- 1College of Animal Science and Veterinary Medicine, Henan Agricultural University, Zhengzhou, China
- 2West China Hospital, Sichuan University, Chengdu, China
A 139,622-bp IncI1 ST71 conjugative plasmid pEC012 from an avian Escherichia coli D-ST117 strain was sequenced, which carried five IS26-bracketed resistance modules: IS26-fosA3-orf1-orf2-Δorf3-IS26, IS26-fip-ΔISEcp1-blaCTX-M-65-IS903D-iroN-IS26, IS26-ΔtnpR-blaTEM-1-rmtB-IS26, IS26-oqxAB-IS26, and IS26-floR-aac(3)-IV-IS26. The backbone of pEC012 was similar to that of several other IncI1 ST71 plasmids: pV408, pM105, and pC271, but these plasmids had different arrangements of multidrug resistance region. In addition, the novel ISEc57 element was identified, which is in the IS21 family. The stepwise emergence of multi-resistance regions demonstrated the accumulation of different resistance determinants through homologous recombination. To the best of our knowledge, this is the first study to identify a multidrug-resistant IncI1 ST71 plasmid carrying blaCTX-M-65, rmtB, fosA3, floR, and oqxAB in an avian E. coli ST117 strain.
Introduction
The emergence and dissemination of antimicrobial resistance have become a major global public health concern. It is associated with mobile genetic elements such as plasmids, transposons, and integrons. Homologous recombination is important in the movement of resistance genes and the creation and evolution of multiresistance region (MRR; Partridge, 2011; van Hoek et al., 2011). In the recent years, blaCTX-Ms has become the most common genes encoding extended-spectrum β-lactamases in multidrug-resistant Enterobacteriaceae worldwide. The blaCTX-Ms genes often coexist with other genes such as armA or rmtB encoding 16S rRNA methylases, or with fosA3, which confers resistance to fosfomycin (Deng et al., 2011; Hou et al., 2012). Recently, multiple resistance genes blaTEM-1, blaCTX-M-65, fosA3, and rmtB were found to coexist on the same IncFII plasmid such as F33:A-:B- plasmid pHN7A8, F33:A-:B- plasmid pEC011, and F2:A-:B- plasmid pXZ, which were obtained from Escherichia coli isolates originating from dogs, chickens and ducks in China, respectively (Sun et al., 2012; He et al., 2013; Pan et al., 2014). Of note, the similar IncFII plasmids carrying these genes were also identified in E. coli and Klebsiella pneumoniae from humans (Xiang et al., 2015; Zhao et al., 2015; Sennati et al., 2016). Moreover, plasmids belonging to IncI1 carrying extended-spectrum and AmpC β-lactamase genes are widespread in Enterobacteriaceae (Garcia-Fernandez et al., 2008). Recently, IncI1 ST71 epidemic plasmid lineage carrying blaTEM-1, blaCTX-M-65, fosA3 was identified in E. coli from humans and animals (Yang et al., 2014; Riccobono et al., 2015). Here, we report the complete sequence of pEC012, a multidrug-resistant IncI1 ST71 plasmid carrying blaTEM-1, blaCTX-M-65, rmtB, fosA3, floR, and oqxAB in an avian E. coli ST117 isolate.
Materials and Methods
Bacterial Strains
In a survey on antimicrobial resistant bacterial strains in China in 2009, one E. coli strain, EC012, was isolated from one chicken on a farm in Changchun Province, Northeast China. The species identification was performed using the VITEK 32 automated identification system (bioMérieux, Marcy l’Etoile, France). E. coli ATCC 25922 was used as the control strain for the antimicrobial susceptibility testing.
Antimicrobial Susceptibility Testing and Antimicrobial Resistance Gene Detection
Antimicrobial susceptibility testing was performed on EC012 using the broth microdilution method, and the minimal inhibitory concentration of fosfomycin was determined using the agar dilution method on Mueller–Hinton agar containing 25 μg/mL glucose-6-phosphate according to the guidelines of the Clinical and Laboratory Standards Institute (Clinical and Laboratory Standards Institute [CLSI], 2013). The presence of antimicrobial resistance genes encoding extended-spectrum β-lactamases, 16S rRNA methylases and the plasmid-encoded fosfomycin-resistance determinants were screened using PCR as described previously (Hou et al., 2012; Pan et al., 2013). The floR gene was amplified by primers (floR-F: 5′-GTATGGGCACCTTCTTCGTCT-3′ and floR-R: 5′- CAGCCCCAACGAAACCAGT-3′) in this study.
