- 1Department of Zoology, Hansraj College, University of Delhi, New Delhi, India
- 2Department of Zoology, Swami Shraddhanand College, University of Delhi, New Delhi, India
- 3Department of Microbiology, Swami Shraddhanand College, University of Delhi, New Delhi, India
- 4Betterhumans Inc., Gainesville, FL, United States
- 5Division Radiopharmaceuticals and Radiation Biology, Institute of Nuclear Medicine and Allied Sciences, New Delhi, India
Exosomes, a subtype of the class of extracellular vesicles and nano-sized particles, have a specific membrane structure that makes them an alternative proposition to combat with cancer through slight modification. As constituents of all most all the primary body fluids, exosomes establish the status of intercellular communication. Exosomes have specific proteins/mRNAs and miRNAs which serve as biomarkers, imparting a prognostic tool in clinical and disease pathologies. They have efficient intrinsic targeting potential and efficacy. Engineered exosomes are employed to deliver therapeutic cargos to the targeted tumor cell or the recipient. Exosomes from cancer cells bring about changes in fibroblast via TGFβ/Smad pathway, augmenting the tumor growth. These extracellular vesicles are multidimensional in terms of the functions that they perform. We herein discuss the uptake and biogenesis of exosomes, their role in various facets of cancer studies, cell-to-cell communication and modification for therapeutic and diagnostic use.
Introduction
Exosomes are recognized as a subtype of the class of Extracellular vesicles (EVs). These nano-sized particles appear as small, flattened hemispheres with a diameter of 40–150 nm and a density of 1.13–1.21 g/ml (Kalluri, 2016; Gilligan and Dwyer, 2017; Kalimuthu et al., 2018; You et al., 2018; Głuszko et al., 2019; Zhao and Xie, 2019). The orientation of the surrounding lipid bilayer membrane can be regarded unique as it serves as a reflection of the intrinsic cell from which the exosomes have originated (Kalluri, 2016; Kalimuthu et al., 2018; Głuszko et al., 2019; Zhao and Xie, 2019). The structural specificity of the membrane confers properties, proving to be useful in cancer treatments through selection of preferably modified exosomes (You et al., 2018).
Exosomes are remarkable constituents of all the major body fluids, including plasma, saliva, urine, cerebrospinal fluid (Zhao and Xie, 2019) and are also present as secretions or discharges of cells such as red blood cells, platelets, lymphocytes, dendritic cells and cancer cells (Głuszko et al., 2019). Nucleic acids like RNA [mRNAs, microRNAs (miRNA) and long noncoding RNA (IncRNA)] (Głuszko et al., 2019; Zhao and Xie, 2019), cellular proteins and lipids represent the contents of exosomes (Kalluri, 2016; Gilligan and Dwyer, 2017; Kalimuthu et al., 2018; You et al., 2018). Noteworthy, the presence of DNA in the exosomes is considered rather contradictory. It is thought to share a relation with the source of the exosomes in consideration (Fais et al., 2013). These contents are protected from degradation and are taken up via fusion by the target cell acting as receiver, thus establishing the status of exosomes as means of intercellular communication (Kalluri, 2016; You et al., 2018; Głuszko et al., 2019).
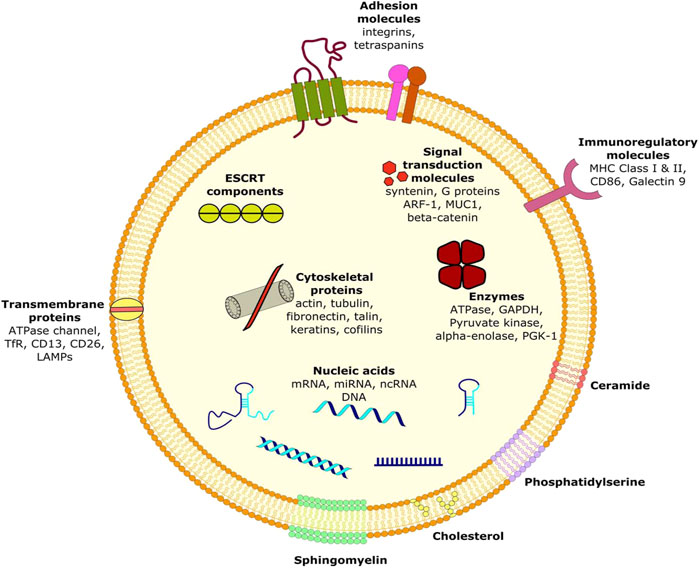
Majority of exosomal proteins are universal and are referred to as “exosome markers” (Głuszko et al., 2019). These include TSG101, ALIX and ESCRT complex (exosome biogenesis), RabGTPases and annexins (exosome delivery and membrane fusion), heat shock proteins (HSP70, HSP90), integrins, tetraspanins (CD9, CD63, CD81 and CD82), MHC class II proteins, epithelial cell adhesion molecules (EpCAM) and members of the human epidermal receptor (HER) family (Fais et al., 2013; Kalluri, 2016; Głuszko et al., 2019). Apart from the signature molecules present within them, there are certain specific proteins and nucleic acids acquired by the exosomes from their native cell types, which can serve as biomarkers for the identification of various diseases including cancer (Fais et al., 2013). Numerous genes and proteins have been identified in lung cancer cells and tissues that can serve as exosomal biomarkers for lung cancer. ZEB1, TRAF4, and TGF-β1 are involved in lung cancer metastasis by EMT proteins while PD-L1, EGFR, TLR7 and TLR8 are involved in inhibiting the immune system. Like other cancers, exosomes derived from breast cancer cells are enriched with certain miNAs that are not abundant in healthy cells. miR-372, miR-101 and miR-373 were not found in significantly higher proportions in exosomes from breast cancer cells. Certain nucleic acid molecules and proteins may also serve as diagnostic biomarkers of colorectal cancer (CRC). Among miRNAs, around 7 to 11 molecules have been identified to be differentially expressed in CRC patients. Similarly, in colorectal Cancer, the cell surface proteoglycan Glyptican1 (GPC1) serves as the most prominent biomarker of pancreatic cancer.
Ovarian cancer cells derived exosomes include membrane proteins, Rab proteins, annexin proteins, tetraspanins, heat shock proteins etc. can be used to potentially identify the malignancy early in its development. Besides, Helicobacter pylori infection is the most common factor that predisposes a person to develop gastric cancer by transporting the virulence factor CagA (mediator of gastric disorders) to epithelial cells and mesenchymal-epithelial transition factor (MET) protein to macrophages. The exosomes are enriched in lipids such as cholesterol, sphingomyelin, hexosylceramides, phosphatidylserine, phosphatidylcholine, phosphatidylethanolamines and saturated fatty acids (Figure 1) (Fais et al., 2013; Kalluri, 2016). Moreover, presence of lipid-raft like domains (due to membrane associated lipid-raft proteins) and/or phospholipid scramblase (responsible for translocating phospholipids of membrane leaflets) have also been consistently reported (Fais et al., 2013).
Exosomes are found to be enriched with the presence numerous classes of RNAs encompassing various expressed and significantly matured miRNAs and mRNAs. They are known to have eminent effects on physiological and developmental aspects of growth, development and regulation of expression in the recipient cell (Kalluri, 2016; Głuszko et al., 2019). Interestingly, recent studies have established a strong notion governing the fact that exosomes are acting as molecular vehicles by showing the presence of placental specific miRNA in the maternal blood exported via mature trophoblast, imparting the ability to modify genetic expressions (Fais et al., 2013).
Exosomes: Biogenesis
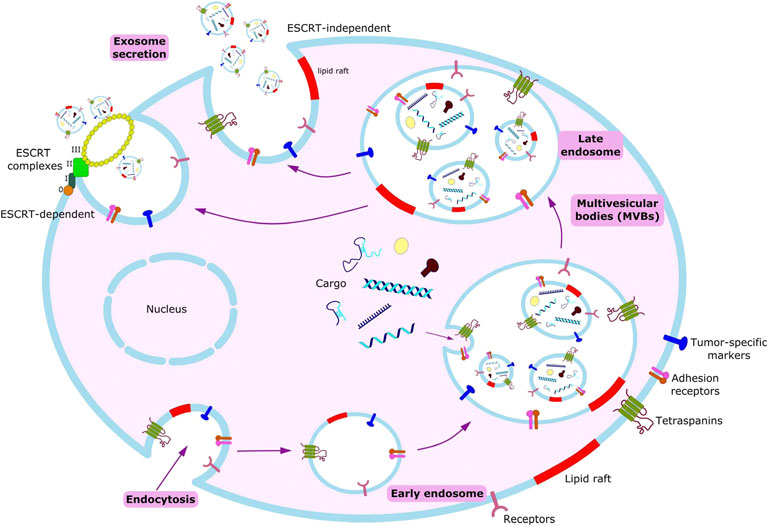
Exosomes are constitutively produced by cells through the inward budding of the plasma membrane leading to the formation of intracellular endosomes, which further fold in to form multivesicular bodies (MVBs). These MVBs contain nano-sized vesicles which after fusing with the plasma membrane release their contents to the extracellular space leading to the release of exosomes (Fais et al., 2013; Kalluri, 2016).
Exosomes are predominantly generated by the aid of endosomal sorting complexes required for transport complexes (ESCRT complexes) (Figure 2). ESCRT is composed of four complexes namely ESCRT-0, ESCRT-I, ESCRT-II and ESCRT-III along with several associated accessory proteins (ALIX, VPS4, Tsg101, VTA1) which sort ubiquitinated cargo proteins on the inner leaflet of the endosomal membrane and caused subsequent scission thereby releasing the exosomes (Kalluri, 2016; Yue et al., 2020).
Biogenesis begins with endocytosis, enclosing bioactive molecules, forming endosomes. Endosomal membrane further undergoes inward budding, enclosing the molecules and forming multivesicular bodies (MVBs). Exosomes form from these MVBs, either through ESCRT-dependent or ESCRT-independent pathways.
Based on recent studies, a key system has been elucidated called the Syndecan-syntenin pathway which is responsible for controlling the generation of endosomal vesicles that release exosomes as well as delivering cargo inside these vesicles. Syntenin bound to syndecans with the help of extracellular heparansulphate chains communicate with a myriad of signaling and adhesion moieties including ALIX protein which links the Syndecan-syntenin pathway with the ESCRT machinery (Fares et al., 2017; Hessvik and Llorente, 2018).
Remarkably, numerous studies have also indicated the presence of an ESCRT-independent pathway for exosome biogenesis and loading which is mediated by lipids and associated proteins. Proteins such as A2, RAB5/7/27, TSG101 have been identified to play important roles in exosome biogenesis (Kalluri, 2016). Several transmembrane proteins of tetraspanin family such as CD9, CD63, CD82, Tspan8 are also involved in ESCR-independent exosome generation. Another protein namely small integral membrane protein of the lysosome/late endosome (SIMPLE) has also been shown to positively influence the release of exosomes. Apart from proteins, various lipids such as Phosphatidic acid, ceramide, glycosphingolipids, lyso-phospholipid are also inducers of exosomebiogenesis (Hessvik and Llorente, 2018; Yue et al., 2020).
ESCRT-dependent or ESCRT-independent mechanisms are not entirely exclusive. Exosome biogenesis is instead a coordinated and synergistic outcome of these mechanisms wherein their presence or absence in a particular cell type and/or cellular homeostasis manipulates intraluminal vesicle number and size in addition to cargo sorting and loading (Yue et al., 2020). Moreover, exosome secretion is eminently regulated by the conditions of the cellular microenvironment. In tumor cells, stressful conditions arise due to several factors such as chemotherapeutics, irradiation, starvation and most notably hypoxia which together lead to increased production of exosomes (Głuszko et al., 2019).
