- 1Laboratory Department, Kamuzu Central Hospital, Lilongwe, Malawi
- 2Department of Biomedical Sciences, School of Health Sciences, University of Zambia, Lusaka, Zambia
- 3Malawi Blood Transfusion Service, Laboratory Department, Lilongwe, Malawi
- 4Malawi-Liverpool-Wellcome Trust (MLW), Blantyre, Malawi
- 5University of North Carolina (UNC) Project, Lilongwe, Malawi
Background: Blood transfusions are vital for treating anemia, yet in malaria-endemic regions such as Malawi, the risk of infection transmission remains significant. Owing to constrained resources and infrastructure, many blood centers have inadequate capacity for malaria detection, leading to the absence of routine screening protocols. This study aimed to explore the prevalence of asymptomatic malaria parasitemia, identify Plasmodium species, and investigate associated factors among blood donors at the Lilongwe Centre of the Malawi Blood Transfusion Service (MBTS).
Methods: This study employed a cross-sectional research design conducted at the Lilongwe Branch of the Malawi Blood Transfusion Service, a pivotal center for blood collection and distribution in Malawi’s central region. The research focused on a population of voluntary asymptomatic blood donors who presented themselves at designated donation centers. The prevalence of asymptomatic malaria parasitemia was determined using two methods: Plasmodium falciparum histidine-rich protein 2 (PfHRP2) rapid diagnostic tests (RDTs) for detecting Plasmodium falciparum parasites and microscopic analysis for confirmation and identification of other Plasmodium species, along with density quantification. Identification of factors associated with malaria transmission among blood donors was done using structured questionnaire. The study was conducted over a two-week period at the MBTS, with data analysis performed using R version 4.3.1, with statistical significance set at p-value < 0.05.
Results: The study identified an overall malaria prevalence of 13.5% (51/377) among the participants, with 13.4% (9/67) of females and 13.5% (42/310) of males testing positive. Of the positive cases, 85.4% (41/48) were due to Plasmodium falciparum, 8.3% (4/48) were mixed infections of Plasmodium falciparum and Plasmodium malariae, and 6.3% (3/48) were solely Plasmodium malariae. Significant demographic factors influencing species distribution included age, occupation and residence. Other significant factors were blood group, repellent use, and travel history. A multivariate logistic regression model was used, but no significant associations were identified at the 0.05 significance level.
Conclusion: The detection of a substantial burden of asymptomatic malaria parasitemia among blood donors highlights a potential risk for transfusion-transmitted malaria in Malawi. This finding underscores the urgent need to strengthen malaria screening protocols and diagnostic capacity within blood transfusion services.
Background
Blood transfusion therapy is a common and essential medical intervention for treating anemia and other life-threatening conditions, involving the transfer of blood or its components from a donor to a recipient (Goodnough and Panigrahi, 2017). Despite its clinical importance, blood transfusion carries inherent risks of transmitting infections, including viral, bacterial, and parasitic pathogens such as Plasmodium species (Buerger and Jain, 2025).
Transfusion-transmitted malaria (TTM) remains a significant yet often underrecognized public health concern in malaria-endemic regions (Ahmadpour et al., 2019). It occurs when malaria parasites are introduced into a recipient through infected donor blood. The condition can have severe clinical consequences, particularly for immunocompromised individuals, pregnant women, and children (Owusu-Ofori et al., 2016; Sumbele et al., 2015). Although the true burden of TTM in sub-Saharan Africa is not well established due to limited post-transfusion surveillance, reports indicate that the risk may be substantial in areas where asymptomatic malaria parasitemia is common among blood donors (De Moraes et al., 2018; Loua et al., 2024). Currently, routine post-transfusion malaria monitoring is not systematically implemented in Malawi, which may result in underreporting of transfusion-related malaria cases.
Malaria-induced anemia further compounds the risk associated with transfusion needs, particularly in vulnerable populations such as pregnant women and children under five (Abdullah and Karunamoorthi, 2016; Iheonu et al., 2018). According to the World Health Organization (WHO), there were an estimated 249 million malaria cases globally in 2022 (WHO, 2023), highlighting the ongoing burden of the disease and the urgent need for improved preventive and diagnostic measures in endemic areas. In Malawi, malaria incidence is estimated at 393 cases per 1,000 population annually, disproportionately affecting young children who account for most of the infections (Gumbo et al., 2022).
Human malaria is caused by five Plasmodium species—P. falciparum, P. malariae, P. ovale, P. vivax, and P. knowlesi—with P. falciparum being the most prevalent and severe (Zekar and Sharman, 2025). However, non-falciparum species also contribute significantly to the disease burden and are increasingly recognized as important causes of both morbidity and mortality (Ranjbar and Tegegn Woldemariam, 2024).In Malawi, non-falciparum infections have been reported at a prevalence of approximately 11.7%, though regional variations exist (Gumbo et al., 2022).
Beyond symptomatic infections, subclinical or asymptomatic malaria parasitemia constitutes a hidden reservoir that perpetuates transmission and poses challenges to malaria elimination efforts (De Moraes et al., 2018). Asymptomatic infections in blood donors are particularly concerning because they may evade standard screening methods and lead to TTM.
Therefore, this study aimed to assess the prevalence of asymptomatic malaria parasitemia among blood donors in Malawi and identify associated factors to inform preventive strategies and strengthen malaria screening protocols within blood transfusion services.
Methods
Study design and setting
This study employed a cross-sectional research design conducted at the Lilongwe Branch of the Malawi Blood Transfusion Service, a central facility for blood collection and distribution in Malawi’s central region. The study population comprised voluntary asymptomatic blood donors who presented themselves at designated blood donation centers.
