- 1College of Marine Sciences, Shanghai Ocean University, Shanghai, China
- 2National Engineering Research Center for Oceanic Fisheries, Shanghai Ocean University, Shanghai, China
- 3Ministry of Education Key Laboratory of Sustainable Exploitation of Oceanic Fisheries Resources, Shanghai Ocean University, Shanghai, China
- 4Collaborative Innovation Center for Distant Water Fisheries, Shanghai, China
- 5Laboratory E3M, Higher National Polytechnic Institute, University of Douala, Douala, Cameroon
A rising trend in catches of non-targeted species has recently been observed in major fisheries including tuna longline fisheries, yet most of these species are unmanaged. Given their importance to local economies and sustainable livelihoods in many coastal countries, there is a need to provide plans for their management. However, most non-targeted species are data-limited which hampers the use of conventional assessment methods. This study applied a novel data-limited length-based Bayesian biomass estimator (LBB) method to assess the stocks of five species from the Atlantic and Pacific Oceans. Estimates of growth, length at first capture and present relative biomass (B/B0, B/BMSY) of these species were gotten from length-frequency (LF) data. Of the ten populations (5 species from two regions) assessed, one has collapsed, one grossly overfished, and three overfished. Six populations had the ratio of mean lengths at first capture (Lc) on the mean length at first capture, which maximizes the catch and biomass (Lc_opt) greater than unity, indicating the presence of large-sized specimens in the populations. Two species faced intense fishing pressure in the Atlantic while one population collapsed in the Pacific Ocean. Our results indicate that even non-targeted pelagic can be prone to over-exploitation. Therefore, there is an urgent need for stakeholders and fisheries managers to focus on improving fishery statistics and to conduct periodic monitoring of stock status indicators for non-target species.
Introduction
Most commercial fisheries target species of high economic values such as tunas and swordfish. However, the fishing gears (longlines, purse seines) used to capture fish specimens do not only capture targeted species but also harvest non-targeted (bycatch) species including sharks, small pelagic species (scombrids) and many others. Some of these bycatches are at times discarded or retained without recording their catch or biological information. Although these species are of low value compared to the highly targeted species, they play an important role in the subsistence (sustenance and food security) and income-generating means to the local populations (Lynch et al., 2016). These non-targeted species also play an important role in regulating the marine trophic level and serve as food for most targeted species.
As indicated in the last FAO (2019) report, only around 12% of the world’s fisheries are properly managed or have sophisticated stock assessments for management purpose. These non-targeted small pelagic are most often considered to be highly abundant and assumed not suffering from population decline given that they reproduce and grow at a faster rate. As a consequence, little or no emphasis is usually put in place to monitor (collect data) and assess their stocks. Robust assessment models demand long-time series of catch-at-age, abundance indices, and fishing effort data which are not available in data-limited fisheries (Methot and Wetzel, 2013; Francis et al., 2016). However, length data are most often the only reported data for most bycatch species in longline vessels targeting tunas and swordfish. Given the lack of data reported for these fisheries, conventional stock assessment approaches cannot be used to assess their stocks.
Consistent time series on historical total catch data is difficult to get for most un-targeted species, while sampling length-frequency (LF) data from captured specimens is easier to record. LF data have been used in fishes to assess their stocks (Munro, 1983; Pauly, 1998; Hordyk et al., 2015; Nadon et al., 2015; Froese et al., 2017, 2018; Rudd and Thorson, 2018). The most abundant un-targeted teleosts in most longline fishing vessels’ catch, particularly that of the Chinese tuna longline vessels operating in all oceanic waters selected for this study are Acanthocybium solandri (Wahoo), Taractes rubescens (Black/dagger Pomfret), Taractichthys steindachneri (Sickle Pomfret), Alepisaurus ferox (Long Snouted Lancetfish), and Coryphaena hippurus (Common Dolphinfish). These five species play important roles in regulating the marine trophic level and also serve as food and income sources for local fishermen in coastal states. They mostly inhabit epipelagic zones in oceanic waters.
Acanthocybium solandri is found to at least 340 m, estimated longevity at 9 years and feeds mostly on small fishes and squids (McBride et al., 2008; Gao et al., 2020). The heaviest captured specimens was 96.4 kg and the largest more than 210 cm recorded in 2006 from the South Pacific Ocean (Zischke, 2012). Kindong et al. (2020) recorded the largest (92 cm FL) and heaviest (17 kg) Taractichthys steindachneri caught in the Pacific Ocean. Smith (1986) reported that this species could live up to 8 years and could be found in waters to at least 700 m. Taractes rubescens is recorded in depths to up to 600 m and has a maximum reported size of 89 cm TL (Jawad et al., 2014; Froese and Pauly, 2020). They feed on small fishes and squids. Alepisaurus ferox can be found in depths ranging from just beneath the surface to 1,830 m. They feed on fish, cephalopods, tunicates and crustaceans, however, diet can vary according to region. The maximum reported length for this species is 215 cm TL and a maximum weight of 9 kg (Froese and Pauly, 2020). Coryphaena hippurus also referred to as “Mahi-mahi” is a marine, brackish, pelagic–neritic and oceanodromous species found in depths ranging between 0 and 85 m and having a maximum reported age of 4 years (Froese and Pauly, 2020; Moltó et al., 2020). Its maximum reported size is 210 cm and reported weight of 40 kg caught in Costa Rica (Froese and Pauly, 2020). They feed on smaller fishes, zooplankton, crustaceans, and squid. This species is of high commercial value and are highly harvested in its range. All five species are batch spawners and are important prey items of tunas, marlins and other large pelagic predators including sharks. They are mainly harvested in recreational and longline fishing vessels, are highly migratory species, and are appreciated as food fish (International Union for Conservation of Nature [IUCN], 2020).
Despite that these small pelagic teleost mostly occur in tropical and subtropical areas of oceanic waters, assessment of their stocks are rare except for Acanthocybium solandri (Zischke and Griffiths, 2015; Pons et al., 2019) and Taractichthys steindachneri (Kindong et al., 2020). A recent report by the FAO (2019) shows a catch drop of Coryphaena hippurus from 125 k tons in 2015 to 85 k tons in 2017 while catches of Acanthocybium solandri slightly increased, around 5,000 tons in 2017. The catches were obtained from artisanal fishers targeting both species and as bycatch reports in some longline vessels; catches for the other three species were not reported (FAO, 2019). The lack of data reporting for most of these species is attributed to the fact that most are been discarded in longline fishing vessels or retained but under-reported (for both artisanal and industrial fisheries). However, given the interest shown by most local fishermen targeting these species, and also the importance of these species to the local communities, it is crucial to perform assessments for these species using available LF data to assure the sustainability of these fishery resources.
