related transcription factor 2, Runx2) over 21 days, cells on randomly oriented fibrous scaffolds showed upregulation of M1 markers (Daghrery et al., 2023). In vivo findings confirmed that F/CaP-coated scaffolds were biocompatible and led to periodontal regeneration when implanted in a rat mandibular periodontal fenestration defect model (Daghrery et al., 2021). The biodegradation of calcium phosphate is influenced by various factors, including porosity, the extent of bone contact, specific surface area, bone type, and animal species (Lu et al., 2002). This section reviews the recent developments, characterization, and biological performance of different CaP-based materials for periodontal tissue regeneration (Table 3).
3.1.1 Hydroxyapatite
Hydroxyapatite (HA) has been a focal point of research since the 1980s because of its chemical resemblance to bone minerals, notable biological activity, and osteoconductivity (Dunaev et al., 2014; Lei et al., 2017). Stoichiometric HA features a dense crystalline network of PO4 tetrahedra, each centered around a P5+ ion coordinated by four oxygen atoms. These tetrahedra form interconnected columns, resulting in two distinct types of unconnected channels within the lattice. HA is commonly synthesized via methods such as solution–precipitation and sol–gel (Ben-Arfa et al., 2017; Mirzaee et al., 2016). These characteristics render HA a preferred bioceramic for periodontal bone tissue repair. Porous chitosan (CS) membranes were fabricated with or without HA using the simple freeze gelation (FG) technique with two different solvent systems: acetic acid (ACa) and ascorbic acid (ASa). Fourier-transform infrared (FT-IR) spectroscopy confirmed the presence of hydroxyapatite and its interactions with chitosan. All the membranes supported cell proliferation, and long-term matrix deposition was supported by the HA-incorporated membranes. These CS and HA composite membranes have potential for use in GTR for periodontal lesions (Qasim et al., 2015). Small intestinal submucosa (SIS) was coated with hydroxyapatite (SIS-HA) and gelatin methacrylate hydroxyapatite (SIS-Gel-HA) using biomineralization and chemical crosslinking. SIS was found to upregulate M2 marker expression, and both SIS-HA and SIS-Gel-HA enhanced the osteogenic differentiation of PDLSCs through the bone morphogenetic protein (BMP)-2/Smad signaling pathway, demonstrating superior in vitro osteogenic activity. In vivo, SIS-HA and SIS-Gel-HA yielded denser and more mature bone, with the highest BMP-2 and Smad expression. SIS-HA and SIS-Gel-HA demonstrated enhanced immunosteogenesis coupling, representing a promising periodontal tissue regeneration approach (Cheng et al., 2024). HA bioceramics with micro-nano-hybrid surfaces (mnHA [a hybrid of nanorods and microrods]) can promote cell adhesion, proliferation, alkaline phosphatase (ALP) activity, and expression of osteogenic/cementogenic-related markers, including Runx2, ALP, osteocalcin (OCN), cementum attachment protein (CAP), and cementum protein (CEMP). Various strategies have been employed to fabricate scaffolds with differing pore parameters (Carotenuto et al., 2022).
The TIPS technique, supported by the salt-leaching process, was employed to investigate the influence of HA content in PLLA/HA scaffolds on various properties, including pore structure, density, porosity, mechanical characteristics, and osteoblast proliferation (Zeinali et al., 2021). It was found that an increased HA content within the scaffold (up to 75 wt%) correlated with enhanced roughness of the pore wall surfaces, elevated porosity (96%–98%), and a higher proliferation rate of osteoblast cells. A more recent study conducted by the same research group demonstrated that apatite whiskers (HAP) completely covered the pore wall surfaces of PCL/HAP scaffolds, resulting in significantly increased surface roughness (Petersen et al., 2018). These scaffolds exhibited high porosity (approximately 90%) along with heterogeneous pores characterized by two distinct size ranges: larger pores with diameters of up to 600 µm and smaller pores located within micropore walls measuring up to 50 µm. Moreover, L-lysine modification of hydroxyapatite enhances the bioactivity of PCL/HAP scaffolds by promoting osteoblast proliferation and differentiation while simultaneously improving mechanical properties and increasing surface roughness of the wall pores. Compared to smooth surfaces, it is suggested that a rough topography may more effectively mimic the mineralized interface encountered by cells adhered to the native bone ECM. These results suggest that HA bioceramics containing mnHA are promising grafts for periodontal tissue regeneration (Mao et al., 2015).
3.1.2 TCP
TCP, a well-studied calcium phosphate, exists in three polymorphs: α, β, and α′, each stable at different temperatures and notable for their bioactivity and degradability (Li et al., 2017). Calcium phosphates are favored as bone substitutes because of their biocompatibility and osteointegration (Stavropoulos et al., 2010). Although sintered hydroxyapatite is widely used in bone-tissue engineering, it is minimally resorbed. In contrast, β-TCP offers superior resorbability in both animal and human studies (Kon et al., 2000). Its three-dimensional structure enhances cell attachment, proliferation, and osteogenic differentiation (Nade et al., 1983; Zambonin et al., 2000). In a recent study, porous β-tricalcium phosphate (β-TCP) scaffolds were fabricated by integrating digital light processing (DLP) printing techniques with an in situ crystal growth process. These bioceramic scaffolds, featuring both macro- and micropores, were produced through DLP printing. Subsequently, an in situ crystal growth process was employed to generate micro-/nano-scale surface topographies. The resulting micro-/nanostructured scaffold enhanced the proliferation and differentiation of rat bone mesenchymal stem cells (rBMSCs), demonstrating significant capacity for skull bone regeneration in a rat model (Di Luca et al., 2016). Native bone tissue exhibits a structural gradient that can be observed radially in long bones and axially in flat bones due to variations in bone density and pore parameters from cancellous to cortical bone. Drawing inspiration from this natural architecture, Di Luca et al. developed three-dimensional polycaprolactone (PCL) scaffolds that feature an axial gradient in pore size and overall porosity using the “fused deposition modeling” (FDM) method. This scaffold notably enhanced osteogenic differentiation of human mesenchymal stem cells (hMSCs), particularly benefiting those residing within larger pores (Diloksumpan et al., 2020). Additionally, three-dimensional printing technology was utilized to create two types of 3D bioceramic scaffolds with distinct internal architectures—either exhibiting a designed porosity gradient or maintaining a uniform pore distribution. Nery and Lynch (1978) first reported the use of TCP-based bone grafts for periodontal defects in 1978. Owing to its high biodegradability, osteoconductivity, and osteoinductivity, β-TCP is a promising scaffold material for bone tissue engineering and is frequently used as an alternative to autologous bone grafts (Zhang J. et al., 2014). β-TCP microspheres with different diameters were fabricated via a solid-in-oil-in-water (S/O/W) emulsion method. BMSCs were co-cultured in vitro with β-TCP microspheres, and SEM and confocal microscope analyses were performed to discover that β-TCP microspheres efficiently promoted BMSC attachment and bone-related gene expression. The co-cultured BMSCs and microspheres were successfully implanted subcutaneously into nude mice for 8 weeks, and abundant new bone-like structures had formed between the β-TCP microspheres, implying that β-TCP microspheres can be used as a cell carrier and bone graft substitute material (Li et al., 2017).
The extensive application of β-TCP in bone tissue regeneration demonstrates its promising potential for periodontal tissue regeneration. The β-TCP scaffold seeded with bone marrow mononuclear cells (BM-MNCs) from C57BL/6J mice, when transplanted into the thigh muscle of syngeneic mice, demonstrated accelerated bone formation and enhanced periodontal tissue regeneration (Uchikawa et al., 2021). Owing to its excellent physical and chemical properties, β-TCP can be integrated with various biomimetic materials to enhance periodontal tissue regeneration. Different concentrations of β-TCP-loaded chitosan hydrogels (0%, 2%, 4%, or 6% β-TCP, 10% β-glycerol phosphate, and 1.5% chitosan) with cells can promote periodontal tissue regeneration (Tan et al., 2023). In the periodontal defect model of beagle dogs, the application of Retro mineral trioxide aggregate (MTA) or a combination of Retro MTA + β-TCP covered with a collagen membrane led to the regeneration of periodontal tissues (Fakheran et al., 2019).
3.1.3 Bioactive glass
Bioactive glass (BG), a class of synthetic alloplastic materials with a silicate base, possesses the distinctive capability to bond with mineralized hard tissues, such as bone, in physiological environments. Developed by Hench et al. in 1969, BG marked a significant advancement in dental applications during the mid-1980s when Clark et al. successfully utilized it in a clinical trial for alveolar ridge preservation in edentulous patients (Välimäki and Aro, 2006). It was not until the 1990s, however, that BG was first used for the treatment of periodontal lesions (Zamet et al., 1997). Recent research has identified BG as an osteoinductive agent capable of inducing osteoprogenitor cell migration into graft structures and promoting cell differentiation by influencing the gene expression of undifferentiated cells (Kargozar et al., 2018; Kargozar et al., 2017). It demonstrated bioactivity along with a relatively high mechanical strength that exhibited only a gradual decline, even under load-bearing conditions within the body (Shue et al., 2012). Schepers and Ducheyne (1997) introduced the term “osseostimulation” to describe the bone-formation process facilitated by BG (Schepers and Ducheyne, 1997). The most extensively used BG over the past 50 years is 45S5 glass, which is characterized by a low SiO2 content and high Na2O and CaO contents, along with a high CaO/P2O5 ratio, enhancing its reactivity with biological fluids (Vitale-Brovarone et al., 2009). In dental applications, 45S5 glass particulate, commercially known as PerioGlas® (NovaBone Products LLC, Alachua, FL, United States), has been demonstrated to inhibit epithelial cell down-growth and promote alveolar bone regeneration (Mengel et al., 2003; Subbaiah and Thomas, 2011). A multi-functional and sustained-release drug delivery system (MB/BG@LG) was developed by encapsulating methylene blue (MB) and BG into a lipid gel (LG) precursor using Macrosol technology. In addition, in vitro and in vivo experiments showed that MB/BG@LG can effectively promote periodontal tissue regeneration by reducing the inflammatory response and promoting cell proliferation and osteogenic differentiation (Chen et al., 2023). The incorporation of nano-BG glass-ceramic particles into alginate composite scaffolds enhances the ALP activity of human periodontal ligament fibroblast cells cultured on these scaffolds. Mesoporous bioactive glass (MBG) is extensively utilized in bone tissue repair and drug delivery applications. However, the occurrence of burst release of drugs and poor compatibility with other materials has limited its broader application. Modifying MBG with a polymer brush presents an effective strategy to enhance its properties. In this study, an alginate (ALG)-modified MBG was synthesized, and the effects of ALG on the characteristics of MBG were systematically investigated. The results demonstrate that ALG significantly improves drug loading efficiency, prolongs drug release duration, and facilitates the orderly deposition of apatite on the surface of MBG (Yao et al., 2022). These results suggest that biocompatible composite scaffolds are promising for periodontal tissue regeneration (Srinivasan et al., 2012).
3.1.4 Calcium sulfate
Calcium sulfate (CaS), commonly referred to as “plaster of Paris,” was first introduced as a synthetic bone graft material by Van Meekeren in 1892. This compound exists in three forms differentiated by the number of water molecules within its crystalline structure: anhydrates, dihydrates, and hemihydrates. The hemihydrate form, which dissolves rapidly, is predominantly utilized in medical-grade products and is available in two variants, α and β. Upon complete degradation in biological fluids, calcium sulfate leaves calcium phosphate deposits that promote bone growth (Shaffer and App, 1971; Zhao et al., 2021). Currently, calcium sulfate and its composites are primarily employed in dentistry and maxillofacial surgery as injectable bone fillers for sinus augmentation and alveolar bone regeneration in small periodontal defects (Thomas and Puleo, 2009).
3.1.5 Calcium silicate
Because of their predominant biocompatibility, bioactivity, and sealing ability, calcium silicate (CSi)-based bioceramics have been widely used in endodontic treatment, including MTA, Biodentine, BioAggregate, and iRoot BP Plus (Parirokh et al., 2018). MTA is characterized by low or no toxicity and excellent biocompatibility. It stimulates repair by allowing cellular adhesion, growth, and proliferation on its surface. MTA possesses good antibacterial properties, but it does not exhibit significant cytotoxic effects on host cells. They possess a large specific surface area and a nanoporous or hollow structure, which contribute to their elevated drug-loading capacity. Additionally, these materials demonstrate pH-responsive drug release behavior and favorable drug release characteristics. Consequently, they hold significant promise for applications in drug delivery. The calcium silicate-based drug delivery systems are characterized by prolonged drug-release times, which can considerably extend the therapeutic effects of administered drugs. Another notable advantage of these systems is their pH-responsive nature regarding drug release; this feature renders them an ideal platform for targeted drug delivery (Zhu et al., 2017). Additionally, it induces cementogenesis while regenerating the periodontal ligament, leading to bone formation.
In recent years, several CSi-based cements have been proven to be potential bioactive materials for bone-substitute applications, and some of them exhibit excellent in vitro and in vivo osteogenesis and odontogenesis (Chen Y. W. et al., 2016). CSi is a common material with excellent mechanical strength (Zhang W. et al., 2021). In addition, CSi cement not only exhibits a good osteoconduction effect but also reduces inflammatory markers in primary human dental pulp stem cells (hDPSCs). CSi-based cements can also stimulate the osteogenic differentiation of various stem cells, such as bone marrow stromal cells, adipose-derived stem cells, human dental pulp cells, and periodontal ligament cells. CSi and β-TCP composite collagen membrane (Retro MTA + β-TCP) has been shown to effectively promote periodontal tissue regeneration in the periodontal defect model of beagle dogs, demonstrating significant potential for periodontal bone regeneration (Fakheran et al., 2019). A fast-setting and controllable degradation of magnesium–calcium silicate (Mg–CSi) cement should be developed via the sol–gel method, and a mechanism should be established to stimulate hPDLSCs using Mg ions. The proliferation, alkaline phosphatase, odontogenesis-related gene (DSPP and DMP-1), and angiogenesis-related protein (vWF and ang-1) secretion of hPDLSCs significantly increased when the Mg content of the specimen was increased. The results of this study suggest that Mg–CSi materials with this modified composition could stimulate hPDLSC behavior and serve as effective bioceramics for bone substitutes and hard tissue regeneration applications, as they stimulate odontogenesis/angiogenesis (Chen et al., 2015; Chen Y. W. et al., 2016).
3.2 Polymer materials
3.2.1 Chitosan
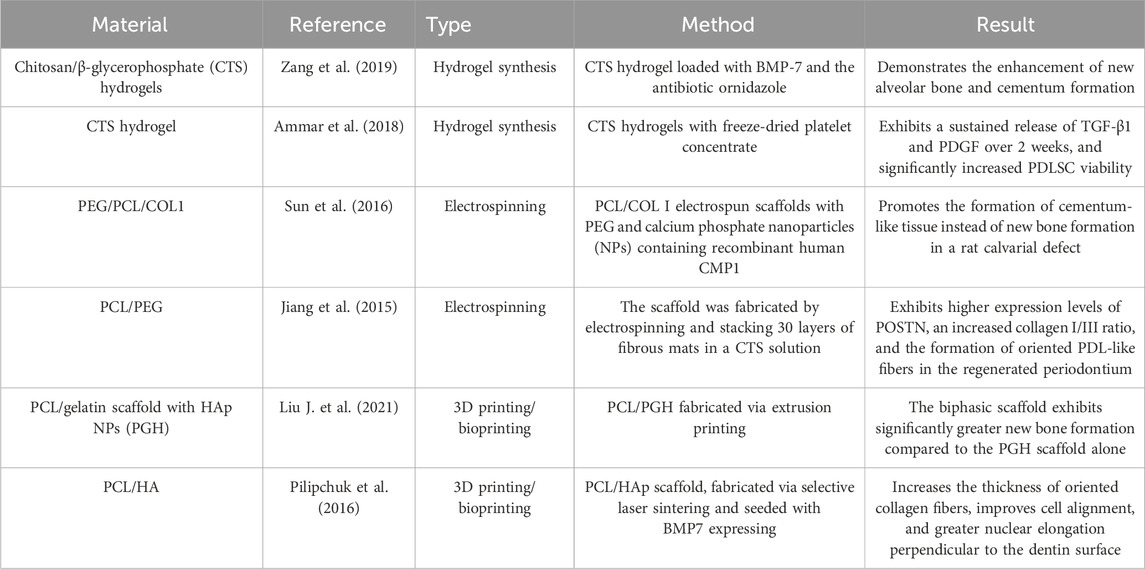
CS, a linear polysaccharide derived from chitin, exhibits notable properties, such as biodegradability, biocompatibility, antimicrobial effects, low antigenicity, hygroscopicity, and moisturizing properties (Bee and Hamid, 2025). Typically sourced from crustacean exoskeletons, chitosan comprises randomly dispersed β-(1,4)-glucosamine and N-acetyl-D-glucosamine units. The deacetylation process of chitin, first identified by Rouget in 1859 and later termed “chitosan” by Hoppe-Seyler in 1894, has demonstrated significant biomedical applications (Duymaz et al., 2025). In recent years, CS has been advocated for GTR/GBR applications because of its exceptional properties, including biocompatibility, biodegradability, anti-inflammatory and antibacterial properties, hydrophilicity, mucoadhesive characteristics, membrane-forming ability, flexibility, and cost-effectiveness. CS has been demonstrated to facilitate cellular adaptation and proliferation of various essential cell types involved in regenerating periodontal tissue, such as osteoprogenitor cells, osteoblasts, periodontal ligament fibroblasts, and gingival fibroblasts. Seol et al. (2004) demonstrated that CS sponges enhance the proliferation and differentiation of osteoblasts. Similarly, Klokkevold et al. (1996) verified that CS promotes the differentiation of osteoprogenitor cells, specifically MSCs, and potentially aids osteogenesis and bone formation. CS nanogels showed no cytotoxicity toward hGF cells at concentrations of 10–80 μL/mL (M et al., 2023).