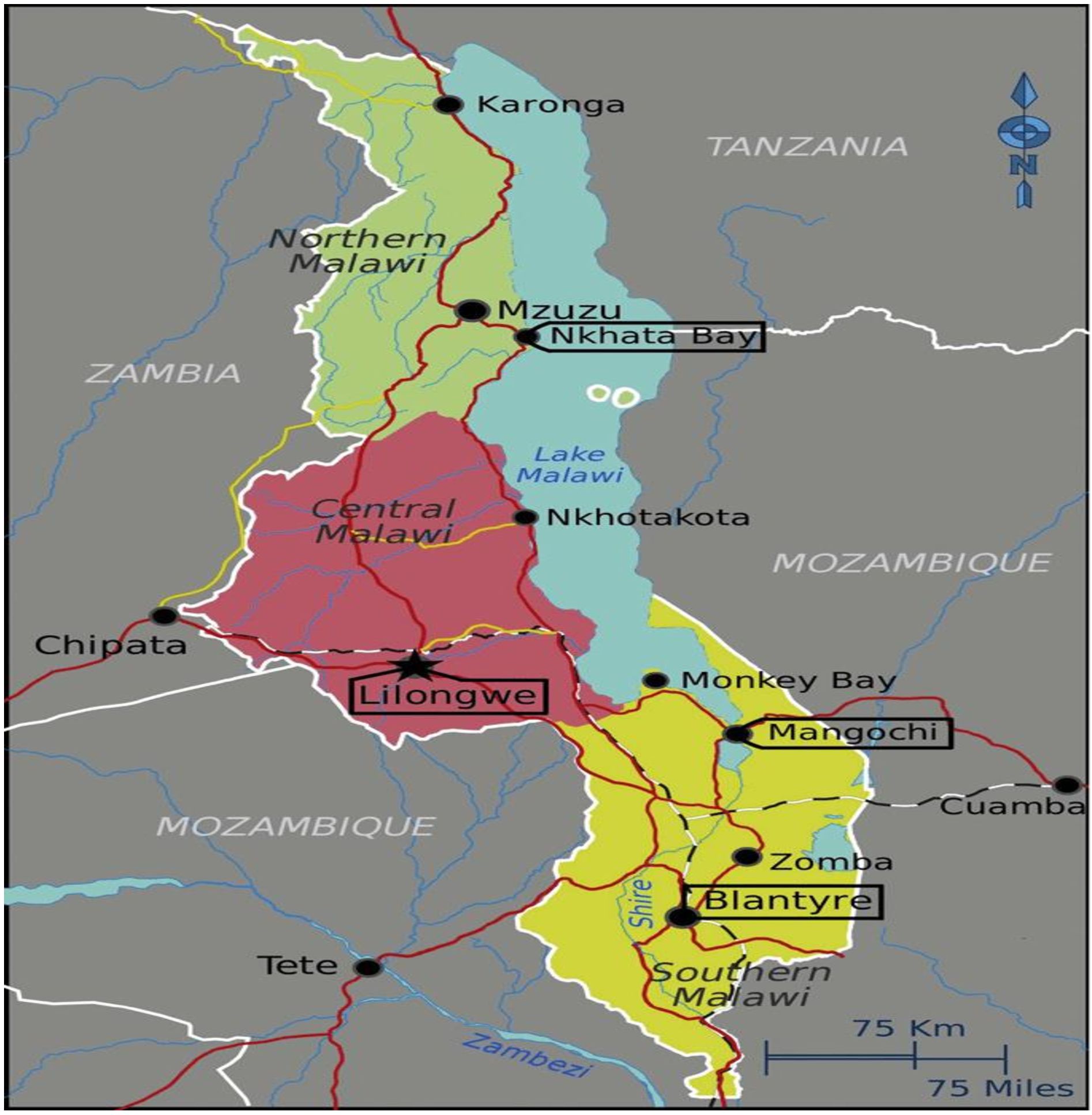
The map of Malawi in Figure 1, highlights the location of Lilongwe District in the Central Region.
Eligibility criteria
Eligible participants included healthy blood donors of all genders aged 18 to 65 years. Individuals were excluded if they had a documented history of symptomatic malaria infection or had used antimalarial medication within six weeks prior to the study.
Sample selection
A systematic sampling approach was used to select 377 participants for the study. The sample size was calculated based on an expected malaria prevalence of 42.8% among Malawian blood donors, as reported in the most recent estimates by Kinge et al. (2019). This calculation assumed an estimated population size of 1,000 donors visiting the blood bank during the study period. Starting from a randomly selected donor, every third consecutive donor was recruited into the study.
Blood sample collection and processing
After obtaining informed consent, eligible blood donors were escorted to the donation room, where qualified nurses from the Malawi Blood Transfusion Service (MBTS) performed blood collection. Approximately one-hour post-donation, the collected blood units were transferred to the blood bank for processing. The research team then accessed these units to obtain samples for malaria screening and further analysis.
From each donor unit, a blood sample of 200–500 μL was collected for malaria testing. These samples were allocated for both rapid diagnostic testing (RDT) and microscopic examination.
Screening methodology
Screening for malaria involved both RDTs and microscopic analysis. For each donor sample, both methods were conducted to ensure comprehensive and accurate detection of malaria parasites.
Rapid diagnostic tests
RDT screening was performed using the ParaHIT f ver. 1.0 Malaria Ag Pf test kit, which detects histidine-rich protein 2 (HRP-2), a specific antigen for P. falciparum. The tests were carried out on-site at the MBTS laboratory, following the manufacturer’s instructions.
Microscopic analysis
For microscopic examination, both thick and thin blood smears were prepared from each donor sample. Thick smears were heat-fixed, while thin smears were fixed using 99% absolute alcohol. The prepared slides were transported to Kamuzu Central Hospital (KCH) laboratory for staining.
At KCH, the slides were stained with 3% Giemsa solution for 45 minutes. Microscopic examination was conducted independently by two experienced biomedical scientists. In cases of discordant results, a third biomedical scientist reviewed the slides to resolve discrepancies.
Parasite density calculation
Parasite density was determined by counting the number of parasites relative to a predetermined number of white blood cells. An average WBC count of 8,000/μL was used as the standard reference. The parasite density was calculated using the following formula:
Identifying factors associated with malaria transmission among blood donors
Study participants were requested to complete a structured questionnaire developed specifically for this research. An English version has been provided as a Supplementary File (Supplementary File 1). This questionnaire was used to collect information about geographic location, socioeconomic factors, and travel history to malaria-endemic districts.
Data analysis
Data were analyzed using R software, version 4.3.1. A p-value of less than 0.05 was considered statistically significant. Both descriptive and inferential statistics were employed to address the study objectives.
Prevalence estimation
The prevalence of asymptomatic malaria parasitemia was calculated as the proportion of donors testing positive (by RDT or microscopy) relative to the total number tested, expressed as a percentage. Descriptive statistics, including frequencies and percentages, were used to summarize prevalence across demographic characteristics such as age, sex, occupation, and residence.
Comparisons of prevalence between categorical variables were assessed using the Chi-square (χ²) test; where expected cell counts were <5, the Fisher’s Exact test was applied.
Distribution of Plasmodium species
The distribution of Plasmodium species identified by microscopy was summarized using frequency tables. Parasite densities were described using medians and interquartile ranges (IQRs), given their non-normal distribution.
Associations between Plasmodium species and demographic variables were analyzed using Chi-square or Fisher’s Exact tests, as appropriate. Differences in parasite density across species were examined using the Kruskal–Wallis test.
Factors associated with asymptomatic malaria parasitemia
Bivariate analyses were first performed to identify potential predictors of asymptomatic malaria parasitemia, including sex, age group, occupation, residence, blood group, repellent use, and travel history.
Variables with a p-value <0.20 in bivariate analysis were subsequently entered into a multivariate binary logistic regression model to determine independent predictors of infection. Results were expressed as adjusted odds ratios (AORs) with 95% confidence intervals (CIs). Model adequacy was assessed using the Hosmer–Lemeshow goodness-of-fit test.
Results
General characteristic
The study population consisted of 377 blood donors, with a majority being male (82.2%). Students made up the largest occupational group (69.8%), while only 6.6% were farmers and 5.3% were health workers. Most participants (80.4%) resided in rural areas. Regarding malaria prevention, repellent use was low, with only 0.3% reporting consistent use, while 69.8% used it occasionally. Travel history outside malaria-endemic areas was uncommon, with only 5.1% reporting recent travel.
The distribution of blood groups among the participants in this study showed a predominance of the O+ group, representing 37.7% of the participants, followed closely by B+ at 36.1%. The least common blood group was B-, with a frequency of 0.3%. Table 1 summarizes the overall proportions of selected socio-demographic factors and blood group diversity of the study population.
