- 1Department of Restorative Dentistry and Endodontology, Osaka University Graduate School of Dentistry, Osaka, Japan
- 2Division of Restorative Dentistry and Periodontology, Trinity College Dublin, Dublin Dental University Hospital, Dublin, Ireland
Vital pulp treatment (VPT) is a therapeutic strategy aimed at conservatively managing deep carious lesions and the exposed pulp. VPT has recently expanded through the use of hydraulic-calcium-silicate cement (HCSC), cone-beam CT, and dental operating microscopy (DOM), as well as an improved understanding of pulpal repair mechanisms. Consensus documents have concluded that non-selective removal of caries is not necessary in the absence of clinical symptoms, as partial or non-selective caries removal techniques reduce the likelihood of pulp exposure. Alternatively, others suggest that leaving carious tissue may help sustain pulpal inflammation and lead to failure of VPT. The presence of a purulent discharge from the exposed pulp is considered indicative of an irreversible damage and a need for pulpectomy. This report documents the successful VPT in a symptomatic molar with deep caries that exhibited purulent discharge from the pulp after exposure. A female patient presented with spontaneous pain in the maxillary left second molar. One year before, she had received a cast-metal restoration over a resin-based-composite (RBC) restoration. The tooth had been largely asymptomatic since; however, after experiencing spontaneous pain, she consulted the hospital clinic. Radiographs indicated that the restoration was not deep with visible dentin between the restoration and the pulp. Pulp sensibility tests yielded a positive response. There was no pain on percussion and no periodontal problems. Removal of the restoration and carious dentin using a DOM resulted in a pulpal exposure with purulent discharge and bleeding. A partial pulpotomy using HCSC was performed. Six months later, the clinical symptoms had completely resolved; however, due to concerns of partial necrosis, the patient was re-assessed. DOM examination revealed an incomplete hard-tissue barrier; moreover, there were also signs of residual tissue with inflammation. As a result, the defected area was enlarged, the pulp tissue debrided, and the HCSC reapplied. After review at 1 year, the tooth remains symptomless, with no apical radiolucency. Notably, this report visually illustrates that exposed pulp tissue containing limited region pus can maintain vitality after VPT. This finding raises two questions: (1) whether leaving infected dentin in situ and avoiding visualization of the pulp exposure is appropriate; and (2) whether the presence of purulent tissue in the pulp warrants a pulpectomy.
Introduction
Vital pulp treatment (VPT) is a biologically based and minimally invasive procedure, which has recently become the focus of extensive research activity and interest in endodontics. Notably, avoiding pulpectomy and maintaining pulp function has been reported to increase tooth retention in teeth with deep caries (1, 2).
VPT management strategies include selective removal techniques for management of deep caries to avoid exposure, as well as exposed pulp management strategies including pulp capping in which the injured tissue is restored without tissue removal, partial pulpotomy, which removes 2–3 mm of pulp tissue, and full/coronal pulpotomy, which removes the coronal pulp to the cervical region of the tooth (3). Recently, VPT has been used to manage deep carious lesions, and the exposed pulp has been expanded through the use of hydraulic-calcium-silicate cement (HCSC), cone-beam CT (CBCT), and the dental operating microscope (DOM), as well as an improved understanding of pulpal repair mechanisms. Additionally, the technical skills of the dental surgeon are likely to be an important variable related to the outcome of the VPT procedure (3).
Conventionally, calcium hydroxide has been applied as the “capping” material in VPT (4). However, with the advent of HCSC, which has better sealing characteristics and biocompatibility than calcium hydroxide (5), the procedure has become more predictable (6). The European Society of Endodontology (ESE) position statement for management of deep caries and the exposed pulp (3) concluded that in the absence of clinical symptoms, non-selective removal of caries is not warranted, as partial or selective caries removal techniques reduce the likelihood of pulp exposure. Alternatively, others have suggested that leaving carious tissue may help sustain pulpal inflammation and lead to failure of the VPT procedure (7). Furthermore, they suggest that in the absence of direct visualization of the exposed pulp, purulent discharge from the exposed pulp may be present, indicating irreversible damage and a need for pulpectomy (7).
Recently, it has been reported that partial pulpotomy can be applied to preserve the pulp in mature teeth as well as in immature teeth diagnosed with irreversible pulpitis (8, 9); however, the visual presence of pulp necrosis has not been described. This case report documents a successful partial pulpotomy of a mildly symptomatic maxillary molar tooth with deep caries that exhibited pus discharge from the pulp after exposure and visualization under the DOM.
Case Report
An overview of the treatment is highlighted in Table 1. On her first visit, a 46-year-old female patient presented with spontaneous pain around her left maxillary second molar, which had previously received an indirect metal restoration above a resin-based-composite (RBC) restoration placed as a liner at a private dental clinic 1 year previously. This tooth was mostly asymptomatic after restoration. However, after experiencing spontaneous pain for several months, she consulted several private dental clinics. Unfortunately, the diagnosis remained unclear, and, as a result, she visited our dental hospital. The patient was a non-smoker and took no medication. General medical history and family history were not noted.