Phylogenetic Group and Multilocus Sequence Typing (MLST)
Phylogenetic typing was done by means of a multiplex PCR-based method with the chuA, yjaA genes and the DNA fragment TSPE4.C2, as described previously (Clermont et al., 2000). Seven housekeeping genes (adk, fumC, gyrB, icd, mdh, purA, and recA) were amplified and sequenced as described previously (Wirth et al., 2006). The sequences were analyzed further by MLST according to the protocols recommended at http://mlst.warwick.ac.uk/mlst/dbs/Ecoli.
Conjugation Experiment and Plasmid Analysis
Conjugation experiment was carried out using E. coli EC012 as the donor and E. coli C600 (resistant to rifampicin) as the recipient, as described previously (Pan et al., 2013). The transconjugants were selected on MacConkey agar supplemented with cefotaxime (4 μg/mL) and rifampicin (450 μg/mL). Transfer frequency was calculated as the number of transconjugants per recipient. Antimicrobial susceptibility testing of the transconjugants was conducted, and the presence of blaCTX-M-65, rmtB, fosA3, and floR was confirmed by PCR as described above. Plasmid DNA was extracted from the transconjugants using the Plasmid Midi Kit (Qiagen, Hilden, Germany), designated as pEC012. The plasmid incompatibility groups of pEC012 were typed by a PCR-based method, as described previously (Carattoli et al., 2005). Plasmid MLST was carried out using published primers with alleles (Garcia-Fernandez et al., 2008) and sequence types assigned according to http://pubmlst.org/plasmid.
Plasmid Sequencing and Annotation
Plasmid pEC012 from the transconjugant TEC012 was fully sequenced by an Illumina Miseq platform. Pair-end index libraries were constructed using NEBNext Ultra DNA Library Prep Kit (Illumina, San Diego, CA, USA). Libraries with different indexes were mixed and loaded on an Illumina MiSeq. Sequencing was carried out using a 2 × 250 paired-end configuration on the Miseq instrument. Sequence data were assembled into eight contigs using Velvet 1.1.06 and CAP3 software (Huang and Madan, 1999; Zerbino and Birney, 2008). Gaps were closed by a PCR-based strategy. The plasmid sequence was initially annotated with the Rapid Annotation using Subsystem Technology (RAST version 2.0) server1 and then curated manually using the BLASTn and BLASTp algorithms2. The comparative analysis of complete nucleotide sequences was performed using the referenced plasmids IncI1 R64 (GenBank accession no. AP005147), IncI1 ST71 plamsid pC193, pM105, pV408, and pC271 (GenBank accession no. LN735558, LN735559, LN735560, LN735561, respectively; Sampei et al., 2010; Riccobono et al., 2015). Physical maps were generated using EasyFig software and DNAPlotter (Carver et al., 2009; Sullivan et al., 2011).
Nucleotide Sequence Accession Number
The complete sequence of pEC012 has been deposited in the GenBank database under accession no. KT282968.
Results and Discussion
Strain and Plasmid Characteristics
The E. coli strain was shown to be resistant to ampicillin, cefotaxime, amikacin, gentamycin, fosfomycin, doxycycline, florfenicol, and ciprofloxacin (Supplementary Table S1), and carried blaTEM-1, blaCTX-M-65, rmtB, fosA3, and floR genes, belonging to D-ST117. E. coli EC012 strain was isolated from the same farm as E. coli D-ST117 EC011, which exhibit the same resistant phenotype (Pan et al., 2014).
The transconjugants were obtained successfully, designated as TEC012, exhibiting resistance to cefotaxime, amikacin, fosfomycin and florfenicol, and low-level resistance to ciprofloxacin (Supplementary Table S1). The conjugation frequency of pEC012 was 5 × 10-3. pEC012 belonged to the incompatibility group IncI1, which was further assigned to ST71, and carried blaTEM-1, blaCTX-M-65, rmtB, fosA3, and floR genes. However, the pEC011 from the strain EC011belonged to IncFII, and carried blaTEM-1, blaCTX-M-65, rmtB, fosA3 genes, not harbored the floR gene (Pan et al., 2014).