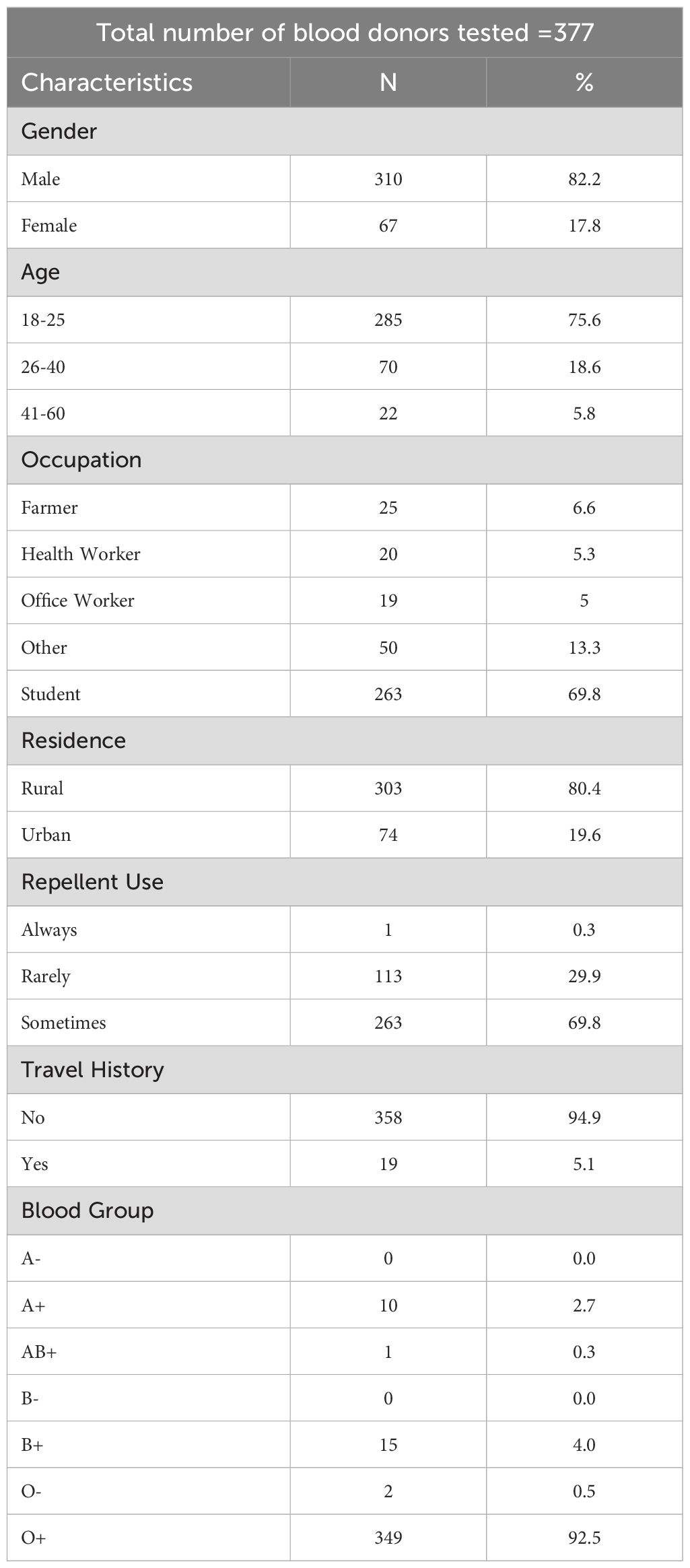
Table 1. Distribution of selected socio-demographic factors and blood group diversity in the study population.
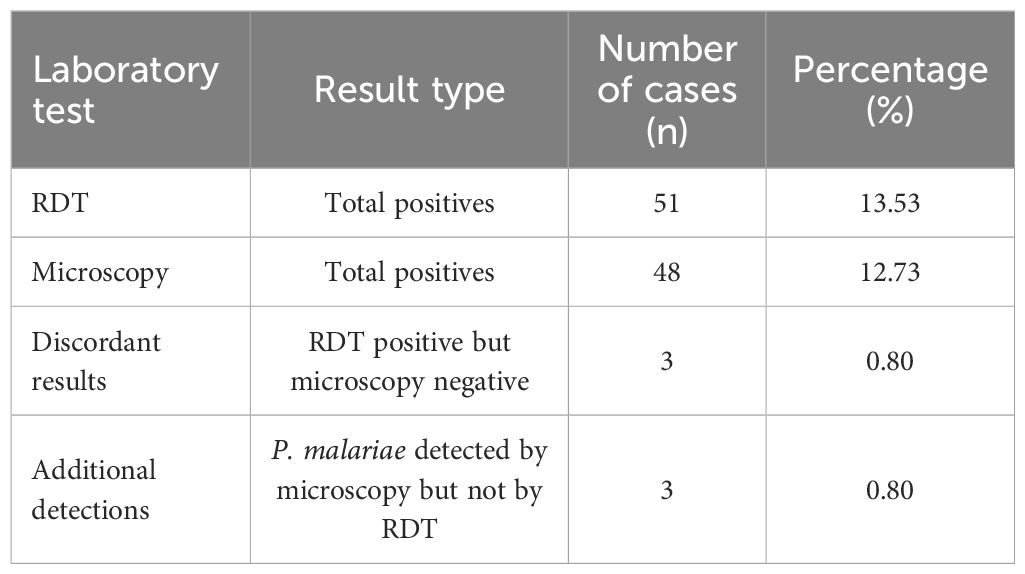
Summary of positive results by laboratory test method
The summary in Table 2 presents the total malaria-positive cases identified by Rapid Diagnostic Test (RDT) and microscopy among 377 blood donors. Overall, 51 (13.53%) samples tested positive by RDT, while 48 (12.73%) were confirmed by microscopy. Of these, three cases were RDT-positive but microscopy-negative, whereas an additional three P. malariae infections were detected exclusively by microscopy. For the overall prevalence estimate, 48 microscopy-confirmed P. falciparum infections were considered, with inclusion of the P. malariae cases bringing the total to 51 confirmed infections across all malaria species.
