- 1Department of Life and Environmental Sciences, Marche Polytechnic University, Ancona, Italy
- 2Institute for Environmental Protection and Research (ISPRA), Livorno, Italy
Multidisciplinary investigations based on integration of chemical and biological measurements, represent an added value to monitoring and management protocols, and their use is recommended by European Directives to evaluate the environmental status of aquatic ecosystems. However, assessing the overall significance of results obtained in different typologies of studies is often a difficult challenge. The aim of this work was to present a quantitative Weight Of Evidence (WOE) model (Sediqualsoft) to integrate huge amounts of heterogeneous data and to validate this approach in complex monitoring scenarios. Using the case-study of an off-shore platform field in the Adriatic Sea, procedures are presented to elaborate different typologies of data (lines of evidence, LOEs), including chemical characterization of sediments, bioavailability, biomarkers, ecotoxicological bioassays and benthic communities around three platforms. These data are initially evaluated by logical flowcharts and mathematical algorithms, which provide specific hazard indices for each considered LOE, before their different weighting and overall integration in an environmental risk index. The monitoring study selected for the WOE elaboration consisted on chemical analyses of trace metals, aliphatic hydrocarbons, polycyclic aromatic hydrocarbons carried out on 60 sediment samples; the same samples were also characterized for the status of benthic communities; bioavailability of metals from sediments was assessed in laboratory conditions on the polychaete Hediste diversicolor, while bioaccumulation of inorganic and organic chemicals and biomarker responses were measured in native and transplanted mussels; ecotoxicological properties of sediments were evaluated through a battery of bioassays determining algal growth of the diatom Phaeodactylum tricornutum, bioluminescence of the marine bacterium Vibrio fischeri, survival of the copepod Acartia tonsa and embryotoxicity of sea urchin Paracentrotus lividus. Overall, almost 7000 analytical results were elaborated and summarized in specific hazard indices. The WOE integration of multiple typologies of data allowed more robust and weighted conclusions compared to the use of individual LOEs, highlighting the feasibility of this procedure for multidisciplinary monitoring and risk assessment approaches. On a practical side, the WOE evidences also suggested a revision of actual monitoring procedures. Overall, the proposed WOE model appeared as a useful tool to summarize large datasets of complex data in integrative indices, and to simplify the interpretation for stakeholders and decision makers, thus supporting a more comprehensive process of “site-oriented” management decisions.
Introduction
Marine ecosystems are challenged by several anthropogenic stressors, including release of chemicals through a variety of sources (i.e., riverine effluents, urban sewers, direct discharge, accidents, and atmospheric deposition). The impact of pollutants is one of the most faceted issues to investigate since mixtures of organic and inorganic compounds can interact through complex mechanisms, modulate responsiveness to other stressors and thus affect several biological responses, from cellular and physiological processes, up to populations dynamics and ecosystem functioning (Newman and Clements, 2007): in this context, the development of accurate monitoring plans is crucial for environmental protection. Initial programs were typically aimed to quantify the presence and distribution of chemicals in the environment, but the importance of assessing biological effects of contaminants has gradually risen and actual monitoring practices recommend the integration of both chemical and biological approaches (Piva et al., 2011; Benedetti et al., 2014; Regoli et al., 2014; Bebianno et al., 2015; Vethaak et al., 2017). The use of multidisciplinary studies for the characterization of aquatic environment quality is highly encouraged also by European Directives such as the Water Framework Directive (WFD, Directive 2000/60/EC) and the Marine Strategy Framework Directive (MFSD, Directive 2008/56/EC), which require member states to evaluate and classify the ecological status of water bodies through the integration of different quality indicators. Strategies for integrated chemical and biological monitoring have been described and recently adopted by various international agencies or working groups such as OSPAR, HELCOM, MEDPOL, ICES (Broeg and Lehtonen, 2006; HELCOM, 2010; Davies and Vethaak, 2012; OSPAR, 2013; Hylland et al., 2017; Lyons et al., 2017; Vethaak et al., 2017): a recognized advantage of such approaches is the added interpretative value derived from the integration of multiple typologies of studies, thus improving our ability to describe and interpret variations of environmental conditions.
The Sediment Quality Triad was the first Weight of Evidence (WOE) approach, combining various lines of evidence (LOEs), such as chemical analysis, toxicity testing, and considering the in-situ benthic community structure (Chapman, 2007), to link contamination with their biological and ecological impacts. Quantification of chemicals is fundamental in environmental monitoring but, by itself, it does not provide information on transfer to biota and biological effects of contaminants (Benedetti et al., 2012). Laboratory bioassays are useful to evaluate the presence of acutely toxic compounds measuring specific biological endpoints in selected organisms: proper batteries integrating various classes of ecotoxicological bioassays have progressively been developed to cover different taxa across the main ecological and trophic positions. Also benthic studies have evolved in the last decade moving from qualitative descriptions, to long-term surveys up to the functioning of benthic communities and development of Ecological Quality Status Descriptors (Dauvin, 2015; Borja et al., 2016, 2017).
Compared to the original triad, additional LoEs have further enhanced our possibility to understand cause-effect relationships between chemical exposure and biological effects, providing a more robust basis for environmental control and management. Bioaccumulation of pollutants in resident or transplanted organisms is a sure proxy of bioavailability and transfer of chemicals from abiotic matrices to biota, being also recognized in some environmental quality standards. Sub-lethal alterations at molecular and cellular level (biomarkers) provide a sensitive indication of early changes which often represent the first warning signals before the onset of long-term toxicological effects or changes at higher levels of biological organization (Broeg and Lehtonen, 2006; Moore et al., 2006; Regoli and Giuliani, 2014; Benedetti et al., 2015). Various health indices have been described in the last years combining complex biomarker data from multiple cellular pathways, to summarize the severity of biological disturbance in a scientifically sound but also user-friendly format for environmental managers and decision makers (Beliaeff and Burgeot, 2002; Broeg and Lehtonen, 2006; Dagnino et al., 2008; Piva et al., 2011; Benedetti et al., 2012; Marigómez et al., 2013).
The inclusion of multiple lines of evidence in a weight of evidence framework would thus represent a powerful approach within national and international strategies for monitoring environmental quality. However, the lack of standardized procedures for the integration of complex datasets of heterogeneous results, often prevents the adoption of such multidisciplinary approaches and their inclusion in decision-supporting procedures (Dagnino et al., 2008; Semenzin et al., 2008; Linkov et al., 2009; Piva et al., 2011; Benedetti et al., 2012). A multistep traffic-light system has been recently proposed for OSPAR, integrating data on contaminant and biological effects in background assessment concentrations (BACs), environmental assessment concentrations (EACs) and their analogous criteria for biological responses. These values are generally derived for North Atlantic areas and species (Vethaak et al., 2017), and for each analyzed parameter a specific color is given if below BAC (blue), between BAC and EAC (green), or above EAC (red): the graphical combination of all the results allows to easily visualize the proportion of parameters assigned to each color, but the relative proportions of various colors in different parameters is not summarized in a quantitative evaluation to facilitate a comparison between samples.
Quantitative criteria would certainly improve the utility of multidisciplinary assessments increasing their capability to compare and discriminate between different environmental conditions. In a recent and quantitative WOE model (Sediqualsoft), logical flowcharts and mathematical algorithms were developed to elaborate data from various LOEs (e.g., sediment chemistry, bioaccumulation, biomarkers, ecotoxicological bioassays and benthic communities): synthetic and quantitative hazard indices are provided for each of the considered line of evidence, before their overall integration in the final WOE assessment (Piva et al., 2011). Independent elaborations for various LOEs are based on different criteria specific for various typologies of data, including the number, magnitude and typology of chemicals exceeding normative guidelines or natural concentrations measured in control organisms, while for biological responses the eco- toxicological relevance of measured endpoints and their variations compared to specific thresholds are considered (Piva et al., 2011). These criteria have been validated in numerous national and international case studies for environmental risk assessment associated with polluted sediments, harbor areas, or complex natural and anthropic impacts on the marine environment (Piva et al., 2011; Benedetti et al., 2012, 2014; Regoli et al., 2014; Bebianno et al., 2015; Mestre et al., 2017; Pittura et al., 2018).
Standardized procedures for integrating heterogeneous data are of utmost importance, particularly in multidisciplinary monitoring scenarios when the overall significance of completely different typologies of results need to be summarized to adequately communicate the risk in a simple way, without reducing the scientific soundness of the overall investigation. In this respect, this work was aimed to demonstrate the feasibility of the quantitative WOE approach as an innovative procedure for quantitative impact assessment of complex anthropogenic activities. The selected case-study was the monitoring of off-shore platforms, which are often regulated by normative requirements, precise sampling strategies and parameters to analyze for demonstrating the lack of negative interactions with several indicators of the marine environment. Despite the extensive analytical effort, a practical challenge remains the absence of effective and transparent approaches to synthesize the overall significance of such huge amounts of different results, ensuring that environmental impacts and hazards are properly quantified and compared on both a geographical and temporal scale. In this study, following prescriptions by the Italian Institute for Environmental Protection and Research (ISPRA), 3 Adriatic platforms were subjected to an extensive multidisciplinary survey including physico-chemical and ecotoxicological characterization of sediments, status of benthic communities, bioavailability of metals from sediments to the polychaete Hediste diversicolor, bioaccumulation of inorganic and organic chemicals, and biomarker responses in natural and transplanted mussels. Despite the main objective of this paper is not a detailed discussion of such results, all these data have been provided to present the elaboration procedure through weighted criteria within the integrated WOE scheme, thus highlighting the importance of advanced criteria in risk assessment procedures, ensuring both scientific reliability and synthetic indices for stakeholders.
Materials and Methods
Sampling Activities and Experimental Design
Sampling of Sediments for Chemical Analyses, Ecotoxicological Characterization, and Bioavailability Assay
The oil field “Rospo Mare” contains three off-shore platforms, RSM-A, RSM-B and RSM-C, in the Southern Adriatic Sea, 20 km east from the city of Vasto (Figure 1). It’s Produced Water is not discharged into the sea but transported on land for treatment. According to requirements of the Italian Ministry of Environment, chemical characterization of sediments and analyses of benthic communities were carried out on 20 samples for platform collected along two orthogonal transects, from NW to SE and from NE to SW, at distances of 25, 50, 100, 250, and 1000 m (Figure 1). Samples were stored at −20°C for chemical analyses, while those for benthic communities were sieved onboard at 0.5 mm, immediately fixed with Formalin Free Fixative, AccustainTM (Sigma-Aldrich), and analyzed within 6 weeks.