Overall Structure of pEC012
pEC012 is 139,622-bp in length with a GC content of 50.8%. The plasmid was composed of a 102,866-bp IncI1 typical backbone fragment encoding genes responsible for plasmid replication, transfer, maintenance, and stability functions, and a 36,756-bp MRR (Supplementary Figure S1). The backbone of pEC012 was similar to other IncI1 ST71 plasmids such as pV408, pM105, and pC271 (>99% at nucleotide level; Figure 1). At least 156 complete open reading frames were predicted within the plasmid.
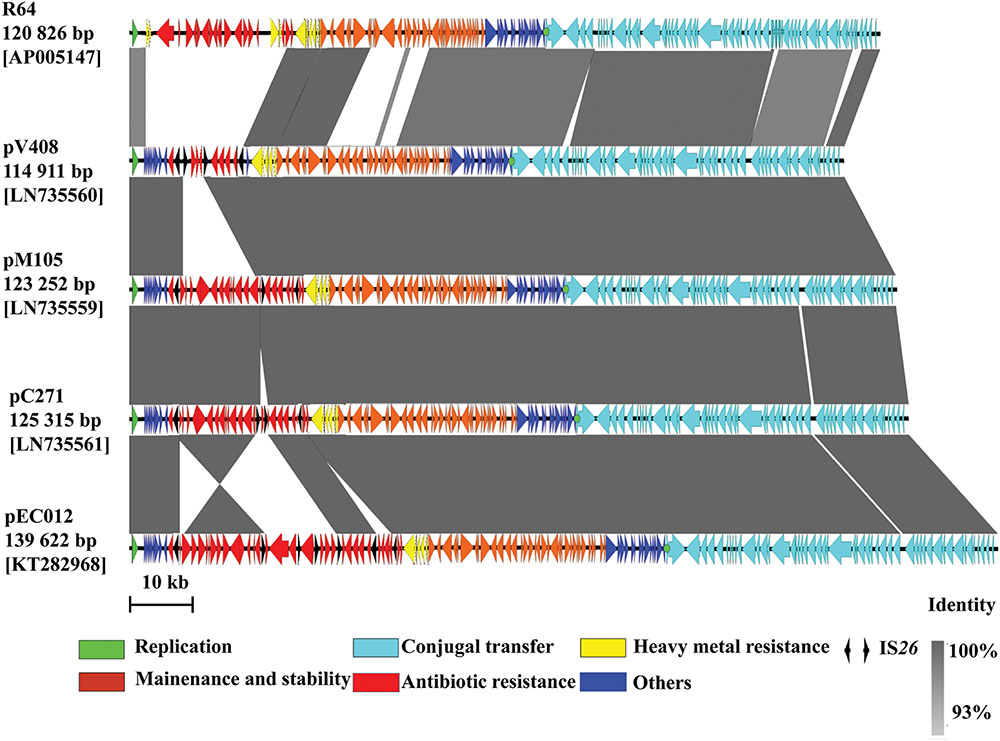
FIGURE 1. Comparative analysis of pEC012 with other IncI1 plasmids. Homologous segments generated by a BLASTn comparison (≥93% identity) are shown as gray boxes. Genes are represented by thick arrows. The location of oriT is indicated by a green dot. The color code equates to that described in this figure legend.
pEC012 MRR
The MRR of pEC012 had complicated structure containing several transposable units with a new arrangement. It consisted of five different segments containing resistance genes. The first segment corresponded to the IS26-formed composite transposon carrying the fosA3 gene, conferring resistance to fosfomycin. The segment ISEcp1-blaCTX-M-65-IS903-iroN was the second transposable unit, in which ISEcp1 was truncated by fip gene. The third segment, including rmtB adjacent to a fragment of Tn2 carrying blaTEM-1, was linked with the second segment by IS26 (Figure 2).
