- 1Departments of Oral Biology and Pediatric Dentistry, School of Dental Medicine, University of Pittsburgh, Pittsburgh, PA, United States
- 2Department of Pedodontics, School of Dentistry, Istanbul University, Istanbul, Turkey
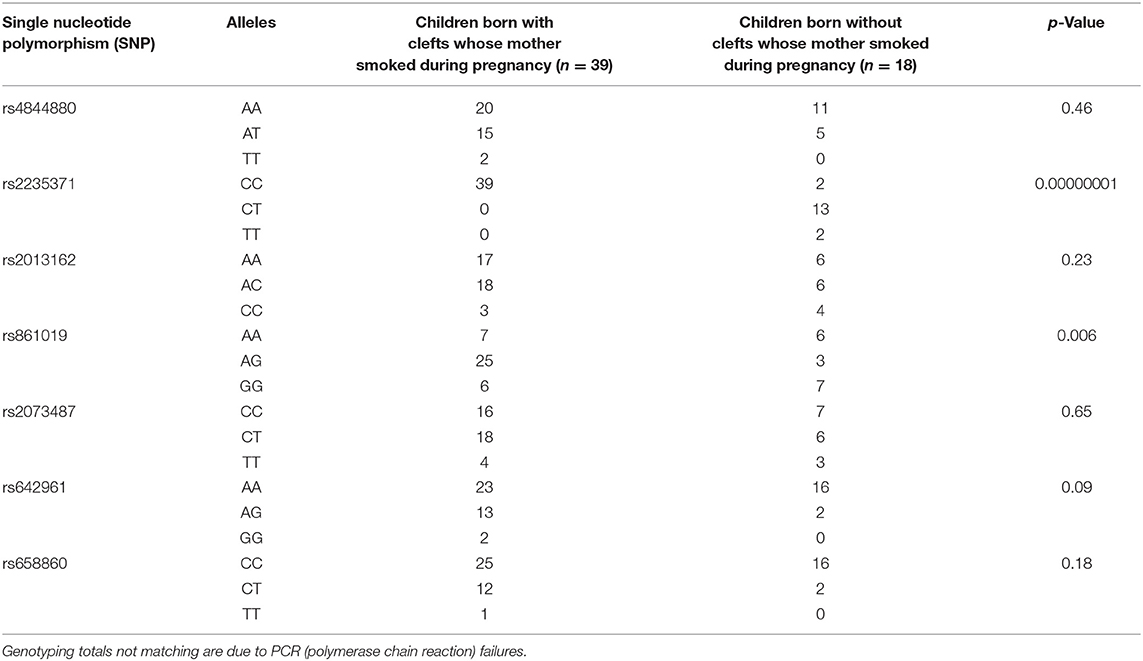
The goal of the present work was to revisit published data to test if genetic variation in interferon regulatory factor 6 (IRF6) is associated with children born with cleft lip with or without cleft palate (CL/P) for cases with positive history of maternal smoking. From the 573 individuals originally studied, this reanalysis focused on 57 who had a positive history of maternal smoking during pregnancy (39 born with CL/P and 18 born without CL/P). Seven IRF6 markers (rs4844880, rs2235371, rs2013162, ra861019, rs2073487, rs642961, and rs658860) were tested for over-transmission of alleles and an alpha of 0.05 was considered statistically significant. All individuals born with CL/P were homozygous for the wild type allele of rs2235371 in comparison to just two individuals born without clefts (p = 0.0000001). For rs861019, individuals born with CL/P were more likely to have the variant allele (p = 0.006). A similar trend was seen for rs642961 (p = 0.09). The results suggest that statistical evidence of over-representation of IRF6 alleles in individuals born with CL/P may be unveiled only when maternal smoking during pregnancy is used as the inclusion criterion in the analysis.
Introduction
Maternal cigarette smoking increases the likelihood of a child being born with cleft lip with or without cleft palate (CL/P) (1), and the population attributable fraction for maternal cigarette smoking was suggested to be between 6.1 and 9.9% (2, 3). A population attributable fraction denotes the fraction of cases that would not have occurred if exposure did not occur, and the association is causal (4). Most cases of CL/P have a multifactorial inheritance, with contributions from more than one gene and possibly from the environment as well (5).
We believe maternal cigarette smoking causing CL/P happens under a multifactorial inheritance framework (5), in which the combination of the presence of certain genetic variation in the individual who smokes leads to clefts. That is why there are many instances of mothers who smoked during pregnancy and who did not have babies with CL/P.
