- 1Department of Orthodontics and Pediatric Dentistry, School of Dentistry, University of Maryland, Baltimore, Baltimore, MD, United States
- 2Department of Neurobiology, University of California, Los Angeles, Los Angeles, CA, United States
The objective was to determine whether hyoid bone elevation induced by an anterior mandibular positioning appliance (AMP) predicts the effectiveness of the AMP in patients with obstructive sleep apnea (OSA). Fifteen patients (12 males and 3 females) underwent polysomnographic recordings and lateral cephalograms before and after AMP use of at least 6 months. Measurements of sleep variables and upper airway morphology were compared between pre-AMP and with-AMP states. The AMP appliance reduced apnea-hypopnea indices (AHI) ~53% (33.77 ± 3.29 vs. 15.85 ± 3.78, P = 0.0013). Cephalograms of the oropharyngeal airway showed that the hyoid bone moved superiorly toward the inferior mandibular border (Δ H-MP) ~5 mm (23.4 ± 1.44 vs. 18.27 ± 1.86, P = 0.0377), with the AMP inserted in the oral cavity; no airway measurement other than hyoid bone position changed. No significant correlations emerged between AHI improvement (Δ AHI) and amounts of hyoid elevation (Δ H-MP) when all patients were pooled. However, when the samples were subcategorized, the correlation coefficients increased significantly (P < 0.01) in both subgroups. This outcome suggests the presence of two distinct types among the “Good-Responders” to AMP appliance use. Overall use of the AMP appliance is effective; however, the effectiveness of the appliance appears to depend on the mode of hyoid elevations, likely resulting from muscle responsiveness in patients with AMP use. The results suggest that Δ H-MP measurements may be a useful marker to segregate patients with tongue and hyoid muscles responsive to AMP from those not-so-responsive.
Introduction
Vertical positioning of the hyoid bone with respect to the inferior mandibular border obtained from lateral cephalograms appears to stratify OSA symptom severity provided that images are carefully collected under natural breathing-phase controlled conditions (1–4). Despite recent advances in three-dimensional imaging techniques, a simple measure of hyoid bone positioned low with respect to the mandible is one of the most practical measurements separating OSA patients from asymptomatic controls (1, 5). This vertical measurement can be easily and consistently achieved by two-dimensional lateral head images as far as a natural head position at the end-expiration phase (end of exhalation) while the mandible is guaranteed to be in a resting position (6).
The optimum pressure generated by nasal CPAP (Continuous Positive Airway Pressure) for patients with OSA is determined, theoretically, by the diameter of the passive pharynx (7). In the absence of inspiratory effort, a negative oropharyngeal airway pressure of −15 cm H2O results in a significant pharyngeal narrowing (~47%), and a displacement of the hyoid bone toward the superior-direction by 5.6 ± 1.8 mm even during wakefulness in normal humans (8). The superior displacement of hyoid bone position (or hyoid elevation) indicates a physiological responsive effort against pharyngeal narrowing by the pharyngeal dilating muscles.
Hyoid elevation may result from a coordinated effort of upper airway (UA) dilators, including the genioglossus and hyoid musculature to maintain pharyngeal patency during inspiration (9–11). Pressure titration for a patient with a nasal CPAP appliance is a critical step for successful treatment (12). Likewise, a popular alternative treatment option, use of an anterior mandibular positioner (AMP) and similar intraoral devices also must be titrated to find the most effective position of the device to advance the mandible relative to the maxilla. Various methods to confirm optimal mandible positioning are used (13–16), including supine cephalometry (16), and nasopharyngoscopy (17, 18). Clinical appliance effectiveness is limited to ~66% of patients (19), and in the remaining cases (34%), the appliance is ineffective although the patient has endured a titration process taking up to several months. Moreover, determining the most efficient mandible position during waking based on the position of the velo- and oropharyngeal wall is pointless if the patient is non-responsive to the AMP during sleep. Thus, considering the increasing popularity of oral appliances, treatment outcome prediction is an important issue (20–23).
Maximizing AMP efficiency depends on optimizing the working processes of the device that include (1) widening the oropharyngeal lumen mechanically, and (2) increasing basal tonicity of the UA dilators to stabilize the lumen, i.e., maintaining patency of the pharynx. Although most clinicians emphasize how much mandibular forward advancement is required to open the retroglossal pharynx, oropharyngeal lumen size in the supine position varies, depending on basal activity of the genioglossus and hyoid muscles during waking and sleep (6, 24). However, no clear guidance is available to determine optimal protrusion of the mandible. This aspect is an important concern, because pharyngeal patency in the supine position is subject to actual pharyngeal lumen size rather than EMG signals exerted from pharyngeal dilators maintaining the lumen size. To understand the processes of AMP action, we propose that changes (Before–After) in hyoid bone position responding to AMP insertion would be a measure of responsiveness of the hyoid/tongue muscles, and thus could indicate effectiveness of the AMP. We initially investigated how the extent of hyoid bone elevation in response to AMP insertion indicates the effectiveness of the AMP in the context of basal responsiveness of the dilating muscles. We hypothesized that the level of responsiveness of neural circuitry to the supra- and infrahyoid muscles following AMP insertion determines the extent of superior elevation of the hyoid bone.