A bunch of data-limited methods that use only length or catch data has recently been developed and their performances tested for managing fisheries of limited data in some areas worldwide (Nadon et al., 2015; Prince et al., 2015; Chong et al., 2019; Pons et al., 2019, 2020). Froese et al. (2018) recently developed a length-based Bayesian biomass (LBB) estimation approach that can be applied to analyze LF data for exploited fishery populations where all appropriate parameters are estimated through Bayesian Monte Carlo Markov Chain (MCMC). This approach assumes constant recruitment, growth and mortality, and can provide the best available information if the LF data is representative of the exploited stock, including data-limited fisheries. The LBB method has recently been applied in the East Asian waters to assess some data-limited stocks (Liang et al., 2020; Wang et al., 2020; Zhang et al., 2020). Thus, even in most non-targeted species which are mostly data-limited, the LBB approach may be applied by fishery managers for precautionary management of these species.
In this study, the LBB method was applied to 10 populations of non-targeted pelagic teleost captured by the Chinese longline fishing vessels operating in the tropical waters of the Atlantic and the Pacific Oceans. This approach was applied to these species to determine life-history parameters and explore the level of biomass depletion caused by fishing on the populations of these species.
Materials and Methods
Precisely in the tropical eastern Atlantic (14°03′N–02°41′N, 33°12′W–21°74′W), two voyages were operated by two Chinese longline vessels targeting tuna and swordfish between June 2019 and May 2020. The power and tonnage of each vessel are 1,000 KW and 619 t. The smaller vessel measured 50 m and larger 54.5 m. In the Central Pacific Ocean (14°03′N–02°41′N, 33°12′W–21°74′W), 8 voyages were also effectuated by 8 Chinese longline vessels targeting tunas and tuna-like species throughout the year 2015. Among these eight vessels operating in the Pacific Ocean, the power, tonnage and size of the smallest vessel was 330 KW, 190 t and 36.6 m, respectively. The largest vessel measured 49 m, a tonnage of 769 t and power of 928 KW. Trained observers deployed on-board these vessels by the Chinese Longline Fisheries Observer Programme had specific tasks to record data (catch and biological) for targeted species and also some major bycatch species.
Length composition data were collected for five bycatch species in these commercial longline fisheries including 1 Scombridae (Acanthocybium solandri, Wahoo), 1 Coryphaenidae (Coryphaena hippurus, Common Dolphinfish), 1 Alepisauridae (Alepisaurus ferox, Long Snouted Lancetfish), and two members of the Bramidae family [Taractes rubescens (Black Pomfret) and Taractichthys steindachneri (Sickle Pomfret)]. For each specimen, the length and when possible, weights were also measured. However, the easiest to measure was the fork length (FL). Therefore, while onboard the vessels, the FL for each captured specimen was measured in a straight line to the nearest lower cm using a measuring tape. Later, specimens were identified to species level, and scientific and common names attributed as referenced in FishBase (Froese and Pauly, 2020). The FL of all caught individuals of the five species were measured during the 10 voyages and used for subsequent analyses. The total number of measured specimens for all five species ranged from 76 to 1,964, with sizes including small-and large-sized individuals, representing a wide size range (Table 1 and Supplementary Tables S2, S3).
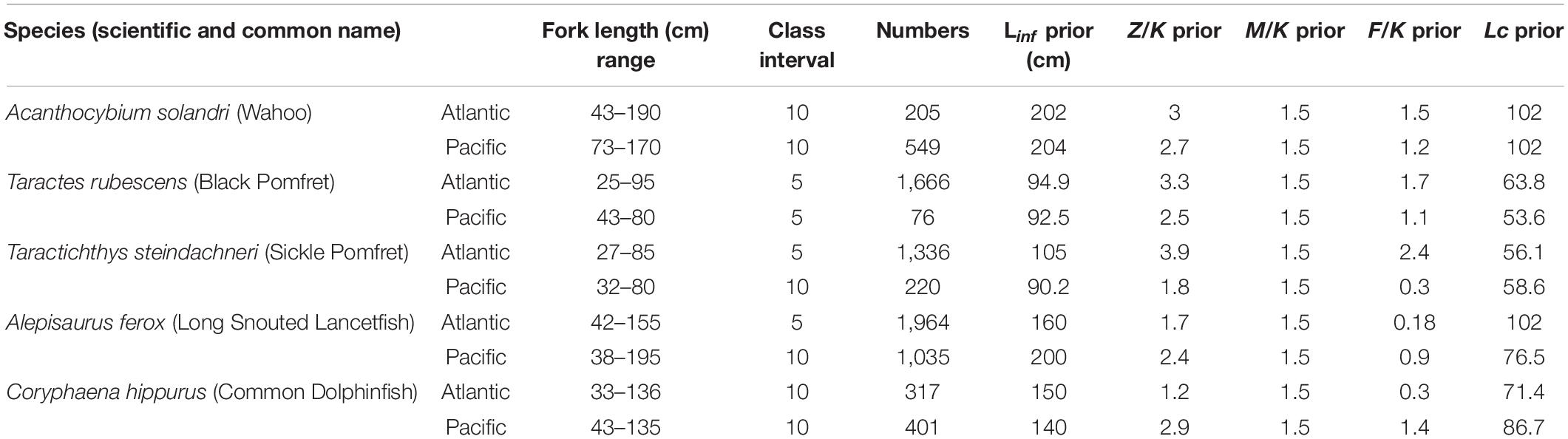
Table 1. Basic information and priors of five small pelagic species from the Atlantic and Pacific Oceans used in the present study.
LBB is a new simple and fast approach that uses a Bayesian Monte Carlo Markov Chain (MCMC) method for estimating relative stock size using length-frequency (LF) data (Froese et al., 2018). The LBB method requires only LF data of a fishery representing the true population structure as input since it uses pre-specified priors on parameters. However, to reduce uncertainty in LBB results, users may manually input required prior parameters including the asymptotic length (Linf), mean length at first capture (Lc), relative natural mortality (M/K) if they have good estimates of these parameters from independent studies (Froese et al., 2018). The LBB method was selected for this analysis among other length-based methods because of its ease in handling and inputting data, and also of the need to explore this new approach in fisheries with limited data. Furthermore, biomass estimated from simulated data through LBB was not significantly different from the true values obtained from conventional stock assessments (Froese et al., 2018).