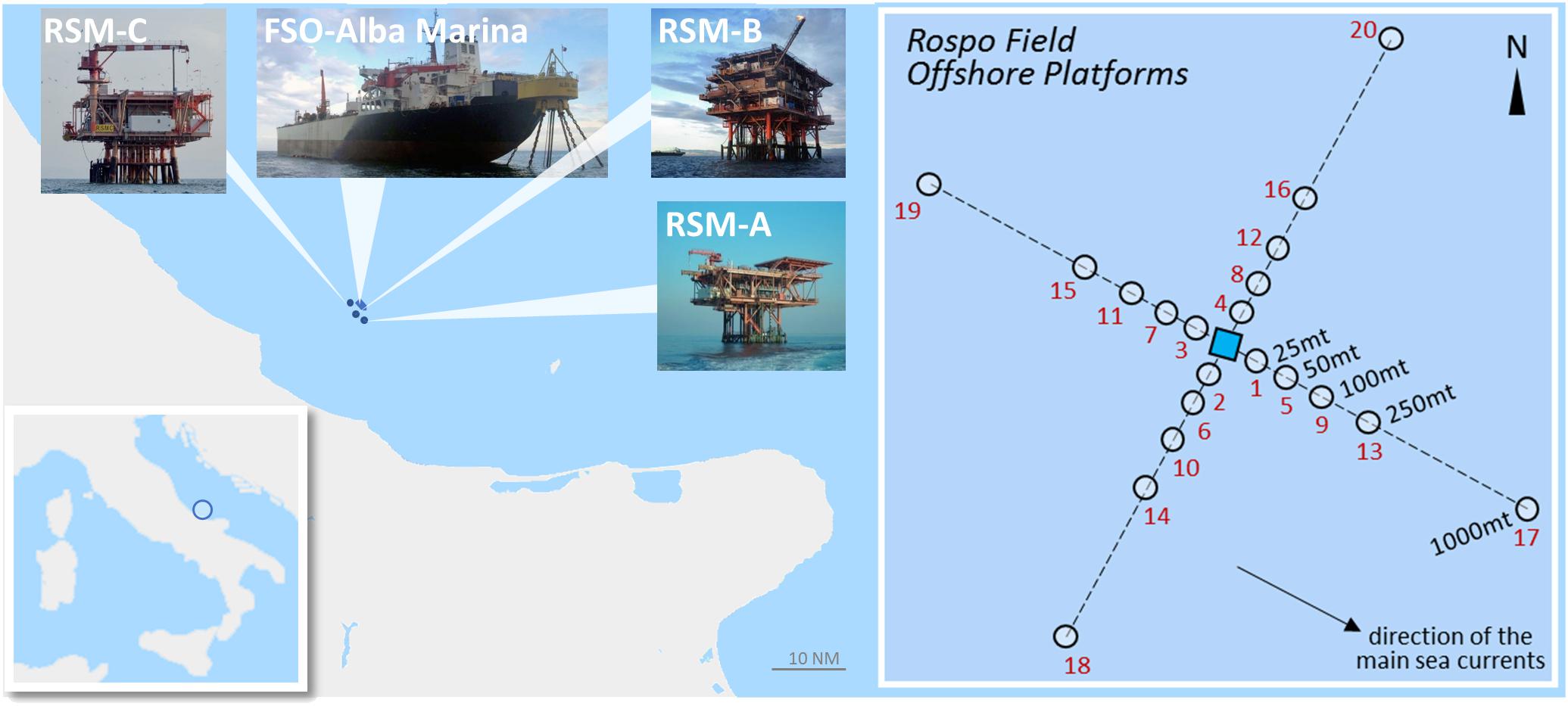
Figure 1. Geographical localization of Rospo Mare field and representative experimental sampling design: scale NM, nautical miles. For each of the three off-shore platforms the following analyses (samples) were performed: chemical characterization of sediments (samples 1–20); benthic communities (samples 1–20 in triplicate); ecotoxicological bioassays (samples 5–8, 17, 17bis, 19, and 19bis); bioavailability of metals from sediments to laboratory exposed polychaetes (samples 5–8, 17, 17bis, 19, and 19bis); bioavailability of metals and organic pollutants in mussels (both native organisms and transplanted on the platforms); biomarkers in mussels (both native organisms and transplanted on the platforms).
Sediments for assessing bioavailability of metals and ecotoxicological characteristics were sampled at eight sites for each platform: of these, four were at 50 m from the structures on the same transects previously identified, while four samples were collected at 1000 m along the transects of dominant currents, two upstream and two downstream (Figure 1). These sediments were stored at +4°C until the preparation of elutriates (within 72 h from the collection), or their use for the bioaccumulation assay with the polychaete H. diversicolor.
Mussel Watch With Native and Transplanted Mussels
Natural mussels, Mytilus galloprovincialis, were sampled from each platform at 3 m depth, while for translocation experiments, control organisms from a local farm were maintained for 4 weeks at the same depth: mussels were deployed in net bags (80 cm height × 25 cm diameter, mesh size 1.5 × 3 cm) secured to a nylon rope, and the system was reinforced by an external net cylinder (2 m height × 80 cm diameter, mesh size 2 × 3 cm) to guarantee free circulation of seawater and protect mussels from fish predation (Gorbi et al., 2008). Approximately 200 adult mussels (6.0 ± 0.5 cm in shell length) were collected; organisms were then wrapped in humid towels, rapidly transported to laboratory and further processed: for bioaccumulation analyses, 10 replicates were prepared from every sampling site, each consisting of whole tissues from 10 organisms, flash frozen in liquid nitrogen and stored at −20°C. For biomarkers, digestive glands were rapidly removed from 30 specimens, pooled in 10 samples (each with tissues of three specimens), frozen in liquid nitrogen and maintained at −80°C; small pieces of digestive glands were rapidly excised from five mussels for histological analyses, placed on cork chucks, frozen in n-hexane precooled to −70°C in liquid nitrogen, and maintained at −80°C; hemolymph was withdrawn from the adductor mussels of 15 specimens, divided in five aliquots, each with fluids of three individuals, and immediately frozen in liquid nitrogen; genotoxic effects were measured in hemocytes of five specimens maintained in Carnoy fixative (acetic acid:methanol 1:3).
Bioaccumulation Assay With the Polychaete Hediste diversicolor
The bioavailability of metals in the eight sediment samples specifically collected at each platform (see above) were tested with the polychaete H. diversicolor. Organisms were obtained from a commercial supplier, acclimated to laboratory conditions for 72 h in aerated artificial seawater (18 ± 2°C, salinity 30 psu, prepared by dissolving InstantOcean® marine salt in reverse osmosis water), and randomly placed in 25 vessels (eight sites for platform and one control sediment), each containing 20 organisms, 600–800 g of sediment and 500 mL of artificial seawater. Water was renewed every week and no mortality was observed after 28 days, when organisms were placed for additional 72 h in artificial seawater to excrete sediment grains. For each treatment, three pools with whole tissues of 6–7 organisms were flash frozen in liquid nitrogen and stored at −20°C until analyses of trace metals.
Analytical Methodologies and Weighted Elaboration of Results
Chemical Characterization of Sediments and Organisms
Concentrations of trace metals (Al, As, Cd, Cu, Cr, Hg, Ni, Pb, Zn), polycyclic aromatic hydrocarbons and aliphatic hydrocarbons were determined in sediments and in mussel tissues, while only metals were measured in polychaetes H. diversicolor exposed to sediments at laboratory conditions. Analytical methods and procedures for quality assurance/quality control were previously described, based on conventional procedures of gas-chromatography with flame ionization detector (FID) for aliphatic hydrocarbons, high performance liquid chromatography (HPLC) with diode array (DAD) and fluorimetric detection for PAHs, atomic absorption spectrophotometry (AAS) for trace metals (Benedetti et al., 2014).
Obtained results have been elaborated through weighted criteria of the WOE model (Sediqualsoft) summarizing specific hazard indices for each typology of data (LOE): conceptual elaborations of the WOE model have been fully detailed elsewhere (Piva et al., 2011). The evaluation of chemical hazards in sediments (LOE-1) is based on the initial calculation, for each pollutant, of the Ratio to Reference (RTR), i.e., the ratio between concentration measured in sediments and threshold indicated by various sediment quality guidelines, SQGs (Figure 2); in the present investigation, SQGs were those from European Water Framework Directive 2000/60 for the achievement of a good ecological status. From the calculated ratio to reference, a RTRw is obtained by the application of a correction factor (w) which, depending to the typology of chemicals, ranges from 1 to 1.3 for “non-priority” (w = 1), “priority” (w = 1.1) or “priority and hazardous” pollutants (w = 1.3) according to EC Directive 2008/105.