In a separate study, CS promoted hPDL cell proliferation at concentrations of 2,000, 1,000, 100, and 50 μg/mL compared to the control group. The morphology of hPDL cells cultured with CS indicated no apoptosis, highlighting its non-toxic properties. Additionally, CS supported initial osteoblast attachment and spreading over fibroblasts, which could be advantageous in GBR procedures for bone regeneration by promoting osteoblast attachment and proliferation. In patients with periodontitis, chitosan implantation has been shown to reduce gingival inflammation due to its antimicrobial characteristics (Xu C. et al., 2012). Yeo et al. (2005) illustrated that a nonwoven chitosan membrane effectively enhances bone and cementum regeneration in surgically induced one-wall intrabony defects in beagle dogs, underscoring its potential in GTR and GBR. Furthermore, a CS/MW/Mel scaffold, a blend of chitosan and mesoporous wollastonite loaded with melatonin, was investigated for biomedical applications (Wang et al., 2025). The mHA/CS scaffold demonstrated enhanced osteoblast differentiation at both the cellular and molecular levels, as evidenced by the increased expression of key osteogenic markers, such as Runx2, Type I collagen, and osteocalcin. CS possesses certain inherent disadvantages, including a high degradation rate and low mechanical strength. When hydroxyapatite is combined with chitosan to form a composite scaffold, the resulting material retains the advantageous properties of enhanced biocompatibility and improved mechanical strength (Rodríguez-Vázquez et al., 2015). Additionally, the scaffold exhibited proangiogenic properties, facilitating vascularization, which is essential for tissue regeneration. This study further revealed the inhibitory effects of the mHA/CS scaffold on the growth of periodontal pathogens. When loaded with 20 μg/mL of recombinant human amelogenin (rhAm), the composite scaffold significantly boosted ALP activity and upregulated the expression of Runx2, OPN, and DLX-5 genes and proteins in vitro (p < 0.05). The GdPO4/Fe3O4/chitosan hybrid scaffold facilitated tumor ablation by enabling rapid heating under near-infrared laser irradiation. The mesoporous structure exhibited a diameter of 7 nm, and its specific surface area measured 33.95 m2/g (Liao et al., 2020). Their large pores and directional structure promoted cell migration and attachment, thereby enhancing bone tissue regeneration via the BMP-2/Smad/runt-related transcription factor 2 pathway (Merkel et al., 2023; Zhao et al., 2020). Furthermore, the scaffold effectively stimulated the formation of cementum-like tissues in vivo (Liao et al., 2020). These findings suggest that the mHA/CS scaffold loaded with 20 μg/mL rhAm has the potential to inhibit the growth of periodontal pathogens while promoting the formation of bone and cementum-like tissues (Table 4).
Natural polymers, particularly chitosan, serve as a diverse range of drug carriers due to their degradation capabilities. Non-toxicity and high biocompatibility are additional specific properties that make chitosan an ideal candidate for drug delivery systems. Chitosan can be modified into various formulations tailored to specific functions and modes of administration. Furthermore, the protonated amino groups present on D-glucosamine in the chitosan structure enable it to adhere to negatively charged mucus layers through electrostatic interactions, allowing penetration into deeper epithelial layers (Aguilar et al., 2019; Bakshi et al., 2020; Qin and Li, 2020). Due to its mucoadhesive properties, chitosan has been utilized as a vehicle for administering drugs via nasal, ocular, buccal, and pulmonary routes (Bernkop-Schnürch and Dünnhaupt, 2012; Khutoryanskiy, 2011; Singh et al., 2023). However, the insolubility of chitosan in physiological environments poses a challenge for effective drug delivery; this limitation can be addressed through chemical modifications such as carboxymethylation, acetylation, thiolation, and quaternization (Chen K. et al., 2017; Wang W. et al., 2020).
3.2.2 Polylactic acid
Polylactic acid (PLA) serves as an ideal polymer scaffold for dental, oral, and craniofacial tissue engineering owing to its hydrolytic degradation into lactic acid, a natural human metabolite (Diaz-Gomez et al., 2020). To enhance the scaffold performance, PLA can be combined with durable polymers, such as PEEK, via selective laser sintering to improve bioactivity, biocompatibility, and cytocompatibility (Feng et al., 2018). Additionally, blending PLA with PCL through 3D electrospinning enhances mechanical properties and promotes bioactivity and osteogenic differentiation (Yao et al., 2017). PLA scaffolds with stromal-derived factor-1 offer sustained release and promote endothelial cell proliferation and neovascularization, thus underscoring the potential of 3D-printed PLA constructs for bone regeneration (Singhvi et al., 2019). A novel biomimetic strategy employs Janus-structured porous PLA membranes (PLAMs) created via unidirectional evaporation-induced pore formation and self-assembled bioactive metal-phenolic network (MPN) nanointerfaces. Through the use of PLGA microspheres loaded with IL-4 (IL-4-PLGA), the composite hydrogel scaffold facilitated the transition of macrophages from the M1 phenotype to the M2 phenotype during the early induction phase. Additionally, it demonstrated a sustained anti-inflammatory effect mediated by PLGA microspheres containing kartogenin (KGN-PLGA) (Cheng et al., 2022). PLA serves as a carrier for doxycycline (Doxy) and nano-hydroxyapatite (nano-HAP), facilitating the gradual release of these substances through its resorption process. Consequently, it is anticipated that PLA will exhibit limited antibacterial properties. The enhanced antibacterial effect observed in the tested nanofibers, which were loaded with half the concentration of Doxy (5.04 µg) compared to the Doxy micro-compresses (10 µg), was evidenced by the larger diameters of the inhibition zones (Andrei et al., 2022). In vitro, MPN modification significantly reduced pro-inflammatory macrophage polarization in murine bone marrow-derived macrophages, stimulated angiogenesis in human umbilical vein endothelial cells, and enhanced the adhesion, migration, and osteogenic differentiation of human periodontal ligament stem cells. In vivo, PLAM-MPN implantation significantly accelerates bone repair in rat periodontal defects (Zhang Y. et al., 2023). This bioactive nanointerface within a Janus porous membrane has diverse capabilities to control cell physiology and promote bone regeneration, showing promise as a membrane for clinical applications in GTR and GBR.
3.2.3 Poly (lactic-co-glycolic acid)
Poly (lactic-co-glycolic acid) (PLGA), a copolymer of lactic and glycolic acids, is widely used as a synthetic biomaterial for drug and growth factor delivery and as a barrier in GTR/GBR. Its degradation rate can be adjusted by varying the lactic-to-glycolic acid ratio, owing to the differing hydrophilicities of the monomers (Washington et al., 2017). In vitro tissue engineering studies have compared various PGA-based polymers, such as PGA–PLA, PGA–PCL, and PGA–poly-4-hydroxybutyrate (P4HB). Among these, the PGA–PLA and PGA–P4HB scaffolds showed superior tissue formation compared to PGA–PCL, likely due to an optimal balance between scaffold degradation and tissue formation, thereby preserving mechanical integrity (Generali et al., 2017). PLGA’s excellent biocompatibility and tunable degradation properties make it versatile for periodontal regeneration applications, including barrier membranes, bone grafts, and delivery systems (Sun et al., 2017). Tetracycline, a widely utilized antibacterial agent, was effectively incorporated into PLGA membranes (Owen et al., 2010). The release profiles demonstrated an initial burst phase during the first week, followed by a sustained drug release over a duration of 14 days. This pattern is advantageous for achieving high antibiotic concentrations in the early stages while ensuring an adequate dose throughout the entire healing process. Notably, the incorporation of tetracycline did not alter the morphology of PDLSCs seeded onto the membranes. Kim et al. (2007) investigated the in vivo release profile of tetracycline-loaded PLGA membranes. The results indicated that sustained tetracycline release occurred with elevated concentrations during the initial 7 days; additionally, a greater extent of tissue attachment was observed compared to PLGA membranes alone. Azithromycin (AZM) was encapsulated in PLGA microspheres (AZM@PLGA) using single emulsion solvent evaporation and then coated with silk fibroin (SF) via electrostatic adsorption to mitigate AZM’s initial burst release (Ouyang et al., 2025). AZM@PLGA-SF microspheres significantly reduced periodontal inflammation and restored periodontal tissue health in vivo. The mechanically formulated microspheres modulated the inflammatory microenvironment in periodontal tissues by reducing pro-inflammatory cytokines (tumor necrosis factor-α, IL-6, interferon-γ, IL-2, and IL-17A) in gingival crevicular fluid while increasing anti-inflammatory cytokines (IL-4 and IL-10). In a rat model of periodontal defects, the superparamagnetic iron oxide nanoparticle (SPION)/PLGA scaffold enhanced IL-10 production and decreased NLRP3 and IL-1β release following Porphyromonas gingivalis infection, resulting in superior periodontal regeneration compared to the PLGA scaffold alone. This antibacterial SPION/PLGA scaffold exhibited anti-inflammatory and anti-adhesive properties, aiding in infection control and promoting periodontal tissue regeneration through immune modulation (Washington et al., 2017).
3.2.4 Alginate
Alginate, a polysaccharide derived from brown algae, is used as a hydrogel in 3D bioprinting and tissue engineering because of its cost-effectiveness, wide compatibility, and similarity to proteins in the ECM. Alginate polymerization is rapid and typically occurs within seconds to minutes of exposure to divalent cations, allowing for various ion-doping options. The introduction of methacrylate groups enables hydrogel production through light-induced radical polymerization. However, the inherent structure of alginate lacks peptide-binding sites for cellular interactions, such as arginine–glycine–aspartate (RGD) and YIGSR peptides (Tolmacheva et al., 2024). Therefore, alginate is commonly combined with other cell-adhesive polymers, such as collagen, and functional polymers, such as agarose or hyaluronic acid, by incorporating an ECM-based protein or RGD peptide sequence to enhance cell adhesion and proliferation. The loading of the drug (spermidine) via dynamic bonding presents a promising avenue, offering a straightforward, cost-effective, and efficient strategy for the sustained release of pharmaceuticals while enhancing the viscoelastic properties of hydrogels (Zhang S. et al., 2023). The drug-loadable calcium alginate hydrogel system is a potential bone defect repair material for clinical dental applications (Chen L. et al., 2017). Bovine serum albumin (BSA) can be directly stored within the 3D pore network structure of hydrogels. However, the positively charged functional groups in BSA can interact with the carboxyl groups present in alginate, leading to the formation of BSA/alginate complexes. Consequently, the release of BSA from these hydrogels is sustained as hydrogel degradation progresses (Zhao et al., 2009). Calcium alginate hydrogels, thus, possess the ability to combine with drugs or proteins through various mechanisms, rendering them highly suitable for drug delivery applications. Collagen provides additional benefits, including low immunogenicity and the promotion of bone regeneration. The use of collagen matrices has been shown to influence osteoblast behavior in various hydrogels (Tharakan et al., 2022). To expedite gelation, post-bioprinting self-healing, and subsequent stabilization via secondary crosslinking, alginate was subjected to methacrylation and oxidation, followed by ionic crosslinking using an eggshell model (Jeon et al., 2022). To enhance alginate hydrogels for bone tissue engineering, inorganic particles, such as bioglass and kaolin, or organic materials, such as cells, drugs, and human allograft tissue, can be integrated (Bhattacharyya et al., 2024; Bhattacharyya et al., 2023; Gharacheh and Guvendiren, 2022). The CS/alginate hydrogel exhibits certain unavoidable limitations, including a high degradation rate and low mechanical strength (Hu et al., 2019; Zupančič et al., 2019). Furthermore, research indicates that the incorporation of carbon nanotubes (CNTs) into the composite scaffold enhances its mechanical properties, potentially making it suitable for periodontal tissue regeneration (Carletti et al., 2011). Consequently, compared to bare hydrogel CS/AL composites, CNT/CS/AL demonstrates superior mechanical and antibacterial properties. Studies have indicated that an RGD-coupled alginate scaffold promotes the differentiation of oral MSCs toward an osteoblast lineage both in vitro and in vivo, as evidenced by the expression of the osteogenic markers Runx2, ALP, and osteocalcin. In summary, these findings represent the first evidence that MSCs from the orofacial tissue, when encapsulated in RGD-modified alginate scaffolds, have the potential for craniofacial bone regeneration (Gharacheh and Guvendiren, 2022). The use of an hPDLSC-based medium enhanced the osteogenic differentiation of hPDLSCs in the Alg + Fib + hPL construct, offering a promising xeno-free strategy for delivering hPDLSCs to boost dental, craniofacial, and orthopedic regenerations (Qiu et al., 2022).
3.3 Hydrogel
Periodontal tissue regeneration is complex. Hydrogels exhibit significant potential for clinical applications in this domain by closely mimicking natural ECM and biological tissues (Sánchez-Cid et al., 2022) (Figure 4). These polymeric materials possess a three-dimensional network of physically or chemically linked polymer chains that enables them to absorb and retain substantial amounts of water while maintaining their solid structure. The high water content of hydrogels contributes to their softness and biocompatibility and creates an optimal environment for cell adhesion, proliferation, differentiation, and growth (Radulescu et al., 2022). Hydrogels have been extensively investigated as scaffolds and/or drug delivery systems because of their ability to integrate cells into their structures and concurrently degrade with new tissue formation (Santos et al., 2023b). Xu et al. (2019) developed an injectable thermosensitive hydrogel that facilitates the continuous release of aspirin and erythropoietin, thereby promoting the healing of inflammation and the repair of alveolar bone. Within the first 3 days of the drug release trial, 86.6% and 69.4% of aspirin and erythropoietin were released, respectively. The rapid release rate of aspirin is particularly beneficial during the early stages of periodontitis treatment, where prompt control of inflammation is essential. The thermosensitive hydrogel designed by Liu et al. (2022) used polyethylene glycol diacrylate as the base scaffold and incorporated additional reactive oxygen species (ROS)-scavenging effects to enhance its functionality. By adding dithiothreitol, a reducing sugar alcohol, this hydrogel also demonstrated oxidative stress-reducing effects on cells, thus providing protection for periodontal cells against inflammatory damage. Furthermore, by incorporating various bioactive molecules, hydrogels can induce crucial cellular processes, such as migration, proliferation, differentiation, vascularization, and mineralization during periodontal regeneration (Gopalakrishna et al., 2023; Ranch et al., 2021; Swain et al., 2019). The hydrogel matrix can be enhanced through the incorporation of organic or inorganic nanomaterials. This addition improves surface reactivity, mechanical properties, and the release kinetics of loaded bioactive agents (Irawan et al., 2018; Radulescu et al., 2022). Additionally, hydrogels offer excellent biocompatibility, water retention, controlled release, and support for cellular interactions, making them promising candidates for periodontal tissue engineering (Zhang W. et al., 2021). The mechanical strength of hydrogels represents a significant limiting factor in their application for periodontal tissue engineering. Striking an optimal balance between mechanical strength and injectability poses considerable challenges, as enhancing the mechanical properties frequently leads to a reduction in injectability (Sun et al., 2024) (Table 5).
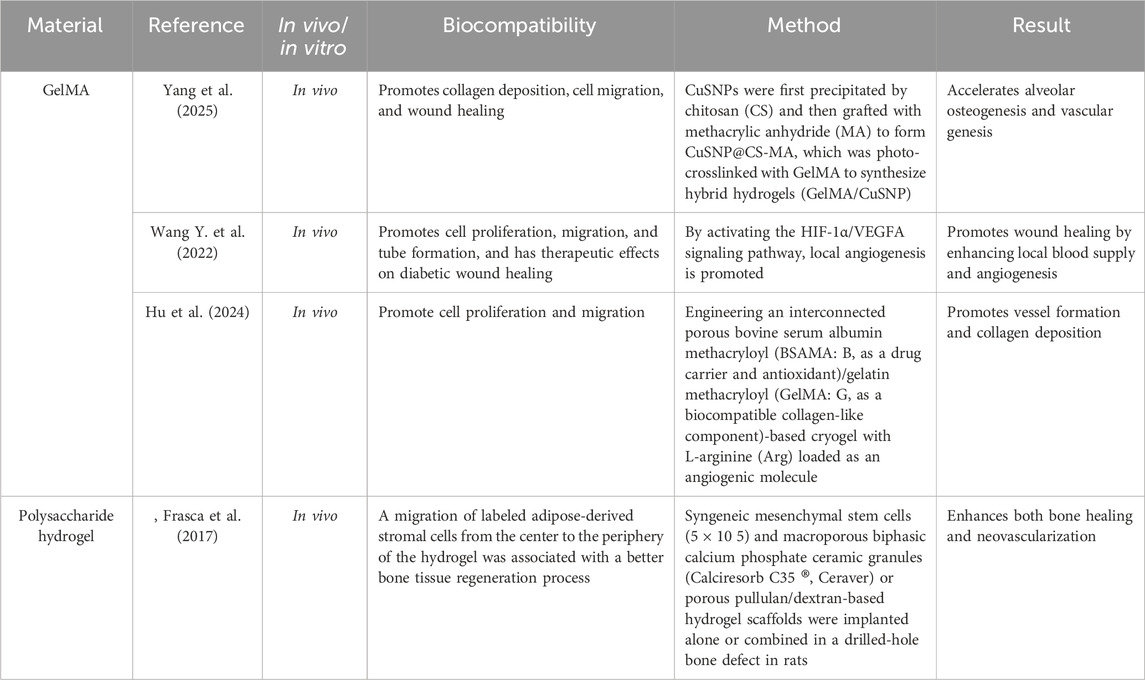
3.3.1 Gelatin methacryloyl (GelMA)
Gelatin methacryloyl (GelMA), akin to the native ECM, features multiple RGD sequences for cell adhesion and MMP-degradable motifs for cell remodeling (Klotz et al., 2016). As a photo-crosslinked hydrogel, GelMA undergoes radical polymerization initiated by light, eliminating the need for potentially harmful chemical cross-linkers (Klotz et al., 2016). Despite its remarkable characteristics, it is primarily constrained by its limited mechanical strength, as certain tissue types inherently exhibit high mechanical stiffness. Although the application of elevated GelMA concentrations can enhance mechanical strength, this improvement comes at the expense of porosity, degradability, and three-dimensional (3D) cell attachment (Sakr et al., 2022). The tunable mechanical properties and favorable biological traits of GelMA make it a versatile scaffold for diverse biomedical applications (Sakr et al., 2022). Studies have shown that GelMA-based scaffolds enhance cell viability and promote osteogenic/odontogenic differentiation of dental stem cells, including dental pulp stem cells (DPSCs), PDLSCs, and stem cells from the apical papilla (SCAPs) (Zhu et al., 2023). In addition to serving as a cell delivery carrier in bioprinting, GelMA facilitates drug and growth factor delivery owing to its degradability. FT-IR spectroscopy confirmed the successful incorporation of irradiated chlorpromazine (CPZ) into the hydrogel matrix. The release profiles of chlorpromazine demonstrated sustained release exclusively in hydrogels containing 1 mg/mL of CPZ. Furthermore, the hydrogel exhibited significant antimicrobial activity against MRSA bacteria compared to penicillin. These findings underscore the potential of CPZ loaded during the photopolymerization process within hydrogels as an effective antimicrobial agent with sustained release properties, rendering it suitable for combating resistant bacterial strains (Tozar et al., 2024). Park et al. (2020) developed a bioink by conjugating BMP peptides to GelMA and found that the BMP-mimicking peptide remained in the bioprinted structure for over 3 weeks and significantly increased odontogenic gene expression in hDPSCs. Pan et al. (2020) evaluated a GelMA hydrogel containing human PDLSCs both in vitro and in vivo, noting enhanced PDLSC proliferation and differentiation within the hydrogel, along with new bone formation in rat alveolar defects treated with hydrogels. GelMA hydrogels have also been improved by using nano-hydroxyapatite (Chen X. et al., 2016). Human PDLSC constructs significantly enhanced osteogenic differentiation, resulting in increased mineralized tissue formation in vivo in a mouse model. GelMA has been combined with materials, such as polyethylene glycol diacrylate (Ma et al., 2017). In diabetic mice, GelMA hydrogels with VH-EVs (Gel-VH-EVs) effectively promoted wound healing by enhancing local blood supply and angiogenesis, potentially by activating the HIF-1α/vascular endothelial growth factor (VEGF)A signaling pathway (Wang Y. et al., 2022). Additionally, GelMA/CuSNP hydrogels accelerate alveolar osteogenesis and vascular genesis, successfully treating periodontitis within 4 weeks in a rat model (Yang et al., 2025).