For the present study, LF data of each fish population throughout the survey period constituting a year were used as input in the LBB software. The length-frequency data obtained for each species had specimens collected for at least 1 year with wide size ranges representing the population structure (Supplementary Tables S2, S3). Furthermore, to reduce uncertainty in results, species having asymptotic lengths obtained from prior studies were also included in the input information database (Table 1 and Supplementary Tables S2, S3).
Details regarding the development of formulae incorporated in the LBB are given in Froese et al. (2018). This study just elaborated on a couple of formulae for this method. LBB assumes that growth in size follows a von Bertalanffy growth function (VBGF; von Bertalanffy, 1938; Pauly, 1998).
where Lt is the length at age t, K is the growth rate (year–1), and t0 is the theoretical age at zero-length (Froese et al., 2018). When the fish specimens are fully selected by the gear used, the curvature of the right side of catch samples is a function of total mortality (Z = M + F) relative to K, expressed by the equation:
where NL is the number of survivors to the length L, NLstart is the number at length Lstart with full selection, from which all specimens encountering the gear are retained. At an unfished state, Z/K becomes M/K, Lstart is zero, and NLstart can be set to 1. The lengths affected by partial selection are, for each species, a function of the fishing gear as shown by the ogive function expressed by the equation below:
where SL is the fraction of individuals that are taken by the gear at length L, and r describes the steepness of the ogive (Quinn and Deriso, 1999; Froese et al., 2018). The framework that approximates the stock status from the estimates of parameters Linf, M/K, F/K and Lc are represented by two equations related to parameters Lopt and Lc_opt (Froese et al., 2016):
Lopt represents the length at which the biomass of the cohort is at its peak. Based on Eq. (4) and taking into consideration the fishing pressure (F/M), the mean length at first capture, which maximizes the catch and biomass (Lc_opt), is gotten from:
The obtained estimates of Lc_opt can later be used to evaluate a proxy for the relative biomass that can produce MSY for a given fishery (Froese et al., 2018).
In this study, the (M/K) value was set to 1.5 as per Hordyk et al. (2015) and Froese et al. (2018), and we let the model estimate Lc and Linf. The Z/K prior was generated based on the equation (Beverton and Holt, 1957; Quinn and Deriso, 1999):
F/K prior equals to Z/K−M/K and the relative fishing mortality F/M = (F/K)/(M/K). Z/K is the ratio of the total mortality rate Z to the somatic growth rate.
Indexes of yield-per-recruit (Y’/R) and catch per unit effort per recruit (CPUE’/R) defined by (Beverton and Holt, 1966) can be evaluated as a function of Lc/Linf, F/K, M/K, and relative fishing mortality F/M as developed in Froese et al. (2018). Hence, given that CPUE is proportionate to biomass in the exploited phase of the population, the derived index of CPUE’/R represents an index of the exploited biomass per recruit B’/R.
In the case where no fishing occurs (F = 0), the relative biomass in the exploited phase of the population is expressed as:
where represents the exploitable fraction (> Lc) of the unfished biomass (B0). According to Beverton and Holt (1966), an index of relative biomass depletion for the exploited part of the population B/B0 can be obtained by:
Fixing parameters F/M = 1 and Lc = Lc_opt and re-calculating expressions for Y’/R, CPUE’/R,, and B/B0, we obtain a proxy for relative biomass at MSY (BMSY/B0). Hence, the current relative stock size (B/BMSY) was calculated, and their estimates converted to define the status of each fishery stock (Palomares et al., 2018). Given the estimated B/B0 and B/BMSY values, the stocks were classified based on the value of B/BMSY as in Palomares et al. (2018); healthy stock status was assigned where B/BMSY > 1.1; a slightly overfished status where 0.8 < B/BMSY ≤ 1.1; overfished where 0.8 ≤ B/BMSY ≤ 0.5; grossly overfished status where 0.5 < B/BMSY ≤ 0.2; and for a collapsed status B/BMSY < 0.2. Froese et al. (2018) also defined B/B0 (0.4–0.5) as the reference limit of a stock’s biomass.
The estimated parameters translate to catch number and catch size rules: if the relative stock size B/B0 < BMSY/B0, catches had better be reduced and if the mean length at first capture Lc is < Lc_opt, fishing would better start at larger sizes. With these two basic and simple rules provided, the application of LBB may directly be used in fisheries with data-limited situations for provisional management of their populations. The length-frequency data constitute wide size ranges of each fish population obtained from the 10 commercial longline vessels operating in the Atlantic and Pacific Oceans were used as input in the LBB package (Table 1 and Supplementary Tables S2, S3). All analyses performed in this study were done using the 33.0 version of the LBB software installed as a package in the R-core environment.
Results
Results obtained from the LBB analyses show that this model fits well to all 10 populations from the Atlantic and Pacific Oceans (Figures 1,2 and Table 2). Values of B/B0 presented by the LBB model for the 10 populations ranged from 0.068 to 0.87 and 0.21 to 11 for F/K (Table 2). Both proxies of B/B0 and B/BMSY defined earlier were considered as the minimum limits of appropriate stock sizes. The results per species using LBB are presented as follows.