Exosomes uptake
Exosomes from the extracellular space can adhere to the cell in its proximity nonspecifically (Tian et al., 2013) or be attached via a specific ligand-receptor complex (Ohno et al., 2013). They can either exert their functional effects by direct activation of a signaling pathway (Al-Nedawi et al., 2008; Cossetti et al., 2014; Patel et al., 2016) or be internalized to transfer their cargo inside the cell, via endosomal maturation: the endosomal-lysosomal degradative pathway (Nakase and Futaki, 2015). Non-specific internalization of exosomes has been shown to occur in both normal and transformed cell lines (Svensson et al., 2013). The uptake depends on the recipient cell and not on the origin of the exosomes (Horibe et al., 2018). It is an energy dependent process, as evidenced by attenuation of the uptake when incubated at 4°C or with compounds interfering with cell function (Morelli et al., 2004). It has been shown that low pH conditions, a consequence of hypoxia in the cell interstitium and a characteristic of tumor microenvironment lead to rupture of the exosomal membrane and subsequent uptake of its cargo through macropinocytosis (Joseph et al., 1996; Gatenby and Gillies, 2004; Taraboletti et al., 2006; Parolini et al., 2009). The two most frequently reported ways of exosomal uptake by cancer cells are Lipid-raft mediated endocytosis and macropinocytosis.
Clathrin-independent endocytosis (CIE) is reported to occur through membrane proteins, CAV-1, flotillin-1 and RhoA that are known to be components of lipid-rafts: the microdomains of plasma membrane that contain high cholesterol and glycosphingolipid concentration, involved in endocytosis (Costa Verdera et al., 2017). Local disruption of actin network and inhibition of dynamin recruitment to plasma membrane: both of which are important in CIE, decreased exosomal uptake, suggesting its role in endocytosis (Tian T. et al., 2014). Although, in a study, CAV1 knock-out cells showed increased uptake of exosomes, in most of them, CAV-1 has been shown to play important role in CIE, as described above (Feng et al., 2010; Chaudhary et al., 2014).
Clathrin-dependent endocytosis (CDE) although shown to be taking part in uptake in some cancer cells, is majorly non-existent (Tian T. et al., 2014).
Macropinocytosis (MP) is another widely discussed pathway of exosomal uptake by cancer cells. Exosomes can themselves induce MP, thus facilitating their uptake (Yan-Liang et al., 2014). EGFR expression has been shown in many tumors, when activated by EFG, enhanced uptake through MP via activation of Rac, leading to cytoskeletal organization and subsequent induction of MP (Nakase et al., 2015).
Role of exosomes in mitigating cancer metastasis
Exosomes and their contents function together as a unit, facilitating the promotion of malignancy and tumorigenic effects, aiding the ability of native epithelial cells. Serving as an alternative to conventional cell-based therapies, exosomes are currently being engineered to deliver therapeutic cargos to the targeted recipient or tumor cells (Gilligan and Dwyer, (2017); You et al., 2018; Głuszko et al., 2019). Furthermore, the most presumptuous property of cancer i.e. metastasis is governed by the localized impression of exosome-mediated signaling. The influence is diverse and can be either due to site-production of exosomes or through uptake by a distant recipient cell (Kalluri, 2016). The mediators proffered between tumor and their microenvironments are under constant modulation and are known to play a key role in cancer immunotherapy. Tumor-derived exosomes (TEXs) are one such modification, suitable for imparting chemotherapeutic resistance, influenced by various other strategies, survival time and tumor growth (You et al., 2018).
A study in 2018 accorded for observing induced apoptosis along with a combined outcome of reduced metastasis and prolonged survival when exosomal cargo, siKrasG12D-1, in the donor cells of Bone marrow MSCs-derived exosomes (BM-MSC), was electroporated with pancreatic cancer as the tumor model (Melo et al., 2014; Mendt et al., 2018; You et al., 2018). The most talked about are the cancer cell-derived exosomes that have been found accountable for the transformation of benign epithelial cells into malignant cells (Kalluri, 2016). Melo SA et al. demonstrated how cancer exosomes differ from normal exosomes as the former possesses the capability of independent transcription of miRNA through a Dicer-dependent pathway (Melo et al., 2014; Kalluri, 2016). It was reported that breast cancer associated exosomes enriched in miRNA exhibit the presence of RISC-Loading Complex (RLC), thus efficiently mediating the process of silencing and miRNA biogenesis (Mendt et al., 2018).
Additionally, a research study in 2015 investigated the chemo-sensitive resistance in tumors displayed by patients with Hepatocellular carcinoma (HCC). A cumulative effect of metastasis, inadequate prognosis and loss of miR-122 encapsulated within exosomes caused the patients to develop a resistance to chemotherapies involving agents such as 5-fluorouracil (5-FU) and doxorubicin (Lou et al., 2015). A subsequent observation was made, while testing, whether a modification revolving around Adipose-derived MSCs (AMSCs) can prove helpful in restoring the lost chemosenstivity via expression of miR-122. Reduction in the tumor mass and volume was observed when the engineered exosomes administered intra-tumoral to BALB/c nude mice with HepG2 tumors, combined with sorafenib treatment, thus increasing the HCC cell sensitivity (Lou et al., 2015; Gilligan and Dwyer, 2017).
Likewise, Tumor-derived exosomes (TEXs) regulate the process of tumor formation due to release of immunosuppressive molecules such as Fas-ligand (FasL), the expression of which contributes to resistance and malignant niche selection. Expression of exosomal secretions can also be traced to the responses and correlated levels of Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL), interleukin 10 (IL-10), programmed death-ligand 1 (PD-L1), neo-angiogenesis factors and several other microenvironment conditioning factors, e.g., transforming growth factor β1 (TGF-β1), prostaglandin E2 (PGE2) and ecto-enzymes engaged in the adenosine pathway (CD39 and CD73) (Głuszko et al., 2019).
The current cancer therapeutics deal with targeted destruction of both cancer stem cells (CSCs) alias cancer-initiating cells (CICs) as well as the non-CSCs. Several investigative experiments suggest that the CSC population is not static, and can be effectively reconfigured. The transformation of non-CSCs to CSCs results in regaining or acquiring stemness phenotype in the non-CSC tumor microenvironment which can be achieved by CSC-derived exosomes (Lin et al., 2013; Hu et al., 2015; Donnarumma et al., 2017).
The progressive inter-conversion establishes an equilibrium between the exosomal derived CSCs and non-CSCs, involving various cellular signaling pathways, bio-active cell cargo, molecular sorting and transport. Thus, a prospective scheme can be developed concerning possible mechanisms like cancer initiation, progression, metastasis, relapsing and resistance to therapies by modifying exosome contents in the cancer surrounding. A controlled and regulated interaction between CSCs and non-CSCs can be accomplished, which will prove to be a more beneficial and novel therapeutic strategy (Sun B. et al., 2018).
Role of exosomes in cancer: Signaling
Exosomes are capable of acting as transporters for different molecules aided by several types of signaling mechanisms and pathways that can operate over varying distances (Whiteside, 2017). Significantly, they play important roles in maintaining cancer-related functions such as metastasis, angiogenesis and regulating tumor micro-environment (Rachel et al., 2017; Whiteside, 2017; Sun Z. et al., 2018). Exosomes generated from Cancer Stem Cells (CSCs) are believed to induce stemness in non-CSCs thereby maintaining a state of equilibria in the tumor-microenvironment (Sun Z. et al., 2018). The ability of exosomes to act as communication vehicles is due to the presence of diverse signaling molecules on their surface derived from their parent cell as well as the presence of various nucleic acids, enzymes and factors inside them, thereby making them capable of acting as efficient mediators of cancer metabolism (Whiteside, 2017). Studies have elucidated that EVs from glioblastoma (GBM) under hypoxic conditions produce growth factors and cytokines which in turn activate directed pericyte movement and PI3K/AKT signaling and induce angiogenesis (Matarredona, 2020). Such EVs carry molecules like VEGF-A which straightaway promote angiogenesis (Are, 2016; Kalluri, 2016; Tomasetti et al., 2017). ECS have also shown enhanced sprouting and bifurcation of vessels after being delivered the components of the Notch pathway via exosome (Whiteside, 2017).
Exosomes from cancer cells have also been noted to bring about changes in fibroblasts via the TGFβ/Smad pathway. Cancer-Associated Fibroblasts (CAFs) can augment or inhibit tumor growth driven by tumor-derived exosomes (Whiteside, 2017; Sun Z. et al., 2018). Another study showed that the transfer of CRE mRNA to normal cells via EVs had pronounced immunosuppressive character (Are, 2016). The oncogenic potential of tumor cells is retained by the removal of tumor suppressor miRNAs via exosomes. Ras-MEK network is involved in maintaining the RNA-Induced Silencing Complex (RISC) which in turn leads to the release of miRNA in exosomes (Rachel et al., 2017). The uptake and attachment of exosomes in target cells are mediated by protein interactions via several adhesion molecules like integrins and ICAMs whereas heparin sulphate proteoglycans, carbohydrate/lectin receptors, T-cell immunoglobinmucin-binding phosphatidylserines, etc. are crucial for their entry inside the cells (Tomasetti et al., 2017). In another tumor microenvironment, the exosomes of prostate cancer cells were rich in H-Ras and K-Ras signaling mechanisms. They contained miRNAs and Rab proteins, which led to aggressive tumors in the recipient target cells. Likewise, cancer-cell derived exosomes having copious presence of Ras and other kinases in the MAP kinase pathway, phosphorylated EGFR, and other growth factors led to an increased longevity of tumor monocytes. It has further been demonstrated that the capability of tumor cells to manipulate healthy distant cells involving exosomes is via a mechanism needing Rho GTPase effectors Rac1/PAK2 which possibly is the basis of metastatic spread of tumors (Rachel et al., 2017). The exosomal signaling is not only dependent on growth factors and cytokines but several metabolites as well, which include lactate, proteins, ketone bodies, etc. (Whiteside, 2017).
As a new and potent method to combat cancer, targeting the precise signaling pathway of exosomes is an exceedingly promising and emergent aspect of cancer therapy (Sun Z. et al., 2018). Refined manipulation of exosomes, for instance by protein fused to a ligand that allows the targeted delivery of exosomes to neuronal cells is one such successful attempt at modifying and channeling the exosomal signaling mechanism for a noble use of therapeutics (Kalluri, 2016).
Role of exosomes in cancer: Biomarkers
The diverse cargo of exosomes consisting of circulating RNAs, proteins with membrane and cell functions are proving to be recent research tools as potential biomarkers. A number of recent studies are looking into the insights of exosomal release as indicative of patho-physiological conditions and not merely riddance of undesirable components (Lin et al., 2015). The results have been suggestive of superior rates of sensitivity and specificity involving diagnosis of various metabolic and infectious diseases and cancer tumors by exosomal cargo biomarkers (Lin et al., 2015; Wong and Chen, 2019). This ability is credited to potentiate and better coordination of intercellular communication exhibited by exosomes, amongst a deck of other responses such as oncogenic growth, tumor progression and signaling pathways (Ruivo et al., 2017; Batista and Melo, 2019; Mathew et al., 2020). Challenges and shortcomings of existing biomarkers such as invasive and predictive nature, limited responsiveness and incompetency of genomic biomarkers in efficiently determining adaptive immune responses (Conway et al., 2018; Mathew et al., 2020); have paved way for development of novel diagnostic tools: the exosomal engineered biomarkers. Islet autoantibodies, like GAD65, IA-2 also known as ICA512, accurately predict development of type 1 diabetes mellitus; likely reduce with the disease progression and the antibodies gets exhausted as soon as insulin therapy is initiated (Towns and Pietropaolo, 2011; Garcia-Contreras et al., 2017). The cytokine stimulated ß-cells releasing EXOs are hence being utilized for TID diagnosis involving analysis of specific proteins and RNAs preceding isolation by using a surface marker (Palmisano et al., 2012; Garcia-Contreras et al., 2017).