Prevalence rate for asymptomatic malaria parasitemia among blood donors
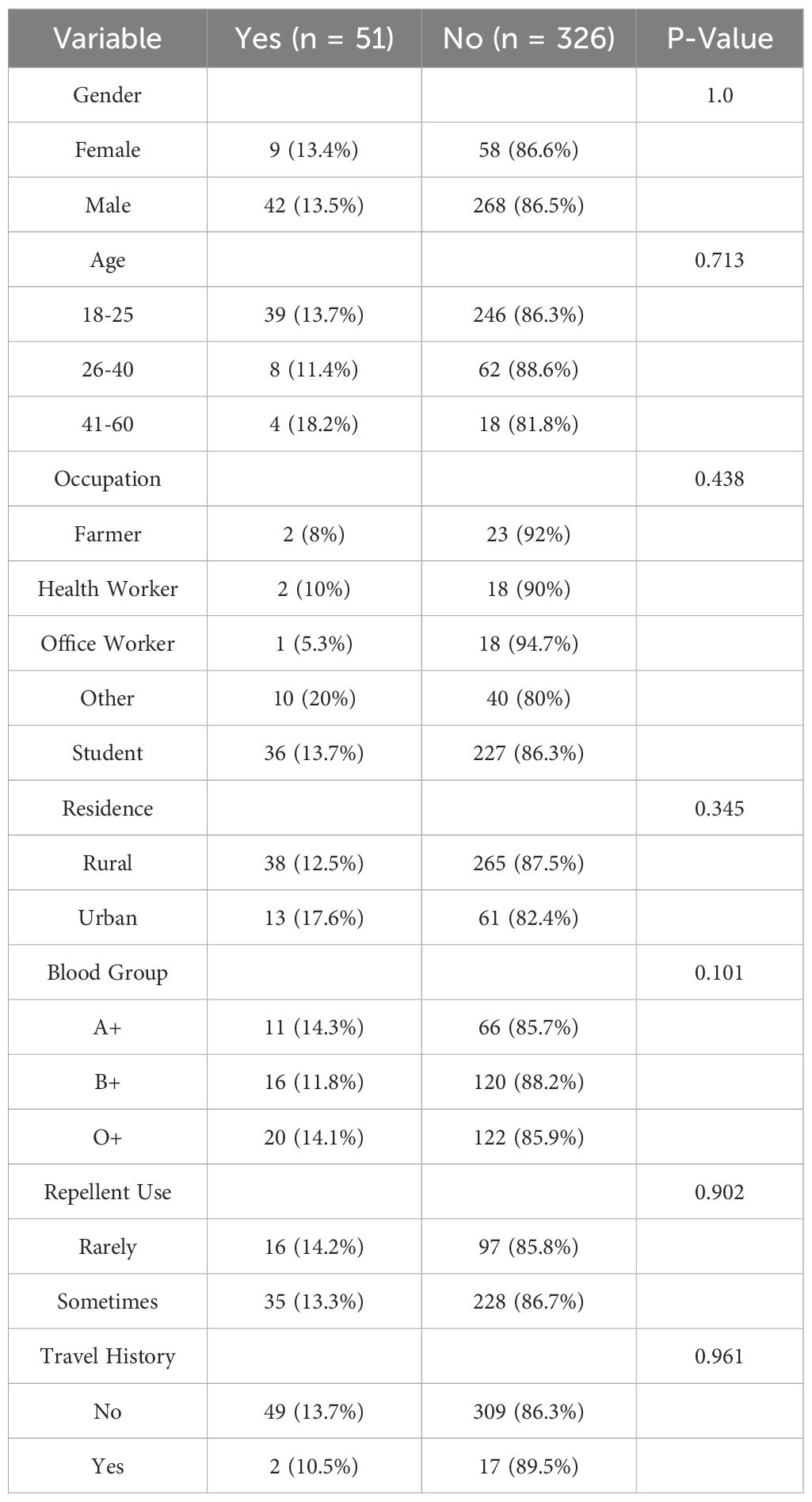
Of the 377 samples, 51 (13.5%) tested positive for malaria. Among the females, 9 (13.4%) tested positive and 58 (86.6%) tested negative. Among the males, 42 (13.5%) tested positive and 268 (86.5%) tested negative for malaria. The highest prevalence was observed in the 41–60 age group (18.2%) and among individuals classified as “Other” in terms of occupation (20%). Rural residents had a prevalence of 12.5%, while urban residents had a prevalence of 17.6%. These demographic characteristics are presented in Table 3.
Determining malaria species associated with asymptomatic malaria parasitemia
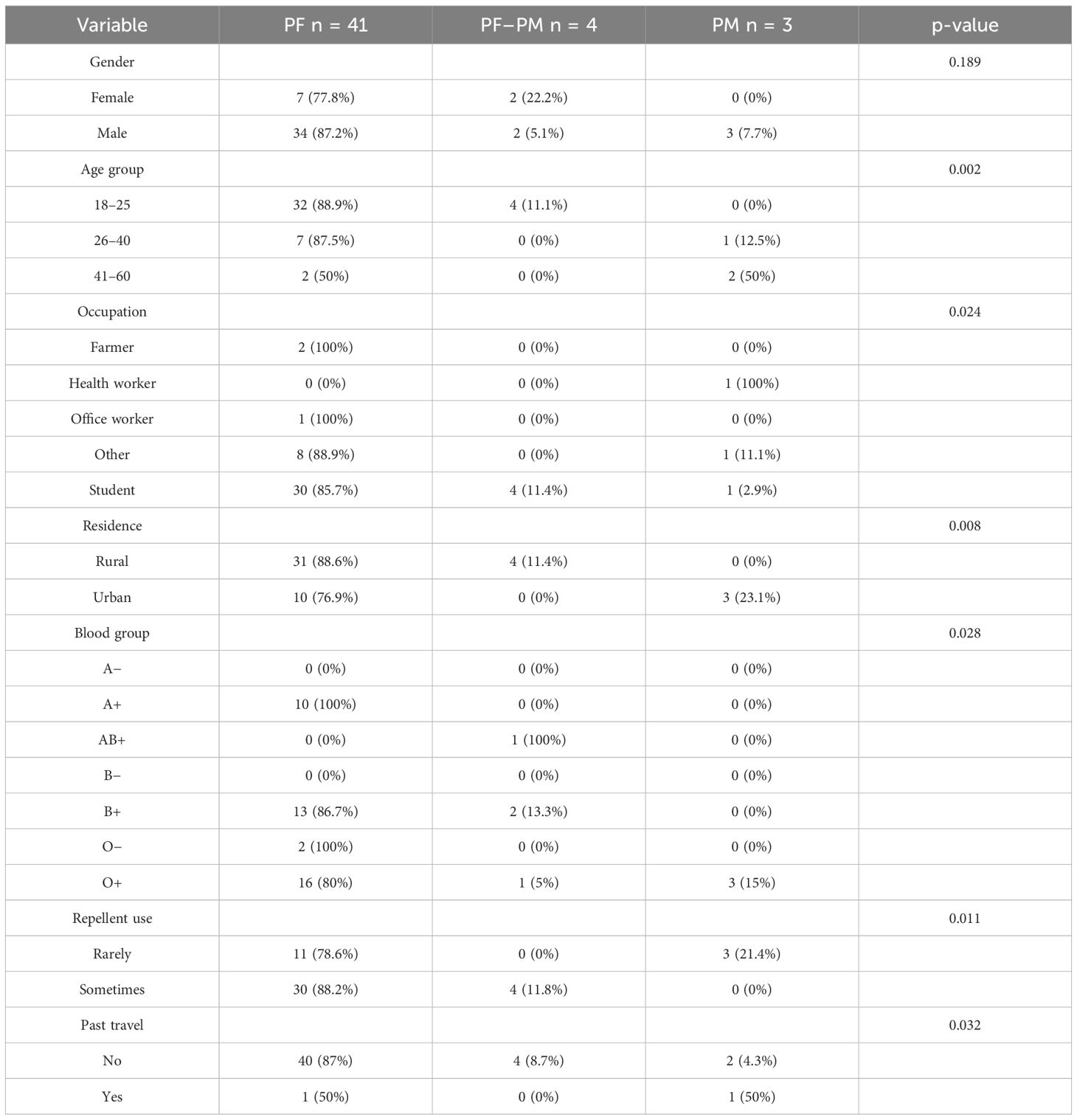
Among the 48 asymptomatic blood donors, P.falciparum dominated with 85.4% of cases, while mixed infections of P. falciparum and P. malariae accounted for 8.3%, and P.malariae alone was found in 6.3% of cases. Mixed infections were more frequent in females (22.2%) than males (5.1%). The age group 41–60 showed a notable presence of P.malariae, with 50% testing positive. Rural areas had a higher prevalence of P.falciparam (88.6%), while urban areas showed a notable 23.1% for P.malariae cases. The demographic breakdowns and corresponding p-values are outlined in Table 4.