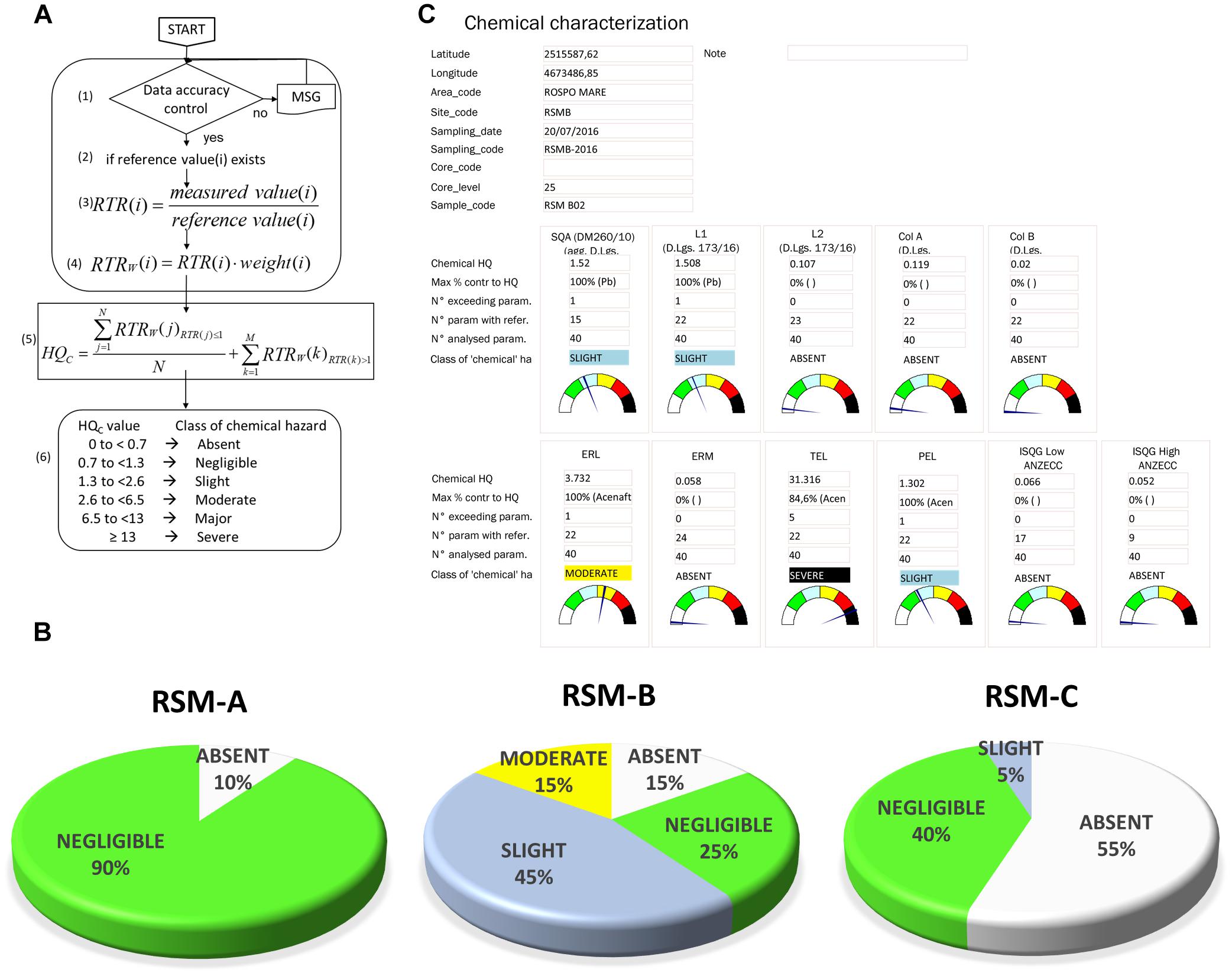
Figure 2. LOE-1: elaboration of chemical results in sediments. (A) Flowchart and calculations of Chemical Hazard Quotient (HQC). (B) Results obtained for the three platforms indicating the percentage distribution of samples assigned to various classes of hazard considering European Sediment Quality Guidelines (EQS) as reference. (C) Model output of chemical characterization (Sediqualsoft).
In the calculation of the specific Hazard Quotient for chemistry (HQC), an average RTRw is obtained for all of the parameters with RTR < 1 (i.e., values below the SQG), while for those with RTR > 1, the RTRw are individually added into the summation ∑:
With this calculation, the HQC increases according to both the number and the magnitude of the exceeding parameters (for which the specific RTRw are individually added), and it is not lowered by the analysis of many “not exceeding” parameters (which are summarized in the averaged RTRw). The values of HQC are assigned to one class of chemical hazard (absent or negligible, slight, moderate, major, severe) depending on the number, typology, and magnitude of exceeding chemicals (see Figure 2 and Piva et al., 2011 for details).
The results on bioaccumulation of chemicals in tissues of wild and caged mussels (LOE-2) were elaborated calculating, for each parameter, the increase of concentration compared to control organisms, corrected for the typology of pollutant and the statistical significance of the difference (Piva et al., 2011 for details). The cumulative Hazard Quotient for bioavailability (HQBA) does not consider parameters with RTRw < 1.3 (tissue concentration ≤ value in control organisms for a priority and hazardous pollutant), calculates the average for those with RTRw ranging between 1.3 and 2.6 (i.e., up to 2-fold increase compared to controls for a priority and hazardous pollutant), and adds the summation (∑) of all those with RTRw ≥ 2.6 (see Figure 3 and Piva et al., 2011 for details):
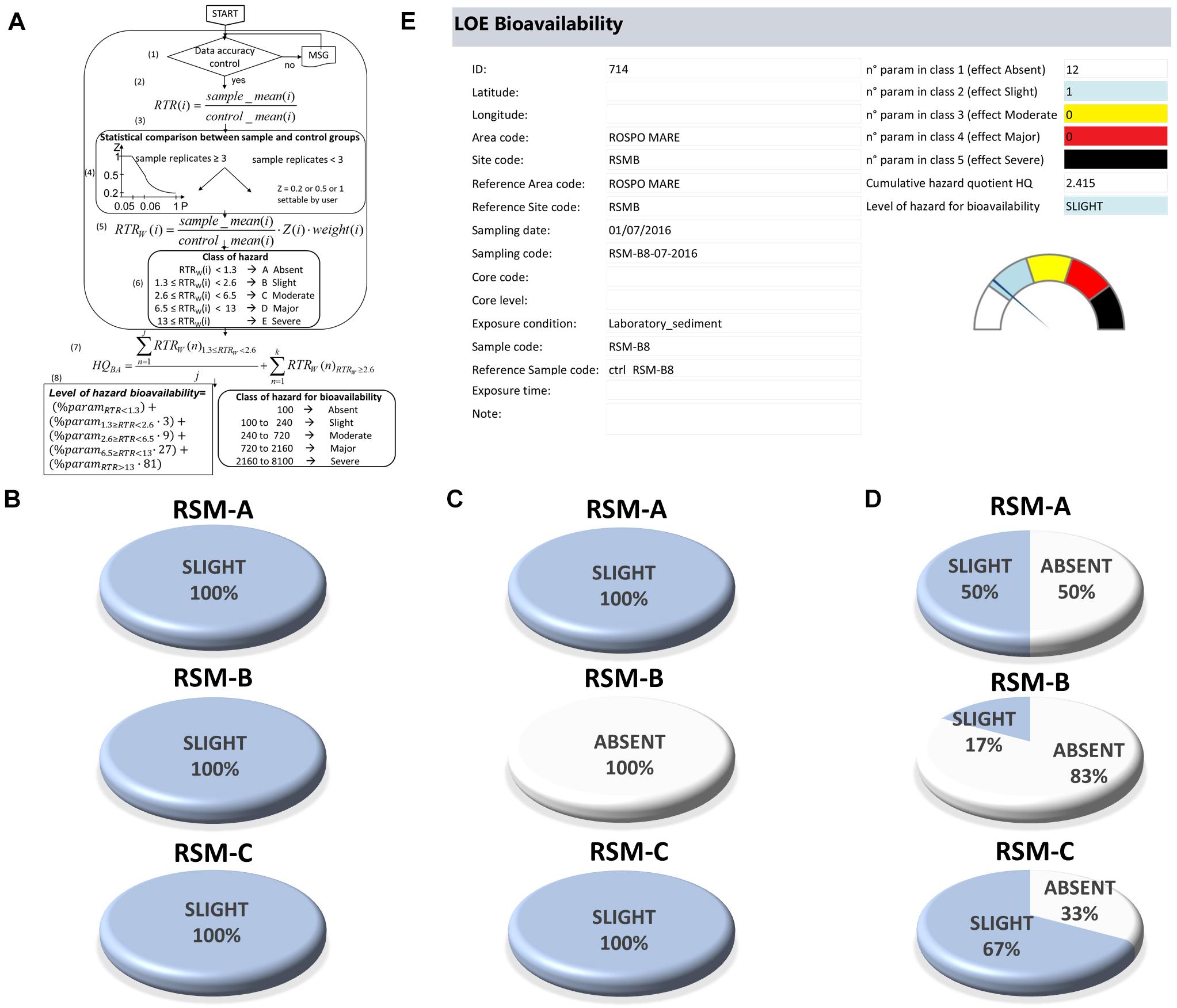
Figure 3. LOE-2: elaboration of bioavailability results. (A) Flowchart and calculations of Bioavailability Hazard Quotient (HQBA). (B–D) Results obtained for the three platforms indicating the percentage distribution of samples assigned to various classes of hazard for native mussels (B), transplanted mussels (C), and laboratory exposed polychaetes (D). (E) Model output for bioavailability characterization.
The level of cumulative HQBA is summarized in one class of hazard for bioavailability, from Absent to Severe, depending on the distribution of analyzed chemicals within the different classes of effect (Piva et al., 2011; Regoli et al., 2014).
Biomarkers Analyses in Native and Transplanted Mussels
Biomarkers were analyzed in native and transplanted mussels. Metallothioneins (MTs), single antioxidant defenses (catalase, glutathione S-transferases, glutathione peroxidases, glutathione reductase, total glutathione), total oxyradical scavenging capacity toward peroxyl radicals (TOSC ROO•) and hydroxyl radicals (TOSC HO•), malondialdehyde content (MDA) were spectrophotometrically measured in digestive glands of mussels. Hemocytes were processed for acetylcholinesterase activity, immune-related alterations, (lysosomal membrane stability, phagocytosis activity, and granulocytes versus hyalinocytes ratio) and genotoxic effects, in terms of percentage of DNA integrity (comet assay) and micronuclei frequency (MN). Standardized protocols have been fully described elsewhere (Benedetti et al., 2014).