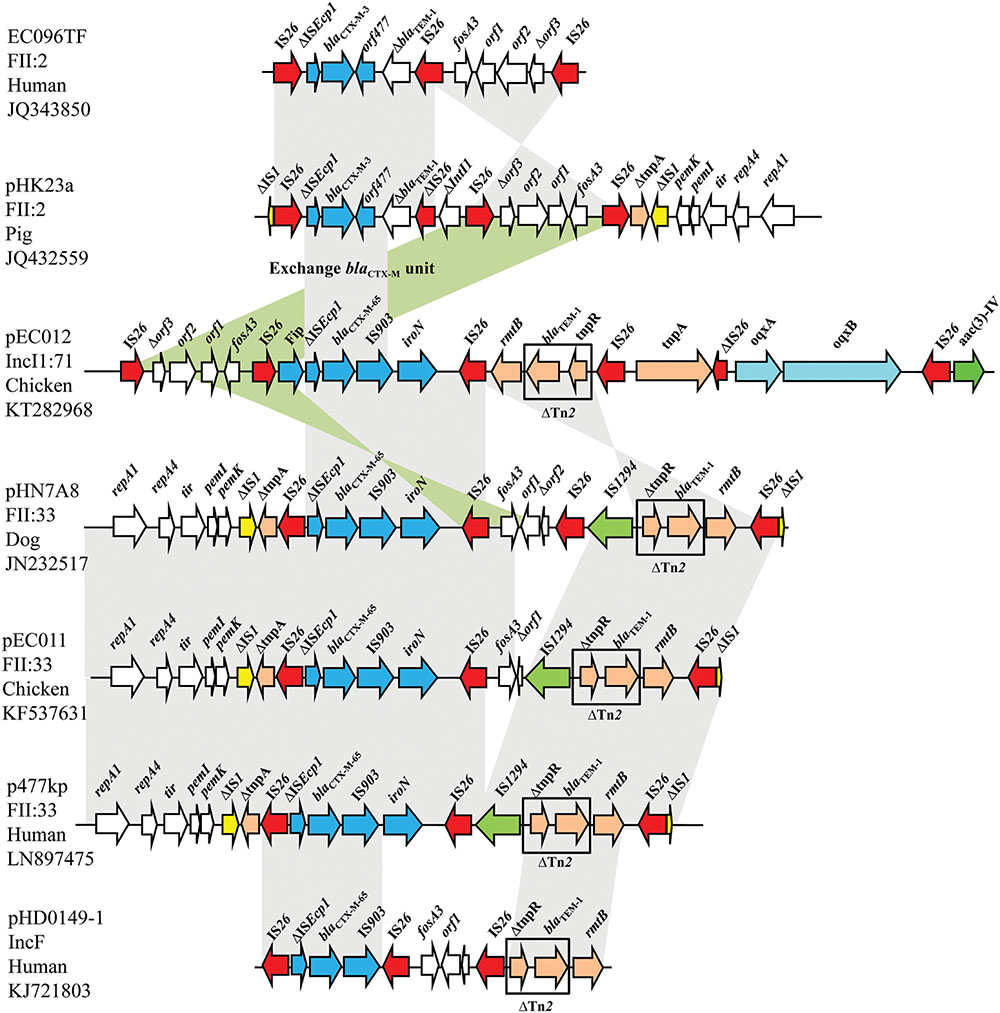
FIGURE 2. Structural features surrounding the blaCTX-M-65 gene in pEC012 compared with other blaCTX-M of IncFII-type plasmids EC096TF (GenBank accession number JQ343850), pHK23a (GenBank accession number JQ432559), pHN7A8 (GenBank accession number JN232517), pEC011 (GenBank accession number KF537631), p477kp (GenBank accession number LN897475), pHD0149-1 (GenBank accession number KJ721803). Similar regions are indicated by gray shading.