Parasite density by species
Figure 2 presents a box plot illustrating the distribution of parasite density among different malaria species. P. falciparum infections exhibited the highest median parasite density at approximately 3,000 parasites/μL (IQR: 1,500–4,800; range: 200–8,000). Mixed P. falciparum–P. malariae infections showed moderate densities, with a median of about 1,800 parasites/μL (IQR: 1,200–2,600; range: 500–3,000). P. malariae infections recorded the lowest median density at around 1,000 parasites/μL (IQR: 400–2,600; range: 0–4,000).
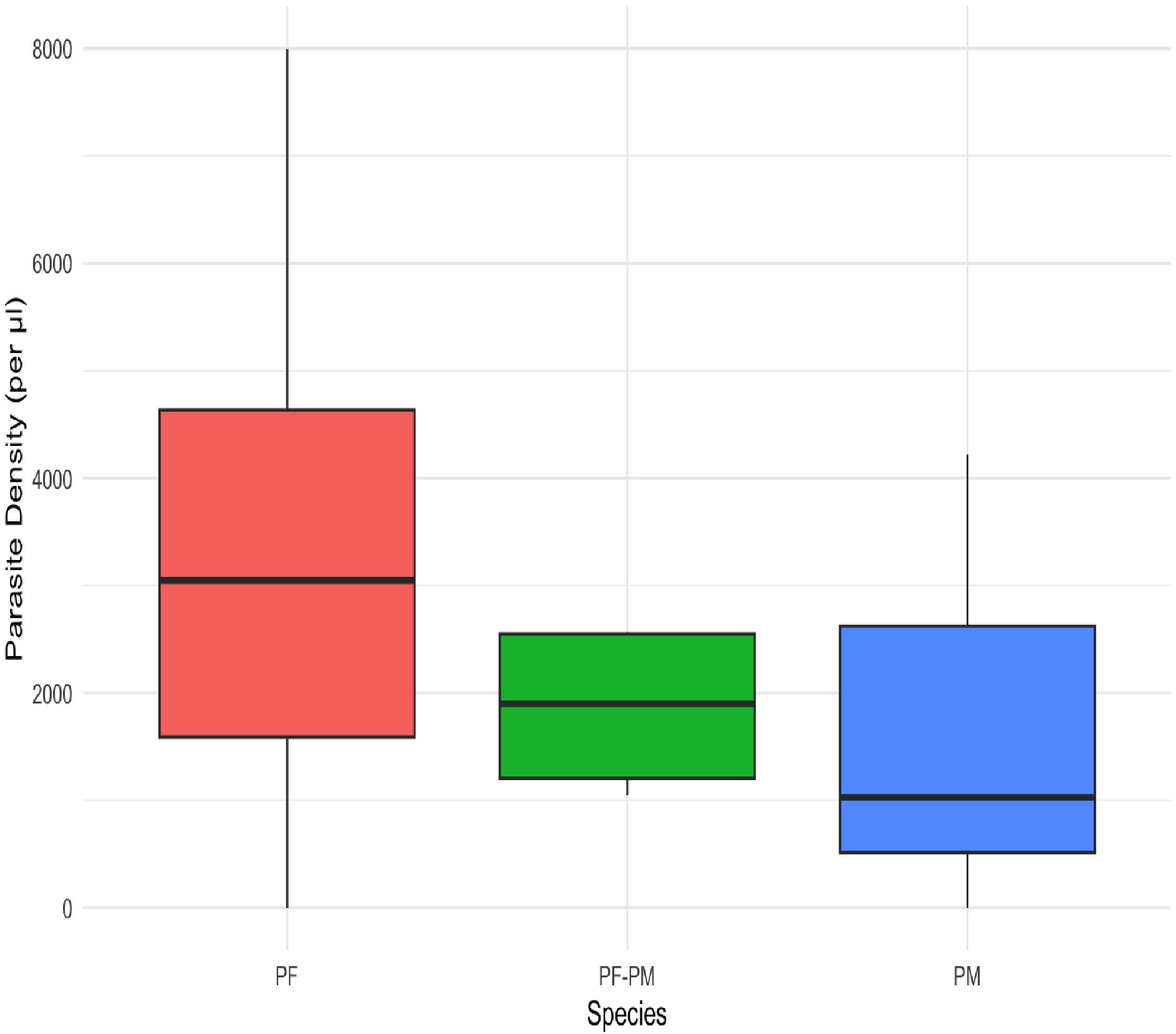
Figure 2. Malaria Parasite Density Distribution Among Asymptomatic Cases. KEY: P. falciparum (Pf), P. malariae (Pm), Mixed infections of P. malariae and P. falciparum (Pf-Pm).
Exploring the factors associated with asymptomatic malaria parasitemia
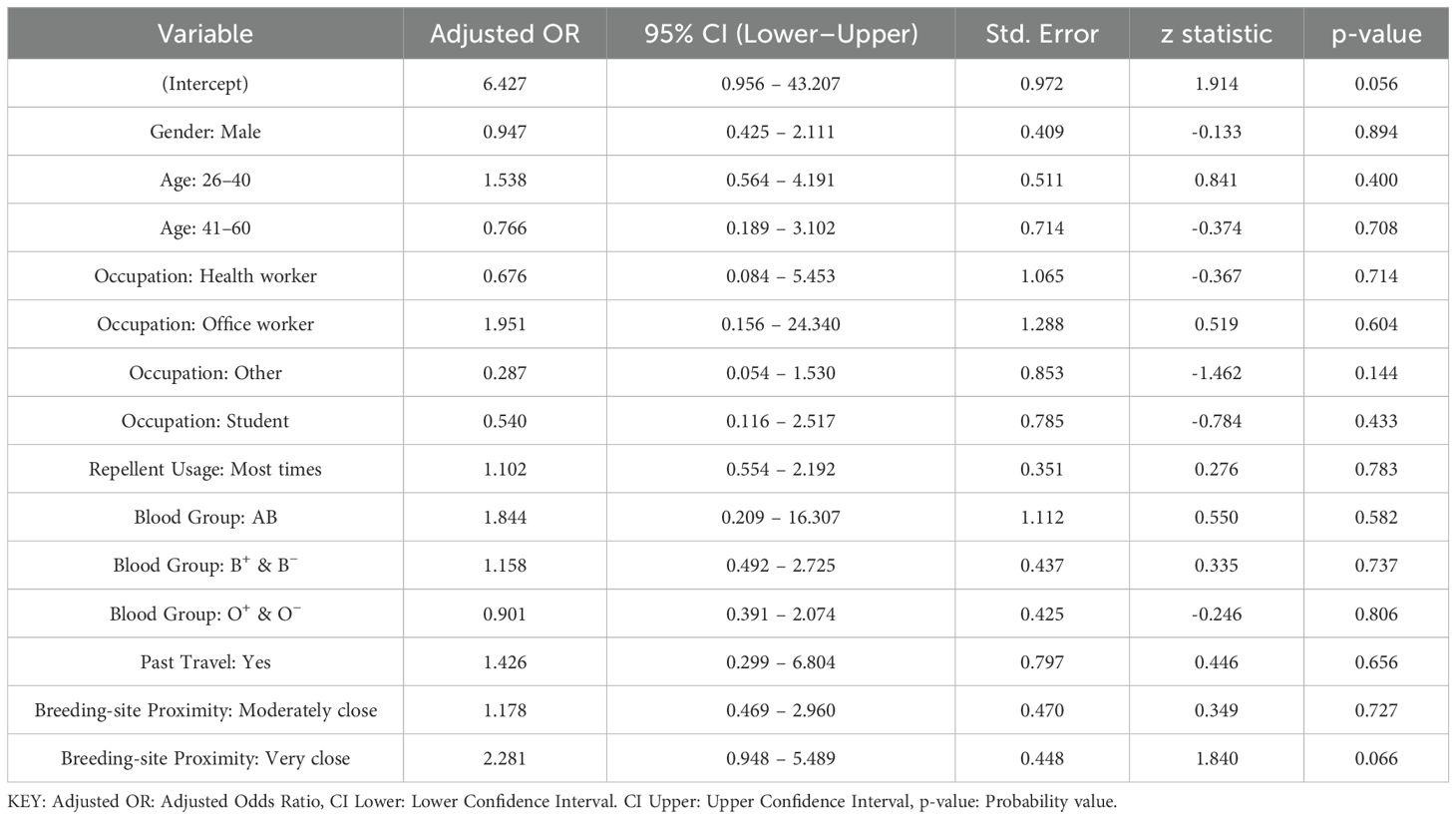
A multivariate logistic regression model is presented in Table 5, predicting asymptomatic malaria parasitemia based on the outcome variable of a positive RDT result. The significance level was set at 0.05, and no significant associations were observed. The analysis revealed no statistically significant associations across all variables. Gender did not significantly affect the likelihood of asymptomatic malaria parasitemia, with males having an adjusted odds ratio (OR) of 0.95 (95% CI: 0.43 - 2.11, p=0.894) compared to females.