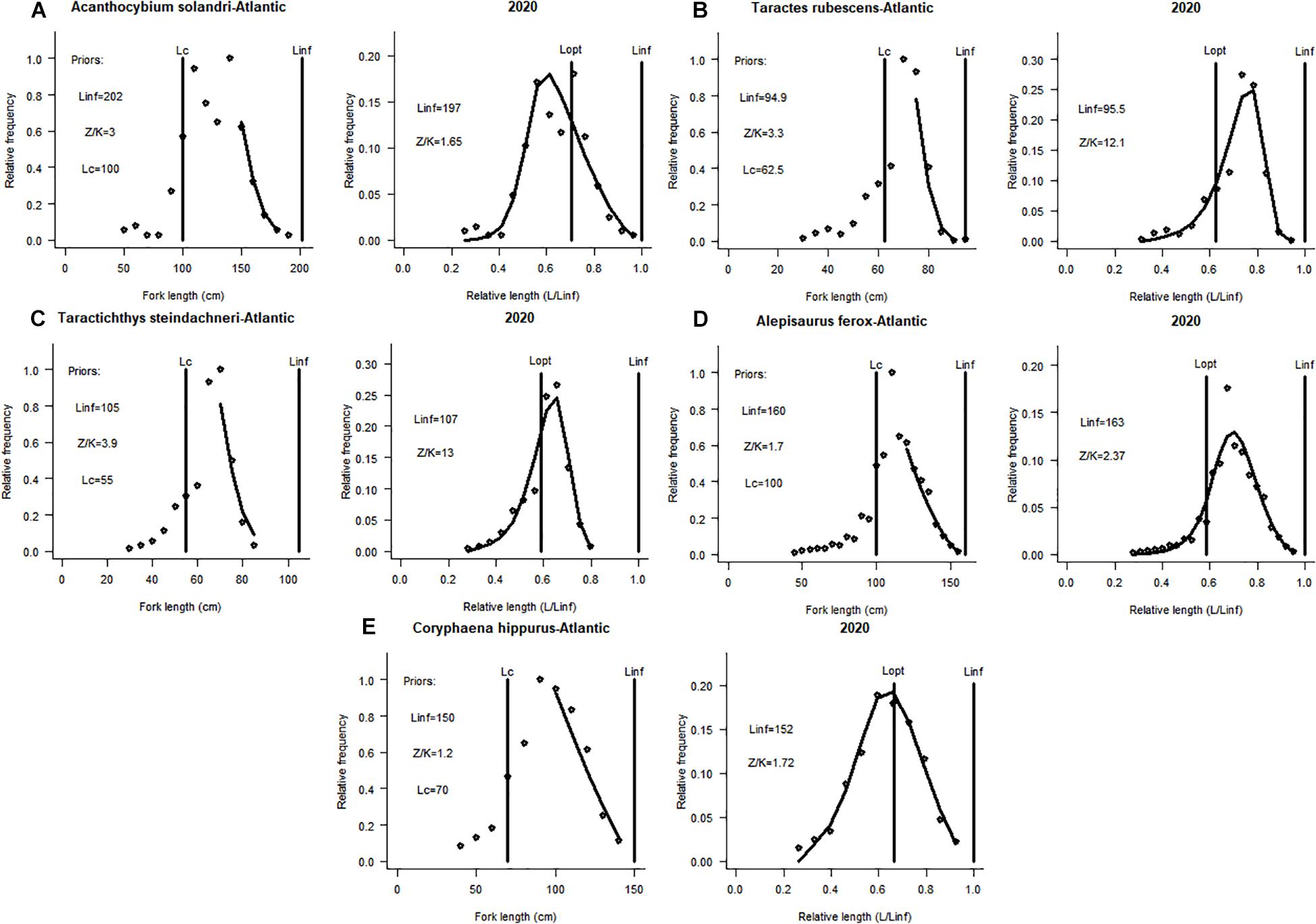
Figure 1. Application of the LBB method to five non-target species (with available length-frequency data) caught by the Chinese tuna longline fisheries vessels operating in the Atlantic Ocean (see also Table 1 and Supplementary Materials). LBB plots for (A) Acanthocybium solandri (B) Taractes rubescens (C) Taractichthys steindachneri (D) Alepisaurus ferox (E) Coryphaena hippurus.
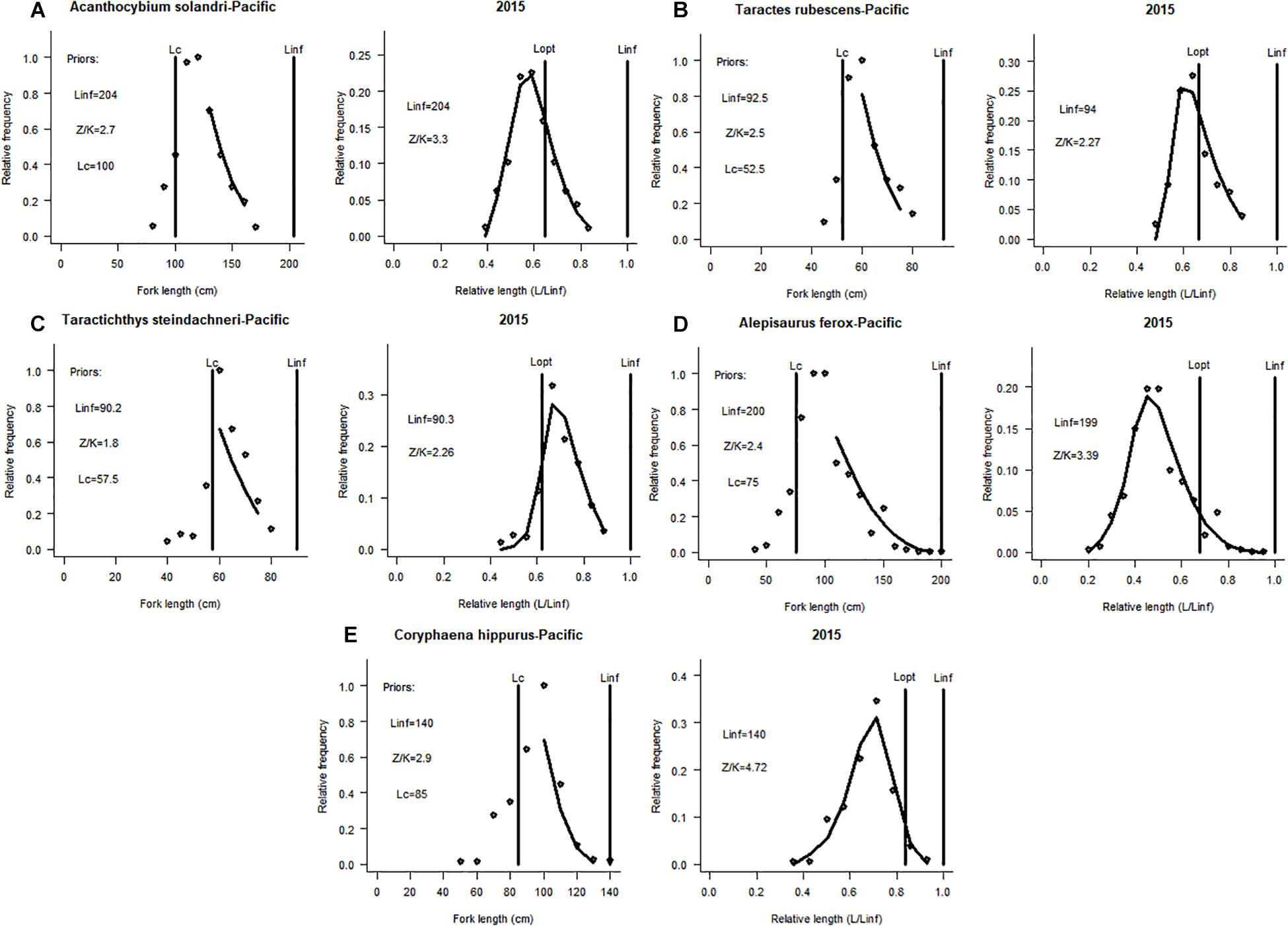
Figure 2. Application of the LBB method to five non-target species (with available length-frequency data) caught by the Chinese tuna longline fisheries vessels operating in the Pacific Ocean (see also Table 1 and Supplementary Materials). LBB plots for (A) Acanthocybium solandri (B) Taractes rubescens (C) Taractichthys steindachneri (D) Alepisaurus ferox (E) Coryphaena hippurus.