Materials and Methods
Study Subjects
We revisited an archived dataset of a cohort (1996–1997) of adult patients who had undergone two sets of overnight polysomnography and lateral cephalometry. Fifteen patients (12 males and 3 females) completed medical, sleep, and physical examinations at a Sleep Disorder Clinic in a university-based teaching hospital. Informed consent was obtained from all individual participants included in the study at the time of study. Some patients from the original cohort were excluded if there was evidence of predominantly central apnea in polysomnographic records, long edentulous span of arches, or temporomandibular joint pain. Also, patients whose apnea were not of an obstructive nature, or who could not wear an AMP appliance, were excluded. Patients who met the criteria, were diagnosed with obstructive sleep apnea (OSA), and who completed the protocol were included. Each of the 15 patients in this cohort who satisfied the inclusion criteria used the same adjustable anterior mandibular positioner (AMP) which was designed to advance the mandible to 75% of the maximum protrusion (25) and to mechanically maintain the mandibular position during sleep. Patients used the AMP for at least 6 months. The research was conducted according to the principles of the Declaration of Helsinki. This study was reportedly approved by the institutional ethics committee, which included patient consent for AMP treatment. Note that ethical appropriateness of using the old data summary was reviewed by the University of Maryland, Baltimore Institutional Review Board that approved this specific research project as non-human subjects research as the analysis was based on an archived dataset and there was no contact with participants for this current study.
Study Design
Observed statistical power was calculated using published mean differences and standard deviations of AHI in the previous report by Ferguson et al. (25). An a priori power analysis was not performed at the initial stage of the current study which was a part of a prior clinical trial published previously (26). Besides, the current analysis here is considered an “interim analysis,” defined as an analysis of data that were obtained before data collection has been completed. This conceptual approach is commonly used when a treatment is uncertain whether the outcome is beneficial or harmful compared to the concurrent treatment method (for instance, nasal CPAP) while the study is on-going. We used a sample size of 15 as if it were the initial endpoint for this retrospective interim analysis on this archived dataset. The current study began as an early clinical trial. The first diagnostic polysomnography (PSG) study was to confirm the basal apnea-hypopnea index (AHI). After the first diagnostic PSG, a lateral cephalometric x-ray was taken. The patients then began using their AMP devices nightly. Patients returned to the sleep laboratory for the second PSG with the AMP appliance after a minimum of 6 months, and the second set of PSG and lateral cephalograms was collected in the same manner with the AMP positioned in the mouth. Both PSG studies included an electroencephalogram (EEG, C3/A2, C4/A1, O2/A1), electrooculogram (EOG), submental electromyogram (EMG), anterior tibialis EMG, electrocardiogram (ECG), chest-abdominal motion, oronasal airflow for expired CO2, and arterial oxygen saturation with pulse oximetry using an earlobe sensor, as previously described (25, 26).
Polysomnography
Standard nocturnal hospital-based PSGs were performed. The PSG results were scored manually, and the total AHI was calculated for each night. Obstructive apnea was defined as no-airflow sensed at the nostril for at least 10 s accompanied by respiratory effort. Hypopneas were defined as a reduction in airflow of at least 50% for at least 10 s, accompanied by a reduction in respiratory effort or by an arterial oxygen desaturation of at least 3%. We used the AHI as the outcome variable for evaluation of efficacy of AMPs. AHI were manually scored by a sleep technician and were finalized by a sleep physician.
Lateral Cephalometry
Upright lateral cephalometry was performed before AMP treatment and immediately after the second overnight sleep study with use of the AMP. During this second x-ray, the AMP appliance was positioned in the mouth. Cephalograms were taken at the end of exhalation to standardize tongue posture and upper airway size with the head at a natural position in accordance with the protocol. Briefly, patients were asked to find their natural head posture first by nodding their head back and forth 3–4 times, gradually reducing the magnitude of the nodding, and stopping at their most comfortable and natural head position while gazing at an imaginary horizon (27). The ear-rod positioners were then adjusted to restrain the head in the machine without disturbing the natural head position. The operator asked the patient to swallow saliva two times and relax the jaw (28). Finally, the operator guided paced breathing for several breaths in accordance with the patient's natural effort, and captured the cephalogram at the end of the third exhalation. Cephalometric tracing and analyses were performed by one trained investigator.
Statistical Analysis
Data were analyzed and graphed using the statistical package GraphPad PRIZM 7 (GraphPad Software, La Jolla, CA) and SPSS v.23 (IBM). Paired and unpaired t-tests were used to compare clinical sleep and cephalometric variables before and after AMP treatment. Two-tailed p-values were calculated, based on t scores and degree of freedom values for inference tests. Selected estimations are presented as mean ± standard error.
Results
Study Samples
The initial analysis on 15 patients was completed with a pair of overnight sleep studies and cephalograms without (Pre-AMP) and with AMP (With-AMP) appliances. Calculated post-hoc achieved power (using GPower 3.0.10) (29) on the mean differences and standard deviations indicated that the sample size of 15 was acceptable, with a power (1–β) of 0.996 (30).
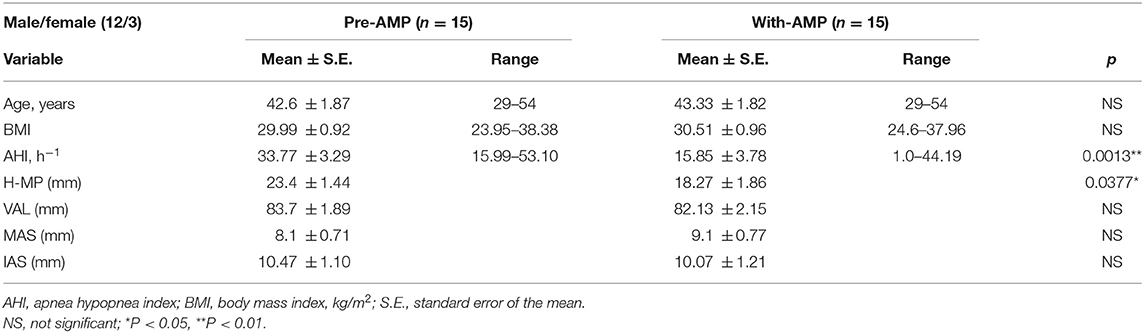
Patient characteristics at baseline and after AMP treatment for at least 6 months are presented in Table 1. There were no significant differences between baseline and after-treatment mean values of Age and BMI variables. The mean mandibular advancement with AMP was 6.05 mm forward and 10.17 mm downward from centric occlusion as each patient protruded the mandible to 75% of their maximum (Figure 1). The average ArcTan value for the degree of mandible-opening measured at the incisal edge was 60.89 ± 2.29 degrees.