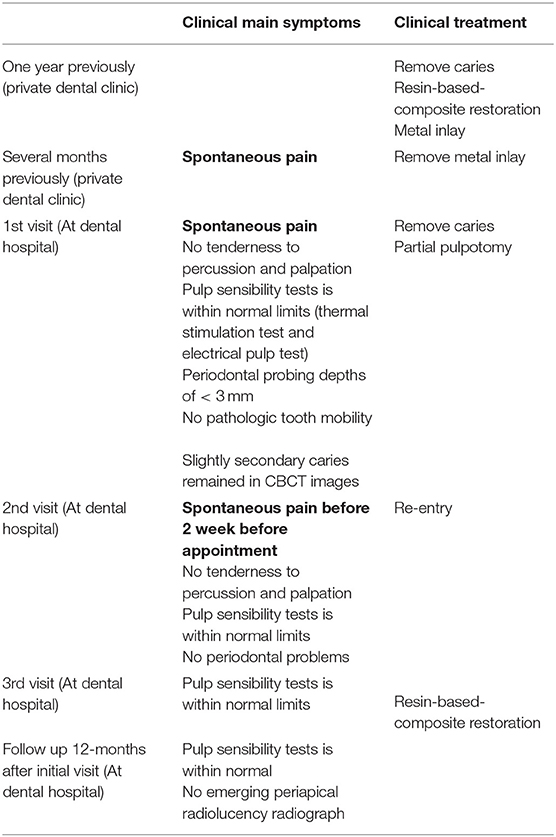
Extraoral examination revealed no facial asymmetry or swelling, while intraoral examination revealed no swelling or sinus tract in the region of the left maxillary second molar. The tooth demonstrated no tenderness to percussion and palpation. Additionally, pulp sensibility tests demonstrated a response within normal limits to thermal stimulation test (“pulper” [cold test] and warmed “temporary stopping” material [heated test] GC, Tokyo, Japan) and the electrical pulp test (Digitest II, Morita, Kyoto, Japan), which was compared with adjacent and contralateral teeth. When applying the tests, the patient was asked to report the persistence of pain by applying sponge pellets cooled with a cooling spray or warmed stick-shape temporary material applied to the tooth surface. The buccal and palatal tooth surfaces were tested, and transient mild pain was reported. Periodontal probing depths of <3 mm were noted around the tooth with no pathologic tooth mobility. During previous examination in a private clinic, bruxism and occlusal interference during lateral movement was suspected to be the cause. As a result, the metal restoration was already removed. Overall, the oral hygiene was good, and plaque levels were well-controlled. To further investigate the etiology, intraoral periapical radiographic images and a limited field CBCT image were taken (Figure 1). The intraoral radiographic image (Figure 1A) revealed that the RBC restoration as a liner remained in situ, and that there was visible radio dense “dentin” between the restoration and the pulp chamber. Conversely, CBCT images of the sagittal section highlighted the resin restoration to be close to the pulp chamber (Figure 1B) and suggested potential residual infected dentin, secondary caries, or artifacts in the “liner” material (Figure 1C). However, it was not possible to make a definite determination at the preoperative stage, and treatment was started after explaining that intraoperative investigation would help to clarify the diagnosis. Thickening of the maxillary sinus mucosa was also evident, but the relationship with the maxillary left second molar remained unknown. No abnormalities were detected in the periapical region. Analysis of the clinical findings concluded that the diagnosis was irreversible pulpitis due to microleakage from the previously placed restoration or remaining infected dentin, which had stimulated inflammation as the infected dentin was in close proximity of the pulp tissue.
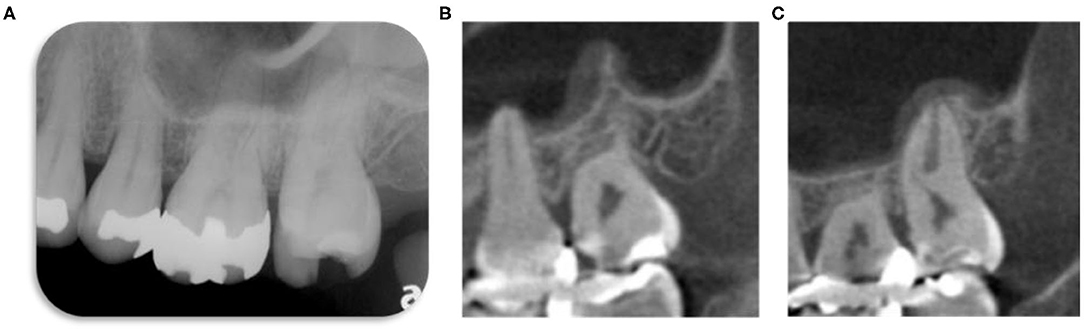
Figure 1. Radiographic image taken during the patient's initial visit to the University Clinic. (A) Intraoral radiographic image. The maxillary left second molar with the restoration already removed. No abnormal findings were discovered on any other teeth. There seems to be a sufficient distance between the restoration and the pulp (≤5 mm), but this remains ambiguous. (B) Selected sagittal images of cone-beam computed tomography (CBCT) volume. Three-dimensional evaluation by CBCT shows that the mesial filling is close to the mesial pulp horn. (C) Sagittal images of CBCT. Thickening of the maxillary sinus mucosa is seen, but the relationship with the maxillary left second molar remains unknown.