Age groups similarly demonstrated no significant differences. The 26–40 years group had an OR of 1.54 (95% CI: 0.56 - 4.19, p=0.400), and the 41–60 years group had an OR of 0.77 (95% CI: 0.19 - 3.10, p=0.708), both compared to the 18–25 years group.
Other variables, including occupation, repellent use, blood group, and past travel history, also showed no significant associations with asymptomatic malaria parasitemia.
Discussion
This study revealed a 13.53% prevalence of asymptomatic malaria parasitemia among blood donors at the Lilongwe Blood Transfusion Center, underscoring a significant but often underestimated public health concern. This prevalence closely mirrors findings from Nigeria, where Olopade et al. (2024) reported an 11.9% infection rate among voluntary donors, indicating that asymptomatic carriage among blood donors is a persistent challenge across sub-Saharan Africa. The consistency between these studies highlights the ongoing risk that asymptomatic infections pose to blood safety and transfusion services in endemic settings.
Comparable research from other regions has reported variable prevalence estimates, reflecting differences in malaria transmission intensity, diagnostic methods, and donor screening policies. For instance, a study in Ghana documented a 2.5% prevalence of asymptomatic malaria parasitemia among blood donors (Tetteh et al., 2023), whereas a systematic review by Ahmadpour et al. (2019) found median prevalence rates of 10.54%, 5.36%, and 0.38% when assessed by microscopy, PCR, and rapid diagnostic tests (RDTs), respectively. Collectively, these findings reaffirm that asymptomatic malaria parasitemia remains a widespread issue, highlighting the need for enhanced surveillance and targeted interventions to mitigate transfusion-transmitted malaria (TTM) and interrupt silent parasite reservoirs within donor populations.
The species distribution observed in this study was dominated by P. falciparum (85.4%), followed by P. malariae (6.3%) and mixed infections (8.3%). The predominance of P. falciparum aligns with national and regional malaria epidemiology but also draws attention to the contribution of non-falciparum species, which are frequently underreported and underprioritized in elimination programs. Previous studies have shown that non-falciparum infections range from 1.4% among children in Zambia (Sitali et al., 2015) to 11.7% among adults in Malawi (Gumbo et al., 2022), emphasizing the need to include these species in diagnostic, treatment, and surveillance frameworks.
The detection of mixed Plasmodium infections in 8.3% of cases further highlights the complex epidemiological landscape of malaria transmission. Similar patterns have been reported in Tanzania, where co-infections with P. falciparum and P. malariae were detected in 23.6% of clinical cases (Popkin-Hall et al., 2023), and in Kenya’s Lake Victoria basin, where PCR analysis identified 23.5% co-infections among children aged 3–15 years (Omondi et al., 2024). These observations are consistent with the systematic review and meta-analysis by Kotepui et al. (2020), which underscored the frequent yet overlooked occurrence of mixed-species malaria infections.
Mixed Plasmodium infections can influence clinical outcomes, immune responses, and treatment efficacy (Kotepui et al., 2020; Tang et al., 2020). The coexistence of multiple species may modify disease progression and complicate therapeutic responses due to differences in drug sensitivities (Van Der Pluijm et al., 2021). A systematic review by Mahittikorn et al. (2021) reported that mixed infections were associated with higher recurrence rates within 28 days following artemisinin-based combination therapy, underscoring the importance of accurate species identification to ensure effective case management.
Analysis of parasite density revealed considerable variation among species, with P. falciparum exhibiting the highest median density. This observation is consistent with findings from Uganda, where Agaba et al. (2022) reported densities exceeding 1,500 parasites/μL among asymptomatic donors, and from Burkina Faso, where Kambou et al. (2024) documented densities ranging from 24 to 42,645 parasites/μL. Such wide variability reflects the heterogeneity of parasite-host interactions in asymptomatic infections.
Differences in parasite density can be influenced by host immunity, parasite biology, and local transmission intensity (Oyibo et al., 2023). In high-transmission settings, repeated exposure induces partial immunity (Bediako et al., 2019), enabling individuals to tolerate substantial parasitemia without clinical symptoms. This immunological adaptation complicates detection and underscores the need for sensitive diagnostic tools capable of identifying low-density infections—especially critical for ensuring transfusion safety and strengthening surveillance systems aimed at malaria elimination. Undetected low-density parasitemia poses a persistent risk of TTM (Buerger and Jain, 2025) and may sustain residual transmission despite apparent clinical control.
No statistically significant associations were identified between malaria infection and the assessed demographic or clinical variables (all p > 0.05). Nonetheless, emerging trends suggested that age and blood group may modulate infection risk. Donors aged 18–24 years exhibited relatively higher odds of infection, consistent with literature suggesting increased exposure among younger adults due to mobility, social behaviors, and incomplete immunity (Gui et al., 2012; Zhou et al., 2021). Although these trends were not statistically significant, they highlight the need to incorporate age-specific risk considerations into donor screening and malaria control strategies.
The exploration of ABO blood group distribution among infected donors revealed that P. falciparum was more frequent among individuals with blood group A+. This observation is consistent with prior hypotheses linking ABO antigens to malaria susceptibility (Nain and Sharma, 2022). Earlier studies have suggested that blood group O confers partial protection against severe malaria (Ai et al., 2022; Panda et al., 2011), though underlying mechanisms remain unclear. Given the limited number of A+ participants in this dataset, these findings should be interpreted with caution, and larger, multi-center studies are warranted to elucidate the role of blood group phenotypes in malaria risk and disease expression.