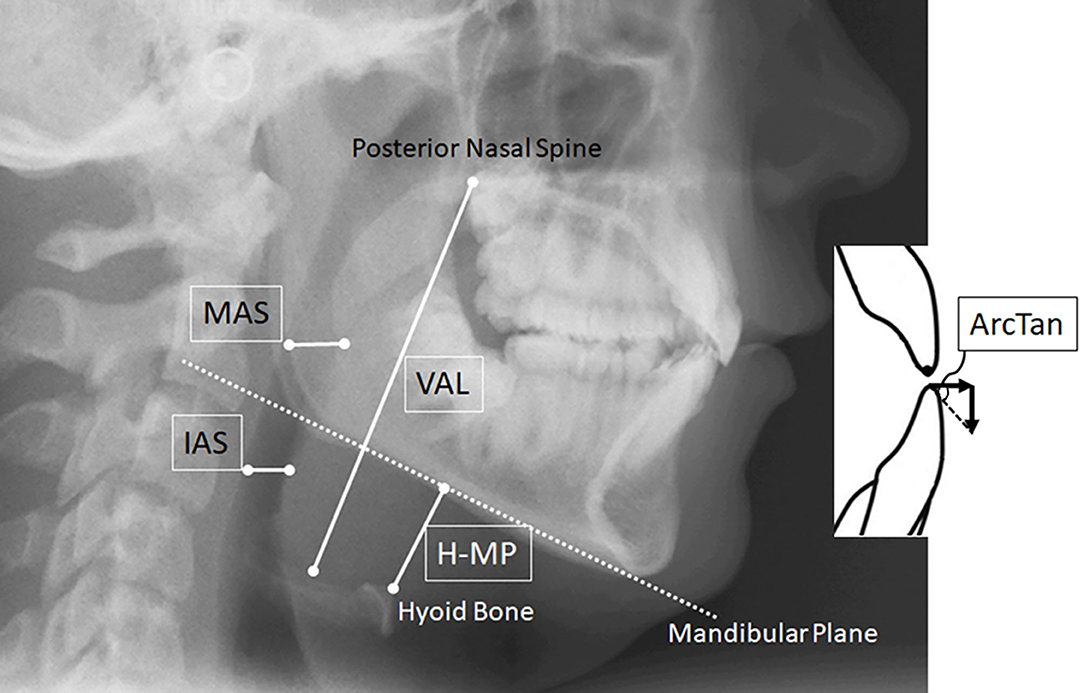
Figure 1. Cephalometric measurements of the pharyngeal airway. Lateral head films of participating patients were taken in the morning after overnight polysomnography at the hospital. VAL (Vertical Airway Length between the base of the epiglottis to posterior nasal spine), MAS (Middle Airway Space between the tongue dorsal surface and the posterior pharyngeal wall measured parallel to the horizontal floor at the tip of the soft palate), and IAS (Inferior Airway Space between the tongue dorsal surface and the posterior pharyngeal wall measured parallel to MAS at the narrowest area). ArcTan values were calculated using the amount of forward mandible advancement (75% of the maximum protrusion on average) seen by superimposing the two lateral cephalograms taken Pre- and With-AMP.
Cephalometric Analysis
Changes in oropharyngeal airway dimensions after AMP use in the oral cavity measured on cephalograms (Figure 1) were tabulated (Table 1). The second set of cephalograms was taken with the appliance in the mouth after the patient had used the appliance for at least 6 months. Among the measurements of pharyngeal airway reported previously, only H-MP, the linear distance between the hyoid bone to the mandibular plane on cephalograms, significantly decreased. The hyoid bone moved ~5 mm superiorly with the AMP in the mouth. Reproducibility on cephalometric measurements performed by an orthodontist was significantly high, as reported previously (6). The correlation coefficient (r) on H-MP measurements was 0.99, indicating the reliability of the procedure.
Amongst several polysomnographic outcome variables, we used AHI measurements for analysis of the relationship with hyoid bone position and symptom severity. AHI values decreased significantly in most patients from a mean of 33.77 to 15.85 after AMP use (Figure 2A). However, in three of the 15 patients, AHI increased or remained the same with the AMP appliance during the second PSG (Figure 2B). Among these three patients, one increased BMI by 4.5, and AHI increased to 44 from an initial AHI of 16 (Figure 2B). AHIs improved with AMP use as the H-MP distance decreased (i.e., the hyoid bone elevated ~5 mm on average) (Figure 2C). Further examination of H-MP measurements identified two patients who had improved their AHI by 11.74 and 11.3 but did not change their hyoid positions (Figure 2D, shaded box). Of the three patients with increased distance between the hyoid bone and the mandibular plane after AMP insertion, one patient showed an AHI increased by 2.02 (Figure 2D, bottom of shaded box). None of these three patients showed a significant change in BMI.