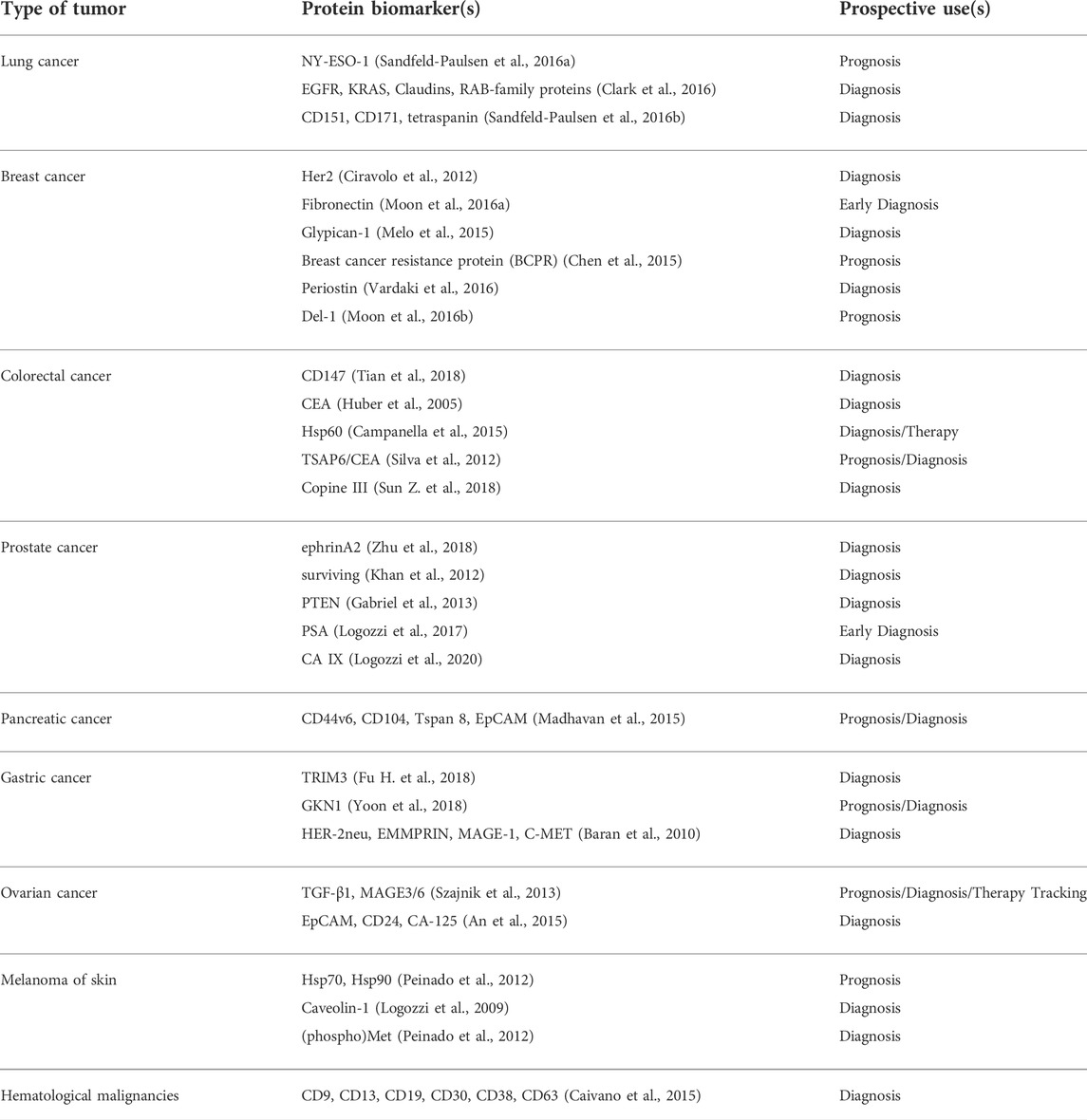
In 2009, a study reported elevated levels of CD63+ (scaffolding membrane protein) exosomes in plasma isolated from melanoma patients; qualifying as a tumor-associated marker based on western blot and flow-cytometric analysis (Logozzi et al., 2009; Lin et al., 2015). The tetraspanin family member was also found helpful in carrying out comparison of various human cancers; when quantification data showed lower levels of CD63 in exosomes derived from non-cancer cells. In addition, they also proposed reliability of CD9 and CD81 as marker proteins based on the fact that both of them were profoundly found in all the (four prostate and five breast) cell lines, that they utilized (Yoshioka et al., 2013; Lin et al., 2015). Several other exosomal proteins have also found their place as suitable prognostic tools in clinical and disease pathologies.
Findings of Taylor et al. regarding exosomal associated eight miRNAs (miR-21, -141, -200a, -200b, -200c, -203, -205, -214) are suggestive of the applications in diseases like ovarian cancer as substitute biomarkers; involving plasma biofluid and are reported to overcome the invasive isolation and profiling of biopsy samples (Taylor and Gercel-Taylor, 2008; Lin et al., 2015). In this regard, cell culture medium biofluids; have found predictive use in metastatic gastric cancers. Let-7 family miRNAs enriched extracellular fractions indicated by signal intensity data, confirming the possibility of selective secretion, and consequently revealed a new disease marker (Ohshima et al., 2010; Lin et al., 2015). Thus, exosomal nucleic acids also possess great potential as biomarkers for cancer diagnosis.
Such expositions by researchers are satisfactory and are coaxing others to take this descriptive nature of the published accounts to a further expanded diagnostic setting, particularly development of ideal biomarkers (Tables 1, 2).
Lung cancer
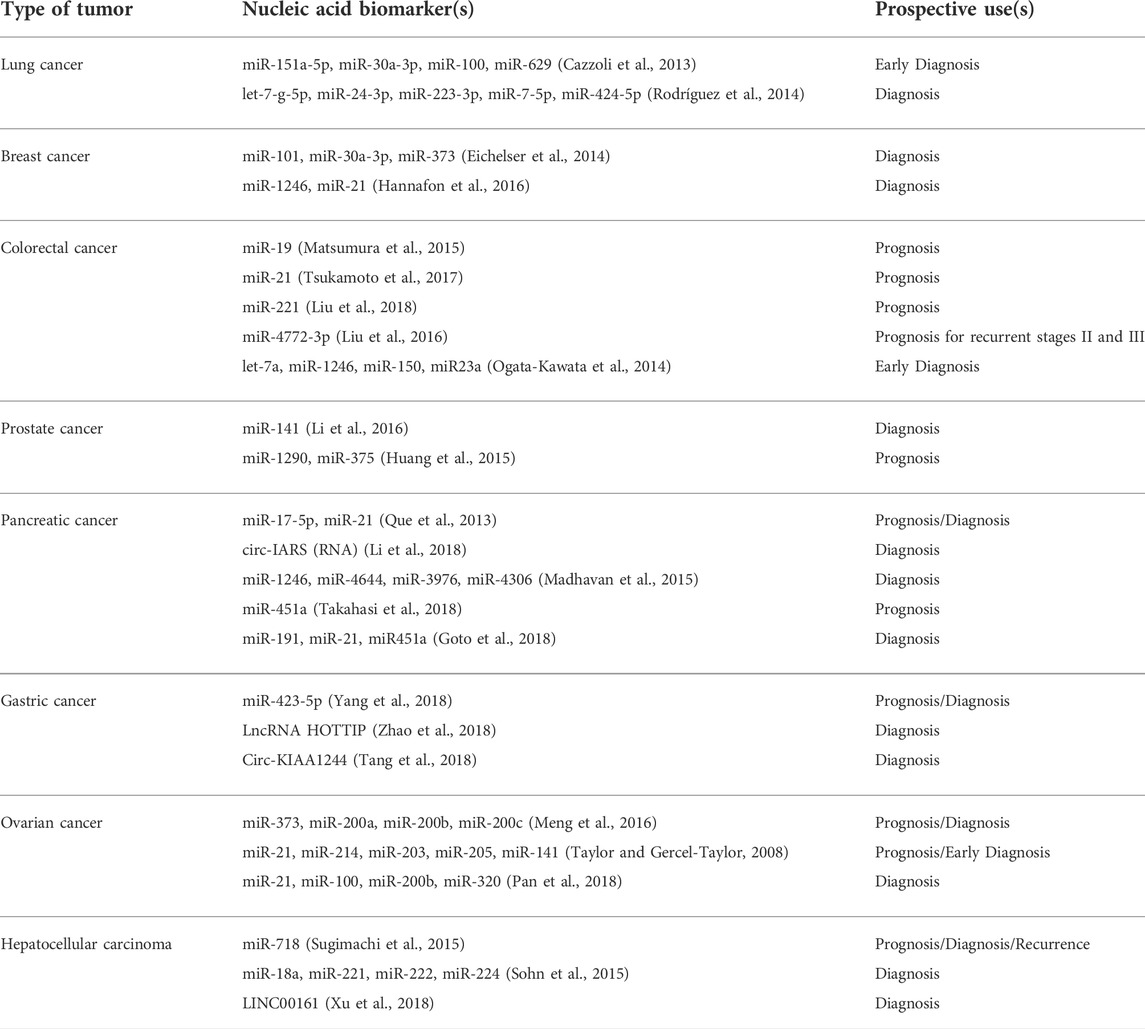
Multiple genes and proteins have been identified in lung cancer cells and tissues that can serve as exosomal biomarkers for lung cancer. The most prominent ones are the proteins (ZEB1, TRAF4, TGF-β, etc.) involved in lung cancer metastasis by EMT, proteins (PD-L1, EGFR, TLR7, TLR8) involved in inhibiting the immune system, and several Wnt proteins (Wnt5b, Wnt3a) and Interleukins (IL-6, IL-8, IL-10) that allow invasion and proliferation of the tumor (Jiang et al., 2021). A number of miRNAs that are an integral part of the exosomal cargo of the lung cancer cells have also been noted. They include miR-660-p5, miR-29a, miR-21 and miR494 (promote proliferation of lung cancer cells), miR-5100, miR-9, miR-23a (promote metastasis), miR-21, miR-29 (promote angiogenesis), and miR-23a (involved in immunomodulation) (Xu et al., 2021).
Breast cancer
As seen with other cancers, exosomes derived from breast cancer cells are enriched with certain miNAs that are not abundant in healthy cells. miR-372, miR-101, and miR-373 were found in significantly higher proportions in exosomes from breast cancer cells. Further, these miRNA are also indicative of metastasizing cancer while miR-373 is a marker of the highly aggressive triple-negative phenotype of breast cancer (Joyce et al., 2016). Expression levels of proteins such as ER (estrogen receptor), Ki67 (a marker of proliferation Ki-67), PR (progesterone receptor), and HER2 (member of the epidermal growth factor receptor family which is involved in the regulation of cell growth, survival, and differentiation via targeting multiple signal transduction pathways) can serve as important biomarkers for breast cancer prognosis and diagnosis (Jafari et al., 2018).
Colorectal cancer
Certain nucleic acid molecules and proteins may also serve as diagnostic biomarkers of colorectal cancer (CRC). Among miRNAs, around 7–11 molecules have been identified to be differentially expressed in CRC patients out of which miR-23a, miR-1246, and miR-21 are considered better markers. Several lncRNAs (colorectal neoplasia differentially expressed-h (CRNDE-h), breast cancer anti-estrogen resistance 4 (BCAR4), mRNA keratin-associated protein 5-4 (KRTAP5-4), and mRNA melanoma antigen family A3 (MAGEA3) have also been found in higher amounts in serum exosomes, thus increasing the scope of using them as predictive as well as diagnostic molecules. Upregulation and downregulation of specific proteins may serve as another method to screen CRC patients. Heat shock protein 60 (a chaperonin involved in tumorigenesis), glypican-1, and the transmembrane protein CD147 are increasingly expressed in Colorectal Cancer, hence could be potential candidates for diagnosis (Balacescu et al., 2018; Xiao et al., 2020).
Pancreatic cancer
Similar to Colorectal Cancer, the cell surface proteoglycan Glyptican1 (GPC1) is the most prominent biomarker of pancreatic cancer. Exosomes enriched in GPC1 are known to positively regulate cancer and thereby serve as the best biomarker for detecting pancreatic cancer (Melo et al., 2015). Further, based on the study of exosomes isolated from pancreatic cancer cell lines and plasma isolated from patients, miRNAs such as miR-196a, miR-1246, miR-191, miR-21, miR-451a, miRNA-483-3p, miR-155, miR-196a, etc. are present in ample amount in the pancreatic cancer tumor microenvironment and may be effectively used to diagnose the same (Gabriel et al., 2020).