The elaboration of biomarkers data is based on a specifically developed algorithm (LOE-3) which assigns to each response a “weight” (based on the toxicological relevance of the endpoint) and a “threshold,” indicating the minimum change above which, depending on species and tissue, the biomarker response should be considered as biologically relevant (Piva et al., 2011). For every analyzed biomarker, the measured variation is normalized by comparison to its specific threshold (effect), then corrected for the weight of the response and the statistical significance of the difference in respect to controls. The calculation of the Hazard Quotient for biomarkers (HQBM) does not consider the contribution of responses with an effect < 1 (lower than threshold), calculates the average for those with an effect up to two-fold compared to the threshold and adds the summation (∑) for the responses more than 2-fold greater than the respective threshold (Figure 4 and Regoli et al., 2014 for details):
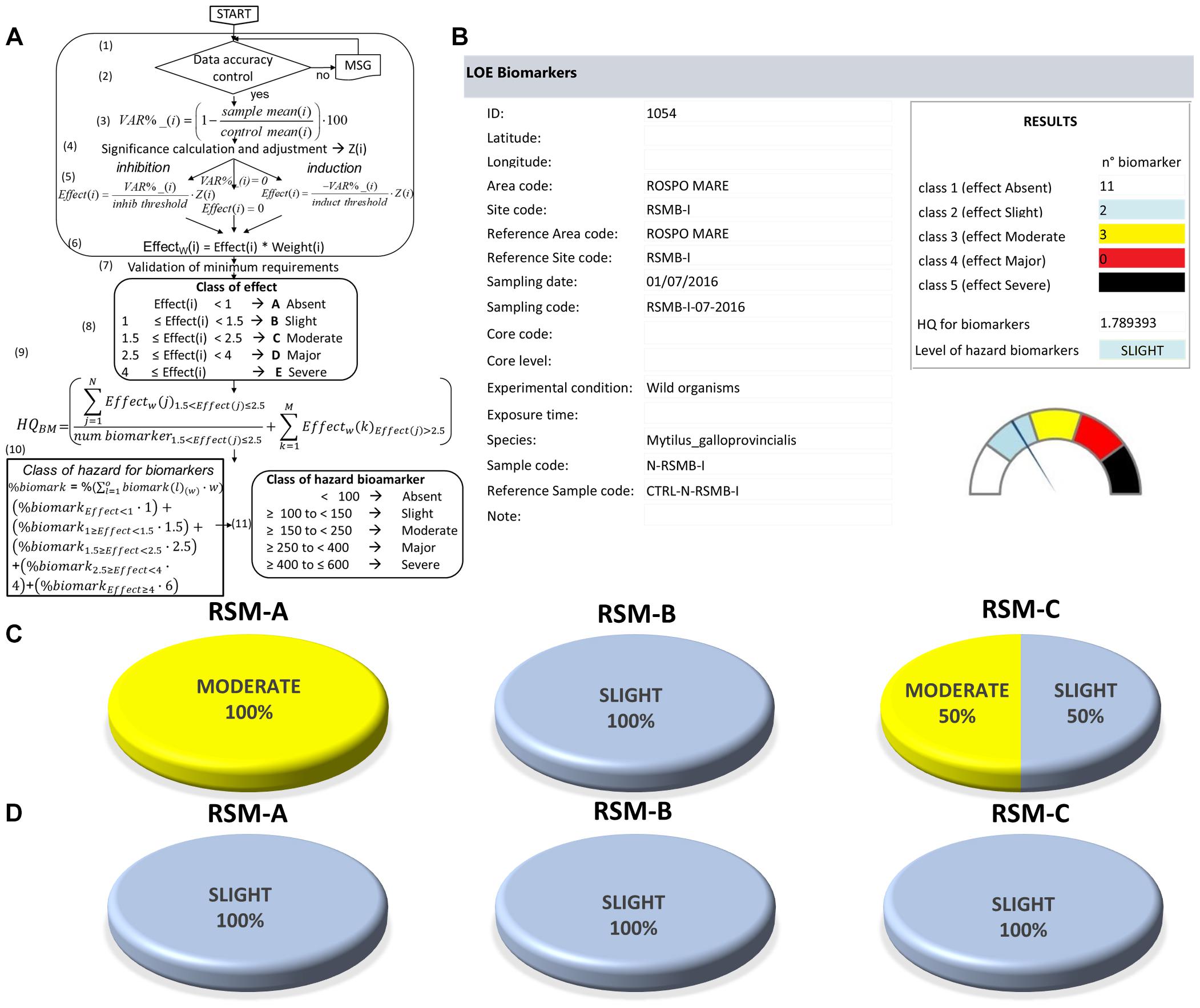
Figure 4. LOE-3: elaboration of biomarker results. (A) Flowchart and calculations of Biomarkers Hazard Quotient (HQBM). (B) Model output of biomarker characterization. (C,D) Results obtained for the three platforms indicating the percentage distribution of samples assigned to various classes of hazard for native (C) and transplanted mussels (D).
According to variations measured for various biomarkers, the Sediqualsoft model summarizes the level of cumulative HQBM in one of five classes of hazard for biomarkers, from Absent to Severe (Piva et al., 2011).
Ecotoxicological Bioassays
A battery of ecotoxicological bioassays was applied to sediments (eight sites for each platform) following standardized procedures. The bioluminescence test with Vibrio fischeri (ISO 11348-3: 200/) and the larval development assay with Acartia tonsa (ISO 16778:2015) were selected for testing the toxicity of the solid phase, while the algal growth inhibition of Phaeodactylum tricornutum (ISO 10253: 2006) and the sea urchin embryotoxicity assay with Paracentrotus lividus (ISPRA, 2017) were used for testing the toxicity of elutriates.
In the weighted elaboration of ecotoxicological bioassays (LOE-4), each test has a weight depending on the biological relevance of the endpoint, and a threshold derived from the sensitivity of the species (Piva et al., 2011). The cumulative hazard quotient (HQBattery) is obtained by the summation (∑) of the weighted effects (Effectw), i.e., the variations measured for each test normalized to its specific thresholds, and corrected for the statistical significance of the difference (w), biological importance of the endpoint, and exposure conditions (w2) (see Figure 5 and Piva et al., 2011 for details):
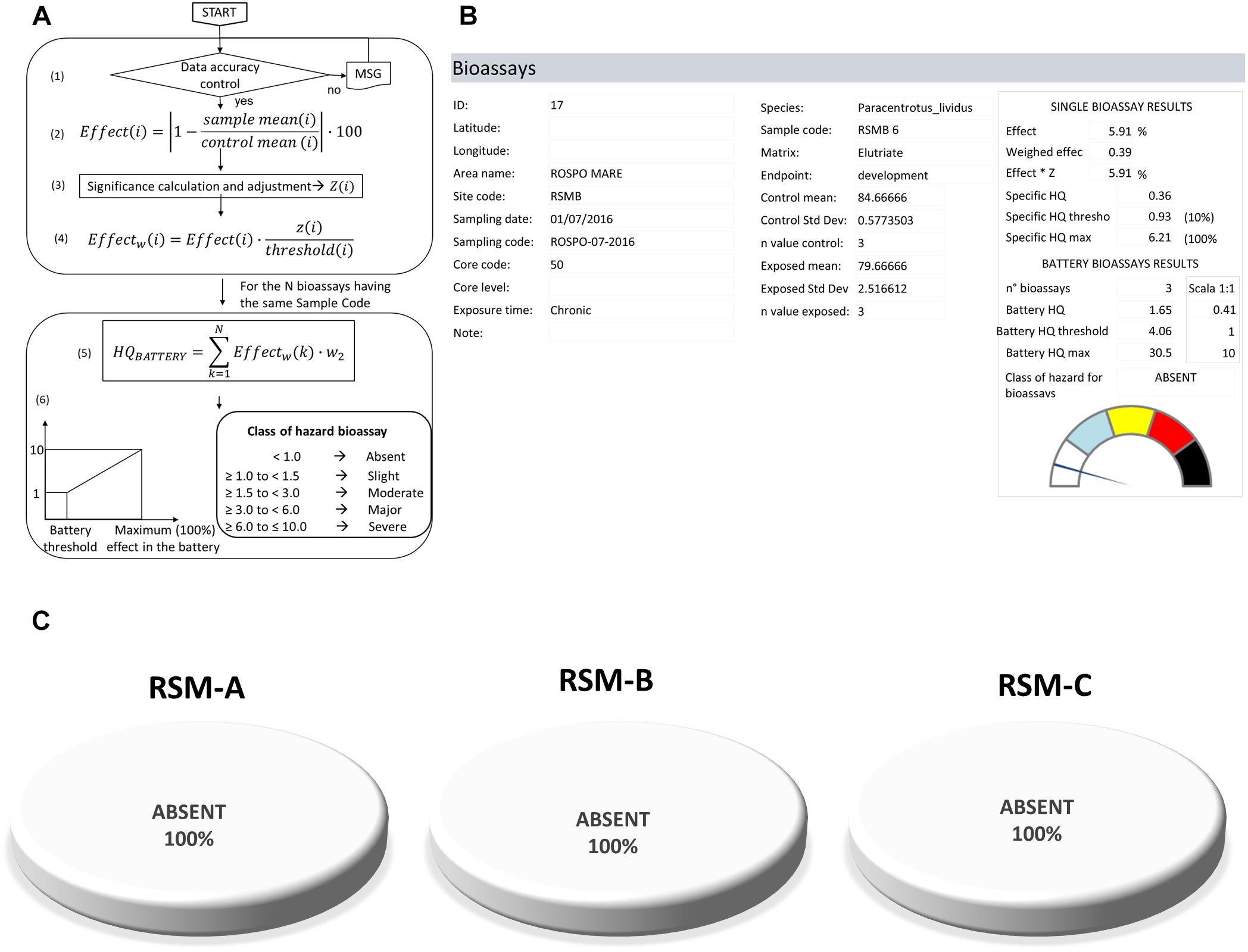
Figure 5. LOE-4: elaboration of ecotoxicological bioassays. (A) Flowchart and calculations of the Battery of Bioassays Hazard Quotient (HQBATTERY). (B) Model output for bioassays elaborations. (C) Results obtained for the three platforms indicating a class of hazard assigned as absent in 100% of samples.
The HQBattery is normalized to a scale ranging from 0 to 10, where 1 is the Battery Threshold (when all the measured bioassays exhibit an effect equal to the threshold, 10 when all the assays exhibit 100% of effect), and then assigned to one of five classes of hazard, from Absent to Severe (Figure 5).
Benthic Communities Analyses
Sediment samples were sorted at the stereomicroscope and the principal animal taxa were generally classified at species level. For each species, whenever possible, the corresponding biocenosis was identified to define the bionomic and ecological settlement.
For the elaboration of data on benthic communities (LOE-5), a specific module has been developed in structured query language (SQL) and Visual Basic to convert the list of identified species in several available univariate and multivariate indices for the classification of ecological quality (Borja et al., 2000; Simboura and Zenetos, 2002; Dauvin and Ruellet, 2007; Muxika et al., 2007; Mistri and Munari, 2008; Sigovini et al., 2013). Such elaborated indices include total abundance (N), species richness (S), Shannon-Weaver Diversity Index (H’), Margalef index (D), Pielou’s evenness index (J), AZTI’ Marine Biotic Index (AMBI), multimetric-AZTI Marine Biotic Index (m-AMBI), Bentic Index (BENTIX), Benthic Index based on Taxonomic Sufficiency (BITS), and Benthic Opportunistic Polychaetes Amphipods (BOPA index). In this work, the AMBI index was chosen for the integration with other LOEs in the final WOE elaboration of ecological risk.