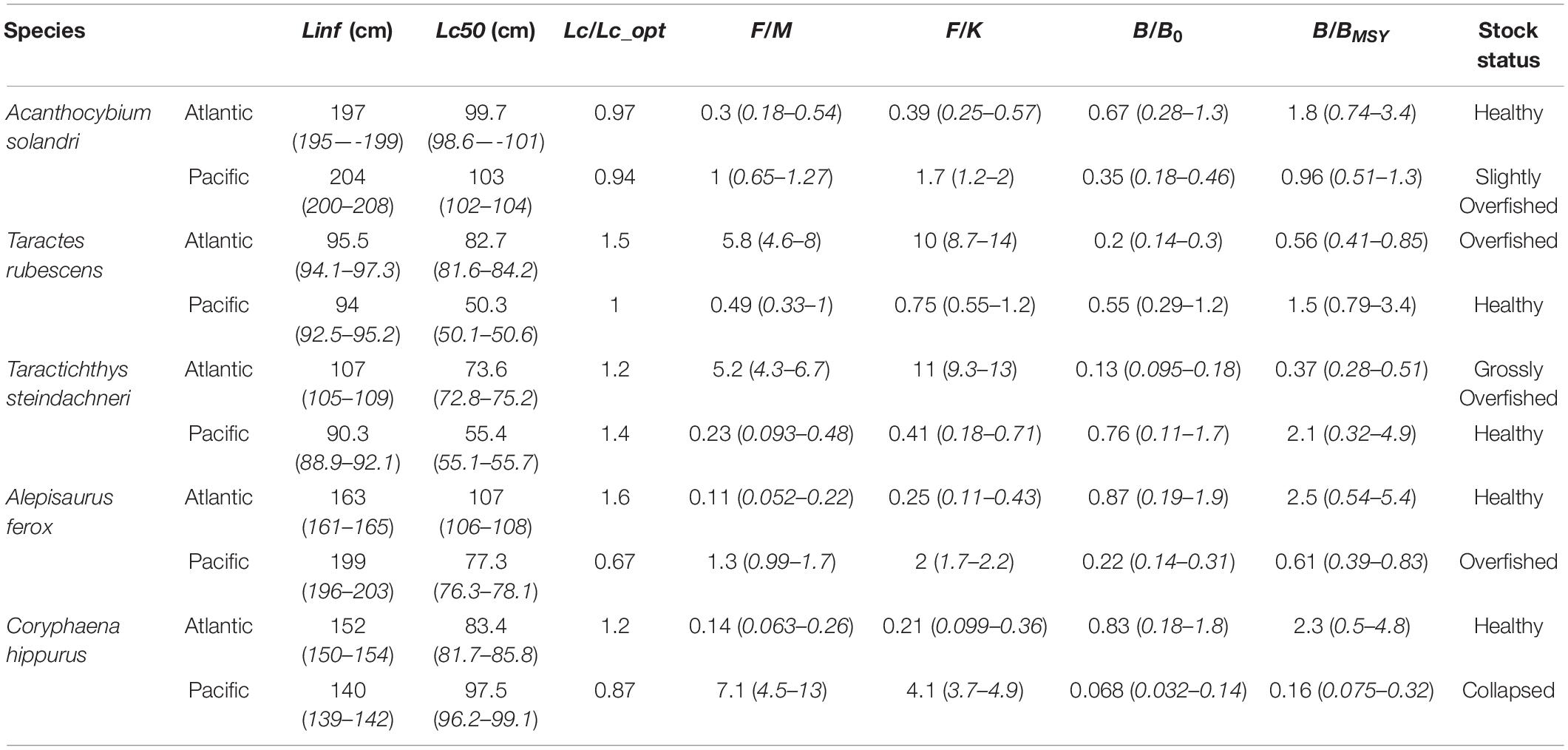
Table 2. Fishery statuses of the five pelagic species assessed in the Atlantic and Pacific Oceans presented by LBB [Linf, Lc50 and Lc/Lc_opt, F/M, F/K, and B/B0 and B/BMSY and their respective 95% confidence intervals (numbers in brackets)].
Acanthocybium solandri (Wahoo)
The stock of this species was in a good state in the tropical eastern Atlantic ocean (F/M = 0.3, B/B0 = 0.67, and B/BMSY = 1.8), meanwhile, in the central Pacific Ocean, its stock was witnessing slight overfishing (F/M = 1, B/B0 = 0.35 and B/BMSY = 0.96). However, the Lc/Lc_opt for this species in both oceans were below unity, signifying a truncated length structure and fishing of small specimens (Figures 1, 2A).
Taractes rubescens (Black Pomfret)
The stock of this pelagic species was under increasing fishing pressure (F/M = 5.8) and its standing biomass was declining (B/B0 = 0.2 and B/BMSY = 0.56) in the Atlantic. In the Pacific, its stock was relatively stable and fishing has less effect on its stock (Table 2). Also, for this species in both oceans, the Lc/Lc_opt ≥ 1 suggesting the presence of larger specimens (Figures 1, 2B).