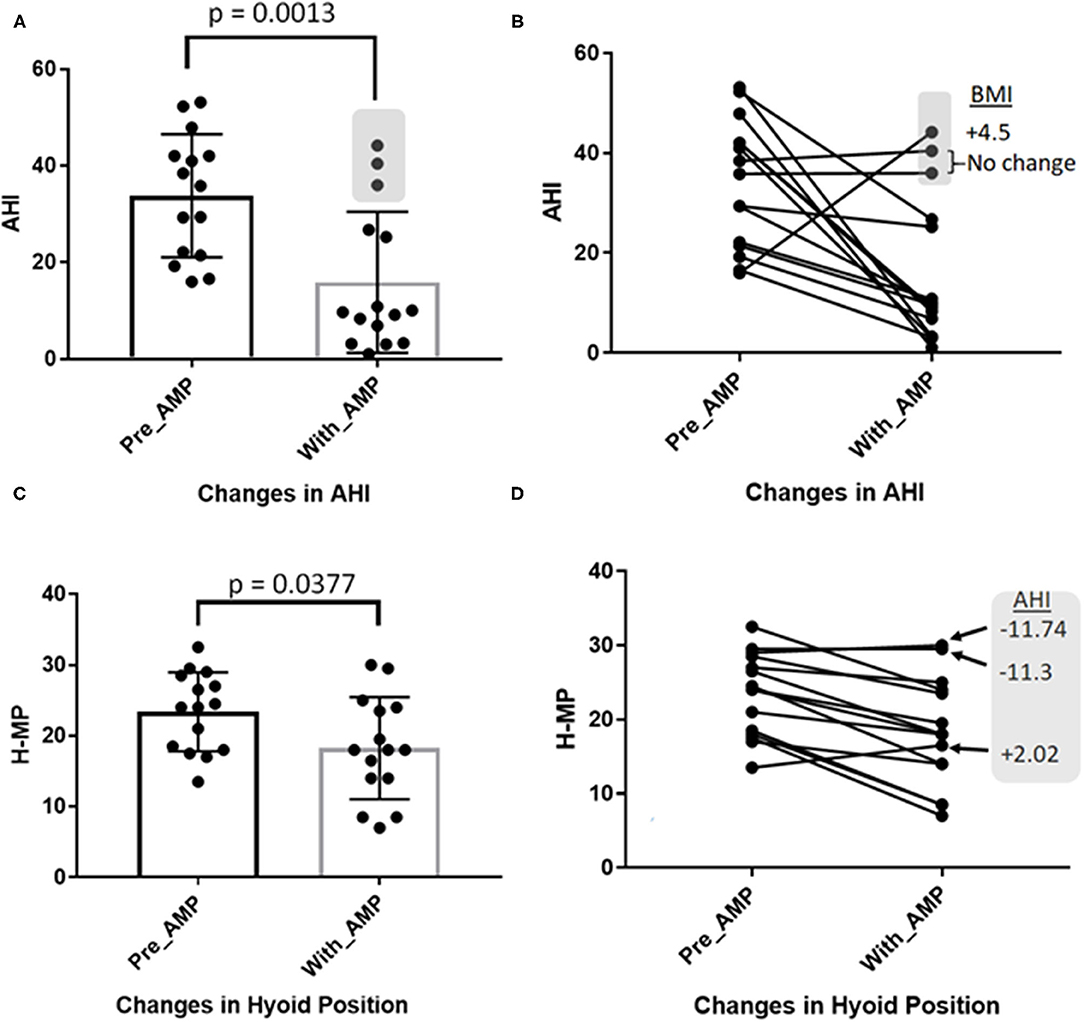
Figure 2. Comparisons of AHI and H-MP measurements between Baseline (Pre-AMP) and after 6 months of AMP use (With-AMP). Each black dot represents one patient. (A) AHI values decreased significantly from a mean of 33.77 to 15.85 (p = 0.0013) after AMP use. (B) AHI values in 3 of 15 patients (gray shading in A and B) increased or remained unchanged. Among these three, one increased BMI by 4.5 (see B) as AHI increased. (C) AHIs improved, with the H-MP distance decreasing from a mean of 23.4 to 18.27 mm (i.e., the hyoid bone elevated ~5 mm with AMP). (D) Two patients with moderately decreased AHI of 11.74 and 11.3 (indicated by arrows in gray shaded box) did not significantly alter their hyoid positions. Of the three patients with increased distances of H-MP after AMP insertion, one patient showed an AHI increase of 2.02 (gray box). BMI values for these three patients were not significantly altered after AMP use.
To visualize potential relationships between the changes in AHI and changes in hyoid bone position after AMP delivery, we calculated “differences” in AHI and H-MP measurements before- and after-appliance use. Δ AHI (WithAMP AHI value subtracted from PreAMP AHI value) and Δ H-MP (WithAMP H-MP values subtracted from PreAMP H-MP values) were calculated, and those values plotted for each patient (Figure 3). A positive value (increased) Δ AHI indicates an AHI improvement. A positive change (increased) Δ H-MP indicates an elevation of the hyoid bone toward the inferior mandible border. Unexpectedly, no significant correlation (r = 0.25, P = 0.37) was found between Δ AHI and Δ H-MP when all patients were included. However, as apparent in the scatter plot, two subgroups emerged among the 15 patients (see Figure 3A). To confirm this segregation statistically, a discriminant analysis was performed on Δ AHI and Δ H-MP measurements. The results showed a significantly high canonical correlation (i.e., correlation between the discriminant scores) of 0.97 (Wilks' Lambda 0.067, Chi-square 29.80, P = 0.000). When means and standard errors were calculated for Δ AHI and Δ H-MP in the two subgroups, Group A showed 24.43 ± 7.39 for Δ AHI and 1.5 ± 1.11 mm for Δ H-MP; Group B showed 16.59 ± 4.89 for Δ AHI and 9.21 ± 0.58 mm for Δ H-MP (Figure 3A). When unpaired t-tests were performed between Group A and B on Δ AHI and Δ H-MP measurements, Δ H-MP measurements showed a significant difference at p < 0.0001 (t score = 6.0, with 12 degrees of freedom). Pearson's correlations between Δ H-MP and Δ AHI measurements in each subgroup were significantly high; i.e., r = 0.84 (P = 0.009) Group A was characterized by small changes (1.5 mm) in hyoid bone position, whereas Group B was characterized by large hyoid elevations, r = 0.97 (P = 0.0003) (shaded box), including some subjects whose AHI deteriorated significantly. Group B demonstrated significantly more elevation (9.21 mm) in hyoid position with AMP use.