Data Elaboration Through Weight of Evidence (WOE) Ecological Risk Assessment Model
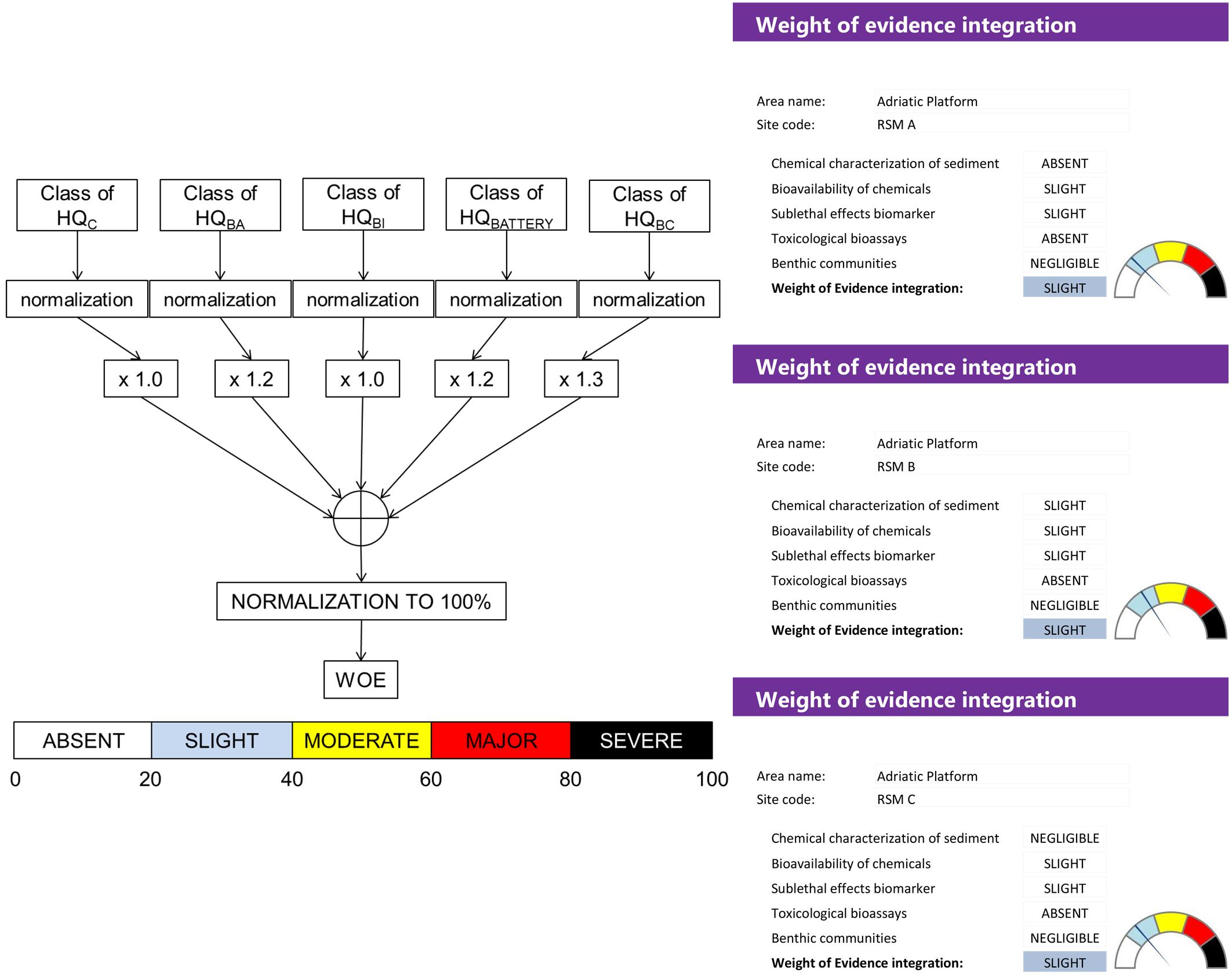
The huge data-sets of results elaborated from the five lines of evidence have been finally integrated through a Weight of Evidence approach (WOE) based on the quantitative model Sediqualsoft. The quantitative hazard quotients (HQs) obtained for each LOEs are normalized to a common scale and given a different weight according to their ecological relevance in the assessment.
In this study, assigned weights to various LOEs were 1.0 for chemical characterization of sediments (LOE-1), 1.2 for bioavailability of chemicals in native and transplanted mussels and 0.6 for bioavailability of metals in laboratory exposed polychaetes (LOE-2), 1.0 for sub-lethal effects on biomarkers in mussels (LOE-3), 1.2 for the ecotoxicological results of the battery of bioassays (LOE-4), 1.3 for composition of benthic communities (LOE-5). An overall WOE level of risk is thus calculated and assigned to 1 of 5 classes of risk from absent to severe (Piva et al., 2011). Scientific criteria, validation of weights and thresholds, expert judgment evaluations and specific flow-charts of each LOE have been validated elsewhere (Piva et al., 2011; Benedetti et al., 2012, 2014).
Results
Chemical Characterization of Sediments (LOE-1)
Chemical analyses of sediments on the three platforms included 60 sampling sites, 53 analytes (individual trace metals, PAHs and aliphatic hydrocarbons), for a total of 3180 analytical results to interpret and compare toward different normative thresholds [i.e., european quality standards (EQS), and other Italian or international sediments guidelines]. Despite the aim of this work is not to discuss the results of the monitoring study but rather the advantages of the integrated WOE elaboration procedure, nonetheless all the measured values are given in Supplementary Materials (Supplementary Table S1). Concentrations of chemical parameters did not reveal critical values, despite some metals were higher than their respective EQS; among PAHs, low molecular weight hydrocarbons largely prevailed, representing typically more than 90% of the total PAH content.
The weighted criteria based on the number, typology and the magnitude of exceeding contaminants (Figure 2A), easily summarized the overall significance of almost 3200 analytical results: the chemical hazard level elaborated toward the European Quality Standards for sediments, ranged mostly between Absent and Negligible in the three platforms without significant differences between sampling distances or different platforms (Figure 2B). The Sediqualsoft model output provides, for all the potentially available sediment normative guidelines, the quantitative value of the calculated HQC, the class of hazard and other useful information on obtained results, such as the chemical contributing more to the HQC, or the number of exceeding chemicals (Figure 2C). A very large scientific information was thus summarized in an easy format for non-expert stakeholders, maintaining the possibility to get important information from the module output: the Moderate chemical hazard observed in 15% of sediments from platform RSM-B was due, for 50–75%, to the contribution of one trace metal (Cr, Cd, or Zn), slightly higher than the corresponding EQS.
Bioaccumulation of Chemicals in Polychaetes and Mussels (LOE-2)
The assessment of bioavailability was evaluated in this study on native and transplanted mussels for both inorganic and organic chemicals and, for trace metals only, also in polychaetes exposed to sediments at laboratory conditions. According to the experimental design on the three platforms and the measured analytes, a total of 2160 results were obtained which are reported in Supplementary Materials (Supplementary Table S2). Data from H. diversicolor revealed often a marked variability in tissue concentrations of metals between organisms exposed to various sediment samples, without clear trends according to platforms or sampling distance. Also bioaccumulation in native and transplanted mussels revealed a few variations, of limited magnitude and without differences between platforms or experimental conditions. The elaboration of bioavailability data through weighted criteria, (Figure 3A) allowed to summarize all the data as representing an Absent or Slight hazard in terms of bioavailability for native mussels, transplanted mussels or laboratory exposed polychaetes (Figure 3B). The model output clearly visualizes the number of parameters assigned to each class of effect, the quantitative HQBA, and the overall level of bioavailability hazard among 1 of 5 possible classes (Figure 3C).
Biomarker Responses in Mussels (LOE-3)
Biomarkers analyzed in mussels included 16 well recognized responses among metallothioneins, acetylcholinesterase, immune parameters, lysosomal responses, antioxidants and total oxyradical scavenging capacity, lipid peroxidation, DNA damage, for a total of almost 600 results to interpret (see Supplementary Materials and Supplementary Table S3). Significant variations were occasionally measured for granulocytes/hyalinocytes ratio, metallothioneins, catalase, total antioxidant capacity, malondialdehyde, DNA integrity. Beside the possibility to discuss on specific pathways and mechanisms of action, the complexity of scientific information is summarized by the flow-chart given in Figure 4A which elaborates data according to weight and threshold assigned to each biomarker. The overall hazard quotient for biomarkers (HQBM) is thus calculated from the number of changed biomarkers, their toxicological relevance, statistical significance and magnitude of such variations, all information which are visualized in the Sediqualsoft model output (Figure 4B). In this study the hazard index for subcellular effects ranged between Slight and Moderate, depending on the experimental approach and platform (Figure 4C), mostly influenced by variations of some immune system responses and antioxidant defenses.
Ecotoxicological Bioassays (LOE-4)
Ecotoxicological characteristics of eight sediment samples for each of the three platforms were evaluated with a battery of four bioassays for a total of almost 100 bioassays tested in triplicate. These results highlighted only a few significant responses for embryotoxicity assay (Supplementary Materials and Supplementary Table S4). The use of weighted criteria did not consider the worst result for the ecotoxicological classification but elaborated the whole battery based on the biological relevance of each endpoint, and the thresholds derived from the sensitivity of various species (Figure 5A). The weighted criteria resulted in an ecotoxicological hazard index which was always summarized as Absent for all the sediment samples of the three platforms: the model output provides all the details on the results obtained for individual bioassays (Figures 5B,C).
Status of Benthic Communities (LOE-5)
For benthic communities the analyses of 180 sediment samples from the three platforms allowed to identify 6473 organisms as belonging to 112 taxonomic groups. The WOE Sediqualsoft model did not develop any new index, but developed the informatic tool to elaborate the list of identified species in all the already available community descriptors, diversity indices and ecological indicators, with corresponding evaluations (Figures 6A,B). Supplementary Materials provide a list of observed species (Supplementary Table S5a) and calculated indices of abundance, richness, Margalef, Shannon, Pielou, AMBI, BENTHIX, BOPA, BITS, mAMBI (Supplementary Table S5b). In this study, the AMBI index, selected as the community descriptor for the investigated site, provided a hazard quotient classified as Absent or Negligible (Figure 6C).