3.3.2 Polysaccharide hydrogels
Polysaccharide hydrogels have emerged as pivotal in bone tissue engineering and regenerative medicine primarily because of their efficacy in enhancing bone repair via cellular and drug delivery. They provide a hydrophilic 3D matrix that enhances osteocyte viability and promotes new bone formation. These hydrogels are extensively used in tissue engineering and wound healing owing to their superior biocompatibility, controlled degradation, and versatile structure (Gonzalez-Fernandez et al., 2022). Their porous polymeric frameworks and high hydration levels emulate the native ECM, supporting cellular proliferation, differentiation, and tissue repair. Furthermore, their biodegradable nature, customizable chemistry, low toxicity, and minimal immune response render them ideal candidates for sustained drug release. In scaffold-based applications, polysaccharide hydrogels induce a foreign body response similar to that of soft tissues, closely mimicking the natural ECM (Taghipour et al., 2020).
Polysaccharide hydrogel scaffolds leverage several key mechanisms to facilitate bone regeneration. First, the hydrogel exhibits low immunogenicity and contains multiple functional groups in its backbone that serve as attachment sites, preventing the displacement of bone cells and therapeutics by blood flow, thereby enabling bone cell proliferation at the fracture site (Koons et al., 2021). Second, the scaffold incorporates MSCs and various osteoinductive proteins, including BMP-2, transforming growth factor-β (TGF-β), and VEGF (Aravamudhan et al., 2013; Gugliandolo et al., 2021; Xu X. et al., 2012). Third, the polysaccharide hydrogel surface is highly microporous, which enhances vascularization and stimulates innate osteogenic processes (Gonzalez-Fernandez et al., 2022). Scaffold-based polysaccharide hydrogels possess porous structures that promote bone regeneration. Studies have demonstrated the remarkable mechanical properties and bone-regenerative capacity of polysaccharide hydrogels. Kolk et al. (2012) integrated regenerated cellulose nanofibers into a chitosan-based polysaccharide hydrogel that enhanced the proliferation and differentiation of osteoblasts. Polysaccharide-based hydrogels were doubly integrated with hydroxyapatite (HAp) nanoparticles and calcium carbonate microspheres (CMs) under physiological conditions. Furthermore, the composite gel scaffolds exhibited favorable sustained drug release and antibacterial properties, as confirmed by calculations of drug release and evaluations of antibacterial activity (Ren et al., 2018). Overall, microporous polysaccharide hydrogels offer excellent biocompatibility, biodegradability, self-repair capabilities, and injectability, making them promising candidates for bone tissue engineering applications.
3.4 Decellularized extracellular matrix
3.4.1 dECM hydrogels
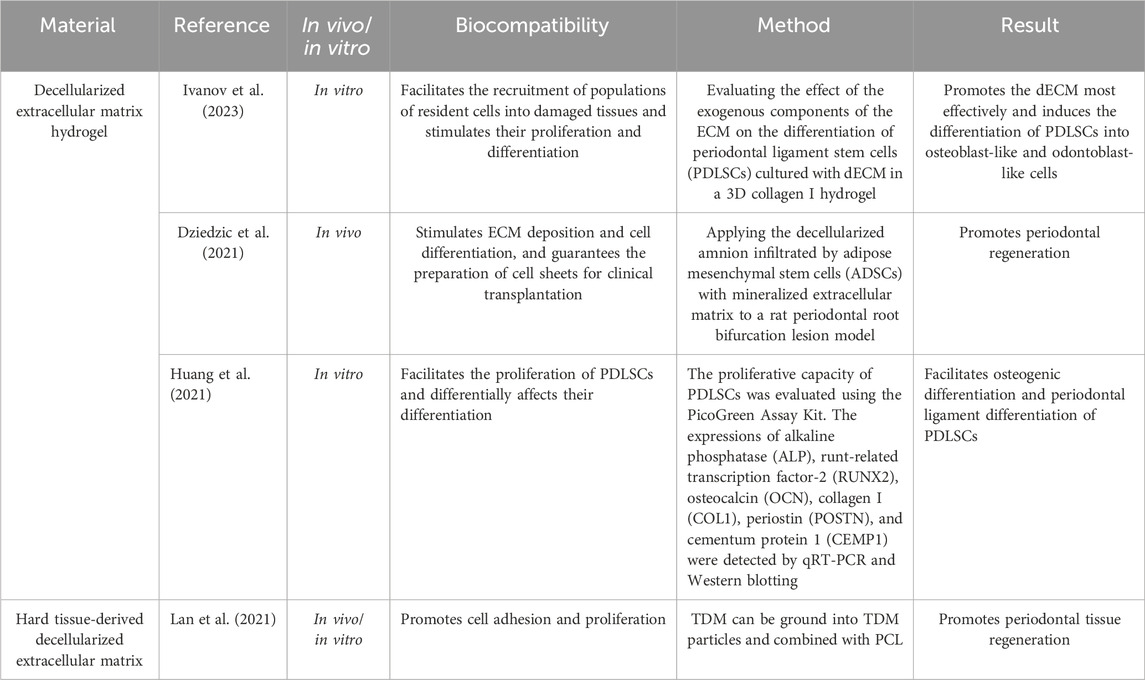
Decellularized extracellular matrix (dECM) hydrogels are increasingly used in tissue engineering, particularly for in vitro organoid development. This intricate macromolecular framework plays a vital role in modulating cellular adhesion, movement, specialization, and functional activity, thereby influencing tissue growth and maintenance (Zhang H. et al., 2021). Among commercial ECM products, Matrigel®—derived from murine tumor secretions—remains widely used due to its rich composition of basement membrane components, including laminin, type IV collagen, entactin, and heparan sulfate. Various tissue sources, such as human, porcine, bovine, and murine tissues, have been processed to yield dECM using mechanical, chemical, or enzymatic decellularization methods (Crapo et al., 2011). The resulting dECM can be gelled by temperature modulation, ionic strength adjustment, pH variation, or chemical crosslinking. As biomimetic materials, dECM-based constructs offer a tissue-specific microenvironment that promotes specialized cell functions and enhances natural regeneration (Gattazzo et al., 2014). Decellularization effectively eliminates cellular material and immunogenic components while retaining key structural proteins (e.g., collagen, elastin, and fibronectin) and macromolecules (e.g., proteoglycans and glycosaminoglycans) (Keane et al., 2015).
The dECM is extensively utilized in regenerative medicine as a scaffold material, owing to its exceptional biological activity and excellent biocompatibility. Advanced manufacturing techniques, such as 3D printing and electrospinning, have been employed to produce materials/scaffolds based on dECM that can replicate the distinct characteristics of periodontal tissues (Schmidt-Schultz and Schultz, 2005). Multiple strategies have been developed to enhance the mechanical strength of the dECM (Jiang et al., 2021). These include functionalized scaffolds embedded with cells that facilitate the harvesting of scaffold-supported dECM through a process of decellularization and crosslinked soluble dECM that can be utilized to form injectable hydrogels for periodontal tissue repair (Liang et al., 2023). This expansion of dECM applications has facilitated the clinical translation of dECM-based materials for periodontal regeneration. Consequently, the dECM serves as a crucial source of bioactive material to initiate the repair of periodontal tissue and regeneration of the periodontium (Papadimitropoulos et al., 2015). For instance, bone dECM serves as a reservoir of pro-inflammatory cytokines, growth factors from the TGF-β family, various bone morphogenetic proteins (BMPs), and angiogenic growth factors, such as VEGF, thereby inducing osteoinduction by controlling different stages of bone regeneration (Wang S. et al., 2022). The immunomodulatory activity of dECM is robust, leading to a reduction in the secretion of pro-inflammatory factors by M1 macrophages and a decrease in local inflammation in Sprague–Dawley rats. In a critical-size model of periodontal defects, the bioprinted module significantly boosted the regeneration of hybrid periodontal tissues in beagles, particularly the anchoring structures of the bone–ligament interface, well-aligned periodontal fibers, and highly mineralized alveolar bone (Yang et al., 2023).
The urinary bladder matrix and SIS are dECM biomimetic materials endorsed by the FDA to create regenerative biomimetic materials for damaged skin, muscle, and gastrointestinal tissues (Brown-Etris et al., 2019). SIS, a commonly used decellularized tissue, shows promise for soft tissue repair but requires enhancement for bone tissue regeneration. Gou et al. (2019) utilized pig SIS as a collagen membrane to guide tissue regeneration. In a rat cranial defect model, SIS dECM facilitated bone tissue regeneration, resulting in an increased bone volume fraction and tissue regeneration, suggesting the potential for bone regeneration in periodontal defects.
3.4.2 Hard tissue-derived dECM
Decellularized bone powder, a common periodontal tissue engineering material, is clinically used in the form of dECM granules (Figure 5). These granules are often combined with hydrogels and a guiding tissue regeneration membrane to enhance periodontal regeneration (Suzuki et al., 2023). Datta et al. (2021) used decellularized bone granules within a chitosan-based hydrogel containing human amniotic mesenchymal stem cells (hAMSCs) to stimulate bone regeneration. Intrinsic growth factors present in decellularized bone granules promote robust cellular viability, proliferation, and osteogenic differentiation of human amnion-derived stem cells (Datta et al., 2021). This biomaterial shows promise for application in periodontal tissue engineering because of its ability to effectively preserve the intricate ECM components found in native tissue, offering optimal cues for the regeneration and repair of damaged periodontal tissue. Son et al. (2019) treated tooth slices from human molars with sodium dodecyl sulfate and Triton X-100, effectively removing nuclear components while preserving the structure and composition (Son et al., 2019). The T-dECMs supported PDLSC regeneration near the cementum by expressing cementum- and PDL-related genes. These findings demonstrate that dentin dECM enhances MSC proliferation and differentiation and show promise for periodontal regeneration in tissue engineering. Dental-derived dECM has been used successfully to promote dental tissue repair (Jeon et al., 2022; Liang J. et al., 2020). The development of soluble dECM, 3D printing, and other advanced fabrication techniques has helped improve the fidelity and strength of dECM-based 3D scaffolds (Park, 2019), focusing on the spatial management in the defect area (Kim et al., 2020; Varoni et al., 2018; Yang et al., 2023). Meanwhile, due to the abundance of certain functional groups in dECM materials—such as hydroxyl, acyl, peptide bonds, and hydrogen bonds—these materials facilitate drug loading through crosslinking reactions (Ding et al., 2023). Notably, this includes amide crosslinking and interactions between aldehyde groups and the amine groups of lysine or hydroxylysine (Gou et al., 2019).
The treated dentin matrix (TDM) exhibits strong odontogenic/osteogenic potential and has been demonstrated to regenerate periodontal ligament-like tissues, including perforating fiber-like tissues, in a beagle canine alveolar socket orthotopic transplantation model. Meanwhile, TDM has the ability to provide sustained drug release, and xenogeneic TDM equipped with the immunomodulatory drug rosiglitazone can achieve a periodontal tissue regeneration effect comparable to that of homogeneous TDM (Lan et al., 2021). Surprisingly, TDM could be ground into TDM particles and combined with PCL, which could be used to fabricate personalized biological roots using low-temperature deposition manufacturing 3D printing technology and regenerate periodontal ligament-like tissue in a beagle canine alveolar socket orthotopic transplantation model. Owing to the superior personalized matching capability of 3D-printed scaffolds, the TDM-PCL scaffold enables the replication of the “immediate implantation” process in the in situ transplantation experiment of the alveolar socket. Therefore, TDM is a type of dECM that plays an important role in periodontal tissue regeneration (Huang et al., 2023) (Table 6).
3.5 Autologous materials as regenerative substitutes for periodontal tissue regeneration
Numerous regenerative techniques have been developed for the treatment of periodontitis. However, current treatment modalities, whether used alone or in combination, face challenges in achieving genuine regeneration, particularly for complex periodontal defects. Consequently, there is a need to identify an optimal material that can facilitate periodontal regeneration with enhanced potential for new attachments and reduced complications. Clinical trials in regenerative medicine have explored the use of allografts, xenografts, alloplasts, or their combinations, based on their documented regenerative capabilities (Liang et al., 2021). Nevertheless, these bone graft materials are hindered by the significant drawback of eliciting foreign body reactions or adverse immune responses, which limits their efficacy in treating periodontal defects. Due to these constraints, clinicians favor autogenous biomimetic materials that promote tissue regeneration with minimal immunogenicity upon introduction into the body. One such biomaterial is the fibrin matrix known as platelet-rich fibrin (PRF), which was initially described by Choukroun. PRF, a second-generation clot derived from a patient’s own blood through centrifugation, transforms into a robust three-dimensional fibrin scaffold containing platelets, leukocytes, and various growth factors (such as platelet-derived growth factor (PDGF), VEGF, TGF-β, insulin-like growth factor 1 and 2 (IGF-1/IGF-2), and epidermal growth factor (EGF)), crucial for tissue regeneration (Christgau et al., 2006; Dohan et al., 2006). The findings highlight the potential of antibiotic-loaded PRF as an effective localized drug delivery system exhibiting robust antibacterial properties (Kobayashi et al., 2016). For patients not undergoing systemic antibiotic therapy, the optimal strategy involves incorporating the antibiotic into PRF or i-PRF only after centrifugation (Tan et al., 2019). Compared to other carriers, such as collagen, PRF consistently demonstrates superior antibacterial effects and enhanced drug-loading capacity (Niemczyk et al., 2025). Biological proteins induce the differentiation of periodontal ligament cells into osteoblasts, thereby promoting bone regeneration. In a clinical trial, Serroni et al. (2022) found that leukocyte platelet-rich fibrin (L-PRF) enhanced the degree II furcation management when combined with autogenous bone grafts compared to autogenous bone grafts and open-flap debridement alone. In a randomized trial, Rexhepi et al. (2021) demonstrated that combining an inorganic bovine bone graft with L-PRF was as effective as combining it with a collagen membrane (CM) in managing unfavorable intrabony defects. Various PRF variants have emerged over time owing to different centrifugation protocols aimed at enhancing growth factor release. Advanced platelet-rich fibrin (A-PRF) developed by Choukroun in 2014 utilizes a low centrifugal speed (150 rpm, 14 min) to achieve a more uniform distribution of neutrophils and an unbound fibrin matrix. Neutrophils in PRF contribute to tissue regeneration by guiding monocyte phagocytosis and producing proteases such as MMP9 involved in wound healing (Fujioka-Kobayashi et al., 2017). Moreover, PRF application in periodontal intrabony treatment led to a reduced pocket probing depth and improved relative attachment levels. Lei et al. (2020) conducted a study utilizing A-PRF and concentrated growth factor (CGF) to treat periodontal defects and demonstrated a consistent and sustained release of growth factors for 2 weeks. The authors noted that this combined approach exhibited comparable efficacy in regenerating periodontal bone and enhanced GTR results in intrabony defects (Lei et al., 2020). Furthermore, the combination of A-PRF+ with xenografts or alloplastic materials may enhance periodontal tissue regeneration, facilitate bone augmentation, and contribute to improved implant stability (Chmielewski et al., 2025). One study utilized human gingival fibroblasts, whereas the other employed human periodontal ligament cells (Miron et al., 2017). Blood samples for these experiments were collected exclusively from human participants (Zheng et al., 2020). One study demonstrated that both PRP and i-PRF exhibited excellent cell viability and biocompatibility (Farshidfar et al., 2025).
4 Physical properties
Successful tissue regeneration requires scaffolds with specific mechanical stability and biodegradability, appropriate dimensions, surface roughness, and porosity to create a suitable microenvironment that supports effective cell–cell interactions, cell migration, proliferation, and differentiation. Biomimetic materials’ physical properties, such as pore size, porosity, particle size, and mechanical strength, play a crucial role in influencing cell attachment and proliferation (Bharadwaz and Jayasuriya, 2020).
4.1 Fabrication for scaffold
Periodontal tissues, which include the gingiva, periodontal ligament, cementum, and alveolar bone, play a critical role in supporting and anchoring teeth within the jawbone. However, the regeneration of the complex structural architecture of periodontal tissues continues to pose a significant challenge. Given the complexity of periodontal tissues and the need to promote coordinated and orderly tissue regeneration, developing hierarchical scaffolds that can simulate various periodontal tissues is an attractive strategy for achieving more synchronous tissue regeneration. Hierarchical scaffolds fabricated by various techniques such as 3D printing/bioprinting (Sufaru et al., 2022), electrospinning (Santos et al., 2023a), and hydrogel synthesis (Santos et al., 2023b) have different designs and compositions and have shown promising results in both in vitro and in vivo studies (Table 7).
To address this, it is essential to carefully design and fabricate suitable scaffolds based on specific clinical requirements in order to fully realize their functional potential. Electrospun nanofibers exhibit high porosity and large surface area, effectively mimicking the natural extracellular matrix and thereby modulating cell attachment, migration, proliferation, and differentiation. Recently, various electrospun nanofibrous membranes have been developed with antibacterial, anti-inflammatory, and osteogenic properties, demonstrating promising potential for periodontal regeneration (Santos et al., 2023a). The 3D bioprinting technology, particularly when utilizing biomaterials embedded with living cells and bioactive molecules, offers precise and controllable regulation of key structural properties such as porosity, connectivity, pore size, and permeability. This additive manufacturing approach constructs complex 3D architectures by systematically depositing cell-laden hydrogels or other biomaterials layer by layer. Although various 3D bioprinting techniques have been developed, each possesses distinct advantages and limitations (Dey and Ozbolat, 2020). Therefore, selecting an appropriate 3D bioprinting method that can accurately mimic the intricate structure and functional properties of native periodontal tissues is crucial for reconstructing a conducive regenerative microenvironment and promoting effective tissue regeneration (Shapira and Dvir, 2021).