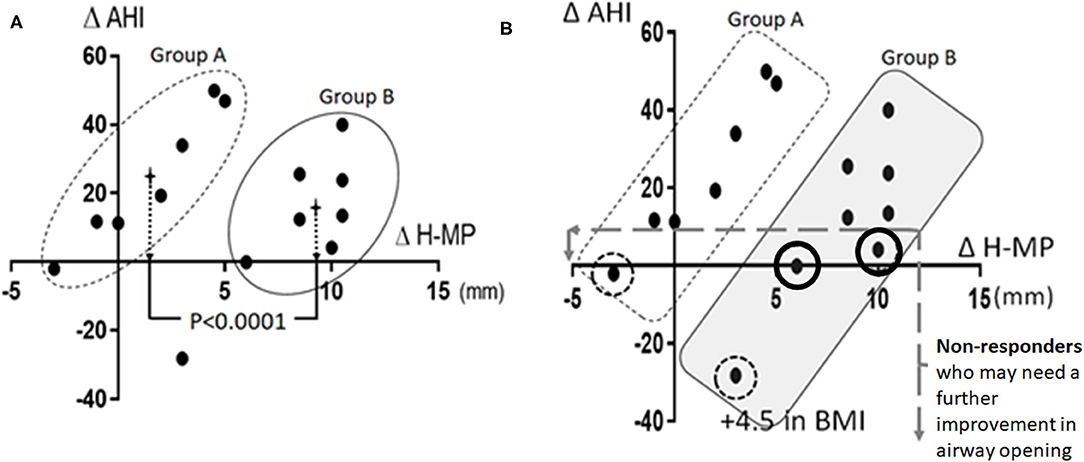
Figure 3. Scatter plots created using two sets of measurements Δ AHI (Pre-AMP—With-AMP) and Δ H-MP (Pre-AMP—With-AMP). A positive value (or increased amount) of Δ AHI indicates an improvement in AHI. A positive value (or increased amount) of Δ H-MP indicates an elevation of the hyoid bone toward the inferior mandible border. (A) 14 subjects are divided into two subgroups, Group A and B as confirmed by the discriminant analysis (SPSS v.23, IBM) (one outlier is excluded). The crosses in the middle of the elliptical circles indicate the mean values of hyoid changes in each group. (B) Four non-responders to AMP use were identified (circled dots). These patients showed Δ AHI lower than 10. In two of these patients, the decrease in AHI after AMP produced a negative Δ AHI value (dotted circles). One of them showed an increased BMI (+4.5).
Discussion
The findings propose the presence of two subgroups among the “Good-Responders” to AMP appliance use. One patient group showed significantly greater hyoid elevation with AMP use than the other group of patients. The outcomes suggest that the extent of hyoid position changes resulting from AMP use in the two subgroups of OSA patients reflects differences in responsiveness of the hyoid muscles to the AMP appliance when the AMP is positioned to achieve 75% of maximum protrusion. This may be a critical view in managing the pathophysiology of OSA.
This clinical report was ramified from the outcomes of a previously-performed randomized crossover study published by Ferguson et al. (25). We found that the AMP oral appliances are effective (defined as >50% reduction in AHI), and in some cases, very effective. For instance, the AHI of one 36-year-old male patient improved markedly from an AHI of 53.1–3.3 after a year of AMP use. This improvement was assisted by a minimal reduction in BMI although the amount of horizontal forward mandibular movement with AMP was only 4 mm. On the other hand, however, in another patient, the appliance enforced an 11 mm protrusion of the mandible, but resulted in no improvement of the AHI (which went from 38.41 to 40.43). This heterogeneity suggests that: (1) the uniformly applied mandible protrusion, i.e., 75% of the maximum protrusion, may not be optimal; (2) there may be two distinct subgroups among our patients distinguished by the extent of repositioning of the hyoid bone after AMP use. Group B showed an extensive repositioning (~9 mm elevation on average) after AMP use for 6 months or longer, while Group A showed minimal vertical changes in hyoid bone position with AMPs majority of which are effective.
Patients with OSA are heterogeneous (31). A previous step-wise cluster analysis performed on 84 OSA subjects recognized two patient subtypes from a pathophysiology perspective; one subtype with skeletal mismatch and another subtype with atypical soft tissue structures (32). Recently, Eckert et al. (33) categorized the etiopathophysiology of OSA patients with respect to four phenotypic traits: collapsibility of the pharynx, muscle responsiveness, arousal threshold, and magnitude of loop gain (31, 33, 34). Of these four traits, hyoid position changes (Δ H-MP) should reflect “UA collapsibility” and “muscle responsiveness” as described by Eckert et al. and others (11).
A recent clinical study reported that hyoid position (H-MP) relative to the mandibular plane linearly correlated with Pcrit (passive critical closing pressure of a pharynx) at r = 0.527, p = 0.001 (35), although the putative one-to-one relationship between Pcrit values and OSA severity remains controversial (36). In fact, Pcrit and pharyngeal dilator muscle activity (which is equivalent to Eckert et al.'s “UA collapsibility” and “muscle responsiveness”) are sleep-stage dependent (37). Predictability of H-MP for OSA severity are high (1, 2) and is greater than those of minimal posterior airway space (PAS) measurements as has been previously reported (38). As reported by Wheatley et al. (8) and this study, the average amount the hyoid bone elevation by AMP use is ~5 mm. Nevertheless, when all samples were included, the correlation between Δ H-MP and Δ AHI was low (Pearson correlation, 0.53). This lack of significant correlation was reported in a similar study by Genta et al. (35), suggesting potential heterogeneity in hyoid positioning with respect to OSA severity.