This study demonstrated a considerable prevalence of asymptomatic malaria parasitemia among blood donors at the Lilongwe Centre of the Malawi Blood Transfusion Service, with P. falciparum accounting for the majority of infections. The presence of asymptomatic parasitemia within a healthy donor population poses a potential risk of transfusion-transmitted malaria, especially in the absence of routine screening protocols. These findings provide critical evidence that malaria transmission through blood transfusion may be an under-recognized contributor to the overall malaria burden in endemic areas such as Malawi.
The implications of these results are far-reaching. First, they underscore the urgent need to strengthen malaria screening protocols within national blood transfusion services to ensure the safety of blood supplies. Integrating malaria testing—particularly using sensitive diagnostic methods—into standard donor screening could substantially reduce the risk of transmission to vulnerable recipients, such as children and pregnant women. Second, the persistence of asymptomatic malaria parasitemia despite ongoing control efforts highlights the importance of including blood safety considerations in broader malaria elimination strategies. Strengthening diagnostic capacity, implementing targeted surveillance among donors, and aligning blood safety programs with national malaria control initiatives will be essential to mitigate transfusion-related risks and support Malawi’s progress toward malaria elimination.
Limitations of the study
This study has several limitations that warrant consideration. First, the use of microscopy and HRP2-based rapid diagnostic tests (RDTs) may have limited the detection of low-density or mixed asymptomatic infections. Although microscopy remains valuable for species identification, its sensitivity declines at low parasitemia levels and is highly dependent on the skill of the examiner. Consequently, the true prevalence of asymptomatic malaria parasitemia among blood donors may have been underestimated. Future studies should incorporate more sensitive molecular tools, such as PCR, to improve the detection of submicroscopic and mixed infections and provide a more accurate estimation of malaria burden.
Additionally, the reliance on HRP2-based RDTs presents another limitation, as Plasmodium falciparum parasites with hrp2 gene deletions are increasingly reported across sub-Saharan Africa. The use of multi-antigen RDTs, such as those combining HRP2 and pLDH targets, would enhance diagnostic reliability and yield more representative prevalence estimates.
Finally, the study population consisted solely of voluntary blood donors, which may have introduced selection bias, as this group may not fully represent the general population. The observed lack of statistical association between gender and malaria infection could also be attributed to the marked imbalance between male and female participants.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The studies involving humans were approved by UNZABREC (University of Zambia Biomedical Research Ethics Committee) (Reference #4813-2023) and NHSREC (National Health Sciences Research Ethics Committee) (Protocol #24/01/4334). The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
Author contributions
CB: Investigation, Data curation, Methodology, Conceptualization, Visualization, Resources, Funding acquisition, Formal analysis, Project administration, Writing – original draft. NK: Writing – review & editing. BM: Writing – review & editing. LS: Supervision, Conceptualization, Project administration, Validation, Investigation, Writing – review & editing, Methodology, Visualization, Software, Formal analysis, Data curation, Funding acquisition, Resources. JS: Writing – review & editing, Data curation, Supervision. EK: Writing – review & editing, Formal analysis, Data curation, Visualization. A-GT: Data curation, Writing – review & editing, Formal analysis.
Funding
The author(s) declare that no financial support was received for the research, and/or publication of this article.
Acknowledgments
The authors would like to acknowledge the voluntary blood donors who participated in this study. We also extend our gratitude to the staff at the Lilongwe Branch of the Malawi Blood Transfusion Service for their support during data collection.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmala.2025.1687355/full#supplementary-material
Abbreviations
EDTA, Ethylenediaminetetraacetic acid; GPPF, Graduate Proposal Presentation Forum; HB, Hemoglobin; HIV, Human Immunodeficiency Virus; LDH, Lactate Dehydrogenase; MBTS, Malawi Blood Transfusion Services; MDHS, Malawi Demographic and Health Survey; MWK, Malawian Kwacha; MRDT, Malaria Rapid Diagnostic Test; NAT, Nucleic Acid Amplification Technology; NHSRC, National Health Science Research Committee; PCR, Polymerase Chain Reaction; RDT, Rapid Diagnostic Test; SDG, Sustainable Development Goals; SPSS, Statistical Package for the Social Sciences; TTI, Transfusion Transmitted Infections; TTM, Transfusion Transmitted Malaria; UNZABREC, University of Zambia Biomedical Research Ethics Committee; USA, The United States of America; WBC, White Blood Cells.
References
Abdullah S. and Karunamoorthi K. (2016). Malaria and blood transfusion: major issues of blood safety in malaria-endemic countries and strategies for mitigating the risk of Plasmodium parasites. Parasitol. Res. 115, 35–47. doi: 10.1007/s00436-015-4808-1
Agaba B. B., Rugera S. P., Mpirirwe R., Atekat M., Okubal S., Masereka K., et al. (2022). Asymptomatic malaria infection, associated factors and accuracy of diagnostic tests in a historically high transmission setting in Northern Uganda. Malaria J. 21, 392. doi: 10.1186/s12936-022-04421-1
Ahmadpour E., Foroutan-Rad M., Majidiani H., Moghaddam S. M., Hatam-Nahavandi K., Hosseini S.-A., et al. (2019). Transfusion-transmitted malaria: A systematic review and meta-analysis. Open Forum Infect. Dis. 6, ofz283. doi: 10.1093/ofid/ofz283
Ai L., Li J., Wang W., and Li Y. (2022). ABO blood group and risk of malaria during pregnancy: a systematic review and meta-analysis. Epidemiol. Infection 150, e25. doi: 10.1017/S095026882200005X
Bediako Y., Adams R., Reid A. J., Valletta J. J., Ndungu F. M., Sodenkamp J., et al. (2019). Repeated clinical malaria episodes are associated with modification of the immune system in children. BMC Med. 17, 60. doi: 10.1186/s12916-019-1292-y
Buerger C. S. and Jain H. (2025). “Infectious complications of blood transfusion,” in StatPearls (StatPearls Publishing, Treasure Island (FL).
De Moraes C. M., Wanjiku C., Stanczyk N. M., Pulido H., Sims J. W., Betz H. S., et al. (2018). Volatile biomarkers of symptomatic and asymptomatic malaria infection in humans. Proc. Natl. Acad. Sci. 115, 5780–5785. doi: 10.1073/pnas.1801512115
Goodnough L. T. and Panigrahi A. K. (2017). Blood transfusion therapy. Med. Clin. North Am. 101, 431–447. doi: 10.1016/j.mcna.2016.09.012
Gui J., Mustachio L. M., Su D.-M., and Craig R. W. (2012). Thymus size and age-related thymic involution: early programming, sexual dimorphism, progenitors and stroma. Aging Dis. 3 (3), 280–290.