Recently, the IncI1 ST71 epidemic plasmid carrying IS26-fip-ΔISEcp1-blaCTX-M-65-IS903D-iroN-IS26 and IS26-fosA3-Δorf1-IS26 modules was reported in the Chaco region of Bolivia, which lacked the tnpR-blaTEM-1-rmtB module (Riccobono et al., 2015). In addition, the IncF plasmids carrying blaTEM-1, blaCTX-M-65, fosA3, and rmtB have been found in E. coli isolates from animals (Sun et al., 2012; He et al., 2013; Pan et al., 2014), and in E. coli and K. pneumoniae from humans (Xiang et al., 2015; Zhao et al., 2015; Sennati et al., 2016). Those plasmids belonging to IncFII possessed a similar MRR structure (Figure 2). In the present study, the IS26-fosA3-IS26 module was located upstream of the ISEcp1-blaCTX-M-65-IS903-iroN module, and Tn2 carrying rmtB was downstream of the ISEcp1-blaCTX-M-65-IS903-iroN module in the opposite orientation with other MRRs of IncF plasmids, such as pHN7A8, pEC011, p477kp, and pHD0149-1 (GenBank accession no. JN232517, KF537631, LN897475, KJ721803, respectively; Figure 2). Seven copies of IS26 were dispersed in the MRR of pEC012, however, target duplication repeats flanking IS26 were not observed. The lack of direct repeats flanking IS26 in the pEC012 MRR suggests that this may have occurred by homologous recombination rather than insertion (Partridge, 2011).
Moreover, pEC012 belonged to IncI1, whereas pEC011 belonged to IncFII, suggesting that the complex MRR was mobilized into different plasmids. However, it was difficult to discriminate if the MRR carrying blaTEM-1, blaCTX-M-65, fosA3, and rmtB was transferred from IncFII to IncI1, or vice versa.
Two IS26 composite transposons located downstream of Tn2 transposon carrying rmtB were also observed. One IS26 composite transposon harbored oqxAB, and another contained floR and aac(3)-IV genes (Figure 3). The typical context of floR is ISCR2-virD2-floR-lysR (Liu et al., 2014; Wang et al., 2014), which could coexists with IS26-oqxAB-IS26, or aac(3)-IV in the other plasmids, such as pS53T, pC271, pK1HV, and pACN001-A (GenBank accession no. KF731829, LN735561, HF545434, KC853434, respectively). In the present study, the IS26-formed composite transposon harboring floR and aac(3)-IV was identical to that on pC271 and pM105 plasmids except for the direction (Figure 1). Nevertheless, no direct repeats flanking IS26 were found. This suggests that two copies of IS26 could form a composite transposon and generate recombination. The IS26-formed composite transposon may have played an important role in generation of this complicated MRR by homologous recombination, and demonstrated the accumulation of different resistant determinants justly.
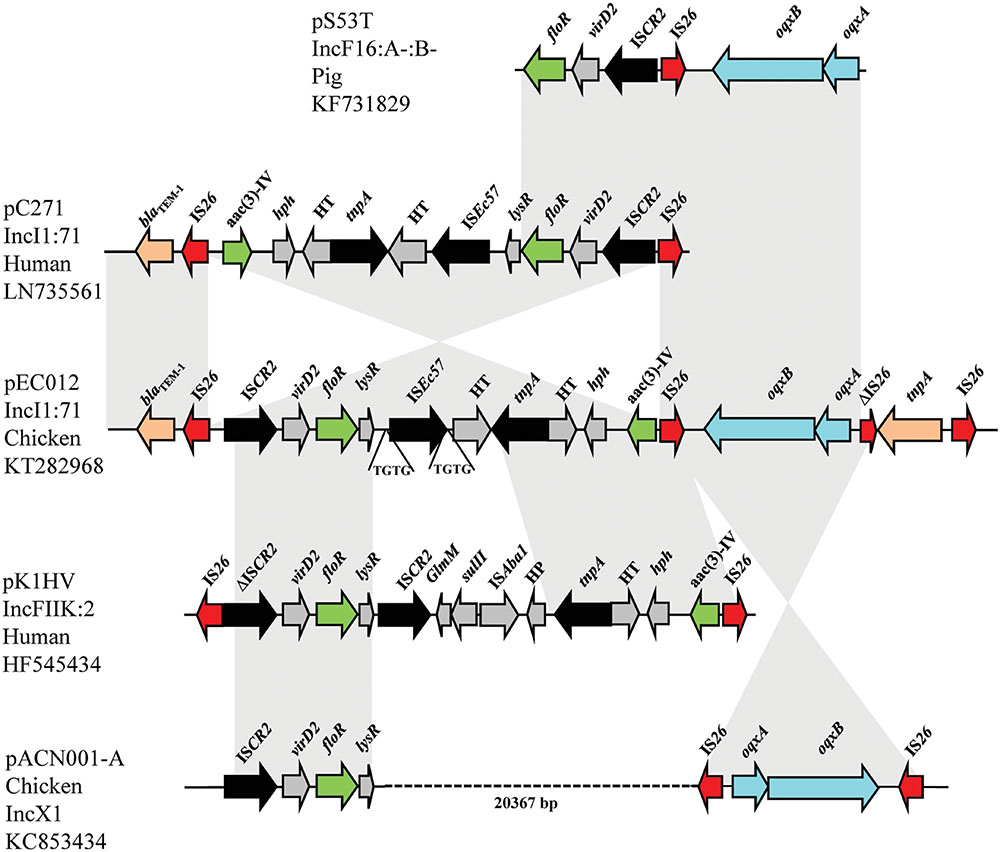
FIGURE 3. Structural features of IS26 composite transposon carrying floR, aac(3)-IV, and oqxAB in pEC012 compared with those of IncFII plasmid pS53T (GenBank accession number KF731829), IncI1 pC271 (GenBank accession number LN735561), IncFIIK pK1HV (GenBank accession number HF545434), and the IncX1 pACN001-A (GenBank accession number KC853434). Similar regions are indicated by gray shading.