Unlike other studies, we analyzed the mean difference (Δ H-MP) in hyoid bone position in the presence or absence of AMP use rather than using two sets of measurements. In so doing, we assumed that we could interpret “functional lengths” of the suprahyoid muscles influencing hyoid position with respect to the pharyngeal patency after 6 months of AMP use. In Group B, patients exhibited “good UA muscle responsiveness” to OSA severity. In contrast, Group A patients showed less reactive upper airway muscles with respect to OSA severity; hence, the hyoid bone position showed minimal changes (1.5 mm on average in Group A) in response to AMP use. When we compared morphology, airway size, or anthropometric variables between groups A and B, no differences emerged. Thus, we postulate that the difference in hyoid bone position changes (Δ H-MP) between groups A and B are perhaps due to a resting muscle tone with the appliance in the oral cavity. Both subgroups include good responders and poor responders to AMP therapy. Therefore, it remains inconclusive why a few patients did not respond to the AMP. However, we postulate that the non-responder belonging to Group B, who showed a significant BMI increase, could improve AHI markedly if the AMP were further advanced or if “muscle factor” were improved.
The hyoid bone moves superiorly in the supine position in asymptomatic subjects, while the tongue mass sinks by 30% due to gravitational pull, as the oropharyngeal space is reduced by 30% (6, 38). In the supine body position, tongue and suprahyoid electromyogram (EMG) activity (principally the genioglossus) increases by 30%. We previously reported that tongue muscle activity increases if the oropharyngeal space is partially blocked by a miniature balloon in normal humans (39). This outcome may underlie why diurnal UA muscle activity remains high in OSA patients (9, 40). Our measurements were carried out in the awake state on two separate occasions. However, the improvements of AHI (Δ AHI) are clearly proportional to ΔH-MP (i.e., “a hypothetical responsiveness of hyoid muscles” to the appliance in both subgroups). Although one could argue that the hyoid bone position changes cannot reflect AHI measured during sleep, our ΔH-MP measurements during the waking state are useful for determining the hypothetical UA muscle responsiveness. The extent of change in hyoid position with AMP appears to reflect tongue muscle responsiveness in patients. Thus, the non-responders would need to secure a more “adequate pharyngeal airway diameter” beyond the size obtained by 75% of the maximum mandible protrusion.
In contrast, those Active-Hyoid non-Responders who showed little or no improvement in AHI despite significant hyoid changes could have “fatigable UA muscles” due to compensatory overwork of the oropharyngeal muscles during wakefulness (41, 42). In such cases, the UA muscles could become more fatigued during sleep. Our previous studies in rodents demonstrated that intermittent hypoxic exposure converts hyoid muscles into more-fatigable composition types, with increased numbers of fast-twitch fibers containing myosin heavy chains IIb (43). This group of OSA patients probably needs nasal CPAP therapy along with weight control to improve fatigability of tongue and hyoid muscles rather than therapy aimed at opening the airway further by additional protrusion of the mandible. Moreover, these patients could be exposed to risk while traveling with a handy AMP appliance, particularly at high altitude, while relying on ineffective airway dilator muscle function, since these OSA patients tend to have fatigued UA muscles consisting of more-fatigable muscle fibers (41).
The outcomes suggest that an appropriate next step would be a prospective patient-based clinical study in which patients who choose to use AMP appliance would obtain a second set of cephalograms and sleep studies with a titratable appliance during the late adjustment period. Comparisons between the Pre-AMP dataset and this second set could offer insights onto whether an AMP appliance is appropriate for the patient and how much advancement of the mandible is optimal.
Conclusions
AMPs for mild-to-moderate OSA patients are sufficiently effective for reducing AHI as shown in this report and others. Thus, AMPs could replace the current non-surgical treatment of choice (nasal CPAP) without the discomfort and difficulty associated with positive pressure intervention. We show that AMP effectiveness could be markedly improved if outcomes could be predicted based on the patient's “hyoid responsiveness,” i.e., hyoid bone elevation with AMP. AMPs appear to be more effective for a specific type of patients, and less so for other types. AMPs may (1) enlarge the oropharyngeal space, and, (2) improve responsivity of hyoid muscles.
Limitations of the Study
We could not report a calibration effort on AHI scoring between the sleep technician and the sleep physician; thus, readers must reference the range values reported in the Table 1 rather than using the mean values, if needed. However, our cephalometric measurements and PSG measurements were mutually blinded between the investigators who worked on cephalometric images and the investigators produced PSG data.
We used a “retrospective interim analysis scheme” because the data collection process was halted unexpectedly. Although post-hoc sample size computation is not encouraged conventionally, sample size re-estimation could provide an opportunity to re-evaluate uncertainties using archived data, and to continue the trial with an updated sample size when necessary. Our post-hoc re-estimation of power was sufficiently high as reported.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics Statement
Ethical review and approval was not required for this study with human participants, in accordance with the local legislation and institutional requirements.
Author Contributions
E-KP supervised and performed entire processes involved in the study including data collection, study design, statistical analysis, and manuscript writing. RH contributed on study design as well as initial and revised manuscripts. Both authors contributed to the article and approved the submitted version.
Funding
Publication of this article is partly supported by American Association of Orthodontists Foundation (AAOF) grant to E-KP.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors thank Dr. K Ferguson who provided patients from her faculty practice and clinical trials. We are also indebted to Dr. Ferguson's previous publications.