Ovarian cancer
Ovarian cancer cells derived exosomes are extracted from either ascites or serum of patients and contain a concoction of specific signature molecules which help in the progress of the tumor. It includes membrane proteins (Alix, TSG 101), Rab proteins, annexin proteins, tetraspanins (CD9, CD82, CD63 and CD81), heat shock proteins (Hsp90, Hsp70), antigens (MHC I and II), Nanog and enzymes (phosphate isomerase, peroxiredoxin, aldehyde reductase, fatty acid synthase), which can be used to potentially identify the malignancy early in itsdevelopment (Feng et al., 2019). Since ovarian cancer is highly lethal yet lacks any early screening test, therefore using exosomalmiRNA biomarkers (miR-100, miR-200b, miR-320, miR-21, miR-362-5p, and miR-1274a etc.) for diagnosis and prognosis of ovarian cancer would be of great clinical utility (Yoshida et al., 2020).
Gastric cancer
Helicobacter pylori infection is the most common factor that predisposes a person to develop gastric cancer. Interestingly, studies have found the role of exosomes in H. pylori infection and tumorigenesis by transporting the virulence factor CagA (mediator of extragstric disorders) to epithelial cells and mesenchymal-epithelial transition factor (MET) protein to macrophages, thus aiding in disease progression (Tang et al., 2021). Other signature molecules that form a part of the exosomes cargo from GC cells include proteins (UBR2, TRIM3, Apolipoprotein E), miRNAs (miR-423-5p, miR-155-5p, miR-27a, etc.), IncRNA (ZFAS1, LINC00152), and circRNA (ciRS-133, circ-KIAA1244) which may be utilized as characteristic biomarker for early diagnosis of gastric cancer (Fu et al., 2019).
Exosomes as therapeutic targets
Modification of exosome content
By default, exosomes are generally engineered under the control and governance of various cellular mechanisms; however, an accelerating number of successful researches are presently being done that involve exploring possibilities of exosomal content modification. The biocompatible traits of exosomes, with several appropriate changes, can trigger the steadiness and efficacy of cellular uptake and prove to be an effective step in improving the picture of current therapeutics. The cognizance of this subject matter is to summarize perspective, passive and lively approaches to unique exosome changes, and examples of the transport molecules (Luan et al., 2017).
Exosomal cargos such as nucleic acid components, heat shock proteins, and various ligand molecules, for example, miR-425-3p, TGF- β, miR-100–5p, and Survivin, are found to be effectively involved with various targeted cells such as TGF- β is with NK cells. These modified exosomes, due to the incorporation of desirable components can prove to be an efficient vehicle in dealing with advancing cases of cancers such as AML, Lung cancers, Hepatocellular cancers, etc. A platinum-based chemotherapeutic approach was carried out to monitor a Lung cancer model, utilizing exosomal content modification and miR-425 as the cargo component. It was rendered ineffective resulting in a resistive response to NSCLC which was an outcome of autophagy due to AKT1 inhibition. Similarly, NK cell-mediated suppression controlled by tumor-derived vesicles proved to be therapeutic in instances of AML accompanied by the opposition of suppression by interleukin-15. The study outcome in the case of A549 cells involved DDP resistance and was observed in resident cancer cells due to modulation of mTOR expression. In another study, Exosomes purified from HeLa Cervical Carcinoma cells exhibited revised survival rates due to assistance from IAP and HSPs (Refer to the table).
One such critical group of derived exosomes comes beneath the mega-group of Tumor-derived exosomes. There are an array of attractive components that contributes to the usage of tumor-derived exosomes for the transport of therapeutics and vaccines for immunotherapy. A stage 1 medical trial has recently been accomplished on the discharge of tumor exosomes, which had been earlier presumed to undergo tumor specificity via antigens equipped for presentation to immune cells and stimulating the immune structures of glioma sufferers to achieve pure and ultimate tumor cells after resection (Thomas Jefferson University, 2000).
For example, for the determination of tumor cells and tumor exosomes in excessive numbers in malignant effusions, it has been established that tumor exosomes convey tumor-related antigens unique to the tumors from which they may be derived, in addition to MHC I molecules. A supply of antigens to dendritic cells by tumor exosomes can result in a T-cellular-mediated immune reaction towards tumor cells (Wolfers et al., 2001). In addition, tumor-focused on selective drug transport involves tumor-derived exosomes and has been proposed as an opportunity due to their unique expression of tetraspanins, which preferentially engage with ligands in specific tissues (Rana et al., 2012). Proteases, including urokinase plasminogen activator, which promotes tumor cellular invasion, and cathepsin D, and adhesion modulators, including vimentin, galectin 3-binding protein, and annexin A1, have additionally been determined in tumor-derived exosomes (Harris et al., 2015); miRNAs and different nucleic acids, that may result in malignant adjustments in target cells, had been identified in tumor cellular exosomes (Melo et al., 2014). An account of exosomal alterations manifested as therapeutic and restorative in specific cancer treatments is enlisted below in Table 3.
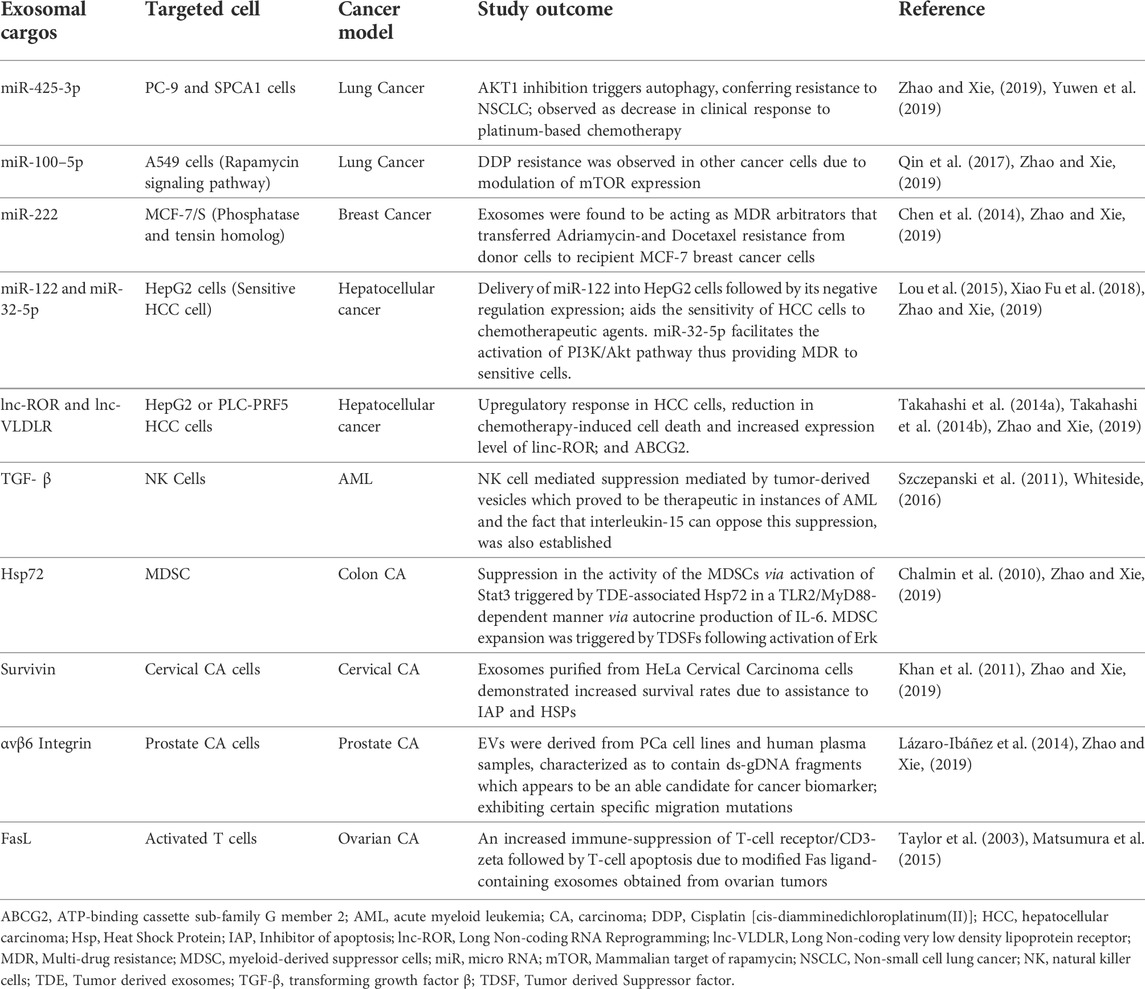
TABLE 3. An account of exosomal alterations manifested as therapeutic and restorative in specific cancer treatments.
Modification of exosome surface
Exosomal surface proteins (ligands) can be modified to aid targeted drug delivery. It is carried out to image and track them, make them better adapted to the target cells vis-à-vis anchorage and uptake, increase their therapeutic value, and give them other possible advantages over unmodified exosomes (Sandfeld-Paulsen et al., 2016a). This specific modification is beneficial in terms of more effective drug delivery, more retention in circulation and more stability. Furthermore, since it augments the targeted delivery, a lesser quantity of exosomes is required for the same effect, thus reducing the need for high yield from parent cells (Zhang et al., 2020).
These modifications are done through various methods, depending upon the requirement, including genetic engineering of parent cells, nanoparticle technology, hydrophobic cargo loading in the lipid bilayer, fusion with liposomes, etc. (Xu et al., 2020). Table 4 summarizes a list of practical changes carried out on exosomal surfaces to assist their involvement in promoting advancements and therapies.
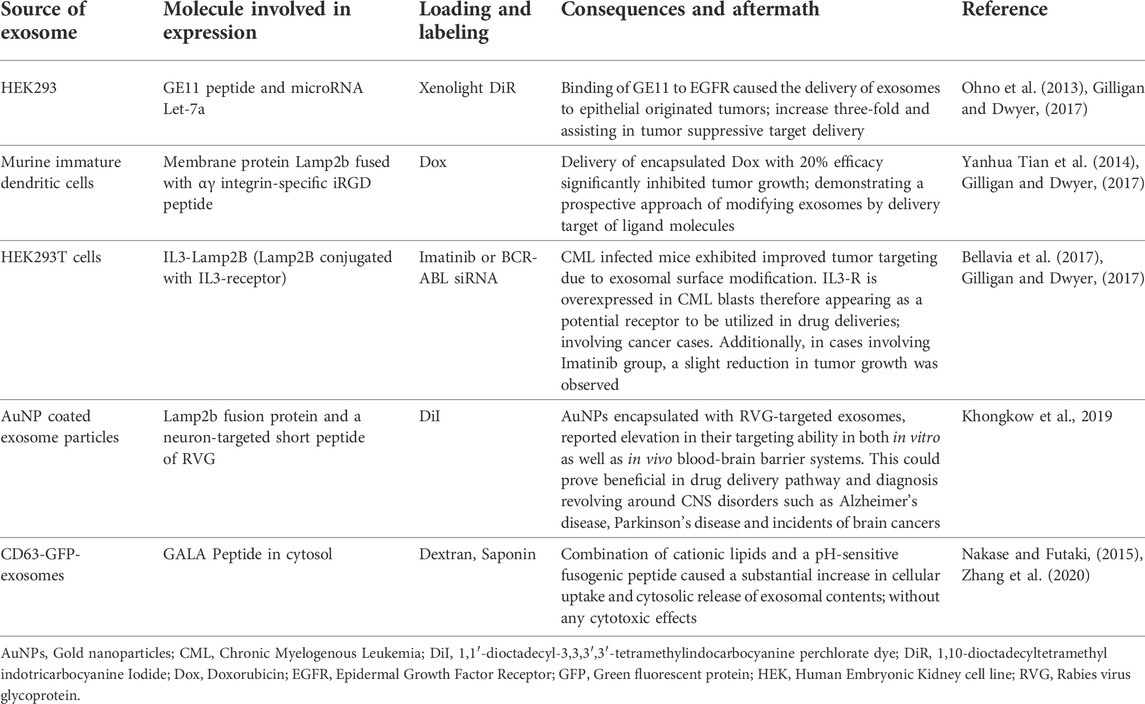
TABLE 4. A list of practical changes carried out on exosomal surfaces to assist their involvement in promoting advancements and therapies.