Based on the clinical assessment, a decision was made to treat the patient using the DOM (OPMI Pico MORA/S100, Carl Zeiss, Oberkochen, Germany). Under local anesthesia and by using rubber dam isolation (Heraeus Kulzer, South Bend, IN, USA), the RBC and remaining infected dentin were removed using a round diamond bur (diameter size 1.4 mm, Shofu, Kyoto, Japan) in a water-cooled high-speed handpiece prior to the use of hand instruments for caries removal (Excavotor round, YDM, Tokyo, Japan). After removing the restoration, the infected dentin was carefully removed from the base of the carious lesion to avoid exposing the pulp while removing the infected dentin. This was guided by both caries detection fluid (Caries Detector, Kuraray, Tokyo, Japan) and the resistance of the dentin to excavators under the DOM. The infected dentin remained deep and localized on the pulpal aspect, rather than around the restoration, suggesting that residual infected dentin was stimulating the pulpitis. Although the infected dentin was thought to have been completely removed, the DOM highlighted areas of spot staining with caries detection fluid (Figure 2D, black arrow). The staining could not be accessed effectively using normal hand instruments (excavator round, YDM, Tokyo, Japan); a custom microextractor was used (YDM, Tokyo, Japan, Figures 2A–C). A microextractor is an instrument normally used for root canal treatment to remove areas that are softer than the surrounding dentin. After the infected dentin was removed and the pulp tissue reached, pus visibly overflowed from the exposure site, accompanied by profuse bleeding (Figures 2E,F). To prevent the ingress of infected dentin debris, the exposed pulp was thoroughly irrigated with 2.5% sodium hypochlorite (Neo Cleaner, NC, Neo Dental Chemical Products Co., LTD, Tokyo, Japan) and 3% ethylenediaminetetraacetic acid (EDTA) solutions (Smear Clean, Nishika, Yamaguchi, Japan) using a root canal syringe (Neo Dental Chemical Products Co., LTD, Tokyo, Japan). The infected dentin was completely removed until the remaining dentin was hard and resistant to hand excavator instruments. The subsequent examination of the pulpal injury site under the DOM revealed pulp tissue with a vascular-like structure (Figure 2G, refer to Supplementary Videos doi: 10.6084/m9.figshare.13228427). Therefore, the intraoperative and postoperative findings altered the preoperative diagnosis of irreversible pulpitis to a case with reversible pulpitis. The volume of tissue resection in the partial pulpotomy using a round diamond bur in a water-cooled high-speed handpiece was deemed sufficient. The amount of pulp resected was about 3.5 mm in depth. After irrigation with 2.5% sodium hypochlorite and EDTA solution, hemostasis was achieved in <3 min. ProRoot MTA (Dentsply Sirona, Ballaigues, Switzerland) was carefully placed on the exposed pulp area and cavity following the manufacturer's instructions. Cotton wool pledgets soaked with purified water were left on the cement to accelerate the setting reaction, and a temporary restoration was placed using glass ionomer cement (GIC) (Glass-ionomer FX Ultra, GC Tokyo, Japan). At this time, a final restoration was considered; however, because of the patient's time constraints and the fact that additional pulpotomy may be considered depending on the condition of the pulp after treatment, a temporary restoration was performed. After treatment, intraoral radiographic images were taken (Figure 2H).
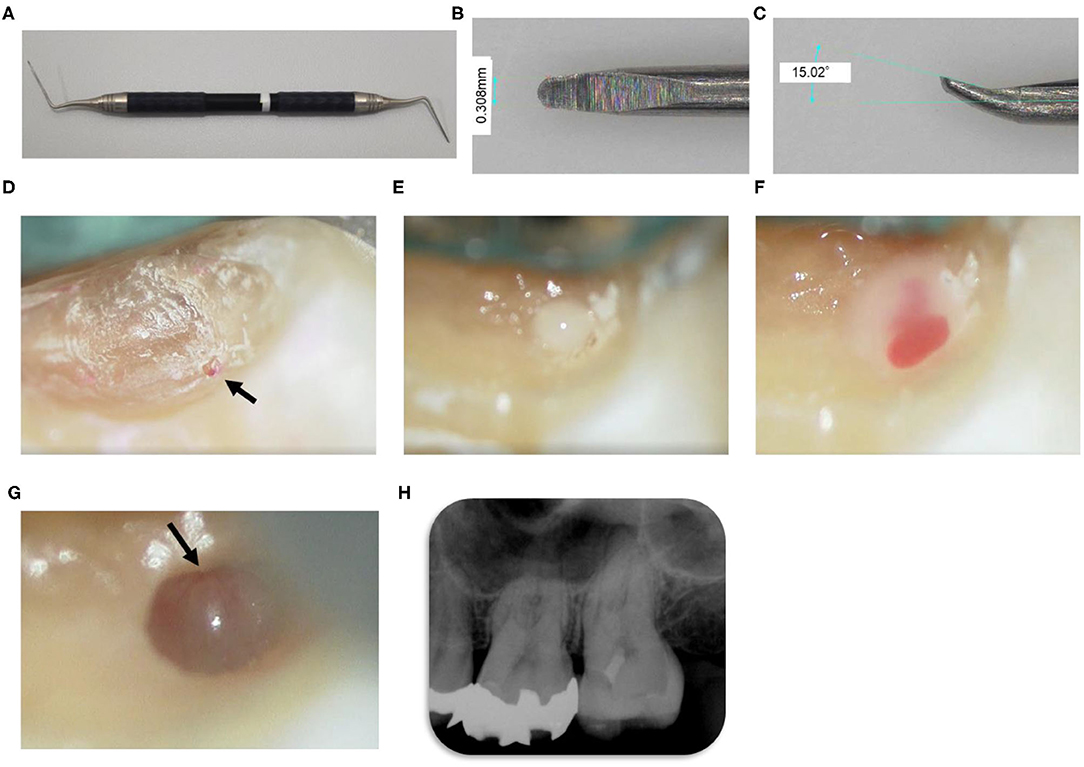
Figure 2. Clinical images extracted from video recording during the first visit. Videos are available online at doi: 10.6084/m9.figshare.13228427 as Supplemental Materials. (A) A custom microextractor. (B) Enlarged image highlighting the size and scale of the microextractor. (C) Enlarged image showing the angle to axis of microextractor. (D) Microscopic image immediately after removal of infected dentin without magnification. The dental operating microscopy demonstrated areas of “spot staining” with caries detection fluid (black arrow). (E,F) Pus overflowed from the exposed pulp tissue and the area bled profusely. (G) After debridement, pulp tissue with vascular-like structures was observed (black arrow). (H) Radiographic images after pulpotomy procedure.