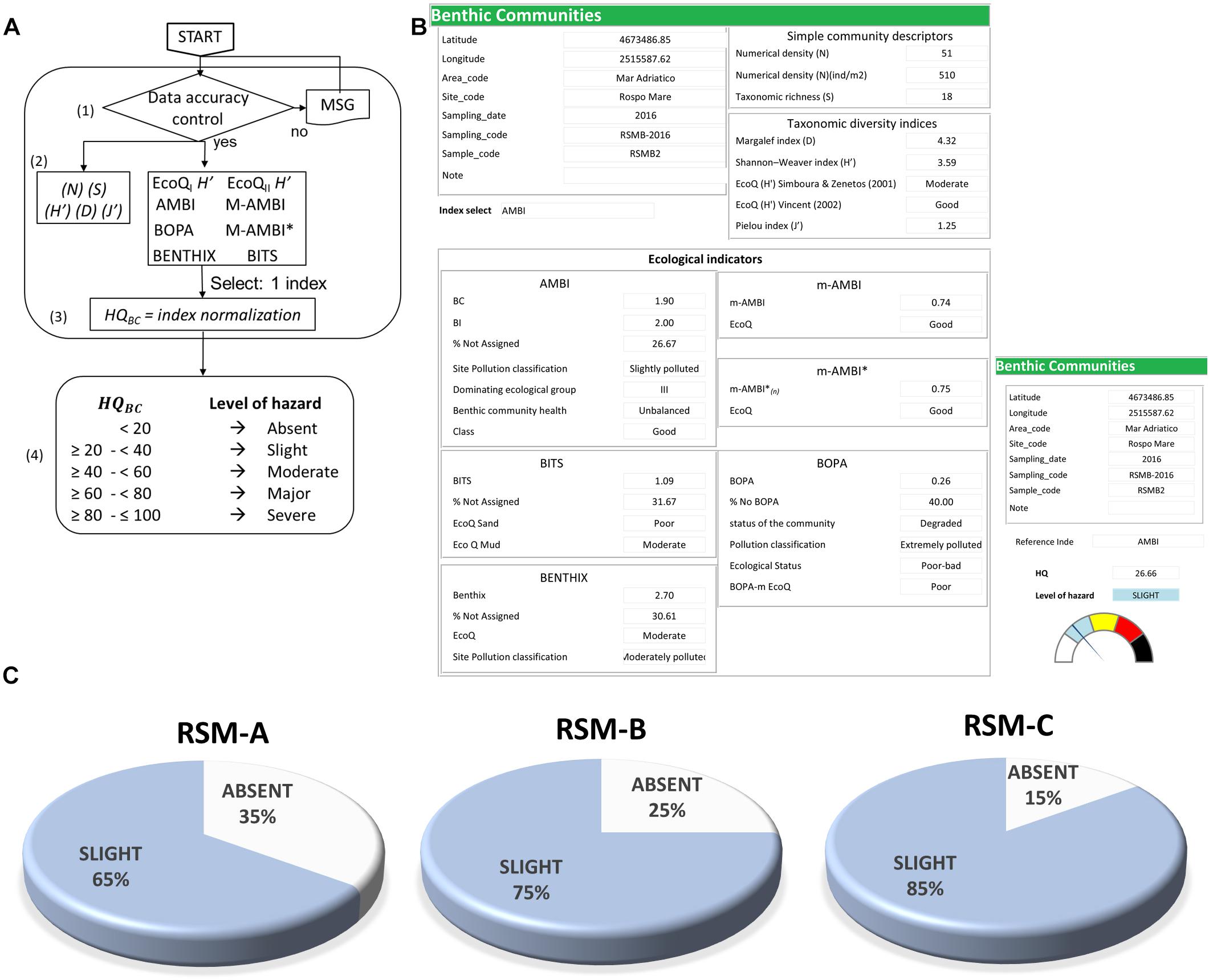
Figure 6. LOE-5: elaboration of benthic communities analyses. (A) Flowchart for the calculation of different indices. (B) Model output for elaborations of benthic communities. (C) Results obtained for the three platforms indicating the percentage of samples assigned to various classes of hazard.
Weight of Evidence Integration (WOE)
The final WOE elaboration, after normalization to a common scale, integrated the Hazard Quotients obtained from various LOEs giving them a different weight according to the ecological relevance of each typology of analyses (Figure 7). A total of approximately 7000 heterogeneous analytical results were summarized in a WOE evaluation of Slight risk (Figure 7), derived from a combination of specific HQs ranging from Absent (i.e., ecotoxicological bioassays or benthic communities) to Moderate (i.e., biomarker responses).
Discussion
It is now worldwide recognized that the characterization of environmental quality and ecological risk assessment must be addressed with multidisciplinary approaches that integrate traditional chemical analyses of abiotic matrices (water and sediments), with those revealing the onset of effects at different levels of biological organization, from bioaccumulation processes and molecular alterations up to population and communities structures (Moore et al., 2004; Hylland, 2006; Chapman, 2007; Viarengo et al., 2007).
The combination of chemical and biological analyses represents an added value for monitoring and management protocols, in line with the recent European Directives which recommend the use of multiple quality indicators for aquatic ecosystems (Lyons et al., 2010, 2017; Lehtonen et al., 2014). Although multidisciplinary approaches are universally accepted from a conceptual point of view, some critical aspects still limit their practical application in standardized procedures of the risk assessment. The interpretation of heterogeneous data often requires expert evaluations, and their integration is even more difficult. The development of quantitative or qualitative scales and indicators is aimed to summarize complex scientific information for an easier interpretation by policy-makers or environmental managers but this approach is generally limited to specific classes of results (Dagnino et al., 2008; Linkov et al., 2009; Piva et al., 2011; Marigómez et al., 2013, Borja et al., 2016, Borja et al., 2017).
The innovative aspect of this study is the application of a quantitative multidisciplinary Weight Of Evidence (WOE) model (Sediqualsoft), able to process and integrate data from five lines of evidence (LOEs) including chemical characterization of sediments, bioavailability, biomarker responses, battery of ecotoxicological assays, status of benthic communities. The presented case-study was the monitoring of an off-shore platforms field in the Adriatic Sea, selected to demonstrate the practical importance of a procedure combining a scientifically sound approach with the possibility to synthesize the overall significance of obtained results.
The most relevant feature of this WOE model is the use of weighted evaluation criteria that for such a huge amount of chemical data allowed to abandon the “pass-to fail” approach, where even a single parameter slightly below or above a threshold, would have determined the chemical classification of sediments. The evaluation of a quantitative index based on the number, typology and magnitude of exceeding chemicals guarantees a more accurate discrimination between samples and comparisons between different areas or periods. Similarly, biological evaluations are not based on variations of individual biomarkers or the worst result for ecotoxicological bioassays, allowing a more integrated assessment of the ecotoxicological hazard. Such weighted criteria have been successfully applied in several field studies (Piva et al., 2011; Benedetti et al., 2012, 2014; Regoli et al., 2014; Bebianno et al., 2015; Mestre et al., 2017) and incorporated in the last Italian law for determining quality class and management options for dredged marine sediments based on their chemical and ecotoxicological characterization (DM 173, 16 July, 2016).
Concerning the different typologies of results obtained in this study, the chemical characterization of sediments around the 3 platforms produced almost 3,200 results to be interpreted according to regulatory references or guidelines. A tabular approach applied to such a massive number of data would have not allowed their overall evaluation, the comparison between different areas or periods, nor any effective synthesis and communication of their environmental significance to non-expert stakeholders. In fact, although concentrations were always within typical ranges of values reported for the Adriatic or non-impacted basins, some values exceeding corresponding EQS were measured for a few metals (Dolenec et al., 1998; Zhang et al., 2008; Tobiszewski and Namieśnik, 2012; Benedetti et al., 2014; Etiope et al., 2014). The substantial uniformity of results suggests that these values represent the typical basal levels of the area, and the weighted elaboration provided a class of chemical hazards generally ranging from Absent to Negligible toward EQS: only for three samples the summarized chemical HQ was Medium due to the concentration of Cd, Cr, or Pb reinforcing the evidence that such individual levels can not to be considered as environmental anomalies due to the presence of the platforms.
The assessment of bioavailability allows to evaluate the biological relevance of chemical data and the application of weighted evaluation criteria is of particular utility in environmental risk assessment. In fact, there are only a few EQS for biota, and their application is often too simplistic and not reliable to represent the marked variability in bioaccumulation phenomena and local geochemical features. As an example, the EQS for mercury in biota is 0.02 μg/g (fresh weight), a value that does not consider differences in tissues or the trophic position, and appears inadequate for Mediterranean organisms which, influenced by natural geochemical anomalies of the basin, typically present higher basal levels of this element. With weighted criteria, tissue concentrations in the organisms from the investigated sites are compared to values measured in control organisms, allowing to eliminate also the effect of seasonal variability. Their elaboration considers the typology of each contaminant, as well as the magnitude and the statistical significance of observed variations. In the present study, the bioaccumulation of more than 30 analytes (between metals, polycyclic aromatic hydrocarbons and aliphatic hydrocarbons) in mussels and polychaetes resulted in approximately 2,200 analytical results that would have been impossible to evaluate based on EQS available for only a few parameters and not always appropriate for Mediterranean organisms. Although obtained results were similar to natural variability intervals already reported for these species (Bocchetti et al., 2008; Fattorini et al., 2008; Gorbi et al., 2008; Benedetti et al., 2014), nonetheless such a large number of data would have also hampered a quantitative comparison between different areas, to discriminate the effects of platforms from natural variability. Based on weighted integration, bioaccumulation data were summarized in a bioavailability hazard ranging between absent and Low depending on the species or experimental conditions, providing a user-friendly format, easy to understand also for non-expert stakeholders.
Biomarkers are recognized for their sensitivity in highlighting the early onset of molecular or cellular alterations, with potential adverse effects on the health conditions of organisms. Widely used to demonstrate the biological impact in polluted environments, they are of particular relevance also in areas with limited anthropogenic pressure since they evaluate integrated responses even when concentrations of individual contaminants are below their respective thresholds of effect (Regoli et al., 2004, 2011, 2014; Regoli and Giuliani, 2014). In this respect, variations of immune and antioxidant biomarkers are widely recognized as early warning signals; however, the complexity of the oxidative network, characterized by several interactions and cascade-effects among different pathways (Regoli and Giuliani, 2014), does not allow to consider variations of individual responses as diagnostic of cellular stress conditions. Routine application of biomarkers in environmental risk assessment procedures is thus often prevented by such difficulty in summarizing data from multiple cellular pathways in simple indices that reflect the overall severity of biological effects. In the present study, the elaboration of results obtained from almost 600 analyses of metabolic systems and biochemical pathways, weighted the toxicological relevance of analyzed biomarkers and normalized the magnitude of observed variations toward specific thresholds for each response. The application of weighted criteria was again fundamental to summarize a large data-set of complex results in a synthetic form; regardless of the significance of variations observed for biomarkers and the mechanistic interactions between immune system and oxidative responses, the overall cellular hazard elaborated from biomarkers ranged from Low to Medium in native mussels, being always Low in transplanted ones. A similar outcome of elaboration does not reduce the scientific relevance of investigated pathways and mechanisms, while being easy to be communicated to stakeholders without an in-depth molecular and cellular knowledge.