Identification of ISEc57, a Novel IS
ISEc57 is 1960-bp long and belongs to the IS21 family3. It possesses 23/27-bp inverted repeats, and creates 4-bp directly repeated sequences of the target site (TGTG in the structure identified; Figure 3). ISEc57 contains two open reading frames (Orf1 and Orf2) encoding proteins of 341 and 257 amino acids, respectively. The deduced amino acid sequences of the Orf1 and Orf2 proteins had 87 and 94% amino acid identity with the transposase subunits of ISAba8, respectively.
Conclusion
We characterized the complete sequence of pEC012, a multidrug-resistant IncI1 ST71 plasmid carrying several resistance determinants, blaTEM, blaCTX-M-65, rmtB, fosA3, oqxAB, floR, and aac(3)-IV in an avian E. coli D-ST117 strain. The stepwise emergence of the MRR demonstrated the accumulation of different resistant determinants. IS26 may play a pivotal role in generation of the complex genetics of resistance genes by homologous recombination. The detection of several resistance determinants on a conjugative plasmid among E. coli of food-producing animal origin may represent an emerging threat to animal and public health. There is an urgent need to monitor the dissemination of this multidrug-resistance plasmid among the Enterobacteriaceae.
Author Contributions
Y-SP is in charge of design of study, acquisition and analysis of data, drafting of article; Z-YZ is in charge of analysis of data and critical revision; LY is in charge of analysis of data and drafting of article; X-DD is in charge of drafting of article; HH is in charge of acquisition of data; X-HZ is in charge of acquisition of data; G-ZH is in charge of conception and design of study, and analysis of data.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors are thankful to the National Natural Science Foundation of China for the financial support (grant No. U1504326 and 31372481) and the Foundation of Henan Educational Committee (grant No. 15A230024).
Supplementary Material
The Supplementary Material for this article can be found online at: http://journal.frontiersin.org/article/10.3389/fmicb.2016.01117
FIGURE S1 | Genetic map of multi-resistance plasmid pEC012. Coding regions are shown indicating the direction of transcription. The different plasmid modules are indicated on the inner circle of the map. The backbone of the plasmid consists of a replication, maintenance and stability (green), conjugal transfer module (gray). The MRR of the plasmid is composed of a 36.7-kb region (red).
Footnotes
References
Carattoli, A., Bertini, A., Villa, L., Falbo, V., Hopkins, K. L., and Threlfall, E. J. (2005). Identification of plasmids by PCR-based replicon typing. J. Microbiol. Methods 63, 219–228. doi: 10.1016/j.mimet.2005.03.018
Carver, T., Thomson, N., Bleasby, A., Berriman, M., and Parkhill, J. (2009). DNAPlotter: circular and linear interactive genome visualization. Bioinformatics 25, 119–120. doi: 10.1093/bioinformatics/btn578
Clermont, O., Bonacorsi, S., and Bingen, E. (2000). Rapid and simple determination of the Escherichia coli phylogenetic group. Appl. Environ. Microbiol. 66, 4555–4558. doi: 10.1128/AEM.66.10.4555-4558.2000
Clinical and Laboratory Standards Institute [CLSI] (2013). Performance Standards for Antimicrobial Susceptibility Testing: Twenty-Third Informational Supplement M100-S23. Wayne, PA: Clinical and Laboratory Standards Institute.