Taractichthys steindachneri (Sickle Pomfret)
This study suggests that the stock of this benthopelagic species distributed globally was seriously been fished down (F/M = 5.2) in the Atlantic; and the ratios B/B0 = 0.13 and B/BMSY = 0.37 were very low, indicating a sharp decline of its standing biomass. Whereas, the population of the same species was healthy in the Pacific. However, the values obtained for Lc/Lc_opt for this species in both oceans suggested the presence of large specimens in both stocks (Table 2 and Figures 1, 2C).
Alepisaurus ferox (Long Snouted Lancetfish)
This species mostly found in deep-waters as compared to the other species had a stable stock in the Atlantic, less influenced by fishing, with the presence of large specimens in its population (B/B0 = 0.87, B/BMSY = 2.5, F/M = 0.11 and Lc/Lc_opt = 1.6). In the Pacific, its stock was overfished and low biomass (F/M = 1.3, B/B0 = 0.22, and B/BMSY = 0.61). The Lc/Lc_opt for this species in the Pacific was below unity, indicating the capture of many small specimens (Figures 1, 2D).
Coryphaena hippurus (Common Dolphinfish)
This species found in oceanic waters worldwide was not under fishing pressure in the Atlantic with healthy biomass and stable stock state (Table 2). Contrarily, in the central region of the Pacific, the estimate of F/M = 7.1 showed that the Common Dolphinfish was under serious fishing pressure, meanwhile the estimate of B/B0 = 0.068 which indicates that the standing biomass of this species has depleted greatly (Figure 2E).
Discussion
Stocks with very limited or unreliable catch data can be assessed using the LBB method. However, the greatest limitation of this approach remains the fact that for an exploited stock, the length-frequency data used must be representative of that stock (Froese et al., 2018). For the present work, all the LF data was obtained from a commercial catch gear for at least 1 year and represented all possible length classes of the species analyzed, thus meeting all LBB requirements. Furthermore, for all 10 populations (5 species × 2 oceans) the LF samples were suitable since their resulting fits presented asymmetric patterns as prescribed in Froese et al. (2018).
The two species in the bramidae family (pomfrets) attained larger sizes in the Atlantic than in the Pacific, which may explain why both fisheries were under intense fishing pressure in the Atlantic Ocean (Table 2). The populations of Acanthocybium solandri, Alepisaurus ferox, and Coryphaena hippurus witnessed intense fishing pressure in the Pacific, reaching maximum lengths earlier than those of the Atlantic. Still in the Pacific, LBB results show that the stock of Coryphaena hippurus collapsed in this same region. Six out of the 10 populations (5 species × 2 oceans) had ratios Lc/Lc_opt ≥ 1 indicating the presence of a good number of large specimens.
Apart from Acanthocybium solandri and Taractichthys steindachneri, no assessment method had previously been applied to the other three stocks (Supplementary Table S1). An ecological risk assessment was applied to all these species in the Western and Central Pacific Ocean (WCPO) to assess the effect of the tuna longline fishery on these populations (Kirby, 2006; Kwon et al., 2009). Reports from this assessment corroborate with the findings obtained in our work; they reported that Acanthocybium solandri, Alepisaurus ferox, and Coryphaena hippurus were under intense fishing threat except for Taractichthys steindachneri and Taractes rubescens. Reports by Zischke and Griffiths (2015) and Kindong et al. (2020) were consistent with the present result; they used different per-recruit models to assess the stocks of Acanthocybium solandri and Taractichthys steindachneri in the Pacific. Kirby (2006) and Kwon et al. (2009) reported that the stock of Coryphaena hippurus was at higher exploitation risk, corroborated by our findings, which showed a collapsed state of this stock in the Pacific. Although no conclusion was reached for this species in the Eastern Pacific Ocean, Aires-da-Silva et al. (2016) still reported that current catches were near the maximum sustainable yield (MSY) estimates from the stock synthesis (SS3) assessment results. These authors insisted on the prioritization of collecting new data for future assessment of Coryphaena hippurus. In the Atlantic Ocean, all except Acanthocybium solandri had no reported stock assessment. Acanthocybium solandri in the Atlantic is grouped under the small tunas species category by the ICCAT (International Commission for the Conservation of Atlantic Tunas) which recently performed initial attempts to assess this species’ stock. Pons et al. (2019) assessed this stock using two length-based methods [(LBSPR: length-based spawning potential ratio (Hordyk et al., 2015) and LIME: length-based integrated mixed-effects (Rudd and Thorson, 2018)] and reported that the stock was stable and not under fishing pressure, which corroborates with results presented by the LBB method in this study. Results from LBB showed that Taractichthys steindachneri was grossly overfished and Taractes rubescens was overfished, with both fisheries having very low biomasses.
As of now, the five pelagic species analyzed in this study are listed as Least Concern (LC) in the IUCN: International Union for Conservation of Nature (International Union for Conservation of Nature [IUCN], 2020) even though a probable increase in catches may have been occurring in recent years (FAO, 2019). However, this classification as LC by the IUCN may be attributed to the limited nature of catches that result as a combination of: data misreporting; low economic value and repeated discarding; an assumed little exposure to fishing stress; and/or the species identification difficulties (Clarke et al., 2014).
Analyses of these species for the Indian Ocean were not taken into consideration because reported LF data for four of these species was less than 50 specimens/year in most cases. However, a review of information for non-targeted pelagic specimens in the Atlantic, Eastern, and Western Central Pacific and Indian Oceans showed that Coryphaena hippurus Acanthocybium solandri and Alepisaurus ferox were commonly encountered in longline fisheries, also including few bramidae (Clarke et al., 2014; FAO, 2019). Coryphaena hippurus seems to be the most documented non-targeted species in the IATTC convention area with catches of 2,500–4,500 tons annually in recent years (Clarke et al., 2014; Aires-da-Silva et al., 2016). Cass-Calay and Ortiz (2009) reported that their stock was not overfished, consistent with our findings (Supplementary Table S1).
Worries have been raised concerning the value of non-target species including Coryphaena hippurus as a source of income and food security to local communities, hence, the urge to safeguard the continuous livelihood supply for artisanal fishers from an apparent disruption from commercial fisheries. This mainly concerns the high catches from purse seine fisheries. Though most un-targeted species are highly productive, many factors of these species still make them vulnerable that may cause their populations to decline (i.e., aggregation, high inter-annual variability, and exploitation before sexual maturity; Moltó et al., 2020). Moreover, due to the lack of quality data, consequently, rare stock assessments, it was unclear if current levels of interactions with tuna longline fisheries were placing these non-targeted fisheries populations at risk. Thus, despite the lack of conservational and management measures to safeguard the stocks of these species, more attention in improving fishery statistics and conducting periodic monitoring of their stocks should be implemented.