References
1. Neelapu BC, Kharbanda OP, Sardana HK, Balachandran R, Sardana V, Kapoor P, et al. Craniofacial and upper airway morphology in adult obstructive sleep apnea patients: a systematic review and meta-analysis of cephalometric studies. Sleep Med Rev. (2017) 31:79–90. doi: 10.1016/j.smrv.2016.01.007
2. Bilici S, Yigit O, Celebi OO, Yasak AG, Yardimci AH. Relations between hyoid-related cephalometric measurements and severity of obstructive sleep apnea. J Craniofac Surg. (2018) 29:1276–81. doi: 10.1097/SCS.0000000000004483
3. Kohno A, Kitamura Y, Kato S, Imai H, Masuda Y, Sato Y, et al. Displacement of the hyoid bone by muscle paralysis and lung volume increase: the effects of obesity and obstructive sleep apnea. Sleep. (2019) 42:zsy198. doi: 10.1093/sleep/zsy198
4. Gulati A, Chate RA, Howes TQ. Can a single cephalometric measurement predict obstructive sleep apnea severity? J Clin Sleep Med. (2010) 6:64–8. doi: 10.5664/jcsm.27712
5. Armalaite J, Lopatiene K. Lateral teleradiography of the head as a diagnostic tool used to predict obstructive sleep apnea. Dentomaxillofac Radiol. (2016) 45:20150085. doi: 10.1259/dmfr.20150085
6. Pae EK, Lowe AA, Sasaki K, Price C, Tsuchiya M, Fleetham JA. A cephalometric and electromyographic study of upper airway structures in the upright and supine positions. Am J Orthod Dentofacial Orthop. (1994) 106:52–9. doi: 10.1016/S0889-5406(94)70021-4
7. Isono S. Obesity and obstructive sleep apnoea: mechanisms for increased collapsibility of the passive pharyngeal airway. Respirology. (2012) 17:32–42. doi: 10.1111/j.1440-1843.2011.02093.x
8. Wheatley JR, Kelly WT, Tully A, Engel LA. Pressure-diameter relationships of the upper airway in awake supine subjects. J Appl Physiol. (1985). (1991) 70:2242–51. doi: 10.1152/jappl.1991.70.5.2242
9. Kwan BCH, McBain RA, Luu BL, Butler JE, Bilston LE, Gandevia SC. Influence of respiratory mechanics and drive on genioglossus movement under ultrasound imaging. PLoS ONE. (2018) 13:e0195884. doi: 10.1371/journal.pone.0195884
10. Cheng S, Brown EC, Hatt A, Butler JE, Gandevia SC, Bilston LE. Healthy humans with a narrow upper airway maintain patency during quiet breathing by dilating the airway during inspiration. J Physiol. (2014) 592:4763–74. doi: 10.1113/jphysiol.2014.279240
11. Sands SA, Eckert DJ, Jordan AS, Edwards BA, Owens RL, Butler JP, et al. Enhanced upper-airway muscle responsiveness is a distinct feature of overweight/obese individuals without sleep apnea. Am J Respir Crit Care Med. (2014) 190:930–7. doi: 10.1164/rccm.201404-0783OC
12. Inoue A, Chiba S, Matsuura K, Osafune H, Capasso R, Wada K. Nasal function and CPAP compliance. Auris Nasus Larynx. (2019) 46:548–58. doi: 10.1016/j.anl.2018.11.006
13. Okuno K, Ikai K, Matsumura-Ai E, Araie T. Predictors of side effects with long-term oral appliance therapy for obstructive sleep apnea. Clin Sleep Med. (2018) 14:119–25. doi: 10.5664/jcsm.6896
14. Cistulli PA, Gotsopoulos H. Single-night titration of oral appliance therapy for obstructive sleep apnea: a step forward? Am J Respir Crit Care Med. (2004) 170:353–4. doi: 10.1164/rccm.2405012
15. Tsai WH, Vazquez JC, Oshima T, Dort L, Roycroft B, Lowe AA, et al. Remotely controlled mandibular positioner predicts efficacy of oral appliances in sleep apnea. Am J Respir Crit Care Med. (2004) 170:366–70. doi: 10.1164/rccm.200310-1446OC
16. Tsuiki S, Almeida FR, Lowe AA, Su J, Fleetham JA. The interaction between changes in upright mandibular position and supine airway size in patients with obstructive sleep apnea. Am J Orthod Dentofacial Orthop. (2005) 128:504–12. doi: 10.1016/j.ajodo.2004.03.040
17. Chan AS, Lee RW, Srinivasan VK, Darendeliler MA, Grunstein RR, Cistulli PA. Nasopharyngoscopic evaluation of oral appliance therapy for obstructive sleep apnoea. Eur Respir J. (2010) 35:836–42. doi: 10.1183/09031936.00077409
18. Sutherland K, Chan ASL, Ngiam J, Darendeliler MA, Cistulli PA. Qualitative assessment of awake nasopharyngoscopy for prediction of oral appliance treatment response in obstructive sleep apnoea. Sleep Breath. (2018) 22:1029–36. doi: 10.1007/s11325-018-1624-8
19. Marco Pitarch R, Selva García M, Puertas Cuesta J, Marco Algarra J, Fernández Julian E, Fons Font A. Effectiveness of a mandibular advancement device in obstructive sleep apnea patients: a prospective clinical trial. Eur Arch Otorhinolaryngol. (2018) 275:1903–11. doi: 10.1007/s00405-018-4978-4
20. Phillips CL, Grunstein RR, Darendeliler MA, Mihailidou AS, Srinivasan VK, Yee BJ, et al. Health outcomes of continuous positive airway pressure versus oral appliance treatment for obstructive sleep apnea: a randomized controlled trial. Am J Respir Crit Care Med. (2013) 187:879–87. doi: 10.1164/rccm.201212-2223OC
21. Sutherland K, Kairaitis K, Yee BJ, Cistulli PA. From CPAP to tailored therapy for obstructive sleep Apnoea. Multidiscip Respir Med. (2018) 13:44. doi: 10.1186/s40248-018-0157-0
22. Sakamoto Y, Yanamoto S, Rokutanda S, Naruse T, Imayama N, Hashimoto M, et al. Predictors of obstructive sleep apnoea-hypopnea severity and oral appliance therapy efficacy by using lateral cephalometric analysis. Oral Rehabil. (2016) 43:649–55. doi: 10.1111/joor.12408
23. Remmers JE, Topor Z, Grosse J, Vranjes N, Mosca EV, Brant R, et al. A feedback-controlled mandibular positioner identifies individuals with sleep apnea who will respond to oral appliance therapy. J Clin Sleep Med. (2017) 13:871–80. doi: 10.5664/jcsm.6656
24. Pae EK, Blasius JJ, Nanda R. Heterogeneity in vertical positioning of the hyoid bone in relation to genioglossal activity in men. Angle Orthod. (2004) 74:343–8. doi: 10.1043/0003-3219(2004)074<0343:HIVPOT>2.0.CO;2
25. Ferguson KA, Ono T, Lowe AA, al-Majed S, Love LL, Fleetham JA. A short-term controlled trial of an adjustable oral appliance for the treatment of mild to moderate obstructive sleep apnoea. Thorax. (1997) 52:362–8. doi: 10.1136/thx.52.4.362
26. Pae EK, Ferguson KA. Cephalometric characteristics of nonobese patients with severe OSA. Angle Orthod. (1999) 69:408–12.