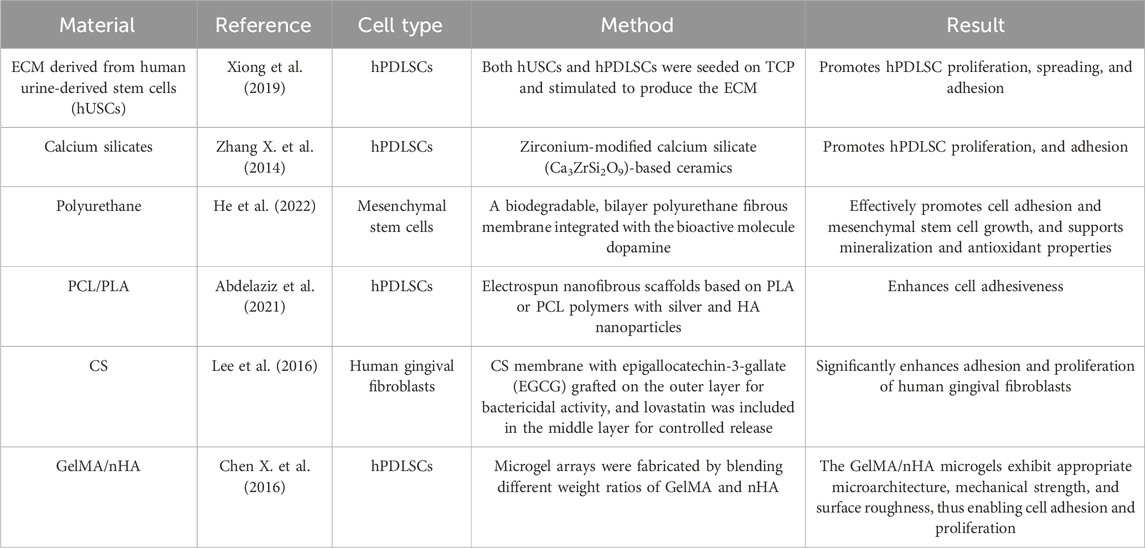
4.2 Cell adhesion
Focal cell adhesion to various scaffold surfaces is crucial for initiating signaling cascades that further promote cell proliferation and differentiation (Lee et al., 2004). Therefore, precise regulation of cell–cell and cell–scaffold interactions can achieve multiple objectives in multifunctional tissue engineering. Biological material scaffolds can provide mechanical support and regulate cell adhesion and motility. Additionally, growth factors delivered by these scaffolds can modulate cellular behavior (Table 8).
The ECM offers significant advantages in regulating cell adhesion. All cells interact with the ECM, which plays a crucial role in determining stem cell fate. Different types of extracellular matrices vary in composition and function (Watt and Huck, 2013). Moreover, upon cell binding to the ECM, intracellular signaling is regulated through cell surface adhesion receptors, among which the integrin family represents a major and essential class. Additionally, various protein domains within the ECM can dynamically bind and release growth factors, thereby synergizing with these factors to modulate cellular behavior (Hynes, 2009). Furthermore, ECM features such as micro-patterned islands, stiffness, physical topography, and deformability can promote distinct biological processes (Watt and Huck, 2013).
4.3 Porosity
The functional performance of implanted cells is largely determined by the pore size of the scaffold. Pore dimensions critically influence the diffusion of nutrients and oxygen, along with the removal of metabolic waste. Furthermore, pore size significantly affects cell adhesion, cell-to-cell interactions, and transmembrane cell migration—processes that vary according to the specific requirements of different tissue regeneration applications (Bružauskaitė et al., 2016). For example, the high porosity and interconnectivity are beneficial for establishing an optimal microenvironment that promotes cell-to-cell communication and facilitates seamless integration of the scaffold into the adjacent tissue at the implant site (Pina et al., 2015). Pore sizes can be categorized into nanoscale (nano-roughness, <100 nm), microscale (micro-roughness, 100 nm–100 μm), and macroscale (100 μm–millimeters) (Vagaská et al., 2010). Different pore sizes may influence distinct cellular processes: nanoscale pores have been shown to play a critical role in collagen fiber formation and ECM deposition, whereas macropores are essential for cell seeding, distribution, migration, and subsequent neovascularization in vivo (Smith et al., 2009). Different pore structures also influence the cellular biological behavior in response to biomaterials and the efficacy of tissue regeneration in periodontal applications (Table 9).
4.4 Drug delivery
Various forms of polymeric scaffolds for cell and drug delivery are available: (1) a conventional, three-dimensional porous matrix, (2) a nanofibrous matrix, (3) a thermosensitive sol–gel transition hydrogel, and (4) porous microspheres. Scaffolds provide a suitable substrate that supports cell attachment, proliferation, migration, and differentiated function. Scaffold matrices enable targeted drug delivery with high loading capacity and efficiency. Furthermore, incorporating therapeutic agents, such as anti-inflammatory inhibitors and antibiotics, into scaffolds can help prevent postoperative infection and manage related diseases over an extended period. Additionally, scaffolds can deliver biological signals—such as adhesion peptides and growth factors—to guide cells toward the desired differentiation state, maintain their viability, and support sustained growth (Garg et al., 2012). Given the tissue heterogeneity and antibacterial requirements inherent in the periodontal tissue regeneration process, the drug delivery capabilities of biomaterials play a critical role (Table 10).
5 Discussion
With the in-depth application of biomimetic materials in tissue engineering, their use in periodontal tissue regeneration has become widespread. This article reviews the application of biomimetic materials and cells in periodontal tissue regeneration after reviewing the literature.
Biomimetic materials can be classified in various ways. Based on the extent to which the material or system mimics natural structures or functions, they can be categorized as bio-inspired, bio-derived, or biomimetic. Bio-inspired materials are not directly derived from biological sources but are synthetically designed to incorporate key functional elements found in nature (Semino, 2008). The inspiration often comes from specific chemical, physical, or topological cues, such as RGD-modified alginate. Bio-derived materials are obtained directly from biological sources such as animals, plants, or humans. They inherently possess many of the essential biological signals but often require processing to remove immunogenic components, such as dECM, collagen, fibrin, and hyaluronic acid. Biomimetic materials are designed not only to present biological cues but also to dynamically respond to their environment in a manner that mimics living systems. These materials often blur the distinction between a material and a machine, such as self-healing hydrogels. Biomimetic materials can be categorized into natural and synthetic polymer systems based on the origin of their polymer chains. Natural polymeric systems are derived from natural sources. They offer excellent biocompatibility and bioactivity; however, they may exhibit issues related to batch-to-batch variability, immunogenicity, and mechanical strength. Examples include collagen, fibrin, silk fibroin, HA, alginate, and chitosan. Synthetic polymeric systems are man-made polymers. They offer precise control over material properties, such as degradation rate, stiffness, and structure, as well as high reproducibility; however, they are often inert and lack biological recognition. Common examples include PEG, PLGA, PCL, and PLA. The selection of the appropriate system depends entirely on the specific application: a simple cell delivery vehicle may utilize a pure natural polymer, whereas a complex organoid model may require a sophisticated, stimuli-responsive synthetic hydrogel engineered with biomimetic intelligence.
Calcium phosphate biomimetic materials have garnered extensive attention in the field of dental implants because of their excellent bone conductivity and bone regeneration capabilities. These materials are chemically and structurally analogous to natural bone, making them ideal for repairing bone defects in periodontal tissue. Furthermore, CaP materials can be combined with other biomimetic materials by applying coatings to enhance the efficiency of tissue regeneration. Polymer materials exhibit biodegradability, biocompatibility, antibacterial properties, low antigenicity, moisture absorption, and moisture retention. These features make them ideal scaffolds for oral, craniofacial, and tissue-engineering applications. Polymer materials are widely utilized as synthetic biomimetic materials for drug and growth factor delivery systems and barriers for GTR and GBR. Hydrogels have advantages over many other biological materials owing to their superior physical and chemical properties. They possess high structural plasticity, enabling them to conform to the personalized architecture of the periodontal tissue. Additionally, hydrogels demonstrate excellent biocompatibility with various cell types and serve as effective sustained-release carriers for controlled drug delivery at different time points. Their printability can also be integrated with 3D printing technology in combination with other biomimetic materials, facilitating their application in periodontal tissue regeneration. dECM is extracted from natural tissues and exhibits excellent biocompatibility and cell-induction capabilities, providing an optimal microenvironment for the regeneration of host stem cells. The dECM hydrogel combines the high plasticity of hydrogel materials, enabling it to carry drugs with sustained-release properties, while effectively adapting to the personalized structural requirements of the periodontal tissue. Hard tissue-derived ECM demonstrates strong osteogenic/dental induction abilities, particularly odontogenic TDM, which not only possesses favorable physicochemical properties but also robust biological induction capabilities, making it a promising biological material for periodontal tissue regeneration.
The aforementioned biomimetic materials have played a significant role in periodontal tissue regeneration, yet they are associated with certain limitations. First and foremost, periodontal tissues—which comprise the gingiva, periodontal ligament, cementum, and alveolar bone—are essential for supporting and anchoring teeth within the jawbone. However, regenerating the complex structural architecture of these tissues remains a major challenge. To address this, scaffolds must be carefully designed and fabricated according to specific clinical requirements to fully realize their functional potential. Novel processing technologies aim to achieve structural features that mimic the extracellular matrix at multiple levels, strategies to emulate cell–extracellular matrix interactions, and biological delivery approaches to recapitulate signaling cascades or developmental and wound-healing programs (Ma, 2008). The rapid evolution of bioprinting has accelerated traditional regenerative medicine, making the fabrication of multilayered scaffolds—essential for targeting the periodontal ligament—conceivable. Physiological mechanical loading is fundamental for generating this complex anatomical structure ex vivo (Roato et al., 2022). The 3D bioprinting technology, particularly when employing biomaterials embedded with living cells and bioactive molecules, enables precise and controllable modulation of key structural parameters, including porosity, interconnectivity, pore size, and permeability. This additive manufacturing approach builds complex 3D architectures by sequentially depositing cell-laden hydrogels or other biomaterials in a layer-by-layer manner. Although various 3D bioprinting techniques have been developed, each has distinct advantages and limitations. Therefore, selecting an appropriate 3D bioprinting method that can accurately replicate the intricate structure and functional characteristics of native periodontal tissues is critical for reconstructing a conducive regenerative microenvironment and promoting effective tissue regeneration. Furthermore, achieving mechanical properties that closely match those of natural periodontal tissues remains a significant challenge. Mechanical loading induces proper alignment of the fibers that constitute the periodontal ligament and maintains tissue homeostasis, whereas excessive loading or inadequate adaptation to mechanical stimuli may partially contribute to aberrant tissue regeneration when using PDLSCs (Roato et al., 2022). With respect to mechanical performance, scaffolds should exhibit Young’s modulus and compressive strength comparable to those of native periodontal tissues. Specifically, physical properties of biomaterials—such as pore size, porosity, particle size, and mechanical strength—play a crucial role in regulating cell attachment and proliferation. For instance, high porosity and interconnectivity facilitate the establishment of an optimal microenvironment that enhances cell-to-cell communication and promotes seamless integration of the scaffold into surrounding tissue at the implant site.
6 Conclusions and future perspectives
Owing to the complex tissue structure and diverse cell composition of periodontal tissue, biomimetic materials used for periodontal tissue regeneration must possess excellent biological properties. They should not only mimic the ECM, participate in the release of signaling molecules, and regulate stem cell behavior but should also exhibit optimal physical and chemical characteristics, such as appropriate mechanical strength, suitable porosity, and a degradation rate compatible with tissue regeneration. Although the application of biomimetic materials in periodontal tissue regeneration has been extensively studied, several key challenges remain: (1) enhancing the biocompatibility of biomimetic materials to more effectively induce the differentiation of odontogenic stem cells; (2) optimizing the physical properties of biomimetic materials to better reconstruct the sandwich-like structure of natural periodontal tissue; (3) developing simpler and more efficient material preparation and preservation methods; (4) optimizing the mechanical properties of the scaffold, which is critically important for both tissue regeneration and the maintenance of long-term physiological function; (5) leveraging 3D printing technology to adapt to the personalized architecture of periodontal tissue by improving the printability of various biomimetic materials and enhancing printing accuracy—this represents a promising research direction; and (6) expanding the application of dECM, which lays a solid foundation for clinical research on periodontal tissue regeneration, although clinical data remain limited and further studies are needed. Although most studies have yielded encouraging results, extensive clinical trials are required to confirm their efficacy. In summary, although biomimetic materials have shown significant promise for periodontal tissue regeneration, substantial efforts are required to translate these findings into routine clinical practice.
Author contributions
QZ: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Supervision, Writing – original draft. CG: Data curation, Formal analysis, Funding acquisition, Resources, Visualization, Writing – original draft. ZZ: Funding acquisition, Project administration, Resources, Visualization, Writing – review and editing.
Funding
The authors declare that financial support was received for the research and/or publication of this article. This research was funded by the Chengdu Medical College Research Program with grants CYFY-GQ75 and CYFY-GQ72.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Abdelaziz, D., Hefnawy, A., Al-Wakeel, E., El-Fallal, A., and El-Sherbiny, I. M. (2021). New biodegradable nanoparticles-in-nanofibers based membranes for guided periodontal tissue and bone regeneration with enhanced antibacterial activity. J. Adv. Res. 28, 51–62. doi:10.1016/j.jare.2020.06.014
Abdo, V. L., Suarez, L. J., De Paula, L. G., Costa, R. C., Shibli, J., Feres, M., et al. (2023). Underestimated microbial infection of resorbable membranes on guided regeneration. Colloids Surfaces B Biointerfaces 226, 113318. doi:10.1016/j.colsurfb.2023.113318
Aguilar, A., Zein, N., Harmouch, E., Hafdi, B., Bornert, F., Offner, D., et al. (2019). Application of chitosan in bone and dental engineering. Molecules 24, 3009. doi:10.3390/molecules24163009
Ammar, M. M., Waly, G. H., Saniour, S. H., and Moussa, T. A. (2018). Growth factor release and enhanced encapsulated periodontal stem cells viability by freeze-dried platelet concentrate loaded thermo-sensitive hydrogel for periodontal regeneration. Saudi Dent. J. 30, 355–364. doi:10.1016/j.sdentj.2018.06.002
Andrei, V., Fiț, N. I., Matei, I., Barabás, R., Bizo, L. A., Cadar, O., et al. (2022). In vitro antimicrobial effect of novel electrospun polylactic acid/hydroxyapatite nanofibres loaded with doxycycline. Mater. (Basel) 15, 6225. doi:10.3390/ma15186225
Aravamudhan, A., M. Ramos, D., Nip, J., Subramanian, A., James, R., D. Harmon, M., et al. (2013). Osteoinductive small molecules: growth factor alternatives for bone tissue engineering. CPD 19, 3420–3428. doi:10.2174/1381612811319190008
Bakshi, P. S., Selvakumar, D., Kadirvelu, K., and Kumar, N. S. (2020). Chitosan as an environment friendly biomaterial - a review on recent modifications and applications. Int. J. Biol. Macromol. 150, 1072–1083. doi:10.1016/j.ijbiomac.2019.10.113