The LBB method requires that the LF samples constitute of small-sized as well as large-sized specimens in order to fully represent all possible size classes of the specie’s population. The lack of either small-or large-sized specimens in the LF data may lead to LBB presenting biased results. Given that longlines represent an important gear type in commercial fisheries, we think the LBB method generated less biased estimates since the sizes caught by this gear for the present study constitutes juvenile and adult specimens from at least 1-year data, thus representative of these species’ populations. Furthermore, Froese et al. (2018) reported that, for LBB to present good results, the LF sample fits must show an asymmetric pattern. In the present study, the LF samples for all populations obtained from longline fishing gears showed asymmetric plot patterns similar to fits presented in Froese et al. (2018). Moreover, the stock status estimated by LBB for the 10 populations were consistent with the few available risk assessment and stock assessment reports (Supplementary Table S1). Therefore, the stock status information presented by LBB are reliable and provide these fisheries with information that may serve for managing and conserving their stocks in these oceanic waters. These LBB results may also serve as proxies in more sophisticated assessment models when more reliable data are available.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Author Contributions
RK and CG analyzed the LF data and completed the first draft and revised the manuscript again. RK and ST guided data analysis and structure of the manuscript. FW and OS provided the original length data. NP and QM modified the manuscript. All authors contributed to the revision of the manuscript.
Funding
This study was supported by the National Key R&D Program of China (2019YFD0901404) and the China National Fishery Observer Programme.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We are grateful to the developer of the LBB method, Rainer Froese. We are also thankful for all China tuna Longline observers for providing samples used in various analyses.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmars.2020.592082/full#supplementary-material
References
Aires-da-Silva, A., Valero, J. L., Maunder, M. N., Minte-Vera, C. V., LennertCody, C., Román, M. H., et al. (2016). “Exploratory stock assessment of dorado (coryphaena hippurus) in the southeastern Pacific Ocean,” in Proceedings of the Inter-American Tropical Tuna Commission, seventh Meeting Scientific Advisory Committee, La Jolla, CA.
Beverton, R. J. H., and Holt, S. J. (1957). On the Dynamics of Exploited Fish Populations. London: Ministry of Agriculture, Fisheries and Food.
Beverton, R. J. H., and Holt, S. J. (1966). Manual of Methods for Fish Stock Assessment, Part II – Tables of Yield Functions. FAO Fisheries Technical Paper No. 38, (Rome: FAO), 10.
Cass-Calay, S. L., and Ortiz, M. (2009). Atlantic and Gulf of Mexico Coastal Pelagic Fisheries. Our Living Oceans: Report on the Status of U.S. Living Marine Resources. U.S. Department of Commerce, NOAA Tech. Memo. NMFS-F/SPO-80, Sixth Edn. Washington, D.C: U.S. Department of Commerce, 369.
Chong, L., Mildenberger, T. K., Rudd, M. B., Taylor, M. H., Cope, J. M., Branch, T. A., et al. (2019). Performance evaluation of data-limited, length-based stock assessment methods. ICES J. Mar. Sci. 77:fsz212. doi: 10.1093/icesjms/fsz212
Clarke, S., Sato, M., Small, C., Sullivan, B., Inoue, Y., and Ochi, D. (2014). Bycatch in Longline Fisheries for Tuna and Tuna-Like Species: A Global Review of Status and Mitigation Measures. FAO Fisheries and Aquaculture Technical Paper No. 588. Rome: FAO, 199.
Francis, R. C., Aires-da-Silva, A. M., Maunder, M. N., Schaefer, K. M., and Fuller, D. W. (2016). Estimating fish growth for stock assessments using both age–length and tagging-increment data. Fish. Res. 180, 113–118. doi: 10.1016/j.fishres.2015.06.011
Froese, D., and Pauly, D. (2020). Editors, FishBase. World Wide Web Electronic Publication. Available online at: www.fishbase.se (accessed September 17, 2020).
Froese, R., Demirel, N., Coro, G., Kleisner, K. M., and Winker, H. (2017). Estimating fisheries reference points from catch and resilience. Fish Fish. 18, 506–526. doi: 10.1111/faf.12190
Froese, R., Winker, H., Coro, G., Demirel, N., Tsikliras, A. C., Dimarchopoulou, D., et al. (2018). A new approach for estimating stock status from length-frequency data. ICES J. Mar. Sci. 75, 2004–2015. doi: 10.1093/icesjms/fsy078
Froese, R., Winker, H., Gascuel, D., Sumaila, U. R., and Pauly, D. (2016). Minimizing the impact of fishing. Fish and Fish. 17:785802. doi: 10.1111/faf.12146
Gao, C. X., Tian, S. Q., Kindong, R., and Dai, X. J. (2020). Biology and Environmental Preferences of Wahoo, Acanthocybium solandri (Cuvier, 1832), in the Western and Central Pacific Ocean (WCPO). J. Mar. Sci. Eng. 8:184. doi: 10.3390/jmse8030184
Hordyk, A., Ono, K., Valencia, S., Loneragan, N., and Prince, J. (2015). A novel length-based empirical estimation method of spawning potential ratio (SPR), and tests of its performance, for small-scale, data-poor fisheries. ICES J. Mar. Sci. 72, 217–231. doi: 10.1093/icesjms/fsu004
International Union for Conservation of Nature [IUCN] (2020). Availble at: https://www.iucnredlist.org/ (accessed August 3, 2020).
Jawad, L. A., AlMamry, J. M., and AlBusaidi, H. K. (2014). New record of the keeltail pomfret, Taractes rubescens (Jordan & Evermann, 1887) (Perciformes: Bramidae) from the Sea of Oman. Intl. J. Mar. Sci. 4, 227–230. doi: 10.5376/ijms.2014.04.0025
Kindong, R., Wu, F., Tian, S. Q., Zhu, J. F., Dai, X. J., Wang, J. Q., et al. (2020). Biological parameters estimate for the sickle pomfret (Taractichthys steindachneri) in the west-central and eastern Pacific Ocean. Indian J. Geo-Mar. Sci. 49, 373–381.
Kirby, D. S. (2006). Ecological risk assessment for species caught in WCPO tuna fisheries: inherent risk as determined by productivity-susceptibility analysis WCPFC-SC2-2006/EB WP-1. Paper Presented at the 2nd Meeting of the Scientific Committee of the Western and Central Pacific Fisheries Commission WCPFC–SC1, (Manila: WCPFC), 24.