27. Sökücü O, Okşayan R, Uyar M, Ademci KE, and Üşümez S. Relationship between head posture and the severity of obstructive sleep apnea. Am J Orthod Dentofacial Orthop. (2016) 150:945–9. doi: 10.1016/j.ajodo.2016.05.011
28. Tingey EM, Buschang PH, Throckmorton GS. Mandibular rest position: a reliable position influenced by head support and body posture. Am J Orthod Dentofacial Orthop. (2001) 120:614–22. doi: 10.1067/mod.2001.119802
29. Faul F, Erdfelder E, Lang AG, Buchner A. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods. (2007) 39:175–91. doi: 10.3758/BF03193146
30. Srinivas TR, Ho B, Kang J, Kaplan B. Post hoc analyses: after the facts. Transplantation. (2015) 99:17–20. doi: 10.1097/TP.0000000000000581
31. Carberry JC, Amatoury J, Eckert DJ. Personalized management approach for OSA. Chest. (2018) 153:744–55. doi: 10.1016/j.chest.2017.06.011
32. Tsuchiya M, Lowe AA, Pae EK, Fleetham JA. Obstructive sleep apnea subtypes by cluster analysis. Am J Orthod Dentofacial Orthop. (1992) 101:533–42. doi: 10.1016/0889-5406(92)70128-W
33. Eckert DJ. Phenotypic approaches to obstructive sleep apnoea - new pathways for targeted therapy. Sleep Med Rev. (2018) 37:45–59. doi: 10.1016/j.smrv.2016.12.003
34. Owens RL, Edwards BA, Eckert DJ, Jordan AS, Sands SA, Malhotra A, et al. An integrative model of physiological traits can be used to predict obstructive sleep apnea and response to non positive airway pressure therapy. Sleep. (2015) 38:961–70. doi: 10.5665/sleep.4750
35. Genta PR, Schorr F, Eckert DJ, Gebrim E, Kayamori F, Moriya HT, et al. Upper airway collapsibility is associated with obesity and hyoid position. Sleep. (2014) 37:1673–8. doi: 10.5665/sleep.4078
36. Azarbarzin A, Sands SA, Taranto-Montemurro L, Oliveira Marques MD, Genta PR, Edwards BA, et al. Estimation of pharyngeal collapsibility during sleep by peak inspiratory airflow. Sleep. (2017) 40:zsw005. doi: 10.1093/sleep/zsw005
37. Carberry JC, Jordan AS, White DP, Wellman A, Eckert DJ. Upper airway collapsibility (Pcrit) and pharyngeal dilator muscle activity are sleep stage dependent. Sleep. (2016) 39:511–21. doi: 10.5665/sleep.5516
38. Hsu WE, Wu TY. Comparison of upper airway measurement by lateral cephalogram in upright position and CBCT in supine position. J Dent Sci. (2019) 4:185–91. doi: 10.1016/j.jds.2019.01.007
39. Pae EK, Blasius JJ, Nanda R. Sex differences in genioglossus muscle response to changes in pharyngeal resistance. Am J Orthod Dentofacial Orthop. (2002) 122:500–5. doi: 10.1067/mod.2002.128862
40. Douglas NJ, Jan MA, Yildirim N, Warren PM, Drummond GB. Effect of posture and breathing route on genioglossal electromyogram activity in normal subjects and in patients with the sleep apnea/hypopnea syndrome. Am Rev Respir Dis. (1993) 148:1341–5. doi: 10.1164/ajrccm/148.5.1341
41. Carrera M, Barbé F, Sauleda J, Tomás M, Gómez C, Agustí AG. Patients with obstructive sleep apnea exhibit genioglossus dysfunction that is normalized after treatment with continuous positive airway pressure. Am J Respir Crit Care Med. (1999) 159:1960–6. doi: 10.1164/ajrccm.159.6.9809052
42. Cori JM, O'Donoghue FJ, Jordan AS. Sleeping tongue: current perspectives of genioglossus control in healthy individuals and patients with obstructive sleep apnea. Nat Sci Sleep. (2018) 10:169–79. doi: 10.2147/NSS.S143296
Keywords: cephalometric analysis, hyoid bone, obstructive sleep apnea, oral appliance, upper airway
Citation: Pae E-K and Harper RM (2021) Elevated Hyoid Bone Position in Response to Mandibular Advancing Appliance Predicts Effectiveness of the Appliance for Obstructive Sleep Apnea. Front. Dent. Med. 2:672936. doi: 10.3389/fdmed.2021.672936
Received: 26 February 2021; Accepted: 08 June 2021;
Published: 29 June 2021.
Edited by:
Francisco Nociti, State University of Campinas, BrazilReviewed by:
Maria Carolina Martins Mussi, Federal University of Western Bahia, BrazilSivaraman Prakasam, Oregon Health & Science University, United States
Copyright © 2021 Pae and Harper. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Eung-Kwon Pae, ZXBhZUB1bWFyeWxhbmQuZWR1
 Eung-Kwon Pae
Eung-Kwon Pae Ronald M. Harper
Ronald M. Harper