The prospects of exosome mimetics
The role of exosomes in multiple cancer types and their potential utility as targeted therapeutics by its alteration or through exosome-mimetics has been widely discussed in this paper. Additionally, since exosomes are functionally involved and produced by almost all types of cells, their application is also omnipresent and is witnessed throughout various ailments. Apart from cancer, exosomes have been studied as ideal vehicles for drug delivery for several neurodegenerative diseases like Alzheimer’s disease and Parkinson’s disease, cardiovascular disorders, musculoskeletal diseases, Kidney ailments, diabetes, etc. (Antimisiaris et al., 2018; Jiang et al., 2019). A revolutionary aspect of exosomes is their ability to act as nanocarriers for the delivery of therapeutic agents in brain to treat disorders pertaining to the CNS (Antimisiaris et al., 2018). Neuron-derived exosomes (NDEs) have also been implied to contain distinguishing biomarkers for HIV-associated neurological disorders (HAND) and Alzheimer’s Disease (AD) (Pulliam et al., 2019).
Studies have also been performed to show how circulating EVs might have a functional role in the pathophysiology of several vascular disorders such as Acute Chest Syndrome (ACS) and Sickle Cell Disease (Lapping-Carr et al., 2020). Experimentally, it has been demonstrated that Mesenchymal Stem Cell Derived-Exosomes (MEX) suppresses lung infection by regulating the lung tissue therefore, thereby lies a possible remedial for Pulmonary Arterial Hypertension (Willis et al., 2018). Urinary exosomes as well give an insight into diverse biomarkers that are pointers to different drug-induced kidney toxicities. Furthermore, this aspect can be scaled up after subsequent research for large-scale drug trials (Griffin et al., 2020).
Preclinical research has displayed the ability of MSC-derived exosomes as an alternative form of therapy for Acute Respiratory Distress Syndrome (ARDS). Furthermore, MSC-derived exosomes behave as silencing complexes; hence they can induce some epigenetic changes in the expression of their cellular receptors, eventually leading to the inability of infection of many RNA viruses like Hepatitis-C, Influenza and Coronavirus. This is suggestive of the underlying aptitude of MSC-derived exosomes to treat COVID-19 infection (Gupta et al., 2020).
\Although the role of exosomes as curative agents is highly promising, there are quite a few limitations that need to be tackled first to unleash its extensive utility. The shortcomings of exosomes therapeutics include scanty yield of exosomes from cells, difficulty in loading drugs and engineering the vesicles, potential unwanted effect at non-target sites, systemic dilution before reaching the target site, change in the conformation of membrane protein (thus affecting organotropism), questionable stability of the engineered-vesicles etc. (Antimisiaris et al., 2018; Hu et al., 2020). Thus, the use of exosomes-mimetics, which are membrane-coated nanoparticles having the same functional characteristics and efficacy as endogenous exosomes appear more promising for the same therapeutic purpose. Additionally, it can be quickly produced, engineered and loaded with our desired drug in a reproducible and relatively cost-effective manner (Hu et al., 2020).
Conclusion
This article has comprehensively discussed exosomes and their quintessential involvement in various facets of Cancer Biology. Firstly, there is enough substantial evidence that solidifies the role of exosomes in cell-to-cell communication and signaling and regulation of tumor micro-environment. Secondly, exosomes aid in malignancy of the disease by promoting metastasis and tumor progression. Moreover, they can be characterized to act as biomarkers for efficient diagnostic applications. Thus, these extra-cellular vesicles are multidimensional in terms of the functions that they perform and have thereby emerged as a highly promising niche for cancer therapeutics. It is remarkable to note their efficient, intrinsic targeting potential, biocompatibility, efficacy and physiological stability.
However, numerous aspects, such as purification, administration, standardization and long-term safety effects, require to be studied and monitored. The current studies also lack clinical trials on human models, which are essential to further advancements in this area. Despite their expanded hopes of applications, more advanced and robust technologies are required to isolate surplus exosomes to counter the scanty yield from the current methods. Also, a scalable and economical method for loading drugs/nucleic acids and modifying the surface of exosome mimetics is awaited that also preserves the integrity and innate characteristics of these engineered vesicles.
We have partly unraveled the search for the perfect, fool-proof tool for the cancer treatment, but the quest is yet incomplete until the abovementioned voids are filled. Conclusively, exosomes undeniably carry a plethora of possibilities to revolutionize and significantly optimize Cancer therapeutics and diagnostics. Even, a multitude of research still needs to be performed and analyzed to apply the pre-clinical proof of concept studies to fruition.
Author contributions
LM- Concept and Manuscript preparation GM- Manuscript Preparation, concept and Corresponding Author UP- Figure construction and Editing AG- Manuscript Preparation JG- Manuscript and Table preparation MS- Proof reading.
Conflict of interest
Author MS was employed by Betterhumans Inc.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations:
ARF, ADP ribosylation factor; LAMP, lysosome-associated membrane protein; MHC, major histocompatibility complex; MUC1, mucin 1; PGK-1, phosphoglycerate kinase 1; TfR, transferrin receptor.
References
Al-Nedawi, K., Meehan, B., Micallef, J., Lhotak, V., May, L., Guha, A., et al. (2008). Intercellular transfer of the oncogenic receptor EGFRvIII by microvesicles derived from tumour cells. Nat. Cell. Biol. 10 (5), 619–624. doi:10.1038/ncb1725
An, T., Qin, S., Xu, Y., Tang, Y., Huang, Y., Situ, B., et al. (2015). Exosomes serve as tumour markers for personalized diagnostics owing to their important role in cancer metastasis. J. Extracell. Vesicles 4, 27522. doi:10.3402/jev.v4.27522
Antimisiaris, S. G., Mourtas, S., and Marazioti, A. (2018). Exosomes and exosome-inspired vesicles for targeted drug delivery. Pharmaceutics Vol. 10, E218. doi:10.3390/pharmaceutics10040218
Are, W. W. (2016). Review communication by extracellular vesicles : Where we are and where we need to go. Cell. 164 (6), 1226–1232. doi:10.1016/j.cell.2016.01.043
Balacescu, O., Sur, D., Cainap, C., Visan, S., Cruceriu, D., Manzat-Saplacan, R., et al. (2018). The impact of miRNA in colorectal cancer progression and its liver metastases. Int. J. Mol. Sci. 19 (12), 3711. doi:10.3390/ijms19123711
Baran, J., Bajkrzyworzeka, M., Weglarczyk, K., Szatanek, R., Zembala, M., Barbasz, J., et al. (2010). Circulating tumour-derived microvesicles in plasma of gastric cancer patients. Cancer Immunol. Immunother. 59 (6), 841–850. doi:10.1007/s00262-009-0808-2
Batista, I. A., and Melo, S. A. (2019). Exosomes and the future of immunotherapy in pancreatic cancer. Int. J. Mol. Sci. 20 (3), 567. doi:10.3390/ijms20030567
Bellavia, D., Raimondo, S., Calabrese, G., Forte, S., Cristaldi, M., Patinella, A., et al. (2017). Interleukin 3- receptor targeted exosomes inhibit in vitro and in vivo Chronic Myelogenous Leukemia cell growth. Theranostics 7 (5), 1333–1345. doi:10.7150/thno.17092
Caivano, A., Laurenzana, I., De Luca, L., La Rocca, F., Simeon, V., Trino, S., et al. (2015). High serum levels of extracellular vesicles expressing malignancy-related markers are released in patients with various types of hematological neoplastic disorders. Tumour Biol. 36, 9739–9752. doi:10.1007/s13277-015-3741-3
Campanella, C., Rappa, F., Sciumè, C., Marino Gammazza, A., Barone, R., Bucchieri, F., et al. (2015). Heat shock protein 60 levels in tissue and circulating exosomes in human large bowel cancer before and after ablative surgery: Exosomal Hsp60 in Large Bowel Cancer. Cancer 121, 3230–3239. doi:10.1002/cncr.29499
Cazzoli, R., Buttitta, F., Di Nicola, M., Malatesta, S., Marchetti, A., Rom, W. N., et al. (2013). microRNAs derived from circulating exosomes as noninvasive biomarkers for screening and diagnosing lung cancer. J. Thorac. Oncol. 8, 1156–1162. doi:10.1097/JTO.0b013e318299ac32
Chalmin, F., Ladoire, S., Mignot, G., Vincent, J., Bruchard, M., Remy-Martin, J. P., et al. (2010). Membrane-associated Hsp72 from tumor-derived exosomes mediates STAT3-dependent immunosuppressive function of mouse and human myeloid-derived suppressor cells. J. Clin. Invest. 120 (2), 457–471. doi:10.1172/JCI40483
Chaudhary, N., Gomez, G. A., Howes, M. T., Lo, H. P., McMahon, K. A., Rae, J. A., et al. (2014). Endocytic crosstalk: Cavins, caveolins, and caveolae regulate clathrin-independent endocytosis. PLoS Biol. 12, e1001832. doi:10.1371/journal.pbio.1001832
Chen, W. X., Liu, X. M., Lv, M. M., Chen, L., Zhao, J. H., Zhong, S. L., et al. (2014). Exosomes from drug-resistant breast cancer cells transmit chemoresistance by a horizontal transfer of microRNAs. PloS one 9 (4), e95240. doi:10.1371/journal.pone.0095240
Chen, Y., Wang, L., Zhu, Y., Chen, Z., Qi, X., Jin, L., et al. (2015). Breast cancer resistance protein (BCRP)-containing circulating microvesicles contribute to chemoresistance in breast cancer. Oncol. Lett. 10, 3742–3748. doi:10.3892/ol.2015.3806
Ciravolo, V., Huber, V., Ghedini, G. C., Venturelli, E., Bianchi, F., Campiglio, M., et al. (2012). Potential role of HER2-overexpressing exosomes in countering trastuzumab-based therapy. J. Cell. Physiol. 227, 658–667. doi:10.1002/jcp.22773
Clark, D. J., Fondrie, W. E., Yang, A., and Mao, L. (2016). Triple SILAC quantitative proteomic analysis reveals differential abundance of cell signaling proteins between normal and lung cancer-derived exosomes. J. Proteomics 133, 161–169. doi:10.1016/j.jprot.2015.12.023
Conway, J. R., Kofman, E., Mo, S. S., Elmarakeby, H., and Van Allen, E. (2018). Genomics of response to immune checkpoint therapies for cancer: Implications for precision medicine. Genome Med. 10, 93. doi:10.1186/s13073-018-0605-7
Cossetti, C., Iraci, N., Mercer, T. R., Leonardi, T., Alpi, E., Drago, D., et al. (2014). Extracellular vesicles from neural stem cells transfer IFN-γ via Ifngr1 to activate Stat1 signaling in target cells. Mol. Cell. 56 (2), 193–204. doi:10.1016/j.molcel.2014.08.020
Costa Verdera, H., Gitz-Francois, J. J., Schiffelers, R. M., and Vader, P. (2017). Cellular uptake of extracellular vesicles is mediated by clathrin-independent endocytosis and macropinocytosis. J. Control. Release 266, 100–108. doi:10.1016/j.jconrel.2017.09.019
Donnarumma, E., Fiore, D., Nappa, M., Roscigno, G., Adamo, A., Iaboni, M., et al. (2017). Cancer-associated fibroblasts release exosomal microRNAs that dictate an aggressive phenotype in breast cancer. Oncotarget 8, 19592–19608. doi:10.18632/oncotarget.14752
Eichelser, C., Stückrath, I., Müller, V., Milde-Langosch, K., Wikman, H., Pantel, K., et al. (2014). Increased serum levels of circulating exosomal microRNA-373 in receptor-negative breast cancer patients. Oncotarget 5, 9650–9663. doi:10.18632/oncotarget.2520
Fais, S., Logozzi, M., Lugini, L., Federici, C., Azzarito, T., Zarovni, N., et al. (2013). Exosomes: The ideal nanovectors for biodelivery. Biol. Chem. 394 (1), 1–15. doi:10.1515/hsz-2012-0236
Fares, J., Kashyap, R., and Zimmermann, P. (2017). Syntenin: Key player in cancer exosome biogenesis and uptake? Cell. adh. Migr. 11, 124–126. doi:10.1080/19336918.2016.1225632
Feng, D., Zhao, W. L., Ye, Y. Y., Bai, X. C., Liu, R. Q., Chang, L. F., et al. (2010). Cellular internalization of exosomes occurs through phagocytosis. Traffic 11, 675–687. doi:10.1111/j.1600-0854.2010.01041.x
Feng, W., Dean, D. C., Hornicek, F. J., Shi, H., and Duan, Z. (2019). Exosomes promote pre-metastatic niche formation in ovarian cancer. Mol. Cancer 18 (1), 124. doi:10.1186/s12943-019-1049-4
Fu, H., Yang, H., Zhang, X., Wang, B., Mao, J., Li, X., et al. (2018). Exosomal TRIM3 is a novel marker and therapy target for gastric cancer. J. Exp. Clin. Cancer Res. 37, 162. doi:10.1186/s13046-018-0825-0
Fu, M., Gu, J., Jiang, P., Qian, H., Xu, W., Zhang, X., et al. (2019). Exosomes in gastric cancer: Roles, mechanisms, and applications. Mol. Cancer 18 (1), 41. doi:10.1186/s12943-019-1001-7
Fu, X., Liu, M., Qu, S., Ma, J., Zhang, Y., Shi, T., et al. (2018). Exosomal microRNA-32-5p induces multidrug resistance in hepatocellular carcinoma via the PI3K/Akt pathway. J. Exp. Clin. Cancer Res. 37 (1), 52. doi:10.1186/s13046-018-0677-7
Gabriel, A. N. A., Wang, F., Jiao, Q., Yvette, U., Yang, X., Al-Ameri, S. A., et al. (2020). The involvement of exosomes in the diagnosis and treatment of pancreatic cancer. Mol. Cancer 19 (1), 132. doi:10.1186/s12943-020-01245-y
Gabriel, K., Ingram, A., Austin, R., Kapoor, A., Tang, D., Majeed, F., et al. (2013). Regulation of the tumor suppressor pten through exosomes: A diagnostic potential for prostate cancer. PLoS ONE 8, e70047. doi:10.1371/journal.pone.0070047
Garcia-Contreras, M., Brooks, R. W., Boccuzzi, L., Robbins, P. D., and Ricordi, C. (2017). Exosomes as biomarkers and therapeutic tools for type 1 diabetes mellitus. Eur. Rev. Med. Pharmacol. Sci. 21 (12), 2940–2956.