Previous work has tested for potential interactions between maternal cigarette smoking and genetic variants (6, 7), and showed that there are differences depending on if the population is Asian or European in origin (7). Interferon regulatory factor 6 (IRF6), a gene which, when mutated, causes Van der Woude and popliteal pterygium syndromes (8), has been included in these analyses because of evidence that it is associated with the occurrence of CL/P in several populations (9). But in general, the proportion of infants exposed to maternal cigarette smoking during pregnancy included in these analyses are very low (6).
There are examples of previous work testing for an association between IRF6 and CL/P that was designed excluding any cases where there was a history of maternal cigarette smoking during pregnancy (10). This approach eliminates any chance for testing gene-environment interactions but creates more discreet test groups and possibly decreases heterogeneity. However, data like maternal smoking during pregnancy are prone to suffer from recall bias, even if the question is asked at birth. It is well-known smoking has detrimental consequences to one's health, including a developing fetus. Not wanting to be judged, people will hide this information. To improve homogeneity, one can suggest that individuals tested should have been exposed to the same risk factor (11), therefore here we tested for association only children that had maternal cigarette smoking positive history. This approach was aimed at minimizing recall bias and at the same time it increases homogeneity. The aim of the present work was performing secondary analysis on a cohort of families that had a child born with CL/P and showed an association with IRF6 to test if associations could still be detected if only individuals with mothers that smoked during pregnancy were considered.
Methods
We performed secondary analysis of published data (12) and details of the studied cohort have been reported elsewhere. We examined 573 individuals, 158 born with CL/P and 161 non-related individuals with no history of syndromic CL/P. All participants were recruited at the Department of Pedodontics clinics, Istanbul University, Turkey. Individuals born with isolated forms of CL/P were invited to participate in this study between October 2007 and October 2009. We also invited at least one unrelated individual not born with CL/P for each cleft case recruited of the same age and sex during the same period. Individuals born with CL/P had an average age of 10.89 years of age and ranged in age from 3 to 23 years. Their ages were no different from the ages of the unaffected individuals (average age, 10.79 years, ranging from 3 to 23 years; p = 0.84). All study participants were examined in a dental office by the same professional (M.K.). Maternal smoking during pregnancy data were originally collected using a standard questionnaire. It was considered any answer that declared smoking was done during any time of pregnancy. These data matched the information available in the dental records. All participants signed an informed consent document prior to entering into this study. Parents consented for their offspring, and age-appropriate consent was obtained from all children older than 7 years of age. This protocol is approved by both the Istanbul University and University of Pittsburgh Institutional Review Boards. Genomic DNA samples were obtained from saliva. Genotyping of seven IRF6 markers were originally performed by the Taqman method (13), with a 7900 automatic instrument and pre-designed probes (Applied Biosystems, Foster City, CA, USA) and were available for this study. These SNPs (rs4844880, rs2235371, rs2013162, rs861019, rs2073487, rs642961, and rs658860) were originally chosen based on linkage disequilibrium and allele frequency. The corresponding probes are marketed, respectively with the following assay numbers: C__2502442_10, C__15952140_10, C__2500165_10, C__2500178_10, C__2500179_1, C__2238941_20, and C__2500208_10.
Over transmission of alleles was tested using PLINK (14) and at first with an alpha set at 0.05 to detect all possible signals. Then, we applied a most strict criterion to account for multiple testing, lowering alpha to 0.007 (0.05/7).
Results
Thirty-nine children were born with CL/P from mothers who smoked during their pregnancy and they were compared to eighteen children born without CL/P from mothers who smoked during their pregnancy. The rs2235371 variant allele was more likely to be present among children born without clefts whose mothers smoked during pregnancy. On the other hand, children born with clefts whose mothers smoked were more likely to have the variant allele of rs861019 and the same trend could be seen for rs642961 (Table 1).
Discussion
The original report of an association between isolated CL/P and IRF6 (9) particularly highlighted the rs2235371 marker. This marker is a non-synonymous change with a valine being substituted by an isoleucine at position 274. However, since the wild type allele was over-transmitted to the cases born with CL/P, it was concluded that it may not have functional consequences. In the present study, testing for association individuals that were exposed to maternal smoking, the same over-transmission of the wild type allele of rs2235371 was unveiled. It is of interest to mention that the initial analysis of the present study population with all individuals born with CL/P, independent of the mother having smoked during pregnancy or not (12), did show an association between IRF6 rs4844880 and CL/P. For the V274I marker (rs2235371), the association could only be detected when the analysis was adjusted for maternal medication use and smoking during pregnancy. In the present reanalysis, the association could be clearly demonstrated when maternal cigarette smoking was used as the inclusion criterion. It was remarkable that all individuals born with clefts were homozygous for the wild type allele [the originally associated allele in (9)]. The original report (9) also showed association particularly for Filipinos and Vietnamese. The European groups studied did not show statistical significant associations likely due to the lower information content of the V274I change. The frequency of the IRF6 V274I change in the public databases (consulted at www.ncbi.nlm.nih.gov/snp/rs 2235371) is reported to be 39% in Koreans and 38% in Vietnamese, whereas 1% in Estonians and Finns, 3% in Swedes, and 0.5% in Qatarians. A study group from South America (derived from the Latin-American Collaborative Study of Congenital Malformations—ECLAMC) also did not show association between clefts and the IRF6 V274I change originally (9). Only when these data were stratified by mitochondrial DNA haplotypes of Amerindians was that an association was unveiled (15). The V274I change may be therefore protective against clefts and IRF6 and the higher frequency of clefts in Amerindians and Asians in comparison to Europeans may not be related to IRF6, which appears to impact equally all populations (16).