A follow-up appointment was scheduled after 1 month. However, as the patient could not re-attend as scheduled due to coronavirus disease (COVID-19) restrictions, the treatment was interrupted for ~5 months. Immediately after the treatment was done at the first visit and 2 weeks prior to the second visit, the patient reported a slight pain that lasted only for a short duration of time. However, these pains were completely tolerable, and no analgesics were required by the patient. By the time of the second visit, however, no discomfort, pain, or untoward clinical symptoms were evident. The pulp test yielded a normal response to thermal and electric pulp sensibility tests, similar to the response noted on the first visit. The radiographic images demonstrated no change in the periapical tissue (Figure 3A). After discussion, a decision was made to reenter the tooth, as the previous finding of purulent discharge, suspected irreversible pulpitis, and two reported incidences of mild pain occurring after the partial pulpotomy and just 2 weeks before the patient appointment suggest that inflamed tissue may have remained in situ.
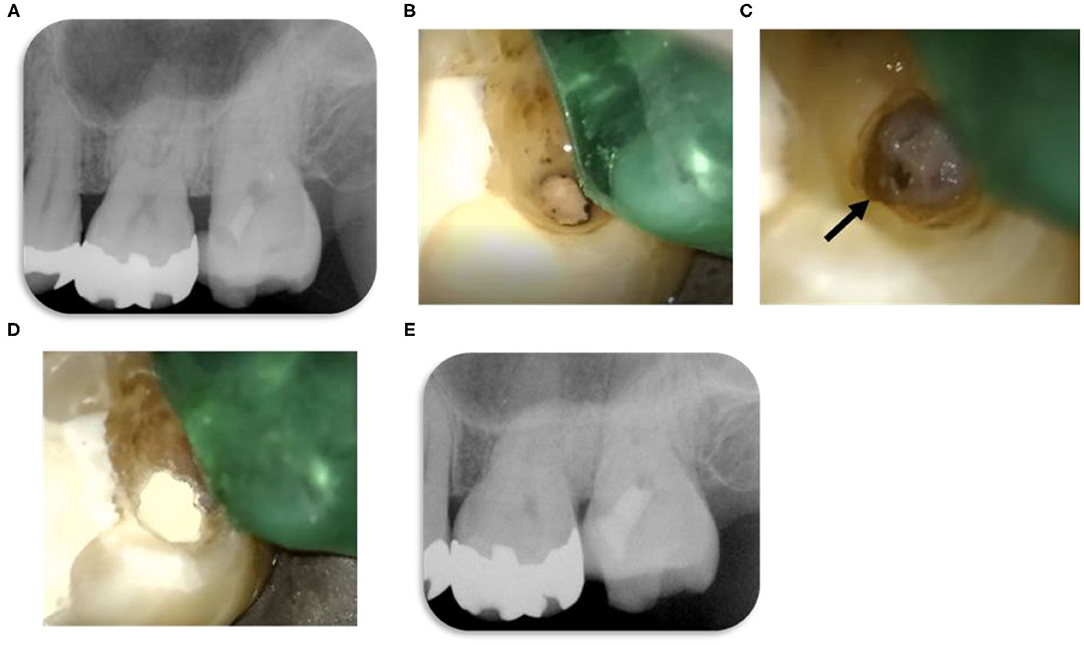
Figure 3. Reentry clinical images extracted from clinical videos during the second visit. Videos are available online at doi: 10.6084/m9.figshare.13228661 as Supplemental Materials. (A) Radiographic image ~6 months after the first visit. There is no significant change in periapical tissue, and hard tissue formation is evident beneath the hydraulic-calcium-silicate cement (HSCS). (B) ProRoot MTA cement exposed after removing the temporary sealing. Black discoloration is observed at the periphery of the material between the cement and the dentin. (C) After removal of the ProRoot MTA cement, hard tissue formation was observed beneath the exposed pulp. Black arrows indicate defects. (D) Exposed pulp and hard tissue were redressed with ProRoot MTA cement. (E) Radiographic image after reentry. The hard tissue formation observed under the dental microscope is not clear on this radiographic image. HCSC volume is visibly higher compared with the first dressing.