Deng, Y., He, L., Chen, S., Zheng, H., Zeng, Z., Liu, Y., et al. (2011). F33:A-:B- and F2:A-:B- plasmids mediate dissemination of rmtB-blaCTX-M-9 group genes and rmtB-qepA in Enterobacteriaceae isolates from pets in China. Antimicrob. Agents Chemother. 55, 4926–4929. doi: 10.1128/AAC.00133-11
Garcia-Fernandez, A., Chiaretto, G., Bertini, A., Villa, L., Fortini, D., Ricci, A., et al. (2008). Multilocus sequence typing of IncI1 plasmids carrying extended-spectrum β-lactamases in Escherichia coli and Salmonella of human and animal origin. J. Antimicrob. Chemother. 61, 1229–1233. doi: 10.1093/jac/dkn131
He, L., Partridge, S. R., Yang, X., Hou, J., Deng, Y., Yao, Q., et al. (2013). Complete nucleotide sequence of pHN7A8, an F33:A-:B- type epidemic plasmid carrying blaCTX-M-65, fosA3 and rmtB from China. J. Antimicrob. Chemother. 68, 46–50. doi: 10.1093/jac/dks369
Hou, J., Huang, X., Deng, Y., He, L., Yang, T., Zeng, Z., et al. (2012). Dissemination of the fosfomycin resistance gene fosA3 with CTX-M β-lactamase genes and rmtB carried on IncFII plasmids among Escherichia coli isolates from pets in China. Antimicrob. Agents Chemother. 56, 2135–2138. doi: 10.1128/AAC.05104-11
Huang, X., and Madan, A. (1999). CAP3: a DNA sequence assembly program. Genome Res. 9, 868–877. doi: 10.1101/gr.9.9.868
Liu, B. T., Li, L., Fang, L. X., Sun, J., Liao, X. P., Yang, Q. E., et al. (2014). Characterization of plasmids carrying oqxAB in blaCTX-M-negative Escherichia coli isolates from food-producing animals. Microb. Drug Resist. 20, 641–650. doi: 10.1089/mdr.2014.0022
Pan, Y. S., Liu, J. H., Hu, H., Zhao, J. F., Yuan, L., Wu, H., et al. (2013). Novel arrangement of the blaCTX-M-55 gene in an Escherichia coli isolate coproducing 16S rRNA methylase. J. Basic Microbiol. 53, 928–933. doi: 10.1002/jobm.201200318
Pan, Y. S., Yuan, L., Zong, Z. Y., Liu, J. H., Wang, L. F., and Hu, G. Z. (2014). A multidrug-resistance region containing blaCTX-M-65, fosA3 and rmtB on conjugative IncFII plasmids in Escherichia coli ST117 isolates from chicken. J. Med. Microbiol. 63, 485–488. doi: 10.1099/jmm.0.070664-0
Partridge, S. R. (2011). Analysis of antibiotic resistance regions in Gram-negative bacteria. FEMS. Microbiol. Rev. 35, 820–855. doi: 10.1111/j.1574-6976.2011.00277.x
Riccobono, E., Di Pilato, V., Di Maggio, T., Revollo, C., Bartoloni, A., Pallecchi, L., et al. (2015). Characterization of IncI1 sequence type 71 epidemic plasmid lineage responsible for the recent dissemination of CTX-M-65 extended-spectrum β-lactamase in the Bolivian Chaco region. Antimicrob. Agents Chemother. 59, 5340–5347. doi: 10.1128/AAC.00589-15
Sampei, G., Furuya, N., Tachibana, K., Saitou, Y., Suzuki, T., and Mizobuchi, K., et al. (2010). Complete genome sequence of the incompatibility group I1 plasmid R64. Plasmid 64, 92–103. doi: 10.1016/j.plasmid.2010.05.005
Sennati, S., Riccobono, E., Di Pilato, V., Villagran, A. L., Pallecchi, L., Bartoloni, A., et al. (2016). pHN7A8-related multiresistance plasmids (blaCTX-M-65, fosA3 and rmtB) detected in clinical isolates of Klebsiella pneumoniae from Bolivia: intercontinental plasmid dissemination? J. Antimicrob. Chemother. 71, 1732–1734. doi: 10.1093/jac/dkv506