Bee, S.-L., and Hamid, Z. A. A. (2025). Chitosan-based dental barrier membrane for periodontal guided tissue regeneration and guided bone regeneration: a review. Int. J. Biol. Macromol. 295, 139504. doi:10.1016/j.ijbiomac.2025.139504
Ben-Arfa, B. A. E., Salvado, I. M. M., Ferreira, J. M. F., and Pullar, R. C. (2017). Novel route for rapid sol-gel synthesis of hydroxyapatite, avoiding ageing and using fast drying with a 50-fold to 200-fold reduction in process time. Mater. Sci. Eng. C 70, 796–804. doi:10.1016/j.msec.2016.09.054
Bernkop-Schnürch, A., and Dünnhaupt, S. (2012). Chitosan-based drug delivery systems. Eur. J. Pharm. Biopharm. 81, 463–469. doi:10.1016/j.ejpb.2012.04.007
Bertsch, C., Maréchal, H., Gribova, V., Lévy, B., Debry, C., Lavalle, P., et al. (2023). Biomimetic bilayered scaffolds for tissue engineering: from current design strategies to medical applications. Adv. Healthc. Mater. 12, 2203115. doi:10.1002/adhm.202203115
Bharadwaz, A., and Jayasuriya, A. C. (2020). Recent trends in the application of widely used natural and synthetic polymer nanocomposites in bone tissue regeneration. Mater Sci. Eng. C Mater Biol. Appl. 110, 110698. doi:10.1016/j.msec.2020.110698
Bhattacharyya, A., Ham, H., Sonh, J., Gunbayar, M., Jeffy, R., Nagarajan, R., et al. (2023). 3D bioprinting of complex tissue scaffolds with in situ homogeneously mixed alginate-chitosan-kaolin bioink using advanced portable biopen. Carbohydr. Polym. 317, 121046. doi:10.1016/j.carbpol.2023.121046
Bhattacharyya, A., Khatun, M. R., Narmatha, S., Nagarajan, R., and Noh, I. (2024). Modulation of 3D bioprintability in polysaccharide bioink by bioglass nanoparticles and multiple metal ions for tissue engineering. Tissue Eng. Regen. Med. 21, 261–275. doi:10.1007/s13770-023-00605-1
Bottino, M. C., Thomas, V., and Janowski, G. M. (2011). A novel spatially designed and functionally graded electrospun membrane for periodontal regeneration. Acta Biomater. 7, 216–224. doi:10.1016/j.actbio.2010.08.019
Brown-Etris, M., Milne, C. T., and Hodde, J. P. (2019). An extracellular matrix graft (Oasis® wound matrix) for treating full-thickness pressure ulcers: a randomized clinical trial. J. Tissue Viability 28, 21–26. doi:10.1016/j.jtv.2018.11.001
Bružauskaitė, I., Bironaitė, D., Bagdonas, E., and Bernotienė, E. (2016). Scaffolds and cells for tissue regeneration: different scaffold pore sizes-different cell effects. Cytotechnology 68, 355–369. doi:10.1007/s10616-015-9895-4
Carletti, E., Motta, A., and Migliaresi, C. (2011). Scaffolds for tissue engineering and 3D cell culture. Methods Mol. Biol. 695, 17–39. doi:10.1007/978-1-60761-984-0_2
Carotenuto, F., Politi, S., Ul Haq, A., De Matteis, F., Tamburri, E., Terranova, M. L., et al. (2022). From soft to hard biomimetic materials: Tuning Micro/nano-architecture of scaffolds for tissue regeneration. Micromachines (Basel) 13, 780. doi:10.3390/mi13050780
Chang, P., Luo, H., Lin, Z., Tai, W., Chang, C., Chang, Y., et al. (2021). Regeneration of critical-sized mandibular defect using a 3D-printed hydroxyapatite-based scaffold: an exploratory study. J. Periodontology 92, 428–435. doi:10.1002/JPER.20-0110
Chen, Y.-W., Yeh, C.-H., and Shie, M.-Y. (2015). Stimulatory effects of the fast setting and suitable degrading ca–si–mg cement on both cementogenesis and angiogenesis differentiation of human periodontal ligament cells. J. Mat. Chem. B 3, 7099–7108. doi:10.1039/C5TB00713E
Chen, Y., Dai, F., Deng, T., Wang, L., Yang, Y., He, C., et al. (2023). An injectable MB/BG@LG sustained release lipid gel with antibacterial and osteogenic properties for efficient treatment of chronic periodontitis in rats. Mater. Today Bio 21, 100699. doi:10.1016/j.mtbio.2023.100699
Chen, H., Ji, X.-R., Zhang, Q., Tian, X.-Z., Zhang, B.-X., and Tang, P.-F. (2016). Effects of calcium sulfate combined with platelet-rich plasma on restoration of long bone defect in rabbits. Chin. Med. J. 129, 557–561. doi:10.4103/0366-6999.176981
Chen, K., Guo, B., and Luo, J. (2017). Quaternized carboxymethyl chitosan/organic montmorillonite nanocomposite as a novel cosmetic ingredient against skin aging. Carbohydr. Polym. 173, 100–106. doi:10.1016/j.carbpol.2017.05.088
Chen, L., Shen, R., Komasa, S., Xue, Y., Jin, B., Hou, Y., et al. (2017). Drug-loadable calcium alginate hydrogel system for use in oral bone tissue repair. Int. J. Mol. Sci. 18, 989. doi:10.3390/ijms18050989
Chen, X., Bai, S., Li, B., Liu, H., Wu, G., Liu, S., et al. (2016). Fabrication of gelatin methacrylate/nanohydroxyapatite microgel arrays for periodontal tissue regeneration. Int. J. Nanomedicine 11, 4707–4718. doi:10.2147/IJN.S111701
Chen, Y. W., Hsu, T.-T., Wang, K., and Shie, M.-Y. (2016). Preparation of the fast setting and degrading ca–si–mg cement with both odontogenesis and angiogenesis differentiation of human periodontal ligament cells. Mater. Sci. Eng. C 60, 374–383. doi:10.1016/j.msec.2015.11.064
Cheng, H., Guo, Q., Zhao, H., Liu, K., Kang, H., Gao, F., et al. (2022). An injectable hydrogel scaffold loaded with dual-drug/sustained-release PLGA microspheres for the regulation of macrophage polarization in the treatment of intervertebral disc degeneration. Int. J. Mol. Sci. 24, 390. doi:10.3390/ijms24010390
Cheng, G., Guo, S., Li, M., Xiao, S., Jiang, B., and Ding, Y. (2024). Hydroxyapatite-coated small intestinal submucosa membranes enhanced periodontal tissue regeneration through immunomodulation and osteogenesis via BMP-2/Smad signaling pathway. Adv. Healthc. Mater. 13, 2301479. doi:10.1002/adhm.202301479
Chmielewski, M., Pilloni, A., and Adamska, P. (2025). Advanced platelet-rich fibrin plus (A-PRF+) as an additive to hard tissue managing protocols in oral surgery: a systematic review. J. Funct. Biomater. 16, 145. doi:10.3390/jfb16040145
Christgau, M., Moder, D., Hiller, K.-A., Dada, A., Schmitz, G., and Schmalz, G. (2006). Growth factors and cytokines in autologous platelet concentrate and their correlation to periodontal regeneration outcomes. J. Clin. Periodontol. 33, 837–845. doi:10.1111/j.1600-051X.2006.00991.x
Conner, K. A., Sabatini, R., Mealey, B. L., Takacs, V. J., Mills, M. P., and Cochrann, D. L. (2003). Guided bone regeneration around titanium plasma-sprayed, acid-etched, and hydroxyapatite-coated implants in the canine model. J. Periodontology 74, 658–668. doi:10.1902/jop.2003.74.5.658
Coura, G. S., Garcez, R. C., De Aguiar, C. B. N. M., Alvarez-Silva, M., Magini, R. S., and Trentin, A. G. (2008). Human periodontal ligament: a niche of neural crest stem cells. J Periodontal Res. 43, 531–536. doi:10.1111/j.1600-0765.2007.01065.x
Crapo, P. M., Gilbert, T. W., and Badylak, S. F. (2011). An overview of tissue and whole organ decellularization processes. Biomaterials 32, 3233–3243. doi:10.1016/j.biomaterials.2011.01.057
Daghrery, A., Ferreira, J. A., De Souza Araújo, I. J., Clarkson, B. H., Eckert, G. J., Bhaduri, S. B., et al. (2021). A highly ordered, nanostructured fluorinated cap-coated melt electrowritten scaffold for periodontal tissue regeneration. Adv. Healthc. Mater. 10, 2101152. doi:10.1002/adhm.202101152
Daghrery, A., Ferreira, J. A., Xu, J., Golafshan, N., Kaigler, D., Bhaduri, S. B., et al. (2023). Tissue-specific melt electrowritten polymeric scaffolds for coordinated regeneration of soft and hard periodontal tissues. Bioact. Mater. 19, 268–281. doi:10.1016/j.bioactmat.2022.04.013
Danhier, F., Ansorena, E., Silva, J. M., Coco, R., Le Breton, A., and Préat, V. (2012). PLGA-Based nanoparticles: an overview of biomedical applications. J. Control. Release 161, 505–522. doi:10.1016/j.jconrel.2012.01.043
Dapeng, L., Xiaojie, L., Ping, G., Yan, D., and Gang, S. (2014). Erk1/2 signalling is involved in the differentiation of periodontal ligament stem cells to schwann cells in dog. Archives Oral Biol. 59, 487–491. doi:10.1016/j.archoralbio.2014.02.010
Datta, S., Rameshbabu, A. P., Bankoti, K., Roy, M., Gupta, C., Jana, S., et al. (2021). Decellularized bone matrix/oleoyl chitosan derived supramolecular injectable hydrogel promotes efficient bone integration. Mater. Sci. Eng. C 119, 111604. doi:10.1016/j.msec.2020.111604
Dey, M., and Ozbolat, I. T. (2020). 3D bioprinting of cells, tissues and organs. Sci. Rep. 10, 14023. doi:10.1038/s41598-020-70086-y
Di Luca, A., Ostrowska, B., Lorenzo-Moldero, I., Lepedda, A., Swieszkowski, W., Van Blitterswijk, C., et al. (2016). Gradients in pore size enhance the osteogenic differentiation of human mesenchymal stromal cells in three-dimensional scaffolds. Sci. Rep. 6, 22898. doi:10.1038/srep22898
Diaz-Gomez, L., Elizondo, M. E., Koons, G. L., Diba, M., Chim, L. K., Cosgriff-Hernandez, E., et al. (2020). Fiber engraving for bioink bioprinting within 3D printed tissue engineering scaffolds. Bioprinting 18, e00076. doi:10.1016/j.bprint.2020.e00076
Diloksumpan, P., Bolaños, R. V., Cokelaere, S., Pouran, B., de Grauw, J., van Rijen, M., et al. (2020). Orthotopic bone regeneration within 3D printed bioceramic scaffolds with region-dependent porosity gradients in an equine model. Adv. Healthc. Mater 9, e1901807. doi:10.1002/adhm.201901807
Ding, S.-L., Zhao, X.-Y., Xiong, W., Ji, L.-F., Jia, M.-X., Liu, Y.-Y., et al. (2023). Cartilage lacuna-inspired microcarriers drive hyaline neocartilage regeneration. Adv. Mater 35, e2212114. doi:10.1002/adma.202212114
Dohan, D. M., Choukroun, J., Diss, A., Dohan, S. L., Dohan, A. J. J., Mouhyi, J., et al. (2006). Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part I: technological concepts and evolution. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 101, e37–e44. doi:10.1016/j.tripleo.2005.07.008
Dos Santos, D. M., Moon, J.-I., Kim, D.-S., Bassous, N. J., Marangon, C. A., Campana-Filho, S. P., et al. (2024). Hierarchical chitin nanocrystal-based 3D printed dual-layer membranes hydrogels: a dual drug delivery nano-platform for periodontal tissue regeneration. ACS Nano 18, 24182–24203. doi:10.1021/acsnano.4c05558
Dunaev, M. V., Kitaev, V. A., Matavkina, M. V., Druzhinin, A. E., and Bubnov, A. S. (2014). Comparative analysis and clinical experience with osteoplastic materials materials based on non-demineralized bone collagen and artificial hydroxylapatite at the close of bone defects in ambulatory surgical dentistry. Vestn. Ross. Akad. Med. Nauk. 2014, 112–120. doi:10.15690/vramn.v69i7-8.1117
Duymaz, D., Kebabci, A. O., and Kizilel, S. (2025). Harnessing the immunomodulatory potential of chitosan and its derivatives for advanced biomedical applications. Int. J. Biol. Macromol. 307, 142055. doi:10.1016/j.ijbiomac.2025.142055
Dziedzic, D. S. M., Mogharbel, B. F., Irioda, A. C., Stricker, P. E. F., Perussolo, M. C., Franco, C. R. C., et al. (2021). Adipose-derived stromal cells and mineralized extracellular matrix delivery by a human decellularized amniotic membrane in periodontal tissue engineering. Membranes 11, 606. doi:10.3390/membranes11080606
Eke, P. I., Borgnakke, W. S., and Genco, R. J. (2020). Recent epidemiologic trends in periodontitis in the USA. Periodontol. 2000 82, 257–267. doi:10.1111/prd.12323
Eliaz, N., and Metoki, N. (2017). Calcium phosphate bioceramics: a review of their history, structure, properties, coating technologies and biomedical applications. Materials 10, 334. doi:10.3390/ma10040334
Fakheran, O., Birang, R., Schmidlin, P. R., Razavi, S. M., and Behfarnia, P. (2019). Retro MTA and tricalcium phosphate/retro MTA for guided tissue regeneration of periodontal dehiscence defects in a dog model: a pilot study. Biomater. Res. 23, 14. doi:10.1186/s40824-019-0163-0
Farshidfar, N., Amiri, M. A., Estrin, N. E., Ahmad, P., Sculean, A., Zhang, Y., et al. (2025). Platelet-rich plasma (PRP) versus injectable platelet-rich fibrin (i-PRF): a systematic review across all fields of medicine. Periodontol 2000, prd.12626. doi:10.1111/prd.12626
Feng, P., Wu, P., Gao, C., Yang, Y., Guo, W., Yang, W., et al. (2018). A multimaterial scaffold with tunable properties: toward bone tissue repair. Adv. Sci. 5, 1700817. doi:10.1002/advs.201700817
Frasca, S., Norol, F., Le Visage, C., Collombet, J.-M., Letourneur, D., Holy, X., et al. (2017). Calcium-phosphate ceramics and polysaccharide-based hydrogel scaffolds combined with mesenchymal stem cell differently support bone repair in rats. J. Mater Sci. Mater Med. 28, 35. doi:10.1007/s10856-016-5839-6
Fujioka-Kobayashi, M., Miron, R. J., Hernandez, M., Kandalam, U., Zhang, Y., and Choukroun, J. (2017). Optimized platelet-rich fibrin with the low-speed concept: growth factor release, biocompatibility, and cellular response. J. Periodontology 88, 112–121. doi:10.1902/jop.2016.160443
Garg, T., Singh, O., Arora, S., and Murthy, R. (2012). Scaffold: a novel carrier for cell and drug delivery. Crit. Rev. Ther. Drug Carr. Syst. 29, 1–63. doi:10.1615/critrevtherdrugcarriersyst.v29.i1.10
Gattazzo, F., Urciuolo, A., and Bonaldo, P. (2014). Extracellular matrix: a dynamic microenvironment for stem cell niche. Biochimica Biophysica Acta (BBA) - General Subj. 1840, 2506–2519. doi:10.1016/j.bbagen.2014.01.010
Generali, M., Kehl, D., Capulli, A. K., Parker, K. K., Hoerstrup, S. P., and Weber, B. (2017). Comparative analysis of poly-glycolic acid-based hybrid polymer starter matrices for in vitro tissue engineering. Colloids Surfaces B Biointerfaces 158, 203–212. doi:10.1016/j.colsurfb.2017.06.046
Gharacheh, H., and Guvendiren, M. (2022). Cell-laden composite hydrogel bioinks with human bone allograft particles to enhance stem cell osteogenesis. Polymers 14, 3788. doi:10.3390/polym14183788
Gonzalez-Fernandez, P., Rodríguez-Nogales, C., Jordan, O., and Allémann, E. (2022). Combination of mesenchymal stem cells and bioactive molecules in hydrogels for osteoarthritis treatment. Eur. J. Pharm. Biopharm. 172, 41–52. doi:10.1016/j.ejpb.2022.01.003
Gopalakrishna, P. K., Jayaramu, R. A., Boregowda, S. S., Eshwar, S., Suresh, N. V., Abu Lila, A. S., et al. (2023). Piperine-loaded in situ gel: formulation, in vitro characterization, and clinical evaluation against periodontitis. Gels 9, 577. doi:10.3390/gels9070577
Gou, M., Huang, Y.-Z., Hu, J.-G., Jiang, Y.-L., Zhang, X.-Z., Su, N.-C., et al. (2019). Epigallocatechin-3-gallate cross-linked small intestinal submucosa for guided bone regeneration. ACS Biomater. Sci. Eng. 5, 5024–5035. doi:10.1021/acsbiomaterials.9b00920
Gou, Y., Huang, Y., Luo, W., Li, Y., Zhao, P., Zhong, J., et al. (2024). Adipose-derived mesenchymal stem cells (MSCs) are a superior cell source for bone tissue engineering. Bioact. Mater. 34, 51–63. doi:10.1016/j.bioactmat.2023.12.003
Gugliandolo, A., Fonticoli, L., Trubiani, O., Rajan, T. S., Marconi, G. D., Bramanti, P., et al. (2021). Oral bone tissue regeneration: mesenchymal stem cells, secretome, and biomaterials. IJMS 22, 5236. doi:10.3390/ijms22105236
Guo, Y., Shi, Z., Han, L., Qin, X., You, J., Zhang, Q., et al. (2024). Infection-sensitive SPION/PLGA scaffolds promote periodontal regeneration via antibacterial activity and macrophage-phenotype modulation. ACS Appl. Mat. Interfaces 16, 41855–41868. doi:10.1021/acsami.4c06430
Hade, M. D., Suire, C. N., and Suo, Z. (2021). Mesenchymal stem cell-derived exosomes: applications in regenerative medicine. Cells 10, 1959. doi:10.3390/cells10081959
Hajishengallis, G., and Chavakis, T. (2021). Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nat. Rev. Immunol. 21, 426–440. doi:10.1038/s41577-020-00488-6
Hao, X., Jiang, B., Wu, J., Xiang, D., Xiong, Z., Li, C., et al. (2024). Nanomaterials for bone metastasis. J. Control. Release 373, 640–651. doi:10.1016/j.jconrel.2024.07.067
He, Y., Wang, X., He, Y., Zhao, X., Lin, J., Feng, Y., et al. (2022). A bioinspired janus polyurethane membrane for potential periodontal tissue regeneration. J. Mater Chem. B 10, 2602–2616. doi:10.1039/d1tb02068d
Hemmati-Sadeghi, S., Ringe, J., Dehne, T., Haag, R., and Sittinger, M. (2018). Hyaluronic acid influence on normal and osteoarthritic tissue-engineered cartilage. IJMS 19, 1519. doi:10.3390/ijms19051519
Ho, M.-H., Huang, K.-Y., Tu, C.-C., Tai, W.-C., Chang, C.-H., Chang, Y.-C., et al. (2022). Functionally graded membrane deposited with PDLLA nanofibers encapsulating doxycycline and enamel matrix derivatives-loaded chitosan nanospheres for alveolar ridge regeneration. Int. J. Biol. Macromol. 203, 333–341. doi:10.1016/j.ijbiomac.2022.01.147