For the reentry procedure, local anesthesia was administered, after which rubber dam isolation was carried out as before. The temporary restoration was removed using a diamond bur and a high-speed handpiece with water spray. Thereafter, the MTA cement was carefully removed using a diamond tip powered by an ultrasonic device (Suprasson Newtron P-MAX2, Satelec, Acteon group, Bordeaux, France). The boundary between the MTA cement and the dentin was discolored and had become black, but no discoloration of the MTA cement itself or the dentin was evident (Figure 3B). After removal of the MTA cement, white hard tissue was observed in the exposed wound under a DOM (Figure 3C, black arrow). However, areas of insufficient hard tissue formation (defects) were identified at the borders of the mineralized repair tissue.
On the basis of visual inspection and clinical tests, i.e., the patient responded normally to percussion pain and electric pulp examination, as compared to neighboring teeth, the partial pulpotomy appeared not to have failed. The placement of a final restoration was considered as an option; however, due to the complaint of slight pain 2 weeks prior to the second visit by the patient and uncertainty involved in the extent of removal of the small portion of the pulp in the case of necrosis, it was decided that a reentry should be performed after explaining the procedure to the patient. As there was a possibility that some infected pulp remained in the defective area, the defective repair was removed to expose the healthy pulp. After enlarging the cavity, the exposed pulp was identified, alongside minimal bleeding, and the patient complained of slight pain in spite of the anesthesia. At this stage, the reentry was considered sufficient and as for the intraoperative diagnosis that supplemented preoperative information with clinical information, it was also concluded reversible pulpitis. After irrigation with sodium hypochlorite and EDTA solutions, hemostasis was again achieved in <3 min. Then, the exposed pulp was covered with ProRoot MTA, and the cavity was restored with MTA cement to preserve sufficient thickness (Figure 3D). The initial setting time of the MTA cement was sufficient to proceed with restoration. After setting with water, the cavity was covered with RBC (Clearfil universal bond Quick ER and Clearfill Majesty ES flow, Kuraray, Tokyo, Japan). Due to time constraints of the patient, a temporary restoration prior to the final restoration was performed using a GIC (Glass-ionomer FX Ultra, Shofu, Kyoto, Japan). After the treatment, intraoral radiographic images were obtained (Figure 3E). The amount of pulp resected was ~4.2 mm in depth from clinical gauging and the radiographic image.
At the third visit, the patient claimed that all the clinical symptoms had completely resolved. There was no spontaneous pain, and the pulp sensitivity test was the same as the adjacent teeth and the preoperative evaluation. After discussion with the patient, RBC filling was selected as the final restoration. A rubber dam was placed and the temporary material removed. The class II cavity was prepared. A metal matrix, wedges, and retainers were adapted and placed on the treating tooth (Composi-tight 3D system, Garrison Dental Solutions, Spring Lake, MI, USA, Figure 4A). Selective etching was performed using a K-etchant gel (Kuraray, Tokyo, Japan). Application of the primer and bonding agent (Clearfil Megabond FA, Kuraray, Tokyo, Japan) was done as per the manufacturer's protocol. After confirming curing by blue light irradiation (Pen cure 2000, Morita, Kyoto Japan), a layered restoration was performed using RBC (Clearfill Majesty ES flow and Clearfil Majesty ES-2, Kuraray, Tokyo, Japan).
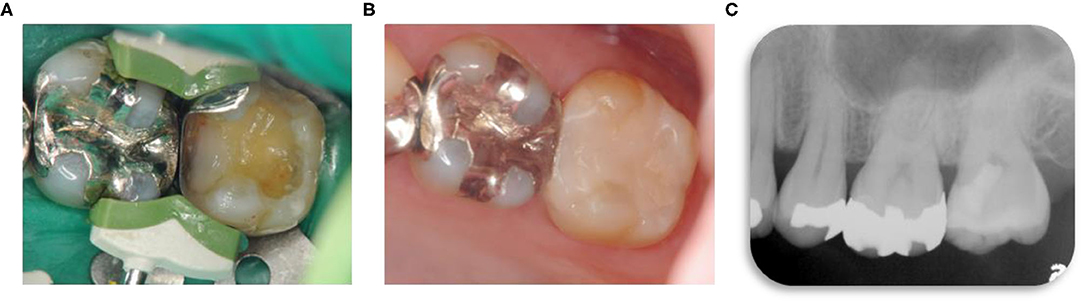
Figure 4. Images at follow-up visit 8 months after completion of the partial pulpotomy. (A) Composite 3D system for resin-based composite. (B) Intraoral image 12 months after the partial pulpotomy. (C) Radiographic image 12 months after the partial pulpotomy revealed the absence of an apical radiolucency indicative of apical periodontitis.
At 12 months after the initial visit, postoperative clinical examination revealed a pulpal response to sensibility test within normal limits. Intraoral photo and radiographic images obtained 12 months after the first partial pulpotomy procedure demonstrated that the treatment had been successful (Figures 4B,C). These series of treatments were performed by a specialist with more than 10 years of experience in the endodontics department.
Discussion
Recently, developments of dental equipment and new biologically based materials have allowed clinicians to maintain pulp tissue in scenarios where, traditionally, pulpectomy would have been the treatment of choice. A well-documented randomized control trial of partial pulpotomy reported a success rate of >80% at 2 years, even in mature teeth diagnosed with irreversible pulpitis (9).
In the current case, the patient had previously visited several dental clinics complaining of mild pain; however, a definitive cause could not be identified. During examination at the previously visited clinics, pain due to occlusal interference was considered. As a result, the existing metal restoration was removed from the infected molar tooth, leaving only the RBC as a liner. However, the pain persisted. In our hospital, clinical examination results demonstrated no abnormalities in the periodontal tissues and no obvious proximity to the pulp, and therefore, a poorly sealing restoration with remaining infected dentin was suspected. Removal of the restoration and replacement with a new restoration was deemed to be a sensible solution for the mild symptoms.