A trend not detected before (12) could be seen for rs642961, with cases born with CL/P having more frequently the variant allele. This marker has better heterozygosity in European groups (17) and was suggested to have functional consequences by disrupting the binding site of transcription factor AP-2alpha.
The role of maternal smoking in clefts may be through hypoxia (5) and we propose here that maternal smoking during pregnancy may also interact with IRF6. IRF6-impaired mice have several key aspects of tissue repair altered, including increased proliferation in the newly formed epidermis and maintenance of myofibroblasts in the granulation tissue (18). It is known that an important aspect of wound healing is oxygen supply and tension in the wound bed (19). IRF6 is expressed in hyperthophic chondrocytes, osteocytes, and bone matrix of craniofacial tissues. When IRF6 is absent, a reduction in the number of lacunae, embedded osteocytes in matrices, and a reduction in mineralization during bone formation can be seen (20). Similarly, hypoxia suppresses hypertrophic differentiation of chondrocytes (21). During facial development, maternal smoking may lead to fluctuations in oxygen supply that are critical at the time the lip and/or palate are having the connective tissue leveled and the middle-edge epithelial apoptosis. This imbalance in the presence of particular IRF6 genetic variation may be the cause in a subset of cases of CL/P. IRF6 rs2235371, rs642961, and/or rs861019 may be useful genomic markers for indicating increased risk of CL/P in pregnancies that are exposed to maternal smoking.
Data Availability Statement
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author/s.
Ethics Statement
The studies involving human participants were reviewed and approved by Istanbul University Ethics Committee. Written informed consent to participate in this study was provided by the participants' legal guardian/next of kin.
Author Contributions
AV provided the concept, designed the study, interpreted and analyzed the data, and wrote the first draft of the manuscript. MK collected the data, obtained the genotypes, and critically revised the manuscript. EB compiled the data and critically revised the manuscript. FS and AM helped to design the study and critically revised the manuscript. All authors contributed to the article and approved the submitted version.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The handling editor declared a past co-authorship with one of the authors AV.
References
1. Xuan Z, Zhongpeng Y, Yanjun G, Jiaqi D, Yuchi Z, Bing S, et al. Maternal active smoking and risk of oral clefts: a meta-analysis. Oral Surg Oral Med Oral Pathol Oral Radiol. (2016) 122:680–90. doi: 10.1016/j.oooo.2016.08.007
2. Honein MA, Devine O, Grosse SD, and Reefhuis J. Prevention of orofacial clefts caused by smoking: implications of the Surgeon General's report. Birth Defects Res A Clin Mol Teratol. (2014) 100:822–5. doi: 10.1002/bdra.23274
3. Sato Y, Yoshioka E, Saijo Y, Miyamoto T, Sengoku K, Azuma H, et al. Population attributable fractions of modifiable risk factors for nonsyndromic orofacial clefts: a prospective cohort study from the Japan Environment and Children's Study. J Epidemiol. (2020). doi: 10.2188/jea.JE20190347. [Epub ahead of print].
4. Greenland S. Applications of stratified analysis methods. In: Rothman KJ, Greenland S, Lash TL, editors. Modern Epidemiology. 3rd ed. Philadelphia, PA: Lippincott Williams & Wilkins (2008). p. 283–302.
5. Vieira AR. Genetic basis of craniofacial deformities and malocclusion, oral clefts, and craniosynostosis. In: Vieira AR, editor. Genetic Basis of Oral Health Conditions. 1st ed. Cham: Springer. (2019). p. 59–72.