Hu, F., Zhou, Z., Xu, Q., Fan, C., Wang, L., Ren, H., et al. (2019). A novel pH-responsive quaternary ammonium chitosan-liposome nanoparticles for periodontal treatment. Int. J. Biol. Macromol. 129, 1113–1119. doi:10.1016/j.ijbiomac.2018.09.057
Hu, J., Chen, Y., Lin, M., Duan, K., Xu, M., Li, T., et al. (2024). Arginine-loaded globular BSAMA/fibrous GelMA biohybrid cryogels with multifunctional features and enhanced healing for soft gingival tissue regeneration. Int. J. Biol. Macromol. 278, 134932. doi:10.1016/j.ijbiomac.2024.134932
Huang, L., Zhou, B., Wu, H., Zheng, L., and Zhao, J. (2017). Effect of apatite formation of biphasic calcium phosphate ceramic (BCP) on osteoblastogenesis using simulated body fluid (SBF) with or without bovine serum albumin (BSA). Mater. Sci. Eng. C 70, 955–961. doi:10.1016/j.msec.2016.05.115
Huang, J., Wu, Y., Liu, J., Zhang, L., Li, B., Chen, L., et al. (2021). Decellularized matrix could affect the proliferation and differentiation of periodontal ligament stem cells in vitro. J Periodontal Res. 56, 929–939. doi:10.1111/jre.12889
Huang, Y., Zhang, Z., Bi, F., Tang, H., Chen, J., Huo, F., et al. (2023). Personalized 3D-Printed scaffolds with multiple bioactivities for bioroot regeneration. Adv. Healthc. Mater. 12, 2300625. doi:10.1002/adhm.202300625
Hynes, R. O. (2009). The extracellular matrix: not just pretty fibrils. Science 326, 1216–1219. doi:10.1126/science.1176009
Irawan, V., Sung, T.-C., Higuchi, A., and Ikoma, T. (2018). Collagen scaffolds in cartilage tissue engineering and relevant approaches for future development. Tissue Eng. Regen. Med. 15, 673–697. doi:10.1007/s13770-018-0135-9
Ivanov, A. A., Kuznetsova, A. V., Popova, O. P., Danilova, T. I., Latyshev, A. V., and Yanushevich, O. O. (2023). Influence of extracellular matrix components on the differentiation of periodontal ligament stem cells in collagen I hydrogel. Cells 12, 2335. doi:10.3390/cells12192335
Jain, N., Dutt, U., Radenkov, I., and Jain, S. (2024). WHO ’s global oral health status report 2022: actions, discussion and implementation. Oral Dis. 30, 73–79. doi:10.1111/odi.14516
Jeon, O., Lee, Y. B., Lee, S. J., Guliyeva, N., Lee, J., and Alsberg, E. (2022). Stem cell-laden hydrogel bioink for generation of high resolution and fidelity engineered tissues with complex geometries. Bioact. Mater. 15, 185–193. doi:10.1016/j.bioactmat.2021.11.025
Jiang, W., Li, L., Zhang, D., Huang, S., Jing, Z., Wu, Y., et al. (2015). Incorporation of aligned PCL–PEG nanofibers into porous chitosan scaffolds improved the orientation of collagen fibers in regenerated periodontium. Acta Biomater. 25, 240–252. doi:10.1016/j.actbio.2015.07.023
Jiang, Y., Liu, J.-M., Huang, J.-P., Lu, K.-X., Sun, W.-L., Tan, J.-Y., et al. (2021). Regeneration potential of decellularized periodontal ligament cell sheets combined with 15-deoxy-Δ12,14-prostaglandin J2 nanoparticles in a rat periodontal defect. Biomed. Mater 16, 045008. doi:10.1088/1748-605X/abee61
Jiang, H., Wu, L., Randsborg, P.-H., Houck, J., Sun, L., Marine, M., et al. (2023). Analysis of polyethylene-related revisions after total ankle replacements reported in US food and drug administration medical device adverse event database. Foot Ankle Int. 44, 13–20. doi:10.1177/10711007221134284
Kargozar, S., Lotfibakhshaiesh, N., Ai, J., Mozafari, M., Brouki Milan, P., Hamzehlou, S., et al. (2017). Strontium- and cobalt-substituted bioactive glasses seeded with human umbilical cord perivascular cells to promote bone regeneration via enhanced osteogenic and angiogenic activities. Acta Biomater. 58, 502–514. doi:10.1016/j.actbio.2017.06.021
Kargozar, S., Baino, F., Hamzehlou, S., Hill, R. G., and Mozafari, M. (2018). Bioactive glasses: sprouting angiogenesis in tissue engineering. Trends Biotechnol. 36, 430–444. doi:10.1016/j.tibtech.2017.12.003
Katebifar, S., Arul, M., Abdulmalik, S., Yu, X., Alderete, J. F., and Kumbar, S. G. (2023). Novel high-strength polyester composite scaffolds for bone regeneration. Polym. Adv. Techs 34, 3770–3791. doi:10.1002/pat.6178
Kawaguchi, H., Hirachi, A., Hasegawa, N., Iwata, T., Hamaguchi, H., Shiba, H., et al. (2004). Enhancement of periodontal tissue regeneration by transplantation of bone marrow mesenchymal stem cells. J. Periodontology 75, 1281–1287. doi:10.1902/jop.2004.75.9.1281
Keane, T. J., Swinehart, I. T., and Badylak, S. F. (2015). Methods of tissue decellularization used for preparation of biologic scaffolds and in vivo relevance. Methods 84, 25–34. doi:10.1016/j.ymeth.2015.03.005
Khutoryanskiy, V. V. (2011). Advances in mucoadhesion and mucoadhesive polymers. Macromol. Biosci. 11, 748–764. doi:10.1002/mabi.201000388
Kim, I.-Y., Jung, U.-W., Kim, C.-S., Lee, Y.-K., Cho, K.-S., Chai, J.-K., et al. (2007). Effects of a tetracycline blended polylactic and polyglycolic acid membrane on the healing of one-wall intrabony defects in beagle dogs. Biomed. Mater 2, S106–S110. doi:10.1088/1748-6041/2/3/S04
Kim, B. S., Das, S., Jang, J., and Cho, D.-W. (2020). Decellularized extracellular matrix-based bioinks for engineering Tissue- and organ-specific microenvironments. Chem. Rev. 120, 10608–10661. doi:10.1021/acs.chemrev.9b00808
Klokkevold, P. R., Vandemark, L., Kenney, E. B., and Bernard, G. W. (1996). Osteogenesis enhanced by Chitosan (Poly-N-Acetyl Glucosaminoglycan) in vitro. J. Periodontology 67, 1170–1175. doi:10.1902/jop.1996.67.11.1170
Klotz, B. J., Gawlitta, D., Rosenberg, A. J. W. P., Malda, J., and Melchels, F. P. W. (2016). Gelatin-methacryloyl hydrogels: towards biofabrication-based tissue repair. Trends Biotechnol. 34, 394–407. doi:10.1016/j.tibtech.2016.01.002
Kobayashi, E., Flückiger, L., Fujioka-Kobayashi, M., Sawada, K., Sculean, A., Schaller, B., et al. (2016). Comparative release of growth factors from PRP, PRF, and advanced-PRF. Clin. Oral Investig. 20, 2353–2360. doi:10.1007/s00784-016-1719-1
Kolk, A., Handschel, J., Drescher, W., Rothamel, D., Kloss, F., Blessmann, M., et al. (2012). Current trends and future perspectives of bone substitute materials – from space holders to innovative biomaterials. J. Cranio-Maxillofacial Surg. 40, 706–718. doi:10.1016/j.jcms.2012.01.002
Kon, E., Muraglia, A., Corsi, A., Bianco, P., Marcacci, M., Martin, I., et al. (2000). Autologous bone marrow stromal cells loaded onto porous hydroxyapatite ceramic accelerate bone repair in critical-size defects of sheep long bones. J. Biomed. Mat. Res. 49, 328–337. doi:10.1002/(sici)1097-4636(20000305)49:3<328::aid-jbm5>3.0.co;2-q
Koons, G. L., Kontoyiannis, P. D., Diba, M., Chim, L. K., Scott, D. W., and Mikos, A. G. (2021). Effect of 3D printing temperature on bioactivity of bone morphogenetic Protein-2 released from polymeric constructs. Ann. Biomed. Eng. 49, 2114–2125. doi:10.1007/s10439-021-02736-9
Lan, T., Chen, J., Zhang, J., Huo, F., Han, X., Zhang, Z., et al. (2021). Xenoextracellular matrix-rosiglitazone complex-mediated immune evasion promotes xenogenic bioengineered root regeneration by altering M1/M2 macrophage polarization. Biomaterials 276, 121066. doi:10.1016/j.biomaterials.2021.121066
Lan, Q., Cao, J., Bi, X., Xiao, X., Li, D., and Ai, Y. (2023). Curcumin-primed periodontal ligament stem cells-derived extracellular vesicles improve osteogenic ability through the Wnt/β-catenin pathway. Front. Cell Dev. Biol. 11, 1225449. doi:10.3389/fcell.2023.1225449
Laubach, M., Suresh, S., Herath, B., Wille, M.-L., Delbrück, H., Alabdulrahman, H., et al. (2022). Clinical translation of a patient-specific scaffold-guided bone regeneration concept in four cases with large long bone defects. J. Orthop. Transl. 34, 73–84. doi:10.1016/j.jot.2022.04.004
Lee, S. J., Choi, J. S., Park, K. S., Khang, G., Lee, Y. M., and Lee, H. B. (2004). Response of MG63 osteoblast-like cells onto polycarbonate membrane surfaces with different micropore sizes. Biomaterials 25, 4699–4707. doi:10.1016/j.biomaterials.2003.11.034
Lee, B.-S., Lee, C.-C., Lin, H.-P., Shih, W.-A., Hsieh, W.-L., Lai, C.-H., et al. (2016). A functional chitosan membrane with grafted epigallocatechin-3-gallate and lovastatin enhances periodontal tissue regeneration in dogs. Carbohydr. Polym. 151, 790–802. doi:10.1016/j.carbpol.2016.06.026
Lei, Y., Xu, Z., Ke, Q., Yin, W., Chen, Y., Zhang, C., et al. (2017). Strontium hydroxyapatite/chitosan nanohybrid scaffolds with enhanced osteoinductivity for bone tissue engineering. Mater. Sci. Eng. C 72, 134–142. doi:10.1016/j.msec.2016.11.063
Lei, L., Yu, Y., Han, J., Shi, D., Sun, W., Zhang, D., et al. (2020). Quantification of growth factors in advanced platelet-rich fibrin and concentrated growth factors and their clinical efficacy as adjunctive to the GTR procedure in periodontal intrabony defects. J. Periodontology 91, 462–472. doi:10.1002/JPER.19-0290
Lemaitre, M., Monsarrat, P., Blasco-Baque, V., Loubières, P., Burcelin, R., Casteilla, L., et al. (2017). Periodontal tissue regeneration using syngeneic adipose-derived stromal cells in a mouse model. Stem Cells Transl. Med. 6, 656–665. doi:10.5966/sctm.2016-0028
Li, Z., Jiang, C., An, S., Cheng, Q., Huang, Y., Wang, Y., et al. (2014). Immunomodulatory properties of dental tissue-derived mesenchymal stem cells. Oral Dis. 20, 25–34. doi:10.1111/odi.12086
Li, B., Liu, Z., Yang, J., Yi, Z., Xiao, W., Liu, X., et al. (2017). Preparation of bioactive β-tricalcium phosphate microspheres as bone graft substitute materials. Mater. Sci. Eng. C 70, 1200–1205. doi:10.1016/j.msec.2016.03.040
Li, G., Zhao, M., Xu, F., Yang, B., Li, X., Meng, X., et al. (2020). Synthesis and biological application of polylactic acid. Molecules 25, 5023. doi:10.3390/molecules25215023
Li, M., Xing, X., Huang, H., Liang, C., Gao, X., Tang, Q., et al. (2022). BMSC-derived ApoEVs promote craniofacial bone repair via ROS/JNK signaling. J. Dent. Res. 101, 714–723. doi:10.1177/00220345211068338
Li, X.-D., Yan, D.-W., Ren, H.-H., Zhang, Q.-Y., and Yan, Y.-G. (2023). Fabricating biodegradable calcium phosphate/calcium sulfate cement reinforced with cellulose: in vitro and in vivo studies. J. Mat. Chem. B 11, 303–315. doi:10.1039/D2TB02191A
Li, S., Guan, X., Yu, W., Zhao, Z., Sun, Y., and Bai, Y. (2024). Effect of human periodontal ligament stem cell-derived exosomes on cementoblast activity. Oral Dis. 30, 2511–2522. doi:10.1111/odi.14671
Liang, Y., Ma, R., Chen, L., Dai, X., Zuo, S., Jiang, W., et al. (2021). Efficacy of i-PRF in regenerative endodontics therapy for mature permanent teeth with pulp necrosis: study protocol for a multicentre randomised controlled trial. Trials 22, 436. doi:10.1186/s13063-021-05401-7
Liang, C., Liao, L., and Tian, W. (2023). Advances focusing on the application of decellularized extracellular matrix in periodontal regeneration. Biomolecules 13, 673. doi:10.3390/biom13040673
Liang, J., Yi, P., Wang, X., Huang, F., Luan, X., Zhao, Z., et al. (2020). Acellular matrix hydrogel for repair of the temporomandibular joint disc. J. Biomed. Mater Res. 108, 2995–3007. doi:10.1002/jbm.b.34629
Liang, Y., Luan, X., and Liu, X. (2020). Recent advances in periodontal regeneration: a biomaterial perspective. Bioact. Mater. 5, 297–308. doi:10.1016/j.bioactmat.2020.02.012
Liao, Y., Li, H., Shu, R., Chen, H., Zhao, L., Song, Z., et al. (2020). Mesoporous hydroxyapatite/chitosan loaded with recombinant-human amelogenin could enhance antibacterial effect and promote periodontal regeneration. Front. Cell Infect. Microbiol. 10, 180. doi:10.3389/fcimb.2020.00180
Limoee, M., Moradipour, P., Godarzi, M., Arkan, E., and Behbood, L. (2019). Fabrication and in-vitro investigation of polycaprolactone - (Polyvinyl alcohol/Collagen) hybrid nanofiber as anti-inflammatory guided tissue regeneration membrane. Curr. Pharm. Biotechnol. 20, 1122–1133. doi:10.2174/1389201020666190722161004
Liu, Z., Yin, X., Ye, Q., He, W., Ge, M., Zhou, X., et al. (2016). Periodontal regeneration with stem cells-seeded collagen-hydroxyapatite scaffold. J. Biomater. Appl. 31, 121–131. doi:10.1177/0885328216637978
Liu, S., Wang, Y., Yu, L., Li, J., and Ge, S. (2022). Development of a thermosensitive hydrogel loaded with DTT and SDF-1 facilitating in situ periodontal bone regeneration. Chem. Eng. J. 432, 134308. doi:10.1016/j.cej.2021.134308
Liu, S., Yu, J.-M., Gan, Y.-C., Qiu, X.-Z., Gao, Z.-C., Wang, H., et al. (2023). Biomimetic natural biomaterials for tissue engineering and regenerative medicine: new biosynthesis methods, recent advances, and emerging applications. Mil. Med. Res. 10, 16. doi:10.1186/s40779-023-00448-w
Liu, J., Zou, Q., Wang, C., Lin, M., Li, Y., Zhang, R., et al. (2021). Electrospinning and 3D printed hybrid bi-layer scaffold for guided bone regeneration. Mater. and Des. 210, 110047. doi:10.1016/j.matdes.2021.110047
Liu, L., Guo, S., Shi, W., Liu, Q., Huo, F., Wu, Y., et al. (2021). Bone marrow mesenchymal stem cell-derived small extracellular vesicles promote periodontal regeneration. Tissue Eng. Part A 27, 962–976. doi:10.1089/ten.tea.2020.0141
Liu, L., Wen, Y., Chen, L., Li, M., Yu, J., Tian, W., et al. (2024). Xenogenous implanted dental follicle stem cells promote periodontal regeneration through inducing the N2 phenotype of neutrophils. Stem Cell Res. Ther. 15, 270. doi:10.1186/s13287-024-03882-2
Liu, Y., Wei, X., Yang, T., Wang, X., Li, T., Sun, M., et al. (2024). Hyaluronic acid methacrylate/Pluronic F127 hydrogel enhanced with spermidine-modified mesoporous polydopamine nanoparticles for efficient synergistic periodontitis treatment. Int. J. Biol. Macromol. 281, 136085. doi:10.1016/j.ijbiomac.2024.136085
Lu, J., Descamps, M., Dejou, J., Koubi, G., Hardouin, P., Lemaitre, J., et al. (2002). The biodegradation mechanism of calcium phosphate biomaterials in bone. J. Biomed. Mater Res. 63, 408–412. doi:10.1002/jbm.10259
Lu, J., Yu, N., Liu, Q., Xie, Y., and Zhen, L. (2023). Periodontal ligament stem cell exosomes key to regulate periodontal regeneration by miR-31-5p in mice model. IJN 18, 5327–5342. doi:10.2147/IJN.S409664
Luo, J., Chen, H., Wang, G., Lyu, J., Liu, Y., Lin, S., et al. (2023). CGRP-loaded porous microspheres protect BMSCs for alveolar bone regeneration in the periodontitis microenvironment. Adv. Healthc. Mater 12, e2301366. doi:10.1002/adhm.202301366
Luo, P., Yuan, Q., Wan, X., Yang, M., and Xu, P. (2023). Effects of immune cells and cytokines on different cells in OA. JIR 16, 2329–2343. doi:10.2147/JIR.S413578
M, A., I, M. A., Ramalingam, K., S, R., and Perumal, E. (2023). Cytocompatibility and wound healing activity of chitosan thiocolchicoside lauric acid nanogel in human gingival fibroblast cells. Cureus 15, e43727. doi:10.7759/cureus.43727
Ma, P. X. (2008). Biomimetic materials for tissue engineering. Adv. Drug Deliv. Rev. 60, 184–198. doi:10.1016/j.addr.2007.08.041
Ma, Y., Ji, Y., Zhong, T., Wan, W., Yang, Q., Li, A., et al. (2017). Bioprinting-based PDLSC-ECM screening for in vivo repair of alveolar bone defect using cell-laden, injectable and photocrosslinkable hydrogels. ACS Biomater. Sci. Eng. 3, 3534–3545. doi:10.1021/acsbiomaterials.7b00601
Ma, L., Rao, N., Jiang, H., Dai, Y., Yang, S., Yang, H., et al. (2022). Small extracellular vesicles from dental follicle stem cells provide biochemical cues for periodontal tissue regeneration. Stem Cell Res. Ther. 13, 92. doi:10.1186/s13287-022-02767-6
Mao, L. X., Liu, J., Zhao, J., Xia, L., Jiang, L., Wang, X., et al. (2015). Effect of micro-nano-hybrid structured hydroxyapatite bioceramics on osteogenic and cementogenic differentiation of human periodontal ligament stem cell via Wnt signaling pathway. IJN 7031, 7031–7044. doi:10.2147/IJN.S90343
Mei, N., Wu, Y., Chen, B., Zhuang, T., Yu, X., Sui, B., et al. (2022). 3D-printed mesoporous bioactive glass/GelMA biomimetic scaffolds for osteogenic/cementogenic differentiation of periodontal ligament cells. Front. Bioeng. Biotechnol. 10, 950970. doi:10.3389/fbioe.2022.950970