The ecotoxicological bioassays carried out with a battery of 4 different species (Vibrio fischeri, Phaeodactylum tricornutum, Paracentrotus lividus, Acartia tonsa) provided rather uniform results between different platforms and distances. The only test showing effects of toxicity was the embryo development of sea urchin (P. lividus), which would have conditioned the ecotoxicological classification of some sediment samples if the “worst result” approach had been followed. On the other hand, the use of weighted criteria to integrate the results of the whole battery of ecotoxicological bioassays (based on weight of endpoints and sensitivity of different species) always indicated the lack of an ecotoxicological hazard for the sediments collected around the three platforms. The usefulness of summarizing the different responsiveness of bioassays in a synthetic index of the battery, led to the adoption of such weighted criteria in Italian normative on classification and management of dredged sediments allowing to better discriminate the ecotoxicological potential of different samples.
For elaboration of data on benthic communities, there are many descriptors, indices of taxonomic diversity and ecological indicators which have been developed in the last three decades due to the ecological value of these analyses, and the need of standardized evaluation procedures. In this respect, the model used in the present study (Sediqualsoft) was set to automatically calculate all the available indices for selecting the more appropriate in the final integration with other LOEs. Different indices provided some contradictory results around Adriatic off-shore platforms because of the differences in their structure and discrepancies in their assignment of species sensitivity (Spagnolo et al., 2014). In our study, the only significant differences among the collected sediment samples, were obtained in terms of total abundance that, however, were attributable more to the granulometric composition of sediments rather than to a possible impact of the platforms. The application of the AMBI index (Borja et al., 2000) which classifies benthic species of soft substrates as belonging to one of five ecological groups with different levels of sensitivity or tolerance to environmental stress, allowed to classify as Absent or Negligible the hazard for benthic communities in all the samples collected from the three platforms.
Besides the novelty to summarize in quantitative hazard indices the different typologies of data, the presented WOE approach allowed for the first time further integration between various LOEs for a more complex level of risk assessment. In this respect, the environmental survey in the off-shore field of Campo Rospo represents a unique case study applying a quantitative WOE approach to such a large number of heterogeneous data in a complex monitoring scenario. More than 7000 analytical results from different LOEs could be summarized in a quantitative risk index, classified as Slight for the three platforms: beside the local significance of the specific result, the presented elaboration procedure has greater general importance in terms of communication and risk management, still maintaining scientifically robust info derived from the weighted elaboration of various results.
Another practical advantage of summarizing quantitative hazard indices is the possibility to suggest variations of consolidated monitoring protocols. Considering the substantial homogeneity of Hazard Quotients elaborated for different typologies of results at various distances from the three platforms, the redundancy in the number of analyzed samples was mathematically calculated from the 95% confidence interval of obtained results. By averaging the minimum sample size for each of the three platforms to estimate the HQ values with the required accuracy, it has been calculated that the minimum, still cautionary, number of stations for chemical and benthic analyses should be eight for each platform (instead of the actual 20), sampled with a random design. For bioaccumulation, the analyses of metals in the polychaete H. diversicolor exposed at laboratory conditions do not appear to be of particular utility, especially in already monitored areas where such assays always gave negative results, and where control activities continue to be carried out in terms of chemical, ecotoxicological and benthic communities. At the same time, bioaccumulation and biomarker analyses in mussels allow a reliable characterization of organisms health status, and comparison between natural and transplanted, despite logistically more complex, provides useful elements to better characterize the origin and significance of the potential impacts.
At this stage, the presented WOE model (Sediqualsoft) demonstrated how to summarize and quantitatively integrate huge amounts of results data from the five analyzed LOEs, chemical characterization of sediments, bioavailability, biomarker responses, battery of ecotoxicological assays, status of benthic communities. Such typologies of analyses are typically included in most environmental quality assessments, and the outputs of the present work can thus be of value to be applied in other monitoring scenarios. In addition, this model should not be considered as a final version since, maintaining its general structure, it can be easily updated or adapted to local or national specifics to include additional parameters, normative limits, species, biomarkers or ecotoxicological bioassays. Further, additional LOEs can be developed and added as specific modules, to elaborate other typologies of data, such as those from sea water analyses or from other investigations which may depend on objectives and specificities of other case-studies.
From the overall results, this work was fundamental for drawing the following conclusions: (i) the multidisciplinary WOE approach allowed for the first time the qualitative and quantitative assessment of environmental quality in a complex monitoring scenario, represented by an off-shore field. Different typologies of data could be summarized in terms of HQs and levels of hazard, allowing an easy comparison between platforms, distances, and future temporal trends. (ii) The WOE model allows the interpretation of large data sets of scientifically complex and heterogeneous data, without the logic of tabular comparison, and thus increasing the capability to discriminate between various environmental conditions. It would have been impossible to interpret and compare almost 7000 heterogeneous data from more than 60 sites based on the pass-to-fail or worst result approach. (iii) The application of a scientifically robust elaboration to summarize synthetic hazard indices in a user-friendly format, supports a more comprehensive process of risk assessment and “site-oriented” management decisions.
Ethics Statement
Only invertebrates (bivalves and polychaetes) were analyzed for bioaccumulation and biomarkers: these species are not listed in the National or European guidelines (Directive 2010/63/EU) on the protection of animals used for scientific purposes. Also species used for ecotoxicological bioassays were tested according to standardized protocols and do not require authorization or approval by ethics committee.
Author Contributions
FR conceived the study and responsible for carrying out the study, edited, and reviewed the final version of the manuscript. AN, DF, Gd’E, MB, and FR participated in the sampling activities. AN, MM, DF, MB, MDC, and SG carried out the laboratory analyses. Gd’E carried out the statistical and weighted elaboration of data. FR, SG, and DP discussed the results. AN, Gd’E, and FR wrote the manuscript. All authors approved the final version of the manuscript before submission.
Funding
This work has been partly supported by funds of the monitoring plan of Edison SpA offshore platforms.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmars.2019.00377/full#supplementary-material
References
Bebianno, M. J., Pereira, C. G., Rey, F., Cravo, A., Duarte, D., D’Errico, G., et al. (2015). Integrated approach to assess ecosystem health in harbor areas. Sci. Total Environ. 514, 92–107. doi: 10.1016/j.scitotenv.2015.01.050
Beliaeff, B., and Burgeot, T. (2002). Integrated biomarker response: a useful tool for ecological risk assessment. Environ. Toxicol. Chem. 21, 1316–1322. doi: 10.1002/etc.5620210629
Benedetti, M., Ciaprini, F., Piva, F., Onorati, F., Fattorini, D., Notti, A., et al. (2012). A multidisciplinary weight of evidence approach toward polluted sediments: integrating sediment chemistry, bioavailability, biomarkers responses and bioassays. Environ. Int. 38, 17–28. doi: 10.1016/j.envint.2011.08.003
Benedetti, M., Giuliani, M. E., and Regoli, F. (2015). Oxidative metabolism of chemical pollutants in marine organisms: molecular and biochemical biomarkers in environmental toxicology. Ann. N. Y. Acad. Sci. 1340, 8–19. doi: 10.1111/nyas.12698
Benedetti, M., Gorbi, S., Fattorini, D., Pacitti, D., D’Errico, G., Piva, F., et al. (2014). Environmental hazards from natural seepage: integrated classification of risk from sediment chemistry, bioavailability and biomarkers responses in sentinel species. Environ. Pollut. 185, 116–126. doi: 10.1016/j.envpol.2013.10.023
Bocchetti, R., Fattorini, D., Pisanelli, B., Macchia, S., Oliviero, L., Pilato, F., et al. (2008). Contaminant accumulation and biomarker responses in caged mussels, Mytilus galloprovincialis, to evaluate bioavailability and toxicological effects of remobilized chemicals during dredging and disposal operations in harbour areas. Aquat. Toxicol. 89, 257–266. doi: 10.1016/j.aquatox.2008.07.011
Borja, A., Elliott, M., Andersen, J. H., Berg, T., Carstensen, J., Halpern, B. S., et al. (2016). Overview of integrative assessment of marine systems: the ecosystem approach in practice. Front. Mar. Sci. 3:20. doi: 10.3389/fmars.2016.00020
Borja, A., Elliott, M., Uyarra, M. C., Carstensen, J., and Mea, M. (2017). Editorial: bridging the gap between policy and science in assessing the health status of marine ecosystems. Front. Mar. Sci. 4:32. doi: 10.3389/fmars.2017.00032
Borja, A., Franco, J., and Pérez, V. (2000). A marine Biotic Index to establish the ecological quality of soft-bottom benthos within European estuarine and coastal environments. Mar. Pollut. Bull. 40, 1100–1114. doi: 10.1016/S0025-326X(00)00061-8
Broeg, K., and Lehtonen, K. K. (2006). Indices for the assessment of environmental pollution of the Baltic Sea coasts: integrated assessment of a multi-biomarker approach. Mar. Pollut. Bull. 53, 508–522. doi: 10.1016/j.marpolbul.2006.02.004
Chapman, P. M. (2007). Determining when contamination is pollution – weight of evidence determinations for sediments and effluents. Environ. Inter. 33, 492–501. doi: 10.1016/j.envint.2006.09.001
Dagnino, A., Sforzini, S., Dondero, F., Fenoglio, S., Bona, E., Jensen, J., et al. (2008). A “Weight-of-Evidence” approach for the integration of environmental “Triad” data to assess ecological risk and biological vulnerability. Integr. Environ. Assess. Manag. 4, 314–326. doi: 10.1897/IEAM_2007-067.1
Dauvin, J.-C. (2015). History of benthic research in the English channel: from general patterns of communities to habitat mosaic description. J. Sea Res. 100, 32–45. doi: 10.1016/j.seares.2014.11.005
Dauvin, J. C., and Ruellet, T. (2007). Polychaete/amphipod ratio revisited. Mar. Pollut. Bull. 55, 215–224. doi: 10.1016/j.marpolbul.2006.08.045
Davies, I. M., and Vethaak, A. D. (2012). Integrated monitoring of chemicals and their effects. ICES Coop. Res. Rep. 315:227.
Dolenec, T., Faganeli, J., and Pirc, S. (1998). Major, minor and trace elements in surficial sediments from the open Adriatic sea: a regional geochemical study. Geol. Croat. 51, 47–58.