6. Wu T, Liang KY, Hetmanski JB, Ruczinski I, Fallin MD, Ingersoll RG, et al. Evidence of gene-environment interaction for the IRF6 gene and maternal multivitamin supplementation in controlling the risk of cleft lip with/without cleft palate. Hum Genet. (2010) 128:401–10. doi: 10.1007/s00439-010-0863-y
7. Beaty TH, Taub MA, Scott AF, Murray JC, Marazita ML, Schwender H, et al. Confirming genes influencing risk to cleft lip with/without cleft palate in a case-parent trio study. Hum Genet. (2013) 132:771–81. doi: 10.1007/s00439-013-1283-6
8. Kondo S, Schutte BC, Richardson RJ, Bjork BC, Knight AS, Watanabe Y, et al. Mutations in IRF6 cause Van der Woude and popliteal pterygium syndromes. Nat Genet. (2002) 32:285–9. doi: 10.1038/ng985
9. Zucchero TM, Cooper ME, Maher BS, Daack-Hirsch S, Nepomuceno B, Ribeiro L, et al. Interferon regulatory factor 6 (IRF6) gene variants and the risk of isolated cleft lip or palate. New Engl J Med. (2004) 351:769–80. doi: 10.1056/NEJMoa032909
10. Jafary F, Nadeali Z, Salehi M, Hosseinzadeh M, Sedghi M, Gholamrezapour T, et al. Significant association between nonsyndromic cleft lip with or without cleft palate and IRF6rs2235371 polymorphism in Iranian familiar population. Mol Biol (Moskow). (2015) 49:848–51. doi: 10.7868/S0026898415060105
11. Henn IW, Alanis LRA, Modesto A, and Vieira AR. The concept of exposure when selecting comparison groups for determining individual susceptibility to addiction to cigarette smoking. PLoS ONE. (2019) 14:e0214946. doi: 10.1371/journal.pone.0214946
12. Yildirim M, Seymen F, Deeley K, Cooper ME, and Vieira AR. Defining predictors of cleft lip and palate risk. J Dent Res. (2012) 91:556–61. doi: 10.1177/0022034512444928
13. Ranade K, Chang MS, Ting CT, Pei D, Hsiao CF, Olivier M, et al. High-throughput genotyping with single nucleotide polymorphisms. Genome Res. (2001) 11:1262–8. doi: 10.1101/gr.157801
14. Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MAR, Bender D, et al. PLINK: a toolset for whole-genome association and population-based linkage analysis. Am J Hum Genet. (2007) 81:559–75. doi: 10.1086/519795
15. Vieira AR, Cooper ME, Marazita ML, Orioli IM, and Castilla EE. Interferon regulatory factor 6 (IRF6) is associated with oral-facial cleft in individuals that originate in South America. Am J Med Genet A. (2007) 143:2075–8. doi: 10.1002/ajmg.a.31884
16. Vieira AR. Genetic and environmental factors in human cleft lip and palate. Front Oral Biol. (2012) 16:19–31. doi: 10.1159/000337521
17. Rahimov F, Marazita ML, Visel A, Cooper ME, Hitchler MJ, Rubini M, et al. Disruption of an AP-2alpha binding site in an IRF6 enhancer is associated with cleft lip. Nat Genet. (2008) 40:1341–7. doi: 10.1038/ng.242
18. Rhea L, Canady FJ, Le M, Reeb T, Canady JW, Kacmarynski DSF, et al. Interferon regulatory factor 6 is required for roper would healing in vivo. Dev Dyn. (2020) 249:509–22. doi: 10.1002/dvdy.134
19. Han G, and Ceillley R. Chronic wound healing: a review of current management and treatments. Adv Ther. (2017) 34:599–610. doi: 10.1007/s12325-017-0478-y
20. Thompson J, Mendoza F, Tan E, Bertol JW, Gaggar AS, Jun G, et al. A cleft lip and palate gene, Irf6, is involved in osteoblast differentiation of craniofacial bone. Dev Dyn. (2019) 248:221–32. doi: 10.1002/dvdy.13
Keywords: cleft lip, cleft palate, smoking, interferon regulatory factors, polymorphis, genetic
Citation: Vieira AR, Koruyucu M, Bekele EK, Seymen F and Modesto A (2021) IRF6 Genetic Variation and Maternal Smoking During Pregnancy in Cleft Lip/Palate. Front. Dent. Med. 2:632055. doi: 10.3389/fdmed.2021.632055
Received: 22 November 2020; Accepted: 09 March 2021;
Published: 31 March 2021.
Edited by:
David John Manton, University Medical Center Groningen, NetherlandsReviewed by:
Yongchu Pan, Nanjing Medical University, ChinaRosa Helena Wanderley Lacerda, Federal University of Paraíba, Brazil
Copyright © 2021 Vieira, Koruyucu, Bekele, Seymen and Modesto. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Alexandre R. Vieira, YXJ2MTFAcGl0dC5lZHU=
 Alexandre R. Vieira
Alexandre R. Vieira Mine Koruyucu
Mine Koruyucu Eyosiyas K. Bekele
Eyosiyas K. Bekele Figen Seymen
Figen Seymen Adriana Modesto
Adriana Modesto