Mengel, R., Soffner, M., and Flores-de-Jacoby, L. (2003). Bioabsorbable membrane and bioactive glass in the treatment of intrabony defects in patients with generalized aggressive periodontitis: results of a 12-Month clinical and radiological study. J. Periodontology 74, 899–908. doi:10.1902/jop.2003.74.6.899
Merkel, A., Chen, Y., Villani, C., and George, A. (2023). GRP78 promotes the osteogenic and angiogenic response in periodontal ligament stem cells. eCM 45, 14–30. doi:10.22203/eCM.v045a02
Miron, R. J., Fujioka-Kobayashi, M., Hernandez, M., Kandalam, U., Zhang, Y., Ghanaati, S., et al. (2017). Injectable platelet rich fibrin (i-PRF): opportunities in regenerative dentistry? Clin. Oral Investig. 21, 2619–2627. doi:10.1007/s00784-017-2063-9
Mirzaee, M., Vaezi, M., and Palizdar, Y. (2016). Synthesis and characterization of silver doped hydroxyapatite nanocomposite coatings and evaluation of their antibacterial and corrosion resistance properties in simulated body fluid. Mater. Sci. Eng. C 69, 675–684. doi:10.1016/j.msec.2016.07.057
Miura, M., Gronthos, S., Zhao, M., Lu, B., Fisher, L. W., Robey, P. G., et al. (2003). SHED: stem cells from human exfoliated deciduous teeth. Proc. Natl. Acad. Sci. U.S.A. 100, 5807–5812. doi:10.1073/pnas.0937635100
Mohd Nor, N. H., Mansor, N. I., Mohd Kashim, M. I. A., Mokhtar, M. H., and Mohd Hatta, F. A. (2023). From teeth to therapy: a review of therapeutic potential within the secretome of stem cells from human exfoliated deciduous teeth. IJMS 24, 11763. doi:10.3390/ijms241411763
Mohd Pu’ad, N. A. S., Koshy, P., Abdullah, H. Z., Idris, M. I., and Lee, T. C. (2019). Syntheses of hydroxyapatite from natural sources. Heliyon 5, e01588. doi:10.1016/j.heliyon.2019.e01588
Morsczeck, C., Götz, W., Schierholz, J., Zeilhofer, F., Kühn, U., Möhl, C., et al. (2005). Isolation of precursor cells (PCs) from human dental follicle of wisdom teeth. Matrix Biol. 24, 155–165. doi:10.1016/j.matbio.2004.12.004
Nade, S., Armstrong, L., McCartney, E., and Baggaley, B. (1983). Osteogenesis after bone and bone marrow transplantation. The ability of ceramic materials to sustain osteogenesis from transplanted bone marrow cells: preliminary studies. Clin. Orthop. Relat. Res. 181, 255–263. doi:10.1097/00003086-198312000-00040
Nagahara, T., Yoshimatsu, S., Shiba, H., Kawaguchi, H., Takeda, K., Iwata, T., et al. (2015). Introduction of a mixture of β-Tricalcium phosphate into a complex of bone marrow mesenchymal stem cells and type I collagen can augment the volume of alveolar bone without impairing cementum regeneration. J. Periodontology 86, 456–464. doi:10.1902/jop.2014.140384
Nery, E. B., and Lynch, K. L. (1978). Preliminary clinical studies of bioceramic in periodontal osseous defects. J. Periodontology 49, 523–527. doi:10.1902/jop.1978.49.10.523
Niemczyk, W., Żurek, J., Niemczyk, S., Kępa, M., Zięba, N., Misiołek, M., et al. (2025). Antibiotic-loaded platelet-rich fibrin (AL-PRF) as a new carrier for antimicrobials: a systematic review of in vitro studies. Int. J. Mol. Sci. 26, 2140. doi:10.3390/ijms26052140
Ortiz, A. E., Ahluwalia, J., Song, S. S., and Avram, M. M. (2020). Analysis of U.S. food and drug administration data on soft-tissue filler complications. Dermatol Surg. 46, 958–961. doi:10.1097/DSS.0000000000002208
Ouyang, Z., Chen, X., Wang, Z., Xu, Y., Deng, Z., Xing, L., et al. (2025). Azithromycin-loaded PLGA microspheres coated with silk fibroin ameliorate inflammation and promote periodontal tissue regeneration. Regen. Biomater. 12, rbae146. doi:10.1093/rb/rbae146
Owen, G. R., Jackson, J. K., Chehroudi, B., Brunette, D. M., and Burt, H. M. (2010). An in vitro study of plasticized poly(lactic-co-glycolic acid) films as possible guided tissue regeneration membranes: material properties and drug release kinetics. J. Biomed. Mater Res. A 95, 857–869. doi:10.1002/jbm.a.32865
Pan, J., Deng, J., Yu, L., Wang, Y., Zhang, W., Han, X., et al. (2020). Investigating the repair of alveolar bone defects by gelatin methacrylate hydrogels-encapsulated human periodontal ligament stem cells. J. Mater Sci. Mater Med. 31, 3. doi:10.1007/s10856-019-6333-8
Papadimitropoulos, A., Scotti, C., Bourgine, P., Scherberich, A., and Martin, I. (2015). Engineered decellularized matrices to instruct bone regeneration processes. Bone 70, 66–72. doi:10.1016/j.bone.2014.09.007
Papapanou, P. N., and Susin, C. (2017). Periodontitis epidemiology: is periodontitis under-recognized, over-diagnosed, or both? Periodontol. 2000 75, 45–51. doi:10.1111/prd.12200
Parirokh, M., Torabinejad, M., and Dummer, P. M. H. (2018). Mineral trioxide aggregate and other bioactive endodontic cements: an updated overview – part I: vital pulp therapy. Int. Endod. J. 51, 177–205. doi:10.1111/iej.12841
Park, C. H. (2019). Biomaterial-based approaches for regeneration of periodontal ligament and cementum using 3D platforms. Int. J. Mol. Sci. 20, 4364. doi:10.3390/ijms20184364
Park, J. H., Gillispie, G. J., Copus, J. S., Zhang, W., Atala, A., Yoo, J. J., et al. (2020). The effect of BMP-mimetic peptide tethering bioinks on the differentiation of dental pulp stem cells (DPSCs) in 3D bioprinted dental constructs. Biofabrication 12, 035029. doi:10.1088/1758-5090/ab9492
Peng, S., Fu, H., Li, R., Li, H., Wang, S., Li, B., et al. (2024). A new direction in periodontitis treatment: biomaterial-mediated macrophage immunotherapy. J. Nanobiotechnol 22, 359. doi:10.1186/s12951-024-02592-4
Petersen, A., Princ, A., Korus, G., Ellinghaus, A., Leemhuis, H., Herrera, A., et al. (2018). A biomaterial with a channel-like pore architecture induces endochondral healing of bone defects. Nat. Commun. 9, 4430. doi:10.1038/s41467-018-06504-7
Pilipchuk, S. P., Monje, A., Jiao, Y., Hao, J., Kruger, L., Flanagan, C. L., et al. (2016). Integration of 3D printed and micropatterned polycaprolactone scaffolds for guidance of oriented collagenous tissue formation in vivo. Adv. Healthc. Mater. 5, 676–687. doi:10.1002/adhm.201500758
Pina, S., Oliveira, J. M., and Reis, R. L. (2015). Natural-based nanocomposites for bone tissue engineering and regenerative medicine: a review. Adv. Mater 27, 1143–1169. doi:10.1002/adma.201403354
Qasim, S. B., Delaine-Smith, R. M., Fey, T., Rawlinson, A., and Rehman, I. U. (2015). Freeze gelated porous membranes for periodontal tissue regeneration. Acta Biomater. 23, 317–328. doi:10.1016/j.actbio.2015.05.001
Qin, Y., and Li, P. (2020). Antimicrobial chitosan conjugates: current synthetic strategies and potential applications. Int. J. Mol. Sci. 21, 499. doi:10.3390/ijms21020499
Qiu, G., Huang, M., Liu, J., Ma, T., Schneider, A., Oates, T. W., et al. (2022). Human periodontal ligament stem cell encapsulation in alginate-fibrin-platelet lysate microbeads for dental and craniofacial regeneration. J. Dent. 124, 104219. doi:10.1016/j.jdent.2022.104219
Radulescu, D.-M., Neacsu, I. A., Grumezescu, A.-M., and Andronescu, E. (2022). New insights of scaffolds based on hydrogels in tissue engineering. Polymers 14, 799. doi:10.3390/polym14040799
Ranch, K. M., Maulvi, F. A., Koli, A. R., Desai, D. T., Parikh, R. K., and Shah, D. O. (2021). Tailored doxycycline hyclate loaded in situ gel for the treatment of periodontitis: optimization, in vitro characterization, and antimicrobial studies. AAPS PharmSciTech 22, 77. doi:10.1208/s12249-021-01950-x
Ren, B., Chen, X., Du, S., Ma, Y., Chen, H., Yuan, G., et al. (2018). Injectable polysaccharide hydrogel embedded with hydroxyapatite and calcium carbonate for drug delivery and bone tissue engineering. Int. J. Biol. Macromol. 118, 1257–1266. doi:10.1016/j.ijbiomac.2018.06.200
Rexhepi, I., Paolantonio, M., Romano, L., Serroni, M., Santamaria, P., Secondi, L., et al. (2021). Efficacy of inorganic bovine bone combined with leukocyte and platelet-rich fibrin or collagen membranes for treating unfavorable periodontal infrabony defects: randomized non-inferiority trial. J. Periodontology 92, 1576–1587. doi:10.1002/JPER.20-0305
Roato, I., Masante, B., Putame, G., Massai, D., and Mussano, F. (2022). Challenges of periodontal tissue engineering: increasing biomimicry through 3D printing and controlled dynamic environment. Nanomater. (Basel) 12, 3878. doi:10.3390/nano12213878
Rodríguez-Vázquez, M., Vega-Ruiz, B., Ramos-Zúñiga, R., Saldaña-Koppel, D. A., and Quiñones-Olvera, L. F. (2015). Chitosan and its potential use as a scaffold for tissue engineering in regenerative medicine. BioMed Res. Int. 2015, 1–15. doi:10.1155/2015/821279
Sakr, M. A., Sakthivel, K., Hossain, T., Shin, S. R., Siddiqua, S., Kim, J., et al. (2022). Recent trends in gelatin methacryloyl nanocomposite hydrogels for tissue engineering. J. Biomed. Mater Res. A 110, 708–724. doi:10.1002/jbm.a.37310
Sánchez-Cid, P., Jiménez-Rosado, M., Romero, A., and Pérez-Puyana, V. (2022). Novel trends in hydrogel development for biomedical applications: a review. Polymers 14, 3023. doi:10.3390/polym14153023
Sándor, G. K., Numminen, J., Wolff, J., Thesleff, T., Miettinen, A., Tuovinen, V. J., et al. (2014). Adipose stem cells used to reconstruct 13 cases with cranio-maxillofacial hard-tissue defects. Stem Cells Transl. Med. 3, 530–540. doi:10.5966/sctm.2013-0173
Santos, M. S., Carvalho, M. S., and Silva, J. C. (2023a). Recent advances on electrospun nanofibers for periodontal regeneration. Nanomaterials 13, 1307. doi:10.3390/nano13081307
Santos, M. S., Dos Santos, A. B., and Carvalho, M. S. (2023b). New insights in hydrogels for periodontal regeneration. JFB 14, 545. doi:10.3390/jfb14110545
Schepers, E. J., and Ducheyne, P. (1997). Bioactive glass particles of narrow size range for the treatment of oral bone defects: a 1-24 month experiment with several materials and particle sizes and size ranges. J. Oral Rehabil. 24, 171–181. doi:10.1111/j.1365-2842.1997.tb00311.x
Schmidt-Schultz, T. H., and Schultz, M. (2005). Intact growth factors are conserved in the extracellular matrix of ancient human bone and teeth: a storehouse for the study of human evolution in health and disease. Biol. Chem. 386, 767–776. doi:10.1515/BC.2005.090
Scott, T. G., Blackburn, G., Ashley, M., Bayer, I. S., Ghosh, A., Biris, A. S., et al. (2013). Advances in bionanomaterials for bone tissue engineering. J. Nanosci. Nanotech. 13, 1–22. doi:10.1166/jnn.2013.6733
Semino, C. E. (2008). Self-assembling peptides: from bio-inspired materials to bone regeneration. J. Dent. Res. 87, 606–616. doi:10.1177/154405910808700710
Seo, B.-M., Miura, M., Gronthos, S., Mark Bartold, P., Batouli, S., Brahim, J., et al. (2004). Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 364, 149–155. doi:10.1016/S0140-6736(04)16627-0
Seol, Y.-J., Lee, J.-Y., Park, Y.-J., Lee, Y.-M., Ku, Y., Rhyu, I.-C., et al. (2004). Chitosan sponges as tissue engineering scaffolds for bone formation. Biotechnol. Lett. 26, 1037–1041. doi:10.1023/B:BILE.0000032962.79531.fd
Serroni, M., Paolantonio, M., Romano, L., Santamaria, P., Rexhepi, I., Sinjari, B., et al. (2022). Added benefit of L-PRF to autogenous bone grafts in the treatment of degree II furcation involvement in mandibular molars. J. Periodontology 93, 1486–1499. doi:10.1002/JPER.21-0369
Shaffer, C. D., and App, G. R. (1971). The use of plaster of paris in treating infrabony periodontal defects in humans. J. Periodontol. 42, 685–690. doi:10.1902/jop.1971.42.11.685
Shapira, A., and Dvir, T. (2021). 3D tissue and organ printing-hope and reality. Adv. Sci. (Weinh) 8, 2003751. doi:10.1002/advs.202003751
Shi, W., Guo, S., Liu, L., Liu, Q., Huo, F., Ding, Y., et al. (2020). Small extracellular vesicles from lipopolysaccharide-preconditioned dental follicle cells promote periodontal regeneration in an inflammatory microenvironment. ACS Biomater. Sci. Eng. 6, 5797–5810. doi:10.1021/acsbiomaterials.0c00882
Shue, L., Yufeng, Z., and Mony, U. (2012). Biomaterials for periodontal regeneration: a review of ceramics and polymers. Biomatter 2, 271–277. doi:10.4161/biom.22948
Si, Z., Wang, X., Sun, C., Kang, Y., Xu, J., Wang, X., et al. (2019). Adipose-derived stem cells: sources, potency, and implications for regenerative therapies. Biomed. and Pharmacother. 114, 108765. doi:10.1016/j.biopha.2019.108765
Singh, S., Chaurasia, A., Rajput, D. S., and Gupta, N. (2023). Mucoadhesive drug delivery system and there future prospective: are a promising approach for effective treatment? Zhongguo Ying Yong Sheng Li Xue Za Zhi 39, e20230005. doi:10.62958/j.cjap.2023.005
Singhvi, M. S., Zinjarde, S. S., and Gokhale, D. V. (2019). Polylactic acid: synthesis and biomedical applications. J. Appl. Microbiol. 127, 1612–1626. doi:10.1111/jam.14290
Smith, I. O., Liu, X. H., Smith, L. A., and Ma, P. X. (2009). Nanostructured polymer scaffolds for tissue engineering and regenerative medicine. Wiley Interdiscip. Rev. Nanomed Nanobiotechnol 1, 226–236. doi:10.1002/wnan.26
Son, H., Jeon, M., Choi, H.-J., Lee, H.-S., Kim, I.-H., Kang, C.-M., et al. (2019). Decellularized human periodontal ligament for periodontium regeneration. PLoS ONE 14, e0221236. doi:10.1371/journal.pone.0221236
Song, A. M., Shu, R., Xie, Y. F., Song, Z. C., Li, H. Y., Liu, X. F., et al. (2007). A study of enamel matrix proteins on differentiation of porcine bone marrow stromal cells into cementoblasts. Cell Prolif. 40, 381–396. doi:10.1111/j.1365-2184.2007.00441.x
Srinivasan, S., Jayasree, R., Chennazhi, K. P., Nair, S. V., and Jayakumar, R. (2012). Biocompatible alginate/nano bioactive glass ceramic composite scaffolds for periodontal tissue regeneration. Carbohydr. Polym. 87, 274–283. doi:10.1016/j.carbpol.2011.07.058
Stavropoulos, A., Windisch, P., Szendröi-Kiss, D., Peter, R., Gera, I., and Sculean, A. (2010). Clinical and histologic evaluation of granular beta-tricalcium phosphate for the treatment of human intrabony periodontal defects: a report on five cases. J. Periodontology 81, 325–334. doi:10.1902/jop.2009.090386
Subbaiah, R., and Thomas, B. (2011). Efficacy of a bioactive alloplast, in the treatment of human periodontal osseous defects-a clinical study. Med. Oral 16, e239–e244. doi:10.4317/medoral.16.e239
Sufaru, I.-G., Macovei, G., Stoleriu, S., Martu, M.-A., Luchian, I., Kappenberg-Nitescu, D.-C., et al. (2022). 3D printed and bioprinted membranes and scaffolds for the periodontal tissue regeneration: a narrative review. Membranes 12, 902. doi:10.3390/membranes12090902
Sun, W., Liu, Y., Miao, L., Wang, Y., Ren, S., Yang, X., et al. (2016). Controlled release of recombinant human cementum protein 1 from electrospun multiphasic scaffold for cementum regeneration. IJN 11, 3145–3158. doi:10.2147/IJN.S104324
Sun, X., Xu, C., Wu, G., Ye, Q., and Wang, C. (2017). Poly(Lactic-Co-Glycolic acid): applications and future prospects for periodontal tissue regeneration. Polymers 9, 189. doi:10.3390/polym9060189
Sun, H., Luan, J., and Dong, S. (2024). Hydrogels promote periodontal regeneration. Front. Bioeng. Biotechnol. 12, 1411494. doi:10.3389/fbioe.2024.1411494
Suo, L., Wu, H., Wang, P., Xue, Z., Gao, J., and Shen, J. (2023). The improvement of periodontal tissue regeneration using a 3D -printed carbon nanotube/chitosan/sodium alginate composite scaffold. J. Biomed. Mater Res. 111, 73–84. doi:10.1002/jbm.b.35133
Suzuki, M., Kimura, T., Nakano, Y., Kobayashi, M., Okada, M., Matsumoto, T., et al. (2023). Preparation of mineralized pericardium by alternative soaking for soft–hard interregional tissue application. J. Biomed. Mater. Res. 111, 198–208. doi:10.1002/jbm.a.37445
Swain, G. P., Patel, S., Gandhi, J., and Shah, P. (2019). Development of moxifloxacin hydrochloride loaded in-situ gel for the treatment of periodontitis: in-vitro drug release study and antibacterial activity. J. Oral Biol. Craniofacial Res. 9, 190–200. doi:10.1016/j.jobcr.2019.04.001
Taghipour, Y. D., Hokmabad, V. R., Del Bakhshayesh, A. R., Asadi, N., Salehi, R., and Nasrabadi, H. T. (2020). The application of hydrogels based on natural polymers for tissue engineering. CMC 27, 2658–2680. doi:10.2174/0929867326666190711103956
Tan, O. L., Safii, S. H., and Razali, M. (2019). Commercial local pharmacotherapeutics and adjunctive agents for nonsurgical treatment of periodontitis: a contemporary review of clinical efficacies and challenges. Antibiot. (Basel) 9, 11. doi:10.3390/antibiotics9010011