Gatenby, R. A., and Gillies, R. J. (2004). Why do cancers have high aerobic glycolysis? Nat. Rev. Cancer 4 (11), 891–899. doi:10.1038/nrc1478
Gilligan, K. E., and Dwyer, R. M. (2017). Engineering exosomes for cancer therapy. Int. J. Mol. Sci. 18 (6), 1122. doi:10.3390/ijms18061122
Głuszko, A., Szczepański, M. J., Ludwig, N., Mirza, S. M., and Olejarz, W. (2019). Exosomes in cancer: Circulating immune-related biomarkers. BioMed Res. Int. 1628029, 9. doi:10.1155/2019/1628029
Goto, T., Fujiya, M., Konishi, H., Sasajima, J., Fujibayashi, S., Hayashi, A., et al. (2018). An elevated expression of serum exosomal microRNA-191, −21, −451a of pancreatic neoplasm is considered to be efficient diagnostic marker. BMC Cancer 18, 116. doi:10.1186/s12885-018-4006-5
Griffin, B. R., Faubel, S., and Edelstein, C. L. (2020). U. S. Department of veterans affairs, 41, 213–226. doi:10.1097/FTD.0000000000000589.Biomarkers
Gupta, A., Kashte, S., Gupta, M., Rodriguez, H. C., Gautam, S. S., Kadam, S., et al. (2020). Mesenchymal stem cells and exosome therapy for COVID-19: Current status and future perspective. Hum. Cell. 4, 907–918. doi:10.1007/s13577-020-00407-w
Hannafon, B. N., Trigoso, Y. D., Calloway, C. L., Zhao, Y. D., Lum, D. H., Welm, A. L., et al. (2016). Plasma exosome microRNAs are indicative of breast cancer. Breast Cancer Res. 18, 90. doi:10.1186/s13058-016-0753-x
Harris, D. A., Patel, S. H., Gucek, M., Hendrix, A., Westbroek, W., and Taraska, J. W. (2015). Exosomes released from breast cancer carcinomas stimulate cell movement. PLoS One 10, e0117495. doi:10.1371/journal.pone.0117495
Hessvik, N. P., and Llorente, A. (2018). Current knowledge on exosome biogenesis and release. Cell. Mol. Life Sci. 75, 193–208. doi:10.1007/s00018-017-2595-9
Horibe, S., Tanahashi, T., Kawauchi, S., Murakami, Y., and Rikitake, Y. (2018). Mechanism of recipient cell-dependent differences in exosome uptake. BMC Cancer 18 (1), 47. doi:10.1186/s12885-017-3958-1
Hu, Q., Su, H., Li, J., Lyon, C., Tang, W., Wan, M., et al. (2020). Clinical applications of exosome membrane proteins. Precis. Clin. Med. 1, 54–66. doi:10.1093/pcmedi/pbaa007
Hu, Y., Yan, C., Mu, L., Huang, K., Li, X., Tao, D., et al. (2015). Fibroblast-derived exosomes contribute to chemoresistance through priming cancer stem cells in colorectal cancer. PLoS One 10, e0125625. doi:10.1371/journal.pone.0125625
Huang, X., Yuan, T., Liang, M., Du, M., Xia, S., Dittmar, R., et al. (2015). Exosomal miR-1290 and miR-375 as prognostic markers in castration-resistant prostate cancer. Eur. Urol. 67, 33–41. doi:10.1016/j.eururo.2014.07.035
Huber, V., Fais, S., Iero, M., Lugini, L., Canese, P., Squarcina, P., et al. (2005). Human colorectal cancer cells induce T-cell death through release of proapoptotic microvesicles: Role in immune escape. Gastroenterology 128, 1796–1804. doi:10.1053/j.gastro.2005.03.045
Jafari, S. H., Saadatpour, Z., Salmaninejad, A., Momeni, F., Mokhtari, M., Nahand, J. S., et al. (2018). Breast cancer diagnosis: Imaging techniques and biochemical markers. J. Cell. Physiol. 233 (7), 5200–5213. doi:10.1002/jcp.26379
Jiang, C., Zhang, N., Hu, X., and Wang, H. (2021). Tumor-associated exosomes promote lung cancer metastasis through multiple mechanisms. Mol. Cancer 20 (1), 117. doi:10.1186/s12943-021-01411-w
Jiang, L., Dong, H., Cao, H., Ji, X., Luan, S., Liu, J., et al. (2019). Exosomes in pathogenesis, diagnosis, and treatment of alzheimer’s disease. Med. Sci. Monit. 25, 3329–3335. doi:10.12659/MSM.914027
Joseph, D., Penland Lolita, B. P. O., Tyagi, S., Kramer, F. R., and Group, N. P. (1996). © 1997 nature publishing group. Group, 4, 303–308. Available at: http://www.nature.com/naturemedicine. https://doi.org/10.1038/nm0798-822
Joyce, D. P., Kerin, M. J., and Dwyer, R. M. (2016). Exosome-encapsulated microRNAs as circulating biomarkers for breast cancer. Int. J. Cancer 139 (7), 1443–1448. doi:10.1002/ijc.30179
Kalimuthu, S., Gangadaran, P., Rajendran, R. L., Zhu, L., Oh, J. M., Lee, H. W., et al. (2018). A new approach for loading anticancer drugs into mesenchymal stem cell-derived exosome mimetics for cancer therapy. Front. Pharmacol. 9, 1116. doi:10.3389/fphar.2018.01116
Kalluri, R. (2016). The biology and function of exosomes in cancer. J. Clin. Invest. 126 (4), 1208–1215. doi:10.1172/JCI81135
Khan, S., Jutzy, J. M. S., Valenzuela, M. M. A., Turay, D., Aspe, J. R., Ashok, A., et al. (2012). Plasma-derived exosomal survivin, a plausible biomarker for early detection of prostate cancer. PLoS One 7 (10), e46737. doi:10.1371/journal.pone.0046737
Khan, S., Jutzy, J. M., Aspe, J. R., McGregor, D. W., Neidigh, J. W., Wall, N. R., et al. (2011). Survivin is released from cancer cells via exosomes. Apoptosis 16 (1), 1–12. doi:10.1007/s10495-010-0534-4
Khongkow, M., Yata, T., Boonrungsiman, S., Ruktanonchai, U. R., Graham, D., Namdee, K., et al. (2019). Surface modification of gold nanoparticles with neuron-targeted exosome for enhanced blood-brain barrier penetration. Sci. Rep. 9 (1), 8278. doi:10.1038/s41598-019-44569-6
Lapping-Carr, G., Gemel, J., Mao, Y., and Beyer, E. C. (2020). Circulating extracellular vesicles and endothelial damage in Sickle cell disease. Front. Physiol. 11. doi:10.3389/fphys.2020.01063
Lázaro-Ibáñez, E., Sanz-Garcia, A., Visakorpi, T., Escobedo-Lucea, C., Siljander, P., Ayuso-Sacido, A., et al. (2014). Different gDNA content in the subpopulations of prostate cancer extracellular vesicles: Apoptotic bodies, microvesicles, and exosomes. Prostate 74 (14), 1379–1390. doi:10.1002/pros.22853
Li, J., Li, Z., Jiang, P., Peng, M., Zhang, X., Chen, K., et al. (2018). Circular RNA IARS (circIARS) secreted by pancreatic cancer cells and located within exosomes regulates endothelial monolayer permeability to promote tumor metastasis. J. Exp. Clin. Cancer Res. 37 (1), 177. doi:10.1186/s13046-018-0822-3
Li, Z., Ma, Y.-Y., Wang, J., Zeng, X.-F., Li, R., Kang, W., et al. (2016). Exosomal microRNA-141 is upregulated in the serum of prostate cancer patients. Onco. Targets. Ther. 9, 139–148. doi:10.2147/OTT.S95565
Lin, J., Li, J., Huang, B., Liu, J., Chen, X., Chen, X. M., et al. (2015). Exosomes: Novel biomarkers for clinical diagnosis. ScientificWorldJournal. 2015, 657086. doi:10.1155/2015/657086
Lin, R., Wang, S., and Zhao, R. C. (2013). Exosomes from human adipose-derived mesenchymal stem cells promote migration through Wnt signaling pathway in a breast cancer cell model. Mol. Cell. Biochem. 383, 13–20. doi:10.1007/s11010-013-1746-z
Liu, C., Eng, C., Shen, J., Lu, Y., Takata, Y., Mehdizadeh, A., et al. (2016). Serum exosomal miR-4772-3p is a predictor of tumor recurrence in stage II and III colon cancer. Oncotarget 7, 76250–76260. doi:10.18632/oncotarget.12841
Liu, L., Meng, T., Yang, X.-H., Sayim, P., Lei, C., Jin, B., et al. (2018). Prognostic and predictive value of long non-coding RNA GAS5 and mircoRNA-221 in colorectal cancer and their effects on colorectal cancer cell proliferation, migration and invasion. Cancer Biomark. 22, 283–299. doi:10.3233/CBM-171011
Logozzi, M., Angelini, D. F., Iessi, E., Mizzoni, D., Di Raimo, R., Federici, C., et al. (2017). Increased PSA expression on prostate cancer exosomes in in vitro condition and in cancer patients. Cancer Lett. 403, 318–329. doi:10.1016/j.canlet.2017.06.036
Logozzi, M., De Milito, A., Lugini, L., Borghi, M., Calabrò, L., Spada, M., et al. (2009). High levels of exosomes expressing CD63 and caveolin-1 in plasma of melanoma patients. PloS one 4 (4), e5219. doi:10.1371/journal.pone.0005219
Logozzi, M., Mizzoni, D., Capasso, C., Del Prete, S., Di Raimo, R., Falchi, M., et al. (2020). Plasmatic exosomes from prostate cancer patients show increased carbonic anhydrase IX expression and activity and low pH. J. Enzyme Inhib. Med. Chem. 35, 280–288. doi:10.1080/14756366.2019.1697249
Lou, G., Song, X., Yang, F., Wu, S., Wang, J., Chen, Z., et al. (2015). Exosomes derived from miR-122-modified adipose tissue-derived MSCs increase chemosensitivity of hepatocellular carcinoma. J. Hematol. Oncol. 8, 122. doi:10.1186/s13045-015-0220-7
Luan, X., Sansanaphongpricha, K., Myers, I., Chen, H., Yuan, H., and Sun, D. (2017). Engineering exosomes as refined biological nanoplatforms for drug delivery. Acta Pharmacol. Sin. 38, 754–763. doi:10.1038/aps.2017.12
Madhavan, B., Yue, S., Galli, U., Rana, S., Gross, W., Müller, M., et al. (2015). Combined evaluation of a panel of protein and miRNA serum-exosome biomarkers for pancreatic cancer diagnosis increases sensitivity and specificity. Int. J. Cancer 136 (11), 2616–2627. doi:10.1002/ijc.29324
Matarredona, E. R., and Pastor, A. M. (2020). Extracellular vesicle-mediated communication. Cells 9 (1), 96–103. doi:10.3390/cells9010096
Mathew, M., Zade, M., Mezghani, N., Patel, R., Wang, Y., Momen-Heravi, F., et al. (2020). Extracellular vesicles as biomarkers in cancer immunotherapy. Cancers 12 (10), 2825. doi:10.3390/cancers12102825
Matsumura, T., Sugimachi, K., Iinuma, H., Takahashi, Y., Kurashige, J., Sawada, G., et al. (2015). Exosomal microRNA in serum is a novel biomarker of recurrence in human colorectal cancer. Br. J. Cancer 113, 275–281. doi:10.1038/bjc.2015.201
Melo, S. A., Sugimoto, H., O'Connell, J. T., Kato, N., Villanueva, A., Vidal, A., et al. (2014). Cancer exosomes perform cell-independent microRNA biogenesis and promote tumorigenesis. Cancer Cell. 26 (5), 707–721. doi:10.1016/j.ccell.2014.09.005
Melo, S. A., Luecke, L. B., Kahlert, C., Fernandez, A. F., Gammon, S. T., Kaye, J., et al. (2015). Glypican-1 identifies cancer exosomes and detects early pancreatic cancer. Nature 523, 177–182. doi:10.1038/nature14581
Mendt, M., Kamerkar, S., Sugimoto, H., McAndrews, K. M., Wu, C. C., Gagea, M., et al. (2018). Generation and testing of clinical-grade exosomes for pancreatic cancer. JCI Insight 3, e99263. doi:10.1172/jci.insight.99263