Etiope, G., Panieri, G., Fattorini, D., Regoli, F., Vannoli, P., Italiano, F., et al. (2014). A thermogenic hydrocarbon seep in shallow Adriatic Sea (Italy): gas origin, sediment contamination and benthic foraminifera. Mar. Petrol. Geol. 57, 283–293. doi: 10.1016/j.marpetgeo.2014.06.006
Fattorini, D., Notti, A., Di Mento, R., Cicero, A. M., Gabellini, M., Russo, A., et al. (2008). Seasonal, spatial and inter-annual variations of trace metals in mussels from the Adriatic Sea: a regional gradient for arsenic and implications for monitoring the impact of off-shore activities. Chemosphere 72, 1524–1533. doi: 10.1016/j.chemosphere.2008.04.071
Gorbi, S., Virno Lamberti, C., Notti, A., Benedetti, M., Fattorini, D., Moltedo, G., et al. (2008). An ecotoxicological protocol with caged mussels, Mytilus galloprovincialis, for monitoring the impact of an offshore platform in the Adriatic sea. Mar. Environ. Res. 65, 34–49. doi: 10.1016/j.marenvres.2007.07.006
HELCOM (2010). Ecosystem Health of the Baltic Sea 2003–2007: HELCOM Initial Holistic Assessment. Helsinki: HELCOM.
Hylland, K. (2006). Biological effects in the management of chemicals in the marine environment. Mar. Pollut. Bull. 53, 614–619. doi: 10.1016/j.marpolbul.2006.08.010
Hylland, K., Burgeot, T., Martínez-Gómez, C., Lang, T., Robinson, C. D., Svavarsson, J., et al. (2017). How can we quantify impacts of contaminants in marine ecosystems? ICON Project. Mar. Environ. Res. 124, 2–10. doi: 10.1016/j.marenvres.2015.11.006
ISPRA (2017). Saggio di fecondazione e saggio di sviluppo embrionale con il riccio di mare Paracentrotus lividus (Lamarck) (Echinodermata: Echinoidea). Rome: ISPRA.
Lehtonen, K. K., Sundelin, B., Lang, T., and Strand, J. (2014). Development of tools for integrated monitoring and assessment of hazardous substances and their biological effects in the Baltic Sea. AMBIO 43, 69–81. doi: 10.1007/s13280-013-0478-3
Linkov, I., Loney, D., Cormier, S., Satterstrom, F. K., and Bridges, T. (2009). Weight-of-evidence evaluation in environmental assessment: review of qualitative and quantitative approaches. Sci. Total Environ. 407, 5199–5205. doi: 10.1016/j.scitotenv.2009.05.004
Lyons, B. P., Bignell, J. P., Stentiford, G. D., Bolam, T. P. C., Rumney, H. S., Bersuder, P., et al. (2017). Determining good environmental status under the marine strategy framework directive: case study for descriptor 8 (chemical contaminants). Mar. Environ. Res. 124, 118–129. doi: 10.1016/j.marenvres.2015.12.010
Lyons, B. P., Thain, J. E., Hylland, K., Davis, I., and Vethaak, A. D. (2010). Using biological effects tools to define good environmental status under the marine strategy framework directive. Mar. Pollut. Bull. 60, 1647–1651. doi: 10.1016/j.marpolbul.2010.06.005
Marigómez, I., Garmendia, L., Soto, M., Orbea, A., Izagirre, U., and Cajaraville, M. P. (2013). Marine ecosystem health status assessment through integrative biomarker indices: a comparative study after the Prestige-oil spill “Mussel Watch”. Ecotoxicology 22, 486–505. doi: 10.1007/s10646-013-1042-4
Mestre, N. C., Rocha, T. L., Canals, M., Cardoso, C., Danovaro, R., Dell’Anno, A., et al. (2017). Environmental hazard assessment of a marine mine tailings deposit site and potential implications for deep-sea mining. Environ. Pollut. 228, 169–178. doi: 10.1016/j.envpol.2017.05.027
Mistri, M., and Munari, C. (2008). BITS: a SMART indicator for soft-bottom, non-tidal lagoons. Mar. Pollut. Bull. 56, 587–599. doi: 10.1016/j.marpolbul.2007.12.002
Moore, M. N., Depledge, M. H., Readman, J. W., and Leonard, P. D. R. (2004). An integrated biomarker-based strategy for ecotoxicological evaluation of risk in environmental management. Mutat. Res. 18, 247–268. doi: 10.1016/j.mrfmmm.2004.06.028
Moore, M. N., Icarus Allen, J., and McVeigh, A. (2006). Environmental prognostics: an integrated model supporting lysosomal stress responses as predictive biomarkers of animal health status. Mar. Environ. Res. 61, 278–304. doi: 10.1016/j.marenvres.2005.10.005
Muxika, I., Borja, Á., and Bald, J. (2007). Using historical data, expert judgement and multivariate analysis in assessing reference conditions and benthic ecological status, according to the European Water Framework Directive. Mar. Pollut. Bull. 55, 16–29. doi: 10.1016/j.marpolbul.2006.05.025
Newman, M. C., and Clements, W. H. (2007). Ecotoxicology: A Comprehensive Treatment, 1st Edition. 880 pag, ISBN 9781420005011. Boca Raton: CRC Press.
OSPAR (2013). Technical Annex on Supporting Parameters for Biological Effects Measurements in Fish and Mussels. Background Document and Technical Annexes for Biological Effects Monitoring, Update 2013, Monitoring and Assessment Series 589/2013. ISBN 978-1-909159-22-8. Paris: OSPAR.
Pittura, L., Avio, C. G., d’Errico, G., Keiter, S., and Cormier, B. (2018). Microplastics as vehicles of environmental PAHs to marine organisms: combined chemical and physical hazards to the Mediterranean mussels, Mytilus galloprovincialis. Front. Mar. Sci. 5:103. doi: 10.3389/fmars.2018.00103
Piva, F., Ciaprini, F., Onorati, F., Benedetti, M., Fattorini, D., Ausili, A., et al. (2011). Assessing sediment hazard through a Weight of Evidence approach with bioindicator organisms: a practical model to elaborate data from sediment chemistry, bioavailability, biomarkers and ecotoxicological bioassays. Chemosphere 83, 475–485. doi: 10.1016/j.chemosphere.2010.12.064
Regoli, F., Frenzilli, G., Bocchetti, R., Annarumma, F., Scarcelli, V., Fattorini, D., et al. (2004). Time-course variations of oxyradical metabolism, DNA integrity and lysosomal stability in mussels, Mytilus galloprovincialis, during a field translocation experiment. Aquat. Toxicol. 68, 167–178. doi: 10.1016/j.aquatox.2004.03.011
Regoli, F., and Giuliani, M. E. (2014). Oxidative pathways of chemical toxicity and oxidative stress biomarkers in marine organisms. Mar. Environ. Res. 93, 106–117. doi: 10.1016/j.marenvres.2013.07.006
Regoli, F., Giuliani, M. E., Benedetti, M., and Arukwe, A. (2011). Molecular and biochemical biomarkers in environmental monitoring: a comparison of biotransformation and antioxidant defense systems in multiple tissues. Aquat. Toxicol. 105, 56–66. doi: 10.1016/j.aquatox.2011.06.014
Regoli, F., Pellegrini, D., Cicero, A. M., Nigro, N., Benedetti, M., Gorbi, S., et al. (2014). A multidisciplinary weight of evidence approach for environmental risk assessment at the Costa Concordia wreck: integrative indices from Mussel Watch. Mar. Environ. Res. 96, 92–104. doi: 10.1016/j.marenvres.2013.09.016
Semenzin, E., Critto, A., Rutgers, M., and Marcomini, A. (2008). Integration of bioavailability, ecology and ecotoxicology by three lines of evidence into ecological risk indexes for contaminated soil assessment. Sci. Total Environ. 389, 71–86. doi: 10.1016/j.scitotenv.2007.08.032
Sigovini, M., Keppel, E., and Tagliapietra, D. (2013). M-AMBI revisited: looking inside a widely-used benthic index. Hydrobiologia 717, 41–50. doi: 10.1007/s10750-013-1565-y
Simboura, N., and Zenetos, A. (2002). Benthic indicators to use in Ecological Quality classification of Mediterranean soft bottom marine ecosystems, including a new Biotic Index. Mediterr. Mar. Sci. 3, 77–111. doi: 10.12681/mms.249
Spagnolo, A., Punzo, E., Santelli, A., Scarcella, G., Strafella, P., Grati, F., et al. (2014). Offshore platforms: comparison of five benthic indicators for assessing the macrozoobenthic stress levels. Mar. Pollut. Bull. 82, 55–65. doi: 10.1016/j.marpolbul.2014.03.023
Tobiszewski, M., and Namieśnik, J. (2012). PAH diagnostic ratios for the identification of pollution emission sources. Environ. Pollut. 162, 110–119. doi: 10.1016/j.envpol.2011.10.025
Vethaak, A. D., Davies, I. M., Thain, J. E., Gubbins, M. J., Martínez-Gómez, C., Robinson, C. D., et al. (2017). Integrated indicator framework and methodology for monitoring and assessment of hazardous substances and their effects in the marine environment. Mar. Environ. Res. 124, 11–20. doi: 10.1016/j.marenvres.2015.09.010
Viarengo, A., Lowe, D., Bolognesi, C., Fabbri, E., and Koehler, A. (2007). The use of biomarkers in biomonitoring: a 2-tier approach assessing the level of pollutant-induced stress syndrome in sentinel organisms. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 146, 281–300. doi: 10.1016/j.cbpc.2007.04.011
Keywords: off-shore platforms, risk assessment, WOE integration, multidisciplinary approaches, monitoring
Citation: Regoli F, d’Errico G, Nardi A, Mezzelani M, Fattorini D, Benedetti M, Di Carlo M, Pellegrini D and Gorbi S (2019) Application of a Weight of Evidence Approach for Monitoring Complex Environmental Scenarios: the Case-Study of Off-Shore Platforms. Front. Mar. Sci. 6:377. doi: 10.3389/fmars.2019.00377
Received: 22 December 2018; Accepted: 17 June 2019;
Published: 08 July 2019.
Edited by:
Thierry Burgeot, Institut Français de Recherche pour l’Exploitation de la Mer (IFREMER), FranceReviewed by:
Gabriela Verónica Aguirre-Martínez, Universidad Arturo Prat, ChileHans Uwe Dahms, Kaohsiung Medical University, Taiwan
Copyright © 2019 Regoli, d’Errico, Nardi, Mezzelani, Fattorini, Benedetti, Di Carlo, Pellegrini and Gorbi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Francesco Regoli, Zi5yZWdvbGlAdW5pdnBtLml0
 Francesco Regoli
Francesco Regoli Giuseppe d’Errico
Giuseppe d’Errico Alessandro Nardi
Alessandro Nardi Marica Mezzelani1
Marica Mezzelani1 Daniele Fattorini
Daniele Fattorini Maura Benedetti
Maura Benedetti Marta Di Carlo
Marta Di Carlo Stefania Gorbi
Stefania Gorbi