After complete removal of the RBC, the infected dentin appeared to be removed without pulp exposure. Therefore, indirect pulp capping seemed to be the most appropriate option to improve the symptoms; however, magnification revealed a “fluid-stained” spot indicative of a microexposure. It is postulated that these “micro” pulp exposure may be prevented during selective caries removal by using magnification, which emphasizes the importance of technical precision when removing infected dentin. Three-dimensional evaluation by CBCT was used to search for the cause of the slight pain. These 3D images showed that the lesion was deeper than thought and that the pulp exposure may have occurred during the previous treatment (Figure 1B). Although the use of liners is no longer routinely advocated, current scientific evidence does not recommend restoring the pulp directly with RBCs for reasons of biocompatibility (10). In addition, the CBCT scan revealed secondary caries and remaining infected dentin (Figure 3A). Therefore, from the results of the CBCT, microleakage of the restoration was suspected as being the cause of the pain. This information can be difficult to establish from conventional 2D intraoral radiographic images. Identifying caries by CBCT is not always accurate (11), but CBCT may contribute to identification of caries by three-dimensional image assessment when the diagnosis is unclear or complex. Therefore, based on the as-low-as reasonably achievable (ALARA) principles, CBCT support may be justified in a case such as this if the patient benefits from the extra information supplied.
The infected dentin was subsequently treated with a manual custom-made microexcavator (Figures 2A–C), and the pus drainage from the exposed pulp tissue was confirmed (Figure 2E). The evidence of pus may have been missed or simply washed away if a water-cooled handpiece or ultrasound device had been used to expose the pulp. It is critical that dental instruments are carefully manipulated in areas close to the pulp using magnification like the DOM. Hand instrumentation increases the visibility when using the DOM and provides tactile sensation of the hardness of the remaining dentin compared to other devices, but there is a possibility of pulpal contamination. Therefore, hand instruments are recommended during the removal of infected dentin. A water-cooled handpiece or an ultrasound device is recommended after the pulp is exposed.
Furthermore, although it was previously thought that pulp with an abscess required complete removal, a recent report suggested that, histologically, the inflammatory reaction may be localized only to the injury site, and that the pulp immediately below the local inflammation is relatively free of inflammatory change and may be preserved (12). In the present case, purulent discharge was of limited duration and location, and the deeper pulp tissues appeared healthy. This implies an actual diagnosis of localized irreversible pulpits with the bulk of the pulp being only reversibly inflamed. In addition, hemostasis (Figure 2G) as well as hard tissue deposition (Figure 3C) could be achieved after disinfection of the wound site. Crucially, a magnified view using the DOM and adaptation of HCSC high-biocompatibility materials are essential in this treatment, which perhaps underlines the view that such cases are more technically difficult than generally considered. In the present case, ProRoot MTA was used as the HCSC as it is the most widely used HCSC and is a reliable biological material, based on extensive evidence; however, it can be associated with tooth discoloration (13–15). Other HCSC with no risk of discoloration should be selected for VPT procedures in aesthetic areas such as the anterior areas of the mouth.
Reentry was mainly justified as during the hiatus in treatment over the course of the COVID-19 pandemic, the patient experienced slight spontaneous pain, albeit only twice. It was difficult to determine whether this pain was due to the healing process, leakage, a residual infected tissue, or a separate dental issue. In this case, as a partial pulpotomy was carried out in the second molar, there was concern that the partial irreversible pulpitis may have been sustained in the residual pulp tissue. We explained to the patient that we would reenter the lesion under strict aseptic conditions in order to verify the healing response. The patient agreed with our opinion. During reentry, hard tissue formation was confirmed beneath the exposed pulp; however, a partial defect was confirmed in the hard tissue. Presumably, a level of pulpitis remained, and complete hard tissue formation could not be achieved. As a result, the defect was further enlarged in order to reach the healthy pulp tissue. A 2.5% sodium hypochlorite and EDTA solution was used to irrigate the exposed pulp prior to washing with saline prior to cement filling. EDTA was used to assist in the removal of any inorganic debris created during entry such as infected dentin small tips produced after hand instrument and hand ultrasonic device that had entered the pulp space. EDTA was expected to release growth factors and bioactive dentin matrix components from dentin, which would be effective in wound healing (16). Note that a chlorhexidine solution could also be advocated as an irrigation solution for pulpal lavage.
The recommended follow-up period for the procedure is after 6 and 12 months. If any concerns arise, it is recommended to wait up to 4 years, if necessary (2). The length of the follow-up period is controversial; it has been reported that most failures of HCSC-applied VPT often occur within 6 months (17); however, another study reported 3 months as an appropriate follow-up based on similar success rates at 3 and 18 months (18). In this case, a follow-up of ~8 months revealed no additional symptoms. Success is predicted based on the fact that it has been over 6 months without any signs or symptoms or evidence of failure. Continued success is predicted based on the fact that it has been over 10 months without any signs or symptoms. Microleakage is a factor that affects long-term prognosis (19); hence, the tooth will continue to be carefully observed by regular review.