Meng, X., Müller, V., Milde-Langosch, K., Trillsch, F., Pantel, K., Schwarzenbach, H., et al. (2016). Diagnostic and prognostic relevance of circulating exosomal miR-373, miR-200a, miR-200b and miR-200c in patients with epithelial ovarian cancer. Oncotarget 7 (13), 16923–16935. doi:10.18632/oncotarget.7850
Moon, P.-G., Lee, J.-E., Cho, Y.-E., Lee, S. J., Chae, Y. S., Jung, J. H., et al. (2016a). Fibronectin on circulating extracellular vesicles as a liquid biopsy to detect breast cancer. Oncotarget 7, 40189–40199. doi:10.18632/oncotarget.9561
Moon, P.-G., Lee, J.-E., Cho, Y.-E., Lee, S. J., Jung, J. H., Chae, Y. S., et al. (2016b). Identification of developmental endothelial locus-1 on circulating extracellular vesicles as a novel biomarker for early breast cancer detection. Clin. Cancer Res. 22, 1757–1766. doi:10.1158/1078-0432.CCR-15-0654
Morelli, A. E., Larregina, A. T., Shufesky, W. J., Sullivan, M. L. G., Stolz, D. B., Papworth, G. D., et al. (2004). Endocytosis, intracellular sorting, and processing of exosomes by dendritic cells. Blood 104 (10), 3257–3266. doi:10.1182/blood-2004-03-0824
Nakase, I., Kobayashi, N. B., Takatani-Nakase, T., and Yoshida, T. (2015). Active macropinocytosis induction by stimulation of epidermal growth factor receptor and oncogenic Ras expression potentiates cellular uptake efficacy of exosomes. Sci. Rep. 5, 10300. doi:10.1038/srep10300
Nakase, I., and Futaki, S. (2015). Combined treatment with a pH-sensitive fusogenic peptide and cationic lipids achieves enhanced cytosolic delivery of exosomes. Sci. Rep. 5, 10112. doi:10.1038/srep10112
Ogata-Kawata, H., Izumiya, M., Kurioka, D., Honma, Y., Yamada, Y., Furuta, K., et al. (2014). Circulating exosomal microRNAs as biomarkers of colon cancer. PLoS ONE 9, e92921. doi:10.1371/journal.pone.0092921
Ohno, S. I., Takanashi, M., Sudo, K., Ueda, S., Ishikawa, A., Matsuyama, N., et al. (2013). Systemically injected exosomes targeted to EGFR deliver antitumor microrna to breast cancer cells. Mol. Ther. 21 (1), 185–191. doi:10.1038/mt.2012.180
Ohshima, K., Inoue, K., Fujiwara, A., Hatakeyama, K., Kanto, K., Watanabe, Y., et al. (2010). Let-7 microRNA family is selectively secreted into the extracellular environment via exosomes in a metastatic gastric cancer cell line. PloS one 5 (10), e13247. doi:10.1371/journal.pone.0013247
Palmisano, G., Jensen, S. S., Le Bihan, M. C., Lainé, J., McGuire, J. N., Pociot, F., et al. (2012). Characterization of membrane-shed microvesicles from cytokine-stimulated β-cells using proteomics strategies. Mol. Cell. Proteomics 11 (8), 230–243. doi:10.1074/mcp.M111.012732
Pan, C., Stevic, I., Müller, V., Ni, Q., Oliveira-Ferrer, L., Pantel, K., et al. (2018). Exosomal microRNAs as tumor markers in epithelial ovarian cancer. Mol. Oncol. 12, 1935–1948. doi:10.1002/1878-0261.12371
Parolini, I., Federici, C., Raggi, C., Lugini, L., Palleschi, S., De Milito, A., et al. (2009). Microenvironmental pH is a key factor for exosome traffic in tumor cells. J. Biol. Chem. 284 (49), 34211–34222. doi:10.1074/jbc.M109.041152
Patel, B., Patel, J., Cho, J. H., Manne, S., Bonala, S., Henske, E., et al. (2016). Exosomes mediate the acquisition of the disease phenotypes by cells with normal genome in tuberous sclerosis complex. Oncogene 35 (23), 3027–3036. doi:10.1038/onc.2015.358
Peinado, H., Alečković, M., Lavotshkin, S., Matei, I., Costasilva, B., Morenobueno, G., et al. (2012). Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat. Med. 18 (6), 883–891. doi:10.1038/nm.2753
Pulliam, L., Sun, B., Mustapic, M., Chawla, S., and Kapogiannis, D. (2019). Plasma neuronal exosomes serve as biomarkers of cognitive impairment in HIV infection and Alzheimer's disease. J. Neurovirol. 25 (5), 702–709. doi:10.1007/s13365-018-0695-4
Qin, X., Yu, S., Zhou, L., Shi, M., Hu, Y., Xu, X., et al. (2017). Cisplatin-resistant lung cancer cell-derived exosomes increase cisplatin resistance of recipient cells in exosomal miR-100-5p-dependent manner. Int. J. Nanomedicine 12, 3721–3733. doi:10.2147/IJN.S131516
Que, R., Ding, G., Chen, J., and Cao, L. (2013). Analysis of serum exosomal microRNAs and clinicopathologic features of patients with pancreatic adenocarcinoma. World J. Surg. Oncol. 11, 219. doi:10.1186/1477-7819-11-219
Rachel, E. S., Gabriel, M., Steve, K., Philip, A. P., and Asfar, S. A. (2017). Ras and exosome signaling Rachel. Physiol. Behav. 176, 139–148. doi:10.1016/j.semcancer.2019.02.004
Rana, S., Yue, S. J., Stadel, D., and Zoller, M. (2012). Toward tailored exosomes: The exosomal tetraspanin web contributes to target cell selection. Int. J. Biochem. Cell. Biol. 44, 1574–1584. doi:10.1016/j.biocel.2012.06.018
Rodríguez, M., Silva, J., Lópezalfonso, A., Lópezmuñiz, M. B., Peña, C., Domínguez, G., et al. (2014). Different exosome cargo from plasma/bronchoalveolar lavage in non-small-cell lung cancer. Genes. Chromosom. Cancer 53 (9), 713–724. doi:10.1002/gcc.22181
Ruivo, C. F., Adem, B., Silva, M., and Melo, S. A. (2017). The biology of cancer exosomes: Insights and new perspectives. Cancer Res. 77 (23), 6480–6488. doi:10.1158/0008-5472.CAN-17-0994
Sandfeld-Paulsen, B., Aggerholm-Pedersen, N., Baek, R., Jakobsen, K. R., Meldgaard, P., Folkersen, B. H., et al. (2016a). Exosomal proteins as prognostic biomarkers in non-small cell lung cancer. Mol. Oncol. 10, 1595–1602. doi:10.1016/j.molonc.2016.10.003
Sandfeld-Paulsen, B., Jakobsen, K. R., Bæk, R., Folkersen, B. H., Rasmussen, T. R., Meldgaard, P., et al. (2016b). Exosomal proteins as a diagnostic biomarkers in lung cancer. J. Thorac. Oncol. 11 (10), 1701–1710. doi:10.1016/j.jtho.2016.05.034
Silva, J., Garcia, V., Rodriguez, M., Compte, M., Cisneros, E., Veguillas, P., et al. (2012). Analysis of exosome release and its prognostic value in human colorectal cancer. Genes. Chromosom. Cancer 51, 409–418. doi:10.1002/gcc.21926
Sohn, W., Kim, J., Kang, S. H., Yang, S. R., Cho, J.-Y., Cho, H. C., et al. (2015). Serum exosomal microRNAs as novel biomarkers for hepatocellular carcinoma. Exp. Mol. Med. 47, e184. doi:10.1038/emm.2015.68
Sugimachi, K., Matsumura, T., Hirata, H., Uchi, R., Ueda, M., Ueo, H., et al. (2015). Identification of a bona fide microRNA biomarker in serum exosomes that predicts hepatocellular carcinoma recurrence after liver transplantation. Br. J. Cancer 112, 532–538. doi:10.1038/bjc.2014.621
Sun, B., Li, Y., Zhou, Y., Ng, T. K., Zhao, C., Gan, Q., et al. (2018). Circulating exosomal CPNE3 as a diagnostic and prognostic biomarker for colorectal cancer. J. Cell. Physiol. 234, 1416–1425. doi:10.1002/jcp.26936
Sun, Z., Wang, L., Dong, L., and Wang, X. (2018). Emerging role of exosome signalling in maintaining cancer stem cell dynamic equilibrium. J. Cell. Mol. Med. 22, 3719–3728. doi:10.1111/jcmm.13676
Svensson, K. J., Christianson, H. C., Wittrup, A., Bourseau-Guilmain, E., Lindqvist, E., Svensson, L. M., et al. (2013). Exosome uptake depends on ERK1/2-heat shock protein 27 signaling and lipid raft-mediated endocytosis negatively regulated by caveolin-1. J. Biol. Chem. 288 (24), 17713–17724. doi:10.1074/jbc.M112.445403
Szajnik, M., Derbis, M., Lach, M., Patalas, P., Michalak, M., Drzewiecka, H., et al. (2013). Exosomes in plasma of patients with ovarian carcinoma: Potential biomarkers of tumor progression and response to therapy. Gynecol. Obstet. 4, 3. doi:10.4172/2161-0932.S4-003
Szczepanski, M. J., Szajnik, M., Welsh, A., Whiteside, T. L., and Boyiadzis, M. (2011). Blast-derived microvesicles in sera from patients with acute myeloid leukemia suppress natural killer cell function via membrane-associated transforming growth factor-beta1. Haematologica 96 (9), 1302–1309. doi:10.3324/haematol.2010.039743
Takahashi, K., Yan, I. K., Kogure, T., Haga, H., and Patel, T. (2014a). Extracellular vesicle-mediated transfer of long non-coding RNA ROR modulates chemosensitivity in human hepatocellular cancer. FEBS open bio 4, 458–467. doi:10.1016/j.fob.2014.04.007
Takahashi, K., Yan, I. K., Wood, J., Haga, H., and Patel, T. (2014b). Involvement of extracellular vesicle long noncoding RNA (linc-VLDLR) in tumor cell responses to chemotherapy. Mol. Cancer Res. 12 (10), 1377–1387. doi:10.1158/1541-7786.MCR-13-0636
Takahasi, K., Iinuma, H., Wada, K., Minezaki, S., Kawamura, S., Kainuma, M., et al. (2018). Usefulness of exosome-encapsulated microRNA-451a as a minimally invasive biomarker for prediction of recurrence and prognosis in pancreatic ductal adenocarcinoma. J. Hepatobiliary. Pancreat. Sci. 25, 155–161. doi:10.1002/jhbp.524
Tang, W., Fu, K., Sun, H., Rong, D., Wang, H., Cao, H., et al. (2018). CircRNA microarray profiling identifies a novel circulating biomarker for detection of gastric cancer. Mol. Cancer 17 (1), 137. doi:10.1186/s12943-018-0888-8
Tang, X. H., Guo, T., Gao, X. Y., Wu, X. L., Xing, X. F., Ji, J. F., et al. (2021). Exosome-derived noncoding RNAs in gastric cancer: Functions and clinical applications. Mol. Cancer 20 (1), 99. doi:10.1186/s12943-021-01396-6
Taraboletti, G., D’Ascenzo, S., Giusti, I., Marchetti, D., Borsotti, P., Millimaggi, D., et al. (2006). Bioavailability of VEGF in tumor-shed vesicles depends on vesicle burst induced by acidic pH. Neoplasia 8 (2), 96–103. doi:10.1593/neo.05583
Taylor, D. D., and Gercel-Taylor, C. (2008). MicroRNA signatures of tumor-derived exosomes as diagnostic biomarkers of ovarian cancer. Gynecol. Oncol. 110 (1), 13–21. doi:10.1016/j.ygyno.2008.04.033
Taylor, D. D., Gerçel-Taylor, C., Lyons, K. S., Stanson, J., and Whiteside, T. L. (2003). T-cell apoptosis and suppression of T-cell receptor/CD3-zeta by Fas ligand-containing membrane vesicles shed from ovarian tumors. Clin. Cancer Res. 9 (14), 5113–5119.