Gumbo A., Topazian H. M., Mwanza A., Mitchell C. L., Puerto-Meredith S., Njiko R., et al. (2022). Occurrence and distribution of nonfalciparum malaria parasite species among adolescents and adults in Malawi. J. Infect. Dis. 225, 257–268. doi: 10.1093/infdis/jiab353
Iheonu F. O., Fajolu I. B., Ezeaka C. V., and Oyibo W. A. (2018). Transfusional malaria in the neonatal period in Lagos, South-West Nigeria. PloS One 13, e0195319. doi: 10.1371/journal.pone.0195319
Kambou S. A. E., Millogo K. S., Sondo P., Kabore B., Kouevi A. F. C., Bouda I., et al. (2024). Prevalence of asymptomatic parasitaemia among household members of children under seasonal malaria chemoprevention coverage and comparison of the performance of standard rapid diagnostic tests versus ultrasensitive RDT for the detection of asymptomatic parasitaemia in Nanoro, Burkina Faso. Parasitol. Res. 123, 383. doi: 10.1007/s00436-024-08380-1
Kinge C. W., Kagura J., M’baya B., Njolomole S., and Chasela C. (2019). Incidence and determinants of transfusion-transmissible infections in voluntary blood donors in Malawi 2005-201. doi: 10.21203/rs.2.18048/v1
Kotepui M., Kotepui K. U., De Jesus Milanez G., and Masangkay F. R. (2020). Plasmodium spp. mixed infection leading to severe malaria: a systematic review and meta-analysis. Sci. Rep. 10, 11068. doi: 10.1038/s41598-020-68082-3
Loua O.-O., Cissoko Y., Chantal Loua S., Konaté I., Issa H. H., Maïga B., et al. (2024). Risk of transfusion-associated malaria in sub-Saharan Africa: The case of Mali. Transfusion Med. 34, 527–536. doi: 10.1111/tme.13101
Mahittikorn A., Masangkay F. R., Kotepui K. U., Milanez G. D. J., and Kotepui M. (2021). The high risk of malarial recurrence in patients with Plasmodium-mixed infection after treatment with antimalarial drugs: a systematic review and meta-analysis. Parasites Vectors 14, 280. doi: 10.1186/s13071-021-04792-5
Nain M. and Sharma A. (2022). Linkages between blood groups and malaria susceptibility. J. Vector Borne Dis. 59, 193. doi: 10.4103/0972-9062.345177
Olopade B. O., Ajeigbe A. K., Owojuyigbe T. O., Ajani A. A., and Fayomi O. D. (2024). Frequency of blood donation and malaria occurrence among blood donors in a tertiary hospital, Nigeria. Ibom Med. J. 17, 103–107. doi: 10.61386/imj.v17i1.385
Omondi P., Musyoka B., Okai T., Kongere J., Kagaya W., Chan C. W., et al. (2024). Non-random distribution of Plasmodium Species infections and associated clinical features in children in the Lake Victoria region, Kenya 2012–2018. Trop. Med. Health 52, 52. doi: 10.1186/s41182-024-00622-3
Owusu-Ofori A., Gadzo D., and Bates I. (2016). Transfusion-transmitted malaria: donor prevalence of parasitaemia and a survey of healthcare workers knowledge and practices in a district hospital in Ghana. Malar J. 15, 234. doi: 10.1186/s12936-016-1289-3
Oyibo W., Latham V., Oladipo O., Ntadom G., Uhomoibhi P., Ogbulafor N., et al. (2023). Malaria parasite density and detailed qualitative microscopy enhances large-scale profiling of infection endemicity in Nigeria. Sci. Rep. 13, 1599. doi: 10.1038/s41598-023-27535-1
Panda A. K., Panda S. K., Sahu A. N., Tripathy R., Ravindran B., and Das B. K. (2011). Association of ABO blood group with severe falciparum malaria in adults: case control study and meta-analysis. Malar J. 10, 309. doi: 10.1186/1475-2875-10-309
Popkin-Hall Z. R., Seth M. D., Madebe R. A., Budodo R., Bakari C., Francis F., et al. (2023). Malaria species positivity rates among symptomatic individuals across regions of differing transmission intensities in mainland Tanzania. J. Infect. Dis. 229, 959. doi: 10.1093/infdis/jiad522
Ranjbar M. and Tegegn Woldemariam Y. (2024). Non-falciparum malaria infections in Uganda, does it matter? A review of the published literature. Malar J. 23, 207. doi: 10.1186/s12936-024-05023-9
Sitali L., Chipeta J., Miller J. M., Moonga H. B., Kumar N., Moss W. J., et al. (2015). Patterns of mixed Plasmodium species infections among children six years and under in selected malaria hyper-endemic communities of Zambia: population-based survey observations. BMC Infect. Dis. 15, 204. doi: 10.1186/s12879-015-0935-7
Sumbele I. U. N., Kimbi H. K., Ndamukong-Nyanga J. L., Nweboh M., Anchang-Kimbi J. K., Lum E., et al. (2015). Malarial anaemia and anaemia severity in apparently healthy primary school children in urban and rural settings in the Mount Cameroon area: cross sectional survey. PloS One 10, e0123549. doi: 10.1371/journal.pone.0123549
Tang J., Templeton T. J., Cao J., and Culleton R. (2020). The Consequences of Mixed-Species Malaria Parasite Co-Infections in Mice and Mosquitoes for Disease Severity, Parasite Fitness, and Transmission Success. Frontiers in Immunology 10, 3072. doi: 10.3389/fimmu.2019.03072
Tetteh J. A., Djissem P. E., and Manyeh A. K. (2023). Prevalence, trends and associated factors of malaria in the Shai-Osudoku District Hospital, Ghana. Malaria J. 22, 131. doi: 10.1186/s12936-023-04561-y
Van Der Pluijm R. W., Amaratunga C., Dhorda M., and Dondorp A. M. (2021). Triple artemisinin-based combination therapies for malaria – A new paradigm? Trends Parasitol. 37, 15–24. doi: 10.1016/j.pt.2020.09.011
World malaria report. (2023). Available online at: https://www.who.int/teams/globalmalaria-programme/reports/world-malaria-report-2023 (Accessed February 20, 25).
Zekar L. and Sharman T. (2025). “Plasmodium falciparum malaria,” in StatPearls (StatPearls Publishing, Treasure Island (FL).
Keywords: transfusion-transmitted malaria, blood transfusion, asymptomatic malaria parasitemia, blood donors, rapid diagnostic tests (RDTs), microscopy, Plasmodium falciparum
Citation: Bowa C, Kalonga N, Mbonshi B, Sitali L, Simwela J, Kudowa E and Tepeka A-G (2025) Prevalence and Plasmodium species distribution of asymptomatic malaria parasitemia among blood donors at the Lilongwe blood transfusion center. Front. Malar. 3:1687355. doi: 10.3389/fmala.2025.1687355
Received: 17 August 2025; Accepted: 07 November 2025; Revised: 04 November 2025;
Published: 27 November 2025.
Edited by:
Tobias Obejum Apinjoh, University of Buea, CameroonReviewed by:
Abhai K. Tripathi, Johns Hopkins University, United StatesLaura Seijas Pereda, University of Alcalá, Spain
Copyright © 2025 Bowa, Kalonga, Mbonshi, Sitali, Simwela, Kudowa and Tepeka. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Collins Bowa, Y29sbGllYm93YUBnbWFpbC5jb20=
†These authors have contributed equally to this work
 Collins Bowa
Collins Bowa Ndagha Kalonga2
Ndagha Kalonga2 Amazing-Grace Tepeka
Amazing-Grace Tepeka