Sullivan, M. J., Petty, N. K., and Beatson, S. A. (2011). Easyfig: a genome comparison visualizer. Bioinformatics 27, 1009–1010. doi: 10.1093/bioinformatics/btr039
Sun, H., Li, S., Xie, Z., Yang, F., Sun, Y., Zhu, Y., et al. (2012). A novel multidrug resistance plasmid isolated from an Escherichia coli strain resistant to aminoglycosides. J. Antimicrob. Chemother. 67, 1635–1638. doi: 10.1093/jac/dks107
van Hoek, A. H., Mevius, D., Guerra, B., Mullany, P., Roberts, A. P., and Aarts, H. J. (2011). Acquired antibiotic resistance genes: an overview. Front. Microbiol. 2:203. doi: 10.3389/fmicb.2011.00203
Wang, X. R., Hao, H. H., Xu, Z. F., Zheng, H. J., Liu, C. Y., Wei, L. Y., et al. (2014). Plasmid-mediated multidrug resistance and virulence in an avian pathogenic Escherichia coli strain isolated in China. J. Glob. Antimicrob. Resist. 2, 57–58. doi: 10.1016/j.jgar.2013.10.004
Wirth, T., Falush, D., Lan, R., Colles, F., Mensa, P., Wieler, L. H., et al. (2006). Sex and virulence in Escherichia coli: an evolutionary perspective. Mol. Microbiol. 60, 1136–1151. doi: 10.1111/j.1365-2958.2006.05172.x
Xiang, D. R., Li, J. J., Sheng, Z. K., Yu, H. Y., Deng, M., Bi, S., et al. (2015). Complete sequence of a novel IncR-F33:A-:B- plasmid, pKP1034, harboring fosA3, blaKPC-2, blaCTX-M-65, blaSHV-12, and rmtB from an epidemic Klebsiella pneumoniae sequence type 11 strain in China. Antimicrob. Agents Chemother. 60, 1343–1348. doi: 10.1128/AAC.01488-15
Yang, X., Liu, W., Liu, Y., Wang, J., Lv, L., Chen, X., et al. (2014). F33: A-: B-, IncHI2/ST3, and IncI1/ST71 plasmids drive the dissemination of fosA3 and blaCTX-M-55/-14/-65 in Escherichia coli from chickens in China. Front. Microbiol. 5:688. doi: 10.3389/fmicb.2014.00688
Zerbino, D. R., and Birney, E. (2008). Velvet: algorithms for de novo short read assembly using de Bruijn graphs. Genome Res. 18, 821–829. doi: 10.1101/gr.074492.107
Keywords: extended-spectrum β-lactamases, 16S rRNA methylase, fosfomycin, Tn2, IS26-composite transposon
Citation: Pan Y-S, Zong Z-Y, Yuan L, Du X-D, Huang H, Zhong X-H and Hu G-Z (2016) Complete Sequence of pEC012, a Multidrug-Resistant IncI1 ST71 Plasmid Carrying blaCTX-M-65, rmtB, fosA3, floR, and oqxAB in an Avian Escherichia coli ST117 Strain. Front. Microbiol. 7:1117. doi: 10.3389/fmicb.2016.01117
Received: 19 February 2016; Accepted: 05 July 2016;
Published: 18 July 2016.
Edited by:
Manuela Caniça, National Institute of Health Doutor Ricardo Jorge, PortugalReviewed by:
Jian-Hua Liu, South China Agricultural University, ChinaInes Barata Moura, Instituto de Tecnologia Química e Biológica, Portugal
Copyright © 2016 Pan, Zong, Yuan, Du, Huang, Zhong and Hu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Gong-Zheng Hu, yaolilab@163.com
 Yu-Shan Pan
Yu-Shan Pan Zhi-Yong Zong
Zhi-Yong Zong Li Yuan
Li Yuan Xiang-Dang Du
Xiang-Dang Du Hui Huang
Hui Huang Xing-Hao Zhong
Xing-Hao Zhong Gong-Zheng Hu
Gong-Zheng Hu