Tan, N., Sabalic-Schoener, M., Nguyen, L., and D’Aiuto, F. (2023). β-Tricalcium phosphate-loaded chitosan-based thermosensitive hydrogel for periodontal regeneration. Polymers 15, 4146. doi:10.3390/polym15204146
Tharakan, S., Khondkar, S., Lee, S., Ahn, S., Mathew, C., Gresita, A., et al. (2022). 3D printed osteoblast–alginate/collagen hydrogels promote survival, proliferation and mineralization at low doses of strontium calcium polyphosphate. Pharmaceutics 15, 11. doi:10.3390/pharmaceutics15010011
Thomas, M. V., and Puleo, D. A. (2009). Calcium sulfate: properties and clinical applications. J. Biomed. Mater Res. 88B, 597–610. doi:10.1002/jbm.b.31269
Tolmacheva, N., Bhattacharyya, A., and Noh, I. (2024). Calcium phosphate biomaterials for 3D bioprinting in bone tissue engineering. Biomimetics (Basel) 9, 95. doi:10.3390/biomimetics9020095
Tozar, T., Nistorescu, S., Gradisteanu Pircalabioru, G., Boni, M., and Staicu, A. (2024). Photopolymerization of chlorpromazine-loaded gelatin methacryloyl hydrogels: characterization and antimicrobial applications. Gels 10, 632. doi:10.3390/gels10100632
Trubiani, O., Di Primio, R., Traini, T., Pizzicannella, J., Scarano, A., Piattelli, A., et al. (2005). Morphological and cytofluorimetric analysis of adult mesenchymal stem cells expanded ex vivo from periodontal ligament. Int. J. Immunopathol. Pharmacol. 18, 213–221. doi:10.1177/039463200501800204
Uchikawa, E., Yoshizawa, M., Li, X., Matsumura, N., Li, N., Chen, K., et al. (2021). Tooth transplantation with a β-tricalcium phosphate scaffold accelerates bone formation and periodontal tissue regeneration. Oral Dis. 27, 1226–1237. doi:10.1111/odi.13634
Vagaská, B., Bacáková, L., Filová, E., and Balík, K. (2010). Osteogenic cells on bio-inspired materials for bone tissue engineering. Physiol. Res. 59, 309–322. doi:10.33549/physiolres.931776
Välimäki, V.-V., and Aro, H. T. (2006). Molecular basis for action of bioactive glasses as bone graft substitute. Scand. J. Surg. 95, 95–102. doi:10.1177/145749690609500204
Varoni, E. M., Vijayakumar, S., Canciani, E., Cochis, A., De Nardo, L., Lodi, G., et al. (2018). Chitosan-based trilayer scaffold for multitissue periodontal regeneration. J. Dent. Res. 97, 303–311. doi:10.1177/0022034517736255
Villaça, J. H., Novaes, Jr., A. B., Souza, S. L. S. D., Taba, Jr., M., Molina, G. O., and Lamano Carvalho, T. L. (2005). Bioactive glass efficacy in the periodontal healing of intrabony defects in monkeys. Braz. Dent. J. 16, 67–74. doi:10.1590/S0103-64402005000100012
Vitale-Brovarone, C., Baino, F., and Verné, E. (2009). High strength bioactive glass-ceramic scaffolds for bone regeneration. J. Mater Sci. Mater Med. 20, 643–653. doi:10.1007/s10856-008-3605-0
Wang, C., Cao, C., Tong, C., Cong, P., Lv, H., and Zhang, Y. (2025). Chitosan scaffold containing melatonin-releasing mesoporous wollastonite intended for periodontal and osseous repair enhances angiogenesis and osteogenesis via SERPINB9P1/miR-545-5p/SIRT6 pathway. Int. J. Biol. Macromol. 310, 142613. doi:10.1016/j.ijbiomac.2025.142613
Wang, M., Li, J., Ye, Y., He, S., and Song, J. (2020). SHED-Derived conditioned exosomes enhance the osteogenic differentiation of PDLSCs via Wnt and BMP signaling in vitro. Differentiation 111, 1–11. doi:10.1016/j.diff.2019.10.003
Wang, S., Niu, Y., Jia, P., Liao, Z., Guo, W., Chaves, R. C., et al. (2022). Alkaline activation of endogenous latent TGFβ1 by an injectable hydrogel directs cell homing for in situ complex tissue regeneration. Bioact. Mater. 15, 316–329. doi:10.1016/j.bioactmat.2021.12.015
Wang, W., Meng, Q., Li, Q., Liu, J., Zhou, M., Jin, Z., et al. (2020). Chitosan derivatives and their application in biomedicine. Int. J. Mol. Sci. 21, 487. doi:10.3390/ijms21020487
Wang, Y., Cao, Z., Wei, Q., Ma, K., Hu, W., Huang, Q., et al. (2022). VH298-loaded extracellular vesicles released from gelatin methacryloyl hydrogel facilitate diabetic wound healing by HIF-1α-mediated enhancement of angiogenesis. Acta Biomater. 147, 342–355. doi:10.1016/j.actbio.2022.05.018
Washington, M. A., Swiner, D. J., Bell, K. R., Fedorchak, M. V., Little, S. R., and Meyer, T. Y. (2017). The impact of monomer sequence and stereochemistry on the swelling and erosion of biodegradable poly(lactic-co-glycolic acid) matrices. Biomaterials 117, 66–76. doi:10.1016/j.biomaterials.2016.11.037
Watt, F. M., and Huck, W. T. S. (2013). Role of the extracellular matrix in regulating stem cell fate. Nat. Rev. Mol. Cell Biol. 14, 467–473. doi:10.1038/nrm3620
Wei, J., Song, Y., Du, Z., Yu, F., Zhang, Y., Jiang, N., et al. (2020). Exosomes derived from human exfoliated deciduous teeth ameliorate adult bone loss in mice through promoting osteogenesis. J. Mol. Histol. 51, 455–466. doi:10.1007/s10735-020-09896-3
Wei, X., Guo, S., Liu, Q., Liu, L., Huo, F., Wu, Y., et al. (2023). Dental follicle stem cells promote periodontal regeneration through periostin-mediated macrophage infiltration and reprogramming in an inflammatory microenvironment. Int J Mol Sci 24, 6353. doi:10.3390/ijms24076353
Wu, J., Chen, L., Wang, R., Song, Z., Shen, Z., Zhao, Y., et al. (2019). Exosomes secreted by stem cells from human exfoliated deciduous teeth promote alveolar bone defect repair through the regulation of angiogenesis and osteogenesis. ACS Biomater. Sci. Eng. 5, 3561–3571. doi:10.1021/acsbiomaterials.9b00607
Wu, H., Li, Y., Shi, L., Liu, Y., and Shen, J. (2025). New advances in periodontal functional materials based on antibacterial, anti-inflammatory, and tissue regeneration strategies. Adv. Healthc. Mater. 14, 2403206. doi:10.1002/adhm.202403206
Xiong, X., Yang, X., Dai, H., Feng, G., Zhang, Y., Zhou, J., et al. (2019). Extracellular matrix derived from human urine-derived stem cells enhances the expansion, adhesion, spreading, and differentiation of human periodontal ligament stem cells. Stem Cell Res. Ther. 10, 396. doi:10.1186/s13287-019-1483-7
Xu, C., Lei, C., Meng, L., Wang, C., and Song, Y. (2012). Chitosan as a barrier membrane material in periodontal tissue regeneration. J. Biomed. Mater Res. 100B, 1435–1443. doi:10.1002/jbm.b.32662
Xu, X., Gu, Z., Chen, X., Shi, C., Liu, C., Liu, M., et al. (2019). An injectable and thermosensitive hydrogel: promoting periodontal regeneration by controlled-release of aspirin and erythropoietin. Acta Biomater. 86, 235–246. doi:10.1016/j.actbio.2019.01.001
Xu, H., Yan, S., Gerhard, E., Xie, D., Liu, X., Zhang, B., et al. (2024). Citric acid: a nexus between cellular mechanisms and biomaterial innovations. Adv. Mater. 36, 2402871. doi:10.1002/adma.202402871
Xu, X., Jha, A. K., Harrington, D. A., Farach-Carson, M. C., and Jia, X. (2012). Hyaluronic acid-based hydrogels: from a natural polysaccharide to complex networks. Soft Matter 8, 3280. doi:10.1039/c2sm06463d
Yamada, S., Shanbhag, S., and Mustafa, K. (2022). Scaffolds in periodontal regenerative treatment. Dent. Clin. N. Am. 66, 111–130. doi:10.1016/j.cden.2021.06.004
Yan, X., Yan, F., Mohammed, H. A. G., and Liu, O. (2021). Maxillofacial-derived mesenchymal stem cells: characteristics and progress in tissue regeneration. Stem Cells Int. 2021, 1–22. doi:10.1155/2021/5516521
Yan, C., Zhang, P., Qin, Q., Jiang, K., Luo, Y., Xiang, C., et al. (2024). 3D-printed bone regeneration scaffolds modulate bone metabolic homeostasis through vascularization for osteoporotic bone defects. Biomaterials 311, 122699. doi:10.1016/j.biomaterials.2024.122699
Yang, R., Ye, F., Cheng, L., Wang, J., Lu, X., Shi, Y., et al. (2011). Osteoinduction by Ca-P biomaterials implanted into the muscles of mice. J. Zhejiang Univ. Sci. B 12, 582–590. doi:10.1631/jzus.B1000204
Yang, Y., Zhang, B., Yang, Y., Peng, B., and Ye, R. (2022). PLGA containing human adipose-derived stem cell-derived extracellular vesicles accelerates the repair of alveolar bone defects via transfer of CGRP. Oxidative Med. Cell. Longev. 2022, 4815284–14. doi:10.1155/2022/4815284
Yang, X., Ma, Y., Wang, X., Yuan, S., Huo, F., Yi, G., et al. (2023). A 3D-Bioprinted functional module based on decellularized extracellular matrix bioink for periodontal regeneration. Adv. Sci. 10, 2205041. doi:10.1002/advs.202205041
Yang, Y., Xu, C., Xu, S., Li, Y., Chen, K., Yang, T., et al. (2025). Injectable hydrogels activated with copper sulfide nanoparticles for enhancing spatiotemporal sterilization and osteogenesis in periodontal therapy. Biomater. Sci. 13, 1434–1448. doi:10.1039/D3BM02134C
Yao, Q., Cosme, J. G. L., Xu, T., Miszuk, J. M., Picciani, P. H. S., Fong, H., et al. (2017). Three dimensional electrospun PCL/PLA blend nanofibrous scaffolds with significantly improved stem cells osteogenic differentiation and cranial bone formation. Biomaterials 115, 115–127. doi:10.1016/j.biomaterials.2016.11.018
Yao, H., Luo, J., Deng, Y., Li, Z., and Wei, J. (2022). Alginate-modified mesoporous bioactive glass and its drug delivery, bioactivity, and osteogenic properties. Front. Bioeng. Biotechnol. 10, 994925. doi:10.3389/fbioe.2022.994925
Yeo, Y., Jeon, D., Kim, C.-S., Choi, S., Cho, K., Lee, Y., et al. (2005). Effects of chitosan nonwoven membrane on periodontal healing of surgically created one-wall intrabony defects in beagle dogs. J. Biomed. Mater Res. 72B, 86–93. doi:10.1002/jbm.b.30121
Yoshino, Y., Miyaji, H., Nishida, E., Kanemoto, Y., Hamamoto, A., Kato, A., et al. (2023). Periodontal tissue regeneration by recombinant human collagen peptide granules applied with β-tricalcium phosphate fine particles. J. Oral Biosci. 65, 62–71. doi:10.1016/j.job.2023.01.002
Zambonin, G., Losito, I., Triffitt, J. T., and Zambonin, C. G. (2000). Detection of collagen synthesis by human osteoblasts on a tricalcium phosphate hydroxyapatite: an X-ray photoelectron spectroscopy investigation. J. Biomed. Mat. Res. 49, 120–126. doi:10.1002/(sici)1097-4636(200001)49:1<120::aid-jbm15>3.0.co;2-q
Zamet, J. S., Darbar, U. R., Griffiths, G. S., Bulman, J. S., Brägger, U., Bürgin, W., et al. (1997). Particulate bioglass® as a grafting material in the treatment of periodontal intrabony defects. J. Clin. Periodontol. 24, 410–418. doi:10.1111/j.1600-051X.1997.tb00205.x
Zang, S., Mu, R., Chen, F., Wei, X., Zhu, L., Han, B., et al. (2019). Injectable chitosan/β-glycerophosphate hydrogels with sustained release of BMP-7 and ornidazole in periodontal wound healing of class III furcation defects. Mater. Sci. Eng. C 99, 919–928. doi:10.1016/j.msec.2019.02.024
Zeinali, R., Del Valle, L. J., Torras, J., and Puiggalí, J. (2021). Recent progress on biodegradable tissue engineering scaffolds prepared by thermally-induced phase separation (TIPS). Int. J. Mol. Sci. 22, 3504. doi:10.3390/ijms22073504
Zhang, Y., Xiong, Y., Chen, X., Chen, C., Zhu, Z., and Li, L. (2018). Therapeutic effect of bone marrow mesenchymal stem cells pretreated with acetylsalicylic acid on experimental periodontitis in rats. Int. Immunopharmacol. 54, 320–328. doi:10.1016/j.intimp.2017.11.028
Zhang, J., Ding, H., Liu, X., Sheng, Y., Liu, X., and Jiang, C. (2019). Dental follicle stem cells: tissue engineering and immunomodulation. Stem Cells Dev. 28, 986–994. doi:10.1089/scd.2019.0012
Zhang, J., Liu, Y., Chen, Y., Yuan, L., Liu, H., Wang, J., et al. (2020). Adipose-derived stem cells: current applications and future directions in the regeneration of multiple tissues. Stem Cells Int. 2020, 1–26. doi:10.1155/2020/8810813
Zhang, H., Jiao, C., Liu, Z., He, Z., Mengxing, G., Wang, C., et al. (2021). 3D-printed composite, calcium silicate ceramic doped with CaSO4·2H2O: degradation performance and biocompatibility. J. Mech. Behav. Biomed. Mater 121, 104642. doi:10.1016/j.jmbbm.2021.104642
Zhang, L., Yu, Z., Liu, S., Liu, F., Zhou, S., Zhang, Y., et al. (2025). Advanced progress of adipose-derived stem cells-related biomaterials in maxillofacial regeneration. Stem Cell Res. Ther. 16, 110. doi:10.1186/s13287-025-04191-y
Zhang, J., Luo, X., Barbieri, D., Barradas, A. M. C., De Bruijn, J. D., Van Blitterswijk, C. A., et al. (2014). The size of surface microstructures as an osteogenic factor in calcium phosphate ceramics. Acta Biomater. 10, 3254–3263. doi:10.1016/j.actbio.2014.03.021
Zhang, S., Jia, Y., Liu, J., Feng, F., Wei, Z., Zhang, M., et al. (2023). A viscoelastic alginate-based hydrogel network coordinated with spermidine for periodontal ligament regeneration. Regen. Biomater. 10, rbad009. doi:10.1093/rb/rbad009
Zhang, W., Du, A., Liu, S., Lv, M., and Chen, S. (2021). Research progress in decellularized extracellular matrix-derived hydrogels. Regen. Ther. 18, 88–96. doi:10.1016/j.reth.2021.04.002
Zhang, X., Han, P., Jaiprakash, A., Wu, C., and Xiao, Y. (2014). A stimulatory effect of Ca3ZrSi2O9 bioceramics on cementogenic/osteogenic differentiation of periodontal ligament cells. J. Mater Chem. B 2, 1415–1423. doi:10.1039/c3tb21663b
Zhang, Y., Chen, Y., Ding, T., Zhang, Y., Yang, D., Zhao, Y., et al. (2023). Janus porous polylactic acid membranes with versatile metal–phenolic interface for biomimetic periodontal bone regeneration. Npj Regen. Med. 8, 28. doi:10.1038/s41536-023-00305-3
Zhao, Y., Li, F., Carvajal, M. T., and Harris, M. T. (2009). Interactions between bovine serum albumin and alginate: an evaluation of alginate as protein carrier. J. Colloid Interface Sci. 332, 345–353. doi:10.1016/j.jcis.2008.12.048
Zhao, P.-P., Ge, Y.-W., Liu, X.-L., Ke, Q.-F., Zhang, J.-W., Zhu, Z.-A., et al. (2020). Ordered arrangement of hydrated GdPO4 nanorods in magnetic chitosan matrix promotes tumor photothermal therapy and bone regeneration against breast cancer bone metastases. Chem. Eng. J. 381, 122694. doi:10.1016/j.cej.2019.122694
Zhao, R., Yang, R., Cooper, P. R., Khurshid, Z., Shavandi, A., and Ratnayake, J. (2021). Bone grafts and substitutes in dentistry: a review of current trends and developments. Molecules 26, 3007. doi:10.3390/molecules26103007
Zhao, B., Chen, Q., Zhao, L., Mao, J., Huang, W., Han, X., et al. (2022). Periodontal ligament stem cell-derived small extracellular vesicles embedded in matrigel enhance bone repair through the adenosine receptor signaling pathway. IJN 17, 519–536. doi:10.2147/IJN.S346755
Zheng, S., Zhang, X., Zhao, Q., Chai, J., and Zhang, Y. (2020). Liquid platelet-rich fibrin promotes the regenerative potential of human periodontal ligament cells. Oral Dis. 26, 1755–1763. doi:10.1111/odi.13501
Zhu, Y.-J., Guo, X.-X., and Sham, T.-K. (2017). Calcium silicate-based drug delivery systems. Expert Opin. Drug Deliv. 14, 215–228. doi:10.1080/17425247.2016.1214566
Zhu, L., Luo, D., and Liu, Y. (2020). Effect of the nano/microscale structure of biomaterial scaffolds on bone regeneration. Int. J. Oral Sci. 12, 6. doi:10.1038/s41368-020-0073-y
Zhu, Y., Wang, W., Chen, Q., Ren, T., Yang, J., Li, G., et al. (2023). Bioprinted PDLSCs with high-concentration GelMA hydrogels exhibit enhanced osteogenic differentiation in vitro and promote bone regeneration in vivo. Clin. Oral Invest. 27, 5153–5170. doi:10.1007/s00784-023-05135-7
Zupančič, Š., Casula, L., Rijavec, T., Lapanje, A., Luštrik, M., Fadda, A. M., et al. (2019). Sustained release of antimicrobials from double-layer nanofiber mats for local treatment of periodontal disease, evaluated using a new micro flow-through apparatus. J. Control Release 316, 223–235. doi:10.1016/j.jconrel.2019.10.008
Keywords: periodontal tissue regeneration, guided tissue regeneration, biomimetic materials, tissue engineering, extracellular matrix
Citation: Zhang Q, Gou C and Zhang Z (2025) Biomimetic materials: a promising strategy for periodontal tissue engineering and regeneration. Front. Bioeng. Biotechnol. 13:1639170. doi: 10.3389/fbioe.2025.1639170
Received: 01 June 2025; Accepted: 06 October 2025;
Published: 25 November 2025.
Edited by:
Xuequan Han, Johns Hopkins University, United StatesReviewed by:
Mahmoud Ebrahimi, University of Maragheh, IranRaj Hazra, North Dakota State University, United States
Copyright © 2025 Zhang, Gou and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhijun Zhang, emhhbmd6aGlqdW5Ac3R1LnNjdS5lZHUuY24=
†These authors have contributed equally to this work and share first authorship
 Qinghua Zhang1,2†
Qinghua Zhang1,2† Zhijun Zhang
Zhijun Zhang