Thomas Jefferson University (2000). “Pilot immunotherapy trial for recurrent malignant gliomas,” in ClinicalTrials.gov [Internet] (Bethesda (MD): National Library of Medicine US).
Tian, T., Zhu, Y. L., Hu, F. H., Wang, Y. Y., Huang, N. P., Xiao, Z. D., et al. (2013). Dynamics of exosome internalization and trafficking. J. Cell. Physiol. 228 (7), 1487–1495. doi:10.1002/jcp.24304
Tian, T., Zhu, Y. L., Zhou, Y. Y., Liang, G. F., Wang, Y. Y., Hu, F. H., et al. (2014). Exosome uptake through clathrin-mediated endocytosis and macropinocytosis and mediating miR-21 delivery. J. Biol. Chem. 289, 22258–22267. doi:10.1074/jbc.M114.588046
Tian, Y., Li, S., Song, J., Ji, T., Zhu, M., Anderson, G. J., et al. (2014). A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials 35 (7), 2383–2390. doi:10.1016/j.biomaterials.2013.11.083
Tian, Y., Ma, L., Gong, M., Su, G., Zhu, S., Zhang, W., et al. (2018). Protein profiling and sizing of extracellular vesicles from colorectal cancer patients via flow cytometry. ACS Nano 12, 671–680. doi:10.1021/acsnano.7b07782
Tomasetti, M., Lee, W., Santarelli, L., and Neuzil, J. (2017). Exosome-derived microRNAs in cancer metabolism: Possible implications in cancer diagnostics and therapy. Exp. Mol. Med. 49, e285. doi:10.1038/emm.2016.153
Towns, R., and Pietropaolo, M. (2011). GAD65 autoantibodies and its role as biomarker of type 1 diabetes and latent autoimmune diabetes in adults (LADA). Drugs Future 36 (11), 847. doi:10.1358/dof.2011.036.11.1710754
Tsukamoto, M., Iinuma, H., Yagi, T., Matsuda, K., and Hashiguchi, Y. (2017). Circulating exosomal MicroRNA-21 as a biomarker in each tumor stage of colorectal cancer. Oncology 92, 360–370. doi:10.1159/000463387
Vardaki, I., Ceder, S., Rutishauser, D., Baltatzis, G., Foukakis, T., Panaretakis, T., et al. (2016). Periostin is identified as a putative metastatic marker in breast cancer-derived exosomes. Oncotarget 7 (46), 74966–74978. doi:10.18632/oncotarget.11663
Whiteside, T. L. (2016). Tumor-derived exosomes and their role in cancer progression. Adv. Clin. Chem. 74, 103–141. doi:10.1016/bs.acc.2015.12.005
Willis, G. R., Fernandez-Gonzalez, A., Reis, M., Mitsialis, S. A., and Kourembanas, S. (2018). Macrophage immunomodulation: The gatekeeper for mesenchymal stem cell derived-exosomes in pulmonary arterial hypertension? Int. J. Mol. Sci. 19. doi:10.3390/ijms19092534
Wolfers, J., Lozier, A., Raposo, G., Regnault, A., Thery, C., and Masurier, C. (2001). Tumor-derived exosomes are a source of shared tumor rejection antigens for CTL cross-priming. Nat. Med. 7, 297–303. doi:10.1038/85438
Wong, C. H., and Chen, Y. C. (2019). Clinical significance of exosomes as potential biomarkers in cancer. World J. Clin. Cases 7 (2), 171–190. doi:10.12998/wjcc.v7.i2.171
Xiao, Y., Zhong, J., Zhong, B., Huang, J., Jiang, L., Jiang, Y., et al. (2020). Exosomes as potential sources of biomarkers in colorectal cancer. Cancer Lett. 476, 13–22. doi:10.1016/j.canlet.2020.01.033
Xu, K., Zhang, C., Du, T., Gabriel, A., Wang, X., Li, X., et al. (2021). Progress of exosomes in the diagnosis and treatment of lung cancer. Biomed. Pharmacother. 134, 111111. doi:10.1016/j.biopha.2020.111111
Xu, M., Su, Y., Liu, X., Xu, M., Chen, X., Zhu, Y., et al. (2018). Serum and exosome long non coding RNAs as potential biomarkers for hepatocellular carcinoma. J. Cancer 9 (15), 2631–2639. doi:10.7150/jca.24978
Xu, M., Sun, X., Wang, Y., Yang, Q., Sun, X., and Wang, Y. (2020). Recent advancements in the loading and modification of therapeutic exosomes. Front. Bioeng. Biotechnol. 8, 586130. doi:10.3389/fbioe.2020.586130
Yan-Liang, Z., Yue-Yuan, Z., Gao-Feng, L., Yuan-Yuan, W., Fei-Hu, H., and Zhong-Dang, X. (2014). Exosome uptake through clathrin-mediated endocytosis and macropinocytosis and mediating miR-21 delivery. J. Biol. Chem. 289 (32), 22258–22267. doi:10.1074/jbc.M114.588046
Yang, H., Fu, H., Wang, B., Zhang, X., Mao, J., Li, X., et al. (2018). Exosomal miR-423-5p targets SUFU to promote cancer growth and metastasis and serves as a novel marker for gastric cancer. Mol. Carcinog. 57, 1223–1236. doi:10.1002/mc.22838
Yoon, J. H., Ham, I.-H., Kim, O., Ashktorab, H., Smoot, D. T., Nam, S. W., et al. (2018). Gastrokine 1 protein is a potential theragnostic target for gastric cancer. Gastric Cancer 21, 956–967. doi:10.1007/s10120-018-0828-8
Yoshida, K., Yokoi, A., Kato, T., Ochiya, T., and Yamamoto, Y. (2020). The clinical impact of intra- and extracellular miRNAs in ovarian cancer. Cancer Sci. 111 (10), 3435–3444. doi:10.1111/cas.14599
Yoshioka, Y., Konishi, Y., Kosaka, N., Katsuda, T., Kato, T., Ochiya, T., et al. (2013). Comparative marker analysis of extracellular vesicles in different human cancer types. J. Extracell. Vesicles 2, 20424. doi:10.3402/jev.v2i0.20424
You, B., Xu, W., and Zhang, B. (2018). Engineering exosomes: A new direction for anticancer treatment. Am. J. Cancer Res. 8 (8), 1332–1342. Published 2018 Aug 1.
Yue, B., Yang, H., Wang, J., Ru, W., Wu, J., Huang, Y., et al. (2020). Exosome biogenesis, secretion and function of exosomal miRNAs in skeletal muscle myogenesis. Cell. Prolif. 53, e12857. doi:10.1111/cpr.12857
Yuwen, D., Ma, Y., Wang, D., Gao, J., Li, X., Xue, W., et al. (2019). Prognostic role of circulating exosomal miR-425-3p for the response of NSCLC to platinum-based chemotherapy. Cancer Epidemiol. Biomarkers Prev. 28 (1), 163–173. doi:10.1158/1055-9965.EPI-18-0569
Zhang, Y., Bi, J., Huang, J., Tang, Y., Du, S., Li, P., et al. (2020). Exosome: A review of its classification, isolation techniques, storage, diagnostic and targeted therapy applications. Int. J. Nanomedicine 15, 6917–6934. doi:10.2147/IJN.S264498
Zhao, R., Zhang, Y., Zhang, X., Yang, Y., Zheng, X., Li, X., et al. (2018). Exosomal long noncoding RNA HOTTIP as potential novel diagnostic and prognostic biomarker test for gastric cancer. Mol. Cancer 17 (1), 68. doi:10.1186/s12943-018-0817-x
Zhao, Y. J., and Xie, L. (2019). Potential role of exosomes in cancer therapy. Precis. Radiat. Oncol. 3, pro6.66. doi:10.1002/pro6.66
Keywords: exosome, targeted delivery, exosome mimetics, biomarker, therapeutics
Citation: Mishra LC, Pandey U, Gupta A, Gupta J, Sharma M and Mishra G (2022) Alternating exosomes and their mimetics as an emergent strategy for targeted cancer therapy. Front. Mol. Biosci. 9:939050. doi: 10.3389/fmolb.2022.939050
Received: 08 May 2022; Accepted: 15 July 2022;
Published: 10 August 2022.
Edited by:
José Alexandre Ferreira, Portuguese Oncology Institute, PortugalReviewed by:
Kamla Kant Shukla, All India Institute of Medical Sciences Jodhpur, IndiaReetobrata Basu, Ohio University, United States
Copyright © 2022 Mishra, Pandey, Gupta, Gupta, Sharma and Mishra. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Gauri Mishra, Z2F1cmlzaHVrbGExQGdtYWlsLmNvbQ==
†These authors have contributed equally to this work
 Lokesh Chandra Mishra
Lokesh Chandra Mishra Utkarsh Pandey
Utkarsh Pandey Abhikarsh Gupta
Abhikarsh Gupta Jyotsna Gupta
Jyotsna Gupta Monal Sharma
Monal Sharma Gauri Mishra
Gauri Mishra