In summary, in this case, the caries process did not appear to reach the pulp chamber from the preoperative intraoral radiographic image at the time of the first examination. Therefore, conventional analysis and pulp tests, including electrical pulp test and thermal stimulation test, are probably inappropriate for the detection of partial pulp abnormalities such as partial necrosis and partial abscesses. A thorough history and potential additional preoperative examination using CBCT is an important consideration for these cases, which are often difficult to diagnose. The current consensus on deep caries is that leaving a small amount of residual infected dentin on the pulpal floor can be tolerated by the pulpal tissues rather than risking its exposure at least in cases with mild symptoms (3). As the patient complained of only minimal pain in this case, indirect pulp capping may have been considered the most obvious treatment option. However, the presence of pulpal abscess and infection would interfere with the success of indirect pulp capping. Recent, histological evaluations suggest that residual infected dentin may lead to persistent chronic inflammation of the pulp tissue and abscess formation in certain cases. Infected dentin may be completely removed in cases with clinical symptoms, even in cases of slight but spontaneous pain. Unfortunately, spontaneous pain is also subjective and difficult to evaluate quantitatively, highlighting the problem in selective caries removal for managing deep caries lesions. Taha et al. (20) reported that “current pulp testing is not reliable to the extent that determining pulpal status has been described as ‘at best an educated guess,' and an urgent need exists for more definitive tests.” A pulp examination method that includes an analysis of pulpal inflammatory biomarkers to assess the level of pulpitis intra-operatively is expected to develop. However, in further understanding pulpal inflammation and repair, the accumulation of illustrative date including video-illustrated case reports like the current study may prove invaluable. There are very few reports or systematic reviews addressing partial pulpotomy in mature teeth with irreversible pulpitis (21). The systematic review by Cushley et al. demonstrated that the success rate of partial pulpotomy for mature teeth diagnosed with irreversible pulpitis is acceptable and could be an alternative method to root canal treatment in terms of clinical time, the operator's skill, and cost-effectiveness (22). Additionally, in the procedure of partial pulpotomy, the pulp tissue can be removed in stages after intraoperative assessment of tissue damage (e.g., 2–3 mm of the coronal pulp at a time). In this case report, the DOM has the potential to contribute to the extent of pulp resection.
As a result, the most appropriate management strategy in reaching a comprehensive diagnosis based on clinical signs and symptoms remains in question, with a case made that in cases with symptoms and deep caries, the pulp tissue should always be visualized using the DOM or magnification.
Conclusion
This report visually illustrates that a symptomatic exposed pulp tissue contained limited region pus can maintain vitality with careful tissue handling and VPT. This raises questions regarding the current thinking of whether to leave the infected dentin in situ and avoid pulp exposure as part of a non-selective caries removal strategy, and, conversely, the notion that the presence of purulent tissue in the pulp space necessitates pulpectomy.
Data Availability Statement
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author/s.
Ethics Statement
The patient provided written informed consent to participate in this report.
Author Contributions
OM and DF: conceptualization. SM: software and investigation. TY and HM: validation. OM: formal analysis, visualization, and data curation. DF: resources. OM, KN, and DF: writing—original draft preparation. TY and DF: writing—review and editing. HM: supervision. OM: funding acquisition. All authors have read and agreed to the published version of the manuscript.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We are grateful to Dr. Madoka Okabe, Shimaoka Tsuyoshi, and Mika Ando from the Department of Restorative Dentistry and Endodontology, Osaka University Graduate School of Dentistry, for many help throughout the treatment. We would like to thank Editage (www.editage.jp) for the English language editing.
References
1. Bjørndal L, Simon S, Tomson PL, and Duncan HF. Management of deep caries and the exposed pulp. Int Endod J. (2019) 52:949–73. doi: 10.1111/iej.13128
2. Duncan HF, Galler KM, Tomson PL, Simon S, El-Karim I, Kundzina R, et al. European society of endodontology position statement: management of deep caries and the exposed pulp. Int Endod J. (2019) 52:923–34. doi: 10.1111/iej.13080
3. Aguilar P, and Linsuwanont P. Vital pulp therapy in vital permanent teeth with cariously exposed pulp: a systematic review. J Endod. (2011) 37:581–7. doi: 10.1016/j.joen.2010.12.004
4. Hosoya N, Takigawa T, Horie T, Maeda H, Yamamoto Y, Momoi Y, et al. A review of the literature on the efficacy of mineral trioxide aggregate in conservative dentistry. Dent Mater J. (2019) 38:693–700. doi: 10.4012/dmj.2018-193