Kwon, Y. J., An, D. H., Moon, D. Y., Hwang, S. J., and Lee, J. B. (2009). An ecological risk assessment for the effect of the Korean tuna longline fishery in the Western and Central Pacific Ocean. J. Kor. Soc. Fish. Tech. 45, 22–33. doi: 10.3796/ksft.2008.45.1.022
Liang, C., Xian, W., Liu, S., and Pauly, D. (2020). Assessments of 14 exploited fish and invertebrate stocks in Chinese waters using the lbb method. Front. Mar. Sci. 7:314. doi: 10.3389/fmars.2020.00314
Lynch, A. J., Cooke, S. J., Deines, A. M., Bower, S. D., Bunnell, D. B., Cowx, I. G., et al. (2016). The social, economic, and environmental importance of inland fish and fisheries. Environ. Rev. 24, 115–121.
McBride, R. S., Richardson, A. K., and Maki, K. L. (2008). Age, growth, and mortality of wahoo, Acanthocybium solandri, from the Atlantic coast of Florida and the Bahamas. Mar. Freshw. Res. 59, 799–807. doi: 10.1071/mf08021
Methot, R. D., and Wetzel, C. R. (2013). Stock synthesis: a biological and statistical framework for fish stock assessment and fishery management. Fish. Res. 142, 86–99. doi: 10.1016/j.fishres.2012.10.012
Moltó, V., Pilar, H., Mauro, S., Amina, B., Raouf, B., Adriano, M., et al. (2020). A Global Review on the Biology of the Dolphinfish (Coryphaena hippurus) and Its fishery in the mediterranean sea: advances in the last two decades. Rev. Fish. Sci. Aquac. 28, 376–420. doi: 10.1080/23308249.2020.1757618
Munro, J. L. (1983). A cost-effective data acquisition system for assessment and management of tropical multispecies, multi-gear fisheries. Fishbyte 1, 7–12.
Nadon, M. O., Ault, J. S., Williams, I. D., Smith, S. G., and DiNardo, G. T. (2015). Length based assessment of coral reef fish populations in the main and northwestern Hawaiian Islands. PLoS One 10:e0133960. doi: 10.1371/journal.pone.0133960
Palomares, M. L. D., Froese, R., Derrick, B., Nöel, S.-L., Tsui, G., Woroniak, J., et al. (2018). A Preliminary Global Assessment of the Status of Exploited Marine Fish and Invertebrate Populations. A Report Prepared by the Sea Around Us for OCEANA. Washington, DC: OCEANA, 64.
Pauly, D. (1998). Beyond our original horizons: the tropicalization of Beverton and Holt. Rev. Fish Biol. Fish. 8, 307–334.
Pons, M., Cope, J. M., and Kell, L. (2020). Comparing performance of catch-based and length-based stock assessment methods in data-limited fisheries. Can. J. Fish. Aquat. Sci. 77, 1026–1037. doi: 10.1139/cjfas-2019-0276
Pons, M., Kell, L., Rudd, M. B., Cope, J. M., and Lucena Fredou, F. (2019). Performance of length-based data-limited methods in a multifleet context: application to small tunas, mackerels, and bonitos in the Atlantic Ocean. ICES J. Mar. Sci. 76, 960–973. doi: 10.1093/icesjms/fsz004
Prince, J., Victor, S., Kloulchad, V., and Hordyk, A. (2015). Length based SPR assessment of eleven Indo-Pacific coral reef fish populations in Palau. Fish. Res. 171, 42–58. doi: 10.1016/j.fishres.2015.06.008
Quinn, T. J., and Deriso, R. B. (1999). Quantitative Fish Dynamics. New York, NY: Oxford University Press.
Rudd, M. B., and Thorson, J. T. (2018). Accounting for variable recruitment and fishing mortality in length-based stock assessments for data-limited fisheries. Can. J. Fish. Aquat. Sci. 75, 1019–1035. doi: 10.1139/cjfas-2017-0143
Smith, M. M. (1986). “Family No. 207. Bramidae,” in Smiths Sea Fishes, eds M. M. Smith and P. C. Heemstra (Berlin: SpringerVerlag), 633–636.
von Bertalanffy, L. (1938). A quantitative theory of organic growth (inquiries on growth laws. II.). Hum. Biol. 10, 181–213.
Wang, Y., Wang, Y., Liu, S., Liang, C., Zhang, H., and Xian, W. (2020). Stock Assessment Using LBB Method for Eight Fish Species From the Bohai and Yellow Seas. Front. Mar. Sci. 7:164. doi: 10.3389/fmars.2020.00164
Zhang, L., Ren, Q., Liu, M., Xu, Q., Kang, B., and Jiang, X. (2020). Fishery Stock Assessments in the Min River Estuary and Its Adjacent Waters in Southern China Using the Length-Based Bayesian Estimation (LBB) Method. Front. Mar. Sci 7:507. doi: 10.3389/fmars.2020.00507
Zischke, M. T. (2012). A review of the biology, stock structure, fisheries and status of wahoo (Acanthocybium solandri), with reference to the Pacific Ocean. Fish. Res. 119-120, 13–22. doi: 10.1016/j.fishres.2011.11.026
Keywords: non-target species, stock assessment, data-limited populations, length-frequency data, biomass estimation, LBB method
Citation: Kindong R, Gao C, Pandong NA, Ma Q, Tian S, Wu F and Sarr O (2020) Stock Status Assessments of Five Small Pelagic Species in the Atlantic and Pacific Oceans Using the Length-Based Bayesian Estimation (LBB) Method. Front. Mar. Sci. 7:592082. doi: 10.3389/fmars.2020.592082
Received: 06 August 2020; Accepted: 19 October 2020;
Published: 06 November 2020.
Edited by:
Hui Zhang, Institute of Oceanology (CAS), ChinaReviewed by:
Cui Liang, Institute of Oceanology (CAS), ChinaJin Gao, Memorial University of Newfoundland, Canada
Copyright © 2020 Kindong, Gao, Pandong, Ma, Tian, Wu and Sarr. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Siquan Tian, c3F0aWFuQHNob3UuZWR1LmNu
 Richard Kindong
Richard Kindong Chunxia Gao1,2,3,4
Chunxia Gao1,2,3,4