5. Kundzina R, Stangvaltaite L, Eriksen HM, and Kerosuo E. Capping carious exposures in adults: a randomized controlled trial investigating mineral trioxide aggregate versus calcium hydroxide. Int Endod J. (2017) 50:924–32. doi: 10.1111/iej.12719
6. Ricucci D, Siqueira JF, Rôças IN, Lipski M, Shiban A, and Tay FR. Pulp and dentine responses to selective caries excavation: A histological and histobacteriological human study. J Dent. (2020) 100:103430. doi: 10.1016/j.jdent.2020.103430
7. Uesrichai N, Nirunsittirat A, Chuveera P, Srisuwan T, Sastraruji T, and Chompu-Inwai P. Partial pulpotomy with two bioactive cements in permanent teeth of 6- to 18-year-old patients with signs and symptoms indicative of irreversible pulpitis: a noninferiority randomized controlled trial. Int Endod J. (2019) 52:749–59. doi: 10.1111/iej.13071
8. Taha NA, and Khazali MA. Partial pulpotomy in mature permanent teeth with clinical signs indicative of irreversible pulpitis: a randomized clinical trial. J Endod. (2017) 43:1417–21. doi: 10.1016/j.joen.2017.03.033
9. Krifka S, Seidenader C, Hiller KA, Schmalz G, and Schweikl H. Oxidative stress and cytotoxicity generated by dental composites in human pulp cells. Clin Oral Investig. (2012) 16:215–24. doi: 10.1007/s00784-010-0508-5
10. Krzyzostaniak J, Kulczyk T, Czarnecka B, and Surdacka A. A comparative study of the diagnostic accuracy of cone beam computed tomography and intraoral radiographic modalities for the detection of noncavitated caries. Clin Oral Investig. (2015) 19:667–72. doi: 10.1007/s00784-014-1282-6
11. Ricucci D, Siqueira JF, Li Y, and Tay FR. Vital pulp therapy: histopathology and histobacteriology-based guidelines to treat teeth with deep caries and pulp exposure. J Dent. (2019) 86:41–52. doi: 10.1016/j.jdent.2019.05.022
12. Bakhtiar H, Nekoofar MH, Aminishakib P, Abedi F, Naghi Moosavi F, Esnaashari E, et al. Human pulp responses to partial pulpotomy treatment with TheraCal as compared with biodentine and ProRoot MTA: a clinical trial. J Endod. (2017) 43:1786–91. doi: 10.1016/j.joen.2017.06.025
13. Kang CM, Kim SH, Shin Y, Lee HS, Lee JH, Kim GT, et al. A randomized controlled trial of ProRoot MTA, OrthoMTA and RetroMTA for pulpotomy in primary molars. Oral Dis. (2015) 21:785–91. doi: 10.1111/odi.12348
14. Suhag K, Duhan J, Tewari S, and Sangwan P. Success of direct pulp capping using mineral trioxide aggregate and calcium hydroxide in mature permanent molars with pulps exposed during carious tissue removal: 1-year follow-up. J Endod. (2019) 45:840–7. doi: 10.1016/j.joen.2019.02.025
15. Harms CS, Schafer E, and Dammaschke T. Clinical evaluation of direct pulp capping using a calcium silicate cement - treatment outcomes over an average period of 2.3 years. Clin Oral Investig. (2019) 23:3491–9. doi: 10.1007/s00784-018-2767-5
16. Widbiller M, Eidt A, Hiller KA, Buchalla W, Schmalz G, and Galler KM. Ultrasonic activation of irrigants increases growth factor release from human dentine. Clin Oral Investig. (2017) 21:879–88. doi: 10.1007/s00784-016-1824-1
17. Kang CM, Sun Y, Song JS, Pang NS, Roh BD, Lee CY, et al. A randomized controlled trial of various MTA materials for partial pulpotomy in permanent teeth. J Dent. (2017) 60:8–13. doi: 10.1016/j.jdent.2016.07.015
18. Matsuo T, Nakanishi T, Shimizu H, and Ebisu S. A clinical study of direct pulp capping applied to carious-exposed pulps. J Endod. (1996) 22:551–6. doi: 10.1016/S0099-2399(96)80017-3
19. Cox CF, Bergenholtz G, Heys DR, Syed SA, Fitzgerald M, and Heys RJ. Pulp capping of dental pulp mechanically exposed to oral microflora: a 1-2 year observation of wound healing in the monkey. J Oral Pathol. (1985) 14:156–68. doi: 10.1111/j.1600-0714
20. Taha NA, About I, Sedgley CM, and Messer HH. Conservative management of mature permanent teeth with carious pulp exposure. J Endod. (2020) 46:S33–41. doi: 10.1016/j.joen.2020.06.025
21. Cushley S, Duncan HF, Lappin MJ, Tomson PL, Lundy FT, Cooper P, et al. Pulpotomy for mature carious teeth with symptoms of irreversible pulpitis: a systematic review. J Dent. (2019) 88:103158. doi: 10.1016/j.jdent.2019.06.005
Keywords: vital pulp therapy, partial pulpotomy, hydraulic calcium-silicate cement, dental operating microscope, cone beam computed tomography, pulpitis
Citation: Motoki O, Fergus DH, Yusuke T, Nanako K, Matsumoto S and Mikako H (2021) Partial Pulpotomy to Successfully Treat a Caries-Induced Pulpal Micro-Abscess: A Case Report. Front. Dent. Med. 2:678632. doi: 10.3389/fdmed.2021.678632
Received: 10 March 2021; Accepted: 26 April 2021;
Published: 22 June 2021.
Edited by:
Ashraf F. Fouad, University of Alabama at Birmingham, United StatesReviewed by:
Waruna Lakmal Dissanayaka, The University of Hong Kong, Hong Kong SAR, ChinaNatalino Lourenço Neto, Universidade de São Paulo, Brazil
Copyright © 2021 Motoki, Fergus, Yusuke, Nanako, Matsumoto and Mikako. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Okamoto Motoki, bW90b2tpQGRlbnQub3Nha2EtdS5hYy5qcA==
 Okamoto Motoki
Okamoto Motoki Duncan Henry Fergus
Duncan Henry Fergus Takahashi Yusuke
Takahashi Yusuke Kuriki Nanako1
Kuriki Nanako1